Molecular Basis of AmpC β-Lactamase Induction by Avibactam in Pseudomonas aeruginosa: PBP Occupancy, Live Cell Binding Dynamics and Impact on Resistant Clinical Isolates Harboring PDC-X Variants
Abstract
1. Introduction
2. Results and Discussion
2.1. Molecular Basis of Avibactam AmpC Induction
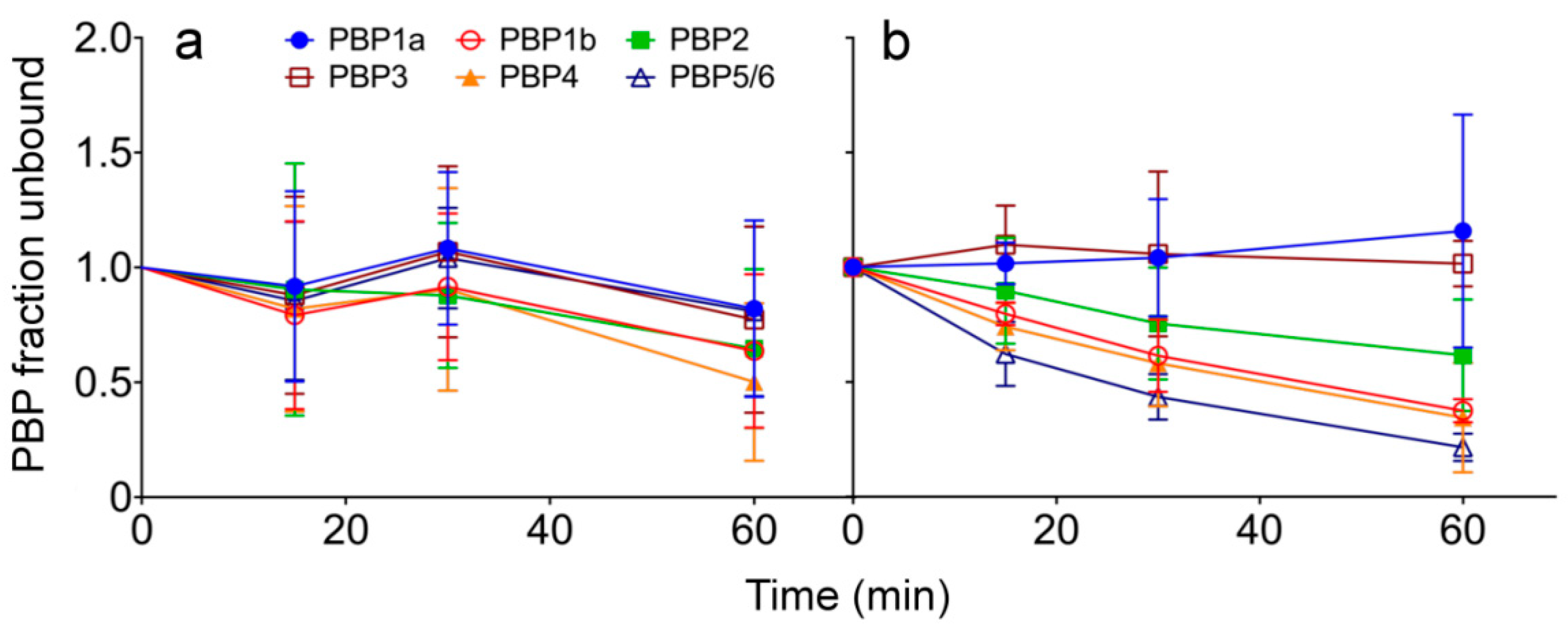
2.2. Avibactam Target Binding in Live P. aeruginosa Cells
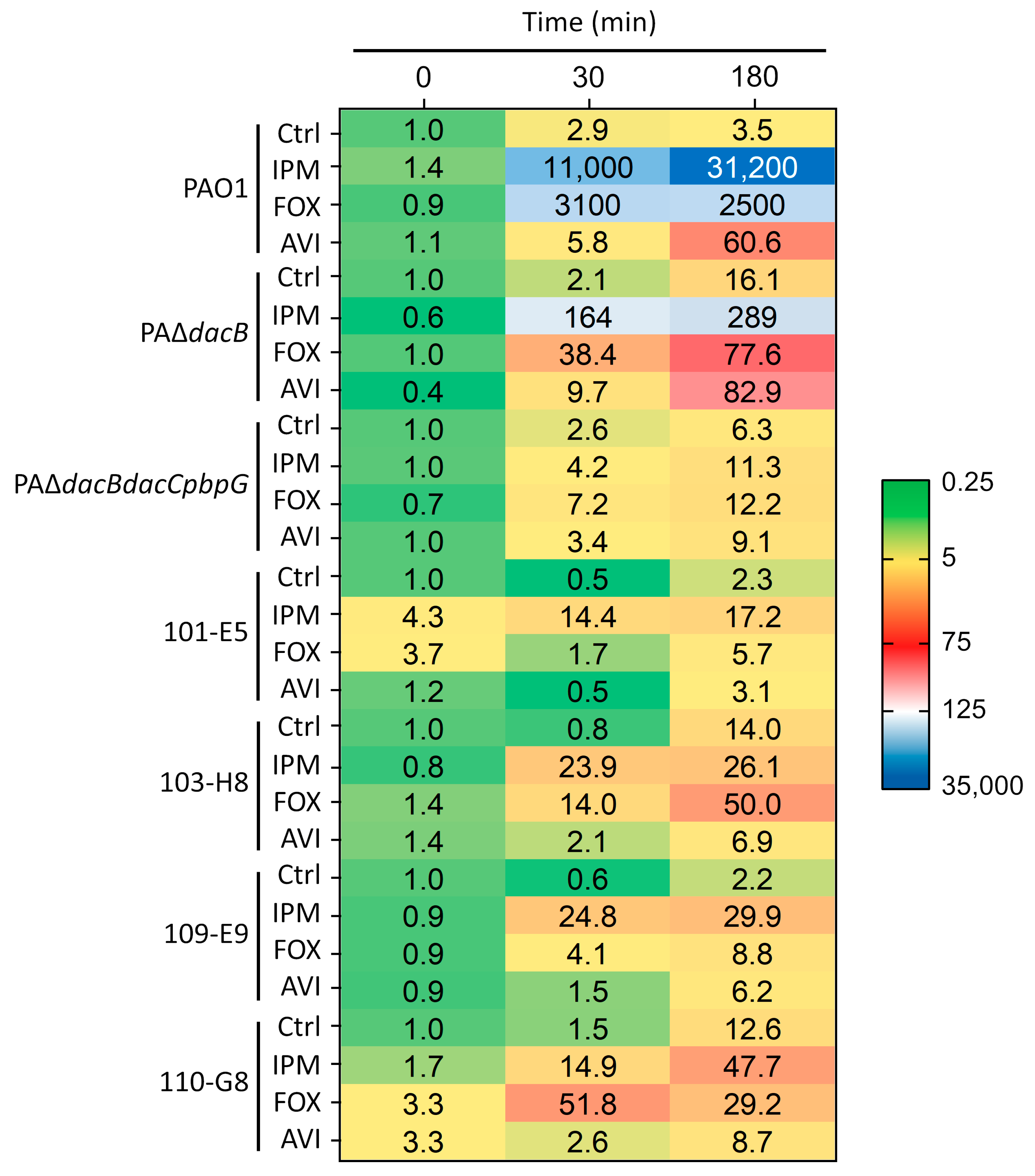
2.3. qRT-PCR-Based ampC Induction Assays
3. Methods
3.1. Bacterial Strains and Antibiotics
3.2. Intact Cells’ Time-Course of PBP-Binding Assay
3.3. Determination of PBP-Binding Affinity (IC50)
3.4. qRT-PCR Quantification of AmpC Induction
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Munoz-Price, L.S.; Poirel, L.; Bonomo, R.A.; Schwaber, M.J.; Daikos, G.L.; Cormican, M.; Cornaglia, G.; Garau, J.; Gniadkowski, M.; Hayden, M.K.; et al. Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect. Dis. 2013, 13, 785–796. [Google Scholar] [CrossRef]
- Jiao, Y.; Moya, B.; Chen, M.J.; Zavascki, A.P.; Tsai, H.; Tao, X.; Sutaria, D.S.; Louie, A.; Boyce, J.D.; Deveson Lucas, D.; et al. Comparable Efficacy and Better Safety of Double β-Lactam Combination Therapy versus β-Lactam plus Aminoglycoside in Gram-Negative Bacteria in Randomized, Controlled Trials. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Juan, C.; Maciá, M.D.; Gutiérrez, O.; Vidal, C.; Pérez, J.L.; Oliver, A. Molecular mechanisms of beta-lactam resistance mediated by AmpC hyperproduction in Pseudomonas aeruginosa clinical strains. Antimicrob. Agents Chemother. 2005, 49, 4733–4738. [Google Scholar] [CrossRef] [PubMed]
- Sanders, C.C.; Bradford, P.A.; Ehrhardt, A.F.; Bush, K.; Young, K.D.; Henderson, T.A.; Sanders, W.E. Penicillin-binding proteins and induction of AmpC beta-lactamase. Antimicrob. Agents Chemother. 1997, 41, 2013–2015. [Google Scholar] [CrossRef]
- Torrens, G.; Hernández, S.B.; Ayala, J.A.; Moya, B.; Juan, C.; Cava, F.; Oliver, A. Regulation of AmpC-Driven β-Lactam Resistance in Pseudomonas aeruginosa: Different Pathways, Different Signaling. mSystems 2019, 4. [Google Scholar] [CrossRef]
- Moya, B.; Dotsch, A.; Juan, C.; Blazquez, J.; Zamorano, L.; Haussler, S.; Oliver, A. Beta-lactam resistance response triggered by inactivation of a nonessential penicillin-binding protein. PLoS Pathog. 2009, 5, e1000353. [Google Scholar] [CrossRef]
- Bhowmick, T.; Weinstein, M.P. Microbiology of Meropenem-Vaborbactam: A Novel Carbapenem Beta-Lactamase Inhibitor Combination for Carbapenem-Resistant Enterobacterales Infections. Infect. Dis. Ther. 2020, 9, 757–767. [Google Scholar] [CrossRef]
- Moya, B.; Bhagwat, S.; Cabot, G.; Bou, G.; Patel, M.; Oliver, A. Effective inhibition of PBPs by cefepime and zidebactam in the presence of VIM-1 drives potent bactericidal activity against MBL-expressing Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2020, 75, 1474–1478. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Estabrook, M.; Jacoby, G.A.; Nichols, W.W.; Testa, R.T.; Bush, K. In vitro susceptibility of characterized β-lactamase-producing strains tested with avibactam combinations. Antimicrob. Agents Chemother. 2015, 59, 1789–1793. [Google Scholar] [CrossRef] [PubMed]
- Moya, B.; Zamorano, L.; Juan, C.; Perez, J.L.; Ge, Y.; Oliver, A. Activity of a new cephalosporin, CXA-101 (FR264205), against beta-lactam-resistant Pseudomonas aeruginosa mutants selected in vitro and after antipseudomonal treatment of intensive care unit patients. Antimicrob. Agents Chemother. 2010, 54, 1213–1217. [Google Scholar] [CrossRef] [PubMed]
- Moya, B.; Zamorano, L.; Juan, C.; Ge, Y.; Oliver, A. Affinity of the new cephalosporin CXA-101 to penicillin-binding proteins of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2010, 54, 3933–3937. [Google Scholar] [CrossRef]
- Moya, B.; Barcelo, I.M.; Bhagwat, S.; Patel, M.; Bou, G.; Papp-Wallace, K.M.; Bonomo, R.A.; Oliver, A. WCK 5107 (Zidebactam) and WCK 5153 Are Novel Inhibitors of PBP2 Showing Potent “β-Lactam Enhancer” Activity against Pseudomonas aeruginosa, Including Multidrug-Resistant Metallo-β-Lactamase-Producing High-Risk Clones. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef]
- Papp-Wallace, K.M.; Nguyen, N.Q.; Jacobs, M.R.; Bethel, C.R.; Barnes, M.D.; Kumar, V.; Bajaksouzian, S.; Rudin, S.D.; Rather, P.N.; Bhavsar, S.; et al. Strategic Approaches to Overcome Resistance against Gram-Negative Pathogens Using β-Lactamase Inhibitors and β-Lactam Enhancers: Activity of Three Novel Diazabicyclooctanes WCK 5153, Zidebactam (WCK 5107), and WCK 4234. J. Med. Chem 2018, 61, 4067–4086. [Google Scholar] [CrossRef]
- MacVane, S.H.; Pandey, R.; Steed, L.L.; Kreiswirth, B.N.; Chen, L. Emergence of Ceftolozane-Tazobactam-Resistant Pseudomonas aeruginosa during Treatment Is Mediated by a Single AmpC Structural Mutation. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Fraile-Ribot, P.A.; Cabot, G.; Mulet, X.; Periañez, L.; Martín-Pena, M.L.; Juan, C.; Pérez, J.L.; Oliver, A. Mechanisms leading to in vivo ceftolozane/tazobactam resistance development during the treatment of infections caused by MDR Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2018, 73, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Haidar, G.; Philips, N.J.; Shields, R.K.; Snyder, D.; Cheng, S.; Potoski, B.A.; Doi, Y.; Hao, B.; Press, E.G.; Cooper, V.S.; et al. Ceftolozane-Tazobactam for the Treatment of Multidrug-Resistant Pseudomonas aeruginosa Infections: Clinical Effectiveness and Evolution of Resistance. Clin. Infect. Dis. 2017, 65, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Slater, C.L.; Winogrodzki, J.; Fraile-Ribot, P.A.; Oliver, A.; Khajehpour, M.; Mark, B.L. Adding Insult to Injury: Mechanistic Basis for How AmpC Mutations Allow Pseudomonas aeruginosa To Accelerate Cephalosporin Hydrolysis and Evade Avibactam. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Lister, P.D.; Gardner, V.M.; Sanders, C.C. Clavulanate induces expression of the Pseudomonas aeruginosa AmpC cephalosporinase at physiologically relevant concentrations and antagonizes the antibacterial activity of ticarcillin. Antimicrob. Agents Chemother. 1999, 43, 882–889. [Google Scholar] [CrossRef]
- Weber, D.A.; Sanders, C.C. Diverse potential of beta-lactamase inhibitors to induce class I enzymes. Antimicrob. Agents Chemother. 1990, 34, 156–158. [Google Scholar] [CrossRef] [PubMed]
- Livermore, D.M.; Jamrozy, D.; Mushtaq, S.; Nichols, W.W.; Young, K.; Woodford, N. AmpC β-lactamase induction by avibactam and relebactam. J. Antimicrob. Chemother. 2017, 72, 3342–3348. [Google Scholar] [CrossRef]
- Tarral, A.; Merdjan, H. Effect of age and sex on the pharmacokinetics and safety of avibactam in healthy volunteers. Clin. Ther. 2015, 37, 877–886. [Google Scholar] [CrossRef]
- Ropy, A.; Cabot, G.; Sanchez-Diener, I.; Aguilera, C.; Moya, B.; Ayala, J.A.; Oliver, A. Role of Pseudomonas aeruginosa low-molecular-mass penicillin-binding proteins in AmpC expression, beta-lactam resistance, and peptidoglycan structure. Antimicrob. Agents Chemother. 2015, 59, 3925–3934. [Google Scholar] [CrossRef] [PubMed]
- Asli, A.; Brouillette, E.; Krause, K.M.; Nichols, W.W.; Malouin, F. Distinctive Binding of Avibactam to Penicillin-Binding Proteins of Gram-Negative and Gram-Positive Bacteria. Antimicrob. Agents Chemother. 2016, 60, 752–756. [Google Scholar] [CrossRef]
- Kocaoglu, O.; Carlson, E.E. Profiling of beta-lactam selectivity for penicillin-binding proteins in Escherichia coli strain DC2. Antimicrob. Agents Chemother. 2015, 59, 2785–2790. [Google Scholar] [CrossRef]
- Pagès, J.M.; Peslier, S.; Keating, T.A.; Lavigne, J.P.; Nichols, W.W. Role of the Outer Membrane and Porins in Susceptibility of β-Lactamase-Producing Enterobacteriaceae to Ceftazidime-Avibactam. Antimicrob. Agents Chemother. 2015, 60, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Miossec, C.; Claudon, M.; Levasseur, P.; Black, M.T. The β-lactamase inhibitor avibactam (NXL104) does not induce ampC β-lactamase in Enterobacter cloacae. Infect. Drug Resist. 2013, 6, 235–240. [Google Scholar] [CrossRef][Green Version]
- Cabot, G.; Ocampo-Sosa, A.A.; Domínguez, M.A.; Gago, J.F.; Juan, C.; Tubau, F.; Rodríguez, C.; Moyà, B.; Peña, C.; Martínez-Martínez, L.; et al. Genetic markers of widespread extensively drug-resistant Pseudomonas aeruginosa high-risk clones. Antimicrob. Agents Chemother. 2012, 56, 6349–6357. [Google Scholar] [CrossRef] [PubMed]
- Cabot, G.; López-Causapé, C.; Ocampo-Sosa, A.A.; Sommer, L.M.; Domínguez, M.; Zamorano, L.; Juan, C.; Tubau, F.; Rodríguez, C.; Moyà, B.; et al. Deciphering the Resistome of the Widespread Pseudomonas aeruginosa Sequence Type 175 International High-Risk Clone through Whole-Genome Sequencing. Antimicrob. Agents Chemother. 2016, 60, 7415–7423. [Google Scholar] [PubMed]
- Tam, V.H.; Chang, K.T.; Zhou, J.; Ledesma, K.R.; Phe, K.; Gao, S.; Van Bambeke, F.; Sánchez-Díaz, A.M.; Zamorano, L.; Oliver, A.; et al. Determining β-lactam exposure threshold to suppress resistance development in Gram-negative bacteria. J. Antimicrob. Chemother. 2017, 72, 1421–1428. [Google Scholar] [CrossRef]
- Fraile-Ribot, P.A.; Zamorano, L.; Orellana, R.; Del Barrio-Tofiño, E.; Sánchez-Diener, I.; Cortes-Lara, S.; López-Causapé, C.; Cabot, G.; Bou, G.; Martínez-Martínez, L.; et al. Activity of Imipenem-Relebactam against a Large Collection of Pseudomonas aeruginosa Clinical Isolates and Isogenic β-Lactam-Resistant Mutants. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Stover, C.K.; Pham, X.Q.; Erwin, A.L.; Mizoguchi, S.D.; Warrener, P.; Hickey, M.J.; Brinkman, F.S.; Hufnagle, W.O.; Kowalik, D.J.; Lagrou, M.; et al. Complete genome sequence of Pseudomonas aeruginosa PAO1, an opportunistic pathogen. Nature 2000, 406, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Meier, T.I.; Kahl, S.D.; Gee, K.R.; Blaszczak, L.C. BOCILLIN FL, a sensitive and commercially available reagent for detection of penicillin-binding proteins. Antimicrob. Agents Chemother. 1999, 43, 1124–1128. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
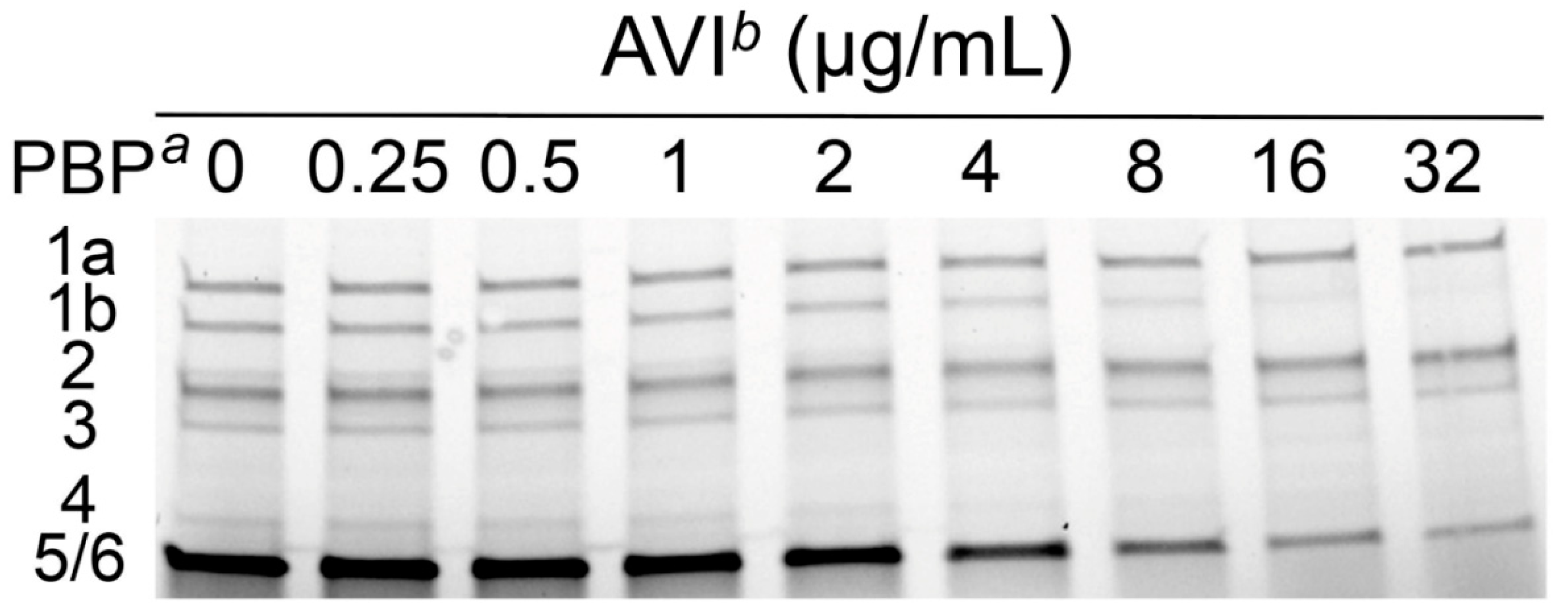
| P. aeruginosa PAO1 PBP a | Mean IC50 ± SD (µg/mL) b | ||
|---|---|---|---|
| Imipenem | Cefoxitin | Avibactam | |
| 1a | 0.2 ± 0.06 | 7.8 ± 0.5 | >32 |
| 1b | 0.1 ± 0.03 | >32 | 3.6 ± 0.1 |
| 2 | 0.05 ± 0.02 | 14.6 ± 2.2 | 4.2 ± 1.6 |
| 3 | 0.3 ± 0.07 | 10.7 ± 1.7 | >32 |
| 4 | 0.1 ± 0.04 | 1.5 ± 0.5 | 3.1 ± 1.3 |
| 5/6 | 0.5 ± 0.2 | 9 ± 2.4 | 2.2 ± 0.7 |
| P. aeruginosa Strain a | ampC Expression (min) b | ||
|---|---|---|---|
| 0 | 30 | 180 | |
| PAO1 | 1 | 2.6 ± 0.5 | 5.8 ± 0.1 |
| PAΔdacB | 110 ± 0.8 | 228 ± 117 | 1100 ± 243 |
| PAΔdacBdacCpbpG | 7500 ± 3800 | 27,000 ± 9400 | 58,000 ± 26,000 |
| 101-E5 | 554 ± 273 | 466 ± 272 | 2400 ± 1100 |
| 103-H8 | 391 ± 345 | 317 ± 21.2 | 5400 ± 2900 |
| 109-E9 | 414 ± 122 | 496 ± 213 | 1700 ± 382 |
| 110-G8 | 306 ± 31.2 | 418 ± 183 | 3500 ± 1600 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Argüello, S.; Montaner, M.; Oliver, A.; Moya, B. Molecular Basis of AmpC β-Lactamase Induction by Avibactam in Pseudomonas aeruginosa: PBP Occupancy, Live Cell Binding Dynamics and Impact on Resistant Clinical Isolates Harboring PDC-X Variants. Int. J. Mol. Sci. 2021, 22, 3051. https://doi.org/10.3390/ijms22063051
López-Argüello S, Montaner M, Oliver A, Moya B. Molecular Basis of AmpC β-Lactamase Induction by Avibactam in Pseudomonas aeruginosa: PBP Occupancy, Live Cell Binding Dynamics and Impact on Resistant Clinical Isolates Harboring PDC-X Variants. International Journal of Molecular Sciences. 2021; 22(6):3051. https://doi.org/10.3390/ijms22063051
Chicago/Turabian StyleLópez-Argüello, Silvia, María Montaner, Antonio Oliver, and Bartolome Moya. 2021. "Molecular Basis of AmpC β-Lactamase Induction by Avibactam in Pseudomonas aeruginosa: PBP Occupancy, Live Cell Binding Dynamics and Impact on Resistant Clinical Isolates Harboring PDC-X Variants" International Journal of Molecular Sciences 22, no. 6: 3051. https://doi.org/10.3390/ijms22063051
APA StyleLópez-Argüello, S., Montaner, M., Oliver, A., & Moya, B. (2021). Molecular Basis of AmpC β-Lactamase Induction by Avibactam in Pseudomonas aeruginosa: PBP Occupancy, Live Cell Binding Dynamics and Impact on Resistant Clinical Isolates Harboring PDC-X Variants. International Journal of Molecular Sciences, 22(6), 3051. https://doi.org/10.3390/ijms22063051







