Disentangling the Molecular Mechanisms of the Antidepressant Activity of Omega-3 Polyunsaturated Fatty Acid: A Comprehensive Review of the Literature
Abstract
1. Introduction
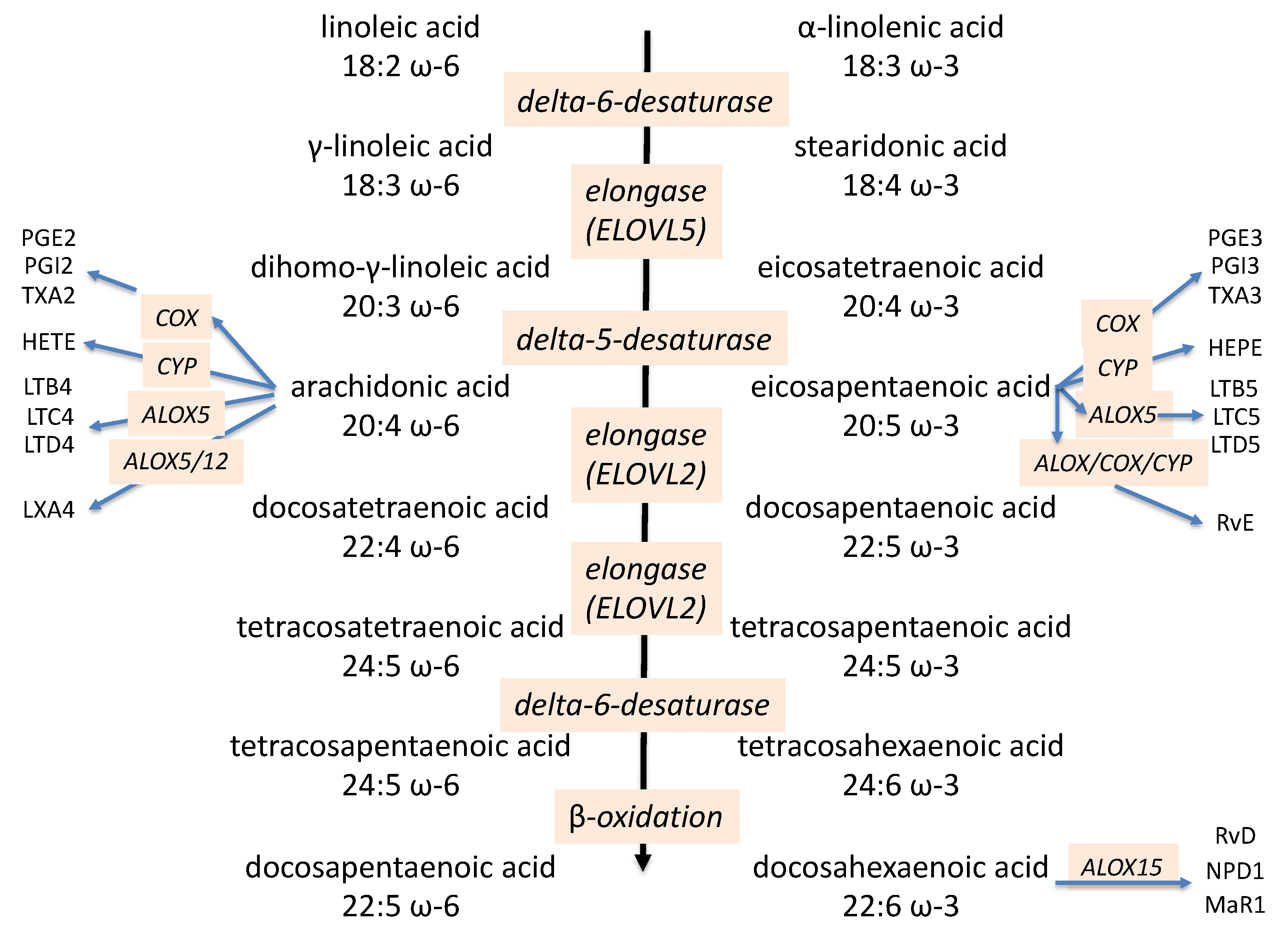
2. Omega-3 PUFAs and Immunomodulation
2.1. Anti-Inflammatory Effects of EPA and DHA
2.2. Pro- and Anti-Inflammatory Oxidation Products from EPA and DHA
2.2.1. Prostaglandins and Leukotrienes
2.2.2. Resolvins and Other Poly-Hydroxyl Products
2.2.3. CYP Metabolism of Double Bonds or the Terminal Carbon Atom
2.2.4. Epoxidation of Endocannabinoids
3. Omega-3 PUFAs, Membrane Structure and Organization
3.1. Differential Effects of EPA and DHA on Lipid Raft Formation
3.2. Preferential Incorporation of EPA and DHA into Different Types of Glycerophospholipids
4. Discussion
| Observation | Citation |
|---|---|
| Last step of DHA synthesis (not EPA synthesis) is in peroxisome | [108,112] |
| Rate-limiting step in DHA biosynthesis is between EPA and DHA | [202,203,204] |
| Brain levels of DHA are kept high, while EPA levels are kept low | [193,194,205,206] |
| Size of lipid rafts is increased by DHA and unaltered by EPA | [185] |
| DHA is inserted in PE1, PS and PC, while EPA is inserted in PI | [199] |
| EPA and DHA are released from lipids by different PLA2 isoenzymes | [191,192] |
| COX and ALOX5 metabolize EPA and AA, not DHA | [108,112] |
| EPA-derived resolvins and DHA-derived resolvins in some cases activate distinct G-protein coupled receptors | [207] |
| RvE1 reduces, while RvD1 increases platelet aggregation | [157] |
| EPA, not DHA activates PPARα | [208] |
| EPA, not DHA increases expression of cPLA2 | [195] |
| EPA, not DHA improves depression | [23,24,25,33] |
| EPA, not DHA ameliorates cardiac illness | [209] |
| EPEA and its epoxide, but not DHEA or its epoxide are highly potent CB2 receptor agonists | [174] |
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AA | arachidonic acid |
| AEA | arachidonic acid-ethanolamide |
| ALOX | arachidonate lipoxygenase |
| CB1 | cannabis receptor-1 |
| CD14 | cluster of differentiation-14 |
| COX | cyclooxygenase |
| CYP | cytochrome P450 |
| DASS21 score | a 21-item rating scale to quantify depression anxiety and stress |
| DHA | docosahexaenoic acid |
| DHEA | docosahexaenoic acid-ethanolamine |
| EPA | eicosapentaenoic acid |
| EPEA | eicosapentaenoic acid-ethanolamine |
| 17,18-EEQ-EA | ethanolamine derivative of EPA, where the omega-3 bond is oxidized to an epoxide |
| ES | effect size |
| GPR or GPCR | G-protein-coupled receptor |
| 20-HETE | 20-hydroxy-tetraenoic acid |
| MCP1 | monocyte chemoattractive protein-1 |
| MDD | major depressive disorder |
| NLRP | acronym for NACHT, LRR and PYD domains-containing protein |
| NFκB | nuclear factor-κB |
| OCD | obsessive-compulsive disorder |
| PC | phosphatidylcholine |
| PE | phosphatidylethanolamine |
| PI | phosphatidylinositol |
| PS | phosphatidylserine |
| PLA2 | phospholipase A2 |
| PPAR | peroxisome proliferator-activated receptor |
| PUFA | polyunsaturated fatty acid |
| RCT | randomized controlled trial |
| RvE1 | Resolvin E1 |
| RvD1 | Resolvin D1 |
| ROS | reactive oxygen species |
| SMD | standardized mean differences |
| SPM | specialized pro-resolving mediators |
| TLR | Toll-like receptor |
References
- Bountziouka, V.; Polychronopoulos, E.; Zeimbekis, A.; Papavenetiou, E.; Ladoukaki, E.; Papairakleous, N.; Gotsis, E.; Metallinos, G.; Lionis, C.; Panagiotakos, D. Long-term fish intake is associated with less severe depressive symptoms among elderly men and women: The MEDIS (MEDiterranean ISlands Elderly) epidemiological study. J. Aging Health 2009, 21, 864–880. [Google Scholar] [CrossRef] [PubMed]
- Tanskanen, A.; Hibbeln, J.R.; Hintikka, J.; Haatainen, K.; Honkalampi, K.; Viinamaki, H. Fish consumption, depression, and suicidality in a general population. Arch. Gen. Psychiatry 2001, 58, 512–513. [Google Scholar] [CrossRef]
- Timonen, M.; Horrobin, D.; Jokelainen, J.; Laitinen, J.; Herva, A.; Rasanen, P. Fish consumption and depression: The Northern Finland 1966 birth cohort study. J. Affect. Disord. 2004, 82, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Suominen-Taipale, A.L.; Partonen, T.; Turunen, A.W.; Mannisto, S.; Jula, A.; Verkasalo, P.K. Fish consumption and omega-3 polyunsaturated fatty acids in relation to depressive episodes: A cross-sectional analysis. PLoS ONE 2010, 5, e10530. [Google Scholar] [CrossRef]
- Appleton, K.M.; Woodside, J.V.; Yarnell, J.W.; Arveiler, D.; Haas, B.; Amouyel, P.; Montaye, M.; Ferrieres, J.; Ruidavets, J.B.; Ducimetiere, P.; et al. Depressed mood and dietary fish intake: Direct relationship or indirect relationship as a result of diet and lifestyle? J. Affect. Disord. 2007, 104, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Miyake, Y.; Sasaki, S.; Tanaka, K.; Arakawa, M. Fish and n-3 polyunsaturated fatty acid intake and depressive symptoms: Ryukyus Child Health Study. Pediatrics 2010, 126, e623–e630. [Google Scholar] [CrossRef]
- Li, Y.; Dai, Q.; Ekperi, L.I.; Dehal, A.; Zhang, J. Fish consumption and severely depressed mood, findings from the first national nutrition follow-up study. Psychiatry Res. 2011, 190, 103–109. [Google Scholar] [CrossRef]
- Sanchez-Villegas, A.; Henriquez, P.; Figueiras, A.; Ortuno, F.; Lahortiga, F.; Martinez-Gonzalez, M.A. Long chain omega-3 fatty acids intake, fish consumption and mental disorders in the SUN cohort study. Eur. J. Nutr. 2007, 46, 337–346. [Google Scholar] [CrossRef]
- Astorg, P.; Couthouis, A.; Bertrais, S.; Arnault, N.; Meneton, P.; Guesnet, P.; Alessandri, J.M.; Galan, P.; Hercberg, S. Association of fish and long-chain n-3 polyunsaturated fatty acid intakes with the occurrence of depressive episodes in middle-aged French men and women. Prostaglandins Leukot. Essent. Fatty Acids 2008, 78, 171–182. [Google Scholar] [CrossRef]
- Hibbeln, J.R. Fish consumption and major depression. Lancet 1998, 351, 1213. [Google Scholar] [CrossRef]
- Noaghiul, S.; Hibbeln, J.R. Cross-national comparisons of seafood consumption and rates of bipolar disorders. Am. J. Psychiatry 2003, 160, 2222–2227. [Google Scholar] [CrossRef]
- Li, F.; Liu, X.; Zhang, D. Fish consumption and risk of depression: A meta-analysis. J. Epidemiol. Commun. Health 2016, 70, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Su, K.P.; Matsuoka, Y.; Pae, C.U. Omega-3 polyunsaturated fatty acids in prevention of mood and anxiety disorders. Clin. Psychopharmacol. Neurosci. 2015, 13, 129–137. [Google Scholar] [CrossRef]
- De Vriese, S.R.; Christophe, A.B.; Maes, M. Lowered serum n-3 polyunsaturated fatty acid (PUFA) levels predict the occurrence of postpartum depression: Further evidence that lowered n-PUFAs are related to major depression. Life Sci. 2003, 73, 3181–3187. [Google Scholar] [CrossRef]
- Lin, P.Y.; Huang, S.Y.; Su, K.P. A meta-analytic review of polyunsaturated fatty acid compositions in patients with depression. Biol. Psychiatry 2010, 68, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Adams, P.B.; Lawson, S.; Sanigorski, A.; Sinclair, A.J. Arachidonic acid to eicosapentaenoic acid ratio in blood correlates positively with clinical symptoms of depression. Lipids 1996, 31, S157–S161. [Google Scholar] [CrossRef]
- Maes, M.; Smith, R.; Christophe, A.; Cosyns, P.; Desnyder, R.; Meltzer, H. Fatty acid composition in major depression: Decreased omega 3 fractions in cholesteryl esters and increased C20: 4 omega 6/C20:5 omega 3 ratio in cholesteryl esters and phospholipids. J. Affect. Disord. 1996, 38, 35–46. [Google Scholar] [CrossRef]
- Edwards, R.; Peet, M.; Shay, J.; Horrobin, D. Omega-3 polyunsaturated fatty acid levels in the diet and in red blood cell membranes of depressed patients. J. Affect. Disord. 1998, 48, 149–155. [Google Scholar] [CrossRef]
- Frasure-Smith, N.; Lesperance, F.; Julien, P. Major depression is associated with lower omega-3 fatty acid levels in patients with recent acute coronary syndromes. Biol. Psychiatry 2004, 55, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Pottala, J.V.; Talley, J.A.; Churchill, S.W.; Lynch, D.A.; von Schacky, C.; Harris, W.S. Red blood cell fatty acids are associated with depression in a case-control study of adolescents. Prostaglandins Leukot. Essent. Fatty Acids 2012, 86, 161–165. [Google Scholar] [CrossRef]
- Huan, M.; Hamazaki, K.; Sun, Y.; Itomura, M.; Liu, H.; Kang, W.; Watanabe, S.; Terasawa, K.; Hamazaki, T. Suicide attempt and n-3 fatty acid levels in red blood cells: A case control study in China. Biol. Psychiatry 2004, 56, 490–496. [Google Scholar] [CrossRef]
- Sublette, M.E.; Hibbeln, J.R.; Galfalvy, H.; Oquendo, M.A.; Mann, J.J. Omega-3 polyunsaturated essential fatty acid status as a predictor of future suicide risk. Am. J. Psychiatry 2006, 163, 1100–1102. [Google Scholar] [CrossRef] [PubMed]
- Martins, J.G. EPA but not DHA appears to be responsible for the efficacy of omega-3 long chain polyunsaturated fatty acid supplementation in depression: Evidence from a meta-analysis of randomized controlled trials. J. Am. Coll. Nutr. 2009, 28, 525–542. [Google Scholar] [CrossRef]
- Sublette, M.E.; Ellis, S.P.; Geant, A.L.; Mann, J.J. Meta-analysis of the effects of eicosapentaenoic acid (EPA) in clinical trials in depression. J. Clin. Psychiatry 2011, 72, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Pajak, A.; Marventano, S.; Castellano, S.; Galvano, F.; Bucolo, C.; Drago, F.; Caraci, F. Role of omega-3 fatty acids in the treatment of depressive disorders: A comprehensive meta-analysis of randomized clinical trials. PLoS ONE 2014, 9, e96905. [Google Scholar] [CrossRef]
- Mocking, R.J.; Harmsen, I.; Assies, J.; Koeter, M.W.; Ruhe, H.G.; Schene, A.H. Meta-analysis and meta-regression of omega-3 polyunsaturated fatty acid supplementation for major depressive disorder. Transl. Psychiatry 2016, 6, e756. [Google Scholar] [CrossRef]
- Parker, G.; Gibson, N.A.; Brotchie, H.; Heruc, G.; Rees, A.M.; Hadzi-Pavlovic, D. Omega-3 fatty acids and mood disorders. Am. J. Psychiatry 2006, 163, 969–978. [Google Scholar] [CrossRef]
- Freeman, M.P.; Fava, M.; Lake, J.; Trivedi, M.H.; Wisner, K.L.; Mischoulon, D. Complementary and alternative medicine in major depressive disorder: The American Psychiatric Association Task Force report. J. Clin. Psychiatry 2010, 71, 669–681. [Google Scholar] [CrossRef]
- Bloch, M.H.; Hannestad, J. Omega-3 fatty acids for the treatment of depression: Systematic review and meta-analysis. Mol. Psychiatry 2012, 17, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.Y.; Mischoulon, D.; Freeman, M.P.; Matsuoka, Y.; Hibbeln, J.; Belmaker, R.H.; Su, K.P. Are omega-3 fatty acids antidepressants or just mood-improving agents? The effect depends upon diagnosis, supplement preparation, and severity of depression. Mol. Psychiatry 2012, 17, 1161–1167. [Google Scholar] [CrossRef]
- Rogers, P.J.; Appleton, K.M.; Kessler, D.; Peters, T.J.; Gunnell, D.; Hayward, R.C.; Heatherley, S.V.; Christian, L.M.; McNaughton, S.A.; Ness, A.R. No effect of n-3 long-chain polyunsaturated fatty acid (EPA and DHA) supplementation on depressed mood and cognitive function: A randomised controlled trial. Br. J. Nutr. 2008, 99, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Marangell, L.B.; Martinez, J.M.; Zboyan, H.A.; Kertz, B.; Kim, H.F.; Puryear, L.J. A double-blind, placebo-controlled study of the omega-3 fatty acid docosahexaenoic acid in the treatment of major depression. Am. J. Psychiatry 2003, 160, 996–998. [Google Scholar] [CrossRef]
- Rapaport, M.H.; Nierenberg, A.A.; Schettler, P.J.; Kinkead, B.; Cardoos, A.; Walker, R.; Mischoulon, D. Inflammation as a predictive biomarker for response to omega-3 fatty acids in major depressive disorder: A proof-of-concept study. Mol. Psychiatry 2016, 21, 71–79. [Google Scholar] [CrossRef]
- Peet, M.; Horrobin, D.F. A dose-ranging study of the effects of ethyl-eicosapentaenoate in patients with ongoing depression despite apparently adequate treatment with standard drugs. Arch. Gen. Psychol. 2002, 59, 913–919. [Google Scholar] [CrossRef]
- Nemets, B.; Stahl, Z.; Belmaker, R.H. Addition of omega-3 fatty acid to maintenance medication treatment for recurrent unipolar depressive disorder. Am. J. Psychiatry 2002, 159, 477–479. [Google Scholar] [CrossRef] [PubMed]
- Su, K.P.; Huang, S.Y.; Chiu, C.C.; Shen, W.W. Omega-3 fatty acids in major depressive disorder. A preliminary double-blind, placebo-controlled trial. Eur. Neuropsychopharmacol. 2003, 13, 267–271. [Google Scholar] [CrossRef]
- Silvers, K.M.; Woolley, C.C.; Hamilton, F.C.; Watts, P.M.; Watson, R.A. Randomised double-blind placebo-controlled trial of fish oil in the treatment of depression. Prostaglandins Leukot. Essent. Fatty Acids 2005, 72, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Nemets, H.; Nemets, B.; Apter, A.; Bracha, Z.; Belmaker, R.H. Omega-3 treatment of childhood depression: A controlled, double-blind pilot study. Am. J. Psychiatry 2006, 163, 1098–1100. [Google Scholar] [CrossRef]
- Grenyer, B.F.; Crowe, T.; Meyer, B.; Owen, A.J.; Grigonis-Deane, E.M.; Caputi, P.; Howe, P.R. Fish oil supplementation in the treatment of major depression: A randomised double-blind placebo-controlled trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 1393–1396. [Google Scholar] [CrossRef] [PubMed]
- Jazayeri, S.; Tehrani-Doost, M.; Keshavarz, S.A.; Hosseini, M.; Djazayery, A.; Amini, H.; Jalali, M.; Peet, M. Comparison of therapeutic effects of omega-3 fatty acid eicosapentaenoic acid and fluoxetine, separately and in combination, in major depressive disorder. Aust. N. Z. J. Psychiatry 2008, 42, 192–198. [Google Scholar] [CrossRef]
- Mischoulon, D.; Papakostas, G.I.; Dording, C.M.; Farabaugh, A.H.; Sonawalla, S.B.; Agoston, A.M.; Smith, J.; Beaumont, E.C.; Dahan, L.E.; Alpert, J.E.; et al. A double-blind, randomized controlled trial of ethyl-eicosapentaenoate for major depressive disorder. J. Clin. Psychiatry 2009, 70, 1636–1644. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Giacosa, A.; Opizzi, A.; Pelucchi, C.; La Vecchia, C.; Montorfano, G.; Negroni, M.; Berra, B.; Politi, P.; Rizzo, A.M. Effect of omega-3 fatty acids supplementation on depressive symptoms and on health-related quality of life in the treatment of elderly women with depression: A double-blind, placebo-controlled, randomized clinical trial. J. Am. Coll. Nutr. 2010, 29, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Giacosa, A.; Opizzi, A.; Pelucchi, C.; La Vecchia, C.; Montorfano, G.; Negroni, M.; Berra, B.; Politi, P.; Rizzo, A.M. Long chain omega 3 polyunsaturated fatty acids supplementation in the treatment of elderly depression: Effects on depressive symptoms, on phospholipids fatty acids profile and on health-related quality of life. J. Nutr. Health Aging 2011, 15, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Lesperance, F.; Frasure-Smith, N.; St-Andre, E.; Turecki, G.; Lesperance, P.; Wisniewski, S.R. The efficacy of omega-3 supplementation for major depression: A randomized controlled trial. J. Clin. Psychiatry 2011, 72, 1054–1062. [Google Scholar] [CrossRef]
- Tajalizadekhoob, Y.; Sharifi, F.; Fakhrzadeh, H.; Mirarefin, M.; Ghaderpanahi, M.; Badamchizade, Z.; Azimipour, S. The effect of low-dose omega 3 fatty acids on the treatment of mild to moderate depression in the elderly: A double-blind, randomized, placebo-controlled study. Eur. Arch. Psychiatry Clin. Neurosci. 2011, 261, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Sinn, N.; Milte, C.M.; Street, S.J.; Buckley, J.D.; Coates, A.M.; Petkov, J.; Howe, P.R. Effects of n-3 fatty acids, EPA v. DHA, on depressive symptoms, quality of life, memory and executive function in older adults with mild cognitive impairment: A 6-month randomised controlled trial. Br. J. Nutr. 2012, 107, 1682–1693. [Google Scholar] [CrossRef]
- Gertsik, L.; Poland, R.E.; Bresee, C.; Rapaport, M.H. Omega-3 fatty acid augmentation of citalopram treatment for patients with major depressive disorder. J. Clin. Psychopharmacol. 2012, 32, 61–64. [Google Scholar] [CrossRef]
- Rizzo, A.M.; Corsetto, P.A.; Montorfano, G.; Opizzi, A.; Faliva, M.; Giacosa, A.; Ricevuti, G.; Pelucchi, C.; Berra, B.; Rondanelli, M. Comparison between the AA/EPA ratio in depressed and non depressed elderly females: Omega-3 fatty acid supplementation correlates with improved symptoms but does not change immunological parameters. Nutr. J. 2012, 11, 82. [Google Scholar] [CrossRef] [PubMed]
- Mozaffari-Khosravi, H.; Yassini-Ardakani, M.; Karamati, M.; Shariati-Bafghi, S.-E. Eicosapentaenoic acid versus docosahexaenoic acid in mild-to-moderate depression: A randomized, double-blind, placebo-controlled trial. Eur. Neuropsychopharmacol. 2013, 23, 636–644. [Google Scholar] [CrossRef]
- Mischoulon, D.; Nierenberg, A.A.; Schettler, P.J.; Kinkead, B.L.; Fehling, K.; Martinson, M.A.; Rapaport, M.H. A double-blind, randomized controlled clinical trial comparing eicosapentaenoic acid versus docosahexaenoic acid for depression. J. Clin. Psychiatry 2014, 76, 54–61. [Google Scholar] [CrossRef]
- Park, Y.; Park, Y.-S.; Kim, S.H.; Oh, D.H.; Park, Y.-C. Supplementation of n-3 polyunsaturated fatty acids for major depressive disorder: A randomized, double-blind, 12-week, placebo-controlled trial in Korea. Ann. Nutr. Metab. 2015, 66, 141–148. [Google Scholar] [CrossRef]
- Ginty, A.T.; Conklin, S.M. Short-term supplementation of acute long-chain omega-3 polyunsaturated fatty acids may alter depression status and decrease symptomology among young adults with depression: A preliminary randomized and placebo controlled trial. Psychiatry Res. 2015, 229, 485–489. [Google Scholar] [CrossRef]
- Young, A.S.; Arnold, L.E.; Wolfson, H.L.; Fristad, M.A. Psychoeducational psychotherapy and omega-3 supplementation improve co-occurring behavioral problems in youth with depression: Results from a pilot RCT. J. Abnorm. Child Psychol. 2017, 45, 1025–1037. [Google Scholar] [CrossRef]
- Gabbay, V.; Freed, R.D.; Alonso, C.M.; Senger, S.; Stadterman, J.; Davison, B.A.; Klein, R.G. A double-blind placebo-controlled trial of omega-3 fatty acids as a monotherapy for adolescent depression. J. Clin. Psychiatry 2018, 79, 11596. [Google Scholar] [CrossRef] [PubMed]
- Trebatická, J.; Hradečná, Z.; Böhmer, F.; Vaváková, M.; Waczulíková, I.; Garaiova, I.; Luha, J.; Škodáček, I.; Šuba, J.; Ďuračková, Z. Emulsified omega-3 fatty-acids modulate the symptoms of depressive disorder in children and adolescents: A pilot study. Child Adolesc. Psychiatry Ment. Health 2017, 11, 1–10. [Google Scholar] [CrossRef]
- Jahangard, L.; Sadeghi, A.; Ahmadpanah, M.; Holsboer-Trachsler, E.; Bahmani, D.S.; Haghighi, M.; Brand, S. Influence of adjuvant omega-3-polyunsaturated fatty acids on depression, sleep, and emotion regulation among outpatients with major depressive disorders-Results from a double-blind, randomized and placebo-controlled clinical trial. J. Psychiatr. Res. 2018, 107, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Tayama, J.; Ogawa, S.; Nakaya, N.; Sone, T.; Hamaguchi, T.; Takeoka, A.; Hamazaki, K.; Okamura, H.; Yajima, J.; Kobayashi, M. Omega-3 polyunsaturated fatty acids and psychological intervention for workers with mild to moderate depression: A double-blind randomized controlled trial. J. Affect. Disord. 2019, 245, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Fristad, M.A.; Vesco, A.T.; Young, A.S.; Healy, K.Z.; Nader, E.S.; Gardner, W.; Seidenfeld, A.M.; Wolfson, H.L.; Arnold, L.E. Pilot randomized controlled trial of omega-3 and individual–family psychoeducational psychotherapy for children and adolescents with depression. J. Clin. Child Adolesc. Psychol. 2019, 48, S105–S118. [Google Scholar] [CrossRef]
- Trebatická, J.; Hradečná, Z.; Surovcová, A.; Katrenčíková, B.; Gushina, I.; Waczulíková, I.; Sušienková, K.; Garaiova, I.; Šuba, J.; Ďuračková, Z. Omega-3 fatty-acids modulate symptoms of depressive disorder, serum levels of omega-3 fatty acids and omega-6/omega-3 ratio in children. A randomized, double-blind and controlled trial. Psychiatry Res. 2020, 287, 112911. [Google Scholar] [CrossRef]
- Stoll, A.L.; Severus, W.E.; Freeman, M.P.; Rueter, S.; Zboyan, H.A.; Diamond, E.; Cress, K.K.; Marangell, L.B. Omega 3 fatty acids in bipolar disorder: A preliminary double-blind, placebo-controlled trial. Arch. Gen. Psychol. 1999, 56, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Hirashima, F.; Parow, A.M.; Stoll, A.L.; Demopulos, C.M.; Damico, K.E.; Rohan, M.L.; Eskesen, J.G.; Zuo, C.S.; Cohen, B.M.; Renshaw, P.F. Omega-3 fatty acid treatment and T(2) whole brain relaxation times in bipolar disorder. Am. J. Psychiatry 2004, 161, 1922–1924. [Google Scholar] [CrossRef]
- Chiu, C.C.; Huang, S.Y.; Chen, C.C.; Su, K.P. Omega-3 fatty acids are more beneficial in the depressive phase than in the manic phase in patients with bipolar I disorder. J. Clin. Psychiatry 2005, 66, 1613–1614. [Google Scholar] [CrossRef]
- Frangou, S.; Lewis, M.; McCrone, P. Efficacy of ethyl-eicosapentaenoic acid in bipolar depression: Randomised double-blind placebo-controlled study. Br. J. Psychiatry J. Ment. Sci. 2006, 188, 46–50. [Google Scholar] [CrossRef]
- Keck, P.E., Jr.; Mintz, J.; McElroy, S.L.; Freeman, M.P.; Suppes, T.; Frye, M.A.; Altshuler, L.L.; Kupka, R.; Nolen, W.A.; Leverich, G.S.; et al. Double-blind, randomized, placebo-controlled trials of ethyl-eicosapentanoate in the treatment of bipolar depression and rapid cycling bipolar disorder. Biol. Psychiatry 2006, 60, 1020–1022. [Google Scholar] [CrossRef] [PubMed]
- Frangou, S.; Lewis, M.; Wollard, J.; Simmons, A. Preliminary in vivo evidence of increased N-acetyl-aspartate following eicosapentanoic acid treatment in patients with bipolar disorder. J. Psychopharmacol. 2007, 21, 435–439. [Google Scholar] [CrossRef]
- Murphy, B.L.; Stoll, A.L.; Harris, P.Q.; Ravichandran, C.; Babb, S.M.; Carlezon, W.A., Jr.; Cohen, B.M. Omega-3 fatty acid treatment, with or without cytidine, fails to show therapeutic properties in bipolar disorder: A double-blind, randomized add-on clinical trial. J. Clin. Psychopharmacol. 2012, 32, 699–703. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Strawn, J.R.; Tallman, M.J.; Welge, J.A.; Patino, L.R.; Blom, T.J.; DelBello, M.P. Effects of fish oil monotherapy on depression and prefrontal neurochemistry in adolescents at high risk for bipolar I disorder: A 12-week placebo-controlled proton magnetic resonance spectroscopy trial. J. Child Adolesc. Psychopharmacol. 2020, 30, 293–305. [Google Scholar] [CrossRef]
- Freeman, M.P.; Davis, M.; Sinha, P.; Wisner, K.L.; Hibbeln, J.R.; Gelenberg, A.J. Omega-3 fatty acids and supportive psychotherapy for perinatal depression: A randomized placebo-controlled study. J. Affect. Disord. 2008, 110, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Rees, A.M.; Austin, M.P.; Parker, G.B. Omega-3 fatty acids as a treatment for perinatal depression: Randomized double-blind placebo-controlled trial. Aust. N. Z. J. Psychiatry 2008, 42, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Nishi, D.; Su, K.-P.; Usuda, K.; Chang, J.P.-C.; Hamazaki, K.; Ishima, T.; Sano, Y.; Ito, H.; Isaka, K.; Tachibana, Y.; et al. Plasma estradiol levels and antidepressant effects of omega-3 fatty acids in pregnant women. Brain Behav. Immun. 2020, 85, 29–34. [Google Scholar] [CrossRef]
- Lucas, M.; Asselin, G.; Merette, C.; Poulin, M.J.; Dodin, S. Ethyl-eicosapentaenoic acid for the treatment of psychological distress and depressive symptoms in middle-aged women: A double-blind, placebo-controlled, randomized clinical trial. Am. J. Clin. Nutr. 2009, 89, 641–651. [Google Scholar] [CrossRef]
- Antypa, N.; Smelt, A.H.; Strengholt, A.; Van der Does, A.J. Effects of omega-3 fatty acid supplementation on mood and emotional information processing in recovered depressed individuals. J. Psychopharmacol. 2012, 26, 738–743. [Google Scholar] [CrossRef]
- Sohrabi, N.; Kashanian, M.; Ghafoori, S.S.; Malakouti, S.K. Evaluation of the effect of omega-3 fatty acids in the treatment of premenstrual syndrome: “A pilot trial”. Complement. Ther. Med. 2013, 21, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Behan, P.O.; Behan, W.M.; Horrobin, D.F. Effect of high doses of essential fatty acids on the postviral fatigue syndrome. Acta Neurol. Scand. 1990, 82, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Warren, G.; McKendrick, M.; Peet, M. The role of essential fatty acids in chronic fatigue syndrome. A case-controlled study of red-cell membrane essential fatty acids (EFA) and a placebo-controlled treatment study with high dose of EFA. Acta Neurol. Scand. 1999, 99, 112–116. [Google Scholar] [CrossRef]
- Zanarini, M.C.; Frankenburg, F.R. omega-3 Fatty acid treatment of women with borderline personality disorder: A double-blind, placebo-controlled pilot study. Am. J. Psychiatry 2003, 160, 167–169. [Google Scholar] [CrossRef]
- Fux, M.; Benjamin, J.; Nemets, B. A placebo-controlled cross-over trial of adjunctive EPA in OCD. J. Psychiatr. Res. 2004, 38, 323–325. [Google Scholar] [CrossRef]
- Hallahan, B.; Hibbeln, J.R.; Davis, J.M.; Garland, M.R. Omega-3 fatty acid supplementation in patients with recurrent self-harm. Single-centre double-blind randomised controlled trial. Br. J. Psychiatry J. Ment. Sci. 2007, 190, 118–122. [Google Scholar] [CrossRef]
- Peet, M.; Horrobin, D.F. A dose-ranging exploratory study of the effects of ethyl-eicosapentaenoate in patients with persistent schizophrenic symptoms. J. Psychiatr. Res. 2002, 36, 7–18. [Google Scholar] [CrossRef]
- Fenton, W.S.; Dickerson, F.; Boronow, J.; Hibbeln, J.R.; Knable, M. A placebo-controlled trial of omega-3 fatty acid (ethyl eicosapentaenoic acid) supplementation for residual symptoms and cognitive impairment in schizophrenia. Am. J. Psychiatry 2001, 158, 2071–2074. [Google Scholar] [CrossRef]
- Qiao, Y.; Mei, Y.; Han, H.; Liu, F.; Yang, X.M.; Shao, Y.; Xie, B.; Long, B. Effects of Omega-3 in the treatment of violent schizophrenia patients. Schizophr. Res. 2018, 195, 283–285. [Google Scholar] [CrossRef]
- Chiu, C.C.; Su, K.P.; Cheng, T.C.; Liu, H.C.; Chang, C.J.; Dewey, M.E.; Stewart, R.; Huang, S.Y. The effects of omega-3 fatty acids monotherapy in Alzheimer’s disease and mild cognitive impairment: A preliminary randomized double-blind placebo-controlled study. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 1538–1544. [Google Scholar] [CrossRef] [PubMed]
- Freund-Levi, Y.; Basun, H.; Cederholm, T.; Faxen-Irving, G.; Garlind, A.; Grut, M.; Vedin, I.; Palmblad, J.; Wahlund, L.O.; Eriksdotter-Jonhagen, M. Omega-3 supplementation in mild to moderate Alzheimer’s disease: Effects on neuropsychiatric symptoms. Int. J. Geriatr. Psychiatry 2008, 23, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, T.M.; Munhoz, R.P.; Alvarez, C.; Naliwaiko, K.; Kiss, A.; Andreatini, R.; Ferraz, A.C. Depression in Parkinson’s disease: A double-blind, randomized, placebo-controlled pilot study of omega-3 fatty-acid supplementation. J. Affect. Disord. 2008, 111, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Carney, R.M.; Freedland, K.E.; Rubin, E.H.; Rich, M.W.; Steinmeyer, B.C.; Harris, W.S. Omega-3 augmentation of sertraline in treatment of depression in patients with coronary heart disease: A randomized controlled trial. JAMA J. Am. Med. Assoc. 2009, 302, 1651–1657. [Google Scholar] [CrossRef]
- Bot, M.; Pouwer, F.; Assies, J.; Jansen, E.H.; Diamant, M.; Snoek, F.J.; Beekman, A.T.; de Jonge, P. Eicosapentaenoic acid as an add-on to antidepressant medication for co-morbid major depression in patients with diabetes mellitus: A randomized, double-blind placebo-controlled study. J. Affect. Disord. 2010, 126, 282–286. [Google Scholar] [CrossRef]
- Giltay, E.J.; Geleijnse, J.M.; Kromhout, D. Effects of n-3 fatty acids on depressive symptoms and dispositional optimism after myocardial infarction. Am. J. Clin. Nutr. 2011, 94, 1442–1450. [Google Scholar] [CrossRef]
- Bot, M.; Pouwer, F.; Assies, J.; Jansen, E.H.; Beekman, A.T.; de Jonge, P. Supplementation with eicosapentaenoic omega-3 fatty acid does not influence serum brain-derived neurotrophic factor in diabetes mellitus patients with major depression: A randomized controlled pilot study. Neuropsychobiology 2011, 63, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Andreeva, V.A.; Galan, P.; Torres, M.; Julia, C.; Hercberg, S.; Kesse-Guyot, E. Supplementation with B vitamins or n-3 fatty acids and depressive symptoms in cardiovascular disease survivors: Ancillary findings from the SUpplementation with FOLate, vitamins B-6 and B-12 and/or OMega-3 fatty acids (SU.FOL.OM3) randomized trial. Am. J. Clin. Nutr. 2012, 96, 208–214. [Google Scholar] [CrossRef]
- Fontani, G.; Corradeschi, F.; Felici, A.; Alfatti, F.; Migliorini, S.; Lodi, L. Cognitive and physiological effects of Omega-3 polyunsaturated fatty acid supplementation in healthy subjects. Eur. J. Clin. Investig. 2005, 35, 691–699. [Google Scholar] [CrossRef]
- van de Rest, O.; Geleijnse, J.M.; Kok, F.J.; van Staveren, W.A.; Hoefnagels, W.H.; Beekman, A.T.; de Groot, L.C. Effect of fish-oil supplementation on mental well-being in older subjects: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2008, 88, 706–713. [Google Scholar] [CrossRef]
- Antypa, N.; Van der Does, A.J.; Smelt, A.H.; Rogers, R.D. Omega-3 fatty acids (fish-oil) and depression-related cognition in healthy volunteers. J. Psychopharmacol. 2009, 23, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Belury, M.A.; Andridge, R.; Malarkey, W.B.; Glaser, R. Omega-3 supplementation lowers inflammation and anxiety in medical students: A randomized controlled trial. Brain Behav. Immun. 2011, 25, 1725–1734. [Google Scholar] [CrossRef]
- DeFina, L.F.; Marcoux, L.G.; Devers, S.M.; Cleaver, J.P.; Willis, B.L. Effects of omega-3 supplementation in combination with diet and exercise on weight loss and body composition. Am. J. Clin. Nutr. 2011, 93, 455–462. [Google Scholar] [CrossRef]
- Kiecolt-Glaser, J.K.; Belury, M.A.; Andridge, R.; Malarkey, W.B.; Hwang, B.S.; Glaser, R. Omega-3 supplementation lowers inflammation in healthy middle-aged and older adults: A randomized controlled trial. Brain Behav. Immun. 2012, 26, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Van der Wurff, I.; Von Schacky, C.; Bergeland, T.; Leontjevas, R.; Zeegers, M.P.; Kirschner, P.; de Groot, R. Effect of one year krill oil supplementation on depressive symptoms and self-esteem of Dutch adolescents: A randomized controlled trial. Prostaglandin Leukot. Essent. Fatty Acids 2020, 163, 102208. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.C.; Huang, S.Y.; Su, K.P. Omega-3 polyunsaturated fatty acids for postpartum depression. Am. J. Obstet. Gynecol. 2004, 190, 582–583. [Google Scholar] [CrossRef]
- Chiu, C.C.; Huang, S.Y.; Shen, W.W.; Su, K.P. Omega-3 fatty acids for depression in pregnancy. Am. J. Psychiatry 2003, 160, 385. [Google Scholar] [CrossRef]
- Leonard, B.; Maes, M. Mechanistic explanations how cell-mediated immune activation, inflammation and oxidative and nitrosative stress pathways and their sequels and concomitants play a role in the pathophysiology of unipolar depression. Neurosci. Biobehav. Rev. 2012, 36, 764–785. [Google Scholar] [CrossRef]
- Lang, U.E.; Borgwardt, S. Molecular mechanisms of depression: Perspectives on new treatment strategies. Cell. Physiol. Biochem. 2013, 31, 761–777. [Google Scholar] [CrossRef]
- Rosenblat, J.D.; Cha, D.S.; Mansur, R.B.; McIntyre, R.S. Inflamed moods: A review of the interactions between inflammation and mood disorders. Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 53, 23–34. [Google Scholar] [CrossRef]
- Lotrich, F.E. Inflammatory cytokine-associated depression. Brain Res. 2015, 1617, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, D.R.; Rapaport, M.H.; Miller, B.J. A meta-analysis of blood cytokine network alterations in psychiatric patients: Comparisons between schizophrenia, bipolar disorder and depression. Mol. Psychiatry 2016, 21, 1696–1709. [Google Scholar] [CrossRef]
- Kopschina Feltes, P.; Doorduin, J.; Klein, H.C.; Juarez-Orozco, L.E.; Dierckx, R.A.; Moriguchi-Jeckel, C.M.; de Vries, E.F. Anti-inflammatory treatment for major depressive disorder: Implications for patients with an elevated immune profile and non-responders to standard antidepressant therapy. J. Psychopharmacol. 2017, 31, 1149–1165. [Google Scholar] [CrossRef] [PubMed]
- Kalkman, H.O.; Feuerbach, D. Antidepressant therapies inhibit inflammation and microglial M1-polarization. Pharmacol. Ther. 2016, 163, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Young, J.J.; Bruno, D.; Pomara, N. A review of the relationship between proinflammatory cytokines and major depressive disorder. J. Affect. Disord. 2014, 169, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Kalkman, H.O. The association between vascular inflammation and depressive disorder. Causality, biomarkers and targeted treatment. Pharmaceuticals 2020, 13, 92. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef] [PubMed]
- Gharekhani, A.; Khatami, M.R.; Dashti-Khavidaki, S.; Razeghi, E.; Noorbala, A.A.; Hashemi-Nazari, S.S.; Mansournia, M.A. The effect of omega-3 fatty acids on depressive symptoms and inflammatory markers in maintenance hemodialysis patients: A randomized, placebo-controlled clinical trial. Eur. J. Clin. Pharmacol. 2014, 70, 655–665. [Google Scholar] [CrossRef]
- Su, K.P.; Lai, H.C.; Yang, H.T.; Su, W.P.; Peng, C.Y.; Chang, J.P.; Chang, H.C.; Pariante, C.M. Omega-3 fatty acids in the prevention of interferon-alpha-induced depression: Results from a randomized, controlled trial. Biol. Psychiatry 2014, 76, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Cipollina, C. Endogenous generation and signaling actions of omega-3 fatty acid electrophilic derivatives. Biomed. Res. Int. 2015, 2015, 501792. [Google Scholar] [CrossRef]
- Dyall, S.C. Long-chain omega-3 fatty acids and the brain: A review of the independent and shared effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Sohn, K.H.; Rhee, S.H.; Hwang, D. Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like receptor 4. J. Biol. Chem. 2001, 276, 16683–16689. [Google Scholar] [CrossRef]
- Shi, H.; Kokoeva, M.V.; Inouye, K.; Tzameli, I.; Yin, H.; Flier, J.S. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Investig. 2006, 116, 3015–3025. [Google Scholar] [CrossRef] [PubMed]
- Russell, F.D.; Burgin-Maunder, C.S. Distinguishing health benefits of eicosapentaenoic and docosahexaenoic acids. Mar. Drugs 2012, 10, 2535–2559. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; Vanden Heuvel, J.P.; Gillies, P.J.; West, S.G.; Kris-Etherton, P.M. Anti-inflammatory effects of polyunsaturated fatty acids in THP-1 cells. Biochem. Biophys. Res. Commun. 2005, 336, 909–917. [Google Scholar] [CrossRef]
- Weldon, S.M.; Mullen, A.C.; Loscher, C.E.; Hurley, L.A.; Roche, H.M. Docosahexaenoic acid induces an anti-inflammatory profile in lipopolysaccharide-stimulated human THP-1 macrophages more effectively than eicosapentaenoic acid. J. Nutr. Biochem. 2007, 18, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, H.; Hoshino, A.; Funayama, M.; Kawahara, K.; Obata, F. Oxidative modulation of the glutathione-redox couple enhances lipopolysaccharide-induced interleukin 12 P40 production by a mouse macrophage cell line, J774A.1. Free Radic. Res. 2003, 37, 293–299. [Google Scholar] [CrossRef]
- Allam-Ndoul, B.; Guenard, F.; Barbier, O.; Vohl, M.C. Effect of n-3 fatty acids on the expression of inflammatory genes in THP-1 macrophages. Lipids Health Dis. 2016, 15, 69. [Google Scholar] [CrossRef]
- Mogensen, T.H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Callaway, J.B.; Ting, J.P. Inflammasomes: Mechanism of action, role in disease, and therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zhang, Y.; Xu, T.; Yin, Y.; Huang, R.; Wang, Y.; Zhang, J.; Huang, D.; Li, W. Chronic dexamethasone treatment results in hippocampal neurons injury due to activate NLRP1 inflammasome in vitro. Int. Immunopharmacol. 2017, 49, 222–230. [Google Scholar] [CrossRef]
- Iwata, M.; Ota, K.T.; Duman, R.S. The inflammasome: Pathways linking psychological stress, depression, and systemic illnesses. Brain Behav. Immun. 2013, 31, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Alcocer-Gomez, E.; de Miguel, M.; Casas-Barquero, N.; Nunez-Vasco, J.; Sanchez-Alcazar, J.A.; Fernandez-Rodriguez, A.; Cordero, M.D. NLRP3 inflammasome is activated in mononuclear blood cells from patients with major depressive disorder. Brain Behav. Immun. 2014, 36, 111–117. [Google Scholar] [CrossRef]
- Akosile, W.; Voisey, J.; Lawford, B.; Colquhounc, D.; Young, R.M.; Mehta, D. The inflammasome NLRP12 is associated with both depression and coronary artery disease in Vietnam veterans. Psychiatry Res. 2018, 270, 775–779. [Google Scholar] [CrossRef]
- Yan, Y.; Jiang, W.; Spinetti, T.; Tardivel, A.; Castillo, R.; Bourquin, C.; Guarda, G.; Tian, Z.; Tschopp, J.; Zhou, R. Omega-3 fatty acids prevent inflammation and metabolic disorder through inhibition of NLRP3 inflammasome activation. Immunity 2013, 38, 1154–1163. [Google Scholar] [CrossRef]
- Zhou, R.; Tardivel, A.; Thorens, B.; Choi, I.; Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 2010, 11, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, L.; Hu, W.; Zheng, Q.; Xiang, W. Mitochondrial dysfunction during in vitro hepatocyte steatosis is reversed by omega-3 fatty acid-induced up-regulation of mitofusin 2. Metabolism 2011, 60, 767–775. [Google Scholar] [CrossRef]
- Lepretti, M.; Martucciello, S.; Burgos Aceves, M.A.; Putti, R.; Lionetti, L. Omega-3 fatty acids and insulin resistance: Focus on the regulation of mitochondria and endoplasmic reticulum stress. Nutrients 2018, 10, 350. [Google Scholar] [CrossRef] [PubMed]
- Rae, C.D.; Williams, S.R. Glutathione in the human brain: Review of its roles and measurement by magnetic resonance spectroscopy. Anal. Biochem. 2017, 529, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.L.; Lagopoulos, J.; Cockayne, N.; Lewis, S.J.; Hickie, I.B.; Hermens, D.F.; Naismith, S.L. The effect of 12-wk omega-3 fatty acid supplementation on in vivo thalamus glutathione concentration in patients “at risk” for major depression. Nutrition 2015, 31, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.E.; Wood, S.J.; Wellard, R.M.; Proffitt, T.M.; McConchie, M.; Amminger, G.P.; Jackson, G.D.; Velakoulis, D.; Pantelis, C.; McGorry, P.D. Ethyl-eicosapentaenoic acid in first-episode psychosis. A 1H-MRS study. Neuropsychopharmacology 2008, 33, 2467–2473. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, D.; Ni, N.; Ren, H.; Luo, C.; He, C.; Kang, J.X.; Wan, J.B.; Su, H. Omega-3 polyunsaturated fatty acids protect neural progenitor cells against oxidative injury. Mar. Drugs 2014, 12, 2341–2356. [Google Scholar] [CrossRef]
- Cai, S.M.; Yang, R.Q.; Li, Y.; Ning, Z.W.; Zhang, L.L.; Zhou, G.S.; Luo, W.; Li, D.H.; Chen, Y.; Pan, M.X.; et al. Angiotensin-(1-7) Improves Liver Fibrosis by Regulating the NLRP3 Inflammasome via Redox Balance Modulation. Antioxid. Redox Signal. 2016, 24, 795–812. [Google Scholar] [CrossRef]
- Gabbs, M.; Leng, S.; Devassy, J.G.; Monirujjaman, M.; Aukema, H.M. Advances in our understanding of oxylipins derived from dietary PUFAs. Adv. Nutr. 2015, 6, 513–540. [Google Scholar] [CrossRef]
- Goldman, D.W.; Pickett, W.C.; Goetzl, E.J. Human neutrophil chemotactic and degranulating activities of leukotriene B5 (LTB5) derived from eicosapentaenoic acid. Biochem. Biophys. Res. Commun. 1983, 117, 282–288. [Google Scholar] [CrossRef]
- Strasser, T.; Fischer, S.; Weber, P.C. Leukotriene B5 is formed in human neutrophils after dietary supplementation with icosapentaenoic acid. Proc. Natl. Acad. Sci. USA 1985, 82, 1540–1543. [Google Scholar] [CrossRef]
- Nieves, D.; Moreno, J.J. Effect of arachidonic and eicosapentaenoic acid metabolism on RAW 264.7 macrophage proliferation. J. Cell Physiol. 2006, 208, 428–434. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Van Dyke, T.E. Resolving inflammation: Dual anti-inflammatory and pro-resolution lipid mediators. Nat. Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.F.; Dona, M.; Fredman, G.; Krishnamoorthy, S.; Irimia, D.; Serhan, C.N. Resolvin E2 formation and impact in inflammation resolution. J. Immunol. 2012, 188, 4527–4534. [Google Scholar] [CrossRef]
- Isobe, Y.; Arita, M.; Matsueda, S.; Iwamoto, R.; Fujihara, T.; Nakanishi, H.; Taguchi, R.; Masuda, K.; Sasaki, K.; Urabe, D.; et al. Identification and structure determination of novel anti-inflammatory mediator resolvin E3, 17,18-dihydroxyeicosapentaenoic acid. J. Biol. Chem. 2012, 287, 10525–10534. [Google Scholar] [CrossRef]
- Arita, M.; Bianchini, F.; Aliberti, J.; Sher, A.; Chiang, N.; Hong, S.; Yang, R.; Petasis, N.A.; Serhan, C.N. Stereochemical assignment, antiinflammatory properties, and receptor for the omega-3 lipid mediator resolvin E1. J. Exp. Med. 2005, 201, 713–722. [Google Scholar] [CrossRef]
- Chiang, N.; Serhan, C.N. Structural elucidation and physiologic functions of specialized pro-resolving mediators and their receptors. Mol. Aspects Med. 2017, 58, 114–129. [Google Scholar] [CrossRef]
- Serhan, C.N. Treating inflammation and infection in the 21st century: New hints from decoding resolution mediators and mechanisms. FASEB J. 2017, 31, 1273–1288. [Google Scholar] [CrossRef]
- Colgan, S.P.; Serhan, C.N.; Parkos, C.A.; Delp-Archer, C.; Madara, J.L. Lipoxin A4 modulates transmigration of human neutrophils across intestinal epithelial monolayers. J. Clin. Investig. 1993, 92, 75–82. [Google Scholar] [CrossRef]
- Serhan, C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef]
- Gilbert, K.; Bernier, J.; Godbout, R.; Rousseau, G. Resolvin D1, a metabolite of omega-3 polyunsaturated fatty acid, decreases post-myocardial infarct depression. Mar. Drugs 2014, 12, 5396–5407. [Google Scholar] [CrossRef]
- Deyama, S.; Ishikawa, Y.; Yoshikawa, K.; Shimoda, K.; Ide, S.; Satoh, M.; Minami, M. Resolvin D1 and D2 reverse lipopolysaccharide-induced depression-like behaviors through the mTORC1 signaling pathway. Int. J. Neuropsychopharmacol. 2017, 20, 575–584. [Google Scholar] [CrossRef]
- Deyama, S.; Shimoda, K.; Suzuki, H.; Ishikawa, Y.; Ishimura, K.; Fukuda, H.; Hitora-Imamura, N.; Ide, S.; Satoh, M.; Kaneda, K.; et al. Resolvin E1/E2 ameliorate lipopolysaccharide-induced depression-like behaviors via ChemR23. Psychopharmacology 2018, 235, 329–336. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Deyama, S.; Shimoda, K.; Yoshikawa, K.; Ide, S.; Satoh, M.; Minami, M. Rapid and sustained antidepressant effects of resolvin D1 and D2 in a chronic unpredictable stress model. Behav. Brain Res. 2017, 332, 233–236. [Google Scholar] [CrossRef]
- Hasturk, H.; Abdallah, R.; Kantarci, A.; Nguyen, D.; Giordano, N.; Hamilton, J.; Van Dyke, T.E. Resolvin E1 (RvE1) attenuates atherosclerotic plaque formation in diet and inflammation-induced atherogenesis. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Salic, K.; Morrison, M.C.; Verschuren, L.; Wielinga, P.Y.; Wu, L.; Kleemann, R.; Gjorstrup, P.; Kooistra, T. Resolvin E1 attenuates atherosclerosis in absence of cholesterol-lowering effects and on top of atorvastatin. Atherosclerosis 2016, 250, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Sima, C.; Montero, E.; Nguyen, D.; Freire, M.; Norris, P.; Serhan, C.N.; Van Dyke, T.E. ERV1 overexpression in myeloid cells protects against high fat diet induced obesity and glucose intolerance. Sci. Rep. 2017, 7, 12848. [Google Scholar] [CrossRef]
- Buckley, C.D.; Gilroy, D.W.; Serhan, C.N. Proresolving lipid mediators and mechanisms in the resolution of acute inflammation. Immunity 2014, 40, 315–327. [Google Scholar] [CrossRef]
- Herova, M.; Schmid, M.; Gemperle, C.; Hersberger, M. ChemR23, the receptor for chemerin and resolvin E1, is expressed and functional on M1 but not on M2 macrophages. J. Immunol. 2015, 194, 2330–2337. [Google Scholar] [CrossRef]
- Schmid, M.; Gemperle, C.; Rimann, N.; Hersberger, M. Resolvin D1 Polarizes primary human macrophages toward a proresolution phenotype through GPR32. J. Immunol. 2016, 196, 3429–3437. [Google Scholar] [CrossRef]
- Lannan, K.L.; Spinelli, S.L.; Blumberg, N.; Phipps, R.P. Maresin 1 induces a novel pro-resolving phenotype in human platelets. J. Thromb. Haemost. 2017, 15, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Rey, C.; Nadjar, A.; Joffre, F.; Amadieu, C.; Aubert, A.; Vaysse, C.; Pallet, V.; Laye, S.; Joffre, C. Maternal n-3 polyunsaturated fatty acid dietary supply modulates microglia lipid content in the offspring. Prostaglandins Leukot. Essent. Fatty Acids 2018, 133, 1–7. [Google Scholar] [CrossRef]
- Rey, C.; Delpech, J.C.; Madore, C.; Nadjar, A.; Greenhalgh, A.D.; Amadieu, C.; Aubert, A.; Pallet, V.; Vaysse, C.; Laye, S.; et al. Dietary n-3 long chain PUFA supplementation promotes a pro-resolving oxylipin profile in the brain. Brain Behav. Immun. 2019, 76, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Waldman, M.; Peterson, S.J.; Arad, M.; Hochhauser, E. The role of 20-HETE in cardiovascular diseases and its risk factors. Prostaglandins Lipid Mediat. 2016, 125, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Elshenawy, O.H.; Shoieb, S.M.; Mohamed, A.; El-Kadi, A.O. Clinical Implications of 20-Hydroxyeicosatetraenoic acid in the kidney, liver, lung and brain: An emerging therapeutic target. Pharmaceutics 2017, 9, 9. [Google Scholar] [CrossRef]
- Arnold, C.; Konkel, A.; Fischer, R.; Schunck, W.H. Cytochrome P450-dependent metabolism of omega-6 and omega-3 long-chain polyunsaturated fatty acids. Pharmacol. Rep. 2010, 62, 536–547. [Google Scholar] [CrossRef]
- Konkel, A.; Schunck, W.H. Role of cytochrome P450 enzymes in the bioactivation of polyunsaturated fatty acids. Biochim. Biophys. Acta 2011, 1814, 210–222. [Google Scholar] [CrossRef]
- Schunck, W.H.; Konkel, A.; Fischer, R.; Weylandt, K.H. Therapeutic potential of omega-3 fatty acid-derived epoxyeicosanoids in cardiovascular and inflammatory diseases. Pharmacol. Ther. 2018, 183, 177–204. [Google Scholar] [CrossRef]
- Ulu, A.; Harris, T.R.; Morisseau, C.; Miyabe, C.; Inoue, H.; Schuster, G.; Dong, H.; Iosif, A.M.; Liu, J.Y.; Weiss, R.H.; et al. Anti-inflammatory effects of omega-3 polyunsaturated fatty acids and soluble epoxide hydrolase inhibitors in angiotensin-II-dependent hypertension. J. Cardiovasc. Pharmacol. 2013, 62, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Spector, A.A.; Kim, H.Y. Cytochrome P450 epoxygenase pathway of polyunsaturated fatty acid metabolism. Biochim. Biophys. Acta 2015, 1851, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Garcia, V.; Gilani, A.; Shkolnik, B.; Pandey, V.; Zhang, F.F.; Dakarapu, R.; Gandham, S.K.; Reddy, N.R.; Graves, J.P.; Gruzdev, A.; et al. 20-HETE signals through g-protein-coupled receptor GPR75 (Gq) to affect vascular function and trigger hypertension. Circ. Res. 2017, 120, 1776–1788. [Google Scholar] [CrossRef]
- Fischer, R.; Konkel, A.; Mehling, H.; Blossey, K.; Gapelyuk, A.; Wessel, N.; von Schacky, C.; Dechend, R.; Muller, D.N.; Rothe, M.; et al. Dietary omega-3 fatty acids modulate the eicosanoid profile in man primarily via the CYP-epoxygenase pathway. J. Lipid Res. 2014, 55, 1150–1164. [Google Scholar] [CrossRef] [PubMed]
- Schuchardt, J.P.; Schmidt, S.; Kressel, G.; Willenberg, I.; Hammock, B.D.; Hahn, A.; Schebb, N.H. Modulation of blood oxylipin levels by long-chain omega-3 fatty acid supplementation in hyper- and normolipidemic men. Prostaglandins Leukot. Essent. Fatty Acids 2014, 90, 27–37. [Google Scholar] [CrossRef]
- Freitas, H.R.; Isaac, A.R.; Malcher-Lopes, R.; Diaz, B.L.; Trevenzoli, I.H.; De Melo Reis, R.A. Polyunsaturated fatty acids and endocannabinoids in health and disease. Nutr. Neurosci. 2018, 21, 695–714. [Google Scholar] [CrossRef]
- Smaga, I.; Bystrowska, B.; Gawlinski, D.; Przegalinski, E.; Filip, M. The endocannabinoid/endovanilloid system and depression. Curr. Neuropharmacol. 2014, 12, 462–474. [Google Scholar] [CrossRef]
- Turcotte, C.; Blanchet, M.R.; Laviolette, M.; Flamand, N. The CB2 receptor and its role as a regulator of inflammation. Cell. Mol. Life Sci. 2016, 73, 4449–4470. [Google Scholar] [CrossRef]
- Balvers, M.G.; Verhoeckx, K.C.; Plastina, P.; Wortelboer, H.M.; Meijerink, J.; Witkamp, R.F. Docosahexaenoic acid and eicosapentaenoic acid are converted by 3T3-L1 adipocytes to N-acyl ethanolamines with anti-inflammatory properties. Biochim. Biophys. Acta 2010, 1801, 1107–1114. [Google Scholar] [CrossRef]
- McDougle, D.R.; Watson, J.E.; Abdeen, A.A.; Adili, R.; Caputo, M.P.; Krapf, J.E.; Johnson, R.W.; Kilian, K.A.; Holinstat, M.; Das, A. Anti-inflammatory omega-3 endocannabinoid epoxides. Proc. Natl. Acad. Sci. USA 2017, 114, E6034–E6043. [Google Scholar] [CrossRef]
- Snider, N.T.; Nast, J.A.; Tesmer, L.A.; Hollenberg, P.F. A cytochrome P450-derived epoxygenated metabolite of anandamide is a potent cannabinoid receptor 2-selective agonist. Mol. Pharmacol. 2009, 75, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Chiurchiu, V.; Battistini, L.; Maccarrone, M. Endocannabinoid signalling in innate and adaptive immunity. Immunology 2015, 144, 352–364. [Google Scholar] [CrossRef] [PubMed]
- Roche, M.; Finn, D.P. Brain CB(2) Receptors: Implications for neuropsychiatric disorders. Pharmaceuticals 2010, 3, 2517–2553. [Google Scholar] [CrossRef]
- Kim, H.Y.; Spector, A.A.; Xiong, Z.M. A synaptogenic amide N-docosahexaenoylethanolamide promotes hippocampal development. Prostaglandins Lipid Mediat. 2011, 96, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Huang, B.X.; Kwon, H.; Rashid, M.A.; Kharebava, G.; Desai, A.; Patnaik, S.; Marugan, J.; Kim, H.Y. Orphan GPR110 (ADGRF1) targeted by N-docosahexaenoylethanolamine in development of neurons and cognitive function. Nat. Commun. 2016, 7, 13123. [Google Scholar] [CrossRef]
- Artmann, A.; Petersen, G.; Hellgren, L.I.; Boberg, J.; Skonberg, C.; Nellemann, C.; Hansen, S.H.; Hansen, H.S. Influence of dietary fatty acids on endocannabinoid and N-acylethanolamine levels in rat brain, liver and small intestine. Biochim. Biophys. Acta 2008, 1781, 200–212. [Google Scholar] [CrossRef]
- Rossmeisl, M.; Jilkova, Z.M.; Kuda, O.; Jelenik, T.; Medrikova, D.; Stankova, B.; Kristinsson, B.; Haraldsson, G.G.; Svensen, H.; Stoknes, I.; et al. Metabolic effects of n-3 PUFA as phospholipids are superior to triglycerides in mice fed a high-fat diet: Possible role of endocannabinoids. PLoS ONE 2012, 7, e38834. [Google Scholar] [CrossRef]
- Boorman, E.; Zajkowska, Z.; Ahmed, R.; Pariante, C.M.; Zunszain, P.A. Crosstalk between endocannabinoid and immune systems: A potential dysregulation in depression? Psychopharmacology 2016, 233, 1591–1604. [Google Scholar] [CrossRef]
- Muller, C.P.; Reichel, M.; Muhle, C.; Rhein, C.; Gulbins, E.; Kornhuber, J. Brain membrane lipids in major depression and anxiety disorders. Biochim. Biophys. Acta 2015, 1851, 1052–1065. [Google Scholar] [CrossRef]
- Gorjao, R.; Azevedo-Martins, A.K.; Rodrigues, H.G.; Abdulkader, F.; Arcisio-Miranda, M.; Procopio, J.; Curi, R. Comparative effects of DHA and EPA on cell function. Pharmacol. Ther. 2009, 122, 56–64. [Google Scholar] [CrossRef] [PubMed]
- McMurray, D.N.; Bonilla, D.L.; Chapkin, R.S. n-3 Fatty acids uniquely affect anti-microbial resistance and immune cell plasma membrane organization. Chem. Phys. Lipids 2011, 164, 626–635. [Google Scholar] [CrossRef]
- Schoeniger, A.; Fuhrmann, H.; Schumann, J. LPS- or Pseudomonas aeruginosa-mediated activation of the macrophage TLR4 signaling cascade depends on membrane lipid composition. PeerJ 2016, 4, e1663. [Google Scholar] [CrossRef]
- Hellwing, C.; Tigistu-Sahle, F.; Fuhrmann, H.; Kakela, R.; Schumann, J. Lipid composition of membrane microdomains isolated detergent-free from PUFA supplemented RAW264.7 macrophages. J. Cell. Physiol. 2018, 233, 2602–2612. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.L.; Zhang, G.; Ji, N.N.; Du, L.; Chen, B.B.; Hua, R.; Zhang, Y.M. Toll-Like Receptor 4 in Paraventricular Nucleus Mediates Visceral Hypersensitivity Induced by Maternal Separation. Front. Pharmacol. 2017, 8, 309. [Google Scholar] [CrossRef]
- Liscovitch, M. Crosstalk among multiple signal-activated phospholipases. Trends Biochem. Sci. 1992, 17, 393–399. [Google Scholar] [CrossRef]
- Law, M.H.; Cotton, R.G.; Berger, G.E. The role of phospholipases A2 in schizophrenia. Mol. Psychiatry 2006, 11, 547–556. [Google Scholar] [CrossRef]
- Basselin, M.; Rosa, A.O.; Ramadan, E.; Cheon, Y.; Chang, L.; Chen, M.; Greenstein, D.; Wohltmann, M.; Turk, J.; Rapoport, S.I. Imaging decreased brain docosahexaenoic acid metabolism and signaling in iPLA(2)beta (VIA)-deficient mice. J. Lipid Res. 2010, 51, 3166–3173. [Google Scholar] [CrossRef] [PubMed]
- Birbes, H.; Pageaux, J.F.; Fayard, J.M.; Lagarde, M.; Laugier, C. Protein kinase C inhibitors stimulate arachidonic and docosahexaenoic acids release from uterine stromal cells through a Ca2+-independent pathway. FEBS Lett. 1998, 432, 219–224. [Google Scholar] [CrossRef]
- McNamara, R.K.; Carlson, S.E. Role of omega-3 fatty acids in brain development and function: Potential implications for the pathogenesis and prevention of psychopathology. Prostaglandins Leukot. Essent. Fatty Acids 2006, 75, 329–349. [Google Scholar] [CrossRef] [PubMed]
- Bazinet, R.P.; Laye, S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Su, K.P.; Yang, H.T.; Chang, J.P.; Shih, Y.H.; Guu, T.W.; Kumaran, S.S.; Galecki, P.; Walczewska, A.; Pariante, C.M. Eicosapentaenoic and docosahexaenoic acids have different effects on peripheral phospholipase A2 gene expressions in acute depressed patients. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 80, 227–233. [Google Scholar] [CrossRef]
- Fordsmann, J.C.; Ko, R.W.; Choi, H.B.; Thomsen, K.; Witgen, B.M.; Mathiesen, C.; Lonstrup, M.; Piilgaard, H.; MacVicar, B.A.; Lauritzen, M. Increased 20-HETE synthesis explains reduced cerebral blood flow but not impaired neurovascular coupling after cortical spreading depression in rat cerebral cortex. J. Neurosci. 2013, 33, 2562–2570. [Google Scholar] [CrossRef]
- Song, C.; Zhang, X.Y.; Manku, M. Increased phospholipase A2 activity and inflammatory response but decreased nerve growth factor expression in the olfactory bulbectomized rat model of depression: Effects of chronic ethyl-eicosapentaenoate treatment. J. Neurosci. 2009, 29, 14–22. [Google Scholar] [CrossRef]
- Chen, C.T.; Domenichiello, A.F.; Trepanier, M.O.; Liu, Z.; Masoodi, M.; Bazinet, R.P. The low levels of eicosapentaenoic acid in rat brain phospholipids are maintained via multiple redundant mechanisms. J. Lipid Res. 2013, 54, 2410–2422. [Google Scholar] [CrossRef]
- Lee, C.H.; Hajra, A.K. Molecular species of diacylglycerols and phosphoglycerides and the postmortem changes in the molecular species of diacylglycerols in rat brains. J. Neurochem. 1991, 56, 370–379. [Google Scholar] [CrossRef]
- Soderberg, M.; Edlund, C.; Kristensson, K.; Dallner, G. Fatty acid composition of brain phospholipids in aging and in Alzheimer’s disease. Lipids 1991, 26, 421–425. [Google Scholar] [CrossRef]
- Tanaka, T.; Iwawaki, D.; Sakamoto, M.; Takai, Y.; Morishige, J.; Murakami, K.; Satouchi, K. Mechanisms of accumulation of arachidonate in phosphatidylinositol in yellowtail. A comparative study of acylation systems of phospholipids in rat and the fish species Seriola quinqueradiata. Eur. J. Biochem. 2003, 270, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Kew, S.; Mesa, M.D.; Tricon, S.; Buckley, R.; Minihane, A.M.; Yaqoob, P. Effects of oils rich in eicosapentaenoic and docosahexaenoic acids on immune cell composition and function in healthy humans. Am. J. Clin. Nutr. 2004, 79, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467S–1476S. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Cameron-Smith, D.; Garg, M.; Sinclair, A.J. Docosapentaenoic acid (22:5n-3): A review of its biological effects. Prog. Lipid Res. 2011, 50, 28–34. [Google Scholar] [CrossRef] [PubMed]
- DeMar, J.C., Jr.; Ma, K.; Bell, J.M.; Rapoport, S.I. Half-lives of docosahexaenoic acid in rat brain phospholipids are prolonged by 15 weeks of nutritional deprivation of n-3 polyunsaturated fatty acids. J. Neurochem. 2004, 91, 1125–1137. [Google Scholar] [CrossRef]
- Liu, J.J.; Green, P.; John Mann, J.; Rapoport, S.I.; Sublette, M.E. Pathways of polyunsaturated fatty acid utilization: Implications for brain function in neuropsychiatric health and disease. Brain Res. 2015, 1597, 220–246. [Google Scholar] [CrossRef]
- Recchiuti, A. Resolvin D1 and its GPCRs in resolution circuits of inflammation. Prostaglandins Lipid Mediat. 2013, 107, 64–76. [Google Scholar] [CrossRef]
- Pawar, A.; Jump, D.B. Unsaturated fatty acid regulation of peroxisome proliferator-activated receptor alpha activity in rat primary hepatocytes. J. Biol. Chem. 2003, 278, 35931–35939. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Wu, J.H. (n-3) fatty acids and cardiovascular health: Are effects of EPA and DHA shared or complementary? J. Nutr. 2012, 142, 614S–625S. [Google Scholar] [CrossRef]
- Di Marzo, V. New approaches and challenges to targeting the endocannabinoid system. Nat. Rev. Drug Discov. 2018, 17, 623–639. [Google Scholar] [CrossRef]
- Correa, F.; Hernangomez, M.; Mestre, L.; Loria, F.; Spagnolo, A.; Docagne, F.; Di Marzo, V.; Guaza, C. Anandamide enhances IL-10 production in activated microglia by targeting CB(2) receptors: Roles of ERK1/2, JNK, and NF-kappaB. Glia 2010, 58, 135–147. [Google Scholar] [CrossRef] [PubMed]
| Indication | Citation |
|---|---|
| Major depressive disorders (MDD) | [32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59] |
| Depressive episodes in bipolar affective disorders | [60,61,62,63,64,65,66,67] |
| Depression during or post pregnancy | [68,69,70] |
| Depression in non-MDD mood disorders (e.g., premenstrual syndrome, dysthymia) | [31,45,49,71,72,73] |
| Depression in other psychiatric conditions (e.g., borderline personality, self-harm, OCD) | [74,75,76,77,78] |
| Depression in established schizophrenia | [79,80,81] |
| Depression in Alzheimer’s dementia/mild cognitive impairment | [46,82,83] |
| Depression in Parkinson’s disease | [84] |
| Depression in medical conditions (cerebrovascular and metabolic diseases or cancer) | [85,86,87,88,89] |
| Depressive features in healthy individuals | [90,91,92,93,94,95,96] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalkman, H.O.; Hersberger, M.; Walitza, S.; Berger, G.E. Disentangling the Molecular Mechanisms of the Antidepressant Activity of Omega-3 Polyunsaturated Fatty Acid: A Comprehensive Review of the Literature. Int. J. Mol. Sci. 2021, 22, 4393. https://doi.org/10.3390/ijms22094393
Kalkman HO, Hersberger M, Walitza S, Berger GE. Disentangling the Molecular Mechanisms of the Antidepressant Activity of Omega-3 Polyunsaturated Fatty Acid: A Comprehensive Review of the Literature. International Journal of Molecular Sciences. 2021; 22(9):4393. https://doi.org/10.3390/ijms22094393
Chicago/Turabian StyleKalkman, Hans O., Martin Hersberger, Suzanne Walitza, and Gregor E. Berger. 2021. "Disentangling the Molecular Mechanisms of the Antidepressant Activity of Omega-3 Polyunsaturated Fatty Acid: A Comprehensive Review of the Literature" International Journal of Molecular Sciences 22, no. 9: 4393. https://doi.org/10.3390/ijms22094393
APA StyleKalkman, H. O., Hersberger, M., Walitza, S., & Berger, G. E. (2021). Disentangling the Molecular Mechanisms of the Antidepressant Activity of Omega-3 Polyunsaturated Fatty Acid: A Comprehensive Review of the Literature. International Journal of Molecular Sciences, 22(9), 4393. https://doi.org/10.3390/ijms22094393






