Pulsed Electric Fields Alter Expression of NF-κB Promoter-Controlled Gene
Abstract
1. Introduction
2. Results
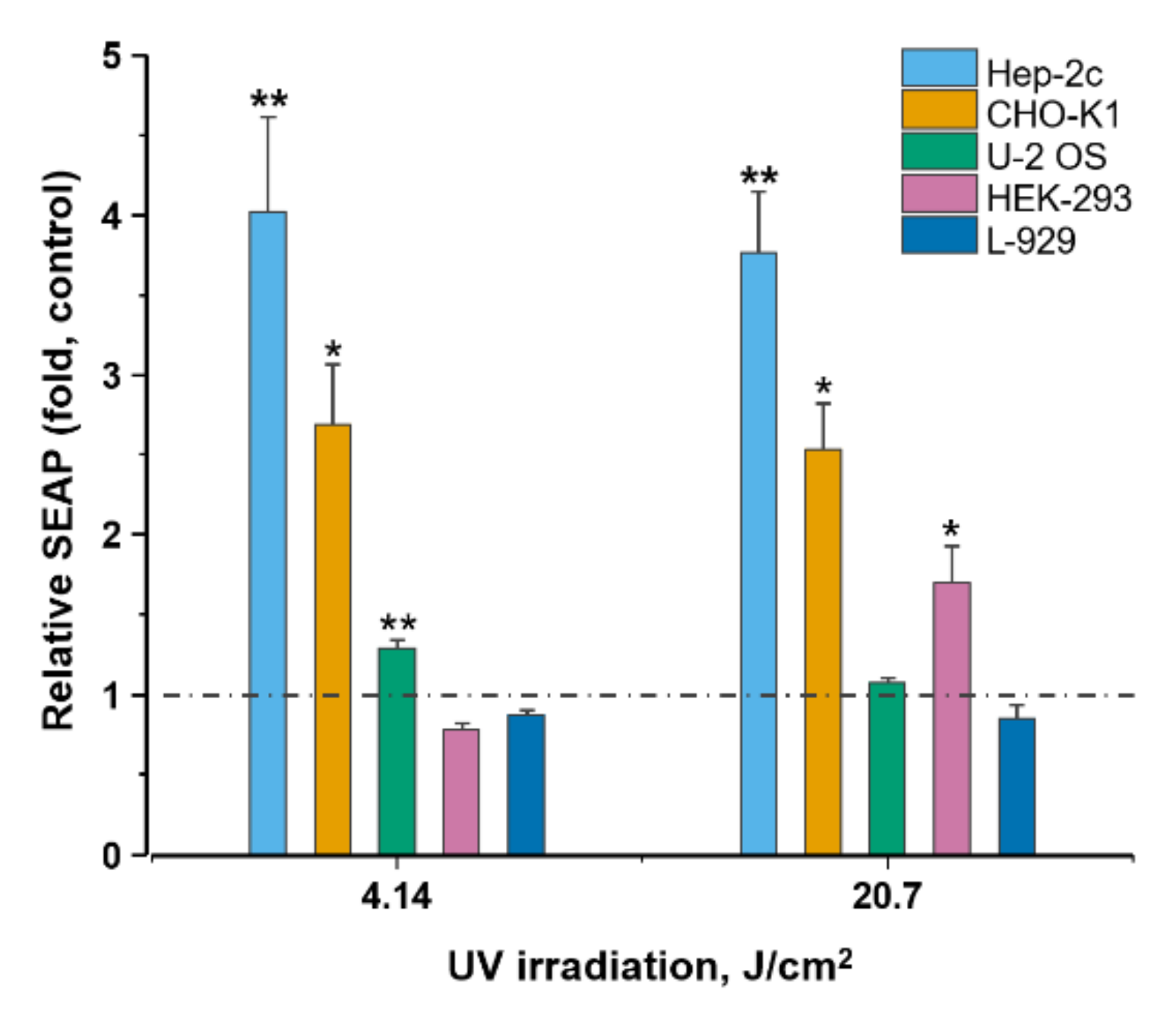
2.1. Validation of Experimental Setup
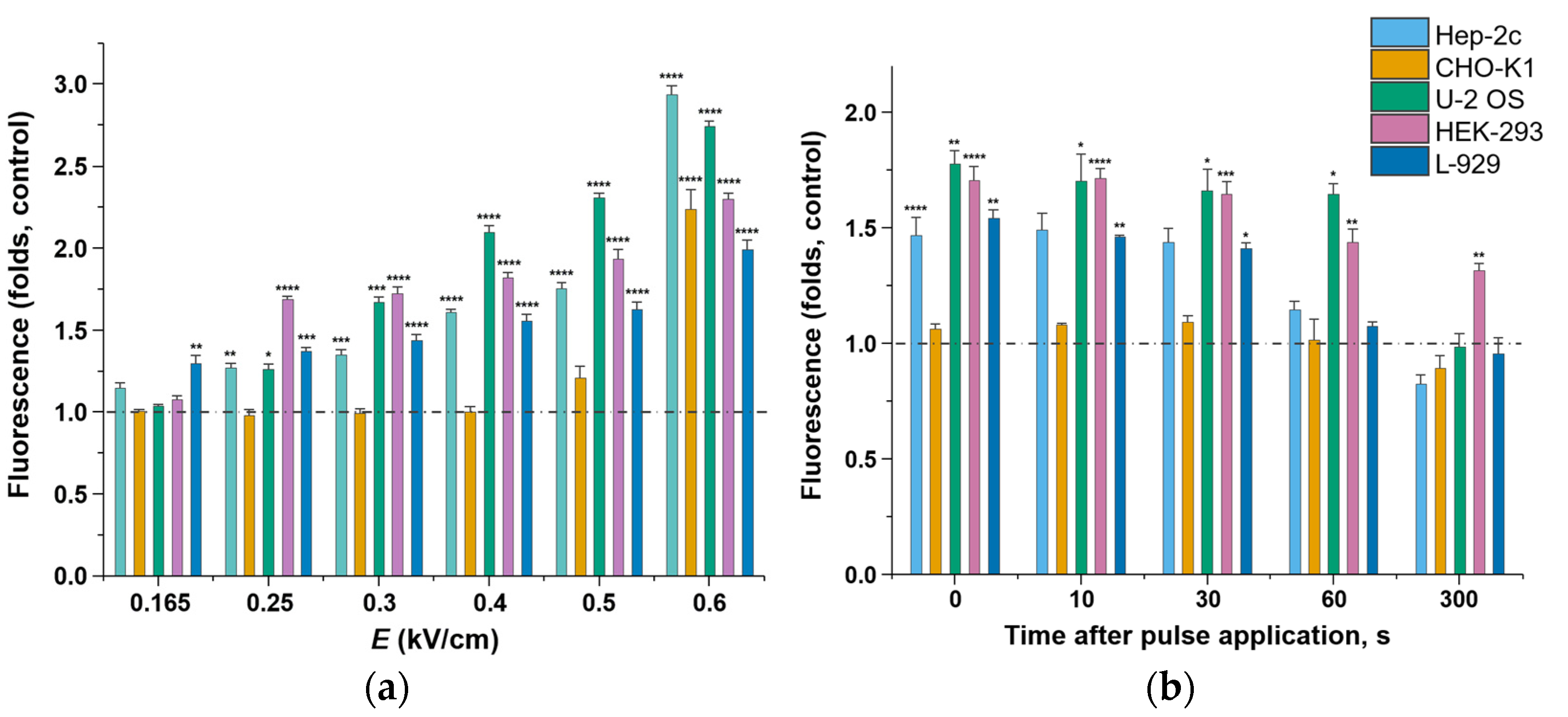
2.2. Membrane Permeability after Microsecond-Duration Pulsed Electric Fields Treatment
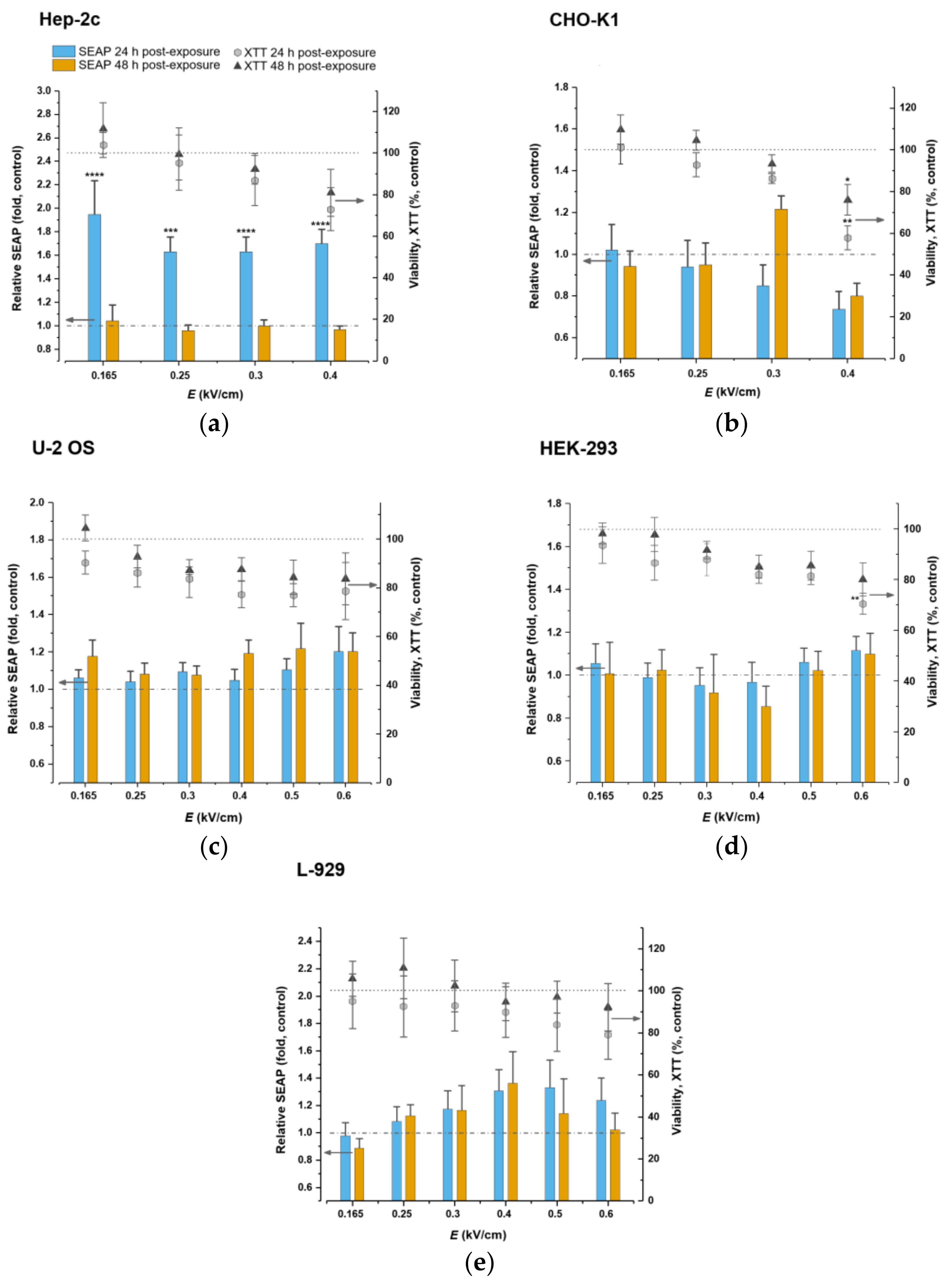
2.3. Cell Viability and NF-kB Pathway-Controlled SEAP Reporter System Response to Microsecond-Duration Pulsed Electric Fields Treatment
3. Discussion
4. Materials and Methods
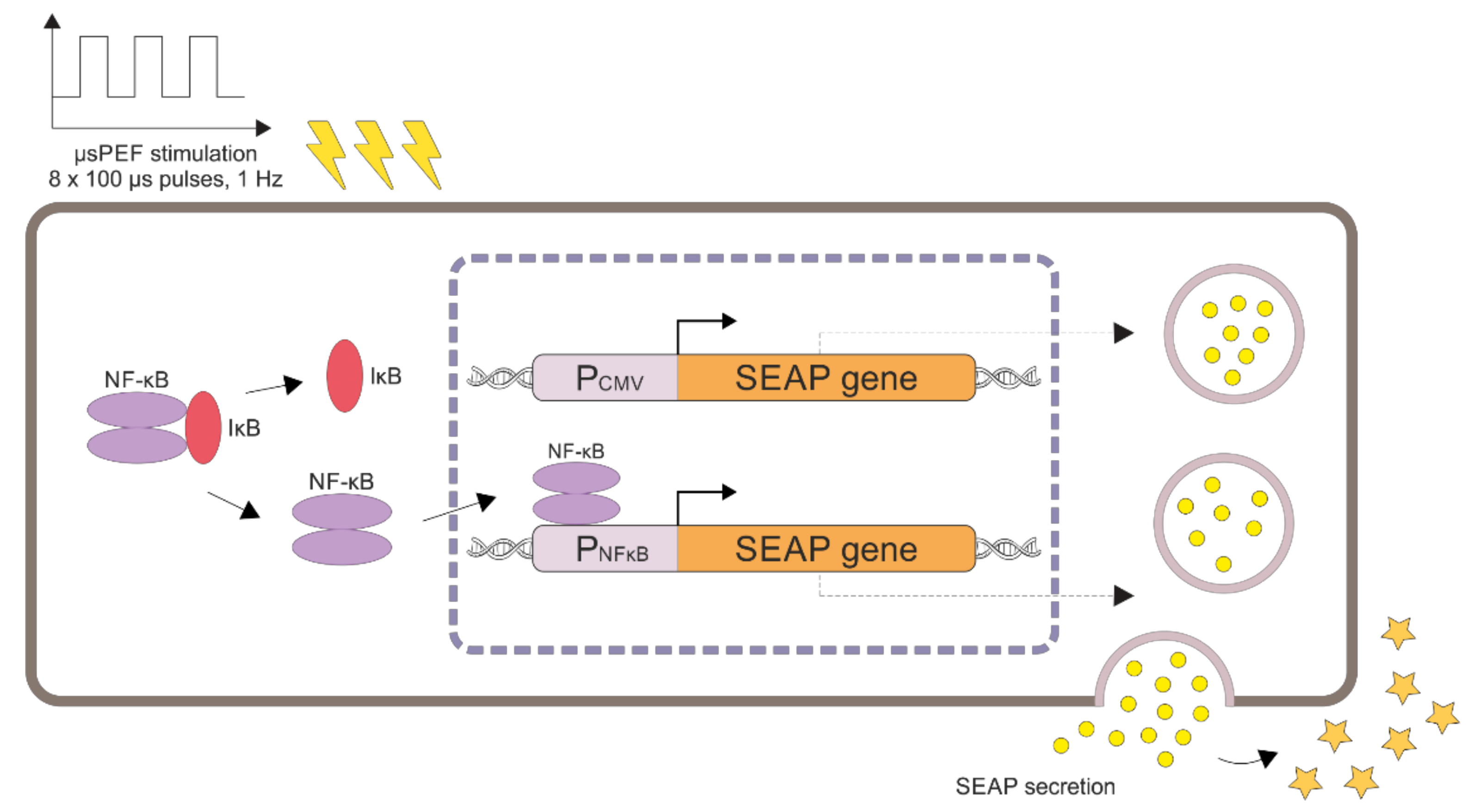
4.1. Experimental Setup
4.2. Cell Culture
4.3. Plasmids
4.4. Plasmid DNA Transfection
4.5. Cell Exposure to Ultraviolet Light
4.6. Cell Exposure to Microsecond-Duration Pulsed Electric Fields
4.7. SEAP Reporter Gene Expression Evaluation
4.8. Metabolic Activity (Viability) Assay
4.9. Membrane Permeabilization Assay
4.10. Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stuible, M.; Burlacu, A.; Perret, S.; Brochu, D.; Paul-Roc, B.; Baardsnes, J.; Loignon, M.; Grazzini, E.; Durocher, Y. Optimization of a high-cell-density polyethylenimine transfection method for rapid protein production in CHO-EBNA1 cells. J. Biotechnol. 2018, 281, 39–47. [Google Scholar] [CrossRef]
- Lillacci, G.; Benenson, Y.; Khammash, M. Synthetic control systems for high performance gene expression in mammalian cells. Nucleic Acids Res. 2018, 46, 9855–9863. [Google Scholar] [CrossRef]
- Rao, R.C.; Zacks, D.N. Cell and Gene Therapy. Cell-Based Ther. Retin. Degener. Dis. 2014, 53, 167–177. [Google Scholar] [CrossRef]
- Lienert, F.; Lohmueller, J.; Garg, A.D.; Silver, P.A. Synthetic biology in mammalian cells: Next generation research tools and therapeutics. Nat. Rev. Mol. Cell Biol. 2014, 15, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Re, A. Synthetic Gene Expression Circuits for Designing Precision Tools in Oncology. Front. Cell Dev. Biol. 2017, 5, 77. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, R.; Berges, B.K.; Solis-Leal, A.; Igbinedion, O.; Strong, C.L.; Schiller, M.R. TALEN gene editing takes aim on HIV. Qual. Life Res. 2016, 135, 1059–1070. [Google Scholar] [CrossRef]
- Cooney, A.; Alaiwa, M.A.; Shah, V.S.; Bouzek, D.C.; Stroik, M.R.; Powers, L.S.; Gansemer, N.D.; Meyerholz, D.; Welsh, M.J.; Stoltz, D.A.; et al. Lentiviral-mediated phenotypic correction of cystic fibrosis pigs. JCI Insight 2016, 1, e88730. [Google Scholar] [CrossRef]
- Samson, S.L.; Gonzalez, E.V.; Yechoor, V.; Bajaj, M.; Oka, K.; Chan, L. Gene Therapy for Diabetes: Metabolic Effects of Helper-dependent Adenoviral Exendin 4 Expression in a Diet-induced Obesity Mouse Model. Mol. Ther. 2008, 16, 1805–1812. [Google Scholar] [CrossRef]
- Pedone, E.; Postiglione, L.; Aulicino, F.; Rocca, D.L.; Montes-Olivas, S.; Khazim, M.; di Bernardo, D.; Cosma, M.P.; Marucci, L. A tunable dual-input system for on-demand dynamic gene expression regulation. Nat. Commun. 2019, 10, 4481. [Google Scholar] [CrossRef]
- Clackson, T. Regulated gene expression systems. Gene Ther. 2000, 7, 120–125. [Google Scholar] [CrossRef]
- Fussenegger, M. The Impact of Mammalian Gene Regulation Concepts on Functional Genomic Research, Metabolic Engineering, and Advanced Gene Therapies. Biotechnol. Prog. 2001, 17, 1–51. [Google Scholar] [CrossRef] [PubMed]
- Ausländer, S.; Fussenegger, M. From gene switches to mammalian designer cells: Present and future prospects. Trends Biotechnol. 2013, 31, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Weber, W.; Fussenegger, M. Artificial mammalian gene regulation networks—Novel approaches for gene therapy and bioengineering. J. Biotechnol. 2002, 98, 161–187. [Google Scholar] [CrossRef]
- Koh, J.-T.; Ge, C.; Zhao, M.; Wang, Z.; Krebsbach, P.H.; Zhao, Z.; Franceschi, R.T. Use of a Stringent Dimerizer-Regulated Gene Expression System for Controlled BMP2 Delivery. Mol. Ther. 2006, 14, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Stieger, K.; Belbellaa, B.; Le Guiner, C.; Moullier, P.; Rolling, F. In vivo gene regulation using tetracycline-regulatable systems. Adv. Drug Deliv. Rev. 2009, 61, 527–541. [Google Scholar] [CrossRef] [PubMed]
- Berens, C.; Hillen, W. Gene regulation by tetracyclines. Constraints of resistance regulation in bacteria shape TetR for application in eukaryotes. JBIC J. Biol. Inorg. Chem. 2003, 270, 3109–3121. [Google Scholar] [CrossRef]
- Zhou, X.; Vink, M.; Klaver, B.; Berkhout, B.; Das, A.T. Optimization of the Tet-On system for regulated gene expression through viral evolution. Gene Ther. 2006, 13, 1382–1390. [Google Scholar] [CrossRef]
- Chua, S.S.; Wang, Y.; DeMayo, F.J.; O’Malley, B.W.; Tsai, S.Y. A novel RU486 inducible system for the activation and repression of genes. Adv. Drug Deliv. Rev. 1998, 30, 23–31. [Google Scholar] [CrossRef]
- Ngan, E.S.; Schillinger, K.; DeMayo, F.; Tsai, S.Y. The mifepristone-inducible gene regulatory system in mouse models of disease and gene therapy. Semin. Cell Dev. Biol. 2002, 13, 143–149. [Google Scholar] [CrossRef]
- Emelyanov, A.; Parinov, S. Mifepristone-inducible LexPR system to drive and control gene expression in transgenic zebrafish. Dev. Biol. 2008, 320, 113–121. [Google Scholar] [CrossRef]
- Toniatti, C.; Bujard, H.; Cortese, R.; Ciliberto, G. Gene therapy progress and prospects: Transcription regulatory systems. Gene Ther. 2004, 11, 649–657. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ye, H.; Daoud-El Baba, M.; Peng, R.-W.; Fussenegger, M. A Synthetic Optogenetic Transcription Device Enhances Blood-Glucose Homeostasis in Mice. Science 2011, 332, 1565–1568. [Google Scholar] [CrossRef]
- Folcher, M.; Oesterle, S.; Zwicky, K.; Thekkottil, T.; Heymoz, J.; Hohmann, M.; Christen, M.; El-Baba, M.D.; Buchmann, P.; Fussenegger, M. Mind-controlled transgene expression by a wireless-powered optogenetic designer cell implant. Nat. Commun. 2014, 5, 5392. [Google Scholar] [CrossRef]
- Andersson, H.A.; Kim, Y.-S.; O’Neill, B.E.; Shi, Z.-Z.; Serda, R.E. HSP70 Promoter-Driven Activation of Gene Expression for Immunotherapy Using Gold Nanorods and Near Infrared Light. Vaccines 2014, 2, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Rome, C.; Couillaud, F.; Moonen, C.T. Spatial and temporal control of expression of therapeutic genes using heat shock protein promoters. Methods 2005, 35, 188–198. [Google Scholar] [CrossRef]
- Ortner, V.; Kaspar, C.; Halter, C.; Töllner, L.; Mykhaylyk, O.; Walzer, J.; Günzburg, W.H.; Dangerfield, J.A.; Hohenadl, C.; Czerny, T. Magnetic field-controlled gene expression in encapsulated cells. J. Control. Release 2012, 158, 424–432. [Google Scholar] [CrossRef]
- Krawczyk, K.; Xue, S.; Buchmann, P.; Charpin-El-Hamri, G.; Saxena, P.; Hussherr, M.-D.; Shao, J.; Ye, H.; Xie, M.; Fussenegger, M. Electrogenetic cellular insulin release for real-time glycemic control in type 1 diabetic mice. Science 2020, 368, 993–1001. [Google Scholar] [CrossRef]
- Bugaj, L.J.; Schaffer, D.V. Bringing next-generation therapeutics to the clinic through synthetic biology. Curr. Opin. Chem. Biol. 2012, 16, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Rubenstrunk, A.; Trollet, C.; Orsini, C.; Scherman, D. Positivein vivo heterologous gene regulation by electric pulses delivery with metallothionein I gene promoter. J. Gene Med. 2005, 7, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Stankevic, V.; Simonis, P.; Zurauskiene, N.; Stirke, A.; Dervinis, A.; Bleizgys, V.; Kersulis, S.; Balevicius, S. Compact Square-Wave Pulse Electroporator with Controlled Electroporation Efficiency and Cell Viability. Symmetry 2020, 12, 412. [Google Scholar] [CrossRef]
- Rossi, A.; Pakhomova, O.N.; Mollica, P.A.; Casciola, M.; Mangalanathan, U.; Pakhomov, A.G.; Muratori, C. Nanosecond Pulsed Electric Fields Induce Endoplasmic Reticulum Stress Accompanied by Immunogenic Cell Death in Murine Models of Lymphoma and Colorectal Cancer. Cancers 2019, 11, 2034. [Google Scholar] [CrossRef]
- Kotnik, T.; Frey, W.; Sack, M.; Meglič, S.H.; Peterka, M.; Miklavčič, D. Electroporation-based applications in biotechnology. Trends Biotechnol. 2015, 33, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Yarmush, M.L.; Golberg, A.; Serša, G.; Kotnik, T.; Miklavčič, D. Electroporation-Based Technologies for Medicine: Principles, Applications, and Challenges. Annu. Rev. Biomed. Eng. 2014, 16, 295–320. [Google Scholar] [CrossRef] [PubMed]
- Murakami, T.; Sunada, Y. Plasmid DNA gene therapy by electroporation: Principles and recent advances. Curr. Gene Ther. 2011, 11, 447–456. [Google Scholar] [CrossRef]
- Pichi, B.; Pellini, R.; De Virgilio, A.; Spriano, G. Electrochemotherapy: A well-accepted palliative treatment by patients with head and neck tumours. Acta Otorhinolaryngol. Ital. 2018, 38, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Kotnik, T.; Kramar, P.; Pucihar, G.; Miklavčič, D.; Tarek, M. Cell membrane electroporation- Part 1: The phenomenon. IEEE Electr. Insul. Mag. 2012, 28, 14–23. [Google Scholar] [CrossRef]
- Kašėta, V.; Kaušylė, A.; Kavaliauskaitė, J.; Petreikytė, M.; Stirkė, A.; Biziulevičienė, G. Detection of intracellular biomarkers in viable cells using millisecond pulsed electric fields. Exp. Cell Res. 2020, 389, 111877. [Google Scholar] [CrossRef]
- Shi, J.; Ma, Y.; Zhu, J.; Chen, Y.; Sun, Y.; Yao, Y.; Yang, Z.; Xie, J. A Review on Electroporation-Based Intracellular Delivery. Molecules 2018, 23, 3044. [Google Scholar] [CrossRef]
- Beebe, S.J.; Schoenbach, K.H. Nanosecond Pulsed Electric Fields: A New Stimulus to Activate Intracellular Signaling. J. Biomed. Biotechnol. 2005, 2005, 301289. [Google Scholar] [CrossRef]
- Roth, C.C.; Glickman, R.D.; Tolstykh, G.P.; Estlack, L.E.; Moen, E.K.; Echchgadda, I.; Beier, H.T.; Barnes, R.A.; Ibey, B.L. Evaluation of the Genetic Response of U937 and Jurkat Cells to 10-Nanosecond Electrical Pulses (nsEP). PLoS ONE 2016, 11, e0154555. [Google Scholar] [CrossRef]
- Beebe, S.J.; Sain, N.M.; Ren, W. Induction of Cell Death Mechanisms and Apoptosis by Nanosecond Pulsed Electric Fields (nsPEFs). Cells 2013, 2, 136–162. [Google Scholar] [CrossRef]
- Zhang, K.; Guo, J.; Ge, Z.; Zhang, J. Nanosecond Pulsed Electric Fields (nsPEFs) Regulate Phenotypes of Chondrocytes through Wnt/β-catenin Signaling Pathway. Sci. Rep. 2014, 4, 5836. [Google Scholar] [CrossRef] [PubMed]
- Morotomi-Yano, K.; Akiyama, H.; Yano, K.-I. Nanosecond pulsed electric fields activate MAPK pathways in human cells. Arch. Biochem. Biophys. 2011, 515, 99–106. [Google Scholar] [CrossRef]
- Nuccitelli, R.; Lui, K.; Kreis, M.; Athos, B.; Nuccitelli, P. Nanosecond pulsed electric field stimulation of reactive oxygen species in human pancreatic cancer cells is Ca2+-dependent. Biochem. Biophys. Res. Commun. 2013, 435, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yao, C.; Sun, C.; Guo, F.; Zhou, W.; Xiong, Z. Dependence on electric field intensities of cell biological effects induced by microsecond pulsed electric fields. IEEE Trans. Dielectr. Electr. Insul. 2011, 18, 2083–2088. [Google Scholar] [CrossRef]
- Kandušer, M.; Šentjurc, M.; Miklavčič, D. Cell membrane fluidity related to electroporation and resealing. Eur. Biophys. J. 2005, 35, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Wäster, P.; Rosdahl, I.; Öllinger, K. Cell fate regulated by nuclear factor-κB- and activator protein-1-dependent signalling in human melanocytes exposed to ultraviolet A and ultraviolet B. Br. J. Dermatol. 2014, 171, 1336–1346. [Google Scholar] [CrossRef]
- Wu, S.; Tan, M.; Hu, Y.; Wang, J.-L.; Scheuner, D.; Kaufman, R.J. Ultraviolet Light Activates NFκB through Translational Inhibition of IκBα Synthesis. J. Biol. Chem. 2004, 279, 34898–34902. [Google Scholar] [CrossRef]
- Lopez-Camarillo, C.; Ocampo, E.A.; Casamichana, M.L.; Perez-Plasencia, C.; Alvarez-Sanchez, E.; Marchat, L.A. Protein Kinases and Transcription Factors Activation in Response to UV-Radiation of Skin: Implications for Carcinogenesis. Int. J. Mol. Sci. 2012, 13, 142–172. [Google Scholar] [CrossRef] [PubMed]
- Bai, F.; Gusbeth, C.; Frey, W.; Nick, P. Nanosecond pulsed electric fields modulate the expression of the astaxanthin biosynthesis genes psy, crtR-b and bkt 1 in Haematococcus pluvialis. Sci. Rep. 2020, 10, 15508. [Google Scholar] [CrossRef]
- Li, K.; Ning, T.; Wang, H.; Jiang, Y.; Zhang, J.; Ge, Z. Nanosecond pulsed electric fields enhance mesenchymal stem cells differentiation via DNMT1-regulated OCT4/NANOG gene expression. Stem Cell Res. Ther. 2020, 11, 308. [Google Scholar] [CrossRef]
- Nesin, V.; Pakhomov, A.G. Inhibition of voltage-gated Na(+) current by nanosecond pulsed electric field (nsPEF) is not mediated by Na(+) influx or Ca(2+) signaling. Bioelectromagnetics 2012, 33, 443–451. [Google Scholar] [CrossRef]
- Roeder, R.G.; Rutter, W.J. Multiple Forms of DNA-dependent RNA Polymerase in Eukaryotic Organisms. Nature 1969, 224, 234–237. [Google Scholar] [CrossRef]
- Nuccitelli, R. Application of Pulsed Electric Fields to Cancer Therapy. Bioelectricity 2019, 1, 30–34. [Google Scholar] [CrossRef]
- Beebe, S. Cell responses without receptors and ligands, using nanosecond pulsed electric fields (nsPEFs). Int. J. Nanomed. 2013, 8, 3401–3404. [Google Scholar] [CrossRef]
- Zhang, J.; Blackmore, P.F.; Hargrave, B.Y.; Xiao, S.; Beebe, S.J.; Schoenbach, K.H. Nanosecond pulse electric field (nanopulse): A novel non-ligand agonist for platelet activation. Arch. Biochem. Biophys. 2008, 471, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Schoenbach, K.H.; Peterkin, F.E.; Alden, R.W., III; Beebe, S.J. The effect of pulsed electric fields on biological cells: Experiments and applications. IEEE Trans. Plasma Sci. 1997, 25, 284–292. [Google Scholar] [CrossRef]
- Tanori, M.; Casciati, A.; Zambotti, A.; Pinto, R.; Gianlorenzi, I.; Pannicelli, A.; Giardullo, P.; Benassi, B.; Marino, C.; Mancuso, M.; et al. Microsecond Pulsed Electric Fields: An Effective Way to Selectively Target and Radiosensitize Medulloblastoma Cancer Stem Cells. Int. J. Radiat. Oncol. 2021, 109, 1495–1507. [Google Scholar] [CrossRef] [PubMed]
- Dermol-Černe, J.; Napotnik, T.B.; Reberšek, M.; Miklavčič, D. Short microsecond pulses achieve homogeneous electroporation of elongated biological cells irrespective of their orientation in electric field. Sci. Rep. 2020, 10, 9149. [Google Scholar] [CrossRef]
- Lewis, N.; Liu, X.; Li, Y.; Nagarajan, H.; Yerganian, G.; O’Brien, E.; Bordbar, A.; Roth, A.M.; Rosenbloom, J.; Bian, C.; et al. Genomic landscapes of Chinese hamster ovary cell lines as revealed by the Cricetulus griseus draft genome. Nat. Biotechnol. 2013, 31, 759–765. [Google Scholar] [CrossRef]
- Webster, G.A.; Perkins, N.D. Transcriptional Cross Talk between NF-κB and p53. Mol. Cell. Biol. 1999, 19, 3485–3495. [Google Scholar] [CrossRef] [PubMed]
- Lechardeur, D.; Sohn, K.-J.; Haardt, M.; Joshi, P.B.; Monck, M.; Graham, R.W.; Beatty, B.; Squire, J.; O’Brodovich, H.; Lukacs, G. Metabolic instability of plasmid DNA in the cytosol: A potential barrier to gene transfer. Gene Ther. 1999, 6, 482–497. [Google Scholar] [CrossRef] [PubMed]
- Bisceglia, B.; Zirpoli, H.; Caputo, M.; Chiadini, F.; Scaglione, A.; Tecce, M.F. Induction of alkaline phosphatase activity by exposure of human cell lines to a low-frequency electric field from apparatuses used in clinical therapies. Bioelectromagnetics 2010, 32, 113–119. [Google Scholar] [CrossRef]
- Caputo, M.; Zirpoli, H.; De Rosa, M.C.; Rescigno, T.; Chiadini, F.; Scaglione, A.; Stellato, C.; Giurato, G.; Weisz, A.; Tecce, M.F.; et al. Effect of low frequency (LF) electric fields on gene expression of a bone human cell line. Electromagn. Biol. Med. 2013, 33, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.J.; Liu, Z.-G. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2011, 21, 103–115. [Google Scholar] [CrossRef]
- Gabriel, B.; Teissie, J. Generation of reactive-oxygen species induced by electropermeabilization of Chinese hamster ovary cells and their consequence on cell viability. JBIC J. Biol. Inorg. Chem. 1994, 223, 25–33. [Google Scholar] [CrossRef]
- Maccarrone, M.; Bladergroen, M.; Rosato, N.; Agrò, A.F.F. Role of Lipid Peroxidation in Electroporation-Induced Cell Permeability. Biochem. Biophys. Res. Commun. 1995, 209, 417–425. [Google Scholar] [CrossRef]
- Martins Antunes de Melo, W.D.; Celiešiūtė-Germanienė, R.; Šimonis, P.; Stirkė, A. Antimicrobial photodynamic therapy (aPDT) for biofilm treatments. Possible synergy between aPDT and pulsed electric fields. Virulence 2021, 12, 2247–2272. [Google Scholar] [CrossRef] [PubMed]
- Hanna, H.; Denzi, A.; Liberti, M.; Andrã©, F.M.; Mir, L.M. Electropermeabilization of Inner and Outer Cell Membranes with Microsecond Pulsed Electric Fields: Quantitative Study with Calcium Ions. Sci. Rep. 2017, 7, 13079. [Google Scholar] [CrossRef]
- Altamirano, F.; López, J.R.; Henríquez-Olguín, C.; Molinski, T.; Allen, P.D.; Jaimovich, E. Increased Resting Intracellular Calcium Modulates NF-κB-dependent Inducible Nitric-oxide Synthase Gene Expression in Dystrophic mdx Skeletal Myotubes. J. Biol. Chem. 2012, 287, 20876–20887. [Google Scholar] [CrossRef]
- Valdés, J.A.; Hidalgo, J.; Galaz, J.L.; Puentes, N.; Silva, M.; Jaimovich, E.; Carrasco, M.A. NF-κB activation by depolarization of skeletal muscle cells depends on ryanodine and IP3 receptor-mediated calcium signals. Am. J. Physiol. Physiol. 2007, 292, C1960–C1970. [Google Scholar] [CrossRef] [PubMed]
- Sée, V.; Rajala, N.K.; Spiller, D.; White, M.R. Calcium-dependent regulation of the cell cycle via a novel MAPK–NF-κB pathway in Swiss 3T3 cells. J. Cell Biol. 2004, 166, 661–672. [Google Scholar] [CrossRef] [PubMed]
- Graybill, P.M.; Davalos, R.V. Cytoskeletal Disruption after Electroporation and Its Significance to Pulsed Electric Field Therapies. Cancers 2020, 12, 1132. [Google Scholar] [CrossRef] [PubMed]
- Németh, Z.H.; Deitch, E.A.; Davidson, M.T.; Szabó, C.; Vizi, E.S.; Haskó, G. Disruption of the actin cytoskeleton results in nuclear factor-κB activation and inflammatory mediator production in cultured human intestinal epithelial cells. J. Cell. Physiol. 2004, 200, 71–81. [Google Scholar] [CrossRef]
- Berger, J.; Hauber, J.; Hauber, R.; Geiger, R.; Cullen, B.R. Secreted placental alkaline phosphatase: A powerful new quantitative indicator of gene expression in eukaryotic cells. Gene 1988, 66, 1–10. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kavaliauskaitė, J.; Kazlauskaitė, A.; Lazutka, J.R.; Mozolevskis, G.; Stirkė, A. Pulsed Electric Fields Alter Expression of NF-κB Promoter-Controlled Gene. Int. J. Mol. Sci. 2022, 23, 451. https://doi.org/10.3390/ijms23010451
Kavaliauskaitė J, Kazlauskaitė A, Lazutka JR, Mozolevskis G, Stirkė A. Pulsed Electric Fields Alter Expression of NF-κB Promoter-Controlled Gene. International Journal of Molecular Sciences. 2022; 23(1):451. https://doi.org/10.3390/ijms23010451
Chicago/Turabian StyleKavaliauskaitė, Justina, Auksė Kazlauskaitė, Juozas Rimantas Lazutka, Gatis Mozolevskis, and Arūnas Stirkė. 2022. "Pulsed Electric Fields Alter Expression of NF-κB Promoter-Controlled Gene" International Journal of Molecular Sciences 23, no. 1: 451. https://doi.org/10.3390/ijms23010451
APA StyleKavaliauskaitė, J., Kazlauskaitė, A., Lazutka, J. R., Mozolevskis, G., & Stirkė, A. (2022). Pulsed Electric Fields Alter Expression of NF-κB Promoter-Controlled Gene. International Journal of Molecular Sciences, 23(1), 451. https://doi.org/10.3390/ijms23010451








