The Role of Porphyrinoid Photosensitizers for Skin Wound Healing
Abstract
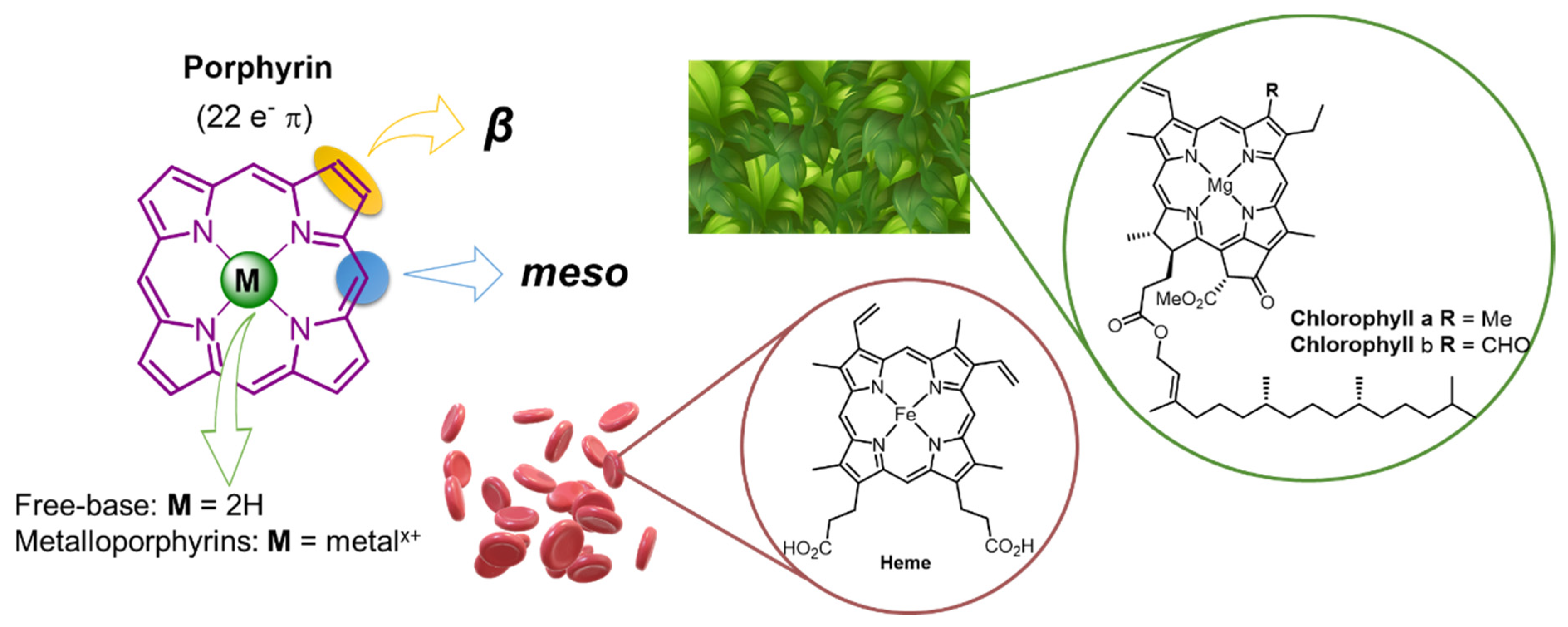
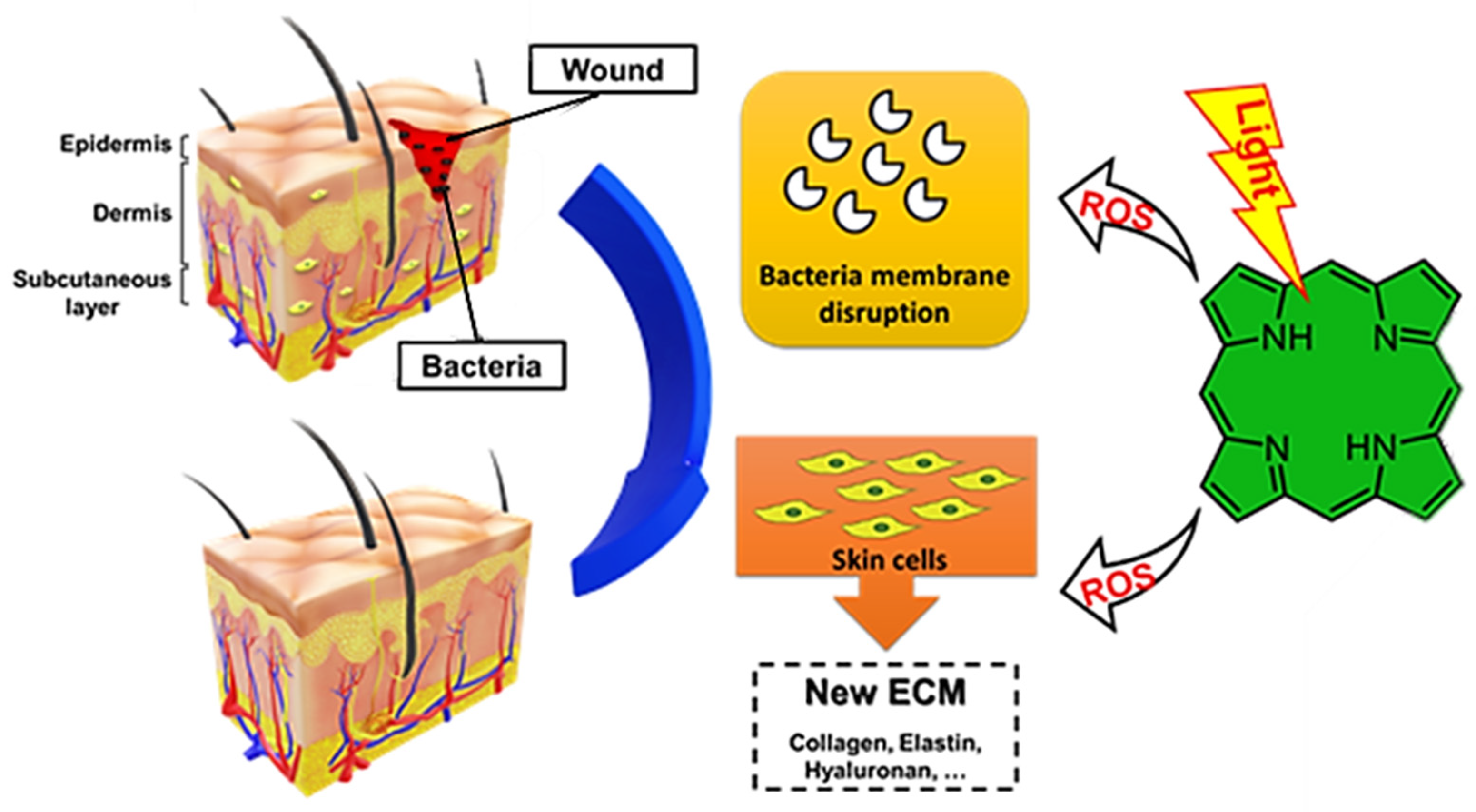
1. Introduction
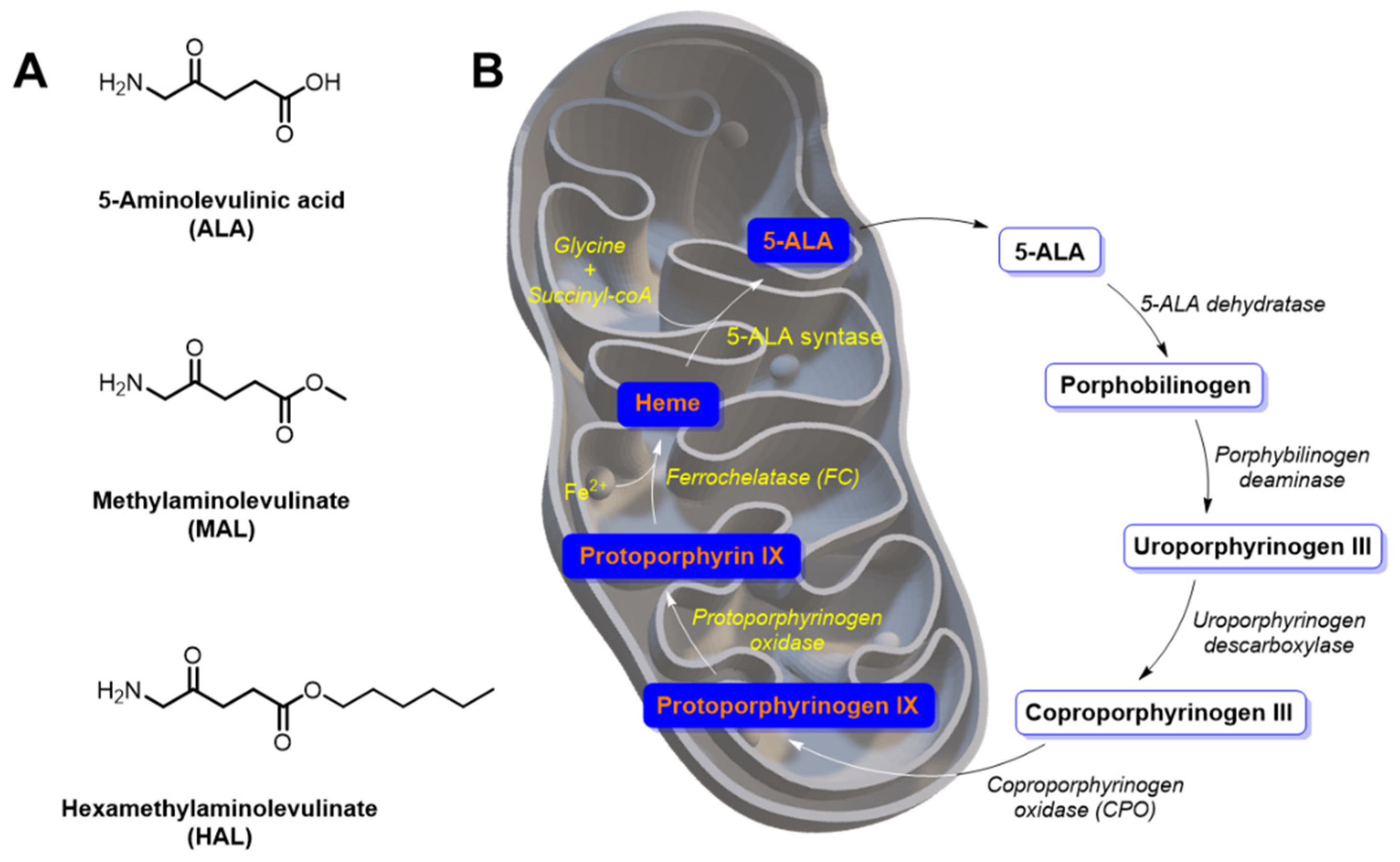
2. 5-Aminolevulinic Acid and Derivatives as Precursors of Endogenous Protoporphyrin-IX
3. Tetrapyrrolic Macrocycles with Natural Origin
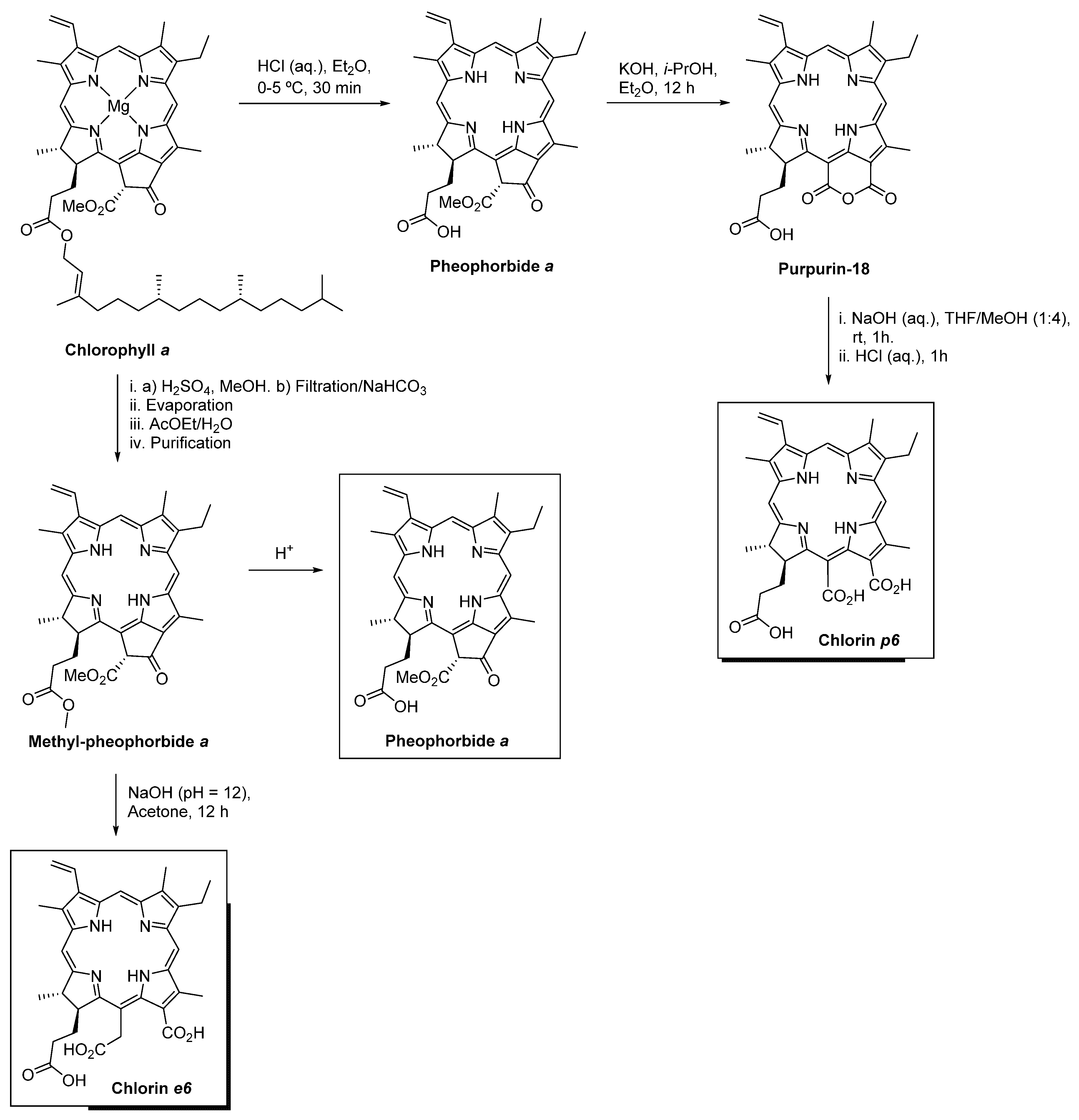
3.1. Pheophorbide a
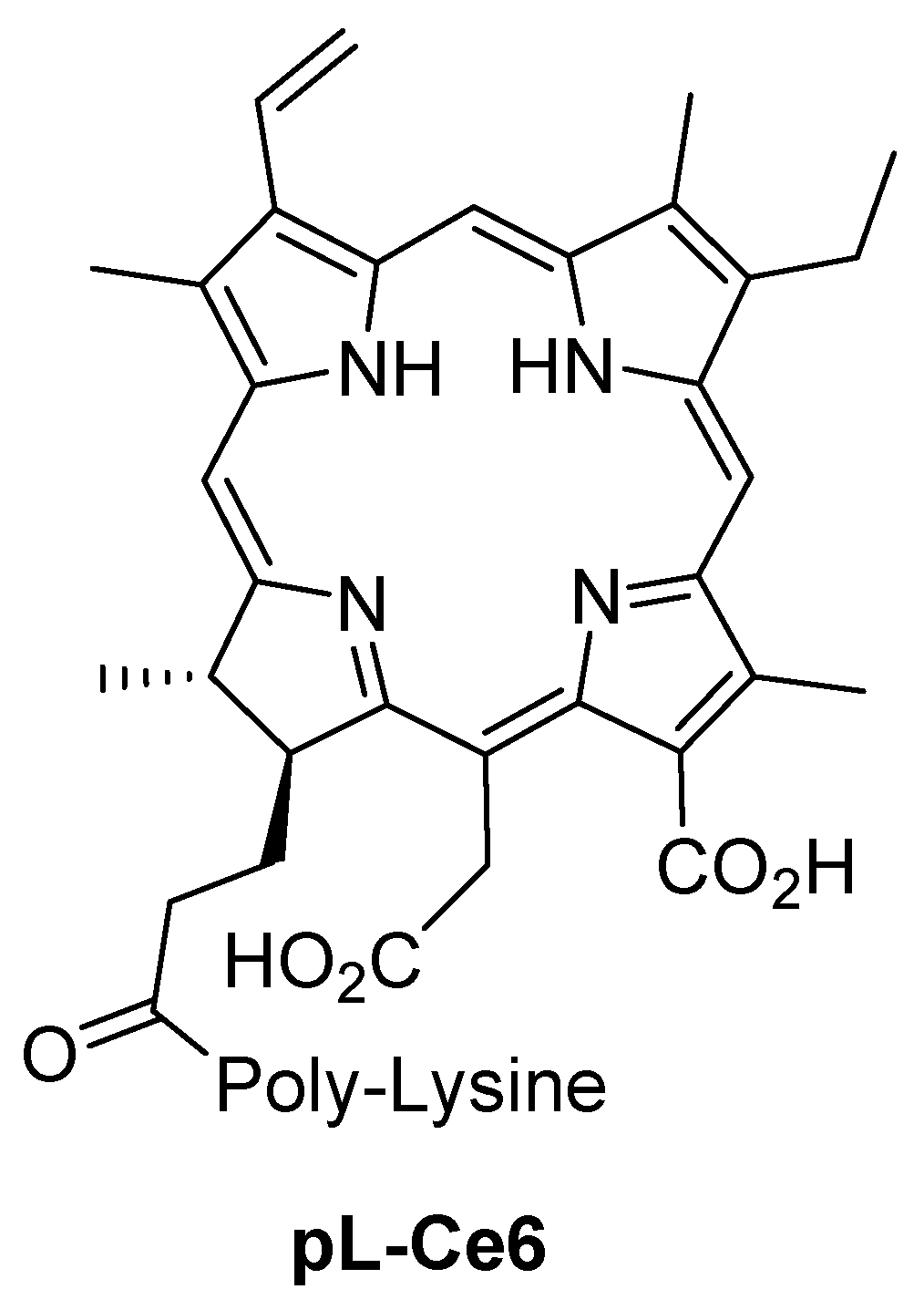
3.2. Chlorin e6
3.3. Chlorin p6
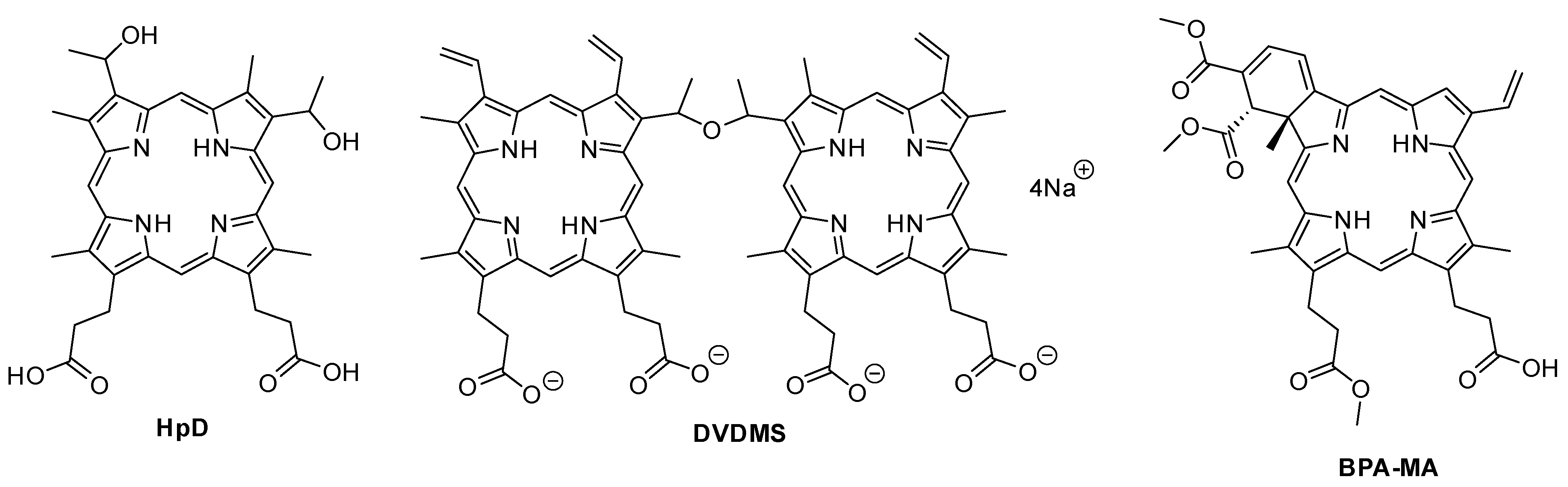
3.4. Sinoporphyrin Sodium
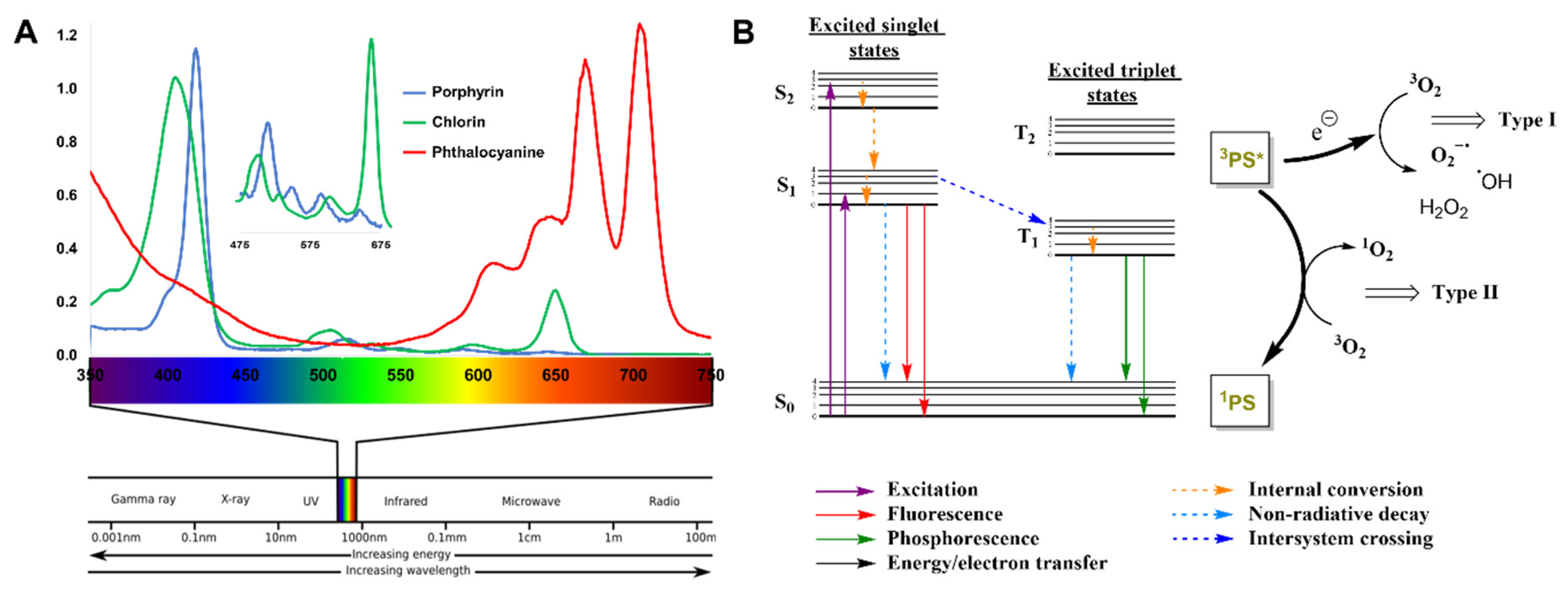
4. Synthetic Tetrapyrrolic Macrocycles
4.1. Porphyrins and Chlorins
4.2. Phthalocyanines
5. Final Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AgSD | Silver sulfadiazine cream |
| ALA | 5-Aminolevulinic acid |
| AlPcCl | Aluminium complex of phthalocyanine |
| BPD-MA | Benzoporphyrin mono-carboxylic acid |
| CASP | Chloroaluminum sulfonated phthalocyanine |
| Ce6 | Chlorin e6 |
| Cp6 | Chlorin p6 |
| CS | Chitosan |
| CVU | Chronic venous ulcers |
| DFUs | Diabetic foot ulcers |
| EDC | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide |
| EDTA | Ethylenediamine tetraacetic acid |
| EGCG | (–)-Epigallocatechin-3-gallate |
| FDA | Food and drug administration |
| GF | Growth factors |
| GTMAC | Glycidyltrimethylammonium chloride |
| HAL | Hexyl aminolevulinate |
| HpD | Hematoporphyrin derivative |
| ICCD | Intensified charge-coupled device |
| IL | Ionic liquid |
| LED | Light-emitting diode |
| LLLT | Low-level laser irradiation |
| MAL | Methyl aminolevulinate |
| MMP | Metalloproteinases |
| MOF | Metal-organic framework |
| MRAB | Multidrug-resistant Acinetobacter baumannii |
| MREC | Multidrug-resistant Escherichia coli |
| MDR | Multidrug-resistant |
| MRSA | Methicillin-resistant Staphylococcus aureus |
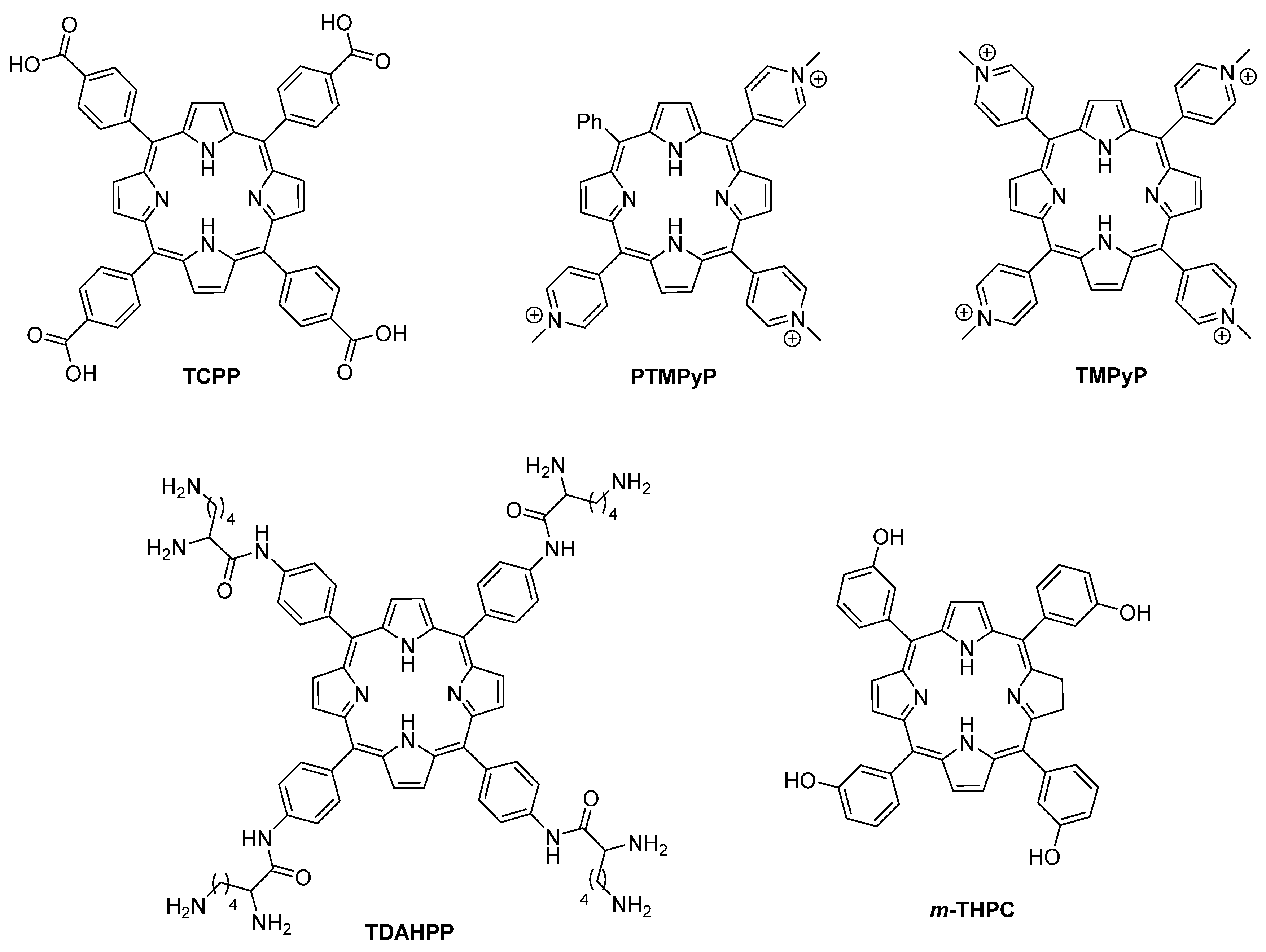
| m-THPC | 5,10,15,20-Tetrakis(3-hydroxyphenyl)chlorin |
| NHS | N-hydroxysuccinimide |
| NMB | New Methylene Blue |
| NP | Nanoparticle |
| Pc | Phthalocyanines |
| PDI | Photodynamic inactivation |
| PF | Pluronic F127 |
| pL-Ce6 | Poly-L-lysine chlorin e6 |
| pL-Cp6 | Poly-L-lysine chlorin p6 |
| PB@MOF | Prussian Blue MOF |
| PEI | Polyethyleneimine |
| PP-IX | Protoporphyrin-IX |
| PTMPP | 5-Phenyl-10,15,20-tris(1-methylpyridinium-4-yl)porphyrin trichloride |
| PVP | Polyvinylpyrrolidone |
| RB | Rose Bengal |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| TBO | Toluidine blue O |
| TCPP | 5,10,15,20-Tetrakis(4-carboxyphenyl)porphyrin |
| TGF | Transforming growth factor |
| TLD141 | [Ru(2,2′-bipyridine)2(2-(2′,2″:5″,2‴-terthiophene)-imidazo-[4,5-f][1,10]phenantroline]2+ |
| TMPyP | 5,10,15,20-Tetrakis(1-methylpyridinium-4-yl)porphyrin |
| TPP | 5,10,15,20-Tetraphenylporphyrin |
| VC12IM | 1-Vinyl-3-dodecylimidazolium bromide |
| VCM | Vancomycin |
| WHO | World Health Organization |
| WHOC | Wound healing organ culture |
| γ-PGA | Poly(γ-glutamic acid) |
References
- Kadish, K.M.; Smith, K.M.; Guilard, R. Handbook of Porphyrin Science; World Scientific Publishing Company: Singapore, 2010. [Google Scholar]
- Cavaleiro, J.A.S.; Tomé, A.C.; Neves, M.G.P.M.S. 9 meso-tetraarylporphyrin derivatives: New synthetic methodologies. In Handbook of Porphyrin Science; Kadish, K.M., Smith, K.M., Guilard, R., Eds.; World Scientific: Singapore, 2010; Volume 2, pp. 193–294. [Google Scholar]
- Cerqueira, A.F.R.; Moura, N.M.M.; Serra, V.V.; Faustino, M.A.F.; Tomé, A.C.; Cavaleiro, J.A.S.; Neves, M.G.P.M.S. β-formyl- and β-vinylporphyrins: Magic building blocks for novel porphyrin derivatives. Molecules 2017, 22, 1269. [Google Scholar] [CrossRef]
- Shepherd, M.; Medlock, A.E.; Dailey, H.A. Porphyrin metabolism. In Encyclopedia of Biological Chemistry, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; pp. 544–549. ISBN 9780123786319. [Google Scholar]
- Taniguchi, M.; Lindsey, J.S. Synthetic chlorins, possible surrogates for chlorophylls, prepared by derivatization of porphyrins. Chem. Rev. 2017, 117, 344–535. [Google Scholar] [CrossRef]
- Di Carlo, G.; Biroli, A.O.; Tessore, F.; Caramori, S.; Pizzotti, M. beta-Substituted Zn-II porphyrins as dyes for DSSC: A possible approach to photovoltaic windows. Coord. Chem. Rev. 2018, 358, 153–177. [Google Scholar] [CrossRef]
- Kundu, S.; Patra, A. Nanoscale strategies for light harvesting. Chem. Rev. 2017, 117, 712–757. [Google Scholar] [CrossRef] [PubMed]
- Di Carlo, G.; Biroli, A.O.; Pizzotti, M.; Tessore, F. Efficient sunlight harvesting by A(4) beta-pyrrolic substituted Zn-II Porphyrins: A mini-review. Front. Chem. 2019, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Birel, O.; Nadeem, S.; Duman, H. Porphyrin-based dye-sensitized solar cells (DSSCs): A review. J. Fluoresc. 2017, 27, 1075–1085. [Google Scholar] [CrossRef]
- Singhal, A. Meso-linked multiporphyrins as model for light harvesting systems: Review. Nat. Prod. Chem. Res. 2017, 5, 259. [Google Scholar] [CrossRef]
- Li, L.L.; Diau, E.W.G. Porphyrin-sensitized solar cells. Chem. Soc. Rev. 2013, 42, 291–304. [Google Scholar] [CrossRef]
- Urbani, M.; Grätzel, M.; Nazeeruddin, M.K.; Torres, T. Meso-substituted porphyrins for dye-sensitized solar cells. Chem. Rev. 2014, 114, 12330–12396. [Google Scholar] [CrossRef]
- Otsuki, J. Supramolecular approach towards light-harvesting materials based on porphyrins and chlorophylls. J. Mater. Chem. A 2018, 6, 6710–6753. [Google Scholar] [CrossRef]
- Guan, W.; Zhou, W.; Lu, J.; Lu, C. Luminescent films for chemo- and biosensing. Chem. Soc. Rev. 2015, 44, 6981–7001. [Google Scholar] [CrossRef]
- Lee, H.; Hong, K.I.; Jang, W.D. Design and applications of molecular probes containing porphyrin derivatives. Coord. Chem. Rev. 2018, 354, 46–73. [Google Scholar] [CrossRef]
- Paolesse, R.; Nardis, S.; Monti, D.; Stefanelli, M.; Di Natale, C. Porphyrinoids for chemical sensor applications. Chem. Rev. 2017, 117, 2517–2583. [Google Scholar] [CrossRef] [PubMed]
- Moura, N.; Núñez, C.; Santos, S.; Faustino, M.; Cavaleiro, J.; Neves, M.G.P.M.S.; Capelo-Martínez, J.L.; Lodeiro, C. Synthesis, spectroscopy studies, and theoretical calculations of new fluorescent probes based on pyrazole containing porphyrins for Zn(II), Cd(II), and Hg(II) optical detection. Inorg. Chem. 2014, 53, 6149–6158. [Google Scholar] [CrossRef] [PubMed]
- Monti, D.; Nardis, S.; Stefanelli, M.; Paolesse, R.; Di Natale, C.; D’Amico, A. Porphyrin-based nanostructures for sensing applications. J. Sens. 2009, 2009, 1–10. [Google Scholar] [CrossRef]
- Ding, Y.; Zhu, W.H.; Xie, Y. Development of ion chemosensors based on porphyrin analogues. Chem. Rev. 2017, 117, 2203–2256. [Google Scholar] [CrossRef]
- Moura, N.M.M.; Núñez, C.; Santos, S.M.; Faustino, M.A.F.; Cavaleiro, J.A.S.; Almeida Paz, F.A.; Neves, M.G.P.M.S.; Capelo, J.L.; Lodeiro, C. A New 3,5-bisporphyrinylpyridine derivative as a fluorescent ratiometric probe for zinc ions. Chem. A Eur. J. 2014, 20, 6684–6692. [Google Scholar] [CrossRef]
- Pegis, M.L.; Wise, C.F.; Martin, D.J.; Mayer, J.M. Oxygen reduction by homogeneous molecular catalysts and electrocatalysts. Chem. Rev. 2018, 118, 2340–2391. [Google Scholar] [CrossRef]
- Ali, A.; Akram, W.; Liu, H.-Y. Reactive cobalt–oxo complexes of tetrapyrrolic macrocycles and N-based ligand in oxidative transformation reactions. Molecules 2018, 24, 78. [Google Scholar] [CrossRef] [PubMed]
- Santos, E.; Carvalho, C.; Terzi, C.; Nakagaki, S. Recent advances in catalyzed sequential reactions and the potential use of tetrapyrrolic macrocycles as catalysts. Molecules 2018, 23, 2796. [Google Scholar] [CrossRef]
- Zhang, W.; Lai, W.; Cao, R. Energy-related small molecule activation reactions: Oxygen reduction and hydrogen and oxygen evolution reactions catalyzed by porphyrin- and corrole-based systems. Chem. Rev. 2017, 117, 3717–3797. [Google Scholar] [CrossRef]
- Costentin, C.; Robert, M.; Savéant, J.M. Current issues in molecular catalysis illustrated by iron porphyrins as catalysts of the CO2-to-CO electrochemical conversion. Acc. Chem. Res. 2015, 48, 2996–3006. [Google Scholar] [CrossRef] [PubMed]
- Costas, M. Selective C-H oxidation catalyzed by metalloporphyrins. Coord. Chem. Rev. 2011, 255, 2912–2932. [Google Scholar] [CrossRef]
- Da Silva, E.S.; Moura, N.M.M.; Neves, M.G.P.M.S.; Coutinho, A.; Prieto, M.; Silva, C.G.; Faria, J.L. Novel hybrids of graphitic carbon nitride sensitized with free-base meso-tetrakis(carboxyphenyl) porphyrins for efficient visible light photocatalytic hydrogen production. Appl. Catal. B Environ. 2018, 221, 56–69. [Google Scholar] [CrossRef]
- Calvete, M.J.F.; Pinto, S.M.A.; Pereira, M.M.; Geraldes, C.F.G.C. Metal coordinated pyrrole-based macrocycles as contrast agents for magnetic resonance imaging technologies: Synthesis and applications. Coord. Chem. Rev. 2017, 333, 82–107. [Google Scholar] [CrossRef]
- Ethirajan, M.; Chen, Y.; Joshi, P.; Pandey, R.K. The role of porphyrin chemistry in tumor imaging and photodynamic therapy. Chem. Soc. Rev. 2011, 40, 340–362. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, L.K.; Bryant, H.E.; Weinstein, J.A. Transition metal complexes as photosensitisers in one- and two-photon photodynamic therapy. Coord. Chem. Rev. 2019, 379, 2–29. [Google Scholar] [CrossRef]
- Mesquita, M.Q.; Dias, C.J.; Neves, M.G.P.M.S.; Almeida, A.; Faustino, M.A.F. Revisiting current photoactive materials for antimicrobial photodynamic therapy. Molecules 2018, 23, 2424. [Google Scholar] [CrossRef]
- Dias, C.J.; Sardo, I.; Moura, N.M.M.; Felgueiras, J.; Neves, M.G.P.M.S.; Fardilha, M.; Faustino, M.A.F. An efficient synthetic access to new uracil-alditols bearing a porphyrin unit and biological assessment in prostate cancer cells. Dye. Pigment. 2020, 173. [Google Scholar] [CrossRef]
- Kou, J.; Dou, D.; Yang, L. Porphyrin photosensitizers in photodynamic therapy and its applications. Oncotarget 2017, 8, 81591–81603. [Google Scholar] [CrossRef]
- Baglia, R.A.; Zaragoza, J.P.T.; Goldberg, D.P. Biomimetic reactivity of oxygen-derived manganese and iron porphyrinoid complexes. Chem. Rev. 2017, 117, 13320–13352. [Google Scholar] [CrossRef]
- Wen, X.; Li, Y.; Hamblin, M.R. Photodynamic therapy in dermatology beyond non-melanoma cancer: An update. Photodiagn. Photodyn. Ther. 2017, 19, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Van Straten, D.; Mashayekhi, V.; de Bruijn, H.; Oliveira, S.; Robinson, D. Oncologic photodynamic therapy: Basic principles, current clinical status and future directions. Cancers 2017, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.; Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef]
- Tanaka, T.; Osuka, A. Conjugated porphyrin arrays: Synthesis, properties and applications for functional materials. Chem. Soc. Rev. 2015, 44, 943–969. [Google Scholar] [CrossRef]
- Castro, K.A.D.F.; Moura, N.M.M.; Simões, M.M.Q.; Cavaleiro, J.A.S.; do Faustino, M.A.F.; Cunha, Â.; Paz, F.A.A.; Mendes, R.F.; Almeida, A.; Freire, C.S.R.; et al. Synthesis and characterization of photoactive porphyrin and poly(2-hydroxyethyl methacrylate) based materials with bactericidal properties. Appl. Mater. Today 2019, 16, 332–341. [Google Scholar] [CrossRef]
- Moura, N.M.M.; Esteves, M.; Vieira, C.; Rocha, G.M.S.R.O.; Faustino, M.A.F.; Almeida, A.; Cavaleiro, J.A.S.; Lodeiro, C.; Neves, M.G.P.M.S. Novel β-functionalized mono-charged porphyrinic derivatives: Synthesis and photoinactivation of Escherichia coli. Dye. Pigment. 2019, 160, 361–371. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Huang, B.; Nawaz, M.H.; Zhang, W. Recent advances of multi-dimensional porphyrin-based functional materials in photodynamic therapy. Coord. Chem. Rev. 2020, 420, 213410. [Google Scholar] [CrossRef]
- Liu, Y.; Qin, R.; Zaat, S.A.J.; Breukink, E.; Heger, M. Antibacterial photodynamic therapy: Overview of a promising approach to fight antibiotic-resistant bacterial infections. J. Clin. Transl. Res. 2015, 1, 140–167. [Google Scholar]
- Vieira, C.; Gomes, A.T.P.C.; Mesquita, M.Q.; Moura, N.M.M.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Almeida, A.; Neves, G.P.M.S.; Faustino, A.F.; Almeida, A. An insight into the potentiation effect of potassium iodide on aPDT efficacy. Front. Microbiol. 2018, 9, 2665. [Google Scholar] [CrossRef]
- Mesquita, M.Q.; Dias, C.J.; Gamelas, S.; Neves, M.G.P.M.S.; Almeida, A.; Faustino, M.A.F. An insight on the role of photosensitizer nanocarriers for Photodynamic Therapy. An. Acad. Bras. Ciênc. 2018, 90, 1101–1130. [Google Scholar] [CrossRef]
- De Freitas, L.F.; Hamblin, M.R. Antimicrobial photoinactivation with functionalized fullerenes. In Nanobiomaterials in Antimicrobial Therapy: Applications of Nanobiomaterials; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 1–27. ISBN 9780323428873. [Google Scholar]
- Almeida, A.; Faustino, M.A.F.; Tomé, J.P.C. Photodynamic inactivation of bacteria: Finding the effective targets. Future Med. Chem. 2015, 7, 1221–1224. [Google Scholar] [CrossRef]
- Ladeira, B.M.F.; Dias, C.J.; Gomes, A.T.P.C.; Tomé, A.C.; Neves, M.G.P.M.S.; Moura, N.M.M.; Almeida, A.; Faustino, M.A.F. Cationic pyrrolidine/pyrroline-substituted porphyrins as efficient photosensitizers against E. coli. Molecules 2021, 26, 464. [Google Scholar] [CrossRef]
- Moreira, X.; Santos, P.; Faustino, M.A.F.; Raposo, M.M.M.; Costa, S.P.G.; Moura, N.M.M.; Gomes, A.T.P.C.; Almeida, A.; Neves, M.G.P.M.S. An insight into the synthesis of cationic porphyrin-imidazole derivatives and their photodynamic inactivation efficiency against Escherichia coli. Dye. Pigment. 2020, 178, 108330. [Google Scholar] [CrossRef]
- Castro, K.A.D.F.; Moura, N.M.M.; Figueira, F.; Ferreira, R.I.; Simões, M.M.Q.; Cavaleiro, J.A.S.; Faustino, M.A.F.; Silvestre, A.J.D.; Freire, C.S.R.; Tomé, J.P.C.; et al. New materials based on cationic porphyrins conjugated to chitosan or titanium dioxide: Synthesis, characterization and antimicrobial efficacy. Int. J. Mol. Sci. 2019, 20, 2522. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Chen, Y.; Shi, J. Reactive oxygen species (ROS)-based nanomedicine. Chem. Rev. 2019, 119, 4881–4985. [Google Scholar] [CrossRef] [PubMed]
- Jaber, G.; Dariush, R.; Shahin, A.; Alireza, T.; Abbas, B. Photosensitizers in antibacterial photodynamic therapy: An overview. Laser Ther. 2018, 27, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, A.; Jesus, P. Low level energy photodynamic therapy for skin processes and regeneration. In Photomedicine—Advances in Clinical Practice; Tanaka, Y., Ed.; IntechOpen: London, UK, 2017; Volume 5, pp. 75–94. ISBN 978-953-51-3156-4. [Google Scholar]
- Vallejo, M.C.S.; Moura, N.M.M.; Ferreira Faustino, M.A.; Almeida, A.; Gonçalves, I.; Serra, V.V.; Neves, M.G.P.M.S. An insight into the role of non-porphyrinoid photosensitizers for skin wound healing. Int. J. Mol. Sci. 2020, 22, 234. [Google Scholar] [CrossRef]
- Silva, J.C.E.; Lacava, Z.G.M.; Kuckelhaus, S.; Silva, L.P.; Neto, L.F.M.; Sauro, E.E.; Tedesco, A.C. Evaluation of the use of low level laser and photosensitizer drugs in healing. Lasers Surg. Med. 2004, 34, 451–457. [Google Scholar] [CrossRef]
- Jang, Y.H.; Koo, G.-B.; Kim, J.-Y.; Kim, Y.-S.; Kim, Y.C. Prolonged activation of ERK contributes to the photorejuvenation effect in photodynamic therapy in human dermal fibroblasts. J. Investig. Dermatol. 2013, 133, 2265–2275. [Google Scholar] [CrossRef]
- Nelson, K.K.; Melendez, J.A. Mitochondrial redox control of matrix metalloproteinases. Free Radic. Biol. Med. 2004, 37, 768–784. [Google Scholar] [CrossRef] [PubMed]
- Rosique, R.G.; Rosique, M.J.; Farina Junior, J.A. Curbing inflammation in skin wound healing: A review. Int. J. Inflamm. 2015, 2015, 316235. [Google Scholar] [CrossRef]
- Nesi-Reis, V.; Lera-Nonose, D.S.S.L.; Oyama, J.; Silva-Lalucci, M.P.P.; Demarchi, I.G.; Aristides, S.M.A.; Teixeira, J.J.V.; Silveira, T.G.V.; Lonardoni, M.V.C. Contribution of photodynamic therapy in wound healing: A systematic review. Photodiagn. Photodyn. Ther. 2018, 21, 294–305. [Google Scholar] [CrossRef]
- Oyama, J.; Fernandes Herculano Ramos-Milaré, Á.C.; Lopes Lera-Nonose, D.S.S.; Nesi-Reis, V.; Galhardo Demarchi, I.; Alessi Aristides, S.M.; Juarez Vieira Teixeira, J.; Gomes Verzignassi Silveira, T.; Campana Lonardoni, M.V. Photodynamic therapy in wound healing in vivo: A systematic review. Photodiagn. Photodyn. Ther. 2020, 30, 101682. [Google Scholar] [CrossRef]
- Lucena, S.R.; Salazar, N.; Gracia-Cazaña, T.; Zamarrón, A.; González, S.; Juarranz, Á.; Gilaberte, Y. Combined treatments with photodynamic therapy for non-melanoma skin cancer. Int. J. Mol. Sci. 2015, 16, 25912–25933. [Google Scholar] [CrossRef] [PubMed]
- Reinhold, U. A review of BF-200 ALA for the photodynamic treatment of mild-to-moderate actinic keratosis. Future Oncol. 2017, 13, 2413–2428. [Google Scholar] [CrossRef]
- Theresia, C.; Zheng, J.; Chen, X.Y. Topical ALA-PDT as alternative therapeutic option in treatment-recalcitrant dermatosis: Report of 4 cases. Photodiagn. Photodyn. Ther. 2017, 20, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Morton, C.A.; Braathen, L.R. Daylight photodynamic therapy for actinic keratoses. Am. J. Clin. Dermatol. 2018, 19, 647–656. [Google Scholar] [CrossRef]
- Mahmoudi, K.; Garvey, K.L.; Bouras, A.; Cramer, G.; Stepp, H.; Jesu Raj, J.G.; Bozec, D.; Busch, T.M.; Hadjipanayis, C.G. 5-aminolevulinic acid photodynamic therapy for the treatment of high-grade gliomas. J. Neurooncol. 2019, 141, 595–607. [Google Scholar] [CrossRef]
- Champeau, M.; Vignoud, S.; Mortier, L.; Mordon, S. Photodynamic therapy for skin cancer: How to enhance drug penetration? J. Photochem. Photobiol. B Biol. 2019, 197, 111544. [Google Scholar] [CrossRef]
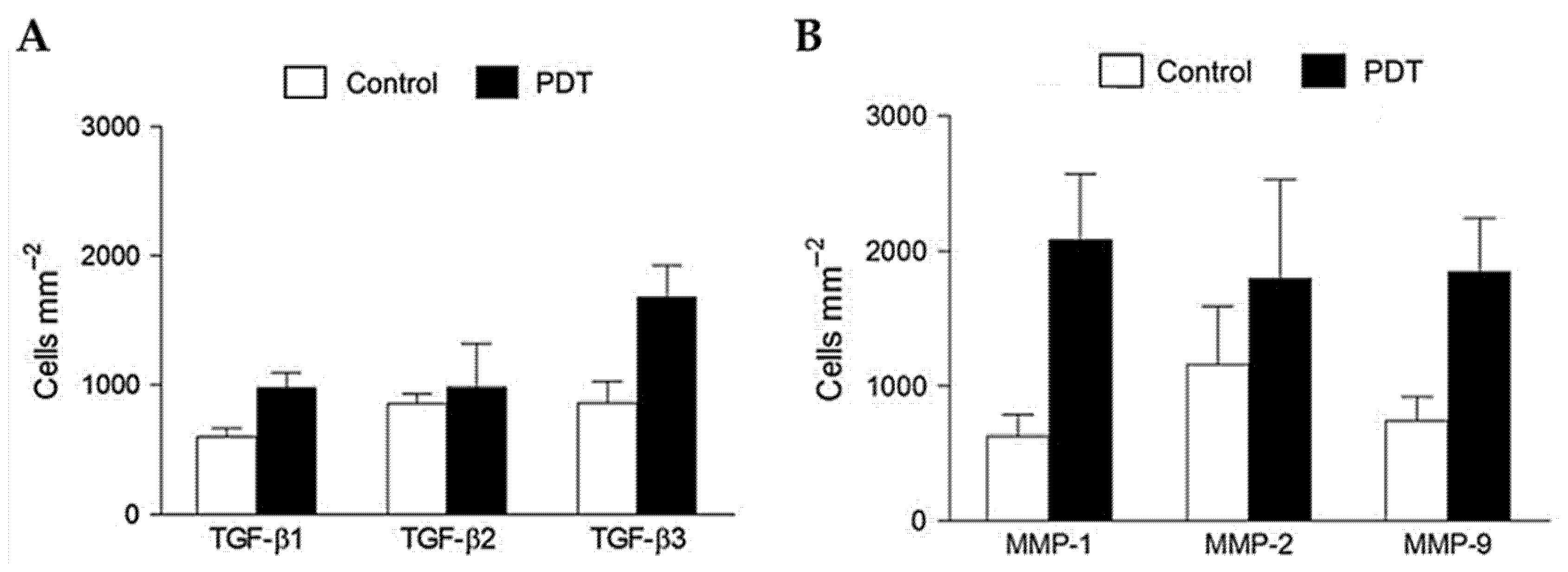
- Mills, S.J.; Farrar, M.D.; Ashcroft, G.S.; Griffiths, C.E.M.; Hardman, M.J.; Rhodes, L.E. Topical photodynamic therapy following excisional wounding of human skin increases production of transforming growth factor-β3 and matrix metalloproteinases 1 and 9, with associated improvement in dermal matrix organization. Br. J. Dermatol. 2014, 171, 55–62. [Google Scholar] [CrossRef]
- Mendoza-Garcia, J.; Sebastian, A.; Alonso-Rasgado, T.; Bayat, A. Optimization of an ex vivo wound healing model in the adult human skin: Functional evaluation using photodynamic therapy. Wound Repair Regen. 2015, 23, 685–702. [Google Scholar] [CrossRef]
- Lei, X.; Liu, B.; Huang, Z.; Wu, J. A clinical study of photodynamic therapy for chronic skin ulcers in lower limbs infected with Pseudomonas aeruginosa. Arch. Dermatol. Res. 2015, 307, 49–55. [Google Scholar] [CrossRef]
- Dyson, M.; Young, S. Effect of laser therapy on wound contraction and cellularity in mice. Lasers Med. Sci. 1986, 1, 125–130. [Google Scholar] [CrossRef]
- Rochkind, S.; Rousso, M.; Nissan, M.; Villarreal, M.; Barr-Nea, L.; Rees, D.G. Systemic effects of low-power laser irradiation on the peripheral and central nervous system, cutaneous wounds, and burns. Lasers Surg. Med. 1989, 9, 174–182. [Google Scholar] [CrossRef]
- Jayasree, R.S.; Gupta, A.K.; Rathinam, K.; Mohanan, P.V.; Mohanty, M. The influence of photodynamic therapy on the wound healing process in rats. J. Biomater. Appl. 2001, 15, 176–186. [Google Scholar] [CrossRef]
- Roatt, B.M.; de Aguiar-Soares, R.D.O.; Coura-Vital, W.; Ker, H.G.; das Moreira, N.D.; Vitoriano-Souza, J.; Giunchetti, R.C.; Carneiro, C.M.; Reis, A.B. Immunotherapy and immunochemotherapy in visceral leishmaniasis: Promising treatments for this neglected disease. Front. Immunol. 2014, 5, 272. [Google Scholar] [CrossRef]
- Abok, K.; Cadenas, E.; Brunk, U. An experimental model system for leishmaniasis. APMIS 1988, 96, 543–551. [Google Scholar] [CrossRef]
- Ghaffarifar, F.; Jorjani, O.; Mirshams, M.; Miranbaygi, M.H.; Hosseini, Z.K. Short communication: Photodynamic therapy as a new treatment of cutaneous leishmaniasis. East Mediterr. Health J. 2006, 12, 902–908. [Google Scholar]
- Evangelou, G.; Krasagakis, K.; Giannikaki, E.; Kruger-Krasagakis, S.; Tosca, A. Successful treatment of cutaneous leishmaniasis with intralesional aminolevulinic acid photodynamic therapy. Photodermatol. Photoimmunol. Photomed. 2011, 27, 254–256. [Google Scholar] [CrossRef]
- Kawczyk-Krupka, A.; Pucelik, B.; Międzybrodzka, A.; Sieroń, A.R.; Dąbrowski, J.M. Photodynamic therapy as an alternative to antibiotic therapy for the treatment of infected leg ulcers. Photodiagn. Photodyn. Ther. 2018, 23, 132–143. [Google Scholar] [CrossRef]
- Clayton, T.H.; Harrison, P.V. Photodynamic therapy for infected leg ulcers. Br. J. Dermatol. 2007, 156, 384–385. [Google Scholar] [CrossRef]
- Morimoto, K.; Ozawa, T.; Awazu, K.; Ito, N.; Honda, N.; Matsumoto, S.; Tsuruta, D. Photodynamic therapy using systemic administration of 5-aminolevulinic acid and a 410-nm wavelength light-emitting diode for methicillin-resistant staphylococcus aureus-infected ulcers in mice. PLoS ONE 2014, 9, e105173. [Google Scholar] [CrossRef] [PubMed]
- Katayama, B.; Ozawa, T.; Morimoto, K.; Awazu, K.; Ito, N.; Honda, N.; Oiso, N.; Tsuruta, D. Enhanced sterilization and healing of cutaneous pseudomonas infection using 5-aminolevulinic acid as a photosensitizer with 410-nm LED light. J. Dermatol. Sci. 2018, 90, 323–331. [Google Scholar] [CrossRef]
- Cappugi, P.; Comacchi, C.; Torchia, D. Photodynamic therapy for chronic venous ulcers. Acta Dermatovenerol. Croat. 2014, 22, 129–131. [Google Scholar]
- Berking, C.; Herzinger, T.; Flaig, M.J.; Brenner, M.; Borelli, C.; Degitz, K. The efficacy of photodynamic therapy in actinic cheilitis of the lower lip: A prospective study of 15 patients. Dermatol. Surg. 2007, 33, 825–830. [Google Scholar] [CrossRef]
- Stender, I.-M.; Wulf, H.C. Photodynamic therapy with 5-aminolevulinic acid in the treatment of actinic cheilitis. Br. J. Dermatol. 1996, 135, 454–456. [Google Scholar] [CrossRef]
- Alexiades-Armenakas, M.R.; Geronemus, R.G. Laser-mediated photodynamic therapy of actinic cheilitis. J. Drugs Dermatol. 2004, 3, 548–551. [Google Scholar]
- Hauschild, A.; Lischner, S.; Lange-Asschenfeldt, B.; Egberts, F. Treatment of actinic cheilitis using photodynamic therapy with methylaminolevulinate. Dermatol. Surg. 2005, 31, 1344–1348. [Google Scholar] [CrossRef]
- Devirgiliis, V.; Panasiti, V.; Fioriti, D.; Anzivino, E.; Bellizzi, A.; Cimillo, M.; Curzio, M.; Melis, L.; Roberti, V.; Gobbi, S.; et al. Antibacterial activity of methyl aminolevulinate photodynamic therapy in the treatment of a cutaneous ulcer. Int. J. Immunopathol. Pharmacol. 2011, 24, 793–795. [Google Scholar] [CrossRef]
- Gilbert, R.; Vickaryous, M.; Viloria-Petit, A. Signalling by transforming growth factor beta isoforms in wound healing and tissue regeneration. J. Dev. Biol. 2016, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Liarte, S.; Bernabé-García, Á.; Nicolás, F.J. Role of TGF-β in skin chronic wounds: A keratinocyte perspective. Cells 2020, 9, 306. [Google Scholar] [CrossRef]
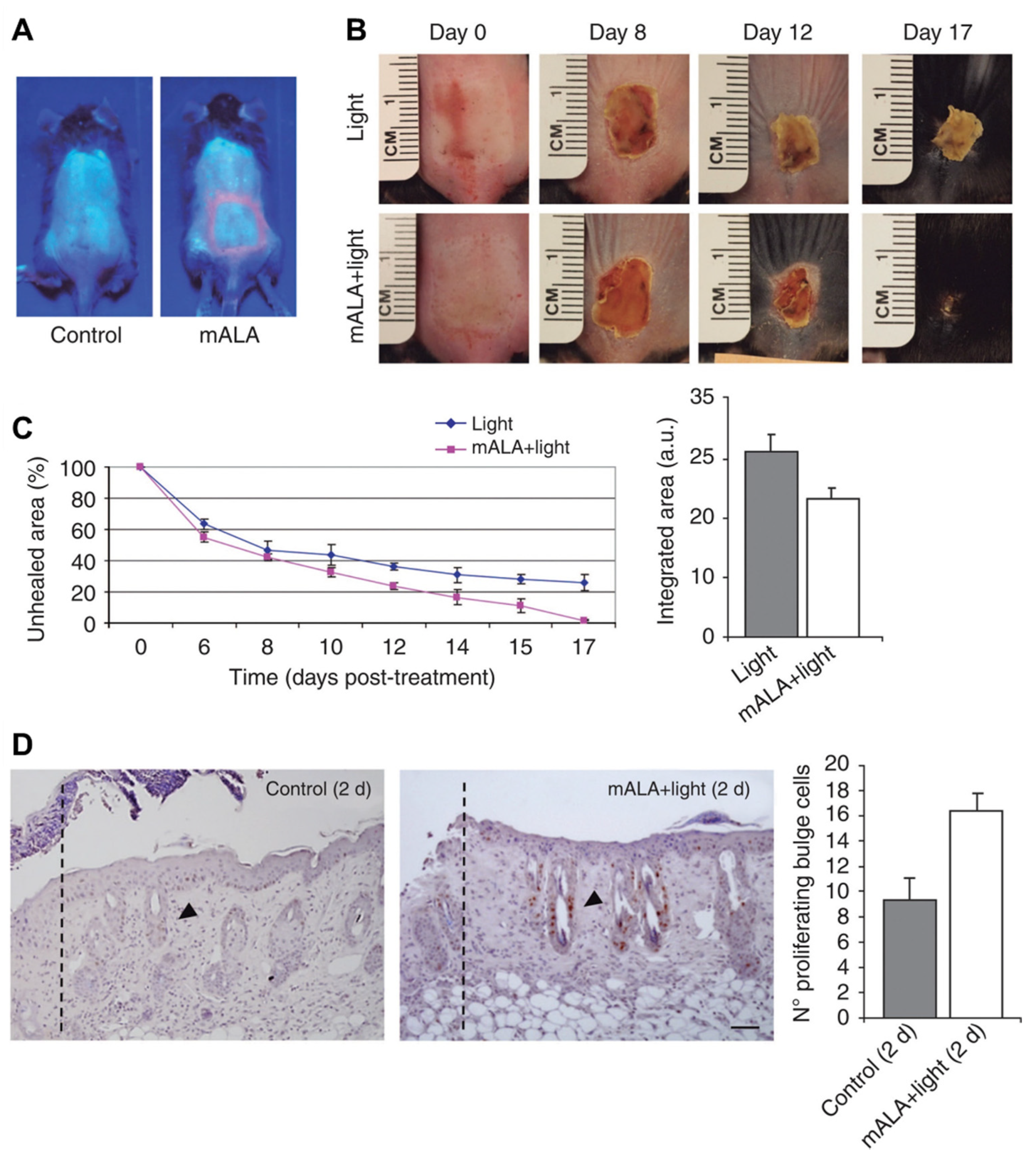
- Carrasco, E.; Calvo, M.I.; Blázquez-Castro, A.; Vecchio, D.; Zamarrón, A.; De Almeida, I.J.D.; Stockert, J.C.; Hamblin, M.R.; Juarranz, Á.; Espada, J. Photoactivation of ROS production in situ transiently activates cell proliferation in mouse skin and in the hair follicle stem cell niche promoting hair growth and wound healing. J. Invest. Dermatol. 2015, 135, 2611–2622. [Google Scholar] [CrossRef]
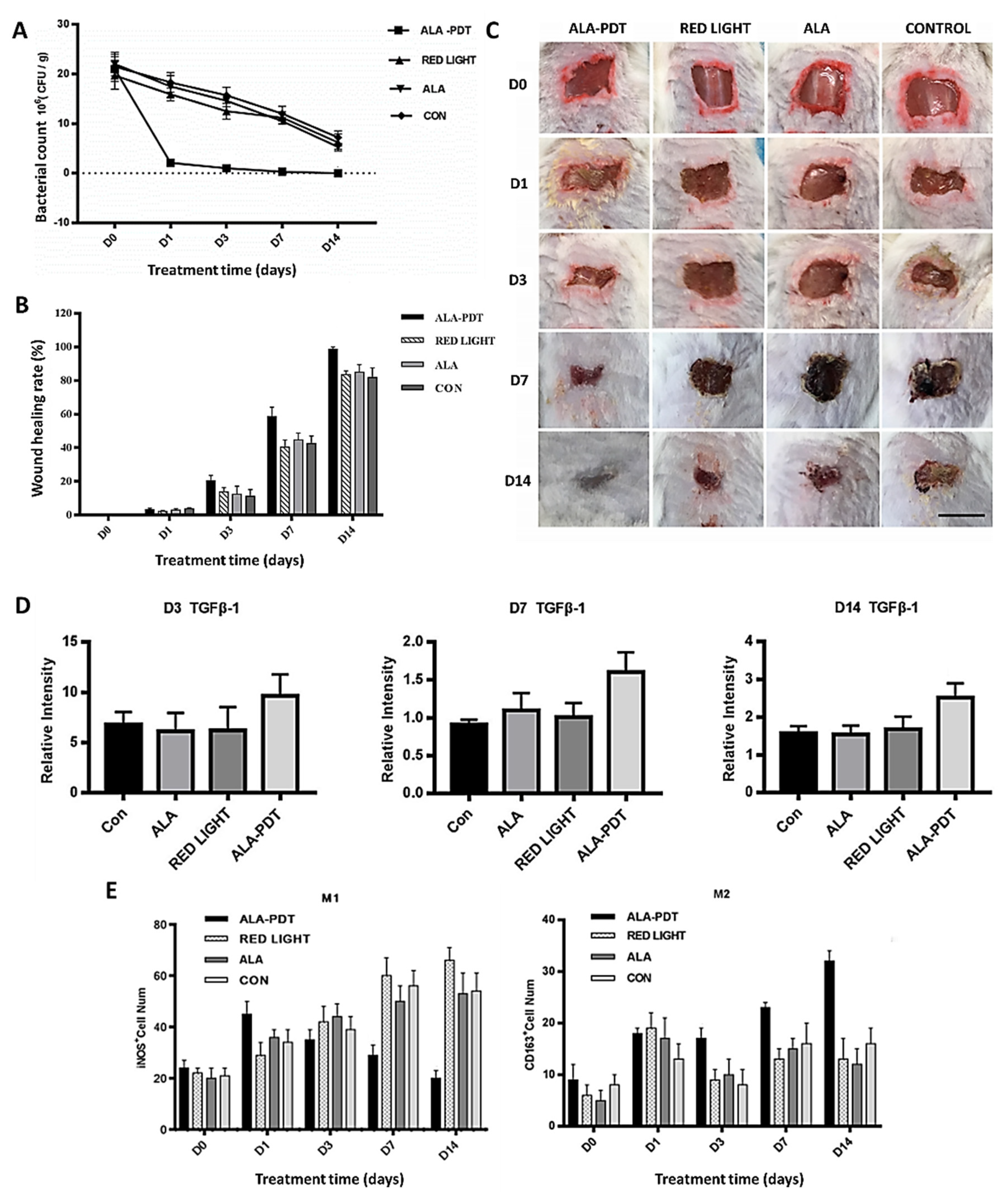
- Yang, T.; Tan, Y.; Zhang, W.; Yang, W.; Luo, J.; Chen, L.; Liu, H.; Yang, G.; Lei, X. Effects of ALA-PDT on the healing of mouse skin wounds infected with pseudomonas aeruginosa and its related mechanisms. Front. Cell Dev. Biol. 2020, 8, 1471. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.T.P.C.; Neves, M.G.P.M.S.; Cavaleiro, J.A.S. Cancer, photodynamic therapy and porphyrin-type derivatives. An. Acad. Bras. Ciênc. 2018, 90, 993–1026. [Google Scholar] [CrossRef]
- Östbring, K.; Rayner, M.; Sjöholm, I.; Otterström, J.; Albertsson, P.Å.; Emek, S.C.; Erlanson-Albertsson, C. The effect of heat treatment of thylakoids on their ability to inhibit in vitro lipase/co-lipase activity. Food Funct. 2014, 5, 2157–2165. [Google Scholar] [CrossRef]
- Uliana, M.P.; Pires, L.; Pratavieira, S.; Brocksom, T.J.; De Oliveira, K.T.; Bagnato, V.S.; Kurachi, C. Photobiological characteristics of chlorophyll a derivatives as microbial PDT agents. Photochem. Photobiol. Sci. 2014, 13, 1137–1145. [Google Scholar] [CrossRef]
- Tamiaki, H.; Fukai, K.; Shimazu, H.; Nishide, K.; Shibata, Y.; Itoh, S.; Kunieda, M. Covalently linked zinc chlorophyll dimers as a model of a chlorophyllous pair in photosynthetic reaction centers. Photochem. Photobiol. Sci. 2008, 7, 1231–1237. [Google Scholar] [CrossRef]
- Saide, A.; Lauritano, C.; Ianora, A. Pheophorbide a: State of the art. Mar. Drugs 2020, 18, 257. [Google Scholar] [CrossRef]
- Pérez-Gálvez, A.; Roca, M. Phyllobilins: A new group of bioactive compounds. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; Volume 52, pp. 159–191. [Google Scholar]
- Lyapina, E.A.; Machneva, T.V.; Larkina, E.A.; Tkachevskaya, E.P.; Osipov, A.N.; Mironov, A.F. Effect of photosensitizers pheophorbide a and protoporphyrin IX on skin wound healing upon low-intensity laser irradiation. Biophysics 2010, 55, 296–300. [Google Scholar] [CrossRef]
- Hamblin, M.R.; O’Donnell, D.A.; Murthy, N.; Contag, C.H.; Hasan, T. Rapid control of wound infections by targeted photodynamic therapy monitored by in vivo bioluminescence imaging. Photochem. Photobiol. 2002, 75, 51–57. [Google Scholar] [CrossRef]
- Hamblin, M.R.; Zahra, T.; Contag, C.H.; McManus, A.T.; Hasan, T. Optical monitoring and treatment of potentially lethal wound infections in vivo. J. Infect. Dis. 2003, 187, 1717–1725. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Tegos, G.P.; Lu, Z.; Huang, L.; Zhiyentayev, T.; Franklin, M.J.; Baer, D.G.; Hamblin, M.R. Photodynamic therapy for Acinetobacter baumannii burn infections in mice. Antimicrob. Agents Chemother. 2009, 53, 3929–3934. [Google Scholar] [CrossRef]
- Dai, T.; Tegos, G.P.; Zhiyentayev, T.; Mylonakis, E.; Hamblin, M.R. Photodynamic therapy for methicillin-resistant Staphylococcus aureus infection in a mouse skin abrasion model. Lasers Surg. Med. 2010, 42, 38–44. [Google Scholar] [CrossRef]
- Sharma, S.K.; Dai, T.; Kharkwal, G.B.; Huang, Y.-Y.; Huang, L.; Bil De Arce, V.J.; Tegos, G.P.; Hamblin, M.R. Drug discovery of antimicrobial photosensitizers using animal models. Curr. Pharm. Des. 2012, 17, 1303–1319. [Google Scholar] [CrossRef]
- Hamblin, M.R. Antimicrobial photodynamic inactivation: A bright new technique to kill resistant microbes. Curr. Opin. Microbiol. 2016, 33, 67–73. [Google Scholar] [CrossRef]
- Mofazzal Jahromi, M.A.; Sahandi Zangabad, P.; Moosavi Basri, S.M.; Sahandi Zangabad, K.; Ghamarypour, A.; Aref, A.R.; Karimi, M.; Hamblin, M.R. Nanomedicine and advanced technologies for burns: Preventing infection and facilitating wound healing. Adv. Drug Deliv. Rev. 2018, 123, 33–64. [Google Scholar] [CrossRef]
- Hu, X.; Huang, Y.Y.; Wang, Y.; Wang, X.; Hamblin, M.R. Antimicrobial photodynamic therapy to control clinically relevant biofilm infections. Front. Microbiol. 2018, 9, 1299. [Google Scholar] [CrossRef] [PubMed]
- Demidova, T.N.; Gad, F.; Zahra, T.; Francis, K.P.; Hamblin, M.R. Monitoring photodynamic therapy of localized infections by bioluminescence imaging of genetically engineered bacteria. J. Photochem. Photobiol. B Biol. 2005, 81, 15–25. [Google Scholar] [CrossRef]
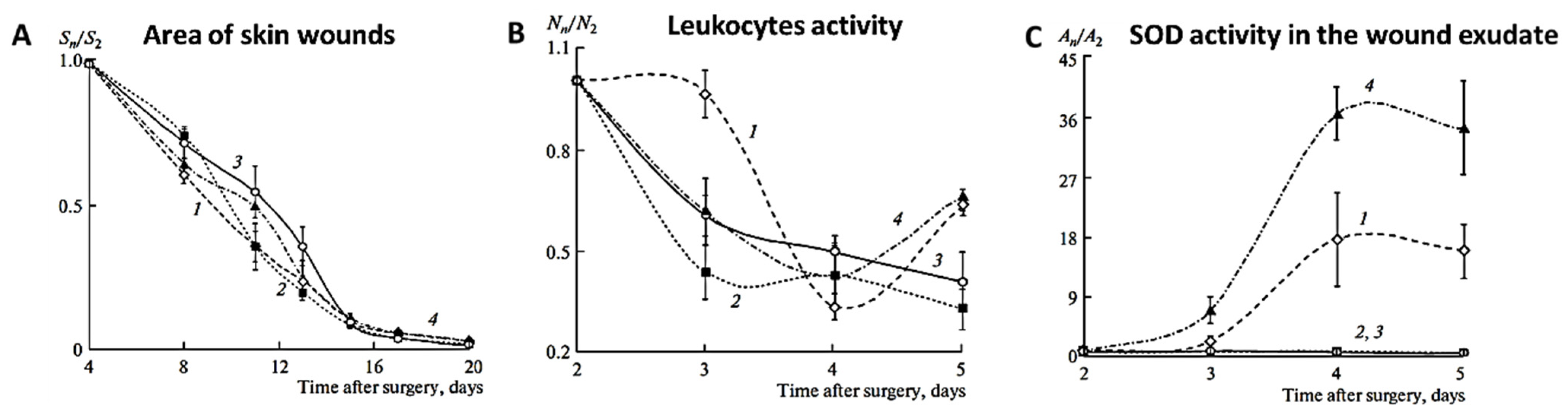
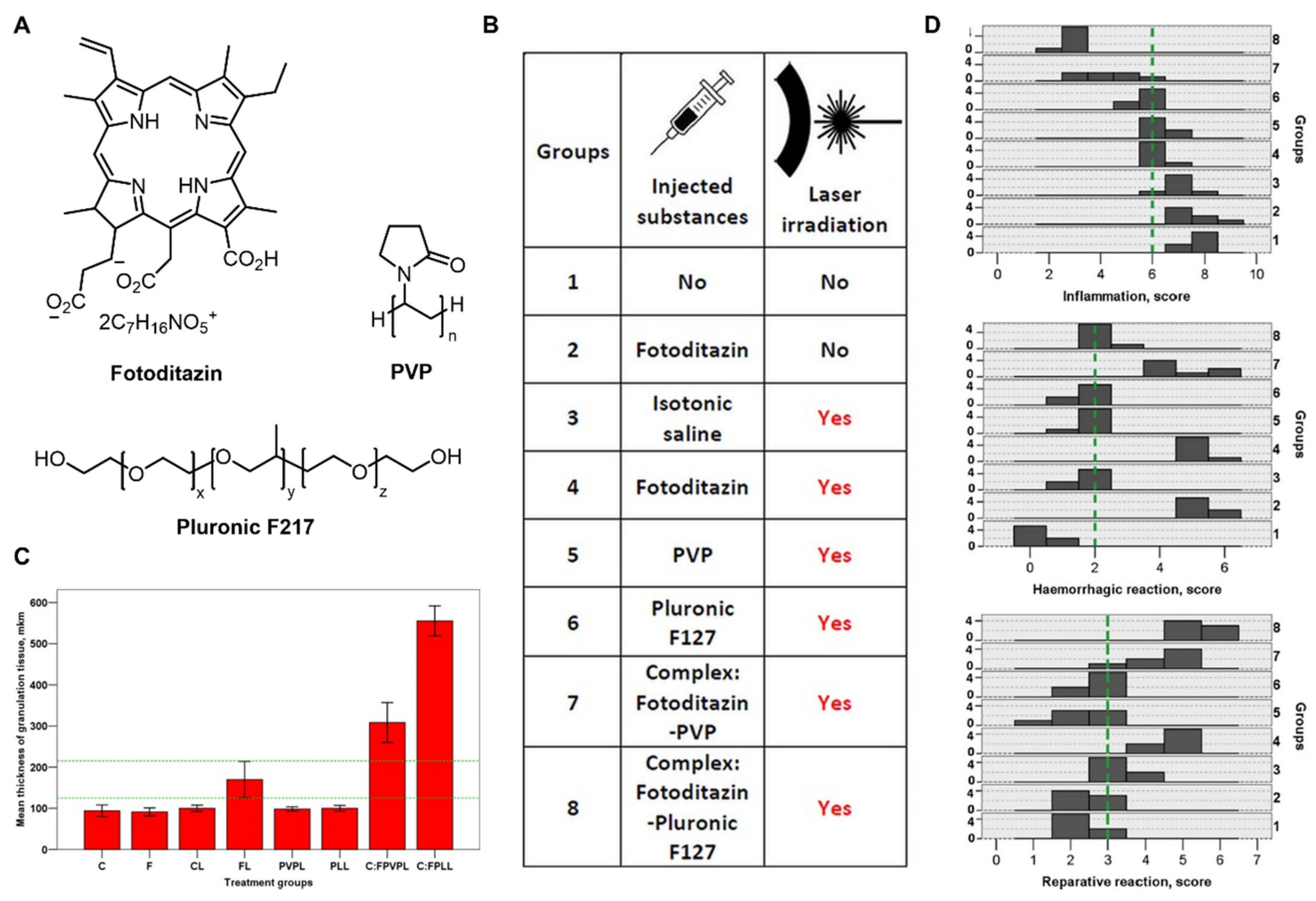
- Rudenko, T.G.; Shekhter, A.B.; Guller, A.E.; Aksenova, N.A.; Glagolev, N.N.; Ivanov, A.V.; Aboyants, R.K.; Kotova, S.L.; Solovieva, A.B. Specific features of early stage of the wound healing process occurring against the background of photodynamic therapy using fotoditazin photosensitizer-amphiphilic polymer complexes. Photochem. Photobiol. 2014, 90, 1413–1422. [Google Scholar] [CrossRef]
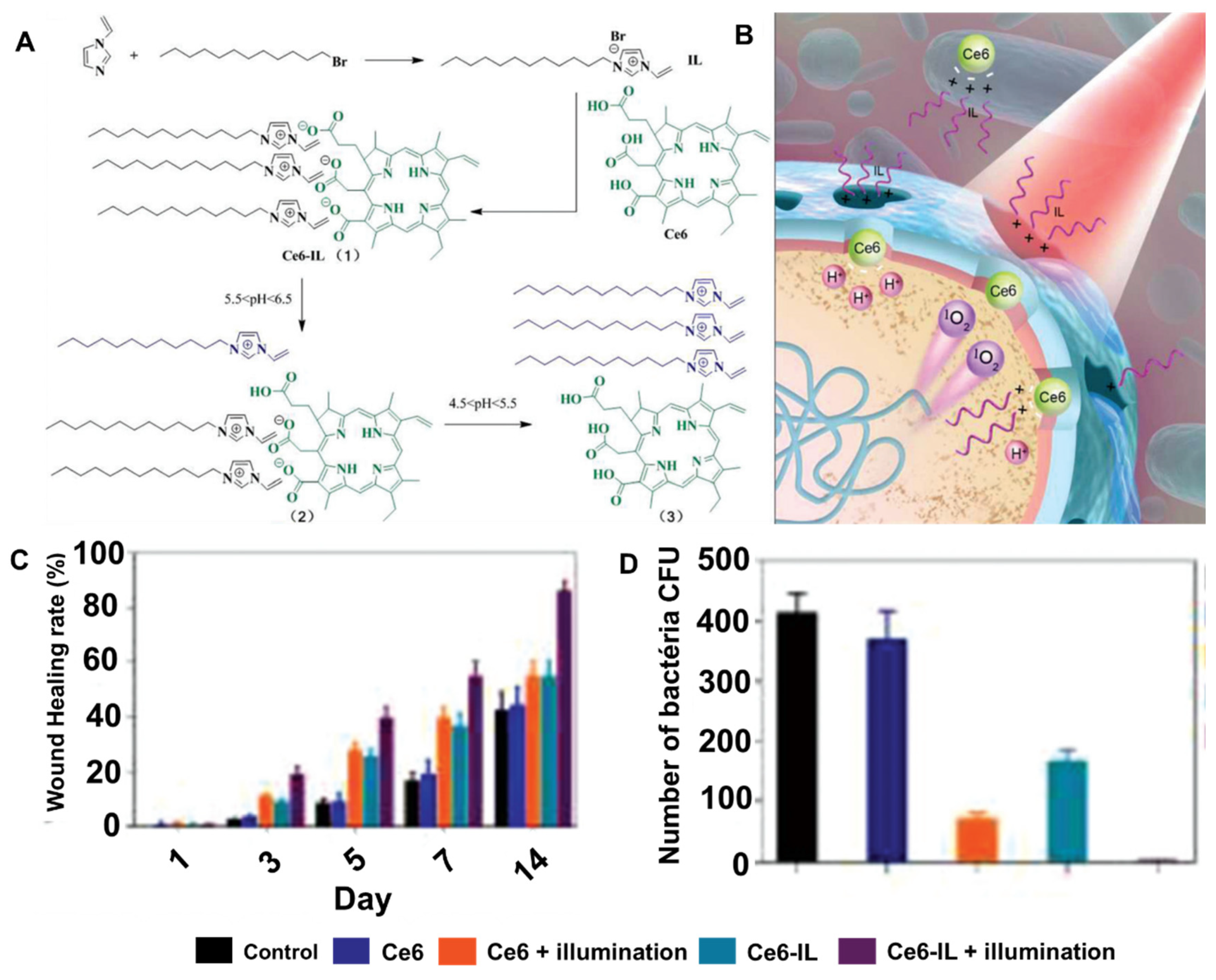
- Wang, C.; Chen, P.; Wang, C.; Chen, P.; Qiao, Y.; Kang, Y.; Guo, S.; Wu, D.; Wangd, J.; Wu, H. Bacteria-activated chlorin e6 ionic liquid based on cation and anion dual-mode antibacterial action for enhanced photodynamic efficacy. Biomater. Sci. 2019, 7, 1399–1410. [Google Scholar] [CrossRef]
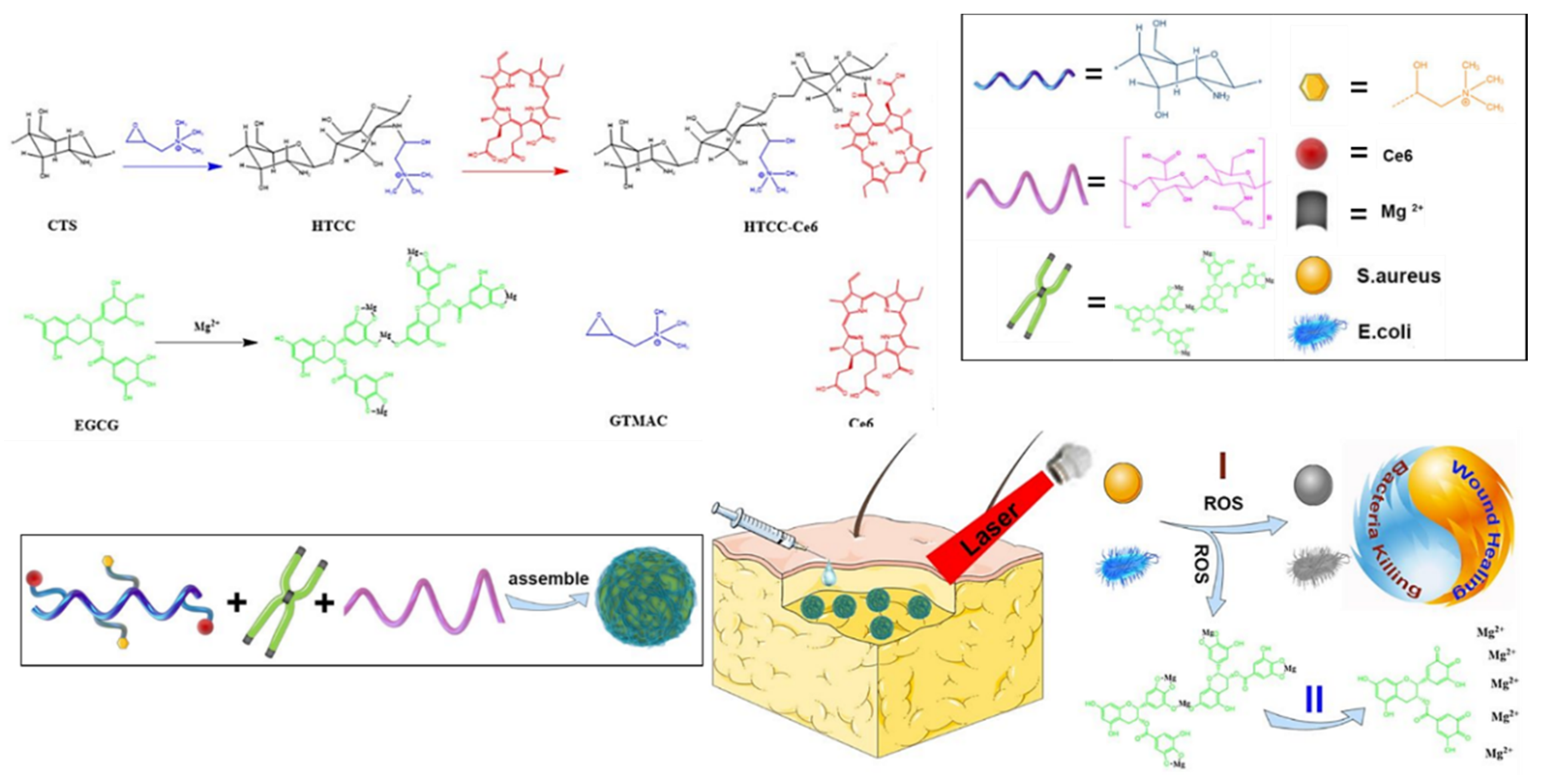
- Hu, C.; Zhang, F.; Kong, Q.; Lu, Y.; Zhang, B.; Wu, C.; Luo, R.; Wang, Y. Synergistic chemical and photodynamic antimicrobial therapy for enhanced wound healing mediated by multifunctional light-responsive nanoparticles. Biomacromolecules 2019, 20, 4581–4592. [Google Scholar] [CrossRef]
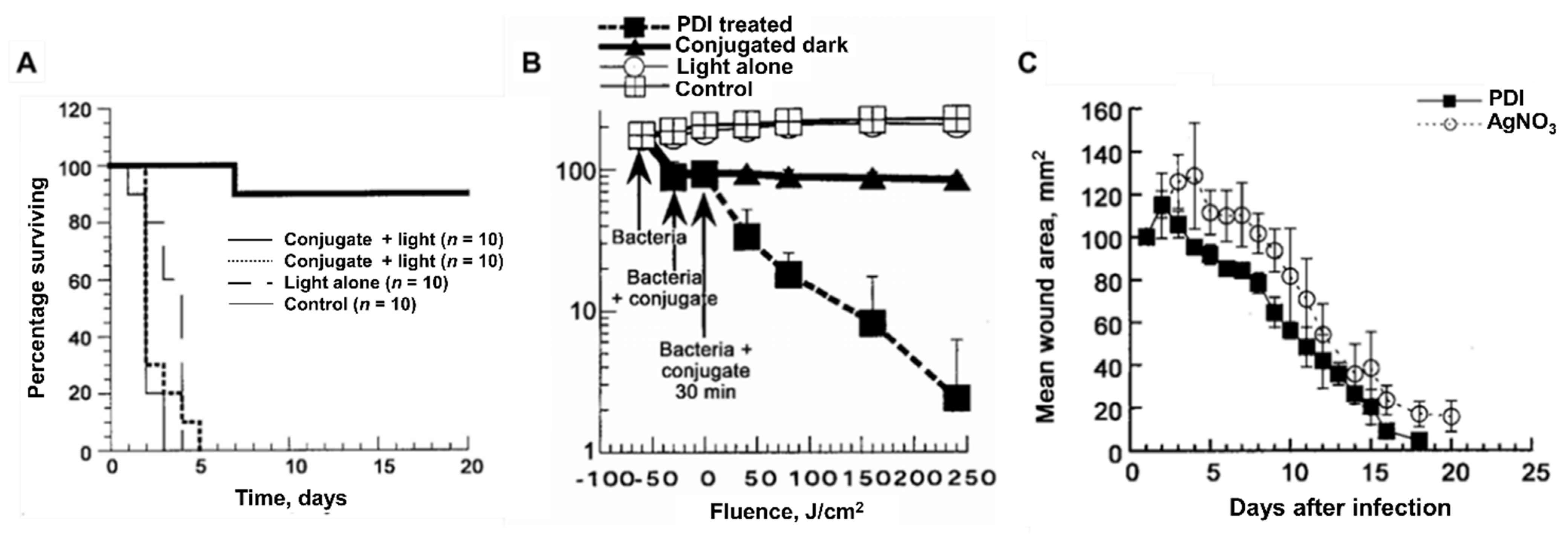
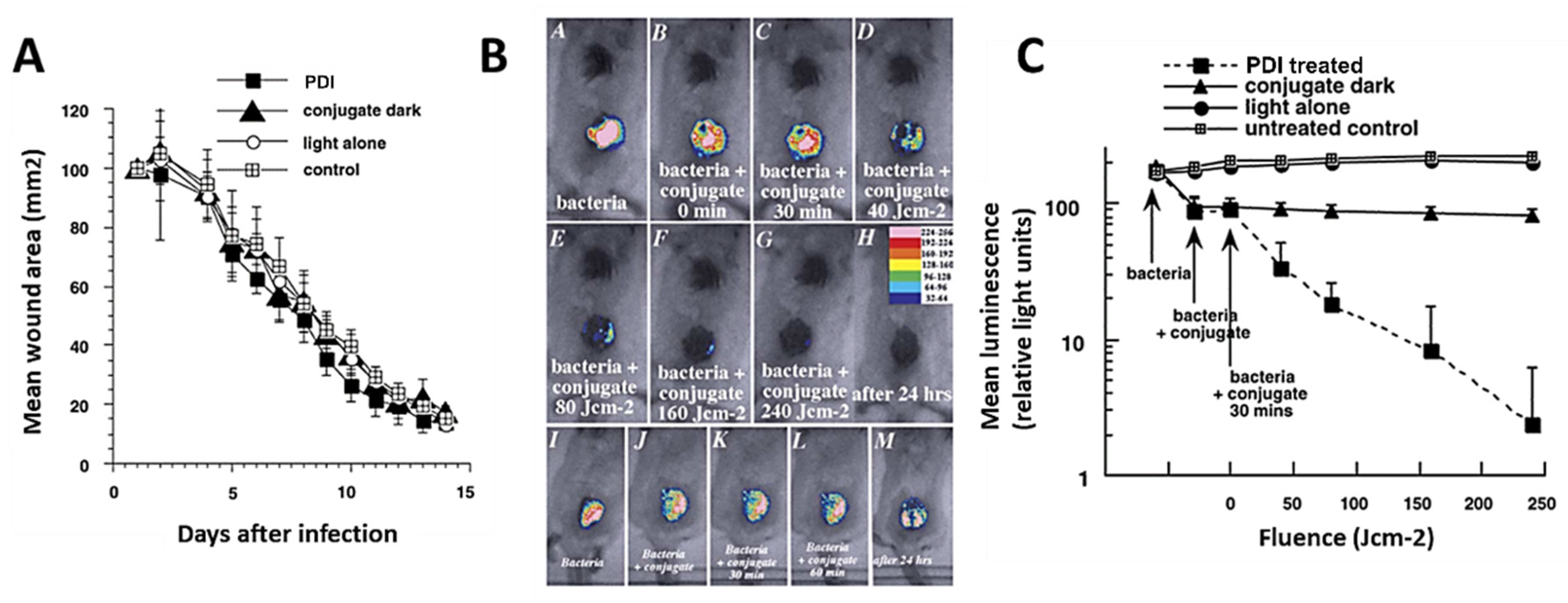
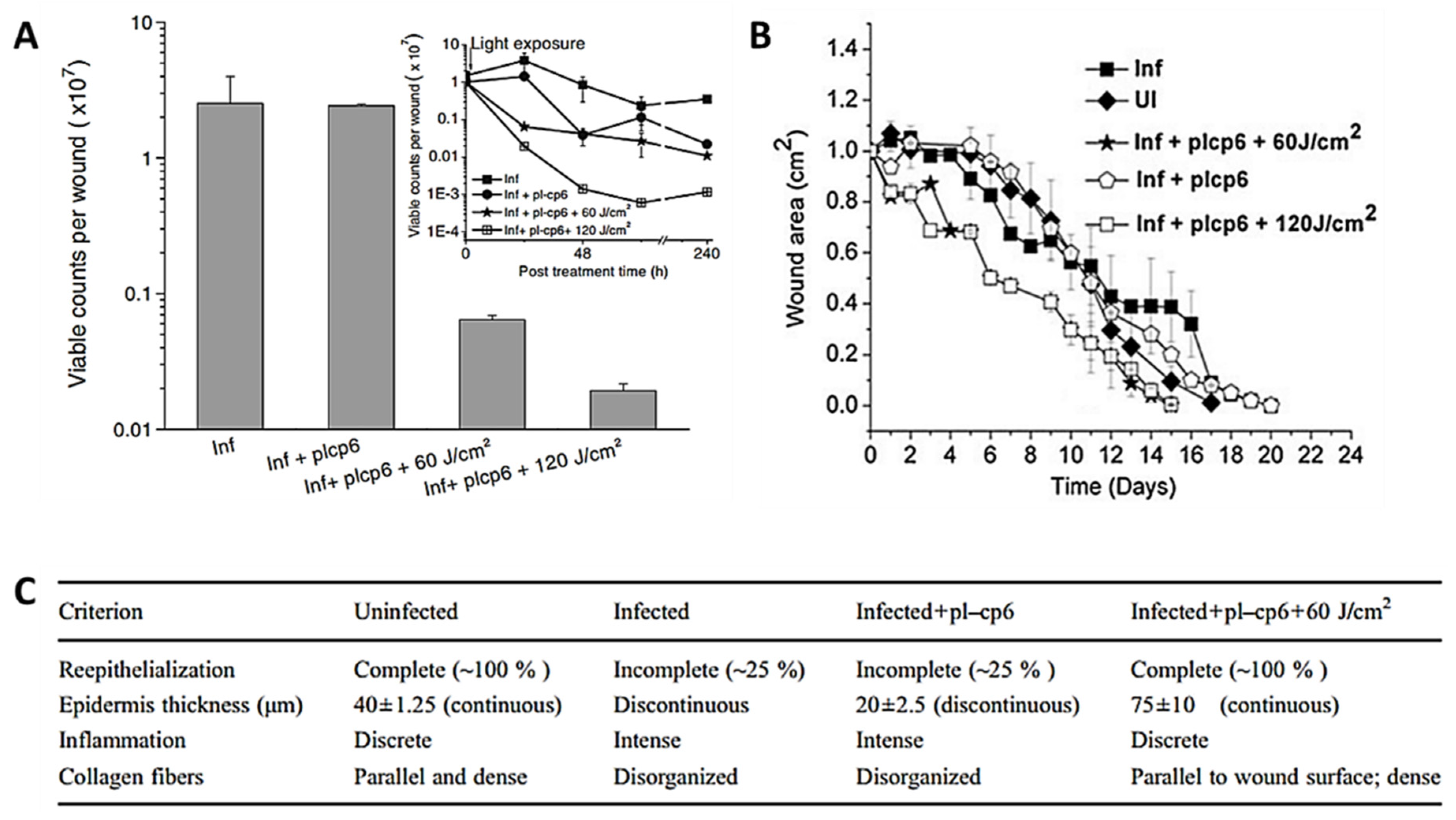
- Sahu, K.; Sharma, M.; Bansal, H.; Dube, A.; Gupta, P.K. Topical photodynamic treatment with poly-l-lysine-chlorin p6 conjugate improves wound healing by reducing hyperinflammatory response in Pseudomonas aeruginosa-infected wounds of mice. Lasers Med. Sci. 2013, 28, 465–471. [Google Scholar] [CrossRef]
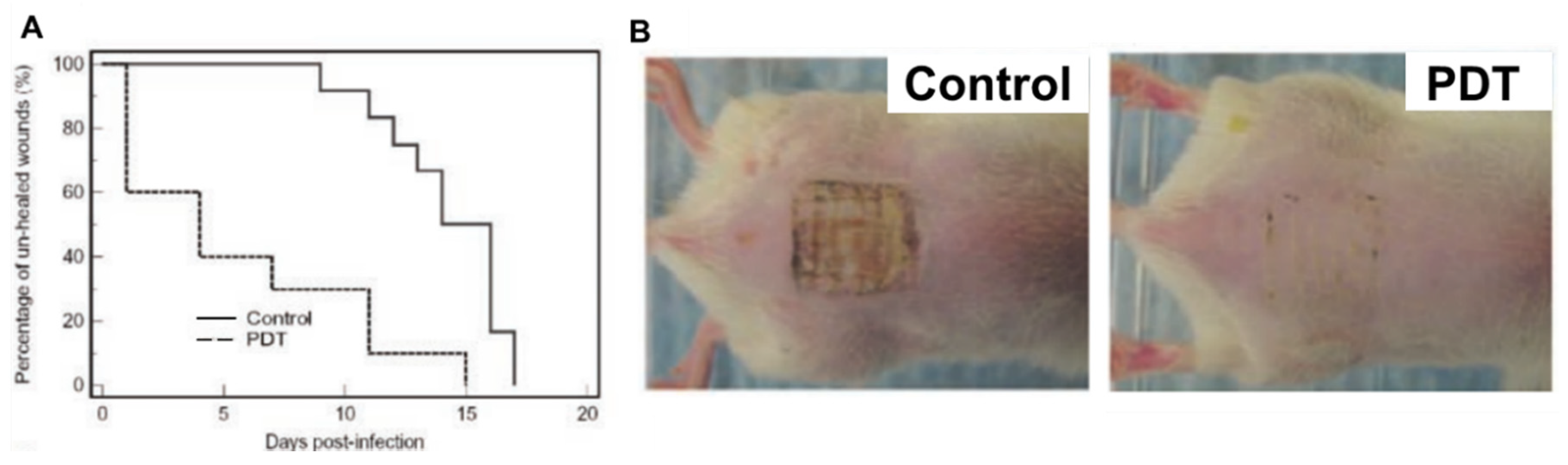
- Sahu, K.; Sharma, M.; Sharma, P.; Verma, Y.; Rao, K.D.; Bansal, H.; Dube, A.; Gupta, P.K. Effect of poly-L-lysine-chlorin P6-mediated antimicrobial photodynamic treatment on collagen restoration in bacteria-infected wounds. Photomed. Laser Surg. 2014, 32, 23–29. [Google Scholar] [CrossRef]
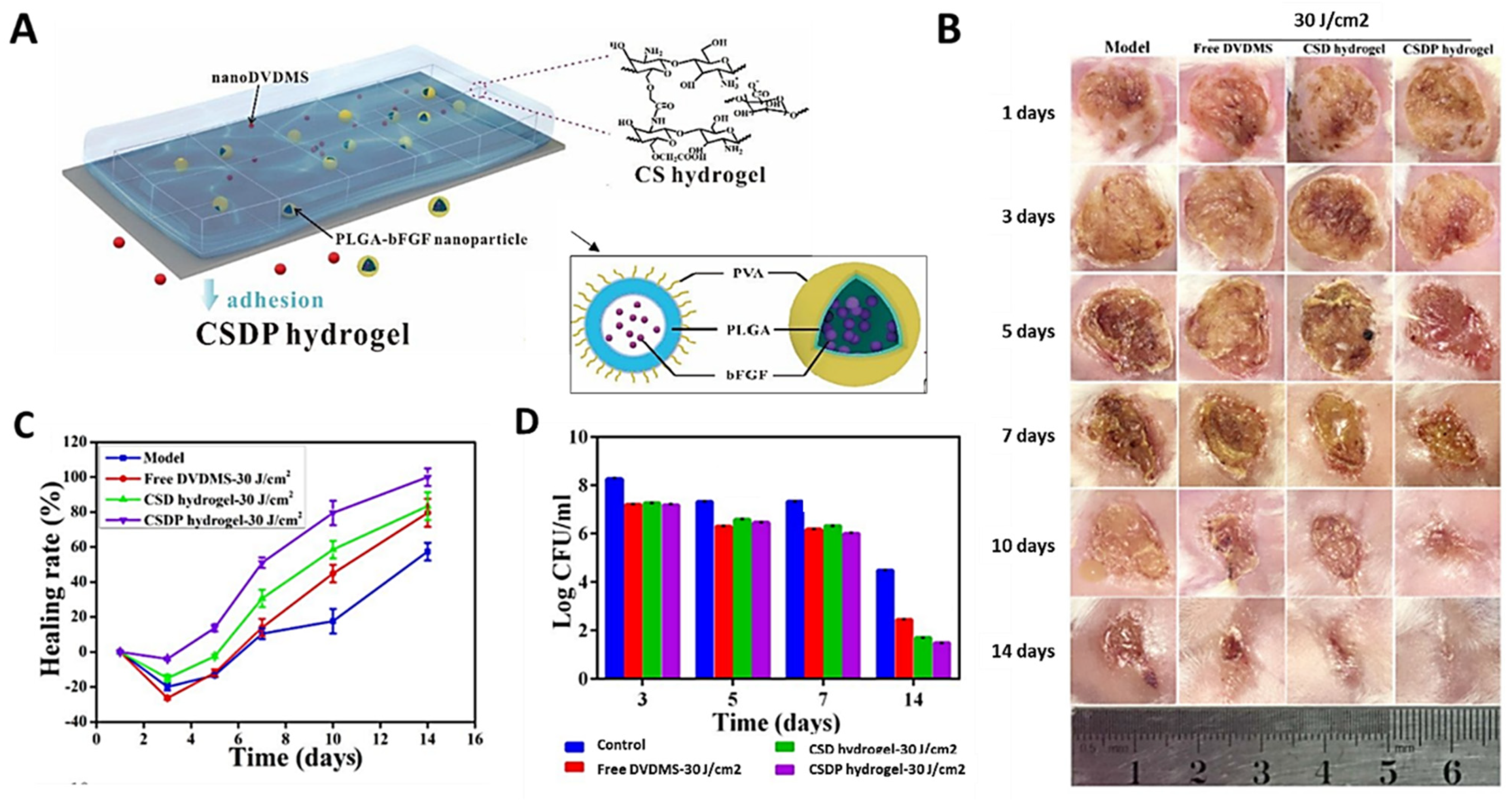
- Mai, B.; Jia, M.; Liu, S.; Sheng, Z.; Li, M.; Gao, Y.; Wang, X.; Liu, Q.; Wang, P. Smart hydrogel-Based DVDMS/bFGF nanohybrids for antibacterial phototherapy with multiple damaging sites and accelerated wound healing. ACS Appl. Mater. Interfaces 2020, 12, 10156–10169. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, S.A.G.; Demidova, T.N.; Aalders, M.C.G.; Hasan, T.; Hamblin, M.R. Photodynamic therapy for Staphylococcus aureus infected burn wounds in mice. Photochem. Photobiol. Sci. 2005, 4, 503–509. [Google Scholar] [CrossRef] [PubMed]
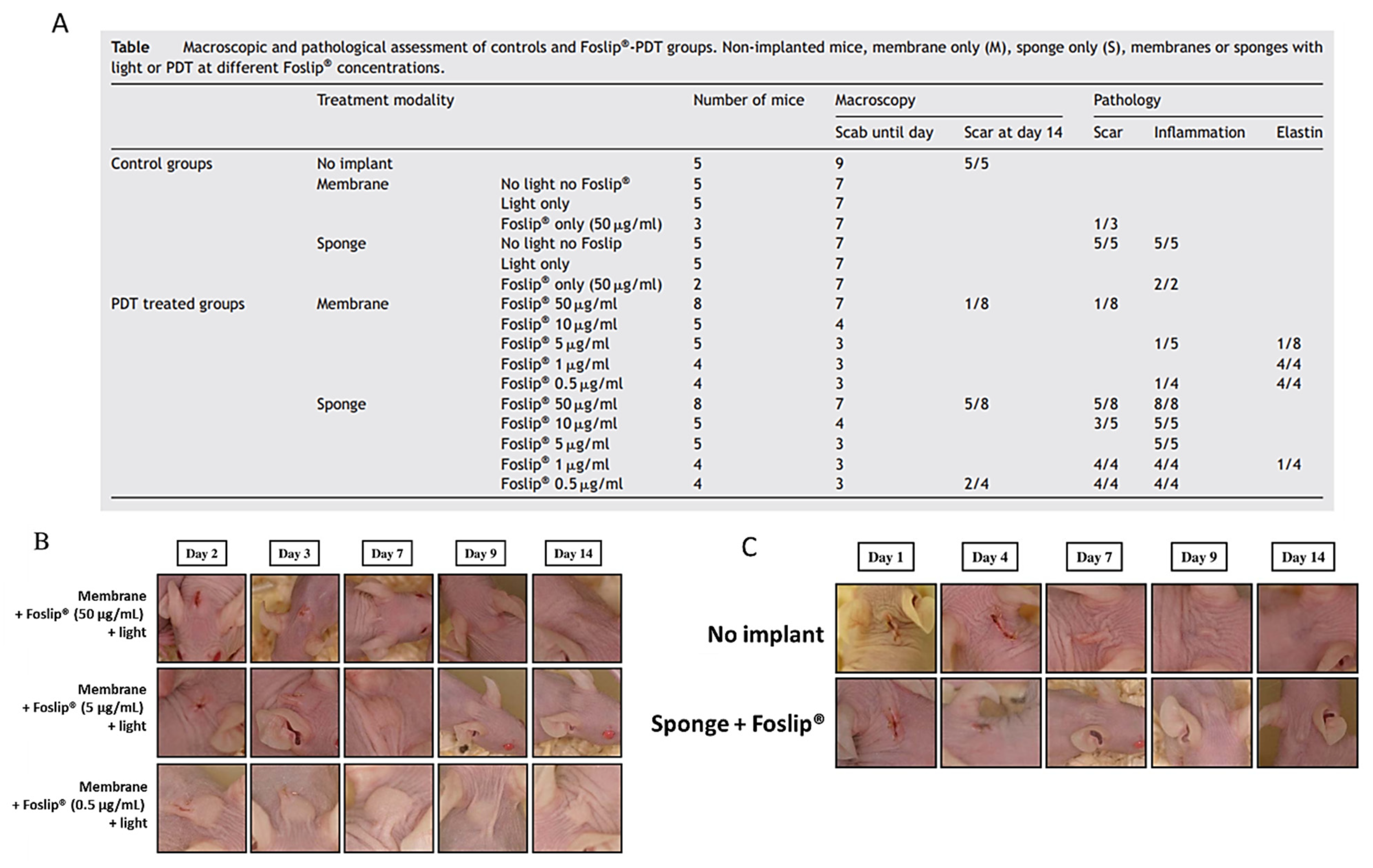
- Garrier, J.; Bezdetnaya, L.; Barlier, C.; Gräfe, S.; Guillemin, F.; D’Hallewin, M.A. Foslip ®-based photodynamic therapy as a means to improve wound healing. Photodiagnosis Photodyn. Ther. 2011, 8, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Arenbergerova, M.; Arenberger, P.; Bednar, M.; Kubat, P.; Mosinger, J. Light-activated nanofibre textiles exert antibacterial effects in the setting of chronic wound healing. Exp. Dermatol. 2012, 21, 619–624. [Google Scholar] [CrossRef]
- Xu, Z.; Gao, Y.; Meng, S.; Yang, B.; Pang, L.; Wang, C.; Liu, T. Mechanism and in vivo evaluation: Photodynamic antibacterial chemotherapy of lysine-porphyrin conjugate. Front. Microbiol. 2016, 7, 242. [Google Scholar] [CrossRef] [PubMed]
- Fila, G.; Kasimova, K.; Arenas, Y.; Nakonieczna, J.; Grinholc, M.; Bielawski, K.P.; Lilge, L. Murine model imitating chronic wound infections for evaluation of antimicrobial photodynamic therapy efficacy. Front. Microbiol. 2016, 7, 1258. [Google Scholar] [CrossRef]
- Luo, Y.; Li, J.; Liu, X.; Tan, L.; Cui, Z.; Feng, X.; Yang, X.; Liang, Y.; Li, Z.; Zhu, S.; et al. Dual metal-organic framework heterointerface. ACS Cent. Sci. 2019, 5, 1591–1601. [Google Scholar] [CrossRef]
- Han, D.; Han, Y.; Li, J.; Liu, X.; Yeung, K.W.K.; Zheng, Y.; Cui, Z.; Yang, X.; Liang, Y.; Li, Z.; et al. Enhanced photocatalytic activity and photothermal effects of cu-doped metal-organic frameworks for rapid treatment of bacteria-infected wounds. Appl. Catal. B Environ. 2020, 261, 118248. [Google Scholar] [CrossRef]
- Li, J.; Zhai, D.; Lv, F.; Yu, Q.; Ma, H.; Yin, J.; Yi, Z.; Liu, M.; Chang, J.; Wu, C. Preparation of copper-containing bioactive glass/eggshell membrane nanocomposites for improving angiogenesis, antibacterial activity and wound healing. Acta Biomater. 2016, 36, 254–266. [Google Scholar] [CrossRef]
- Han, D.; Li, Y.; Liu, X.; Yeung, K.W.K.; Zheng, Y.; Cui, Z.; Liang, Y.; Li, Z.; Zhu, S.; Wang, X.; et al. Photothermy-strengthened photocatalytic activity of polydopamine-modified metal-organic frameworks for rapid therapy of bacteria-infected wounds. J. Mater. Sci. Technol. 2021, 62, 83–95. [Google Scholar] [CrossRef]
- Sun, L.; Song, L.; Zhang, X.; Zhou, R.; Yin, J.; Luan, S. Poly(γ-glutamic acid)-based electrospun nanofibrous mats with photodynamic therapy for effectively combating wound infection. Mater. Sci. Eng. C 2020, 113, 110936. [Google Scholar] [CrossRef]
- Mack, J.; Kobayashi, N. Low symmetry phthalocyanines and their analogues. Chem. Rev. 2011, 111, 281–321. [Google Scholar] [CrossRef] [PubMed]
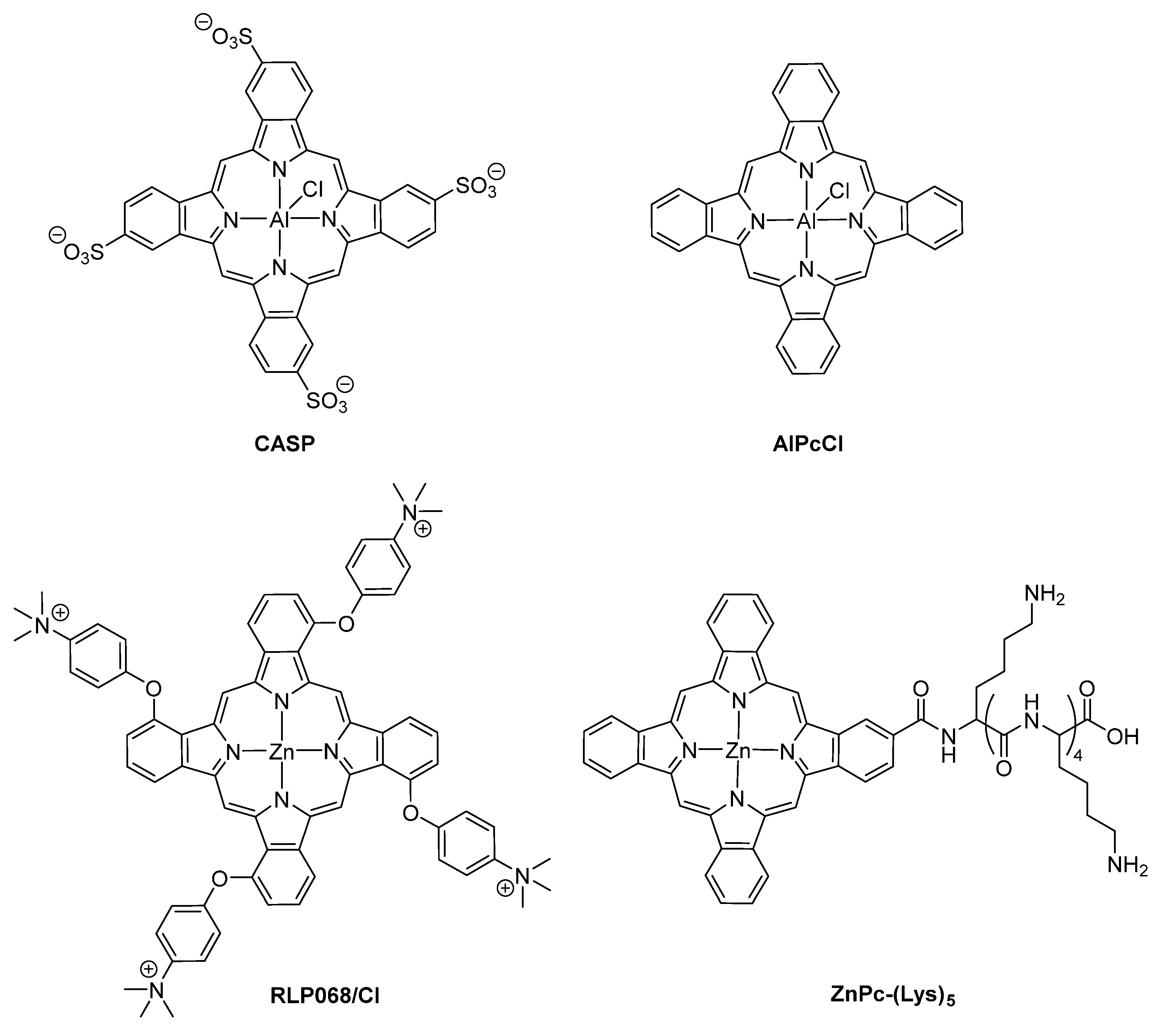
- Stern, S.J.; Flock, S.T.; Small, S.; Thomsen, S.; Jacques, S. Photodynamic therapy with chloroaluminum sulfonated phthalocyanine in the rat window chamber. Am. J. Surg. 1990, 160, 360–364. [Google Scholar] [CrossRef]
- Parekh, S.G.; Trauner, K.B.; Zarins, B.; Foster, T.E.; Anderson, R.R. Photodynamic modulation of wound healing with BPD-MA and CASP. Lasers Surg. Med. 1999, 24, 375–381. [Google Scholar] [CrossRef]
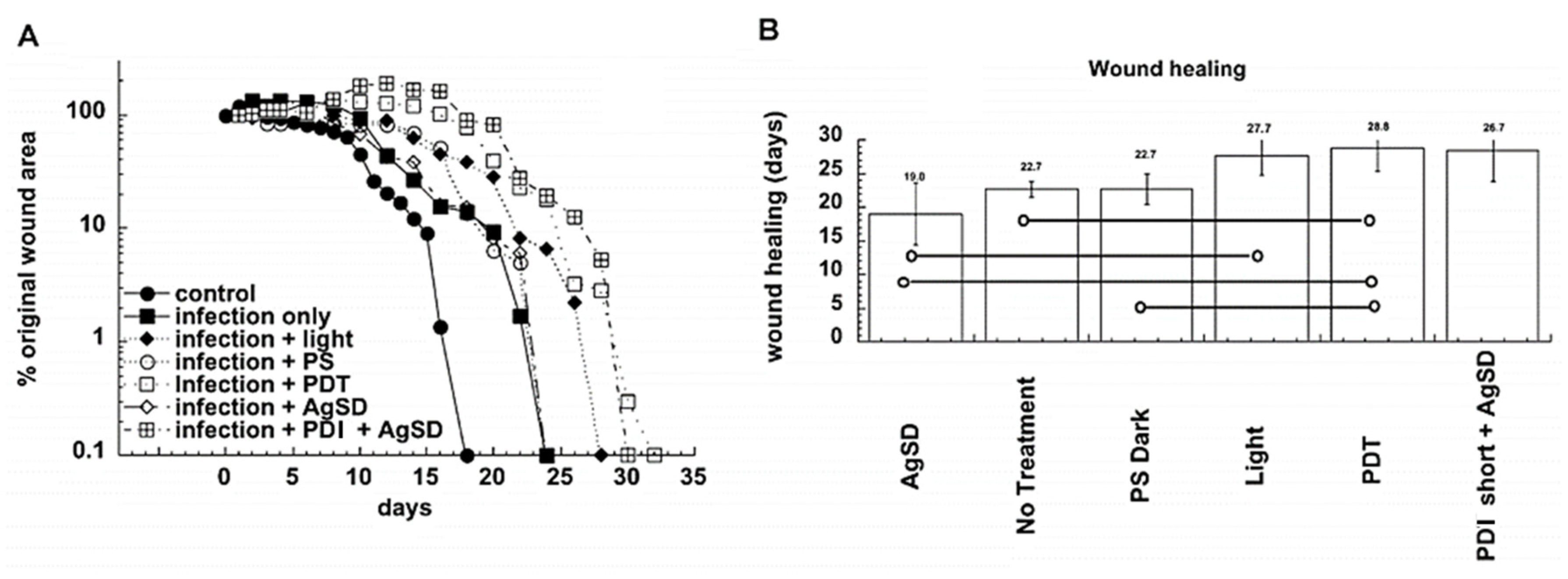
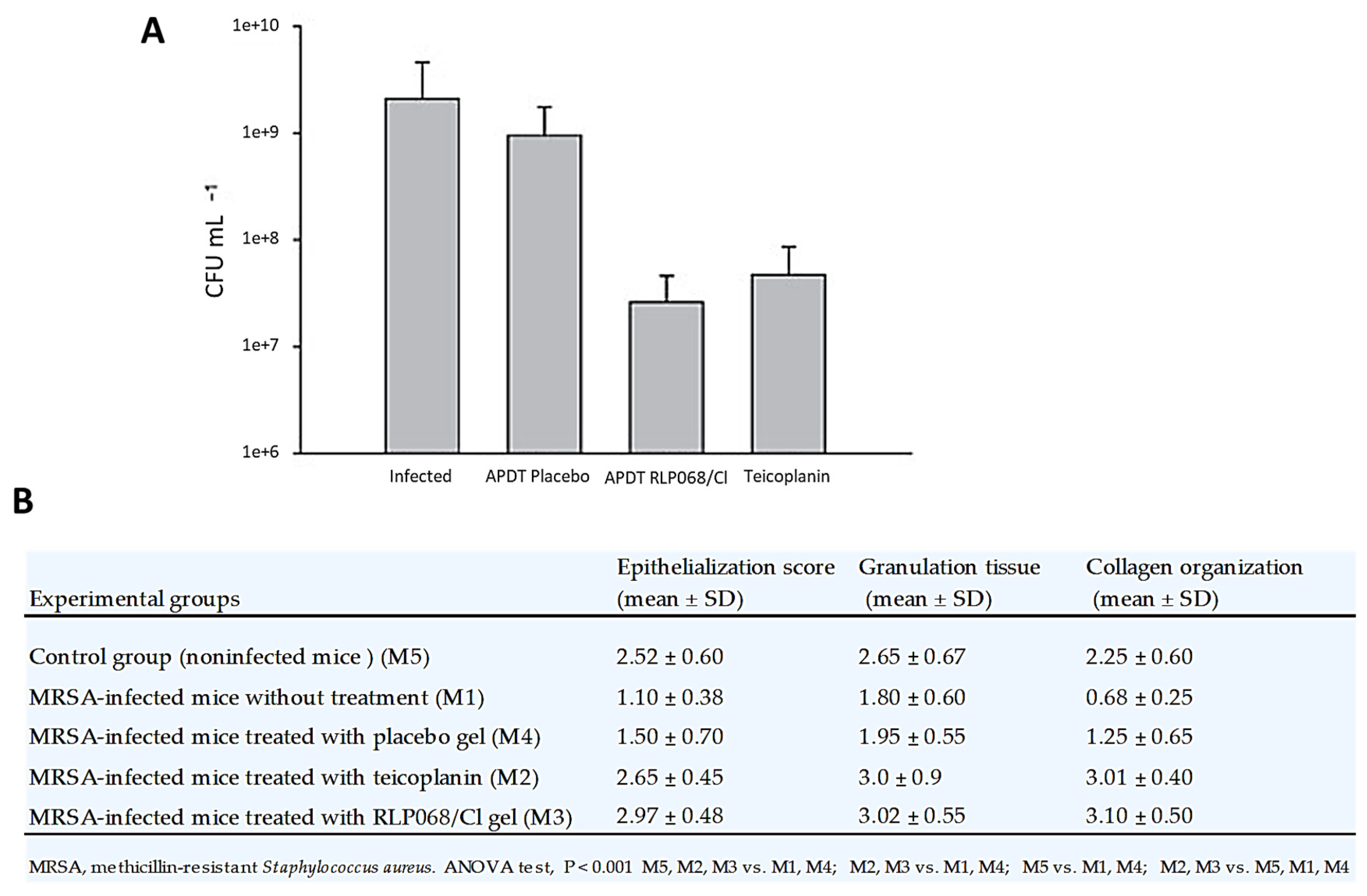
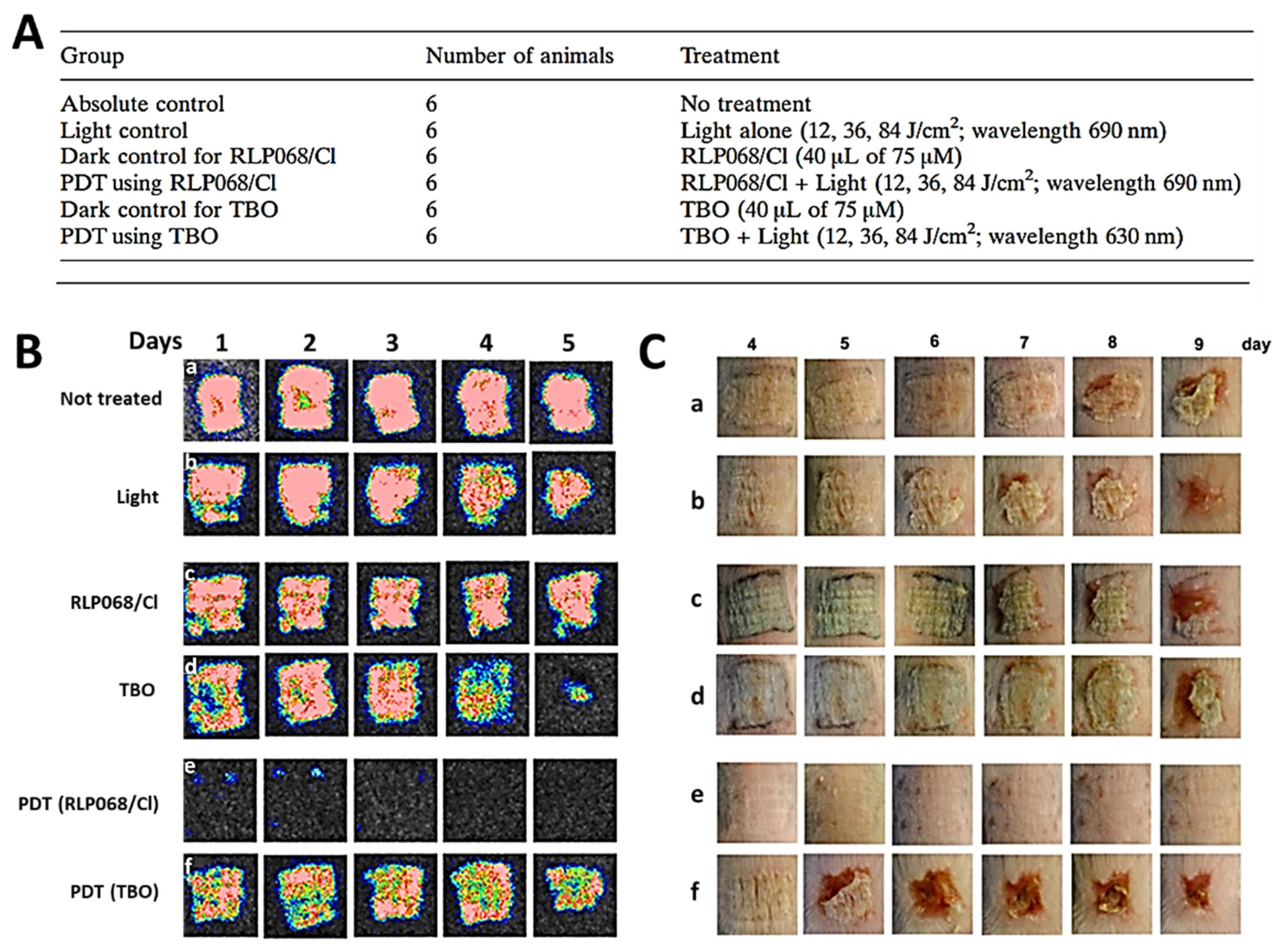
- Simonetti, O.; Cirioni, O.; Orlando, F.; Alongi, C.; Lucarini, G.; Silvestri, C.; Zizzi, A.; Fantetti, L.; Roncucci, G.; Giacometti, A.; et al. Effectiveness of antimicrobial photodynamic therapy with a single treatment of RLP068/Cl in an experimental model of Staphylococcus aureus wound infection. Br. J. Dermatol. 2011, 164, 987–995. [Google Scholar] [CrossRef]
- Vecchio, D.; Dai, T.; Huang, L.; Fantetti, L.; Roncucci, G.; Hamblin, M.R. Antimicrobial photodynamic therapy with RLP068 kills methicillin-resistant Staphylococcus aureus and improves wound healing in a mouse model of infected skin abrasion PDT with RLP068/Cl in infected mouse skin abrasion. J. Biophoton. 2013, 6, 733–742. [Google Scholar] [CrossRef]
- Mosti, G.; Picerni, P.; Licau, M.; Mattaliano, V. Photodynamic therapy in infected venous and mixed leg ulcers: A pilot experience. J. Wound Care 2018, 27, 816–821. [Google Scholar] [CrossRef]
- Mannucci, E.; Genovese, S.; Monami, M.; Navalesi, G.; Dotta, F.; Anichini, R.; Romagnoli, F.; Gensini, G. Photodynamic topical antimicrobial therapy for infected foot ulcers in patients with diabetes: A randomized, double-blind, placebo-controlled study—The D.A.N.T.E (Diabetic ulcer Antimicrobial New Topical treatment Evaluation) study. Acta Diabetol. 2014, 51, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, Y.; Yan, S.; Chen, Z.; Deng, Y.; Xu, P.; Chen, J.; Liu, W.; Hu, P.; Huang, M.; et al. An effective zinc phthalocyanine derivative against multidrug-resistant bacterial infection. J. Porphyr. Phthalocyan. 2017, 21, 205–210. [Google Scholar] [CrossRef]
- Chen, Z.; Zhou, S.; Chen, J.; Deng, Y.; Luo, Z.; Chen, H.; Hamblin, M.R.; Huang, M. Pentalysine β-carbonylphthalocyanine zinc: An effective tumor-targeting photosensitizer for photodynamic therapy. ChemMedChem 2010, 5, 890–898. [Google Scholar] [CrossRef]
- Chen, J.; Yang, L.; Chen, J.; Liu, W.; Zhang, D.; Xu, P.; Dai, T.; Shang, L.; Yang, Y.; Tang, S.; et al. Composite of silver nanoparticles and photosensitizer leads to mutual enhancement of antimicrobial efficacy and promotes wound healing. Chem. Eng. J. 2019, 374, 1373–1381. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Gould, L. Opportunities and challenges of the management of chronic wounds: A multidisciplinary viewpoint. Chron. Wound Care Manag. Res. 2020, 7, 27–36. [Google Scholar] [CrossRef]
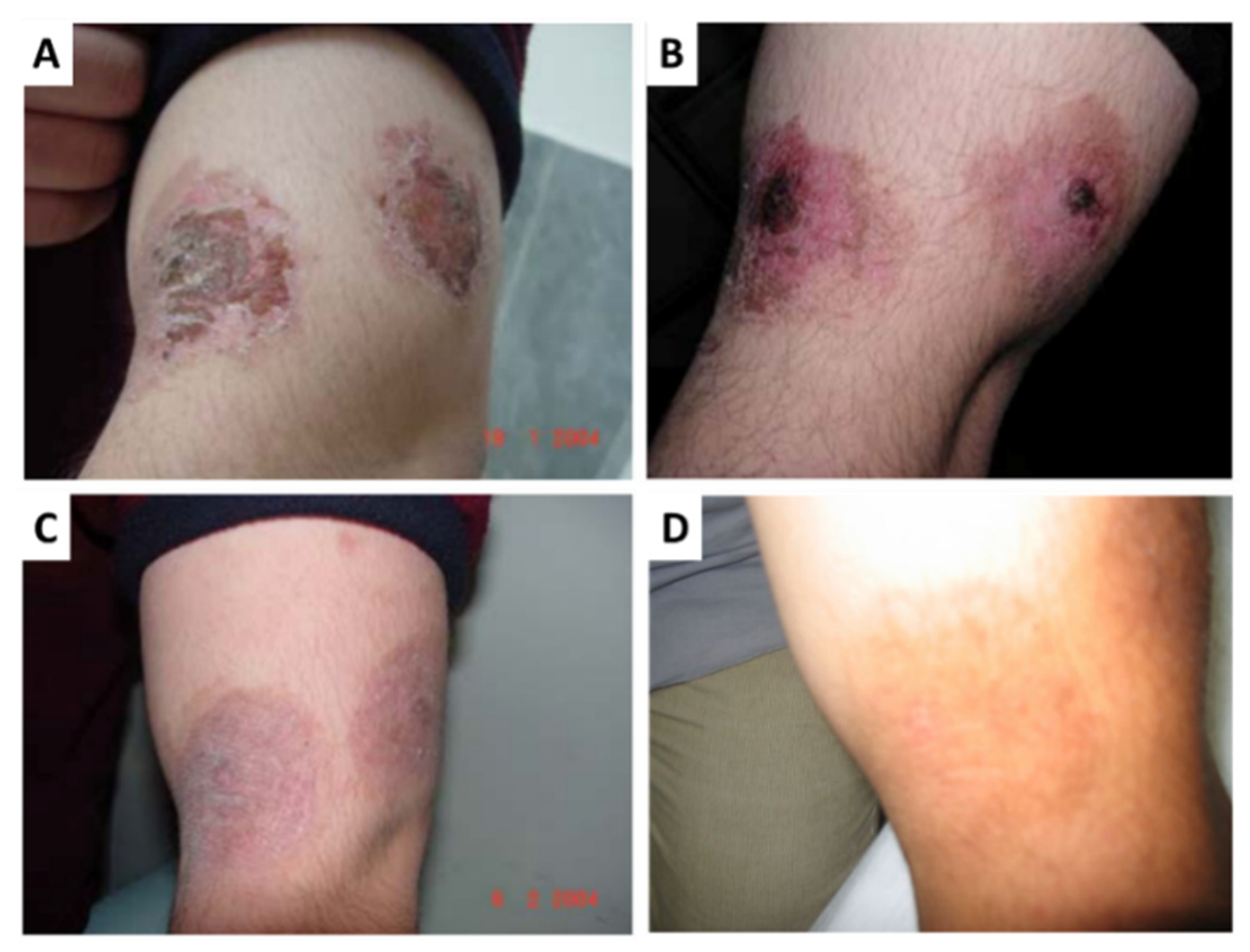
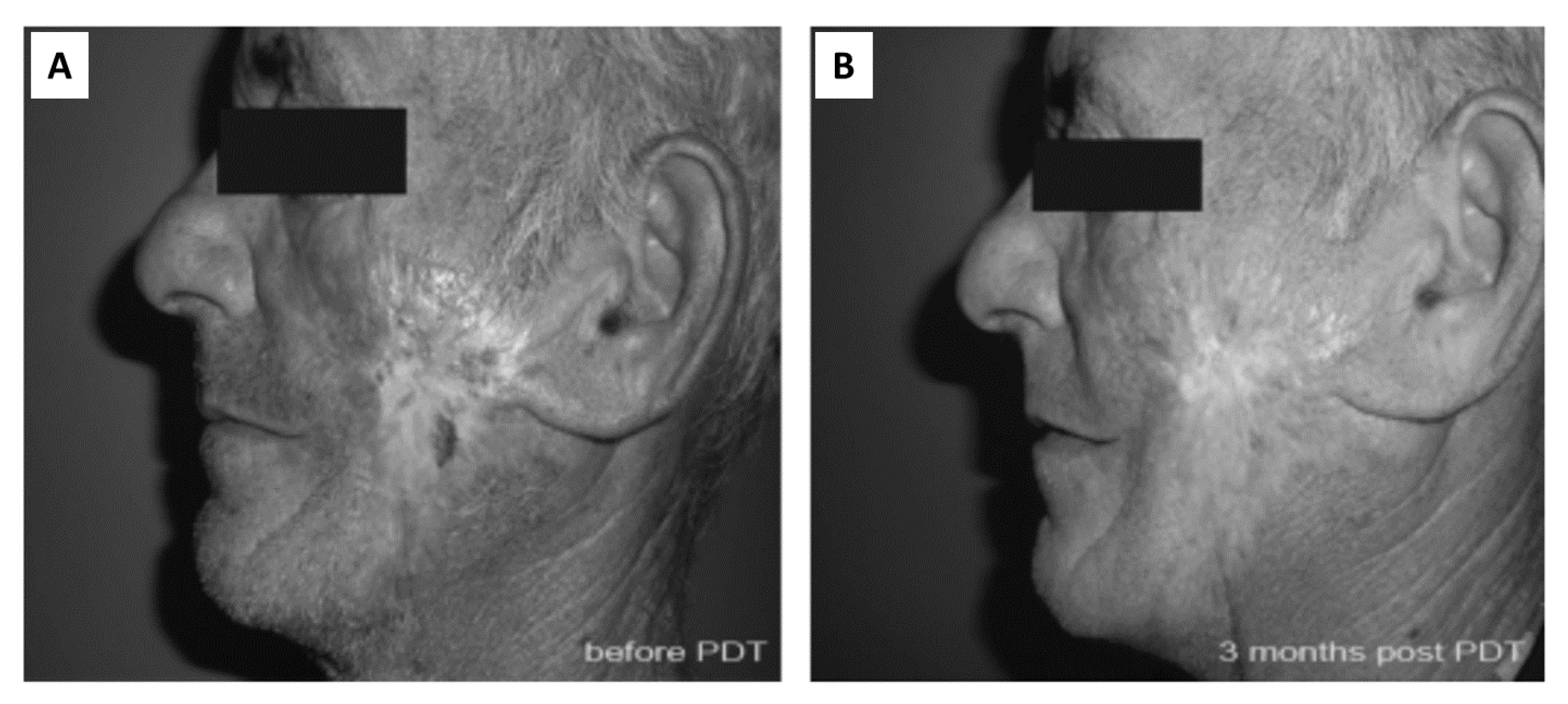
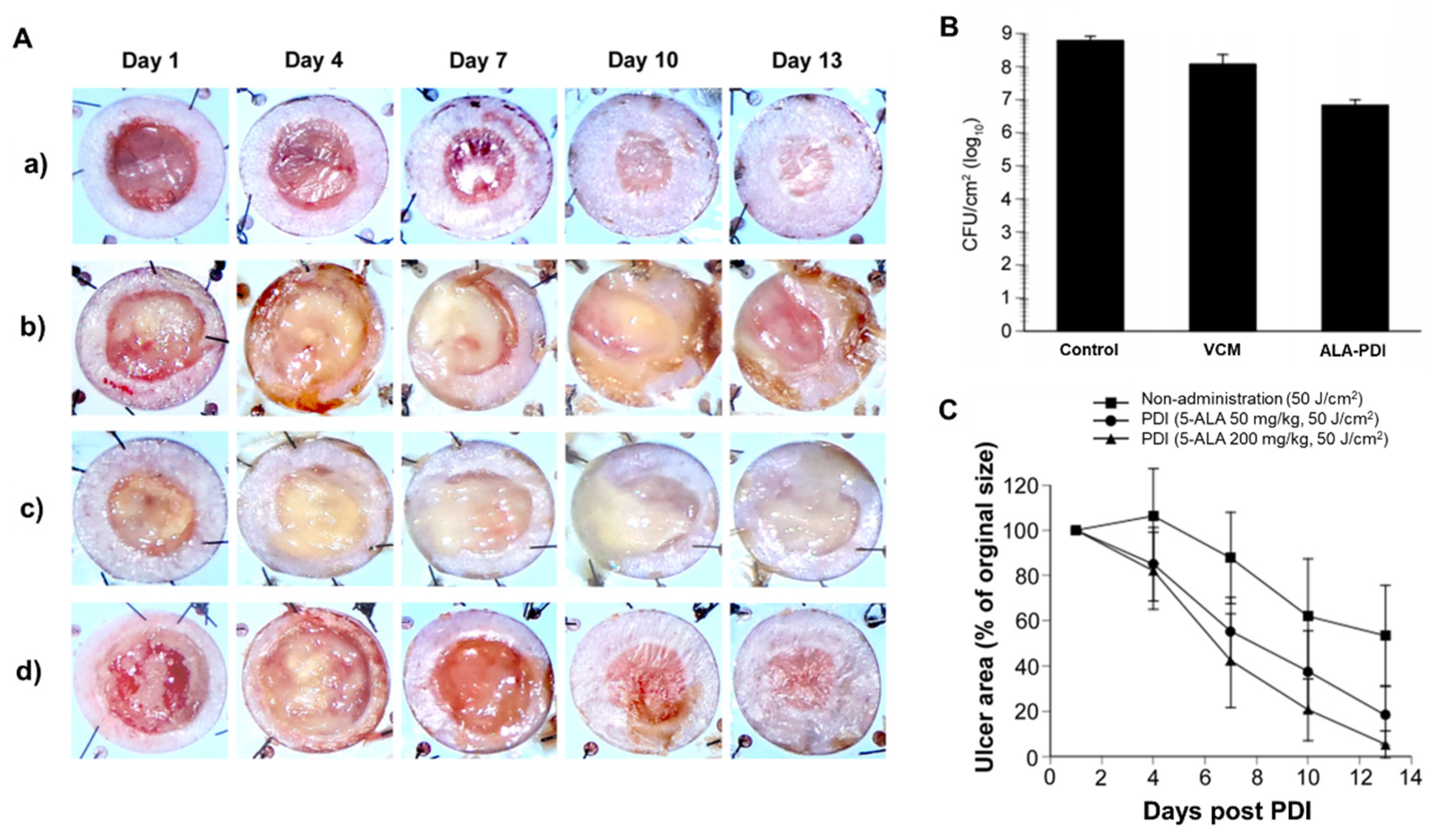
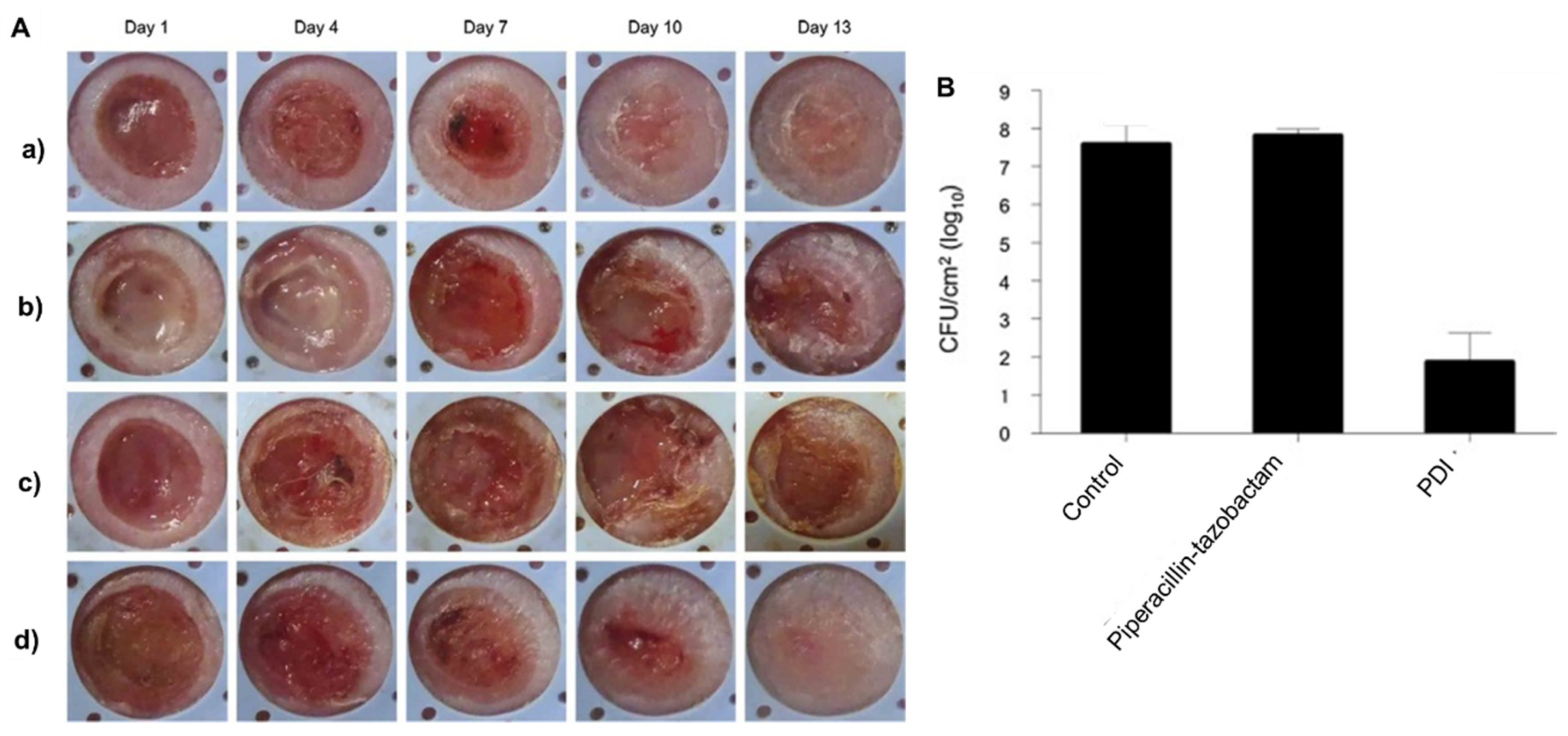
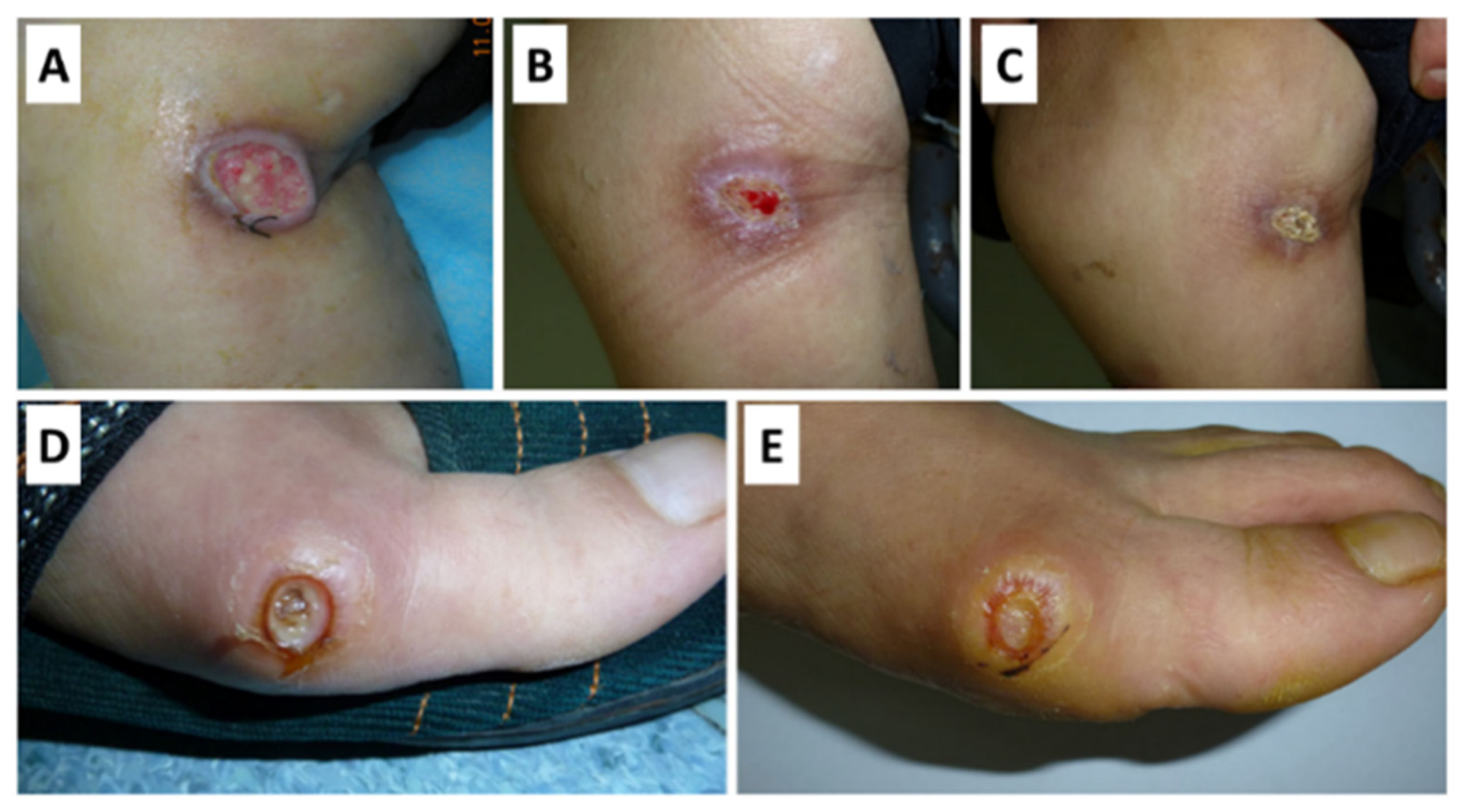
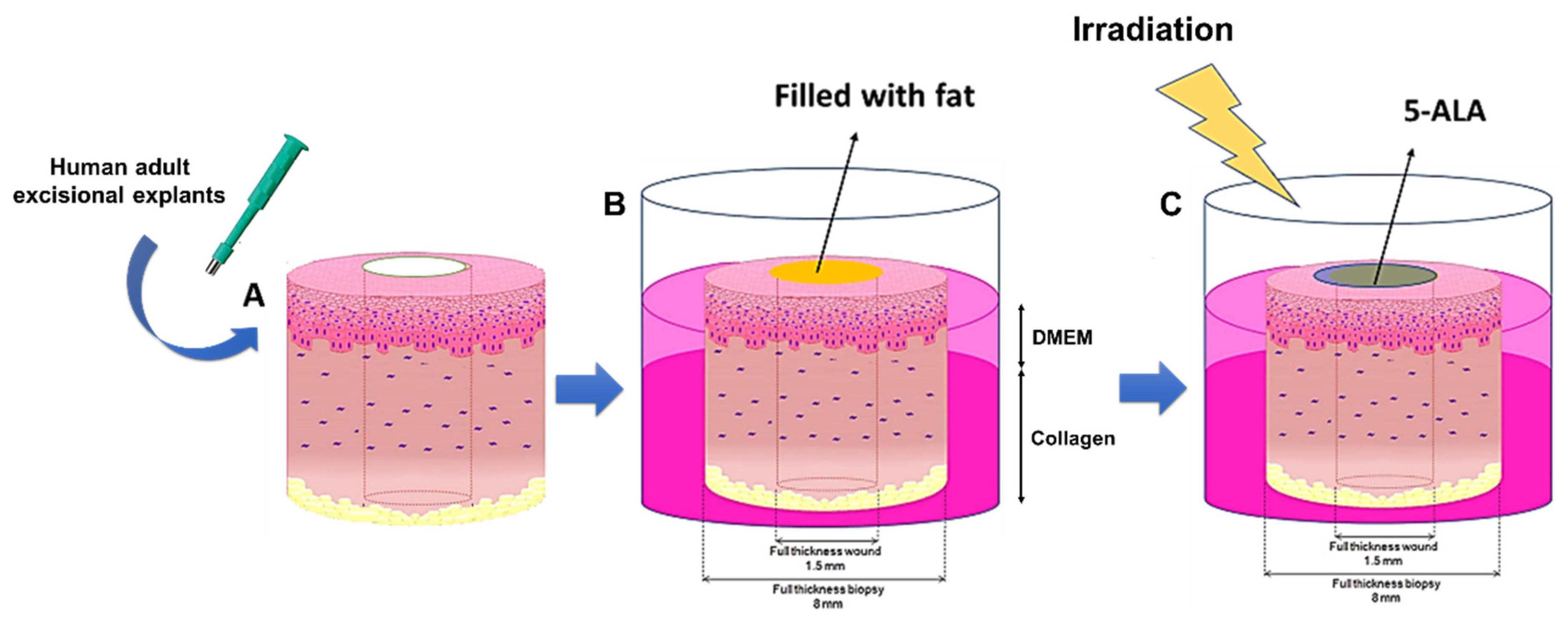
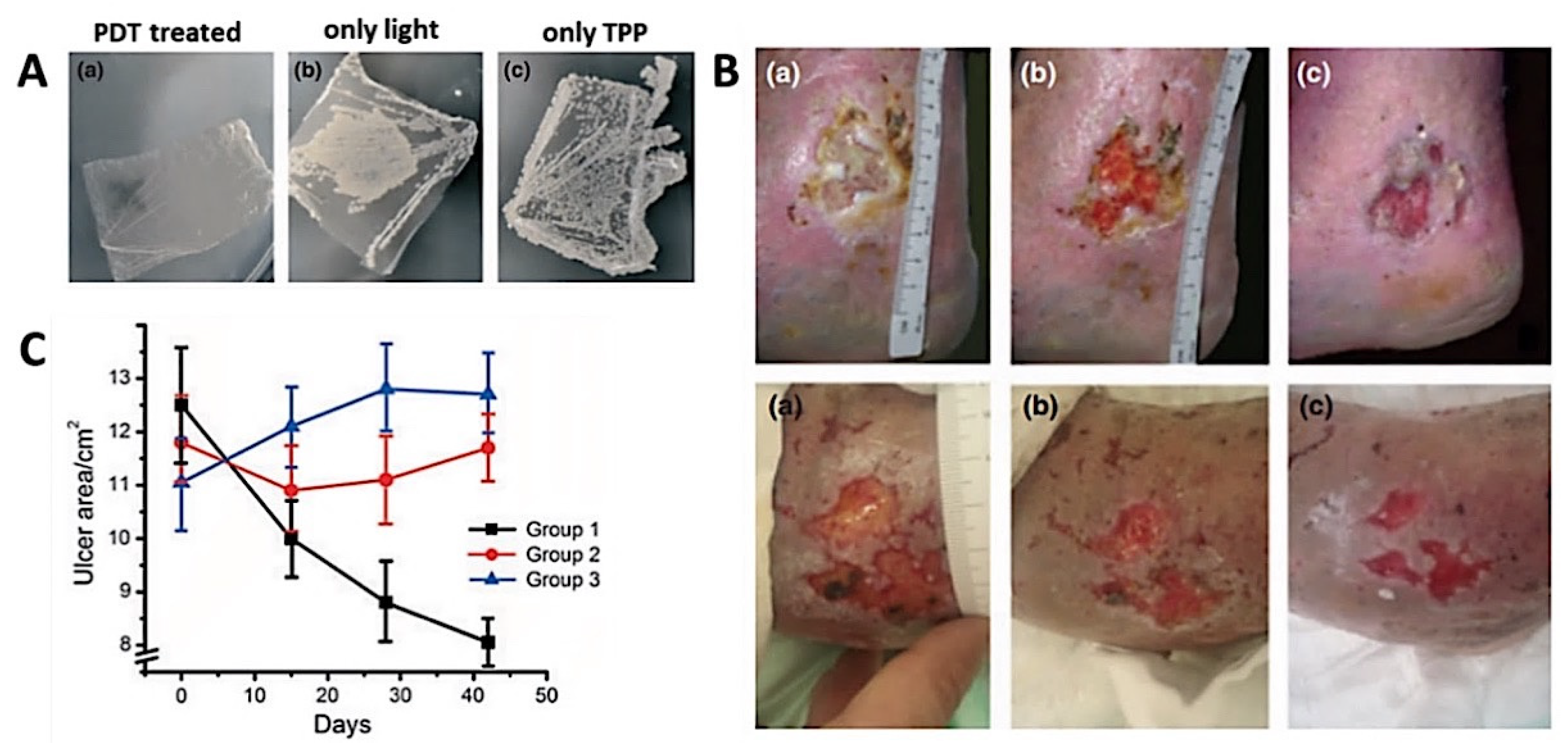
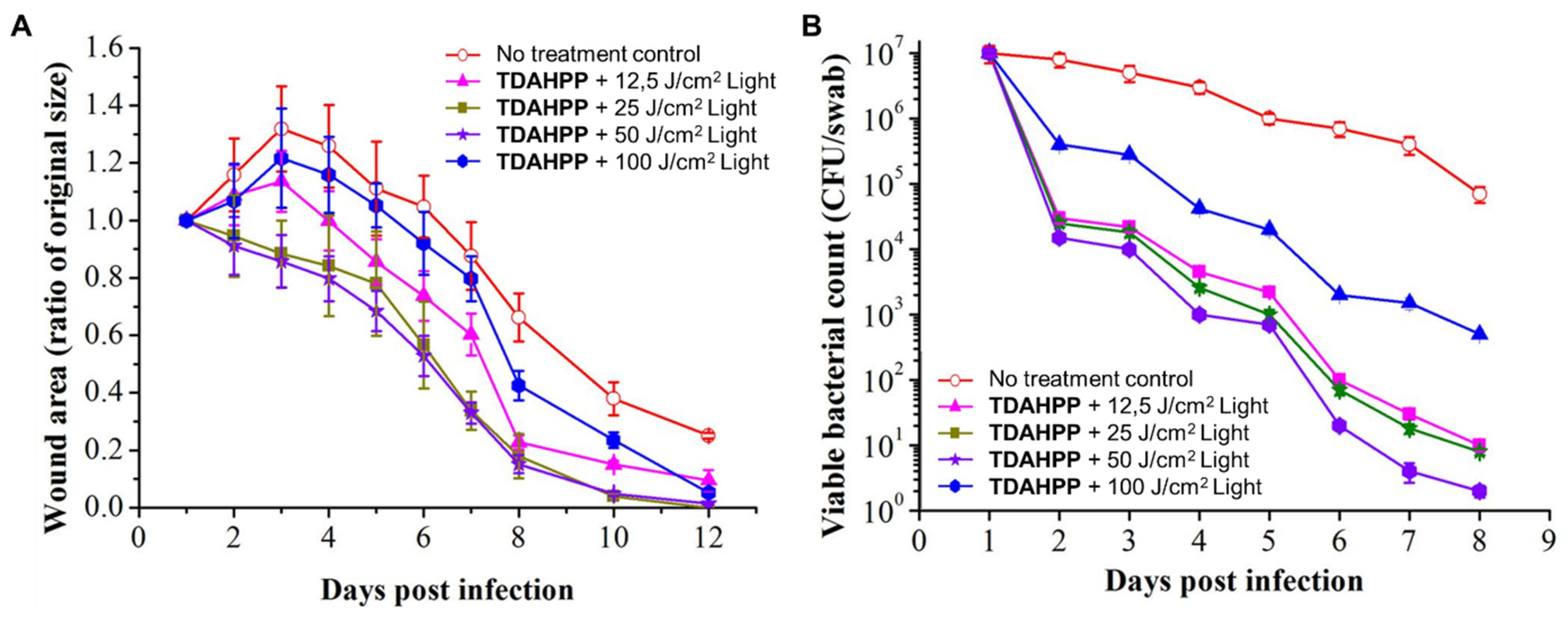
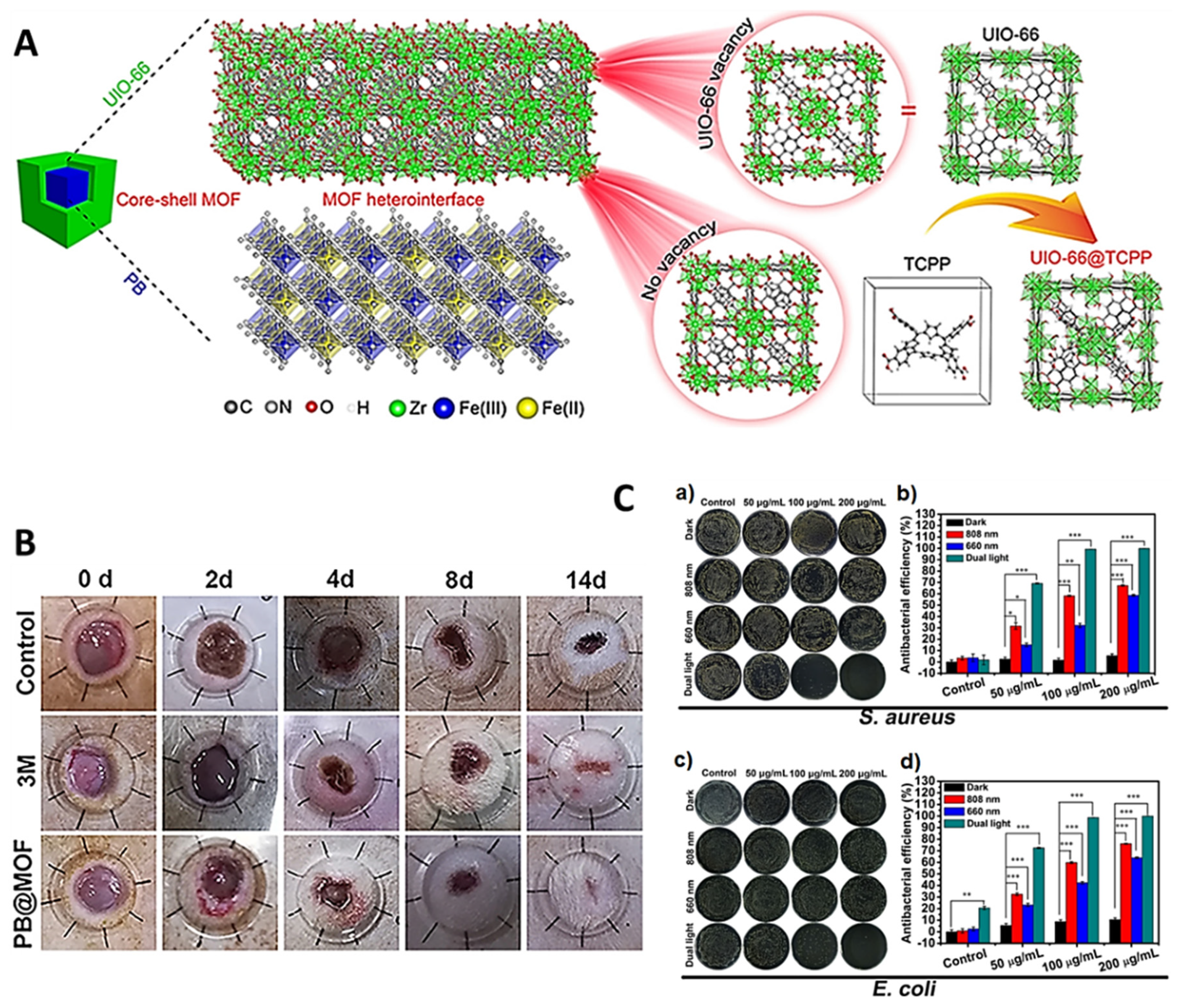
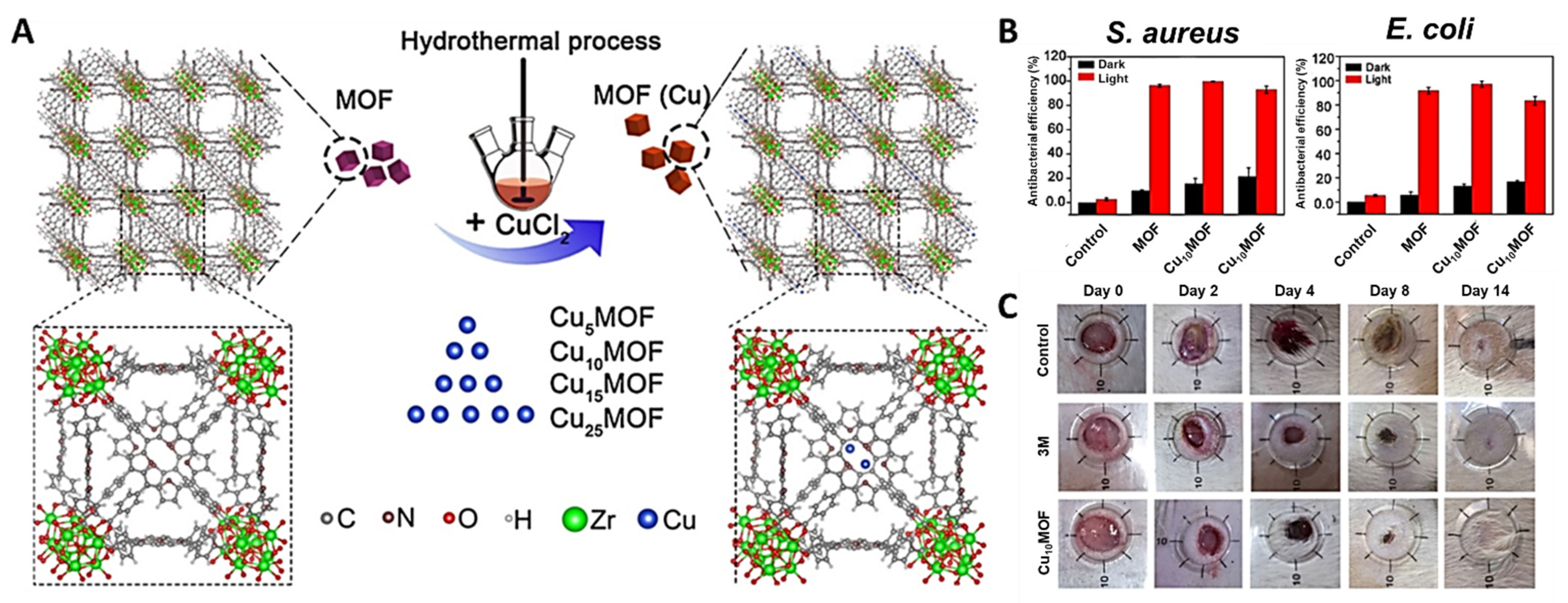
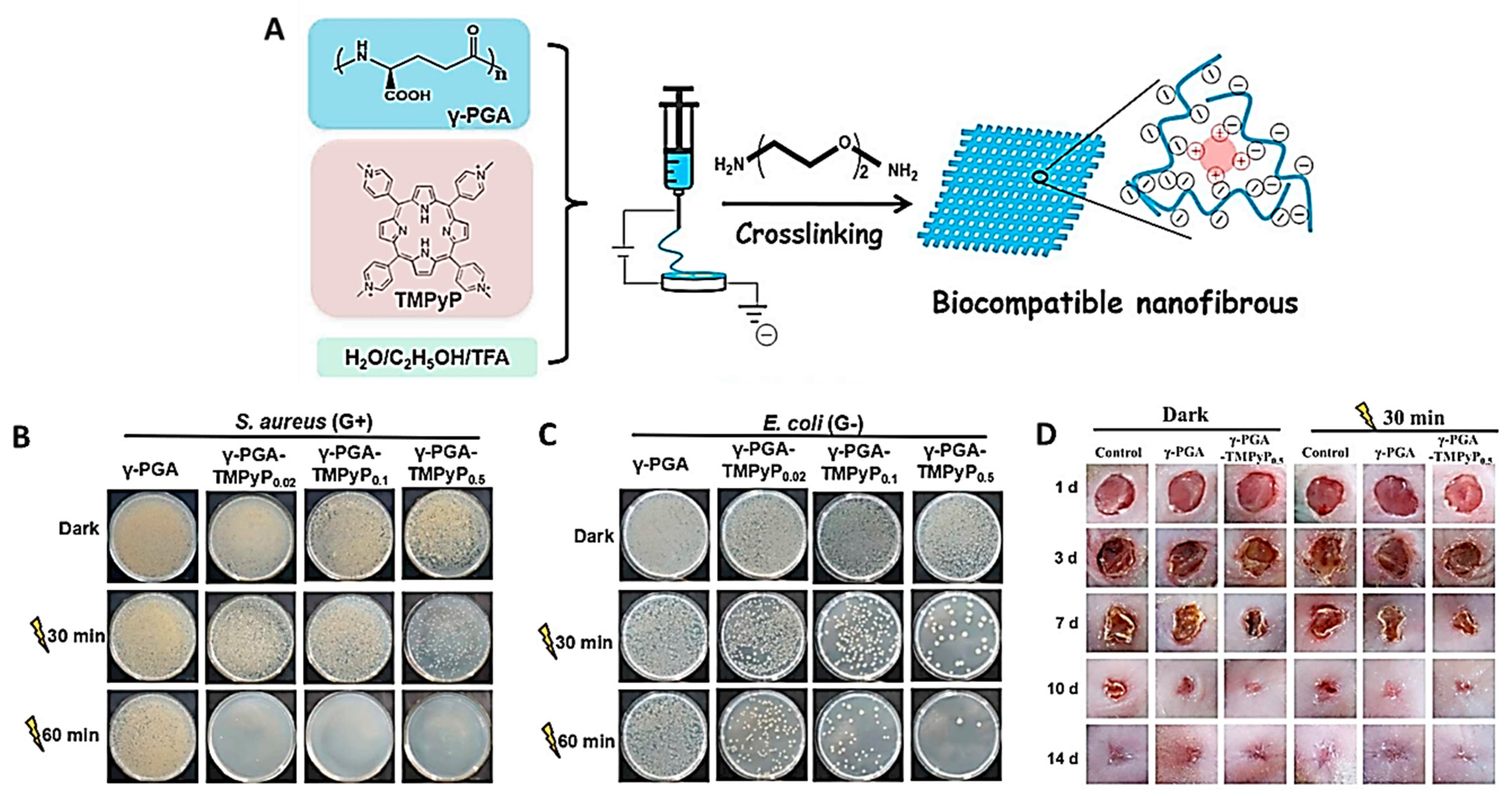
| Entry | Sample | Wound Healing Period (days) a |
|---|---|---|
| 1 | Control-1 | 19 ± 0 |
| 2 | Control-2 (ALA only) | 20 ± 1 |
| 3 | Control-3 (HpD only) | 18 ± 2 |
| 4 | Test-1 (ALA + He-Ne) | 13 ± 1 |
| 5 | Test-2 (ALA + He-Ne + Nd:YAG) | 17 ± 1 |
| 6 | Test-3 (HpD + He-Ne) | 16 ± 2 |
| 7 | Test-4 (HpD + He-Ne + Nd:YAG) | 14 ± 1 |
| Wound Type | Infection | PS | Log Reduction | Improved Healing | Ref. | |
|---|---|---|---|---|---|---|
| Ex Vivo | Wound healing organ culture | Not infected | ALA | n.a. | YES | [68] |
| In vivo animal studies | Excisions | Not infected | ALA | n.a. | YES | [72] |
| AlPcCl-PEG | n.a. | YES | [55] | |||
| E. coli | pLCe6 | YES | inconclusive | [98] | ||
| Ce6-IL | YES | YES | [108] | |||
| TCPP-PB@MOF | YES | YES | [118] | |||
| TCPP-PCN224 | YES | YES | [119] | |||
| MOF-TCPP-Dopamine | YES | YES | [121] | |||
| PGA-TMPyP nanofibrous mat | YES | YES | [122] | |||
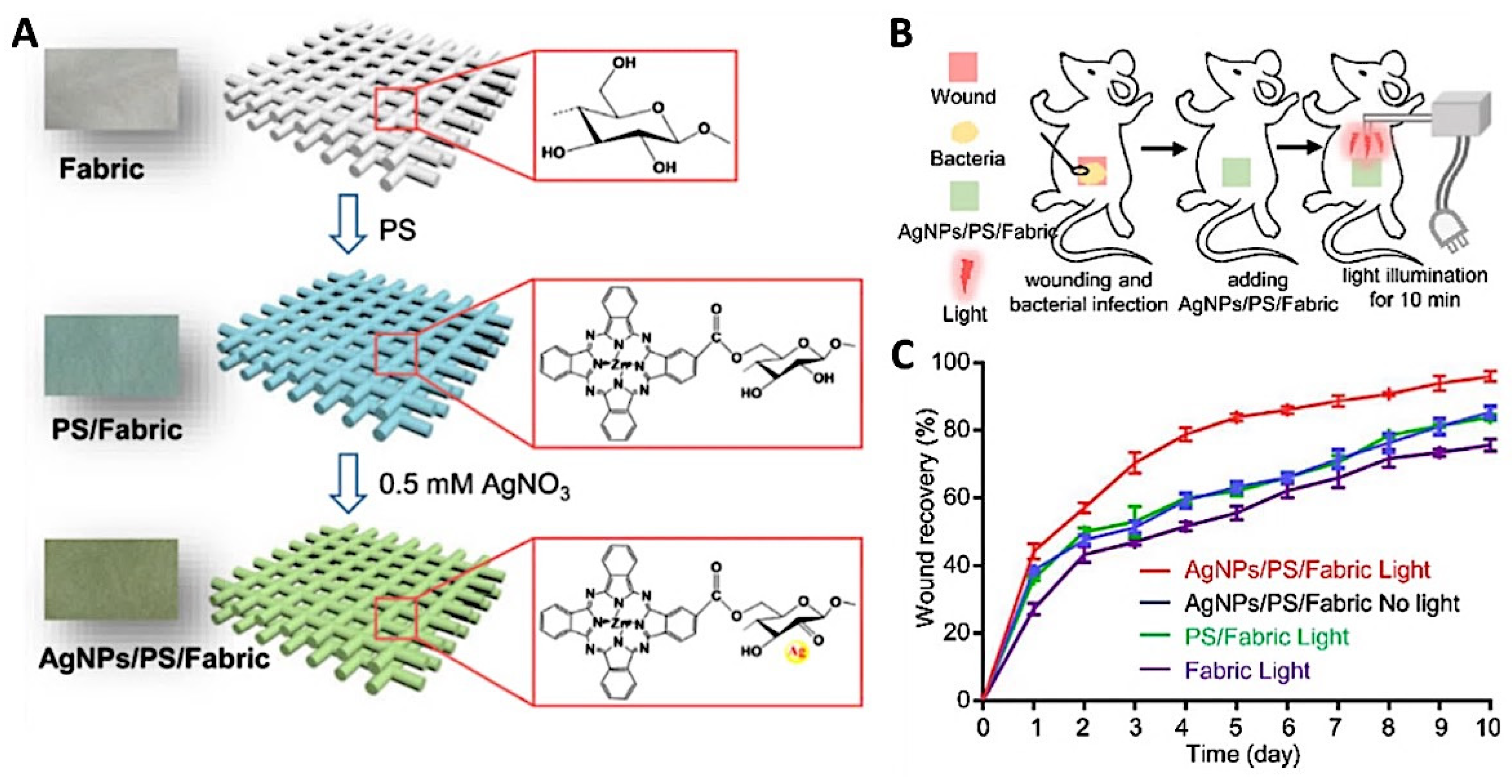
| AgNP/ZnPcCO2H fabric | YES | YES | [132] | |||
| P. aeruginosa | pL-Ce6 | YES | YES | [99] | ||
| pL–Cp6 | YES | NO | [110] | |||
| S. aureus | Ce6-IL | YES | YES | [108] | ||
| TCPP-PB@MOF | YES | YES | [118] | |||
| TCPP-PCN224 | YES | YES | [119] | |||
| MOF-TCPP-dopamine | YES | YES | [121] | |||
| PGA-TMPyP nanofibrous mat | YES | YES | [122] | |||
| MRSA; P. aeruginosa | pL–Cp6 | n.d. | NO | [111] | ||
| TMPyP | YES | inconclusive | [117] | |||
| MRSA; E. coli; P. aeruginosa | TDAHPP | YES | YES | [116] | ||
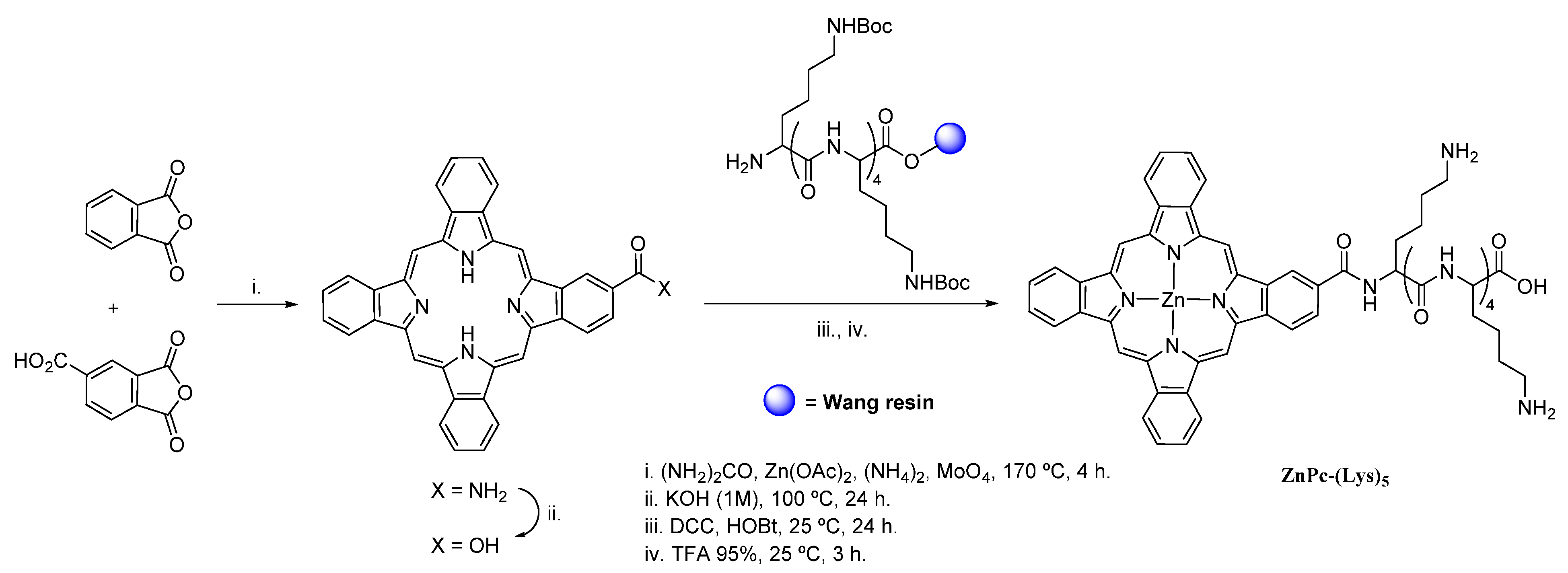
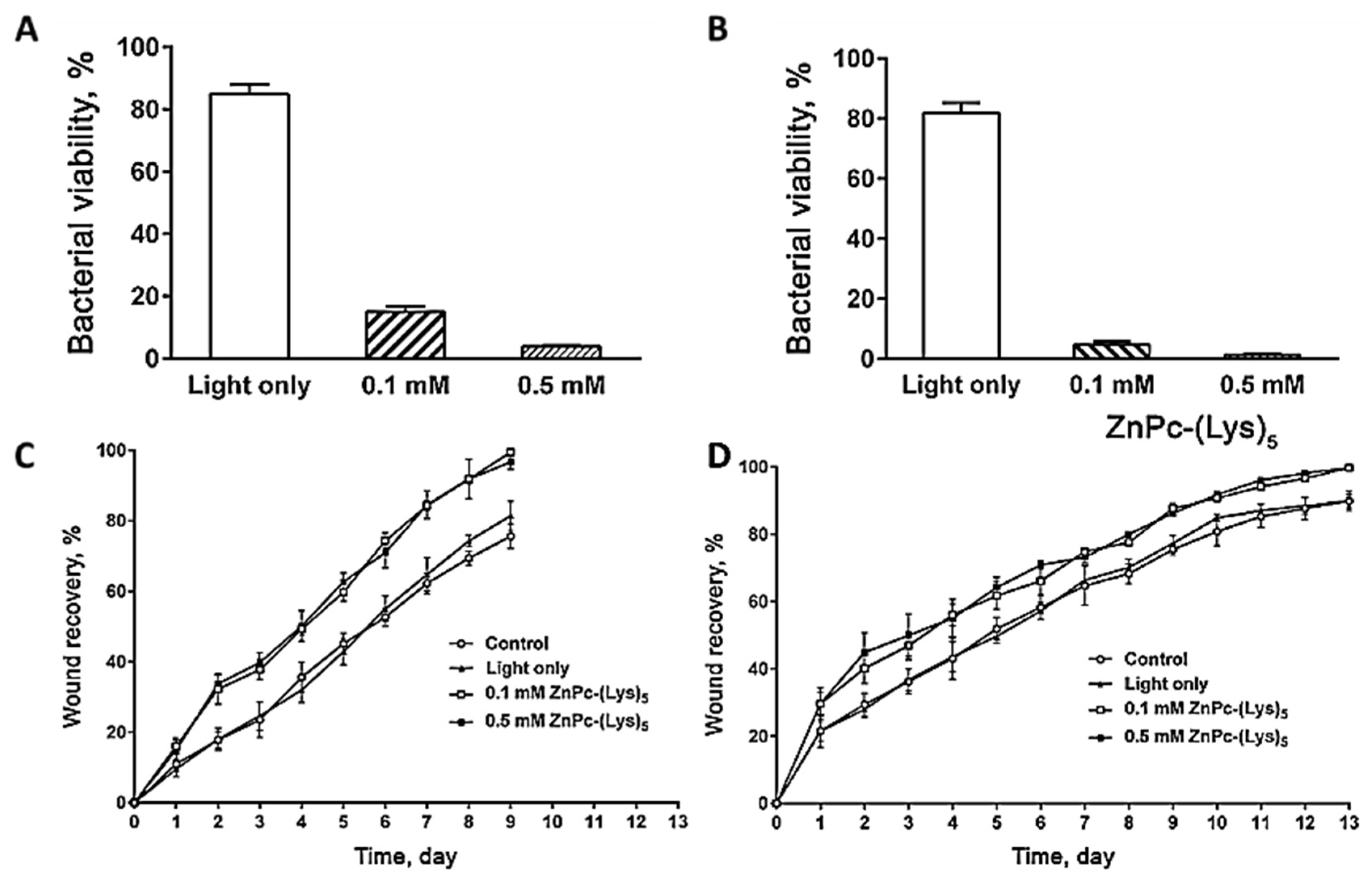
| MREC; MRAB | ZnPc-(Lys)5 | YES | YES | [130] | ||
| MRSA | RLP068/Cl-cellulose gel | YES | YES | [126] | ||
| Abscesses | S. aureus | pLCe6 | YES | inconclusive | [106] | |
| E. coli | YES | inconclusive | [106] | |||
| P. aeruginosa | YES | inconclusive | [106] | |||
| A. baumannii | YES | inconclusive | [100] | |||
| Burns | Not infected | MAL | n.a. | YES | [89] | |
| MRSA | PEI-Ce6 | YES | YES | [101] | ||
| RLP068/Cl | YES | YES | [127] | |||
| MDR S. aureus | Sinoporphyrin derivative | YES | YES | [112] | ||
| S. aureus | PTMPyP | YES | NO | [113] | ||
| Ulcers | MRSA | ALA | YES | YES | [79] | |
| Skin wounds (not specified) | Not infected | Pheophorbide a | n.a. | YES | [97] | |
| Fotoditazin–Pluronic F127 | n.a. | YES | [107] | |||
| P. aeruginosa | ALA | YES | YES | [80,90] | ||
| S. aureus | HTCC-Ce6-Mg-EGCG NP | YES | YES | [109] | ||
| Incisions | Not infected | m-THPC-collagen scaffolds | n.a. | YES | [114] | |
| CASP | n.a. | YES | [121] | |||
| MPD-MA | n.a. | NO | [122] | |||
| In vivo human studies | Cutaneous Leishmaniasis lesions | Leishmania major; Leishmania tropica | ALA | n.d. | YES | [75] [76] |
| Leg Ulcer | MRSA | ALA | n.d. | YES | [78] | |
| P. aeruginosa | ALA | n.d. | YES | [69] | ||
| RLP068/Cl | YES | YES | [128] | |||
| S. aureus; P. aeruginosa E. coli | TPP | YES | YES | [115] | ||
| S. aureus | RLP068/Cl | YES | YES | [128] | ||
| Chronic Venous Ulcers | E. faecalis | ALA | n.d. | YES | [81] | |
| MAL | n.d. | YES | [86] | |||
| S. aureus | ALA | n.d. | YES | [81] | ||
| MAL | n.d. | YES | [86] | |||
| Actinic Keratosis | Not infected | MAL | n.a. | YES | [82] | |
| Excisional Wounds | Not infected | MAL | n.a. | inconclusive | [67] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vallejo, M.C.S.; Moura, N.M.M.; Gomes, A.T.P.C.; Joaquinito, A.S.M.; Faustino, M.A.F.; Almeida, A.; Gonçalves, I.; Serra, V.V.; Neves, M.G.P.M.S. The Role of Porphyrinoid Photosensitizers for Skin Wound Healing. Int. J. Mol. Sci. 2021, 22, 4121. https://doi.org/10.3390/ijms22084121
Vallejo MCS, Moura NMM, Gomes ATPC, Joaquinito ASM, Faustino MAF, Almeida A, Gonçalves I, Serra VV, Neves MGPMS. The Role of Porphyrinoid Photosensitizers for Skin Wound Healing. International Journal of Molecular Sciences. 2021; 22(8):4121. https://doi.org/10.3390/ijms22084121
Chicago/Turabian StyleVallejo, Mariana C. S., Nuno M. M. Moura, Ana T. P. C. Gomes, Ana S. M. Joaquinito, Maria Amparo F. Faustino, Adelaide Almeida, Idalina Gonçalves, Vanda Vaz Serra, and Maria Graça P. M. S. Neves. 2021. "The Role of Porphyrinoid Photosensitizers for Skin Wound Healing" International Journal of Molecular Sciences 22, no. 8: 4121. https://doi.org/10.3390/ijms22084121
APA StyleVallejo, M. C. S., Moura, N. M. M., Gomes, A. T. P. C., Joaquinito, A. S. M., Faustino, M. A. F., Almeida, A., Gonçalves, I., Serra, V. V., & Neves, M. G. P. M. S. (2021). The Role of Porphyrinoid Photosensitizers for Skin Wound Healing. International Journal of Molecular Sciences, 22(8), 4121. https://doi.org/10.3390/ijms22084121












