Targeting lncRNA H19/miR-29b/COL1A1 Axis Impedes Myofibroblast Activities of Precancerous Oral Submucous Fibrosis
Abstract
1. Introduction
2. Results
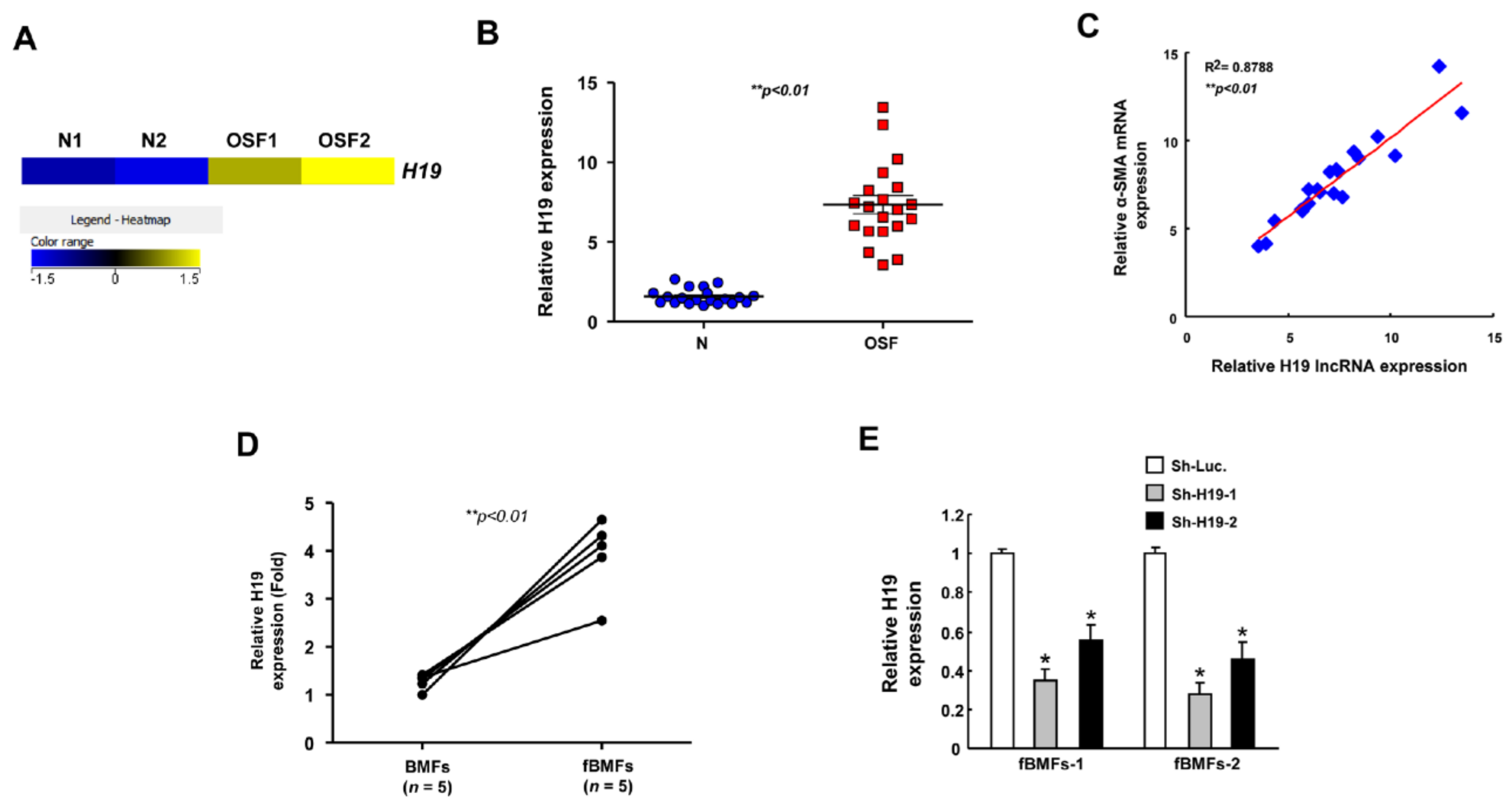
2.1. H19 Is Overexpressed in OSF Tissues and Associated with the Expression of Myofibroblast Markers
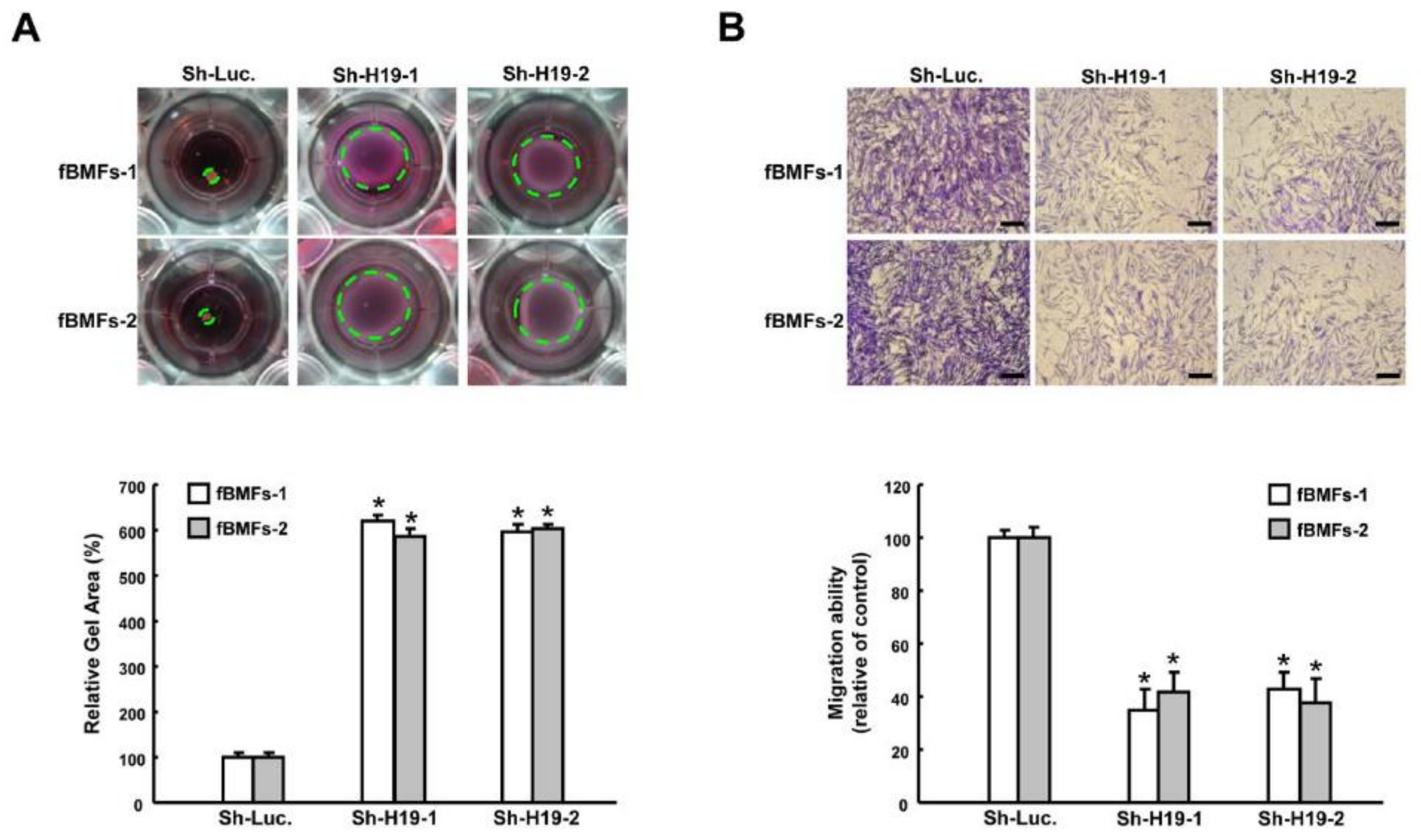
2.2. Silencing H19 Inhibits Myofibroblast Activities
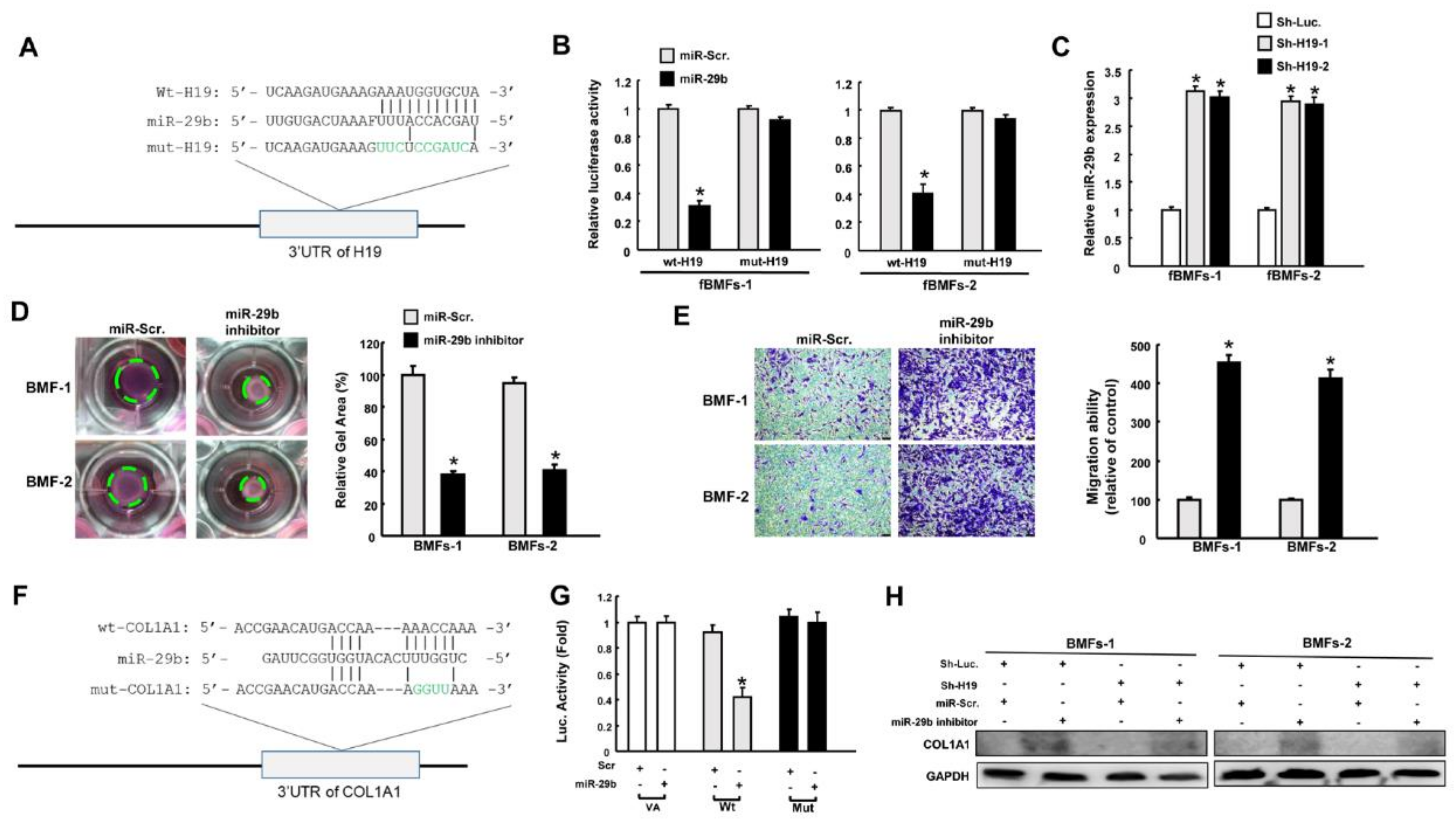
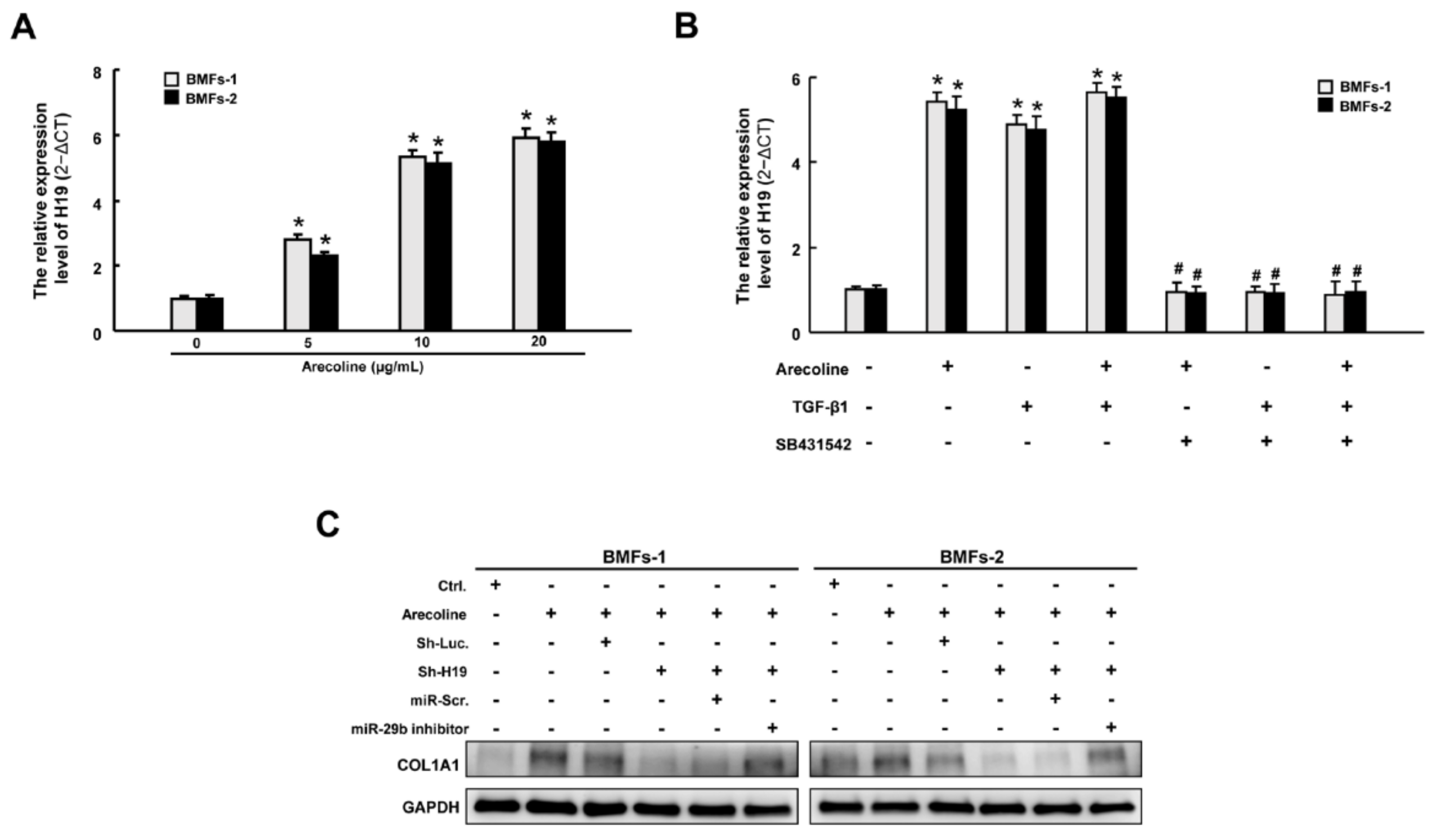
2.3. H19 Promotes Myofibroblast Activation through the Suppression of miR-29b
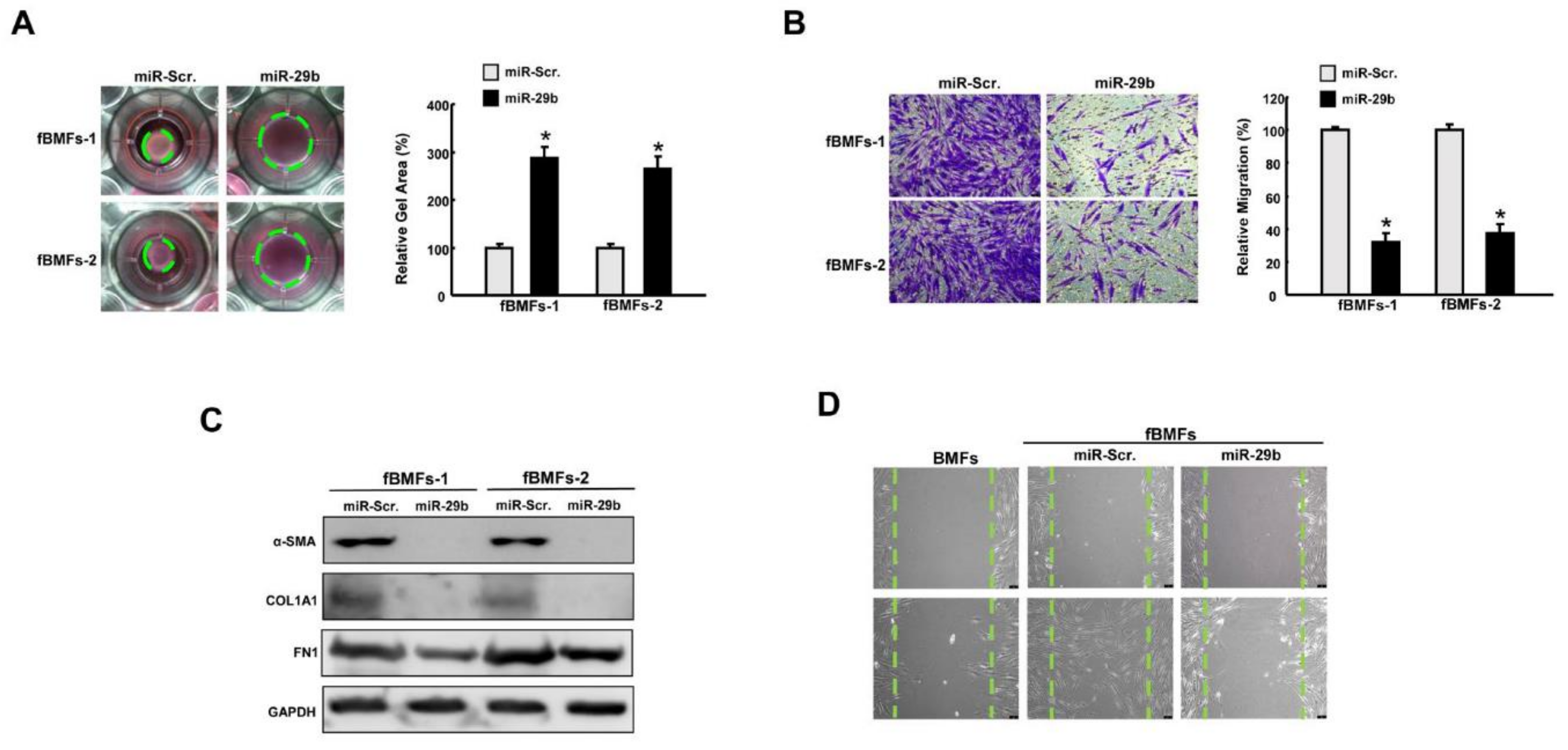
2.4. Overexpression of miR-29b Suppresses Myofibroblast Phenotypes and Expression of Fibrosis Markers
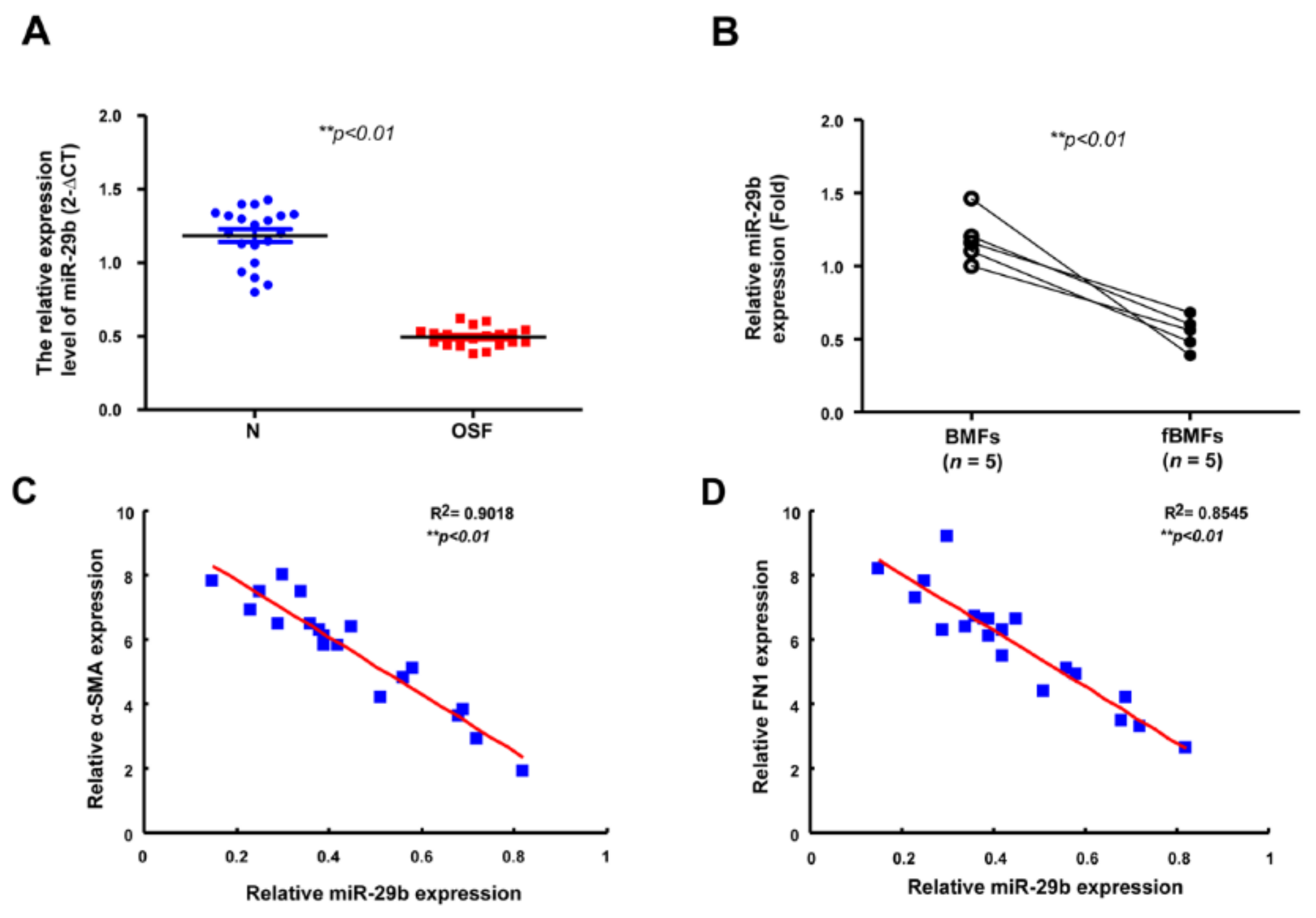
2.5. The Expression of miR-29b Is Downregulated in OSF Specimens and Negatively Correlated with Fibrosis Markers
2.6. H19 Is Induced by Arecoline-Activated TGF-β Pathway
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Tissue Acquisition
4.3. Cell Culture
4.4. RNA Sequencing
4.5. Quantitative Real-Time PCR
4.6. Inhibition and Overexpression of H19
4.7. Collagen Gel Contraction
4.8. Transwell Migration Assay
4.9. Luciferase Reporter Assay
4.10. Western Blot Analysis
4.11. Wound Healing Assay
4.12. Statistical Analysis
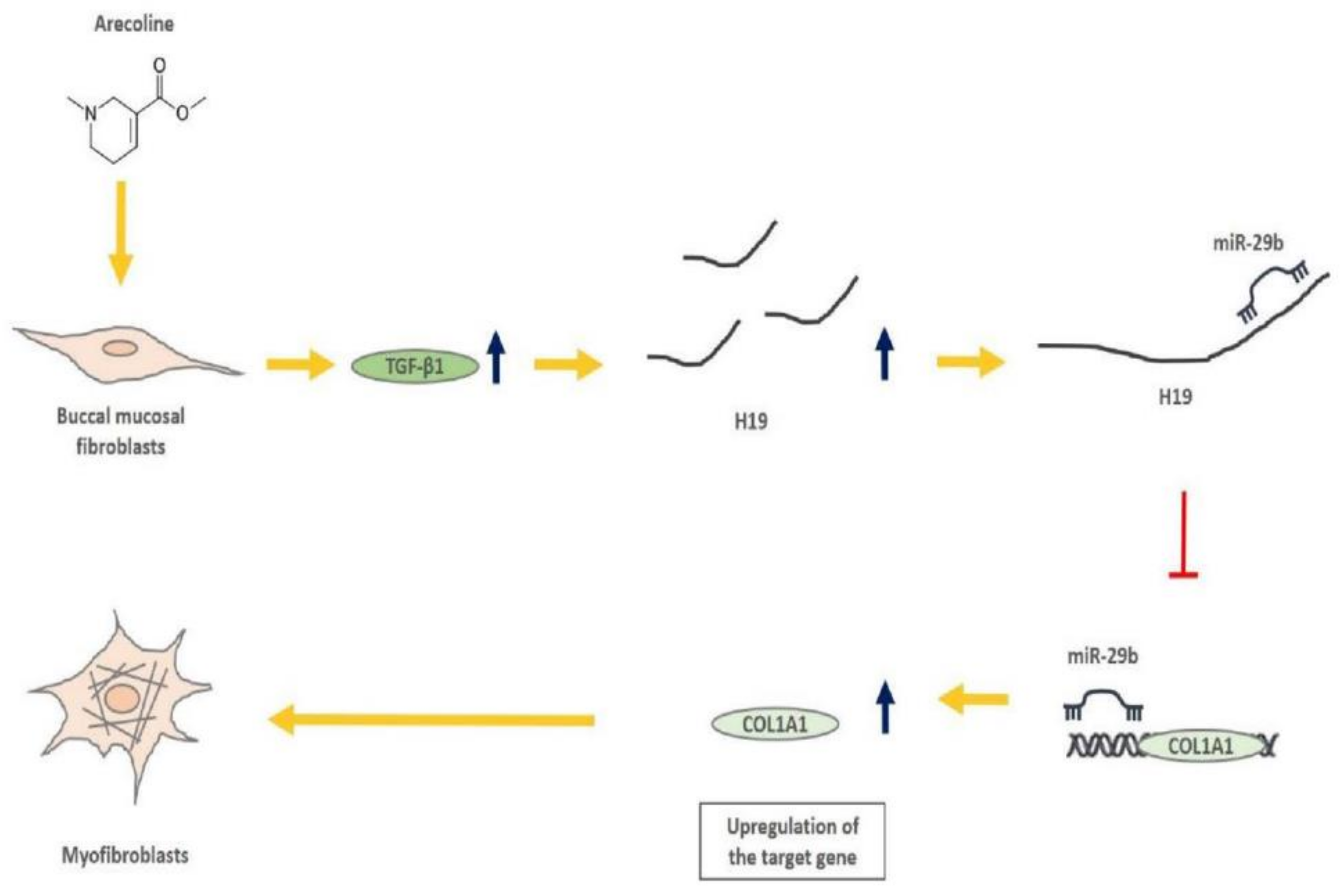
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Warnakulasuriya, S.; Johnson, N.W.; van der Waal, I. Nomenclature and classification of potentially malignant disorders of the oral mucosa. J. Oral Pathol. Med. 2007, 36, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-C.; Tsai, C.-H.; Lai, Y.-L.; Yu, C.-C.; Chi, W.-Y.; Li, J.J.; Chang, W.-W. Arecoline-induced myofibroblast transdifferentiation from human buccal mucosal fibroblasts is mediated by ZEB1. J. Cell. Mol. Med. 2014, 18, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Pant, I.; Kumar, N.; Khan, I.; Rao, S.G.; Kondaiah, P. Role of areca nut induced TGF-β and epithelial-mesenchymal interaction in the pathogenesis of oral submucous fibrosis. PLoS ONE 2015, 10, e0129252. [Google Scholar] [CrossRef] [PubMed]
- Hinz, B.; Celetta, G.; Tomasek, J.J.; Gabbiani, G.; Chaponnier, C. Alpha-smooth muscle actin expression upregulates fibroblast contractile activity. Mol. Biol. Cell 2001, 12, 2730–2741. [Google Scholar] [CrossRef] [PubMed]
- Angadi, P.V.; Kale, A.D.; Hallikerimath, S. Evaluation of myofibroblasts in oral submucous fibrosis: Correlation with disease severity. J. Oral Pathol. Med. 2011, 40, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-J.; Tao, H.; Deng, Z.Y.; Lu, C.; Li, J. Non-coding RNA-mediated epigenetic regulation of liver fibrosis. Metabolism 2015, 64, 1386–1394. [Google Scholar] [CrossRef]
- Tang, P.M.; Zhang, Y.-Y.; Mak, T.S.; Tang, P.C.; Huang, X.-R.; Lan, H.-Y. Transforming growth factor-β signalling in renal fibrosis: From Smads to non-coding RNAs. J. Physiol. 2018, 596, 3493–3503. [Google Scholar] [CrossRef] [PubMed]
- Yao, R.-W.; Wang, Y.; Chen, L.-L. Cellular functions of long noncoding RNAs. Nat. Cell Biol. 2019, 21, 542–551. [Google Scholar] [CrossRef]
- Liao, Y.-W.; Yu, C.-C.; Hsieh, P.-L.; Chang, Y.-C. miR-200b ameliorates myofibroblast transdifferentiation in precancerous oral submucous fibrosis through targeting ZEB2. J. Cell. Mol. Med. 2018, 22, 4130–4138. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Liao, Y.-W.; Hsieh, P.-L.; Lu, M.-Y.; Peng, C.-Y.; Chu, P.-M.; Yang, H.-W.; Huang, Y.-F.; Yu, C.-C.; Yu, C.-H. LncRNA GAS5-AS1 inhibits myofibroblasts activities in oral submucous fibrosis. J. Formos. Med. Assoc. 2018, 117, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Zhu, Y.; He, Z.; Zhang, D.; Guo, F.; Jian, X.; Zhang, C. Long non-coding RNA expression profile associated with malignant progression of oral submucous fibrosis. J. Oncol. 2019, 2019, 6835176. [Google Scholar] [CrossRef]
- Gabory, A.; Ripoche, M.-A.; Yoshimizu, T.; Dandolo, L. The H19 gene: Regulation and function of a non-coding RNA. Cytogenet. Genome Res. 2006, 113, 188–193. [Google Scholar] [CrossRef]
- El-Naggar, A.K.; Lai, S.; Tucker, S.A.; Clayman, G.L.; Goepfert, H.; Hong, W.K.; Huff, V. Frequent loss of imprinting at the IGF2 and H19 genes in head and neck squamous carcinoma. Oncogene 1999, 18, 7063–7069. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cheng, Z.; Dai, L.; Jiang, T.; Jia, L.; Jing, X.; An, L.; Wang, H.; Liu, M. Knockdown of long noncoding RNA H19 represses the progress of pulmonary fibrosis through the Transforming Growth Factor β/Smad3 pathway by regulating microRNA 140. Mol. Cell. Biol. 2019, 39, e00143-19. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Guo, Z.; Xie, W.; Jin, W.; Zhu, D.; Chen, S.; Ren, T. The lncRNA H19 mediates pulmonary fibrosis by regulating the miR-196a/COL1A1 axis. Inflammation 2018, 41, 896–903. [Google Scholar] [CrossRef]
- Tang, Y.; He, R.; An, J.; Deng, P.; Huang, L.; Yang, W. The effect of H19-miR-29b interaction on bleomycin-induced mouse model of idiopathic pulmonary fibrosis. Biochem. Biophys. Res. Commun. 2016, 479, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, I.; Kikkawa, N.; Matsushita, R.; Kato, M.; Kurozumi, A.; Nishikawa, R.; Goto, Y.; Koshizuka, K.; Hanazawa, T.; Enokida, H. Tumor-suppressive microRNAs (miR-26a/b, miR-29a/b/c and miR-218) concertedly suppressed metastasis-promoting LOXL2 in head and neck squamous cell carcinoma. J. Hum. Genet. 2016, 61, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-W.; Lu, M.-Y.; Chiu, Y.-W.; Liao, Y.-W.; Huang, Y.-F.; Chueh, P.-J.; Hsieh, P.-L.; Yu, C.-C. Hinokitiol ablates myofibroblast activation in precancerous oral submucous fibrosis by targeting Snail. Environ. Toxicol. 2018, 33, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wang, L.; Lu, L.; Jiang, P.; Sun, H.; Wang, H. Inhibition of miR-29 by TGF-beta-Smad3 signaling through dual mechanisms promotes transdifferentiation of mouse myoblasts into myofibroblasts. PLoS ONE 2012, 7, e33766. [Google Scholar] [CrossRef]
- Khan, I.; Kumar, N.; Pant, I.; Narra, S.; Kondaiah, P. Activation of TGF-β pathway by areca nut constituents: A possible cause of oral submucous fibrosis. PLoS ONE 2012, 7, e51806. [Google Scholar] [CrossRef] [PubMed]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef]
- Hao, Y.; Crenshaw, T.; Moulton, T.; Newcomb, E.; Tycko, B. Tumour-suppressor activity of H19 RNA. Nature 1993, 365, 764–767. [Google Scholar] [CrossRef] [PubMed]
- Raveh, E.; Matouk, I.J.; Gilon, M.; Hochberg, A. The H19 Long non-coding RNA in cancer initiation, progression and metastasis—A proposed unifying theory. Mol. Cancer 2015, 14, 184. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-M.; Lin, Z.-Y.; Yang, Z.-H.; Wang, Y.-Y.; Wan, D.; Zhong, J.-L.; Zhuang, P.-L.; Huang, Z.-Q.; Zhou, B.; Chen, W.-L. IncRNA H19 promotes tongue squamous cell carcinoma progression through β-catenin/GSK3β/EMT signaling via association with EZH2. Am. J. Transl. Res. 2017, 9, 3474–3486. [Google Scholar]
- Wu, T.; Qu, L.; He, G.; Tian, L.; Li, L.; Zhou, H.; Jin, Q.; Ren, J.; Wang, Y.; Wang, J.; et al. Regulation of laryngeal squamous cell cancer progression by the lncRNA H19/miR-148a-3p/DNMT1 axis. Oncotarget 2016, 7, 11553–11566. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.J.; Liu, L.P.; Tao, H.; Hu, W.; Shi, P.; Deng, Z.Y.; Li, J. MeCP2 silencing of LncRNA H19 controls hepatic stellate cell proliferation by targeting IGF1R. Toxicology 2016, 359–360, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Choong, O.K.; Chen, C.-Y.; Zhang, J.; Lin, J.-H.; Lin, P.-J.; Ruan, S.-C.; Kamp, T.J.; Hsieh, C.H. Hypoxia-induced H19/YB-1 cascade modulates cardiac remodeling after infarction. Theranostics 2019, 9, 6550–6567. [Google Scholar] [CrossRef]
- Song, Y.; Liu, C.; Liu, X.; Trottier, J.; Beaudoin, M.; Zhang, L.; Pope, C.; Peng, G.; Barbier, O.; Zhong, X.; et al. H19 promotes cholestatic liver fibrosis by preventing ZEB1-mediated inhibition of epithelial cell adhesion molecule. Hepatology 2017, 66, 1183–1196. [Google Scholar] [CrossRef] [PubMed]
- Sekiya, Y.; Ogawa, T.; Yoshizato, K.; Ikeda, K.; Kawada, N. Suppression of hepatic stellate cell activation by microRNA-29b. Biochem. Biophys. Res. Commun. 2011, 412, 74–79. [Google Scholar] [CrossRef]
- Van Rooij, E.; Sutherland, L.B.; Thatcher, J.E.; DiMaio, J.M.; Naseem, R.H.; Marshall, W.S.; Hill, J.A.; Olson, E.N. Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc. Natl. Acad. Sci. USA 2008, 105, 13027–13032. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Meng, X.-M.; Huang, X.R.; Chung, A.C.; Feng, Y.L.; Hui, D.S.; Yu, C.-M.; Sung, J.J.; Lan, H.Y. miR-29 inhibits bleomycin-induced pulmonary fibrosis in mice. Mol. Ther. 2012, 20, 1251–1260. [Google Scholar] [CrossRef]
- Qin, W.; Chung, A.C.; Huang, X.R.; Meng, X.M.; Hui, D.S.; Yu, C.-M.; Sung, J.J.; Lan, H.Y. TGF-β/Smad3 signaling promotes renal fibrosis by inhibiting miR-29. J. Am. Soc. Nephrol. 2011, 22, 1462–1474. [Google Scholar] [CrossRef]
- Postlethwaite, A.E.; Seyer, J.M.; Kang, A.H. Chemotactic attraction of human fibroblasts to type I, II, and III collagens and collagen-derived peptides. Proc. Natl. Acad. Sci. USA 1978, 75, 871–875. [Google Scholar] [CrossRef]
- Yu, C.H.; Fang, C.Y.; Yu, C.C.; Hsieh, P.L.; Liao, Y.W.; Tsai, L.L.; Chu, P.M. LINC00312/YBX1 axis regulates myofibroblast activities in oral submucous fibrosis. Int. J. Mol. Sci. 2020, 21, 2979. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, C.-C.; Liao, Y.-W.; Hsieh, P.-L.; Chang, Y.-C. Targeting lncRNA H19/miR-29b/COL1A1 Axis Impedes Myofibroblast Activities of Precancerous Oral Submucous Fibrosis. Int. J. Mol. Sci. 2021, 22, 2216. https://doi.org/10.3390/ijms22042216
Yu C-C, Liao Y-W, Hsieh P-L, Chang Y-C. Targeting lncRNA H19/miR-29b/COL1A1 Axis Impedes Myofibroblast Activities of Precancerous Oral Submucous Fibrosis. International Journal of Molecular Sciences. 2021; 22(4):2216. https://doi.org/10.3390/ijms22042216
Chicago/Turabian StyleYu, Cheng-Chia, Yi-Wen Liao, Pei-Ling Hsieh, and Yu-Chao Chang. 2021. "Targeting lncRNA H19/miR-29b/COL1A1 Axis Impedes Myofibroblast Activities of Precancerous Oral Submucous Fibrosis" International Journal of Molecular Sciences 22, no. 4: 2216. https://doi.org/10.3390/ijms22042216
APA StyleYu, C.-C., Liao, Y.-W., Hsieh, P.-L., & Chang, Y.-C. (2021). Targeting lncRNA H19/miR-29b/COL1A1 Axis Impedes Myofibroblast Activities of Precancerous Oral Submucous Fibrosis. International Journal of Molecular Sciences, 22(4), 2216. https://doi.org/10.3390/ijms22042216








