Cholinergic Modulation of Neuroinflammation: Focus on α7 Nicotinic Receptor
Abstract
1. Introduction
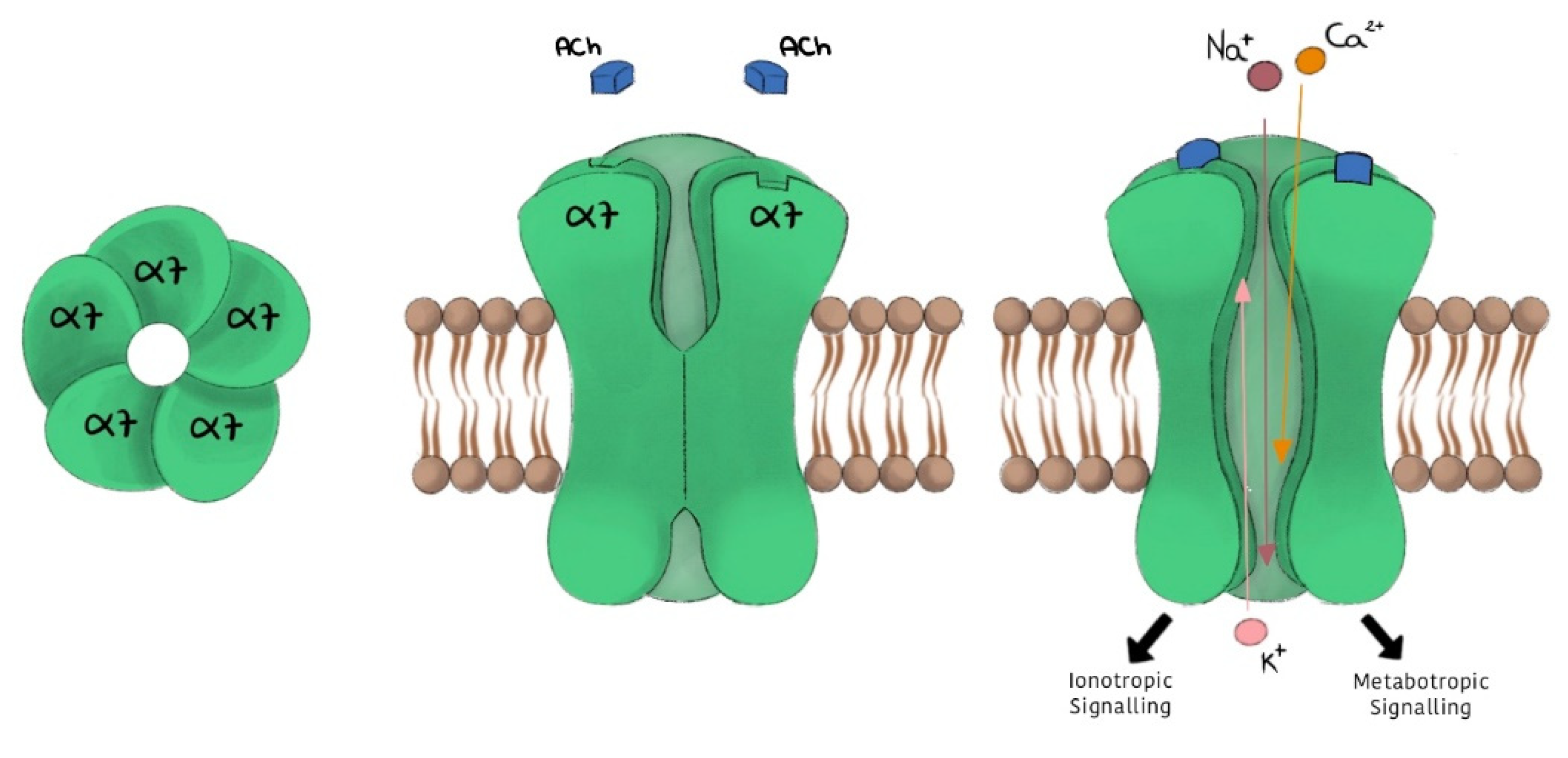
2. α7 nAChR Subtype
- -
- high permeability to calcium;
- -
- rapid activation and desensitization phase (in the order of milliseconds);
- -
- selective inhibition given by α-bungarotoxin (α-BTX) and methyllycaconitine (MLA), and a low affinity for nicotine.
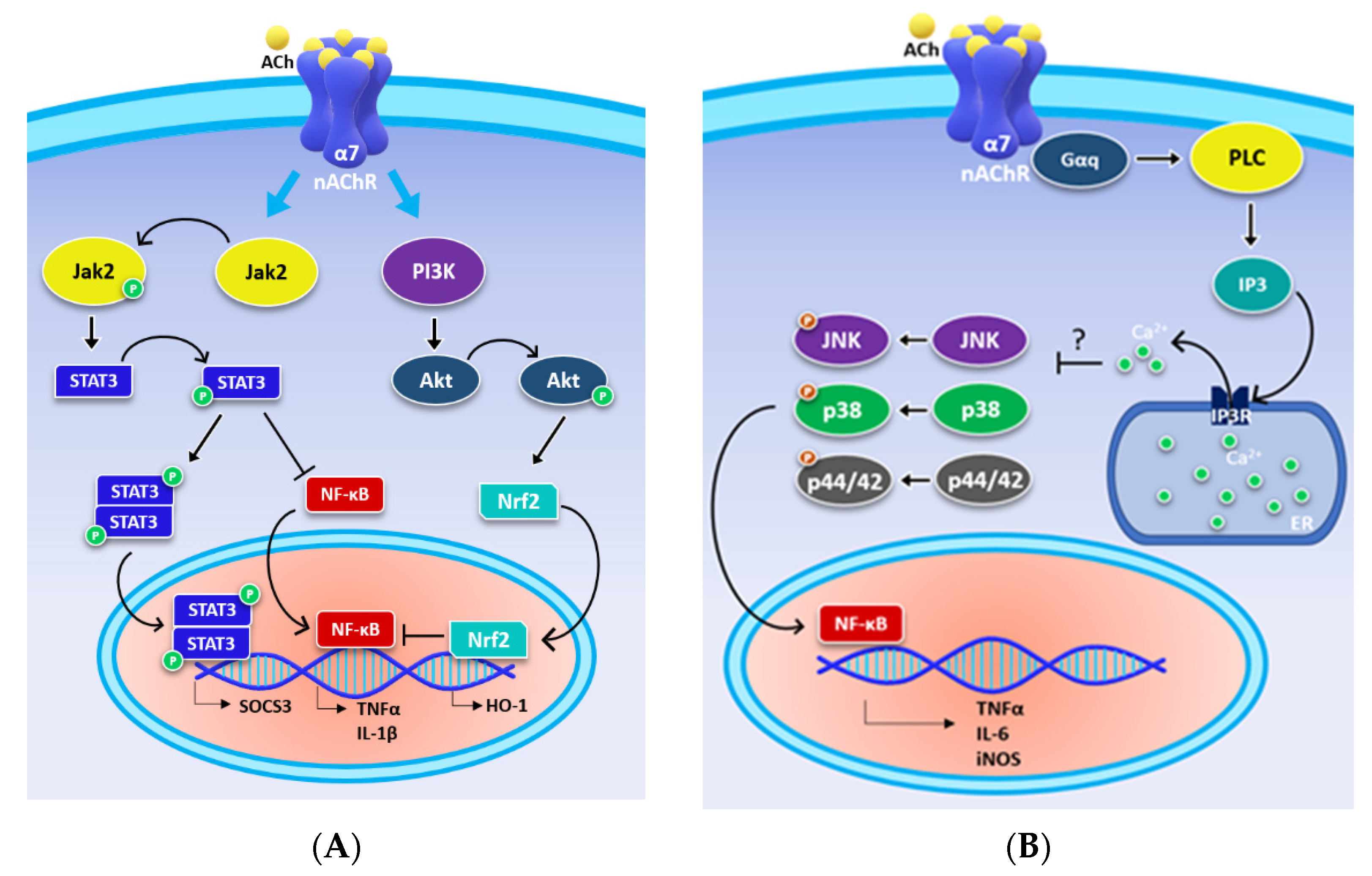
3. α7 Metabotropic Signalling Mediates the Cholinergic Anti-Inflammatory Pathway
4. α7 nAChR Effects in the Glial Cells
5. Role of α7 nAChR in the Control of Neuroinflammation in the CNS Pathologies
6. α7 nAChR Neuropharmacology
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Jonge, W.J.; Ulloa, L. The alpha7 nicotinic acetylcholine receptor as a pharmacological target for inflammation. Br. J. Pharmacol. 2007, 151, 915–929. [Google Scholar] [CrossRef]
- Wang, H.H.; Yu, M.; Ochani, M.; Amelia, C.A.; Tanovic, M.; Susarla, S.; Li, J.H.; Wang, H.H.; Yang, N.; Ulloa, L.; et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature 2003, 421, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Egea, J.; Buendia, I.; Parada, E.; Navarro, E.; León, R.; Lopez, M.G. Anti-inflammatory role of microglial alpha7 nAChRs and its role in neuroprotection. Biochem. Pharmacol. 2015, 97, 463–472. [Google Scholar] [CrossRef]
- Corradi, J.; Bouzat, C. Understanding the Bases of Function and Modulation of α 7 Nicotinic Receptors: Implications for Drug Discovery. Mol. Pharmacol. 2016, 90, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Jurado-Coronel, J.C.; Avila-Rodriguez, M.; Capani, F.; Gonzalez, J.; Echeverria Moran, V.; Barreto, G.E. Targeting the Nicotinic Acetylcholine Receptors (nAChRs) in Astrocytes as a Potential Therapeutic Target in Parkinson’s Disease. Curr. Pharm. Des. 2016, 22, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Mashimo, M.; Moriwaki, Y.; Misawa, H.; Ono, S.; Horiguchi, K.; Kawashima, K. Expression and Function of the Cholinergic System in Immune Cells. Front. Immunol. 2017, 8, 1085. [Google Scholar] [CrossRef] [PubMed]
- Bouzat, C.; Gumilar, F.; Spitzmaul, G.; Wang, H.L.; Rayes, D.; Hansen, S.B.; Taylor, P.; Sine, S.M. Coupling of agonist binding to channel gating in an ACh-binding protein linked to an ion channel. Nature 2004, 430, 896–900. [Google Scholar] [CrossRef]
- Lee, W.Y.; Sine, S.M. Principal pathway coupling agonist binding to channel gating in nicotinic receptors. Nature 2005, 438, 243–247. [Google Scholar] [CrossRef]
- Bartos, M.; Corradi, J.; Bouzat, C. Structural Basis of Activation of Cys-Loop Receptors: The Extracellular–Transmembrane Interface as a Coupling Region. Mol. Neurobiol. 2009, 40, 236–252. [Google Scholar] [CrossRef]
- Kume, T.; Takada-Takatori, Y. Nicotinic Acetylcholine Rreceptor Signaling: Roles in Neuroprotection; Springer: Singapore, 2018; ISBN 9789811084881. [Google Scholar]
- Rangwala, F.; Drisdel, R.C.; Rakhilin, S.; Ko, E.; Atluri, P.; Harkins, A.B.; Fox, A.P.; Salman, S.B.; Green, W.N. Neuronal α-Bungarotoxin Receptors Differ Structurally from Other Nicotinic Acetylcholine Receptors. J. Neurosci. 1997, 17, 8201–8212. [Google Scholar] [CrossRef]
- Drisdel, R.C.; Green, W.N. Neuronal α-Bungarotoxin Receptors Are α7 Subunit Homomers. J. Neurosci. 2000, 20, 133–139. [Google Scholar] [CrossRef]
- King, J.R.; Nordman, J.C.; Bridges, S.P.; Lin, M.-K.; Kabbani, N. Identification and Characterization of a G Protein-binding Cluster in α7 Nicotinic Acetylcholine Receptors. J. Biol. Chem. 2015, 290, 20060–20070. [Google Scholar] [CrossRef] [PubMed]
- King, J.R.; Kabbani, N. Alpha 7 nicotinic receptor coupling to heterotrimeric G proteins modulates RhoA activation, cytoskeletal motility, and structural growth. J. Neurochem. 2016, 138, 532–545. [Google Scholar] [CrossRef]
- King, J.R.; Ullah, A.; Bak, E.; Jafri, M.S.; Kabbani, N. Ionotropic and Metabotropic Mechanisms of Allosteric Modulation of α 7 Nicotinic Receptor Intracellular Calcium. Mol. Pharmacol. 2018, 93, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Yakel, J.L. Nicotinic acetylcholine receptor-mediated calcium signaling in the nervous system. Acta Pharmacol. Sin. 2009, 30, 673–680. [Google Scholar] [CrossRef]
- Chang, K.T.; Berg, D.K. Voltage-Gated Channels Block Nicotinic Regulation of CREB Phosphorylation and Gene Expression in Neurons. Neuron 2001, 32, 855–865. [Google Scholar] [CrossRef]
- de Jonge, W.J.; van der Zanden, E.P.; The, F.O.; Bijlsma, M.F.; van Westerloo, D.J.; Bennink, R.J.; Berthoud, H.-R.; Uematsu, S.; Akira, S.; van den Wijngaard, R.M.; et al. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway. Nat. Immunol. 2005, 6, 844–851. [Google Scholar] [CrossRef]
- Bencherif, M.; Lippiello, P.M.; Lucas, R.; Marrero, M.B. Alpha7 nicotinic receptors as novel therapeutic targets for inflammation-based diseases. Cell. Mol. Life Sci. 2011, 68, 931–949. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.; Vargas, J.; Hoffmann, A. Signaling via the NFκB system. Wiley Interdiscip. Rev. Syst. Biol. Med. 2016, 8, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Parada, E.; Egea, J.; Buendia, I.; Negredo, P.; Cunha, A.C.; Cardoso, S.; Soares, M.P.; López, M.G. The Microglial α7-Acetylcholine Nicotinic Receptor Is a Key Element in Promoting Neuroprotection by Inducing Heme Oxygenase-1 via Nuclear Factor Erythroid-2-Related Factor 2. Antioxid. Redox Signal. 2013, 19, 1135–1148. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Role of Nrf2 in Oxidative Stress and Toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Shytle, R.D.; Mori, T.; Townsend, K.; Vendrame, M.; Sun, N.; Zeng, J.; Ehrhart, J.; Silver, A.A.; Sanberg, P.R.; Tan, J. Cholinergic modulation of microglial activation by α7 nicotinic receptors. J. Neurochem. 2004, 89, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.; McIntire, J.; Ryan, S.; Dunah, A.; Loring, R. Anti-inflammatory effects of astroglial α7 nicotinic acetylcholine receptors are mediated by inhibition of the NF-κB pathway and activation of the Nrf2 pathway. J. Neuroinflamm. 2017, 14, 192. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Hide, I.; Matsubara, A.; Hama, C.; Harada, K.; Miyano, K.; Andrä, M.; Matsubayashi, H.; Sakai, N.; Kohsaka, S.; et al. Microglial α7 nicotinic acetylcholine receptors drive a phospholipase C/IP3 pathway and modulate the cell activation toward a neuroprotective role. J. Neurosci. Res. 2006, 83, 1461–1470. [Google Scholar] [CrossRef]
- Koistinaho, M.; Koistinaho, J. Role of p38 and p44/42 mitogen-activated protein kinases in microglia. Glia 2002, 40, 175–183. [Google Scholar] [CrossRef]
- Youssef, M.; Ibrahim, A.; Akashi, K.; Hossain, M.S. PUFA-Plasmalogens Attenuate the LPS-Induced Nitric Oxide Production by Inhibiting the NF-kB, p38 MAPK and JNK Pathways in Microglial Cells. Neuroscience 2019, 397, 18–30. [Google Scholar] [CrossRef]
- Lee, Y.B.; Schrader, J.W.; Kim, S.U. p38 MAP Kinase regulates TNF-α production in Human astrocytes and microglia by multiple mechanisms. Cytokine 2000, 12, 874–880. [Google Scholar] [CrossRef]
- You, P.; Fu, S.; Yu, K.; Xia, Y.; Wu, H.; Yang, Y.; Ma, C.; Liu, D.; Chen, X.; Wang, J.; et al. Scutellarin suppresses neuroinflammation via the inhibition of the AKT/NF-κB and p38/JNK pathway in LPS-induced BV-2 microglial cells. Naunyn. Schmiedebergs. Arch. Pharmacol. 2018, 391, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Molas, S.; Dierssen, M. The role of nicotinic receptors in shaping and functioning of the glutamatergic system: A window into cognitive pathology. Neurosci. Biobehav. Rev. 2014, 46, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, E.X.; Pereira, E.F.; Castro, N.G.; Alkondon, M.; Reinhardt, S.; Schröder, H.; Maelicke, A. Nicotinic receptor function in the mammalian central nervous system. Ann. N. Y. Acad. Sci. 1995, 757, 48–72. [Google Scholar] [CrossRef]
- Takada, Y.; Yonezawa, A.; Kume, T.; Katsuki, H.; Kaneko, S.; Sugimoto, H.; Akaike, A. Nicotinic acetylcholine receptor-mediated neuroprotection by donepezil against glutamate neurotoxicity in rat cortical neurons. J. Pharmacol. Exp. Ther. 2003, 306, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Akaike, A.; Tamura, Y.; Yokota, T.; Shimohama, S.; Kimura, J. Nicotine-induced protection of cultured cortical neurons againstN-methyl-D-aspartate receptor-mediated glutamate cytotoxicity. Brain Res. 1994, 644, 181–187. [Google Scholar] [CrossRef]
- Liu, Q.; Zhao, B. Nicotine attenuates β -amyloid peptide-induced neurotoxicity, free radical and calcium accumulation in hippocampal neuronal cultures. Br. J. Pharmacol. 2004, 141, 746–754. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, M.; Katsuki, H.; Takagi, M.; Kume, T.; Akaike, A. Long-term treatment with nicotine suppresses neurotoxicity of, and microglial activation by, thrombin in cortico-striatal slice cultures. Eur. J. Pharmacol. 2009, 602, 288–293. [Google Scholar] [CrossRef]
- Takeuchi, H.; Yanagida, T.; Inden, M.; Takata, K.; Kitamura, Y.; Yamakawa, K.; Sawada, H.; Izumi, Y.; Yamamoto, N.; Kihara, T.; et al. Nicotinic receptor stimulation protects nigral dopaminergic neurons in rotenone-induced Parkinson’s disease models. J. Neurosci. Res. 2009, 87, 576–585. [Google Scholar] [CrossRef]
- Toborek, M.; Son, K.W.; Pudelko, A.; King-Pospisil, K.; Wylegala, E.; Malecki, A. ERK 1/2 signaling pathway is involved in nicotine-mediated neuroprotection in spinal cord neurons. J. Cell. Biochem. 2007, 100, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Vijayaraghavan, S. Nicotinic cholinergic signaling in hippocampal astrocytes involves calcium-induced calcium release from intracellular stores. Proc. Natl. Acad. Sci. USA 2001, 98, 4148–4153. [Google Scholar] [CrossRef]
- Kalkman, H.O.; Feuerbach, D. Modulatory effects of α7 nAChRs on the immune system and its relevance for CNS disorders. Cell. Mol. Life Sci. 2016, 73, 2511–2530. [Google Scholar] [CrossRef]
- Han, B.; Li, X.; Hao, J. The cholinergic anti-inflammatory pathway: An innovative treatment strategy for neurological diseases. Neurosci. Biobehav. Rev. 2017, 77, 358–368. [Google Scholar] [CrossRef]
- Zhang, J.; Rivest, S. Anti-inflammatory effects of prostaglandin E2 in the central nervous system in response to brain injury and circulating lipopolysaccharide. J. Neurochem. 2001, 76, 855–864. [Google Scholar] [CrossRef]
- Revathikumar, P.; Bergqvist, F.; Gopalakrishnan, S.; Korotkova, M.; Jakobsson, P.-J.; Lampa, J.; Le Maître, E. Immunomodulatory effects of nicotine on interleukin 1β activated human astrocytes and the role of cyclooxygenase 2 in the underlying mechanism. J. Neuroinflamm. 2016, 13, 256. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, X.; Hui, Y.; Zhu, C.; Wu, J.; Taylor, D.H.; Ji, J.; Fan, W.; Huang, Z.; Hu, J. Activation of α7 nicotinic acetylcholine receptors protects astrocytes against oxidative stress-induced apoptosis: Implications for Parkinson’s disease. Neuropharmacology 2015, 91, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhu, N.; Wang, K.; Zhang, Z.; Wang, Y. Identification of α7 nicotinic acetylcholine receptor on hippocampal astrocytes cultured in vitro and its role on inflammatory mediator secretion. Neural Regen. Res. 2012, 7, 1709–1714. [Google Scholar] [CrossRef] [PubMed]
- Gamage, R.; Wagnon, I.; Rossetti, I.; Childs, R.; Niedermayer, G.; Chesworth, R.; Gyengesi, E. Cholinergic Modulation of Glial Function During Aging and Chronic Neuroinflammation. Front. Cell. Neurosci. 2020, 14, 318. [Google Scholar] [CrossRef] [PubMed]
- Kuzumaki, N.; Ikegami, D.; Imai, S.; Narita, M.; Tamura, R.; Yajima, M.; Suzuki, A.; Miyashita, K.; Niikura, K.; Takeshima, H.; et al. Enhanced IL-1β Production in response to the activation of hippocampal glial cells impairs neurogenesis in aged mice. Synapse 2010, 64, 721–728. [Google Scholar] [CrossRef]
- Báez-Pagán, C.A.; Delgado-Vélez, M.; Lasalde-Dominicci, J.A. Activation of the Macrophage α7 Nicotinic Acetylcholine Receptor and Control of Inflammation. J. Neuroimmune Pharmacol. 2015, 10, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Gatta, V.; Mengod, G.; Reale, M.; Tata, A.M. Possible Correlation between Cholinergic System Alterations and Neuro/Inflammation in Multiple Sclerosis. Biomedicines 2020, 8, 153. [Google Scholar] [CrossRef]
- Dumas, J.A.; Newhouse, P.A. The cholinergic hypothesis of cognitive aging revisited again: Cholinergic functional compensation. Pharmacol. Biochem. Behav. 2011, 99, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Caruso, A.; Nicoletti, F.; Mango, D.; Saidi, A.; Orlando, R.; Scaccianoce, S. Stress as risk factor for Alzheimer’s disease. Pharmacol. Res. 2018, 132, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.L.; Rodriguez-Ortiz, C.J.; Kitazawa, M. Infection, systemic inflammation, and Alzheimer’s disease. Microbes Infect. 2015, 17, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Tata, A.M.; Velluto, L.; Reale, C.D. and M. Cholinergic System Dysfunction and Neurodegenerative Diseases: Cause or Effect? CNS Neurol. Disord. Drug Targets 2014, 13, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Confaloni, A.; Tosto, G.; Tata, A.M. Promising Therapies for Alzheimer’s Disease. Curr. Pharm. Des. 2016, 22, 2050–2056. [Google Scholar] [CrossRef]
- De Jaco, A.; Bernardini, L.; Rosati, J.; Tata, A.M. Alpha-7 Nicotinic Receptors in Nervous System Disorders: From Function to Therapeutic Perspectives. Cent. Nerv. Syst. Agents Med. Chem. 2017, 17, 1–9. [Google Scholar] [CrossRef]
- Bencherif, M.; Lippiello, P.M. Alpha7 neuronal nicotinic receptors: The missing link to understanding Alzheimer’s etiopathology? Med. Hypotheses 2010, 74, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Dineley, K.T. Beta-amyloid peptide—nicotinic acetylcholine receptor interaction: The two faces of health and disease. Front. Biosci. 2007, 12, 5030–5080. [Google Scholar] [CrossRef] [PubMed]
- Barykin, E.P.; Garifulina, A.I.; Kruykova, E.V.; Spirova, E.N.; Anashkina, A.A.; Adzhubei, A.A.; Shelukhina, I.V.; Kasheverov, I.E.; Mitkevich, V.A.; Kozin, S.A.; et al. Isomerization of Asp7 in Beta-Amyloid Enhances Inhibition of the α7 Nicotinic Receptor and Promotes Neurotoxicity. Cells 2019, 8, 771. [Google Scholar] [CrossRef]
- Takata, K.; Amamiya, T.; Mizoguchi, H.; Kawanishi, S.; Kuroda, E.; Kitamura, R.; Ito, A.; Saito, Y.; Tawa, M.; Nagasawa, T.; et al. Alpha7 nicotinic acetylcholine receptor-specific agonist DMXBA (GTS-21) attenuates Aβ accumulation through suppression of neuronal γ-secretase activity and promotion of microglial amyloid-β phagocytosis and ameliorates cognitive impairment in a mouse mode. Neurobiol. Aging 2018, 62, 197–209. [Google Scholar] [CrossRef]
- Medeiros, R.; Castello, N.A.; Cheng, D.; Kitazawa, M.; Baglietto-Vargas, D.; Green, K.N.; Esbenshade, T.A.; Bitner, R.S.; Decker, M.W.; LaFerla, F.M. α7 Nicotinic Receptor Agonist Enhances Cognition in Aged 3xTg-AD Mice with Robust Plaques and Tangles. Am. J. Pathol. 2014, 184, 520–529. [Google Scholar] [CrossRef]
- Maurer, S.V.; Williams, C.L. The Cholinergic System Modulates Memory and Hippocampal Plasticity via Its Interactions with Non-Neuronal Cells. Front. Immunol. 2017, 8, 1489. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, J.; Wu, J.; Zhu, C.; Hui, Y.; Han, Y.; Huang, Z.; Ellsworth, K.; Fan, W. α7 nicotinic acetylcholine receptor-mediated neuroprotection against dopaminergic neuron loss in an MPTP mouse model via inhibition of astrocyte activation. J. Neuroinflamm. 2012, 9, 617. [Google Scholar] [CrossRef]
- Hua, Y.; Yang, B.; Chen, Q.; Zhang, J.; Hu, J.; Fan, Y. Activation of α7 Nicotinic Acetylcholine Receptor Protects Against 1-Methyl-4-Phenylpyridinium-Induced Astroglial Apoptosis. Front. Cell. Neurosci. 2019, 13, 507. [Google Scholar] [CrossRef] [PubMed]
- Nizri, E.; Irony-Tur-Sinai, M.; Lory, O.; Orr-Urtreger, A.; Lavi, E.; Brenner, T. Activation of the Cholinergic Anti-Inflammatory System by Nicotine Attenuates Neuroinflammation via Suppression of Th1 and Th17 Responses. J. Immunol. 2009, 183, 6681–6688. [Google Scholar] [CrossRef]
- Nizri, E.; Brenner, T. Modulation of inflammatory pathways by the immune cholinergic system. Amino Acids 2013, 45, 73–85. [Google Scholar] [CrossRef]
- Nizri, E.; Hamra-Amitay, Y.; Sicsic, C.; Lavon, I.; Brenner, T. Anti-inflammatory properties of cholinergic up-regulation: A new role for acetylcholinesterase inhibitors. Neuropharmacology 2006, 50, 540–547. [Google Scholar] [CrossRef]
- Nizri, E.; Irony-Tur-Sinai, M.; Faranesh, N.; Lavon, I.; Lavi, E.; Weinstock, M.; Brenner, T. Suppression of neuroinflammation and immunomodulation by the acetylcholinesterase inhibitor rivastigmine. J. Neuroimmunol. 2008, 203, 12–22. [Google Scholar] [CrossRef]
- Reale, M.; de Angelis, F.; di Nicola, M.; Capello, E.; di Ioia, M.; de Luca, G.; Lugaresi, A.; Tata, A.M. Relation between pro-inflammatory cytokines and acetylcholine levels in relapsing-remitting multiple sclerosis patients. Int. J. Mol. Sci. 2012, 13, 12656–12664. [Google Scholar] [CrossRef]
- Di Pinto, G.; Di Bari, M.; Martin-Alvarez, R.; Sperduti, S.; Serrano-Acedo, S.; Gatta, V.; Tata, A.M.; Mengod, G. Comparative study of the expression of cholinergic system components in the CNS of experimental autoimmune encephalomyelitis mice: Acute vs. remitting phase. Eur. J. Neurosci. 2018, 48, 2165–2181. [Google Scholar] [CrossRef] [PubMed]
- Reale, M.; Costantini, E.; Di Nicola, M.; D’Angelo, C.; Franchi, S.; D’Aurora, M.; Di Bari, M.; Orlando, V.; Galizia, S.; Ruggieri, S.; et al. Butyrylcholinesterase and Acetylcholinesterase polymorphisms in Multiple Sclerosis patients: Implication in peripheral inflammation. Sci. Rep. 2018, 8, 1319. [Google Scholar] [CrossRef] [PubMed]
- Di Bari, M.; Di Pinto, G.; Reale, M.; Mengod, G.; Tata, A.M. Cholinergic System and Neuroinflammation: Implication in Multiple Sclerosis. Cent. Nerv. Syst. Agents Med. Chem. 2017, 17, 109–115. [Google Scholar] [CrossRef]
- Reale, M.; Di Bari, M.; Di Nicola, M.; D’Angelo, C.; De Angelis, F.; Velluto, L.; Tata, A.M. Nicotinic receptor activation negatively modulates pro-inflammatory cytokine production in multiple sclerosis patients. Int. Immunopharmacol. 2015, 29, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Martin-Ruiz, C.M.; Haroutunian, V.H.; Long, P.; Young, A.H.; Davis, K.L.; Perry, E.K.; Court, J.A. Dementia rating and nicotinic receptor expression in the prefrontal cortex in schizophrenia. Biol. Psychiatry 2003, 54, 1222–1233. [Google Scholar] [CrossRef]
- Severance, E.G.; Yolken, R.H. Novel α7 nicotinic receptor isoforms and deficient cholinergic transcription in schizophrenia. Genes, Brain Behav. 2008, 7, 37–45. [Google Scholar] [CrossRef]
- Bacchelli, E.; Battaglia, A.; Cameli, C.; Lomartire, S.; Tancredi, R.; Thomson, S.; Sutcliffe, J.S.; Maestrini, E. Analysis of CHRNA7 rare variants in autism spectrum disorder susceptibility. Am. J. Med. Genet. A 2015, 167A, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xiao, C.; Indersmitten, T.; Freedman, R.; Leonard, S.; Lester, H.A. The Duplicated α7 Subunits Assemble and Form Functional Nicotinic Receptors with the Full-length α7. J. Biol. Chem. 2014, 289, 26451–26463. [Google Scholar] [CrossRef]
- Gault, J.; Hopkins, J.; Berger, R.; Drebing, C.; Logel, J.; Walton, C.; Short, M.; Vianzon, R.; Olincy, A.; Ross, R.G.; et al. Comparison of polymorphisms in the α7 nicotinic receptor gene and its partial duplication in schizophrenic and control subjects. Am. J. Med. Genet. 2003, 123B, 39–49. [Google Scholar] [CrossRef]
- Hashimoto, K. Targeting of α7 Nicotinic Acetylcholine Receptors in the Treatment of Schizophrenia and the Use of Auditory Sensory Gating as a Translational Biomarker. Curr. Pharm. Des. 2015, 21, 3797–3806. [Google Scholar] [CrossRef]
- Fan, H.; Gu, R.; Wei, D. The α7 nAChR selective agonists as drug candidates for Alzheimer’s disease. Adv. Exp. Med. Biol. 2015, 827, 353–365. [Google Scholar] [CrossRef]
- Dallanoce, C.; Magrone, P.; Matera, C.; Frigerio, F.; Grazioso, G.; De Amici, M.; Fucile, S.; Piccari, V.; Frydenvang, K.; Pucci, L.; et al. Design, Synthesis, and Pharmacological Characterization of Novel Spirocyclic Quinuclidinyl-Δ2-Isoxazoline Derivatives as Potent and Selective Agonists of α7 Nicotinic Acetylcholine Receptors. ChemMedChem 2011, 6, 889–903. [Google Scholar] [CrossRef]
- Pernarella, M.; Piovesana, R.; Matera, C.; Faroni, A.; Fiore, M.; Dini, L.; Reid, A.J.; Dallanoce, C.; Tata, A.M. Effects mediated by the α7 nicotinic acetylcholine receptor on cell proliferation and migration in rat adipose-derived stem cells. Eur. J. Histochem. 2020, 64, 61–70. [Google Scholar] [CrossRef]
- Jones, I.W.; Barik, J.; O’Neill, M.J.; Wonnacott, S. Alpha bungarotoxin-1.4nm gold: A novel conjugate for visualising the precise subcellular distribution of alpha 7* nicotinic acetylcholine receptors. J. Neurosci. Methods 2004, 134, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Meyer, E.M.; Tay, E.T.; Papke, R.L.; Meyers, C.; Huang, G.L.; de Fiebre, C.M. 3-[2,4-Dimethoxybenzylidene]anabaseine (DMXB) selectively activates rat alpha7 receptors and improves memory-related behaviors in a mecamylamine-sensitive manner. Brain Res. 1997, 768, 49–56. [Google Scholar] [CrossRef]
- Lopez-Hernandez, G.; Placzek, A.N.; Thinschmidt, J.S.; Lestage, P.; Trocme-Thibierge, C.; Morain, P.; Papke, R.L. Partial agonist and neuromodulatory activity of S 24795 for alpha7 nAChR responses of hippocampal interneurons. Neuropharmacology 2007, 53, 134–144. [Google Scholar] [CrossRef]
- Horenstein, N.A.; Papke, R.L. Anti-inflammatory Silent Agonists. ACS Med. Chem. Lett. 2017, 8, 989–991. [Google Scholar] [CrossRef] [PubMed]
- Godin, J.-R.; Roy, P.; Quadri, M.; Bagdas, D.; Toma, W.; Narendrula-Kotha, R.; Kishta, O.A.; Damaj, M.I.; Horenstein, N.A.; Papke, R.L.; et al. A silent agonist of α7 nicotinic acetylcholine receptors modulates inflammation ex vivo and attenuates EAE. Brain. Behav. Immun. 2020, 87, 286–300. [Google Scholar] [CrossRef]
- Eskildsen, J.; Redrobe, J.P.; Sams, A.G.; Dekermendjian, K.; Laursen, M.; Boll, J.B.; Papke, R.L.; Bundgaard, C.; Frederiksen, K.; Bastlund, J.F. Discovery and optimization of Lu AF58801, a novel, selective and brain penetrant positive allosteric modulator of alpha-7 nicotinic acetylcholine receptors: Attenuation of subchronic phencyclidine (PCP)-induced cognitive deficits in rats following oral ad. Bioorg. Med. Chem. Lett. 2014, 24, 288–293. [Google Scholar] [CrossRef]
- Unal, G.; Bekci, H.; Cumaoglu, A.; Yerer, M.B.; Aricioglu, F. Alpha 7 nicotinic receptor agonist and positive allosteric modulators improved social and molecular deficits of MK-801 model of schizophrenia in rats. Pharmacol. Biochem. Behav. 2020, 193, 172916. [Google Scholar] [CrossRef] [PubMed]
| Nicotinic Agonists | Receptor Selectivity | Ki |
|---|---|---|
| SEN 12333 | Full agonist α7 subunit | 260 nM |
| PNU-282907 | Full agonist α7 subunit | 27 nM |
| PNU-120596 | Full agonist α7 subunit | 0.9 µM |
| TC 5619 | Full agonist α7 subunit | 1 nM |
| ICH3 | Partial agonist α7 subunit | 4.6 nM |
| S24795 | Partial agonist α7 subunit | 34 nM |
| A-582941 | Partial agonist α7 subunit | 16 nM |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piovesana, R.; Salazar Intriago, M.S.; Dini, L.; Tata, A.M. Cholinergic Modulation of Neuroinflammation: Focus on α7 Nicotinic Receptor. Int. J. Mol. Sci. 2021, 22, 4912. https://doi.org/10.3390/ijms22094912
Piovesana R, Salazar Intriago MS, Dini L, Tata AM. Cholinergic Modulation of Neuroinflammation: Focus on α7 Nicotinic Receptor. International Journal of Molecular Sciences. 2021; 22(9):4912. https://doi.org/10.3390/ijms22094912
Chicago/Turabian StylePiovesana, Roberta, Michael Sebastian Salazar Intriago, Luciana Dini, and Ada Maria Tata. 2021. "Cholinergic Modulation of Neuroinflammation: Focus on α7 Nicotinic Receptor" International Journal of Molecular Sciences 22, no. 9: 4912. https://doi.org/10.3390/ijms22094912
APA StylePiovesana, R., Salazar Intriago, M. S., Dini, L., & Tata, A. M. (2021). Cholinergic Modulation of Neuroinflammation: Focus on α7 Nicotinic Receptor. International Journal of Molecular Sciences, 22(9), 4912. https://doi.org/10.3390/ijms22094912








