Molecular Identification of Endophytic Bacteria in Leucojum aestivum In Vitro Culture, NMR-Based Metabolomics Study and LC-MS Analysis Leading to Potential Amaryllidaceae Alkaloid Production
Abstract
1. Introduction
2. Results and Discussion
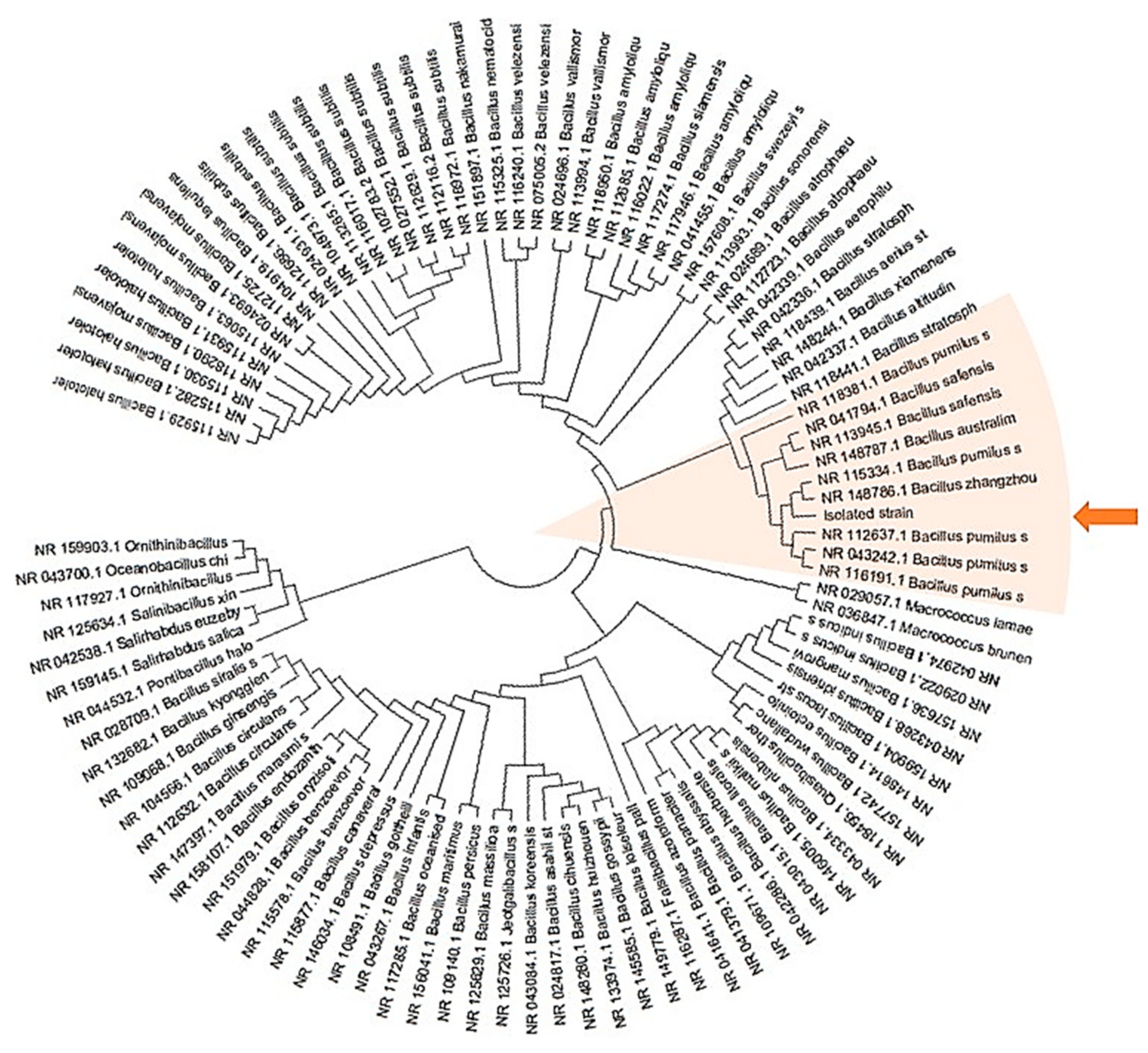
2.1. Isolation and Identification of Endophytic Bacteria from Leucojum aestivum In Vitro Bulblets
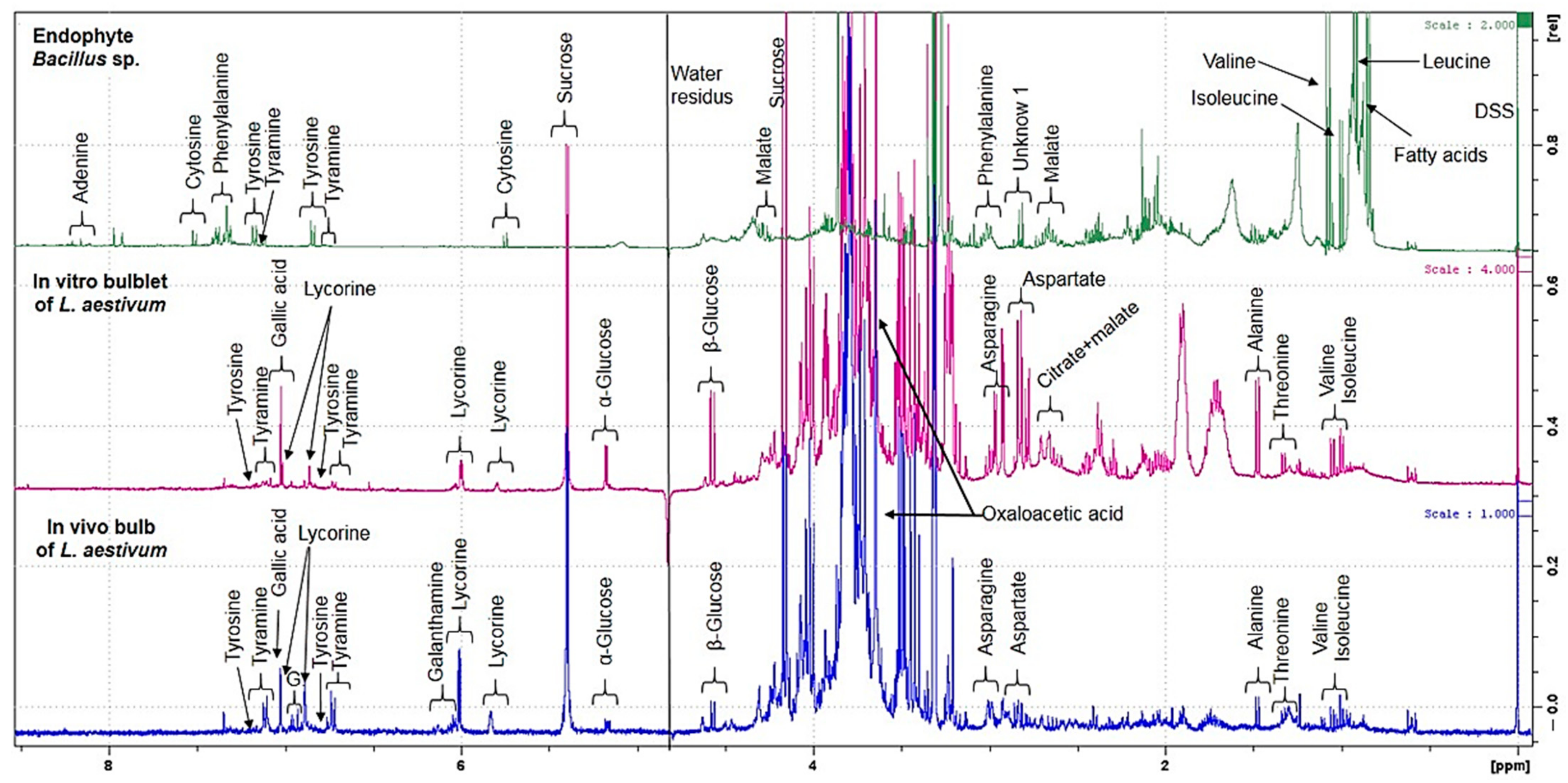
2.2. Untargeted 1H NMR-Based Metabolomics: Identification and Quantification
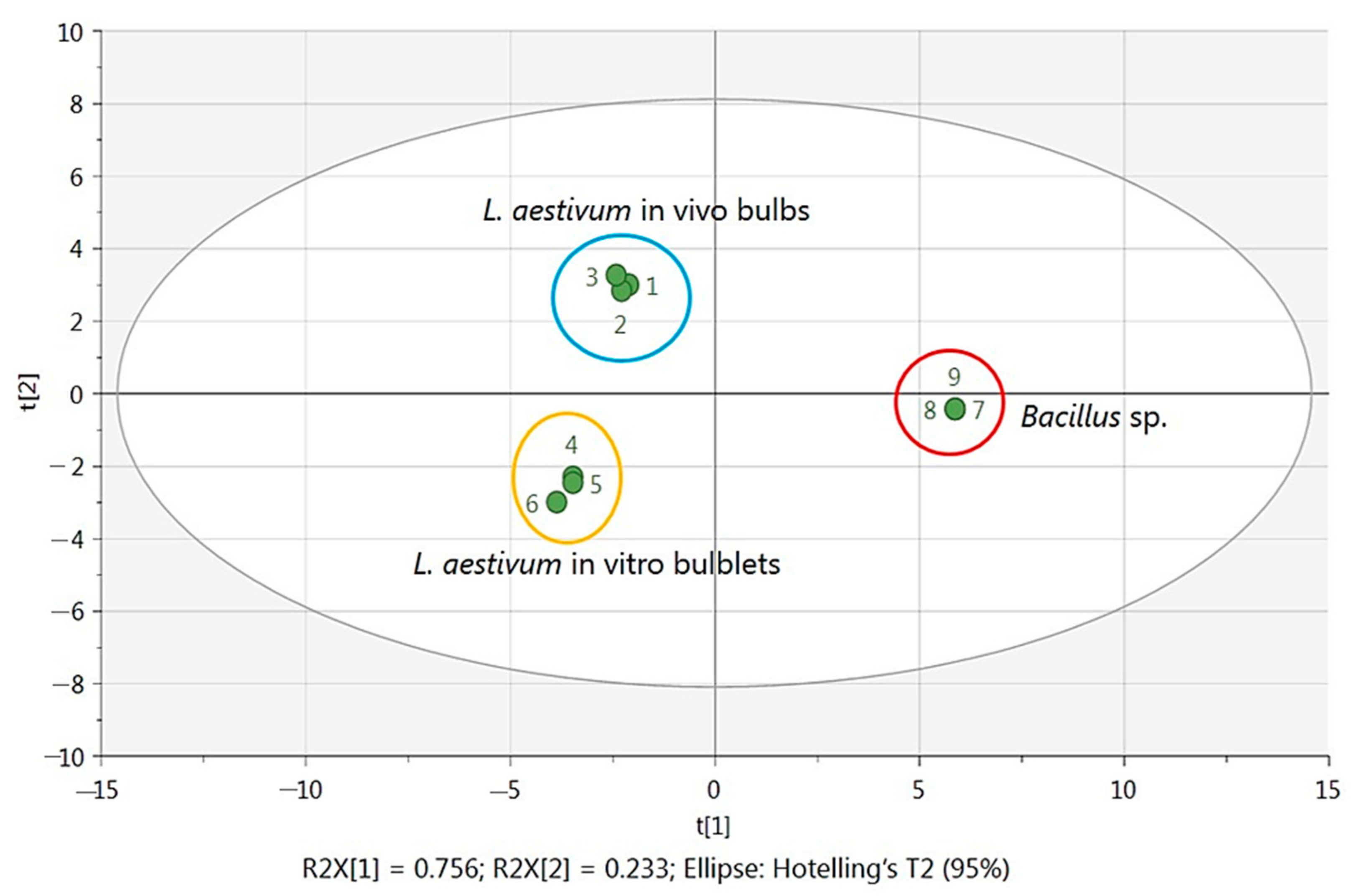
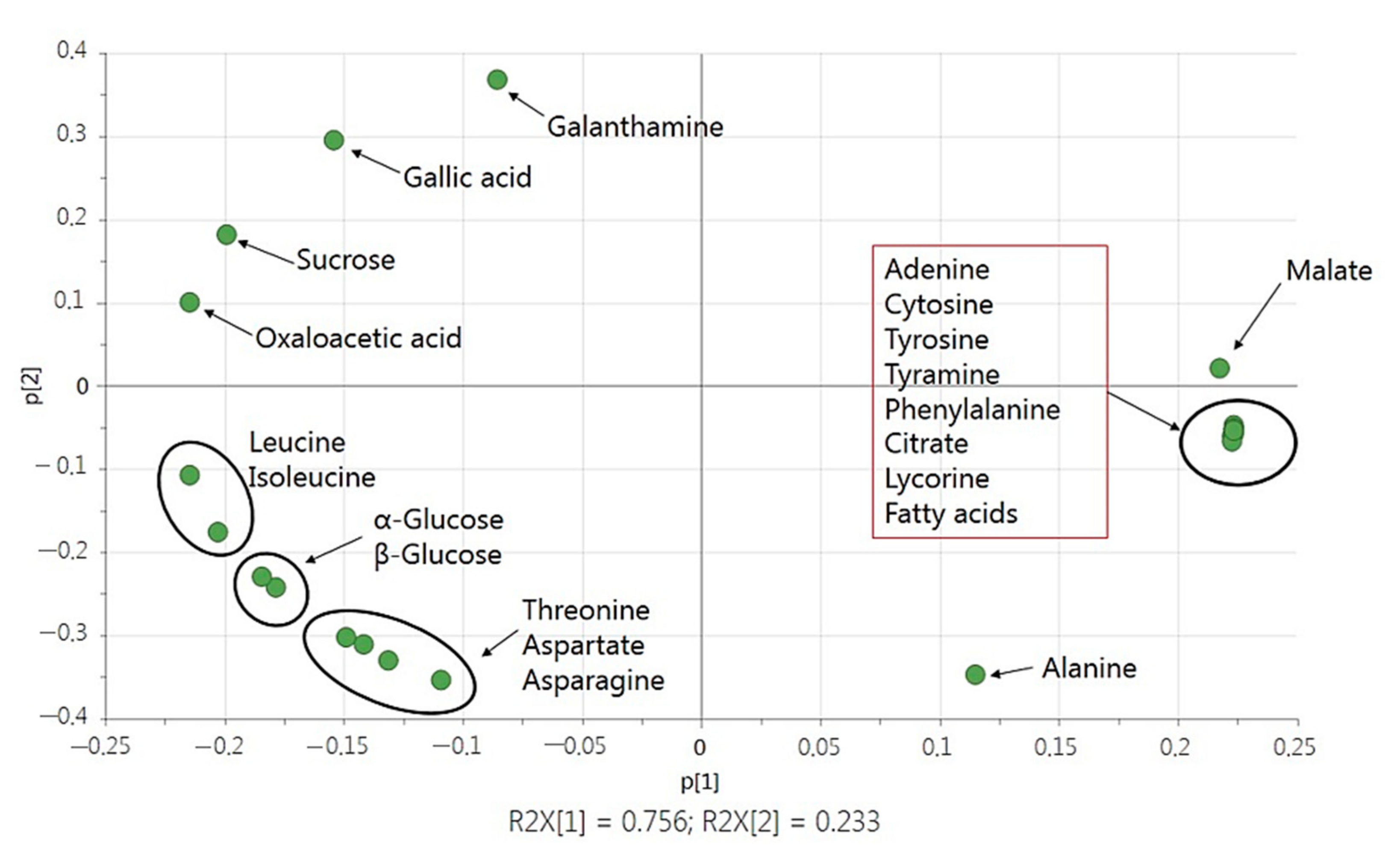
2.3. Statistical Analysis with Multivariate Data Analysis
2.4. Quantification by NMR
2.5. Target Research of Amaryllidaceae Alkaloids by 1H NMR, LC-MS and GC-MS Analysis in Bacterial Endophyte Bacillus sp.
3. Materials and Methods
3.1. Chemicals
3.2. Plant Materials, Preparation of In Vitro Culture of L. aestivum
3.3. Isolation of Endophytic Bacteria
3.4. Identification of Endophytic Bacteria and PCR Amplification
3.5. In Silico Analysis
3.6. Preparation of Plants Samples and Endophytic Bacteria for Untargeted NMR-Based Metabolomics
3.7. Preparation of Enriched Endophytic Bacteria Sample for NMR, LC-MS and GC-MS Analysis
3.8. NMR Equipment and Experimental Conditions
3.9. Softwares and the Statistical Analysis
3.10. LC-MS and GC-MS Equipments and Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Glossary
| MeOH | Methanol |
| CH3CN | Acetonitrile |
| HCOOH | Formic acid |
| NH4OH | Ammonium hydroxide |
| LA | Leucojum aestivum |
| MS | Murashige and Skoog |
| NA | Nutrient agar |
| DW | Dry weight |
| SPE | Solid phase extraction |
| NMR | Nuclear Magnetic Resonance |
| ESI-HRMS | Electrospray Ionization—High Resolution Mass Spectrometry |
| MS | Mass spectrometry |
| LC | Liquid chromatography |
| GC | Gas chromatography |
References
- Rinner, U.; Dank, C.; Hudlicky, T. Galanthamine. Targets Heterocycl. Syst. 2016, 283. [Google Scholar] [CrossRef]
- He, M.; Qu, C.; Gao, O.; Hu, X.; Hong, X. Biological and Pharmacological Activities of Amaryllidaceae Alkaloids. Rsc Adv. 2015, 5, 16562–16574. [Google Scholar] [CrossRef]
- Szlávik, L.; Gyuris, Á.; Minárovits, J.; Forgo, P.; Molnár, J.; Hohmann, J. Alkaloids from Leucojum vernum and Antiretroviral Activity of Amaryllidaceae Alkaloids. Planta Med. 2004, 70, 871–873. [Google Scholar] [CrossRef] [PubMed]
- Khalifa, M.F.; Attia, E.Z.; Fahim, J.R.; Kamel, M.S. An Overview on the Chemical and Biological Aspects of Lycorine Alkaloid. J. Adv. Biomed. Pharm. Sci. 2018, 1, 41–49. [Google Scholar] [CrossRef]
- Cao, Z.; Yang, P.; Zhou, Q. Multiple Biological Functions and Pharmacological Effects of Lycorine. Sci. China Chem. 2013, 56, 1382–1391. [Google Scholar] [CrossRef]
- Osorio, E.J.; Robledo, S.M.; Bastida, J. Chapter 2, Alkaloids with Antiprotozoal Activity. In The Alkaloids: Chemistry and Biology; Cordell, G.A., Ed.; Academic Press: Cambridge, MA, USA, 2008; Volume 66, pp. 113–190. [Google Scholar]
- Ying, X.; Huang, A.; Xing, Y.; Lan, L.; Yi, Z.; He, P. Lycorine Inhibits Breast Cancer Growth and Metastasis via Inducing Apoptosis and Blocking Src/FAK-Involved Pathway. Sci. China Life Sci. 2017, 60, 417–428. [Google Scholar] [CrossRef]
- Gouda, S.; Das, G.; Sen, S.K.; Shin, H.-S.; Patra, J.K. Endophytes: A Treasure House of Bioactive Compounds of Medicinal Importance. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Khiralla, A.; Spina, R.; Saliba, S.; Laurain-Mattar, D. Diversity of Natural Products of the Genera Curvularia and Bipolaris. Fungal Biol. Rev. 2019, 33, 101–122. [Google Scholar] [CrossRef]
- Na, R.; Jiajia, L.; Dongliang, Y.; Yingzi, P.; Juan, H.; Xiong, L.; Nana, Z.; Jing, Z.; Yitian, L. Indentification of Vincamine Indole Alkaloids Producing Endophytic Fungi Isolated from Nerium indicum, Apocynaceae. Microbiol. Res. 2016, 192, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Khiralla, A.; Spina, R.; Varbanov, M.; Philippot, S.; Lemiere, P.; Slezack-Deschaumes, S.; André, P.; Mohamed, I.; Yagi, S.M.; Laurain-Mattar, D. Evaluation of Antiviral, Antibacterial and Antiproliferative Activities of the Endophytic Fungus Curvularia papendorfii, and Isolation of a New Polyhydroxyacid. Microorganisms 2020, 8, 1353. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, J.; Li, Y.; Wen, J.; Wang, R. Bacterial Endophytes from Lycoris radiata Promote the Accumulation of Amaryllidaceae Alkaloids. Microbiol. Res. 2020, 239, 126501. [Google Scholar] [CrossRef]
- Sebola, T.E.; Uche-Okereafor, N.C.; Tapfuma, K.I.; Mekuto, L.; Green, E.; Mavumengwana, V. Evaluating Antibacterial and Anticancer Activity of Crude Extracts of Bacterial Endophytes from Crinum macowanii Baker Bulbs. MicrobiologyOpen 2019, 8, e914. [Google Scholar] [CrossRef] [PubMed]
- Rebotiloe, F.M.; Eunice, U.-J.; Mahloro, H.S.-D. Isolation and Identification of Bacterial Endophytes from Crinum macowanii Baker. Afr. J. Biotechnol. 2018, 17, 1040–1047. [Google Scholar] [CrossRef]
- Wang, W.-X.; Kusari, S.; Sezgin, S.; Lamshöft, M.; Kusari, P.; Kayser, O.; Spiteller, M. Hexacyclopeptides Secreted by an Endophytic Fungus Fusarium solani N06 Act as Crosstalk Molecules in Narcissus tazetta. Appl. Microbiol. Biotechnol. 2015, 99, 7651–7662. [Google Scholar] [CrossRef] [PubMed]
- Deborde, C.; Moing, A.; Roch, L.; Jacob, D.; Rolin, D.; Giraudeau, P. Plant Metabolism as Studied by NMR Spectroscopy. Prog. Nucl. Magn. Reson. Spectrosc. 2017, 102–103, 61–97. [Google Scholar] [CrossRef] [PubMed]
- Vignoli, A.; Ghini, V.; Meoni, G.; Licari, C.; Takis, P.G.; Tenori, L.; Turano, P.; Luchinat, C. High-Throughput Metabolomics by 1D NMR. Angew. Chem. Int. Ed. 2019, 58, 968–994. [Google Scholar] [CrossRef]
- Courant, F.; Antignac, J.-P.; Dervilly-Pinel, G.; Bizec, B.L. Basics of Mass Spectrometry Based Metabolomics. Proteomics 2014, 14, 2369–2388. [Google Scholar] [CrossRef]
- Covington, B.C.; McLean, J.A.; Bachmann, B.O. Comparative Mass Spectrometry-Based Metabolomics Strategies for the Investigation of Microbial Secondary Metabolites. Nat. Prod. Rep. 2017, 34, 6–24. [Google Scholar] [CrossRef]
- Kostidis, S.; Addie, R.D.; Morreau, H.; Mayboroda, O.A.; Giera, M. Quantitative NMR Analysis of Intra- and Extracellular Metabolism of Mammalian Cells: A Tutorial. Anal. Chim. Acta 2017, 980, 1–24. [Google Scholar] [CrossRef]
- Kosmides, A.K.; Kamisoglu, K.; Calvano, S.E.; Corbett, S.A.; Androulakis, I.P. Metabolomic Fingerprinting: Challenges and Opportunities. Crit. Rev. Biomed. Eng. 2013, 41, 205–221. [Google Scholar] [CrossRef] [PubMed]
- Berkov, S.; Bastida, J.; Viladomat, F.; Codina, C. Development and Validation of a GC–MS Method for Rapid Determination of Galanthamine in Leucojum aestivum and Narcissus ssp.: A Metabolomic Approach. Talanta 2011, 83, 1455–1465. [Google Scholar] [CrossRef]
- Singh, A.; Desgagné-Penix, I. Transcriptome and Metabolome Profiling of Narcissus pseudonarcissus ‘King Alfred’ Reveal Components of Amaryllidaceae Alkaloid Metabolism. Sci. Rep. 2017, 7, 17356. [Google Scholar] [CrossRef]
- Holmes, E.; Tang, H.; Wang, Y.; Seger, C. The Assessment of Plant Metabolite Profiles by NMR-Based Methodologies. Planta Med. 2006, 72, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Lubbe, A.; Pomahačová, B.; Choi, Y.H.; Verpoorte, R. Analysis of Metabolic Variation and Galanthamine Content in Narcissus Bulbs by 1H NMR. Phytochem. Anal. 2010, 21, 66–72. [Google Scholar] [CrossRef]
- Lubbe, A.; Gude, H.; Verpoorte, R.; Choi, Y.H. Seasonal Accumulation of Major Alkaloids in Organs of Pharmaceutical Crop Narcissus Carlton. Phytochemistry 2013, 88, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Akram, M.N.; Verpoorte, R.; Pomahačová, B. Effect of Bulb Age on Alkaloid Contents of Narcissus pseudonarcissus Bulbs. S. Afr. J. Bot. 2021, 136, 182–189. [Google Scholar] [CrossRef]
- Ferdausi, A.; Chang, X.; Hall, A.; Jones, M. Galanthamine Production in Tissue Culture and Metabolomic Study on Amaryllidaceae Alkaloids in Narcissus pseudonarcissus cv. Carlton. Ind. Crop. Prod. 2020, 144, 112058. [Google Scholar] [CrossRef]
- Saliba, S.; Ptak, A.; Boisbrun, M.; Spina, R.; Dupire, F.; Laurain-Mattar, D. Stimulating Effect of Both 4’-O-Methylnorbelladine Feeding and Temporary Immersion Conditions on Galanthamine and Lycorine Production by Leucojum aestivum L. Bulblets. Eng. Life Sci. 2016, 16, 731–739. [Google Scholar] [CrossRef]
- Ptak, A.; El Tahchy, A.; Skrzypek, E.; Wójtowicz, T.; Laurain-Mattar, D. Influence of Auxins on Somatic Embryogenesis and Alkaloid Accumulation in Leucojum aestivum Callus. Cent. Eur. J. Biol. 2013, 8, 591–599. [Google Scholar] [CrossRef]
- Ptak, A.; Morańska, E.; Skrzypek, E.; Warchoł, M.; Spina, R.; Laurain-Mattar, D.; Simlat, M. Carbohydrates Stimulated Amaryllidaceae Alkaloids Biosynthesis in Leucojum aestivum L. Plants Cultured in RITA® Bioreactor. PeerJ 2020, 8, e8688. [Google Scholar] [CrossRef]
- Lata, H.; Li, X.C.; Silva, B.; Moraes, R.M.; Halda-Alija, L. Identification of IAA-Producing Endophytic Bacteria from Micropropagated Echinacea Plants Using 16S RRNA Sequencing. Plant Cell Tissue Organ Cult. 2006, 85, 353–359. [Google Scholar] [CrossRef]
- Pham, N.T.; Meier-Dinkel, A.; Höltken, A.M.; Quambusch, M.; Mahnkopp, F.; Winkelmann, T. Endophytic Bacterial Communities in in Vitro Shoot Cultures Derived from Embryonic Tissue of Hybrid Walnut (Juglans × intermedia). Plant Cell Tissue Organ Cult. 2017, 130, 153–165. [Google Scholar] [CrossRef]
- Quambusch, M.; Brümmer, J.; Haller, K.; Winkelmann, T.; Bartsch, M. Dynamics of Endophytic Bacteria in Plant in Vitro Culture: Quantification of Three Bacterial Strains in Prunus avium in Different Plant Organs and in Vitro Culture Phases. Plant Cell Tissue Organ Cult. 2016, 126, 305–317. [Google Scholar] [CrossRef]
- Orlikowska, T.; Nowak, K.; Reed, B. Bacteria in the Plant Tissue Culture Environment. Plant Cell Tissue Organ Cult. 2017, 128, 487–508. [Google Scholar] [CrossRef]
- Thomas, P. A Three-Step Screening Procedure for Detection of Covert and Endophytic Bacteria in Plant Tissue Cultures. Curr. Sci. 2004, 87, 67–72. [Google Scholar]
- Fongicides Sonata®: Bayer-Agri, Traitement Phytopharmaceutique Pour La Protection Des Cultures—Sonata®. Available online: https://www.bayer-agri.fr/produits/fiche/fongicides-sonata/ (accessed on 26 August 2020).
- Araújo, W.L.; Marcon, J.; Maccheroni, W.; van Elsas, J.D.; van Vuurde, J.W.L.; Azevedo, J.L. Diversity of Endophytic Bacterial Populations and Their Interaction with Xylella fastidiosa in Citrus Plants. Appl. Environ. Microbiol. 2002, 68, 4906–4914. [Google Scholar] [CrossRef]
- Jasim, B.; Joseph, A.A.; John, C.J.; Mathew, J.; Radhakrishnan, E.K. Isolation and Characterization of Plant Growth Promoting Endophytic Bacteria from the Rhizome of Zingiber officinale. 3 Biotech 2014, 4, 197–204. [Google Scholar] [CrossRef]
- Ali, M.; Abbasi, B.H.; Ahmad, N.; Ali, S.S.; Ali, S.; Ali, G.S. Sucrose-Enhanced Biosynthesis of Medicinally Important Antioxidant Secondary Metabolites in Cell Suspension Cultures of Artemisia absinthium L. Bioprocess Biosyst. Eng. 2016, 39, 1945–1954. [Google Scholar] [CrossRef] [PubMed]
- Bastida, J.; Berkov, S.; Torras, L.; Pigni, N.B.; de Andrade, J.P.; Martínez, V.; Codina, C.; Viladomat, F. 3. Chemical and Biological Aspects of Amaryllidaceae Alkaloids. In Recent Advances in Pharmaceutical Sciences; Transworld Research Network: Trivandrum, India, 2011; pp. 65–100. ISBN 978-81-7895-528-5. [Google Scholar]
- Petruczynik, A.; Misiurek, J.; Tuzimski, T.; Uszyński, R.; Szymczak, G.; Chernetskyy, M.; Waksmundzka-Hajnos, M. Comparison of Different HPLC Systems for Analysis of Galantamine and Lycorine in Various Species of Amaryllidaceae Family. J. Liq. Chromatogr. Relat. Technol. 2016, 39, 574–579. [Google Scholar] [CrossRef]
- Gussev, C.; Bosseva, Y.; Pandova, B.; Yanev, S.; Stanilova, M. Resource Assessment of Leucojum aestivum L. (Amaryllidaceae) Populations in Bulgaria. Bocconea 2007, 21, 405–411. [Google Scholar]
- Georgiev, V.; Ivanov, I.; Pavlov, A. Recent Progress in Amaryllidaceae Biotechnology. Molecules 2020, 25, 4670. [Google Scholar] [CrossRef] [PubMed]
- Laurain-Mattar, D.; Ptak, A. Amaryllidaceae Alkaloid Accumulation by Plant In Vitro Systems. In Bioprocessing of Plant In Vitro Systems; Pavlov, A., Bley, T., Eds.; Reference Series in Phytochemistry; Springer: Cham, Switzerland, 2018; pp. 203–223. ISBN 978-3-319-54600-1. [Google Scholar]
- Wang, M.; Carver, J.J.; Phelan, V.V.; Sanchez, L.M.; Garg, N.; Peng, Y.; Nguyen, D.D.; Watrous, J.; Kapono, C.A.; Luzzatto-Knaan, T.; et al. Sharing and Community Curation of Mass Spectrometry Data with Global Natural Products Social Molecular Networking. Nat. Biotechnol. 2016, 34, 828–837. [Google Scholar] [CrossRef]
- Guijas, C.; Montenegro-Burke, J.R.; Domingo-Almenara, X.; Palermo, A.; Warth, B.; Hermann, G.; Koellensperger, G.; Huan, T.; Uritboonthai, W.; Aisporna, A.E.; et al. METLIN: A Technology Platform for Identifying Knowns and Unknowns. Anal. Chem. 2018, 90, 3156–3164. [Google Scholar] [CrossRef]
- MassBank Japan. Mass Spectral DataBase. Available online: http://www.massbank.jp/ (accessed on 16 April 2020).
- Dührkop, K.; Fleischauer, M.; Ludwig, M.; Aksenov, A.A.; Melnik, A.V.; Meusel, M.; Dorrestein, P.C.; Rousu, J.; Böcker, S. SIRIUS 4: A Rapid Tool for Turning Tandem Mass Spectra into Metabolite Structure Information. Nat. Methods 2019, 16, 299–302. [Google Scholar] [CrossRef] [PubMed]
- Tarakemeh, A.; Azizi, M.; Rowshan, V.; Salehi, H.; Spina, R.; Dupire, F.; Arouie, H.; Laurain-Mattar, D. Screening of Amaryllidaceae Alkaloids in Bulbs and Tissue Cultures of Narcissus papyraceus and Four Varieties of N. tazetta. J. Pharm. Biomed. Anal. 2019, 172, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Berkov, S.; Codina, C.; Viladomat, F.; Bastida, J. Alkaloids from Galanthus nivalis. Phytochemistry 2007, 68, 1791–1798. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Kumar, A.; Singh, R.; Pandey, K.D. Endophytic Bacteria: A New Source of Bioactive Compounds. 3 Biotech 2017, 7, 315. [Google Scholar] [CrossRef] [PubMed]
- Soujanya, K.N.; Siva, R.; Mohana Kumara, P.; Srimany, A.; Ravikanth, G.; Mulani, F.A.; Aarthy, T.; Thulasiram, H.V.; Santhoshkumar, T.R.; Nataraja, K.N.; et al. Camptothecin-Producing Endophytic Bacteria from Pyrenacantha volubilis Hook. (Icacinaceae): A Possible Role of a Plasmid in the Production of Camptothecin. Phytomedicine Int. J. Phytother. Phytopharm. 2017, 36, 160–167. [Google Scholar] [CrossRef]
- Ek-Ramos, M.J.; Gomez-Flores, R.; Orozco-Flores, A.A.; Rodríguez-Padilla, C.; González-Ochoa, G.; Tamez-Guerra, P. Bioactive Products From Plant-Endophytic Gram-Positive Bacteria. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Villarreal-Delgado, M.F.; Villa-Rodríguez, E.D.; Cira-Chávez, L.A.; Estrada-Alvarado, M.I.; Parra-Cota, F.I.; de los Santos-Villalobos, S.; Villarreal-Delgado, M.F.; Villa-Rodríguez, E.D.; Cira-Chávez, L.A.; Estrada-Alvarado, M.I.; et al. The Genus Bacillus as a Biological Control Agent and Its Implications in the Agricultural Biosecurity. Rev. Mex. Fitopatol. 2018, 36, 95–130. [Google Scholar] [CrossRef]
- NIST Standard Reference Databases: Analytical Chemistry. Available online: https://www.nist.gov/srd/nist-standard-reference-databases-analytical-chemistry (accessed on 25 April 2020).
- Berkov, S.; Osorio, E.; Viladomat, F.; Bastida, J. Chapter Two—Chemodiversity, chemotaxonomy and chemoecology of Amaryllidaceae alkaloids. In The Alkaloids: Chemistry and Biology; Knölker, H.-J., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 83, pp. 113–185. [Google Scholar]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Ptak, A.; El Tahchy, A.; Dupire, F.; Boisbrun, M.; Henry, M.; Chapleur, Y.; Moś, M.; Laurain-Mattar, D. LCMS and GCMS for the Screening of Alkaloids in Natural and in vitro Extracts of Leucojum aestivum. J. Nat. Prod. 2009, 72, 142–147. [Google Scholar] [CrossRef]
- El Tahchy, A.; Ptak, A.; Boisbrun, M.; Barre, E.; Guillou, C.; Dupire, F.; Chrétien, F.; Henry, M.; Chapleur, Y.; Laurain-Mattar, D. Kinetic Study of the Rearrangement of Deuterium-Labeled 4′-O-Methylnorbelladine in Leucojum aestivum Shoot Cultures by Mass Spectrometry. Influence of Precursor Feeding on Amaryllidaceae Alkaloid Accumulation. J. Nat. Prod. 2011, 74, 2356–2361. [Google Scholar] [CrossRef]
- Ptak, A.; Simlat, M.; Kwiecień, M.; Laurain-Mattar, D. Leucojum aestivum Plants Propagated in in vitro Bioreactor Culture and on Solid Media Containing Cytokinins. Eng. Life Sci. 2013, 13, 261–270. [Google Scholar] [CrossRef]
- Gürtler, V.; Stanisich, V.A. New Approaches to Typing and Identification of Bacteria Using the 16S-23S RDNA Spacer Region. Microbiol. Read. Engl. 1996, 142, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Nei, M. Estimation of the Number of Nucleotide Substitutions in the Control Region of Mitochondrial DNA in Humans and Chimpanzees. Mol. Biol. Evol. 1993, 10, 512–526. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Choi, Y.H.; Verpoorte, R. NMR-Based Metabolomic Analysis of Plants. Nat. Protoc. 2010, 5, 536–549. [Google Scholar] [CrossRef]
- Verpoorte, R.; Choi, Y.H.; Kim, H.K. NMR-Based Metabolomics at Work in Phytochemistry. Phytochem. Rev. 2007, 6, 3–14. [Google Scholar] [CrossRef]
- Larive, C.K.; Barding, G.A.; Dinges, M.M. NMR Spectroscopy for Metabolomics and Metabolic Profiling. Anal. Chem. 2015, 87, 133–146. [Google Scholar] [CrossRef]
- Ullrich, S.F.; Averesch, N.J.H.; Castellanos, L.; Choi, Y.H.; Rothauer, A.; Kayser, O. Discrimination of Wild Types and Hybrids of Duboisia myoporoides and Duboisia leichhardtii at Different Growth Stages Using 1H NMR-Based Metabolite Profiling and Tropane Alkaloids-Targeted HPLC-MS Analysis. Phytochemistry 2016, 131, 44–56. [Google Scholar] [CrossRef]
- Pan, Q.; Dai, Y.; Nuringtyas, T.R.; Mustafa, N.R.; Schulte, A.E.; Verpoorte, R.; Choi, Y.H. Investigation of the Chemomarkers Correlated with Flower Colour in Different Organs of Catharanthus roseus Using NMR-Based Metabolomics. Phytochem. Anal. 2014, 25, 66–74. [Google Scholar] [CrossRef]
- Palama, T.L.; Canard, I.; Rautureau, G.J.P.; Mirande, C.; Chatellier, S.; Elena-Herrmann, B. Identification of Bacterial Species by Untargeted NMR Spectroscopy of the Exo-Metabolome. Analyst 2016, 141, 4558–4561. [Google Scholar] [CrossRef]
- Pariyani, R.; Ismail, I.S.; Ahmad Azam, A.; Abas, F.; Shaari, K. Identification of the Compositional Changes in Orthosiphon stamineus Leaves Triggered by Different Drying Techniques Using 1H NMR Metabolomics. J. Sci. Food Agric. 2017, 97, 4169–4179. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, G.; Zhang, J.; Zhang, Y.; Li, J.; Xiong, C.; Zhang, Q.; Li, X.; Lai, X. Metabolic Discrimination of Sea Buckthorn from Different Hippophaë Species by 1H NMR Based Metabolomics. Sci. Rep. 2017, 7, 1585. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Roy, R. Quantitative 1H NMR Spectroscopy. TrAC Trends Anal. Chem. 2012, 35, 5–26. [Google Scholar] [CrossRef]
- Jacob, D.; Deborde, C.; Lefebvre, M.; Maucourt, M.; Moing, A. NMRProcFlow: A Graphical and Interactive Tool Dedicated to 1D Spectra Processing for NMR-Based Metabolomics. Metab. Off. J. Metab. Soc. 2017, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- De Meyer, T.; Sinnaeve, D.; Van Gasse, B.; Tsiporkova, E.; Rietzschel, E.R.; De Buyzere, M.L.; Gillebert, T.C.; Bekaert, S.; Martins, J.C.; Van Criekinge, W. NMR-Based Characterization of Metabolic Alterations in Hypertension Using an Adaptive, Intelligent Binning Algorithm. Anal. Chem. 2008, 80, 3783–3790. [Google Scholar] [CrossRef] [PubMed]

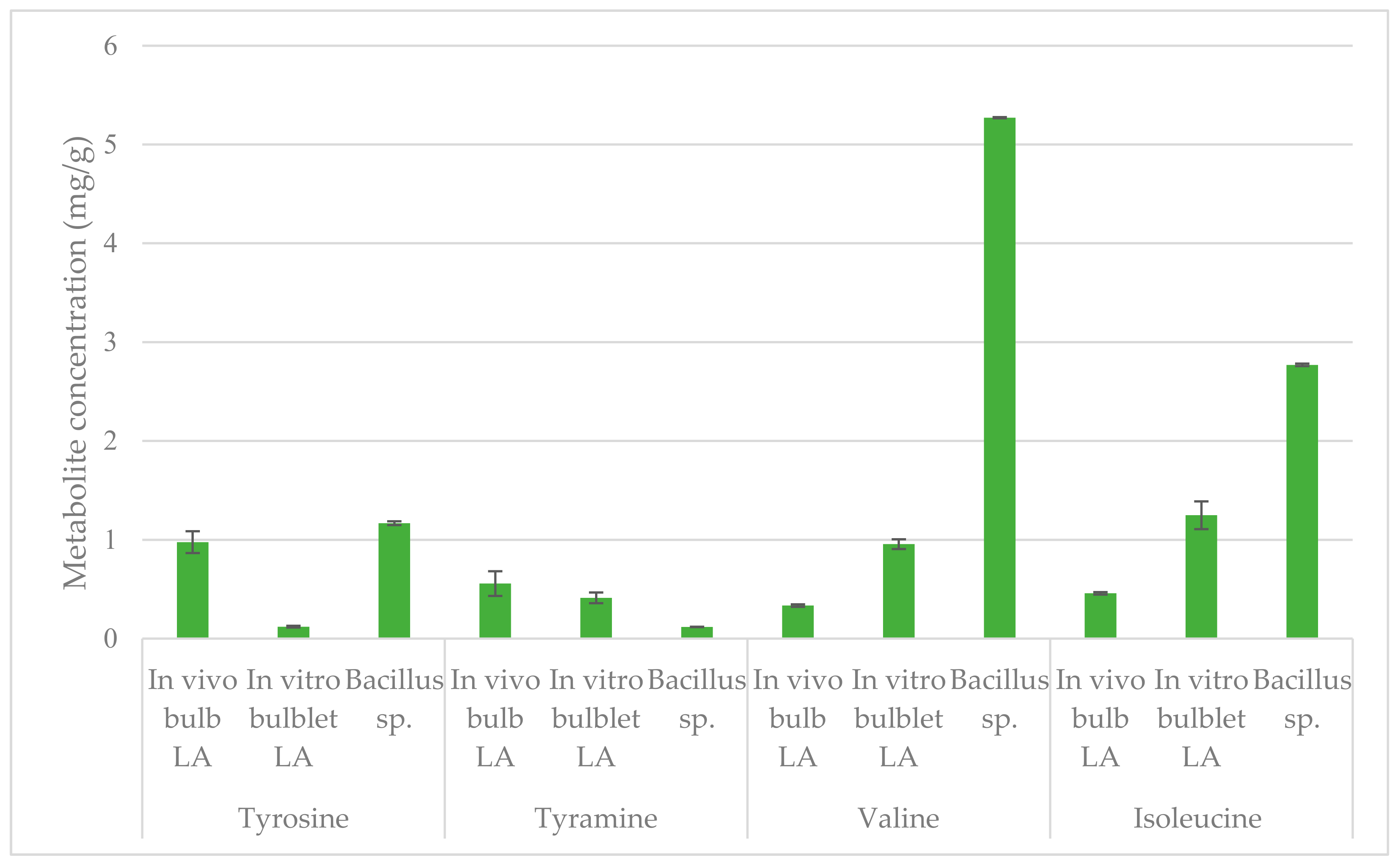
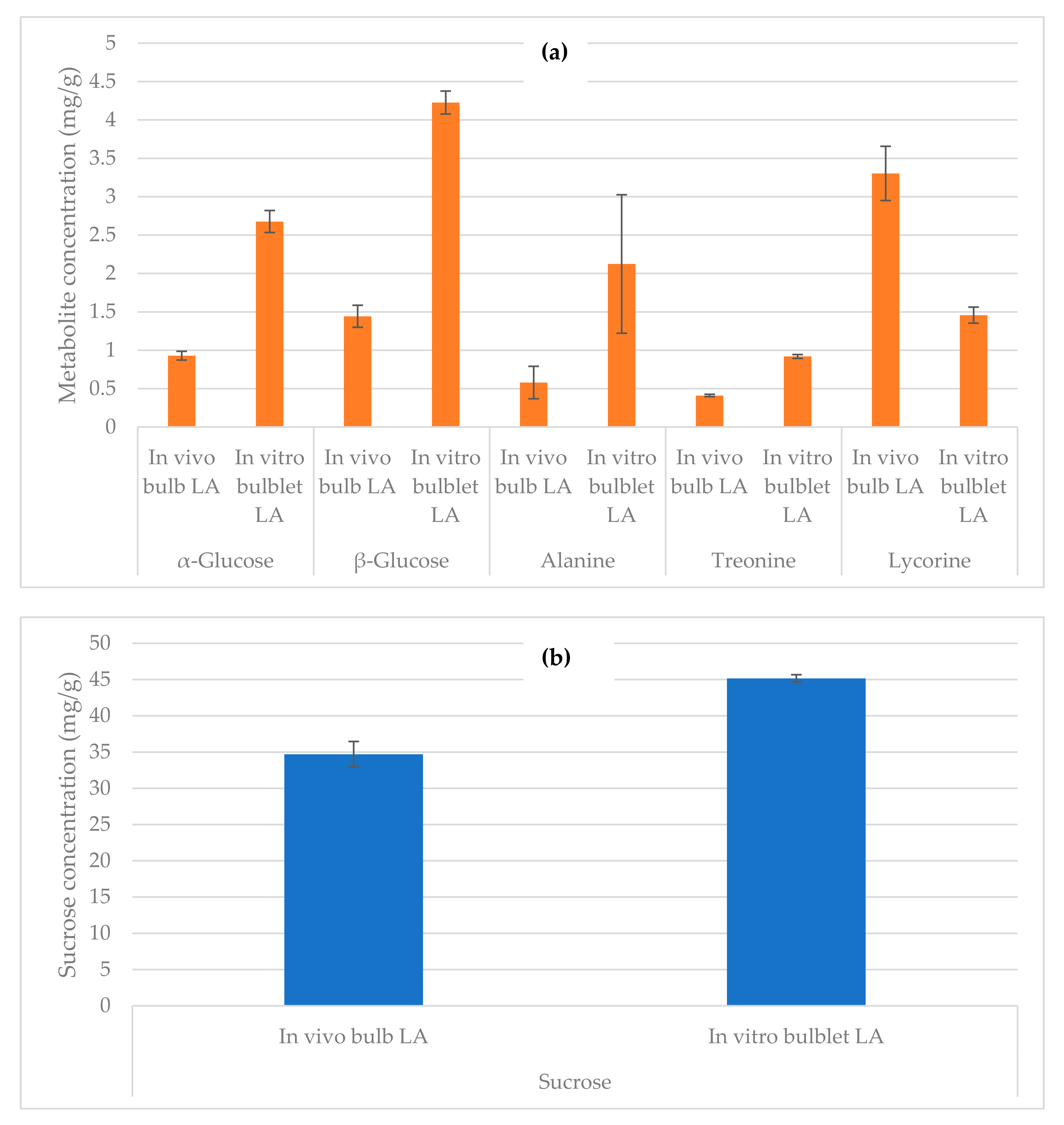

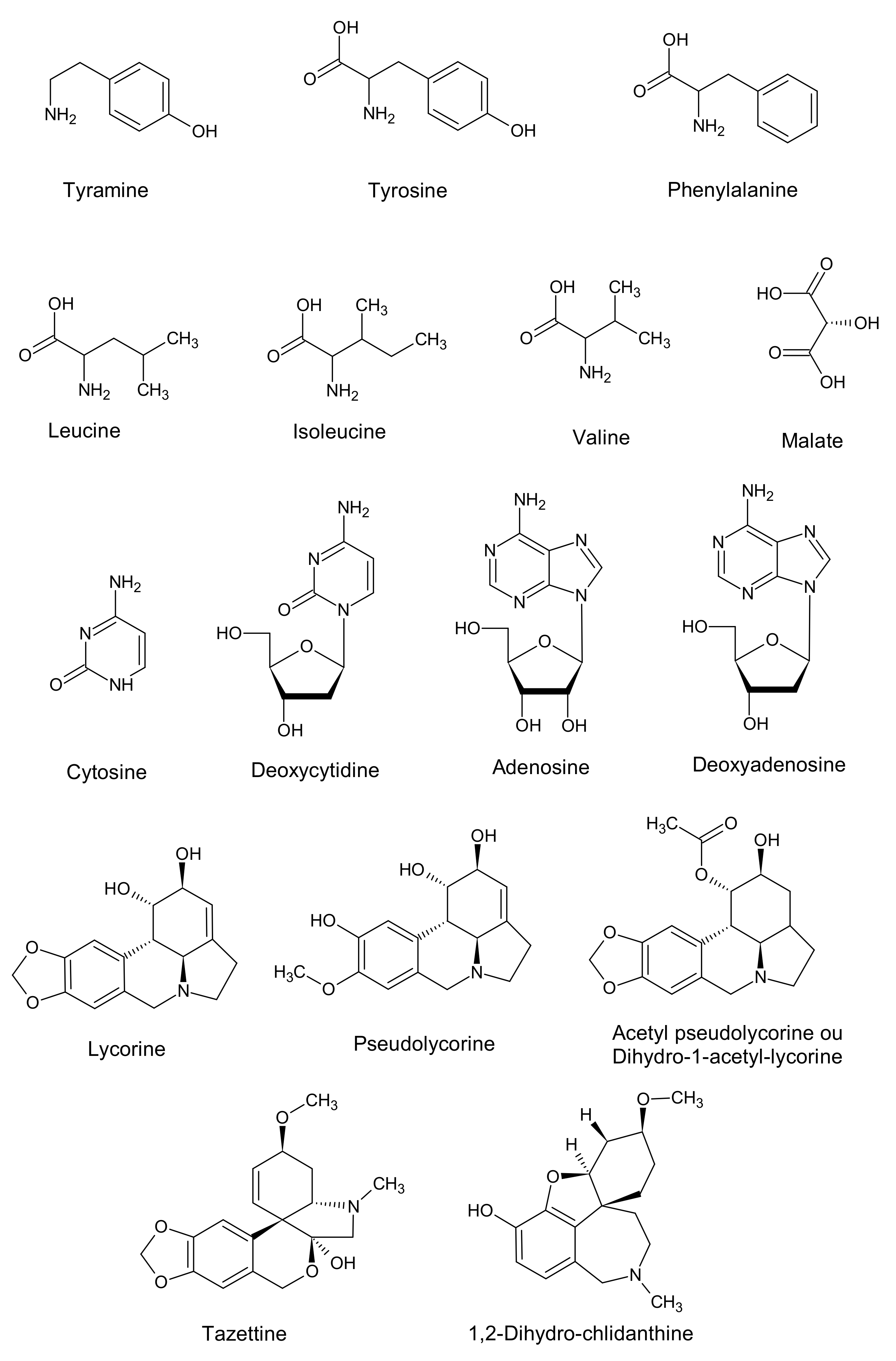
| Molecules | Molecular Weight | Molecular Formula | Chemical Shifts, Multiplicity (ppm), J (Hz) | Extract from In Vivo L. aestivum Bulb | Extract from In Vitro L. aestivum Bulblet | Extract from Bacillus sp. |
|---|---|---|---|---|---|---|
| Adenine | 135,113 | C5H5N5 | 8.2 (s), 8.21 (s) | − | − | Y |
| Cytosine | 111.1 | C4H5N3O | 7.52 (s), 7.51 (s) | − | − | Y |
| Phenylalanine | 165.19 | C9H11NO2 | 7.42–7.33 (m), 3.09 (dd, J = 14.8, 8.3) | − | − | Y |
| Tyrosine | 181.19 | C9H11NO3 | 7.16 (d, J = 8.4), 6.83 (d, J = 8.5) | Y | Y | Y |
| Tyramine | 137.18 | C8H11NO | 7.11 (d, J = 8.6) 6.71,(d, J = 8.5) | Y | Y | Y |
| Gallic acid | 170.12 | C7H6O5 | 7.02 (s) | Y | Y | − |
| Lycorine | 287.32 | C16H17NO4 | 7.00 (s), 6.85 (s) | Y | Y | − |
| Galanthamine | 287.35 | C17H21NO3 | 6.95 (d, J = 8.5) 6.89 (d, J = 8.5) | Y | − | − |
| Sucrose | 342.30 | C12H22O11 | 5.39 (d, J = 3.8) | Y | Y | − |
| α-Glucose | 180.16 | C6H12O6 | 5.17 (d, J = 3.8) | Y | Y | − |
| β-Glucose | 180.16 | C6H12O6 | 4.57 (d, J = 7.9) | Y | Y | − |
| Malate | 134.09 | C4H6O5 | 4.34 (dd, J = 6.6 Hz, 4.7 Hz) | − | − | Y |
| Oxaloacetic acid | 132.07 | C4H4O5 | 3.64 (s) | Y | Y | − |
| Asparagine | 132.11 | C4H8N2O3 | 3.94 (dd, J = 8.0, 4.0) | Y | Y | − |
| Aspartate | 133.10 | C4H7NO4 | 2.78 (dd, J = 1.7, 5.3) | Y | Y | − |
| Citrate | 192.12 | C6H8O7 | 2.74 (d, J = 17.6 Hz), | − | Y | − |
| Alanine | 89.09 | C3H7NO2 | 1.46 (d, J = 7.2) | Y | Y | − |
| Threonine | 119.12 | C4H9NO3 | 1.33 (d, J = 6.6) | Y | Y | − |
| Valine | 117.15 | C5H11NO2 | 1.00 (d, J = 6.8) 1.05 (d, J = 6.8) | Y | Y | Y |
| Isoleucine | 131.17 | C6H13NO2 | 0.96 (t, J = 7.4) 1.03 (d, J = 6.8) | Y | Y | Y |
| Leucine | 131.17 | C6H13NO2 | 1.05 (d, J = 7) 1.01 (t, J = 7) | − | − | Y |
| Fatty acids (as oleic acid) | 282.5 | C18H34O2 | 0.89 (d, J = 7.1) | Y | Y | Y |
| Compounds Name | Retention Time (min) | Molecular Formula | Molecular Mass [M+H]+ | Identification Methods |
|---|---|---|---|---|
| Methyl-phenylalanine | 1.9 | C10H13NO2 | 180.1025 | b, d, e, f |
| Tazettine | 3.2 | C18H21NO5 | 332.1486 | c, e, f |
| Deoxyadenosine | 4.6 | C10H13N5O3 | 252.1097 | c, d, e |
| Adenosine | 4.9 | C10H13N5O4 | 268.10481 | b, c, d, e |
| Lycorine | 6.1 | C16H17NO4 | 288.1231 | a, c, e, f |
| Deoxycytidine | 6.4 | C9H13N3O4 | 228.0997 | c, d, e |
| Pseudolycorine | 8 | C16H19NO4 | 290.14 | e, f |
| Acetyl pseudolycorine | 11.5 | C18H21NO5 | 332.1498 | c, e |
| Compound Name | Retention Time (min) | Molecular Mass | Molecular Formula | Retention Index |
|---|---|---|---|---|
| Tazettine | 12.84 | 331 | C18H21NO5 | 2415 |
| 1,2-Dihydro-chlidanthine | 15.15 | 289 | C17H23NO3 | 2211 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spina, R.; Saliba, S.; Dupire, F.; Ptak, A.; Hehn, A.; Piutti, S.; Poinsignon, S.; Leclerc, S.; Bouguet-Bonnet, S.; Laurain-Mattar, D. Molecular Identification of Endophytic Bacteria in Leucojum aestivum In Vitro Culture, NMR-Based Metabolomics Study and LC-MS Analysis Leading to Potential Amaryllidaceae Alkaloid Production. Int. J. Mol. Sci. 2021, 22, 1773. https://doi.org/10.3390/ijms22041773
Spina R, Saliba S, Dupire F, Ptak A, Hehn A, Piutti S, Poinsignon S, Leclerc S, Bouguet-Bonnet S, Laurain-Mattar D. Molecular Identification of Endophytic Bacteria in Leucojum aestivum In Vitro Culture, NMR-Based Metabolomics Study and LC-MS Analysis Leading to Potential Amaryllidaceae Alkaloid Production. International Journal of Molecular Sciences. 2021; 22(4):1773. https://doi.org/10.3390/ijms22041773
Chicago/Turabian StyleSpina, Rosella, Sahar Saliba, François Dupire, Agata Ptak, Alain Hehn, Séverine Piutti, Sophie Poinsignon, Sebastien Leclerc, Sabine Bouguet-Bonnet, and Dominique Laurain-Mattar. 2021. "Molecular Identification of Endophytic Bacteria in Leucojum aestivum In Vitro Culture, NMR-Based Metabolomics Study and LC-MS Analysis Leading to Potential Amaryllidaceae Alkaloid Production" International Journal of Molecular Sciences 22, no. 4: 1773. https://doi.org/10.3390/ijms22041773
APA StyleSpina, R., Saliba, S., Dupire, F., Ptak, A., Hehn, A., Piutti, S., Poinsignon, S., Leclerc, S., Bouguet-Bonnet, S., & Laurain-Mattar, D. (2021). Molecular Identification of Endophytic Bacteria in Leucojum aestivum In Vitro Culture, NMR-Based Metabolomics Study and LC-MS Analysis Leading to Potential Amaryllidaceae Alkaloid Production. International Journal of Molecular Sciences, 22(4), 1773. https://doi.org/10.3390/ijms22041773









