Skp2 and Slug Are Coexpressed in Aggressive Prostate Cancer and Inhibited by Neddylation Blockade
Abstract
1. Introduction
2. Results
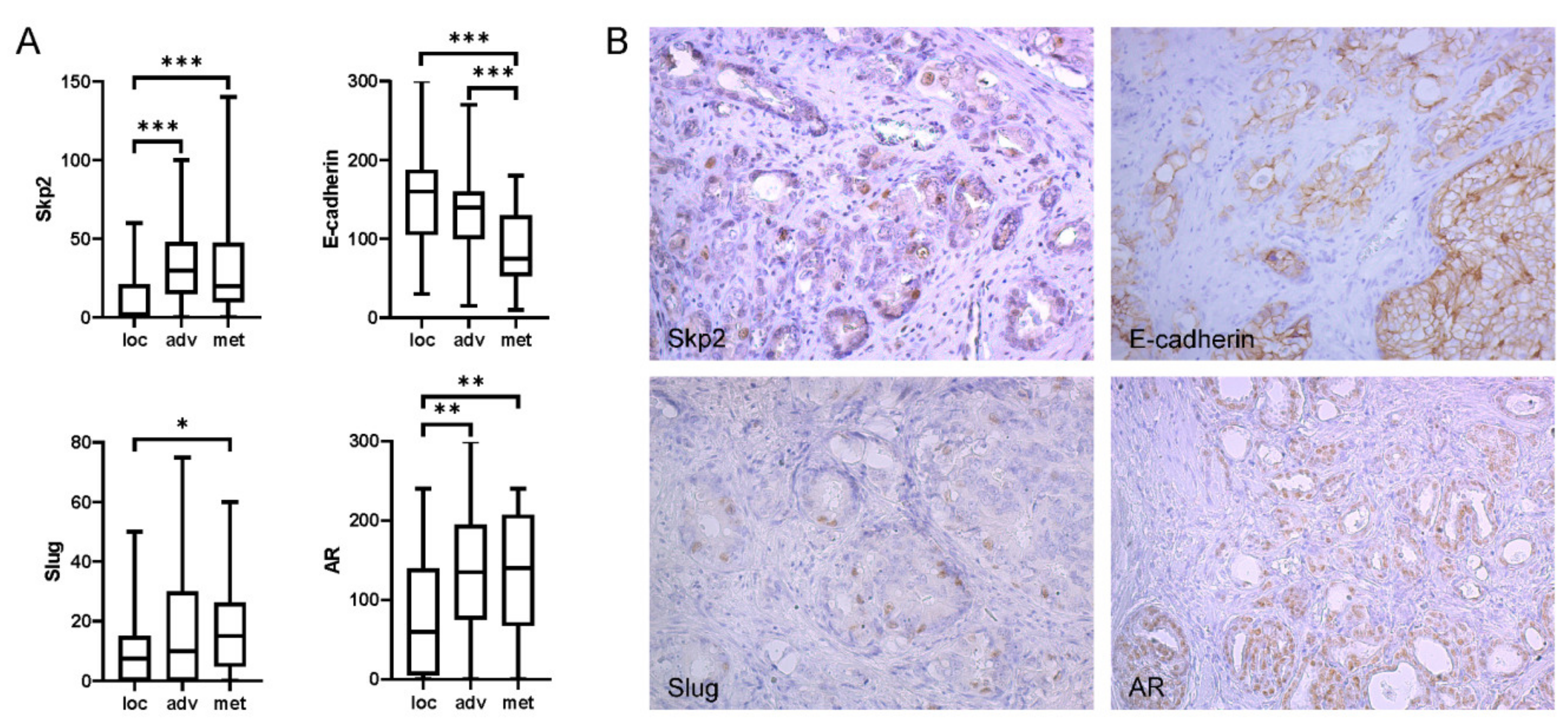
2.1. Expression of Skp2 Correlated with Slug in Patients with High Gleason Score and Metastasis to Lymph Nodes
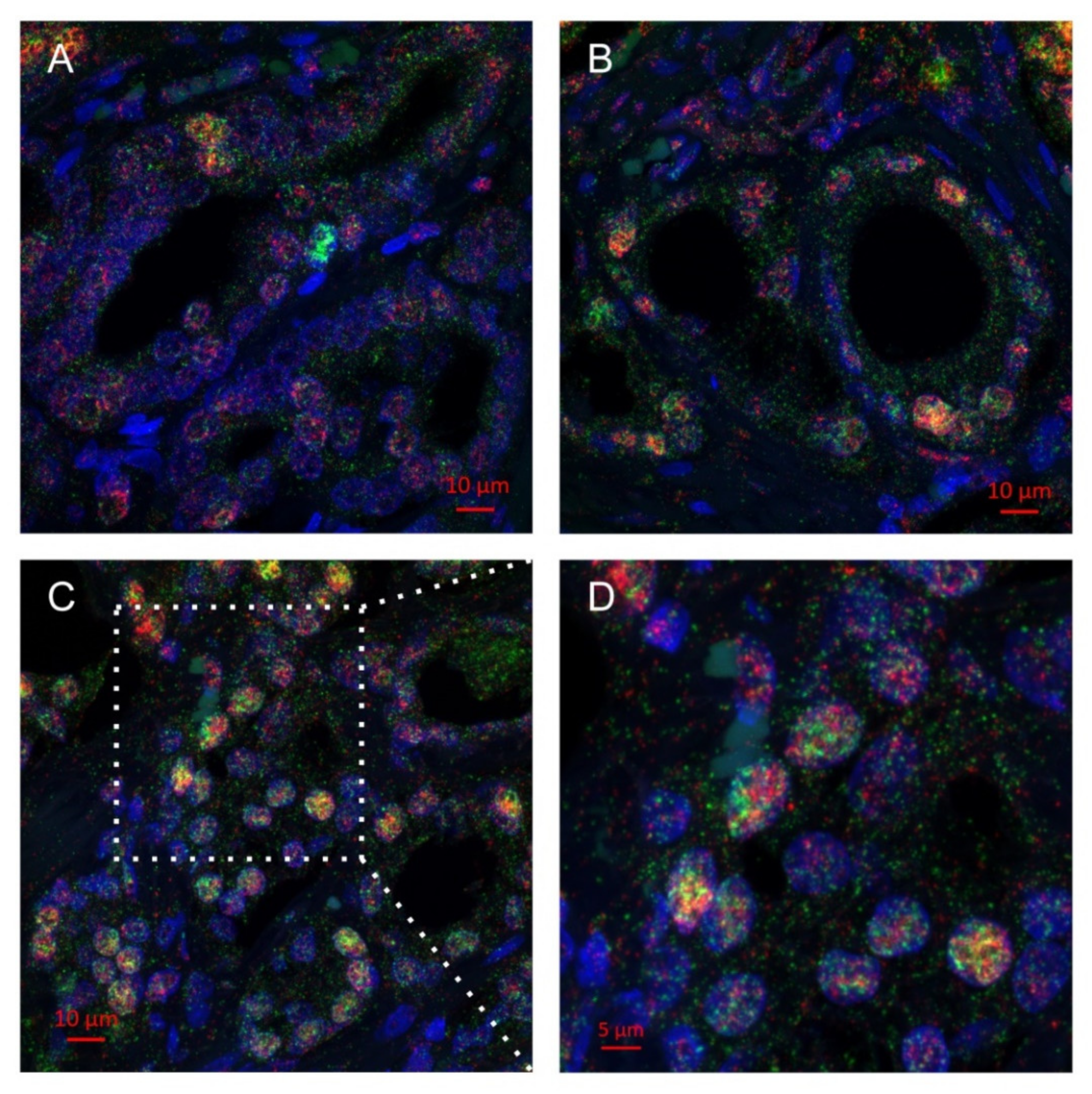
2.2. Multiplex Immunohistochemistry Revealed Colocalization of Skp2 and Slug in Prostate Cancer Tissue
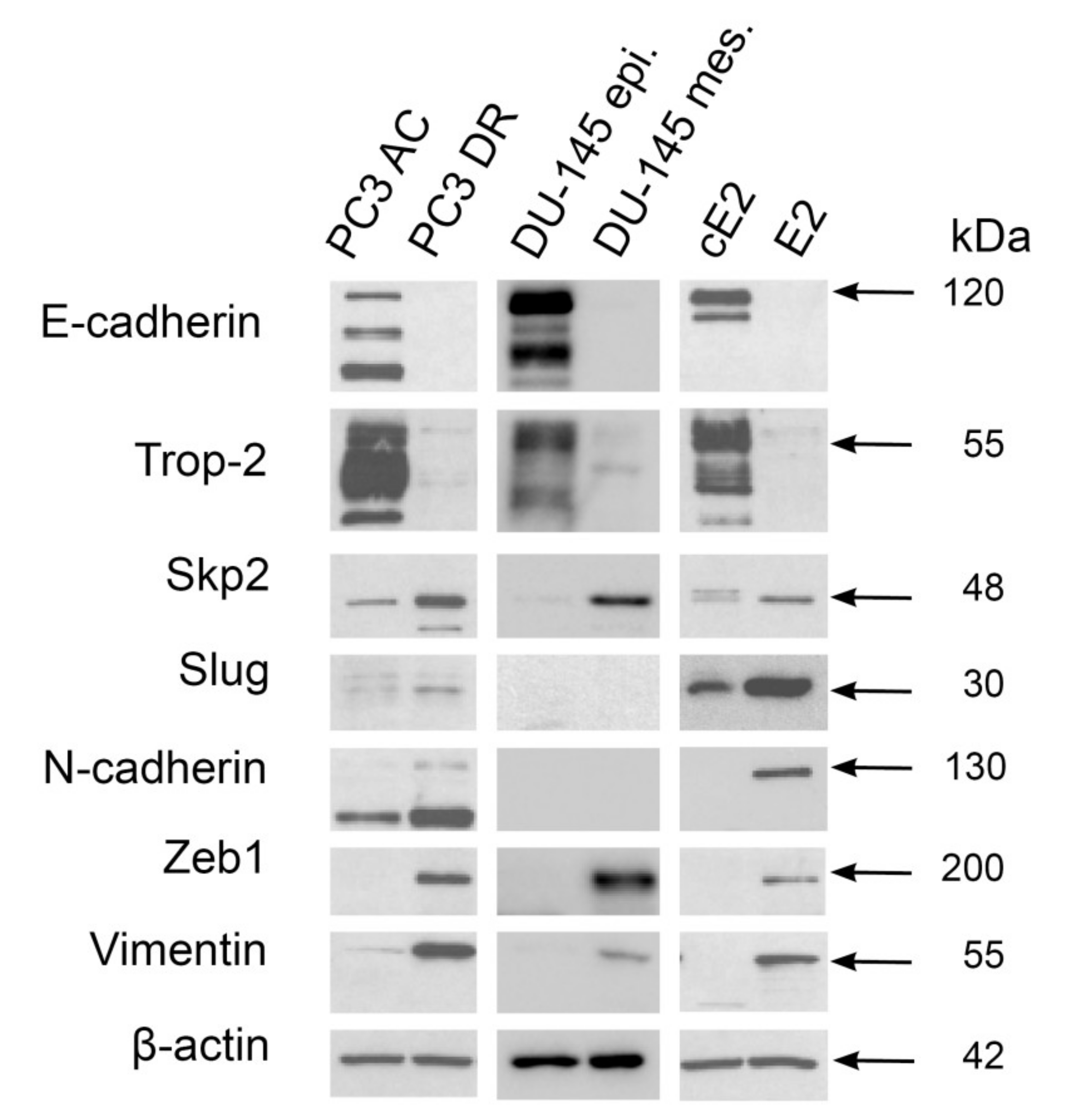
2.3. Mesenchymal Phenotype Is Associated with Enhanced Expression of Both Skp2 and Slug in Human PC3 and Mouse E2 Prostate Cancer Cell Lines
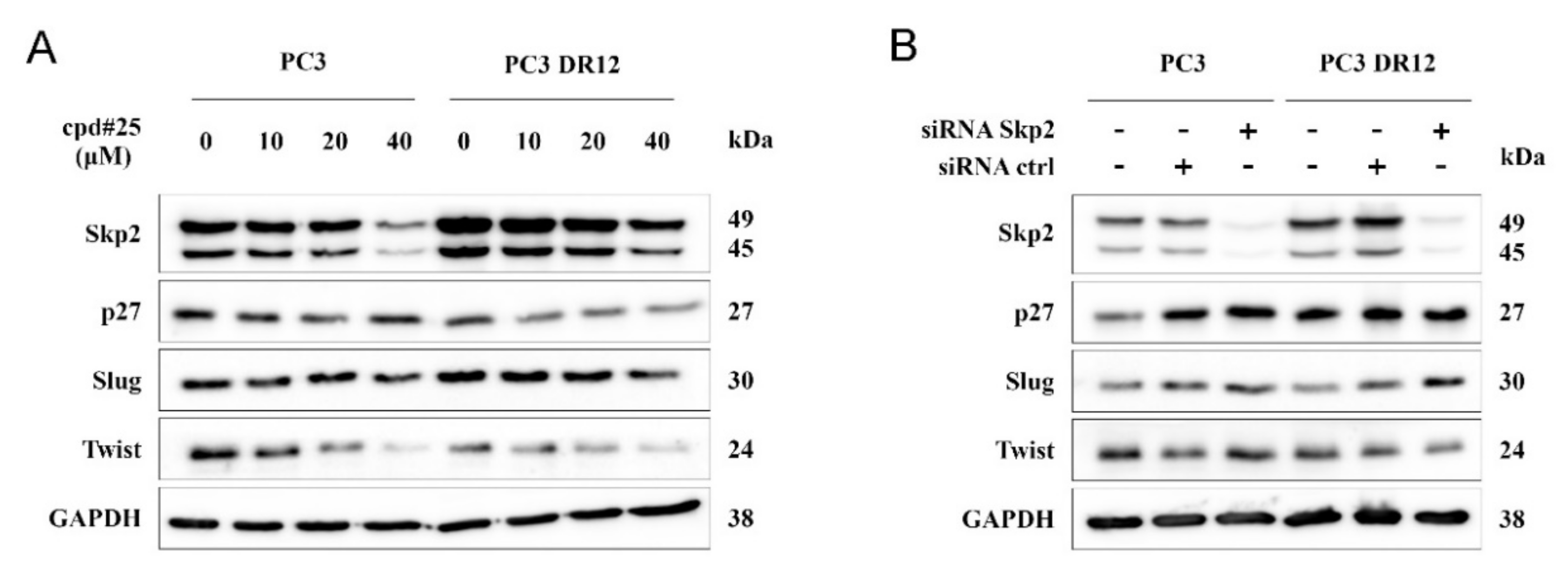
2.4. Chemical Inhibition of Skp2, but Not RNA Interference, Modestly Decreased Slug Protein Levels
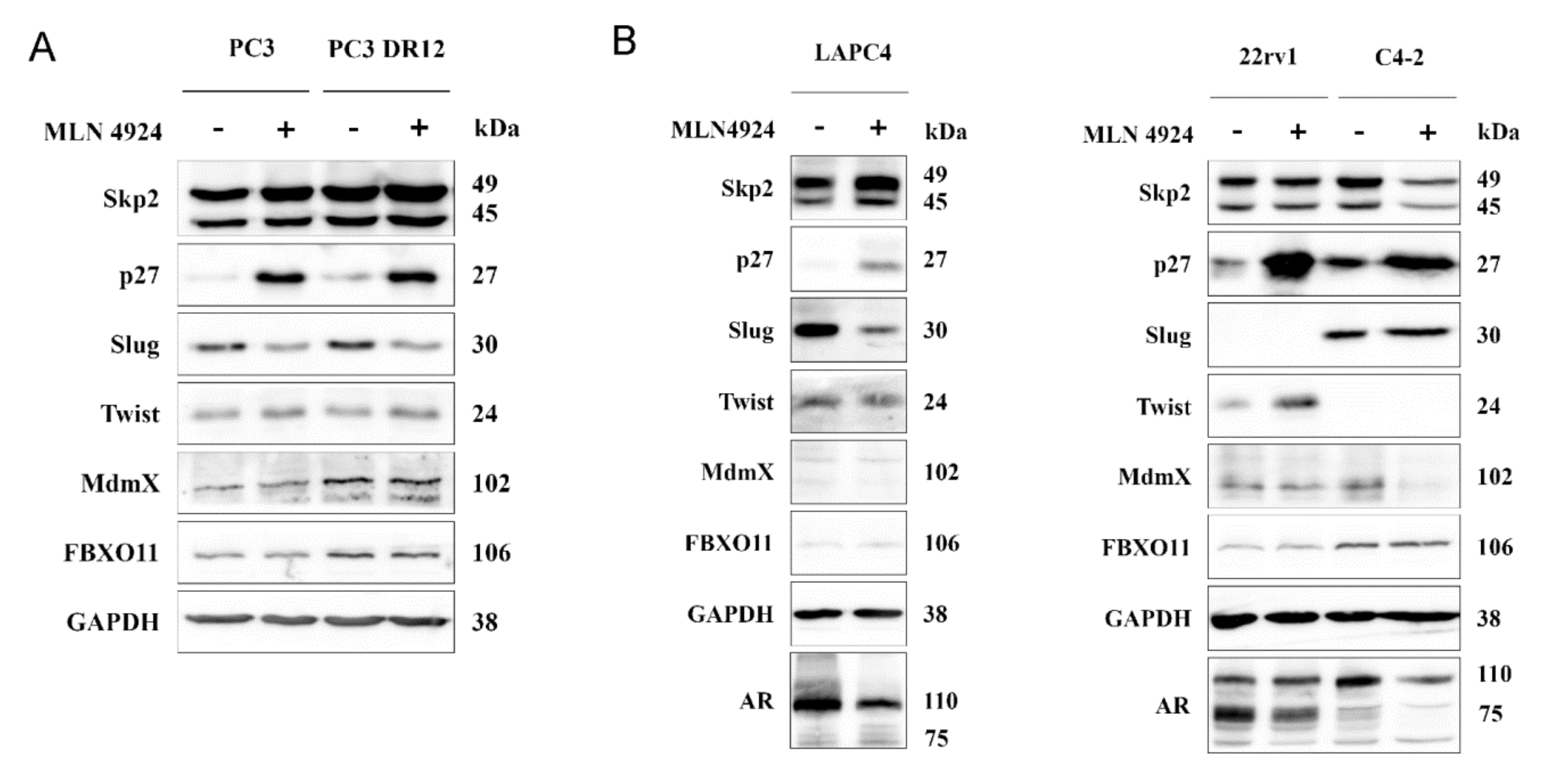
2.5. Neddylation Blockade Downregulated Slug Expression in PC3 and LAPC4 Cells
3. Discussion
4. Materials and Methods
4.1. Patients and Standard Immunohistochemistry
4.2. Multiplex Immunohistochemistry
4.3. Cell Culture
4.4. Western Blot Analysis and Immunodetection
4.5. Chemical Inhibitors
4.6. RNA Interference
4.7. Cell Fractionation
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Weinberg, R.A. Epithelial-to-mesenchymal transition in cancer: Complexity and opportunities. Front. Med. 2018, 12, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Tian, X.-J.; Xing, J. Signal Transduction Pathways of EMT Induced by TGF-β, SHH, and WNT and Their Crosstalks. J. Clin. Med. 2016, 5. [Google Scholar] [CrossRef]
- Marín-Aguilera, M.; Codony-Servat, J.; Reig, Ò.; Lozano, J.J.; Fernández, P.L.; Pereira, M.V.; Jiménez, N.; Donovan, M.; Puig, P.; Mengual, L.; et al. Epithelial-to-mesenchymal transition mediates docetaxel resistance and high risk of relapse in prostate cancer. Mol. Cancer Ther. 2014, 13, 1270–1284. [Google Scholar] [CrossRef] [PubMed]
- Bolós, V.; Peinado, H.; Pérez-Moreno, M.A.; Fraga, M.F.; Esteller, M.; Cano, A. The transcription factor Slug represses E-cadherin expression and induces epithelial to mesenchymal transitions: A comparison with Snail and E47 repressors. J. Cell Sci. 2003, 116, 499–511. [Google Scholar] [CrossRef]
- Cobaleda, C.; Pérez-Caro, M.; Vicente-Dueñas, C.; Sánchez-García, I. Function of the zinc-finger transcription factor SNAI2 in cancer and development. Annu. Rev. Genet. 2007, 41, 41–61. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, J.; Chai, K.; Ying, X.; Zhou, B.P. The Role of Snail in EMT and Tumorigenesis. Curr. Cancer Drug Targets 2013, 13, 963–972. [Google Scholar] [CrossRef]
- Esposito, S.; Russo, M.V.; Airoldi, I.; Tupone, M.G.; Sorrentino, C.; Barbarito, G.; Di Meo, S.; Di Carlo, E. SNAI2/Slug gene is silenced in prostate cancer and regulates neuroendocrine differentiation, metastasis-suppressor and pluripotency gene expression. Oncotarget 2015, 6, 17121–17134. [Google Scholar] [CrossRef]
- Kahounová, Z.; Remšík, J.; Fedr, R.; Bouchal, J.; Mičková, A.; Slabáková, E.; Binó, L.; Hampl, A.; Souček, K. Slug-expressing mouse prostate epithelial cells have increased stem cell potential. Stem Cell Res. 2020, 46, 101844. [Google Scholar] [CrossRef]
- Cui, D.; Xiong, X.; Zhao, Y. Cullin-RING ligases in regulation of autophagy. Cell Div. 2016, 11, 8. [Google Scholar] [CrossRef]
- Cai, Z.; Moten, A.; Peng, D.; Hsu, C.-C.; Pan, B.-S.; Manne, R.; Li, H.-Y.; Lin, H.-K. The Skp2 Pathway: A Critical Target for Cancer Therapy. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Senft, D.; Qi, J.; Ronai, Z.A. Ubiquitin ligases in oncogenic transformation and cancer therapy. Nat. Rev. Cancer 2018, 18, 69–88. [Google Scholar] [CrossRef]
- Brown, J.S.; Jackson, S.P. Ubiquitylation, neddylation and the DNA damage response. Open Biol. 2015, 5, 150018. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lu, Y.; Wang, L.; Mizokami, A.; Keller, E.T.; Zhang, J.; Fu, J. Skp2 is associated with paclitaxel resistance in prostate cancer cells. Oncol. Rep. 2016, 36, 559–566. [Google Scholar] [CrossRef]
- Ben-Izhak, O.; Lahav-Baratz, S.; Meretyk, S.; Ben-Eliezer, S.; Sabo, E.; Dirnfeld, M.; Cohen, S.; Ciechanover, A. Inverse relationship between Skp2 ubiquitin ligase and the cyclin dependent kinase inhibitor p27Kip1 in prostate cancer. J. Urol. 2003, 170, 241–245. [Google Scholar] [CrossRef]
- Jang, W.; Kim, T.; Koo, J.S.; Kim, S.-K.; Lim, D.-S. Mechanical cue-induced YAP instructs Skp2-dependent cell cycle exit and oncogenic signaling. EMBO J. 2017, 36, 2510–2528. [Google Scholar] [CrossRef]
- Simeckova, S.; Kahounová, Z.; Fedr, R.; Remšík, J.; Slabáková, E.; Suchánková, T.; Procházková, J.; Bouchal, J.; Kharaishvili, G.; Král, M.; et al. High Skp2 expression is associated with a mesenchymal phenotype and increased tumorigenic potential of prostate cancer cells. Sci. Rep. 2019, 9, 5695. [Google Scholar] [CrossRef]
- Urbanucci, A.; Sahu, B.; Seppälä, J.; Larjo, A.; Latonen, L.M.; Waltering, K.K.; Tammela, T.L.J.; Vessella, R.L.; Lähdesmäki, H.; Jänne, O.A.; et al. Overexpression of androgen receptor enhances the binding of the receptor to the chromatin in prostate cancer. Oncogene 2012, 31, 2153–2163. [Google Scholar] [CrossRef]
- Tomlins, S.A.; Mehra, R.; Rhodes, D.R.; Cao, X.; Wang, L.; Dhanasekaran, S.M.; Kalyana-Sundaram, S.; Wei, J.T.; Rubin, M.A.; Pienta, K.J.; et al. Integrative molecular concept modeling of prostate cancer progression. Nat. Genet. 2007, 39, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.-H.; Morrow, J.K.; Li, C.-F.; Gao, Y.; Jin, G.; Moten, A.; Stagg, L.J.; Ladbury, J.E.; Cai, Z.; Xu, D.; et al. Pharmacological inactivation of Skp2 SCF ubiquitin ligase restricts cancer stem cell traits and cancer progression. Cell 2013, 154, 556–568. [Google Scholar] [CrossRef]
- Zhou, W.; Ni, T.K.; Wronski, A.; Glass, B.; Skibinski, A.; Beck, A.; Kuperwasser, C. The SIRT2 Deacetylase Stabilizes Slug to Control Malignancy of Basal-like Breast Cancer. Cell Rep. 2016, 17, 1302–1317. [Google Scholar] [CrossRef]
- Jin, Y.; Zhang, P.; Wang, Y.; Jin, B.; Zhou, J.; Zhang, J.; Pan, J. Neddylation Blockade Diminishes Hepatic Metastasis by Dampening Cancer Stem-Like Cells and Angiogenesis in Uveal Melanoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 3741–3754. [Google Scholar] [CrossRef]
- Hauck, P.M.; Wolf, E.R.; Olivos, D.J., III; McAtarsney, C.P.; Mayo, L.D. The fate of murine double minute X (MdmX) is dictated by distinct signaling pathways through murine double minute 2 (Mdm2). Oncotarget 2017, 8, 104455–104466. [Google Scholar] [CrossRef]
- Wang, Y.V.; Wade, M.; Wahl, G.M. Guarding the guardian: Mdmx plays important roles in setting p53 basal activity and determining biological responses in vivo. Cell Cycle 2009, 21, 21–3444. [Google Scholar] [CrossRef][Green Version]
- Lu, L.; Schulz, H.; Wolf, D.A. The F-box protein SKP2 mediates androgen control of p27 stability in LNCaP human prostate cancer cells. BMC Cell Biol. 2002, 3, 22. [Google Scholar] [CrossRef]
- Tsai, Y.-S.; Lai, C.-L.; Lai, C.-H.; Chang, K.-H.; Wu, K.; Tseng, S.-F.; Fazli, L.; Gleave, M.; Xiao, G.; Gandee, L.; et al. The role of homeostatic regulation between tumor suppressor DAB2IP and oncogenic Skp2 in prostate cancer growth. Oncotarget 2014, 5, 6425–6436. [Google Scholar] [CrossRef] [PubMed]
- Pernicová, Z.; Slabáková, E.; Kharaishvili, G.; Bouchal, J.; Král, M.; Kunická, Z.; Machala, M.; Kozubík, A.; Souček, K. Androgen depletion induces senescence in prostate cancer cells through down-regulation of Skp2. Neoplasia 2011, 13, 526–536. [Google Scholar] [CrossRef]
- Wang, H.; Sun, D.; Ji, P.; Mohler, J.; Zhu, L. An AR-Skp2 pathway for proliferation of androgen-dependent prostate-cancer cells. J. Cell Sci. 2008, 121, 2578–2587. [Google Scholar] [CrossRef] [PubMed]
- Ruan, D.; He, J.; Li, C.-F.; Lee, H.-J.; Liu, J.; Lin, H.-K.; Chan, C.-H. Skp2 deficiency restricts the progression and stem cell features of castration-resistant prostate cancer by destabilizing Twist. Oncogene 2017, 36, 4299–4310. [Google Scholar] [CrossRef]
- Lu, W.; Liu, S.; Li, B.; Xie, Y.; Izban, M.G.; Ballard, B.R.; Sathyanarayana, S.A.; Adunyah, S.E.; Matusik, R.J.; Chen, Z. SKP2 loss destabilizes EZH2 by promoting TRAF6-mediated ubiquitination to suppress prostate cancer. Oncogene 2017, 36, 1364–1373. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Lee, S.H.; Song, K.Y.; Na, S.J.; Joo, H.O.; Park, J.M.; Jung, E.S.; Choi, M.-G.; Park, C.H. Evaluation of Slug expression is useful for predicting lymph node metastasis and survival in patients with gastric cancer. BMC Cancer 2017, 17, 670. [Google Scholar] [CrossRef]
- Shioiri, M.; Shida, T.; Koda, K.; Oda, K.; Seike, K.; Nishimura, M.; Takano, S.; Miyazaki, M. Slug expression is an independent prognostic parameter for poor survival in colorectal carcinoma patients. Br. J. Cancer 2006, 94, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
- Mikami, S.; Katsube, K.-I.; Oya, M.; Ishida, M.; Kosaka, T.; Mizuno, R.; Mukai, M.; Okada, Y. Expression of Snail and Slug in renal cell carcinoma: E-cadherin repressor Snail is associated with cancer invasion and prognosis. Lab. Investig. J. Tech. Methods Pathol. 2011, 91, 1443–1458. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Huang, J.; Wu, Q.; Cai, Y.; Zhu, L.; Lu, X.; Chen, S.; Chen, C.; Wang, Z. Acquisition of epithelial-mesenchymal transition is associated with Skp2 expression in paclitaxel-resistant breast cancer cells. Br. J. Cancer 2014, 110, 1958–1967. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Han, S.; Wilder-Romans, K.; Sun, G.Y.; Zhu, H.; Liu, X.; Tan, M.; Wang, G.; Feng, F.Y.; Sun, Y. Neddylation inactivation represses androgen receptor transcription and inhibits growth, survival and invasion of prostate cancer cells. Neoplasia 2020, 22, 192–202. [Google Scholar] [CrossRef]
- Lockhart, A.C.; Bauer, T.M.; Aggarwal, C.; Lee, C.B.; Harvey, R.D.; Cohen, R.B.; Sedarati, F.; Nip, T.K.; Faessel, H.; Dash, A.B.; et al. Phase Ib study of pevonedistat, a NEDD8-activating enzyme inhibitor, in combination with docetaxel, carboplatin and paclitaxel, or gemcitabine, in patients with advanced solid tumors. Investig. New Drugs 2019, 37, 87–97. [Google Scholar] [CrossRef]
- Zhou, L.; Jiang, Y.; Luo, Q.; Li, L.; Jia, L. Neddylation: A novel modulator of the tumor microenvironment. Mol. Cancer 2019, 18, 77. [Google Scholar] [CrossRef] [PubMed]
- Puhr, M.; Hoefer, J.; Schäfer, G.; Erb, H.H.H.; Oh, S.J.; Klocker, H.; Heidegger, I.; Neuwirt, H.; Culig, Z. Epithelial-to-mesenchymal transition leads to docetaxel resistance in prostate cancer and is mediated by reduced expression of miR-200c and miR-205. Am. J. Pathol. 2012, 181, 2188–2201. [Google Scholar] [CrossRef]
- Liao, C.-P.; Liang, M.; Cohen, M.B.; Flesken-Nikitin, A.; Jeong, J.H.; Nikitin, A.Y.; Roy-Burman, P. Mouse prostate cancer cell lines established from primary and postcastration recurrent tumors. Horm Cancer 2010, 1, 44–54. [Google Scholar] [CrossRef][Green Version]
- Remšík, J.; Binó, L.; Kahounová, Z.; Kharaishvili, G.; Šimecková, Š.; Fedr, R.; Kucírková, T.; Lenárt, S.; Muresan, X.M.; Slabáková, E.; et al. Trop-2 plasticity is controlled by epithelial-to-mesenchymal transition. Carcinogenesis 2018, 39, 1411–1418. [Google Scholar] [CrossRef]
- Simkova, D.; Kharaishvili, G.; Korinkova, G.; Ozdian, T.; Suchánková-Kleplová, T.; Soukup, T.; Krupka, M.; Galandakova, A.; Dzubak, P.; Janikova, M.; et al. The dual role of asporin in breast cancer progression. Oncotarget 2016, 7, 52045–52060. [Google Scholar] [CrossRef] [PubMed]
- Bouchal, J.; Baumforth, K.R.N.; Sváchová, M.; Murray, P.G.; von Angerer, E.; Kolár, Z. Microarray analysis of bicalutamide action on telomerase activity, p53 pathway and viability of prostate carcinoma cell lines. J. Pharm. Pharmacol. 2005, 57, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.H.; Zhang, Y.; Jiang, L.Q.; Wang, S.; Lei, C.Q.; Sun, M.S.; Shu, H.B.; Liu, Y. WDFY1 mediates TLR3/4 signaling by recruiting TRIF. EMBO Rep. 2015, 16, 447–455. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Subgroups | N | % |
|---|---|---|---|
| Age | 49–60 | 36 | 35.6 |
| 61–70 | 57 | 56.4 | |
| 71–76 | 8 | 7.9 | |
| Serum PSA (ng/mL) | <4 | 12 | 11.9 |
| 4–10 | 40 | 39.6 | |
| >10 | 48 | 47.5 | |
| missing | 1 | 1.0 | |
| Gleason score | <7 | 22 | 21.8 |
| 7 | 49 | 48.5 | |
| >7 | 30 | 29.7 | |
| Cancer stage | pT2a-c | 42 | 41.6 |
| pT3a-b | 50 | 49.5 | |
| pT4 | 9 | 8.9 | |
| Lymph node status | pN1 | 29 | 28.7 |
| pN0 | 65 | 64.4 | |
| pNx* | 7 | 6.9 |
| Protein 1 | Protein 2 | Whole Cohort (N = 101) | Gleason > 7 (N = 30) | Positive LN (N = 29) |
|---|---|---|---|---|
| Skp2 | Slug | 0.322 | 0.557 | 0.559 |
| androgen receptor | 0.370 | 0.535 | 0.372 | |
| Slug | androgen receptor | 0.154 | 0.374 | 0.285 |
| Ki-67 | androgen receptor | 0.054 | 0.246 | 0.504 |
| beta-catenin membrane | −0.193 | −0.234 | −0.418 | |
| E-cadherin | beta-catenin membrane | 0.106 | 0.423 | 0.615 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mickova, A.; Kharaishvili, G.; Kurfurstova, D.; Gachechiladze, M.; Kral, M.; Vacek, O.; Pokryvkova, B.; Mistrik, M.; Soucek, K.; Bouchal, J. Skp2 and Slug Are Coexpressed in Aggressive Prostate Cancer and Inhibited by Neddylation Blockade. Int. J. Mol. Sci. 2021, 22, 2844. https://doi.org/10.3390/ijms22062844
Mickova A, Kharaishvili G, Kurfurstova D, Gachechiladze M, Kral M, Vacek O, Pokryvkova B, Mistrik M, Soucek K, Bouchal J. Skp2 and Slug Are Coexpressed in Aggressive Prostate Cancer and Inhibited by Neddylation Blockade. International Journal of Molecular Sciences. 2021; 22(6):2844. https://doi.org/10.3390/ijms22062844
Chicago/Turabian StyleMickova, Alena, Gvantsa Kharaishvili, Daniela Kurfurstova, Mariam Gachechiladze, Milan Kral, Ondrej Vacek, Barbora Pokryvkova, Martin Mistrik, Karel Soucek, and Jan Bouchal. 2021. "Skp2 and Slug Are Coexpressed in Aggressive Prostate Cancer and Inhibited by Neddylation Blockade" International Journal of Molecular Sciences 22, no. 6: 2844. https://doi.org/10.3390/ijms22062844
APA StyleMickova, A., Kharaishvili, G., Kurfurstova, D., Gachechiladze, M., Kral, M., Vacek, O., Pokryvkova, B., Mistrik, M., Soucek, K., & Bouchal, J. (2021). Skp2 and Slug Are Coexpressed in Aggressive Prostate Cancer and Inhibited by Neddylation Blockade. International Journal of Molecular Sciences, 22(6), 2844. https://doi.org/10.3390/ijms22062844






