Biological Properties and Prospects for the Application of Eugenol—A Review
Abstract
1. Introduction
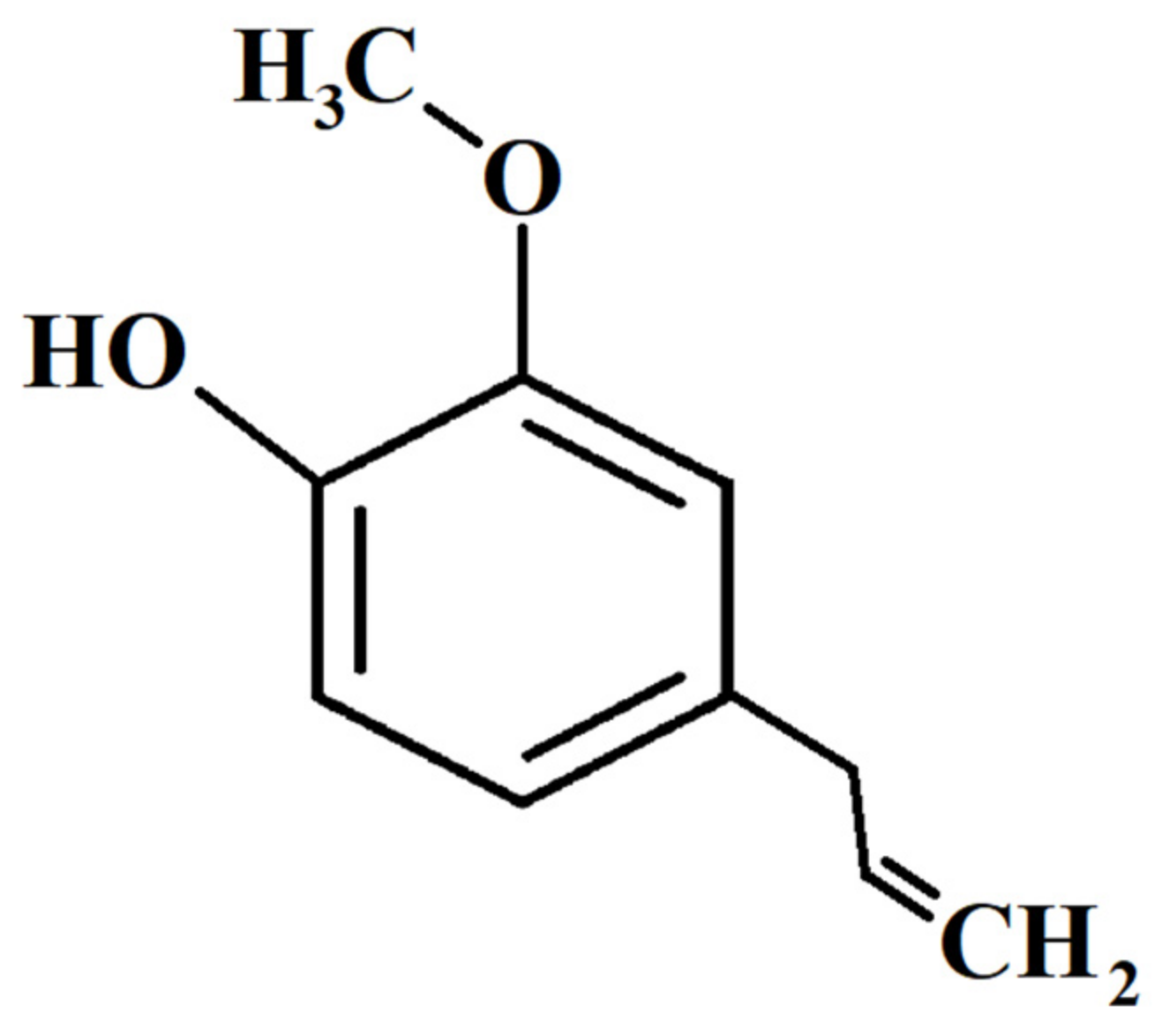
2. Characteristics of Eugenol
3. Antioxidant Activity
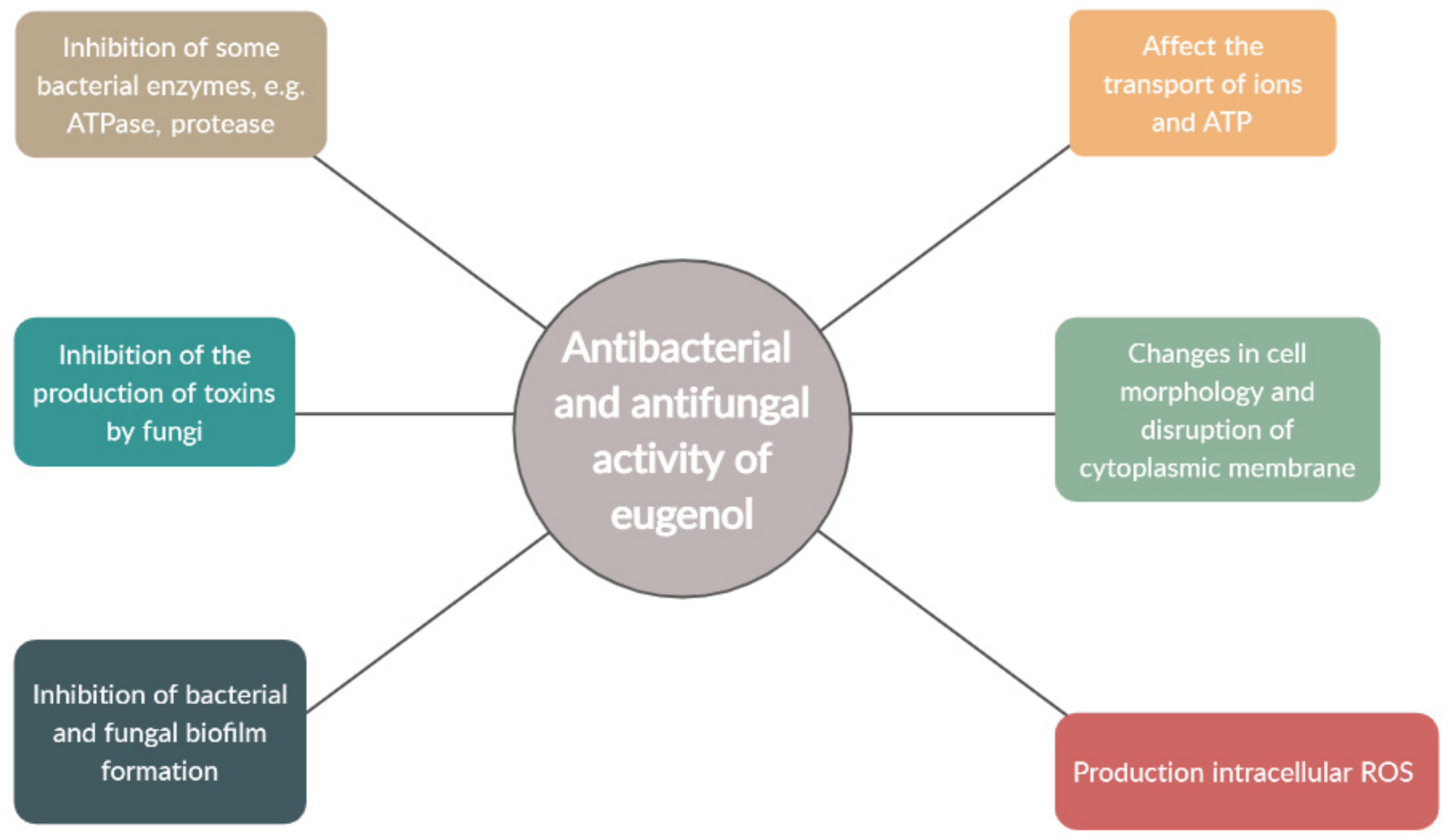
4. Antimicrobial Activity of Eugenol
5. Anticancer Activity of Eugenol
6. Anti-Inflammatory and Analgesic Activities of Eugenol
7. Other Properties
8. Eugenol Derivatives
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| A549 | lung cancer adenocarcinoma cells |
| ALDH | aldehyde dehydrogenases |
| CCl4 | carbon tetrachloride |
| COX-2 | cyclooxygenase-2 |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| Eos | essential oils |
| FAO | Food and Agriculture Organization |
| GBS | group B streptococci- strain |
| HeLa | human immortal cell line from cervical cancer |
| HL-60 | human promyelocytic leukemia cells |
| HSV | herpes simplex virus |
| H2O2 | hydrogen peroxide |
| IC50 | the half maximal inhibitory concentration |
| IL | interleukin |
| iNOS | inducible oxide nitrate synthase |
| MAO | monoamine oxidase |
| MIC | the minimum inhibitory concentration |
| MMP | matrix metalloproteinase |
| MRC-5 | human embryonic lung fibroblast |
| NF-κB | nuclear factor kappa B |
| NSAIDs | nonsteroidal anti-inflammatory drugs |
| PGE2 | prostaglandin E2 |
| ROS | reactive oxygen species |
| TNF-α | tumor necrosis factor alpha |
| VGSC | voltage-gated sodium channels |
| WHO | World Health Organization |
| 5-LOX | 5-lipoxygenase and fungicides |
References
- Tammannavar, P.; Pushpalatha, C.; Jain, S.; Sowmya, S.V. An unexpected positive hypersensitive reaction to eugenol. Case Rep. 2013, 2013, bcr2013009464. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Barbieri, R.; Coppo, E.; Orhan, I.E.; Daglia, M.; Nabavi, S.F.; Izadi, M.; Abdollahi, M.; Nabavi, S.M.; Ajami, M. Antimicrobial activity of eugenol and essential oils containing eugenol: A mechanistic viewpoint. Crit. Rev. Microbiol. 2017, 43, 668–689. [Google Scholar] [CrossRef]
- Fangjun, L.; Zhijia, Y. Tumor suppressive roles of eugenol in human lung cancer cells. Thorac. Cancer 2018, 9, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Batiha, G.E.; Alkazmi, L.M.; Wasef, L.G.; Beshbishy, A.M.; Nadwa, E.H.; Rashwan, E.K. Syzygium aromaticum L. (Myrtaceae): Traditional Uses, Bioactive Chemical Constituents, Pharmacological and Toxicological Activities. Biomolecules 2020, 10, 202. [Google Scholar] [CrossRef] [PubMed]
- Daniel, A.N.; Sartoretto, S.M.; Schmidt, G.; Caparroz-Assef, S.M.; Bersani-Amado, C.A.; Cuman, R.K.N. Anti-inflammatory and antinociceptive activities of eugenol essential oil in experimental animal models. Rev. Bras. Farmacogn. 2009, 19, 212–217. [Google Scholar] [CrossRef]
- Pramod, K.; Ansari, S.H.; Ali, J. Eugenol: A Natural Compound with Versatile Pharmacological Actions. Nat. Prod. Commun. 2010, 5, 1999–2006. [Google Scholar] [CrossRef]
- Khalil, A.A.; Rahman, U.; Khan, M.R.; Sahar, A.; Mehmood, T.; Khan, M. Essential oil eugenol: Sources, extraction techniques and nutraceutical perspectives. RSC Adv. 2017, 7, 32669–32681. [Google Scholar] [CrossRef]
- Mohammadi Nejad, S.; Özgüneş, H.; Başaran, N. Pharmacological and Toxicological Properties of Eugenol. Turk. J. Pharm. Sci. 2017, 14, 201–206. [Google Scholar] [CrossRef]
- Mak, K.K.; Kamal, M.B.; Ayuba, S.B.; Sakirolla, R.; Kang, Y.B.; Mohandas, K.; Balijepalli, M.K.; Ahmad, S.H.; Pichika, M.R. A comprehensive review on eugenol’s antimicrobial properties and industry applications: A transformation from ethnomedicine to industry. Phcog. Rev. 2019, 13, 1–9. [Google Scholar]
- Raja, M.R.C.; Srinivasan, V.; Selvaraj, S.; Mahapatra, S.K. Versatile and synergistic potential of eugenol: A review. Pharm. Anal. Acta 2015, 6, 1–6. [Google Scholar]
- Da Silva, F.F.M.; Monte, F.J.Q.; de Lemos, T.L.G.; Garcia do Nascimento, P.G.; de Medeiros Costa, A.K.; de Paiva, L.M.M. Eugenol derivatives: Synthesis, characterization, and evaluation of antibacterial and antioxidant activities. Chem. Cent. J. 2018, 12, 34–42. [Google Scholar] [CrossRef]
- Navarro Triviño, F.J.; Barrales, C.; Ruiz-Villaverde, R. Eugenol allergy mimicking aphtous oral recurrent and burning mouth syndrome. Contact Dermat. 2019, 2019, 1–2. [Google Scholar]
- Kabuto, H.; Tada, M.; Kohno, M. Eugenol [2-Methoxy-4-(2-propenyl)phenol] Prevents 6-HydroxydopamineInduced Dopamine Depression and Lipid Peroxidation Inductivity in Mouse Striatum. Biol. Pharm. Bull. 2007, 30, 423–427. [Google Scholar] [CrossRef]
- Pavithra, B. Eugenol—A Review. J. Pharm. Sci. Res. 2014, 6, 153–154. [Google Scholar]
- Gülçin, I. Antioxidant activity of eugenol: A structure-activity relationship study. J. Med. Food. 2011, 14, 975–985. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Rosés, R.; Risco, E.; Vila, R.; Peñalver, P.; Cañigueral, S. Biological and Nonbiological Antioxidant Activity of Some Essential Oils. J. Agric. Food Chem. 2016, 64, 4716–4724. [Google Scholar] [CrossRef]
- Fathy, M.; Fawzy, M.A.; Hintzsche, H.; Nikaido, T.; Dandekar, T.; Othman, E.M. Eugenol Exerts Apoptotic Effect and Modulates the Sensitivity of HeLa Cells to Cisplatin and Radiation. Molecules 2019, 24, 3979. [Google Scholar] [CrossRef] [PubMed]
- Devi, K.P.; Nisha, S.A.; Sakthivel, R.; Pandian, S.K. Eugenol (an essential oil of clove) acts as an antibacterial agent against Salmonella typhi by disrupting the cellular membrane. J. Ethnopharmacol. 2010, 130, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Gill, A.O.; Holley, R.A. Inhibition of membrane bound ATPases of Escherichia coli and Listeria monocytogenes by plant oil aromatics. Int. J. Food Microbiol. 2006, 111, 170–174. [Google Scholar] [CrossRef]
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 3, 1–24. [Google Scholar] [CrossRef]
- Perugini Biasi-Garbin, R.; Saori Otaguiri, E.; Morey, A.T.; Fernandes da Silva, M.; Belotto Morguette, A.E.; Armando Contreras Lancheros, C.; Kian, D.; Perugini, M.R.; Nakazato, G.; Durán, N.; et al. Effect of Eugenol against Streptococcus agalactiae and Synergistic Interaction with Biologically Produced Silver Nanoparticles. Evid. Based Complement. Altern. Med. 2015, 2015, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sharifzadeh, A.; Shokri, H. In vitro synergy of eugenol on the antifungal effects of voriconazole against Candida tropicalis and Candida krusei strains isolated from the genital tract of mares. Equine Vet. J. 2021, 53, 94–101. [Google Scholar] [CrossRef]
- Hemaiswarya, S.; Doble, M. Synergistic interaction of eugenol with antibiotics against Gram negative bacteria. Phytomedicine 2009, 16, 997–1005. [Google Scholar] [CrossRef]
- Olszewska, M.A.; Gędas, A.; Simões, M. The Effects of Eugenol, Trans-Cinnamaldehyde, Citronellol, and Terpineol on Escherichia coli Biofilm Control as Assessed by Culture-Dependent and -Independent Methods. Molecules 2020, 25, 2641. [Google Scholar] [CrossRef]
- Furowicz, A.J.; Boroñ-Kaczmarska, A.; Ferlas, M.; Czernomysy-Furowicz, D.; Pobucewicz, A. Bacterial biofilm as well as other microbiol elements and mechanisms of survival in extreme conditions. Medycyna Wet. 2010, 66, 444–448. [Google Scholar]
- Qian, W.; Sun, Z.; Wang, T.; Yang, M.; Liu, M.; Zhang, J.; Li, Y. Antimicrobial activity of eugenol against carbapenem-resistant Klebsiella pneumoniae and its effect on biofilms. Microb. Pathog. 2019, 139, 103924. [Google Scholar] [CrossRef]
- Yoo, C.B.; Han, K.T.; Cho, K.S.; Ha, J.; Park, H.J.; Nam, J.H.; Kil, U.H.; Lee, K.T. Eugenol isolated from the essential oil of Eugenia caryophyllata induces a reactive oxygen species-mediated apoptosis in HL-60 human promyelocytic leukemia cells. Cancer Lett. 2005, 225, 41–52. [Google Scholar] [CrossRef]
- Islam, S.S.; Al-Sharif, I.; Sultan, A.; Al-Mazrou, A.; Remmal, A.; Aboussekhra, A. Eugenol potentiates cisplatin anti-cancer activity through inhibition of ALDH-positive breast cancer stem cells and the NF-κB signaling pathway. Mol. Carcinog. 2018, 57, 333–346. [Google Scholar] [CrossRef]
- Park, C.K.; Li, H.Y.; Yeon, K.Y.; Jung, S.J.; Choi, S.Y.; Lee, S.J.; Lee, S.; Park, K.; Kim, J.S.; Oh, S.B. Eugenol inhibits sodium currents in dental afferent neurons. J. Dent. Res. 2006, 85, 900–904. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.M.; Lee, K.; Im, S.T.; Go, E.J.; Kim, Y.H.; Park, C.K. Co-Application of Eugenol and QX-314 Elicits the Prolonged Blockade of Voltage-Gated Sodium Channels in Nociceptive Trigeminal Ganglion Neurons. Biomolecules 2020, 10, 1513. [Google Scholar] [CrossRef] [PubMed]
- De Andrade, F.D.C.P.; Mendes, A.N. Computational analysis of eugenol inhibitory activity in lipoxygenase and cyclooxygenase pathways. Sci. Rep. 2020, 10, 16204–16217. [Google Scholar] [CrossRef]
- Pytko-Polończyk, J.; Muszyńska, B. Surowce naturalne w stomatologii. Med. Int. Rev. 2016, 27, 68–75. [Google Scholar]
- Pino-Otín, M.R.; Ballestero, D.; Navarro, E.; González-Coloma, A.; Val, J.; Mainar, A.M. Ecotoxicity of a novel biopesticide from Artemisia absinthium on non-target aquatic organisms. Chemosphere 2019, 216, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, I.A.T.A.; Silva, R.; Silva, A.G.; Milet-Pinheiro, P.; Paiva, P.M.G.; Navarro, D.M.A.F.; Silva, M.V.; Napoleao, T.H.; Correia, M.T.S. Chemical characterization and insecticidal effect against Sitophilus zeamais (maize weevil) of essential oil from Croton rudolphianus leaves. Crop Prot. 2020, 129, 105043. [Google Scholar] [CrossRef]
- Sharma, A.; Shukla, A.; Attri, K.; Kumar, M.; Kumar, P.; Suttee, A.; Singh, G.; Barnwal, R.P.; Singla, N. Global trends in pesticides: A looming threat and viable alternatives. Ecotoxicol. Environ. Saf. 2020, 201, 110812. [Google Scholar] [CrossRef]
- Peres, M.C.; de Souza Costa, G.C.; dos Reis, L.E.L.; da Silva, L.D.; Peixoto, M.F.; Alves, C.C.F.; Forim, M.R.; Quintela, E.D.; Araújo, W.L.; Cazal, C.M. In natura and nanoencapsulated essential oils from Xylopia aromatica reduce oviposition of Bemisia tabaci in Phaseolus vulgaris. J. Pest. Sci. 2020, 93, 807–821. [Google Scholar] [CrossRef]
- Rong, S.; Xu, H.; Li, L.; Chen, R.; Gao, X.; Xu, Z. Antifungal activity of endophytic Bacillus safensis B21 and its potential application as a biopesticide to control rice blast. Pestic. Biochem. Physiol. 2020, 162, 69–77. [Google Scholar] [CrossRef]
- Pavela, R.; Maggi, F.; Iannarelli, R.; Benelli, G. Plant extracts for developing mosquito larvicides: From laboratory to the field, with insights on the modes of action. Acta Trop. 2019, 193, 236–271. [Google Scholar] [CrossRef] [PubMed]
- Matos, L.F.; Barbosa, D.R.S.; Lima, E.C.; Dutra, K.A.; Navarro, D.M.A.F.; Alves, J.L.R.; Silva, G.N. Chemical composition and insecticidal e_ect of essential oils from Illicium verum and Eugenia caryophyllus on Callosobruchus maculatus in cowpea. Ind. Crop. Prod. 2020, 145, 112088. [Google Scholar] [CrossRef]
- Saroj, A.; Oriyomi, O.V.; Nayak, A.K.; Haider, S.Z. Phytochemicals of Plant-Derived Essential Oils: A Novel Green Approach Against Pests. In Natural Remedies for Pest, Disease and Weed Control; Egbuna, C., Sawicka, B., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2020; pp. 65–79. [Google Scholar]
- Lengai, G.M.W.; Muthom, J.W.; Mbega, E.R. Phytochemical activity and role of botanical pesticides in pest management for sustainable agricultural crop production. Sci. Afr. 2020, 7, e00239. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y. Eugenol nanoemulsion stabilized with zein and sodium caseinate by self-assembly. J. Agric. Food Chem. 2017, 65, 2990–2998. [Google Scholar] [CrossRef] [PubMed]
- Ju, J.; Xie, Y.; Yu, H.; Guo, Y.; Cheng, Y.; Qian, H.; Yao, W. Analysis of the synergistic antifungal mechanism of eugenol and citral. LWT Food Sci. Technol. 2020, 123, 109128. [Google Scholar] [CrossRef]
- Novato, T.; Gomes, G.A.; Zeringóta, V.; Franco, C.T.; Oliveira, D.R.; Melo, D.; Carvalho, M.G.; Daemon, E.; Monteiro, C.M.O. In vitro assessment of the acaricidal activity of carvacrol, thymol, eugenol and their acetylated derivatives on Rhipicephalus microplus (Acari: Ixodidae). Vet. Parasitol. 2018, 260, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-H.; Tung, S.-H.; Jeng, R.-J.; Abu-Omar, M.M.; Lin, C.-H. A facile strategy to achieve fully bio-based epoxy thermosets from eugenol. Green Chem. 2019, 21, 4475–4488. [Google Scholar] [CrossRef]
- Fernandes, M.J.G.; Pereira, R.B.; Pereira, D.M.; Fortes, A.G.; Castanheira, E.M.S.; Gonçalves, M.S.T. New Eugenol Derivatives with Enhanced Insecticidal Activity. Int. J. Mol. Sci. 2020, 21, 9257. [Google Scholar] [CrossRef] [PubMed]
- Maximino, S.C.; Dutra, J.A.P.; Rodrigues, R.P.; de Cássia Ribeiro Gonçalves, R.; Morais, P.A.B.; Ventura, J.A.; Schuenck, R.P.; Júnior, V.L.; Kitagwa, R.R.; de Souza Borges, W. Synthesis of Eugenol Derivatives and Evaluation of their Antifungal Activity Against Fusarium solani f. sp. piperis. Curr. Pharm. Des. 2020, 26, 1532–1542. [Google Scholar] [CrossRef]
- Zore, G.B.; Thakre, A.D.; Jadhav, S.; Karuppayil, S.M.; Candida, A. Terpenoids inhibit Candida albicans growth by affecting membrane integrity and arrest of cell cycle. Phytomedicine 2011, 18, 1181–1190. [Google Scholar] [CrossRef]
- Pavela, R. Efficacy of naphthoquinones as insecticides against the house fly, Musca Domestica L. Ind. Crop. Prod. 2013, 43, 745–750. [Google Scholar] [CrossRef]
- Sánchez-Calvo, J.M.; Barbero, G.R.; Guerrero-Vásquez, G.; Durán, A.; Macías, M.; Rodríguez-Iglesias, M.; Molinillo, J.M.G.; Macias, F. Synthesis, antibacterial and antifungal activities of naphthoquinone derivatives: A structure–activity relationship study. Med. Chem. Res. 2016, 25, 1274–1285. [Google Scholar] [CrossRef]
- Ma, Y.; Xu, F.; Chen, C.; Li, Q.; Wang, M. The beneficial use of essential oils from buds and fruit of Syzygium aromaticum to combat pathogenic fungi of Panax notoginseng. Ind. Crop. Prod. 2019, 133, 185–192. [Google Scholar] [CrossRef]
- Olea, A.F.; Bravo, A.; Martínez, R.; Thomas, M.; Sedan, C.; Espinoza, L.; Zambrano, E.; Carvajal, D.; Silva-Moreno, E.; Carrasco, H. Antifungal Activity of Eugenol Derivatives against Botrytis Cinerea. Molecules 2019, 24, 1239. [Google Scholar] [CrossRef]
- Romanazzi, G.; Droby, S. Control Strategies for Postharvest Grey Mould on Fruit Crops. In Botrytis—The Fungus, the Pathogen and Its Management in Agricultural Systems; Fillinger, S., Elad, Y., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 217–228. [Google Scholar]

| Plant | Part | Concentration (mg/g) |
|---|---|---|
| Clove, Clovetree | Flower Leaf, stem | 1809 |
| Clover pepper | Fruit | 36 |
| Betel Pepper | Leaf | 17.85 |
| Carrot | Seed | 7 |
| Tulsi | Leaf | 4.2–4.97 |
| Ceylon Cinnamon, Cinnamon | Bark | 3.52 |
| Turmeric | Leaf, essential oil | 2.1 |
| Bay, Bay Laurel | Leaf | 1.34 |
| Chinese Ginger | Rhizome | 0.4 |
| Nutmeg | Seed | 0.32 |
| Small-Flowered Oregano | Shoot | 0.055–0.125 |
| Strain | Application | Activity |
|---|---|---|
| Aspergillus ochraceus | Grains | Inhibition Ochratoxin A synthesis |
| Phlyctena vagabunda, Penicillium expansum, Botrytis cinerea, Monilinia fructigena, | Apples | Fungicidal |
| Sclerotinia sclerotiorum, Rhizopus stolonifer, Mucor spp. | Peaches | Fungicidal |
| Saccharomyces bayanus, Rhodotorula bacarum, Pichia membranifaciens | Fruit juice | Fungicidal |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ulanowska, M.; Olas, B. Biological Properties and Prospects for the Application of Eugenol—A Review. Int. J. Mol. Sci. 2021, 22, 3671. https://doi.org/10.3390/ijms22073671
Ulanowska M, Olas B. Biological Properties and Prospects for the Application of Eugenol—A Review. International Journal of Molecular Sciences. 2021; 22(7):3671. https://doi.org/10.3390/ijms22073671
Chicago/Turabian StyleUlanowska, Magdalena, and Beata Olas. 2021. "Biological Properties and Prospects for the Application of Eugenol—A Review" International Journal of Molecular Sciences 22, no. 7: 3671. https://doi.org/10.3390/ijms22073671
APA StyleUlanowska, M., & Olas, B. (2021). Biological Properties and Prospects for the Application of Eugenol—A Review. International Journal of Molecular Sciences, 22(7), 3671. https://doi.org/10.3390/ijms22073671