Abstract
Mushroom poisoning has always been a threat to human health. There are a large number of reports about ingestion of poisonous mushrooms every year around the world. It attracts the attention of researchers, especially in the aspects of toxin composition, toxic mechanism and toxin application in poisonous mushroom. Inocybe is a large genus of mushrooms and contains toxic substances including muscarine, psilocybin, psilocin, aeruginascin, lectins and baeocystin. In order to prevent and remedy mushroom poisoning, it is significant to clarify the toxic effects and mechanisms of these bioactive substances. In this review article, we summarize the chemistry, most known toxic effects and mechanisms of major toxic substances in Inocybe mushrooms, especially muscarine, psilocybin and psilocin. Their available toxicity data (different species, different administration routes) published formerly are also summarized. In addition, the treatment and medical application of these toxic substances in Inocybe mushrooms are also discussed. We hope that this review will help understanding of the chemistry and toxicology of Inocybe mushrooms as well as the potential clinical application of its bioactive substances to benefit human beings.
1. Introduction
Poisoning incidents caused by ingesting poisonous mushrooms occur every year all over the world, and a large number of poisoning incidents have been reported. Accidental poisoning by foraged mushrooms can affect health or even lead to death in some cases. Mushroom poisoning is a worldwide food safety issue and it has been one of the main causes of food poisoning deaths.
In recent years, more than 100 kinds of poisonous mushrooms have been found, of which more than 30 have fatal toxicity. Among the many poisonous mushrooms, the genus Inocybe are common poisonous mushrooms. Many species of the genus Inocybe, a highly diverse genus of ectomycorrhizal Agaricales, are world-wide mycorrhizal mushrooms. At present, we mainly distinguish edible fungi and toadstools by morphology, but Inocybe mushrooms are often confused with edible species because of their similar appearance, which is also often the cause of mushroom poisoning [1]. People and animals may show some toxic symptoms after ingesting these mushrooms. The main toxic substances of these fungi are muscarine, psilocybin and psilocin. They act primarily on the central nervous system and may cause a range of neurological symptoms. Muscarine increases parasympathetic tone by M-type of post-ganglionic parasympathetic receptors located in muscles and glands, and it causes parasympathetic nervous excitement, heart rate slows down and weakens, muscle contractions, increased gland secretion and pupils become constricted. Furthermore, it can act on the central nervous system and cause nerve excitement [2]. Muscarine is fatal to human beings if too much is ingested at once. According to the available data, the human lethal dose of muscarine in the aforementioned mushrooms is about 500 g of raw mushroom. However, 10 to 20 g of raw mushroom is enough to lead to poisoning [3]. Thus, eating a small number of poisonous mushrooms by mistake is very likely to be poisonous.
Psilocybin and psilocin are neurohallucinogenic toxins and they cause people to experience hallucinations, uncontrollable laughter, extreme excitement and other strange feelings after ingestion. Therefore, they are also known as “laughing mushrooms” and are banned in many western countries as drugs because of these strange neurological symptoms.
Although these toxins have toxic effects and may threaten human life and health, they have been applied to scientific research and biomedicine with the development and maturity of scientific research. Psilocybin and psilocin, particularly, are widely used in the treatment of neurological diseases, drug addiction and psychotherapy due to their mild toxic effects. Psilocybin and psilocin can change the state of neuroconsciousness and reduce anxiety and depression in psychiatric patients. They are also used as a treatment for Alzheimer’s disease, schizophrenia and other mental and psychiatric disorders. Due to their low toxicity and short duration of action, they are often used as a drug substitute for drug treatment to reduce the pain and dependence of drug addicts.
Mushroom poisoning is closely related to people’s health and life, so it is of great significance to understanding toxic components and toxic types of poisonous mushrooms for prevention and treatment of mushroom poisoning. In this review article, we summarized the chemistry, most known toxic effects and mechanisms of major toxic substances in Inocybe mushrooms, especially muscarine, psilocybin and psilocin. We also discuss the treatment and applications of these toxins. It is hoped that this review can help us to better understand the main bioactive substances and the clinical application of Inocybe, so as to provide some reference for toxin related researchers.
2. Mycology
Inocybe is a large genus of mushrooms in the order Agaricales and family Cortinariaceae (Figure 1). Index Fungorum includes 848 species in Inocybe [4]. The genus Inocybe (Inocybaceae) has a wide distribution in temperate and tropical regions of the Northern Hemisphere. As for morphological characteristics, the fruiting bodies are small. The cap is deep cinnamon-brown to rust-brown oblate with hairy scales, up to 4 cm in diameter and have margin without cracks. Mushroom meat is white and its fold is straight to nearly extended, light rust color, with white fold margin. The mushroom has a cylindrical almost-white stipe. Its ecological habit is to grow in groups in forests. Ryberg and collaborators considered that large parts of Inocybe taxonomy and evolutionary history remain poorly explored [5]. As new species of Inocybe mushrooms are discovered, some confusion around this group of mushrooms is quite understandable.

Figure 1.
Inocybe mushrooms.
3. Inocybe Poisoning
Inocybe mushrooms contain toxic substances, which can cause poisoning at low dose. Due to the similar appearance to many edible mushrooms, they are often eaten by humans and animals by mistake. Published reports of poisoning with Inocybe poisonous fungi are limited to intoxication of dogs and humans.
3.1. Animal Poisoning
In general, the number of reported animal poisonings is relatively low. Toxicology testing is only available for a limited number of fungal toxins. Three cases of dog poisoning with Inocybe asterospora Quel were recorded in China [6,7], and five cases were recorded in Norway [8]. Common clinical findings in dogs were diarrhea, vomiting, ptyalism and tachycardia. The results of the therapy have shown that the prognosis of Inocybe poisoning is very good. All dogs only fully recovered after supportive care.
3.2. Human Poisoning
Poisoning by people with Inocybe mushrooms is not very common. The diagnosis uses the clinical picture and anamnestic data, mycological and toxicological examination of the residues of mushrooms, their spores and toxins [9]. Several published reports of poisonous Inocybe mushrooms were often limited by insufficient identification of the species [1]. Inocybe erubescens (syn. I. patouillardii) (Figure 2) is the most common type of Inocybe associated with toxicity in Europe. Other poisoning of humans was reported in species Inocybe fastigiata [10], Inocybe tristis [11], Inocybe asterospora [7], and Inocybe aeruginascens [12].

Figure 2.
Inocybe erubescens, the most common Inocybe species associated with toxicity in Europe.
Although awareness of poisonous mushrooms is increasing, hospitals annually treat many patients diagnosed with poisoning by toxic fungal species. Inocybe fungal poisoning has been reported in India in 2015 [13]. A total of 11 people were poisoned, including a 6-month-old child. All those treated were hospitalized. Respiratory distress, vomiting, diarrhea and visual disturbances were observed. It is not known how they were treated, but everyone was released from hospital the next day after intoxication. Tropical Inocybe carnosibulbosa CK Pradeep & Matheny, previously unknown in India, have been identified as the cause of poisoning [14]. In 2019 in the province of Ningxia in China, two patients exhibited typical muscarinic symptons after consuming wild mushrooms. The clinical manifestations included chills, sweating, salivation and diarrhea. The incubation period was approximately 2 h. After emergency treatment, they recovered 24 h later. The specimen was identified as Inocybe serotina and muscarine was detected [15]. In China, one of the food poisoning accidents in Panyu City, Guangdong province, attracted people’s attention. Ten migrant workers on a construction site ingested mushrooms which grew on cow dung as their dinner. After eating for about 5 min, some people experienced nausea, dizziness, headache and limb weakness. Two hours later, all the workers had the same symptons. The doctor immediately induced emesis, gastrolavage, activated carbon adsorption and other emergency measures; all the workers recovered and were discharged 3 d later. Experts determined that the cause of poisoning was the psilocybin contained in the mushrooms they ate.
The latency to first signs of poisoning varies between 15 min and 2 h after consumption. Clinical manifestations include nausea, vomiting, abdominal pain, diarrhea, hypersalivation, diaphoresis, hypotension, tearing, blurred vision, miosis, tremors, restlessness, flushing and syncope. Additionally, this sickness has a good prognosis after supportive treatment, including intravenous fluids, antiemetics and 1 mg atropine intravenously. Full recovery is usually within 12 h [16].
4. Inocybe Poisonous Substances
In order to understand the causes and pathogenesis of human poisoning by ingestion of poisonous mushrooms, many researchers have made great progress in exploring the main toxic substances in mushroom. Based on current knowledge, alkaloids are the main toxic principles of the Inocybe mushrooms [17]. The following toxic alkaloids have been found in the Inocybe mushrooms: muscarine (I), psilocin (II) and psilocybin (III) and, aeruginascin (IV) and baeocystin (V). The chemical structures of all known Inocybe-alkaloids are shown in Figure 3.

Figure 3.
Structures of compounds isolated from the mushrooms of the genus Inocybe: I. muscarine; II. psilocin; III. psilocybin; IV. aeruginascin and V. baeocystin.
4.1. Muscarine
Muscarine is named mainly for its isolation from Amanita muscaria and is a kind of neurosymptomatic toxin. Muscarine is also found in the genera Inocybe, Clitocybe, Mycena and Omphalotus [18]. The main poisonous substance of Inocybe mushrooms is alkaloid muscarine. The actual muscarine content of Amanita. muscaria is only around 0.0003% of the fresh weight [1]. Instead, muscarine concentrations are much higher in Inocybe reaching as high as 1.6%. Additionally, the paper chromatography method applied to 34 Inocybe species revealed detectable quantities of muscarine ranging from 0.01 to 0.80% in approximately 75% of them [19]. Alkaloid muscarine is a colorless and odorless ammonium quaternary molecule that stimulates the parasympathetic nervous system of animals [20].
The mechanism is similar with acetylcholine, which binds to acetylcholine receptors and generates a characteristic series of symptoms including central nervous system disorder, hallucination-induced inhibition or excitation. Specifically, those include sweating, tearing and bradycardia. These symptoms generally occur rapidly within two hours of eating. Muscarine is particularly widespread in Inocybe mushrooms taken from North America and Europe while widespread in east Asia [2,21,22,23]. About their application, some studies have shown that the cholinergic receptor is activated by binding to the muscarine. After activation, it can promote the invasion and metastasis of colon cancer or bile duct cancer cells [24]. That may provide a new therapeutic direction to inhibit tumor growth and metastasis by knocking out the receptor or inhibiting the activation of the receptor, so muscarine has potential clinical value.
4.1.1. Chemistry and Stereochemistry
Muscarine(L-(+)-muscarinee,(2S,4R,5S)-(4-hydroxy-5-methyl-tetrahydrofuran-2-ylmethyl) trimethylamonium, C9H20NO2, CAS number 300-54-9) is a toxic alkaloid found in fly agaric and other mushrooms of the Inocybe species. It is the first parasympathomimetic substance ever studied and causes profound parasympathetic activation that may end in convulsions and death. Muscarinee was firstly isolated and found to exist in four stereoisomers: (+)-muscarinee, (+)-epi-muscarinee, (-)-allo-muscarinee and (+)-epiallo-muscarinee (Figure 4). L-(+)-muscarinee is a natural product found in certain mushrooms, particularly in Inocybe and Clitocybe species [25]. The distribution and relative concentrations of muscarinee and its stereoisomers in Inocybe species were determined by Catalfomo and Eugster. Notable was the finding that Inocybe cinnamomea, Inocybe geophylla, and Inocybe lacera contained concentrations of epi-muscarinee equal to or higher than muscarinee itself [22].
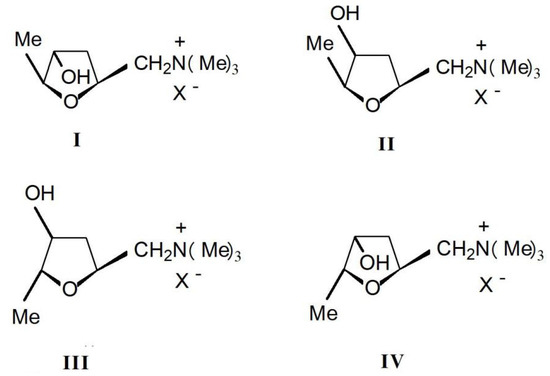
Figure 4.
Four stereoisomers of muscarinee: I. muscarine; II.epi-muscarinee; III. allo-muscarinee and IV. epiallo-muscarinee.
4.1.2. Poisonous Mechanism
The mechanism of muscarinic poisoning is to mimic the action of the neurotransmitter acetylcholine by binding to muscarinic acetylcholine receptors, resulting in continuous impulses of cholinergic nerves [26]. Muscarine acts in the peripheral nervous system, competing with acetylcholine at its receptor binding site. Once bound to the receptor, muscarinic mimics the effects of acetylcholine. Muscarine cannot inactivate acetylcholinesterase, and uncontrolled receptor hyperstimulation occurs. There are five different types of muscarinic receptors: M1–M5, and most tissues express a mixture of subtypes. The M2 and M3 subtypes mediate the muscarinic response of peripheral autonomic tissues. The M1 and M4 subtypes are more abundant in the brain and autonomic ganglia [27]. M1, M3 and M5 interact with Gq protein to stimulate the hydrolysis of phosphoinositide and release intracellular calcium [28]. The M2 and M4 receptors interact with the Gi protein to inhibit adenylate cyclase, resulting in a decrease in the intracellular cyclic adenosine monophosphate concentration. Muscarinic receptors play a leading role in mediating the role of acetylcholine in the brain, and indirectly produce excitatory and inhibitory effects by combining with a series of unique receptor subtypes. Muscarinic receptors are found in both presynapses and postsynapses. Ultimately, their main neuronal effects seem to be mediated by changing the properties of ion channels. Presynaptic muscarinic receptors participate in an important feedback loop that regulates neurotransmitter release [29]. The acetylcholine released from the presynaptic terminal can bind to muscarinic receptors in the same nerve terminal to activate the enzymatic process that regulates subsequent neurotransmitter release.
4.1.3. Clinical Response
Muscarine is a toxic neurotoxic alkaloid that enhances parasympathetic excitability. Muscarinee can cause parasympathetic nervous system disorder around the body and inhibit the role of the acetylcholine in the choline receptor [30]. The symptoms of intoxication with Inocybe mushrooms rich in muscarinee are very typical. First, symptoms start early after eating (15–120 min) with headache, nausea and vomiting. Second, salivation and lacrimation follows, combined with miosis, reduced vision and disturbed accommodation. Other signs of poisoning are gastric colic, diarrhea, bronchoconstriction and severe dyspnea. Bradycardia combined with rapid hypotension and vasodilation leads to circulatory shock. Death occurs after approximately 8 h. Usually, muscarine rarely causes death and most of the poisoned people survive without treatment.
4.1.4. Experimental Toxicity
Muscarine is a toxic alkaloid. Published acute and subchronic toxicity parameters of muscarinee are summarized in Table 1.

Table 1.
Published acute and subacute toxic parameters of muscarinee.
4.1.5. Human Toxicity
Patients generally recover after muscarineism, but ingestion of large quantities of mushrooms containing the toxin can also cause death. The lethal dose of muscarinee in humans was estimated to be between 180 and 300 mg. Therefore, ingestion of a single mushroom containing 0.33% muscarinee on a dry weight basis can be lethal [38].
4.1.6. Treatment
Effective therapy consists of rapid administration of atropine [39]. Serious poisoning is rare because of the low gastrointestinal bioavailability of muscarinee from the mushrooms and the low concentration of muscarinee. Treatment includes induction of emesis and administration of activated charcoal and rehydration to restore fluid balance and electrolytes is a key component of treatment. If clinical signs of life are present, atropine should be given. After giving a test dose of 0.04 mg/kg (1/4 i.v. and 3/4 i.m. or s.c.) to determine its efficacy, atropine can be given repeatedly until symptoms are abolished or until cessation of salivation [23].
4.2. Psilocybin and Psilocin
4.2.1. Chemical Structure
Psilocybin and psilocin are hallucinogenic substances isolated from the fruiting bodies of Psilocybe mexicana cultivated by Swiss chemist Hofmann in 1957, and it has been proven that psilocybin and psilocin are kinds of mushroom toxins that can cause neurohallucinogenic effects [40]. These two kinds of toxins are mainly distributed in the genera Inocybe, Agrocybe, Conocybe, Copelandia, Galerina, Gymnopilus and Hypholoma [41]. Psilocybin is also known as psilocybine. Its scientific name is 4-phospho-N, N-dimethylamine, which is a small molecule indole alkaloid with hallucinogenic activity. Psilocybin is primarily a pro-drug that is rapidly converted in the body through dephosphorylation by alkaline phosphatase to active metabolite psilocin [42]. Because Psilocybin is unstable in the blood, it is easy to dephosphorylate and exists in the form of psilocybin dephosphorization.
4.2.2. Physico-Chemical Characteristics
Pure psilocybin and psilocin are colorless crystals and sensitive to temperature. When stored for a few months at room temperature, both will be completely deactivated. Whereas, freeze-dried mushrooms will remain active after being stored for more than two years at −5 °C [43]. Cytochrome oxidase oxidizes the dephosphorization of psilocybin to produce the blue product, and hallucinogenic mushrooms that contain the toxin often turn blue after being picked [44]. So, we can identify the hallucinogenic mushroom by this way.
4.2.3. Poisonous Mechanism
The toxicological mechanism of psilocybin is not fully understood. The psychedelic effect of psilocybin is related to the activity of 5-hydroxytryptamine (5-HT), dopamine, epinephrine and other bioamines [45]. In animal pharmacological research data, it was found that dopamine and 5-HT inhibited each other. The imbalance of 5-HT and dopamine in the thalamic striatal cortical margin was a key factor causing neurological symptoms [46]. Electron-ray tomography proved that psilocybin can unbalance 5-HT and dopamine [47]. It has also been suggested that psilocybin poisoning is caused by activation of the 5-HT2A receptor and has nothing to do with dopamine stimulation. Overactivation of the 5-HT2A receptor causes neurological symptoms and inhibition of this receptor leads to a calming effect [48]. The strange hallucinations caused by psilocybin are mainly due to the activation of serotonergic (5-HT2A) receptors in the body. Psilocybin induces neural excitatory effects by increasing the receptors of 5-HT2A in the postsynaptic membrane [49].
Psilocin is a mixed serotonergic receptor agonist [50]. It has a high affinity for the 5-HT2A receptor in the brain, where it mimics the effect of serotonin [51]. Psilocin binds less strongly with other serotonergic receptors 5-HT1A, 5-HT2C and 5-HT1D [52]. Published acute toxic parameters of psilocin are summarized in Table 2.

Table 2.
Published acute and subacute toxic parameters of psilocin.
4.2.4. Clinical Response
Psychiatric symptoms are associated with dose, environment, previous hallucinogenic experiences, emotions and personality [55]. Psilocybin binds and activates serotonergic (5-HT2A) receptors, leading to the physiological and neurotoxic effects. Psilocybin can only cause physiological symptoms at doses of 8–10 mg (0.1–0.2 mg/kg, oral), including mydriasis, light elevated blood pressure, increased heart rate and paresthesia [56,57]. Hallucination occurs when the dose is more than 15 mg [58].
The symptoms of psilocybin poisoning can be divided into three stages. The first stage starts from 20 to 30 min after oral administration and lasts for 10 to 15 min, with implant neurological disorder accompanied by sensory and perceptual interference. The second stage is the peak stage of neurological symptoms, during which the feeling is relatively strong, including personality solution, derealization, spatiotemporal change and weightlessness. Sensory changes mainly include visual disturbances, such as bright and warm colors, especially red and green. Physical sensations including brain vertigo and mental depression are accompanied by anxiety and restlessness. In the final stage, the mental symptoms gradually disappear, while the vegetative nerve interference continues for a period of time. This period can be accompanied by lethargy, extreme exhaustion and mental decadence. In severe cases, delusional syndrome occurs [59].
4.2.5. Treatment
Psilocybin mainly acts on the autonomic nerves, causing nerve excitation, hallucination, tachycardia, dilated pupils and dysuria, but is generally not life-threatening [60]. In general, taking poisonous mushrooms can cause central nervous poisoning (within 20 min) [61]. If poisonous mushrooms are provided in the form of liquid (such as soup), poisoning phenomenon will occur earlier (about 5–10 min) and lasting 2–4 h [62]. Usually, the peak period of poisoning lasts only a little more than an hour [63]. People who have eaten psilocybin mushrooms generally recover completely.
Treatment usually includes induction of emesis and gastrolavage. Vomiting inducing can be used when people ingest psilocybin containing mushrooms within one hour with syrupus ipecacuanhae. However, gastric lavage is generally not used because the stirring produces an unpleasant sensation and whole mushrooms may clog the stomach tube [59]. As for the patients who are in extreme anxiety or have some seriously aggressive behavior, benzodiazepines or chlorpromazine may be taken. When using these anticholinergic agents, respiratory failure should be observed and intubation or pulmonary ventilation devices should be used if necessary [64]. In addition, for most patients, the effective way for them is to rest and recover in a dark and quiet room.
4.2.6. Application
Psilocybin was used in religious practices by native Americans in Central and South America for thousands of years. Psilocybin and psilocin are regulated substances in many countries and are the two major hallucinogenic compounds of “magic mushrooms” [65]. Psilocybin is very popular and often misused.
Medical
Although psilocybin has not been widely developed and utilized, a lot of research work has been done on the prevention and treatment of diseases. Psilocybin has been used in various forms for the treatment of mental disorders and addiction since its discovery. Psilocybin is devoted to the scientific community as a research tool for modeling psychosis as well as for possible therapeutic effects [66]. Furthermore, psilocybin can be used in end-of-life care for patients with advanced cancer to ease the pain [67]. Psilocybin is suitable for psychotherapy because it alters consciousness and state of mind and arouses emotions while being low in toxicity. Low-dose psilocybin can be used to treat patients with depression, anxiety and other physical and mental disorders; high dose psilocybin has a definite effect on getting rid of bad habits such as smoking, alcohol and crime [68]. These show the dual value of “psychedelic mushrooms” in both humanities and medicine.
Physiological Research
As psilocybin can affect many physiological activities such as hearing, vision, memory and emotion, it is widely used for research basis, formation causes, regulation modes and mutual connections of these physiological activities. It is proven that visual cortex V1 is a structural model composed of interconnected polygonal column units, each of which contains cylinders from many different sources and the lateral connections of these columns should be symmetrical [69]. Carter’s team used psilocybin to explore the relationship between attention and working memory, concluding that attention and working memory are likely to be separate and that there is no obvious link between them [70]. Carter and co-workers conducted binocular vision competition experiment with psilocybin, and the results showed that the tryptamine pathway participated in binocular vision competition [71]. A lot of literatures show that psilocybin and other psychedelics are very helpful for the study of 5-HT receptor subtype [72]. In short, psilocybin is greatly applied to physiological research.
4.3. Aeruginascin
Aeruginascin (N,N,N-trimethyl-4-phosphoryloxytryptamine, C13H19N2O4P, Mr 298,279 g/mol, CAS Number 114264-95-8) is an indoleamine alkaloid which occurs naturally only within the mushroom I. aeruginascens [73]. It is the N-trimethyl analogue of psilocybin [74], and it is closely related to the frog-skin toxin bufotenidine [75].
4.4. Baeocystin
Baeocystin, a methyl analogue of psilocybin, is a tryptamine toxin. It was first isolated from the extracts of Psilocybe baeocystis [76,77] and Psilocybe semilanceata [78,79]. Firstly, it was synthesized by Troxler and his partners in 1959 [80]. Additionally, they are thought to be precursors to psilocybin. We can find that baeocystin is an N-demethylated derivative of psilocybin. The hydrogen on the -OH of one methylpsilocybin is also replaced by phosphoric acid, and the -CH3 is removed from the -N. Additionally, Figure 3 illustrates that the structure of baeocystin (V) exists in its zwitterionic form. The mechanism of baeocystin is similar to psilocybin and psilocin. It can also bind to specific subtypes of the 5-HT receptor to produce hallucinogenic effects [81].
4.5. Lectins
Mushrooms are a valuable source of lectins with unique traits and potential for biotechnology and biomedical applications [82]. Mushroom lectins are a significant group of biologically active glycoproteins that have chemopreventive activity against cancer. A new lectin with a molecular weight of 17 kDa and carbohydrate specificity against lactose and galactose has been isolated from Inocybe umbrinella dried fruits. This lectin has a unique N-terminal amino acid sequence, DGVLATNAVA, and it is active against hepatocellular carcinoma cell line (HepG2, 3.5 ± 0.2 μmol/L). However, the production of this lectin from fresh mushroom is impractical, because only 15 mg lectin per 100 g fruiting bodies of I. umbrinella was isolated [83].
5. Conclusions
People have the habit of eating mushrooms because of its delicious taste, rich nutrition and high medicinal value. However, there are so many kinds of mushrooms which look similar. The species diversity of poisonous mushrooms is quite rich. If edible and poisonous mushrooms cannot be identified, it may cause toxic mushroom poisoning and even lead to death. Mushroom poisoning is closely related to people’s health and lives. Mushroom poisoning is still a threat to human life. The incidence of mushroom poisoning is high, the mortality rate is high and the toxins contained in mushrooms are complex and varied. The clinical manifestations of poisoning are varied. Currently, there is no specific antidote for mushroom poisoning, and only medication and blood purification can be given as symptomatic supportive treatment. For instance, toxins found in the genus Inocybe show signs of affecting the central nervous system, the parasympathetic system and the gastrointestinal system. The effects of toxins in the above-mentioned mushroom species are generally mild, but the symptoms depend on both the dose of the toxins consumed and the individual sensitivity. In most cases, treatment is symptomatic.
Currently, there is a lack of basic research on mushroom poisoning. Clinical research is mainly focused on the general epidemiology and case reports of poisonous mushrooms, and there is a lack of exploration of toxins and research on the pathogenesis of the toxins. Actually, poisonous mushrooms and their toxins have broad development prospects and they have been used in many aspects such as biological control, anti-cancer medicine and biotechnology. Exploitation and utilization of toxic mushroom resources will benefit people. The genus Inocybe is very numerous and new species are still being discovered. So far, toxicological analysis has only been performed on a small number of species, and it can be expected that further unknown biochemicals will be gradually discovered. Therefore, it is assumed that new substances with interesting biological effects will be discovered. With the progress of toxin detection technology, especially the development and application of metabonomic technology, toxins caused by toadstool poisoning will be easier to detect, and more new toxins will be gradually discovered, so as to better study the mechanisms of action of different toxins causing poisoning. At the same time, it is of great help to determine the toxic components, poisoning types and poisoning mechanisms of poisonous mushrooms, and also reduce the harm of poisonous mushrooms to human health.
Funding
This work was supported by the long-term organization development plan (University Hospital, Hradec Kralove, Czech Republic), National Key R & D Program (2016YFD0501207, 2016YFD0501009), NSFC (31972741, 31572576), China Postdoctoral Science Foundation (2016T90477), PAPD. The authors would also like to acknowledge the funding received from UHK VT2019-2021 and from the Ministry of Health of the Czech Republic (FN HK 00179906) and the Charles University in Prague, Czech Republic (PROGRES Q40/15).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that they have no competing interests.
References
- Lurie, Y.; Wasser, S.P.; Taha, M.; Shehade, H.; Nijim, J.; Hoffmann, Y.; Basis, F.; Vardi, M.; Lavon, O.; Suaed, S.; et al. Mushroom poisoning from species of genus Inocybe (fiber head mushroom): A case series with exact species identification. Clin. Toxicol. 2009, 47, 562–565. [Google Scholar] [CrossRef]
- Hasuo, H.; Akasu, T.; Gallagher, J.P. Muscarine activates a nonselective cation current through a M3 muscarinic receptor subtype in rat dorsolateral septal nucleus neurons. J. Neurophysiol. 1996, 76, 2221–2230. [Google Scholar] [CrossRef] [PubMed]
- Klaassen, C.D.; Casarett, L.J.; Doull, J. Casarett and Doull’s Toxicology: The Basic Science of Poisons, 8th ed.; McGraw-Hill Education: New York, NY, USA, 2013; ISBN 9780071769235. [Google Scholar]
- Kirk, P.M. Species fungorum (version 20 November 2015). In Species 2000 & ITIS Catalogue of Life; The Catalogue of Life Partnership: Leiden, The Netherlands, 2019. [Google Scholar]
- Ryberg, M.; Kristiansson, E.; Sjökvist, E.; Nilsson, R.H. An outlook on the fungal internal transcribed spacer sequences in GenBank and the introduction of a web-based tool for the exploration of fungal diversity. New Phytol. 2009, 181, 471–477. [Google Scholar] [CrossRef]
- Puschner, B.; Wegenast, C. Mushroom poisoning cases in dogs and cats: Diagnosis and treatment of hepatotoxic, neurotoxic, gastroenterotoxic, nephrotoxic, and muscarineic mushrooms. Vet. Clin. N. Am. Small Anim. Pract. 2018, 48, 1053–1067. [Google Scholar] [CrossRef]
- Lin, H.T. Mushroom poisoning (Inocybe asterospora Quel.)—Report of 3 cases. Zhonghua Yu Fang Yi Xue Za Zhi 1984, 18, 231–233. (in Chinese). [Google Scholar]
- Seljetun, K.O.; von Krogh, A. Acute Inocybe mushroom toxicosis in dogs: 5 cases (2010–2014). J. Vet. Emerg. Crit. Care 2017, 27, 212–217. [Google Scholar] [CrossRef]
- Kohn, R.; Mot’ovská, Z. Mushroom poisoning--classification, symptoms and therapy. Vnitr. Lek. 1997, 43, 230–233. (in Slovak). [Google Scholar]
- Wilson, D. Poisoning by Inocybe fastigiata. Br. Med. J. 1947, 2, 297. [Google Scholar] [CrossRef][Green Version]
- Amitai, I.; Peleg, O.; Ariel, I.; Binyamini, N. Severe poisoning in a child by the mushroom Inocybe tristis, Malençon and Bertault. Isr. J. Med. Sci. 1982, 18, 798–801. [Google Scholar] [PubMed]
- Gartz, J.; Drewitz, G. The green discolored mushroom Inocybe aeruginascens--an Inocybe species with hallucinogenic effects. Z Arztl Fortbild 1986, 80, 551–553. (in German). [Google Scholar]
- Pradeep, C.K.; Vrinda, K.B.; Shibu, P.V.; Hailee, B.K.; Brandon Matheny, P. New species of Inocybe (Agaricales) from tropical India. Mycol. Prog. 2016, 15, 2–25. [Google Scholar] [CrossRef]
- Bijeesh, C.; Vrinda, K.B.; Pradeeo, C.K. Inocybe poisoning in Kerala—A case study. J. Mycopathol. Res. 2020, 57, 255–258. [Google Scholar]
- Xu, F.; Zhang, Y.Z.; Zhang, Y.H.; Guan, G.Y.; Zhang, K.P.; Li, H.J.; Wang, J.J. Mushroom poisoning from Inocybe serotina: A case report from Ningxia, northwest China with exact species identification and muscarine detection. Toxicon 2020, 179, 72–75. [Google Scholar] [CrossRef]
- Beuhler, M.C. Overview of mushroom poisoning. In Critical Care Toxicology: Diagnosis and Management of the Critically Poisoned Patient; Brent, J., Burkhart, K., Dargan, P., Hatten, B., Megarbane, B., Palmer, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 2103–2128. [Google Scholar]
- Hossain, M.A.; Park, S.C. A review on mushroom toxins. In Food Toxicology; Chapter 14; Bagchi, D., Swaroop, A., Eds.; CRC Press: Boca Raton, FL, USA, 2016; 584p, ISBN 9781498708753. [Google Scholar]
- Jin, Z. Muscarine, imidazole, oxazole, and thiazole alkaloids. Nat. Prod. Rep. 2003, 20, 584–605. [Google Scholar] [CrossRef]
- Brown, J.K.; Malone, M.H.; Stuntz, D.E.; Tyler, V.E., Jr. Paper chromatographic determination of muscarinee in Inocybe species. J. Pharm. Sci. 1962, 51, 853–856. [Google Scholar] [PubMed]
- Young, A. Muscarinee-Containing mushrooms. In Handbook of Mushroom Poisoning: Diagnosis and Treatment; Spoerke, D.G., Rumack, B.H., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 289–299. [Google Scholar]
- Malone, M.H.; Robichaud, R.; Tyler, V.E., Jr.; Brady, L. Relative muscarineic potency of thirty Inocybe species. Lloydia 1962, 25, 231–237. [Google Scholar]
- Catalfomo, P.; Eugster, C.H. Muscarinee and muscarinee isomers in selected Inocybe species. Helv. Chim. Acta 1970, 53, 848–851. [Google Scholar] [CrossRef]
- Gurevich, L.S.; Nezdoiminogo, E.L. Psilocybin and muscarinee as possible chemotaxonomic markers for the genus Inocybe (Fr) Fr. Mycol. Phytopathol. 1992, 262, 88–97. [Google Scholar]
- Raufman, J.P.; Samimi, R.; Shah, N.; Shah, N.; Khurana, S.; Shant, J.; Drachenberg, C.; Xie, G.; Wess, J.; Cheng, K. Genetic ablation of M3 muscarinic receptors attenuates murine colon epithelial cell proliferation and neoplasia. J. Cancer Res. 2008, 68, 3573–3578. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, B.; Ferenc, T.; Kusowska, J.; Ciećwierz, J.; Kowalczyk, E. Poisoning with selected mushrooms with neurotropic and hallucinogenic effect. Med. Pr. 2010, 61, 583–595. (in Polish). [Google Scholar]
- White, J.; Weinstein, S.; Haro, L.; Bedry, R.; Schaper, A.; Rumack, B.; Zilker, T. Mushroom poisoning: A proposed new clinical classification. Toxicon 2019, 157, 53–65. [Google Scholar] [CrossRef]
- Colloby, S.J.; Nathan, P.J.; McKeith, I.G.; Bakker, G.; O’Brien, J.T.; Taylor, J.P. Cholinergic muscarinic M(1)/M(4) receptor networks in dementia with Lewy bodies. Brain Commun. 2020, 2, fcaa098. [Google Scholar] [CrossRef] [PubMed]
- Matera, M.G.; Rinaldi, B.; Berardo, C.; Rinaldi, M.; Cazzola, M. A review of the pharmacokinetics of M(3) muscarinic receptor antagonists used for the treatment of asthma. Expert. Opin. Drug Metab. Toxicol. 2020, 16, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Sevilla, D.; Núñez, A.; Buño, W. Muscarinic receptors, from synaptic plasticity to its role in network activity. Neuroscience 2021, 456, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.M.; Katzung, B.B. Základni a Klinická Farmakologie; Katzung, B.G., Ed.; H&H, Bom: Ružinov, Slovakia, 1992; p. 83. [Google Scholar]
- Kögl, F.; Duisberg, H.; Erxleben, H. Untersuchungen über Pilzgifte. I. Über das muscarine. I. Justus Leibigs Ann. der Chem. 1931, 489, 156–192. [Google Scholar] [CrossRef]
- Bovet, D.; Bovet-Nitti, F. Structure et Activite Pharmacodyanmique des Medicaments du Systeme Nerveux Vegetatif; Karge, S., Ed.; AAAS: New York, NY, USA, 1948; p. 406. [Google Scholar]
- Staib, A.H.; Schröder, M. Participation of the cholinergic system in temperature regulation by the mouse. Arch. Int. Pharmacodyn. Ther. 1971, 192, 88–95. (in German). [Google Scholar]
- Habermehl, G. Chemistry and biochemistry of amphibian poisons. Naturwissenschaften 1969, 56, 615–622. (in German). [Google Scholar] [CrossRef]
- Eltherington, C.D.; Barnes, L.G. Drug Dosages in Laboratory Animals—A Handbook; University California Press: Berkeley, CA, USA, 1973; 348p. [Google Scholar]
- Mosher, H.S.; Fuhrman, F.A.; Buchwald, H.D.; Fischer, H.G. Tarichatoxin—Tetrodotoxin: A potent neurotoxin. Science 1964, 144, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Arena, J.M. Poisoning: Toxicology, Symptoms, Treatments, 2nd ed.; C.C. Thomas: Springfield, IL, USA, 1970; Volume 2, p. 73. [Google Scholar]
- Bresinsky, H.; Besl, A. A Colour Atlas of Poisonous Fungi, 1st ed.; Manson Publishing Ltd.: London, UK, 1989; 295p, ISBN-10: 9780723415763, ISBN-13: 978-0723415763. [Google Scholar]
- Jahodár, L. Prírodní Toxiny a Jedy; Houdek, L., Ed.; Galén: Prague, Czech Republic, 2004; p. 109. [Google Scholar]
- Hofman, A.; Heim, R.; Brack, A.; Kobel, H. Psilocybin einpsychotroper Wirkstoff aus dem mexikanischen Rauschpilz Psilocybe mexieana Heim. Experientia 1958, 14, 107. [Google Scholar] [CrossRef]
- Stebelska, K. Fungal hallucinogens psilocin, ibotenic acid, and muscimol: Analytical methods and biologic activities. Ther. Drug Monit. 2013, 35, 420–442. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J. Metabolism of psilocybin and psilocin: Clinical and forensic toxicological relevance. Drug Metab. Rev. 2017, 49, 84–91. [Google Scholar] [CrossRef]
- Beug, M.W.; Bigwood, J. Quantitive analysis of psilocybin andpsilocin in Psilocybe baeocystis by HPLC and TLC. J. Chromatogr. 1981, 207, 379–385. [Google Scholar] [CrossRef]
- Lenz, C.; Wick, J.; Braga, D.; García-Altares, M.; Lackner, G.; Hertweck, C.; Gressler, M.; Hoffmeister, D. Injury-triggered blueing reactions of psilocybe “Magic” mushrooms. Angew Chem. Int. Ed. Engl. 2020, 59, 1450–1454. [Google Scholar] [CrossRef] [PubMed]
- Leikin, J.B.; Krantz, A.J.; Zell-Kanter, M.; Barkin, R.L.; Hryhorczuk, D.O. Clinical features andmanagement of intoxication due to hallucingenic drugs. Med. Toxicol. Advers. Drug Exp. 1989, 4, 324–350. [Google Scholar] [CrossRef]
- Vollenweider, F.X.; Geyer, M.A. A systems model of altered con-sciousness: Integrating natural and grug-induced psychoses. BrainResBull 2001, 56, 495–507. [Google Scholar]
- Vollenweider, F.X.; Vontobel, P.; Hell, D.; Leenders, K.L. 5-HT modulationof dopamine release in basal ganglia in psilocybin—Induced psy-chosis in man---aPET study with [11C] raclopride. Neuropsychopharmacology 1999, 20, 424–433. [Google Scholar] [CrossRef]
- Vollenweider, F.X.; Vollenweider-Schhherpenhuyzen, M.F.; Babler, A.; Vogel, H.; Hell, D. Psilocybin induces schizophrenia-like psychosisin humanvia a serotonin-2 agonist action. Neuroreport 1998, 9, 3897–3902. [Google Scholar] [CrossRef]
- Buckholtz, N.S.; Zhou, D.F.; Freedman, D.X.; Potter, W.Z. LSD administration selectively down regulates serotonin2 receptors in rat brain. Neuropsychopharmacology 1990, 3, 137–148. [Google Scholar] [PubMed]
- Rambousek, L.; Páleníček, T.; Valeš, K.; Stuchlík, A. The effect of psilocin on memory acquisition, retrieval, and consolidation in the rat. Front. Behav. Neurosci. 2014, 8, 180. [Google Scholar] [CrossRef]
- López-Giménez, J.; González-Maeso, J. Hallucinogens and serotonin 5-HT(2A) receptor-mediated signaling pathways. Curr. Top. Behav. Neurosci. 2018, 36, 45–73. [Google Scholar]
- Passie, T.; Seifert, J.; Schneider, U.; Emrich, H.M. The pharmacology of psilocybin. Addict. Biol. 2002, 7, 357–364. [Google Scholar] [CrossRef]
- Bosin, T.R.; Campaigne, E.; Dinner, A.; Rogers, R.B.; Maickel, R.P. Comparative toxicological studies of indole, benzo[b] thiophene, and 1-methylindole derivatives. J. Toxicol. Environ. Health 1976, 1, 515–520. [Google Scholar] [CrossRef]
- Usdin, E.; Efron, D.H. Psychotropic Drugs and Related Compounds, 2nd ed.; Pergamon Pr: Washington, DC, USA, 1972; p. 138. [Google Scholar]
- Mills, P.R.; Lesinskas, D.; Watkinson, G. The danger of hallucinogenic mushrooms. Scot. Med. J. 1979, 24, 316–317. [Google Scholar] [CrossRef]
- Griffiths, R.R. Psilocybin can occasion mystical-type experiences having substantial and sustained personal meaning and spiritual significance. Psychopharmacology 2006, 187, 268–283. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, R.R. Psilocybin occasioned mystical-type experiences: Immediate and persisting dose-related effects. Psychopharmacology 2011, 218, 649–665. [Google Scholar] [CrossRef]
- Hasler, F. Acute psychological and physiological effects of psilocybin in healthy humans: A double-blind, placebo-controlled dose-effect study. Psychopharmacology 2004, 172, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Smolinske, S.C. Psilocybin-Containing mushrooms. In Handbook of Mushroom Poisoning Diagnosis and Treatment; Spoerke, D.G., Rumack, B.H., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 309–324. [Google Scholar]
- Allen, J.W.; Merlin, M.D.; Jansen, K. An ethnomycological review of psychoactive agarics in Australia and New Iealand. Psychoact. Drugs 1991, 23, 39. [Google Scholar] [CrossRef] [PubMed]
- Lampe, K.F. Toxic fungi. Rev. Pharmacol. Toxicol. 1979, 19, 85. [Google Scholar] [CrossRef]
- Shulgin, A.T. Profiles of psychedelic drugs: 8 psilocybin. Psychedelic Drugs 1980, 12, 79. [Google Scholar] [CrossRef]
- Young, R.E.; Hutchison, S.; Milroy, R. The rising price of mushrooms. Lancet 1982, 1, 213. [Google Scholar] [CrossRef]
- Benjamin, D.R. Mushrooms: Poisons and panaceas:a handbook fornaturalists, mycologosts, and physicans. Nature 1996, 379, 503–504. [Google Scholar]
- Shirota, O.; Hakamata, W.; Goda, Y. Concise large-scale synthesis of psilocin and psilocybin, principal hallucinogenic constituents of “magic mushroom”. J. Nat. Prod. 2003, 66, 885–887. [Google Scholar] [CrossRef] [PubMed]
- Tylš, F.; Páleníček, T.; Horáček, J. Psilocybin–Summary of knowledge and new perspectives. Eur. Neuropsychopharmacol. 2014, 24, 342–356. [Google Scholar] [CrossRef] [PubMed]
- Horgan, J. The electric koolaid clinical trial: LSD and other hallucinogens were once considered promising psychiatric treatments. New Sci. 2005, 185, 36–39. [Google Scholar]
- Grinspoon, L.; Dobin, R. Psychedelics as catalysts of insight-oriented psychotherapy. Soc. Res. 2001, 68, 667–695. [Google Scholar]
- Bressloff, P.C.; Cowan, J.D.; Golubitsky, M.; Thomas, P.J.; Wiener, M.C. What geometric visual hallucinations tell us about the visual cortex. Neural. Comput. 2002, 14, 473–491. [Google Scholar] [CrossRef] [PubMed]
- Carter, O.L.; Burr, D.C.; Pettigrew, J.D.; Wallis, G.M.; Hasler, F.; Vollenweider, F.X. Using psilocybin to investigate the relationship between attention, working memory, and the serotonin 1A and 2A receptors. J. Cogn. Neurosci. 2005, 17, 1497–1508. [Google Scholar] [CrossRef]
- Carter, O.L.; Pettigrew, J.D.; Hasler, F.; Wallis, G.M.; Liu, G.B.; Hell, D.; Vollenweider, F.X. Modulating the rate and rhythmicity of perceptual rivalry alternationswith the mixed 5-HT2A and 5-HTlA agonist psilocybin. Neuropsychopharmacology 2005, 30, 1154–1162. [Google Scholar] [CrossRef]
- Hery, F.; Hamon, M. Neuroleptics and serotonin. L’Encephale 1993, 19, 525–532. [Google Scholar]
- Gartz, J. Analysis of aeruginascin in fruit bodies of the mushroom Inocybe aeruginascens. Int. J. Crude Drug Res. 1989, 27, 141–144. [Google Scholar] [CrossRef]
- Jensen, N.; Gartz, J.; Laatsch, H. Aeruginascin, a trimethylammonium analogue of psilocybin from the hallucinogenic mushroom Inocybe aeruginascens. Planta Med. 2006, 72, 665–666. [Google Scholar] [CrossRef] [PubMed]
- Wieland, H.; Konz, W.; Mittasch, H. Toad poisons. VII. Constitution of bufotenin and bufotenidine. Justus Leibigs Ann. der Chem. 1934, 513, 1–25. [Google Scholar] [CrossRef]
- Leung, A.Y.; Paul, A.G. Baeocystin, a mono-methyl analog of psilocybin from Psilocybe baeocystis saprophytic culture. J. Pharm. Sci. 1967, 56, 146. [Google Scholar] [CrossRef]
- Leung, A.Y.; Paul, A.G. Baeocystin and norbaeocystin: New analogs of psilocybin from Psilocybe baeocystis. J. Pharm. Sci. 1968, 57, 1667–1671. [Google Scholar] [CrossRef]
- Repke, D.B.; Leslie, D.T. Baeocystin in Psilocybe semilanceata. J. Pharm. Sci. 1977, 66, 113–114. [Google Scholar] [CrossRef] [PubMed]
- Repke, D.B.; Leslie, D.T.; Guzman, G. Baeocystin in psilocybe, conocybe and panaeolus. Lloydia 1977, 40, 566–578. [Google Scholar] [PubMed]
- Troxler, F.; Seeman, F.; Hofmann, A. Abwandlungsprodukte von Psilocybin und Psilocin. 2. Mitteilung über synthetische Indolverbindungen. Helvetica Chimica Acta 1959, 42, 2073–2103. (in German). [Google Scholar] [CrossRef]
- Mckenna, D.J.; Repke, D.B.; Lo, L.; Peroutka, S.J. Differential interactions of indolealkylamines with 5-hydroxytryptamine receptor subtypes. Neuropharmacology 1990, 29, 193–198. [Google Scholar] [CrossRef]
- Sarup Singh, R.; Preet Kaur, H.; Rakesh Kanwar, J. Mushroom lectins as promising anticancer substances. Curr. Protein Pept. Sci. 2016, 17, 797–807. [Google Scholar] [CrossRef]
- Zhao, J.K.; Wang, H.X.; Ng, T.B. Purification and characterization of a novel lectin from the toxic wild mushroom Inocybe umbrinella. Toxicon 2009, 53, 360–366. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).