New Insight into Justicidin B Pathway and Production in Linum austriacum
Abstract
1. Introduction
2. Results
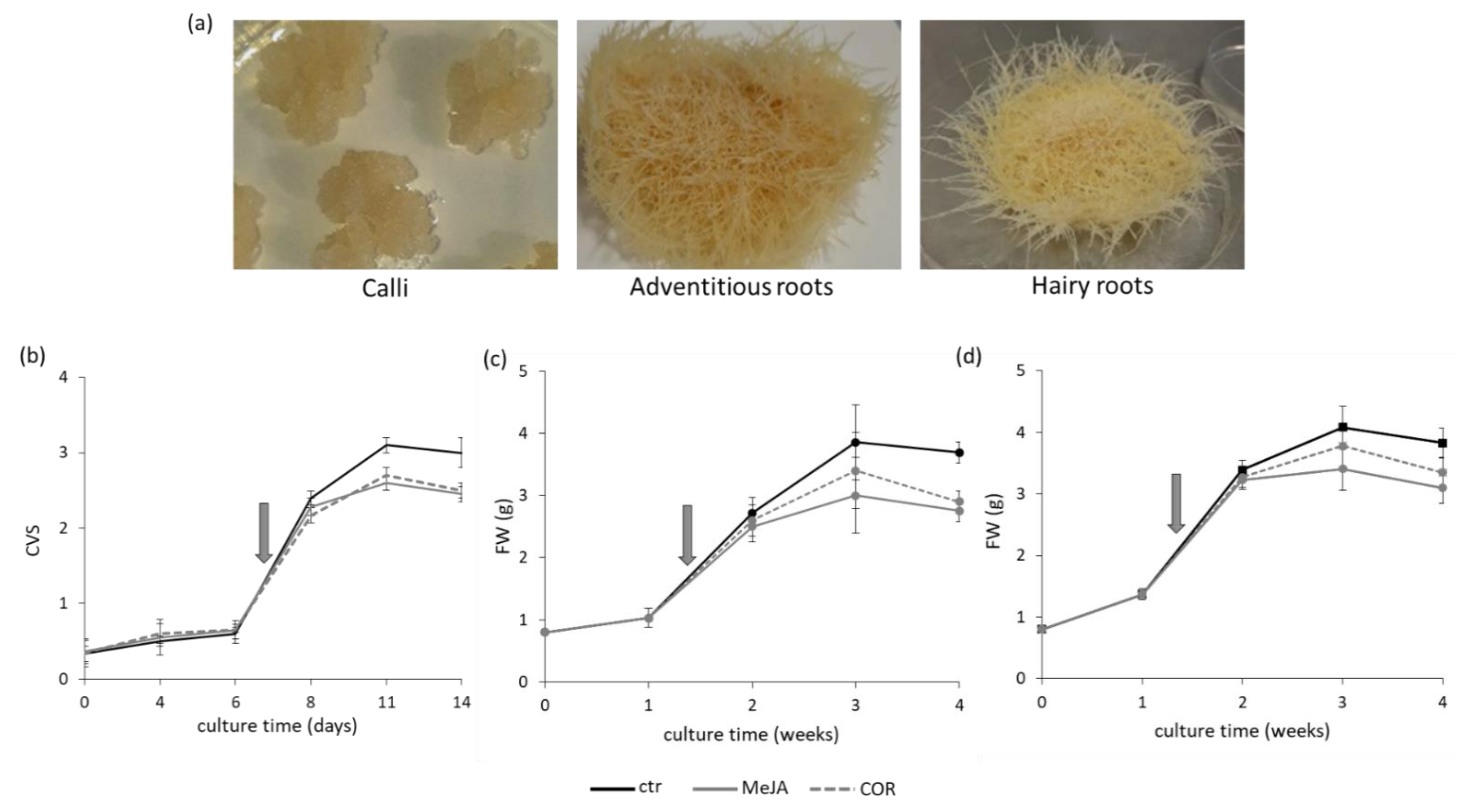
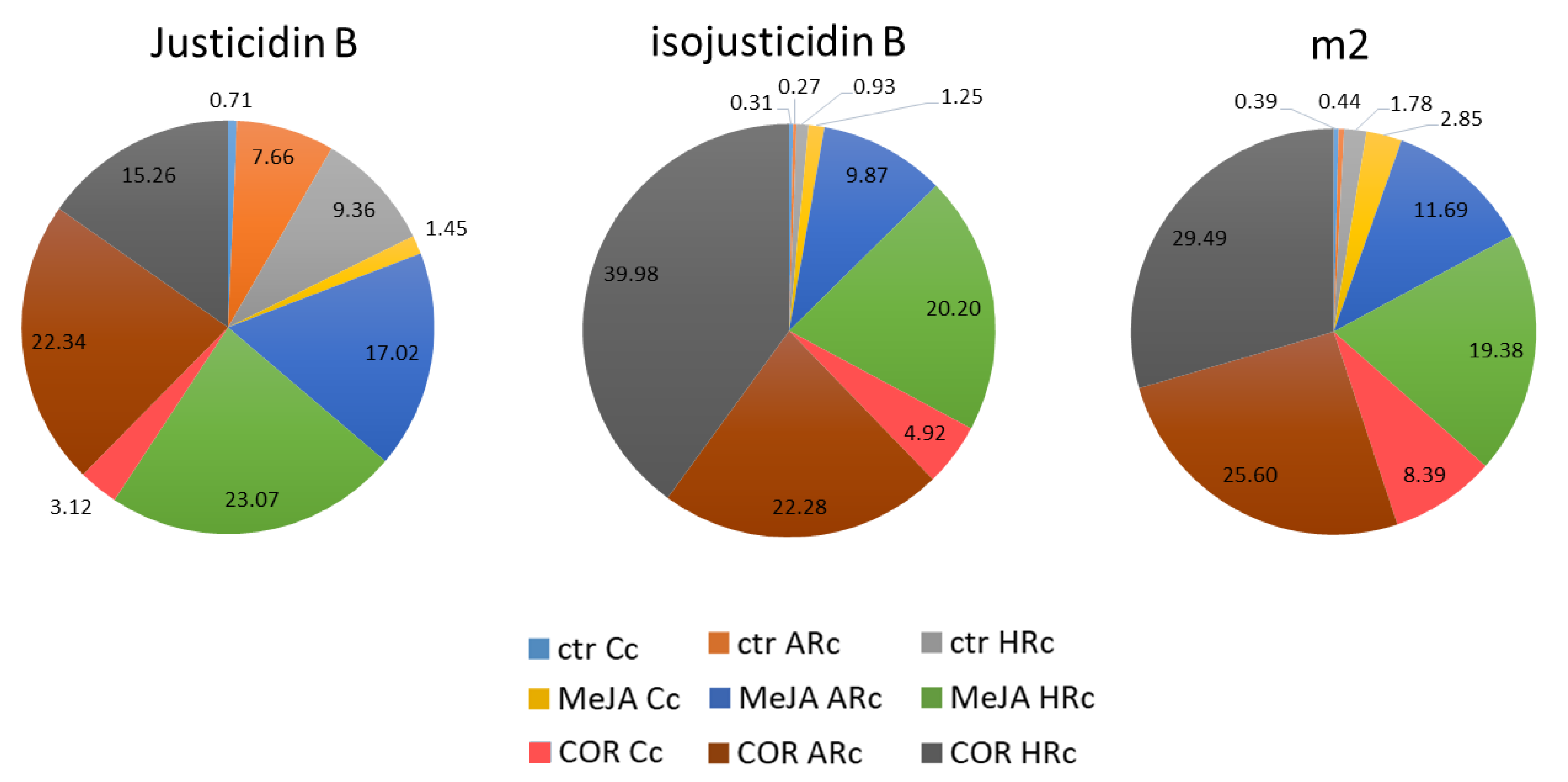
2.1. Tissue Cultures, Elicitors Treatment and Phenolic Content
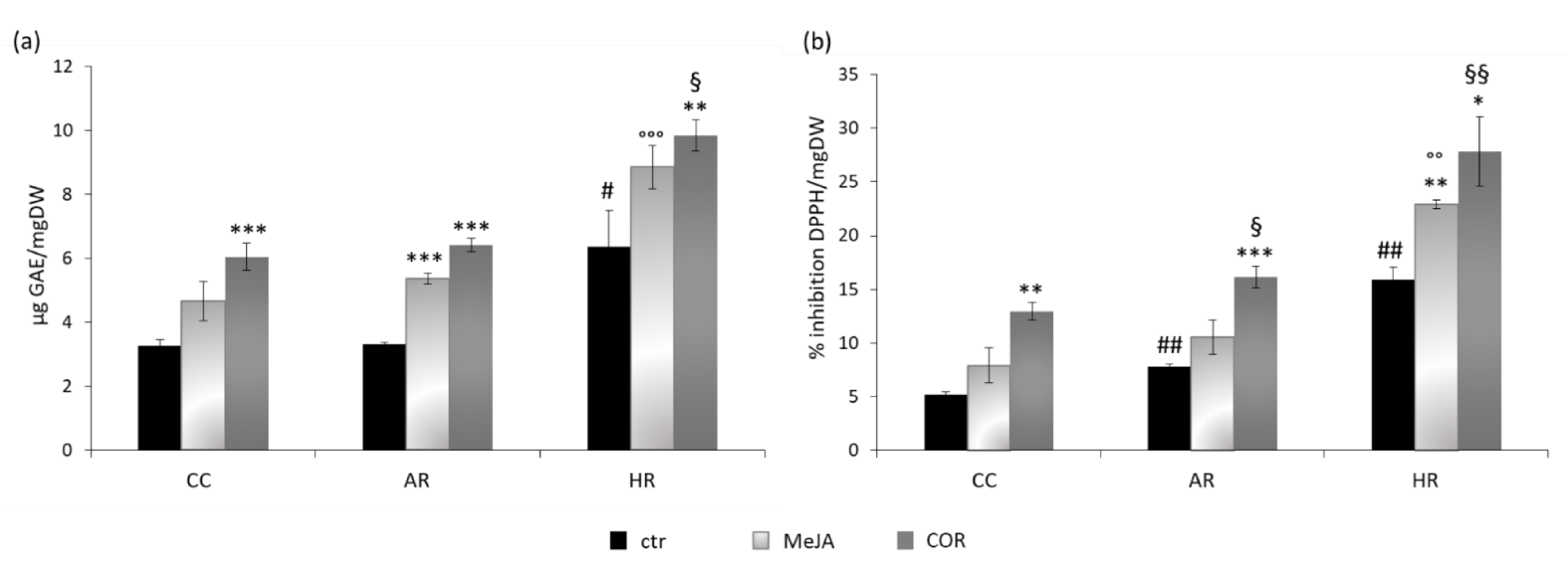
2.2. Total Phenolic Content and DPPH Radical Scavenging Activity
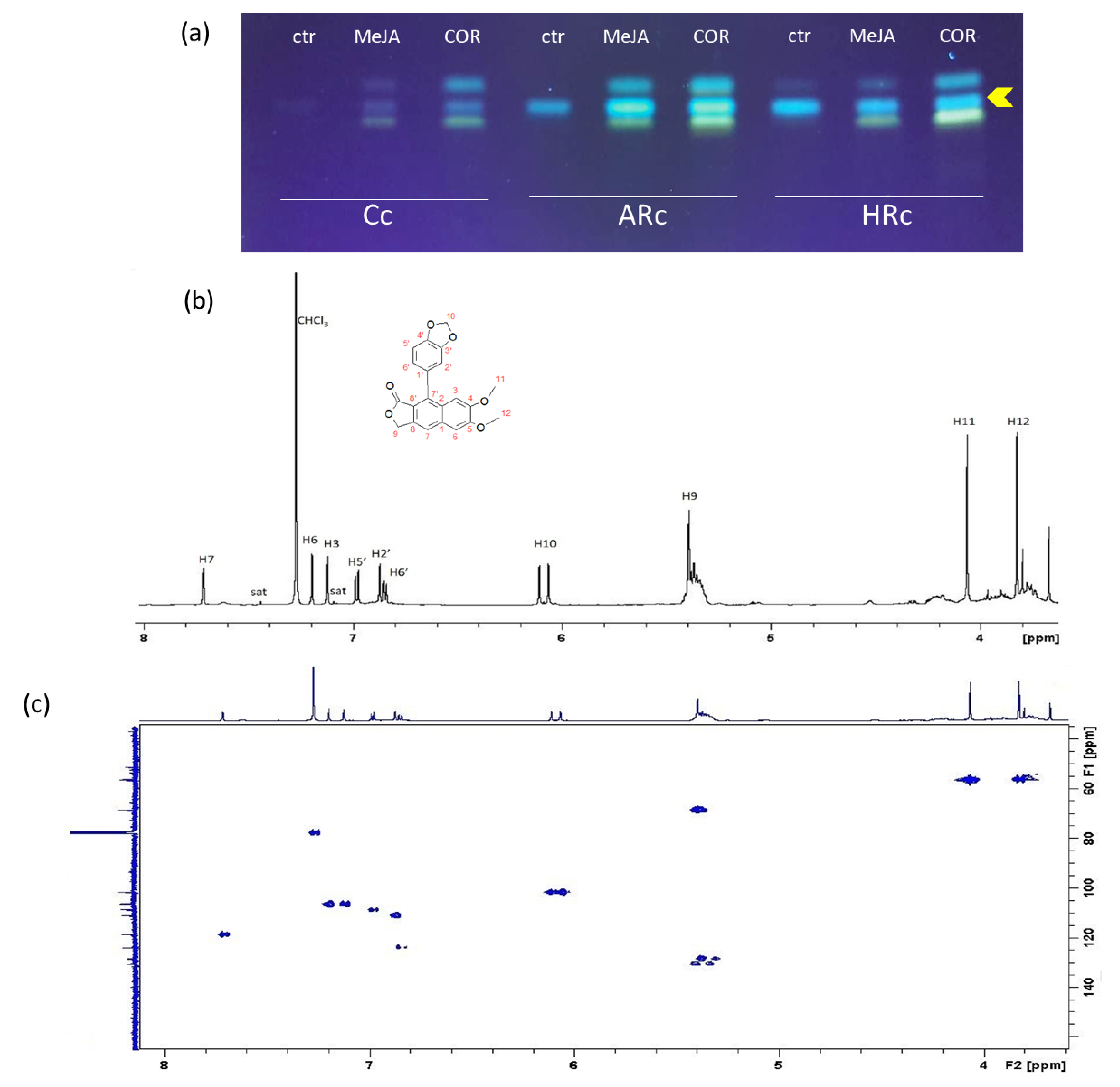
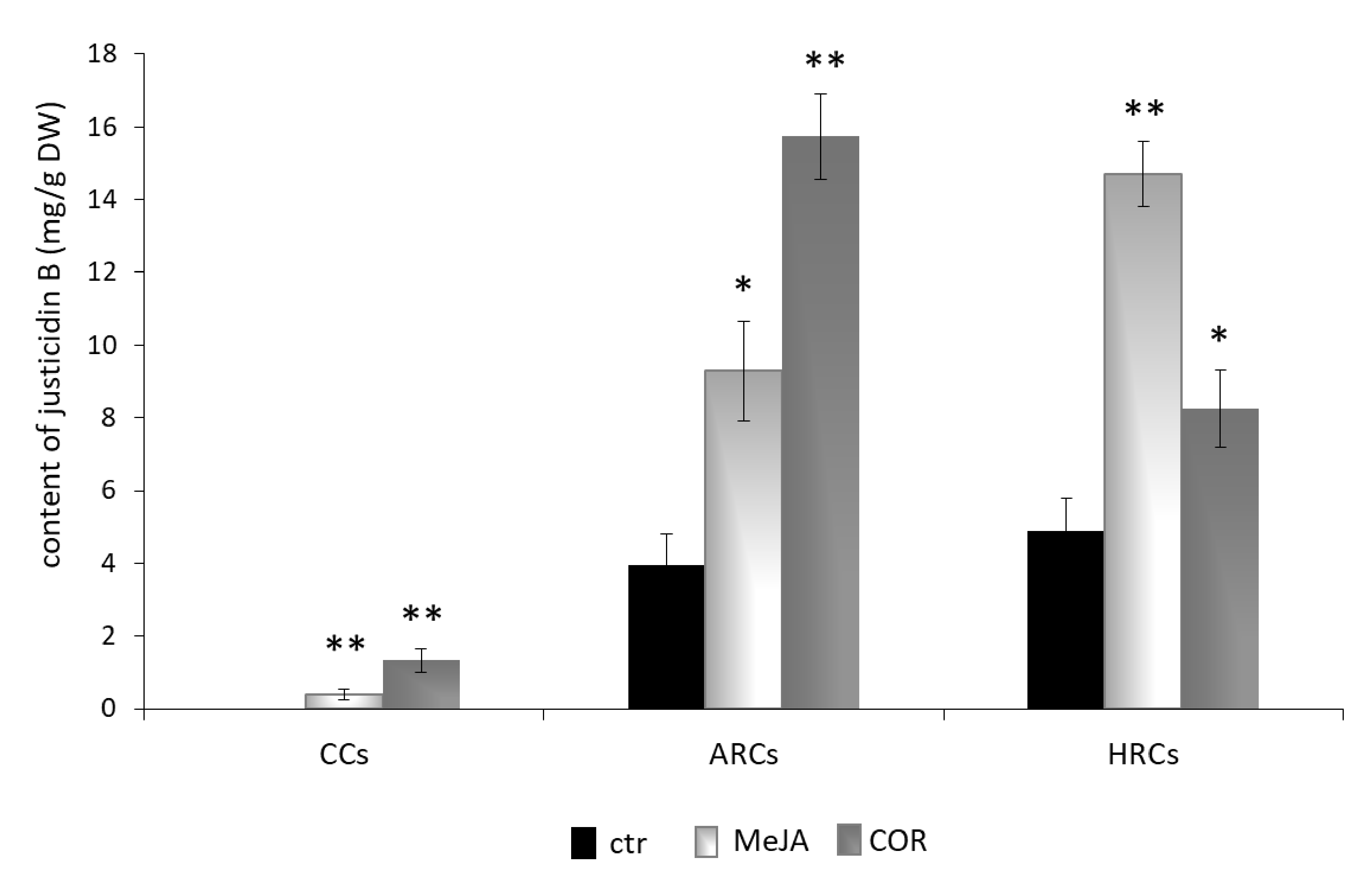
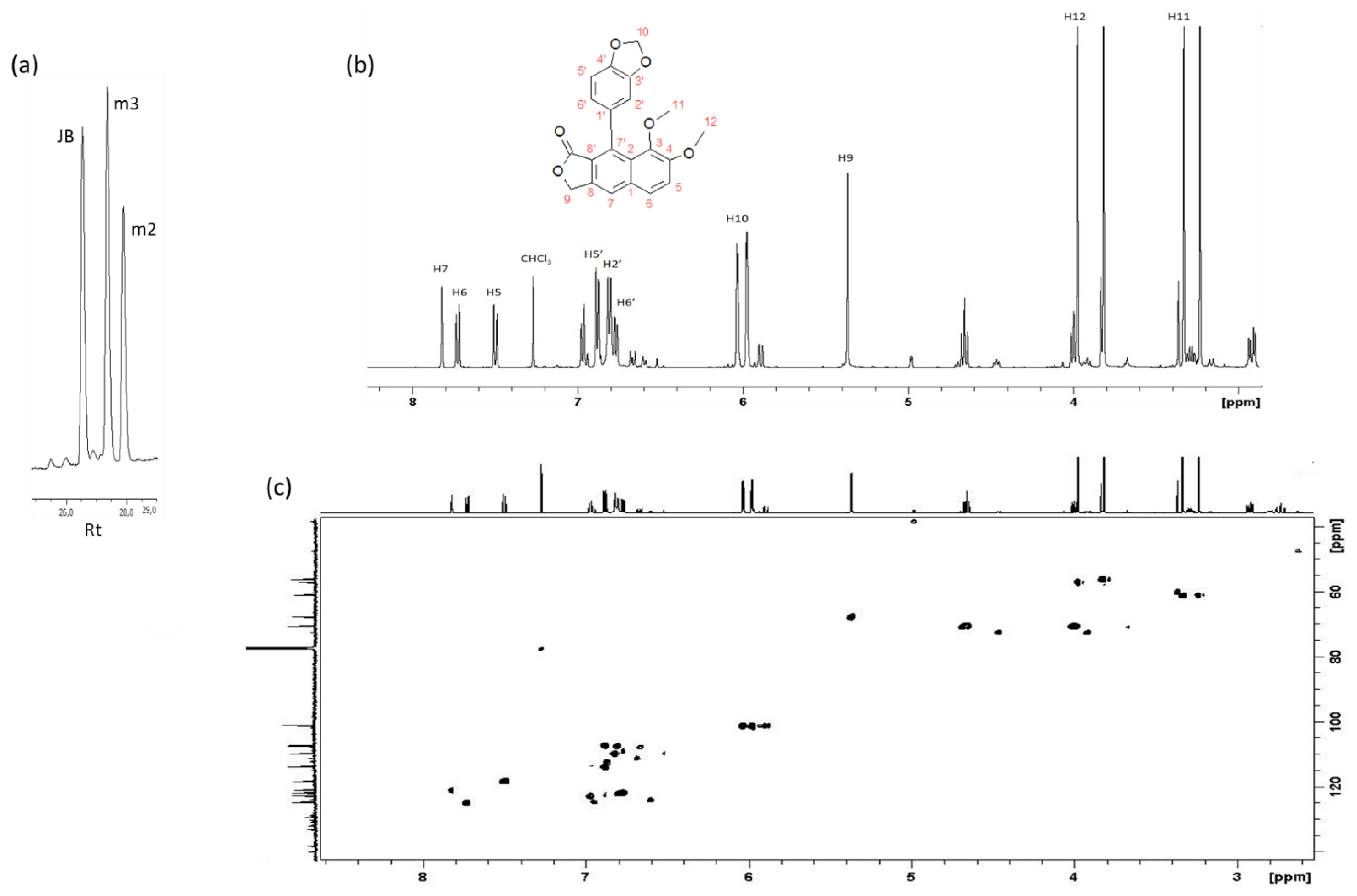
2.3. Isolation, Identification and Quantification of Justicidin B
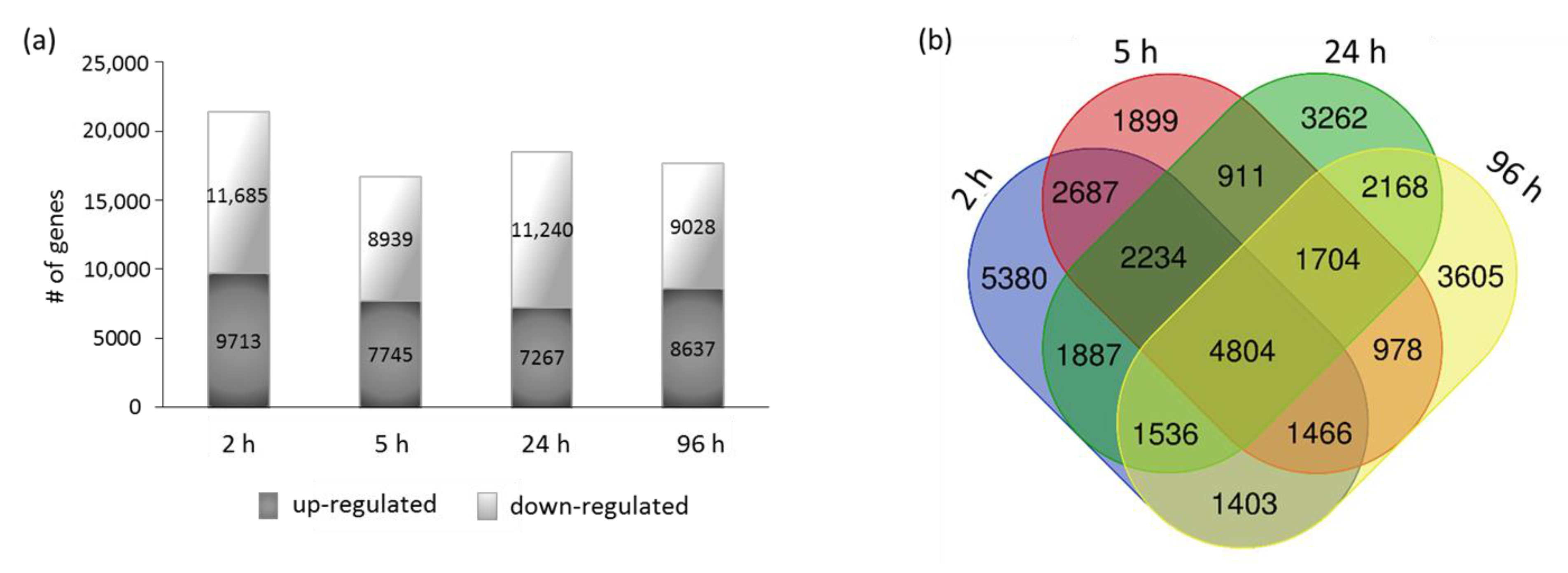
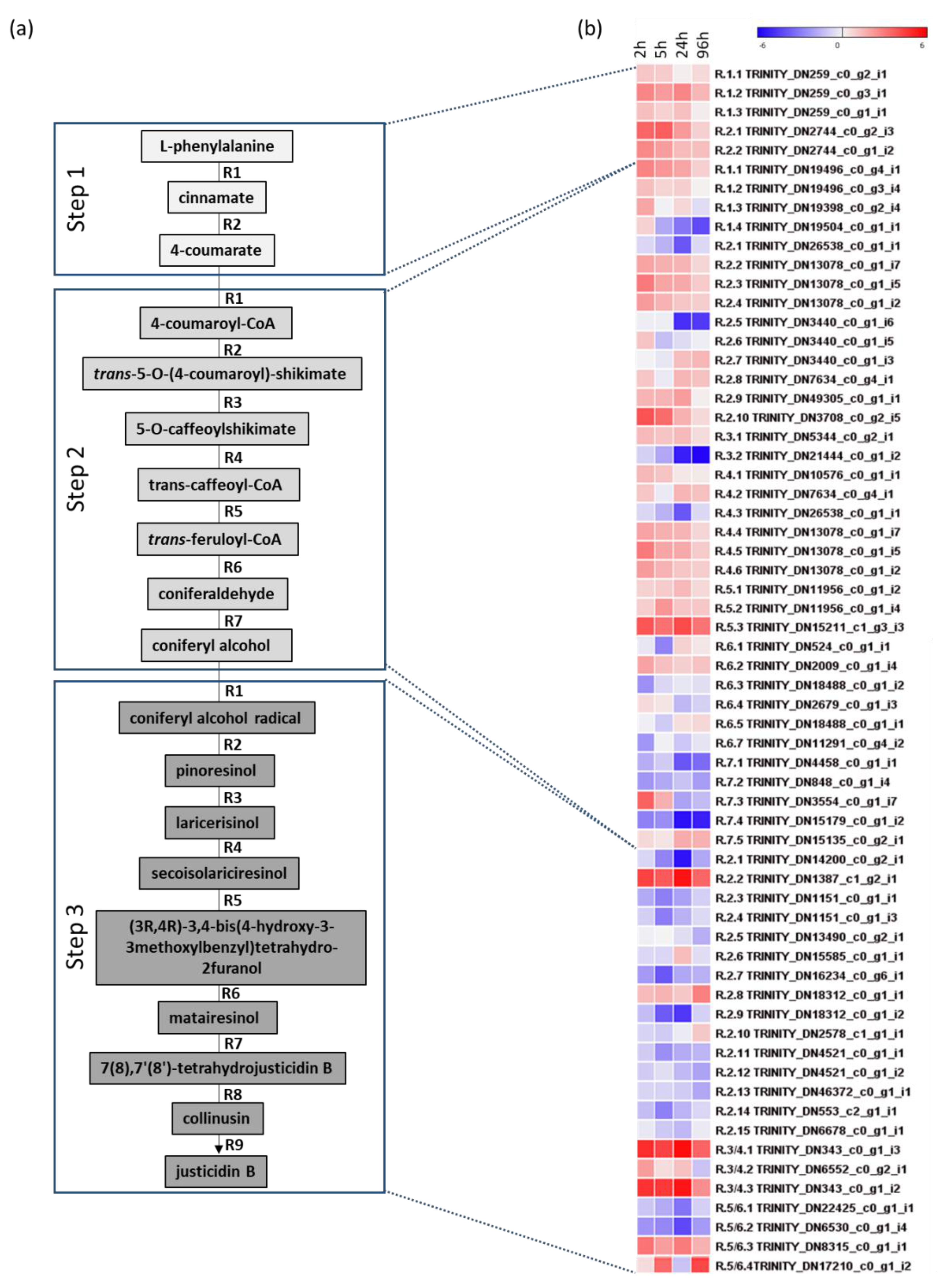
2.4. RNA-Seq Analysis
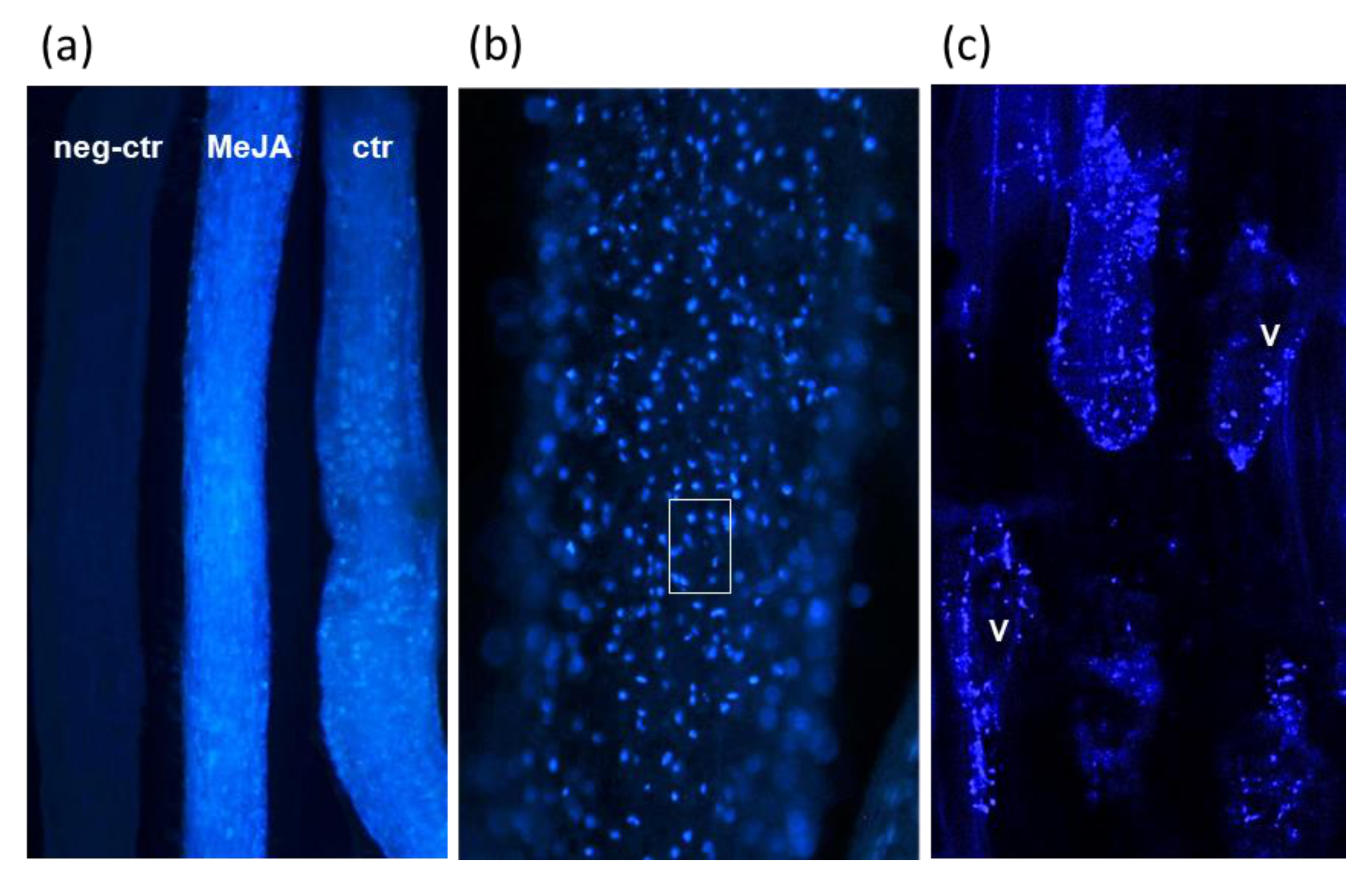
2.5. Microscopic Analysis of Justicidin B in Adventitious Roots
2.6. Justicidin B Production in Bioreactor
3. Discussion
3.1. Elicitation, Time Course of Growth, Phenols Accumulation and Antioxidant Activity
3.2. Identification, Isolation and Quantification of Justicidin B
3.3. Bioreactor Culture
3.4. Transcriptome Analysis of Justicidin B Pathway
3.5. Justicidin B Cellular Localization
4. Conclusions
5. Materials and Methods
5.1. Plant Material and Cultures
5.2. Growth Measurement and Kinetics of Cc, ARc and HRc
5.3. Elicitor Treatments
5.4. Lignans Extraction
5.5. Total Phenolic Content and DPPH Radical Scavenging Activity
5.6. Chromatographic Analysis
5.6.1. TLC experimental conditions
5.6.2. Analytical HPLC
5.6.3. Semi-preparative HPLC
5.7. 1H-NMR Analysis
5.8. RNA Sequencing, Sequence Assembly and Functional Annotation
5.9. Fluorescence and Confocal Microscopic Analysis of Justicidin B in Adventitious Root Cultures
5.10. Hairy Roots Cultivation in Bioreactor System
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Namdeo, A.G. Plant cell elicitation for production of secondary metabolites: A review. Pharmacogn. Rev. 2007, 1, 69–79. [Google Scholar]
- Guerriero, G.; Berni, R.; Muñoz-Sánchez, J.A.; Apone, F.; Abdel-Salam, E.M.; Qahtan, A.A.; Alatar, A.A.; Cantini, C.; Cai, G.; Hausman, J.-F.; et al. Production of Plant Secondary Metabolites: Examples, Tips and Suggestions for Biotechnologists. Genes 2018, 9, 309. [Google Scholar] [CrossRef]
- Niraula, N.P.; Kim, S.-H.; Sohng, J.K.; Kim, E.-S. Biotechnological doxorubicin production: Pathway and regulation engineering of strains for enhanced production. Appl. Microbiol. Biotechnol. 2010, 87, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Umezawa, T. Biosynthesis of lignans and norlignans. J. Wood Sci. 2007, 53, 273–284. [Google Scholar] [CrossRef]
- Bennett, R.N.; Wallsgrove, R.M. Secondary metabolites in plant defence mechanisms. New Phytol. 1994, 127, 617–633. [Google Scholar] [CrossRef]
- Gordaliza, M.; Garcıa, P.A.; Del Corral, J.M.; Castro, M.A.; Gómez-Zurita, M.A. Podophyllotoxin: Distribution, sources, applications and new cytotoxic derivatives. Toxicon 2004, 44, 441–459. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.; Sánchez, I.; Pujol, M.D. Synthesis and Biological Activity of New Class of Dioxygenated Anticancer Agents. Curr. Med. Chem. Agents 2005, 5, 215–237. [Google Scholar] [CrossRef]
- Saleem, M.; Kim, H.J.; Ali, M.S.; Lee, Y.S. An update on bioactive plant lignans. Nat. Prod. Rep. 2005, 22, 696–716. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.-H.; Jin, H.-B.; Chen, Y.-H.; Cui, L.-J.; Ren, W.-W.; Gong, Y.-F.; Tang, K.-X. Terpenoid Indole Alkaloids Biosynthesis and Metabolic Engineering inCatharanthus roseus. J. Integr. Plant Biol. 2007, 49, 961–974. [Google Scholar] [CrossRef]
- Malik, S.; Bíba, O.; Grúz, J.; Arroo, R.R.J.; Strnad, M. Biotechnological approaches for producing aryltetralin lignans from Linum species. Phytochem. Rev. 2014, 13, 893–913. [Google Scholar] [CrossRef]
- Schmidt, T.J.; Hemmati, S.; Klaes, M.; Konuklugil, B.; Mohagheghzadeh, A.; Ionkova, I.; Fuss, E.; Alfermann, A.W. Lignans in flowering aerial parts of Linum species—Chemodiversity in the light of systematics and phylogeny. Phytochemistry 2010, 71, 1714–1728. [Google Scholar] [CrossRef]
- Rogers, C.M. The systematics of Linum sect. Linopsis (Linaceae). Plant Syst. Evol. 1982, 140, 225. [Google Scholar] [CrossRef]
- Diederichsen, A.; Richards, K. Cultivated flax and the genus Linum L.: Taxonomy and germplasm conservation. In Flax, 1st ed.; CRC Press: Boca Raton, FL, USA, 2003; pp. 34–66. [Google Scholar]
- Schmidt, T.J.; Klaes, M.; Sendker, J. Lignans in seeds of Linum species. Phytochemistry 2012, 82, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Hemmati, S.; Seradj, H. Justicidin B: A Promising Bioactive Lignan. Molecules 2016, 21, 820. [Google Scholar] [CrossRef] [PubMed]
- Ai, Z.Z.; Zhou, S.S.; Wu, S.T.; Hong, Z.C.; Zhang, Y.; Wang, M.F.; Yang, Y.F.; Wu, H.Z. Justicidin B inhibits PDGF-BB-induced proliferation and ECM accumulation in mesangial cells via Nrf2/HO-1 and Akt/mTOR signaling pathway. Nat. Prod. Commun. 2020, 15, 1–10. [Google Scholar] [CrossRef]
- Mohagheghzadeh, A.; Schmidt, T.J.; Alfermann, A.W. Arylnaphthalene Lignans from in Vitro Cultures ofLinum austriacum†. J. Nat. Prod. 2002, 65, 69–71. [Google Scholar] [CrossRef]
- Ionkova, I.; Sasheva, P.; Ionkov, T.; Momekov, G. Linum narbonense: A new valuable tool for biotechnological production of a potent anticancer lignan Justicidine B. Pharmacogn. Mag. 2013, 9, 39–44. [Google Scholar] [CrossRef]
- Konuklugil, B.; Ionkova, I.; Vasilev, N.; Schmidt, T.J.; Windhövel, J.; Fuss, E.; Alfermann, A.W. Lignans from Linum species of sections Syllinum and Linum. Nat. Prod. Res. 2007, 21, 1–6. [Google Scholar] [CrossRef]
- Vasilev, N.; Elfahmi Bos, R.; Kayser, O.; Momekov, G.; Konstantinov, S.; Ionkova, I. Production of Justicidin B, a cytotoxic arylnaphthalene lignan from genetically transformed root cultures of Linum leonii. J. Nat. Prod. 2006, 69, 1014–1017. [Google Scholar] [CrossRef]
- Vasilev, N.; Ionkova, I. Lignan Production by Cell Cultures of Linum setaceum. and Linum campanulatum. Pharm. Biol. 2005, 43, 509–511. [Google Scholar] [CrossRef]
- Mohagheghzadeh, A.; Dehshahri, S.; Hemmati, S. Accumulation of Lignans by in vitro Cultures of Three Linum Species. Z. Naturforsch. C 2009, 64, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Hemmati, S.; Schmidt, T.J.; Fuss, E. (+)-Pinoresinol/(−)-lariciresinol reductase fromLinum perenneHimmelszelt involved in the biosynthesis of justicidin B. FEBS Lett. 2007, 581, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Bourgaud, F.; Gravot, A.; Milesi, S.; Gontier, E. Production of plant secondary metabolites: A historical perspective. Plant Sci. 2001, 161, 839–851. [Google Scholar] [CrossRef]
- Radman, R.; Saez, T.; Bucke, C.; Keshavarz, T. Elicitation of plants and microbial cell systems. Biotechnol. Appl. Biochem. 2003, 37, 91–102. [Google Scholar] [CrossRef]
- McConn, M.; Creelman, R.A.; Bell, E.; Mullet, J.E.; Browse, J. Jasmonate is essential for insect defense in Arabidopsis. Proc. Natl. Acad. Sci. USA 1997, 94, 5473–5477. [Google Scholar] [CrossRef]
- Ionkova, I. Effect of methyl jasmonate on production of ariltetralin lignans in hairy root cultures of Linum tauricum. Pharmacognosy Res. 2009, 1, 102. [Google Scholar]
- Bender, C.L.; Alarcón-Chaidez, F.; Gross, D.C. Pseudomonas syringae Phytotoxins: Mode of Action, Regulation, and Biosynthesis by Peptide and Polyketide Synthetases. Microbiol. Mol. Biol. Rev. 1999, 63, 266–292. [Google Scholar] [CrossRef] [PubMed]
- Onrubia, M.; Moyano, E.; Bonfill, M.; Cusidó, R.M.; Goossens, A.; Palazón, J. Coronatine, a more powerful elicitor for inducing taxane biosynthesis in Taxus x media cell cultures than methyl jasmonate. J. Plant Physiol. 2013, 170, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Katsir, L.; Schilmiller, A.L.; Staswick, P.E.; He, S.Y.; Howe, G.A. COI1 is a critical component of a receptor for jasmonate and the bacterial virulence factor coronatine. Proc. Natl. Acad. Sci. USA 2008, 105, 7100–7105. [Google Scholar] [CrossRef]
- Lindfors, A.; Kuusela, H.; Hohtola, A.; Kupila-Ahvenniemi, S. Molecular correlates of tissue browning and deterioration in Scots pine calli. Biol. Plant. 1990, 32, 171–180. [Google Scholar] [CrossRef]
- Zhao, J.-L.; Zhou, L.-G.; Wu, J.-Y. Effects of biotic and abiotic elicitors on cell growth and tanshinone accumulation in Salvia miltiorrhiza cell cultures. Appl. Microbiol. Biotechnol. 2010, 87, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, J.; Wang, X.D.; Zhao, B.; Wang, Y. C Enhanced production of phenylethanoid glycosides by precursor feeding to cell culture of Cistanche deserticola. Process Biochem. 2005, 40, 3480–3484. [Google Scholar] [CrossRef]
- Vasilev, N.; Nedialkov, P.; Ionkova, I.; Ninov, S. HPTLC densitomeric determination of justicidin B in Linum in vitro cultures. Die Pharm. 2004, 59, 528–529. [Google Scholar]
- Da Silva, R.; Ruas, M.M.; Donate, P.M. Complete assignments of 1 H and 13 C NMR spectral data for arylnaphthalene lignan lactones. Magn. Reson. Chem. 2007, 45, 902–904. [Google Scholar] [CrossRef]
- Eom, S.H.; Kim, H.; Hyun, T.K. The cinnamyl alcohol dehydrogenase (CAD) gene family in flax (Linum usitatissimum L.): In-sight from expression profiling of cads induced by elicitors in cultured flax cells. Arch. Biol. Sci. 2016, 68, 603–612. [Google Scholar] [CrossRef]
- Le Roy, J.; Blervacq, A.-S.; Créach, A.; Huss, B.; Hawkins, S.; Neutelings, G. Spatial regulation of monolignol biosynthesis and laccase genes control developmental and stress-related lignin in flax. BMC Plant Biol. 2017, 17, 1–20. [Google Scholar] [CrossRef]
- Corbin, C.; Drouet, S.; Markulin, L.; Auguin, D.; Lainé, É.; Davin, L.B.; Cort, J.R.; Lewis, N.G.; Hano, C. A genome-wide analysis of the flax (Linum usitatissimum L.) dirigent protein family: From gene identification and evolution to differential regulation. Plant Mol. Biol. 2018, 97, 73–101. [Google Scholar] [CrossRef]
- Dalisay, D.S.; Kim, K.W.; Lee, C.; Yang, H.; Rübel, O.; Bowen, B.P.; Davin, L.B.; Lewis, N.G. Dirigent Protein-Mediated Lignan and Cyanogenic Glucoside Formation in Flax Seed: Integrated Omics and MALDI Mass Spectrometry Imaging. J. Nat. Prod. 2015, 78, 1231–1242. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A.T.; Gang, D.R.; Davin, L.B.; Bedgar, D.L.; Chu, A.; Lewis, N.G. (+)-Pinoresinol/(+)-Lariciresinol Reductase from Forsythia intermedia. J. Biol. Chem. 1996, 271, 29473–29482. [Google Scholar] [CrossRef]
- Xia, Z.-Q.; Costa, M.A.; Pélissier, H.C.; Davin, L.B.; Lewis, N.G. Secoisolariciresinol Dehydrogenase Purification, Cloning, and Functional Expression. J. Biol. Chem. 2001, 276, 12614–12623. [Google Scholar] [CrossRef]
- Gang, D.R.; Costa, M.A.; Fujita, M.; Dinkova-Kostova, A.T.; Wang, H.-B.; Burlat, V.; Martin, W.; Sarkanen, S.; Davin, L.B.; Lewis, N.G. Regiochemical control of monolignol radical coupling: A new paradigm for lignin and lignan biosynthesis. Chem. Biol. 1999, 6, 143–151. [Google Scholar] [CrossRef]
- Xia, Z.-Q.; Costa, M.A.; Proctor, J.; Davin, L.B.; Lewis, N.G. Dirigent-mediated podophyllotoxin biosynthesis in Linum flavum and Podophyllum peltatum. Phytochemistry 2000, 55, 537–549. [Google Scholar] [CrossRef]
- Hoffmann, L.; Maury, S.; Martz, F.; Geoffroy, P.; Legrand, M. Purification, cloning, and properties of an acyltransferase controlling shikimate and quinate ester intermediates in phenylpropanoid metabolism. J. Biol. Chem. 2003, 278, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Lepelley, M.; Cheminade, G.; Tremillon, N.; Simkin, A.; Caillet, V.; McCarthy, J. Chlorogenic acid synthesis in coffee: An analysis of CGA content and real-time RT-PCR expression of HCT, HQT, C3H1, and CCoAOMT1 genes during grain development in C. canephora. Plant Sci. 2007, 172, 978–996. [Google Scholar] [CrossRef]
- Nakatsubo, T.; Mizutani, M.; Suzuki, S.; Hattori, T.; Umezawa, T. Characterization of Arabidopsis thaliana Pinoresinol Reductase, a New Type of Enzyme Involved in Lignan Biosynthesis. J. Biol. Chem. 2008, 283, 15550–15557. [Google Scholar] [CrossRef] [PubMed]
- Murthy, H.N.; Lee, E.-J.; Paek, K.-Y. Production of secondary metabolites from cell and organ cultures: Strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tissue Organ Cult. (PCTOC) 2014, 118, 1–16. [Google Scholar] [CrossRef]
- Ahmad, T.A.; Al-Mahdawe, M.M.; Nadir, D.S. Effect of methyl jasmonate on the production of furanocoumarins in cell suspension cultures of Ruta graveolens L. PCTOC 2020, 143, 565–571. [Google Scholar] [CrossRef]
- Wasternack, C.; Hause, B. Jasmonates: Biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann. Bot. 2013, 111, 1021–1058. [Google Scholar] [CrossRef]
- Weiler, E.W.; Kutchan, T.M.; Gorba, T.; Brodschelm, W.; Niesel, U.; Bublitz, F. The Pseudomonas phytotoxin coronatine mimics octadecanoid signalling molecules of higher plants. FEBS Lett. 1994, 345, 9–13. [Google Scholar] [CrossRef]
- Baldi, A.; Dixit, V. Yield enhancement strategies for artemisinin production by suspension cultures of Artemisia annua. Bioresour. Technol. 2008, 99, 4609–4614. [Google Scholar] [CrossRef]
- Vaccaro, M.C.; Mariaevelina, A.; Malafronte, N.; De Tommasi, N.; Leone, A. Increasing the synthesis of bioactive abietane diterpenes in Salvia sclarea hairy roots by elicited transcriptional reprogramming. Plant Cell Rep. 2016, 36, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, L.; Morreel, K.; De Witte, E.; Lammertyn, F.; Van Montagu, M.; Boerjan, W.; Inzé, D.; Goossens, A. Mapping methyl jasmonate-mediated transcriptional reprogramming of metabolism and cell cycle progression in cultured Arabidopsis cells. Proc. Natl. Acad. Sci. USA 2008, 105, 1380–1385. [Google Scholar] [CrossRef] [PubMed]
- Almagro, L.; Belchí-Navarro, S.; Martínez-Márquez, A.; Bru, R.; Pedreño, M.A. Enhanced extracellular production of trans-resveratrol in Vitis vinifera suspension cultured cells by using cyclodextrins and coronatine. Plant Physiol. Biochem. 2015, 97, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Ali, R.; Ma, W.; Lemtiri-Chlieh, F.; Tsaltas, D.; Leng, Q.; Von Bodman, S.; Berkowitz, G.A. Death Don’t Have No Mercy and Neither Does Calcium: Arabidopsis cyclic nucleotide gated channel2 and Innate Immunity. Plant Cell 2007, 19, 1081–1095. [Google Scholar] [CrossRef]
- Belhadj, A.; Telef, N.; Saigne, C.; Cluzet, S.; Barrieu, F.; Hamdi, S.; Mérillon, J.-M. Effect of methyl jasmonate in combination with carbohydrates on gene expression of PR proteins, stilbene and anthocyanin accumulation in grapevine cell cultures. Plant Physiol. Biochem. 2008, 46, 493–499. [Google Scholar] [CrossRef]
- Ramirez-Estrada, K.; Vidal-Limon, H.; Hidalgo, D.; Moyano, E.; Golenioswki, M.; Cusidó, R.M.; Palazon, J. Elicitation, an Effective Strategy for the Biotechnological Production of Bioactive High-Added Value Compounds in Plant Cell Factories. Molecules 2016, 21, 182. [Google Scholar] [CrossRef]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef]
- Gabr, A.M.; Mabrok, H.B.; Ghanem, K.Z.; Blaut, M.; Smetanska, I. Lignan accumulation in callus and Agrobacterium rhizogenes-mediated hairy root cultures of flax (Linum usitatissimum). PCTOC 2016, 126, 255–267. [Google Scholar] [CrossRef]
- Ali, M.B.; Abbasi, B.H.; Haq, I.U. Production of commercially important secondary metabolites and antioxidant activity in cell suspension cultures of Artemisia absinthium L. Ind. Crop. Prod. 2013, 49, 400–406. [Google Scholar] [CrossRef]
- Zahir, A.; Abbasi, B.H.; Adil, M.; Anjum, S.; Zia, M.; Haq, I.U. Synergistic Effects of Drought Stress and Photoperiods on Phenology and Secondary Metabolism of Silybum marianum. Appl. Biochem. Biotechnol. 2014, 174, 693–707. [Google Scholar] [CrossRef]
- Stiles, A.R.; Liu, C.Z. Hairy Root Culture: Bioreactor design and process intensification. Adv. Biochem. Eng. Biotechnol. 2013, 134, 91–114. [Google Scholar]
- Momekov, G.; Yossifov, D.; Guenova, M.; Michova, A.; Stoyanov, N.; Konstantinov, S.; Ionkov, T.; Sacheva, P.; Ionkova, I. Apoptotic mechanisms of the biotechnologically produced arylnaphtalene lignan justicidin B in the acute myeloid leukemia-derived cell line HL-60. Pharmacol. Rep. 2014, 66, 1073–1076. [Google Scholar] [CrossRef]
- Rontein, D.; Dieuaide-Noubhani, M.; Dufourc, E.J.; Raymond, P.; Rolin, D. The metabolic architecture of plant cells: Stability of central metabolism and flexibility of anabolic pathways during the growth cycle of tomato cells. J. Biol. Chem. 2002, 277, 43948–43960. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, L.; Wang, L.; Tan, M.; Ogutu, C.O.; Yin, Z.; Zhou, J.; Wang, J.; Wang, L.; Yan, X. Transcriptome analysis and molecular mechanism of linseed (Linum usitatissimum L.) drought tolerance under repeated drought using single-molecule long-read sequencing. BMC Genom. 2021, 22, 1–23. [Google Scholar] [CrossRef]
- Uppalapati, S.R.; Ayoubi, P.; Weng, H.; Palmer, D.A.; Mitchell, R.E.; Jones, W.; Bender, C.L. The phytotoxin coronatine and methyl jasmonate impact multiple phytohormone pathways in tomato. Plant J. 2005, 42, 201–217. [Google Scholar] [CrossRef]
- Wang, Z.; Hobson, N.; Galindo, L.; Zhu, S.; Shi, D.; McDill, J.; Yang, L.; Hawkins, S.; Neutelings, G.; Datla, R.; et al. The genome of flax (Linum usitatissimum) assembled de novo from short shotgun sequence reads. Plant J. 2012, 72, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Dash, P.K.; Rai, R.; Mahato, A.K.; Gaikwad, K.; Singh, N.K. Transcriptome Landscape at Different Developmental Stages of a Drought Tolerant Cultivar of Flax (Linum usitatissimum). Front. Chem. 2017, 5, 82. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, A.A.; Kudryavtseva, A.V.; Krasnov, G.S.; Koroban, N.V.; Speranskaya, A.S.; Krinitsina, A.A.; Belenikin, M.S.; Snezhkina, A.V.; Sadritdinova, A.F.; Kishlyan, N.V.; et al. Gene expression profiling of flax (Linum usitatissimum L.) under edaphic stress. BMC Plant Biol. 2016, 16, 139–146. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, X.; Yan, H.; Li, W.; Li, Y.; Cai, R.; Xiang, Y. The Lipoxygenase Gene Family in Poplar: Identification, Classification, and Expression in Response to MeJA Treatment. PLoS ONE 2015, 10, e0125526. [Google Scholar] [CrossRef]
- Sun, G.; Yang, Y.; Xie, F.; Wen, J.F.; Wu, J.; Wilson, I.W.; Tang, Q.; Liu, H.; Qiu, D. Deep sequencing reveals transcriptome re-programming of Taxus × media cells to the elicitation with methyl jasmonate. PLoS ONE 2013, 8, e62865. [Google Scholar]
- Cai, Z.; Kastell, A.; Knorr, D.; Smetanska, I. Exudation: An expanding technique for continuous production and release of secondary metabolites from plant cell suspension and hairy root cultures. Plant Cell Rep. 2011, 31, 461–477. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Kajala, K.; Coil, D.A.; Brady, S.M. Draft Genome Sequence of Rhizobium rhizogenes Strain ATCC 15834. Genome Announc. 2014, 2, e01108–e01114. [Google Scholar] [CrossRef] [PubMed]
- Vaira, A.M.; Semeria, L.; Crespi, S.; Lisa, V.; Allavena, A.; Accotto, G.P. Resistance to tospoviruses in Nicotiana benthamiana transformed with the N gene of tomato spotted wilt virus: Correlation between transgene expression and protection in primary transformants. Mol. Plant Microbe Interact. 1995, 8, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Blom, T.J.M.; Kreis, W.; Van Iren, F.; Libbenga, K.R. A non-invasive method for the routine-estimation of fresh weight of cells grown in batch suspension cultures. Plant Cell Rep. 1992, 11, 146–149. [Google Scholar] [CrossRef]
- Baker, C.J.; Mock, N.M. An improved method for monitoring cell death in cell suspension and leaf disc assays using evans blue. Plant Cell Tissue Organ Cult. 1994, 39, 7–12. [Google Scholar] [CrossRef]
- Steponkus, P.L.; Lanphear, F.O. Refinement of the Triphenyl Tetrazolium Chloride Method of Determining Cold Injury. Plant Physiol. 1967, 42, 1423–1426. [Google Scholar] [CrossRef] [PubMed]
- Narayani, M.; Srivastava, S. Elicitation: A stimulation of stress in in vitro plant cell/tissue cultures for enhancement of secondary metabolite production. Phytochem. Rev. 2017, 16, 1227–1252. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Gillespie, K.M. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin–Ciocalteu reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef]
- Cheng, Z.; Moore, A.J.; Yu, L. High-Throughput Relative DPPH Radical Scavenging Capacity Assay. J. Agric. Food Chem. 2006, 54, 7429–7436. [Google Scholar] [CrossRef]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, H.P.L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 2016, 34, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Tarazona, S.; Furió-Tarí, P.; Turrà, D.; Pietro, A.D.; Nueda, M.J.; Ferrer, A.; Conesa, A. Data quality aware analysis of differential expression in RNA-seq with NOISeq R/Bioc package. Nucleic Acids Res. 2015, 43, e140. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mascheretti, I.; Alfieri, M.; Lauria, M.; Locatelli, F.; Consonni, R.; Cusano, E.; Dougué Kentsop, R.A.; Laura, M.; Ottolina, G.; Faoro, F.; et al. New Insight into Justicidin B Pathway and Production in Linum austriacum. Int. J. Mol. Sci. 2021, 22, 2507. https://doi.org/10.3390/ijms22052507
Mascheretti I, Alfieri M, Lauria M, Locatelli F, Consonni R, Cusano E, Dougué Kentsop RA, Laura M, Ottolina G, Faoro F, et al. New Insight into Justicidin B Pathway and Production in Linum austriacum. International Journal of Molecular Sciences. 2021; 22(5):2507. https://doi.org/10.3390/ijms22052507
Chicago/Turabian StyleMascheretti, Iride, Michela Alfieri, Massimiliano Lauria, Franca Locatelli, Roberto Consonni, Erica Cusano, Roméo A. Dougué Kentsop, Marina Laura, Gianluca Ottolina, Franco Faoro, and et al. 2021. "New Insight into Justicidin B Pathway and Production in Linum austriacum" International Journal of Molecular Sciences 22, no. 5: 2507. https://doi.org/10.3390/ijms22052507
APA StyleMascheretti, I., Alfieri, M., Lauria, M., Locatelli, F., Consonni, R., Cusano, E., Dougué Kentsop, R. A., Laura, M., Ottolina, G., Faoro, F., & Mattana, M. (2021). New Insight into Justicidin B Pathway and Production in Linum austriacum. International Journal of Molecular Sciences, 22(5), 2507. https://doi.org/10.3390/ijms22052507









