Melanin Transfer in the Epidermis: The Pursuit of Skin Pigmentation Control Mechanisms
Abstract
1. Introduction
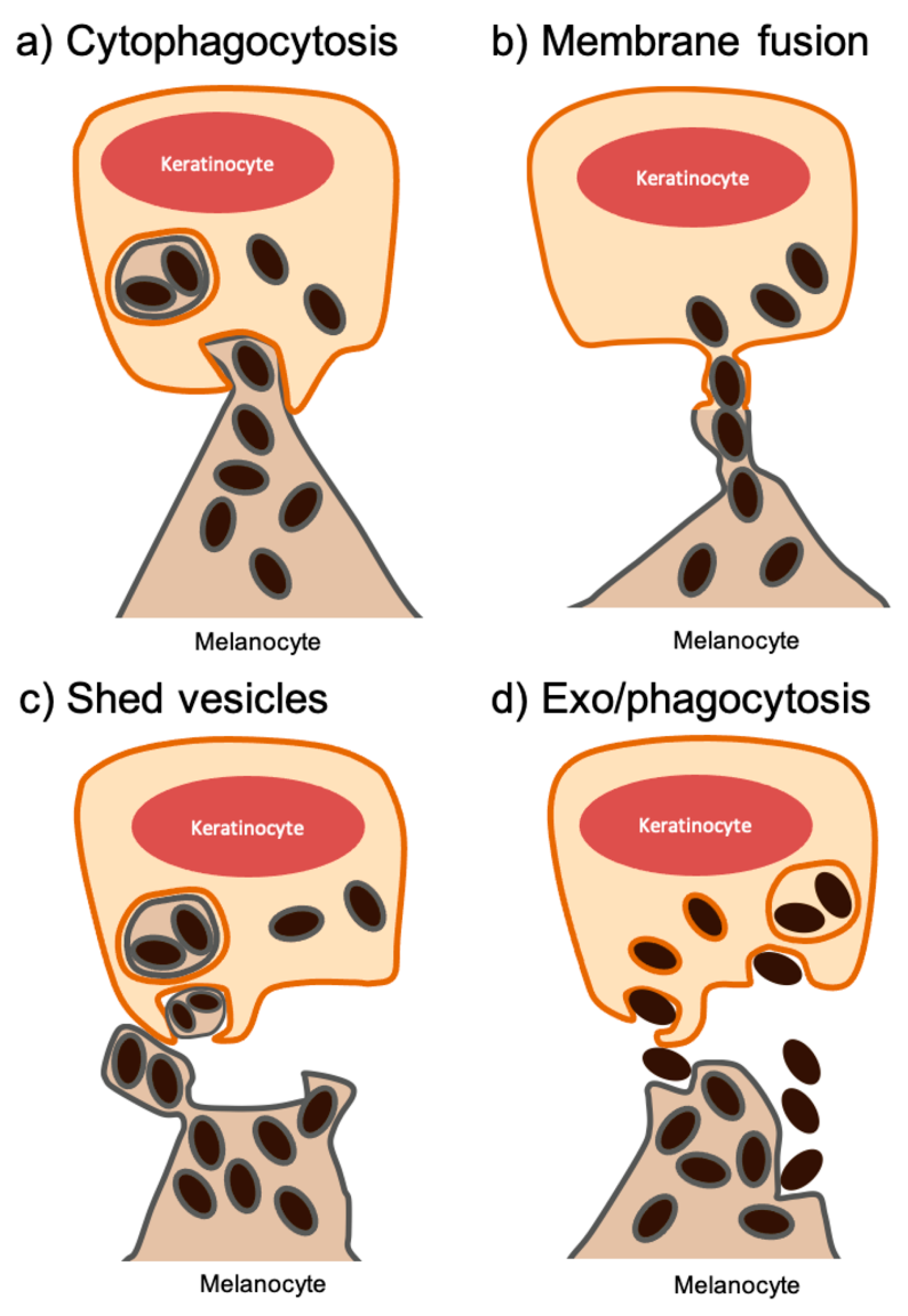
2. Melanin Transfer between Melanocytes and Keratinocytes
2.1. Cytophagocytosis of Melanocyte Dendrite Tips by Basal Keratinocytes
2.2. Membrane Fusion of Melanocyte and Keratinocyte Membranes
2.3. Transfer of Shed Melanosome-Loaded Vesicles
2.4. Coupled Exocytosis of the Melanin Core by Melanocytes and Phagocytosis by Keratinocytes
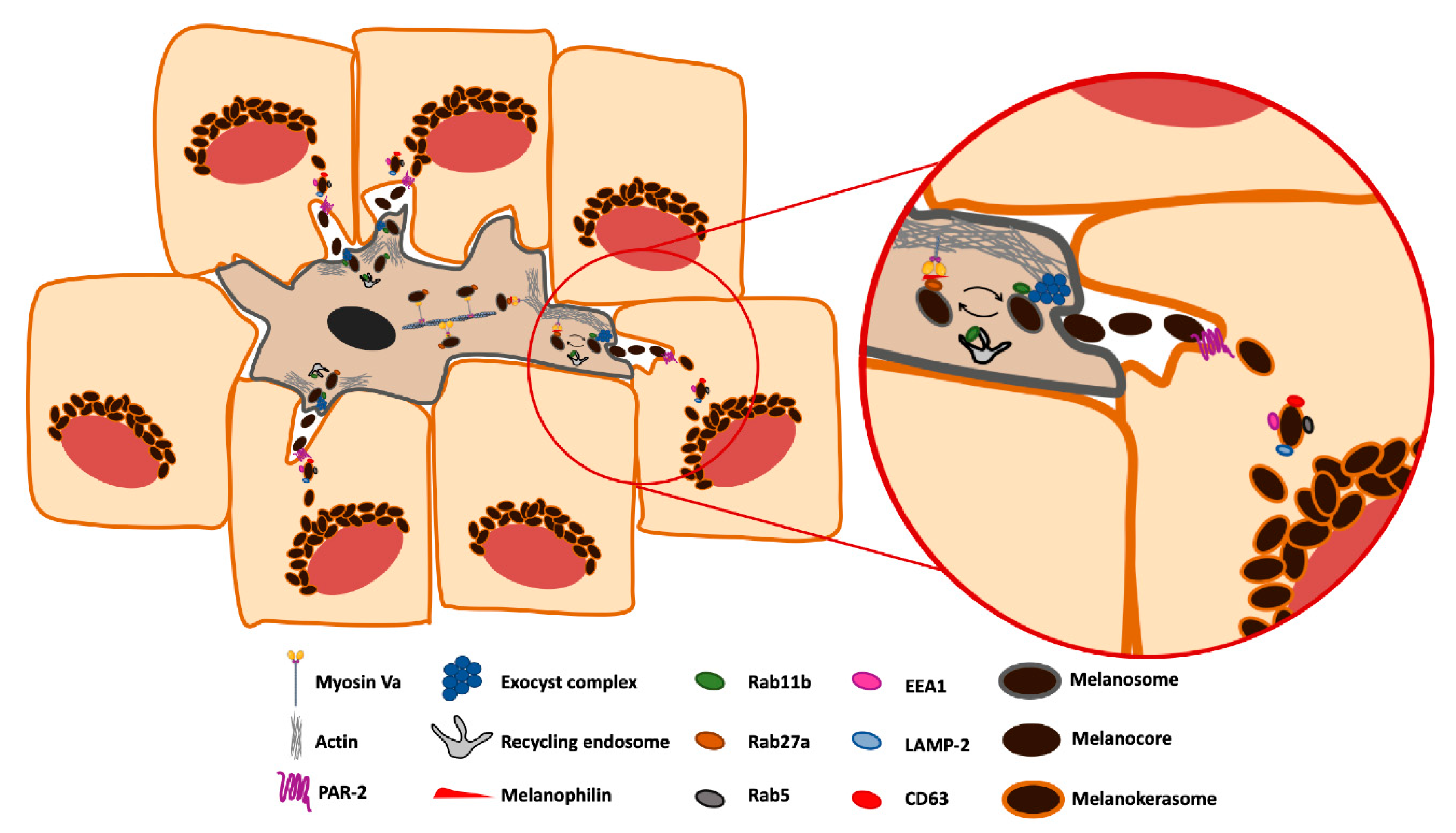
3. Melanin Secretion from Melanocytes
4. Melanin Uptake by Keratinocytes
5. Melanin Processing within Keratinocytes
6. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Lai-Cheong, J.E.; McGrath, J.A. Structure and Function of Skin, Hair and Nails. Medicine 2013, 41, 317–320. [Google Scholar] [CrossRef]
- Nicol, N.H. Anatomy and Physiology of the Skin. Dermatol. Nurs. Dermatol. Nurses Assoc. 2005, 17, 62. [Google Scholar] [CrossRef]
- Cichorek, M.; Wachulska, M.; Stasiewicz, A.; Tymińska, A. Skin Melanocytes: Biology and Development. Postepy Dermatol. I Alergol. 2013, 30, 30–41. [Google Scholar] [CrossRef]
- Cichorek, M.; Wachulska, M.; Stasiewicz, A. Heterogeneity of Neural Crest-Derived Melanocytes. Cent. Eur. J. Biol. 2013, 8, 315–330. [Google Scholar] [CrossRef]
- Lapedriza, A.; Petratou, K.; Kelsh, R.N. Neural Crest Cells and Pigmentation. Neural Crest Cells Evol. Dev. Dis. 2014, 287–311. [Google Scholar] [CrossRef]
- Grice, E.A.; Segre, J.A. The Skin Microbiome. Nat. Rev. Microbiol. 2011, 9, 244–253. [Google Scholar] [CrossRef]
- Solanas, G.; Benitah, S.A. Regenerating the Skin: A Task for the Heterogeneous Stem Cell Pool and Surrounding Niche. Nat. Rev. Mol. Cell Biol. 2013, 14, 737–748. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B.; Breathnach, A.S. [The Epidermal Melanin Unit System]. Dermatol. Wochenschr. 1963, 147, 481–489. [Google Scholar]
- Frenk, E.; Schellhorn, J.P. [Morphology of the Epidermal Melanin Unit]. Dermatologica 1969, 139, 271–277. [Google Scholar] [CrossRef]
- Del Bino, S.; Duval, C.; Bernerd, F. Clinical and Biological Characterization of Skin Pigmentation Diversity and Its Consequences on UV Impact. Int. J. Mol. Sci. 2018, 19, 2668. [Google Scholar] [CrossRef]
- Cox, N.H. Fitzpatrick’s Dermatology in General Medicine. Br. J. Dermatol. 2004, 150, 794. [Google Scholar] [CrossRef]
- Ito, S.; Wakamatsu, K. Quantitative Analysis of Eumelanin and Pheomelanin in Humans, Mice, and Other Animals: A Comparative Review. Pigment Cell Res. 2003, 16, 523–531. [Google Scholar] [CrossRef]
- Ito, S.; Wakamatsu, K.; Sarna, T. Photodegradation of Eumelanin and Pheomelanin and Its Pathophysiological Implications. Photochem. Photobiol. 2018, 94, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Hurbain, I.; Romao, M.; Sextius, P.; Bourreau, E.; Marchal, C.; Bernerd, F.; Duval, C.; Raposo, G. Melanosome Distribution in Keratinocytes in Different Skin Types: Melanosome Clusters Are Not Degradative Organelles. J. Investig. Dermatol. 2018, 138, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, G.; Bolasco, G.; Aspite, N.; Lucania, G.; Lotti, L.V.; Torrisi, M.R.; Picardo, M. Melanosome Transfer Promoted by Keratinocyte Growth Factor in Light and Dark Skin-Derived Keratinocytes. J. Investig. Dermatol. 2008, 128, 558–567. [Google Scholar] [CrossRef]
- Thong, H.-Y.; Jee, S.-H.; Sun, C.-C.; Boissy, R.E. The Patterns of Melanosome Distribution in Keratinocytes of Human Skin as One Determining Factor of Skin Colour. Br. J. Dermatol. 2003, 149, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Hachiya, A.; Sriwiriyanont, P.; Ohuchi, A.; Kitahara, T.; Takema, Y.; Visscher, M.O.; Boissy, R.E. Functional Analysis of Keratinocytes in Skin Color Using a Human Skin Substitute Model Composed of Cells Derived from Different Skin Pigmentation Types. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2007, 21, 2829–2839. [Google Scholar] [CrossRef]
- Raposo, G.; Marks, M.S. The Dark Side of Lysosome-Related Organelles: Specialization of the Endocytic Pathway for Melanosome Biogenesis. Traffic (Cph. Den.) 2002, 3, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Marks, M.S. Melanosomes—Dark Organelles Enlighten Endosomal Membrane Transport. Nat. Rev. Mol. Cell Biol. 2007, 8, 786–797. [Google Scholar] [CrossRef]
- Delevoye, C.; Marks, M.S.; Raposo, G. Lysosome-Related Organelles as Functional Adaptations of the Endolysosomal System. Curr. Opin. Cell Biol. 2019, 59, 147–158. [Google Scholar] [CrossRef]
- Raposo, G.; Tenza, D.; Murphy, D.M.; Berson, J.F.; Marks, M.S. Distinct Protein Sorting and Localization to Premelanosomes, Melanosomes, and Lysosomes in Pigmented Melanocytic Cells. J. Cell Biol. 2001, 152, 809–823. [Google Scholar] [CrossRef]
- Hurbain, I.; Geerts, W.J.C.; Boudier, T.; Marco, S.; Verkleij, A.J.; Marks, M.S.; Raposo, G. Electron Tomography of Early Melanosomes: Implications for Melanogenesis and the Generation of Fibrillar Amyloid Sheets. Proc. Natl. Acad. Sci. USA 2008, 105, 19726–19731. [Google Scholar] [CrossRef] [PubMed]
- Marks, M.S.; Seabra, M.C. The Melanosome: Membrane Dynamics in Black and White. Nat. Rev. Mol. Cell Biol. 2001, 2, 738–748. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Yaar, M.; Byers, H.R.; Goukassian, D.; Fine, R.E.; Gonsalves, J.; Gilchrest, B.A. Kinesin Participates in Melanosomal Movement along Melanocyte Dendrites. J. Investig. Dermatol. 2000, 114, 438–443. [Google Scholar] [CrossRef]
- Jordens, I.; Westbroek, W.; Marsman, M.; Rocha, N.; Mommaas, M.; Huizing, M.; Lambert, J.; Naeyaert, J.M.; Neefjes, J. Rab7 and Rab27a Control Two Motor Protein Activities Involved in Melanosomal Transport. Pigment Cell Res. 2006, 19, 412–423. [Google Scholar] [CrossRef]
- Hume, A.N.; Ushakov, D.S.; Tarafder, A.K.; Ferenczi, M.A.; Seabra, M.C. Rab27a and MyoVa Are the Primary Mlph Interactors Regulating Melanosome Transport in Melanocytes. J. Cell Sci. 2007, 120, 3111–3122. [Google Scholar] [CrossRef]
- Nagashima, K.; Torii, S.; Yi, Z.; Igarashi, M.; Okamoto, K.; Takeuchi, T.; Izumi, T. Melanophilin Directly Links Rab27a and Myosin Va through Its Distinct Coiled-Coil Regions. FEBS Lett. 2002, 517, 233–238. [Google Scholar] [CrossRef]
- Gross, S.P.; Carolina Tuma, M.; Deacon, S.W.; Serpinskaya, A.S.; Reilein, A.R.; Gelfand, V.I. Interactions and Regulation of Molecular Motors in Xenopus Melanophores. J. Cell Biol. 2002, 156, 855–865. [Google Scholar] [CrossRef]
- Alzahofi, N.; Welz, T.; Robinson, C.L.; Page, E.L.; Briggs, D.A.; Stainthorp, A.K.; Reekes, J.; Elbe, D.A.; Straub, F.; Kallemeijn, W.W.; et al. Rab27a Co-Ordinates Actin-Dependent Transport by Controlling Organelle-Associated Motors and Track Assembly Proteins. Nat. Commun. 2020, 11, 3495. [Google Scholar] [CrossRef]
- Jiang, M.; Paniagua, A.E.; Volland, S.; Wang, H.; Balaji, A.; Li, D.G.; Lopes, V.S.; Burgess, B.L.; Williams, D.S. Microtubule Motor Transport in the Delivery of Melanosomes to the Actin-Rich Apical Domain of the Retinal Pigment Epithelium. J. Cell Sci. 2020, 133. [Google Scholar] [CrossRef] [PubMed]
- Aktary, Z.; Conde-Perez, A.; Rambow, F.; Di Marco, M.; Amblard, F.; Hurbain, I.; Raposo, G.; Delevoye, C.; Coscoy, S.; Larue, L. A Role for Dynlt3 in Melanosome Movement, Distribution, Acidity and Transfer. Commun. Biol. 2021, 4, 423. [Google Scholar] [CrossRef]
- Okazaki, K.; Uzuka, M.; Morikawa, F.; Toda, K.; Seiji, M. Transfer Mechanism of Melanosomes in Epidermal Cell Culture. J. Investig. Dermatol. 1976, 67, 541–547. [Google Scholar] [CrossRef]
- Wolff, K.; Konrad, K. Phagocytosis of Latex Beads by Epidermal Keratinocytes In Vivo. J. Ultrastruct. Res. 1972, 39, 262–280. [Google Scholar] [CrossRef]
- Sharlow, E.R.; Paine, C.S.; Babiarz, L.; Eisinger, M.; Shapiro, S.; Seiberg, M. The Protease-Activated Receptor-2 Upregulates Keratinocyte Phagocytosis. J. Cell Sci. 2000, 113, 3093–3101. [Google Scholar]
- Mottaz, J.H.; Zelickson, A.S. Melanin Transfer: A Possible Phagocytic Process. J. Investig. Dermatol. 1967, 49, 605–610. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Birbeck, M.S.C.; Mercer, E.H.; Barnicot, N.A. The Structure and Formation of Pigment Granules in Human Hair. Exp. Cell Res. 1956, 10, 505–514. [Google Scholar] [CrossRef]
- YAMAMOTO, O.; BHAWAN, J. Three Modes of Melanosome Transfers in Caucasian Facial Skin: Hypothesis Based on an Ultrastructural Study. Pigment Cell Res. 1994, 7, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Scott, G.; Leopardi, S.; Printup, S.; Madden, B.C. Filopodia Are Conduits for Melanosome Transfer to Keratinocytes. J. Cell Sci. 2002, 115, 1441–1451. [Google Scholar] [PubMed]
- Singh, S.K.; Kurfurst, R.; Nizard, C.; Schnebert, S.; Perrier, E.; Tobin, D.J. Melanin Transfer in Human Skin Cells Is Mediated by Filopodia—A Model for Homotypic and Heterotypic Lysosome-Related Organelle Transfer. FASEB J. 2010, 24, 3756–3769. [Google Scholar] [CrossRef]
- Beaumont, K.A.; Hamilton, N.A.; Moores, M.T.; Brown, D.L.; Ohbayashi, N.; Cairncross, O.; Cook, A.L.; Smith, A.G.; Misaki, R.; Fukuda, M.; et al. The Recycling Endosome Protein Rab17 Regulates Melanocytic Filopodia Formation and Melanosome Trafficking. Traffic 2011, 12, 627–643. [Google Scholar] [CrossRef] [PubMed]
- Van Den Bossche, K.; Naeyaert, J.M.; Lambert, J. The Quest for the Mechanism of Melanin Transfer. Traffic 2006, 7, 769–778. [Google Scholar] [CrossRef]
- Ni, J.; Wang, N.; Gao, L.; Li, L.; Zheng, S.; Liu, Y.; Ozukum, M.; Nikiforova, A.; Zhao, G.; Song, Z. The Effect of the NMDA Receptor-Dependent Signaling Pathway on Cell Morphology and Melanosome Transfer in Melanocytes. J. Dermatol. Sci. 2016, 84, 296–304. [Google Scholar] [CrossRef]
- Singh, S.K.; Baker, R.; Sikkink, S.K.; Nizard, C.; Schnebert, S.; Kurfurst, R.; Tobin, D.J. E-Cadherin Mediates Ultraviolet Radiation- and Calcium-Induced Melanin Transfer in Human Skin Cells. Exp. Dermatol. 2017, 26, 1125–1133. [Google Scholar] [CrossRef]
- Tarafder, A.K.; Bolasco, G.; Correia, M.S.; Pereira, F.J.C.; Iannone, L.; Hume, A.N.; Kirkpatrick, N.; Picardo, M.; Torrisi, M.R.; Rodrigues, I.P.; et al. Rab11b Mediates Melanin Transfer between Donor Melanocytes and Acceptor Keratinocytes via Coupled Exo/Endocytosis. J. Investig. Dermatol. 2014, 134, 1056–1066. [Google Scholar] [CrossRef]
- Cerdan, D.; Redziniak, G.; Bourgeois, C.A.; Monsigny, M.; Kieda, C. C32 Human Melanoma Cell Endogenous Lectins: Characterization and Implication in Vesicle-Mediated Melanin Transfer to Keratinocytes. Exp. Cell Res. 1992, 203, 164–173. [Google Scholar] [CrossRef]
- Aspengren, S.; Hedberg, D.; Wallin, M. Studies of Pigment Transfer between Xenopus Laevis Melanophores and Fibroblasts In Vitro and In Vivo. Pigment Cell Res. 2006, 19, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Tadokoro, R.; Murai, H.; Sakai, K.I.; Okui, T.; Yokota, Y.; Takahashi, Y. Melanosome Transfer to Keratinocyte in the Chicken Embryonic Skin Is Mediated by Vesicle Release Associated with Rho-Regulated Membrane Blebbing. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Wäster, P.; Eriksson, I.; Vainikka, L.; Rosdahl, I.; Öllinger, K. Extracellular Vesicles Are Transferred from Melanocytes to Keratinocytes after UVA Irradiation. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef]
- Ando, H.; Niki, Y.; Yoshida, M.; Ito, M.; Akiyama, K.; Kim, J.; Yoon, T.; Matsui, M.S.; Yarosh, D.B.; Ichihashi, M. Involvement of Pigment Globules Containing Multiple Melanosomes in the Transfer of Melanosomes from Melanocytes to Keratinocytes. Cell. Logist. 2011, 1, 12–20. [Google Scholar] [CrossRef]
- Ando, H.; Niki, Y.; Ito, M.; Akiyama, K.; Matsui, M.S.; Yarosh, D.B.; Ichihashi, M. Melanosomes Are Transferred from Melanocytes to Keratinocytes through the Processes of Packaging, Release, Uptake, and Dispersion. J. Investig. Dermatol. 2012, 132, 1222–1229. [Google Scholar] [CrossRef]
- Cardinali, G.; Ceccarelli, S.; Kovacs, D.; Aspite, N.; Lotti, L.V.; Torrisi, M.R.; Picardo, M. Keratinocyte Growth Factor Promotes Melanosome Transfer to Keratinocytes. J. Investig. Dermatol. 2005, 125, 1190–1199. [Google Scholar] [CrossRef]
- Seiberg, M.; Paine, C.; Sharlow, E.; Eisinger, M.; Shapiro, S.S.; Andrade-Gordon, P.; Costanzo, M. Inhibition of Melanosome Transfer Results in Skin Lightening1. J. Investig. Dermatol. 2000, 115, 162–167. [Google Scholar] [CrossRef]
- Seiberg, M.; Paine, C.; Sharlow, E.; Andrade-Gordon, P.; Costanzo, M.; Eisinger, M.; Shapiro, S.S. The Protease-Activated Receptor 2 Regulates Pigmentation via Keratinocyte-Melanocyte Interactions. Exp. Cell Res. 2000, 254, 25–32. [Google Scholar] [CrossRef]
- Correia, M.S.; Moreiras, H.; Pereira, F.J.C.; Neto, M.V.M.V.; Festas, T.C.; Tarafder, A.K.; Ramalho, J.S.; Seabra, M.C.; Barral, D.C. Melanin Transferred to Keratinocytes Resides in Nondegradative Endocytic Compartments. J. Investig. Dermatol. 2018, 138, 637–646. [Google Scholar] [CrossRef]
- SWIFT, J.A. Transfer of Melanin Granules from Melanocytes to the Cortical Cells of Human Hair. Nature 1964, 203, 976–977. [Google Scholar] [CrossRef]
- Virador, V.M.; Muller, J.; Wu, X.; Abdel-Malek, Z.A.; Yu, Z.X.; Ferrans, V.J.; Kobayashi, N.; Wakamatsu, K.; Ito, S.; Hammer, J.A.; et al. Influence of Alpha-Melanocyte-Stimulating Hormone and Ultraviolet Radiation on the Transfer of Melanosomes to Keratinocytes. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2002, 16, 105–107. [Google Scholar]
- Potter, B.; Medenica, M. Ultramicroscopic Phagocytosis of Synthetic Melanin by Epidermal Cells in Vivo. J. Investig. Dermatol. 1968, 51, 300–303. [Google Scholar] [CrossRef][Green Version]
- Moreiras, H.; Pereira, F.J.C.; Neto, M.V.; Bento-Lopes, L.; Festas, T.C.; Seabra, M.C.; Barral, D.C. The Exocyst Is Required for Melanin Exocytosis from Melanocytes and Transfer to Keratinocytes. Pigment Cell Melanoma Res. 2020, 33, 366–371. [Google Scholar] [CrossRef]
- Moreiras, H.; Neto, M.V.; Bento-Lopes, L.; Escrevente, C.; Ramalho, J.S.; Seabra, M.C.; Barral, D.C. Melanocore Uptake by Keratinocytes Occurs through Phagocytosis and Involves Protease-Activated Receptor-2 Activation. bioRxiv 2021. [Google Scholar] [CrossRef]
- Domingues, L.; Hurbain, I.; Gilles-Marsens, F.; Sirés-Campos, J.; André, N.; Dewulf, M.; Romao, M.; Viaris de Lesegno, C.; Macé, A.S.; Blouin, C.; et al. Coupling of Melanocyte Signaling and Mechanics by Caveolae Is Required for Human Skin Pigmentation. Nat. Commun. 2020, 11, 2988. [Google Scholar] [CrossRef]
- Ménager, M.M.; Ménasché, G.; Romao, M.; Knapnougel, P.; Ho, C.-H.; Garfa, M.; Raposo, G.; Feldmann, J.; Fischer, A.; de Saint Basile, G. Secretory Cytotoxic Granule Maturation and Exocytosis Require the Effector Protein HMunc13-4. Nat. Immunol. 2007, 8, 257–267. [Google Scholar] [CrossRef]
- Wu, B.; Guo, W. The Exocyst at a Glance. J. Cell Sci. 2015, 128, 2957–2964. [Google Scholar] [CrossRef] [PubMed]
- Heider, M.R.; Munson, M. Exorcising the Exocyst Complex. Traffic (Cph. Den.) 2012, 13, 898–907. [Google Scholar] [CrossRef]
- Lu, H.; Liu, S.; Zhang, G.; Kwong, L.N.; Zhu, Y.; Miller, J.P.; Hu, Y.; Zhong, W.; Zeng, J.; Wu, L.; et al. Oncogenic BRAF-Mediated Melanoma Cell Invasion. Cell Rep. 2016, 15, 2012–2024. [Google Scholar] [CrossRef] [PubMed]
- Koike, S.; Yamasaki, K.; Yamauchi, T.; Shimada-Omori, R.; Tsuchiyama, K.; Aiba, S. Toll-like Receptor 2 Utilizes RAB11A for Melanosome Transfer from Melanocytes to Keratinocytes. J. Dermatol. Sci. 2019, 94, 310–312. [Google Scholar] [CrossRef] [PubMed]
- Paul, D.; Achouri, S.; Yoon, Y.Z.; Herre, J.; Bryant, C.E.; Cicuta, P. Phagocytosis Dynamics Depends on Target Shape. Biophys. J. 2013, 105, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Ter, H. Endocytosis of Particle Formulations by Macrophages and Its Application to Clinical Treatment. In Molecular Regulation of Endocytosis; InTech: London, UK, 2012; pp. 413–428. ISBN 978-953-51-0662-3. [Google Scholar]
- Lin, C.B.; Chen, N.; Scarpa, R.; Guan, F.; Babiarz-Magee, L.; Liebel, F.; Li, W.-H.; Kizoulis, M.; Shapiro, S.; Seiberg, M. LIGR, a Protease-Activated Receptor-2-Derived Peptide, Enhances Skin Pigmentation without Inducing Inflammatory Processes. Pigment Cell Melanoma Res. 2008, 21, 172–183. [Google Scholar] [CrossRef]
- Boissy, R.E. Melanosome Transfer to and Translocation in the Keratinocyte. Exp. Dermatol. 2003, 12, 5–12. [Google Scholar] [CrossRef]
- Borovanský, J.; Elleder, M. Melanosome Degradation: Fact or Fiction. Pigment Cell Res. 2003, 16, 280–286. [Google Scholar] [CrossRef]
- Murase, D.; Hachiya, A.; Takano, K.; Hicks, R.; Visscher, M.O.; Kitahara, T.; Hase, T.; Takema, Y.; Yoshimori, T. Autophagy Has a Significant Role in Determining Skin Color by Regulating Melanosome Degradation in Keratinocytes. J. Investig. Dermatol. 2013, 133, 2416–2424. [Google Scholar] [CrossRef]
- Du, C.; Zhang, T.; Xiao, X.; Shi, Y.; Duan, H.; Ren, Y. Protease-Activated Receptor-2 Promotes Kidney Tubular Epithelial Inflammation by Inhibiting Autophagy via the PI3K/Akt/MTOR Signalling Pathway. Biochem. J. 2017, 474, 2733–2747. [Google Scholar] [CrossRef] [PubMed]
- Akinduro, O.; Sully, K.; Patel, A.; Robinson, D.J.; Chikh, A.; McPhail, G.; Braun, K.M.; Philpott, M.P.; Harwood, C.A.; Byrne, C.; et al. Constitutive Autophagy and Nucleophagy during Epidermal Differentiation. J. Investig. Dermatol. 2016, 136, 1460–1470. [Google Scholar] [CrossRef] [PubMed]
- Byers, H.R.; Maheshwary, S.; Amodeo, D.M.; Dykstra, S.G. Role of Cytoplasmic Dynein in Perinuclear Aggregation of Phagocytosed Melanosomes and Supranuclear Melanin Cap Formation in Human Keratinocytes. J. Investig. Dermatol. 2003, 121, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Castellano-Pellicena, I.; Morrison, C.G.; Bell, M.; O’Connor, C.; Tobin, D.J. Melanin Distribution in Human Skin: Influence of Cytoskeletal, Polarity, and Centrosome-Related Machinery of Stratum Basale Keratinocytes. Int. J. Mol. Sci. 2021, 22, 3143. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreiras, H.; Seabra, M.C.; Barral, D.C. Melanin Transfer in the Epidermis: The Pursuit of Skin Pigmentation Control Mechanisms. Int. J. Mol. Sci. 2021, 22, 4466. https://doi.org/10.3390/ijms22094466
Moreiras H, Seabra MC, Barral DC. Melanin Transfer in the Epidermis: The Pursuit of Skin Pigmentation Control Mechanisms. International Journal of Molecular Sciences. 2021; 22(9):4466. https://doi.org/10.3390/ijms22094466
Chicago/Turabian StyleMoreiras, Hugo, Miguel C. Seabra, and Duarte C. Barral. 2021. "Melanin Transfer in the Epidermis: The Pursuit of Skin Pigmentation Control Mechanisms" International Journal of Molecular Sciences 22, no. 9: 4466. https://doi.org/10.3390/ijms22094466
APA StyleMoreiras, H., Seabra, M. C., & Barral, D. C. (2021). Melanin Transfer in the Epidermis: The Pursuit of Skin Pigmentation Control Mechanisms. International Journal of Molecular Sciences, 22(9), 4466. https://doi.org/10.3390/ijms22094466






