Unlocking the Health Potential of Microalgae as Sustainable Sources of Bioactive Compounds
Abstract
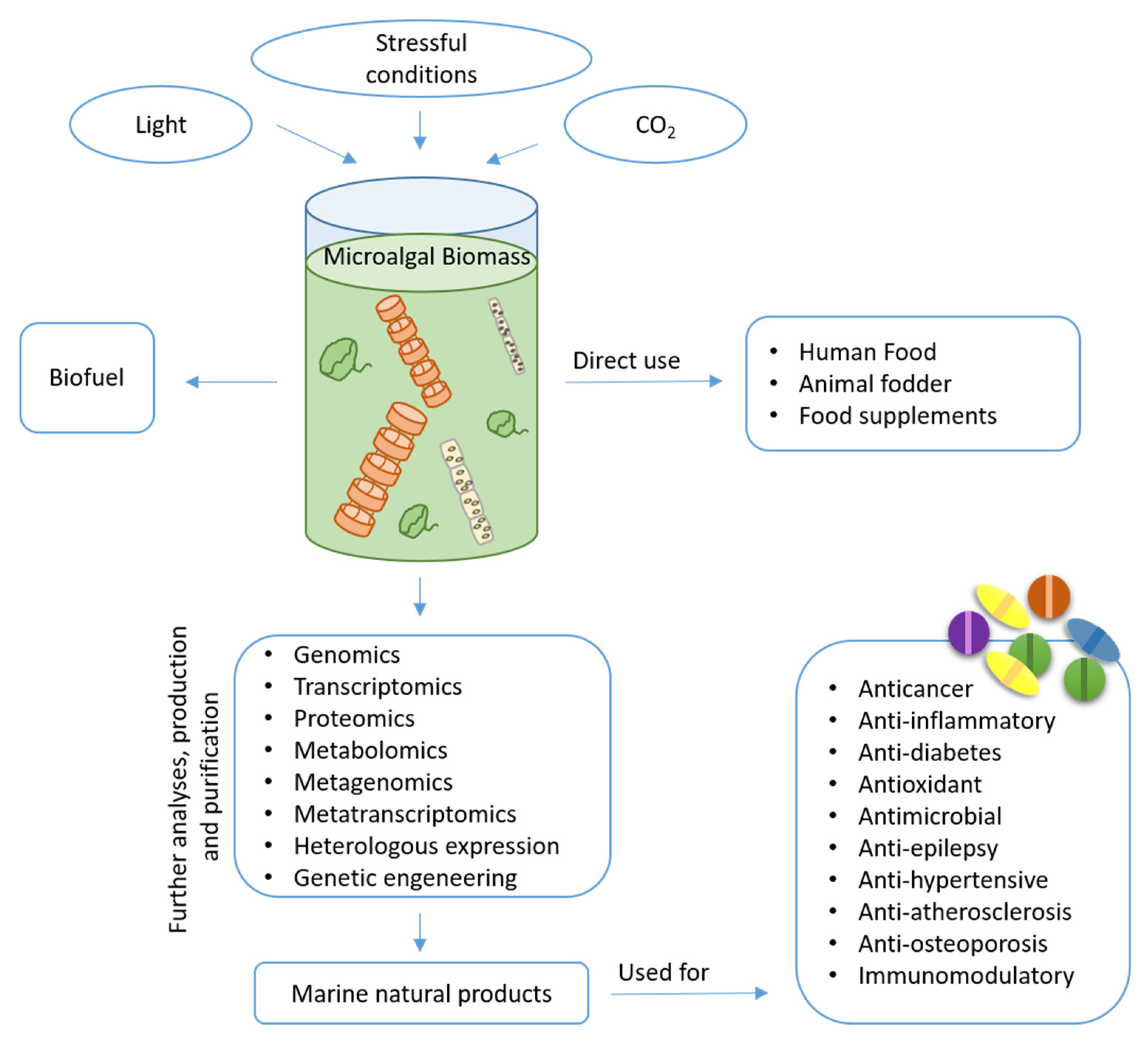
1. Introduction
2. Microalgal Bioactive Compounds
2.1. Pigments
2.1.1. Fucoxanthin
2.1.2. β-Carotene
2.1.3. Astaxanthin
2.1.4. Violaxanthin
2.1.5. Lutein and Minor Carotenoids
| Compound | Microalgae | Bioactivity | Concentration | Mechanism of Action | Ref. |
|---|---|---|---|---|---|
| Fucoxanthin | Phaeodactylm tricornutum | Anti-obesity: (C57B/6 mice a high-fat diet). | In vivo: 771.1 and 1273.18 μg/g of diet for 15 and 30% PT powder. | Activation of AMPK and HMGCR pathways. | [34] |
| Anticancer: (Caco-2, HeLa and HepG2). | In vitro: Dose–dependent manner (0.1, 1, 10 and 50 μg/mL). | Increased the caspase activity up to 4.6-fold. | [36] | ||
| Antioxidant: (Human primary blood cells) | In vitro: IC50 value of 201.2 ± 21.4 μg/mL. | Inhibit the oxidative burst in human PMLs, scavenge radicals and increase the GSH to GSSH ratio. | [36] | ||
| Anti-diabetic: (3T3-L1 cells) | In vitro: IC50 value of 0.68 mmol/L and 4.75 mmol/L | Inhibiting carbohydrate-hydrolyzing enzymes and lipid accumulation. | [47] | ||
| β-carotene | Dunaliella salina | Antioxidant: (Albino rats of either sex of the Wister strain weighing 180–220 gm) | In vivo: 125 μg/Kg and 250 μg/Kg. | Restores the activity of hepatic enzymes. | [58] |
| Anticancer: (MCF-7 breast cancer) | In vitro: 250 μg/mL | Remains unclear. | [61] | ||
| Astaxanthin | Haematococcus pluvialis | Antioxidant: (Thirty-five healthy adults age 35–69 years) | In vivo: 6 mg/day | Remains unclear. | [68] |
| Anti-inflammatory: (young healthy adult human female). | In vivo: 2 or 8 mg/daily. | Shifting the T-lymphocyte response from a Th1 response dominated by IFN-γ to a Th1/Th2 response dominated by IFN- γ and IL-4. | [72] | ||
| Anti-diabetic: (C57BL/KsJ-db/db mice). | In vivo: 10 mg/mouse/day. | Preservation of β cell function. | [73] | ||
| Anticancer: (HCT116 colon cancer). | In vitro: 25 μg/mL. | Increase of p53, p21WAF-1/CIP-1 and p27 expression, decrease of cyclin D1 expression and AKT phosphorylation. | [79] | ||
| Violaxanthin | Dunaliella tertiolecta | Anticancer: (MCF-7, LNcaP cell lines) | In vitro: From 0.1 μg/mL to 40 μg/mL. | Phophatidylserines translocation. | [80] |
| Chlorella ellipsoidea | Anti-inflammatory: (Raw 264.7 cell lines). | In vitro: 60 μM | Inhibition of NF-κB | [81] | |
| Lutein | Chlorella vulgaris | Anticancer: (HCT116 cell lines). | In vitro: IC50 values of 40.41 ± 4.43 μg/mL. | Apoptosis-inducing activity. | [84] |
| Antioxidant: (human neuroblastoma cells, rat trachea epitelial cells). | In vitro: 40–50 μM. | Remain unclear | [92] |
2.2. Polyphenols
2.3. Polysaccharides
| Compound | Microalgae | Bioactivity | Concentration | Mechanism of Action | Ref. |
|---|---|---|---|---|---|
| Polyphenols | |||||
| Flavonoids and alkaloid | Scenedesmus bajacalifornicus BBKLP-07 | Antioxidant | In vitro: Radical scavenging effects of 60.45 and 63.57% at 50 μg/mL. | Reduction of methanolic solution of colored free radical DPPH by free radical scavengers. | [103] |
| Anti-diabetic | In vitro: IC50 80.21 μg/mL | Inhibitory activity of α-amylase. | [103] | ||
| Anti-inflammatory | In vitro: 67.35% protein denaturation at 100 μg/mL | [103] | |||
| Polysaccharides | |||||
| Calcium spirulan and dextran sulphate | Porphyridium cruentum | Anti-viral | In vitro: IC50 9.3 and 9.6 μm/mL. | Inhibitory effect on the replication of HIV-1 and HSV-1. | [108] |
| Porphyridium UTEX 637 | Antioxidant: (3T3 cells) | In vitro: 7.5 μg/well: 41.4% of inhibition. 19 μg/well: 65% of inhibition. 37.5 μg/well: 79.7% of inhibition. | Autooxidation of linoleic acid, and oxidative damage to 3T3. | [111] | |
| Chlorella stigmatophora and Phaeodactylm tricornutum | Anti-inflammatory: Female C57BI mice | In vivo: Intraperitoneally crude polysaccharide extract 5 or 10 mg/kg. | Colloidal carbon clearance (in vivo assay) Phagocytic activity (in vitro assay) | [113] | |
| Porphyridium cruentum | Anticancer: Golden Syrian race Graffi Myeloid tumor | In vitro: Dose dependent manner at different time. | Increased both, spreading and phagocytic activity of peritoneal macrophages in healthy and GTBH in a dose dependent manner. | [116] |
2.4. Lipids
| Compound | Microalgae | Bioactivity | Concentration | Ref. |
|---|---|---|---|---|
| Fatty acids | ||||
| Palmitoleic acid and hexadecatrienoic acid (HTA) | Phaeodactylum tricornutum | Antimicrobial: Palmitoleic acid inhibited the growth of staphylococcal species, including multidrug-resistant Staphylococcus aureus MRSA. HTA inhibited the growth of Gram-positive and Gram-negative | In vitro: IC50 values of palmitoleic acid and HTA against S. aureus were 10–20 and 20–40 μM, respectively | [128] |
| EPA-enriched fraction | Cocconeis scutellum Ehrenberg | Antiproliferative activity on breast carcinoma (BT20) cells, activation of caspases-3 and caspase-8, and cell cycle progression block from S to G2-M phases | In vitro: Tests at 0–1.7 and 0.1–4 μg/well | [127] |
| EPA and DHA | Chlorophyceae and Eustigmatophyceae, species names not specified | Antidiabetes: increase of the cytokines IL17A, IL-12, IL-4, IL-6, IL-10, and TGF-β but the decrease of IFN-ɣ, TNF-α, and IL-5 in diabetic mice | In vivo: 1 mg/g of mouse weight. | [129] |
| Polar lipids | ||||
| Two monogalactosyldiacyl glycerolipids (MGDGs | Phaeodactylum tricornutum | Pro-apoptotic activity on immortal mouse epithelial cell lines (W2 cells). | In vitro: 52 μM and 64 μM | [130] |
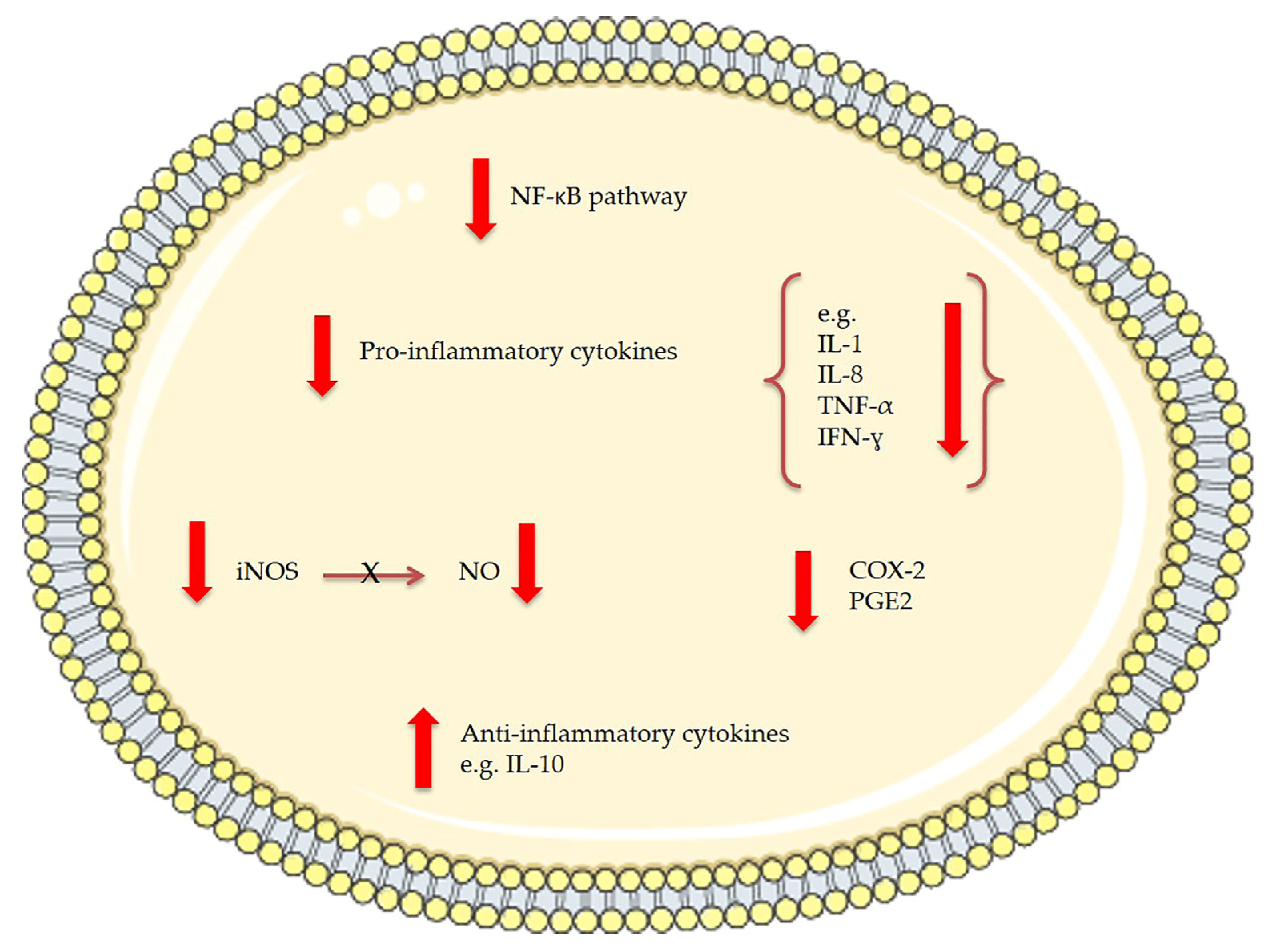
| Two MGDGs | Tetraselmis chuii | Anti-inflammatory: reduce nitric oxide (NO) production and inducible nitric oxide synthase (iNOS) protein levels in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophage cells | In vitro: 50 μg/mL | [131] |
| MGDGs and digalactosyl diacylglycerolipids (DGDGs) | Nannochloropsis granulata | Anti-inflammatory: reduce nitric oxide (NO) production and inducible nitric oxide synthase (iNOS) protein levels in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophage cells | In vitro: 50 μg/mL | [132] |
| sulfoquinovosyl diacylglycerolipids (SQDGs) | Tetradesmus lagerheimii, Scenedesmus producto-capitatus, Pectinodesmus pectinatus, Tetradesmus wisconsinensis | inhibit the glutaminyl cyclase (QC) | In vitro: 0.2 mg/mL | [133] |
| A synthetic sulfolipid (Sulfavant) SQDG18 | Thalassiosira weissflogii CCMP1336 | It triggered an effective immune response against cancer cells to improve dendritic cell (DC) maturation and increase CD83-positive DC. SQDG18 stimulated the production of the pro-inflammatory cytokines IL-12 and INF-ɣ | [134,135] | |
| MGDGs DGDGs | Chlorella vulgaris | Antitumor: Epstein-Barr virus-associated early antigen (EBV-EA) activation on Raji cells induced by 12-O-tetradecanoylphorbol-13- acetate (TPA) | In vitro: 500–2500 mol ratio/TPA | [136] |
| SQDG | Porphyridium cruentum | Inhibition of the growth cancer cell-lines on human colon (DLD-1), breast (MCF-7), prostate adenocarcinoma (PC-3) and malignant melanoma (M4 Beu) cancer cells; | In vitro: IC50: 20–46 µg/mL | [137] |
| SQDG | Porphyridium cruentum | Inhibition of DNA α-polymerase; | In vitro: IC50: 378 µg/mL | [137] |
| Lipid extracts containing EPA, SQDG, MGDG, DGDG and others | Pavlova lutheri | Down-regulation of the production of cytokine IL-6 in lipopolysaccharide (LPS)-stimulated human THP-1 macrophages; Down-regulation of Toll-like receptor 8, Toll-like receptor 1, TNF receptor-associated factor 5, Mitogen-activated protein kinase 1; Increase of Prostaglandin E receptor 1 | In vitro: 3 µg/mL total fatty acids | [138] |
| Oil containing eicosapentaenoic acid (EPA), phospholipids and glycolipids | Nannochloropsis oculata | Glycolipids in the algal oil may increase Long-chain omega-3 polyunsaturated fatty acids (LC n-3 PUFA) bioavailability | In vivo: 5 mL algal oil per kg body weight each day per 7 days in rats | [139] |
| Oxilipins | ||||
| PUAs (2-trans,4-trans-decadienal, 2-trans,4-trans-octadienal and 2-trans,4-trans-heptadienal) | Pure compounds from Sigma-Aldrich Inc. | Anticancer (COLO 205 and A549 cells) | In vitro: 2–10 µM | [140] |
| 2-trans-4-cis-7-cis-decatrienal, 2-trans-4-trans-7-cis-decatrienal and 2-trans-4-trans-decadienal | Thalassiosira rotula, Skeletonema costatum and Pseudonitzschia delicatissima | Anticancer (Caco-2 cells) | In vitro: 11–17 µg/mL | [141] |
| Oxylipin-containing lyophilised (OLM) biomass | Chlamydomonas debaryana | Anti-inflammatory activities on a recurrent 2,4,6-trinitrobenzenesulfonic acid (TNBS)-induced colitis mice model; significant decrease of TNF-α, iNOS and COX-2 | In vivo: 300 and 600 mg/kg | [142] |
| Oxylipin 13-HOTE | Chlamydomonas debaryana, | Anticancer: UACC-62 (melanoma) than towards HT-29 (colon adenocarcinoma) cells | In vitro: 68.2 ± 0.2 µM (UACC-62) >100 µM (HT29) | [142] |
| 15-HEPE | Nannochloropsis gaditana | Anticancer: UACC-62 (melanoma) than towards HT-29 (colon adenocarcinoma) cells | In vitro: 78.8 ± 4.6 µM (UACC-62) >100 µM (HT29) | [142] |
| Steroids | ||||
| Ergosterol, 7-Dehydroporiferasterol, Ergosterol peroxide, 7-Dehydroporiferasterol peroxide, 7-oxocholesterol | Chlorella vulgaris | Anti-inflammatory (12-O-tetradecanoylphorbol-13-acetate (TPA)-induced inflammation in mice) | In vivo: 0.2–0.7 mg/ear | [143] |
| Ergosterol peroxide | Chlorella vulgaris | Anticancer (TPA tumor-promoting effect in 7,12-dimethylbenz[a]anthracene-initiated mice) | In vivo: 2 μmol | [143] |
| Ergosterol, 7-Dehydroporiferasterol, mixture | Dunaliella tertiolecta | Anti-inflammatory activity on peripheral blood mononuclear cells (PBMC; isolated from sheep) treated with Concanavalin A (Con A) and lipopolysaccharide (LPS); increase of the anti-inflammatory cytokine interleukin 10 (IL-10) | In vitro: 0.4 mg/mL mixture; 0.8 mg/mL for ergosterol alone | [144] |
| Ergosterol, 7-Dehydroporiferasterol | Dunaliella tertiolecta | Neuromodulatory action was found in selective brain areas of rats | In vivo: 20–30 mg/kg | [145] |
| 24-Oxocholesterol acetate, Ergost-5-en-3β-ol, Cholest-5-en-24-1,3-(acetyloxy)-, 3β-ol and others | Isochrysis galbana | Antituberculosis | In vitro: Minimum inhibitory concentration of 50–60 μg/mL | [146] |
| Stigmasterol, 5β-Hydroxysitostanol | Navicula incerta | Anti-cancer in human hepatoma HepG2 cells | 40%, 43% and 54% toxicity at 5, 10 and 20 μM, respectively | [147,148] |
2.5. Glycolipids
2.6. Steroids
2.7. Oxylipins
2.8. Proteins and Peptides
| Compound | Microalgae | Mechanism of Action | Ref. |
|---|---|---|---|
| VECYGPNRPQF | Chlorella vulgaris | Superoxide radical quenching | [178] |
| LNGDVW | Chlorella ellipsoidea | Free radical scavenging | [179] |
| PGWNQWFL, VEVLPPAEL | Navicula incerta | Cytotoxicity in HepG2/CYP2E1 cells | [180] |
| VECYGPNRPQF | Chlorella vulgaris | AGS cells | [181] |
| polypeptide CPAP | Chlorella pyrenoidosa | HepG2 cells | [182] |
| VECYGPNRPQF | Chlorella vulgaris | ACE inhibitor | [178] |
| GMNNLTP; LEQ | Nannochloropsis oculata | ACE inhibitor | [183] |
| VECYGPNRPQF | Chlorella sp | Gene expression inhibition of vascular adhesion molecules (E-selectin, ICAM, VCAM, MCP-1 and ET-1) | [186] |
| Chlorella derived peptide | Chlorella sp. | Inhibition of MMP-1, CYR61 | [184] |
| MPDW | Nannochloropsis oculata | Increase of ALP, osteocalcin, collagen type I, BMP-2, BMP2/4; phosphorylation of MAPK/Smad pathways | [185] |
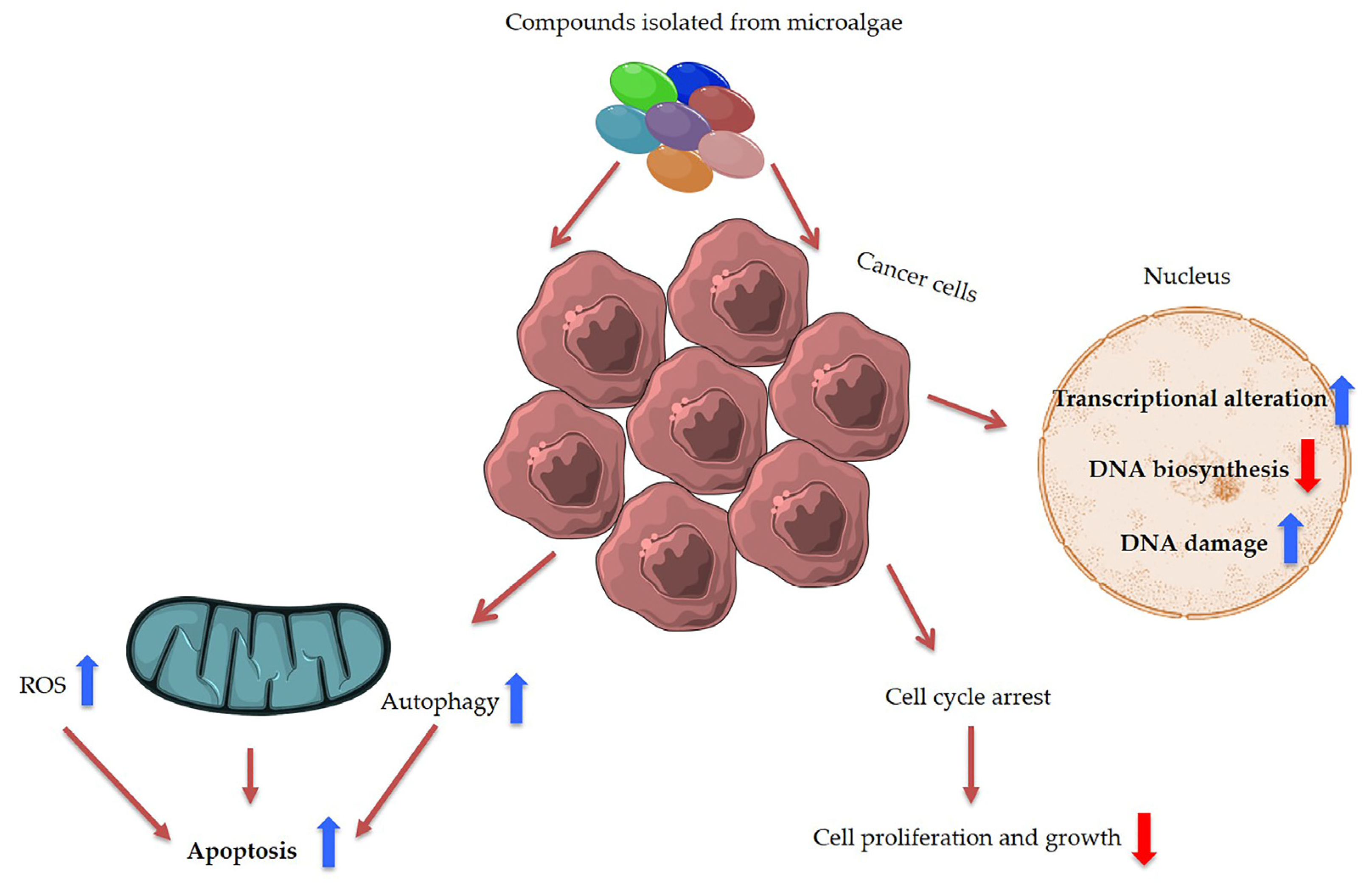
2.9. Bioactive Polyketides and Macrolides
Toxins with Potential Human Health Applications
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal Triacylglycerols as Feedstocks for Biofuel Production: Perspectives and Advances. Plant J. 2008, 54, 19. [Google Scholar] [CrossRef]
- Microalgae Immobilization: Current Techniques and Uses-ScienceDirect. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0960852407004567 (accessed on 17 February 2021).
- Lauritano, C.; Andersen, J.H.; Hansen, E.; Albrigtsen, M.; Escalera, L.; Esposito, F.; Helland, K.; Hanssen, K.Ø.; Romano, G.; Ianora, A. Bioactivity Screening of Microalgae for Antioxidant, Anti-Inflammatory, Anticancer, Anti-Diabetes, and Antibacterial Activities. Front. Mar. Sci. 2016, 3, 68. [Google Scholar] [CrossRef]
- Romano, G.; Costantini, M.; Sansone, C.; Lauritano, C.; Ruocco, N.; Ianora, A. Marine Microorganisms as a Promising and Sustainable Source of Bioactive Molecules. Mar. Environ. Res. 2017, 128, 58–69. [Google Scholar] [CrossRef]
- Andrade, K.M.; Lauritano, C.; Romano, G.; Ianora, A. Marine Microalgae with Anti-Cancer Properties. Mar. Drugs 2018, 16, 165. [Google Scholar] [CrossRef]
- Saide, A.; Lauritano, C.; Ianora, A. Pheophorbide a: State of the Art. Mar. Drugs 2020, 18, 257. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, C.; Helland, K.; Riccio, G.; Andersen, J.H.; Ianora, A.; Hansen, E.H. Lysophosphatidylcholines and Chlorophyll-Derived Molecules from the Diatom Cylindrotheca closterium with Anti-Inflammatory Activity. Mar. Drugs 2020, 18, 166. [Google Scholar] [CrossRef] [PubMed]
- Elagoz, A.M.; Ambrosino, L.; Lauritano, C. De Novo Transcriptome of the Diatom Cylindrotheca closterium Identifies Genes Involved in the Metabolism of Anti-Inflammatory Compounds. Sci. Rep. 2020, 10, 4138. [Google Scholar] [CrossRef]
- Lauritano, C.; Ianora, A. Marine Organisms with Anti-Diabetes Properties. Mar. Drugs 2016, 14, 220. [Google Scholar] [CrossRef]
- Giordano, D.; Costantini, M.; Coppola, D.; Lauritano, C.; Pons, L.N.; Ruocco, N.; di Prisco, G.; Ianora, A.; Verde, C. Biotechnological Applications of Bioactive Peptides from Marine Sources. Adv. Microb. Physiol. 2018, 51. [Google Scholar] [CrossRef]
- Lauritano, C. First Identification of Marine Diatoms with Anti-Tuberculosis Activity. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Brillatz, T.; Lauritano, C.; Jacmin, M.; Khamma, S.; Marcourt, L.; Righi, D.; Romano, G.; Esposito, F.; Ianora, A.; Queiroz, E.F.; et al. Zebrafish-Based Identification of the Antiseizure Nucleoside Inosine from the Marine Diatom Skeletonema marinoi. PLoS ONE 2018, 13, e0196195. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; Lauritano, C. Microalgae with Immunomodulatory Activities. Mar. Drugs 2019, 18, 2. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; De Luca, D.; Lauritano, C. Monogalactosyldiacylglycerol and Sulfolipid Synthesis in Microalgae. Mar. Drugs 2020, 18, 237. [Google Scholar] [CrossRef] [PubMed]
- Ingebrigtsen, R.A.; Hansen, E.; Andersen, J.H.; Eilertsen, H.C. Light and Temperature Effects on Bioactivity in Diatoms. J. Appl. Phycol. 2016, 28, 939–950. [Google Scholar] [CrossRef] [PubMed]
- Osvik, R.D.; Andersen, J.H.; Eilertsen, H.C.; Geneviere, A.-M.; Hansen, E.H. Bioactivity of a Marine Diatom (Porosira glacialis [Grunow] Jörgensen 1905) Cultivated with and Without Factory Smoke CO2. Ind. Biotechnol. 2021, 17, 38–48. [Google Scholar] [CrossRef]
- Lauritano, C.; De Luca, D.; Ferrarini, A.; Avanzato, C.; Minio, A.; Esposito, F.; Ianora, A. De Novo Transcriptome of the Cosmopolitan Dinoflagellate Amphidinium carterae to Identify Enzymes with Biotechnological Potential. Sci. Rep. 2017, 7, 11701. [Google Scholar] [CrossRef]
- Lauritano, C.; De Luca, D.; Amoroso, M.; Benfatto, S.; Maestri, S.; Racioppi, C.; Esposito, F.; Ianora, A. New Molecular Insights on the Response of the Green Alga Tetraselmis suecica to Nitrogen Starvation. Sci. Rep. 2019, 9, 3336. [Google Scholar] [CrossRef] [PubMed]
- Vingiani, G.M.; De Luca, P.; Ianora, A.; Dobson, A.D.W.; Lauritano, C. Microalgal Enzymes with Biotechnological Applications. Mar. Drugs 2019, 17, 459. [Google Scholar] [CrossRef] [PubMed]
- Osvik, R.D.; Ingebrigtsen, R.A.; Norrbin, M.F.; Andersen, J.H.; Eilertsen, H.C.; Hansen, E.H. Adding Zooplankton to the OSMAC Toolkit: Effect of Grazing Stress on the Metabolic Profile and Bioactivity of a Diatom. Mar. Drugs 2021, 19, 87. [Google Scholar] [CrossRef] [PubMed]
- Cutignano, A.; Nuzzo, G.; Ianora, A.; Luongo, E.; Romano, G.; Gallo, C.; Sansone, C.; Aprea, S.; Mancini, F.; D’Oro, U.; et al. Development and Application of a Novel SPE-Method for Bioassay-Guided Fractionation of Marine Extracts. Mar. Drugs 2015, 13, 5736–5749. [Google Scholar] [CrossRef]
- Lauritano, C.; Ferrante, M.I.; Rogato, A. Marine Natural Products from Microalgae: An -Omics Overview. Mar. Drugs 2019, 17, 269. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine Natural Products. Nat. Prod. Rep. 2018, 35, 8–53. [Google Scholar] [CrossRef]
- Dolganyuk, V.; Belova, D.; Babich, O.; Prosekov, A.; Ivanova, S.; Katserov, D.; Patyukov, N.; Sukhikh, S. Microalgae: A Promising Source of Valuable Bioproducts. Biomolecules 2020, 10, 1153. [Google Scholar] [CrossRef] [PubMed]
- Ambati, R.R.; Gogisetty, D.; Aswathanarayana, R.G.; Ravi, S.; Bikkina, P.N.; Bo, L.; Yuepeng, S. Industrial Potential of Carotenoid Pigments from Microalgae: Current Trends and Future Prospects. Crit. Rev. Food Sci. Nutr. 2019, 59, 1880–1902. [Google Scholar] [CrossRef] [PubMed]
- Bowler, C.; Allen, A.E.; Badger, J.H.; Grimwood, J.; Jabbari, K.; Kuo, A.; Maheswari, U.; Martens, C.; Maumus, F.; Otillar, R.P. The Phaeodactylum Genome Reveals the Evolutionary History of Diatom Genomes. Nature 2008, 456, 239–244. [Google Scholar] [CrossRef]
- Kuczynska, P.; Jemiola-Rzeminska, M.; Strzalka, K. Photosynthetic Pigments in Diatoms. Mar. Drugs 2015, 13, 5847–5881. [Google Scholar] [CrossRef] [PubMed]
- Manfellotto, F.; Stella, G.R.; Falciatore, A.; Brunet, C.; Ferrante, M.I. Engineering the Unicellular Alga Phaeodactylum tricornutum for Enhancing Carotenoid Production. Antioxidants 2020, 9, 757. [Google Scholar] [CrossRef] [PubMed]
- Lohr, M.; Wilhelm, C. Xanthophyll Synthesis in Diatoms: Quantification of Putative Intermediates and Comparison of Pigment Conversion Kinetics with Rate Constants Derived from a Model. Planta 2001, 212, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Dambek, M.; Eilers, U.; Breitenbach, J.; Steiger, S.; Büchel, C.; Sandmann, G. Biosynthesis of Fucoxanthin and Diadinoxanthin and Function of Initial Pathway Genes in Phaeodactylum tricornutum. J. Exp. Bot. 2012, 63, 5607–5612. [Google Scholar] [CrossRef]
- Veith, T.; Büchel, C. The Monomeric Photosystem I-Complex of the Diatom Phaeodactylum tricornutum Binds Specific Fucoxanthin Chlorophyll Proteins (FCPs) as Light-Harvesting Complexes. Biochim. Biophys. Acta BBA-Bioenerg. 2007, 1767, 1428–1435. [Google Scholar] [CrossRef]
- Tsukui, T.; Konno, K.; Hosokawa, M.; Maeda, H.; Sashima, T.; Miyashita, K. Fucoxanthin and Fucoxanthinol Enhance the Amount of Docosahexaenoic Acid in the Liver of KKAy Obese/Diabetic Mice. J. Agric. Food Chem. 2007, 55, 5025–5029. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, N.; Gemello, E.; Gammone, M.; de Girolamo, M.; Ficoneri, C.; Riccioni, G. Fucoxantin: A Treasure from the Sea. Mar. Drugs 2012, 10, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, S.M.; Cha, K.H.; Mok, I.-K.; Koo, S.Y.; Pan, C.-H.; Lee, J.K. Evaluation of the Anti-Obesity Effect of the Microalga Phaeodactylum tricornutum. Appl. Biol. Chem. 2016, 59, 283–290. [Google Scholar] [CrossRef]
- Kotake-Nara, E.; Kushiro, M.; Zhang, H.; Sugawara, T.; Miyashita, K.; Nagao, A. Carotenoids Affect Proliferation of Human Prostate Cancer Cells. J. Nutr. 2001, 131, 3303–3306. [Google Scholar] [CrossRef] [PubMed]
- Neumann, U.; Derwenskus, F.; Flaiz Flister, V.; Schmid-Staiger, U.; Hirth, T.; Bischoff, S. Fucoxanthin, A Carotenoid Derived from Phaeodactylum tricornutum Exerts Antiproliferative and Antioxidant Activities In Vitro. Antioxidants 2019, 8, 183. [Google Scholar] [CrossRef]
- Murakami, A.; Nakashima, M.; Koshiba, T.; Maoka, T.; Nishino, H.; Yano, M.; Sumida, T.; Kyung Kim, O.; Koshimizu, K.; Ohigashi, H. Modifying Effects of Carotenoids on Superoxide and Nitric Oxide Generation from Stimulated Leukocytes. Cancer Lett. 2000, 149, 115–123. [Google Scholar] [CrossRef]
- Heo, S.-J.; Ko, S.-C.; Kang, S.-M.; Kang, H.-S.; Kim, J.-P.; Kim, S.-H.; Lee, K.-W.; Cho, M.-G.; Jeon, Y.-J. Cytoprotective Effect of Fucoxanthin Isolated from Brown Algae Sargassum siliquastrum against H2O2-Induced Cell Damage. Eur. Food Res. Technol. 2008, 228, 145–151. [Google Scholar] [CrossRef]
- Zheng, J.; Piao, M.J.; Keum, Y.S.; Kim, H.S.; Hyun, J.W. Fucoxanthin Protects Cultured Human Keratinocytes against Oxidative Stress by Blocking Free Radicals and Inhibiting Apoptosis. Biomol. Ther. 2013, 21, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Zhang, Y.; Ruan, J.; Yang, Z.; Wang, C.; Hong, Z.; Zuo, Z. Protective Effects of Fucoxanthin and Fucoxanthinol against Tributyltin-Induced Oxidative Stress in HepG2 Cells. Environ. Sci. Pollut. Res. 2018, 25, 5582–5589. [Google Scholar] [CrossRef]
- Wang, X.; Cui, Y.; Qi, J.; Zhu, M.; Zhang, T.; Cheng, M.; Liu, S.; Wang, G. Fucoxanthin Exerts Cytoprotective Effects against Hydrogen Peroxide-Induced Oxidative Damage in L02 Cells. BioMed Res. Int. 2018, 2018, 1–11. [Google Scholar] [CrossRef]
- Sangeetha, R.K.; Bhaskar, N.; Baskaran, V. Comparative Effects of β-Carotene and Fucoxanthin on Retinol Deficiency Induced Oxidative Stress in Rats. Mol. Cell. Biochem. 2009, 331, 59–67. [Google Scholar] [CrossRef]
- Xia, S.; Wang, K.; Wan, L.; Li, A.; Hu, Q.; Zhang, C. Production, Characterization, and Antioxidant Activity of Fucoxanthin from the Marine Diatom Odontella aurita. Mar. Drugs 2013, 11, 2667–2681. [Google Scholar] [CrossRef]
- Hosokawa, M.; Miyashita, T.; Nishikawa, S.; Emi, S.; Tsukui, T.; Beppu, F.; Okada, T.; Miyashita, K. Fucoxanthin Regulates Adipocytokine MRNA Expression in White Adipose Tissue of Diabetic/Obese KK-Ay Mice. Arch. Biochem. Biophys. 2010, 504, 17–25. [Google Scholar] [CrossRef]
- Park, H.J.; Lee, M.K.; Park, Y.B.; Shin, Y.C.; Choi, M.S. Beneficial Effects of Undaria Pinnatifida Ethanol Extract on Diet-Induced-Insulin Resistance in C57BL/6J Mice. Food Chem. Toxicol. 2011, 49, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Murakami-Funayama, K.; Miyashita, K. Maeda Anti-Obesity and Anti-Diabetic Effects of Fucoxanthin on Diet-Induced Obesity Conditions in a Murine Model. Mol. Med. Rep. 2009, 2, 897–902. [Google Scholar] [CrossRef]
- Kawee-Ai, A.; Kim, A.T.; Kim, S.M. Inhibitory Activities of Microalgal Fucoxanthin against α-Amylase, α-Glucosidase, and Glucose Oxidase in 3T3-L1 Cells Linked to Type 2 Diabetes. J. Oceanol. Limnol. 2019, 37, 928–937. [Google Scholar] [CrossRef]
- Rasmussen, R.S.; Morrissey, M.T. Marine Biotechnology for Production of Food Ingredients. In Advances in Food and Nutrition Research; Elsevier: Amsterdam, The Netherlands, 2007; Volume 52, pp. 237–292. ISBN 978-0-12-373711-3. [Google Scholar]
- Prieto, A.; Pedro Cañavate, J.; García-González, M. Assessment of Carotenoid Production by Dunaliella salina in Different Culture Systems and Operation Regimes. J. Biotechnol. 2011, 151, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Crupi, P.; Toci, A.T.; Mangini, S.; Wrubl, F.; Rodolfi, L.; Tredici, M.R.; Coletta, A.; Antonacci, D. Determination of Fucoxanthin Isomers in Microalgae (Isochrysis sp.) by High-Performance Liquid Chromatography Coupled with Diode-Array Detector Multistage Mass Spectrometry Coupled with Positive Electrospray Ionization: Fucoxanthin Isomers in Isochrysis sp. Rapid Commun. Mass Spectrom. 2013, 27, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Harvey, P.J.; Ben-Amotz, A. Towards a Sustainable Dunaliella salina Microalgal Biorefinery for 9-Cis β-Carotene Production. Algal Res. 2020, 50, 102002. [Google Scholar] [CrossRef]
- Goiris, K.; Muylaert, K.; Fraeye, I.; Foubert, I.; De Brabanter, J.; De Cooman, L. Antioxidant Potential of Microalgae in Relation to Their Phenolic and Carotenoid Content. J. Appl. Phycol. 2012, 24, 1477–1486. [Google Scholar] [CrossRef]
- Grune, T.; Lietz, G.; Palou, A.; Ross, A.C.; Stahl, W.; Tang, G.; Thurnham, D.; Yin, S.; Biesalski, H.K. β-Carotene Is an Important Vitamin A Source for Humans. J. Nutr. 2010, 140, 2268S–2285S. [Google Scholar] [CrossRef]
- Stahl, W.; Schwarz, W.; Sundquist, A.R.; Sies, H. Cis-Trans Isomers of Lycopene and β-Carotene in Human Serum and Tissues. Arch. Biochem. Biophys. 1992, 294, 173–177. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant Activity of Carotenoids. Mol. Aspects Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Schmidt, R. Deactivation of O 2 (1Δg) Singlet Oxygen by Carotenoids: Internal Conversion of Excited Encounter Complexes. J. Phys. Chem. A 2004, 108, 5509–5513. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. Beta-carotene: An unusual type of lipid antioxidant. Science 1984, 224, 569–573. [Google Scholar] [CrossRef]
- Murthy, K.N.C.; Vanitha, A.; Rajesha, J.; Swamy, M.M.; Sowmya, P.R.; Ravishankar, G.A. In Vivo Antioxidant Activity of Carotenoids from Dunaliella salina—A Green Microalga. Life Sci. 2005, 76, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Nishino, H.; Murakoshi, M.; Ii, T.; Takemura, M.; Kuchide, M.; Kanazawa, M.; Mou, X.Y.; Wada, S.; Masuda, M.; Ohsaka, Y. Carotenoids in cancer chemoprevention. Cancer Metastasis Rev. 2002, 21, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-D.; Liu, C.; Bronson, R.T.; Smith, D.E.; Krinsky, N.I.; Russell, R.M. Retinoid Signaling and Activator Protein-1 Expression in Ferrets Given β-Carotene Supplements and Exposed to Tobacco Smoke. J. Natl. Cancer Inst. 1999, 91, 60–66. [Google Scholar] [CrossRef]
- Singh, P.; Baranwal, M.; Reddy, S.M. Antioxidant and Cytotoxic Activity of Carotenes Produced by Dunaliella salina under Stress. Pharm. Biol. 2016, 54, 2269–2275. [Google Scholar] [CrossRef]
- Amaro, H.M.; Barros, R.; Guedes, A.C.; Sousa-Pinto, I.; Malcata, F.X. Microalgal Compounds Modulate Carcinogenesis in the Gastrointestinal Tract. Trends Biotechnol. 2013, 31, 92–98. [Google Scholar] [CrossRef]
- Lorenz, R.T.; Cysewski, G.R. Commercial Potential for Haematococcus Microalgae as a Natural Source of Astaxanthin. Trends Biotechnol. 2000, 18, 160–167. [Google Scholar] [CrossRef]
- Seabra, L.M.J.; Pedrosa, L.F.C. Astaxanthin: Structural and Functional Aspects. Rev. Nutr. 2010, 23, 1041–1050. [Google Scholar] [CrossRef]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial Applications of Microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Palozza, P.; Krinsky, N.I. Astaxanthin and Canthaxanthin Are Potent Antioxidants in a Membrane Model. Arch. Biochem. Biophys. 1992, 297, 291–295. [Google Scholar] [CrossRef]
- Ranga Rao, A.; Baskaran, V.; Sarada, R.; Ravishankar, G.A. In Vivo Bioavailability and Antioxidant Activity of Carotenoids from Microalgal Biomass—A Repeated Dose Study. Food Res. Int. 2013, 54, 711–717. [Google Scholar] [CrossRef]
- Spiller, G.A.; Dewell, A. Safety of an Astaxanthin-Rich Haematococcus pluvialis Algal Extract: A Randomized Clinical Trial. J. Med. Food 2003, 6, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.H.; Lee, Y.K. Effect of Total Secondary Carotenoids Extracts from Chlorococcum sp. on Helicobacter pylori-Infected BALB/c Mice. Int. Immunopharmacol. 2003, 3, 979–986. [Google Scholar] [CrossRef]
- Bennedsen, M.; Wang, X.; Willén, R.; Wadström, T.; Andersen, L.P. Treatment of H. Pylori Infected Mice with Antioxidant Astaxanthin Reduces Gastric Inflammation, Bacterial Load and Modulates Cytokine Release by Splenocytes. Immunol. Lett. 2000, 70, 185–189. [Google Scholar] [CrossRef]
- Kamath, B.S.; Srikanta, B.M.; Dharmesh, S.M.; Sarada, R.; Ravishankar, G.A. Ulcer Preventive and Antioxidative Properties of Astaxanthin from Haematococcus pluvialis. Eur. J. Pharmacol. 2008, 590, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Chyun, J.; Kim, Y.; Line, L.L.; Chew, B.P. Astaxanthin Decreased Oxidative Stress and Inflammation and Enhanced Immune Response in Humans. Nutr. Metab. 2010, 7, 18. [Google Scholar] [CrossRef]
- Uchiyama, K.; Naito, Y.; Hasegawa, G.; Nakamura, N.; Takahashi, J.; Yoshikawa, T. Astaxanthin Protects β-Cells against Glucose Toxicity in Diabetic Db/Db Mice. Redox Rep. 2002, 7, 290–293. [Google Scholar] [CrossRef]
- Otton, R.; Marin, D.P.; Bolin, A.P.; dos Santos, R.D.C.M.; Polotow, T.G.; Sampaio, S.C.; de Barros, M.P. Astaxanthin Ameliorates the Redox Imbalance in Lymphocytes of Experimental Diabetic Rats. Chem. Biol. Interact. 2010, 186, 306–315. [Google Scholar] [CrossRef]
- Naito, Y.; Uchiyama, K.; Aoi, W.; Hasegawa, G.; Nakamura, N.; Yoshida, N.; Maoka, T.; Takahashi, J.; Yoshikawa, T. Prevention of Diabetic Nephropathy by Treatment with Astaxanthin in Diabetic Db/Db Mice. BioFactors 2004, 20, 49–59. [Google Scholar] [CrossRef]
- Mularczyk, M.; Michalak, I.; Marycz, K. Astaxanthin and Other Nutrients from Haematococcus pluvialis—Multifunctional Applications. Mar. Drugs 2020, 18, 459. [Google Scholar] [CrossRef] [PubMed]
- Landon, R.; Gueguen, V.; Petite, H.; Letourneur, D.; Pavon-Djavid, G.; Anagnostou, F. Impact of Astaxanthin on Diabetes Pathogenesis and Chronic Complications. Mar. Drugs 2020, 18, 357. [Google Scholar] [CrossRef]
- Faraone, I.; Sinisgalli, C.; Ostuni, A.; Armentano, M.F.; Carmosino, M.; Milella, L.; Russo, D.; Labanca, F.; Khan, H. Astaxanthin Anticancer Effects Are Mediated through Multiple Molecular Mechanisms: A Systematic Review. Pharmacol. Res. 2020, 155, 104689. [Google Scholar] [CrossRef]
- Palozza, P.; Torelli, C.; Boninsegna, A.; Simone, R.; Catalano, A.; Mele, M.C.; Picci, N. Growth-Inhibitory Effects of the Astaxanthin-Rich Alga Haematococcus pluvialis in Human Colon Cancer Cells. Cancer Lett. 2009, 283, 108–117. [Google Scholar] [CrossRef]
- Pasquet, V.; Morisset, P.; Ihammouine, S.; Chepied, A.; Aumailley, L.; Berard, J.-B.; Serive, B.; Kaas, R.; Lanneluc, I.; Thiery, V.; et al. Antiproliferative Activity of Violaxanthin Isolated from Bioguided Fractionation of Dunaliella tertiolecta Extracts. Mar. Drugs 2011, 9, 819–831. [Google Scholar] [CrossRef]
- Soontornchaiboon, W.; Joo, S.S.; Kim, S.M. Anti-Inflammatory Effects of Violaxanthin Isolated from Microalga Chlorella ellipsoidea in RAW 264.7 Macrophages. Biol. Pharm. Bull. 2012, 35, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Low, K.L.; Idris, A.; Mohd Yusof, N. Novel Protocol Optimized for Microalgae Lutein Used as Food Additives. Food Chem. 2020, 307, 125631. [Google Scholar] [CrossRef] [PubMed]
- Kotake-Nara, E.; Asai, A.; Nagao, A. Neoxanthin and Fucoxanthin Induce Apoptosis in PC-3 Human Prostate Cancer Cells. Cancer Lett. 2005, 220, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Cha, K.H.; Koo, S.Y.; Lee, D.-U. Antiproliferative Effects of Carotenoids Extracted from Chlorella ellipsoidea and Chlorella vulgaris on Human Colon Cancer Cells. J. Agric. Food Chem. 2008, 56, 10521–10526. [Google Scholar] [CrossRef]
- Alves-Rodrigues, A.; Shao, A. The Science behind Lutein. Toxicol. Lett. 2004, 150, 57–83. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Sevilla, J.M.; Acién Fernández, F.G.; Molina Grima, E. Biotechnological Production of Lutein and Its Applications. Appl. Microbiol. Biotechnol. 2010, 86, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Safafar, H.; van Wagenen, J.; Møller, P.; Jacobsen, C. Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater. Mar. Drugs 2015, 13, 7339–7356. [Google Scholar] [CrossRef] [PubMed]
- Barredo, J.L. Microbial Carotenoids from bacteria and microalgae. In Methods and Protocols; Humana Press: New York, NY, USA, 2012; p. 5. [Google Scholar]
- Gwak, Y.; Hwang, Y.; Wang, B.; Kim, M.; Jeong, J.; Lee, C.-G.; Hu, Q.; Han, D.; Jin, E. Comparative Analyses of Lipidomes and Transcriptomes Reveal a Concerted Action of Multiple Defensive Systems against Photooxidative Stress in Haematococcus pluvialis. J. Exp. Bot. 2014, 65, 4317–4334. [Google Scholar] [CrossRef] [PubMed]
- Granadolorencio, F.; Herrerobarbudo, C.; Acienfernandez, G.; Molinagrima, E.; Fernandezsevilla, J.; Perezsacristan, B.; Blanconavarro, I. In Vitro Bioaccesibility of Lutein and Zeaxanthin from the Microalgae Scenedesmus almeriensis. Food Chem. 2009, 114, 747–752. [Google Scholar] [CrossRef]
- Jin, E.; Polle, J.; Lee, H.K.; Hyun, S.M.; Chang, A. Xanthophylls in Microalgae: From Biosynthesis to Biotechnological Mass Production and Application. J. Microbiol. Biotechnol. 2003, 13, 165–174. [Google Scholar]
- Santocono, M.; Zurria, M.; Berrettini, M.; Fedeli, D.; Falcioni, G. Influence of Astaxanthin, Zeaxanthin and Lutein on DNA Damage and Repair in UVA-Irradiated Cells. J. Photochem. Photobiol. B 2006, 85, 205–215. [Google Scholar] [CrossRef]
- Natrah, F.M.I.; Yusoff, F.M.; Shariff, M.; Abas, F.; Mariana, N.S. Screening of Malaysian Indigenous Microalgae for Antioxidant Properties and Nutritional Value. J. Appl. Phycol. 2007, 19, 711–718. [Google Scholar] [CrossRef]
- Plant Polyphenols as Dietary Antioxidants in Human Health and Disease. Available online: https://www.hindawi.com/journals/omcl/2009/897484/ (accessed on 23 February 2021).
- Fernando, I.P.S.; Nah, J.-W.; Jeon, Y.-J. Potential Anti-Inflammatory Natural Products from Marine Algae. Environ. Toxicol. Pharmacol. 2016, 48, 22–30. [Google Scholar] [CrossRef]
- Besednova, N.N.; Andryukov, B.G.; Zaporozhets, T.S.; Kryzhanovsky, S.P.; Fedyanina, L.N.; Kuznetsova, T.A.; Zvyagintseva, T.N.; Shchelkanov, M.Y. Antiviral Effects of Polyphenols from Marine Algae. Biomedicines 2021, 9, 200. [Google Scholar] [CrossRef] [PubMed]
- Zern, T.L.; Fernandez, M.L. Cardioprotective Effects of Dietary Polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar] [CrossRef] [PubMed]
- Soleas, G.J.; Grass, L.; Josephy, P.D.; Goldberg, D.M.; Diamandis, E.P. A Comparison of the Anticarcinogenic Properties of Four Red Wine Polyphenols. Clin. Biochem. 2006, 39, 492–497. [Google Scholar] [CrossRef]
- Sabu, M.C.; Smitha, K.; Kuttan, R. Anti-Diabetic Activity of Green Tea Polyphenols and Their Role in Reducing Oxidative Stress in Experimental Diabetes. J. Ethnopharmacol. 2002, 83, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Del Mondo, A.; Smerilli, A.; Ambrosino, L.; Albini, A.; Noonan, D.M.; Sansone, C.; Brunet, C. Insights into Phenolic Compounds from Microalgae: Structural Variety and Complex Beneficial Activities from Health to Nutraceutics. Crit. Rev. Biotechnol. 2021, 41, 155–171. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cheng, K.; Wong, C.; Fan, K.; Chen, F.; Jiang, Y. Evaluation of Antioxidant Capacity and Total Phenolic Content of Different Fractions of Selected Microalgae. Food Chem. 2007, 102, 771–776. [Google Scholar] [CrossRef]
- Hajimahmoodi, M.; Faramarzi, M.A.; Mohammadi, N.; Soltani, N.; Oveisi, M.R.; Nafissi-Varcheh, N. Evaluation of Antioxidant Properties and Total Phenolic Contents of Some Strains of Microalgae. J. Appl. Phycol. 2010, 22, 43–50. [Google Scholar] [CrossRef]
- Patil, L.; Kaliwal, B.B. Microalga Scenedesmus bajacalifornicus BBKLP-07, a New Source of Bioactive Compounds with in Vitro Pharmacological Applications. Bioprocess. Biosyst. Eng. 2019, 42, 979–994. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidantsby means of folin-ciocalteu reagent. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Nakamura, C.V.; Ueda-Nakamura, T.; Bando, E.; Melo, A.F.N.; Cortez, D.A.G.; Dias Filho, B.P. Antibacterial Activity of Ocimum Gratissimum, L. Essential Oil. Memórias Inst. Oswaldo Cruz 1999, 94, 675–678. [Google Scholar] [CrossRef] [PubMed]
- De Jesus Raposo, M.F.; de Morais, A.M.M.B.; de Morais, R.M.S.C. Bioactivity and Applications of Polysaccharides from Marine Microalgae. In Polysaccharides; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2014; pp. 1–38. ISBN 978-3-319-03751-6. [Google Scholar]
- Keidan, M.; Friedlander, M.; (Malis) Arad, S. Effect of Brefeldin A on Cell-Wall Polysaccharide Production in the Red Microalga Porphyridium sp. (Rhodophyta) through Its Effect on the Golgi Apparatus. J. Appl. Phycol. 2009, 21, 707. [Google Scholar] [CrossRef]
- Hayashi, K.; Hayashi, T.; Kojima, I. A Natural Sulfated Polysaccharide, Calcium Spirulan, Isolated from Spirulina Platensis: In Vitro and Ex Vivo Evaluation of Anti-Herpes Simplex Virus and Anti-Human Immunodeficiency Virus Activities. AIDS Res. Hum. Retrovir. 1996, 12, 1463–1471. [Google Scholar] [CrossRef] [PubMed]
- Raposo, M.; De Morais, R.; Bernardo de Morais, A. Bioactivity and Applications of Sulphated Polysaccharides from Marine Microalgae. Mar. Drugs 2013, 11, 233–252. [Google Scholar] [CrossRef]
- Radonić, A.; Thulke, S.; Achenbach, J.; Kurth, A.; Vreemann, A.; König, T.; Walter, C.; Possinger, K.; Nitsche, A. Anionic Polysaccharides from Phototrophic Microorganisms Exhibit Antiviral Activities to Vaccinia Virus. J. Antivir. Antiretrovir. 2011, 2, 51–55. [Google Scholar] [CrossRef]
- Tannin-Spitz, T.; Bergman, M.; van-Moppes, D.; Grossman, S.; Arad, S. (Malis) Antioxidant Activity of the Polysaccharide of the Red Microalga Porphyridium Sp. J. Appl. Phycol. 2005, 17, 215–222. [Google Scholar] [CrossRef]
- Chen, B.; You, W.; Huang, J.; Yu, Y.; Chen, W. Isolation and Antioxidant Property of the Extracellular Polysaccharide from Rhodella reticulata. World. J. Microbiol. Biotechnol. 2010, 26, 833–840. [Google Scholar] [CrossRef]
- Guzmán, S.; Gato, A.; Lamela, M.; Freire-Garabal, M.; Calleja, J.M. Anti-Inflammatory and Immunomodulatory Activities of Polysaccharide from Chlorella stigmatophora and Phaeodactylum tricornutum: Anti-inflammatory and immunomodulatory activities. Phytother. Res. 2003, 17, 665–670. [Google Scholar] [CrossRef]
- Tabarsa, M.; Shin, I.-S.; Lee, J.H.; Surayot, U.; Park, W.; You, S. An Immune-Enhancing Water-Soluble α-Glucan from Chlorella vulgaris and Structural Characteristics. Food Sci. Biotechnol. 2015, 24, 1933–1941. [Google Scholar] [CrossRef]
- Geresh, S.; Mamontov, A.; Weinstein, J. Sulfation of Extracellular Polysaccharides of Red Microalgae: Preparation, Characterization and Properties. J. Biochem. Biophys. Methods 2002, 50, 179–187. [Google Scholar] [CrossRef]
- Gardeva, E.; Toshkova, R.; Minkova, K.; Gigova, L. Cancer Protective Action of Polysaccharide, Derived from Red Microalga Porphyridium cruentum—A Biological Background. Biotechnol. Biotechnol. Equip. 2009, 23, 783–787. [Google Scholar] [CrossRef]
- Gujar, A.; Cui, H.; Ji, C.; Kubar, S.; Li, R. Development, Production and Market Value of Microalgae Products. Appl. Microbiol. Open Access 2019, 5, 162. [Google Scholar]
- Thompson Thomas, E. Lipid. Available online: https://www.britannica.com/science/lipid (accessed on 3 March 2021).
- Yi, Z.; Xu, M.; Di, X.; Brynjolfsson, S.; Fu, W. Exploring Valuable Lipids in Diatoms. Front. Mar. Sci. 2017, 4, 17. [Google Scholar] [CrossRef]
- Mangas-Sánchez, J.; Adlercreutz, P. Highly Efficient Enzymatic Biodiesel Production Promoted by Particle-Induced Emulsification. Biotechnol. Biofuels 2015, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Peltomaa, E.; Johnson, M.; Taipale, S. Marine Cryptophytes Are Great Sources of EPA and DHA. Mar. Drugs 2017, 16, 3. [Google Scholar] [CrossRef]
- Sapieha, P.; Stahl, A.; Chen, J.; Seaward, M.R.; Willett, K.L.; Krah, N.M.; Dennison, R.J.; Connor, K.M.; Aderman, C.M.; Liclican, E.; et al. 5-Lipoxygenase metabolite 4-HDHA is a mediator of the antiangiogenic effect ofω-3 polyunsaturated fatty acids. Sci. Transl. Med. 2011, 3, 69ra12. [Google Scholar] [CrossRef]
- Hallahan, B.; Garland, M.R. Essential Fatty Acids and Mental Health. Br. J. Psychiatry 2005, 186, 275–277. [Google Scholar] [CrossRef]
- Lafourcade, M.; Larrieu, T.; Mato, S.; Duffaud, A.; Sepers, M.; Matias, I.; De Smedt-Peurusse, V.; Labrousse, V.F.; Bretillon, L.; Matute, C.; et al. Nutritional Omega-3 Deficiency Abolishes Endocannabinoid-Mediated Neuronal Functions. Nat. Neurosci. 2011, 14, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Weldon, S.; Mitchell, S.; Kelleher, D.; Gibney, M.J.; Roche, H.M. Conjugated Linoleic Acid and Atherosclerosis: No Effect on Molecular Markers of Cholesterol Homeostasis in THP-1 Macrophages. Atherosclerosis 2004, 174, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Mullen, A.; Loscher, C.E.; Roche, H.M. Anti-Inflammatory Effects of EPA and DHA Are Dependent upon Time and Dose-Response Elements Associated with LPS Stimulation in THP-1-Derived Macrophages. J. Nutr. Biochem. 2010, 21, 444–450. [Google Scholar] [CrossRef]
- Nappo, M.; Berkov, S.; Massucco, C.; Di Maria, V.; Bastida, J.; Codina, C.; Avila, C.; Messina, P.; Zupo, V.; Zupo, S. Apoptotic Activity of the Marine Diatom Cocconeis scutellum and Eicosapentaenoic Acid in BT20 Cells. Pharm. Biol. 2012, 50, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.P.; Lebl, T.; Yan, L.; Smith, V.J. Isolation and Structural Characterisation of Two Antibacterial Free Fatty Acids from the Marine Diatom, Phaeodactylum tricornutum. Appl. Microbiol. Biotechnol. 2008, 81, 755–764. [Google Scholar] [CrossRef]
- Gutiérrez-Pliego, L.E.; Martínez-Carrillo, B.E.; Reséndiz-Albor, A.A.; Arciniega-Martínez, I.M.; Escoto-Herrera, J.A.; Rosales-Gómez, C.A.; Valdés-Ramos, R. Effect of Supplementation with n-3 Fatty Acids Extracted from Microalgae on Inflammation Biomarkers from Two Different Strains of Mice. J. Lipids 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Andrianasolo, E.H.; Haramaty, L.; Vardi, A.; White, E.; Lutz, R.; Falkowski, P. Apoptosis-Inducing Galactolipids from a Cultured Marine Diatom, Phaeodactylum tricornutum. J. Nat. Prod. 2008, 71, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.H.; Gallant, P.; Stefanova, R.; Melanson, R.; O’Leary, S.J.B. Monogalactosyldiacylglycerols, Potent Nitric Oxide Inhibitors from the Marine Microalga Tetraselmis Chui. Nat. Prod. Res. 2013, 27, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.H.; Stefanova, R.; Gallant, P.; McGinn, P.J. Mono-and Digalactosyldiacylglycerols: Potent Nitric Oxide Inhibitors from the Marine Microalga Nannochloropsis granulata. J. Appl. Phycol. 2013, 25, 349–357. [Google Scholar] [CrossRef]
- Hielscher-Michael, S.; Griehl, C.; Buchholz, M.; Demuth, H.-U.; Arnold, N.; Wessjohann, L.A. Natural Products from Microalgae with Potential against Alzheimer’s Disease: Sulfolipids Are Potent Glutaminyl Cyclase Inhibitors. Mar. Drugs 2016, 14, 203. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Cutignano, A.; Pagano, D.; Gallo, C.; Barra, G.; Nuzzo, G.; Sansone, C.; Ianora, A.; Urbanek, K.; Fenoglio, D. A New Marine-Derived Sulfoglycolipid Triggers Dendritic Cell Activation and Immune Adjuvant Response. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Fioretto, L.; Gallo, C.; Ziaco, M.; Nuzzo, G.; D’Ippolito, G.; Borzacchiello, A.; Fabozzi, A.; De Palma, R.; Fontana, A. Preparation, Supramolecular Aggregation and Immunological Activity of the Bona Fide Vaccine Adjuvant Sulfavant, S. Mar. Drugs 2020, 18, 451. [Google Scholar] [CrossRef]
- Morimoto, T.; Nagatsu, A.; Murakami, N.; Sakakibara, J.; Tokuda, H.; Nishino, H.; Iwashima, A. Anti-Tumour-Promoting Glyceroglycolipids from the Green Alga, Chlorella vulgaris. Phytochemistry 1995, 40, 1433–1437. [Google Scholar] [CrossRef]
- Bergé, J.P.; Debiton, E.; Dumay, J.; Durand, P.; Barthomeuf, C. In Vitro Anti-Inflammatory and Anti-Proliferative Activity of Sulfolipids from the Red Alga Porphyridium cruentum. J. Agric. Food Chem. 2002, 50, 6227–6232. [Google Scholar]
- Robertson, R.; Guihéneuf, F.; Bahar, B.; Schmid, M.; Stengel, D.; Fitzgerald, G.; Ross, R.; Stanton, C. The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages. Mar. Drugs 2015, 13, 5402–5424. [Google Scholar] [CrossRef]
- Kagan, M.L.; Levy, A.; Leikin-Frenkel, A. Comparative Study of Tissue Deposition of Omega-3 Fatty Acids from Polar-Lipid Rich Oil of the Microalgae Nannochloropsis oculata with Krill Oil in Rats. Food Funct. 2015, 6, 185–191. [Google Scholar] [CrossRef]
- Sansone, C.; Braca, A.; Ercolesi, E.; Romano, G.; Palumbo, A.; Casotti, R.; Francone, M.; Ianora, A. Diatom-Derived Polyunsaturated Aldehydes Activate Cell Death in Human Cancer Cell Lines but Not Normal Cells. PLoS ONE 2014, 9, e101220. [Google Scholar] [CrossRef] [PubMed]
- Miralto, A.; Barone, G.; Romano, G.; Poulet, S.A.; Ianora, A.; Russo, G.L.; Buttino, I.; Mazzarella, G.; Laabir, M.; Cabrini, M.; et al. The Insidious Effect of Diatoms on Copepod Reproduction. Nature 1999, 402, 173–176. [Google Scholar] [CrossRef]
- Ávila-Román, J.; Talero, E.; De los Reyes, C.; Zubía, E.; Motilva, V.; García-Mauriño, S. Cytotoxic activity of microalgal-derived oxylipins against human cancer cell lines and their impact on ATP levels. Nat. Prod. Commun. 2016, 11, 1871–1875. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, K.; Akihisa, T.; Kanno, H.; Kaminaga, T.; Izumida, M.; Sakoh, T.; Tamura, T.; Takido, M. Inhibitory Effects of Sterols Isolated from Chlorella Vulgaris on 12-O-Tetradecanoylphorbol-13-Acetate-Induced Inflammation and Tumor Promotion in Mouse Skin. Biol. Pharm. Bull. 1996, 19, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Caroprese, M.; Albenzio, M.; Ciliberti, M.G.; Francavilla, M.; Sevi, A. A Mixture of Phytosterols from Dunaliella tertiolecta Affects Proliferation of Peripheral Blood Mononuclear Cells and Cytokine Production in Sheep. Vet. Immunol. Immunopathol. 2012, 150, 27–35. [Google Scholar] [CrossRef]
- Francavilla, M.; Colaianna, M.; Zotti, M.; Morgese, M.G.; Trotta, P.; Tucci, P.; Schiavone, S.; Cuomo, V.; Trabace, L. Extraction, characterization and In Vivo neuromodulatory activity of phytosterols from microalga Dunaliella tertiolecta. Curr. Med. Chem. 2012, 19, 3058–3067. [Google Scholar] [CrossRef]
- Prakash, S.; Sasikala, S.L.; Aldous, V.H.J. Isolation and Identification of MDR–Mycobacterium Tuberculosis and Screening of Partially Characterised Antimycobacterial Compounds from Chosen Marine Micro Algae. Asian Pac. J. Trop. Med. 2010, 3, 655–661. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Li, X.-F.; Kang, K.-H.; Ryu, B.; Kim, S.K. Stigmasterol Isolated from Marine Microalgae Navicula incerta Induces Apoptosis in Human Hepatoma HepG2 Cells. BMB Rep. 2014, 47, 433. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; KANG, K.H.; Kim, Y.S. Pharmaceutical Composition for Preventing or Treating Liver Cancer Comprising Stigmasterol and 5 beta-hydroxysitostanol Isolated from Navicula incerta. U.S. Patent 2014/0057884 A1, 27 February 2014. [Google Scholar]
- Glycolipid. Available online: https://phys.libretexts.org/Courses/University_of_California_Davis/UCD%3A_Biophysics_241_-_Membrane_Biology/01%3A_Lipids/1.04%3A_Glycolipids (accessed on 3 March 2021).
- Harwood, J.L.; Guschina, I.A. The Versatility of Algae and Their Lipid Metabolism. Biochimie 2009, 91, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Hölzl, G.; Dörmann, P. Structure and Function of Glycoglycerolipids in Plants and Bacteria. Prog. Lipid Res. 2007, 46, 225–243. [Google Scholar] [CrossRef] [PubMed]
- Morawski, M.; Schilling, S.; Kreuzberger, M.; Waniek, A.; Jäger, C.; Koch, B.; Cynis, H.; Kehlen, A.; Arendt, T.; Hartlage-Rübsamen, M. Glutaminyl Cyclase in Human Cortex: Correlation with (PGlu)-Amyloid-β Load and Cognitive Decline in Alzheimer’s Disease. J. Alzheimers Dis. 2014, 39, 385–400. [Google Scholar] [CrossRef]
- Fontana, A.; Manzo, E.; Cutignano, A.; de Palma, R. Beta-glycolipids for use as adjuvants. U.S. Patent EP3007725 A1, 20 April 2016. [Google Scholar]
- Clayton, R.B.; Kluger, R.H. Steroid. Available online: https://www.britannica.com/science/steroid (accessed on 3 March 2021).
- Luo, X.; Su, P.; Zhang, W. Advances in Microalgae-Derived Phytosterols for Functional Food and Pharmaceutical Applications. Mar. Drugs 2015, 13, 4231–4254. [Google Scholar] [CrossRef]
- Ryckebosch, E.; Bruneel, C.; Termote-Verhalle, R.; Muylaert, K.; Foubert, I. Influence of Extraction Solvent System on Extractability of Lipid Components from Different Microalgae Species. Algal Res. 2014, 3, 36–43. [Google Scholar] [CrossRef]
- Ahmed, F.; Zhou, W.; Schenk, P.M. Pavlova lutheri Is a High-Level Producer of Phytosterols. Algal Res. 2015, 10, 210–217. [Google Scholar] [CrossRef]
- Fontana, A.; d’Ippolito, G.; Cutignano, A.; Romano, G.; Ianora, A.; Miralto, A.; Cimino, G. Oxylipin Pathways in Marine Diatoms: A Look at the Chemical Aspects. Pure Appl. Chem. 2007, 79, 481–490. [Google Scholar] [CrossRef]
- Lauritano, C.; Romano, G.; Roncalli, V.; Amoresano, A.; Fontanarosa, C.; Bastianini, M.; Braga, F.; Carotenuto, Y.; Ianora, A. New Oxylipins Produced at the End of a Diatom Bloom and Their Effects on Copepod Reproductive Success and Gene Expression Levels. Harmful Algae 2016, 55, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Ianora, A.; Miralto, A. Toxigenic Effects of Diatoms on Grazers, Phytoplankton and Other Microbes: A Review. Ecotoxicology 2010, 19, 493–511. [Google Scholar] [CrossRef] [PubMed]
- Ianora, A.; Miralto, A.; Poulet, S.A.; Carotenuto, Y.; Buttino, I.; Romano, G.; Casotti, R.; Pohnert, G.; Wichard, T.; Colucci-D’Amato, L. Aldehyde Suppression of Copepod Recruitment in Blooms of a Ubiquitous Planktonic Diatom. Nature 2004, 429, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, C.; Borra, M.; Carotenuto, Y.; Biffali, E.; Miralto, A.; Procaccini, G.; Ianora, A. First Molecular Evidence of Diatom Effects in the Copepod Calanus helgolandicus. J. Exp. Mar. Biol. Ecol. 2011, 404, 79–86. [Google Scholar] [CrossRef]
- Lauritano, C.; Borra, M.; Carotenuto, Y.; Biffali, E.; Miralto, A.; Procaccini, G.; Ianora, A. Molecular Evidence of the Toxic Effects of Diatom Diets on Gene Expression Patterns in Copepods. PLoS ONE 2011, 6, e26850. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, C.; Carotenuto, Y.; Miralto, A.; Procaccini, G.; Ianora, A. Copepod Population-Specific Response to a Toxic Diatom Diet. PLoS ONE 2012, 7, e47262. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, C.; Carotenuto, Y.; Vitiello, V.; Buttino, I.; Romano, G.; Hwang, J.-S.; Ianora, A. Effects of the Oxylipin-Producing Diatom Skeletonema marinoi on Gene Expression Levels of the Calanoid Copepod Calanus sinicus. Mar. Genom. 2015, 24, 89–94. [Google Scholar] [CrossRef]
- Carotenuto, Y.; Dattolo, E.; Lauritano, C.; Pisano, F.; Sanges, R.; Miralto, A.; Procaccini, G.; Ianora, A. Insights into the Transcriptome of the Marine Copepod Calanus helgolandicus Feeding on the Oxylipin-Producing Diatom Skeletonema marinoi. Harmful Algae 2014, 31, 153–162. [Google Scholar] [CrossRef]
- Asai, S.; Sanges, R.; Lauritano, C.; Lindeque, P.K.; Esposito, F.; Ianora, A.; Carotenuto, Y. De Novo Transcriptome Assembly and Gene Expression Profiling of the Copepod Calanus helgolandicus Feeding on the PUA-Producing Diatom Skeletonema marinoi. Mar. Drugs 2020, 18, 392. [Google Scholar] [CrossRef] [PubMed]
- Romano, G.; Miralto, A.; Ianora, A. Teratogenic Effects of Diatom Metabolites on Sea Urchin Paracentrotus lividus Embryos. Mar. Drugs 2010, 8, 950–967. [Google Scholar] [CrossRef] [PubMed]
- Sartori, D.; Gaion, A. Toxicity of Polyunsaturated Aldehydes of Diatoms to Indo-Pacific Bioindicator Organism Echinometra mathaei. Drug Chem. Toxicol. 2016, 39, 124–128. [Google Scholar] [PubMed]
- Varrella, S.; Romano, G.; Ruocco, N.; Ianora, A.; Bentley, M.G.; Costantini, M. First Morphological and Molecular Evidence of the Negative Impact of Diatom-Derived Hydroxyacids on the Sea Urchin Paracentrotus lividus. Toxicol. Sci. 2016, 151, 419–433. [Google Scholar] [CrossRef] [PubMed]
- Ruocco, N.; Costantini, S.; Zupo, V.; Lauritano, C.; Caramiello, D.; Ianora, A.; Budillon, A.; Romano, G.; Nuzzo, G.; D’Ippolito, G. Toxigenic Effects of Two Benthic Diatoms upon Grazing Activity of the Sea Urchin: Morphological, Metabolomic and de Novo Transcriptomic Analysis. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Ruocco, N.; Annunziata, C.; Ianora, A.; Libralato, G.; Manfra, L.; Costantini, S.; Costantini, M. Toxicity of Diatom-Derived Polyunsaturated Aldehyde Mixtures on Sea Urchin Paracentrotus lividus Development. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Esposito, R.; Ruocco, N.; Albarano, L.; Ianora, A.; Manfra, L.; Libralato, G.; Costantini, M. Combined Effects of Diatom-Derived Oxylipins on the Sea Urchin Paracentrotus lividus. Int. J. Mol. Sci. 2020, 21, 719. [Google Scholar] [CrossRef]
- Ruocco, N.; Albarano, L.; Esposito, R.; Zupo, V.; Costantini, M.; Ianora, A. Multiple Roles of Diatom-Derived Oxylipins within Marine Environments and Their Potential Biotechnological Applications. Mar. Drugs 2020, 18, 342. [Google Scholar] [CrossRef] [PubMed]
- Ávila-Román, J.; Talero, E.; Alcaide, A.; de los Reyes, C.; Zubía, E.; García-Mauriño, S.; Motilva, V. Preventive Effect of the Microalga Chlamydomonas debaryana on the Acute Phase of Experimental Colitis in Rats. Br. J. Nutr. 2014, 112, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Bai, L.; Zhu, L.; Yang, L.; Zhang, X. Marine Algae-Derived Bioactive Peptides for Human Nutrition and Health. J. Agric. Food Chem. 2014, 62, 9211–9222. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed Marine Natural Products in the Pharmaceutical and Cosmeceutical Industries: Tips for Success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- Sheih, I.-C.; Wu, T.-K.; Fang, T.J. Antioxidant Properties of a New Antioxidative Peptide from Algae Protein Waste Hydrolysate in Different Oxidation Systems. Bioresour. Technol. 2009, 100, 3419–3425. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.-C.; Kim, D.; Jeon, Y.-J. Protective Effect of a Novel Antioxidative Peptide Purified from a Marine Chlorella ellipsoidea Protein against Free Radical-Induced Oxidative Stress. Food Chem. Toxicol. 2012, 50, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.-H.; Qian, Z.-J.; Ryu, B.; Karadeniz, F.; Kim, D.; Kim, S.-K. Antioxidant Peptides from Protein Hydrolysate of Microalgae Navicula Incerta and Their Protective Effects in HepG2/CYP2E1 Cells Induced by Ethanol. Phytother. Res. 2012, 26, 1555–1563. [Google Scholar] [CrossRef]
- Sheih, I.-C.; Fang, T.J.; Wu, T.-K.; Lin, P.-H. Anticancer and Antioxidant Activities of the Peptide Fraction from Algae Protein Waste. J. Agric. Food Chem. 2010, 58, 1202–1207. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, X. Separation, Antitumor Activities, and Encapsulation of Polypeptide from Chlorella pyrenoidosa. Biotechnol. Prog. 2013, 29, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Samarakoon, K.W.; Kwon, O.-N.; Ko, J.-Y.; Lee, J.-H.; Kang, M.-C.; Kim, D.; Lee, J.B.; Lee, J.-S.; Jeon, Y.-J. Purification and Identification of Novel Angiotensin-I Converting Enzyme (ACE) Inhibitory Peptides from Cultured Marine Microalgae (Nannochloropsis oculata) Protein Hydrolysate. J. Appl. Phycol. 2013, 25, 1595–1606. [Google Scholar] [CrossRef]
- Chen, C.-L.; Liou, S.-F.; Chen, S.-J.; Shih, M.-F. Protective Effects of Chlorella-Derived Peptide on UVB-Induced Production of MMP-1 and Degradation of Procollagen Genes in Human Skin Fibroblasts. Regul. Toxicol. Pharmacol. 2011, 60, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.H.T.; Qian, Z.-J.; Nguyen, V.-T.; Choi, I.-W.; Heo, S.-J.; Oh, C.H.; Kang, D.-H.; Kim, G.H.; Jung, W.-K. Tetrameric Peptide Purified from Hydrolysates of Biodiesel Byproducts of Nannochloropsis oculata Induces Osteoblastic Differentiation through MAPK and Smad Pathway on MG-63 and D1 Cells. Process. Biochem. 2013, 48, 1387–1394. [Google Scholar] [CrossRef]
- Shih, M.F.; Chen, L.C.; Cherng, J.Y. Chlorella 11-Peptide Inhibits the Production of Macrophage-Induced Adhesion Molecules and Reduces Endothelin-1 Expression and Endothelial Permeability. Mar. Drugs 2013, 11, 3861–3874. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Yamada, A.; Noda, K.; Hasegawa, T.; Okuda, M.; Shoyama, Y.; Nomoto, K. A Novel Glycoprotein Obtained from Chlorella vulgaris Strain CK22 Shows Antimetastatic Immunopotentiation. Cancer Immunol. Immunother. 1998, 45, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.; Ohno, N.; Tanaka, K.; Kamiya, N.; Okuda, M.; Yadomae, T.; Nomoto, K. A Water-Soluble Antitumor Glycoprotein from Chlorella vulgaris. Planta Med. 2002, 4, 423–426. [Google Scholar]
- Hasegawa, T.; Matsuguchi, T.; Noda, K.; Tanaka, K.; Kumamoto, S.; Shoyama, Y.; Yoshikai, Y. Toll-like Receptor 2 Is at Least Partly Involved in the Antitumor Activity of Glycoprotein from Chlorella vulgaris. Int. Immunopharmacol. 2002, 2, 579–589. [Google Scholar] [CrossRef]
- Bellou, S.; Baeshen, M.N.; Elazzazy, A.M.; Aggeli, D.; Sayegh, F.; Aggelis, G. Microalgal Lipids Biochemistry and Biotechnological Perspectives. Biotechnol. Adv. 2014, 32, 1476–1493. [Google Scholar] [CrossRef] [PubMed]
- Tevatia, R.; Allen, J.; Rudrappa, D.; White, D.; Clemente, T.E.; Cerutti, H.; Demirel, Y.; Blum, P. The Taurine Biosynthetic Pathway of Microalgae. Algal Res. 2015, 9, 21–26. [Google Scholar] [CrossRef]
- Clifford, E.L.; Varela, M.M.; De Corte, D.; Bode, A.; Ortiz, V.; Herndl, G.J.; Sintes, E. Taurine Is a Major Carbon and Energy Source for Marine Prokaryotes in the North Atlantic Ocean off the Iberian Peninsula. Microb. Ecol. 2019, 78, 299–312. [Google Scholar] [CrossRef]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino Acids, Fatty Acids, and Dietary Fibre in Edible Seaweed Products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Houston, M. Nutraceuticals, Vitamins, Antioxidants, and Minerals in the Prevention and Treatment of Hypertension. Prog. Cardiovasc. Dis. 2005, 47, 396–449. [Google Scholar] [CrossRef]
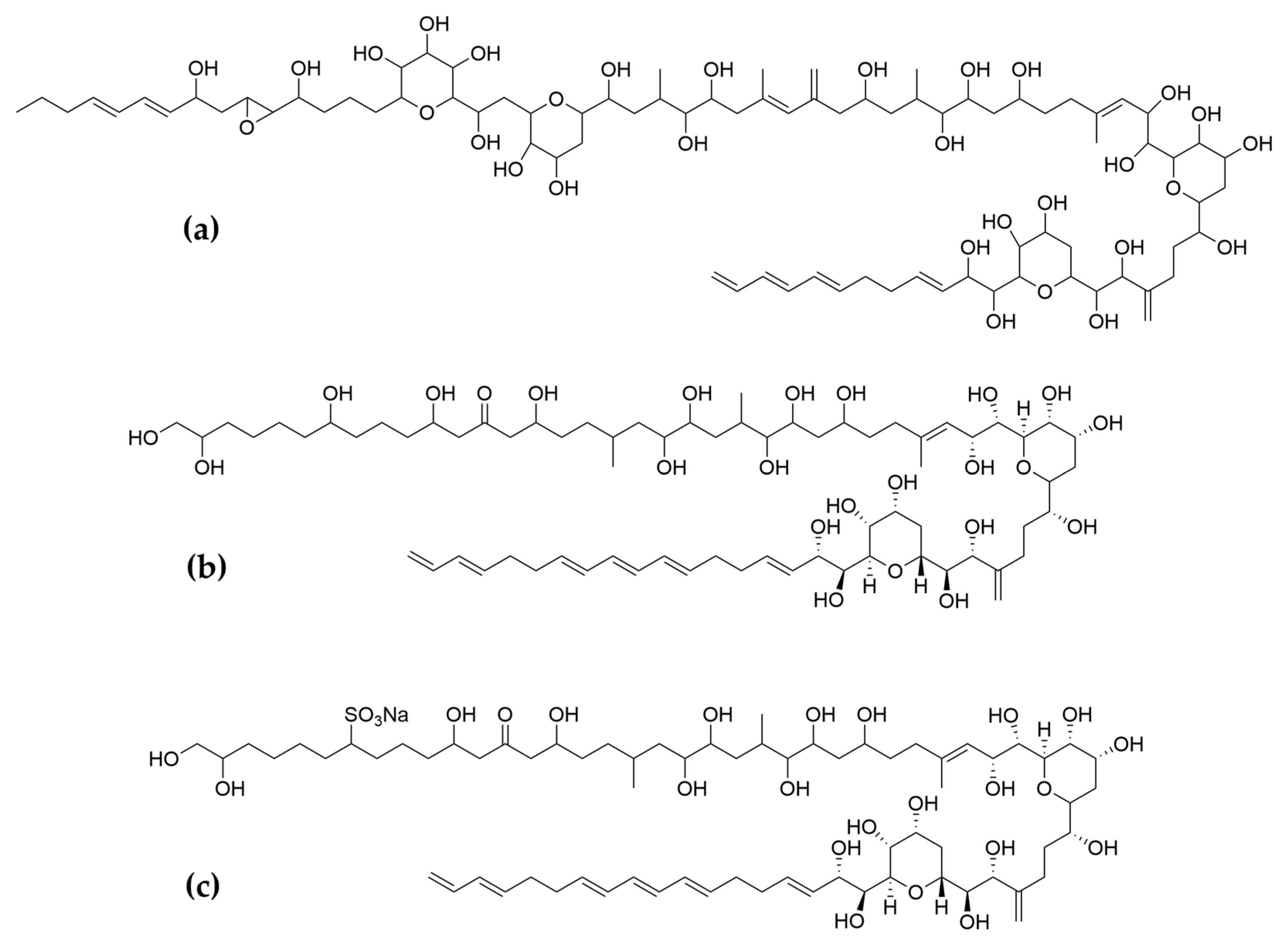
- Kobayashi, J.; Kubota, T. Bioactive Macrolides and Polyketides from Marine Dinoflagellates of the Genus Amphidinium. J. Nat. Prod. 2007, 70, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.-H.; Lee, H.-G.; Jo, J.; Kim, H.M.; Kim, S.-M.; Park, J.Y.; Jeon, C.B.; Kang, H.-S.; Park, M.G.; Park, C.; et al. The Exceptionally Large Genome of the Harmful Red Tide Dinoflagellate Cochlodinium polykrikoides Margalef (Dinophyceae): Determination by Flow Cytometry. Algae 2016, 31, 373–378. [Google Scholar] [CrossRef]
- Rein, K.S.; Gibbs, P.D.L.; Palacios, A.; Abiy, L.; Dickey, R.; Snyder, R.V.; Lopez, J.V. Polyketide Synthase Genes from Marine Dinoflagellates. Mar. Biotechnol. 2003, 5, 1–12. [Google Scholar] [CrossRef]
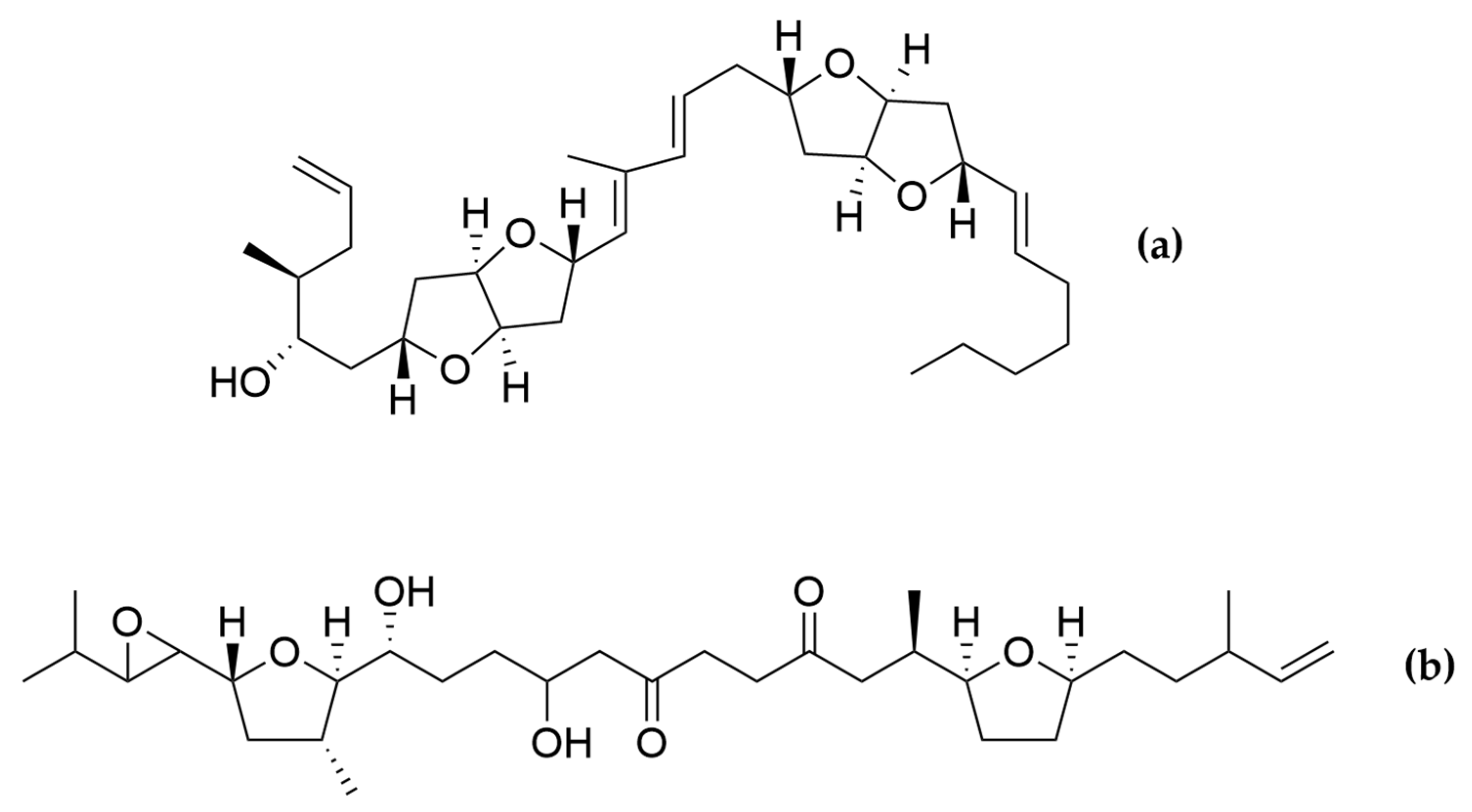
- Satake, M.; Murata, M.; Yasumoto, T.; Fujita, T.; Naoki, H. Amphidinol, a Polyhydroxy-Polyene Antifungal Agent with an Unprecedented Structure, from a Marine Dinoflagellate, Amphidinium klebsii. J. Am. Chem. Soc. 1991, 113, 9859–9861. [Google Scholar] [CrossRef]
- Morsy, N.; Konoki, K.; Houdai, T.; Matsumori, N.; Oishi, T.; Murata, M.; Aimoto, S. Roles of Integral Protein in Membrane Permeabilization by Amphidinols. Biochim. Biophys. Acta BBA-Biomembr. 2008, 1778, 1453–1459. [Google Scholar] [CrossRef] [PubMed]
- Echigoya, R.; Rhodes, L.; Oshima, Y.; Satake, M. The Structures of Five New Antifungal and Hemolytic Amphidinol Analogs from Amphidinium carterae Collected in New Zealand. Harmful Algae 2005, 4, 383–389. [Google Scholar] [CrossRef]
- Nuzzo, G.; Cutignano, A.; Sardo, A.; Fontana, A. Antifungal Amphidinol 18 and Its 7-Sulfate Derivative from the Marine Dinoflagellate Amphidinium carterae. J. Nat. Prod. 2014, 77, 1524–1527. [Google Scholar] [CrossRef]
- Satake, M.; Cornelio, K.; Hanashima, S.; Malabed, R.; Murata, M.; Matsumori, N.; Zhang, H.; Hayashi, F.; Mori, S.; Kim, J.S.; et al. Structures of the Largest Amphidinol Homologues from the Dinoflagellate Amphidinium carterae and Structure–Activity Relationships. J. Nat. Prod. 2017, 80, 2883–2888. [Google Scholar] [CrossRef] [PubMed]
- Martínez, K.A.; Lauritano, C.; Druka, D.; Romano, G.; Grohmann, T.; Jaspars, M.; Martín, J.; Díaz, C.; Cautain, B.; de la Cruz, M.; et al. Amphidinol 22, a New Cytotoxic and Antifungal Amphidinol from the Dinoflagellate Amphidinium carterae. Mar. Drugs 2019, 17, 385. [Google Scholar] [CrossRef] [PubMed]
- Paul, G.K.; Matsumori, N.; Konoki, K. Chemical Structures of Amphidinols 5 and 6 Isolated from Marine Dinof1agellate Amphidinium klebsii and Their Cholesterol-Dependent Membrane Disruption. J. Mar. Biotechnol. 1997, 5, 124–128. [Google Scholar]
- Espiritu, R.A.; Tan, M.C.S.; Oyong, G.G. Evaluation of the Anti-Cancer Potential of Amphidinol 2, a Polyketide Metabolite from the Marine Dinoflagellate. Jordan J. Biol. Sci. 2017, 10, 6. [Google Scholar]
- Houdai, T.; Matsuoka, S.; Matsumori, N.; Murata, M. Membrane-Permeabilizing Activities of Amphidinol 3, Polyene-Polyhydroxy Antifungal from a Marine Dinoflagellate. Biochim. Biophys. Acta BBA-Biomembr. 2004, 1667, 91–100. [Google Scholar] [CrossRef]
- Morsy, N.; Matsuoka, S.; Houdai, T.; Matsumori, N.; Adachi, S.; Murata, M.; Iwashita, T.; Fujita, T. Isolation and Structure Elucidation of a New Amphidinol with a Truncated Polyhydroxyl Chain from Amphidinium klebsii. Tetrahedron 2005, 61, 8606–8610. [Google Scholar] [CrossRef]
- Meng, Y.; Van Wagoner, R.M.; Misner, I.; Tomas, C.; Wright, J.L.C. Structure and Biosynthesis of Amphidinol 17, a Hemolytic Compound from Amphidinium carterae. J. Nat. Prod. 2010, 73, 409–415. [Google Scholar] [CrossRef]
- Cutignano, A.; Nuzzo, G.; Sardo, A.; Fontana, A. The Missing Piece in Biosynthesis of Amphidinols: First Evidence of Glycolate as a Starter Unit in New Polyketides from Amphidinium carterae. Mar. Drugs 2017, 15, 157. [Google Scholar] [CrossRef]
- Washida, K.; Koyama, T.; Yamada, K.; Kita, M.; Uemura, D. Karatungiols A and B, Two Novel Antimicrobial Polyol Compounds, from the Symbiotic Marine Dinoflagellate Amphidinium Sp. Tetrahedron Lett. 2006, 47, 2521–2525. [Google Scholar] [CrossRef]
- Kobayashi, J.; Tsuda, M. Amphidinolides, Bioactive Macrolides from Symbiotic Marine Dinoflagellates. ChemInform 2004, 35. [Google Scholar] [CrossRef]
- Kumagai, K. Amphirionin-2, a Novel Linear Polyketide with Potent Cytotoxic Activity from a Marine Dinoflagellate Amphidinium Species. Bioorg. Med. Chem. Lett. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Satake, M.; Yasumoto, T. Antimicrobial Activities of Polyether Compounds of Dinoflagellate Origins. J. Appl. Phycol. 1990, 2, 305–308. [Google Scholar] [CrossRef]
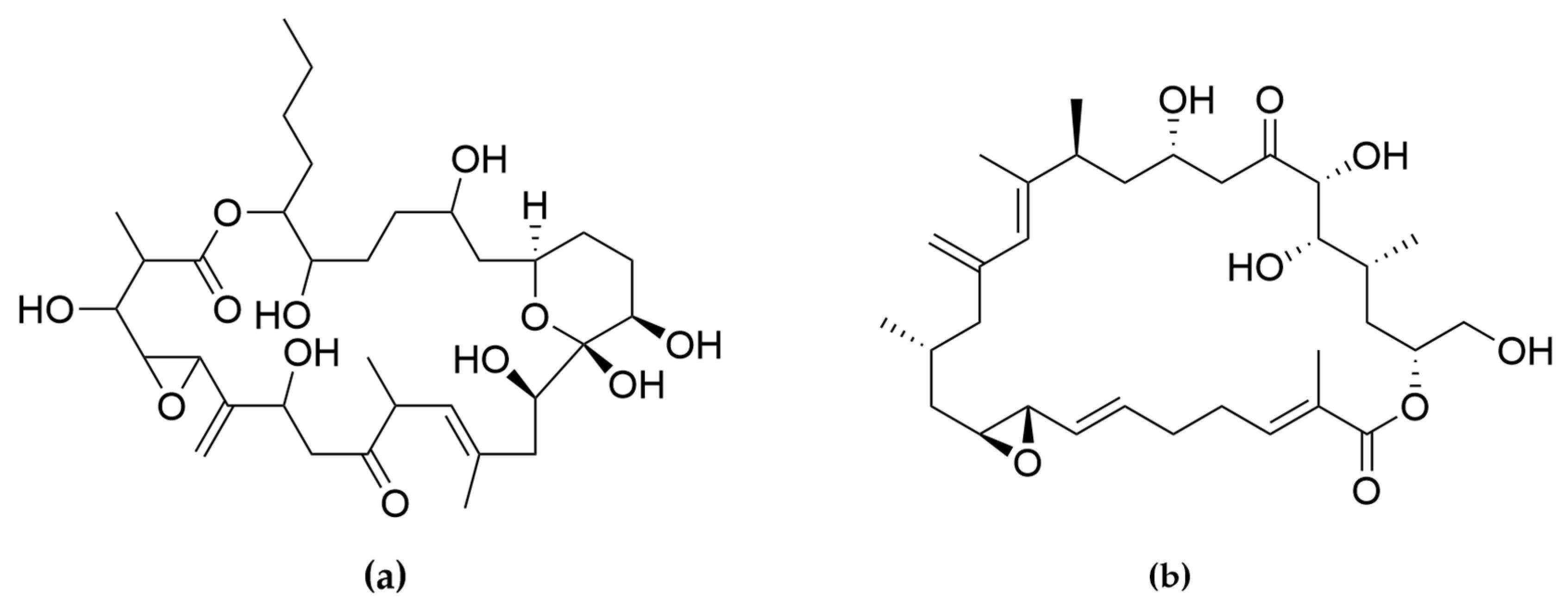
- Murakami, M.; Makabe, K.; Yamaguchi, K.; Konosu, S.; Wälchli, M.R. Goniodomin a, a Novel Polyether Macrolide from the Dinoflagellate Goniodoma pseudogoniaulax. Tetrahedron Lett. 1988, 29, 1149–1152. [Google Scholar] [CrossRef]
- Espiña, B.; Cagide, E.; Louzao, M.C.; Vilariño, N.; Vieytes, M.R.; Takeda, Y.; Sasaki, M.; Botana, L.M. Cytotoxicity of Goniodomin A and B in Non Contractile Cells. Toxicol. Lett. 2016, 250–251, 10–20. [Google Scholar] [CrossRef]
- Akakabe, M.; Kumagai, K.; Tsuda, M.; Konishi, Y.; Tominaga, A.; Tsuda, M.; Fukushi, E.; Kawabata, J. Amphirionin-5, a Novel Linear Polyketide from a Cultured Marine Dinoflagellate Amphidinium Species with a Potent Cell Proliferation-Promoting Activity. Tetrahedron Lett. 2014, 55, 3491–3494. [Google Scholar] [CrossRef]
- Kobayashi, J.; Ishibashi, M.; Nakamura, H.; Ohizumi, Y.; Yamasu, T.; Sasaki, T.; Hirata, Y. Amphidinolide-A, a Novel Antineoplastic Macrolide from the Marine Dinoflagellate Amphidinium Sp. Tetrahedron Lett. 1986, 27, 5755–5758. [Google Scholar] [CrossRef]
- Usui, T.; Kazami, S.; Dohmae, N.; Mashimo, Y.; Kondo, H.; Tsuda, M.; Terasaki, A.G.; Ohashi, K.; Kobayashi, J.; Osada, H. Amphidinolide H, a Potent Cytotoxic Macrolide, Covalently Binds on Actin Subdomain 4 and Stabilizes Actin Filament. Chem. Biol. 2004, 11, 1269–1277. [Google Scholar] [CrossRef]
- Caruana, A.M.N.; Amzil, Z. Microalgae and Toxins. In Microalgae in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2018; pp. 263–305. ISBN 978-0-12-811405-6. [Google Scholar]
- Kudela, R.M.; Bickel, A.; Carter, M.L.; Howard, M.D.A.; Rosenfeld, L. The Monitoring of Harmful Algal Blooms through Ocean Observing. In Coastal Ocean Observing Systems; Elsevier: Amsterdam, The Netherlands, 2015; pp. 58–75. ISBN 978-0-12-802022-7. [Google Scholar]
- Kellmann, R.; Stüken, A.; Orr, R.J.S.; Svendsen, H.M.; Jakobsen, K.S. Biosynthesis and Molecular Genetics of Polyketides in Marine Dinoflagellates. Mar. Drugs 2010, 8, 1011–1048. [Google Scholar] [CrossRef]
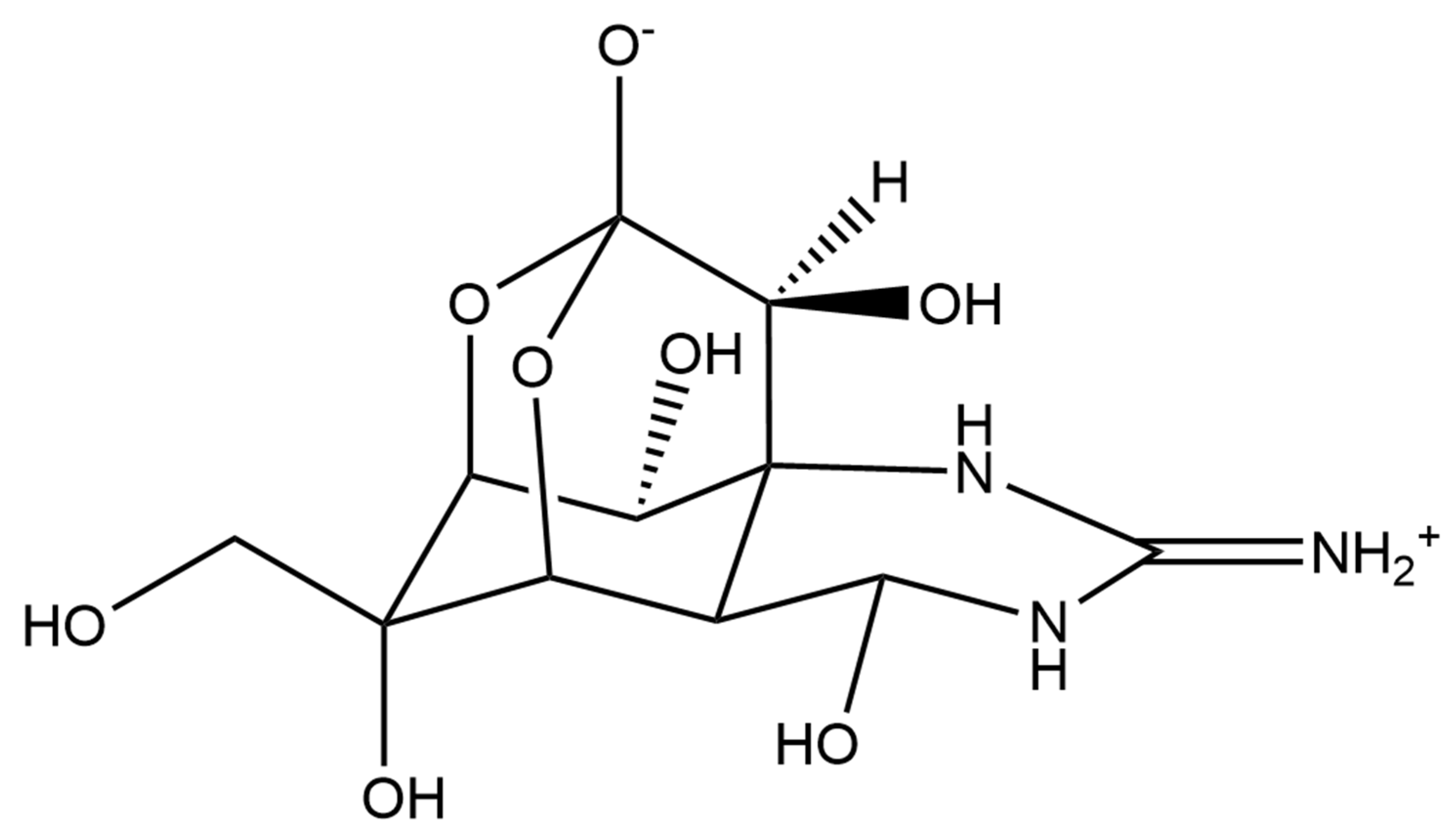
- Kohane, D.S.; Lu, N.T.; Gökgöl-Kline, A.C.; Shubina, M.; Kuang, Y.; Hall, S.; Strichartz, G.R.; Berde, C.B. The Local Anesthetic Properties and Toxicity of Saxitonin Homologues for Rat Sciatic Nerve Block In Vivo. Reg. Anesth. Pain Med. 2000, 25, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Lattes, K.; Venegas, P.; Lagos, N.; Lagos, M.; Pedraza, L.; Rodriguez-Navarro, A.J.; García, C. Local Infiltration of Gonyautoxin Is Safe and Effective in Treatment of Chronic Tension-Type Headache. Neurol. Res. 2009, 31, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Nieto, F.R.; Cobos, E.J.; Tejada, M.Á.; Sánchez-Fernández, C.; González-Cano, R.; Cendán, C.M. Tetrodotoxin (TTX) as a Therapeutic Agent for Pain. Mar. Drugs 2012, 10, 281–305. [Google Scholar] [CrossRef] [PubMed]
- Hagen, N.A.; Cantin, L.; Constant, J.; Haller, T.; Blaise, G.; Ong-Lam, M.; du Souich, P.; Korz, W.; Lapointe, B. Tetrodotoxin for Moderate to Severe Cancer-Related Pain: A Multicentre, Randomized, Double-Blind, Placebo-Controlled, Parallel-Design Trial. Pain Res. Manag. 2017, 2017, 1–7. [Google Scholar] [CrossRef]
- Alfonso, A.; Vieytes, M.; Botana, L. Yessotoxin, a Promising Therapeutic Tool. Mar. Drugs 2016, 14, 30. [Google Scholar] [CrossRef] [PubMed]
- Korsnes, M.S.; Kolstad, H.; Kleiveland, C.R.; Korsnes, R.; Ørmen, E. Autophagic Activity in BC3H1 Cells Exposed to Yessotoxin. Toxicol. In Vitro 2016, 32, 166–180. [Google Scholar] [CrossRef]
- Tobío, A.; Alfonso, A.; Madera-Salcedo, I.; Botana, L.M.; Blank, U. Yessotoxin, a Marine Toxin, Exhibits Anti-Allergic and Anti-Tumoural Activities Inhibiting Melanoma Tumour Growth in a Preclinical Model. PLoS ONE 2016, 11, e0167572. [Google Scholar] [CrossRef] [PubMed]
- ES2363975 Uso de La Yesotoxina. Analogos y Derivados Para El Tratamiento y/o La Prevencion de Enfermedades Neurodegeneratives Relacionadas Con Tau y Beta-Amiloide. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=ES32281349&docAn=201030162 (accessed on 4 March 2021).
- Jung, J.H.; Sim, C.J.; Lee, C.-O. Cytotoxic Compounds from a Two-Sponge Association. J. Nat. Prod. 1995, 58, 1722–1726. [Google Scholar] [CrossRef] [PubMed]
- Boente-Juncal, A.; Álvarez, M.; Antelo, Á.; Rodríguez, I.; Calabro, K.; Vale, C.; Thomas, O.; Botana, L. Structure Elucidation and Biological Evaluation of Maitotoxin-3, a Homologue of Gambierone, from Gambierdiscus belizeanus. Toxins 2019, 11, 79. [Google Scholar] [CrossRef]
- Zhao, Y.; Huang, J.; Yuan, X.; Peng, B.; Liu, W.; Han, S.; He, X. Toxins Targeting the KV1.3 Channel: Potential Immunomodulators for Autoimmune Diseases. Toxins 2015, 7, 1749–1764. [Google Scholar] [CrossRef]
- Murrell, R.N.; Gibson, J.E. Brevetoxins 2, 3, 6, and 9 Show Variability in Potency and Cause Significant Induction of DNA Damage and Apoptosis in Jurkat E6-1 Cells. Arch. Toxicol. 2009, 83, 1009–1019. [Google Scholar] [CrossRef]
- Keeler, D.; Grandal, M.; McCall, J. Brevenal, a Marine Natural Product, Is Anti-Inflammatory and an Immunomodulator of Macrophage and Lung Epithelial Cells. Mar. Drugs 2019, 17, 184. [Google Scholar] [CrossRef]
- Alonso, E.; Vale, C.; Vieytes, M.R.; Laferla, F.M.; Giménez-Llort, L.; Botana, L.M. The Cholinergic Antagonist Gymnodimine Improves Aβ and Tau Neuropathology in an in Vitro Model of Alzheimer Disease. Cell. Physiol. Biochem. 2011, 27, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.F. Designing Drugs to Avoid Toxicity. In Progress in Medicinal Chemistry; Elsevier: Amsterdam, The Netherlands, 2011; Volume 50, pp. 1–47. ISBN 978-0-12-381290-2. [Google Scholar]
- Kodama, M.; Sato, S.; Sakamoto, S.; Ogata, T. Occurrence of Tetrodotoxin in Alexandrium tamarense, a Causative Dinoflagellate of Paralytic Shellfish Poisonin. Toxicon 1996, 34, 1101–1105. [Google Scholar] [CrossRef]
- Geffeney, S.; Ruben, C. The Structural Basis and Functional Consequences of Interactions Between Tetrodotoxin and Voltage-Gated Sodium Channels. Mar. Drugs 2006, 4, 143–156. [Google Scholar] [CrossRef]
- Miles, C.O.; Samdal, I.A.; Aasen, J.A.G.; Jensen, D.J.; Quilliam, M.A.; Petersen, D.; Briggs, L.M.; Wilkins, A.L.; Rise, F.; Cooney, J.M.; et al. Evidence for Numerous Analogs of Yessotoxin in Protoceratium reticulatum. Harmful Algae 2005, 4, 1075–1091. [Google Scholar] [CrossRef]
- Seki, T.; Satake, M.; Mackenzie, L.; Kaspar, H.F.; Yasumoto, T. Gymnodimine, a New Marine Toxin of Unprecedented Structure Isolated from New Zealand Oysters and the Dinoflagellate, Gymnodinium sp. Tetrahedron Lett. 1995, 36, 7093–7096. [Google Scholar] [CrossRef]
- Baden, D.; Adams, D. Brevetoxins: Chemistry, Mechanism of Action, and Methods of Detection. In Seafood and Freshwater Toxins; Botana, L., Ed.; CRC Press: Boca Raton, FL, USA, 2000; Volume 103, ISBN 978-0-8247-8956-5. [Google Scholar]
- Naar, J.P.; Flewelling, L.J.; Lenzi, A.; Abbott, J.P.; Granholm, A.; Jacocks, H.M.; Gannon, D.; Henry, M.; Pierce, R.; Baden, D.G.; et al. Brevetoxins, like Ciguatoxins, Are Potent Ichthyotoxic Neurotoxins That Accumulate in Fish. Toxicon 2007, 50, 707–723. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Prego-Faraldo, M.; Pásaro, E.; Méndez, J.; Laffon, B. Okadaic Acid: More than a Diarrheic Toxin. Mar. Drugs 2013, 11, 4328–4349. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Nelson, D.R.; Yi, Z.; Xu, M.; Khraiwesh, B.; Jijakli, K.; Chaiboonchoe, A.; Alzahmi, A.; Al-Khairy, D.; Brynjolfsson, S.; et al. Bioactive Compounds from Microalgae: Current Development and Prospects. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; Volume 54, pp. 199–225. ISBN 978-0-444-63929-5. [Google Scholar]
- Rosenberg, J.N.; Oyler, G.A.; Wilkinson, L.; Betenbaugh, M.J. A Green Light for Engineered Algae: Redirecting Metabolism to Fuel a Biotechnology Revolution. Curr. Opin. Biotechnol. 2008, 19, 430–436. [Google Scholar] [CrossRef]
- León-Bañares, R.; González-Ballester, D.; Galván, A.; Fernández, E. Transgenic Microalgae as Green Cell-Factories. Trends Biotechnol. 2004, 22, 45–52. [Google Scholar] [CrossRef]
- Rasala, B.A.; Muto, M.; Lee, P.A.; Jager, M.; Cardoso, R.M.F.; Behnke, C.A.; Kirk, P.; Hokanson, C.A.; Crea, R.; Mendez, M.; et al. Production of Therapeutic Proteins in Algae, Analysis of Expression of Seven Human Proteins in the Chloroplast of Chlamydomonas reinhardtii. Plant Biotechnol. J. 2010, 8, 719–733. [Google Scholar] [CrossRef]
- Ng, I.-S.; Tan, S.-I.; Kao, P.-H.; Chang, Y.-K.; Chang, J.-S. Recent Developments on Genetic Engineering of Microalgae for Biofuels and Bio-Based Chemicals. Biotechnol. J. 2017, 12, 1600644. [Google Scholar] [CrossRef]
- Santhosh, S.; Dhandapani, R.; Hemalatha, N. Bioactive Compounds from Microalgae and Its Different Applications-A Review. Adv. Appl. Sci. Res. 2016, 7, 153–158. [Google Scholar]
- Orejuela-Escobar, L.; Gualle, A.; Ochoa-Herrera, V.; Philippidis, G.P. Prospects of Microalgae for Biomaterial Production and Environmental Applications at Biorefineries. Sustainability 2021, 13, 3063. [Google Scholar] [CrossRef]
- Vieira, M.V.; Pastrana, L.M.; Fuciños, P. Microalgae Encapsulation Systems for Food, Pharmaceutical and Cosmetics Applications. Mar. Drugs 2020, 18, 644. [Google Scholar] [CrossRef] [PubMed]
- AstaPure® EyeQ: Natural Astaxanthin for Eyes and Brain-Algatech. Available online: https://www.algatech.com/astapure-eyeq-natural-astaxanthin-for-eyes-and-brain-download-the-ebook/ (accessed on 3 March 2021).
- Betatene 7.5% N: Natural Mixed Carotenoids. BASF Personal Care and Nutrition. Available online: https://www.google.com/search?client=firefox-b-e&q=Betatene+7.5%25+N%3A+Natural+Mixed+Carotenoids.+BASF+Personal+Care+and+Nutrition (accessed on 3 March 2021).
- Fu, W.; Chaiboonchoe, A.; Khraiwesh, B.; Nelson, D.R.; Al-Khairy, D.; Mystikou, A.; Alzahmi, A.; Salehi-Ashtiani, K. Algal Cell Factories: Approaches, Applications, and Potentials. Mar. Drugs 2016, 14, 225. [Google Scholar] [CrossRef] [PubMed]
- Naumann, T.; Çebi, Z.; Podola, B.; Melkonian, M. Growing Microalgae as Aquaculture Feeds on Twin-Layers: A Novel Solid-State Photobioreactor. J. Appl. Phycol. 2012, 25, 1413–1420. [Google Scholar] [CrossRef]
- Pilot Scale of Microalgal Production Using Photobioreactor|IntechOpen. Available online: https://www.intechopen.com/books/photosynthesis-from-its-evolution-to-future-improvements-in-photosynthetic-efficiency-using-nanomaterials/pilot-scale-of-microalgal-production-using-photobioreactor (accessed on 25 February 2021).

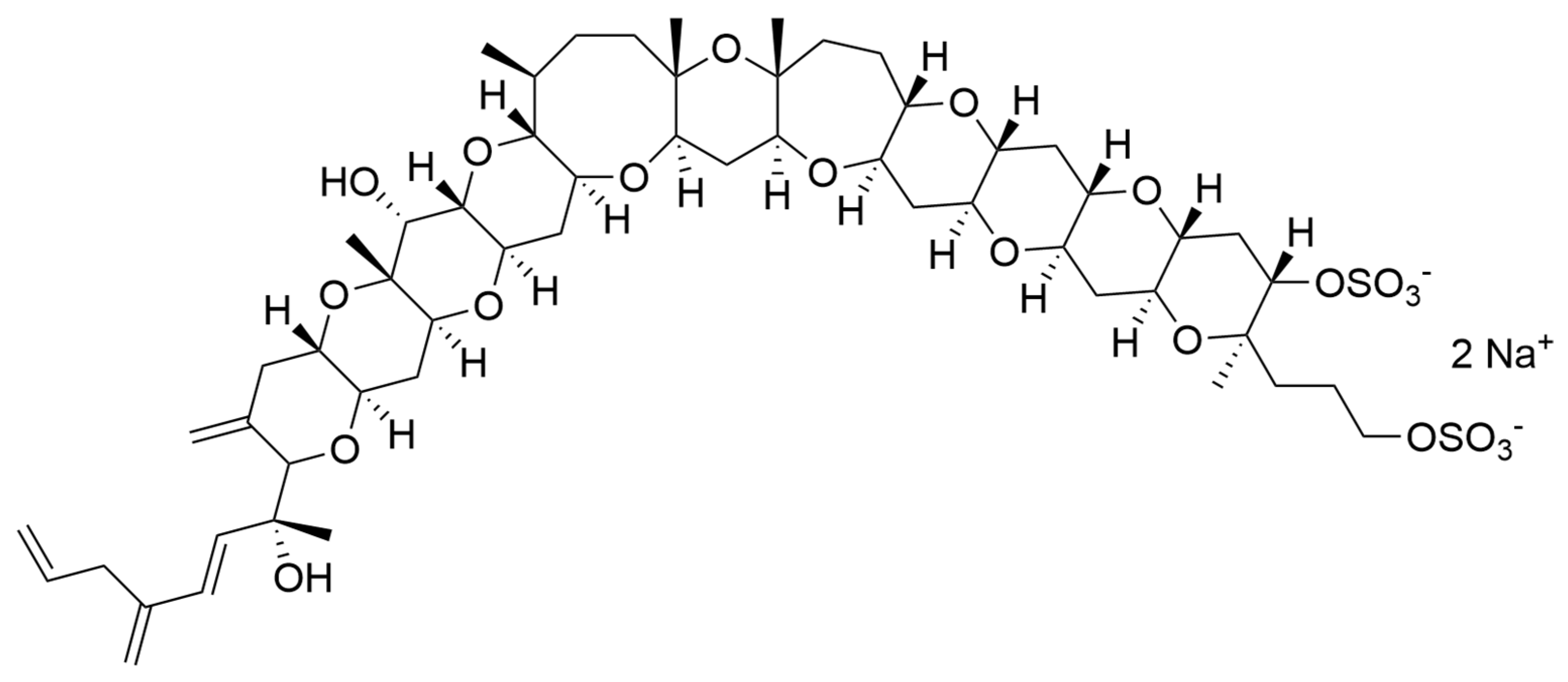
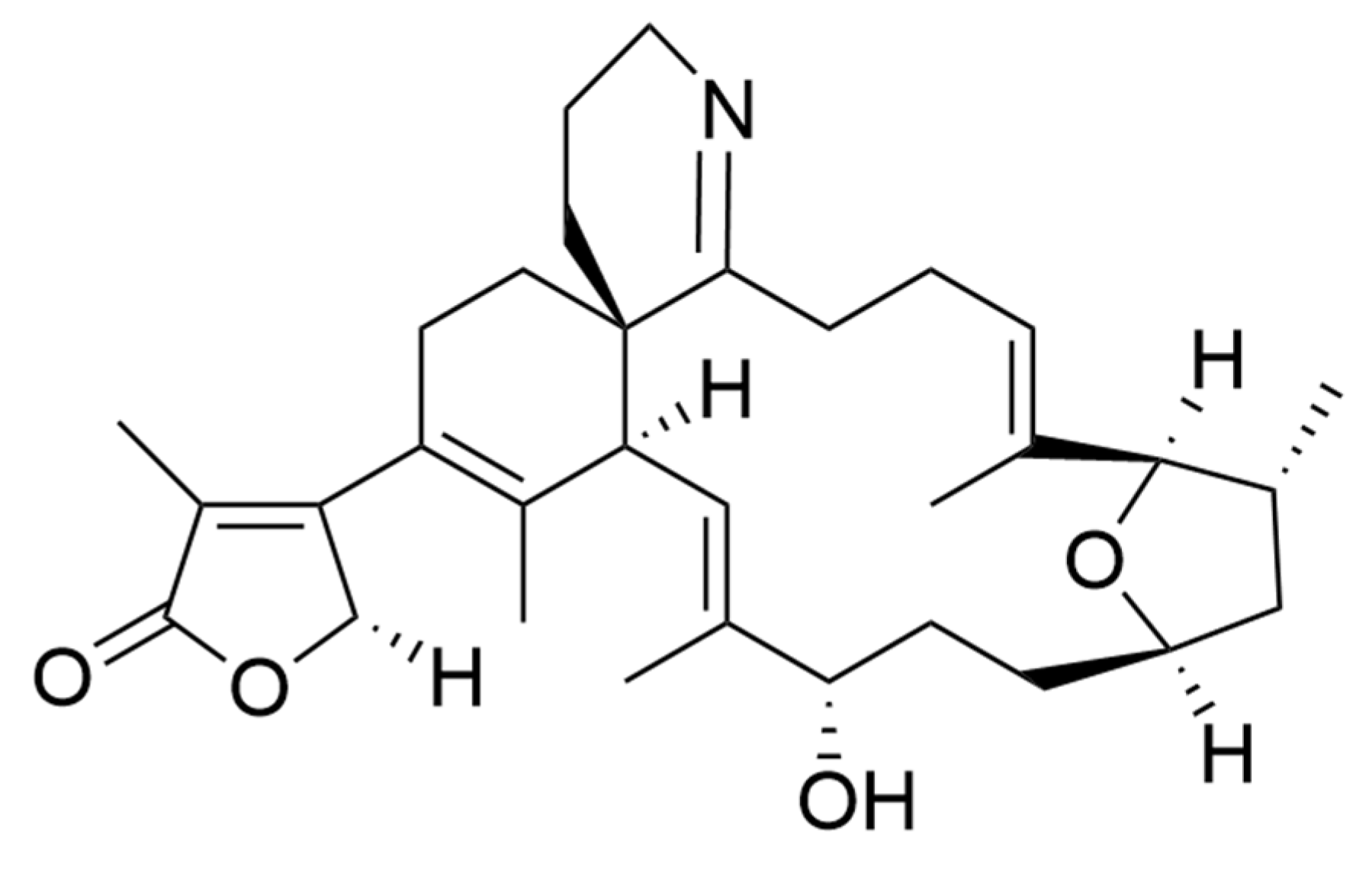

| Compound | Microalgae | Bioactivity | Concentration | Ref. |
|---|---|---|---|---|
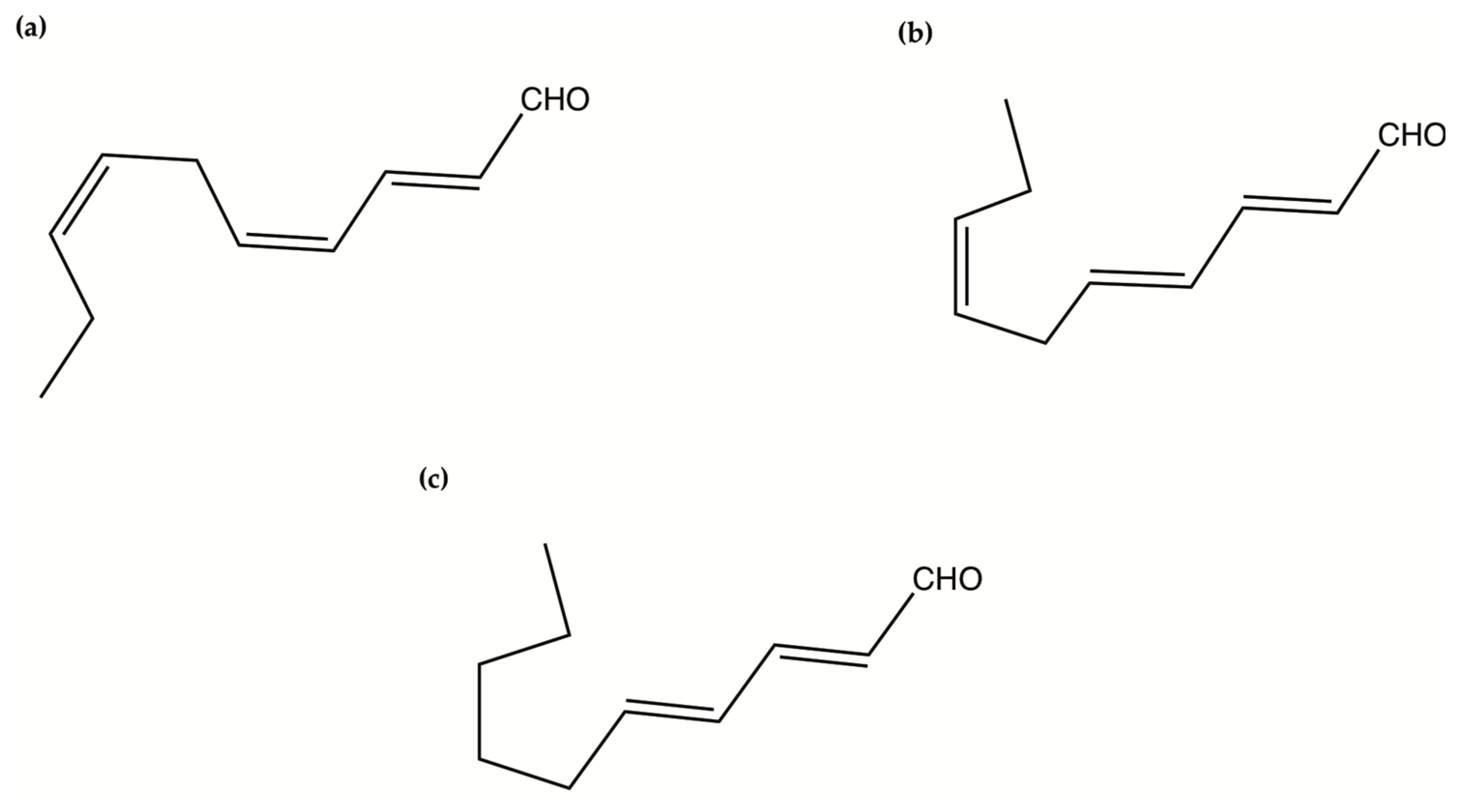
| Amphidinols 1, 2, 4, 5 and 6 (AM1, AM2, AM4, AM5, AM6) | Amphidinium klebsii | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: Minimum effect concentration (MEC) value 4 to 6 µg/disk In vitro Half maximum effective concentrations (EC50 values): 50 nM (AM1) 910 nM (AM2) 185 nM (AM4) 230 nM (AM5) 580 nM (AM6) | [204] |
| Amphidinol 2 (AM2) | Amphidinium klebsii | Anticancer activity against colon cancer cells HCT116, HT29, and breast cancer cells MCF7 | Half maximal inhibitory concentration (IC50) values: 6.18 µM (HCT116) 0.87 µM (HT29) 2.98 µM (MCF7) | [205] |
| Amphidinol 3 (AM3) | Amphidinium klebsii | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: MEC value 9 µg/disk In vitro EC50 value: 250 nM | [206] |
| Amphidinol 7 (AM7) | Amphidinium klebsii | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: MEC value 10 µg/disk In vitro EC50 value: 300 nM | [207] |
| Amphidinol 9 (AM9) | Amphidinium carterae | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: MEC value 10 µg/disk In vitro EC50 value: 300 nM | [200] |
| Amphidinols 10, 11, 12 and 13 (AM10, AM11, AM12 and AM13) | Amphidinium carterae | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: MEC value >100 µg/disk In vitro EC50 value: >2000 nM | [200] |
| Amphidinols 14 and 15 (AM14 and AM15) | Amphidinium klebsii | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: MEC value >60 µg/disk In vitro EC50 value: >50 µM | [199] |
| Amphidinol 17 (AM17) | Amphidinium carterae | Antifungal activity against Aspergillus niger, and Candida kefyr. Haemolytic activity on human erythrocytes | In vitro disk assay: not detectable antifungal activity In vitro EC50 value: >4.5 µM | [208] |
| Amphidinols 18 and 19 (AM18 and AM19) | Amphidinium carterae | Antifungal activity against Candida albicans | In vitro growth inhibition assay: MIC values 9 µg/mL (AM18) Not detectable activity (AM19) | [201] |
| Amphidinols 20 and 21 (AM20 and AM21) | Amphidinium carterae | Antifungal activity against Aspergillus niger Haemolytic activity on human erythrocytes | In vitro disk assay: MEC value >15 µg/disk In vitro EC50 value: 1–3 µM (AM20) >10 µM (AM21) | [202] |
| Amphidinol 22 (AM22) | Amphidinium carterae | Antifugal activity against Candida albicans and Aspergillus fumigatus Anticancer activity against several cancer cell lines | Minimum inhibitory concentration value (MIC): 64 µg/mL In vitro: Half maximal inhibitory concentration (IC50) values: from 6 to 16 µM | [203] |
| Amphidinols A and B (AM-A and AM-B) | Amphidinium carterae | Antifungal activity against Candida albicans | In vitro growth inhibition assay: MIC values 19 µg/mL (AM-A) >150 µg/mL (AM-B) | [209] |
| Karantungiol A | Amphidinium sp. | Antifungal activity against NBRC4407 Aspergillus niger Antiprotozoal activity against Trichomonas foetus | In vitro disk assay: 12 µg/disc In vitro Antiprotozoal assay: MIC 1 µg/mL | [210] |
| Amphidinolides H and N (AMP-H and AMP-N) | Amphidinium sp. | Anticancer activity against L1210 murine leukemia cells and KB human epidermoid carcinoma cells | Half maximal inhibitory concentration (IC50) values: 0.48 ng/mL (AMP-H, L1210) 0.52 ng/mL (AMP-H, KB) 0.05 ng/mL (AMP-N, L1210) 0.06 ng/mL (AMP-N, KB) | [211] |
| Amphirionin 2 | Amphidinium sp. | Anticancer activity against colon cancer Caco-2 and lung cancer A549 cells | Half maximal inhibitory concentration (IC50) values: 100 ng/mL (Caco-2) 600 ng/mL (A549) | [212] |
| Gambieric acids A and B | Gambierdiscus toxicus | Antifungal activity against several fungal strains | In vitro growth inhibition assay: MIC values 0.2 to 6.25 µg/mL depending on compound/strain | [213] |
| Goniodomin A | Goniodoma pseudogoniaulax | Antifungal activity against Mortierella ramannianus and Candida albicans Reduction on metabolic rate of BE(2)-M17 human neuroblastoma cells | Growth inhibition at a concentration of 0.5 µg/mL In vitro: 50% decreased metabolic rate after 6-h incubation with 15 μM | [214,215] |
| Compound | Microalgae | Bioactivity | Concentration/dosing | Ref. |
|---|---|---|---|---|
| Saxitoxin | Alexandrium sp. | Local anesthetic (Rat sciatic nerve) | In vivo: 58 ± 3 nmol/mL (for 60 min analgesia) | [222] |
| Neosaxitoxin | Alexandrium sp. | Local anesthetic (Rat sciatic nerve) Bladder pain blocker (Human) | In vivo: 34 ± 2 nmol/mL (for 60 min analgesia) In vivo: dose of 80 µg, with successful prolonged pain reduction in all the patients. | [222] |
| Gonyautoxins 2/3 | Alexandrium sp. | Chronic headache (Human) | In vivo: dose of 50 µg (70% of the patients responded, long lasting effect) | [223] |
| Tetrodotoxin | Alexandrium tamarense | Acute, inflammatory and neuropathic pain (animal models) Several to moderate cancer-related pain (human) | In vivo: several different doses/ application methods In vivo: eight doses of 30 µg (during 4 days) for prolonged pain relief | [224,225] |
| Okadaic acid | Porocentrum lima | Antifungal activity on Aspergillus niger and Penicillium funiculosum | Disc test: 10 µg/disc (inhibition circle observed) | [213] |
| Yessotoxin | Protoceratium reticulatum | Cell death inducer in several cancer cell lines (27 out of 58 cell lines) Antiproliferative activity on BC3H1 myoblast cells Antiproliferative activity on RBL-2H3 and B16F10 melanoma cells Anti-Alzheimer activity | In vitro: nanomolar scale concentrations, different mechanisms In vitro: 100 nM induced autophagy in BC3H1 cells In vitro: 10–100 nM drastically reduced cell viability by apoptotic cell death In vitro: 1 nM reduced β-amyloid deposition and Tau protein hyperphosphorylation | [226,227,228,229] |
| Pectenotoxin 2 | Dinophysis sp. | Antiproliferative activity of several cancer cell lines, particularly strong for COLO205 colon cancer cells. | In vitro: LC50 value of 8 nM for COLO205 | [230] |
| Maitotoxin 3 | Gambierdiscus belizeanus | Antiproliferative activity on SH-SY5Y human neuroblastoma cells | In vitro: IC50 value of 0.7 µM | [231] |
| Gambierol | Gambierdiscus toxicus | Immunomodulatory activity on immune cells expressing K+ channels | In vitro Kv1.1–1.5 channel inhibition: IC50 values at the nanomolar scale | [232] |
| Brevetoxins 2, 3, 6, 9 | Karenia brevis | Antiproliferative activity on leukemic cells Jurkat E6-1 | In vitro: from 5 to 60 mM. Brevetoxin 2 was the most potent with IC50 5.6 mM | [233] |
| Brevenal | Karenia brevis | Anti-Inflammatory activity on adenocarcinoma cell line A549 Anti-Inflammatory activity on murine macrophages RAW 264.7 | 0.1 and 1 nM reduced the level of pro-inflammatory chemokine IL-8 0.1 and 1 pM reduced the level of pro-inflammatory cytokine TNF-α | [234] |
| Gymnodimine | Gymnodinium sp. | Anti-Alzheimer activity by reduction on intracellular amyloid-beta levels and reduction of tau hyperphosphorylation | 50 nM reduced β-amyloid expression by 20.9 ± 0.6% (3–7 days in culture) and decreased tau hyperphosphorylation by 34–37% | [235] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saide, A.; Martínez, K.A.; Ianora, A.; Lauritano, C. Unlocking the Health Potential of Microalgae as Sustainable Sources of Bioactive Compounds. Int. J. Mol. Sci. 2021, 22, 4383. https://doi.org/10.3390/ijms22094383
Saide A, Martínez KA, Ianora A, Lauritano C. Unlocking the Health Potential of Microalgae as Sustainable Sources of Bioactive Compounds. International Journal of Molecular Sciences. 2021; 22(9):4383. https://doi.org/10.3390/ijms22094383
Chicago/Turabian StyleSaide, Assunta, Kevin A. Martínez, Adrianna Ianora, and Chiara Lauritano. 2021. "Unlocking the Health Potential of Microalgae as Sustainable Sources of Bioactive Compounds" International Journal of Molecular Sciences 22, no. 9: 4383. https://doi.org/10.3390/ijms22094383
APA StyleSaide, A., Martínez, K. A., Ianora, A., & Lauritano, C. (2021). Unlocking the Health Potential of Microalgae as Sustainable Sources of Bioactive Compounds. International Journal of Molecular Sciences, 22(9), 4383. https://doi.org/10.3390/ijms22094383








