The Immunology of Syncytialized Trophoblast
Abstract
1. The Hemochorial Placenta Poses Unique Immune Challenges
2. Immune Interactions at STB Interfaces
2.1. A Possible Role for Glycosylation
2.2. Toll-Like Receptors (TLRs) and STB Immune Responses
2.3. STB and Immune Checkpoint Molecules
2.4. A Role for STB-Derived Extracellular Vesicles
2.5. Immune Modulation by Human Endogenous Retroviral Proteins
2.6. Gestational Age-Specific Susceptibility of STB to Infection
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Soares, M.J.; Varberg, K.M.; Iqbal, K. Hemochorial placentation: Development, function, and adaptations. Biol. Reprod. 2018, 99, 196–211. [Google Scholar] [CrossRef]
- Medawar, P.B. Some immunological and endocrinological problems raised by the evolution of viviparity in vertebrates. Symp. Soc. Exp. Biol. 1953, 7, 320–338. [Google Scholar]
- Hyde, K.J.; Schust, D.J. Immunologic challenges of human reproduction: An evolving story. Fertil. Steril. 2016, 106, 499–510. [Google Scholar] [CrossRef]
- Roberts, V.H.; Morgan, T.; Bednarek, P.; Morita, M.; Burton, G.; Lo, J.; Frias, A. Early first trimester uteroplacental flow and the progressive disintegration of spiral artery plugs: New insights from contrast-enhanced ultrasound and tissue histopathology. Hum. Reprod. 2017, 32, 2382–2393. [Google Scholar] [CrossRef] [PubMed]
- Tersigni, C.; Meli, F.; Neri, C.; Iacoangeli, A.; Franco, R.; Lanzone, A.; Scambia, G.; Di Simone, N. Role of Human Leukocyte Antigens at the Feto-Maternal Interface in Normal and Pathological Pregnancy: An Update. Int. J. Mol. Sci. 2020, 21, 4756. [Google Scholar] [CrossRef] [PubMed]
- PrabhuDas, M.; Bonney, E.; Caron, K.; Dey, S.; Erlebacher, A.; Fazleabas, A.; Fisher, S.; Golos, T.; Matzuk, M.; McCune, J.M.; et al. Immune mechanisms at the maternal-fetal interface: Perspectives and challenges. Nat. Immunol. 2015, 16, 328–334. [Google Scholar] [CrossRef]
- Ander, S.E.; Diamond, M.S.; Coyne, C.B. Immune responses at the maternal-fetal interface. Sci. Immunol. 2019, 4, eaat6114. [Google Scholar] [CrossRef]
- Genbačev, O.; Vićovac, L.; Larocque, N. The role of chorionic cytotrophoblasts in the smooth chorion fusion with parietal decidua. Placenta 2015, 36, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Slukvin, I.; Lunn, D.; Watkins, D.; Golos, T. Placental expression of the nonclassical MHC class I molecule Mamu-AG at implantation in the rhesus monkey. Proc. Natl. Acad. Sci. USA 2000, 97, 9104–9109. [Google Scholar] [CrossRef] [PubMed]
- Hsi, B.; Yeh, C.; Faulk, W. Class I antigens of the major histocompatibility complex on cytotrophoblast of human chorion laeve. Immunology 1984, 52, 621. [Google Scholar]
- Shorter, S.; Starkey, P.; Ferry, B.; Clover, L.; Sargent, I.; Redman, C. Antigenic heterogeneity of human cytotrophoblast and evidence for the transient expression of MHC class I antigens distinct from HLA-G. Placenta 1993, 14, 571–582. [Google Scholar] [CrossRef]
- Solders, M.; Gorchs, L.; Gidlöf, S.; Tiblad, E.; Lundell, A.-C.; Kaipe, H. Maternal adaptive immune cells in decidua parietalis display a more activated and coinhibitory phenotype compared to decidua basalis. Stem Cells Int. 2017, 2017, 8010961. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, D.W.; Zickwolf, G.K.; Weil, G.J.; Sylvester, S.; DeMaria, M.A. Male fetal progenitor cells persist in maternal blood for as long as 27 years postpartum. Proc. Natl. Acad. Sci. USA 1996, 93, 705–708. [Google Scholar] [CrossRef]
- Kinder, J.M.; Stelzer, I.A.; Arck, P.C.; Way, S.S. Immunological implications of pregnancy-induced microchimerism. Nat. Rev. Immunol. 2017, 17, 483. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J.L. Autoimmune disease and the long-term persistence of fetal and maternal microchimerism. Lupus 1999, 8, 493–496. [Google Scholar] [CrossRef]
- Nassar, D.; Khosrotehrani, K.; Aractingi, S. Fetal microchimerism in skin wound healing. Chimerism 2012, 3, 45–47. [Google Scholar] [CrossRef] [PubMed]
- Fjeldstad, H.E.; Johnsen, G.M.; Staff, A.C. Fetal microchimerism and implications for maternal health. Obstet. Med. 2020, 13, 112–119. [Google Scholar] [CrossRef]
- Sachini, N.; Papamatheakis, J. NF-Y and the immune response: Dissecting the complex regulation of MHC genes. Biochim. Biophys. Acta Gene Regul. Mech. 2017, 1860, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Luo, Y. Histone acetylation and the regulation of major histocompatibility class II gene expression. Adv. Protein Chem. Struct. Biol. 2017, 106, 71–111. [Google Scholar]
- Geirsson, A.; Paliwal, I.; Lynch, R.J.; Bothwell, A.L.; Hammond, G.L. Class II transactivator promoter activity is suppressed through regulation by a trophoblast noncoding RNA1. Transplantation 2003, 76, 387–394. [Google Scholar] [CrossRef]
- Tilburgs, T.; Meissner, T.B.; Ferreira, L.M.; Mulder, A.; Musunuru, K.; Ye, J.; Strominger, J.L. NLRP2 is a suppressor of NF-ƙB signaling and HLA-C expression in human trophoblasts. Biol. Reprod. 2017, 96, 831–842. [Google Scholar] [CrossRef]
- Lefebvre, S.; Moreau, P.; Dausset, J.; Carosella, E.; Paul, P. Downregulation of HLA class I gene transcription in choriocarcinoma cells is controlled by the proximal promoter element and can be reversed by CIITA. Placenta 1999, 20, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Ljunggren, H.-G.; Kärre, K. In search of the ‘missing self’: MHC molecules and NK cell recognition. Immunol. Today 1990, 11, 237–244. [Google Scholar] [CrossRef]
- Belisle, J.A.; Horibata, S.; Jennifer, G.A.; Petrie, S.; Kapur, A.; André, S.; Gabius, H.-J.; Rancourt, C.; Connor, J.; Paulson, J.C.; et al. Identification of Siglec-9 as the receptor for MUC16 on human NK cells, B cells, and monocytes. Mol. Cancer 2010, 9, 1–14. [Google Scholar] [CrossRef]
- Chen, Q.; Pang, P.-C.; Cohen, M.E.; Longtine, M.S.; Schust, D.J.; Haslam, S.M.; Blois, S.M.; Dell, A.; Clark, G.F. Evidence for differential glycosylation of trophoblast cell types. Mol. Cell. Proteom. 2016, 15, 1857–1866. [Google Scholar] [CrossRef] [PubMed]
- Nishima, W.; Miyashita, N.; Yamaguchi, Y.; Sugita, Y.; Re, S. Effect of bisecting GlcNAc and core fucosylation on conformational properties of biantennary complex-type N-glycans in solution. J. Phys. Chem. B 2012, 116, 8504–8512. [Google Scholar] [CrossRef] [PubMed]
- El Ouagari, K.; Teissié, J.; Benoist, H. Glycophorin A Protects K562 Cells from Natural Killer Cell Attack ROLE OF OLIGOSACCHARIDES. J. Biol. Chem. 1995, 270, 26970–26975. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Ihara, Y.; Ohnishi, A.; Ijuhin, N.; Nishiura, T.; Kanakura, Y.; Matsuzawa, Y.; Taniguchi, N. Bisecting N-acetylglucosamine on K562 cells suppresses natural killer cytotoxicity and promotes spleen colonization. Cancer Res. 1996, 56, 412–418. [Google Scholar] [PubMed]
- Borowski, S.; Tirado-Gonzalez, I.; Freitag, N.; Garcia, M.G.; Barrientos, G.; Blois, S.M. Altered glycosylation contributes to placental dysfunction upon early disruption of the NK cell-DC dynamics. Front. Immunol. 2020, 11, 1316. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, M.; Gao, R.; Liu, X.; Chen, X.; Geng, Y.; Ding, Y.; Wang, Y.; He, J. Altered β1, 6-GlcNAc and bisecting GlcNAc-branched N-glycan on integrin β1 are associated with early spontaneous miscarriage in humans. Hum. Reprod. 2015, 30, 2064–2075. [Google Scholar] [CrossRef]
- Marini, M.; Bonaccini, L.; Thyrion, G.D.Z.; Vichi, D.; Parretti, E.; Sgambati, E. Distribution of sugar residues in human placentas from pregnancies complicated by hypertensive disorders. Acta Histochem. 2011, 113, 815–825. [Google Scholar] [CrossRef]
- Bulmer, J.N.; Lash, G.E. Uterine natural killer cells: Time for a re-appraisal? F1000research 2019, 8, F1000. [Google Scholar] [CrossRef] [PubMed]
- Al-Lamki, R.; Skepper, J.; Burton, G. Are human placental bed giant cells merely aggregates of small mononuclear trophoblast cells? An ultrastructural and immunocytochemical study. Hum. Reprod. 1999, 14, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Ibeto, L.; Antonopoulos, A.; Grassi, P.; Pang, P.-C.; Panico, M.; Bobdiwala, S.; Al-Memar, M.; Davis, P.; Davis, M.; Norman Taylor, J.; et al. Insights into the hyperglycosylation of human chorionic gonadotropin revealed by glycomics analysis. PLoS ONE 2020, 15, e0228507. [Google Scholar] [CrossRef] [PubMed]
- Sulke, A.; Jones, D.; Wood, P. Hormonal modulation of human natural killer cell activity in vitro. J. Reprod. Immunol. 1985, 7, 105–110. [Google Scholar] [CrossRef]
- Koga, K.; Cardenas, I.; Aldo, P.; Abrahams, V.M.; Peng, B.; Fill, S.; Romero, R.; Mor, G. Activation of TLR3 in the trophoblast is associated with preterm delivery. Am. J. Reprod. Immunol. 2009, 61, 196–212. [Google Scholar] [CrossRef]
- Nakada, E.; Walley, K.; Nakada, T.; Hu, Y.; Von Dadelszen, P.; Boyd, J. Toll-like receptor-3 stimulation upregulates sFLT-1 production by trophoblast cells. Placenta 2009, 30, 774–779. [Google Scholar] [CrossRef]
- Gonzalez, J.M.; Xu, H.; Ofori, E.; Elovitz, M.A. Toll-like receptors in the uterus, cervix, and placenta: Is pregnancy an immunosuppressed state? Am. J. Obstet. Gynecol. 2007, 197, e1–e6. [Google Scholar] [CrossRef]
- Vidya, M.K.; Kumar, V.G.; Sejian, V.; Bagath, M.; Krishnan, G.; Bhatta, R. Toll-like receptors: Significance, ligands, signaling pathways, and functions in mammals. Int. Rev. Immunol. 2018, 37, 20–36. [Google Scholar] [CrossRef]
- Olmos-Ortiz, A.; Flores-Espinosa, P.; Mancilla-Herrera, I.; Vega-Sánchez, R.; Díaz, L.; Zaga-Clavellina, V. Innate Immune Cells and Toll-like Receptor–Dependent Responses at the Maternal–Fetal Interface. Int. J. Mol. Sci. 2019, 20, 3654. [Google Scholar] [CrossRef]
- Pudney, J.; He, X.; Masheeb, Z.; Kindelberger, D.W.; Kuohung, W.; Ingalls, R.R. Differential expression of toll-like receptors in the human placenta across early gestation. Placenta 2016, 46, 1–10. [Google Scholar] [CrossRef]
- Abrahams, V.M.; Bole-Aldo, P.; Kim, Y.M.; Straszewski-Chavez, S.L.; Chaiworapongsa, T.; Romero, R.; Mor, G. Divergent trophoblast responses to bacterial products mediated by TLRs. J. Immunol. 2004, 173, 4286–4296. [Google Scholar] [CrossRef] [PubMed]
- Holmlund, U.; Cebers, G.; Dahlfors, A.R.; Sandstedt, B.; Bremme, K.; EkstrÖm, E.S.; Scheynius, A. Expression and regulation of the pattern recognition receptors Toll-like receptor-2 and Toll-like receptor-4 in the human placenta. Immunology 2002, 107, 145–151. [Google Scholar] [CrossRef]
- Kumazaki, K.; Nakayama, M.; Yanagihara, I.; Suehara, N.; Wada, Y. Immunohistochemical distribution of Toll-like receptor 4 in term and preterm human placentas from normal and complicated pregnancy including chorioamnionitis. Hum. Pathol. 2004, 35, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Krikun, G.; Abrahams, V.M.; Mor, G.; Guller, S. Cell type-specific expression and function of toll-like receptors 2 and 4 in human placenta: Implications in fetal infection. Placenta 2007, 28, 1024–1031. [Google Scholar] [CrossRef] [PubMed]
- Rindsjö, E.; Holmlund, U.; Sverremark-Ekström, E.; Papadogiannakis, N.; Scheynius, A. Toll-like receptor-2 expression in normal and pathologic human placenta. Hum. Pathol. 2007, 38, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Patni, S.; Wynen, L.P.; Seager, A.L.; Morgan, G.; White, J.O.; Thornton, C.A. Expression and activity of Toll-like receptors 1–9 in the human term placenta and changes associated with labor at term. Biol. Reprod. 2009, 80, 243–248. [Google Scholar] [CrossRef]
- Mitsunari, M.; Yoshida, S.; Shoji, T.; Tsukihara, S.; Iwabe, T.; Harada, T.; Terakawa, N. Macrophage-activating lipopeptide-2 induces cyclooxygenase-2 and prostaglandin E2 via toll-like receptor 2 in human placental trophoblast cells. J. Reprod. Immunol. 2006, 72, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Bryant, A.H.; Menzies, G.E.; Scott, L.M.; Spencer-Harty, S.; Davies, L.B.; Smith, R.A.; Jones, R.H.; Thornton, C.A. Human gestation-associated tissues express functional cytosolic nucleic acid sensing pattern recognition receptors. Clin. Exp. Immunol. 2017, 189, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Tangerås, L.H.; Silva, G.B.; Stødle, G.S.; Gierman, L.M.; Skei, B.; Collett, K.; Beversmark, A.-L.; Skråstad, R.B.; Thomsen, L.C.V.; Bjørge, L.; et al. Placental inflammation by HMGB1 activation of TLR4 at the syncytium. Placenta 2018, 72, 53–61. [Google Scholar] [CrossRef]
- Afkham, A.; Eghbal-Fard, S.; Heydarlou, H.; Azizi, R.; Aghebati-Maleki, L.; Yousefi, M. Toll-like receptors signaling network in pre-eclampsia: An updated review. J. Cell. Physiol. 2019, 234, 2229–2240. [Google Scholar] [CrossRef]
- Kolben, T.M.; Rogatsch, E.; Hester, A.; Kuhn, C.; Schmoeckel, E.; Czogalla, B.; Mahner, S.; Jeschke, U.; Kolben, T. Involvement of ILR4α and TLR4 in miscarriages. J. Reprod. Immunol. 2019, 131, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Y.; Wang, S.C.; Li, D.J.; Du, M.R. Co-Signaling Molecules in Maternal-Fetal Immunity. Trends Mol. Med. 2017, 23, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Veras, E.; Kurman, R.J.; Wang, T.L.; Shih, I.M. PD-L1 Expression in Human Placentas and Gestational Trophoblastic Diseases. Int. J. Gynecol. Pathol. 2017, 36, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, M.; Mezawa, H.; Kawai, T.; Urashima, M. Elevated Soluble PD-L1 in Pregnant Women’s Serum Suppresses the Immune Reaction. Front. Immunol. 2019, 10, 86. [Google Scholar] [CrossRef]
- Holets, L.M.; Hunt, J.S.; Petroff, M.G. Trophoblast CD274 (B7-H1) is differentially expressed across gestation: Influence of oxygen concentration. Biol. Reprod. 2006, 74, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Petroff, M.G.; Kharatyan, E.; Torry, D.S.; Holets, L. The immunomodulatory proteins B7-DC, B7-H2, and B7-H3 are differentially expressed across gestation in the human placenta. Am. J. Pathol. 2005, 167, 465–473. [Google Scholar] [CrossRef]
- Vento-Tormo, R.; Efremova, M.; Botting, R.A.; Turco, M.Y.; Vento-Tormo, M.; Meyer, K.B.; Park, J.E.; Stephenson, E.; Polanski, K.; Goncalves, A.; et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature 2018, 563, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Yabe, S.; Alexenko, A.P.; Amita, M.; Yang, Y.; Schust, D.J.; Sadovsky, Y.; Ezashi, T.; Roberts, R.M. Comparison of syncytiotrophoblast generated from human embryonic stem cells and from term placentas. Proc. Natl. Acad. Sci. USA 2016, 113, E2598–E2607. [Google Scholar] [CrossRef]
- Yang, Y.; Adachi, K.; Sheridan, M.A.; Alexenko, A.P.; Schust, D.J.; Schulz, L.C.; Ezashi, T.; Roberts, R.M. Heightened potency of human pluripotent stem cell lines created by transient BMP4 exposure. Proc. Natl. Acad. Sci. USA 2015, 112, E2337–E2346. [Google Scholar] [CrossRef]
- Amita, M.; Adachi, K.; Alexenko, A.P.; Sinha, S.; Schust, D.J.; Schulz, L.C.; Roberts, R.M.; Ezashi, T. Complete and unidirectional conversion of human embryonic stem cells to trophoblast by BMP4. Proc. Natl. Acad. Sci. USA 2013, 110, E1212–E1221. [Google Scholar] [CrossRef]
- Roberts, R.M.; Yabe, S.; Yang, Y.; Ezashi, T. A human stem cell model for creating placental syncytiotrophoblast, the major cellular barrier that limits fetal exposure to xenobiotics. In Stem Cells in Toxicology and Medicine, 1st ed.; Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 179–195. [Google Scholar]
- Wang, L.Q.; Yan, C.F.; Zhao, Y.; Chu, J.; Yu, X.W. Reduced CD200 and CD200R1 expression in human chorionic villi contributes to early spontaneous abortion. Acta Obs. Gynecol. Scand. 2014, 93, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Gu, Y.; Sun, J.; Zhu, H.; Lewis, D.F.; Wang, Y. Reduced CD200 expression is associated with altered Th1/Th2 cytokine production in placental trophoblasts from preeclampsia. Am. J. Reprod. Immunol. 2018, 79. [Google Scholar] [CrossRef] [PubMed]
- Wykes, M.N.; Lewin, S.R. Immune checkpoint blockade in infectious diseases. Nat. Rev. Immunol. 2018, 18, 91–104. [Google Scholar] [CrossRef]
- Wei, S.C.; Duffy, C.R.; Allison, J.P. Fundamental Mechanisms of Immune Checkpoint Blockade Therapy. Cancer Discov. 2018, 8, 1069–1086. [Google Scholar] [CrossRef] [PubMed]
- Mincheva-Nilsson, L.; Baranov, V. Placenta-derived exosomes and syncytiotrophoblast microparticles and their role in human reproduction: Immune modulation for pregnancy success. Am. J. Reprod. Immunol. 2014, 72, 440–457. [Google Scholar] [CrossRef] [PubMed]
- Huang-Doran, I.; Zhang, C.-Y.; Vidal-Puig, A. Extracellular vesicles: Novel mediators of cell communication in metabolic disease. Trends Endocrinol. Metab. 2017, 28, 3–18. [Google Scholar] [CrossRef]
- Zondler, L.; Müller, K.; Khalaji, S.; Bliederhäuser, C.; Ruf, W.P.; Grozdanov, V.; Thiemann, M.; Fundel-Clemes, K.; Freischmidt, A.; Holzmann, K. Peripheral monocytes are functionally altered and invade the CNS in ALS patients. Acta Neuropathol. 2016, 132, 391–411. [Google Scholar] [CrossRef]
- Heijnen, H.F.; Schiel, A.E.; Fijnheer, R.; Geuze, H.J.; Sixma, J.J. Activated Platelets Release Two Types of Membrane Vesicles: Microvesicles by Surface Shedding and Exosomes Derived From Exocytosis of Multivesicular Bodies and alpha-Granules. Blood J. Am. Soc. Hematol. 1999, 94, 3791–3799. [Google Scholar]
- Szajnik, M.; Czystowska, M.; Szczepanski, M.J.; Mandapathil, M.; Whiteside, T.L. Tumor-derived microvesicles induce, expand and up-regulate biological activities of human regulatory T cells (Treg). PLoS ONE 2010, 5, e11469. [Google Scholar] [CrossRef]
- Morel, O.; Jesel, L.; Freyssinet, J.-M.; Toti, F. Cellular mechanisms underlying the formation of circulating microparticles. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 15–26. [Google Scholar] [CrossRef]
- Redman, C.; Sargent, I. Circulating microparticles in normal pregnancy and pre-eclampsia. Placenta 2008, 29, 73–77. [Google Scholar] [CrossRef]
- Bernischke, K.; Kaufmann, P.; Baergen, R. Pathology of the Human Placenta, 4th ed.; Springer: New York, NY, USA, 2000; pp. 42–49. [Google Scholar]
- Huppertz, B.; Frank, H.-G.; Reister, F.; Kaufmann, P. Villous cytotrophoblast regulation of the syncytial apoptotic cascade in the human placenta. Histochem. Cell Biol. 1998, 110, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.D.; Akyol, S.; Gercel-Taylor, C. Pregnancy-associated exosomes and their modulation of T cell signaling. J. Immunol. 2006, 176, 1534–1542. [Google Scholar] [CrossRef] [PubMed]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Chen, X.; Liang, H.; Zhang, J.; Zen, K.; Zhang, C.-Y. Horizontal transfer of microRNAs: Molecular mechanisms and clinical applications. Protein Cell 2012, 3, 28–37. [Google Scholar] [CrossRef]
- Bhatti, G.; Romero, R.; Rice, G.E.; Fitzgerald, W.; Pacora, P.; Gomez-Lopez, N.; Kavdia, M.; Tarca, A.L.; Margolis, L. Compartmentalized profiling of amniotic fluid cytokines in women with preterm labor. PLoS ONE 2020, 15, e0227881. [Google Scholar] [CrossRef] [PubMed]
- Kshirsagar, S.; Alam, S.; Jasti, S.; Hodes, H.; Nauser, T.; Gilliam, M.; Billstrand, C.; Hunt, J.; Petroff, M. Immunomodulatory molecules are released from the first trimester and term placenta via exosomes. Placenta 2012, 33, 982–990. [Google Scholar] [CrossRef] [PubMed]
- McCoy-Simandle, K.; Hanna, S.J.; Cox, D. Exosomes and nanotubes: Control of immune cell communication. Int. J. Biochem. Cell Biol. 2016, 71, 44–54. [Google Scholar] [CrossRef]
- Aliotta, J.M.; Pereira, M.; Sears, E.H.; Dooner, M.S.; Wen, S.; Goldberg, L.R.; Quesenberry, P.J. Lung-derived exosome uptake into and epigenetic modulation of marrow progenitor/stem and differentiated cells. J. Extracell. Vesicles 2015, 4, 26166. [Google Scholar] [CrossRef]
- Redman, C.; Sargent, I. Microparticles and immunomodulation in pregnancy and pre-eclampsia. J. Reprod. Immunol. 2007, 76, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Song, G.; Lim, W. Effects of extracellular vesicles on placentation and pregnancy disorders. Reproduction 2019, 158, R189–R196. [Google Scholar] [CrossRef] [PubMed]
- Kurian, N.K.; Modi, D. Extracellular vesicle mediated embryo-endometrial cross talk during implantation and in pregnancy. J. Assist. Reprod. Genet. 2019, 36, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Bannert, N.; Kurth, R. Retroelements and the human genome: New perspectives on an old relation. Proc. Natl. Acad. Sci. USA 2004, 101, 14572–14579. [Google Scholar] [CrossRef] [PubMed]
- Reiss, D.; Zhang, Y.; Mager, D.L. Widely variable endogenous retroviral methylation levels in human placenta. Nucleic Acids Res. 2007, 35, 4743–4754. [Google Scholar] [CrossRef]
- Blaise, S.; de Parseval, N.; Bénit, L.; Heidmann, T. Genomewide screening for fusogenic human endogenous retrovirus envelopes identifies syncytin 2, a gene conserved on primate evolution. Proc. Natl. Acad. Sci. USA 2003, 100, 13013–13018. [Google Scholar] [CrossRef] [PubMed]
- Esnault, C.; Priet, S.; Ribet, D.; Vernochet, C.; Bruls, T.; Lavialle, C.; Weissenbach, J.; Heidmann, T. A placenta-specific receptor for the fusogenic, endogenous retrovirus-derived, human syncytin-2. Proc. Natl. Acad. Sci. USA 2008, 105, 17532–17537. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, J.; Schust, D.J.; Kinjo, T.; Aoki, Y.; Jinno, Y.; Kudo, Y. Suppressyn localization and dynamic expression patterns in primary human tissues support a physiologic role in human placentation. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Cianciolo, G.J.; Copeland, T.D.; Oroszlan, S.; Snyderman, R. Inhibition of lymphocyte proliferation by a synthetic peptide homologous to retroviral envelope proteins. Science 1985, 230, 453–455. [Google Scholar] [CrossRef]
- Gröger, V.; Cynis, H. Human endogenous retroviruses and their putative role in the development of autoimmune disorders such as multiple sclerosis. Front. Microbiol. 2018, 9, 265. [Google Scholar]
- Hurst, T.P.; Magiorkinis, G. Activation of the innate immune response by endogenous retroviruses. J. Gen. Virol. 2015, 96, 1207–1218. [Google Scholar] [CrossRef]
- Mangeney, M.; Renard, M.; Schlecht-Louf, G.; Bouallaga, I.; Heidmann, O.; Letzelter, C.; Richaud, A.; Ducos, B.; Heidmann, T. Placental syncytins: Genetic disjunction between the fusogenic and immunosuppressive activity of retroviral envelope proteins. Proc. Natl. Acad. Sci. USA 2007, 104, 20534–20539. [Google Scholar] [CrossRef]
- Holder, B.S.; Tower, C.L.; Forbes, K.; Mulla, M.J.; Aplin, J.D.; Abrahams, V.M. Immune cell activation by trophoblast-derived microvesicles is mediated by syncytin 1. Immunology 2012, 136, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Lokossou, A.G.; Toudic, C.; Nguyen, P.T.; Elisseeff, X.; Vargas, A.; Rassart, É.; Lafond, J.; Leduc, L.; Bourgault, S.; Gilbert, C.; et al. Endogenous retrovirus-encoded Syncytin-2 contributes to exosome-mediated immunosuppression of T cells. Biol. Reprod. 2020, 102, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Dupressoir, A.; Marceau, G.; Vernochet, C.; Bénit, L.; Kanellopoulos, C.; Sapin, V.; Heidmann, T. Syncytin-A and syncytin-B, two fusogenic placenta-specific murine envelope genes of retroviral origin conserved in Muridae. Proc. Natl. Acad. Sci. USA 2005, 102, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, J.; Sugimoto, M.; Bernstein, H.; Jinno, Y.; Schust, D. A novel human endogenous retroviral protein inhibits cell-cell fusion. Sci. Rep. 2013, 3, 1462. [Google Scholar] [CrossRef]
- Nethe, M.; Berkhout, B.; Van der Kuyl, A.C. Retroviral superinfection resistance. Retrovirology 2005, 2, 52. [Google Scholar] [CrossRef] [PubMed]
- Marin, M.; Tailor, C.S.; Nouri, A.; Kabat, D. Sodium-dependent neutral amino acid transporter type 1 is an auxiliary receptor for baboon endogenous retrovirus. J. Virol. 2000, 74, 8085–8093. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.; Genbacev, O.; Maidji, E.; Pereira, L. Human cytomegalovirus infection of placental cytotrophoblasts in vitro and in utero: Implications for transmission and pathogenesis. J. Virol. 2000, 74, 6808–6820. [Google Scholar] [CrossRef]
- Koi, H.; Zhang, J.; Makrigiannakis, A.; Getsios, S.; MacCalman, C.D.; Kopf, G.S.; Strauss, J.F., III; Parry, S. Differential expression of the coxsackievirus and adenovirus receptor regulates adenovirus infection of the placenta. Biol. Reprod. 2001, 64, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- Maidji, E.; McDonagh, S.; Genbacev, O.; Tabata, T.; Pereira, L. Maternal antibodies enhance or prevent cytomegalovirus infection in the placenta by neonatal Fc receptor-mediated transcytosis. Am. J. Pathol. 2006, 168, 1210–1226. [Google Scholar] [CrossRef]
- Robbins, J.R.; Skrzypczynska, K.M.; Zeldovich, V.B.; Kapidzic, M.; Bakardjiev, A.I. Placental syncytiotrophoblast constitutes a major barrier to vertical transmission of Listeria monocytogenes. PLoS Pathog. 2010, 6, e1000732. [Google Scholar] [CrossRef] [PubMed]
- Delorme-Axford, E.; Donker, R.B.; Mouillet, J.-F.; Chu, T.; Bayer, A.; Ouyang, Y.; Wang, T.; Stolz, D.B.; Sarkar, S.N.; Morelli, A.E.; et al. Human placental trophoblasts confer viral resistance to recipient cells. Proc. Natl. Acad. Sci. USA 2013, 110, 12048–12053. [Google Scholar] [CrossRef]
- Bayer, A.; Delorme-Axford, E.; Sleigher, C.; Frey, T.K.; Trobaugh, D.W.; Klimstra, W.B.; Emert-Sedlak, L.A.; Smithgall, T.E.; Kinchington, P.R.; Vadia, S.; et al. Human trophoblasts confer resistance to viruses implicated in perinatal infection. Am. J. Obstet. Gynecol. 2015, 212, e1–e71. [Google Scholar] [CrossRef]
- Faure-Bardon, V.; Magny, J.-F.; Parodi, M.; Couderc, S.; Garcia, P.; Maillotte, A.-M.; Benard, M.; Pinquier, D.; Astruc, D.; Patural, H.; et al. Sequelae of congenital cytomegalovirus following maternal primary infections are limited to those acquired in the first trimester of pregnancy. Clin. Infect. Dis. 2019, 69, 1526–1532. [Google Scholar] [CrossRef]
- Pass, R.F.; Fowler, K.B.; Boppana, S.B.; Britt, W.J.; Stagno, S. Congenital cytomegalovirus infection following first trimester maternal infection: Symptoms at birth and outcome. J. Clin. Virol. 2006, 35, 216–220. [Google Scholar] [CrossRef]
- Mendes, A.K.T.; Ribeiro, M.R.C.; Lamy-Filho, F.; Amaral, G.A.; Borges, M.C.R.; Costa, L.C.; Cavalcante, T.B.; Batista, R.F.L.; Sousa, P.d.S.; Silva, A.A.M.d.; et al. Congenital Zika syndrome: Association between the gestational trimester of maternal infection, severity of brain computed tomography findings and microcephaly at birth. Rev. Inst. Med. Trop. São Paulo 2020, 62, e56. [Google Scholar] [CrossRef]
- Sheridan, M.A.; Balaraman, V.; Schust, D.J.; Ezashi, T.; Roberts, R.M.; Franz, A.W. African and Asian strains of Zika virus differ in their ability to infect and lyse primitive human placental trophoblast. PLoS ONE 2018, 13, e0200086. [Google Scholar] [CrossRef] [PubMed]
- Bayer, A.; Lennemann, N.J.; Ouyang, Y.; Bramley, J.C.; Morosky, S.; Marques Jr, E.T.D.A.; Cherry, S.; Sadovsky, Y.; Coyne, C.B. Type III interferons produced by human placental trophoblasts confer protection against Zika virus infection. Cell Host Microbe 2016, 19, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, M.A.; Yunusov, D.; Balaraman, V.; Alexenko, A.P.; Yabe, S.; Verjovski-Almeida, S.; Schust, D.J.; Franz, A.W.; Sadovsky, Y.; Ezashi, T.; et al. Vulnerability of primitive human placental trophoblast to Zika virus. Proc. Natl. Acad. Sci. USA 2017, 114, E1587–E1596. [Google Scholar] [CrossRef]
- Pique-Regi, R.; Romero, R.; Tarca, A.L.; Luca, F.; Xu, Y.; Alazizi, A.; Leng, Y.; Hsu, C.-D.; Gomez-Lopez, N. Does the human placenta express the canonical cell entry mediators for SARS-CoV-2? Elife 2020, 9, e58716. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.M.; Green, J.A.; Schulz, L.C. The evolution of the placenta. Reproduction 2016, 152, R179–R189. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, G.; Funk, M.; Vernochet, C.; Leal, F.; Tarazona, O.A.; Meurice, G.; Heidmann, O.; Dupressoir, A.; Miralles, A.; Ramirez-Pinilla, M.P.; et al. An endogenous retroviral envelope syncytin and its cognate receptor identified in the viviparous placental Mabuya lizard. Proc. Natl. Acad. Sci. USA 2017, 114, E10991–E11000. [Google Scholar] [CrossRef] [PubMed]

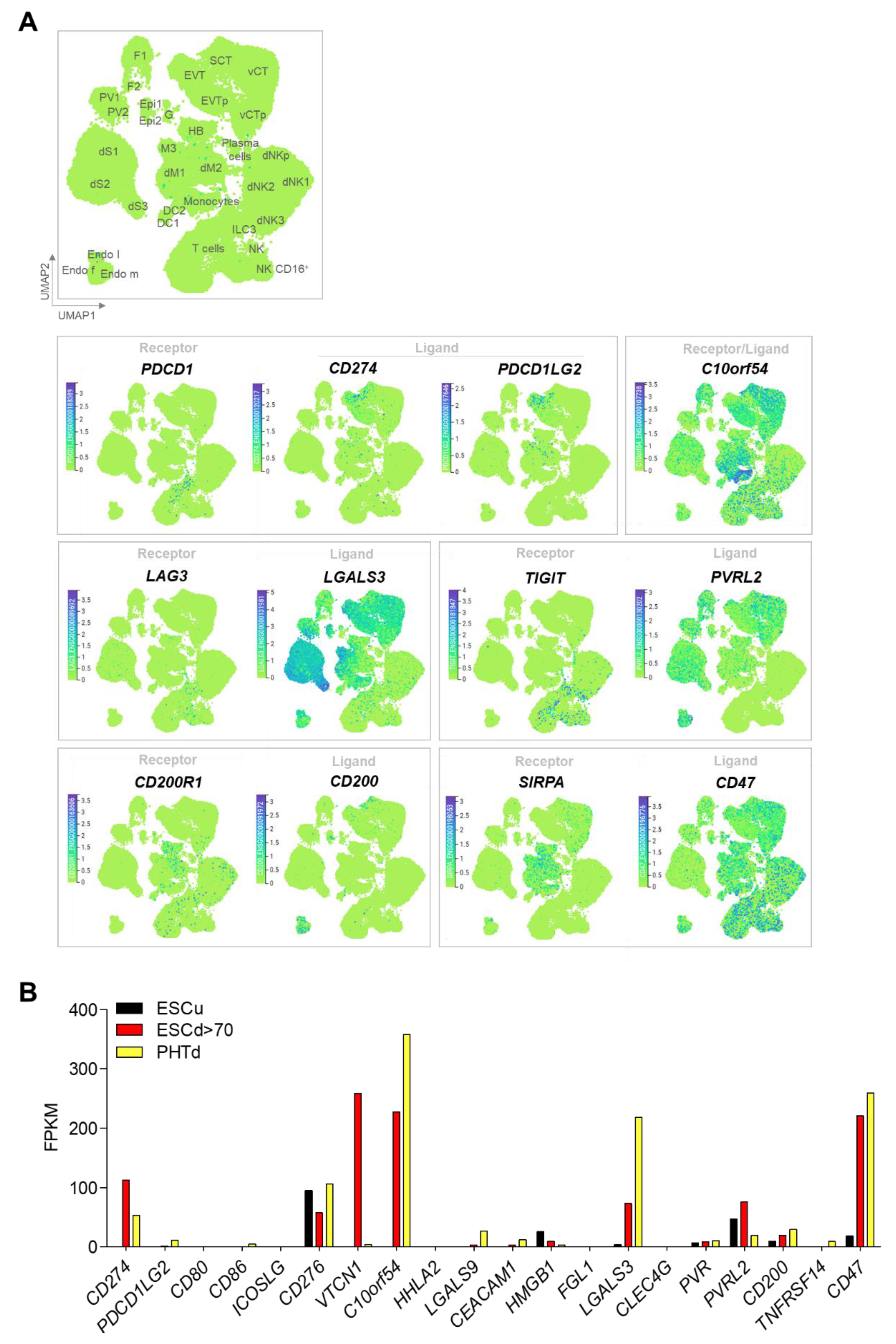
| Receptors | Ligands | ||
|---|---|---|---|
| Protein | Gene | Protein | Gene |
| PD1 | PDCD1 | PD-L1 | CD274 |
| PD-L2 | PDCD1LG2 | ||
| CTLA4 | CTLA4 | B7-1 | CD80 |
| B7-2 | CD86 | ||
| ICOS | ICOS | B7-H2 | ICOSLG |
| B7-H3 | CD276 | ||
| B7-H4 | VTCN1 | ||
| B7-H5 | C10orf54 | B7-H5 | C10orf54 (VSIR) |
| TMIGD2 | TMIGD2 | B7-H7 | HHLA2 |
| TIM3 | HAVCR2 | GAL9 | LGALS9 |
| CEACAM1 | CEACAM1 | ||
| HMGB1 | HMGB1 | ||
| LAG3 | LAG3 | FGL1 | FGL1 |
| GAL3 | LGALS3 | ||
| CLEC4G | CLEC4G | ||
| TIGIT | TIGIT | CD155 | PVR |
| CD112 | PVRL2 (NECTIN2) | ||
| CD200R1 | CD200R1 | CD200 | CD200 |
| BTLA | BTLA | HVEM | TNFRSF14 |
| SIRPα | SIRPA | CD47 | CD47 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schust, D.J.; Bonney, E.A.; Sugimoto, J.; Ezashi, T.; Roberts, R.M.; Choi, S.; Zhou, J. The Immunology of Syncytialized Trophoblast. Int. J. Mol. Sci. 2021, 22, 1767. https://doi.org/10.3390/ijms22041767
Schust DJ, Bonney EA, Sugimoto J, Ezashi T, Roberts RM, Choi S, Zhou J. The Immunology of Syncytialized Trophoblast. International Journal of Molecular Sciences. 2021; 22(4):1767. https://doi.org/10.3390/ijms22041767
Chicago/Turabian StyleSchust, Danny J., Elizabeth A. Bonney, Jun Sugimoto, Toshi Ezashi, R. Michael Roberts, Sehee Choi, and Jie Zhou. 2021. "The Immunology of Syncytialized Trophoblast" International Journal of Molecular Sciences 22, no. 4: 1767. https://doi.org/10.3390/ijms22041767
APA StyleSchust, D. J., Bonney, E. A., Sugimoto, J., Ezashi, T., Roberts, R. M., Choi, S., & Zhou, J. (2021). The Immunology of Syncytialized Trophoblast. International Journal of Molecular Sciences, 22(4), 1767. https://doi.org/10.3390/ijms22041767






