1. Introduction
In the last decade, the use of cold atmospheric plasma (CAP), which is an ionized gas near room temperature, has become a novel method to treat cancer. Both direct application of CAP (e.g., by the clinically approved kINPenMED
® plasma jet) and indirect treatment by application of plasma-treated liquids (PTLs) have been shown to provide a significant anti-cancer effect [
1]. Van Boxem et al. [
2] showed that PTLs have an anti-cancer effect for a number of different CAP and liquid conditions, and Lin et al. [
3] found that CAP can induce immunogenic cancer cell death. This mode of cell death induced by CAP was later attributed to the CAP generated short-lived reactive species [
4]. Moreover, CAP and PTLs have been reported to cause a selective anti-cancer effect [
5], although selectivity depends on the cell type, the type of cancer, and the culturing medium [
6]. Bekeschus et al. [
7] demonstrated, using an
in ovo model, that CAP is a safe cancer treatment modality with respect to possible metastasis formation. A number of promising results of the clinical application of CAP for cancer treatment have also been published (see e.g., [
8,
9]).
It is widely believed that the processes leading to cancer cell death are initiated by reactive oxygen and/or nitrogen species (RONS), in particular hydrogen peroxide (), but the knowledge about the specific mechanisms underlying cell death induced by CAP and PTL is still very limited. The lack of understanding of the combined effect of RONS contained in CAP and PTL in terms of the cellular response to exposure is problematic in the development of CAP/PTL treatment as a standardized cancer therapy for clinical use. Ultimately, it should be possible to predict and quantify the susceptibility to CAP/PTL of a particular cell line in terms of features specifically associated with those cells.
So far, the vast majority of the literature in plasma oncology are experimental studies. As a complement, another approach to increase the understanding of complex biological systems such as the interaction between cells and PTL, is to develop a mathematical model that includes all the known information (of major importance in the given context) about the system and use it to investigate the system’s response to various conditions. Especially, the system’s response to a perturbation of the “normal” conditions can be analyzed. Furthermore, the development of the mathematical model itself can be seen as a way to summarize the current state of knowledge on the matter in a compact manner; it can be seen as the current “working hypothesis” of the mechanisms and processes governing the system dynamics.
Mathematical modeling has indeed proven to be a useful approach to increase our knowledge about the mechanisms of the cell’s antioxidant defense and redox signaling. Some examples are the range of diffusion of
in the cytosol [
10,
11] and the cellular decomposition of exogenous
[
12,
13,
14]. In the context of plasma oncology, two catalase-dependent apoptotic pathways associated with cancer cells, which possibly could be reactivated by CAP and thus explain the anti-cancer effect of CAP, have been investigated by mathematical modeling [
15]. It was found that these pathways are unlikely to account for the anti-cancer effect of CAP and thus the underlying cause has to be studied further.
In the present study, we develop a mathematical model that includes the species and mechanisms of major importance in the context of a cell system exposed to PTL. The ultimate aim is to find a measure in terms of key features and characteristics of cells, which is able to quantify a particular cell system’s susceptibility toward PTL and thus explain differences in response between normal cells and cancer cells. To the best of our knowledge, this is a completely novel approach in the field of plasma oncology, and we believe that our study will provide a new perspective and new insights as a complement to the experimental studies. An extensive summary of the background, leading to the more detailed research question, is provided in
Section 2.
2. Experimentally Observed Cytotoxic Effects of CAP and PTL and Possible Features Determining Cancer Cell Susceptibility
An immediate effect of CAP treatment of cancer cells is an increase in intracellular RONS [
16,
17,
18,
19,
20]. The significance of this RONS accumulation has been verified by the observation that the treatment does not succeed if the cancer cells have been pre-treated with intracellular RONS scavengers [
17,
21,
22]. The origin of the increase in intracellular RONS after CAP treatment is still under investigation, but a hypothesis consistent with experimental observations is that it is caused by a diffusion of extracellular CAP-originated RONS across the cell membrane [
17,
18,
23,
24].
It has been demonstrated that the anti-cancer effect of CAP can also be induced by the species in PTL. In PTL, which mainly consists of
,
and
[
25,
26,
27],
has been shown to be of major importance [
2,
25,
26,
27,
28,
29,
30,
31]. It has been demonstrated that the
consumption rate, which is cell specific, of cancer cells after PTL treatment, is a key factor determining the specific susceptibility of cancer cell lines to PTL. More explicitly, it has been reported that the higher the
consumption rate of cancer cells, the lower the susceptibility toward CAP/PTL [
32]. The susceptibility of cancer cells toward exogenous
has also been shown in [
33,
34,
35,
36]. However, it has been found that
alone cannot account for the total anti-cancer effect observed for PTL [
27]. In this context, there are some reports of a synergistic effect of
and
in PTL [
25,
26]. Thus, the cytotoxic effect of
seems to be enhanced in the presence of
. The study in [
26] found a
selective, synergistic anti-cancer effect for
in the μM-range and
in the mM-range, whereas in [
25], a
non-selective, synergistic anti-cancer effect was reported when
and
were both in the mM-range. Since
and
in PTL may react to form
[
37], which is known to be highly toxic to cells, it has been speculated whether
is the species causing the synergistic effect of
and
. Its formation could thus potentially increase the cytotoxicity of PTL compared to an equal concentration of
only.
To summarize, some key points of the observed cytotoxic effects of CAP or PTL are:
An intracellular increase of RONS, which is likely to be caused by diffusion of CAP-originated constituents through the cell membrane, is crucial for cell cytotoxicity.
The key species in the anti-cancer effect of PTL is
(note that this may be different for direct CAP treatment, where short-lived RONS also play a crucial role [
4,
38]), and the corresponding cytotoxicity is inversely proportional to the extracellular consumption rate of
.
The effect of extracellular is enhanced in the presence of , which can be a clue to understanding why PTL enables a more efficient treatment than a mock solution of only.
Thus, from the information presented in literature, we can conclude that the cellular response to an addition of extracellular
, with and without a simultaneous addition of
, is crucial to understanding the anti-cancer effect of PTL. In
Section 2.1, we introduce the key parameters to predict the response toward extracellular
of cells. We also relate this to general differences between normal cells and cancer cells. Based on this information and knowledge, we introduce our approach, and formulate our research question and aim in detail in
Section 2.2.
2.1. Differences in Cellular Response to Exogenous Hydrogen Peroxide
In particular, two factors determine whether a certain cell line is susceptible to the exposure of exogenous :
Several cancer cell lines have shown a common phenotype of
decreased catalase expression and
increased aquaporin expression (that facilitates the transport of
through the cell membrane [
39,
40,
41,
42] and thus determines the
membrane diffusion rate) compared to normal cells. Hence, cancer cells in general can be assumed to be more susceptible to exogenous
.
2.1.1. Membrane Diffusion Rate of Hydrogen Peroxide in Normal versus Cancer Cells
Aquaporins are proteins that form pores in the cell membrane. Primarily, they facilitate the transport of water between cells, but they also enable the trans-membrane diffusion of
(due to the chemical similarities between both molecules). Thus, the aquaporin expression in the cell membrane relates to the membrane diffusion rate of
. Many aquaporins have been found to be overexpressed in tumors of different origins, especially in aggressive tumors [
43]. Since different cancer cell lines express aquaporins to various extents [
43,
44], the different responses of
-exposure by different cancer cell lines can at least partly be explained by the non-identical levels of aquaporin expression. In [
45], it was found that aquaporin 3 accounted for nearly 80% of the membrane diffusion of
in a human pancreatic cancer cell line. For cells with a decreased aquaporin 3 expression, the rate of
-uptake from the extracellular compartment was significantly decreased. It has furthermore been shown that for glioblastoma tumor cells, the anti-cancer effect of PTL as well as the increase of the concentration of intracellular RONS was significantly inhibited when aquaporin 8 was inhibited [
46].
2.1.2. Catalase Activity in Normal versus Cancer Cells
Catalase is one of the main enzymes of the antioxidant defense system of cells of almost all aerobic organisms. The biological role of catalase is to regulate intracellular steady-state concentrations of
, and experimental investigations and kinetic models using in vitro data have demonstrated that catalase is the major enzyme involved in the antioxidant defense against high concentrations of
[
12,
47,
48,
49]. In particular, catalase has been shown to be responsible for the clearance of
exogenous in vitro and in vivo [
12,
50,
51,
52].
Although catalase levels vary widely across cell lines, the total concentration of catalase (extracellular and intracellular) is frequently reported to be lower in cancer cells than in normal cells [
36,
53,
54,
55,
56,
57,
58,
59,
60]. In [
61], it was found that the catalase activity in various cancer cells is up to an order of magnitude lower compared to normal cells, and in [
62], it was shown that normal cells had a better capacity to remove extracellular
than cancer cells; the rate constants for removal of extracellular
were on average two times higher in normal cells than in cancer cells. Furthermore, it was reported in [
62] that the rate constants for
removal by different cell lines correlated with the number of active catalase monomers per cell.
However, while in general, the levels of catalase are low in cancer cells, catalase activity appears to vary greatly across different cancer cell lines [
63]. In [
34], it was found that three cancer cell lines (glioblastoma) that were extremely susceptible to
(generated by ascorbic acid) had reduced activity of intracellular catalase. Ascorbic acid-resistant cancer cell lines, on the other hand, exhibited significantly higher levels of catalase, but catalase knockdown sensitized these cell lines to extracellular
.
An additional aspect of catalase that may be of interest in the context of cytotoxicity of CAP and PTL, is that it has been shown to decompose
[
64]. Thus, if the synergistic effect of
and
is to be found in the formation of
, catalase might have a double function (i.e., as a protective factor toward exogenous exposure of both
and
).
2.2. Approach and Research Question
In this study, we develop a mathematical model of the kinetics of the key species of PTL (i.e.,
and
) as well as of the processes governing the interaction with a cell system, which are given in terms of the
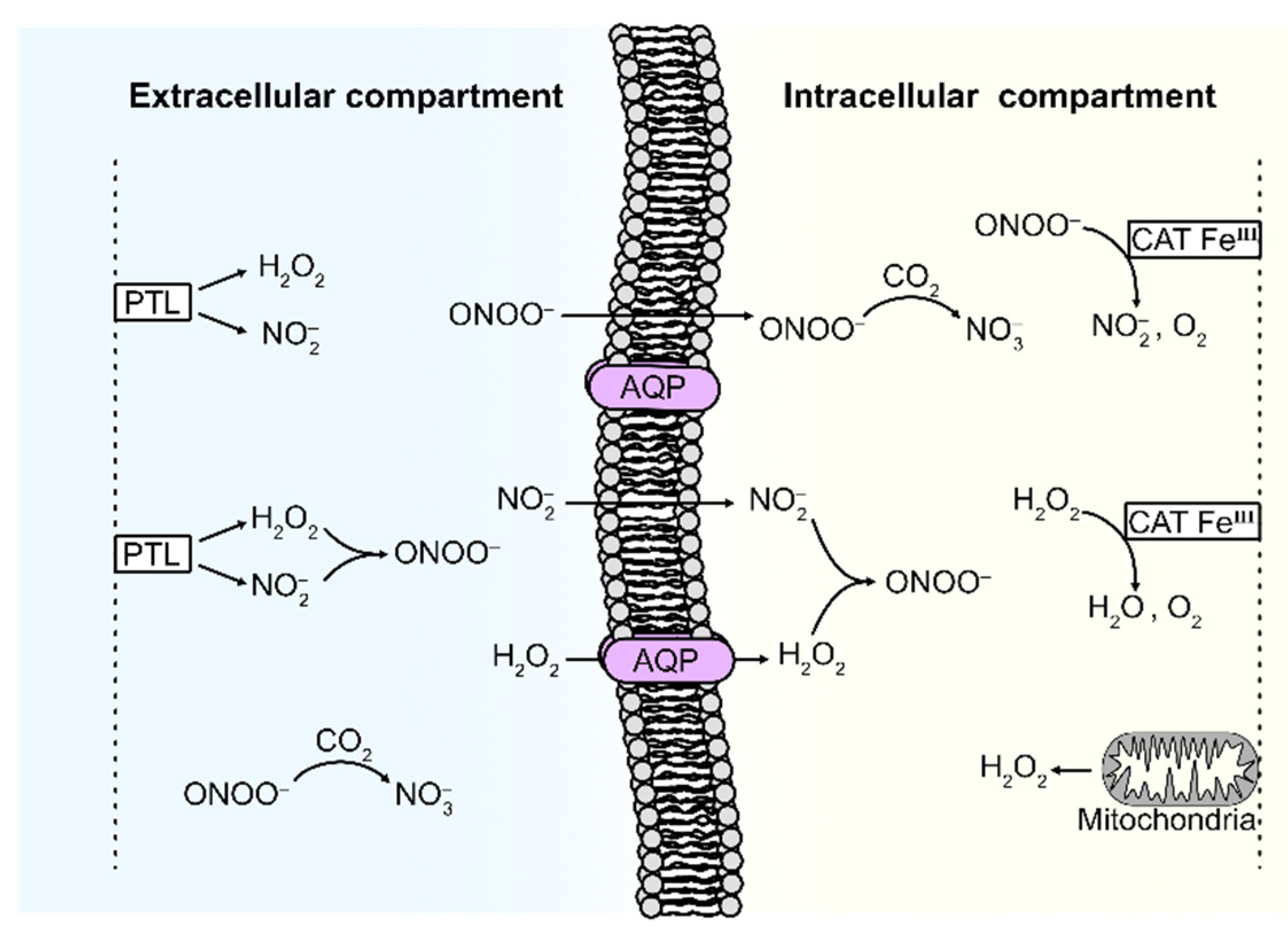
membrane diffusion rate constant and the intracellular catalase concentration. The system modeled is illustrated in
Figure 1.
As can be seen, the system consists of two compartments: the extracellular compartment (EC) and the intracellular compartment (IC). The two compartments are separated by the cell membrane, which some species in the system can diffuse through. Our mathematical model is explained in detail in
Section 5, with references to all input data and assumptions made. Briefly, it takes into account (i) the diffusion of
and
from the EC (where these species are supplied by PTL) to the IC, (ii) the formation of
from
and
(in both the IC and EC), (iii) the mitochondrial production of
(in the IC), and (iv) the decomposition of
and
(in the IC). Furthermore, (v) since the
-catalyzed consumption is considered to be the main route for
-decay in biological systems (due to a high
-concentration) [
65,
66,
67,
68,
69], this reaction is also included.
There have already been attempts to capture the susceptibility toward exogenous
of different cell lines in terms of their
membrane diffusion rate constant and intracellular catalase concentration [
13,
14]. Two dependent variables that have been investigated recently are the intracellular steady-state concentration of
and the so-called latency (which describes the reduced average reaction rates for the observed decomposition of
due to the localization of encapsulated catalase in the peroxisomes). In [
13], a lumped-parameter mathematical model, assuming that catalase is the major
-removal enzyme, was developed and used to calculate the intracellular steady-state
concentration for several cell lines. The model was calibrated to the experimental values of the measured critical parameters, and the resulting intracellular steady-state
concentration was related to observed cell specific susceptibility to extracellular exposure of
. The results showed that despite the fact that the experimental parameters including catalase concentration and
membrane diffusion rate constant, in particular, varied significantly across cell lines, the calculated steady-state intracellular-to-extracellular
ratio did not vary significantly across cell lines. In [
14], it was investigated whether variations in the latency of peroxisomal catalase across cancer cell lines correlates with observed in vitro susceptibility to ascorbate at equivalent dosing of extracellular
. The so-called effectiveness factor, which takes both the membrane diffusion rate and the overall reduced activity for encapsulated catalase into account, was used to quantify the effect of latency. The results suggest that latency alone is not a reliable parameter for predicting cell susceptibility to ascorbate (and hence,
).
In this study, we explore new dependent variables that could possibly explain the difference in cell susceptibility to an external addition of
, with and without a simultaneous addition of
, and ultimately, quantify the effect in terms of the
membrane diffusion rate constant (
) and the intracellular catalase concentration (
). Since we cannot distinguish a cancer cell from a normal cell solely by their
membrane diffusion rate constant and intracellular catalase concentration, we will have to work under the notations “cancer-like cells” (i.e., systems in the higher range of
membrane diffusion rate constant and the lower range of catalase concentration) and “normal-like cells” (i.e., systems in the lower range of
membrane diffusion rate constant and the higher range of catalase concentration). We investigate different regimes of the supplied extracellular
- and
concentrations according to experimental observations of the regimes of selective/non-selective and synergistic/non-synergistic anti-cancer effect of PTL [
25,
26]. The dependent variables that we investigate are:
The temporal maximum ofandin the IC. As opposed to the steady-state value of the intracellular concentration, the temporal maximum can be expected to be dependent on both and . These dependent variables may be related to the maximal intracellular oxidative power of the extracellularly added (and ), and thus it would be of interest to study whether a certain extracellularly added concentration of (and ) would result in a higher oxidative power in a more cancer-like cell than in a more normal-like cell.
The system response time (i.e., the time out of equilibrium) with respect toin the IC. In order to achieve tumor progression, it is essential for cancer cells to optimize their RONS concentration and maintain the RONS equilibrium. For our mathematical model, this is translated into the question: does a more cancer-like cell have a longer response time compared to a more normal-like cell?
The “load” of intracellularand(i.e., the time integral ofandin the IC). As the temporal maximum of and cannot capture any information about the total “load” of and (i.e., how much the intracellular and is increased over a period of time), it could be of interest to study such a dependent variable as a complement. The load can be seen as a measure that combines the temporal maximum concentration and the system response time. Another possible way to define the load of intracellular would be to only consider the concentration of over a “baseline”. Here, the steady-state intracellular , before the perturbation of an addition of extracellular and at the upper limit of , is used as the baseline.
The inverse of the average and maximal rate of extracellularconsumption. Since the cell susceptibility of CAP and PTL has been found to be inversely proportional to the (extracellular) consumption rate of it is of interest to explore a dependent variable quantifying the system susceptibility in terms of the consumption. We investigate two such candidates where one was defined in terms of the inverse of the average consumption rate, and the other one in terms of the inverse of the maximal consumption rate.
For all proposed dependent variables, we will analyze the dependence on and and whether a more cancer-like cell is associated with a higher “response” than a more normal-like cell. Our main research question is thus: Can the difference in cell susceptibility toward PTL be understood, and even quantified, by one of these dependent variables?
To the best of our knowledge, this is the first study of its kind, and our aim is to take some initial steps in the direction of an increased understanding of the mechanisms underlying the selective and synergistic anti-cancer effect of PTL, and ultimately, be able to predict the response of different cells.
3. Results
As introduced in
Section 2.2, in order to try to understand the combined role of the
membrane diffusion rate constant and the intracellular catalase concentration in determining the susceptibility of cells toward exogenous
, we have to go beyond the steady-state value of the intracellular
concentration [
13] (as well as latency [
14]) and examine dependent variables that take the system’s temporal response of a
perturbation in the EC into account. To be able to present the results in a more compact manner, the variables not yet introduced but of importance, and their denotations, are presented in
Table 1. Details about the independent and dependent variables can be found in
Section 5, where the mathematical model is presented. Likewise, details about the numerical calculations such as the values of the independent variables and parameters used in the model, can be found in
Section 6.
For the analysis and interpretation of the results, we mainly consider three important features of the dependent variable of interest:
Does it account for selectivity with respect to different regimes of ?
Does it account for a synergistic effect when is added to the system?
Does it represent a feasible measure to quantify the susceptibility to exogenous of a cell system in terms of and ?
To qualify as a “measure” (i.e., as a quantification of the susceptibility in terms of
and
), the dependent variable should be associated with a higher value for cells with a higher susceptibility and a lower value for cells with a lower susceptibility. Thus, in accordance with experimental observations, a feasible measure should result in a higher value for more cancer-like cells than for more normal-like cells, at least in the expected regime of selectivity (that is, for
in the μM-range [
26]). However, it should be noted that in this study, we do not follow strict mathematical criteria for a function to be categorized as a measure.
3.1. The Temporal Maximum of the Intracellular Hydrogen Peroxide Concentration: A Possible Measure of the Cell Susceptibility to Exogenous Hydrogen Peroxide
Our calculation results suggest that the temporal maximum of
(i.e.,
) is the dependent variable of major interest in terms of our requirements. Therefore, we focus our analysis on this variable. The results of the other dependent variables are presented in
Appendix A.
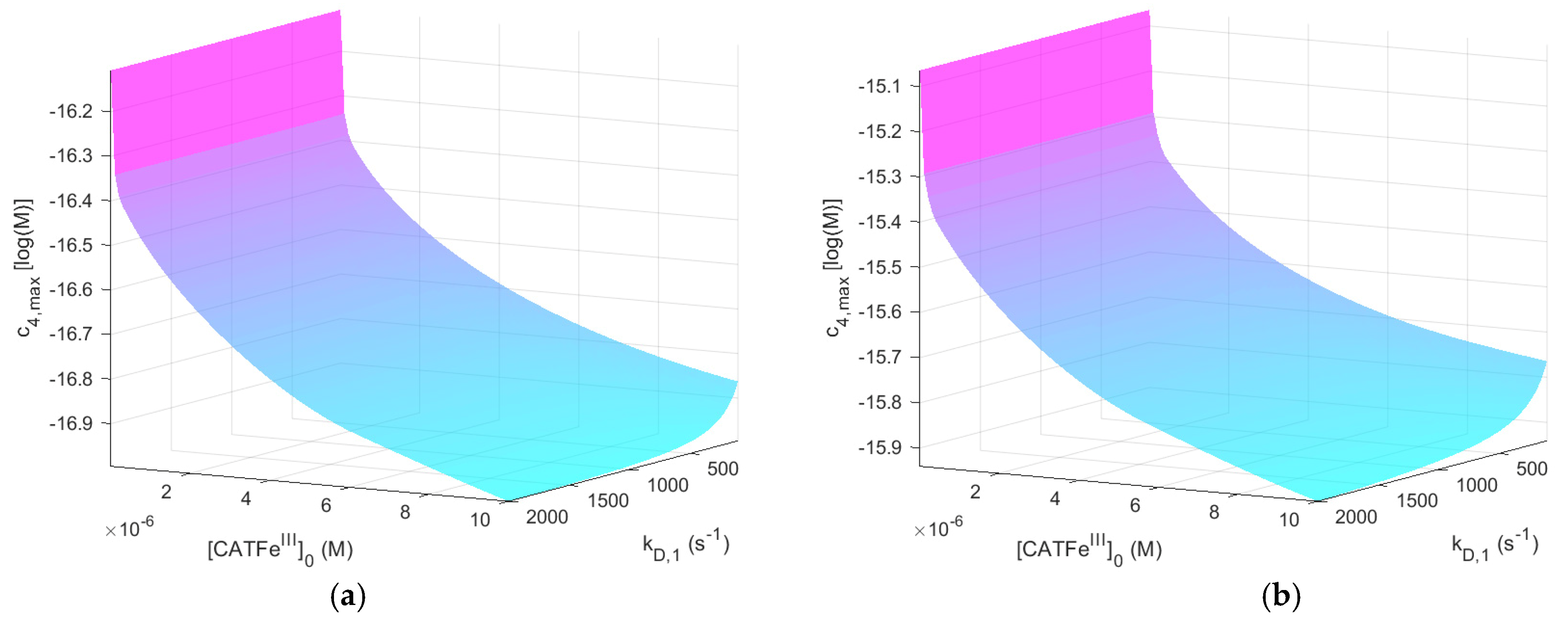
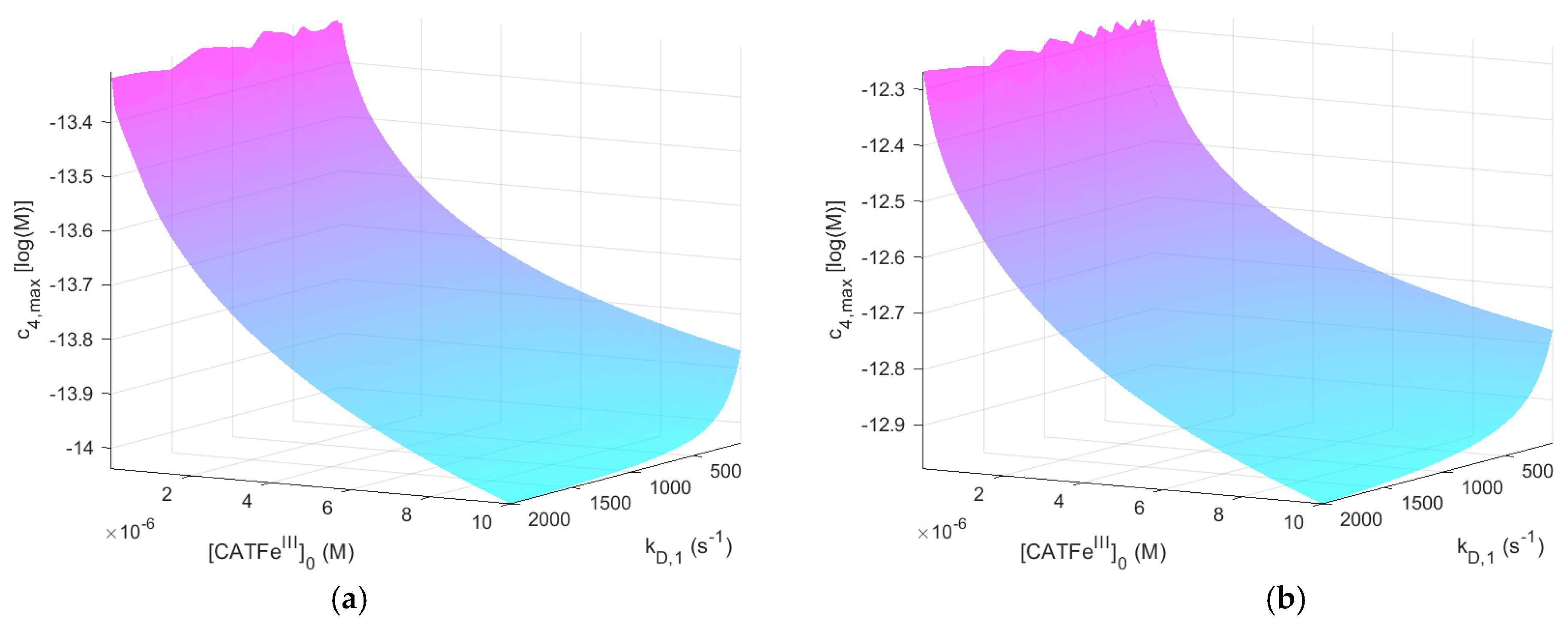
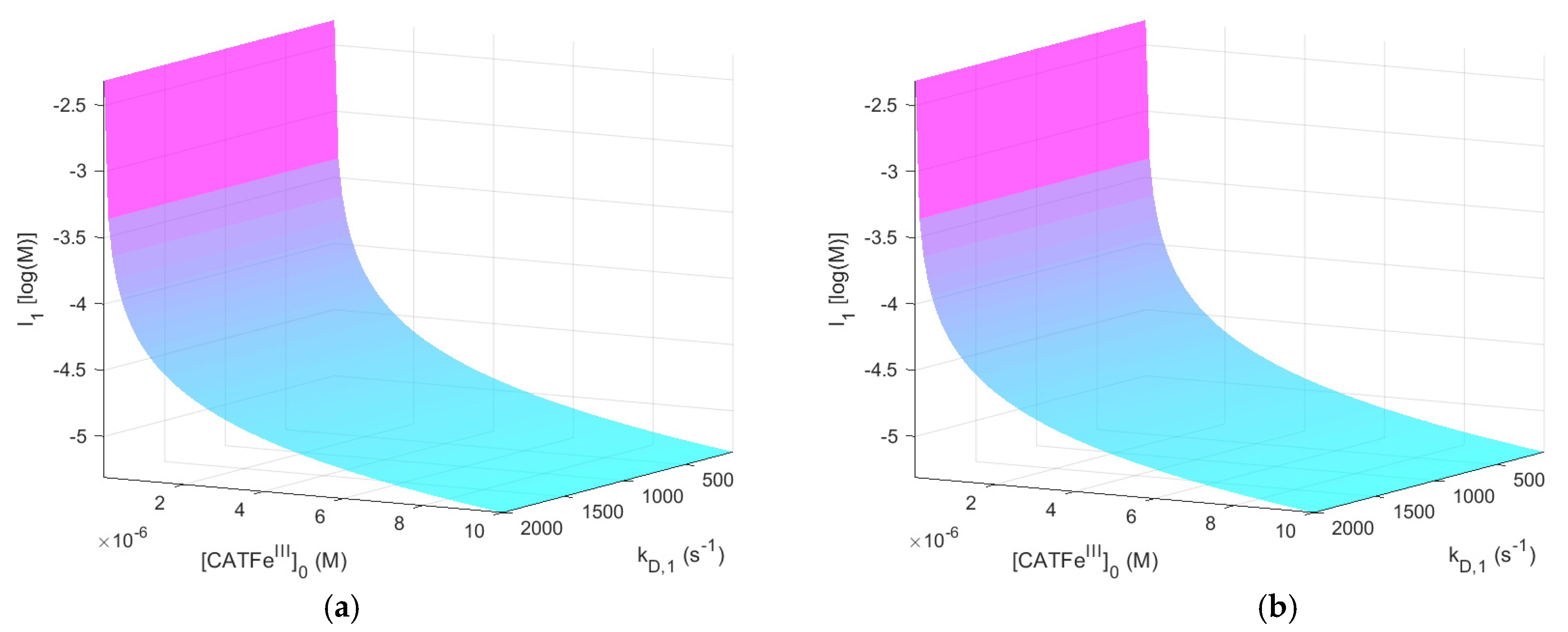
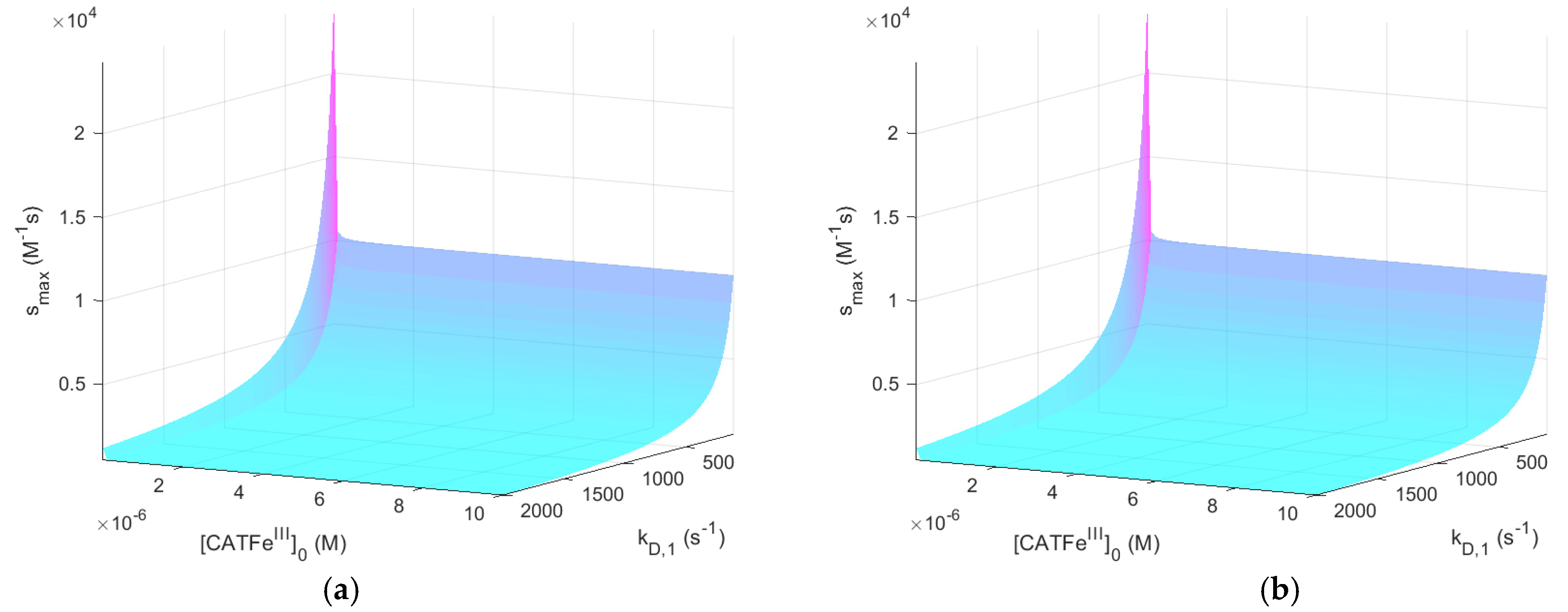
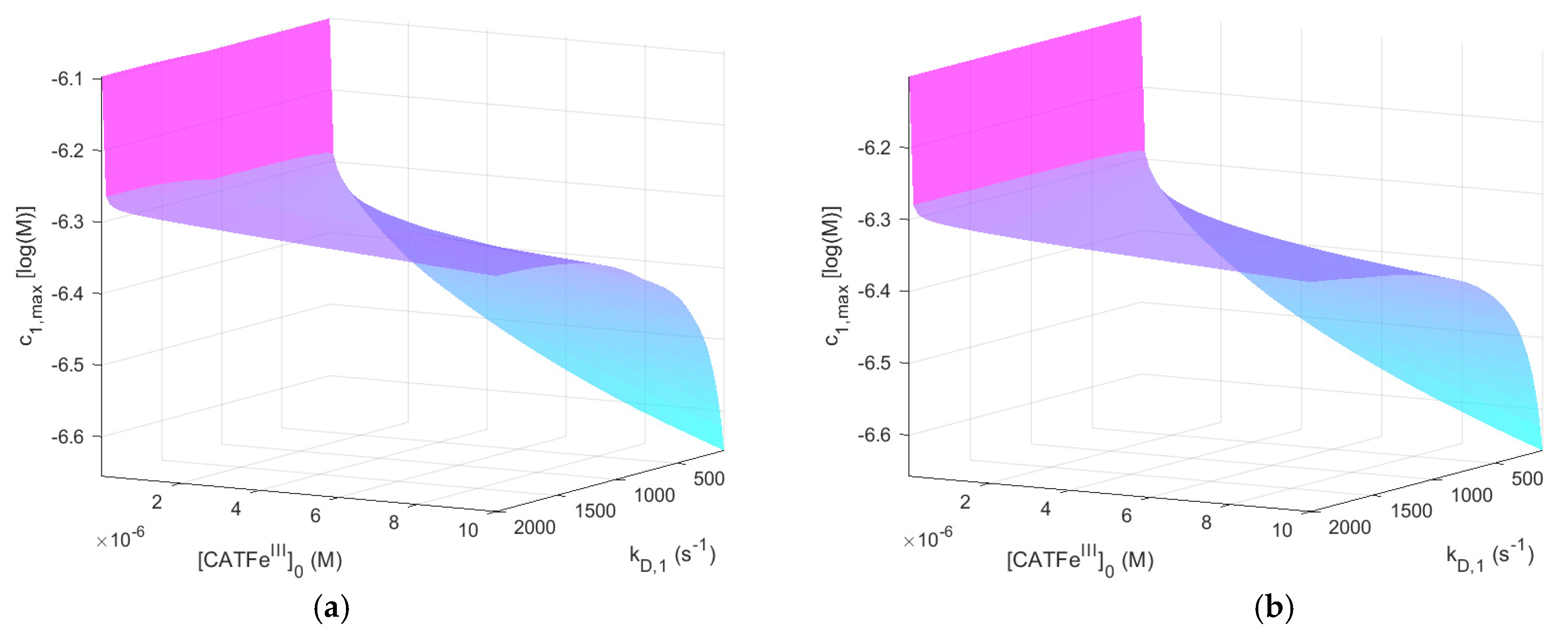
Figure 2 shows
as a function of
and
for
with and without
. The same results, but for
, are shown in
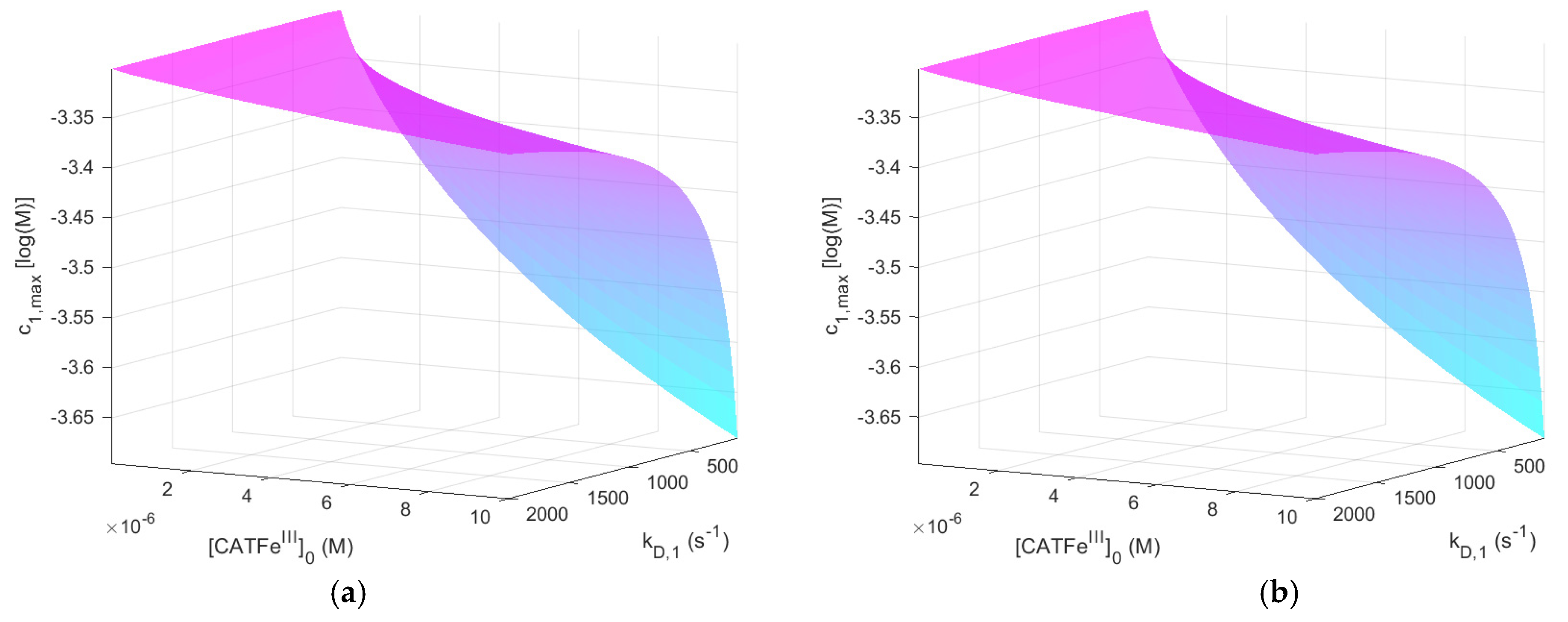
Figure 3.
When comparing the result for the different
-regimes for
(see
Figure 2a and
Figure 3a), we see that
is also in different concentration regimes, which is logical. Indeed, for
,
whereas for
,
. Thus, by assuming that there exists a threshold value
for which all types of cells undergo cell death, selectivity could be accounted for. However, there is no obvious synergetic effect; when comparing
Figure 2a,b,
is almost identical. Thus, the addition of
does not change
significantly. The same is true for
Figure 3a,b.
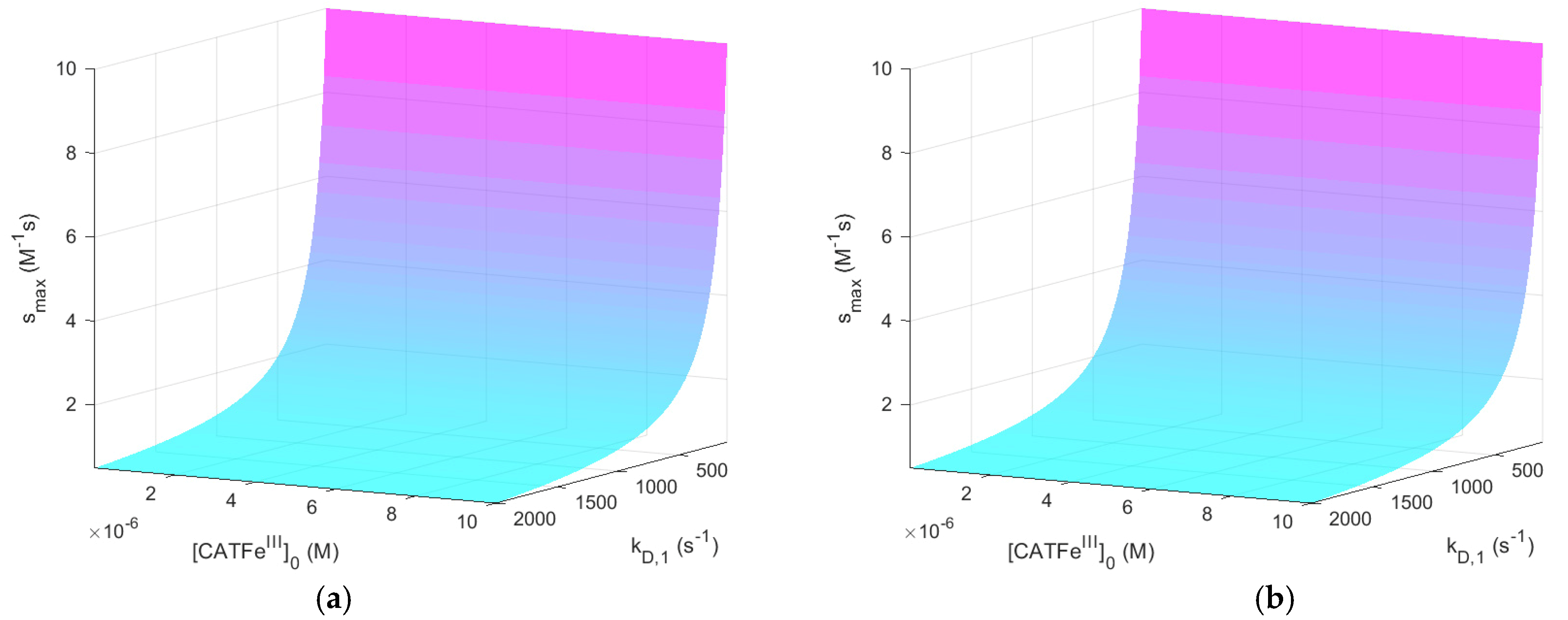
For
shows an increased
-dependence with increasing
. The lowest value of
is for the lowest values of
and highest values of
, as would be expected for a dependent variable that would qualify as a measure of the cell susceptibility in terms of
and
. In addition, the highest value of
is associated with the lowest value of
. However, in this regime, the dependence on
is insignificant. Here, in contrast, there exists a significant
-dependence and by changing the scale on the
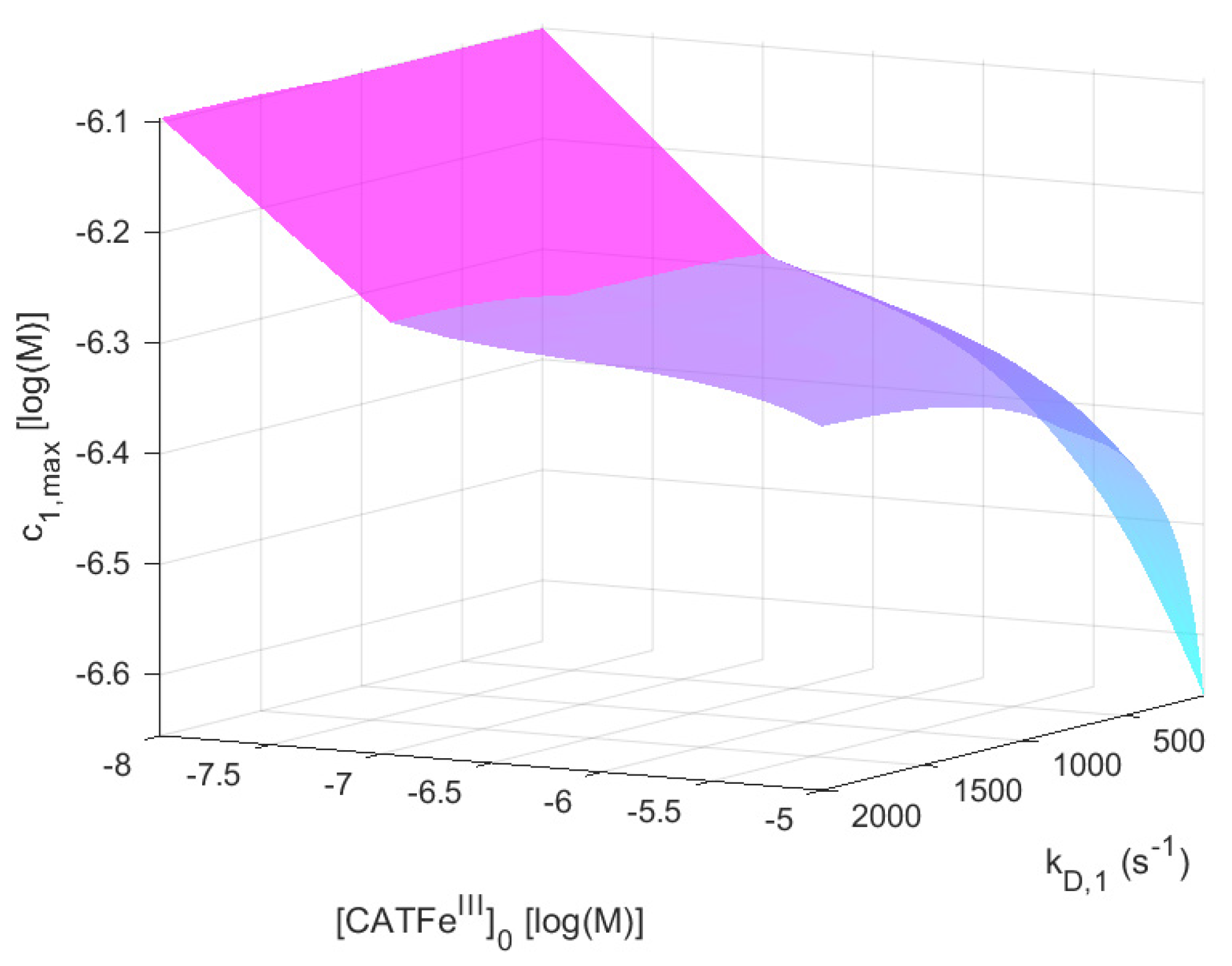
-axis to a log-scale (see
Figure 4), we see that there are two distinct regimes with a clear shift from one regime to another at about
. The regimes of
and
with the most profound difference between the value of
is between cells with
(for all
) and cells with the lowest possible
and highest possible
. Thus,
(i.e., the temporal maximum of
) is associated with a higher value for cancer-like cells than for normal-like cells. Indeed,
is about four times greater for the most susceptible cells compared to the most resistant cells.
In summary, does capture the dependence of and in a manner that is consistent with experimental observations and could thus represent a feasible measure to quantify the susceptibility of different cells in terms of their membrane diffusion rate constant and intracellular catalase concentration. However, in our model, it cannot yet account for the synergistic effect when is added.
To the best of our knowledge, there are not yet any experimental results to support our findings. We hope that our theoretical work will inspire future experimental studies. In the next section (
Section 3.2), we discuss possible opportunities to experimentally quantify
.
3.2. Physical Interpretation and the Use of the Temporal Maximum of the Intracellular Hydrogen Peroxide Concentration as an Experimental Probe
Our model, with all the equations, is explained in detail in
Section 5. Here we use the equations to better understand how we can use
as a measure to quantify the response of different cells. In order to analyze and write the equations in a more compact manner, we first introduce some short notations as well as some new notations (see
Table 2).
The temporal maximum of
(i.e.,
) occurs when the production and consumption of intracellular
are equal and
(as opposed to the steady-state value of
, which is governed by the same rate equation but for which
). If we exclude in Equation (10) (see
Section 5.2.2) the term representing the formation of
from
and
(since it is much smaller than the other terms), we have
Thus,
Here, we can furthermore use the constraint
since the total catalase concentration will be constant. By noting that
(see
Section 6.2.1), we can use the approximate expression
From our numerical calculations, we know that for low , is independent on whereas for high , is highly dependent on Furthermore (for ), for low , is highly dependent on , whereas for high , is independent on . The question is whether this behavior can be understood.
In the analysis, we first note that the implicit importance of
and
in determining the value of
at the time of
, and thus
, is hidden. The dependence on
originates from the fact that in our model,
is determined by
. Equation (3) (in
Section 5.2.1) can be approximated as:
Hence, the initial rate, or driving force, of
-consumption in the EC will crucially depend on
, and thus,
. In fact,
for
, whereas
for
(see Equation (18),
Section 6). It means that the initial driving force is about ten times higher in the latter case compared to the former. This could explain why
is seemingly independent on
at low values of
; if
for all values of
will be the dominant factor of the build up of
in the IC. For higher values of
, it seems like somewhen at
,
becomes increasingly dependent on
. It is thus reasonable to believe that the term
is becoming increasingly dominant and that the larger the value of
, the larger the value of
. For a fixed value of
,
will thus increase with increasing values of
.
In summary, this means that the lower the value of , the less important is the value of , and the other way around. Thus, the susceptibility (toward exogenous ) of cancer-like cells is not much influenced by the membrane diffusion rate constant and this is due to their much higher level of intracellular prior to the perturbation by the addition of exogenous . Normal-like cells, on the other hand, are more sensitive to the value of the membrane diffusion rate constant, since the difference in concentration between the intracellular and extracellular will be much larger.
Another aspect of
is whether it could provide an opportunity to extract information about different cell lines in terms of their
membrane diffusion rate constant and intracellular catalase concentration. By measuring
and the corresponding
for different
it could be possible to roughly quantify
and
. There are many experimental techniques for the detection and quantification of the
concentration in vitro and in vivo. The intracellular
concentration has been detected and measured by a chemoselective fluorescent naphthylimide peroxide probe [
70], by a genetically encoded red fluorescent sensor [
71], and by fluorescent reporter proteins [
72]. Thus, even if
does not represent a feasible measure of the cell susceptibility in terms of
and
, it could still possibly be used to gain more knowledge about the correlation between
and
and cell susceptibility toward exogenous
and PTLs.
4. Discussion
In this study, we use a theoretical approach to increase the knowledge about possible underlying causes of the anti-cancer effect of PTL. Although the model is fairly simple, it does include the major pathways for species production and consumption relevant for such a cell system. It also puts emphasis on two important features (i.e., the membrane diffusion rate constant and the intracellular catalase concentration), possibly explaining the different cell responses and cell susceptibility toward PTL when comparing normal cells to cancer cells, but also when comparing resistant vs. sensitive cancer cells. Nevertheless, it is important to keep in mind that in our model, different cells are only defined in terms of these two features, which are independent variables in our analysis, whereas in reality, there are countless of other features characteristic for different types of cells that could play an important role in the context of the anti-cancer effect of PTL. Here, we merely analyze the immediate cell response determined by the scavenging system active at high concentrations of . However, we do believe that our results contribute to a better understanding of some mechanisms probably underlying the anti-cancer effect of PTL. It brings novelty to the field of plasma oncology, and more broadly to the field of redox biology by using a theoretical approach and by proposing new ways to quantify the selective and synergistic anti-cancer effect of PTL in terms of inherent factors of cells. Here, we discuss each of our main findings and their potential implications. We also highlight what we believe are the most important limitations of the model.
As opposed to the steady-state intracellular concentration of
, which has been evaluated in previous studies [
13], our results suggest that the temporal maximal concentration of intracellular
could be a measure feasible to quantify the cell susceptibility toward exogenous
in terms of the
membrane diffusion rate constant and the intracellular catalase concentration. This result furthermore enables us to speculate whether the mode of action of
is as a signaling molecule rather than as a toxic substance causing necrosis. It is known that the intracellular concentration of a signaling molecule rises and falls within a short period. Indeed, whether a signaling molecule is effective or not is determined by how rapidly it is produced, how rapidly it is removed, and the concentration it must reach to alter the activity of its target effector. Of particular relevance in our context is that several reports have demonstrated that the rate of
generation and its concentration as a function of time play a key role in determining target cell damage or destruction [
73,
74,
75]. RONS are regulators of signaling pathways such as the extracellular signal-regulated kinase (ERK) mitogen activated protein kinase (MAPK) pathway, which is important for cell proliferation, and a number of studies have demonstrated the ability of exogenous oxidants to activate the ERK MAPK pathway [
76,
77,
78,
79,
80]. As in the general case, the duration and intensity of the ERK MAPK signal determine the outcome of the cellular response; there is a connection between the levels of ROS in a cell and the levels of MAPK signaling. In particular, MAPKs are activated in response to
[
81,
82,
83].
Based on our modeling results (presented in the
Appendix A; i.e.,
Figure A1 and
Figure A9), we do not think that the formation of
itself plays a major role in the explanation of the synergistic effect of
and
. This is because although the overall intracellular concentration of
is increased with about one order of magnitude when
is added to the system, the dependence on the
membrane diffusion rate constant is such that cells with a higher value of the
membrane diffusion rate constant (i.e., cancer-like cells) are associated with a lower maximal intracellular
concentration than more normal-like cells (i.e., cells with a lower value of the
membrane diffusion rate constant). In addition, the load of
is independent of the
membrane diffusion rate constant. However, an important aspect to keep in mind regarding our results for
and the choice to include
-catalyzed consumption of
in our model, is that
redirects much of the
produced in vivo toward radical mechanisms [
65]. Indeed, many of the reactions of
in vivo are more likely to be mediated by reactive intermediates derived from the reaction of
with
than by
itself [
84,
85]. Thus, if the production of such reactive intermediates were to be monitored instead of
, our results might be different. In this context, especially the formation of
should be considered; a fraction (about 30%) of the formed
will produce cage-escaped
and
radicals according to [
86,
87,
88]
where
and
[
89]. A possibly important target in the context of our study is catalase; catalase is so far the best known protein target for
and the rate constant of the reaction of bovine liver catalase with
is
at
[
90]. Since the temporal maximum of intracellular
(i.e.,
) is inversely dependent on the catalase concentration (i.e.,
) with an increasingly steeper incline for lower catalase concentrations in the regime
(see
Figure 2), cancer-like cells would be more vulnerable to a decrease in the catalase concentration than normal-like cells, which are associated with higher values of
. Thus, including these reaction pathways may possibly also make the dependent variable
able to account for the synergetic effect of
. Such an extension of our model was out of the scope for this study, but would be highly interesting in a future model development.
In experiments, the consumption rate of extracellular
has been found to inversely correlate with the susceptibility of cancer cells toward exogenous
[
32]. Thus, cancer cell lines with a high consumption rate were less susceptible. Our results cannot yet account for this correlation; when cells are defined in terms of their
membrane diffusion rate constant and their intracellular catalase concentration, susceptibility in terms of the inverse of the extracellular
consumption rate is not consistent with the experimental observations of cancer cells having a higher
membrane diffusion rate constant and a lower catalase concentration (see
Section 2.1). The fact that our model does not reproduce these patterns leaves an open question of how to construct a dependent variable in terms of the inverse of the extracellular
consumption rate so that it corresponds to the experimental correlation.
The fact that our mathematical model, as well as our criteria for a dependent variable to represent a feasible measure of the cell susceptibility, does not select the system response time as a good candidate does not necessarily indicate that this variable, in general, cannot capture cell susceptibility toward exogenous . Indeed, in our definition of this dependent variable, we assume a tolerance of a 10% increase of the intracellular steady-state concentration, and a different assumption of the tolerance might give a different result.
Except for the limitations of the model already mentioned in this discussion, some other model assumptions could hamper a realistic representation of a cell system in interaction with PTL. One such limitation is that in our model, the rate of mitochondrial
production is constant. Although it can be argued that this assumption is a valid starting point, in a model development, it could be important to modify this aspect to represent a cancer cell in a more realistic manner. Indeed, it has been shown that in some cancer cells, the mitochondrial respiration is decreased (in favor of aerobic glycolysis), and moreover, this shift seems to be a dynamic process (see [
91] and references therein). We believe that future models could benefit from trying to take such variation of the rate of mitochondrial
production into account, but this was out of the scope for this study.
Another aspect to take into account in a more realistic model is the fact that the
membrane diffusion rate constant is not a static but dynamic property. In [
92], it was shown that cellular stress conditions reversibly inhibit the diffusion of
(and
) of aquaporin 8. Thus, a more complex model taking the implicit time-dependence of the
membrane diffusion rate (caused by the increased intracellular
concentration after the addition of exogenous
) could potentially produce results different from our model.
A third aspect to be aware of is that in our model, we assume that the addition of PTL does not affect the membrane diffusion rate constants. However, a number of studies have reported an enhanced cell membrane permeability (and thus, increased membrane diffusion rate constants) after CAP/PTL treatment [
93,
94,
95]. For the aim and approach of our study, where the membrane diffusion rate constant of the key species
is varied within a range of possible values, we believe that our assumption is a valid starting point. Nevertheless, for future model extensions and developments, this aspect might be important to take into account.
Finally, it should be mentioned that the rate equations used to model the system are derived from information (collected from the literature) about rate constants and reaction orders for each reaction as they appear in the experiments. Most likely, the experimental conditions will deviate from the conditions of cells treated with PTL, which will affect the accuracy of the results produced by the model. However, for the purpose of our study, we believe that parameter values of the correct order of magnitude are sufficient at this stage.
7. Conclusions
With this study, we aim to gain insights about the possible mechanisms underlying the anti-cancer effect of plasma-treated liquids (PTLs). In particular, we are interested in whether cell susceptibility toward PTL can be quantified in terms of cell-specific features, how selectivity arises, and why combined with (as in PTL) offers a synergistic and thus enhanced anti-cancer effect compared with only. By developing a mathematical model describing the kinetics of the species in PTL-treated cells, we analyze four different dependent variables as a function of the membrane diffusion rate constant and the intracellular catalase concentration. Ultimately, one or more of these dependent variables could be used to quantify selective and synergistic effects of PTLs for different types of cells. In accordance with experimental observations, cancer cells are supposed to be associated with a higher membrane diffusion rate constant and a lower intracellular catalase concentration compared to normal cells, and we use this knowledge in the evaluation of our proposed dependent variables.
The model is built up ab initio based on the species, reactions, and processes of major importance in the context of cell susceptibility toward PTL, and parameter values such as rate constants are extracted from the literature. Thus, the model itself summarizes the current state of knowledge on the matter in a compact and descriptive manner. This type of mathematical modeling to gain insight into the underlying mechanisms of the anti-cancer effect of PTL is novel and this study is the first of its kind in the field of plasma oncology. Furthermore, we propose new ways to quantify the selective and synergistic anti-cancer effect of PTL in terms of inherent cell features, which is also an innovative approach in the ongoing research on the mode of action of PTL.
We find that the temporal maximal intracellular concentration shows a dependency of the membrane diffusion rate constant and the intracellular catalase concentration, so that it could possibly be used to quantify the anti-cancer effect of exogenous , but it does not account for the synergistic effect of and in PTL. However, by including the reactions where is produced in the catalyzed consumption of , and the interaction between and catalase, the dependent variable could possibly also be able to account for the synergetic effect of .
We believe that our model is an important step to unveil the underlying mechanisms of the anti-cancer effect of CAP and PTLs, but more efforts are needed in order to understand the full picture of the causes and action. Here, both positive and negative results are important to share, in order to increase our collective knowledge of which clues may lead us forward in our search, and which clues we can leave behind, at least for now. Theoretical and experimental approaches to investigate possible key features of cells and their interaction with CAP and PTLs play complementary roles in our aim to push the limit of knowledge further. We hope, and believe, that our study contributes to the quest to quantify selective and synergistic effects of plasma for cancer treatment.