The Putative RNA-Binding Protein Dri1 Promotes the Loading of Kinesin-14/Klp2 to the Mitotic Spindle and Is Sequestered into Heat-Induced Protein Aggregates in Fission Yeast
Abstract
1. Introduction
2. Results
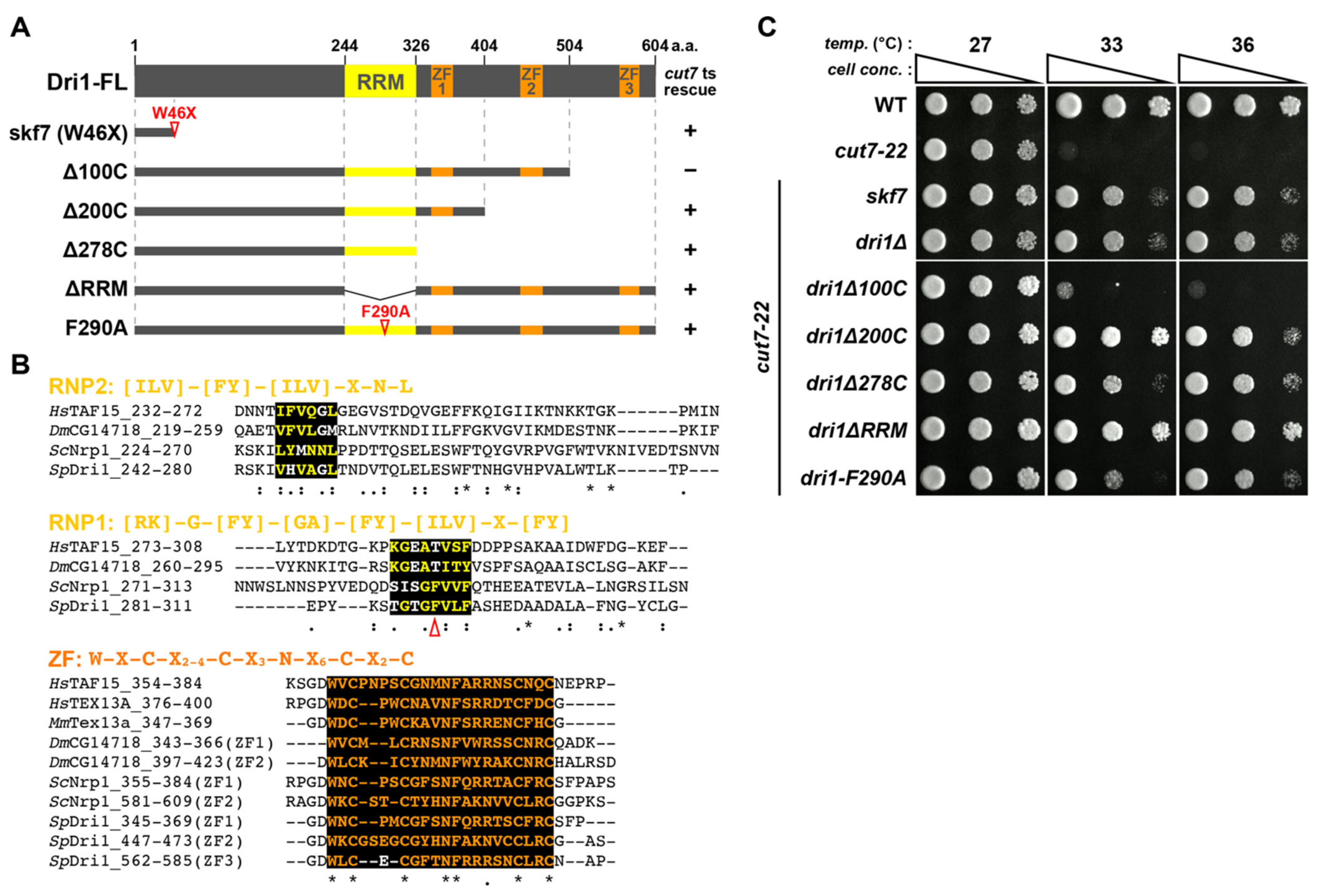
2.1. Loss of Function of a Putative RNA Binding Protein Dri1/Skf7 Suppresses the Cut7-22 Temperature-Sensitive Growth Defect
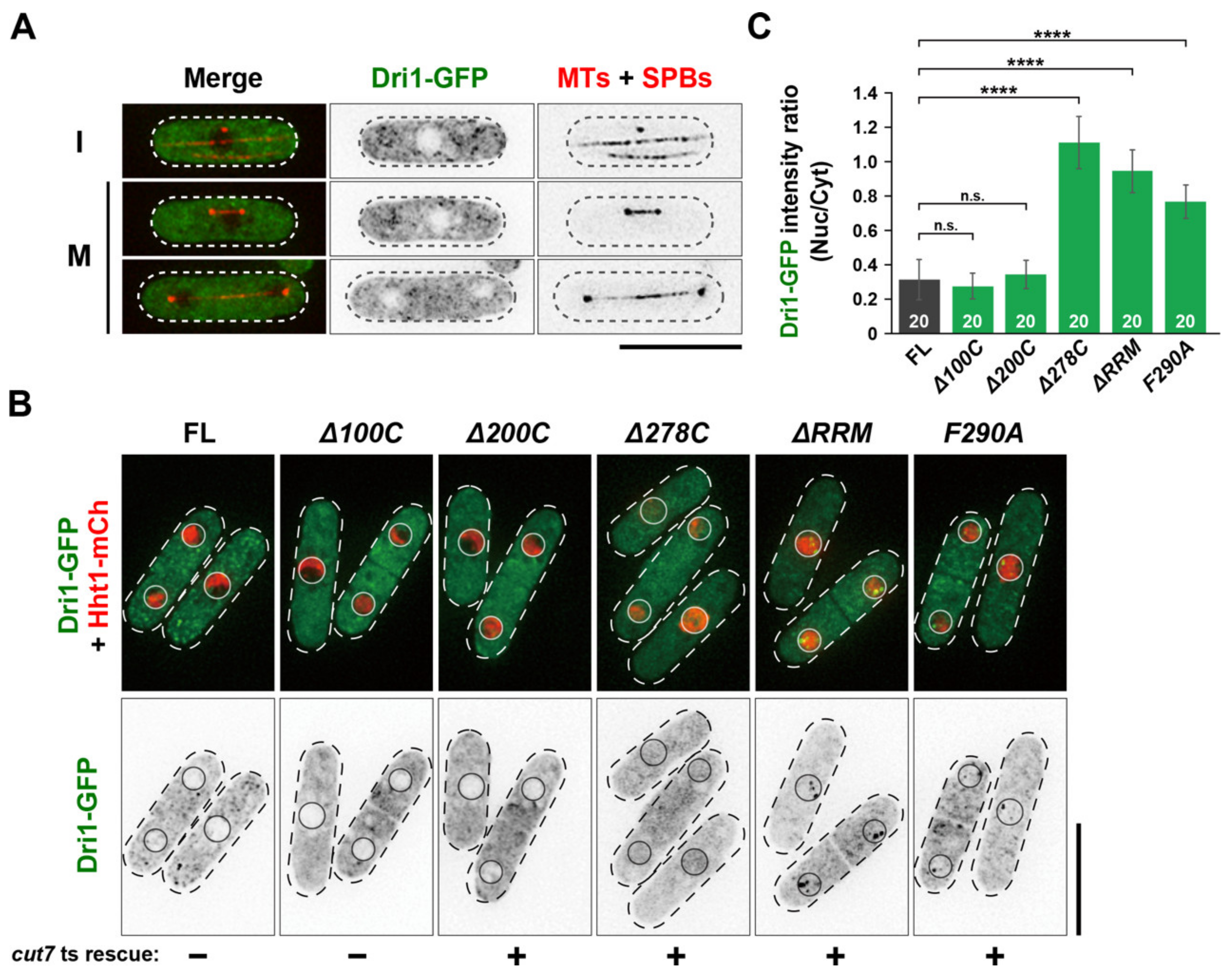
2.2. The RNA-Binding Domains of Dri1 Are Required for Its Function
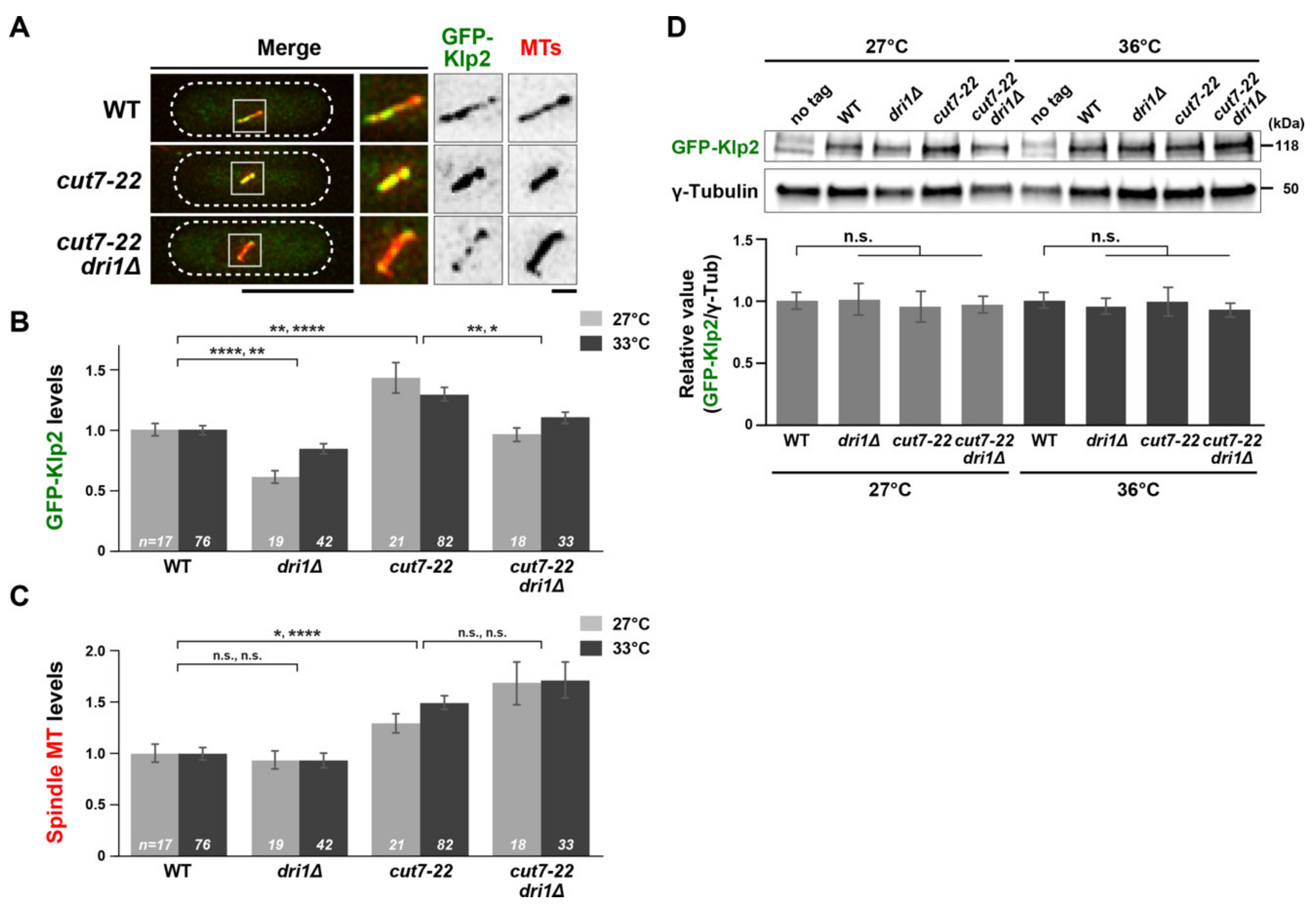
2.3. Dri1 Is Required for Proper Localization of Kinesin-14/Klp2 on the Spindle Microtubules
2.4. Dri1 Is Dispensable for the Spindle Localization of EB1/Mal3
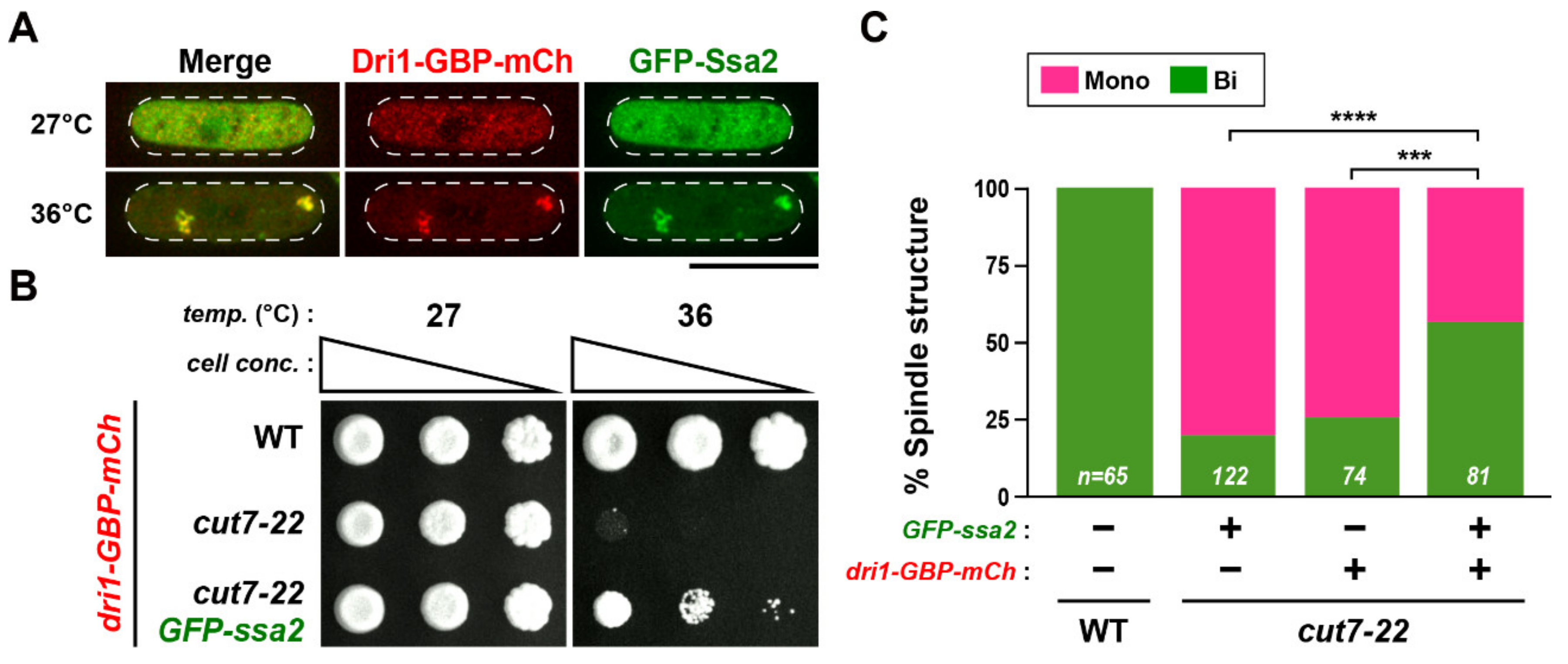
2.5. Cytoplasmic Dri1 Counteracts the Kinesin-5/Cut7 Function
2.6. Dri1 Is Exported from the Nucleus in a Rae1-Dependent Manner
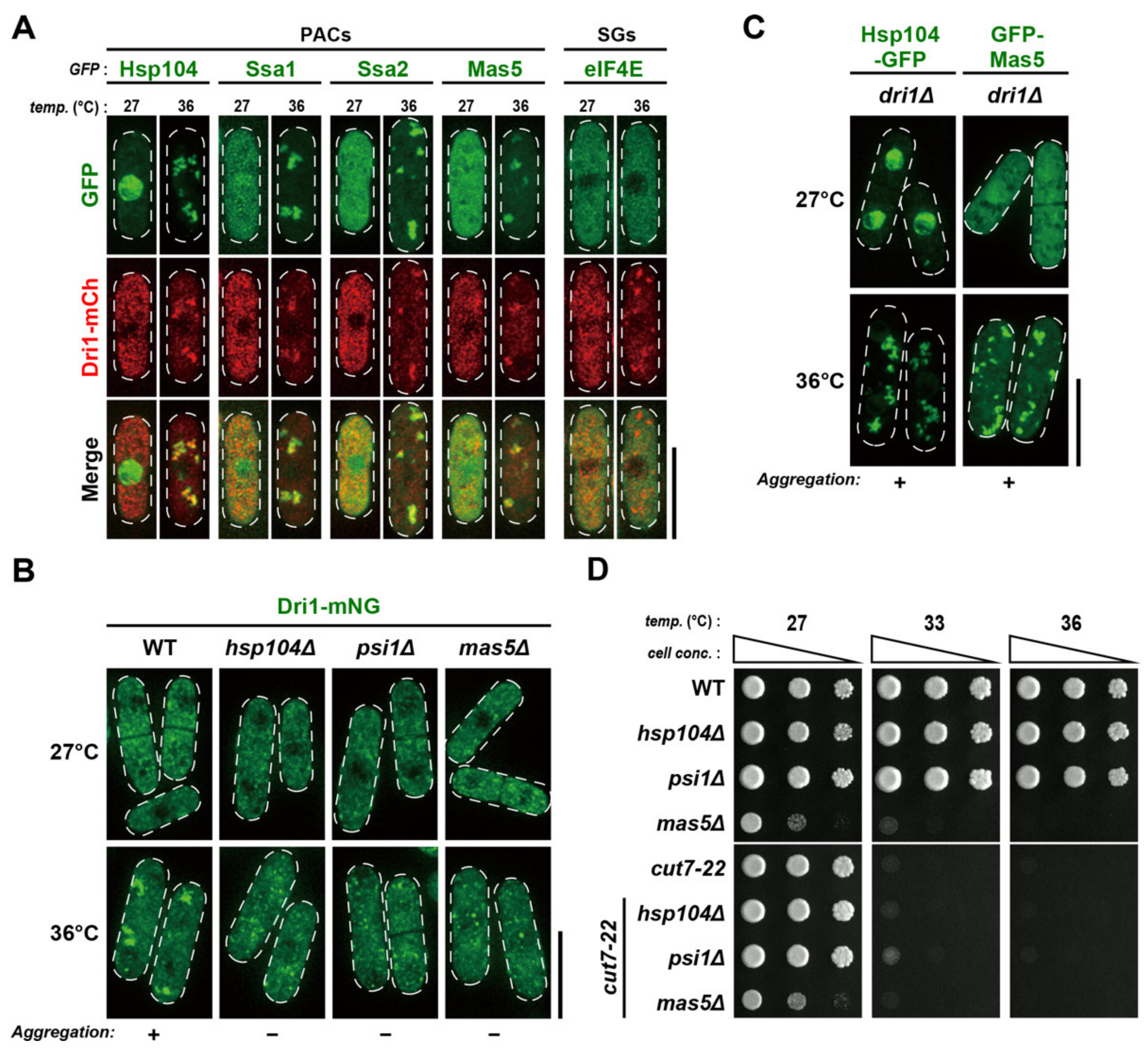
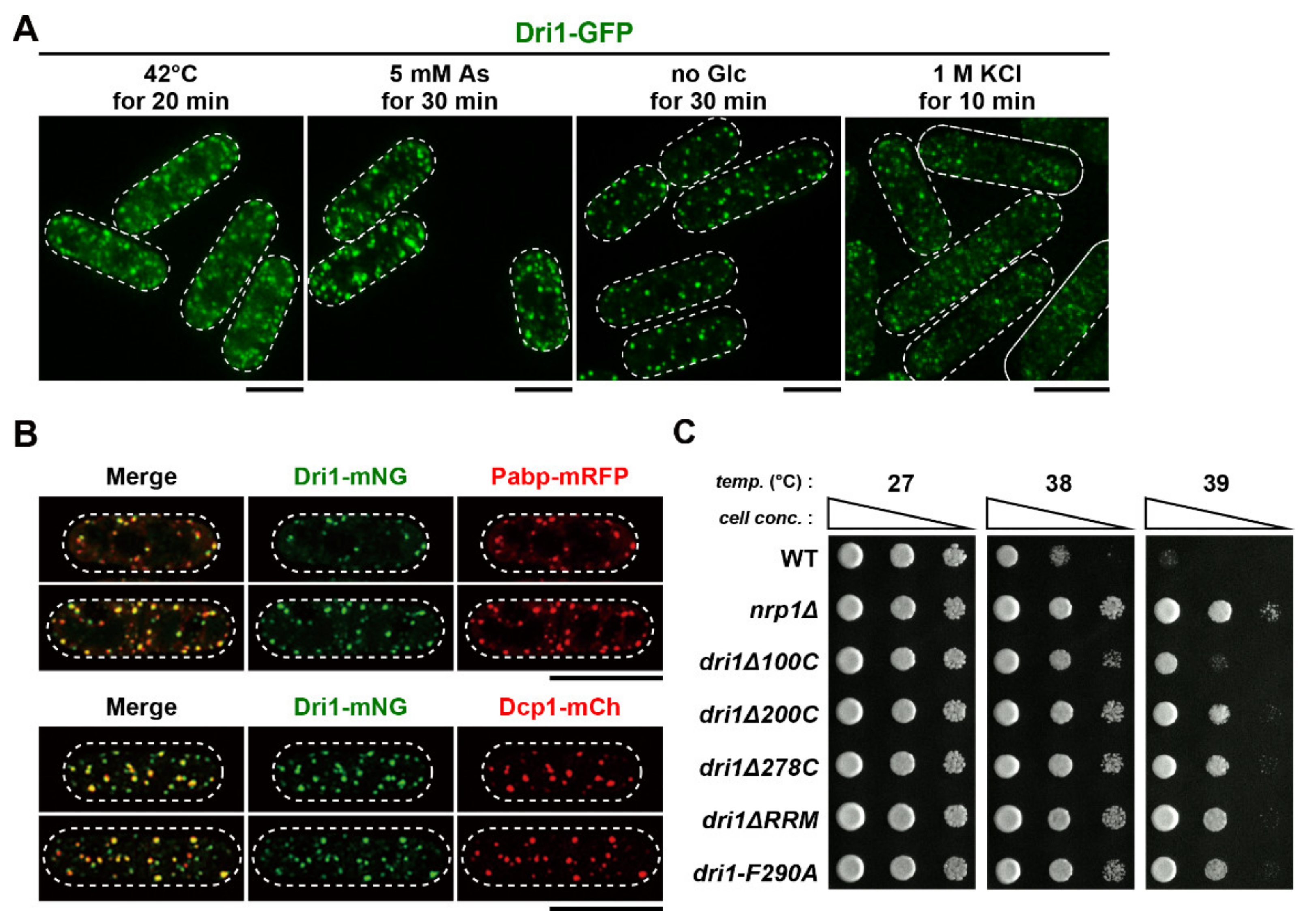
2.7. Dri1 Assembles into Protein Aggregate Centers upon Mild Temperature Shift-Up
2.8. PACs Spatially and Functionally Sequester Dri1 upon Mild Heat Stress
2.9. Dri1 Accumulates at Stress Granules and Limits the Maximum Temperature for Growth
3. Discussion
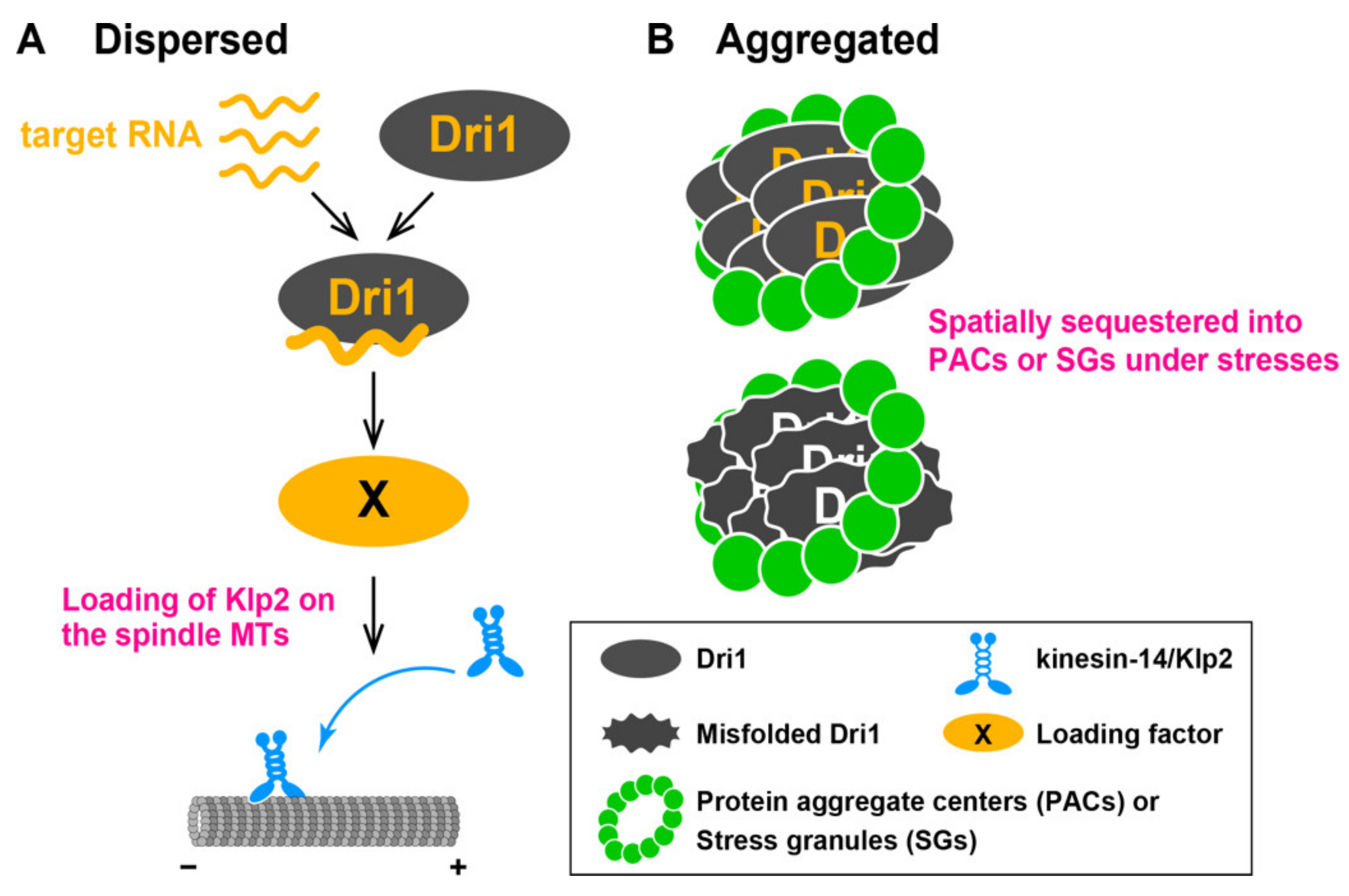
3.1. Dri1 Promotes Loading of Kinesin-14/Klp2 onto Spindle MTs
3.2. How Does Dri1 Play a Role through Its RNA-Binding Ability?
3.3. Identification of Dri1 as a Component of Protein Aggregate Centers and Stress Granules under Heat Stress
3.4. Conservation of the Tex13/Dri1 Protein Family
4. Materials and Methods
4.1. Strains, Media, and Genetic Methods
4.2. Next Generation Sequencing and Annotation of skf7 Genes
4.3. Stress Treatment
4.4. Preparation and Manipulation of Nucleic Acids
4.5. Strain Construction, Gene Disruption and Epitope Tagging
4.6. Construction of Truncated/Mutated dri1 Mutants
4.7. Western Blot Analysis
4.8. Fluorescence Microscopy
4.9. Quantification of Fluorescent Signal Intensities
4.10. Statistical Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MT | Microtubule |
| MAP | Microtubule-associated protein |
| SPB | Spindle pole body |
| RBP | RNA-binding protein |
| ncRNA | Non-coding RNA |
| SG | Stress granule |
| P body | Processing body |
| PAC | Protein aggregation center |
| ts | Temperature sensitive |
| RRM | RNA recognition motif |
| ZF | Zinc finger |
| GBP | GFP-binding protein |
References
- Desai, A.; Mitchison, T.J. Microtubule polymerization dynamics. Annu. Rev. Cell Dev. Biol. 1997, 13, 83–117. [Google Scholar] [CrossRef] [PubMed]
- Heald, R.; Khodjakov, A. Thirty years of search and capture: The complex simplicity of mitotic spindle assembly. J. Cell Biol. 2015, 211, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Akhmanova, A. Microtubule tip-interacting proteins: A view from both ends. Curr. Opin. Cell Biol. 2011, 23, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Kapitein, L.C.; Peterman, E.J.; Kwok, B.H.; Kim, J.H.; Kapoor, T.M.; Schmidt, C.F. The bipolar mitotic kinesin Eg5 moves on both microtubules that it crosslinks. Nature 2005, 435, 114–118. [Google Scholar] [CrossRef]
- Kashina, A.S.; Baskin, R.J.; Cole, D.G.; Wedaman, K.P.; Saxton, W.M.; Scholey, J.M. A bipolar kinesin. Nature 1996, 379, 270–272. [Google Scholar] [CrossRef]
- Avunie-Masala, R.; Movshovich, N.; Nissenkorn, Y.; Gerson-Gurwitz, A.; Fridman, V.; Koivomagi, M.; Loog, M.; Hoyt, M.A.; Zaritsky, A.; Gheber, L. Phospho-regulation of kinesin-5 during anaphase spindle elongation. J. Cell Sci. 2011, 124 Pt 6, 873–878. [Google Scholar] [CrossRef]
- Goldstein, A.; Siegler, N.; Goldman, D.; Judah, H.; Valk, E.; Koivomagi, M.; Loog, M.; Gheber, L. Three Cdk1 sites in the kinesin-5 Cin8 catalytic domain coordinate motor localization and activity during anaphase. Cell Mol. Life Sci. 2017, 74, 3395–3412. [Google Scholar] [CrossRef]
- Blangy, A.; Lane, H.A.; d’Herin, P.; Harper, M.; Kress, M.; Nigg, E.A. Phosphorylation by p34cdc2 regulates spindle association of human Eg5, a kinesin-related motor essential for bipolar spindle formation in vivo. Cell 1995, 83, 1159–1169. [Google Scholar] [CrossRef]
- Bertran, M.T.; Sdelci, S.; Regue, L.; Avruch, J.; Caelles, C.; Roig, J. Nek9 is a Plk1-activated kinase that controls early centrosome separation through Nek6/7 and Eg5. EMBO J. 2011, 30, 2634–2647. [Google Scholar] [CrossRef]
- Drummond, D.R.; Hagan, I.M. Mutations in the bimC box of Cut7 indicate divergence of regulation within the bimC family of kinesin related proteins. J. Cell Sci. 1998, 111, 853–865. [Google Scholar] [CrossRef]
- Smith, E.; Hegarat, N.; Vesely, C.; Roseboom, I.; Larch, C.; Streicher, H.; Straatman, K.; Flynn, H.; Skehel, M.; Hirota, T.; et al. Differential control of Eg5-dependent centrosome separation by Plk1 and Cdk1. EMBO J. 2011, 30, 2233–2245. [Google Scholar] [CrossRef]
- Hagan, I.; Yanagida, M. Kinesin-related cut7 protein associates with mitotic and meiotic spindles in fission yeast. Nature 1992, 356, 74–76. [Google Scholar] [CrossRef]
- Hagan, I.; Yanagida, M. Novel potential mitotic motor protein encoded by the fission yeast cut7+ gene. Nature 1990, 347, 563–566. [Google Scholar] [CrossRef]
- Olmsted, Z.T.; Colliver, A.G.; Riehlman, T.D.; Paluh, J.L. Kinesin-14 and kinesin-5 antagonistically regulate microtubule nucleation by γ-TuRC in yeast and human cells. Nat. Commun. 2014, 5, 5339. [Google Scholar] [CrossRef]
- Syrovatkina, V.; Tran, P.T. Loss of kinesin-14 results in aneuploidy via kinesin-5-dependent microtubule protrusions leading to chromosome cut. Nat. Commun. 2015, 6, 7322. [Google Scholar] [CrossRef]
- Yukawa, M.; Ikebe, C.; Toda, T. The Msd1-Wdr8-Pkl1 complex anchors microtubule minus ends to fission yeast spindle pole bodies. J. Cell Biol. 2015, 209, 549–562. [Google Scholar] [CrossRef]
- Troxell, C.L.; Sweezy, M.A.; West, R.R.; Reed, K.D.; Carson, B.D.; Pidoux, A.L.; Cande, W.Z.; McIntosh, J.R. pkl1+: And klp2+: Two kinesins of the Kar3 subfamily in fission yeast perform different functions in both mitosis and meiosis. Mol. Biol. Cell 2001, 12, 3476–3488. [Google Scholar] [CrossRef]
- Pidoux, A.L.; LeDizet, M.; Cande, W.Z. Fission yeast pkl1 is a kinesin-related protein involved in mitotic spindle function. Mol. Biol. Cell 1996, 7, 1639–1655. [Google Scholar] [CrossRef]
- Yukawa, M.; Kawakami, T.; Okazaki, M.; Kume, K.; Tang, N.H.; Toda, T. A microtubule polymerase cooperates with the kinesin-6 motor and a microtubule cross-linker to promote bipolar spindle assembly in the absence of kinesin-5 and kinesin-14 in fission yeast. Mol. Biol. Cell 2017, 28, 3647–3659. [Google Scholar] [CrossRef]
- Yukawa, M.; Teratani, Y.; Toda, T. How essential Kinesin-5 becomes non-essential in fission yeast: Force balance and microtubule dynamics matter. Cells 2020, 9, 1154. [Google Scholar] [CrossRef]
- Rincon, S.A.; Lamson, A.; Blackwell, R.; Syrovatkina, V.; Fraisier, V.; Paoletti, A.; Betterton, M.D.; Tran, P.T. Kinesin-5-independent mitotic spindle assembly requires the antiparallel microtubule crosslinker Ase1 in fission yeast. Nat. Commun. 2017, 8, 15286. [Google Scholar] [CrossRef]
- Shirasugi, Y.; Sato, M. Kinetochore-mediated outward force promotes spindle pole separation in fission yeast. Mol. Biol. Cell 2019, 30, 2802–2813. [Google Scholar] [CrossRef]
- Yukawa, M.; Yamada, Y.; Yamauchi, T.; Toda, T. Two spatially distinct kinesin-14 proteins, Pkl1 and Klp2, generate collaborative inward forces against kinesin-5 Cut7 in S. pombe. J. Cell Sci. 2018, 131, jcs.210740. [Google Scholar] [CrossRef]
- Ito, K.K.; Watanabe, K.; Kitagawa, D. The emerging role of ncRNAs and RNA-binding proteins in mitotic apparatus formation. Non-Coding RNA 2020, 6, 13. [Google Scholar] [CrossRef]
- Grenfell, A.W.; Heald, R.; Strzelecka, M. Mitotic noncoding RNA processing promotes kinetochore and spindle assembly in Xenopus. J. Cell Biol. 2016, 214, 133–141. [Google Scholar] [CrossRef]
- Hussain, S.; Benavente, S.B.; Nascimento, E.; Dragoni, I.; Kurowski, A.; Gillich, A.; Humphreys, P.; Frye, M. The nucleolar RNA methyltransferase Misu (NSun2) is required for mitotic spindle stability. J. Cell Biol. 2009, 186, 27–40. [Google Scholar] [CrossRef]
- Jambhekar, A.; Emerman, A.B.; Schweidenback, C.T.; Blower, M.D. RNA stimulates Aurora B kinase activity during mitosis. PLoS ONE 2014, 9, e100748. [Google Scholar] [CrossRef]
- Hassine, S.; Bonnet-Magnaval, F.; Benoit Bouvrette, L.P.; Doran, B.; Ghram, M.; Bouthillette, M.; Lecuyer, E.; DesGroseillers, L. Staufen1 localizes to the mitotic spindle and controls the localization of RNA populations to the spindle. J. Cell Sci. 2020, 133, jcs247155. [Google Scholar] [CrossRef]
- Blower, M.D.; Nachury, M.; Heald, R.; Weis, K. A Rae1-containing ribonucleoprotein complex is required for mitotic spindle assembly. Cell 2005, 121, 223–234. [Google Scholar] [CrossRef]
- Hyman, A.A.; Weber, C.A.; Julicher, F. Liquid-liquid phase separation in biology. Annu. Rev. Cell Dev. Biol. 2014, 30, 39–58. [Google Scholar] [CrossRef] [PubMed]
- Protter, D.S.W.; Parker, R. Principles and properties of stress granules. Trends Cell Biol. 2016, 26, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Eulalio, A.; Behm-Ansmant, I.; Izaurralde, E. P bodies: At the crossroads of post-transcriptional pathways. Nat. Rev. Mol. Cell Biol. 2007, 8, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Boronat, S.; Marte, L.; Vega, M.; Garcia-Santamarina, S.; Cabrera, M.; Ayte, J.; Hidalgo, E. The Hsp40 Mas5 connects protein quality control and the general stress response through the thermo-sensitive Pyp1. iScience 2020, 23, 101725. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, M.; Boronat, S.; Marte, L.; Vega, M.; Perez, P.; Ayte, J.; Hidalgo, E. Chaperone-facilitated aggregation of thermo-sensitive proteins shields them from degradation during heat stress. Cell Rep. 2020, 30, 2430–2443.e4. [Google Scholar] [CrossRef]
- Boronat, S.; Cabrera, M.; Hidalgo, E. Spatial sequestration of misfolded proteins as an active chaperone-mediated process during heat stress. Curr. Genet. 2021. [Google Scholar] [CrossRef]
- Yukawa, M.; Yamada, Y.; Toda, T. Suppressor analysis uncovers that MAPs and microtubule dynamics balance with the Cut7/Kinesin-5 motor for mitotic spindle assembly in Schizosaccharomyces pombe. G3 (Bethesda) 2019, 9, 269–280. [Google Scholar] [CrossRef]
- Toda, T.; Adachi, Y.; Hiraoka, Y.; Yanagida, M. Identification of the pleiotropic cell division cycle gene NDA2 as one of two different α-tubulin genes in Schizosaccharomyces pombe. Cell 1984, 37, 233–242. [Google Scholar] [CrossRef]
- Beinhauer, J.D.; Hagan, I.M.; Hegemann, J.H.; Fleig, U. Mal3, the fission yeast homologue of the human APC-interacting protein EB-1 is required for microtubule integrity and the maintenance of cell form. J. Cell Biol. 1997, 139, 717–728. [Google Scholar] [CrossRef]
- Wright, M.W.; Eyre, T.A.; Lush, M.J.; Povey, S.; Bruford, E.A. HCOP: The HGNC comparison of orthology predictions search tool. Mamm. Genome 2005, 16, 827–828. [Google Scholar] [CrossRef]
- Eyre, T.A.; Wright, M.W.; Lush, M.J.; Bruford, E.A. HCOP: A searchable database of human orthology predictions. Brief. Bioinform. 2007, 8, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Burd, C.G.; Dreyfuss, G. Conserved structures and diversity of functions of RNA-binding proteins. Science 1994, 265, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Mana-Capelli, S.; McLean, J.R.; Chen, C.T.; Gould, K.L.; McCollum, D. The kinesin-14 Klp2 is negatively regulated by the SIN for proper spindle elongation and telophase nuclear positioning. Mol. Biol. Cell 2012, 23, 4592–4600. [Google Scholar] [CrossRef] [PubMed]
- Szczesna, E.; Kasprzak, A.A. Insights into the process of EB1-dependent tip-tracking of kinesin-14 Ncd. The role of the microtubule. Eur. J. Cell Biol. 2016, 95, 521–530. [Google Scholar] [CrossRef]
- Kornakov, N.; Mollers, B.; Westermann, S. The EB1-Kinesin-14 complex is required for efficient metaphase spindle assembly and kinetochore bi-orientation. J. Cell Biol. 2020, 219, e202003072. [Google Scholar] [CrossRef]
- Braun, M.; Lansky, Z.; Bajer, S.; Fink, G.; Kasprzak, A.A.; Diez, S. The human kinesin-14 HSET tracks the tips of growing microtubules in vitro. Cytoskeleton 2013, 70, 515–521. [Google Scholar] [CrossRef]
- Chang, L.; Gould, K.L. Sid4p is required to localize components of the septation initiation pathway to the spindle pole body in fission yeast. Proc. Natl. Acad. Sci. USA 2000, 97, 5249–5254. [Google Scholar] [CrossRef]
- Choe, J.; Schuster, T.; Grunstein, M. Organization, primary structure, and evolution of histone H2A and H2B genes of the fission yeast Schizosaccharomyces pombe. Mol. Cell Biol. 1985, 5, 3261–3269. [Google Scholar] [CrossRef]
- Matsumoto, S.; Yanagida, M. Histone gene organization of fission yeast: A common upstream sequence. EMBO J. 1985, 4, 3531–3538. [Google Scholar] [CrossRef]
- Yoon, J.H.; Whalen, W.A.; Bharathi, A.; Shen, R.; Dhar, R. Npp106p, a Schizosaccharomyces pombe nucleoporin similar to Saccharomyces cerevisiae Nic96p, functionally interacts with Rae1p in mRNA export. Mol. Cell Biol. 1997, 17, 7047–7060. [Google Scholar] [CrossRef][Green Version]
- Satoh, R.; Matsumura, Y.; Tanaka, A.; Takada, M.; Ito, Y.; Hagihara, K.; Inari, M.; Kita, A.; Fukao, A.; Fujiwara, T.; et al. Spatial regulation of the KH domain RNA-binding protein Rnc1 mediated by a Crm1-independent nuclear export system in Schizosaccharomyces pombe. Mol. Microbiol. 2017, 104, 428–448. [Google Scholar] [CrossRef]
- Brown, J.A.; Bharathi, A.; Ghosh, A.; Whalen, W.; Fitzgerald, E.; Dhar, R. A mutation in the Schizosaccharomyces pombe rae1 gene causes defects in poly(A)+ RNA export and in the cytoskeleton. J. Biol. Chem. 1995, 270, 7411–7419. [Google Scholar] [CrossRef]
- Thakurta, A.G.; Yoon, J.H.; Dhar, R. Schizosaccharomyces pombe spPABP, a homologue of Saccharomyces cerevisiae Pab1p, is a non-essential, shuttling protein that facilitates mRNA export. Yeast 2002, 19, 795–802. [Google Scholar] [CrossRef]
- Satoh, R.; Hagihara, K.; Sugiura, R. Rae1-mediated nuclear export of Rnc1 is an important determinant in controlling MAPK signaling. Curr. Genet. 2018, 64, 103–108. [Google Scholar] [CrossRef]
- Kedersha, N.; Anderson, P. Stress granules: Sites of mRNA triage that regulate mRNA stability and translatability. Biochem. Soc. Trans. 2002, 30 Pt 6, 963–969. [Google Scholar] [CrossRef]
- Rothbauer, U.; Zolghadr, K.; Muyldermans, S.; Schepers, A.; Cardoso, M.C.; Leonhardt, H. A versatile nanotrap for biochemical and functional studies with fluorescent fusion proteins. Mol. Cell Proteom. 2008, 7, 282–289. [Google Scholar] [CrossRef]
- Nilsson, D.; Sunnerhagen, P. Cellular stress induces cytoplasmic RNA granules in fission yeast. RNA 2011, 17, 120–133. [Google Scholar] [CrossRef]
- Wen, W.L.; Stevenson, A.L.; Wang, C.Y.; Chen, H.J.; Kearsey, S.E.; Norbury, C.J.; Watt, S.; Bahler, J.; Wang, S.W. Vgl1, a multi-KH domain protein, is a novel component of the fission yeast stress granules required for cell survival under thermal stress. Nucleic Acids Res. 2010, 38, 6555–6566. [Google Scholar] [CrossRef]
- Satoh, R.; Tanaka, A.; Kita, A.; Morita, T.; Matsumura, Y.; Umeda, N.; Takada, M.; Hayashi, S.; Tani, T.; Shinmyozu, K.; et al. Role of the RNA-binding protein Nrd1 in stress granule formation and Its implication in the stress response in fission yeast. PLoS ONE 2012, 7, e29683. [Google Scholar] [CrossRef]
- Advani, V.M.; Ivanov, P. Stress granule subtypes: An emerging link to neurodegeneration. Cell Mol. Life Sci. 2020, 77, 4827–4845. [Google Scholar] [CrossRef]
- Hsiao, W.Y.; Wang, Y.T.; Wang, S.W. Fission yeast Puf2, a Pumilio and FBF family of RNA-binding protein, links stress granules to processing bodies. Mol. Cell Biol. 2020, 40, e00589-19. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Chen, W.L.; Wang, S.W. Pdc1 functions in the assembly of P bodies in Schizosaccharomyces pombe. Mol. Cell Biol. 2013, 33, 1244–1253. [Google Scholar] [CrossRef] [PubMed]
- Ban, H.; Sun, W.; Chen, Y.; Chen, Y.; Li, F. Dri1 mediates heterochromatin assembly via RNAi and histone deacetylation. Genetics 2021. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.J.; McCarrey, J.R.; Yang, F.; Page, D.C. An abundance of X-linked genes expressed in spermatogonia. Nat. Genet. 2001, 27, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.D.; Mansfield, R.E.; Leung, W.; Vaz, P.M.; Loughlin, F.E.; Grant, R.P.; Mackay, J.P. Characterization of a family of RanBP2-type zinc fingers that can recognize single-stranded RNA. J. Mol. Biol. 2011, 407, 273–283. [Google Scholar] [CrossRef]
- Kwon, J.T.; Jin, S.; Choi, H.; Kim, J.; Jeong, J.; Kim, J.; Kim, Y.; Cho, B.N.; Cho, C. Identification and characterization of germ cell genes expressed in the F9 testicular teratoma stem cell line. PLoS ONE 2014, 9, e103837. [Google Scholar] [CrossRef]
- Buchan, J.R.; Muhlrad, D.; Parker, R. P bodies promote stress granule assembly in Saccharomyces cerevisiae. J. Cell Biol. 2008, 183, 441–455. [Google Scholar] [CrossRef]
- Moreno, S.; Klar, A.; Nurse, P. Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol. 1991, 194, 795–823. [Google Scholar]
- Sato, M.; Dhut, S.; Toda, T. New drug-resistant cassettes for gene disruption and epitope tagging in Schizosaccharomyces pombe. Yeast 2005, 22, 583–591. [Google Scholar] [CrossRef]
- Bahler, J.; Wu, J.Q.; Longtine, M.S.; Shah, N.G.; McKenzie, A., 3rd; Steever, A.B.; Wach, A.; Philippsen, P.; Pringle, J.R. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 1998, 14, 943–951. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yukawa, M.; Ohishi, M.; Yamada, Y.; Toda, T. The Putative RNA-Binding Protein Dri1 Promotes the Loading of Kinesin-14/Klp2 to the Mitotic Spindle and Is Sequestered into Heat-Induced Protein Aggregates in Fission Yeast. Int. J. Mol. Sci. 2021, 22, 4795. https://doi.org/10.3390/ijms22094795
Yukawa M, Ohishi M, Yamada Y, Toda T. The Putative RNA-Binding Protein Dri1 Promotes the Loading of Kinesin-14/Klp2 to the Mitotic Spindle and Is Sequestered into Heat-Induced Protein Aggregates in Fission Yeast. International Journal of Molecular Sciences. 2021; 22(9):4795. https://doi.org/10.3390/ijms22094795
Chicago/Turabian StyleYukawa, Masashi, Mitsuki Ohishi, Yusuke Yamada, and Takashi Toda. 2021. "The Putative RNA-Binding Protein Dri1 Promotes the Loading of Kinesin-14/Klp2 to the Mitotic Spindle and Is Sequestered into Heat-Induced Protein Aggregates in Fission Yeast" International Journal of Molecular Sciences 22, no. 9: 4795. https://doi.org/10.3390/ijms22094795
APA StyleYukawa, M., Ohishi, M., Yamada, Y., & Toda, T. (2021). The Putative RNA-Binding Protein Dri1 Promotes the Loading of Kinesin-14/Klp2 to the Mitotic Spindle and Is Sequestered into Heat-Induced Protein Aggregates in Fission Yeast. International Journal of Molecular Sciences, 22(9), 4795. https://doi.org/10.3390/ijms22094795






