The Influence of UV Light on Photodegradation of Acetylsalicylic Acid
Abstract
1. Introduction
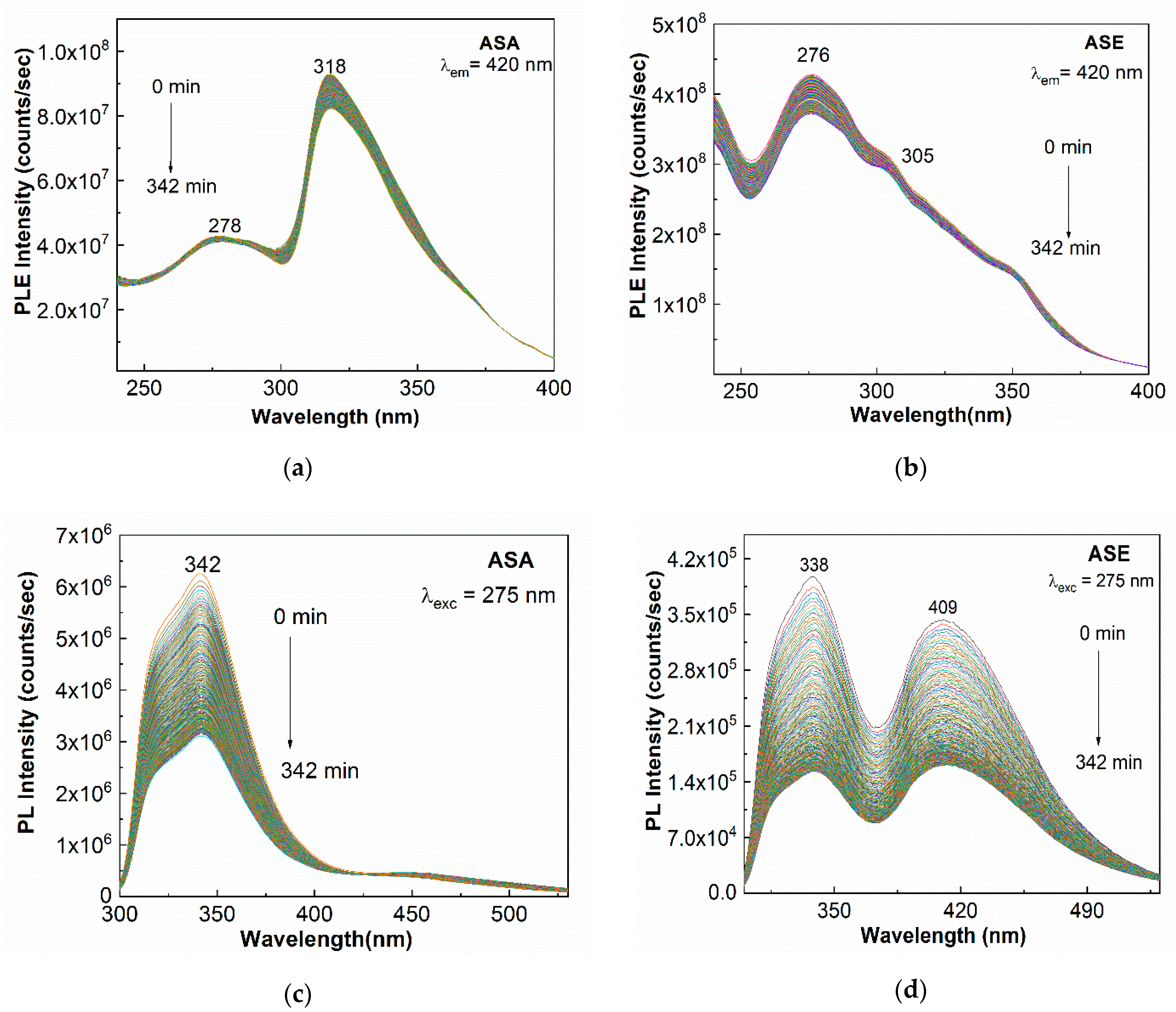
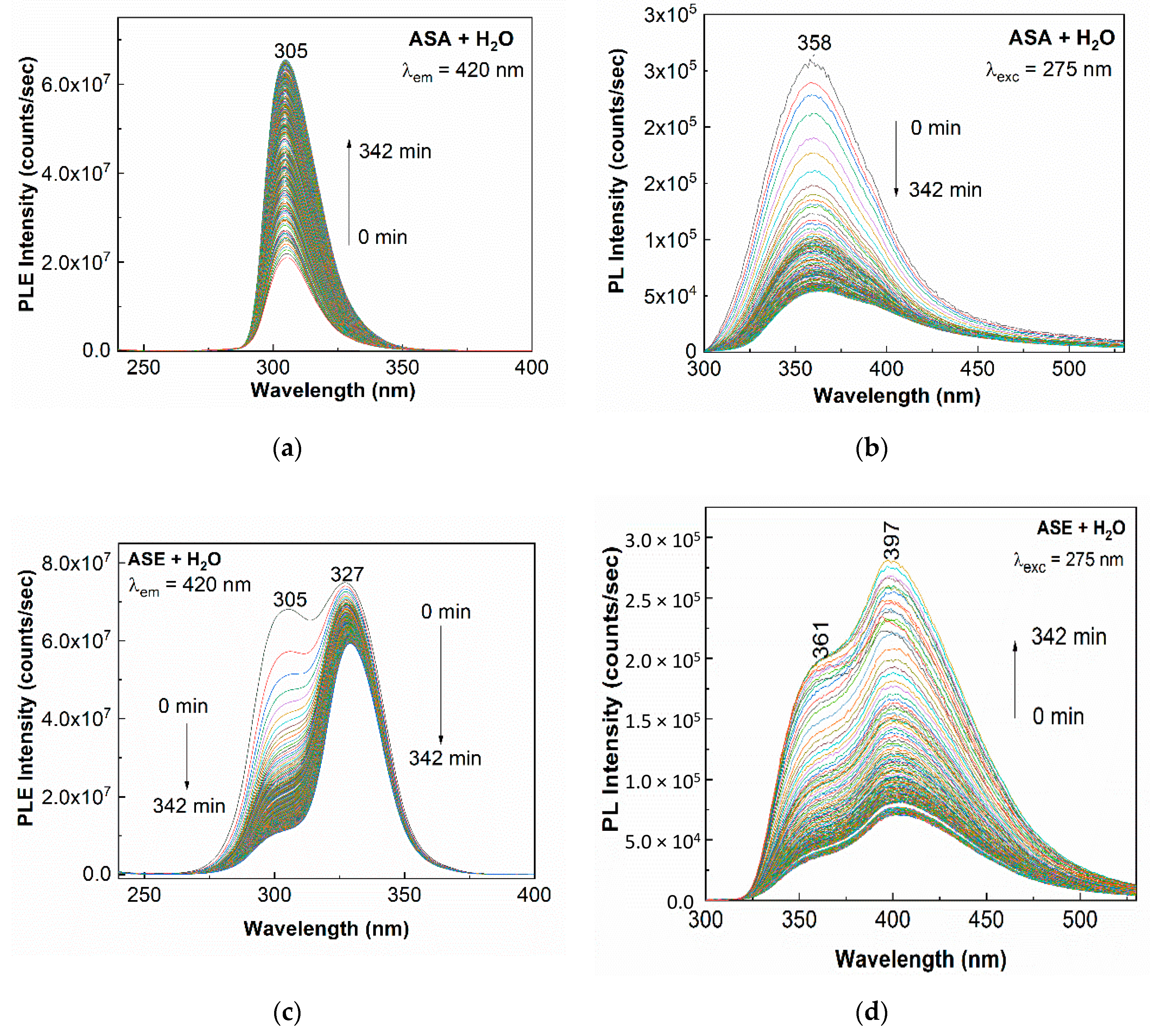
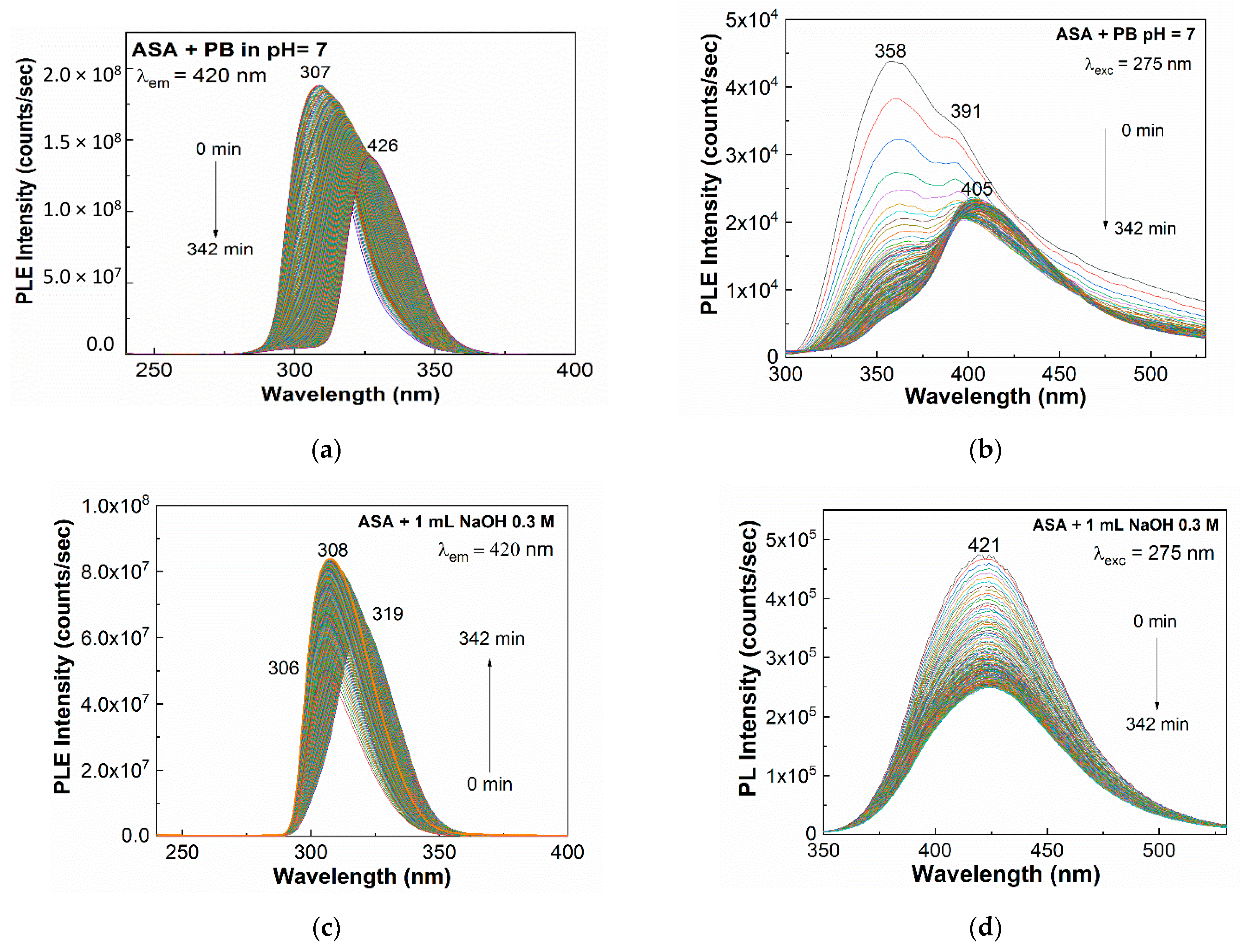
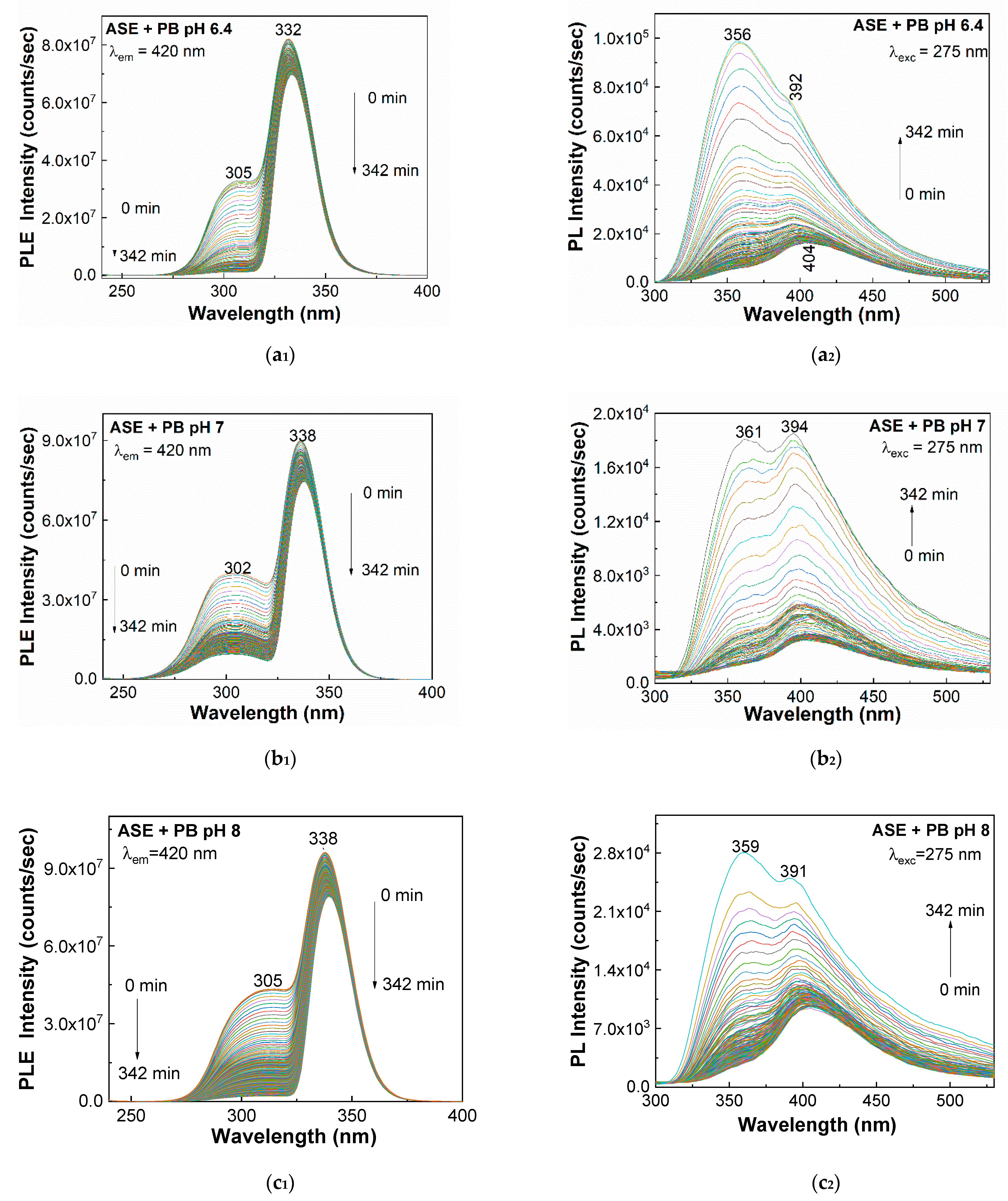
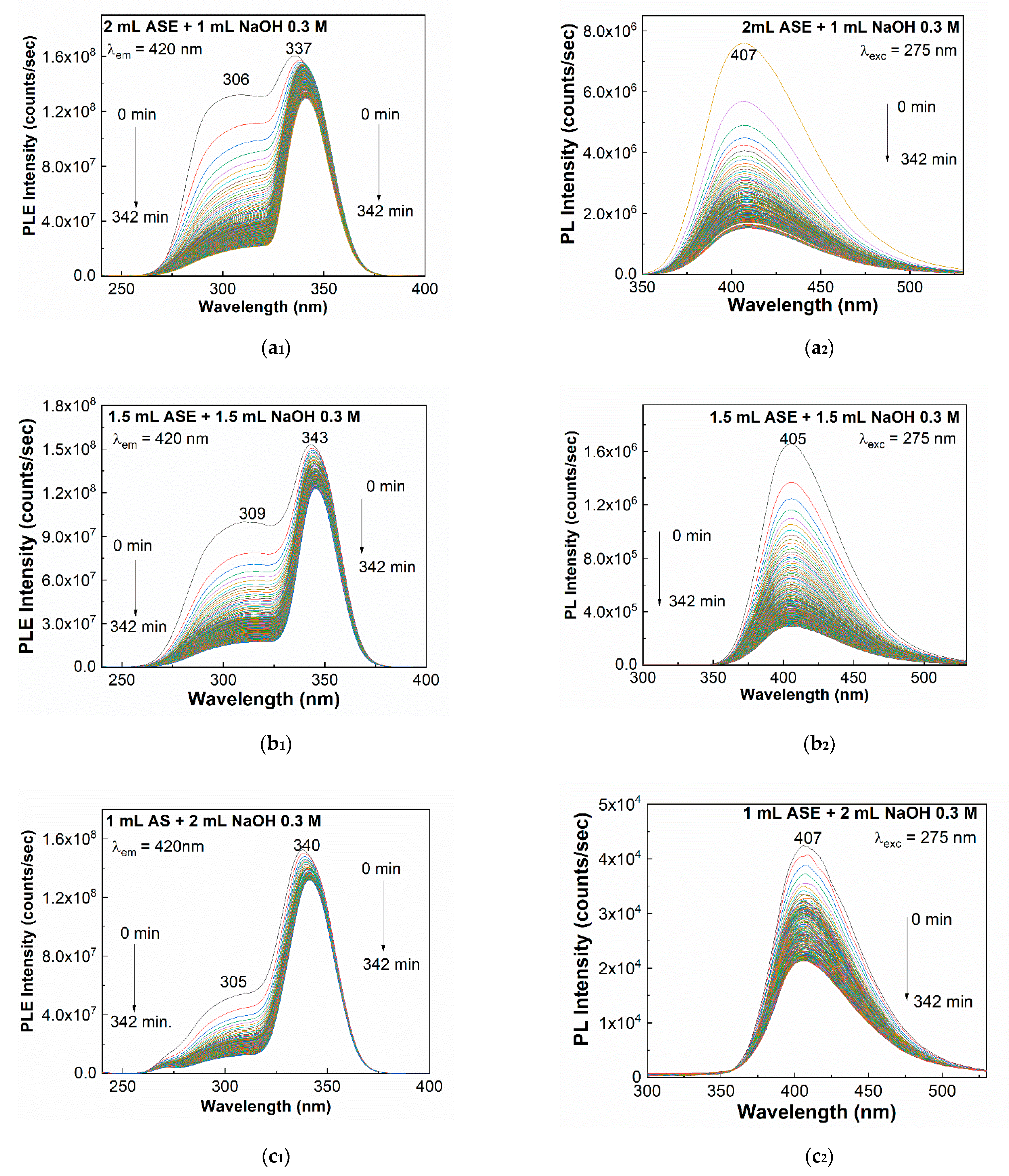
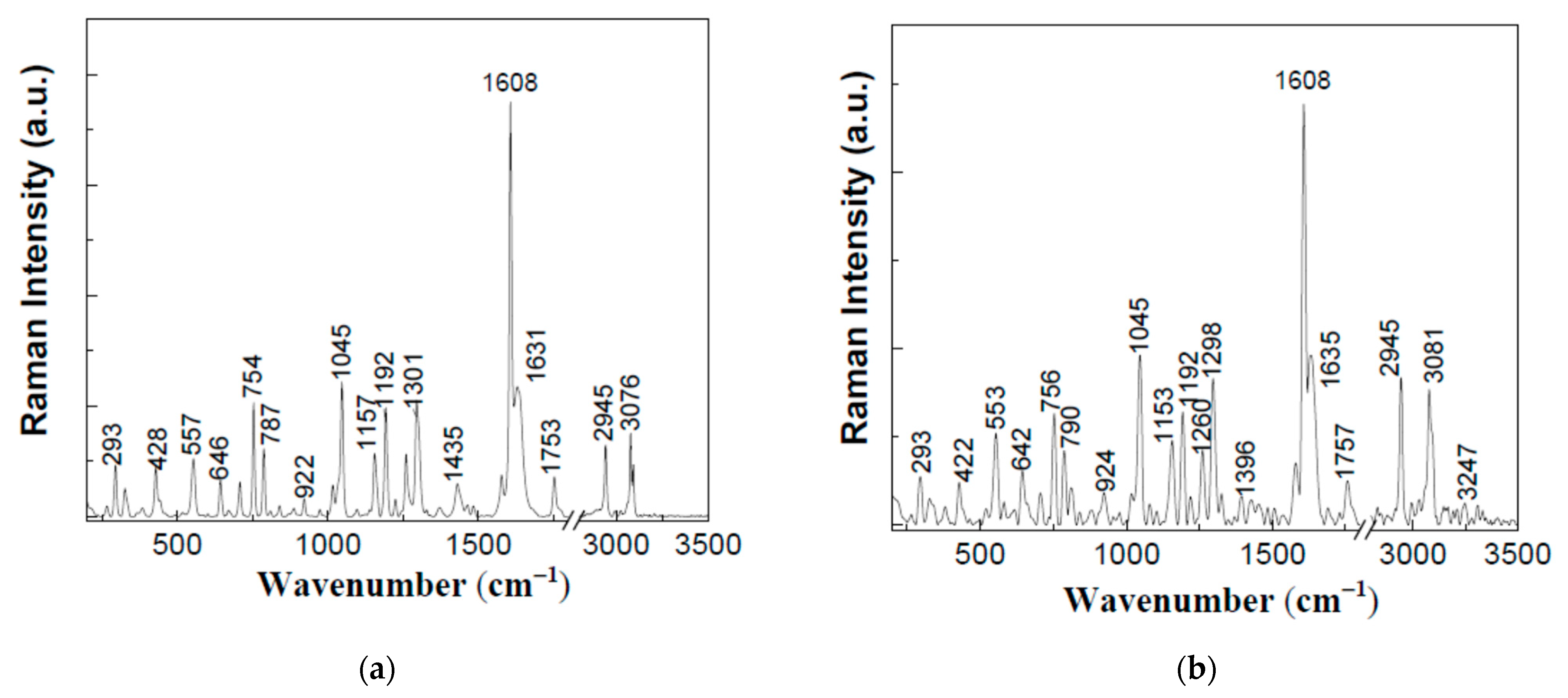
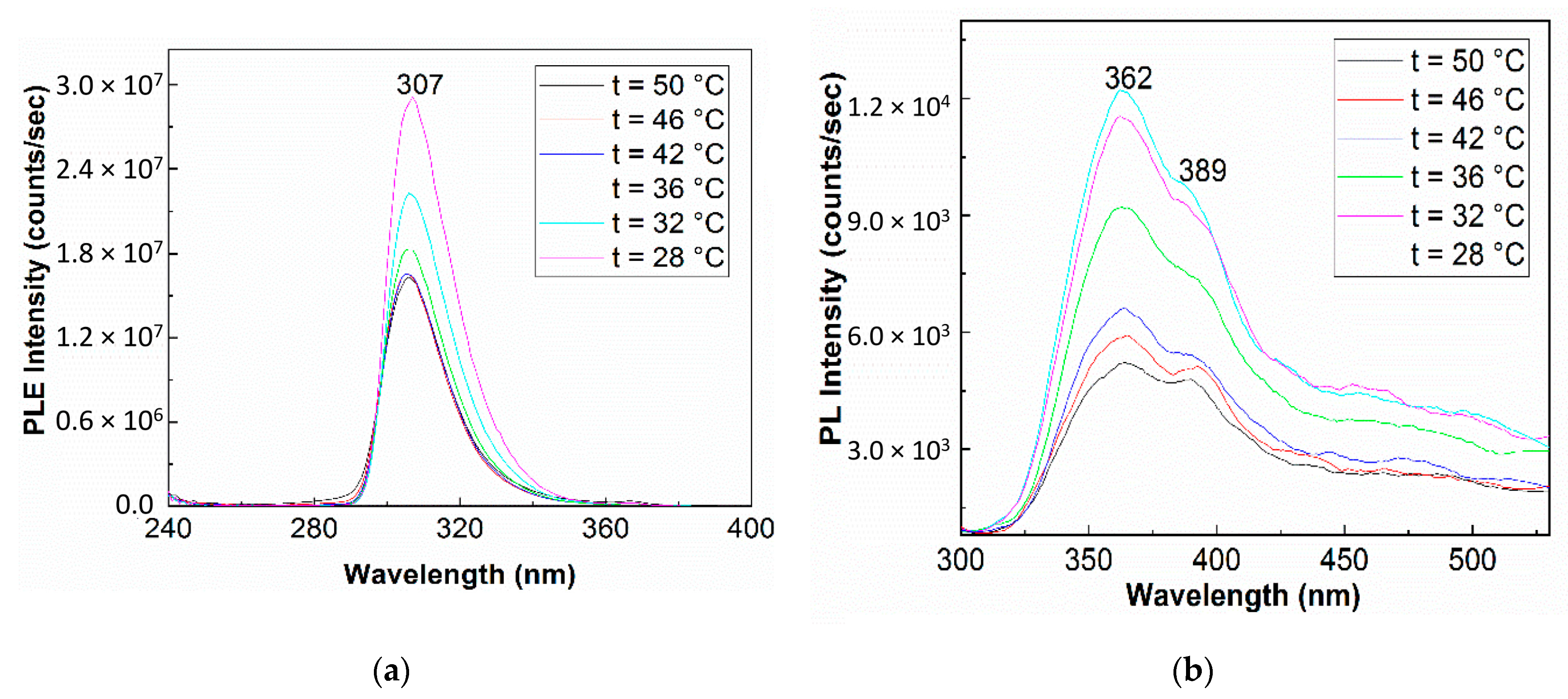
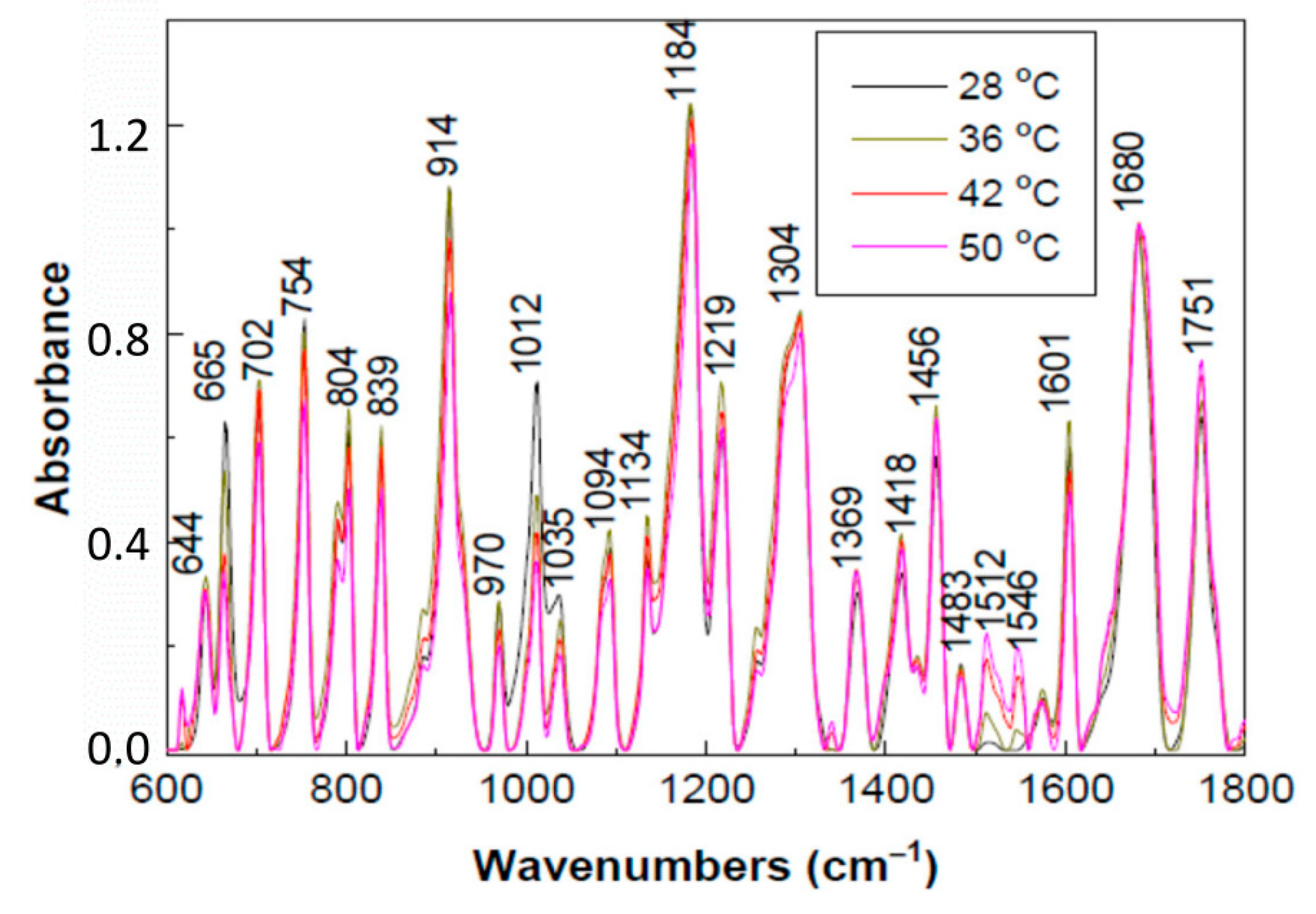
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Parolini, M. Toxicity of the Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) acetylsalicylic acid, paracetamol, diclofenac, ibuprofen and naproxen towards freshwater invertebrates: A review. Sci. Total Environ. 2020, 740, 140043. [Google Scholar] [CrossRef] [PubMed]
- Fokunang, C.N.; Fokunang, E.T.; Frederick, K.; Ngameni, B.; Ngadjui, B. Overview of non-steroidal anti-inflammatory drugs (nsaids) in resource limited countries. MOJ Toxicol. 2018, 4, 5–13. [Google Scholar] [CrossRef]
- Bianconi, V.; Violi, F.; Fallarino, F.; Pignatelli, P.; Sahebkar, A.; Pirro, M. Is Acetylsalicylic Acid a Safe and Potentially Useful Choice for Adult Patients with COVID-19? Drugs 2020, 80, 1383–1396. [Google Scholar] [CrossRef] [PubMed]
- Osbone, T.F.; Veigulis, Z.P.; Arreola, D.M.; Mahajan, S.M.; Roosli, E.; Curtin, C.M. Association of mortality and aspirin prescription for COVID-19 patients at the veterans health administration. PLoS ONE 2021, 16, e0246825. [Google Scholar]
- Tsai, S.C.; Lu, C.C.; Bau, D.T.; Chiu, Y.J.; Yen, Y.T.; Hsu, Y.M.; Fu, C.W.; Kuo, S.C.; Lo, Y.S.; Chiu, H.Y.; et al. Approaches towards fighting the COVID-19 pandemic (review). Int. J. Molec. Med. 2021, 47, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Bortolasci, C.G.; Puri, B.K.; Olive, L.; Marx, W.; O’Neil, A.; Athan, E.; Carvalho, A.; Maes, M.; Walder, K.; et al. Preventing the development of severe COVID-19 by modifying immunothrombosis. Life Sci. 2021, 264, 118617. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.; Zhong, L.; Duan, T.; Zhang, R.H.; Wang, X.; Wang, G.; Hu, K.; Lv, X.; Kang, T. Asprin suppresses the growth and metastasis of osteosarcoma through the NF-kB pathway. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015, 21, 5349–5359. [Google Scholar] [CrossRef]
- Ornelas, A.; Zacharias-Millward, N.; Menter, D.G.; Davis, J.S.; Lichtenberger, L.; Hawke, D.; Hawk, E.; Vilar, E.; Bhattacharya, P.; Millward, S. Beyond COX-1: The effects of aspirin on platelet biology and potential mechanisms of chemoprevention. Cancer Metastasis Rev. 2017, 36, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Alfonso, L.; Ai, G.; Spittle, R.C.; Bhat, G.J. Molecular targets of aspirin and cancer prevention. Brit. J. Cancer 2014, 111, 61–67. [Google Scholar] [CrossRef]
- Verardi, L.; De Lorenzis, E.; Natalello, G. Tolerability and safety of acetylsalicylic acid in patients with systemic sclerosis. Ann. Rheum. Dis. 2020, 79, 1606. [Google Scholar] [CrossRef]
- Loomans-Kropp, H.A.; Pinsky, P.; Cao, Y.; Chan, A.T.; Umar, A. Association of Aspirin Use With Mortality Risk Among Older Adult Participants in the Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial. JAMA Netw. Open 2019, 2, 1916729. [Google Scholar] [CrossRef] [PubMed]
- Farias, S.; Boateng, J.S. In vitro, ex vivo and in vivo evaluation of taste masked low dose acetylsalicylic acid loaded composite wafers as platforms for buccal administration in geriatric patients with dysphagia. Int. J. Pharm. 2020, 589, 119807. [Google Scholar] [CrossRef] [PubMed]
- Bugiardini, R.; Pavasović, S.; Yoon, J.; van der Schaar, M.; Kedev, S.; Vavlukis, M.; Badimon, L. Aspirin for primary prevention of ST segment elevation myocardial infarction in persons with diabetes and multiple risk factors. E-Clin. Med. 2020, 27, 100548. [Google Scholar]
- Wang, Z.; Tang, Z.; Peng, X.; Zhu, W.; Ying-jia, X.; Ge, L.; Ge, J. Network Meta-analysis: Aspirin plus Traditional Chinese Medicine for stroke prevention in Patients with Atrial Fibrillation. J. Herb. Med. 2020, 22, 100355. [Google Scholar] [CrossRef]
- Wang, S.; Yu, Y.; Ryan, P.M.; Dang, M.; Clark, C.; Kontogiannis, V.; Rahmani, J.; Varkaneh, H.K.; Salehisahlabadi, A.; Day, A.S.; et al. Association of aspirin therapy with risk of hepatocellular carcinoma: A systematic review and dose-response analysis of cohort studies with 2.5 million participants. Pharmacol. Res. 2020, 151, 104585. [Google Scholar] [CrossRef]
- Dos Santos, F.D.A.G.; Leite-Andrade, M.C.; de Sousa Brandão, I.; da Silva Alves, A.I.; Buonafina, M.D.S.; Nunes, M.; de Araújo-Neto, L.N.; de Freitas, M.A.; Brayner, F.A.; Alves, L.C.; et al. Anti-biofilm effect by the combined action of fluconazole and acetylsalicylic acid against species of Candida parapsilosis complex. Infect. Genet. Evol. 2020, 84, 104378. [Google Scholar] [CrossRef]
- Azboy, I.; Groff, H.; Goswami, K.; Vahedian, M.; Parvizi, J. Low-Dose Aspirin Is Adequate for Venous Thromboembolism Prevention Following Total Joint Arthroplasty: A Systematic Review. J. Arthroplasty 2020, 35, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xiong, C.; Liu, J.; Sun, T.; Ren, Z.; Li, Y.; Geng, J.; Li, X. Aspirin exerts anti-tumor effect through inhibiting Blimp1 and activating ATF4/CHOP pathway in multiple myeloma. Biomed. Pharm. 2020, 125, 110005. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.; Daniel, A.; Gyadangi, B.; Ramsamy, S. Isolation, Characterization of a Potential Degradation Product of Aspirin and an HPLC Method for Quantitative Estimation of Its Impurities. J. Chromatogr. Sci. 2015, 53, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.; Ali, M.; Ahuja, A.; Ali, J. Stability Testing of Pharmaceutical Products—Comparison of Stability Testing Guidelines. Curr. Pharm. Anal. 2010, 6, 142–150. [Google Scholar] [CrossRef]
- Singh, R.; Tiwari, M.K.; Gangopadhayay, D.; Mishra, P.C.; Mishra, H.; Srivastava, A.; Singh, R.K. Detection and monitoring of in vitro formation of salicylic acid from aspirin using fluorescence spectroscopic technique and DFT calculations. J. Photoch. Photobiolgy B 2018, 189, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, P.P.; Li, X.X.; Nie, K.; Tuo, M.-F.; Kong, B.; Chen, J. Monitoring the hydrolyzation of aspirin during the dissolution testing for aspirin delayed-release tablets with a fiber-optic dissolution system. J. Pharm. Anal. 2012, 2, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Husain, M.A.; Rehman, S.U.; Ishqi, H.M.; Sarwar, T.; Tabish, M. Spectroscopic and molecular docking evidence of aspirin and diflunisal binding to DNA: A comparative study. RSC Adv. 2015, 5, 64335–64345. [Google Scholar] [CrossRef]
- Muthuselvi, C.; Dhavachitra, M.; Pandiarajan, S. Growth and Characterization of Aspirin Crystal in the Phosphoric acid Medium. J. Chem. Pharm. Res. 2016, 8, 804–814. [Google Scholar]
- Zhen, X.-E.; Zong, M.; Gao, S.-N.; Cao, Y.-G.; Jiang, L.; Chen, S.-X.; Li, S. Preparation and Characterization of a Novel Aspirin Derivative with Anti-Thrombotic and Gastric Mucosal Protection Properties. PLoS ONE 2014, 9, e98513. [Google Scholar]
- Motan, G.; Puia, A. Studies of different types of aspirin by spectrophotometric methods. Acta Chem. Iasi 2014, 22, 155–164. [Google Scholar] [CrossRef]
- Szostak, R.; Mazurek, S. Quantitative determination of acetylsalicylic acid and acetaminophen in tablets by FT-Raman spectroscopy Electronic Supplementary Information available. Analyst 2001, 127, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Heng, J.Y.Y.; Bismarck, A.; Lee, A.F.; Wilson, K.; Williams, D.R. Anisotropic Surface Chemistry of Aspirin Crystals. J. Pharm. Sci. 2007, 96, 2134–2144. [Google Scholar] [CrossRef] [PubMed]
- Ignaczak, A.; Porwański, S.; Szyszka, M. Deeper insight into the properties of the newly synthesized macrocycles as drug receptors – some preliminary quantum chemical studies. New J. Chem. 2017, 41, 521–529. [Google Scholar] [CrossRef]
- De Aguiar, J.L.N.; Leandro, K.C.; Abrantes, S.D.M.P.; Albert, A.L.M. Development of a new analytical method for determination of acetylsalicylic and salicylic acids in tablets by reversed phase liquid chromatography. Braz. J. Pharm. Sci. 2009, 45, 723–727. [Google Scholar] [CrossRef]
- Ramjith, S.; Sunith, D.K.; Radhakrishnan, S.; Sameer, P.A. HPLC study of aspirin and aspirin derivatives. Int. J. Res. Pharm. Chem. 2013, 3, 1–5. [Google Scholar]
- Cofan, C.; Radovan, C. Anodic Determination of Acetylsalicylic Acid at a Mildly Oxidized Boron-Doped Diamond Electrode in Sodium Sulphate Medium. Int. J. Electrochem. 2011, 2011, 1–9. [Google Scholar] [CrossRef][Green Version]
- Mukherjee, D.; Ray, A.; Barghi, S. Mechanism of Acetyl Salicylic Acid (Aspirin) Degradation under Solar Light in Presence of a TiO2-Polymeric Film Photocatalyst. Processes 2016, 4, 13. [Google Scholar] [CrossRef]
- Dai, Q.; Xia, Y.; Jiang, L.; Li, W.; Wang, J.; Chen, J. Enhanced Degradation of Aspirin by Electrochemical oxidation with Modified PbO2Electrode and Hydrogen Peroxide. Int. J. Electrochem. Sci. 2012, 7, 12895–12906. [Google Scholar]
- Daescu, M.; Matea, A.; Negrila, C.; Serbschi, C.; Ion, A.C.; Baibarac, M. Photoluminescence as a valuable tool in the optical characterization of acetaminophen and the monitoring of its photodegradation reactions. Molecules 2020, 25, 4571. [Google Scholar] [CrossRef]
- Daescu, M.; Toulbe, N.; Baibarac, M.; Mogos, A.; Lorinczi, A.; Logofatu, C. Photoluminescence as a complementary tool for UV-VIS spectroscopy to highlight the photodegradation of drugs: A case study on melatonin. Molecules 2020, 25, 3820. [Google Scholar] [CrossRef] [PubMed]
- Baibarac, M.; Smaranda, I.; Nila, A.; Serbschi, C. Optical properties of folic acid in phosphate buffer solutions: The influence of pH and UV irradiation on the UV-VIS absorption spectra and photoluminescence. Sci. Rep. 2019, 9, 14278. [Google Scholar] [CrossRef] [PubMed]
- Smaranda, I.; Nila, A.; Manta, C.M.; Samohvalov, D.; Gherca, D.; Baibarac, M. The influence of UV light on the azathioprine photodegradation: New evidences by photoluminescence. Results Phys. 2019, 14, 102443. [Google Scholar] [CrossRef]
- Thiessen, J.J. Aspirin: Plasma concentration and effects. Thromb. Res. 1983, 29 (Suppl. IV), 105–111. [Google Scholar] [CrossRef]
- Edwards, I.J. The hydrolysis of aspirin, Part 2. Trans. Faraday Soc. 1952, 48, 696–699. [Google Scholar] [CrossRef]
- Kozma, L.; Hornak, I.; Eroshtak, I.; Nemet, B. Study of the fluorescent properties of salicylic acid derivates on solutions. Transl. Zheermal Prikl. Specktroscopii 1990, 53, 259–269. [Google Scholar]
- Dumitrescu, V.; Surmeian, M.; Doneanu, C.; Stanescu, S. Computer program for the determination of salicylic acid content in aspirin by derivative spectrometry. Anal. Chim. Acta 1996, 333, 181–186. [Google Scholar] [CrossRef]
- Premkumar, R.; Premkumar, S.; Rekha, T.N.; Parameswari, A.; Mathavan, T.; Benial, A.M.F. Surface enhanced Raman spectroscopic studies on aspirin: An experimental and theoretical approach. AIP Conf. Proc. 2016, 1728, 020613. [Google Scholar]
- Binev, I.G.; Stamboliyska, B.A.; Binev, Y.I. The infrared spectra and structure of acetylsalicylic acid (aspirin) and its oxyanion: An ab initio force field treatment. J. Molec. Struct. 1996, 378, 189–197. [Google Scholar]
- Balci, A.K.; Koksal, O.; Kose, A.; Armagan, E.; Ozdemir, F.; Inal, T.; Oner, N. General characteristics of patients with electrolyte imbalance admitted to emergency department. World J. Emerg. Med. 2013, 4, 113–116. [Google Scholar] [CrossRef] [PubMed]
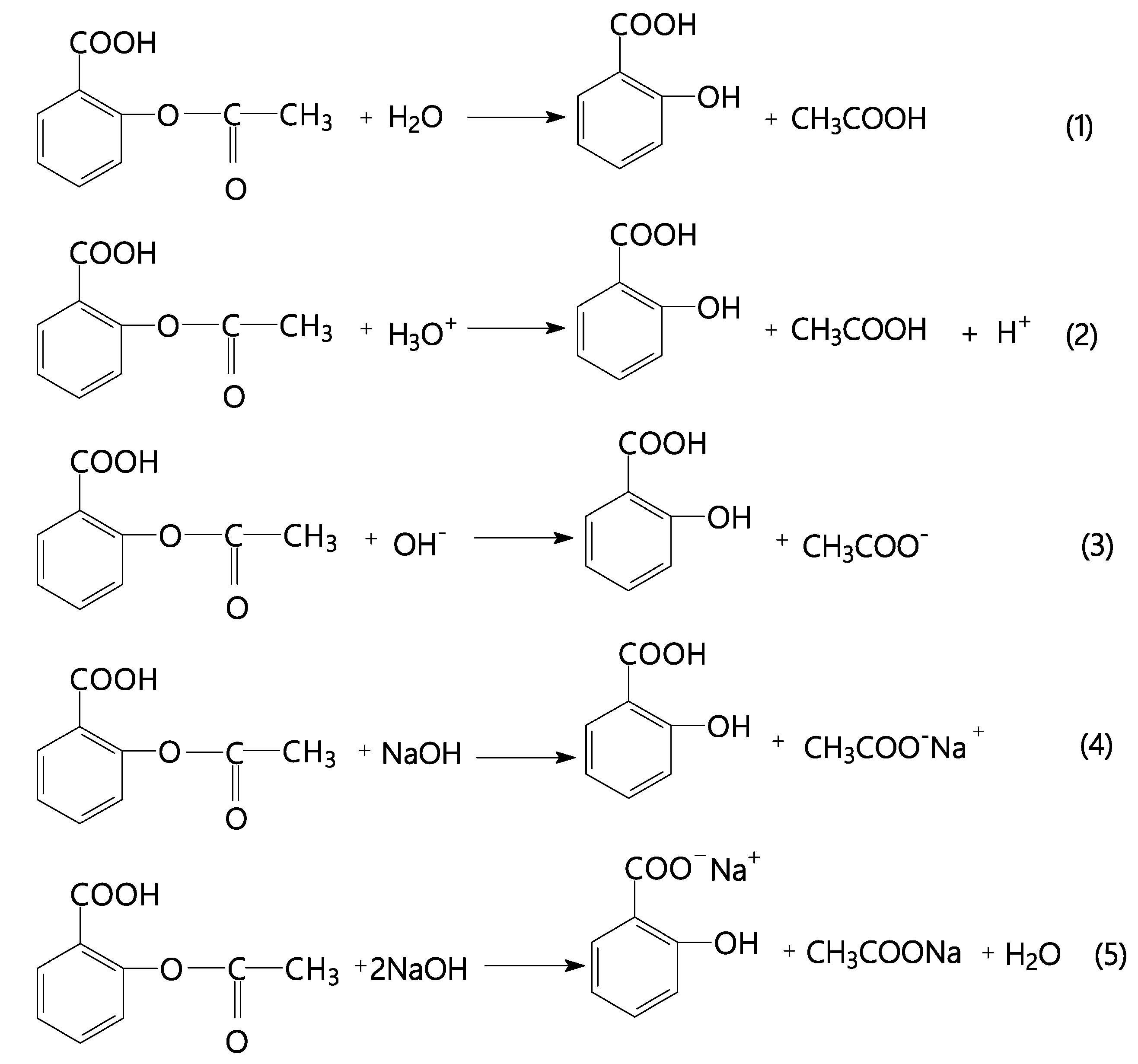

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daescu, M.; Iota, M.; Serbschi, C.; Ion, A.C.; Baibarac, M. The Influence of UV Light on Photodegradation of Acetylsalicylic Acid. Int. J. Mol. Sci. 2021, 22, 4046. https://doi.org/10.3390/ijms22084046
Daescu M, Iota M, Serbschi C, Ion AC, Baibarac M. The Influence of UV Light on Photodegradation of Acetylsalicylic Acid. International Journal of Molecular Sciences. 2021; 22(8):4046. https://doi.org/10.3390/ijms22084046
Chicago/Turabian StyleDaescu, Monica, Miruna Iota, Constantin Serbschi, Alina C. Ion, and Mihaela Baibarac. 2021. "The Influence of UV Light on Photodegradation of Acetylsalicylic Acid" International Journal of Molecular Sciences 22, no. 8: 4046. https://doi.org/10.3390/ijms22084046
APA StyleDaescu, M., Iota, M., Serbschi, C., Ion, A. C., & Baibarac, M. (2021). The Influence of UV Light on Photodegradation of Acetylsalicylic Acid. International Journal of Molecular Sciences, 22(8), 4046. https://doi.org/10.3390/ijms22084046






