Membrane Melatonin Receptors Activated Cell Signaling in Physiology and Disease
Abstract
:1. Introduction
2. Melatonin Receptors
2.1. Melatonin Receptor Subtypes—Classification and Physiological Significance
2.2. Melatonin Receptors MT1 and MT2 Belong to a Distinct Family of GPCR
2.3. MT1 and MT2 Functional Capacity Depends on the Formation of Homo- and Heterodimers
2.4. MT Receptors Are Widely Distributed in Central and Peripheral Tissue
2.5. MT1 and MT2 Receptor Sensitivity Is Regulated by GPCR Common Mechanisms and Circadian Fluctuation of Melatonin Concentration
3. MT1 and MT2 -Activated Signaling Pathways Are Involved in Physiological Processes
3.1. cAMP Signaling Pathway
3.2. PLC Signaling Pathway
3.3. Melatonin-Induced Activation of MEK/ERK Kinases
4. Do MT Signaling Matters to Human Reproductive Function?
5. What Is behind the Neuroimmunomodulatory Action of Melatonin
6. MT—Dependent Signaling of Cell Growth—An Important Clue for the Oncostatic Activity of Melatonin
7. Melatonin Receptors and Melatonin Receptor Polymorphisms—Clinical Impact
7.1. Diabetes Mellitus, Melatonin and Melatonin Receptors
7.1.1. Melatonin and Diabetes Mellitus
7.1.2. Melatonin Receptors, Pancreas and Diabetes Mellitus
7.1.3. Melatonin Receptor Polymorphisms and Glucose Abnormalities
7.2. Oncological Diseases, Melatonin and Melatonin Receptors
7.2.1. Melatonin, Melatonin Receptor Polymorphisms and Breast Cancer
7.2.2. Melatonin, Melatonin Receptor Polymorphisms and Other Malignancies
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, J.; Clough, S.J.; Hutchinson, A.J.; Adamah-Biassi, E.B.; Popovska-Gorevski, M.; Dubocovich, M.L. MT1 and MT2 Melatonin Receptors: A Therapeutic Perspective. Annu. Rev. Pharm. Toxicol. 2016, 56, 361–383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar Terron, M.; Flores, L.J.; Koppisepi, S. Medical Implications of Melatonin: Receptor-Mediated and Receptor-Independent Actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar]
- Waly, N.E.; Hallworth, R. Circadian Pattern of Melatonin MT1 and MT2 Receptor Localization in the Rat Suprachiasmatic Nucleus. J. Circadian Rhythm. 2015, 13, 1. [Google Scholar] [CrossRef] [Green Version]
- Arendt, J.; Deacon, S.; English, J.; Hampton, S.; Morgan, L. Melatonin and Adjustment to Phase Shift. J. Sleep Res. 1995, 4, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Vanecek, J. Melatonin Inhibits Release of Luteinizing Hormone (LH) via Decrease of [Ca2+]I and Cyclic AMP. Physiol. Res. 1998, 47, 329–335. [Google Scholar]
- Cecon, E.; Oishi, A.; Jockers, R. Melatonin Receptors: Molecular Pharmacology and Signalling in the Context of System Bias. Br. J. Pharm. 2018, 175, 3263–3280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reppert, S.M.; Weaver, D.R. Melatonin Madness. Cell 1995, 83, 1059–1062. [Google Scholar] [CrossRef] [Green Version]
- Dubocovich, M.L.; Delagrange, P.; Krause, D.N.; Sugden, D.; Cardinali, D.P.; Olcese, J. International Union of Basic and Clinical Pharmacology. LXXV. Nomenclature, Classification, and Pharmacology of G Protein-Coupled Melatonin Receptors. Pharm. Rev. 2010, 62, 343–380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubocovich, M.L.; Rivera-Bermudez, M.A.; Gerdin, M.J.; Masana, M.I. Molecular Pharmacology, Regulation and Function of Mammalian Melatonin Receptors. Front. Biosci. 2003, 8, D1093–D1108. [Google Scholar] [CrossRef] [Green Version]
- Pandi-Perumal, S.R.; Trakht, I.; Srinivasan, V.; Spence, D.W.; Maestroni, G.J.M.; Zisapel, N.; Cardinali, D.P. Physiological Effects of Melatonin: Role of Melatonin Receptors and Signal Transduction Pathways. Prog. Neurobiol. 2008, 85, 335–353. [Google Scholar] [CrossRef]
- Smirnov, A.N. Nuclear Melatonin Receptors. Biochemistry 2001, 66, 19–26. [Google Scholar] [CrossRef]
- Slominski, R.M.; Reiter, R.J.; Schlabritz-Loutsevitch, N.; Ostrom, R.S.; Slominski, A.T. Melatonin Membrane Receptors in Peripheral Tissues: Distribution and Functions. Mol. Cell. Endocrinol. 2012, 351, 152–166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reppert, S.M.; Weaver, D.R.; Ebisawa, T. Cloning and Characterization of a Mammalian Melatonin Receptor That Mediates Reproductive and Circadian Responses. Neuron 1994, 13, 1177–1185. [Google Scholar] [CrossRef]
- Browning, C.; Beresford, I.; Fraser, N.; Giles, H. Pharmacological Characterization of Human Recombinant Melatonin Mt(1) and MT(2) Receptors. Br. J. Pharm. 2000, 129, 877–886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubocovich, M.L.; Markowska, M. Functional MT1 and MT2 Melatonin Receptors in Mammals. Endocrine 2005, 27, 101–110. [Google Scholar] [CrossRef]
- Dubocovich, M.L. Melatonin Is a Potent Modulator of Dopamine Release in the Retina. Nature 1983, 306, 782–784. [Google Scholar] [CrossRef]
- Lerner, A.B.; Case, J.D.; Mori, W.; Wright, M.R. Melatonin in Peripheral Nerve. Nature 1959, 183, 1821. [Google Scholar] [CrossRef]
- Cardinali, D.P. Melatonin. A Pineal Modulatory Signal Affecting Pituitary Function. Prog. Clin. Biol. Res. 1981, 74, 179–198. [Google Scholar]
- Dubocovich, M.L. Pharmacology and Function of Melatonin Receptors. FASEB J. 1988, 2, 2765–2773. [Google Scholar] [CrossRef]
- Tauschanova, P.; Georgiev, G.; Manchev, S.; Konakchieva, R. Effect of Glucocorticoids on Melatonin Receptor Expression under T-Cell Activated Immune Response. Comptes Rendus L’Academie Bulg. Des Sci. 2007, 60, 899–902. [Google Scholar]
- Alexander, S.P.; Christopoulos, A.; Davenport, A.P.; Kelly, E.; Mathie, A.; Peters, J.A.; Veale, E.L.; Armstrong, J.F.; Faccenda, E.; Harding, S.D.; et al. THE concise guide to pharmacology 2021/22: G Protein-Coupled Receptors. Br. J. Pharm. 2021, 178 (Suppl. S1), S27–S156. [Google Scholar] [CrossRef]
- Cardinali, D.P. An Assessment of Melatonin’s Therapeutic Value in the Hypoxic-Ischemic Encephalopathy of the Newborn. Front. Synaptic Neurosci. 2019, 11, 34. [Google Scholar] [CrossRef] [Green Version]
- Tan, D.-X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Tamura, H.; Reiter, R.J. Melatonin as a Naturally Occurring Co-Substrate of Quinone Reductase-2, the Putative MT3 Melatonin Membrane Receptor: Hypothesis and Significance. J. Pineal Res. 2007, 43, 317–320. [Google Scholar] [CrossRef]
- Jockers, R.; Maurice, P.; Boutin, J.A.; Delagrange, P. Melatonin Receptors, Heterodimerization, Signal Transduction and Binding Sites: What’s New? Br. J. Pharm. 2008, 154, 1182–1195. [Google Scholar] [CrossRef] [Green Version]
- Levoye, A.; Dam, J.; Ayoub, M.A.; Guillaume, J.-L.; Couturier, C.; Delagrange, P.; Jockers, R. The Orphan GPR50 Receptor Specifically Inhibits MT1 Melatonin Receptor Function through Heterodimerization. EMBO J. 2006, 25, 3012–3023. [Google Scholar] [CrossRef]
- Dufourny, L.; Levasseur, A.; Migaud, M.; Callebaut, I.; Pontarotti, P.; Malpaux, B.; Monget, P. GPR50 Is the Mammalian Ortholog of Mel1c: Evidence of Rapid Evolution in Mammals. BMC Evol. Biol. 2008, 8, 105. [Google Scholar] [CrossRef] [Green Version]
- Witt-Enderby, P.A.; Bennett, J.; Jarzynka, M.J.; Firestine, S.; Melan, M.A. Melatonin Receptors and Their Regulation: Biochemical and Structural Mechanisms. Life Sci. 2003, 72, 2183–2198. [Google Scholar] [CrossRef]
- Masana, M.I.; Dubocovich, M.L. Melatonin Receptor Signaling: Finding the Path through the Dark. Sci. STKE 2001, 2001, pe39. [Google Scholar] [CrossRef] [PubMed]
- Brydon, L.; Barrett, P.; Morgan, P.J.; Strosberg, A.D.; Jockers, R. Investigation of the Human Mel 1a Melatonin Receptor Using Anti-Receptor Antibodies. Adv. Exp. Med. Biol. 1999, 460, 215–220. [Google Scholar] [CrossRef]
- Chan, A.S.L.; Lai, F.P.L.; Lo, R.K.H.; Voyno-Yasenetskaya, T.A.; Stanbridge, E.J.; Wong, Y.H. Melatonin Mt1 and MT2 Receptors Stimulate C-Jun N-Terminal Kinase via Pertussis Toxin-Sensitive and -Insensitive G Proteins. Cell. Signal. 2002, 14, 249–257. [Google Scholar] [CrossRef]
- Petit, L.; Lacroix, I.; de Coppet, P.; Strosberg, A.D.; Jockers, R. Differential Signaling of Human Mel1a and Mel1b Melatonin Receptors through the Cyclic Guanosine 3’-5’-Monophosphate Pathway. Biochem. Pharm. 1999, 58, 633–639. [Google Scholar] [CrossRef]
- Chen, L.; He, X.; Zhang, Y.; Chen, X.; Lai, X.; Shao, J.; Shi, Y.; Zhou, N. Melatonin Receptor Type 1 Signals to Extracellular Signal-Regulated Kinase 1 and 2 via Gi and Gs Dually Coupled Pathways in HEK-293 Cells. Biochemistry 2014, 53, 2827–2839. [Google Scholar] [CrossRef] [PubMed]
- Arnis, S.; Fahmy, K.; Hofmann, K.P.; Sakmar, T.P. A Conserved Carboxylic Acid Group Mediates Light-Dependent Proton Uptake and Signaling by Rhodopsin. J. Biol. Chem. 1994, 269, 23879–23881. [Google Scholar] [CrossRef]
- Ballesteros, J.; Palczewski, K. G Protein-Coupled Receptor Drug Discovery: Implications from the Crystal Structure of Rhodopsin. Curr. Opin. Drug Discov. Devel. 2001, 4, 561–574. [Google Scholar] [PubMed]
- Conway, S.; Mowat, E.S.; Drew, J.E.; Barrett, P.; Delagrange, P.; Morgan, P.J. Serine Residues 110 and 114 Are Required for Agonist Binding but Not Antagonist Binding to the Melatonin MT(1) Receptor. Biochem. Biophys. Res. Commun. 2001, 282, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Ward, S.D.C.; Hamdan, F.F.; Bloodworth, L.M.; Siddiqui, N.A.; Li, J.H.; Wess, J. Use of an in Situ Disulfide Cross-Linking Strategy to Study the Dynamic Properties of the Cytoplasmic End of Transmembrane Domain VI of the M3 Muscarinic Acetylcholine Receptor. Biochemistry 2006, 45, 676–685. [Google Scholar] [CrossRef]
- Noda, K.; Saad, Y.; Graham, R.M.; Karnik, S.S. The High Affinity State of the Beta 2-Adrenergic Receptor Requires Unique Interaction between Conserved and Non-Conserved Extracellular Loop Cysteines. J. Biol. Chem. 1994, 269, 6743–6752. [Google Scholar] [CrossRef]
- Mseeh, F.; Gerdin, M.J.; Dubocovich, M.I. Identification of Cysteines Involved in Ligand Binding to the Human Melatonin MT(2) Receptor. Eur. J. Pharm. 2002, 449, 29–38. [Google Scholar] [CrossRef]
- Gerdin, M.J.; Mseeh, F.; Dubocovich, M.L. Mutagenesis Studies of the Human MT2 Melatonin Receptor. Biochem. Pharm. 2003, 66, 315–320. [Google Scholar] [CrossRef]
- Barrett, P.; Conway, S.; Morgan, P.J. Digging Dee–-Structure-Function Relationships in the Melatonin Receptor Family. J. Pineal. Res. 2003, 35, 221–230. [Google Scholar] [CrossRef]
- Ayoub, M.A.; Couturier, C.; Lucas-Meunier, E.; Angers, S.; Fossier, P.; Bouvier, M.; Jockers, R. Monitoring of Ligand-Independent Dimerization and Ligand-Induced Conformational Changes of Melatonin Receptors in Living Cells by Bioluminescence Resonance Energy Transfer. J. Biol. Chem. 2002, 277, 21522–21528. [Google Scholar] [CrossRef] [Green Version]
- Ayoub, M.A.; Levoye, A.; Delagrange, P.; Jockers, R. Preferential Formation of MT1/MT2 Melatonin Receptor Heterodimers with Distinct Ligand Interaction Properties Compared with MT2 Homodimers. Mol. Pharm. 2004, 66, 312–321. [Google Scholar] [CrossRef]
- Oishi, A.; Cecon, E.; Jockers, R. Melatonin Receptor Signaling: Impact of Receptor Oligomerization on Receptor Function. Int. Rev. Cell. Mol. Biol. 2018, 338, 59–77. [Google Scholar] [CrossRef]
- Baba, K.; Benleulmi-Chaachoua, A.; Journé, A.-S.; Kamal, M.; Guillaume, J.-L.; Dussaud, S.; Gbahou, F.; Yettou, K.; Liu, C.; Contreras-Alcantara, S.; et al. Heteromeric MT1/MT2 Melatonin Receptors Modulate Photoreceptor Function. Sci. Signal. 2013, 6, ra89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morgan, P.J.; Mercer, J.G. Control of Seasonality by Melatonin. Proc. Nutr. Soc. 1994, 53, 483–493. [Google Scholar] [CrossRef] [Green Version]
- Konakchieva, R.; Kyurkchiev, S.; Kehayov, I.; Taushanova, P.; Kanchev, L. Selective Effect of Methoxyindoles on the Lymphocyte Proliferation and Melatonin Binding to Activated Human Lymphoid Cells. J. Neuroimmunol. 1995, 63, 125–132. [Google Scholar] [CrossRef]
- Georgiev, G.N.; Mourdjeva, M.; Oreshkova, T.; Pankov, R.; Konakchieva, R. MT1 and MT2 Melatonin Receptor Expression and in Vitro Melatonin Effect on the Pha-Dependent Activation of Human PBMC. Comptes Rendus L’Academie Bulg. Des Sci. 2019, 72, 1500–1506. [Google Scholar] [CrossRef]
- Dimitrov, R.; Georgiev, G.; Todorov, P.; Dimitrov, Y.; Konakchieva, R. Membrane Melatonin Receptor Type MT1 Expression in Human Ejaculated Spermatozoa. Comptes Rendus L’Academie Bulg. Des Sci. 2012, 65, 947–952. [Google Scholar]
- Reppert, S.M.; Godson, C.; Mahle, C.D.; Weaver, D.R.; Slaugenhaupt, S.A.; Gusella, J.F. Molecular Characterization of a Second Melatonin Receptor Expressed in Human Retina and Brain: The Mel1b Melatonin Receptor. Proc. Natl. Acad. Sci. USA 1995, 92, 8734–8738. [Google Scholar] [CrossRef] [Green Version]
- Slominski, A.; Fischer, T.W.; Zmijewski, M.A.; Wortsman, J.; Semak, I.; Zbytek, B.; Slominski, R.M.; Tobin, D.J. On the Role of Melatonin in Skin Physiology and Pathology. Endocrine 2005, 27, 137–148. [Google Scholar] [CrossRef] [Green Version]
- Ferguson, S.S. Evolving Concepts in G Protein-Coupled Receptor Endocytosis: The Role in Receptor Desensitization and Signaling. Pharm. Rev. 2001, 53, 1–24. [Google Scholar] [PubMed]
- Reppert, S.M.; Weaver, D.R.; Ebisawa, T.; Mahle, C.D.; Kolakowski, L.F. Cloning of a Melatonin-Related Receptor from Human Pituitary. FEBS Lett. 1996, 386, 219–224. [Google Scholar] [CrossRef] [Green Version]
- Dubocovich, M.L.; Masana, M.I.; Iacob, S.; Sauri, D.M. Melatonin Receptor Antagonists That Differentiate between the Human Mel1a and Mel1b Recombinant Subtypes Are Used to Assess the Pharmacological Profile of the Rabbit Retina ML1 Presynaptic Heteroreceptor. Naunyn Schmiedebergs Arch. Pharm. 1997, 355, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Dollins, A.B.; Zhdanova, I.V.; Wurtman, R.J.; Lynch, H.J.; Deng, M.H. Effect of Inducing Nocturnal Serum Melatonin Concentrations in Daytime on Sleep, Mood, Body Temperature, and Performance. Proc. Natl. Acad. Sci. USA 1994, 91, 1824–1828. [Google Scholar] [CrossRef] [Green Version]
- Mulchahey, J.J.; Goldwater, D.R.; Zemlan, F.P. A Single Blind, Placebo Controlled, across Groups Dose Escalation Study of the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of the Melatonin Analog Beta-Methyl-6-Chloromelatonin. Life Sci. 2004, 75, 1843–1856. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Ushinohama, K.; Ikesue, S.; Yukawa, E.; Higuchi, S.; Hamase, K.; Zaitsu, K.; Ohdo, S. Chronopharmacology of Melatonin in Mice to Maximize the Antitumor Effect and Minimize the Rhythm Disturbance Effect. J. Pharm. Exp. Ther. 2004, 308, 378–384. [Google Scholar] [CrossRef] [Green Version]
- Radio, N.M.; Doctor, J.S.; Witt-Enderby, P.A. Melatonin Enhances Alkaline Phosphatase Activity in Differentiating Human Adult Mesenchymal Stem Cells Grown in Osteogenic Medium via MT2 Melatonin Receptors and the MEK/ERK (1/2) Signaling Cascade. J. Pineal Res. 2006, 40, 332–342. [Google Scholar] [CrossRef]
- Gauer, F.; Masson-Pevet, M.; Stehle, J.; Pevet, P. Daily Variations in Melatonin Receptor Density of Rat Pars Tuberalis and Suprachiasmatic Nuclei Are Distinctly Regulated. Brain Res. 1994, 641, 92–98. [Google Scholar] [CrossRef]
- Schuster, C.; Gauer, F.; Malan, A.; Recio, J.; Pévet, P.; Masson-Pévet, M. The Circadian Clock, Light/Dark Cycle and Melatonin Are Differentially Involved in the Expression of Daily and Photoperiodic Variations in Mt(1) Melatonin Receptors in the Siberian and Syrian Hamsters. Neuroendocrinology 2001, 74, 55–68. [Google Scholar] [CrossRef]
- Gauer, F.; Schuster, C.; Poirel, V.J.; Pevet, P.; Masson-Pevet, M. Developmental Expression of Both Melatonin Receptor Mt1 MRNA and Melatonin Binding Sites in Syrian Hamster Suprachiasmatic Nuclei. Adv. Exp. Med. Biol. 1999, 460, 271–278. [Google Scholar] [CrossRef]
- Konakchieva, R.; Manchev, S.; Pevét, P.; Masson-Pevét, M. Autoradiographic Detection of 2-(125I)-Iodomelatonin Binding Sites in Immune Tissue of Rats. Adv. Exp. Med. Biol. 1999, 460, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, J.M.; Pozo, D.; García-Mauriño, S.; Osuna, C.; Molinero, P.; Calvo, J.R. Involvement of Nuclear Receptors in the Enhanced IL-2 Production by Melatonin in Jurkat Cells. Ann. N. Y. Acad. Sci. 2000, 917, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Tomova, A.; Kumanov, P.; Robeva, R.; Manchev, S.; Konakchieva, R. Melatonin Secretion and Non-Specific Immune Responses Are Differentially Expressed in Corticotropin-Dependent and Corticotropin-Independent Cushing’s Syndrome. Med. Sci. Monit. 2008, 14, CR327–CR332. [Google Scholar] [PubMed]
- New, D.C.; Tsim, S.T.; Wong, Y.H. G Protein-Linked Effector and Second Messenger Systems Involved in Melatonin Signal Transduction. Neurosignals 2003, 12, 59–70. [Google Scholar] [CrossRef]
- Travnickova-Bendova, Z.; Cermakian, N.; Reppert, S.M.; Sassone-Corsi, P. Bimodal Regulation of mPeriod Promoters by CREB-Dependent Signaling and CLOCK/BMAL1 Activity. Proc. Natl. Acad. Sci. USA 2002, 99, 7728–7733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, X.; von Gall, C.; Pieschl, R.L.; Gribkoff, V.K.; Stehle, J.H.; Reppert, S.M.; Weaver, D.R. Targeted Disruption of the Mouse Mel(1b) Melatonin Receptor. Mol. Cell. Biol. 2003, 23, 1054–1060. [Google Scholar] [CrossRef] [Green Version]
- Witt-Enderby, P.A.; MacKenzie, R.S.; McKeon, R.M.; Carroll, E.A.; Bordt, S.L.; Melan, M.A. Melatonin Induction of Filamentous Structures in Non-Neuronal Cells That Is Dependent on Expression of the Human Mt1 Melatonin Receptor. Cell Motil. Cytoskelet. 2000, 46, 28–42. [Google Scholar] [CrossRef]
- Lépinay, J.; Taragnat, C.; Dubois, J.-P.; Chesneau, D.; Jockers, R.; Delagrange, P.; Bozon, V. Negative Regulation of Melatonin Secretion by Melatonin Receptors in Ovine Pinealocytes. PLoS ONE 2021, 16, e0255249. [Google Scholar] [CrossRef]
- Chen, M.; Cecon, E.; Karamitri, A.; Gao, W.; Gerbier, R.; Ahmad, R.; Jockers, R. Melatonin MT1 and MT2 Receptor ERK Signaling Is Differentially Dependent on Gi/o and Gq/11 Proteins. J. Pineal Res. 2020, 68, e12641. [Google Scholar] [CrossRef]
- Georgiev, G.N.; Marinova, E.; Konakchieva, R.; Todorov, P. Melatonin Selectively Influences the Transcription of Pluripotency and Differentiation Markers in Human Non-Cancer Cells. Biotechnol. Biotechnol. Equip. 2019, 33, 286–293. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Meng, K.; He, Y.; Wang, H.; Zhang, Y.; Quan, F. Melatonin Stimulates STAR Expression and Progesterone Production via Activation of the PI3K/AKT Pathway in Bovine Theca Cells. Int. J. Biol. Sci. 2019, 15, 404–415. [Google Scholar] [CrossRef]
- Shu, T.; Wu, T.; Pang, M.; Liu, C.; Wang, X.; Wang, J.; Liu, B.; Rong, L. Effects and Mechanisms of Melatonin on Neural Differentiation of Induced Pluripotent Stem Cells. Biochem. Biophys. Res. Commun. 2016, 474, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Song, C.; Zhang, J.; Zhao, J.; Fu, B.; Mao, T.; Zhang, Y. Melatonin-Mediated Upregulation of GLUT1 Blocks Exit from Pluripotency by Increasing the Uptake of Oxidized Vitamin C in Mouse Embryonic Stem Cells. FASEB J. 2017, 31, 1731–1743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stazi, M.; Negro, S.; Megighian, A.; D’Este, G.; Solimena, M.; Jockers, R.; Lista, F.; Montecucco, C.; Rigoni, M. Melatonin Promotes Regeneration of Injured Motor Axons via MT1 Receptors. J. Pineal Res. 2021, 70, e12695. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.-H.; Tung, Y.-T.; Chen, H.-Y.; Chiang, Y.-F.; Hong, H.-C.; Huang, K.-C.; Hsu, S.-P.; Huang, T.-C.; Hsia, S.-M. Melatonin Activates Cell Death Programs for the Suppression of Uterine Leiomyoma Cell Proliferation. J. Pineal Res. 2020, 68, e12620. [Google Scholar] [CrossRef] [PubMed]
- Pang, S.F.; Pang, C.S.; Poon, A.M.; Lee, P.P.; Liu, Z.M.; Shiu, S.Y. Melatonin: A Chemical Photoperiodic Signal with Clinical Significance in Humans. Chin. Med. J. 1998, 111, 197–203. [Google Scholar] [PubMed]
- Díaz López, B.; Díaz Rodríguez, E.; Urquijo, C.; Fernández Alvarez, C. Melatonin Influences on the Neuroendocrine-Reproductive Axis. Ann. N. Y. Acad. Sci. 2005, 1057, 337–364. [Google Scholar] [CrossRef]
- Woo, M.M.; Tai, C.J.; Kang, S.K.; Nathwani, P.S.; Pang, S.F.; Leung, P.C. Direct Action of Melatonin in Human Granulosa-Luteal Cells. J. Clin. Endocrinol. Metab. 2001, 86, 4789–4797. [Google Scholar] [CrossRef]
- Yie, S.M.; Brown, G.M.; Liu, G.Y.; Collins, J.A.; Daya, S.; Hughes, E.G.; Foster, W.G.; Younglai, E.V. Melatonin and Steroids in Human Pre-Ovulatory Follicular Fluid: Seasonal Variations and Granulosa Cell Steroid Production. Hum. Reprod. 1995, 10, 50–55. [Google Scholar] [CrossRef]
- Brzezinski, A.; Fibich, T.; Cohen, M.; Schenker, J.G.; Laufer, N. Effects of Melatonin on Progesterone Production by Human Granulosa Lutein Cells in Culture. Fertil. Steril. 1992, 58, 526–529. [Google Scholar] [CrossRef]
- Niles, L.P.; Wang, J.; Shen, L.; Lobb, D.K.; Younglai, E.V. Melatonin Receptor MRNA Expression in Human Granulosa Cells. Mol. Cell. Endocrinol. 1999, 156, 107–110. [Google Scholar] [CrossRef]
- Rönnberg, L.; Kauppila, A.; Leppäluoto, J.; Martikainen, H.; Vakkuri, O. Circadian and Seasonal Variation in Human Preovulatory Follicular Fluid Melatonin Concentration. J. Clin. Endocrinol. Metab. 1990, 71, 492–496. [Google Scholar] [CrossRef]
- Tamura, H.; Takasaki, A.; Taketani, T.; Tanabe, M.; Kizuka, F.; Lee, L.; Tamura, I.; Maekawa, R.; Aasada, H.; Yamagata, Y.; et al. The Role of Melatonin as an Antioxidant in the Follicle. J. Ovarian Res. 2012, 5, 5. [Google Scholar] [CrossRef] [Green Version]
- Itoh, M.T.; Ishizuka, B.; Kuribayashi, Y.; Amemiya, A.; Sumi, Y. Melatonin, Its Precursors, and Synthesizing Enzyme Activities in the Human Ovary. Mol. Hum. Reprod. 1999, 5, 402–408. [Google Scholar] [CrossRef] [Green Version]
- Adriaens, I.; Jacquet, P.; Cortvrindt, R.; Janssen, K.; Smitz, J. Melatonin Has Dose-Dependent Effects on Folliculogenesis, Oocyte Maturation Capacity and Steroidogenesis. Toxicology 2006, 228, 333–343. [Google Scholar] [CrossRef]
- Tanavde, V.S.; Maitra, A. In Vitro Modulation of Steroidogenesis and Gene Expression by Melatonin: A Study with Porcine Antral Follicles. Endocr. Res. 2003, 29, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Mediavilla, M.D.; Sanchez-Barcelo, E.J.; Tan, D.X.; Manchester, L.; Reiter, R.J. Basic Mechanisms Involved in the Anti-Cancer Effects of Melatonin. Curr. Med. Chem. 2010, 17, 4462–4481. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.-K.; Lee, J.-H.; Moon, J.-H.; Lee, Y.-J.; Park, S.-Y. Melatonin-Mediated β-Catenin Activation Protects Neuron Cells against Prion Protein-Induced Neurotoxicity. J. Pineal Res. 2014, 57, 427–434. [Google Scholar] [CrossRef]
- Parakh, T.N.; Hernandez, J.A.; Grammer, J.C.; Weck, J.; Hunzicker-Dunn, M.; Zeleznik, A.J.; Nilson, J.H. Follicle-Stimulating Hormone/CAMP Regulation of Aromatase Gene Expression Requires Beta-Catenin. Proc. Natl. Acad. Sci. USA 2006, 103, 12435–12440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikolov, G.; Georgiev, G.N.; Marinova, E.; Mourdjeva, M.; Konakchieva, R. UP-Regulation of MT1 and MT2 Receptors by in Vitro Melatonin and Modulation of Alpha-Tubulin and Aromatase P450 Expression in Human Granulosa-Lutein Cells. Comptes Rendus L’Academie Bulg. Des Sci. 2020, 73, 348–354. [Google Scholar] [CrossRef]
- Huerto-Delgadillo, L.; Antón-Tay, F.; Benítez-King, G. Effects of Melatonin on Microtubule Assembly Depend on Hormone Concentration: Role of Melatonin as a Calmodulin Antagonist. J. Pineal Res. 1994, 17, 55–62. [Google Scholar] [CrossRef]
- Benítez-King, G. Melatonin as a Cytoskeletal Modulator: Implications for Cell Physiology and Disease. J. Pineal Res. 2006, 40, 1–9. [Google Scholar] [CrossRef]
- Jarzynka, M.J.; Passey, D.K.; Johnson, D.A.; Konduru, N.V.; Fitz, N.F.; Radio, N.M.; Rasenick, M.; Benloucif, S.; Melan, M.A.; Witt-Enderby, P.A. Microtubules Modulate Melatonin Receptors Involved in Phase-Shifting Circadian Activity Rhythms: In Vitro and in Vivo Evidence. J. Pineal Res. 2009, 46, 161–171. [Google Scholar] [CrossRef] [Green Version]
- Yan, K.; Popova, J.S.; Moss, A.; Shah, B.; Rasenick, M.M. Tubulin Stimulates Adenylyl Cyclase Activity in C6 Glioma Cells by Bypassing the Beta-Adrenergic Receptor: A Potential Mechanism of G Protein Activation. J. Neurochem. 2001, 76, 182–190. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Mayo, J.C.; Sainz, R.M.; Leon, J.; Czarnocki, Z. Melatonin as an Antioxidant: Biochemical Mechanisms and Pathophysiological Implications in Humans. Acta Biochim. Pol. 2003, 50, 1129–1146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espino, J.; Ortiz, Á.; Bejarano, I.; Lozano, G.M.; Monllor, F.; García, J.F.; Rodríguez, A.B.; Pariente, J.A. Melatonin Protects Human Spermatozoa from Apoptosis via Melatonin Receptor- and Extracellular Signal-Regulated Kinase-Mediated Pathways. Fertil. Steril. 2011, 95, 2290–2296. [Google Scholar] [CrossRef] [PubMed]
- Konakchieva, R.; Todorov, P. Melatonin Protects Human Spermatozoa from Apoptosis via Melatonin Receptor- and Extracellular Signal-Regulated Kinase-Mediated Pathways. Fertil. Steril. 2011, 96, e159. [Google Scholar] [CrossRef]
- Fujinoki, M. Melatonin-Enhanced Hyperactivation of Hamster Sperm. Reproduction 2008, 136, 533–541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luboshitzky, R.; Shen-Orr, Z.; Herer, P. Seminal Plasma Melatonin and Gonadal Steroids Concentrations in Normal Men. Arch. Androl. 2002, 48, 225–232. [Google Scholar] [CrossRef] [Green Version]
- Van Vuuren, R.J.; Pitout, M.J.; van Aswegen, C.H.; Theron, J.J. Putative Melatonin Receptor in Human Spermatozoa. Clin. Biochem. 1992, 25, 125–127. [Google Scholar] [CrossRef]
- Dadoune, J.P.; Pawlak, A.; Alfonsi, M.F.; Siffroi, J.P. Identification of Transcripts by Macroarrays, RT-PCR and in Situ Hybridization in Human Ejaculate Spermatozoa. Mol. Hum. Reprod. 2005, 11, 133–140. [Google Scholar] [CrossRef]
- Calvo, J.R.; González-Yanes, C.; Maldonado, M.D. The Role of Melatonin in the Cells of the Innate Immunity: A Review. J. Pineal Res. 2013, 55, 103–120. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Reiter, R.J.; Lardone, P.J.; Herrera, J.L.; Fernández-Montesinos, R.; Guerrero, J.M.; Pozo, D. The Modulatory Role of Melatonin on Immune Responsiveness. Curr. Opin. Investig. Drugs 2006, 7, 423–431. [Google Scholar]
- Dubocovich, M.L. Melatonin Receptors: Are There Multiple Subtypes? Trends Pharm. Sci. 1995, 16, 50–56. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; García-Mauriño, S.; Calvo, J.R.; Guerrero, J.M. Melatonin Counteracts the Inhibitory Effect of PGE2 on IL-2 Production in Human Lymphocytes via Its Mt1 Membrane Receptor. FASEB J. 2003, 17, 755–757. [Google Scholar] [CrossRef]
- Lardone, P.J.; Carrillo-Vico, A.; Naranjo, M.C.; De Felipe, B.; Vallejo, A.; Karasek, M.; Guerrero, J.M. Melatonin Synthesized by Jurkat Human Leukemic T Cell Line Is Implicated in IL-2 Production. J. Cell. Physiol. 2006, 206, 273–279. [Google Scholar] [CrossRef]
- Calvo, J.R.; Rafii-el-Idrissi, M.; Pozo, D.; Guerrero, J.M. Immunomodulatory Role of Melatonin: Specific Binding Sites in Human and Rodent Lymphoid Cells. J. Pineal Res. 1995, 18, 119–126. [Google Scholar] [CrossRef] [PubMed]
- García-Pergañeda, A.; Pozo, D.; Guerrero, J.M.; Calvo, J.R. Signal Transduction for Melatonin in Human Lymphocytes: Involvement of a Pertussis Toxin-Sensitive G Protein. J. Immunol. 1997, 159, 3774–3781. [Google Scholar]
- Barjavel, M.J.; Mamdouh, Z.; Raghbate, N.; Bakouche, O. Differential Expression of the Melatonin Receptor in Human Monocytes. J. Immunol. 1998, 160, 1191–1197. [Google Scholar]
- García-Pergañeda, A.; Guerrero, J.M.; Rafii-El-Idrissi, M.; Paz Romero, M.; Pozo, D.; Calvo, J.R. Characterization of Membrane Melatonin Receptor in Mouse Peritoneal Macrophages: Inhibition of Adenylyl Cyclase by a Pertussis Toxin-Sensitive G Protein. J. Neuroimmunol. 1999, 95, 85–94. [Google Scholar] [CrossRef]
- Pozo, D.; Delgado, M.; Fernandez-Santos, J.M.; Calvo, J.R.; Gomariz, R.P.; Martin-Lacave, I.; Ortiz, G.G.; Guerrero, J.M. Expression of the Mel1a-Melatonin Receptor MRNA in T and B Subsets of Lymphocytes from Rat Thymus and Spleen. FASEB J. 1997, 11, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Drazen, D.L.; Bilu, D.; Bilbo, S.D.; Nelson, R.J. Melatonin Enhancement of Splenocyte Proliferation Is Attenuated by Luzindole, a Melatonin Receptor Antagonist. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 280, R1476–R1482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markowska, M.; Mrozkowiak, A.; Pawlak, J.; Skwarło-Sońta, K. Intracellular Second Messengers Involved in Melatonin Signal Transduction in Chicken Splenocytes in Vitro. J. Pineal Res. 2004, 37, 207–212. [Google Scholar] [CrossRef]
- Espino, J.; Rodríguez, A.B.; Pariente, J.A. The Inhibition of TNF-α-Induced Leucocyte Apoptosis by Melatonin Involves Membrane Receptor MT1/MT2 Interaction. J. Pineal Res. 2013, 54, 442–452. [Google Scholar] [CrossRef] [PubMed]
- Radogna, F.; Cristofanon, S.; Paternoster, L.; D’Alessio, M.; De Nicola, M.; Cerella, C.; Dicato, M.; Diederich, M.; Ghibelli, L. Melatonin Antagonizes the Intrinsic Pathway of Apoptosis via Mitochondrial Targeting of Bcl-2. J. Pineal Res. 2008, 44, 316–325. [Google Scholar] [CrossRef]
- Radogna, F.; Paternoster, L.; Albertini, M.C.; Cerella, C.; Accorsi, A.; Bucchini, A.; Spadoni, G.; Diamantini, G.; Tarzia, G.; De Nicola, M.; et al. Melatonin Antagonizes Apoptosis via Receptor Interaction in U937 Monocytic Cells. J. Pineal Res. 2007, 43, 154–162. [Google Scholar] [CrossRef]
- Luchetti, F.; Betti, M.; Canonico, B.; Arcangeletti, M.; Ferri, P.; Galli, F.; Papa, S. ERK MAPK Activation Mediates the Antiapoptotic Signaling of Melatonin in UVB-Stressed U937 Cells. Free Radic. Biol. Med. 2009, 46, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Weaver, D.R.; Stehle, J.H.; Stopa, E.G.; Reppert, S.M. Melatonin Receptors in Human Hypothalamus and Pituitary: Implications for Circadian and Reproductive Responses to Melatonin. J. Clin. Endocrinol. Metab. 1993, 76, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Tam, P.C.; Poon, A.M.; Brown, G.M.; Pang, S.F. 2-[125I]Iodomelatonin-Binding Sites in the Human Kidney and the Effect of Guanosine 5’-O-(3-Thiotriphosphate). J. Clin. Endocrinol. Metab. 1995, 80, 1560–1565. [Google Scholar] [CrossRef]
- Conway, S.; Drew, J.E.; Canning, S.J.; Barrett, P.; Jockers, R.; Strosberg, A.D.; Guardiola-Lemaitre, B.; Delagrange, P.; Morgan, P.J. Identification of Mel1a Melatonin Receptors in the Human Embryonic Kidney Cell Line HEK293: Evidence of G Protein-Coupled Melatonin Receptors Which Do Not Mediate the Inhibition of Stimulated Cyclic AMP Levels. FEBS Lett. 1997, 407, 121–126. [Google Scholar] [CrossRef]
- Collins, A.R.; Annangi, B.; Rubio, L.; Marcos, R.; Dorn, M.; Merker, C.; Estrela-Lopis, I.; Cimpan, M.R.; Ibrahim, M.; Cimpan, E.; et al. High Throughput Toxicity Screening and Intracellular Detection of Nanomaterials. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1413. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, M.; Reiter, R.J.; Meltz, M.L.; Herman, T.S. Effect of Melatonin on Cell Growth, Metabolic Activity, and Cell Cycle Distribution. J. Pineal Res. 2001, 31, 228–233. [Google Scholar] [CrossRef]
- Martín-Renedo, J.; Mauriz, J.L.; Jorquera, F.; Ruiz-Andrés, O.; González, P.; González-Gallego, J. Melatonin Induces Cell Cycle Arrest and Apoptosis in Hepatocarcinoma HepG2 Cell Line. J. Pineal Res. 2008, 45, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Madrid, J.A.; Tan, D.-X.; Reiter, R.J. Melatonin, the Circadian Multioscillator System and Health: The Need for Detailed Analyses of Peripheral Melatonin Signaling. J. Pineal Res. 2012, 52, 139–166. [Google Scholar] [CrossRef]
- Kepka, M.; Szwejser, E.; Pijanowski, L.; Verburg-van Kemenade, B.M.; Chadzinska, M. A Role for Melatonin in Maintaining the Pro- and Anti-Inflammatory Balance by Influencing Leukocyte Migration and Apoptosis in Carp. Dev. Comp. Immunol. 2015, 53, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Dinic, J.; Riehl, A.; Adler, J.; Parmryd, I. The T Cell Receptor Resides in Ordered Plasma Membrane Nanodomains That Aggregate upon Patching of the Receptor. Sci. Rep. 2015, 5, 10082. [Google Scholar] [CrossRef] [Green Version]
- Yusuf, I.; Fruman, D.A. Regulation of Quiescence in Lymphocytes. Trends Immunol. 2003, 24, 380–386. [Google Scholar] [CrossRef]
- Babich, A.; Burkhardt, J.K. Coordinate Control of Cytoskeletal Remodeling and Calcium Mobilization during T-Cell Activation. Immunol. Rev. 2013, 256, 80–94. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.-H.; Cho, S.; Wirtz, D. Tight Coupling between Nucleus and Cell Migration through the Perinuclear Actin Cap. J. Cell Sci. 2014, 127, 2528–2541. [Google Scholar] [CrossRef] [Green Version]
- Versaevel, M.; Grevesse, T.; Gabriele, S. Spatial Coordination between Cell and Nuclear Shape within Micropatterned Endothelial Cells. Nat. Commun. 2012, 3, 671. [Google Scholar] [CrossRef] [Green Version]
- Chang, T.; Niu, C.; Sun, C.; Ma, Y.; Guo, R.; Ruan, Z.; Gao, Y.; Lu, X.; Li, H.; Lin, Y.; et al. Melatonin Exerts Immunoregulatory Effects by Balancing Peripheral Effector and Regulatory T Helper Cells in Myasthenia Gravis. Aging 2020, 12, 21147–21160. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Yang, Y.; Fan, C.; Han, J.; Wang, D.; Di, S.; Hu, W.; Liu, D.; Li, X.; Reiter, R.J.; et al. Melatonin as a Potential Anticarcinogen for Non-Small-Cell Lung Cancer. Oncotarget 2016, 7, 46768–46784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aneiros-Fernandez, J.; Arias-Santiago, S.; Arias-Santiago, B.; Herrero-Fernández, M.; Carriel, V.; Aneiros-Cachaza, J.; López-Valverde, A.; Cutando-Soriano, A. MT1 Melatonin Receptor Expression in Warthin’s Tumor. Pathol. Oncol. Res. 2013, 19, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Collins, A.R.; Dai, J.; Dubocovich, M.L.; Hill, S.M. MT(1) Melatonin Receptor Overexpression Enhances the Growth Suppressive Effect of Melatonin in Human Breast Cancer Cells. Mol. Cell. Endocrinol. 2002, 192, 147–156. [Google Scholar] [CrossRef]
- Hill, S.M.; Blask, D.E.; Xiang, S.; Yuan, L.; Mao, L.; Dauchy, R.T.; Dauchy, E.M.; Frasch, T.; Duplesis, T. Melatonin and Associated Signaling Pathways That Control Normal Breast Epithelium and Breast Cancer. J. Mammary Gland Biol. Neoplasia 2011, 16, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.M.; Belancio, V.P.; Dauchy, R.T.; Xiang, S.; Brimer, S.; Mao, L.; Hauch, A.; Lundberg, P.W.; Summers, W.; Yuan, L.; et al. Melatonin: An Inhibitor of Breast Cancer. Endocr. Relat. Cancer 2015, 22, R183–R204. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, G.M.; Martinez, M.; Camargo, I.C.C.; Domeniconi, R.F.; Martinez, F.E.; Chuffa, L.G.A. Melatonin Attenuates Her-2, P38 MAPK, p-AKT, and MTOR Levels in Ovarian Carcinoma of Ethanol-Preferring Rats. J. Cancer 2014, 5, 728–735. [Google Scholar] [CrossRef]
- Zhou, Q.; Gui, S.; Zhou, Q.; Wang, Y. Melatonin Inhibits the Migration of Human Lung Adenocarcinoma A549 Cell Lines Involving JNK/MAPK Pathway. PLoS ONE 2014, 9, e101132. [Google Scholar] [CrossRef] [Green Version]
- Lu, Y.-X.; Chen, D.-L.; Wang, D.-S.; Chen, L.-Z.; Mo, H.-Y.; Sheng, H.; Bai, L.; Wu, Q.-N.; Yu, H.-E.; Xie, D.; et al. Melatonin Enhances Sensitivity to Fluorouracil in Oesophageal Squamous Cell Carcinoma through Inhibition of Erk and Akt Pathway. Cell Death Dis. 2016, 7, e2432. [Google Scholar] [CrossRef] [Green Version]
- Benítez-King, G.; Soto-Vega, E.; Ramírez-Rodriguez, G. Melatonin Modulates Microfilament Phenotypes in Epithelial Cells: Implications for Adhesion and Inhibition of Cancer Cell Migration. Histol. Histopathol. 2009, 24, 789–799. [Google Scholar] [CrossRef]
- Shen, Y.; Guo, S.; Chen, G.; Ding, Y.; Wu, Y.; Tian, W. Hyperglycemia Induces Osteoclastogenesis and Bone Destruction Through the Activation of Ca2+/Calmodulin-Dependent Protein Kinase II. Calcif. Tissue Int. 2019, 104, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Schaller, M.D. Calcium-Dependent Pyk2 Activation: A Role for Calmodulin? Biochem. J. 2008, 410, e3–e4. [Google Scholar] [CrossRef] [Green Version]
- Ortíz-López, L.; Morales-Mulia, S.; Ramírez-Rodríguez, G.; Benítez-King, G. ROCK-Regulated Cytoskeletal Dynamics Participate in the Inhibitory Effect of Melatonin on Cancer Cell Migration. J. Pineal Res. 2009, 46, 15–21. [Google Scholar] [CrossRef]
- American Diabetes Association. Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2014, 37 (Suppl. S1), S81–S90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Diabetes Association 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2021. Diabetes Care 2021, 44, S15–S33. [Google Scholar] [CrossRef] [PubMed]
- Banday, M.Z.; Sameer, A.S.; Nissar, S. Pathophysiology of Diabetes: An Overview. Avicenna J. Med. 2020, 10, 174–188. [Google Scholar] [CrossRef]
- Roden, M.; Shulman, G.I. The Integrative Biology of Type 2 Diabetes. Nature 2019, 576, 51–60. [Google Scholar] [CrossRef] [Green Version]
- Defronzo, R.A. Banting Lecture. From the Triumvirate to the Ominous Octet: A New Paradigm for the Treatment of Type 2 Diabetes Mellitus. Diabetes 2009, 58, 773–795. [Google Scholar] [CrossRef] [Green Version]
- James, S.M.; Honn, K.A.; Gaddameedhi, S.; Van Dongen, H.P.A. Shift Work: Disrupted Circadian Rhythms and Sleep-Implications for Health and Well-Being. Curr. Sleep Med. Rep. 2017, 3, 104–112. [Google Scholar] [CrossRef]
- Knutsson, A.; Kempe, A. Shift Work and Diabete—A Systematic Review. Chronobiol. Int. 2014, 31, 1146–1151. [Google Scholar] [CrossRef]
- Brum, M.C.B.; Filho, F.F.D.; Schnorr, C.C.; Bottega, G.B.; Rodrigues, T.C. Shift Work and Its Association with Metabolic Disorders. Diabetol. Metab.Syndr. 2015, 7, 45. [Google Scholar] [CrossRef] [Green Version]
- Chalernvanichakorn, T.; Sithisarankul, P.; Hiransuthikul, N. Shift Work and Type 2 Diabetic Patients’ Health. J. Med. Assoc. Thai. 2008, 91, 1093–1096. [Google Scholar]
- Young, J.; Waclawski, E.; Young, J.A.; Spencer, J. Control of Type 1 Diabetes Mellitus and Shift Work. Occup. Med. 2013, 63, 70–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dumont, M.; Paquet, J. Progressive Decrease of Melatonin Production over Consecutive Days of Simulated Night Work. Chronobiol. Int. 2014, 31, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Dijk, D.-J.; Duffy, J.F.; Silva, E.J.; Shanahan, T.L.; Boivin, D.B.; Czeisler, C.A. Amplitude Reduction and Phase Shifts of Melatonin, Cortisol and Other Circadian Rhythms after a Gradual Advance of Sleep and Light Exposure in Humans. PLoS ONE 2012, 7, e30037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cagnacci, A.; Arangino, S.; Renzi, A.; Paoletti, A.M.; Melis, G.B.; Cagnacci, P.; Volpe, A. Influence of Melatonin Administration on Glucose Tolerance and Insulin Sensitivity of Postmenopausal Women. Clin. Endocrinol. 2001, 54, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Sastre, P.; Scheer, F.A.J.L.; Gómez-Abellán, P.; Madrid, J.A.; Garaulet, M. Acute Melatonin Administration in Humans Impairs Glucose Tolerance in Both the Morning and Evening. Sleep 2014, 37, 1715–1719. [Google Scholar] [CrossRef] [Green Version]
- Kampmann, U.; Lauritzen, E.S.; Grarup, N.; Jessen, N.; Hansen, T.; Møller, N.; Støy, J. Acute Metabolic Effects of Melatonin—A Randomized Crossover Study in Healthy Young Men. J. Pineal Res. 2021, 70, e12706. [Google Scholar] [CrossRef]
- Goyal, A.; Terry, P.D.; Superak, H.M.; Nell-Dybdahl, C.L.; Chowdhury, R.; Phillips, L.S.; Kutner, M.H. Melatonin Supplementation to Treat the Metabolic Syndrome: A Randomized Controlled Trial. Diabetol. Metab. Syndr. 2014, 6, 124. [Google Scholar] [CrossRef] [Green Version]
- Rezvanfar, M.R.; Heshmati, G.; Chehrei, A.; Haghverdi, F.; Rafiee, F.; Rezvanfar, F. Effect of Bedtime Melatonin Consumption on Diabetes Control and Lipid Profile. Int. J. Diabetes Dev. Ctries. 2017, 37, 74–77. [Google Scholar] [CrossRef]
- Delpino, F.M.; Figueiredo, L.M.; Nunes, B.P. Effects of Melatonin Supplementation on Diabetes: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Clin. Nutr. 2021, 40, 4595–4605. [Google Scholar] [CrossRef]
- Robeva, R.; Kirilov, G.; Tomova, A.; Kumanov, P. Melatonin-Insulin Interactions in Patients with Metabolic Syndrome. J. Pineal Res. 2008, 44, 52–56. [Google Scholar] [CrossRef]
- McMullan, C.J.; Schernhammer, E.S.; Rimm, E.B.; Hu, F.B.; Forman, J.P. Melatonin Secretion and the Incidence of Type 2 Diabetes. JAMA 2013, 309, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Reutrakul, S.; Siwasaranond, N.; Nimitphong, H.; Saetung, S.; Chirakalwasan, N.; Chailurkit, L.-O.; Srijaruskul, K.; Ongphiphadhanakul, B.; Thakkinstian, A. Associations between Nocturnal Urinary 6-Sulfatoxymelatonin, Obstructive Sleep Apnea Severity and Glycemic Control in Type 2 Diabetes. Chronobiol. Int. 2017, 34, 382–392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulder, H. Melatonin Signalling and Type 2 Diabetes Risk: Too Little, Too Much or Just Right? Diabetologia 2017, 60, 826–829. [Google Scholar] [CrossRef]
- Garaulet, M.; Qian, J.; Florez, J.C.; Arendt, J.; Saxena, R.; Scheer, F.A.J.L. Melatonin Effects on Glucose Metabolism: Time To Unlock the Controversy. Trends Endocrinol. Metab. 2020, 31, 192–204. [Google Scholar] [CrossRef]
- Peschke, E.; Stumpf, I.; Bazwinsky, I.; Litvak, L.; Dralle, H.; Mühlbauer, E. Melatonin and Type 2 Diabetes—A Possible Link? J. Pineal Res. 2007, 42, 350–358. [Google Scholar] [CrossRef]
- Zibolka, J.; Bazwinsky-Wutschke, I.; Mühlbauer, E.; Peschke, E. Distribution and Density of Melatonin Receptors in Human Main Pancreatic Islet Cell Types. J. Pineal Res. 2018, 65, e12480. [Google Scholar] [CrossRef] [PubMed]
- Lyssenko, V.; Nagorny, C.L.F.; Erdos, M.R.; Wierup, N.; Jonsson, A.; Spégel, P.; Bugliani, M.; Saxena, R.; Fex, M.; Pulizzi, N.; et al. Common Variant in MTNR1B Associated with Increased Risk of Type 2 Diabetes and Impaired Early Insulin Secretion. Nat. Genet. 2009, 41, 82–88. [Google Scholar] [CrossRef]
- Ramracheya, R.D.; Muller, D.S.; Squires, P.E.; Brereton, H.; Sugden, D.; Huang, G.C.; Amiel, S.A.; Jones, P.M.; Persaud, S.J. Function and Expression of Melatonin Receptors on Human Pancreatic Islets. J. Pineal Res. 2008, 44, 273–279. [Google Scholar] [CrossRef]
- Kemp, D.M.; Ubeda, M.; Habener, J.F. Identification and Functional Characterization of Melatonin Mel 1a Receptors in Pancreatic Beta Cells: Potential Role in Incretin-Mediated Cell Function by Sensitization of CAMP Signaling. Mol. Cell. Endocrinol. 2002, 191, 157–166. [Google Scholar] [CrossRef]
- Derlacz, R.A.; Poplawski, P.; Napierala, M.; Jagielski, A.K.; Bryla, J. Melatonin-Induced Modulation of Glucose Metabolism in Primary Cultures of Rabbit Kidney-Cortex Tubules. J. Pineal Res. 2005, 38, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, T.C.; Lellis-Santos, C.; Jesus, D.S.; Taneda, M.; Rodrigues, S.C.; Amaral, F.G.; Lopes, A.M.S.; Cipolla-Neto, J.; Bordin, S.; Anhê, G.F. Absence of Melatonin Induces Night-Time Hepatic Insulin Resistance and Increased Gluconeogenesis Due to Stimulation of Nocturnal Unfolded Protein Response. Endocrinology 2011, 152, 1253–1263. [Google Scholar] [CrossRef] [Green Version]
- Faria, J.A.; Kinote, A.; Ignacio-Souza, L.M.; de Araújo, T.M.; Razolli, D.S.; Doneda, D.L.; Paschoal, L.B.; Lellis-Santos, C.; Bertolini, G.L.; Velloso, L.A.; et al. Melatonin Acts through MT1/MT2 Receptors to Activate Hypothalamic Akt and Suppress Hepatic Gluconeogenesis in Rats. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E230–E242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müssig, K.; Staiger, H.; Machicao, F.; Häring, H.-U.; Fritsche, A. Genetic Variants in MTNR1B Affecting Insulin Secretion. Ann. Med. 2010, 42, 387–393. [Google Scholar] [CrossRef]
- Staiger, H.; Machicao, F.; Schäfer, S.A.; Kirchhoff, K.; Kantartzis, K.; Guthoff, M.; Silbernagel, G.; Stefan, N.; Häring, H.-U.; Fritsche, A. Polymorphisms within the Novel Type 2 Diabetes Risk Locus MTNR1B Determine Beta-Cell Function. PLoS ONE 2008, 3, e3962. [Google Scholar] [CrossRef] [Green Version]
- Prokopenko, I.; Langenberg, C.; Florez, J.C.; Saxena, R.; Soranzo, N.; Thorleifsson, G.; Loos, R.J.F.; Manning, A.K.; Jackson, A.U.; Aulchenko, Y.; et al. Variants in MTNR1B Influence Fasting Glucose Levels. Nat. Genet. 2009, 41, 77–81. [Google Scholar] [CrossRef]
- Sparsø, T.; Bonnefond, A.; Andersson, E.; Bouatia-Naji, N.; Holmkvist, J.; Wegner, L.; Grarup, N.; Gjesing, A.P.; Banasik, K.; Cavalcanti-Proença, C.; et al. G-Allele of Intronic Rs10830963 in MTNR1B Confers Increased Risk of Impaired Fasting Glycemia and Type 2 Diabetes through an Impaired Glucose-Stimulated Insulin Release: Studies Involving 19,605 Europeans. Diabetes 2009, 58, 1450–1456. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Liu, L.; Zhao, J.; Cui, G.; Chen, C.; Ding, H.; Wang, D.W. Large Scale Meta-Analyses of Fasting Plasma Glucose Raising Variants in GCK, GCKR, MTNR1B and G6PC2 and Their Impacts on Type 2 Diabetes Mellitus Risk. PLoS ONE 2013, 8, e67665. [Google Scholar] [CrossRef]
- Shen, L.-L.; Jin, Y. Effects of MTNR1B Genetic Variants on the Risk of Type 2 Diabetes Mellitus: A Meta-Analysis. Mol. Genet. Genom. Med. 2019, 7, e611. [Google Scholar] [CrossRef] [Green Version]
- Walford, G.A.; Green, T.; Neale, B.; Isakova, T.; Rotter, J.I.; Grant, S.F.A.; Fox, C.S.; Pankow, J.S.; Wilson, J.G.; Meigs, J.B.; et al. Common Genetic Variants Differentially Influence the Transition from Clinically Defined States of Fasting Glucose Metabolism. Diabetologia 2012, 55, 331–339. [Google Scholar] [CrossRef] [Green Version]
- Olsson, L.; Pettersen, E.; Ahlbom, A.; Carlsson, S.; Midthjell, K.; Grill, V. No Effect by the Common Gene Variant Rs10830963 of the Melatonin Receptor 1B on the Association between Sleep Disturbances and Type 2 Diabetes: Results from the Nord-Trøndelag Health Study. Diabetologia 2011, 54, 1375–1378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haljas, K.; Lahti, J.; Tuomi, T.; Isomaa, B.; Eriksson, J.G.; Groop, L.; Räikkönen, K. Melatonin Receptor 1B Gene Rs10830963 Polymorphism, Depressive Symptoms and Glycaemic Traits. Ann. Med. 2018, 50, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Rönn, T.; Wen, J.; Yang, Z.; Lu, B.; Du, Y.; Groop, L.; Hu, R.; Ling, C. A Common Variant in MTNR1B, Encoding Melatonin Receptor 1B, Is Associated with Type 2 Diabetes and Fasting Plasma Glucose in Han Chinese Individuals. Diabetologia 2009, 52, 830–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chambers, J.C.; Zhang, W.; Zabaneh, D.; Sehmi, J.; Jain, P.; McCarthy, M.I.; Froguel, P.; Ruokonen, A.; Balding, D.; Jarvelin, M.-R.; et al. Common Genetic Variation near Melatonin Receptor MTNR1B Contributes to Raised Plasma Glucose and Increased Risk of Type 2 Diabetes among Indian Asians and European Caucasians. Diabetes 2009, 58, 2703–2708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langenberg, C.; Pascoe, L.; Mari, A.; Tura, A.; Laakso, M.; Frayling, T.M.; Barroso, I.; Loos, R.J.F.; Wareham, N.J.; Walker, M.; et al. Common Genetic Variation in the Melatonin Receptor 1B Gene (MTNR1B) Is Associated with Decreased Early-Phase Insulin Response. Diabetologia 2009, 52, 1537–1542. [Google Scholar] [CrossRef] [Green Version]
- Kelliny, C.; Ekelund, U.; Andersen, L.B.; Brage, S.; Loos, R.J.F.; Wareham, N.J.; Langenberg, C. Common Genetic Determinants of Glucose Homeostasis in Healthy Children: The European Youth Heart Study. Diabetes 2009, 58, 2939–2945. [Google Scholar] [CrossRef] [Green Version]
- Kan, M.Y.; Zhou, D.Z.; Zhang, D.; Zhang, Z.; Chen, Z.; Yang, Y.F.; Guo, X.Z.; Xu, H.; He, L.; Liu, Y. Two Susceptible Diabetogenic Variants near/in MTNR1B Are Associated with Fasting Plasma Glucose in a Han Chinese Cohort. Diabet. Med. 2010, 27, 598–602. [Google Scholar] [CrossRef]
- Takeuchi, F.; Katsuya, T.; Chakrewarthy, S.; Yamamoto, K.; Fujioka, A.; Serizawa, M.; Fujisawa, T.; Nakashima, E.; Ohnaka, K.; Ikegami, H.; et al. Common Variants at the GCK, GCKR, G6PC2-ABCB11 and MTNR1B Loci Are Associated with Fasting Glucose in Two Asian Populations. Diabetologia 2010, 53, 299–308. [Google Scholar] [CrossRef]
- Liu, C.; Wu, Y.; Li, H.; Qi, Q.; Langenberg, C.; Loos, R.J.F.; Lin, X. MTNR1B Rs10830963 Is Associated with Fasting Plasma Glucose, HbA1C and Impaired Beta-Cell Function in Chinese Hans from Shanghai. BMC Med. Genet. 2010, 11, 59. [Google Scholar] [CrossRef] [Green Version]
- Tam, C.H.T.; Ho, J.S.K.; Wang, Y.; Lee, H.M.; Lam, V.K.L.; Germer, S.; Martin, M.; So, W.Y.; Ma, R.C.W.; Chan, J.C.N.; et al. Common Polymorphisms in MTNR1B, G6PC2 and GCK Are Associated with Increased Fasting Plasma Glucose and Impaired Beta-Cell Function in Chinese Subjects. PLoS ONE 2010, 5, e11428. [Google Scholar] [CrossRef]
- Ohshige, T.; Iwata, M.; Omori, S.; Tanaka, Y.; Hirose, H.; Kaku, K.; Maegawa, H.; Watada, H.; Kashiwagi, A.; Kawamori, R.; et al. Association of New Loci Identified in European Genome-Wide Association Studies with Susceptibility to Type 2 Diabetes in the Japanese. PLoS ONE 2011, 6, e26911. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-Y.; Wang, H.-J.; Ma, J.; Xu, Z.-Y.; Hinney, A.; Hebebrand, J.; Wang, Y. Association of the Rs10830963 Polymorphism in MTNR1B with Fasting Glucose Levels in Chinese Children and Adolescents. Obes. Facts 2011, 4, 197–203. [Google Scholar] [CrossRef]
- Reinehr, T.; Scherag, A.; Wang, H.-J.; Roth, C.L.; Kleber, M.; Scherag, S.; Boes, T.; Vogel, C.; Hebebrand, J.; Hinney, A. Relationship between MTNR1B (Melatonin Receptor 1B Gene) Polymorphism Rs10830963 and Glucose Levels in Overweight Children and Adolescents. Pediatr. Diabetes 2011, 12, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Barker, A.; Sharp, S.J.; Timpson, N.J.; Bouatia-Naji, N.; Warrington, N.M.; Kanoni, S.; Beilin, L.J.; Brage, S.; Deloukas, P.; Evans, D.M.; et al. Association of Genetic Loci with Glucose Levels in Childhood and Adolescence: A Meta-Analysis of over 6000 Children. Diabetes 2011, 60, 1805–1812. [Google Scholar] [CrossRef] [Green Version]
- Renström, F.; Shungin, D.; Johansson, I.; MAGIC Investigators; Florez, J.C.; Hallmans, G.; Hu, F.B.; Franks, P.W. Genetic Predisposition to Long-Term Nondiabetic Deteriorations in Glucose Homeostasis: Ten-Year Follow-up of the GLACIER Study. Diabetes 2011, 60, 345–354. [Google Scholar] [CrossRef] [Green Version]
- Holzapfel, C.; Siegrist, M.; Rank, M.; Langhof, H.; Grallert, H.; Baumert, J.; Irimie, C.; Klopp, N.; Wolfarth, B.; Illig, T.; et al. Association of a MTNR1B Gene Variant with Fasting Glucose and HOMA-B in Children and Adolescents with High BMI-SDS. Eur. J. Endocrinol. 2011, 164, 205–212. [Google Scholar] [CrossRef] [Green Version]
- Comuzzie, A.G.; Cole, S.A.; Laston, S.L.; Voruganti, V.S.; Haack, K.; Gibbs, R.A.; Butte, N.F. Novel Genetic Loci Identified for the Pathophysiology of Childhood Obesity in the Hispanic Population. PLoS ONE 2012, 7, e51954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Florez, J.C.; Jablonski, K.A.; McAteer, J.B.; Franks, P.W.; Mason, C.C.; Mather, K.; Horton, E.; Goldberg, R.; Dabelea, D.; Kahn, S.E.; et al. Effects of Genetic Variants Previously Associated with Fasting Glucose and Insulin in the Diabetes Prevention Program. PLoS ONE 2012, 7, e44424. [Google Scholar] [CrossRef]
- DeMenna, J.; Puppala, S.; Chittoor, G.; Schneider, J.; Kim, J.Y.; Shaibi, G.Q.; Mandarino, L.J.; Duggirala, R.; Coletta, D.K. Association of Common Genetic Variants with Diabetes and Metabolic Syndrome Related Traits in the Arizona Insulin Resistance Registry: A Focus on Mexican American Families in the Southwest. Hum. Hered. 2014, 78, 47–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jonsson, A.; Ladenvall, C.; Ahluwalia, T.S.; Kravic, J.; Krus, U.; Taneera, J.; Isomaa, B.; Tuomi, T.; Renström, E.; Groop, L.; et al. Effects of Common Genetic Variants Associated with Type 2 Diabetes and Glycemic Traits on α- and β-Cell Function and Insulin Action in Humans. Diabetes 2013, 62, 2978–2983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Semiz, S.; Dujic, T.; Velija-Asimi, Z.; Prnjavorac, B.; Bego, T.; Ostanek, B.; Marc, J.; Causevic, A. Effects of Melatonin Receptor 1B Gene Variation on Glucose Control in Population from Bosnia and Herzegovina. Exp. Clin. Endocrinol. Diabetes 2014, 122, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Dalla Man, C.; Cobelli, C.; Groop, L.; Zhao, H.; Bale, A.E.; Shaw, M.; Duran, E.; Pierpont, B.; Caprio, S.; et al. A Common Variant in the MTNR1b Gene Is Associated with Increased Risk of Impaired Fasting Glucose (IFG) in Youth with Obesity. Obesity 2015, 23, 1022–1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marouli, E.; Kanoni, S.; Mamakou, V.; Hackinger, S.; Southam, L.; Prins, B.; Rentari, A.; Dimitriou, M.; Zengini, E.; Gonidakis, F.; et al. Evaluating the Glucose Raising Effect of Established Loci via a Genetic Risk Score. PLoS ONE 2017, 12, e0186669. [Google Scholar] [CrossRef] [Green Version]
- Patel, R.; Rathwa, N.; Palit, S.P.; Ramachandran, A.V.; Begum, R. Association of Melatonin &MTNR1B Variants with Type 2 Diabetes in Gujarat Population. Biomed. Pharm. 2018, 103, 429–434. [Google Scholar] [CrossRef]
- Caro-Gomez, M.A.; Naranjo-González, C.A.; Gallego-Lopera, N.; Parra-Marín, M.V.; Valencia, D.M.; Arcos, E.G.; Villegas-Perrasse, A.; Bedoya-Berrío, G. Association of Native American Ancestry and Common Variants in ACE, ADIPOR2, MTNR1B, GCK, TCF7L2 and FTO Genes with Glycemic Traits in Colombian Population. Gene 2018, 677, 198–210. [Google Scholar] [CrossRef]
- Çöl, N.; Nacak, M.; Araz, M. Association of Melatonin Receptor 1 B Gene (Rs10830963 and Rs9192552) Polymorphısm with Adolescent Obesity and Related Comorbidities in Turkey. J. Int. Med. Res. 2018, 46, 3086–3096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Luis Román, D.A.; Primo, D.; Aller, R.; Izaola, O. Association of the Rs10830963 Polymorphism in MTNR1B with Fasting Glucose, Serum Adipokine Levels and Components of Metabolic Syndrome in Adult Obese Subjects. Nutr. Hosp. 2019, 36, 60–65. [Google Scholar] [CrossRef]
- Sorlí, J.V.; Barragán, R.; Coltell, O.; Portolés, O.; Pascual, E.C.; Ortega-Azorín, C.; González, J.I.; Estruch, R.; Saiz, C.; Pérez-Fidalgo, A.; et al. Chronological Age Interacts with the Circadian Melatonin Receptor 1B Gene Variation, Determining Fasting Glucose Concentrations in Mediterranean Populations. Additional Analyses on Type-2 Diabetes Risk. Nutrients 2020, 12, 3323. [Google Scholar] [CrossRef]
- Oh, S.-W.; Lee, J.-E.; Shin, E.; Kwon, H.; Choe, E.K.; Choi, S.-Y.; Rhee, H.; Choi, S.H. Genome-Wide Association Study of Metabolic Syndrome in Korean Populations. PLoS ONE 2020, 15, e0227357. [Google Scholar] [CrossRef]
- Fujita, H.; Hara, K.; Shojima, N.; Horikoshi, M.; Iwata, M.; Hirota, Y.; Tobe, K.; Seino, S.; Kadowaki, T. Variations with Modest Effects Have an Important Role in the Genetic Background of Type 2 Diabetes and Diabetes-Related Traits. J. Hum. Genet. 2012, 57, 776–779. [Google Scholar] [CrossRef] [Green Version]
- Palmer, N.D.; Goodarzi, M.O.; Langefeld, C.D.; Wang, N.; Guo, X.; Taylor, K.D.; Fingerlin, T.E.; Norris, J.M.; Buchanan, T.A.; Xiang, A.H.; et al. Genetic Variants Associated With Quantitative Glucose Homeostasis Traits Translate to Type 2 Diabetes in Mexican Americans: The GUARDIAN (Genetics Underlying Diabetes in Hispanics) Consortium. Diabetes 2015, 64, 1853–1866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, C.; Zhang, R.; Wang, C.; Yu, W.; Lu, J.; Ma, X.; Wang, J.; Jiang, F.; Tang, S.; Bao, Y.; et al. Effects of GCK, GCKR, G6PC2 and MTNR1B Variants on Glucose Metabolism and Insulin Secretion. PLoS ONE 2010, 5, e11761. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.; Li, X.; Gu, Q.; Chen, H.; Lu, D.; Gao, X. A Common Polymorphism Rs3781637 in MTNR1B Is Associated with Type 2 Diabetes and Lipids Levels in Han Chinese Individuals. Cardiovasc. Diabetol. 2011, 10, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rees, S.D.; Hydrie, M.Z.I.; O’Hare, J.P.; Kumar, S.; Shera, A.S.; Basit, A.; Barnett, A.H.; Kelly, M.A. Effects of 16 Genetic Variants on Fasting Glucose and Type 2 Diabetes in South Asians: ADCY5 and GLIS3 Variants May Predispose to Type 2 Diabetes. PLoS ONE 2011, 6, e24710. [Google Scholar] [CrossRef] [PubMed]
- Been, L.F.; Hatfield, J.L.; Shankar, A.; Aston, C.E.; Ralhan, S.; Wander, G.S.; Mehra, N.K.; Singh, J.R.; Mulvihill, J.J.; Sanghera, D.K. A Low Frequency Variant within the GWAS Locus of MTNR1B Affects Fasting Glucose Concentrations: Genetic Risk Is Modulated by Obesity. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 944–951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salman, M.; Dasgupta, S.; Cholendra, A.; Venugopal, P.N.; Lakshmi, G.L.; Xaviour, D.; Rao, J.; D’Souza, C.J.M. MTNR1B Gene Polymorphisms and Susceptibility to Type 2 Diabetes: A Pilot Study in South Indians. Gene 2015, 566, 189–193. [Google Scholar] [CrossRef]
- Gao, K.; Wang, J.; Li, L.; Zhai, Y.; Ren, Y.; You, H.; Wang, B.; Wu, X.; Li, J.; Liu, Z.; et al. Polymorphisms in Four Genes (KCNQ1 Rs151290, KLF14 Rs972283, GCKR Rs780094 and MTNR1B Rs10830963) and Their Correlation with Type 2 Diabetes Mellitus in Han Chinese in Henan Province, China. Int. J. Environ. Res. Public Health 2016, 13, 260. [Google Scholar] [CrossRef] [Green Version]
- Gao, K.; Ren, Y.; Wang, J.; Liu, Z.; Li, J.; Li, L.; Wang, B.; Li, H.; Wang, Y.; Cao, Y.; et al. Interactions between Genetic Polymorphisms of Glucose Metabolizing Genes and Smoking and Alcohol Consumption in the Risk of Type 2 Diabetes Mellitus. Appl. Physiol. Nutr. Metab. 2017, 42, 1316–1321. [Google Scholar] [CrossRef]
- Gaulton, K.J.; Ferreira, T.; Lee, Y.; Raimondo, A.; Mägi, R.; Reschen, M.E.; Mahajan, A.; Locke, A.; Rayner, N.W.; Robertson, N.; et al. Genetic Fine Mapping and Genomic Annotation Defines Causal Mechanisms at Type 2 Diabetes Susceptibility Loci. Nat. Genet. 2015, 47, 1415–1425. [Google Scholar] [CrossRef]
- Hong, K.-W.; Chung, M.; Cho, S.B. Meta-Analysis of Genome-Wide Association Study of Homeostasis Model Assessment β Cell Function and Insulin Resistance in an East Asian Population and the European Results. Mol. Genet. Genomics 2014, 289, 1247–1255. [Google Scholar] [CrossRef]
- Stancáková, A.; Kuulasmaa, T.; Paananen, J.; Jackson, A.U.; Bonnycastle, L.L.; Collins, F.S.; Boehnke, M.; Kuusisto, J.; Laakso, M. Association of 18 Confirmed Susceptibility Loci for Type 2 Diabetes with Indices of Insulin Release, Proinsulin Conversion, and Insulin Sensitivity in 5,327 Nondiabetic Finnish Men. Diabetes 2009, 58, 2129–2136. [Google Scholar] [CrossRef] [Green Version]
- Simonis-Bik, A.M.; Nijpels, G.; van Haeften, T.W.; Houwing-Duistermaat, J.J.; Boomsma, D.I.; Reiling, E.; van Hove, E.C.; Diamant, M.; Kramer, M.H.H.; Heine, R.J.; et al. Gene Variants in the Novel Type 2 Diabetes Loci CDC123/CAMK1D, THADA, ADAMTS9, BCL11A, and MTNR1B Affect Different Aspects of Pancreatic Beta-Cell Function. Diabetes 2010, 59, 293–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingelsson, E.; Langenberg, C.; Hivert, M.-F.; Prokopenko, I.; Lyssenko, V.; Dupuis, J.; Mägi, R.; Sharp, S.; Jackson, A.U.; Assimes, T.L.; et al. Detailed Physiologic Characterization Reveals Diverse Mechanisms for Novel Genetic Loci Regulating Glucose and Insulin Metabolism in Humans. Diabetes 2010, 59, 1266–1275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.Y.; Cheong, H.S.; Park, B.-L.; Baik, S.H.; Park, S.; Lee, S.W.; Kim, M.-H.; Chung, J.H.; Choi, J.S.; Kim, M.-Y.; et al. Melatonin Receptor 1 B Polymorphisms Associated with the Risk of Gestational Diabetes Mellitus. BMC Med. Genet. 2011, 12, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, H.; Li, Q.; Gao, S. Meta-Analysis of the Relationship between Common Type 2 Diabetes Risk Gene Variants with Gestational Diabetes Mellitus. PLoS ONE 2012, 7, e45882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, S.; Liu, Y.; Tan, Y.; Gan, L.; Mei, J.; Song, W.; Chi, S.; Dong, X.; Chen, X.; Deng, S. Association of Genetic Variants of Melatonin Receptor 1B with Gestational Plasma Glucose Level and Risk of Glucose Intolerance in Pregnant Chinese Women. PLoS ONE 2012, 7, e40113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Bao, W.; Rong, Y.; Yang, H.; Bowers, K.; Yeung, E.; Kiely, M. Genetic Variants and the Risk of Gestational Diabetes Mellitus: A Systematic Review. Hum. Reprod. Update 2013, 19, 376–390. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Qiao, B.; Zhan, Y.; Peng, W.; Chen, Z.-J.; Sun, L.; Zhang, J.; Zhao, L.; Gao, Q. Association between Genetic Variations in MTNR1A and MTNR1B Genes and Gestational Diabetes Mellitus in Han Chinese Women. Gynecol. Obs. Invest. 2013, 76, 221–227. [Google Scholar] [CrossRef]
- Huopio, H.; Cederberg, H.; Vangipurapu, J.; Hakkarainen, H.; Pääkkönen, M.; Kuulasmaa, T.; Heinonen, S.; Laakso, M. Association of Risk Variants for Type 2 Diabetes and Hyperglycemia with Gestational Diabetes. Eur. J. Endocrinol. 2013, 169, 291–297. [Google Scholar] [CrossRef] [Green Version]
- Stuebe, A.M.; Wise, A.; Nguyen, T.; Herring, A.; North, K.E.; Siega-Riz, A.M. Maternal Genotype and Gestational Diabetes. Am. J. Perinatol. 2014, 31, 69–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, J.; Xiang, A.H.; Trigo, E.; Takayanagi, M.; Beale, E.; Lawrence, J.M.; Hartiala, J.; Richey, J.M.; Allayee, H.; Buchanan, T.A.; et al. Genetic Variation in MTNR1B Is Associated with Gestational Diabetes Mellitus and Contributes Only to the Absolute Level of Beta Cell Compensation in Mexican Americans. Diabetologia 2014, 57, 1391–1399. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Huang, Z.; Li, H.; Bai, J.; Liu, X.; Ye, H. Relationship between Melatonin Receptor 1B (Rs10830963 and Rs1387153) with Gestational Diabetes Mellitus: A Case-Control Study and Meta-Analysis. Arch. Gynecol. Obs. 2016, 294, 55–61. [Google Scholar] [CrossRef]
- Grotenfelt, N.E.; Wasenius, N.S.; Rönö, K.; Laivuori, H.; Stach-Lempinen, B.; Orho-Melander, M.; Schulz, C.-A.; Kautiainen, H.; Koivusalo, S.B.; Eriksson, J.G. Interaction between Rs10830963 Polymorphism in MTNR1B and Lifestyle Intervention on Occurrence of Gestational Diabetes. Diabetologia 2016, 59, 1655–1658. [Google Scholar] [CrossRef] [PubMed]
- Tarnowski, M.; Malinowski, D.; Safranow, K.; Dziedziejko, V.; Pawlik, A. MTNR1A and MTNR1B Gene Polymorphisms in Women with Gestational Diabetes. Gynecol. Endocrinol. 2017, 33, 395–398. [Google Scholar] [CrossRef]
- Rosta, K.; Al-Aissa, Z.; Hadarits, O.; Harreiter, J.; Nádasdi, Á.; Kelemen, F.; Bancher-Todesca, D.; Komlósi, Z.; Németh, L.; Rigó, J.; et al. Association Study with 77 SNPs Confirms the Robust Role for the Rs10830963/G of MTNR1B Variant and Identifies Two Novel Associations in Gestational Diabetes Mellitus Development. PLoS ONE 2017, 12, e0169781. [Google Scholar] [CrossRef]
- Ding, M.; Chavarro, J.; Olsen, S.; Lin, Y.; Ley, S.H.; Bao, W.; Rawal, S.; Grunnet, L.G.; Thuesen, A.C.B.; Mills, J.L.; et al. Genetic Variants of Gestational Diabetes Mellitus: A Study of 112 SNPs among 8722 Women in Two Independent Populations. Diabetologia 2018, 61, 1758–1768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alharbi, K.K.; Al-Sulaiman, A.M.; Shedaid, K.M.B.; Al-Shangiti, A.M.; Marie, M.; Al-Sheikh, Y.A.; Ali Khan, I. MTNR1B Genetic Polymorphisms as Risk Factors for Gestational Diabetes Mellitus: A Case-Control Study in a Single Tertiary Care Center. Ann. Saudi Med. 2019, 39, 309–318. [Google Scholar] [CrossRef] [Green Version]
- Xie, K.; Zhang, Y.; Wen, J.; Chen, T.; Kong, J.; Zhang, J.; Wu, X.; Hu, C.; Xu, B.; Ji, C.; et al. Genetic Predisposition to Gestational Glucose Metabolism and Gestational Diabetes Mellitus Risk in a Chinese Population. J. Diabetes 2019, 11, 869–877. [Google Scholar] [CrossRef]
- Shen, Y.; Jia, Y.; Li, Y.; Gu, X.; Wan, G.; Zhang, P.; Zhang, Y.; Jiang, L. Genetic Determinants of Gestational Diabetes Mellitus: A Case-Control Study in Two Independent Populations. Acta Diabetol. 2020, 57, 843–852. [Google Scholar] [CrossRef]
- Jia, Y.; Shen, Y.; Shi, X.; Gu, X.; Zhang, P.; Liu, Y.; Zhu, A.; Jiang, L. MTNR1B Gene on Susceptibility to Gestational Diabetes Mellitus: A Two-Stage Hospital-Based Study in Southern China. Mol. Genet. Genom. 2020, 295, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Nie, M.; Li, W.; Ping, F.; Hu, Y.; Ma, L.; Gao, J.; Liu, J. Association of Six Single Nucleotide Polymorphisms with Gestational Diabetes Mellitus in a Chinese Population. PLoS ONE 2011, 6, e26953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Zhou, Y.; Qiao, B.; Xu, L.; Li, Y.; Li, C. Association Between a Melatonin Receptor 1B Genetic Polymorphism and Its Protein Expression in Gestational Diabetes Mellitus. Reprod. Sci. 2019, 26, 1382–1388. [Google Scholar] [CrossRef]
- Beaumont, R.N.; Warrington, N.M.; Cavadino, A.; Tyrrell, J.; Nodzenski, M.; Horikoshi, M.; Geller, F.; Myhre, R.; Richmond, R.C.; Paternoster, L.; et al. Genome-Wide Association Study of Offspring Birth Weight in 86 577 Women Identifies Five Novel Loci and Highlights Maternal Genetic Effects That Are Independent of Fetal Genetics. Hum. Mol. Genet. 2018, 27, 742–756. [Google Scholar] [CrossRef]
- Andersen, M.K.; Sterner, M.; Forsén, T.; Käräjämäki, A.; Rolandsson, O.; Forsblom, C.; Groop, P.-H.; Lahti, K.; Nilsson, P.M.; Groop, L.; et al. Type 2 Diabetes Susceptibility Gene Variants Predispose to Adult-Onset Autoimmune Diabetes. Diabetologia 2014, 57, 1859–1868. [Google Scholar] [CrossRef]
- Hotta, K.; Kitamoto, A.; Kitamoto, T.; Mizusawa, S.; Teranishi, H.; So, R.; Matsuo, T.; Nakata, Y.; Hyogo, H.; Ochi, H.; et al. Association between Type 2 Diabetes Genetic Susceptibility Loci and Visceral and Subcutaneous Fat Area as Determined by Computed Tomography. J. Hum. Genet. 2012, 57, 305–310. [Google Scholar] [CrossRef] [Green Version]
- Peter, I.; McCaffery, J.M.; Kelley-Hedgepeth, A.; Hakonarson, H.; Reis, S.; Wagenknecht, L.E.; Kopin, A.S.; Huggins, G.S. Genetics Subgroup of the Look AHEAD Study Association of Type 2 Diabetes Susceptibility Loci with One-Year Weight Loss in the Look AHEAD Clinical Trial. Obesity 2012, 20, 1675–1682. [Google Scholar] [CrossRef]
- Goni, L.; Cuervo, M.; Milagro, F.I.; Martínez, J.A. Gene-Gene Interplay and Gene-Diet Interactions Involving the MTNR1B Rs10830963 Variant with Body Weight Loss. J. Nutr. Nutr. 2014, 7, 232–242. [Google Scholar] [CrossRef] [PubMed]
- de Luis, D.A.; Izaola, O.; Primo, D.; Aller, R. Association of the Rs10830963 Polymorphism in Melatonin Receptor Type 1B (MTNR1B) with Metabolic Response after Weight Loss Secondary to a Hypocaloric Diet Based in Mediterranean Style. Clin. Nutr. 2018, 37, 1563–1568. [Google Scholar] [CrossRef]
- De Luis, D.A.; Izaola, O.; Primo, D.; Aller, R. A Circadian Rhythm-Related MTNR1B Genetic Variant (Rs10830963) Modulate Body Weight Change and Insulin Resistance after 9 months of a High Protein/Low Carbohydrate vs a Standard Hypocaloric Diet. J. Diabetes Complicat. 2020, 34, 107534. [Google Scholar] [CrossRef] [PubMed]
- Goni, L.; Sun, D.; Heianza, Y.; Wang, T.; Huang, T.; Cuervo, M.; Martínez, J.A.; Shang, X.; Bray, G.A.; Sacks, F.M.; et al. Macronutrient-Specific Effect of the MTNR1B Genotype on Lipid Levels in Response to 2 Year Weight-Loss Diets. J. Lipid Res. 2018, 59, 155–161. [Google Scholar] [CrossRef] [Green Version]
- Goni, L.; Sun, D.; Heianza, Y.; Wang, T.; Huang, T.; Martínez, J.A.; Shang, X.; Bray, G.A.; Smith, S.R.; Sacks, F.M.; et al. A Circadian Rhythm-Related MTNR1B Genetic Variant Modulates the Effect of Weight-Loss Diets on Changes in Adiposity and Body Composition: The POUNDS Lost Trial. Eur. J. Nutr. 2019, 58, 1381–1389. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.; Zhang, X.; Shi, J.; Wang, M.; Wei, Z.; Zhao, A.; Li, B.; Zhao, X.; Xing, Q.; et al. Common Genetic Variation in MTNR1B Is Associated with Serum Testosterone, Glucose Tolerance, and Insulin Secretion in Polycystic Ovary Syndrome Patients. Fertil. Steril. 2010, 94, 2486–2489. [Google Scholar] [CrossRef]
- Song, X.; Sun, X.; Ma, G.; Sun, Y.; Shi, Y.; Du, Y.; Chen, Z.-J. Family Association Study between Melatonin Receptor Gene Polymorphisms and Polycystic Ovary Syndrome in Han Chinese. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 195, 108–112. [Google Scholar] [CrossRef]
- Li, C.; Shi, Y.; You, L.; Wang, L.; Chen, Z.-J. Association of Rs10830963 and Rs10830962 SNPs in the Melatonin Receptor (MTNR1B) Gene among Han Chinese Women with Polycystic Ovary Syndrome. Mol. Hum. Reprod. 2011, 17, 193–198. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.-H.; Kou, L.-C.; Wang, H.-M.; Bo, C.-M.; Song, X.-C. Genetic Polymorphisms of Melatonin Receptors 1A and 1B May Result in Disordered Lipid Metabolism in Obese Patients with Polycystic Ovary Syndrome. Mol. Med. Rep. 2019, 19, 2220–2230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, S.; Xu, J.; Shi, H.; Li, W.; Li, Q.; Sun, Y.-P. Association between Melatonin Receptor Gene Polymorphisms and Polycystic Ovarian Syndrome: A Systematic Review and Meta-Analysis. Biosci. Rep. 2020, 40, BSR20200824. [Google Scholar] [CrossRef] [PubMed]
- Nelson, L.M.; Ward, K.; Ogilvie, J.W. Genetic Variants in Melatonin Synthesis and Signaling Pathway Are Not Associated with Adolescent Idiopathic Scoliosis. Spine 2011, 36, 37–40. [Google Scholar] [CrossRef]
- Yang, M.; Wei, X.; Yang, W.; Li, Y.; Ni, H.; Zhao, Y.; Chen, Z.; Bai, Y.; Li, M. The Polymorphisms of Melatonin Receptor 1B Gene (MTNR1B) (Rs4753426 and Rs10830963) and Susceptibility to Adolescent Idiopathic Scoliosis: A Meta-Analysis. J. Orthop. Sci. 2015, 20, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Mahanna-Gabrielli, E.; Miano, T.A.; Augoustides, J.G.; Kim, C.; Bavaria, J.E.; Kofke, W.A. Does the Melatonin Receptor 1B Gene Polymorphism Have a Role in Postoperative Delirium? PLoS ONE 2018, 13, e0207941. [Google Scholar] [CrossRef]
- Machiela, M.J.; Lindström, S.; Allen, N.E.; Haiman, C.A.; Albanes, D.; Barricarte, A.; Berndt, S.I.; Bueno-de-Mesquita, H.B.; Chanock, S.; Gaziano, J.M.; et al. Association of Type 2 Diabetes Susceptibility Variants with Advanced Prostate Cancer Risk in the Breast and Prostate Cancer Cohort Consortium. Am. J. Epidemiol. 2012, 176, 1121–1129. [Google Scholar] [CrossRef]
- Deming, S.L.; Lu, W.; Beeghly-Fadiel, A.; Zheng, Y.; Cai, Q.; Long, J.; Shu, X.O.; Gao, Y.-T.; Zheng, W. Melatonin Pathway Genes and Breast Cancer Risk among Chinese Women. Breast Cancer Res. Treat. 2012, 132, 693–699. [Google Scholar] [CrossRef] [Green Version]
- Zienolddiny, S.; Haugen, A.; Lie, J.-A.S.; Kjuus, H.; Anmarkrud, K.H.; Kjærheim, K. Analysis of Polymorphisms in the Circadian-Related Genes and Breast Cancer Risk in Norwegian Nurses Working Night Shifts. Breast Cancer Res. 2013, 15, R53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, S.-C.; Ho, Y.-C.; Liu, Y.-F.; Reiter, R.J.; Chou, C.-H.; Yeh, C.-M.; Lee, H.-L.; Chung, W.-H.; Hsieh, M.-J.; Yang, S.-F. Association of Melatonin Membrane Receptor 1A/1B Gene Polymorphisms with the Occurrence and Metastasis of Hepatocellular Carcinoma. Oncotarget 2017, 8, 85655–85669. [Google Scholar] [CrossRef] [Green Version]
- Pierce, B.L.; Austin, M.A.; Ahsan, H. Association Study of Type 2 Diabetes Genetic Susceptibility Variants and Risk of Pancreatic Cancer: An Analysis of PanScan-I Data. Cancer Causes Control 2011, 22, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-D.; Yang, S.-F.; Wang, Y.-H.; Fang, W.-F.; Lin, Y.-C.; Liou, B.-C.; Lin, Y.-F.; Tang, K.-T.; Cheng, C.-W. Associations of Melatonin Receptor Gene Polymorphisms with Graves’ Disease. PLoS ONE 2017, 12, e0185529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanev, D.; Robeva, R.; Andonova, S.; Decheva, V.; Tomova, A.; Kumanov, P.; Savov, A.; Rashkov, R.; Kolarov, Z. Melatonin Receptor 1b Polymorphisms in Women with Systemic Lupus Erythematosus. Acta Reumatol. Port. 2016, 41, 62–67. [Google Scholar]
- Fayad, A.; Hassan, E.; Salem, T. Melatonin Receptor 1β Gene Polymorphism Rs10830963, Serum Melatonin, TNF-α, IL-6, IL-1β, in Egyptian Patients with Systemic Lupus Erythematosus. Egypt J. Immunol. 2019, 26, 101–112. [Google Scholar] [PubMed]
- Wu, L.; Cui, L.; Tam, W.H.; Ma, R.C.W.; Wang, C.C. Genetic Variants Associated with Gestational Diabetes Mellitus: A Meta-Analysis and Subgroup Analysis. Sci. Rep. 2016, 6, 30539. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Wang, Y.-K.; Qin, L.-Y.; Wei, Q.; Liu, N.; Jiang, M.; Yu, H.-P.; Yu, X.-Y. A Functional Polymorphism Rs10830963 in Melatonin Receptor 1B Associated with the Risk of Gestational Diabetes Mellitus. Biosci. Rep. 2019, 39, BSR20190744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, Y.; Tang, L.; Li, L.; Li, L. The Roles of ADIPOQ Rs266729 and MTNR1B Rs10830963 Polymorphisms in Patients with Gestational Diabetes Mellitus: A Meta-Analysis. Gene 2020, 730, 144302. [Google Scholar] [CrossRef] [PubMed]
- Jia, G.; Gao, Y.; Li, C.; Zhang, Y. Effects of MTNR1B Genetic Variants on Individual Susceptibility to Gestational Diabetes Mellitus: A Meta-Analysis. Am. J. Perinatol. 2020, 37, 607–612. [Google Scholar] [CrossRef]
- Lane, J.M.; Chang, A.-M.; Bjonnes, A.C.; Aeschbach, D.; Anderson, C.; Cade, B.E.; Cain, S.W.; Czeisler, C.A.; Gharib, S.A.; Gooley, J.J.; et al. Impact of Common Diabetes Risk Variant in MTNR1B on Sleep, Circadian, and Melatonin Physiology. Diabetes 2016, 65, 1741–1751. [Google Scholar] [CrossRef] [Green Version]
- Tan, X.; Ciuculete, D.-M.; Schiöth, H.B.; Benedict, C. Associations between Chronotype, MTNR1B Genotype and Risk of Type 2 Diabetes in UK Biobank. J. Intern. Med. 2020, 287, 189–196. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Minguez, J.; Saxena, R.; Bandín, C.; Scheer, F.A.; Garaulet, M. Late Dinner Impairs Glucose Tolerance in MTNR1B Risk Allele Carriers: A Randomized, Cross-over Study. Clin. Nutr. 2018, 37, 1133–1140. [Google Scholar] [CrossRef]
- Tuomi, T.; Nagorny, C.L.F.; Singh, P.; Bennet, H.; Yu, Q.; Alenkvist, I.; Isomaa, B.; Östman, B.; Söderström, J.; Pesonen, A.-K.; et al. Increased Melatonin Signaling Is a Risk Factor for Type 2 Diabetes. Cell Metab. 2016, 23, 1067–1077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garaulet, M.; Gómez-Abellán, P.; Rubio-Sastre, P.; Madrid, J.A.; Saxena, R.; Scheer, F.A.J.L. Common Type 2 Diabetes Risk Variant in MTNR1B Worsens the Deleterious Effect of Melatonin on Glucose Tolerance in Humans. Metabolism 2015, 64, 1650–1657. [Google Scholar] [CrossRef] [Green Version]
- Tan, X.; Benedict, C. Increased Risk of Myocardial Infarction among Patients with Type 2 Diabetes Who Carry the Common Rs10830963 Variant in the MTNR1B Gene. Diabetes Care 2020, 43, 2289–2292. [Google Scholar] [CrossRef] [PubMed]
- Bouatia-Naji, N.; Bonnefond, A.; Cavalcanti-Proença, C.; Sparsø, T.; Holmkvist, J.; Marchand, M.; Delplanque, J.; Lobbens, S.; Rocheleau, G.; Durand, E.; et al. A Variant near MTNR1B Is Associated with Increased Fasting Plasma Glucose Levels and Type 2 Diabetes Risk. Nat. Genet. 2009, 41, 89–94. [Google Scholar] [CrossRef]
- Xia, Q.; Chen, Z.-X.; Wang, Y.-C.; Ma, Y.-S.; Zhang, F.; Che, W.; Fu, D.; Wang, X.-F. Association between the Melatonin Receptor 1B Gene Polymorphism on the Risk of Type 2 Diabetes, Impaired Glucose Regulation: A Meta-Analysis. PLoS ONE 2012, 7, e50107. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Xie, L.; Zhong, M.; Yang, B.; Yang, Q.; Yang, H.; Xie, C. The Association between Melatonin Receptor 1B Gene Polymorphisms and Type 2 Diabetes Mellitus (T2DM) in Chinese Populations: A Meta-Analysis. Ann. Palliat. Med. 2020, 9, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.H.; Kim, S.-H.; Cho, Y.M.; Go, M.J.; Cho, Y.S.; Choi, S.H.; Moon, M.K.; Jung, H.S.; Shin, H.D.; Kang, H.M.; et al. A Genome-Wide Association Study of Gestational Diabetes Mellitus in Korean Women. Diabetes 2012, 61, 531–541. [Google Scholar] [CrossRef] [Green Version]
- Bonnefond, A.; Clément, N.; Fawcett, K.; Yengo, L.; Vaillant, E.; Guillaume, J.-L.; Dechaume, A.; Payne, F.; Roussel, R.; Czernichow, S.; et al. Rare MTNR1B Variants Impairing Melatonin Receptor 1B Function Contribute to Type 2 Diabetes. Nat. Genet. 2012, 44, 297–301. [Google Scholar] [CrossRef]
- Anim-Koranteng, C.; Shah, H.E.; Bhawnani, N.; Ethirajulu, A.; Alkasabera, A.; Onyali, C.B.; Mostafa, J.A. Melatonin—A New Prospect in Prostate and Breast Cancer Management. Cureus 2021, 13, e18124. [Google Scholar] [CrossRef]
- Tam, C.W.; Mo, C.W.; Yao, K.-M.; Shiu, S.Y.W. Signaling Mechanisms of Melatonin in Antiproliferation of Hormone-Refractory 22Rv1 Human Prostate Cancer Cells: Implications for Prostate Cancer Chemoprevention. J. Pineal Res. 2007, 42, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Carbajo-Pescador, S.; Martín-Renedo, J.; García-Palomo, A.; Tuñón, M.J.; Mauriz, J.L.; González-Gallego, J. Changes in the Expression of Melatonin Receptors Induced by Melatonin Treatment in Hepatocarcinoma HepG2 Cells. J. Pineal Res. 2009, 47, 330–338. [Google Scholar] [CrossRef]
- Sánchez-Hidalgo, M.; Lee, M.; de la Lastra, C.A.; Guerrero, J.M.; Packham, G. Melatonin Inhibits Cell Proliferation and Induces Caspase Activation and Apoptosis in Human Malignant Lymphoid Cell Lines. J. Pineal Res. 2012, 53, 366–373. [Google Scholar] [CrossRef]
- Pawlikowski, M.; Winczyk, K.; Karasek, M. Oncostatic Action of Melatonin: Facts and Question Marks. Neuro Endocrinol. Lett. 2002, 23 (Suppl. S1), 24–29. [Google Scholar]
- Srinivasan, V.; Spence, D.W.; Pandi-Perumal, S.R.; Trakht, I.; Cardinali, D.P. Therapeutic Actions of Melatonin in Cancer: Possible Mechanisms. Integr. Cancer Ther. 2008, 7, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, S.; Zhou, Y.; Meng, X.; Zhang, J.-J.; Xu, D.-P.; Li, H.-B. Melatonin for the Prevention and Treatment of Cancer. Oncotarget 2017, 8, 39896–39921. [Google Scholar] [CrossRef] [Green Version]
- Kiefer, T.; Ram, P.T.; Yuan, L.; Hill, S.M. Melatonin Inhibits Estrogen Receptor Transactivation and CAMP Levels in Breast Cancer Cells. Breast Cancer Res. Treat. 2002, 71, 37–45. [Google Scholar] [CrossRef]
- Girgert, R.; Hanf, V.; Emons, G.; Gründker, C. Membrane-Bound Melatonin Receptor MT1 down-Regulates Estrogen Responsive Genes in Breast Cancer Cells. J. Pineal Res. 2009, 47, 23–31. [Google Scholar] [CrossRef]
- Blask, D.E.; Hill, S.M.; Dauchy, R.T.; Xiang, S.; Yuan, L.; Duplessis, T.; Mao, L.; Dauchy, E.; Sauer, L.A. Circadian Regulation of Molecular, Dietary, and Metabolic Signaling Mechanisms of Human Breast Cancer Growth by the Nocturnal Melatonin Signal and the Consequences of Its Disruption by Light at Night. J. Pineal Res. 2011, 51, 259–269. [Google Scholar] [CrossRef]
- Sanchez-Barcelo, E.J.; Mediavilla, M.D.; Alonso-Gonzalez, C.; Rueda, N. Breast Cancer Therapy Based on Melatonin. Recent Pat. Endocr. Metab. Immune Drug Discov. 2012, 6, 108–116. [Google Scholar] [CrossRef]
- Hill, S.M.; Cheng, C.; Yuan, L.; Mao, L.; Jockers, R.; Dauchy, B.; Frasch, T.; Blask, D.E. Declining Melatonin Levels and MT1 Receptor Expression in Aging Rats Is Associated with Enhanced Mammary Tumor Growth and Decreased Sensitivity to Melatonin. Breast Cancer Res. Treat. 2011, 127, 91–98. [Google Scholar] [CrossRef]
- De Castro, T.B.; Bordin-Junior, N.A.; de Almeida, E.A.; de Campos Zuccari, D.A.P. Evaluation of Melatonin and AFMK Levels in Women with Breast Cancer. Endocrine 2018, 62, 242–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schernhammer, E.S.; Hankinson, S.E. Urinary Melatonin Levels and Postmenopausal Breast Cancer Risk in the Nurses’ Health Study Cohort. Cancer Epidemiol. Biomark. Prev. 2009, 18, 74–79. [Google Scholar] [CrossRef] [Green Version]
- Devore, E.E.; Warner, E.T.; Eliassen, A.H.; Brown, S.B.; Beck, A.H.; Hankinson, S.E.; Schernhammer, E.S. Urinary Melatonin in Relation to Postmenopausal Breast Cancer Risk According to Melatonin 1 Receptor Status. Cancer Epidemiol. Biomark. Prev. 2017, 26, 413–419. [Google Scholar] [CrossRef] [Green Version]
- Basler, M.; Jetter, A.; Fink, D.; Seifert, B.; Kullak-Ublick, G.A.; Trojan, A. Urinary Excretion of Melatonin and Association with Breast Cancer: Meta-Analysis and Review of the Literature. Breast Care 2014, 9, 182–187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.; Huang, L.; Sun, G.-P. Urinary 6-Sulfatoxymelatonin Level and Breast Cancer Risk: Systematic Review and Meta-Analysis. Sci. Rep. 2017, 7, 5353. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, K.; Pula, B.; Zemla, A.; Owczarek, T.; Wojnar, A.; Rys, J.; Ambicka, A.; Podhorska-Okolow, M.; Ugorski, M.; Dziegiel, P. Expression of Melatonin Receptor MT1 in Cells of Human Invasive Ductal Breast Carcinoma. J. Pineal Res. 2013, 54, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Oprea-Ilies, G.; Haus, E.; Sackett-Lundeen, L.; Liu, Y.; McLendon, L.; Busch, R.; Adams, A.; Cohen, C. Expression of Melatonin Receptors in Triple Negative Breast Cancer (TNBC) in African American and Caucasian Women: Relation to Survival. Breast Cancer Res. Treat. 2013, 137, 677–687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González-González, A.; Mediavilla, M.D.; Sánchez-Barceló, E.J. Melatonin: A Molecule for Reducing Breast Cancer Risk. Molecules 2018, 23, 336. [Google Scholar] [CrossRef] [Green Version]
- Pham, T.-T.; Lee, E.-S.; Kong, S.-Y.; Kim, J.; Kim, S.-Y.; Joo, J.; Yoon, K.-A.; Park, B. Night-Shift Work, Circadian and Melatonin Pathway Related Genes and Their Interaction on Breast Cancer Risk: Evidence from a Case-Control Study in Korean Women. Sci. Rep. 2019, 9, 10982. [Google Scholar] [CrossRef] [Green Version]
- Rabstein, S.; Harth, V.; Justenhoven, C.; Pesch, B.; Plöttner, S.; Heinze, E.; Lotz, A.; Baisch, C.; Schiffermann, M.; Brauch, H.; et al. Polymorphisms in Circadian Genes, Night Work and Breast Cancer: Results from the GENICA Study. Chronobiol. Int. 2014, 31, 1115–1122. [Google Scholar] [CrossRef]
- Hasan, M.; Marzouk, M.A.; Adhikari, S.; Wright, T.D.; Miller, B.P.; Matossian, M.D.; Elliott, S.; Wright, M.; Alzoubi, M.; Collins-Burow, B.M.; et al. Pharmacological, Mechanistic, and Pharmacokinetic Assessment of Novel Melatonin-Tamoxifen Drug Conjugates as Breast Cancer Drugs. Mol. Pharm. 2019, 96, 272–296. [Google Scholar] [CrossRef]
- Tam, C.W.; Shiu, S.Y.W. Functional Interplay between Melatonin Receptor-Mediated Antiproliferative Signaling and Androgen Receptor Signaling in Human Prostate Epithelial Cells: Potential Implications for Therapeutic Strategies against Prostate Cancer. J. Pineal Res. 2011, 51, 297–312. [Google Scholar] [CrossRef]
- Shiu, S.Y.W.; Leung, W.Y.; Tam, C.W.; Liu, V.W.S.; Yao, K.-M. Melatonin MT1 Receptor-Induced Transcriptional up-Regulation of P27(Kip1) in Prostate Cancer Antiproliferation Is Mediated via Inhibition of Constitutively Active Nuclear Factor Kappa B (NF-ΚB): Potential Implications on Prostate Cancer Chemoprevention and Therapy. J. Pineal Res. 2013, 54, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Liu, V.W.S.; Yau, W.L.; Tam, C.W.; Yao, K.-M.; Shiu, S.Y.W. Melatonin Inhibits Androgen Receptor Splice Variant-7 (AR-V7)-Induced Nuclear Factor-Kappa B (NF-ΚB) Activation and NF-ΚB Activator-Induced AR-V7 Expression in Prostate Cancer Cells: Potential Implications for the Use of Melatonin in Castration-Resistant Prostate Cancer (CRPC) Therapy. Int. J. Mol. Sci. 2017, 18, 1130. [Google Scholar] [CrossRef] [Green Version]
- Shen, D.; Ju, L.; Zhou, F.; Yu, M.; Ma, H.; Zhang, Y.; Liu, T.; Xiao, Y.; Wang, X.; Qian, K. The Inhibitory Effect of Melatonin on Human Prostate Cancer. Cell Commun. Signal. 2021, 19, 34. [Google Scholar] [CrossRef]
- Jung-Hynes, B.; Schmit, T.L.; Reagan-Shaw, S.R.; Siddiqui, I.A.; Mukhtar, H.; Ahmad, N. Melatonin, a Novel Sirt1 Inhibitor, Imparts Antiproliferative Effects against Prostate Cancer in Vitro in Culture and in Vivo in TRAMP Model. J. Pineal Res. 2011, 50, 140–149. [Google Scholar] [CrossRef] [Green Version]
- Sigurdardottir, L.G.; Markt, S.C.; Rider, J.R.; Haneuse, S.; Fall, K.; Schernhammer, E.S.; Tamimi, R.M.; Flynn-Evans, E.; Batista, J.L.; Launer, L.; et al. Urinary Melatonin Levels, Sleep Disruption, and Risk of Prostate Cancer in Elderly Men. Eur. Urol. 2015, 67, 191–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lissoni, P.; Cazzaniga, M.; Tancini, G.; Scardino, E.; Musci, R.; Barni, S.; Maffezzini, M.; Meroni, T.; Rocco, F.; Conti, A.; et al. Reversal of Clinical Resistance to LHRH Analogue in Metastatic Prostate Cancer by the Pineal Hormone Melatonin: Efficacy of LHRH Analogue plus Melatonin in Patients Progressing on LHRH Analogue Alone. Eur. Urol. 1997, 31, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Zharinov, G.M.; Bogomolov, O.A.; Chepurnaya, I.V.; Neklasova, N.Y.; Anisimov, V.N. Melatonin Increases Overall Survival of Prostate Cancer Patients with Poor Prognosis after Combined Hormone Radiation Treatment. Oncotarget 2020, 11, 3723–3729. [Google Scholar] [CrossRef]
- Chuffa, L.G.d.A.; Reiter, R.J.; Lupi, L.A. Melatonin as a Promising Agent to Treat Ovarian Cancer: Molecular Mechanisms. Carcinogenesis 2017, 38, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Chuffa, L.G.A.; Fioruci-Fontanelli, B.A.; Mendes, L.O.; Fávaro, W.J.; Pinheiro, P.F.F.; Martinez, M.; Martinez, F.E. Characterization of Chemically Induced Ovarian Carcinomas in an Ethanol-Preferring Rat Model: Influence of Long-Term Melatonin Treatment. PLoS ONE 2013, 8, e81676. [Google Scholar] [CrossRef]
- Sivas, M.C.; Tapisiz, O.L.; Ayik, R.T.; Kahraman, D.; Kiykac Altinbas, S.; Moraloglu Tekin, O. Effects of Melatonin on Uterine Hypertrophy/Hyperplasia: A Preliminary Experimental Rat Study. Heliyon 2020, 6, e05142. [Google Scholar] [CrossRef]
- Gu, C.; Yang, H.; Chang, K.; Zhang, B.; Xie, F.; Ye, J.; Chang, R.; Qiu, X.; Wang, Y.; Qu, Y.; et al. Melatonin Alleviates Progression of Uterine Endometrial Cancer by Suppressing Estrogen/Ubiquitin C/SDHB-Mediated Succinate Accumulation. Cancer Lett. 2020, 476, 34–47. [Google Scholar] [CrossRef]
- Dauchy, R.T.; Blask, D.E.; Dauchy, E.M.; Davidson, L.K.; Tirrell, P.C.; Greene, M.W.; Tirrell, R.P.; Hill, C.R.; Sauer, L.A. Antineoplastic Effects of Melatonin on a Rare Malignancy of Mesenchymal Origin: Melatonin Receptor-Mediated Inhibition of Signal Transduction, Linoleic Acid Metabolism and Growth in Tissue-Isolated Human Leiomyosarcoma Xenografts. J. Pineal Res. 2009, 47, 32–42. [Google Scholar] [CrossRef]
- Pariente, R.; Pariente, J.A.; Rodríguez, A.B.; Espino, J. Melatonin Sensitizes Human Cervical Cancer HeLa Cells to Cisplatin-Induced Cytotoxicity and Apoptosis: Effects on Oxidative Stress and DNA Fragmentation. J. Pineal Res. 2016, 60, 55–64. [Google Scholar] [CrossRef]
- Shafabakhsh, R.; Reiter, R.J.; Mirzaei, H.; Teymoordash, S.N.; Asemi, Z. Melatonin: A New Inhibitor Agent for Cervical Cancer Treatment. J. Cell Physiol. 2019, 234, 21670–21682. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.A.; Tan, D.-X.; Acuna-Castroviejo, D.; Qin, L.; Yang, S.-F.; Xu, K. Melatonin, a Full Service Anti-Cancer Agent: Inhibition of Initiation, Progression and Metastasis. Int. J. Mol. Sci. 2017, 18, 843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, F.-Y.; Lin, C.-W.; Yang, S.-F.; Lee, W.-J.; Lin, Y.-W.; Lee, L.-M.; Chang, J.-L.; Weng, W.-C.; Lin, C.-H.; Chien, M.-H. Interactions between Environmental Factors and Melatonin Receptor Type 1A Polymorphism in Relation to Oral Cancer Susceptibility and Clinicopathologic Development. PLoS ONE 2015, 10, e0121677. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, E.; Kozaki, K.; Tsuda, H.; Suzuki, E.; Pimkhaokham, A.; Yamamoto, G.; Irie, T.; Tachikawa, T.; Amagasa, T.; Inazawa, J.; et al. Frequent Silencing of a Putative Tumor Suppressor Gene Melatonin Receptor 1 A (MTNR1A) in Oral Squamous-Cell Carcinoma. Cancer Sci. 2008, 99, 1390–1400. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-W.; Wang, S.-S.; Wen, Y.-C.; Tung, M.-C.; Lee, L.-M.; Yang, S.-F.; Chien, M.-H. Genetic Variations of Melatonin Receptor Type 1A Are Associated with the Clinicopathologic Development of Urothelial Cell Carcinoma. Int. J. Med. Sci. 2017, 14, 1130–1135. [Google Scholar] [CrossRef] [PubMed]
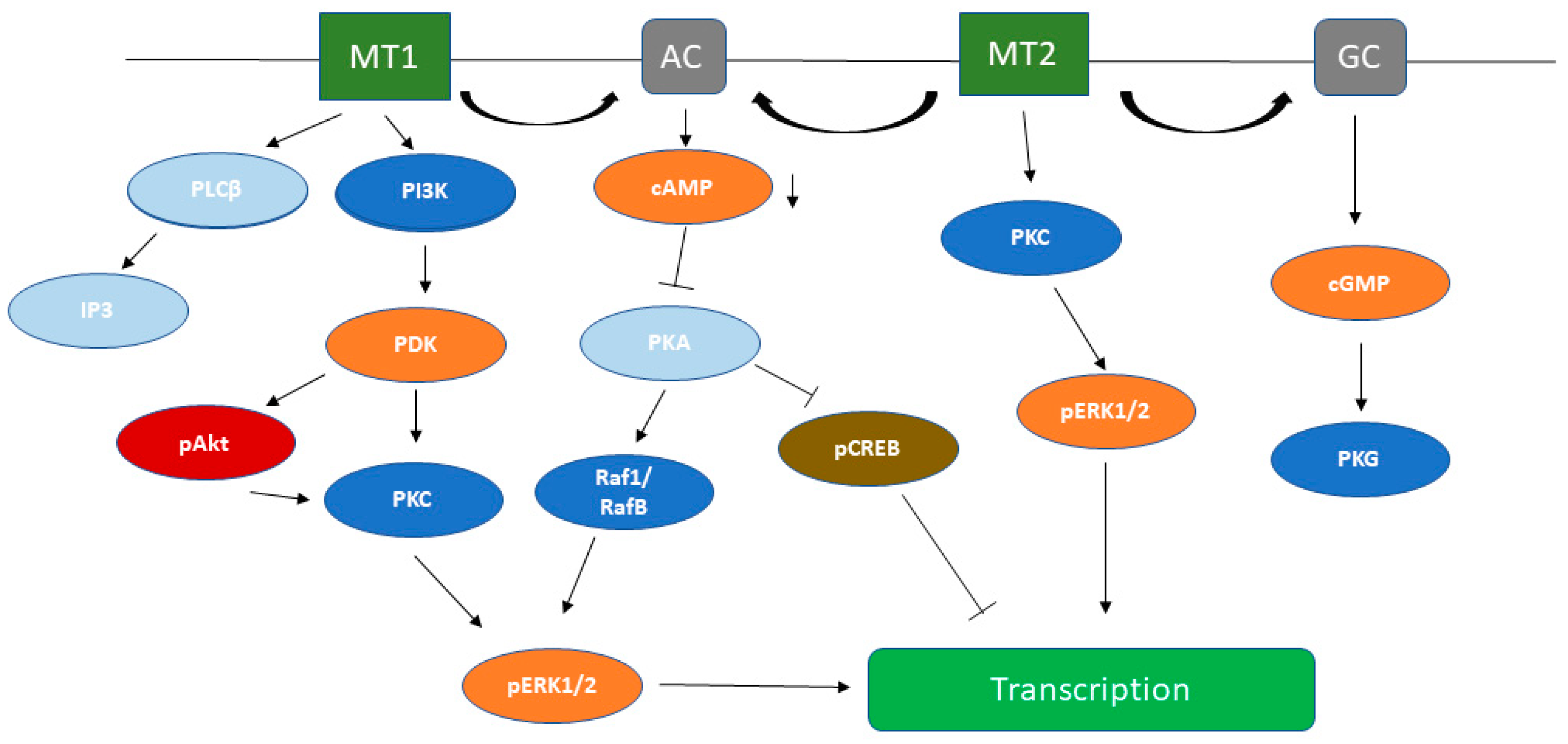
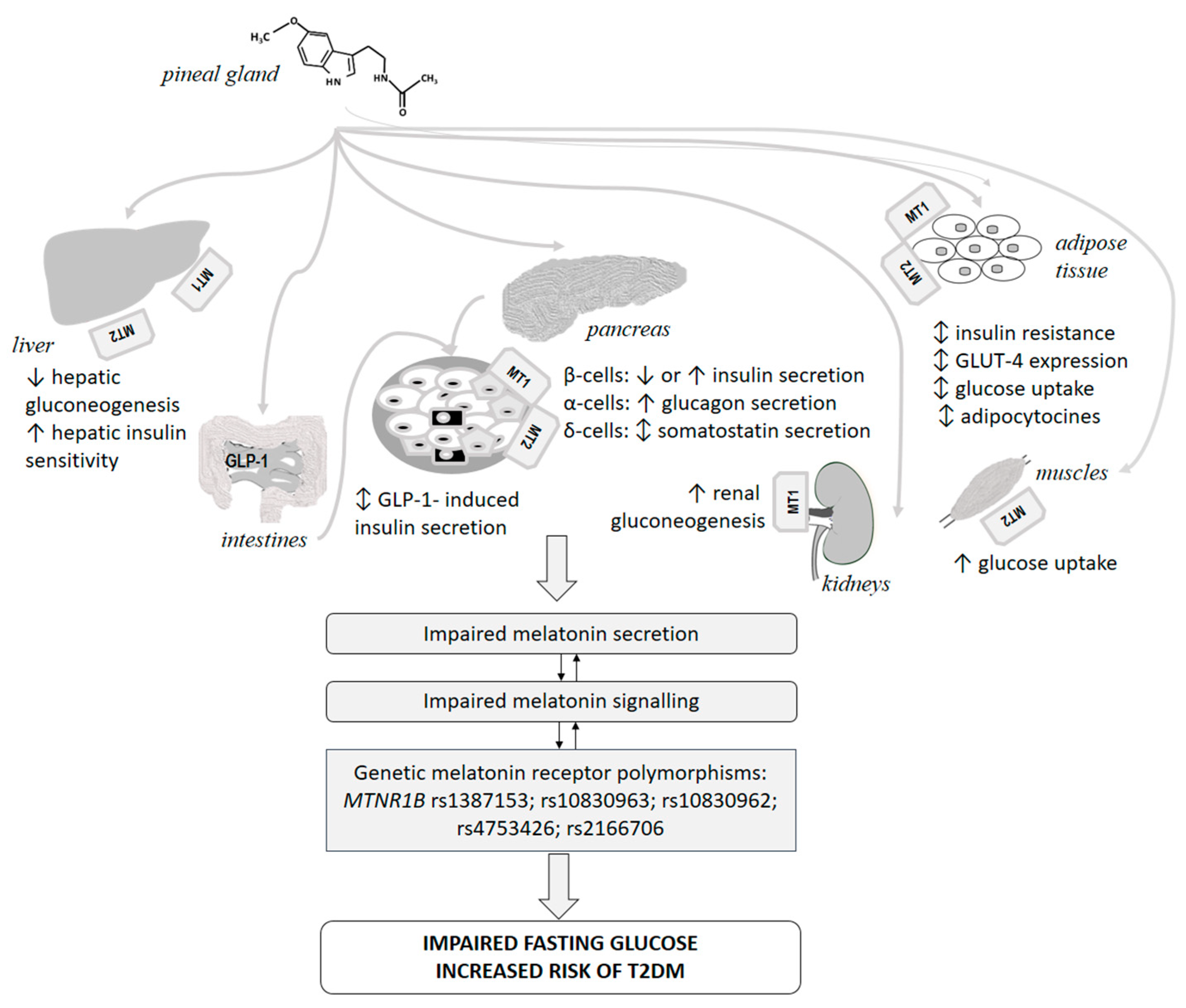
| Receptor Type | Distribution | Function | Signaling Pathway | Reference |
|---|---|---|---|---|
| MT1 | Human peripheral blood mononuclear cells | Regulation of interleukin-2 production Cytoskeletal actin rearrangement CD25+ expression Regulation of interleukin-2 production | Inhibition of cAMP accumulation Inhibition of T-mitogen dependent proliferation Counteraction of prostaglandin EP3 receptor induced Ca2+ influx | [46,47,105] |
| Jurkat cells | Regulation of interleukin-2 production | [106] | ||
| Human T cells | Regulation of interleukin-2 and interleukin-2 receptor (CD25) production | Inhibition of forskolin-stimulated cyclic AMP (cAMP) production, cyclic GMP (cGMP) production and diacylglycerol (DAG) production | [107,108] | |
| Human monocytes | [109] | |||
| Mouse peritoneal macrophages | Pertussis toxin-sensitive G protein inhibition of adenylyl cyclase | [110] | ||
| Rat B cells and T cells | [111] | |||
| Rat spleen and thymus | Maturation of secondary immune response | [20,61] | ||
| Mouse splenocytes | Increase of proliferation | [112] | ||
| MT2 | Rat leukocytes | Inhibition of motility (rolling) | [12] | |
| Chick spleen | Regulation of splenocyte proliferation | [113] | ||
| Jurkat cells | Regulation of of interleukin-2 production | [62] | ||
| Human T cells | Regulation of interleukin-2 and interleukin-2 receptor (CD25) production | cAMP—dependent signaling | [108] | |
| Human peripheral blood mononuclear cells | Modulation of cell life/death balance of human leucocytes | Inhibition of TNF-α Activation of ERK signaling | [114] | |
| MT1/MT2 | U937 cells | Prevention of apoptosis induced by ultraviolet irradiation | Activation of extracellular signal-regulated protein kinase (ERK) and mitogen-activated protein kinase (MAPK) pathway | [115,116,117] |
| Clinical Condition | MTNR1B, rs10830963 C>G Effects | References |
|---|---|---|
| Glucose abnormalities, Beta-cells function, Insulin resistance, Type 2 diabetes mellitus | Minor G-allele is associated with increased fasting glucose as well as with increased prevalence of impaired fasting glucose in children, adolescents and adults. The effect is more pronounced in younger individuals. | [169,176,177,178,184,185,186,187,188,189,190,191,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206,207,208,209,210] |
| G-allele is associated with increased risk of type 2 diabetes mellitus in most but not all ethnic groups, and especially in elderly patients. | [169,177,178,182,184,185,188,190,192,202,208,209,211,212,213,214,215,216,217,218,219] | |
| G allele is associated with increased pancreatic islet MTNR1B expression and decreased beta cell glucose sensitivity. | [169,186,191,195,220,221] | |
| G-allele is associated with decreased early phase insulin secretion after glucose load. | [169,176,178,203,213,222,223,224] | |
| G allele is not associated with peripheral insulin sensitivity, but is related to hepatic insulin resistance. | [178,186,190] | |
| G allele determines a significantly higher risk of transition from normal fasting glucose to impaired fasting glucose than from impaired fasting glucose to T2DM. | [181] | |
| Gestational Diabetes Mellitus | G allele is associated with gestational DM in almost all studies. The expression level of MT2 in placenta is significantly higher in the GDM patients than in controls as well as in G allele carriers in comparison to C allele carriers. | [225,226,227,228,229,230,231,232,233,234,235,236,237,238,239,240,241,242,243] |
| G allele determines lower effect of lifestyle intervention on the development of GDM. | [234] | |
| Maternal G allele is associated with higher offspring birth weight. | [244] | |
| Autoimmune diabetes mellitus | G allele is associated with increased risk of low GAD autoantibodies LADA. | [245] |
| Obesity and weight loss | G allele is not associated with obesity in adults. The polymorphism might exert gender-specific modulation effect on body weight in children. | [176,205,207,246,247] |
| G allele determines lower weight loss and weaker improvement of metabolic parameters on hypocaloric diet. | [248,249,250] | |
| G allele carriers might benefit from hypocaloric low-fat diet leading to a decreased weight, better fat distribution and pronounced decrease of total and LDL- cholesterol in comparison to high-fat diet. | [251,252] | |
| PCOS | G-allele is not associated with increased prevalence of PCOS according to some studies, but is a risk factor for PCOS development according to other studies and a meta-analysis. | [253,254,255,256,257] |
| The variant modulates serum glucose and insulin concentrations during OGTT in women with PCOS. G-allele is associated with increased testosterone levels in one, but not other studies. | [253,255,256] | |
| Adolescent idiopathic scoliosis | No significant associations with adolescent idiopathic scoliosis. | [258,259] |
| Neuro-psychological disturbances | GG genotype associated with an increased risk of postoperative delirium. | [260] |
| Oncological and autoimmune diseases | Nominal association with increased prostate cancer risk, not significant after correction for multiple comparisons. | [261] |
| Possible association with breast cancer risk in some but not all studies, especially in individuals exposed to night-shift work. | [262,263] | |
| No association with hepatocellular carcinoma risk. | [264] | |
| Nominal association with increased pancreatic cancer risk. | [265] | |
| No association with Graves’ disease. | [266] | |
| G-allele is associated with increased risk of systemic lupus erythematosus in one population, but not in other. | [267,268] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikolaev, G.; Robeva, R.; Konakchieva, R. Membrane Melatonin Receptors Activated Cell Signaling in Physiology and Disease. Int. J. Mol. Sci. 2022, 23, 471. https://doi.org/10.3390/ijms23010471
Nikolaev G, Robeva R, Konakchieva R. Membrane Melatonin Receptors Activated Cell Signaling in Physiology and Disease. International Journal of Molecular Sciences. 2022; 23(1):471. https://doi.org/10.3390/ijms23010471
Chicago/Turabian StyleNikolaev, Georgi, Ralitsa Robeva, and Rossitza Konakchieva. 2022. "Membrane Melatonin Receptors Activated Cell Signaling in Physiology and Disease" International Journal of Molecular Sciences 23, no. 1: 471. https://doi.org/10.3390/ijms23010471
APA StyleNikolaev, G., Robeva, R., & Konakchieva, R. (2022). Membrane Melatonin Receptors Activated Cell Signaling in Physiology and Disease. International Journal of Molecular Sciences, 23(1), 471. https://doi.org/10.3390/ijms23010471







