Gut Susceptibility to Viral Invasion: Contributing Roles of Diet, Microbiota and Enteric Nervous System to Mucosal Barrier Preservation
Abstract
1. Introduction
2. What Types of Viruses Are Found in the Gut?
| Family | Genus | Clinical Manifestation | Reference |
|---|---|---|---|
| Adenoviridae | Mastadenovirus C, F and others | Gastroenteritis | [7] |
| Anelloviridae | Anellovirus | Enteritis a | [8] |
| Astroviridae | Astrovirus | Gastroenteritis, meningitis, encephalitis | [9] |
| Caliciviridae | Norovirus | Gastroenteritis a | [10] |
| Sapovirus | Gastroenteritis a | [11] | |
| Circoviridae | Circovirus | Unclear | [12] |
| Cyclovirus | Unclear | [12] | |
| Coronaviridae | Torovirus | Gastroenteritis a,b | [13] |
| Parvoviridae | Bocavirus | Gastroenteritis b | [14] |
| Bufavirus | Unclear | [15] | |
| Picobirnaviridae | Picobirnavirus | Gastroenteritis a | [16] |
| Picornaviridae | Enteroviruses including Poliovirus and Echovirus | Enteritis, neurologic syndrome | [17] |
| Parechovirus | Gastroenteritis, respiratory infection, sepsis-like illness, CNS infection | [18] | |
| Cardiovirus | Gastroenteritis, respiratory infection, myocarditis | [19] | |
| Salivirus | Gastroenteritis | [20] | |
| Polyomaviridae | Polyomavirus | Unclear | [21] |
| Reoviridae | Rotavirus | Gastroenteritis | [22] |
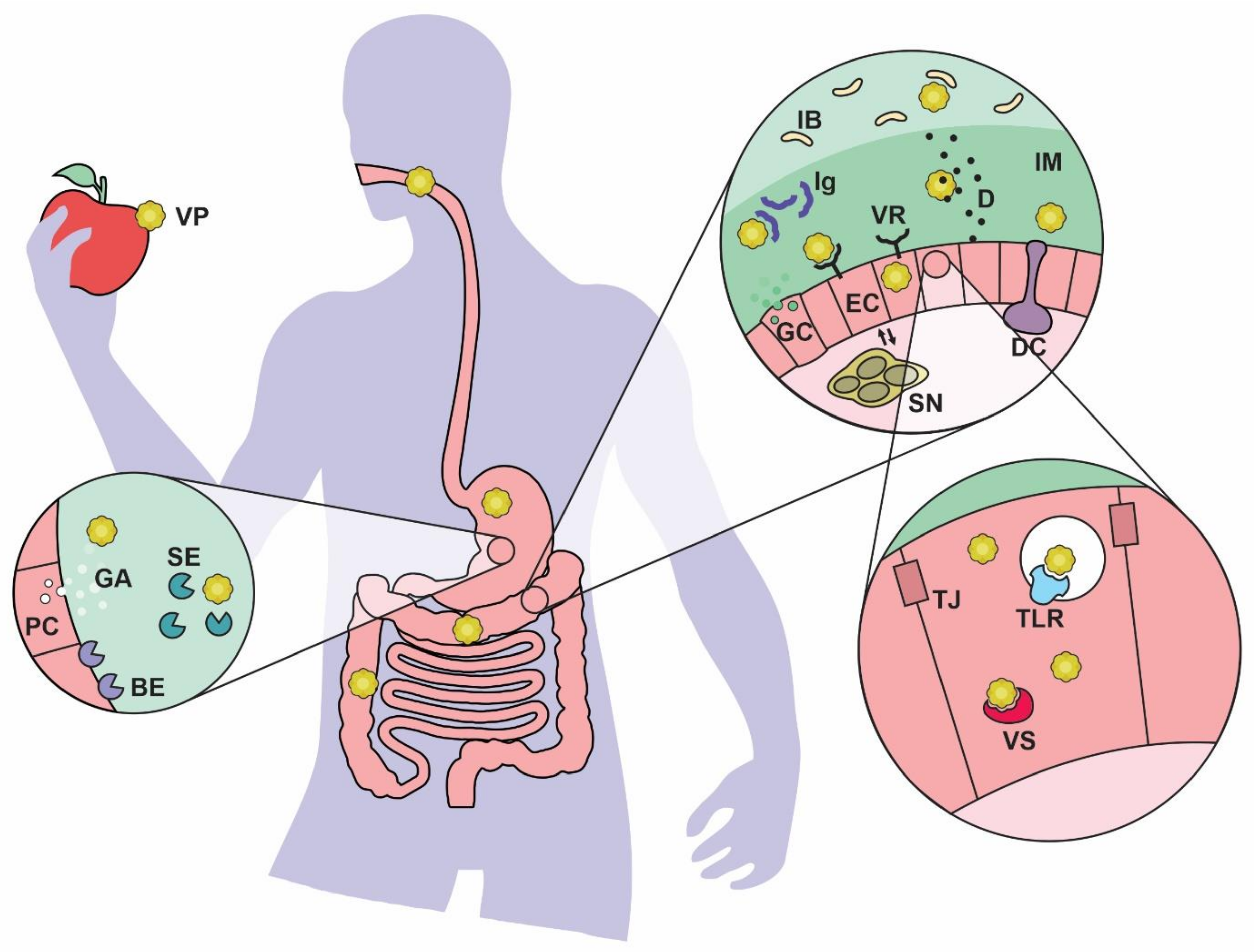
3. The Enteric Virus Road
4. General Effects of Enteric Viruses in Gut Health
5. Mucosal Barrier Changes under Viral Infection
6. Interactions between Gut Bacteria and Potentially Pathogenic Viruses
7. Malnutrition and Gut Viral Infections
8. Enteric Neurons and Mucosal Barrier under Viral Attack
9. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shkoporov, A.N.; Clooney, A.G.; Sutton, T.D.; Ryan, F.J.; Daly, K.M.; Nolan, J.A.; McDonnell, S.A.; Khokhlova, E.V.; Draper, L.A.; Forde, A.; et al. The human gut virome is highly diverse, stable, and individual specific. Cell Host Microbe 2019, 26, 527–541. [Google Scholar] [CrossRef] [PubMed]
- Carding, S.R.; Davis, N.; Hoyles, L. Review article: The human intestinal virome in health and disease. Aliment. Pharmacol. Ther. 2017, 46, 800–815. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.S.; Zhou, Y.; Zhao, G.; Bauer, I.K.; Droit, L.; Ndao, I.M.; Warner, B.B.; Tarr, P.I.; Wang, D.; Holtz, L.R. Early life dynamics of the human gut virome and bacterial microbiome in infants. Nat. Med. 2015, 21, 1228–1234. [Google Scholar] [CrossRef] [PubMed]
- Reyes, A.; Haynes, M.; Hanson, N.; Angly, F.E.; Heath, A.C.; Rohwer, F.; Gordon, J.I. Viruses in the faecal microbiota of monozygotic twins and their mothers. Nat. Cell Biol. 2010, 466, 334–338. [Google Scholar] [CrossRef]
- Maqsood, R.; Rodgers, R.; Rodriguez, C.; Handley, S.A.; Ndao, I.M.; Tarr, P.I.; Warner, B.B.; Lim, E.S.; Holtz, L.R. Discordant transmission of bacteria and viruses from mothers to babies at birth. Microbiome 2019, 7, 156. [Google Scholar] [CrossRef]
- Virgin, H.W. The virome in mammalian physiology and disease. Cell 2014, 157, 142–150. [Google Scholar] [CrossRef]
- Lun, J.H.; Crosbie, N.D.; White, P.A. Genetic diversity and quantification of human mastadenoviruses in wastewater from Sydney and Melbourne, Australia. Sci. Total. Environ. 2019, 675, 305–312. [Google Scholar] [CrossRef]
- Brassard, J.; Gagné, M.-J.; Leblanc, D.; Poitras, Élyse; Houde, A.; Boras, V.F.; Inglis, G.D. Association of age and gender with Torque teno virus detection in stools from diarrheic and non-diarrheic people. J. Clin. Virol. 2015, 72, 55–59. [Google Scholar] [CrossRef]
- Reuter, G.; Pankovics, P.; Boros, A. Nonsuppurative (Aseptic) meningoencephalomyelitis associated with neurovirulent astro-virus infections in humans and animals. Clin. Microbiol. Rev. 2018, 31. [Google Scholar] [CrossRef]
- de Graaf, M.; van Beek, J.; Koopmans, M.P. Human norovirus transmission and evolution in a changing world. Nat. Rev. Micro-biol. 2016, 14, 421–433. [Google Scholar] [CrossRef]
- Oka, T.; Wang, Q.; Katayama, K.; Saif, L.J. Comprehensive review of human sapoviruses. Clin. Microbiol. Rev. 2015, 28, 32–53. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Kapoor, A.; Slikas, B.; Bamidele, O.S.; Wang, C.; Shaukat, S.; Alam Masroor, M.; Wilson, M.L.; Ndjango, J.-B.N.; Peeters, M.; et al. Multiple diverse circoviruses infect farm animals and are commonly found in human and chimpanzee feces. J. Virol. 2009, 84, 1674–1682. [Google Scholar] [CrossRef]
- Lodha, A.; De Silva, N.; Petric, M.; Moore, A.M. Human torovirus: A new virus associated with neonatal necrotizing enterocolitis. Acta Paediatr. 2007, 94, 1085–1088. [Google Scholar] [CrossRef] [PubMed]
- De, R.; Liu, L.; Qian, Y.; Zhu, R.; Deng, J.; Wang, F.; Sun, Y.; Dong, H.; Jia, L.; Zhao, L. Risk of acute gastroenteritis associated with human bocavirus infection in children: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0184833. [Google Scholar] [CrossRef] [PubMed]
- Väisänen, E.; Mohanraj, U.; Kinnunen, P.M.; Jokelainen, P.; Al, E.V.E.; Barakat, A.M.; Sadeghi, M.; Jalilian, F.A.; Majlesi, A.; Masika, M.; et al. Global distribution of human protoparvoviruses. Emerg. Infect. Dis. 2018, 24, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, B.; Bányai, K.; Martella, V.; Jakab, F.; Masachessi, G.; Kobayashi, N. Picobirnavirus infections: Viral persistence and zoonotic potential. Rev. Med. Virol. 2012, 22, 245–256. [Google Scholar] [CrossRef]
- Berlin, L.E.; Rorabaugh, M.L.; Heidrich, F.; Roberts, K.; Doran, T.; Modlin, J.F. Aseptic meningitis in infants <2 years of age: Diagnosis and etiology. J. Infect. Dis 1993, 168, 888–892. [Google Scholar] [CrossRef]
- Olijve, L.; Jennings, L.; Walls, T. Human parechovirus: An increasingly recognized cause of sepsis-like illness in young infants. Clin. Microbiol. Rev. 2017, 31, e00047–17. [Google Scholar] [CrossRef]
- Tan, S.Z.K.; Tan, M.Z.Y.; Prabakaran, M. Saffold virus, an emerging human cardiovirus. Rev. Med. Virol. 2017, 27, e1908. [Google Scholar] [CrossRef] [PubMed]
- Reuter, G.; Pankovics, P.; Boros, Á. Saliviruses—The first knowledge about a newly discovered human picornavirus. Rev. Med. Virol. 2016, 27, e1904. [Google Scholar] [CrossRef]
- Li, K.; Guo, J.; Zhao, R.; Xue, Y.; Chen, L.; Yang, J.; Peng, J.; Jin, Q. Prevalence of 10 human polyomaviruses in fecal samples from children with acute gastroenteritis: A case-control study. J. Clin. Microbiol. 2013, 51, 3107–3109. [Google Scholar] [CrossRef] [PubMed]
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Rotavirus infection. Nat. Rev. Dis. Primers 2017, 3, 17083. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, J.K. Innate host barriers to viral trafficking and population diversity: Lessons learned from poliovirus. Adv. Virus Res. 2010, 77, 85–118. [Google Scholar]
- Michelangeli, F.; Ruiz, M.C. I, 2. Physiology and pathophysiology of the gut in relation to viral diarrhea. Perspect. Med. Virol. 2003, 9, 23–50. [Google Scholar] [CrossRef] [PubMed]
- Kotwal, G.; Cannon, J.L. Environmental persistence and transfer of enteric viruses. Curr. Opin. Virol. 2014, 4, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Amerongen, H.M.; Wilson, G.A.; Fields, B.N.; Neutra, M.R. Proteolytic processing of reovirus is required for adherence to intestinal M cells. J. Virol. 1994, 68, 8428–8432. [Google Scholar] [CrossRef]
- Johansson, M.E.V.; Sjövall, H.; Hansson, G.C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 352–361. [Google Scholar] [CrossRef]
- Wilson, S.S.; Wiens, M.E.; Smith, J.G. Antiviral mechanisms of human defensins. J. Mol. Biol. 2013, 425, 4965–4980. [Google Scholar] [CrossRef]
- Ochoa, T.J.; Cleary, T.G. Effect of lactoferrin on enteric pathogens. Biochimie 2009, 91, 30–34. [Google Scholar] [CrossRef]
- Wells, A.I.; Coyne, C.B. Enteroviruses: A gut-wrenching game of entry, detection, and evasion. Viruses 2019, 11, 460. [Google Scholar] [CrossRef]
- Davis, I.C.; Owen, R.L. The immunopathology of M cells. Springer Semin. Immunopathol. 1997, 18, 421–448. [Google Scholar] [CrossRef] [PubMed]
- Sharif, M.; Baek, Y.-B.; Naveed, A.; Stalin, N.; Kang, M.-I.; Park, S.-I.; Soliman, M.; Cho, K.-O. Porcine sapovirus-induced tight junction dissociation via activation of RhoA/ROCK/MLC signaling pathway. J. Virol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kuss, S.K.; Etheredge, C.A.; Pfeiffer, J.K. Multiple host barriers restrict poliovirus trafficking in mice. PLoS Pathog. 2008, 4, e1000082. [Google Scholar] [CrossRef] [PubMed]
- Cui, T.; Theuns, S.; Xie, J.; Nauwynck, H.J. Porcine rotavirus mainly infects primary porcine enterocytes at the basolateral surface. Veter. Res. 2019, 50, 110. [Google Scholar] [CrossRef] [PubMed]
- Bird, S.W.; Maynard, N.D.; Covert, M.W.; Kirkegaard, K. Nonlytic viral spread enhanced by autophagy components. Proc. Natl. Acad. Sci. USA 2014, 111, 13081–13086. [Google Scholar] [CrossRef]
- Segrist, E.; Cherry, S. Using diverse model systems to define intestinal epithelial defenses to enteric viral infections. Cell Host Microbe 2020, 27, 329–344. [Google Scholar] [CrossRef]
- Sun, L.; Miyoshi, H.; Origanti, S.; Nice, T.J.; Barger, A.C.; Manieri, N.A.; Fogel, L.A.; French, A.R.; Piwnica-Worms, D.; Piwnica-Worms, H.; et al. Type I interferons link viral infection to enhanced epithelial turnover and repair. Cell Host Microbe 2015, 17, 85–97. [Google Scholar] [CrossRef]
- Hartman, S.; Brown, E.; Loomis, E.; Russell, H.A. Gastroenteritis in children. Am. Fam. Physician 2019, 99, 159–165. [Google Scholar]
- Pires, S.M.; Fischer-Walker, C.L.; Lanata, C.F.; Devleesschauwer, B.; Hall, A.J.; Kirk, M.D.; Duarte, A.S.R.; Black, R.E.; Angulo, F.J. Aetiology-specific estimates of the global and regional incidence and mortality of diarrhoeal diseases commonly transmitted through food. PLoS ONE 2015, 10, e0142927. [Google Scholar] [CrossRef]
- Kernbauer, E.; Ding, Y.; Cadwell, K. An enteric virus can replace the beneficial function of commensal bacteria. Nat. Cell Biol. 2014, 516, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Kim, M.S.; Kim, E.; Cheon, J.H.; Lee, Y.S.; Kim, Y.; Lee, S.H.; Seo, S.U.; Shin, S.H.; Choi, S.S.; et al. Enteric viruses ameliorate gut inflammation via toll-like receptor 3 and toll-like receptor 7-mediated interferon-beta production. Immunity 2016, 44, 889–900. [Google Scholar] [CrossRef]
- Moyano-Porcile, V.; Olavarría-Ramírez, L.; González-Arancibia, C.; Bravo, J.A.; Julio-Pieper, M. Short-term effects of Poly(I:C) on gut permeability. Pharmacol. Res. 2015, 101, 130–136. [Google Scholar] [CrossRef]
- Homayun, B.; Lin, X.; Choi, H.-J. Challenges and recent progress in oral drug delivery systems for biopharmaceuticals. Pharmaceutics 2019, 11, 129. [Google Scholar] [CrossRef]
- Sen, A.; Namsa, N.D.; Feng, N.; Greenberg, H.B. Rotavirus reprograms multiple interferon receptors and restricts their intestinal antiviral and inflammatory functions. J. Virol. 2020, 94. [Google Scholar] [CrossRef]
- Kaufman, S.S.; Chatterjee, N.K.; Fuschino, M.E.; Morse, D.L.; Morotti, R.A.; Magid, M.S.; Gondolesi, G.E.; Florman, S.S.; Fishbein, T.M. Characteristics of human calicivirus enteritis in intestinal transplant recipients. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 328–333. [Google Scholar] [CrossRef]
- Troeger, H.; Loddenkemper, C.; Schneider, T.; Schreier, E.; Epple, H.-J.; Zeitz, M.; Fromm, M.; Schulzke, J.-D.; Troeger, H.; Loddenkemper, C.; et al. Structural and functional changes of the duodenum in human norovirus infection. Gut 2008, 58, 1070–1077. [Google Scholar] [CrossRef] [PubMed]
- Cortez, V.; Boyd, D.F.; Crawford, J.C.; Sharp, B.; Livingston, B.; Rowe, H.M.; Davis, A.; Alsallaq, R.; Robinson, C.G.; Vogel, P.; et al. Astrovirus infects actively secreting goblet cells and alters the gut mucus barrier. Nat. Commun. 2020, 11, 2097. [Google Scholar] [CrossRef] [PubMed]
- Deitz, S.B.; Dodd, D.A.; Cooper, S.; Parham, P.; Kirkegaard, K. MHC I-dependent antigen presentation is inhibited by poliovirus protein 3A. Proc. Natl. Acad. Sci. USA 2000, 97, 13790–13795. [Google Scholar] [CrossRef] [PubMed]
- Madan, V.; Sánchez-Martínez, S.; Vedovato, N.; Rispoli, G.; Carrasco, L.; Nieva, J.L. Plasma membrane-porating domain in poliovirus 2B protein. A short peptide mimics viroporin activity. J. Mol. Biol. 2007, 374, 951–964. [Google Scholar] [CrossRef]
- Pickard, J.M.; Zeng, M.Y.; Caruso, R.; Núñez, G. Gut microbiota: Role in pathogen colonization, immune responses, and inflammatory disease. Immunol. Rev. 2017, 279, 70–89. [Google Scholar] [CrossRef]
- Karst, S.M. The influence of commensal bacteria on infection with enteric viruses. Nat. Rev. Genet. 2016, 14, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Neu, U.; Mainou, B.A. Virus interactions with bacteria: Partners in the infectious dance. PLoS Pathog. 2020, 16, e1008234. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Ma, W.-T.; Pang, M.; Fan, Q.-L.; Hua, J.-L. The commensal microbiota and viral infection: A comprehensive review. Front. Immunol. 2019, 10, 1551. [Google Scholar] [CrossRef]
- Mori, K.; Konishi, N.; Suzuki, Y.; Harada, S.; Maeda, M.; Akase, S.; Obata, H.; Monma, C.; Nagano, M.; Kimoto, K.; et al. Comparison between patients with norovirus-related gastroenteritis and asymptomatic carriers with respect to distribu-tion of antibody-complexed viral particles and intestinal flora. J. Med. Virol. 2018, 90, 1882–1887. [Google Scholar] [CrossRef]
- Nagata, S.; Asahara, T.; Ohta, T.; Yamada, T.; Kondo, S.; Bian, L.; Wang, C.; Yamashiro, Y.; Nomoto, K. Effect of the continuous intake of probiotic-fermented milk containing Lactobacillus casei strain Shirota on fever in a mass outbreak of norovirus gastroenteritis and the faecal microflora in a health service facility for the aged. Br. J. Nutr. 2011, 106, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Nagata, S.; Asahara, T.; Wang, C.; Suyama, Y.; Chonan, O.; Takano, K.; Daibou, M.; Takahashi, T.; Nomoto, K.; Yamashiro, Y. The effectiveness of lactobacillus beverages in controlling infections among the residents of an aged care facility: A randomized placebo-controlled double-blind trial. Ann. Nutr. Metab. 2015, 68, 51–59. [Google Scholar] [CrossRef]
- Mathew, S.; Smatti, M.K.; Al Ansari, K.; Nasrallah, G.K.; Al Thani, A.A.; Yassine, H.M. Mixed viral-bacterial infections and their effects on gut microbiota and clinical illnesses in children. Sci. Rep. 2019, 9, 865. [Google Scholar] [CrossRef]
- Giuffrè, M.; Campigotto, M.; Campisciano, G.; Comar, M.; Crocè, L.S. A story of liver and gut microbes: How does the intestinal flora affect liver disease? A review of the literature. Am. J. Physiol. Liver Physiol. 2020, 318, G889–G906. [Google Scholar] [CrossRef]
- Nelson, A.M.; Walk, S.T.; Taube, S.; Taniuchi, M.; Houpt, E.R.; Wobus, C.E.; Young, V.B. Disruption of the human gut microbiota following norovirus infection. PLoS ONE 2012, 7, e48224. [Google Scholar] [CrossRef]
- Almand, E.A.; Moore, M.D.; Outlaw, J.; Jaykus, L.-A. Human norovirus binding to select bacteria representative of the human gut microbiota. PLoS ONE 2017, 12, e0173124. [Google Scholar] [CrossRef] [PubMed]
- Rowe, H.M.; Meliopoulos, V.A.; Iverson, A.; Bomme, P.; Schultz-Cherry, S.; Rosch, J.W. Direct interactions with influenza promote bacterial adherence during respiratory infections. Nat. Microbiol. 2019, 4, 1328–1336. [Google Scholar] [CrossRef]
- Scrimshaw, N.S.; Taylor, E.C.; Gordon, E.J. Interactions of Nutrition and Infection; World Health Organization: Geneva, Switzerland, 1968; Volume 57, pp. 3–329. [Google Scholar]
- Solomons, N.W. Malnutrition and infection: An update. Br. J. Nutr. 2007, 98, S5–S10. [Google Scholar] [CrossRef][Green Version]
- UNICEF; WHO. Levels and Trends in Child Mortality, Report 2011; United Nations International Children’s Fund: New York, NY, USA, 2011. [Google Scholar]
- Tickell, K.D.; Sharmin, R.; Deichsel, E.L.; Lamberti, L.M.; Walson, J.L.; Faruque, A.S.G.; Pavlinac, P.B.; Kotloff, K.L.; Chisti, M.J. The effect of acute malnutrition on enteric pathogens, moderate-to-severe diarrhoea, and associated mortality in the Global Enteric Multicenter Study cohort: A post-hoc analysis. Lancet Glob. Health 2020, 8, e215–e224. [Google Scholar] [CrossRef]
- Parker, E.P.; Ramani, S.; Lopman, B.A.; Church, J.A.; Iturriza-Gómara, M.; Prendergast, A.J.; Grassly, N.C. Causes of impaired oral vaccine efficacy in developing countries. Futur. Microbiol. 2018, 13, 97–118. [Google Scholar] [CrossRef]
- Bucak, I.H.; Ozturk, A.B.; Almis, H.; Cevik, M.Ö.; Tekin, M.; Konca, Ç.; Turgut, M.; Bulbul, M. Is there a relationship between low vitamin D and rotaviral diarrhea? Pediatr. Int. 2016, 58, 270–273. [Google Scholar] [CrossRef]
- Zhao, Y.; Yu, B.; Mao, X.; He, J.; Huang, Z.; Zheng, P.; Yu, J.; Han, G.; Liang, X.; Chen, D. Effect of 25-hydroxyvitamin D3on rotavirus replication and gene expressions of RIG-I signalling molecule in porcine rotavirus–infected IPEC-J2 cells. Arch. Anim. Nutr. 2015, 69, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Broquet, A.H.; Hirata, Y.; McAllister, C.S.; Kagnoff, M.F. RIG-I/MDA5/MAVS are required to signal a protective IFN response in rotavirus-infected intestinal epithelium. J. Immunol. 2010, 186, 1618–1626. [Google Scholar] [CrossRef] [PubMed]
- Afolabi, O.F.; Saka, A.O.; Ojuawo, A.; Biliaminu, S.A. Serum zinc levels of hospitalized children with acute diarrhea differ by the isolated viruses. Int. J. Health Sci. 2019, 13, 4–10. [Google Scholar]
- Das, S.K.; Chisti, M.J.; Huq, S.; Malek, M.A.; Vanderlee, L.; Kaur, G.; Salam, M.A.; Ahmed, T.; Faruque, A.S.G.; Al Mamun, A. Clinical characteristics, etiology and antimicrobial susceptibility among overweight and obese individuals with diarrhea: Observed at a large diarrheal disease hospital, Bangladesh. PLoS ONE 2013, 8, e70402. [Google Scholar] [CrossRef]
- Salvatoni, A.; Baj, A.; Bianchi, G.; Federico, G.; Colombo, M.; Toniolo, A. Intrafamilial spread of enterovirus infections at the clinical onset of type 1 diabetes. Pediatr. Diabetes 2013, 14, 407–416. [Google Scholar] [CrossRef] [PubMed]
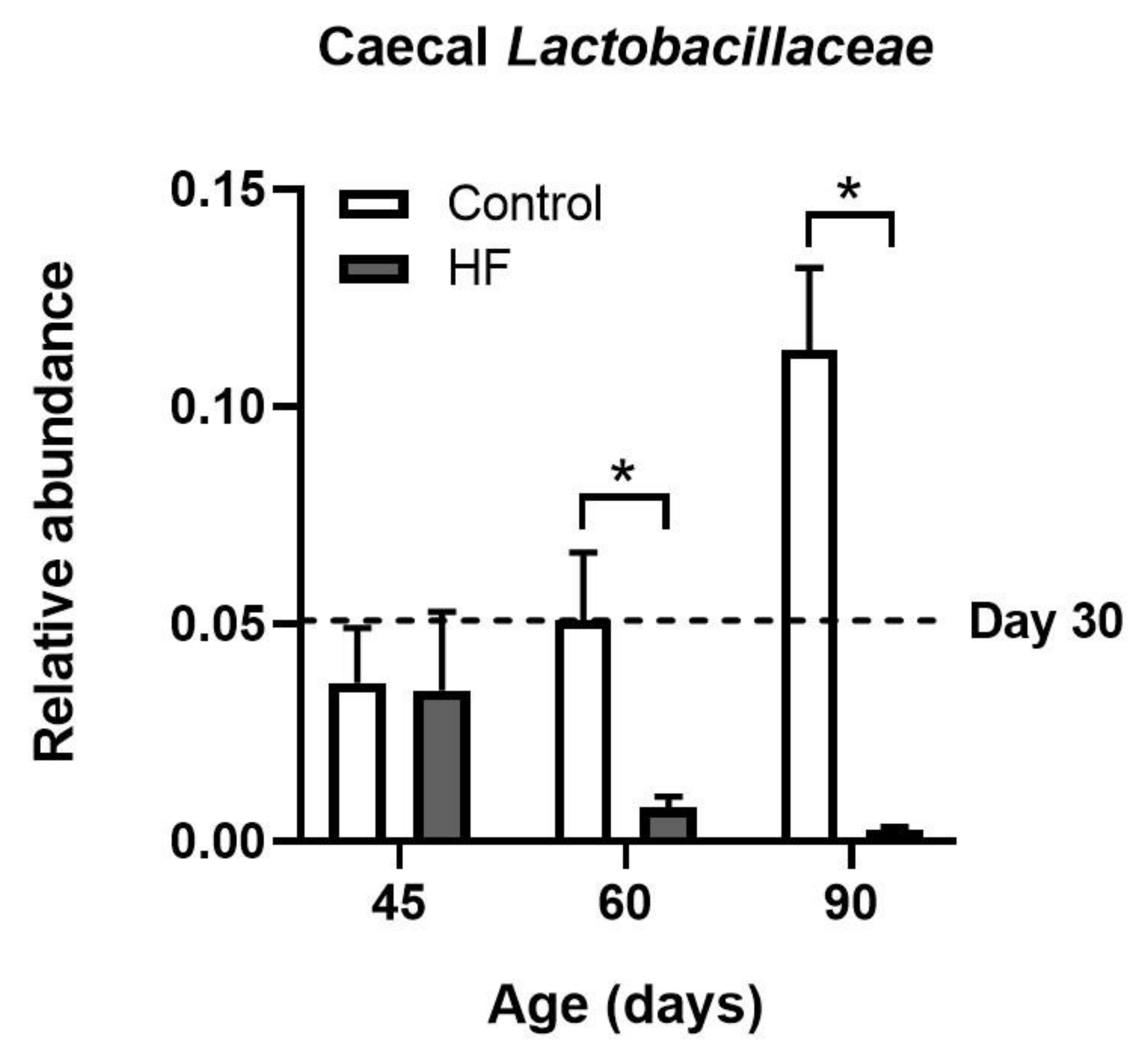
- Singh, R.K.; Chang, H.-W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Farahnik, B.; Nakamura, M.; Zhu, T.H.; et al. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef] [PubMed]
- LeComte, V.; Kaakoush, N.O.; Maloney, C.A.; Raipuria, M.; Huinao, K.D.; Mitchell, H.M.; Morris, M.J. Changes in gut microbiota in rats fed a high fat diet correlate with obesity-associated metabolic parameters. PLoS ONE 2015, 10, e0126931. [Google Scholar] [CrossRef] [PubMed]
- Maragkoudakis, P.A.; Chingwaru, W.; Gradisnik, L.; Tsakalidou, E.; Cencic, A. Lactic acid bacteria efficiently protect human and animal intestinal epithelial and immune cells from enteric virus infection. Int. J. Food Microbiol. 2010, 141, S91–S97. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Wu, X.; Nawaz, M.; Li, J.; Yu, P.; Moore, J.E.; Xu, J. Molecular characterization of fecal microbiota in patients with viral diarrhea. Curr. Microbiol. 2011, 63, 259–266. [Google Scholar] [CrossRef]
- Vergnolle, N.; Cirillo, C. Neurons and glia in the enteric nervous system and epithelial barrier function. Physiology 2018, 33, 269–280. [Google Scholar] [CrossRef]
- Spencer, N.J.; Hu, H. Enteric nervous system: Sensory transduction, neural circuits and gastrointestinal motility. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 338–351. [Google Scholar] [CrossRef]
- Lundgren, O. Role of the enteric nervous system in the fluid and electrolyte secretion of rotavirus diarrhea. Science 2000, 287, 491–495. [Google Scholar] [CrossRef]
- Holland-Cunz, S.; Göppl, M.; Rauch, U.; Bär, C.; Klotz, M.; Schäfer, K.-H. Acquired intestinal aganglionosis after a lytic infection with varicella-zoster virus. J. Pediatr. Surg. 2006, 41, e29–e31. [Google Scholar] [CrossRef]
- Gershon, A.A.; Chen, J.; Gershon, M.D. Use of saliva to identify varicella zoster virus infection of the gut. Clin. Infect. Dis. 2015, 61, 536–544. [Google Scholar] [CrossRef]
- Gesser, R.M.; Koo, S.C. Oral inoculation with herpes simplex virus type 1 infects enteric neuron and mucosal nerve fibers within the gastrointestinal tract in mice. J. Virol. 1996, 70, 4097–4102. [Google Scholar] [CrossRef] [PubMed]
- Giuffrè, M.; Moretti, R.; Campisciano, G.; Da Silveira, A.B.M.; Monda, V.M.; Comar, M.; Di Bella, S.; Antonello, R.M.; Luzzati, R.; Crocè, L.S. You talking to me? Says the Enteric Nervous System (ENS) to the microbe. How intestinal microbes interact with the ENS. J. Clin. Med. 2020, 9, 3705. [Google Scholar] [CrossRef] [PubMed]
- Fenrich, M.; Mrdenovic, S.; Balog, M.; Tomic, S.; Zjalic, M.; Roncevic, A.; Mandic, D.; Debeljak, Z.; Heffer, M. SARS-CoV-2 dissemination through peripheral nerves explains multiple organ injury. Front. Cell. Neurosci. 2020, 14, 229. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Julio-Pieper, M.; López-Aguilera, A.; Eyzaguirre-Velásquez, J.; Olavarría-Ramírez, L.; Ibacache-Quiroga, C.; Bravo, J.A.; Cruz, G. Gut Susceptibility to Viral Invasion: Contributing Roles of Diet, Microbiota and Enteric Nervous System to Mucosal Barrier Preservation. Int. J. Mol. Sci. 2021, 22, 4734. https://doi.org/10.3390/ijms22094734
Julio-Pieper M, López-Aguilera A, Eyzaguirre-Velásquez J, Olavarría-Ramírez L, Ibacache-Quiroga C, Bravo JA, Cruz G. Gut Susceptibility to Viral Invasion: Contributing Roles of Diet, Microbiota and Enteric Nervous System to Mucosal Barrier Preservation. International Journal of Molecular Sciences. 2021; 22(9):4734. https://doi.org/10.3390/ijms22094734
Chicago/Turabian StyleJulio-Pieper, Marcela, Alejandra López-Aguilera, Johana Eyzaguirre-Velásquez, Loreto Olavarría-Ramírez, Claudia Ibacache-Quiroga, Javier A. Bravo, and Gonzalo Cruz. 2021. "Gut Susceptibility to Viral Invasion: Contributing Roles of Diet, Microbiota and Enteric Nervous System to Mucosal Barrier Preservation" International Journal of Molecular Sciences 22, no. 9: 4734. https://doi.org/10.3390/ijms22094734
APA StyleJulio-Pieper, M., López-Aguilera, A., Eyzaguirre-Velásquez, J., Olavarría-Ramírez, L., Ibacache-Quiroga, C., Bravo, J. A., & Cruz, G. (2021). Gut Susceptibility to Viral Invasion: Contributing Roles of Diet, Microbiota and Enteric Nervous System to Mucosal Barrier Preservation. International Journal of Molecular Sciences, 22(9), 4734. https://doi.org/10.3390/ijms22094734






