Glucosylceramide Changes Bacterial Metabolism and Increases Gram-Positive Bacteria through Tolerance to Secondary Bile Acids In Vitro †
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains
2.2. Materials
2.3. Bacterial Cultures
2.4. Coculture of Glucosylceramide and Microbes
2.5. Analysis of Metabolites of Cultured Broth Using Gas Chromatography
2.6. Analysis of Lactic Acid
2.7. Next-Generation Sequencing Analysis of Cell-Mock-1 Incubated with Glucosylceramide
2.8. Statistical Analysis
3. Results
3.1. Glucosylceramide Addition Alters Metabolism of Blautia coccoides
3.2. Glucosylceramide Addition Increases Gram-Positive Intestinal Microbes
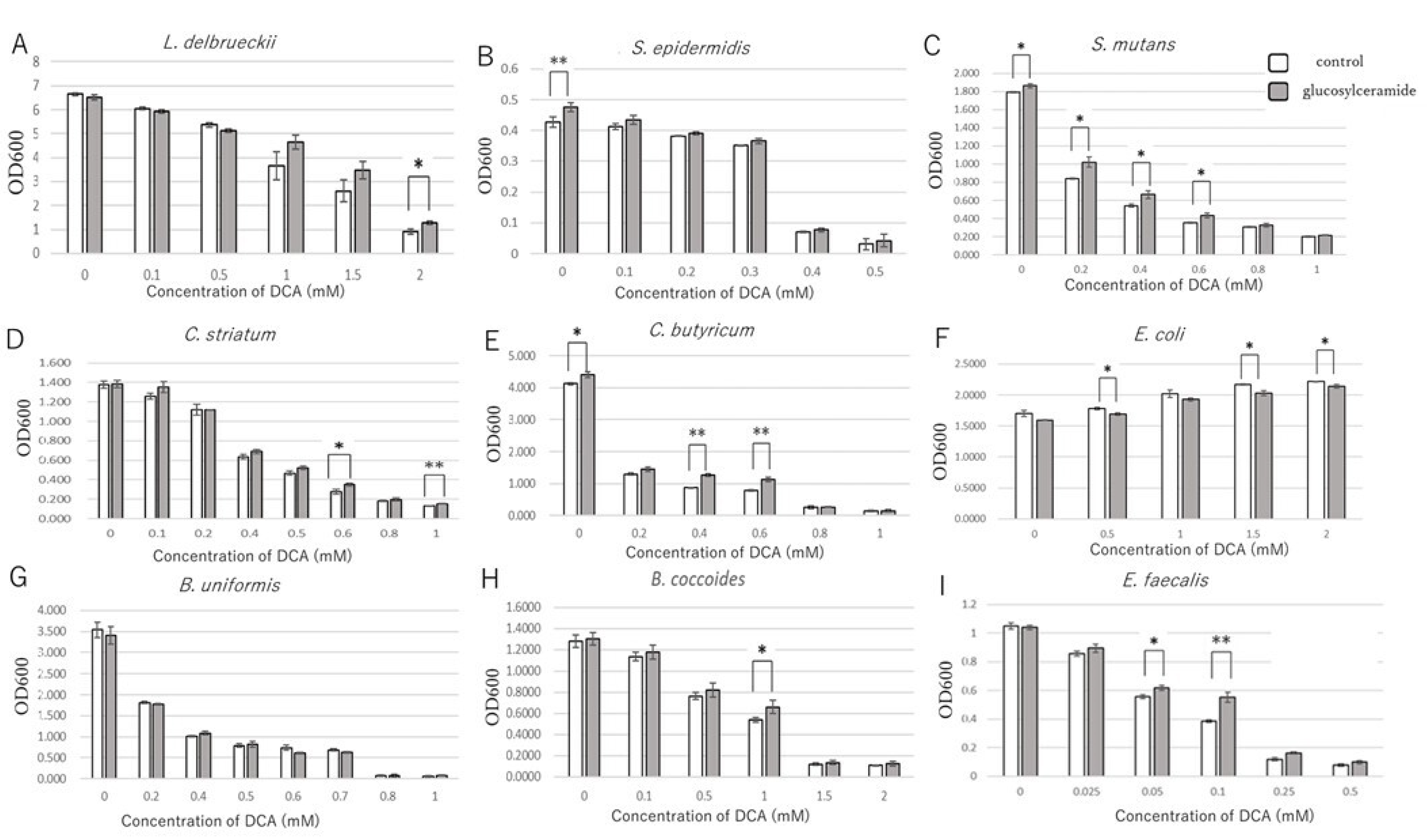
3.3. Glucosylceramide Confers Tolerance of Intestinal Microbes to Secondary Bile Acids
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lilly, D.M.; Stillwell, R.H. Probiotics: Growth-promoting factors produced by microorganisms. Science 1965, 147, 747–748. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R. Dietary modulation of the human gut microflora using prebiotics. Br. J. Nutr. 1998, 80, S209–S212. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Han, F.; Hu, B.; Li, J.; Yu, W. In vivo prebiotic properties of alginate oligosaccharides prepared through enzymatic hydrolysis of alginate. Nutr. Res. 2006, 26, 597–603. [Google Scholar] [CrossRef]
- Kukkonen, K.; Savilahti, E.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Poussa, T.; Tuure, T.; Kuitunen, M. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: A randomized, double-blind, placebo-controlled trial. J. Allergy Clin. Immunol. 2007, 119, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Joyce, S.A.; Kamil, A.; Fleige, L.; Fleige, L.; Gahan, C.G. The cholesterol-lowering effect of oats and oat beta glucan: Modes of action and potential role of bile acids and the microbiome. Front. Nutr. 2019, 6, 171. [Google Scholar] [CrossRef] [PubMed]
- Kankaanpää, P.; Yang, B.; Kallio, H.; Lsolauri, E.; Saiminen, S. Effects of polyunsaturated fatty acids in growth medium on lipid composition and on physicochemical surface properties of lactobacilli. Appl. Environ. Microbiol. 2004, 70, 129–136. [Google Scholar] [CrossRef]
- Guo, T.; Song, D.; Cheng, L.; Xin, Z. Interactions of tea catechins with intestinal microbiota and their implication for human health. Food Sci. Biotechnol. 2019, 28, 1617–1625. [Google Scholar] [CrossRef]
- Ito, Y.; Ichikawa, T.; Morohoshi, Y.; Nakamura, T.; Saegusa, Y.; Ishihara, K. Effect of tea catechins on body fat accumulation in rats fed a normal diet. Biomed. Res. 2008, 29, 27–32. [Google Scholar] [CrossRef][Green Version]
- Jung, E.S.; Park, H.M.; Hyun, S.M.; Shon, J.C.; Singh, D.; Liu, K.-H.; Whon, T.W.; Bae, J.-W.; Hwang, J.S.; Lee, C.H. The green tea modulates large intestinal microbiome and exo/endogenous metabolome altered through chronic UVB-exposure. PLoS ONE 2017, 12, e0187154. [Google Scholar] [CrossRef]
- Hidalgo, M.; Oruna-Concha, M.J.; Kolida, S.; Walton, G.E.; Kallithraka, S.; Spencer, J.P.E.; Gibson, G.R.; Pascual-Teresa, S.D. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. J. Agric. Food Chem. 2012, 60, 3882–3890. [Google Scholar] [CrossRef]
- Naqash, F.; Masoodi, F.A.; Rather, S.A.; Wani, S.M.; Gani, A. Emerging concepts in the nutraceutical and functional properties of pectin—A review. Carbohydr. Polym. 2017, 168, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Kumrungsee, T.; Kato, N.; Fukuda, S.; Kuroda, M.; Yamaguchi, S. Supplemental Aspergillus lipase and protease preparations display powerful bifidogenic effects and modulate the gut microbiota community of rats. Fermentation 2021, 7, 294. [Google Scholar] [CrossRef]
- Yang, Y.; Kumrungsee, T.; Kato, N.; Fukuda, S.; Kuroda, M.; Yamaguchi, S. Aspergillus-derived cellulase preparation exhibits prebiotic-like effects on gut microbiota in rats. Fermentation 2022, 8, 71. [Google Scholar] [CrossRef]
- Yang, Y.; Iwamoto, A.; Kumrungsee, T.; Okazaki, Y.; Kuroda, M.; Yamaguchi, S.; Kato, N. Consumption of an acid protease derived from Aspergillus oryzae causes bifidogenic effect in rats. Nutr. Res. 2017, 44, 60–66. [Google Scholar] [CrossRef]
- Platt, F.M. Sphingolipid lysosomal storage disorders. Nature 2014, 510, 68–75. [Google Scholar] [CrossRef]
- Hannun, Y.A.; Bell, R.M. Functions of sphingolipids and sphingolipid breakdown products in cellular regulation. Science 1989, 243, 500–507. [Google Scholar] [CrossRef]
- Hannun, Y.A.; Obeid, L.M. Principles of bioactive lipid signalling: Lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 2008, 9, 139–150. [Google Scholar] [CrossRef]
- Fujino, Y.; Ohnishi, M. Structure of cerebroside in Aspergillus oryzae. Biochim. Biophys. Acta 1976, 486, 161–171. [Google Scholar] [CrossRef]
- Hirata, M.; Tsuge, K.; Jayakody, L.N.; Urano, Y.; Sawada, K.; Inaba, S.; Nagao, K.; Kitagaki, H. Structural determination of glucosylceramides in the distillation remnants of shochu, the Japanese traditional liquor, and its production by Aspergillus kawachii. J. Agric. Food Chem. 2012, 60, 11473–11482. [Google Scholar] [CrossRef]
- Hamajima, H.; Fujikawa, A.; Yamashiro, M.; Ogami, T.; Kitamura, S.; Tsubata, M.; Tan, S.; Matsunaga, H.; Sawada, K.; Kumagai, S.; et al. Chemical analysis of the sugar moiety of monohexosylceramide contained in koji, Japanese traditional rice fermented with Aspergillus. Fermentation 2016, 2, 2. [Google Scholar] [CrossRef]
- Tani, Y.; Amaishi, Y.; Funatsu, T.; Ito, M.; Itonori, S.; Hata, Y.; Ashida, H.; Yamamoto, K. Structural analysis of cerebrosides from Aspergillus fungi: The existence of galactosylceramide in A. oryzae. Biotechnol. Lett. 2014, 36, 2507–2513. [Google Scholar] [CrossRef] [PubMed]
- Del Poeta, M.; Nimrichter, L.; Rodrigues, M.L.; Luberto, C. Synthesis and biological properties of fungal glucosylceramide. PLoS Pathog. 2014, 10, e1003832. [Google Scholar] [CrossRef] [PubMed]
- Hamajima, H.; Matsunaga, H.; Fujikawa, A.; Sato, T.; Mitsutake, S.; Yanagita, T.; Nagao, K.; Nakayama, J.; Kitagaki, H. Japanese traditional dietary fungus koji Aspergillus oryzae functions as a prebiotic for Blautia coccoides through glycosylceramide: Japanese dietary fungus koji is a new prebiotic. Springer Plus 2016, 5, 1321. [Google Scholar] [CrossRef]
- Takahashi, K.; Izumi, K.; Nakahata, E.; Hirata, M.; Sawada, K.; Tsuge, K.; Nagao, K.; Kitagaki, H. Quantitation and structural determination of glucosylceramides contained in sake lees. J. Oleo Sci. 2014, 63, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Takakuwa, N.; Saito, K.; Ohnishi, M.; Oda, Y. Determination of glucosylceramide contents in crop tissues and by-products from their processing. Bioresour. Technol. 2005, 96, 1089–1092. [Google Scholar] [CrossRef]
- Schmelz, E.M.; Sullards, M.C.; Dillehay, D.L.; Merrill, A.H., Jr. Colonic cell proliferation and aberrant crypt foci formation are inhibited by dairy glycosphingolipids in 1,2-dimethylhydrazine-treated CF1 mice. J. Nutr. 2000, 130, 522–527. [Google Scholar] [CrossRef]
- Symolon, H.; Schmelz, E.M.; Dillehay, D.L.; Merrill, A.H., Jr. Dietary soy sphingolipids suppress tumorigenesis and gene expression in 1, 2-dimethylhydrazine-treated CF1 mice and Apc Min/+ mice. J. Nutr. 2004, 134, 1157–1161. [Google Scholar] [CrossRef]
- Fujiwara, K.; Yazama, H.; Donishi, R.; Koyama, S.; Fukuhara, T.; Takeuchi, H. Inhibitory effects of glucosylceramide on tumorigenesis induced by a carcinogen in mice. Laryngoscope 2020, 130, E593–E597. [Google Scholar] [CrossRef]
- Yazama, H.; Kitatani, K.; Fujiwara, K.; Kato, M.; Hashimoto-Nishimura, M.; Kawamoto, K.; Hasegawa, K.; Kitano, H.; Bielawska, A.; Bielawski, J.; et al. Dietary glucosylceramides suppress tumor growth in a mouse xenograft model of head and neck squamous cell carcinoma by the inhibition of angiogenesis through an increase in ceramide. Int. J. Clin. Oncol. 2015, 20, 438–446. [Google Scholar] [CrossRef]
- Fujiwara, K.; Kitatani, K.; Fukushima, K.; Yazama, H.; Umehara, H.; Kikuchi, M.; Igarashi, Y.; Kitano, H.; Okazaki, T. Inhibitory effects of dietary glucosylceramides on squamous cell carcinoma of the head and neck in NOD/SCID mice. Int. J. Clin. Oncol. 2011, 16, 133–140. [Google Scholar] [CrossRef]
- Hossain, Z.; Sugawara, T.; Aida, K.; Hirata, T. Effect of dietary glucosylceramide from sea cucumber on plasma and liver lipids in cholesterol-fed mice. Fish. Sci. 2011, 77, 1081–1085. [Google Scholar] [CrossRef]
- Tsuji, K.; Mitsutake, S.; Ishikawa, J.; Takagi, Y.; Akiyama, M.; Shimizu, H.; Tomiyama, T.; Igarashi, Y. Dietary glucosylceramide improves skin barrier function in hairless mice. J. Dermatol. Sci. 2006, 44, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, M.; Fujikawa, A.; Nagadome, M.; Kohama, K.; Ogami, T.; Kitamura, S.; Kitagaki, H. Glycosylceramides purified from the Japanese traditional non-pathogenic fungus Aspergillus and koji increase the expression of genes involved in tight junctions and ceramide delivery in normal human epidermal keratinocytes. Fermentation 2019, 5, 43. [Google Scholar] [CrossRef]
- Yamashita, S.; Seino, T.; Aida, K.; Kinoshita, M. Effects of plant sphingolipids on inflammatory stress in differentiated caco-2 cells. J. Oleo Sci. 2017, 66, 1337–1342. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sugawara, T.; Kinoshita, M.; Ohnishi, M.; Miyazawa, T. Apoptosis induction by wheat-flour sphingoid bases in DLD-1 human colon cancer cells. Biosci. Biotechnol. Biochem. 2002, 66, 2228–2231. [Google Scholar] [CrossRef] [PubMed]
- Aida, K.; Kinoshita, M.; Sugawara, T.; Ono, J.; Miyazawa, T.; Ohnishi, M. Apoptosis inducement by plant and fungus sphingoid bases in human colon cancer cells. J. Oleo Sci. 2004, 53, 503–510. [Google Scholar] [CrossRef]
- Nilsson, Å. Metabolism of cerebroside in the intestinal tract of the rat. Biochim. Biophys. Acta BBA-Lipids Lipid Metab. 1969, 187, 113–121. [Google Scholar] [CrossRef]
- Sugawara, T.; Tsuduki, T.; Yano, S.; Hirose, M.; Duan, J.; Aida, K.; Ikeda, I.; Hirata, T. Intestinal absorption of dietary maize glucosylceramide in lymphatic duct cannulated rats. J. Lipid Res. 2010, 51, 1761–1769. [Google Scholar] [CrossRef]
- Kawata, M.; Tsukamoto, A.; Isozaki, R.; Nobukawa, S.; Kawahara, N.; Akutsu, S.; Suzuki, M.; Asanuma, N. Glucerabacter Canisensis gen. nov., sp. nov., isolated from dog feces and its effect on the hydrolysis of plant glucosylceramide in the intestine of dogs. Arch. Microbiol. 2018, 200, 505–515. [Google Scholar] [CrossRef]
- Zwicker, B.L.; Agellon, L.B. Transport and biological activities of bile acids. Int. J. Biochem. Cell Biol. 2013, 45, 1389–1398. [Google Scholar] [CrossRef]
- Yokota, A.; Fukiya, S.; Islam, K.B.M.S.; Ooka, T.; Ogura, Y.; Hayashi, T.; Hagio, M.; Ishizuka, S. Is bile acid a determinant of the gut microbiota on a high-fat diet? Gut Microbes 2012, 3, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Umar, S.; Rust, B.; Lazarova, D.; Bordonaro, M. Secondary bile acids and short chain fatty acids in the colon: A focus on colonic microbiome, cell proliferation, inflammation, and cancer. Int. J. Mol. Sci. 2019, 20, 1214. [Google Scholar] [CrossRef] [PubMed]
- Mouzaki, M.; Wang, A.Y.; Bandsma, R.; Comelli, E.M.; Arendt, B.M.; Zhang, L.; Fung, S.; Fischer, S.E.; Mcgilvray, L.G.; Allard, J.P. Bile acids and dysbiosis in non-alcoholic fatty liver disease. PLoS ONE 2016, 11, e0151829. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Hold, G.L.; Harmsen, H.; Stewart, C.S.; Flint, H.J. Growth requirements and fermentation products of Fusobacterium prausnitzii, and a proposal to reclassify it as Faecalibacterium prausnitzii gen. nov., comb. nov. Int. J. Syst. Evol. Microbiol. 2002, 52, 2141–2146. [Google Scholar] [CrossRef]
- Kusaba, Y.; Otsuka, A.; Dai, H.; Inaba, S.; Kitagaki, H. Induction of chromosomal aneuploids from brewery shochu yeast with novel brewery characteristics. Fermentation 2022, 8, 62. [Google Scholar] [CrossRef]
- Motomura, S.; Horie, K.; Kitagaki, H. Mitochondrial activity of sake brewery yeast affects malic and succinic acid production during alcoholic fermentation. J. Inst. Brew. 2012, 118, 22–26. [Google Scholar] [CrossRef]
- Takahashi, S.; Tomita, J.; Nishioka, K.; Hisada, T.; Nishijima, M. Development of a prokaryotic universal primer for simultaneous analysis of bacteria and archaea using next-generation sequencing. PLoS ONE 2014, 9, e105592. [Google Scholar] [CrossRef]
- Aronesty, E. Comparison of sequencing utility programs. Open Bioinform. J. 2013, 7, 1–8. [Google Scholar] [CrossRef]
- Gordon, A.; Hannon, G.J. FASTX-Toolkit FASTQ/A Short-Reads Preprocessing Tools [Software]. Available online: http://hannonlab.cshl.edu/fastx_toolkit/index.html (accessed on 5 May 2022).
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Chong, J.; Soufan, O.; Li, C.; Caraus, L.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
- Ozato, N.; Saito, S.; Yamaguchi, T.; Katashima, M.; Tokuda, I.; Sawada, K.; Katsuragi, Y.; Kakuta, M.; Imoto, S.; Ihara, K.; et al. Blautia genus associated with visceral fat accumulation in adults 20–76 years of age. NPJ Biofilms Microbiomes 2019, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Sawada, K.; Sato, T.; Hamajima, H.; Jayakody, L.N.; Hirata, M.; Yamashiro, M.; Tajima, M.; Mitsutake, S.; Nagao, K.; Tsuge, K.; et al. Glucosylceramide contained in koji mold-cultured cereal confers membrane and flavor modification and stress tolerance to Saccharomyces cerevisiae during coculture fermentation. Appl. Environ. Microbiol. 2015, 81, 3688–3698. [Google Scholar] [CrossRef] [PubMed]
- Olsson, O. The capacity of the large intestine. Acta Radiol. 1952, 37, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.K.; Koo, B.G.; Sharma, C.; Kang, S.C. Characterization of Weissella confusa DD_A7 isolated from kimchi. LWT 2019, 111, 663–672. [Google Scholar] [CrossRef]
- Liu, X.; Mao, B.; Gu, J.; Wu, J.; Cui, S.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. Blautia—A new functional genus with potential probiotic properties? Gut Microbes 2021, 13, 1875796. [Google Scholar] [CrossRef]
- Yang, J.; Bindels, L.B.; Segura Munoz, R.R.; Martínez, I.; Walter, J.; Ramer-Tait, A.E.; Rose, D.J. Disparate metabolic responses in mice fed a high-fat diet supplemented with maize-derived non-digestible feruloylated oligo- and polysaccharides are linked to changes in the gut microbiota. PLoS ONE 2016, 11, e0146144. [Google Scholar] [CrossRef]
- Panasevich, M.R.; Kerr, K.R.; Dilger, R.N.; Fahey, G.C.; Guérin-Deremaux, L., Jr.; Lynch, G.L.; Wils, D.; Suchodolski, J.S.; Steer, J.M.; Dowd, S.E.; et al. Modulation of the faecal microbiome of healthy adult dogs by inclusion of potato fibre in the diet. Br. J. Nutr. 2015, 113, 125–133. [Google Scholar] [CrossRef]
- Lee, S.M.; Han, H.W.; Yim, S.Y. Beneficial effects of soy milk and fiber on high cholesterol diet-induced alteration of gut microbiota and inflammatory gene expression in rats. Food Funct. 2015, 6, 492–500. [Google Scholar] [CrossRef]
- Odamaki, T.; Kato, K.; Sugahara, H.; Hashikura, N.; Takahashi, S.; Xiao, J.Z.; Abe, F.; Osawa, R. Age-related changes in gut microbiota composition from newborn to centenarian: A cross-sectional study. BMC Microbiol. 2016, 16, 90. [Google Scholar] [CrossRef]
- Nakayama, J.; Watanabe, K.; Jiang, J.; Matsuda, K.; Chao, S.H.; Haryono, P.; La-Ongkham, O.; Sarwoko, M.A.; Sujaya, I.N.; Zhao, L.; et al. Diversity in gut bacterial community of school-age children in Asia. Sci. Rep. 2015, 5, 8397. [Google Scholar] [CrossRef]
- Bonder, M.J.; Kurilshikov, A.; Tigchelaar, E.F.; Mujagic, Z.; Imhann, F.; Vila, A.V.; Deelen, P.; Vatanen, T.; Schirmer, M.; Smeekens, S.P.; et al. The effect of host genetics on the gut microbiome. Nat. Genet. 2016, 48, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-C.; Gurram, B.; Baldridge, M.; Head, R.; Lam, V.; Luo, C.; Cao, Y.; Simpson, P.; Hayward, M.; Holte, M.; et al. O-011 Paneth cell phenotypes define a subtype of pediatric Crohn’s disease through alterations in host–microbial interactions. Inflamm. Bowel Dis. 2016, 22, S4. [Google Scholar] [CrossRef]
- Torres, J.; Bao, X.; Goel, A.; Colombel, J.-F.; Pekow, J.; Jabri, B.; Williams, K.M.; Castillo, A.; Odin, J.A.; Meckel, K.; et al. The features of mucosa-associated microbiota in primary sclerosing cholangitis. Aliment. Pharmacol. Ther. 2016, 43, 790–801. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, A.; Valdés, L.; Salazar, N.; de los Reyes-Gavilán, C.G.; Ruas-Madiedo, P.; Gueimonde, M.; González, S. Pilot study of diet and microbiota: Interactive associations of fibers and polyphenols with human intestinal bacteria. J. Agric. Food Chem. 2014, 62, 5330–5336. [Google Scholar] [CrossRef]
- Suda, Y.; Kagawa, K.; Fukuyama, K.; Elean, M.; Zhou, B.; Tomokiyo, M.; Islam, M.A.; Rajoka, M.; Kober, A.; Shimazu, T.; et al. Soymilk-fermented with Lactobacillus delbrueckii subsp. delbrueckii TUA4408L improves immune-health in pigs. Benef. Microbes 2022, 13, 61–72. [Google Scholar] [CrossRef]
- Mogna, L.; Deidda, F.; Nicola, S.; Amoruso, A.; Del Piano, M.; Mogna, G. In vitro inhibition of Klebsiella pneumoniae by Lactobacillus delbrueckii subsp. delbrueckii LDD01 (DSM 22106): An innovative strategy to possibly counteract such infections in humans? J. Clin. Gastroenterol. 2016, 50, S136–S139. [Google Scholar] [CrossRef]
- Kanai, T.; Mikami, Y.; Hayashi, A. A breakthrough in probiotics: Clostridium butyricum regulates gut homeostasis and anti-inflammatory response in inflammatory bowel disease. J. Gastroenterol. 2015, 50, 928–939. [Google Scholar] [CrossRef]
- Tomita, Y.; Ikeda, T.; Sakata, S.; Saruwatari, K.; Sato, R.; Iyama, S.; Jodai, T.; Akaike, K.; Ishizuka, S.; Saeki, S.; et al. Association of probiotic Clostridium butyricum therapy with survival and response to immune checkpoint blockade in patients with lung cancer. Cancer Immunol. Res. 2020, 8, 1236–1242. [Google Scholar] [CrossRef]
- Boltin, D.; Katzir, M.; Bugoslavsky, V.; Yalashvili, I.; Brosh-Nissimov, T.; Fried, M.; Elkayam, O. Corynebacterium striatum--a classic pathogen eluding diagnosis. Eur. J. Intern. Med. 2009, 20, e49–e52. [Google Scholar] [CrossRef]
- Krzyściak, W.; Pluskwa, K.K.; Jurczak, A.; Koscielniak, D. The pathogenicity of the Streptococcus genus. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1361–1376. [Google Scholar] [CrossRef]
- O’Gara, J.P.; Humphreys, H. Staphylococcus epidermidis biofilms: Importance and implications. J. Med. Microbiol. 2001, 50, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Miwa, T.; Taniguchi, H.; Nagano, T.; Shimarmura, K.; Tanaka, T.; Kumagai, H. Binding specificity of Lactobacillus to glycolipids. Biochem. Biophys. Res. Commun. 1996, 228, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Neeser, J.R.; Granato, D.; Rouvet, M.; Servin, A.; Teneberg, S.; Karlsson, K.-A. Lactobacillus johnsonii La1 shares carbohydrate-binding specificities with several enteropathogenic bacteria. Glycobiology 2000, 10, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Strömberg, N.; Ryd, M.; Lindberg, A.A.; Karlsson, K.-A. Studies on the binding of bacteria to glycolipids. Two species of Propionibacterium apparently recognize separate epitopes on lactose of lactosylceramide. FEBS Lett. 1988, 232, 193–198. [Google Scholar] [CrossRef]
- Fikry, A.M.; Attia, A.I.; Ismail, I.E.; Alagawany, M.; Reda, F.M. Dietary citric acid enhances growth performance, nutrient digestibility, intestinal microbiota, antioxidant status, and immunity of Japanese quails. Poult. Sci. 2021, 100, 101326. [Google Scholar] [CrossRef]
- Liu, F.; Li, P.; Chen, M.; Luo, Y.; Prabhakar, M.; Zheng, H.; He, Y.; Qi, Q.; Long, H.; Zhang, Y.; et al. Fructooligosaccharide (FOS) and Galactooligosaccharide (GOS) increase bifidobacterium but reduce butyrate producing bacteria with adverse glycemic metabolism in healthy young population. Sci. Rep. 2017, 7, 11789. [Google Scholar] [CrossRef]
- An, D.; Na, C.; Bielawski, J.; Hannun, Y.A.; Kasper, D.L. Membrane sphingolipids as essential molecular signals for Bacteroides survival in the intestine. Proc. Natl. Acad. Sci. USA 2011, 108, 4666–4671. [Google Scholar] [CrossRef]
- Yamashita, S.; Soga, M.; Nguma, E.; Kinoshita, M.; Miyazawa, T. Protective mechanism of rice-derived lipids and glucosylceramide in an in vitro intestinal tract model. J. Agric. Food Chem. 2021, 69, 10206–10214. [Google Scholar] [CrossRef]
- Patterson, L.; Allen, J.; Posey, I.; Shaw, J.J.P.; Costa-Pinheiro, P.; Walker, S.J.; Gademsey, A.; Wu, X.; Wu, S.; Zachos, N.C.; et al. Glucosylceramide production maintains colon integrity in response to Bacteroides fragilis toxin-induced colon epithelial cell signaling. FASEB J. 2020, 34, 15922–15945. [Google Scholar] [CrossRef]
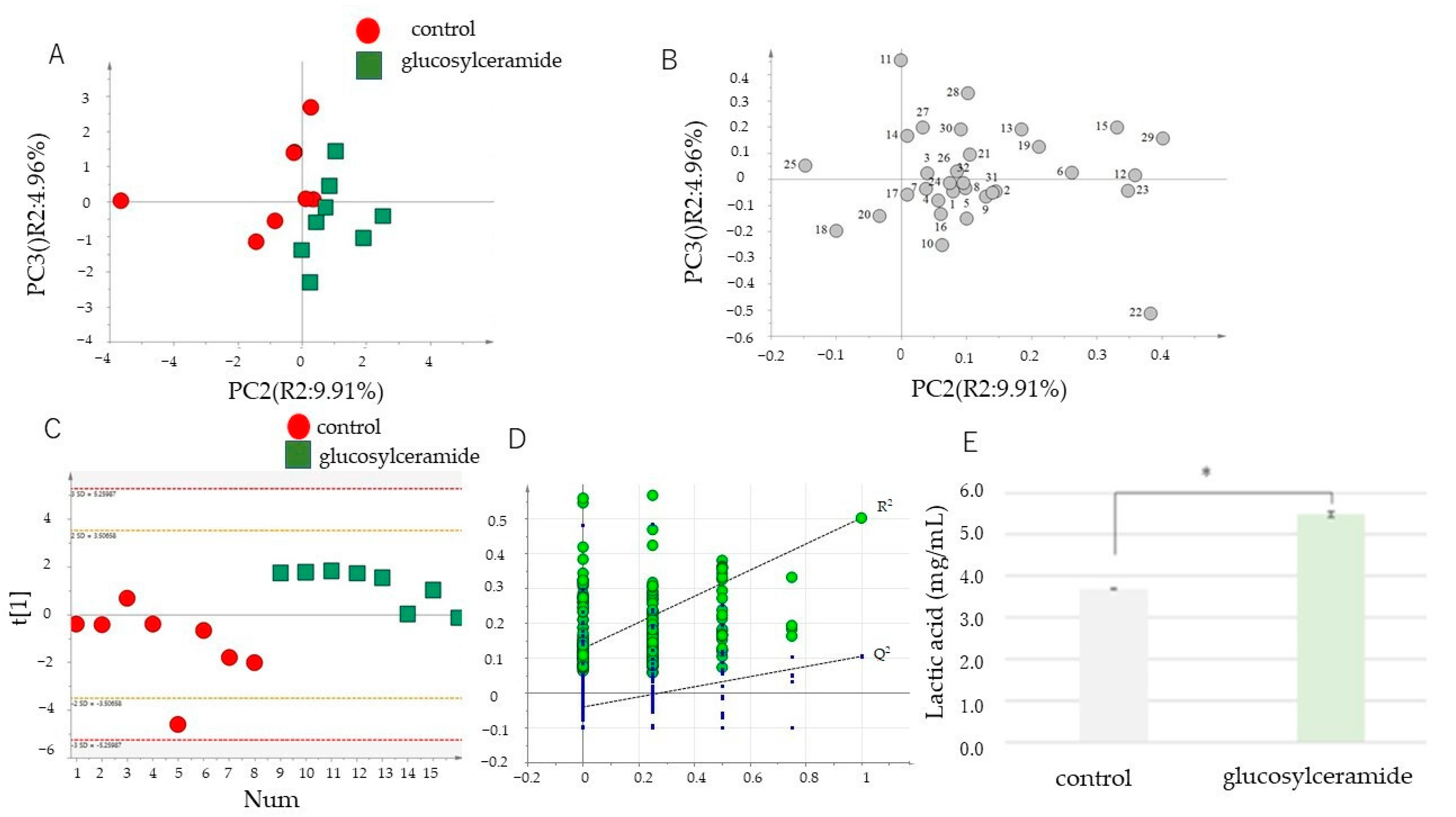
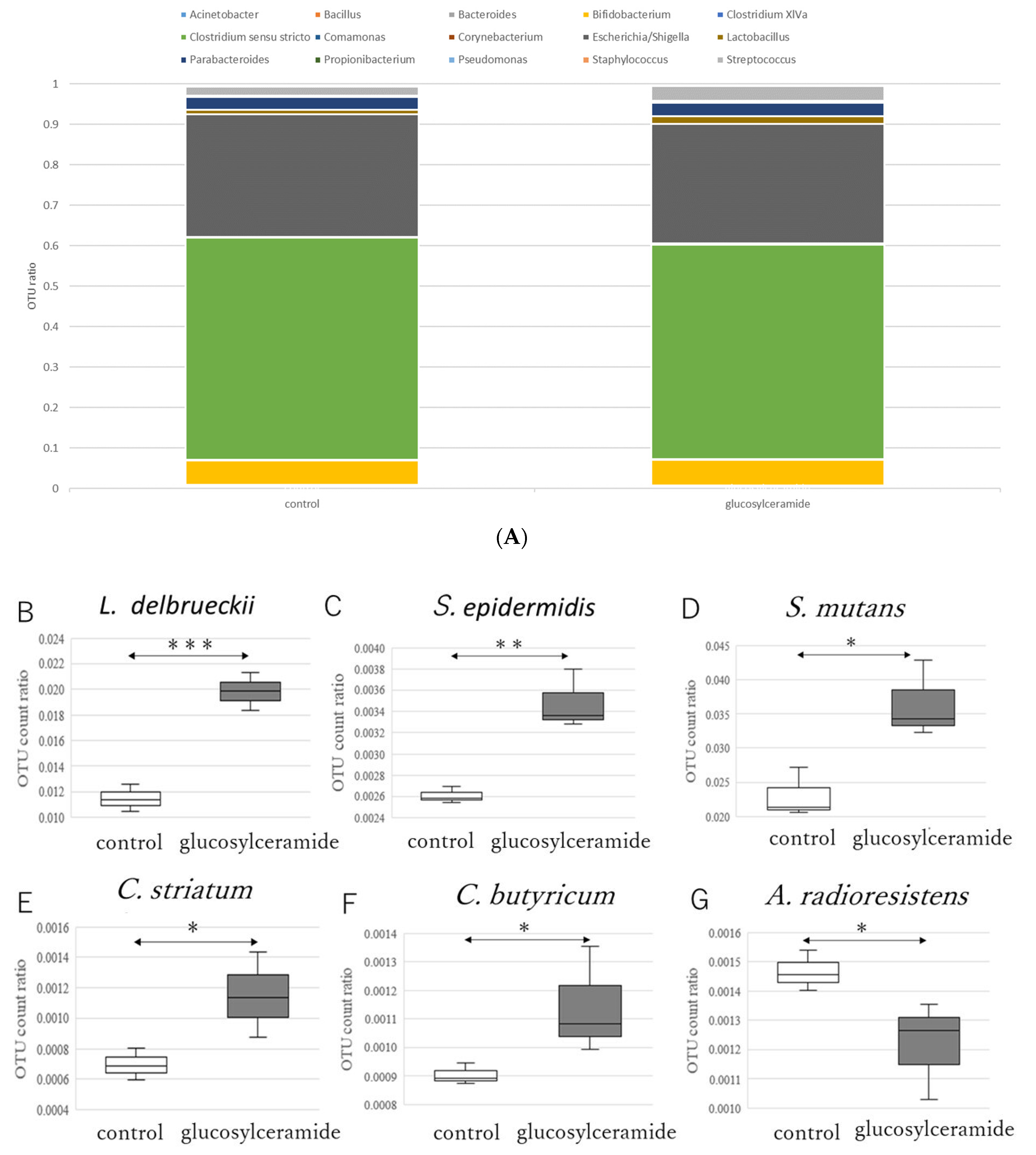
| No. | Retention Time (min) | Peak Name |
|---|---|---|
| 1 | 6.923 | Lactic acid |
| 2 | 6.994 | Unknown001 |
| 3 | 7.086 | Unknown002 |
| 4 | 7.595 | Alanine1 |
| 5 | 8.813 | Isoleucine1 |
| 6 | 9.191 | Valine2 |
| 7 | 11.896 | Methionine1 |
| 8 | 12.016 | Aspartic acid1 |
| 9 | 12.285 | Unknown003 |
| 10 | 13.244 | L-proline |
| 11 | 13.341 | Butanoic acid |
| 12 | 13.378 | Glutamic acid1 |
| 13 | 13.504 | Unknown004 |
| 14 | 13.613 | Phenylalanine1 |
| 15 | 13.797 | Unknown005 |
| 16 | 13.994 | Unknown006 |
| 17 | 14.398 | Glutamic acid2 |
| 18 | 14.494 | Phenylalanine2 |
| 19 | 14.819 | Unknown007 |
| - | 15.587 | Ribitol(IS) |
| 20 | 16.126 | Phosphoric acid |
| 21 | 16.835 | Unknown008 |
| 22 | 17.029 | Fructose1 |
| 23 | 17.142 | Unknown009 |
| 24 | 17.41 | Tyrosine |
| 25 | 17.595 | Unknown010 |
| 26 | 18.2 | Gluconic acid |
| 27 | 18.527 | Unknown011 |
| 28 | 19.364 | M-inosito1 |
| 29 | 20.277 | Tryptophan2 |
| 30 | 20.455 | Unknown012 |
| 31 | 20.672 | Unknown013 |
| 32 | 20.779 | Unknown014 |
| Peak Name | VIP Score | Coefficient |
|---|---|---|
| Citric acid | 3.18 | 0.227 ** |
| Threonine2 | 1.82 | −0.130 |
| Lysine1 | 1.69 | 0.121 |
| Unknown3 | 1.60 | 0.114 |
| Unknown1 | 1.51 | 0.108 |
| Lactic acid2 | 1.27 | 0.091 |
| Valine2 | 1.17 | 0.083 |
| Proline, isolecine1 | 1.11 | 0.079 |
| IC50 (mM) | |||
|---|---|---|---|
| Bacteria | Control | Glucosylceramide | p Value |
| L.delbrueckii | 1.17 ± 0.16 | 1.47 ± 0.07 | 0.078 |
| S. epidermidis | 0.343 ± 0.01 | 0.337 ± 0.00 | 0.131 |
| S. mutans | 0.175 ± 0.002 | 0.251 ± 0.014 | 0.003 ** |
| C. striatum | 0.369 ± 0.014 | 0.393 ± 0.020 | 0.197 |
| C. butyricum | 0.088 ± 0.003 | 0.117 ± 0.014 | 0.053 |
| E. coli | - | - | - |
| B. uniformis | 0.21 ± 0.02 | 0.22 ± 0.02 | 0.344 |
| B. coccoides | 0.67 ±0.07 | 0.81 ± 0.06 | 0.086 |
| E. faecalis | 0.06 ± 0.00 | 0.08 ± 0.00 | 0.007 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, H.; Otsuka, A.; Tanabe, K.; Yanagita, T.; Nakayama, J.; Kitagaki, H. Glucosylceramide Changes Bacterial Metabolism and Increases Gram-Positive Bacteria through Tolerance to Secondary Bile Acids In Vitro. Int. J. Mol. Sci. 2022, 23, 5300. https://doi.org/10.3390/ijms23105300
Dai H, Otsuka A, Tanabe K, Yanagita T, Nakayama J, Kitagaki H. Glucosylceramide Changes Bacterial Metabolism and Increases Gram-Positive Bacteria through Tolerance to Secondary Bile Acids In Vitro. International Journal of Molecular Sciences. 2022; 23(10):5300. https://doi.org/10.3390/ijms23105300
Chicago/Turabian StyleDai, Huanghuang, Akira Otsuka, Kurumi Tanabe, Teruyoshi Yanagita, Jiro Nakayama, and Hiroshi Kitagaki. 2022. "Glucosylceramide Changes Bacterial Metabolism and Increases Gram-Positive Bacteria through Tolerance to Secondary Bile Acids In Vitro" International Journal of Molecular Sciences 23, no. 10: 5300. https://doi.org/10.3390/ijms23105300
APA StyleDai, H., Otsuka, A., Tanabe, K., Yanagita, T., Nakayama, J., & Kitagaki, H. (2022). Glucosylceramide Changes Bacterial Metabolism and Increases Gram-Positive Bacteria through Tolerance to Secondary Bile Acids In Vitro. International Journal of Molecular Sciences, 23(10), 5300. https://doi.org/10.3390/ijms23105300







