Acute Tubulointerstitial Nephritis in Clinical Oncology: A Comprehensive Review
Abstract
1. Introduction
2. Drug-Related ATIN
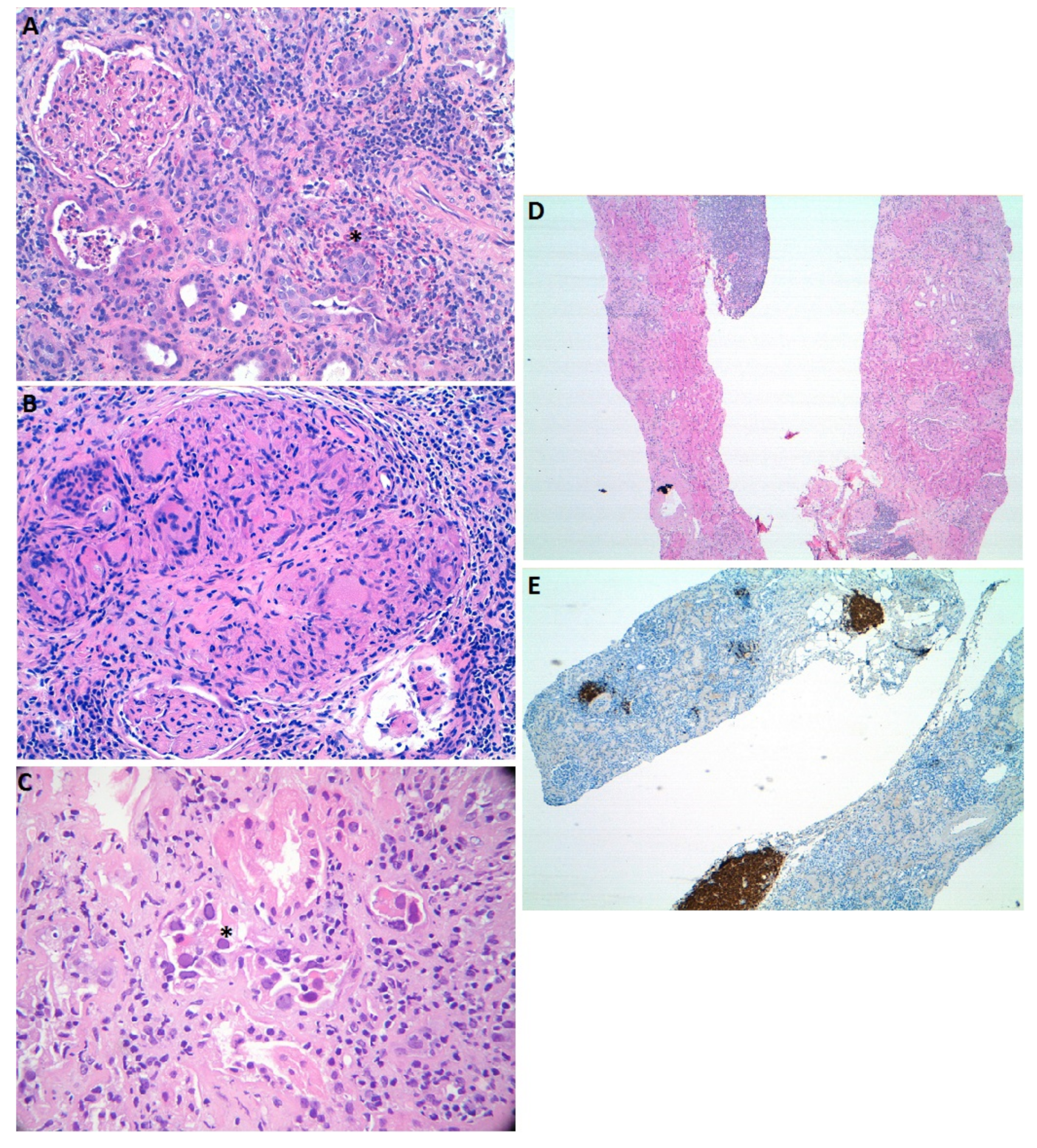
2.1. Immune Checkpoint Inhibitors
2.2. Platinum Agents
2.3. Tyrosine Kinase Inhibitors
2.4. Ifosfamide
2.5. Lenalidomide
2.6. Bacillus Calmette–Guerin (BCG)
2.7. Chimeric Antigen Receptor–Modified T Cells (CAR-T Cells)
3. Infection-Associated ATIN
4. Hematological Malignancies and ATIN
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Raghavan, R.; Eknoyan, G. Acute interstitial nephritis—A reappraisal and update. Clin. Nephrol. 2014, 82, 149–162. [Google Scholar] [CrossRef]
- Eddy, A.A. Drug-induced tubulointerstitial nephritis: Hypersensitivity and necroinflammatory pathways. Pediatr. Nephrol. 2019, 35, 547–554. [Google Scholar] [CrossRef]
- Praga, M.; González, E. Acute interstitial nephritis. Kidney Int. 2010, 77, 956–961. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.L.C.; de Brito, B.B.; da Silva, F.A.F.; dos Santos Botelho, A.C.; de Melo, F.F. Nephrotoxicity in cancer treatment: An overview. World J. Clin. Oncol. 2020, 11, 190–204. [Google Scholar] [CrossRef] [PubMed]
- Chambers, W.H.; Rabinowich, H.; Herberman, R.B. Mechanisms of Immunosuppression. In Holland-Frei Cancer Medicine, 6th ed.; BC Decker Inc.: Hamilton, ON, Canada, 2003. [Google Scholar]
- Iványi, B.; Hamilton-Dutoit, S.J.; Hansen, H.E.; Olsen, S. Acute tubulointerstitial nephritis: Phenotype of infiltrating cells and prognostic impact of tubulitis. Virchows Arch. 1996, 428, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Kang, K.P.; Kim, W.; Park, S.K.; Lee, S. Nonsteroidal antiinflammatory drug induced acute granulomatous interstitial nephritis. BMC Res. Notes 2015, 8, 793. [Google Scholar] [CrossRef] [PubMed]
- Härmark, L.; van der Wiel, H.E.; de Groot, M.C.H.; van Grootheest, A.C. Proton pump inhibitor-induced acute interstitial nephritis. Br. J. Clin. Pharmacol. 2007, 64, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Mac, K.; Chavada, R.; Paull, S.; Howlin, K.; Wong, J. Cefepime induced acute interstitial nephritis—A case report. BMC Nephrol. 2015, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Chatzikyrkou, C.; Hamwi, I.; Clajus, C.; Becker, J.; Hafer, C.; Kielstein, J.T. Biopsy proven acute interstitial nephritis after treatment with moxifloxacin. BMC Nephrol. 2010, 11, 19. [Google Scholar] [CrossRef]
- Sury, K.; Perazella, M.A. The nephrotoxicity of new immunotherapies. Expert Rev. Clin. Pharmacol. 2019, 12, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-P.; Jung, H. Clinical pharmacologic aspects of immune checkpoint inhibitors in cancer therapy. Transl. Clin. Pharmacol. TCP 2016, 7, 7–12. [Google Scholar] [CrossRef]
- Abdel-Rahman, O.; Fouad, M. A network meta-analysis of the risk of immune-related renal toxicity in cancer patients treated with immune checkpoint inhibitors. Immunotherapy 2016, 8, 665–674. [Google Scholar] [CrossRef]
- Mamlouk, O.; Selamet, U.; Machado, S.; Abdelrahim, M.; Glass, W.F.; Tchakarov, A.; Gaber, L.; Lahoti, A.; Workeneh, B.; Chen, S.; et al. Nephrotoxicity of immune checkpoint inhibitors beyond tubulointerstitial nephritis: Single-center experience. J. Immunother. Cancer 2019, 7, 2. [Google Scholar] [CrossRef]
- Cortazar, F.B.; Marrone, K.A.; Troxell, M.L.; Ralto, K.M.; Hoenig, M.P.; Brahmer, J.R.; Le, D.T.; Lipson, E.J.; Glezerman, I.G.; Wolchok, J.; et al. Clinicopathological features of acute kidney injury associated with immune checkpoint inhibitors HHS Public Access. Kidney Int. 2016, 90, 638–647. [Google Scholar] [CrossRef]
- Koda, R.; Watanabe, H.; Tsuchida, M.; Iino, N.; Suzuki, K.; Hasegawa, G.; Imai, N.; Narita, I. Immune checkpoint inhibitor (nivolumab)-associated kidney injury and the importance of recognizing concomitant medications known to cause acute tubulointerstitial nephritis: A case report. BMC Nephrol. 2018, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Seethapathy, H.; Zhao, S.; Chute, D.F.; Zubiri, L.; Oppong, Y.; Strohbehn, I.; Cortazar, F.B.; Leaf, D.E.; Mooradian, M.J.; Villani, A.-C.; et al. The Incidence, Causes, and Risk Factors of Acute Kidney Injury in Patients Receiving Immune Checkpoint Inhibitors. Clin. J. Am. Soc. Nephrol. 2019, 14, 1692–1700. [Google Scholar] [CrossRef] [PubMed]
- Cortazar, F.B.; Kibbelaar, Z.A.; Glezerman, I.G.; Abudayyeh, A.; Mamlouk, O.; Motwani, S.S.; Murakami, N.; Herrmann, S.M.; Manohar, S.; Shirali, A.C.; et al. Clinical Features and Outcomes of Immune Checkpoint Inhibitor–Associated AKI: A Multicenter Study. J. Am. Soc. Nephrol. 2020, 31, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Draibe, J.B.; García-Carro, C.; Martinez-Valenzuela, L.; Agraz, I.; Fulladosa, X.; Bolufer, M.; Tango, A.; Torras, J.; Soler, M.J. Acute tubulointerstitial nephritis induced by checkpoint inhibitors versus classical acute tubulointerstitial nephritis: Are they the same disease? Clin. Kidney J. 2020. [Google Scholar] [CrossRef]
- Yamada, S.; Yazawa, M.; Yamamoto, M.; Koitabashi, K.; Ichikawa, D.; Koike, J.; Shibagaki, Y. A case of biopsy-proven oxaliplatin-induced acute tubulointerstitial nephritis with thrombocytopenia and anemia. CEN Case Rep. 2019, 8, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-J.; Oh, K.-H.; Kang, H.-R.; Lee, S.-J. Oxaliplatin-induced acute tubulointerstitial nephritis: Two case reports. Clin. Nephrol. 2018, 89, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Asai, A.; Katsuno, T.; Yamaguchi, M.; Iwagaitsu, S.; Nobata, H.; Kinashi, H.; Kitamura, H.; Banno, S.; Ito, Y. Carboplatin-related acute interstitial nephritis in a patient with pancreatic neuroendocrine tumor. CEN Case Rep. 2019, 9, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.T.; Haap, M.; Kopp, H.-G.; Lipp, H.-P. Tyrosine Kinase Inhibitors—A Review on Pharmacology, Metabolism and Side Effects. Curr. Drug Metab. 2009, 10, 470–481. [Google Scholar] [CrossRef]
- Winn, S.K.; Ellis, S.; Savage, P.; Sampson, S.; Marsh, J.E. Biopsy-proven acute interstitial nephritis associated with the tyrosine kinase inhibitor sunitinib: A class effect? Nephrol. Dial. Transplant. 2009, 24, 673–675. [Google Scholar] [CrossRef]
- Azar, I.; Esfandiarifard, S.; Sinai, P.; Wazir, A.; Foulke, L.; Mehdi, S. Sunitinib-Induced Acute Interstitial Nephritis in a Thrombocytopenic Renal Cell Cancer Patient. Case Rep. Oncol. Med. 2017, 2017, 1–4. [Google Scholar] [CrossRef]
- Izzedine, H.; Brocheriou, I.; Rixe, O.; Deray, G. Interstitial nephritis in a patient taking sorafenib. Nephrol. Dial. Transplant. 2007, 22, 2411. [Google Scholar] [CrossRef]
- Khurana, A. Allergic interstitial nephritis possibly related to sunitinib use. Am. J. Geriatr. Pharmacother. 2007, 5, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Prabhash, K.; Noronha, V.; Punatar, S.; Joshi, A.; Desphande, R.V. Sunitinib-induced thrombotic microangiopathy. J. Cancer Res. Ther. 2016, 12, 6. [Google Scholar] [CrossRef] [PubMed]
- Higgs, D.; Nagy, C.; Einhorn, L.H. Ifosfamide: A clinical review. Semin. Oncol. Nurs. 1989, 5, 70–77. [Google Scholar] [CrossRef]
- Baraldi, O.; Comai, G.; Cuna, V.; Palmerini, E.; Gasperoni, L.; Chiocchini, A.L.C.; Malvi, D.; Fabbrizio, B.; Ferrari, D.; La Manna, G. Ifosfamide-induced karyomegalic interstitial nephritis in Ewing’s sarcoma and osteosarcoma: Report of two cases. Gazz. Med. Ital. Arch. Sci. Med. 2018, 177, 651–657. [Google Scholar]
- Priyamvada, P.S.; Jayasurya, R.; Srinivas, B.H.; Ponraj, M.; Haridasan, S.; Parameswaran, S. Karyomegalic interstitial nephropathy following ifosfamide therapy. Indian J. Nephrol. 2016, 26, 294–297. [Google Scholar] [CrossRef]
- Matsuura, T.; Wakino, S.; Yoshifuji, A.; Nakamura, T.; Tokuyama, H.; Hashiguchi, A.; Konishi, K.; Iwasa, T.; Shoji, M.; Hosono, A.; et al. Improvement in karyomegalic interstitial nephritis three years after ifosfamide and cisplatin therapy by corticosteroid. CEN Case Rep. 2014, 3, 226–231. [Google Scholar] [CrossRef]
- Kheder El-Fekih, R.; Izzedine, H. Lenalidomide nephrotoxicity. Bull. Cancer 2016, 103, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Dinh, A.; Palmer, M.B.; Svoboda, J.; Hogan, J.J. Lenalidomide-induced tubulointerstitial nephritis in Hodgkin lymphoma. Leuk. Lymphoma 2019, 60, 3084–3086. [Google Scholar] [CrossRef] [PubMed]
- Shaaban, H.; Layne, T.; Guron, G. A case of DRESS (drug reaction with eosinophilia and systemic symptoms) with acute interstitial nephritis secondary to lenalidomide. J. Oncol. Pharm. Pract. 2013, 20, 302–304. [Google Scholar] [CrossRef]
- Shanbhag, A.; Pritchard, E.R.; Chatterjee, K.; Hammond, D.A. Highly Probable Drug Reaction with Eosinophilia and Systemic Symptoms Syndrome Associated with Lenalidomide. Hosp. Pharm. 2017, 52, 408–411. [Google Scholar] [CrossRef]
- Morales, A. BCG: A throwback from the stone age of vaccines opened the path for bladder cancer immunotherapy. Can. J. Urol. 2017, 24, 8788–8793. [Google Scholar]
- Mohammed, A.; Arastu, Z. Emerging concepts and spectrum of renal injury following Intravesical BCG for non-muscle invasive bladder cancer. BMC Urol. 2017, 17, 114. [Google Scholar] [CrossRef]
- Bernatowska, E.A.; Wolska-Kuśnierz, B.; Pac, M.; Kurenko-Deptuch, M.; Zwolska, Z.; Casanova, J.-L.; Piątosa, B.; Van Dongen, J.; Roszkowski, K.; Mikoluc, B.; et al. Disseminated Bacillus Calmette-Guérin Infection and Immunodeficiency. Emerg. Infect. Dis. 2007, 13, 799–801. [Google Scholar] [CrossRef]
- Ziegler, J.; Ho, J.; Gibson, I.W.; Nayak, J.G.; Stein, M.; Walkty, A.; Orr, P. Disseminated Mycobacterium bovis infection post-kidney transplant following remote intravesical BCG therapy for bladder cancer. J. Urol. 2019, 202, 457. [Google Scholar] [CrossRef] [PubMed]
- Kheder-Elfekih, R.; Selmi, Y.; Jebali, H.; Ben Fatma, L.; Smaoui, W.; Krid, M.; Beji, S.; Rais, L.; Zouaghi, M.K. A case of renal granulomatosis with polyangiitis following intravesical bacillus Calmette-Guérin therapy. Saudi J. Kidney Dis. Transplant. 2018, 29, 185–188. [Google Scholar] [CrossRef]
- Singh, N.P.; Prakash, A.; Kubba, S.; Ganguli, A.; Agarwal, S.K.; Dinda, A.K.; Aggarwal, P.N. Nephrotic Syndrome as a Complication of Intravesical BCG Treatment of Transitional Cell Carcinoma of Urinary Bladder. Ren. Fail. 2007, 29, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.; Xu, H. A new insight in chimeric antigen receptor-engineered T cells for cancer immunotherapy. J. Hematol. Oncol. 2017, 10, 1–11. [Google Scholar] [CrossRef]
- Mori, K.; Yoshihara, T.; Nishimura, Y.; Uchida, M.; Katsura, K.; Kawase, Y.; Hatano, I.; Ishida, H.; Chiyonobu, T.; Kasubuchi, Y.; et al. Acute renal failure due to adenovirus-associated obstructive uropathy and necrotizing tubulointerstitial nephritis in a bone marrow transplant recipient. Bone Marrow Transplant. 2003, 31, 1173–1176. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Keddis, M.; Leung, N.; Herrmann, S.; El-Zoghby, Z.; Sethi, S. Adenovirus-Induced Interstitial Nephritis Following Umbilical Cord Blood Transplant for Chronic Lymphocytic Leukemia. Am. J. Kidney Dis. 2012, 59, 886–890. [Google Scholar] [CrossRef]
- Lion, T. Adenovirus Infections in Immunocompetent and Immunocompromised Patients. Clin. Microbiol. Rev. 2014, 27, 441–462. [Google Scholar] [CrossRef] [PubMed]
- Rady, K.; Walters, G.; Brown, M.; Talaulikar, G. Allograft adenovirus nephritis. Clin. Kidney J. 2014, 7, 289–292. [Google Scholar] [CrossRef]
- Lamarche, C.; Orio, J.; Collette, S.; Senécal, L.; Hébert, M.J.; Renoult, É.; Tibbles, L.A. and Delisle, J.S. BK Polyomavirus and the Transplanted Kidney: Immunopathology and Therapeutic Approaches. Transplantation 2016, 100, 2276–2287. [Google Scholar] [CrossRef]
- Stracke, S.; Helmchen, U.; Von Müller, L.; Bunjes, D.; Keller, F. Polyoma virus-associated interstitial nephritis in a patient with acute myeloic leukaemia and peripheral blood stem cell transplantation. Nephrol. Dial. Transplant. 2003, 18, 2431–2433. [Google Scholar] [CrossRef][Green Version]
- Yamanaka, K.; Oka, K.; Nakazawa, S.; Hirai, T.; Kishikawa, H.; Nishimura, K.; Kyo, M.; Ichikawa, Y. Immunohistochemical features of BK virus nephropathy in renal transplant recipients. Clin. Transplant. 2012, 26, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Ogura, M.; Kagami, S.; Nakao, M.; Kono, M.; Kanetsuna, Y.; Hosoya, T. Fungal granulomatous interstitial nephritis presenting as acute kidney injury diagnosed by renal histology including PCR assay. Clin. Kidney J. 2012, 5, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Shalin, S.C.; Ferringer, T.; Cassarino, D.S. PAS and GMS utility in dermatopathology: Review of the current medical literature. J. Cutan. Pathol. 2020, 47, 1096–1102. [Google Scholar] [CrossRef]
- Heggermont, W.A.; Verhoef, G.; Evenepoel, P.; Sprangers, B.; Lerut, E.; Tousseyn, T.; Claes, K. Clinical case report: A rare cause of acute kidney failure—Tissue is the issue. Acta Clin. Belg. Int. J. Clin. Lab. Med. 2016, 72, 201–204. [Google Scholar] [CrossRef]
- DeDe, F.; Yılmaz, B.; Aylı, D.; Kayatas, M.; Atılgan, G.; Caner, S.; Akyurek, N.; Odabaş, A.R. Acute Tubulo-Interstitiel Nephritis Associated with Diffuse Large B-Cell Lymphoma Presenting as Acute Renal Failure. Ren. Fail. 2008, 30, 465–467. [Google Scholar] [CrossRef] [PubMed]
- Biro, E.; Szikszay, E.; Pethő-Orosz, P.; Bigida, L.; Balla, G.; Szabó, T. Acute interstitial nephritis in T-cell leukemia in a pediatric patient. Pediatr. Int. 2016, 58, 940–942. [Google Scholar] [CrossRef]
- Gu, X.; Herrera, G.A. Light-Chain–Mediated Acute Tubular Interstitial Nephritis: A Poorly Recognized Pattern of Renal Disease in Patients with Plasma Cell Dyscrasia. Arch. Pathol. Lab. Med. 2006, 130, 165–169. [Google Scholar] [CrossRef]
- Herrera, G.A.; Joseph, L.; Gu, X.; Hough, A.; Barlogie, B. Renal Pathologic Spectrum in an Autopsy Series of Patients with Plasma Cell Dyscrasia. Arch. Pathol. Lab. Med. 2004, 128, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Momeni, A.; Hajigholami, A.; Nasri, H. Multiple myeloma presenting as acute tubulointerstitial nephritis and normal serum protein electrophoresis. Iran. J. Kidney Dis. 2012, 6, 69–72. [Google Scholar] [PubMed]
- Ardalan, M.R.; Shoja, M.M. Multiple myeloma presented as acute interstitial nephritis and rheumatoid arthritis-like polyarthritis. Am. J. Hematol. 2007, 82, 309–313. [Google Scholar] [CrossRef]
- Kakareko, K.; Rydzewska-Rosołowska, A.; Hryszko, T.; Zalewski, G.; Naumnik, B. One kidney biopsy—double trouble: Acute tubulo-interstitial nephritis associated with mantle cell lymphoma. Pol. Arch. Intern. Med. 2018, 128, 709–710. [Google Scholar] [CrossRef]
- Kamat, A.V.; Goldsmith, D.; O’Donnell, P.; Van Der Walt, J.; Carr, R. Renal Failure with Granulomatous Interstitial Nephritis and Diffuse Leukemic Renal Infiltration in Chronic Lymphocytic Leukemia. Ren. Fail. 2007, 29, 763–765. [Google Scholar] [CrossRef] [PubMed]
- Nassereddine, S.; Rafei, H.; Elbahesh, E.; Tabbara, I. Acute Graft Versus Host Disease: A Comprehensive Review. Anticancer. Res. 2017, 37, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Wanchoo, R.; Stotter, B.R.; Bayer, R.L.; Jhaveri, K.D. Acute kidney injury in hematopoietic stem cell transplantation. Curr. Opin. Crit. Care 2019, 25, 531–538. [Google Scholar] [CrossRef]
- El Seisi, S.; Gupta, R.; Clase, C.M.; Forrest, D.L.; Milandinovic, M.; Couban, S. Renal pathology at autopsy in patients who died after hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2003, 9, 683–688. [Google Scholar] [CrossRef]
- Homma, C.I.; Kami, M.; Masuo, S.; Sakiyama, M.; Kojima, R.; Hori, A.; Kusumi, E.; Katori, H.; Takeuchi, K.; Kishi, Y.; et al. Graft-versus-host disease of the kidney after rapid tapering of cyclosporin following reduced intensity hematopoietic stem cell transplantation. Bone Marrow Transplant. 2005, 35, 929–930. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kusumi, E.; Kami, M.; Hara, S.; Hoshino, J.; Yamaguchi, Y.; Murashige, N.; Kishi, Y.; Shibagaki, Y.; Shibata, T.; Matsumura, T.; et al. Postmortem examination of the kidney in allogeneic hematopoietic stem cell transplantation recipients: Possible involvement of graft-versus-host disease. Int. J. Hematol. 2008, 87, 225–230. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Valenzuela, L.; Draibe, J.; Fulladosa, X.; Gomà, M.; Gómez, F.; Antón, P.; Cruzado, J.M.; Torras, J. Acute Tubulointerstitial Nephritis in Clinical Oncology: A Comprehensive Review. Int. J. Mol. Sci. 2021, 22, 2326. https://doi.org/10.3390/ijms22052326
Martínez-Valenzuela L, Draibe J, Fulladosa X, Gomà M, Gómez F, Antón P, Cruzado JM, Torras J. Acute Tubulointerstitial Nephritis in Clinical Oncology: A Comprehensive Review. International Journal of Molecular Sciences. 2021; 22(5):2326. https://doi.org/10.3390/ijms22052326
Chicago/Turabian StyleMartínez-Valenzuela, Laura, Juliana Draibe, Xavier Fulladosa, Montserrat Gomà, Francisco Gómez, Paula Antón, Josep María Cruzado, and Joan Torras. 2021. "Acute Tubulointerstitial Nephritis in Clinical Oncology: A Comprehensive Review" International Journal of Molecular Sciences 22, no. 5: 2326. https://doi.org/10.3390/ijms22052326
APA StyleMartínez-Valenzuela, L., Draibe, J., Fulladosa, X., Gomà, M., Gómez, F., Antón, P., Cruzado, J. M., & Torras, J. (2021). Acute Tubulointerstitial Nephritis in Clinical Oncology: A Comprehensive Review. International Journal of Molecular Sciences, 22(5), 2326. https://doi.org/10.3390/ijms22052326





