Selected Aspects Related to Medicinal and Aromatic Plants as Alternative Sources of Bioactive Compounds
Abstract
1. Introduction
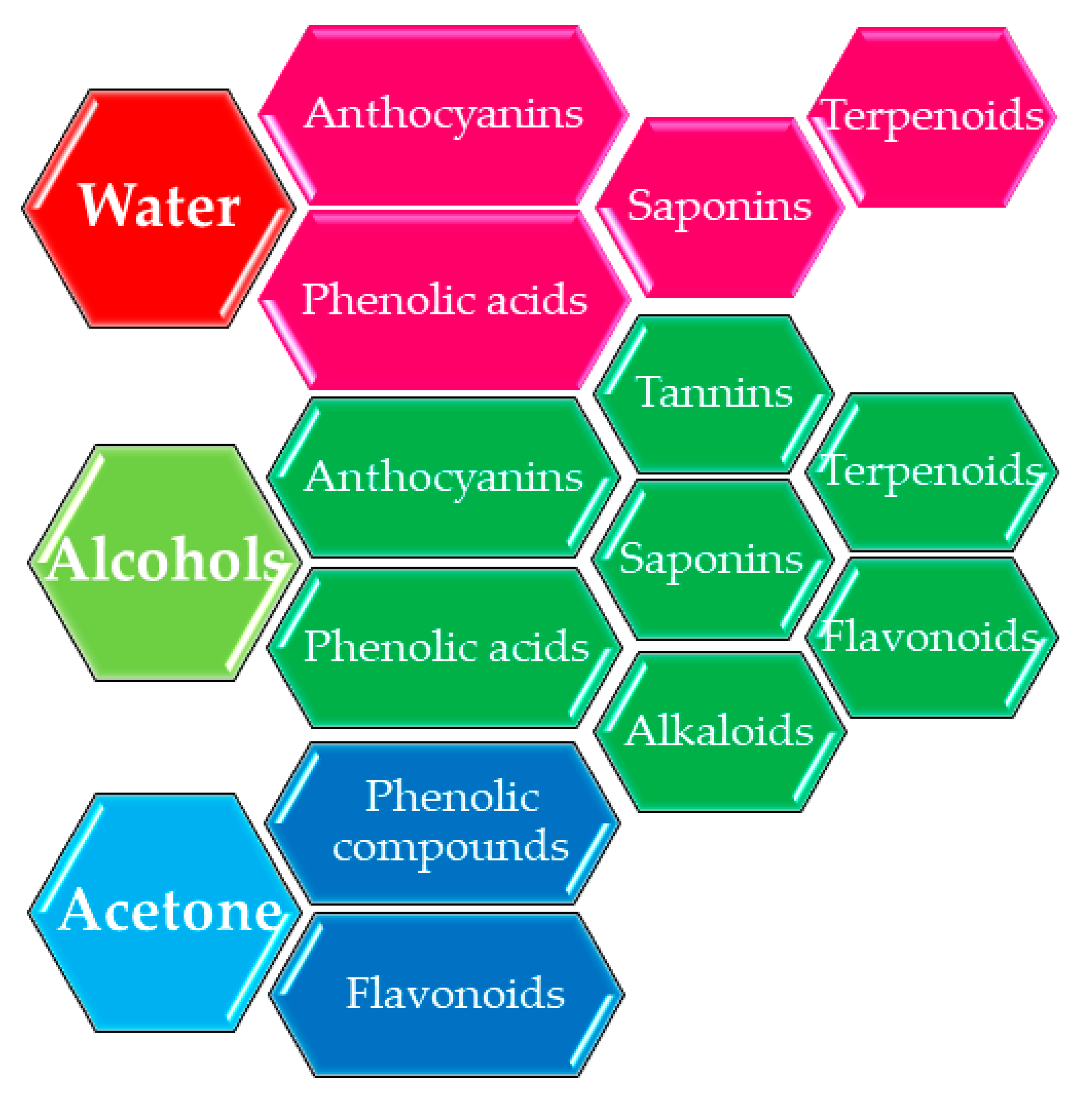
2. Classical Extraction Versus Modern Extraction Techniques: Possibilities to Scale up
3. The Influence of Extraction Conditions
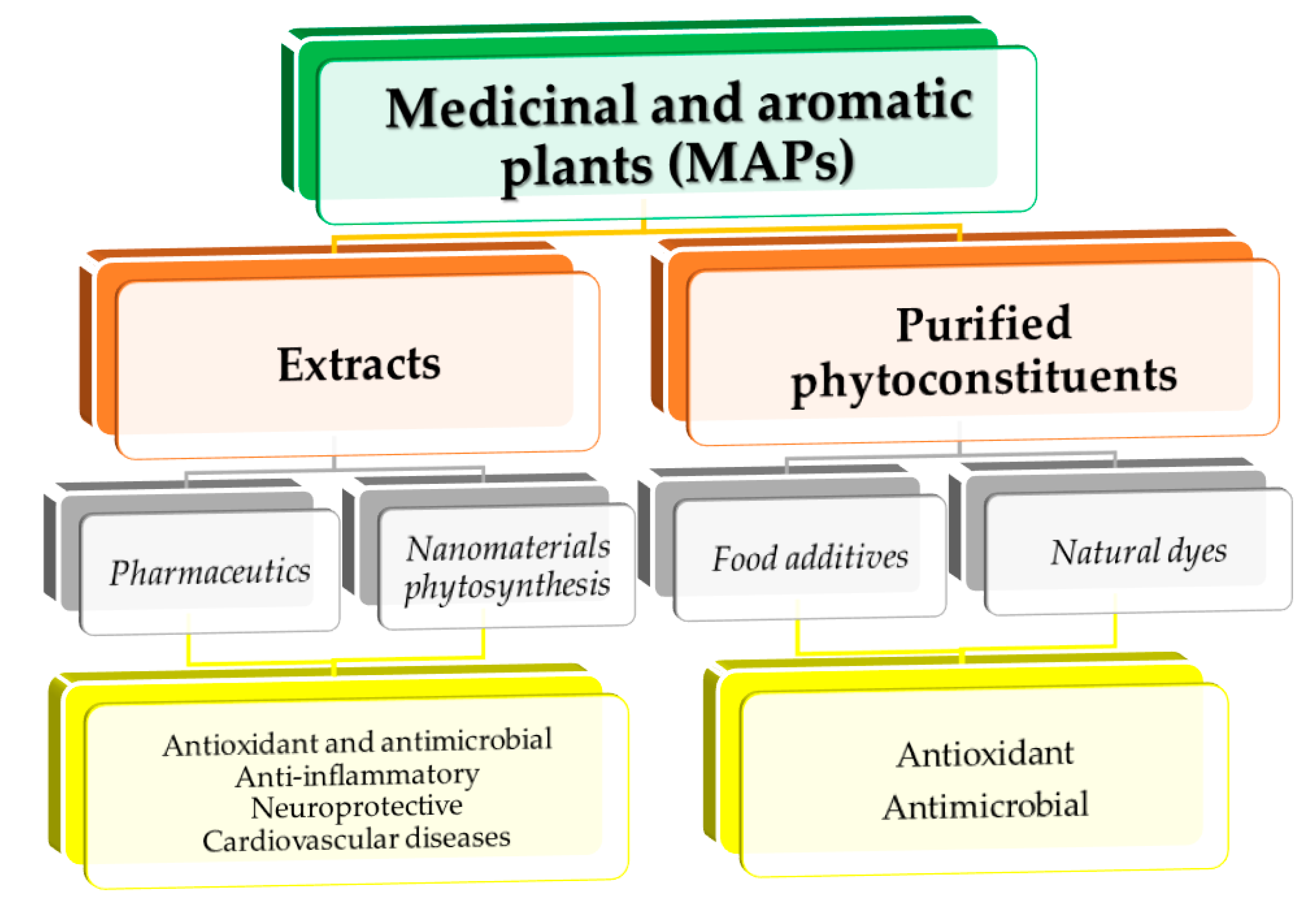
4. Different Medicinal and Aromatic Plants—Different Applications
4.1. Medical Applications
4.1.1. Origanum spp.
4.1.2. Thymus spp.
4.1.3. Salvia spp.
4.2. Industrial Applications
4.3. Applications in Nanotechnology
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fierascu, R.C.; Fierascu, I.; Ortan, A.; Georgiev, M.I.; Sieniawska, E. Innovative approaches for recovery of phytoconstituents from medicinal/aromatic plants and biotechnological production. Molecules 2020, 25, 309. [Google Scholar] [CrossRef] [PubMed]
- Manousi, N.; Sarakatsianos, I.; Samanidou, V. Extraction techniques of phenolic compounds and other bioactive compounds from medicinal and aromatic plants. In Engineering Tools in the Beverage Industry. Volume 3: The Science of Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing: Duxford, UK, 2019; pp. 283–314. [Google Scholar]
- Saha, A.; Basak, B.B. Scope of value addition and utilization of residual biomass from medicinal and aromatic plants. Ind. Crops Prod. 2020, 145, 111979. [Google Scholar] [CrossRef]
- Zengin, G.; Mollica, A.; Aumeeruddy, M.Z.; Rengasamy, K.R.; Mahomoodally, M.F. Phenolic profile and pharmacological propensities of Gynandriris sisyrinchium through in vitro and in silico perspectives. Ind. Crops Prod. 2018, 121, 328–337. [Google Scholar] [CrossRef]
- Fierascu, I.; Georgiev, M.I.; Ortan, A.; Fierascu, R.C.; Avramescu, S.M.; Ionescu, D.; Sutan, A.; Brinzan, A.; Ditu, L.M. Phyto-mediated metallic nano-architectures via Melissa officinalis L.: Synthesis, characterization and biological properties. Sci. Rep. 2017, 7, 12428. [Google Scholar] [CrossRef]
- Saha, A.; Tripathy, V.; Basak, B.B.; Kumar, J. Entrapment of distilled palmarosa (Cymbopogon martinii) wastes in alginate beads for adsorptive removal of methylene blue from aqueous solution. Environ. Prog. Sustain. Energy 2018, 37, 1942–1953. [Google Scholar] [CrossRef]
- Elfalleh, W.; Kirkan, B.; Sarikurkcu, C. Antioxidant potential and phenolic composition of extracts from Stachys tmolea: An endemic plant from Turkey. Ind. Crops Prod. 2019, 127, 212–216. [Google Scholar] [CrossRef]
- Volenzo, T.; Odiyo, J. Integrating endemic medicinal plants into the global value chains: The ecological degradation challenges and opportunities. Heliyon 2020, 6, e04970. [Google Scholar] [CrossRef]
- Bazak, R.; Houri, M.; Achy, S.E.; Hussein, W.; Refaat, T. Passive targeting of nanoparticles to cancer: A comprehensive review of the literature. Mol. Clin. Oncol. 2014, 2, 904908. [Google Scholar] [CrossRef]
- Chen, R.J.Y.; Jinn, T.R.; Chen, Y.C.; Chung, T.Y.; Yang, W.H.; Tzen, J.T.C. Active ingredients in Chinese medicines promoting blood circulation as Na+/K+-ATPase inhibitors. Acta Pharmacol. Sin. 2011, 32, 141–151. [Google Scholar] [CrossRef]
- Pacher, P.; Steffens, S.; Haskó, G.; Schindler, T.H.; Kunos, G. Cardiovascular effects of marijuana and synthetic cannabinoids: The good, the bad, and the ugly. Nat. Rev. Cardiol. 2018, 15, 151–166. [Google Scholar] [CrossRef]
- Chandrashekhar, V.M.; Ranpariya, V.L.; Ganapaty, S.; Parashar, A.; Muchandi, A.A. Neuroprotective activity of Matricaria recutita L. against global model ofischemia in rats. J. Ethnopharmacol. 2010, 127, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Berger, R.G.; Lunkenbein, S.; Ströhle, A.; Hahn, A. Antioxidants in food: Mere myth or magic medicine? Crit. Rev. Food Sci. 2012, 52, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Bohn, T. Exogenous antioxidants—double-edged swords in cellular redox state: Health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid. Med. Cell. Longev. 2010, 3, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Bessada, S.M.F.; Barreira, J.C.M.; Oliveira, M.B.P.P. Asteraceae species with most prominent bioactivity and their potential applications: A review. Ind. Crops Prod. 2015, 76, 604–615. [Google Scholar] [CrossRef]
- El-Sayed, S.M.; Youssef, A.M. Potential application of herbs and spices and their effects in functional dairy products. Heliyon 2019, 5, e01989. [Google Scholar] [CrossRef]
- Faccio, G. Plant complexity and cosmetic innovation. IScience 2020, 23, 101358. [Google Scholar] [CrossRef]
- Jha, P.; Sen, R.; Jobby, R.; Sachar, S.; Bhatkalkar, S.; Desai, N. Biotransformation of xenobiotics by hairy roots. Phytochemistry 2020, 176, 112421. [Google Scholar] [CrossRef]
- Rassem, H.H.A.; Nour, A.H.; Yunus, R.M. Techniques for extraction of essential oils from plants: A review. Aust. J. Basic Appl. Sci. 2016, 10, 117–127. [Google Scholar]
- Frohlich, P.C.; Santos, K.A.; Palú, F.; Cardozo-Filho, L.; da Silva, C.; da Silva, E.A. Evaluation of the effects of temperature and pressure on the extraction of eugenol from clove (Syzygium aromaticum) leaves using supercritical CO2. J. Supercrit. Fluids 2019, 143, 313–320. [Google Scholar] [CrossRef]
- Saleh, I.A.; Vinatoru, M.; Mason, T.J.; Abdel-Azim, N.S.; Aboutabl, E.A.; Hammouda, F.M. A possible general mechanism for ultrasound-assisted extraction (UAE) suggested from the results of UAE of chlorogenic acid from Cynara scolymus L. (artichoke) leaves. Ultrason. Sonochem. 2016, 31, 330–336. [Google Scholar] [CrossRef]
- Kimbaris, A.C.; Siatis, M.G.; Daferera, D.J.; Tarantilis, P.A.; Pappas, C.S.; Polissiou, M.G. Comparison of distillation and ultrasound-assisted extraction methods for the isolation of sensitive aroma compounds from garlic (Allium sativum). Ultrason. Sonochem. 2016, 13, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Périno-Issartier, S.; Zill-e-Huma; Abert-Vian, M.; Chemat, F. Solvent free microwave-assisted extraction of antioxidants from sea buckthorn (Hippophae rhamnoides) food by-products. Food Bioproc. Technol. 2011, 4, 1020–1028. [Google Scholar]
- Cvetanović, A.; Švarc-Gajić, J.; Zeković, Z.; Gašić, U.; Tešić, Z.; Zengin, G.; Mašković, P.; Mahomoodally, M.F.; Đurović, S. Subcritical water extraction as a cutting edge technology for the extraction of bioactive compounds from chamomile: Influence of pressure on chemical composition and bioactivity of extracts. Food Chem. 2018, 266, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Harbourne, N.; Jacquier, J.C.; O’Riordan, D. Optimisation of the extraction and processing conditions of chamomile (Matricaria chamomilla L.) for incorporation into a beverage. Food Chem. 2009, 115, 15–19. [Google Scholar] [CrossRef]
- Vian, M.A.; Fernandez, V.; Visinoni, F.; Chemat, F. Microwave hydrodiffusion and gravity, a new technique for extraction of essential oils. J. Chromatogr. A 2008, 1190, 14–17. [Google Scholar] [CrossRef]
- Patonay, K.; Szalontai, H.; Csugány, J.; Szabó-Hudák, O.; Kónya, E.P.; Németh, E.Z. Comparison of extraction methods for the assessment of total polyphenol content and in vitro antioxidant capacity of horsemint (Mentha longifolia (L.). J. Appl. Res. Med. Aromat. Plants 2019, 15, 100220. [Google Scholar] [CrossRef]
- Fornari, T.; Ruiz-Rodriguez, A.; Vicente, G.; Vázquez, E.; García-Risco, M.R.; Reglero, G. Kinetic study of the supercritical CO2 extraction of different plants from Lamiaceae family. J. Supercrit. Fluids 2012, 64, 1–8. [Google Scholar] [CrossRef]
- Binello, A.; Orio, L.; Pignata, G.; Nicola, S.; Chemat, F.; Cravotto, G. Effect of microwaves on the in situ hydrodistillation of four different Lamiaceae. Compt. Rendus Chim. 2014, 17, 181–186. [Google Scholar] [CrossRef]
- Wellwood, C.R.L.; Cole, R.A. Relevance of carnosic acid concentrations to the selection of rosemary, Rosmarinus officinalis (L.), accessions for optimization of antioxidant yield. J. Agric. Food Chem. 2004, 52, 6101–6107. [Google Scholar] [CrossRef]
- Carvalho, R.N.; Moura, L.S.; Rosa, P.T.V.; Meireles, M.A.A. Supercritical fluid extraction from rosemary (Rosmarinus officinalis): Kinetic data, extract’s global yield, composition, and antioxidant activity. J. Supercrit. Fluids 2005, 35, 197–204. [Google Scholar] [CrossRef]
- Rodríguez-Rojo, S.; Visentin, A.; Maestri, D.; Cocero, M.J. Assisted extraction of rosemary antioxidants with green solvents. J. Food Eng. 2012, 109, 98–103. [Google Scholar] [CrossRef]
- Jokić, S.; Molnar, M.; Jakovljević, M.; Aladić, K.; Jerković, I. Optimization of supercritical CO2 extraction of Salvia officinalis L. leaves targeted on oxygenated monoterpenes, α-humulene, viridiflorol and Manool. J. Supercrit. Fluids 2018, 133, 253–262. [Google Scholar] [CrossRef]
- Miguel, G.; Cruz, C.; Faleiro, M.L.; Simões, M.T.F.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G. Salvia officinalis L. essential oils: Effect of hydrodistillation time on the chemical composition, antioxidant and antimicrobial activities. Nat. Prod. Res. 2011, 25, 526–541. [Google Scholar] [CrossRef] [PubMed]
- Ollanketo, M.; Peltoketo, A.; Hartonen, K.; Hiltunen, R.; Riekkola, M.L. Extraction of sage (Salvia officinalis L.) by pressurized hot water and conventional methods: Antioxidant activity of the extracts. Eur. Food Res. Technol. 2002, 215, 158–163. [Google Scholar] [CrossRef]
- Mašković, P.; Veličković, V.; Mitić, M.; Đurović, S.; Zeković, Z.; Radojković, M.; Cvetanović, A.; Švarc-Gajić, J.; Vujić, J. Summer savory extracts prepared by novel extraction methods resulted in enhanced biological activity. Ind. Crops Prod. 2017, 109, 875–881. [Google Scholar]
- Nickavar, B.; Mojab, F.; Dolat-Abadi, R. Analysis of the essential oils of two Thymus species from Iran. Food Chem. 2005, 90, 609–611. [Google Scholar] [CrossRef]
- Bendif, H.; Adouni, K.; Miara, M.D.; Baranauskienė, R.; Kraujalis, P.; Venskutonis, P.R.; Nabavi, S.M.; Maggi, F. Essential oils (EOs), pressurized liquid extracts (PLE) and carbon dioxide supercritical fluid extracts (SFE-CO2) from Algerian Thymus munbyanus as valuable sources of antioxidants to be used on an industrial level. Food Chem. 2018, 260, 289–298. [Google Scholar] [CrossRef]
- Armenta, S.; Garrigues, S.; de la Guardia, M. The role of green extraction techniques in green analytical chemistry. TrAC Trends Anal. Chem. 2015, 71, 2–8. [Google Scholar] [CrossRef]
- Armenta, S.; de la Guardia, M.; Namiesnik, J. Green microextraction. In Analytical Microextraction Techniques; Valcarcel, M., Ed.; Bentham Science Publishers: Sharjah, UAE, 2017; pp. 3–27. [Google Scholar]
- Armenta, S.; Esteve-Turrillas, F.A.; Garrigues, S.; de la Guardia, M. Green Analytical Chemistry: The Role of Green Extraction Techniques. In Green Extraction Techniques Principles, Advances and Applications; Ibáñez, E., Cifuente, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 76, pp. 1–25. [Google Scholar]
- Bravo, J.; Monente, C.; Juániz, I.; De Peña, M.P.; Cid, C. Influence of extraction process on antioxidant capacity of spent coffee. Food Res. Int. 2013, 50, 610–616. [Google Scholar] [CrossRef]
- Espino, M.; de los Ángeles Fernández, M.; Gomez, F.J.V.; Silva, M.F. Natural designer solvents for greening analytical chemistry. TrAC Trends Anal. Chem. 2016, 76, 126–136. [Google Scholar] [CrossRef]
- Espino, M.; de los Ángeles Fernández, M.; Gomez, F.J.V.; Boiteux, J.; Silva, M.F. Green analytical chemistry metrics: Towards a sustainable phenolics extraction from medicinal plants. Microchem. J. 2018, 141, 438–443. [Google Scholar] [CrossRef]
- Khaw, K.Y.; Parat, M.O.; Shaw, P.N.; Falconer, J.R. Solvent supercritical fluid technologies to extract bioactive compounds from natural sources: A review. Molecules 2017, 22, 1186. [Google Scholar] [CrossRef]
- Lefebvre, T.; Destandau, E.; Lesellier, E. Selective extraction of bioactive compounds from plants using recent extraction techniques: A review. J. Chromatogr. A 2021, 1635, 461770. [Google Scholar] [CrossRef] [PubMed]
- Latif, S.; Anwar, F. Physicochemical studies of hemp (Cannabis sativa) seed oil using enzyme-assisted cold-pressing. Eur. J. Lipid Sci. Technol. 2009, 111, 1042–1048. [Google Scholar] [CrossRef]
- Cvetanović, A.; Švarc-Gajić, J.; Mašković, P.; Savić, S.; Nikolić, L.J. Antioxidant and biological activity of chamomile extracts obtained by different techniques: Perspective of using superheated water for isolation of biologically active compounds. Ind. Crops Prod. 2015, 65, 582–591. [Google Scholar]
- Belwal, T.; Chemat, F.; Venskutonis, P.R.; Cravotto, G.; Jaiswal, D.K.; Bhatt, I.D.; Devkota, H.P.; Luo, Z. Recent advances in scaling-up of non-conventional extraction techniques: Learning from successes and failures. TrAC Trend Anal. Chem. 2020, 127, 115895. [Google Scholar] [CrossRef]
- Kotnik, P.; Skerget, M.; Knez, Z. Supercritical fluid extraction of chamomile flower heads: Comparison with conventional extraction, kinetics and scale-up. J. Supercrit. Fluids 2017, 43, 192–198. [Google Scholar] [CrossRef]
- Meullemiestre, A.; Petitcolas, E.; Maache-Rezzoug, Z.; Chemat Rezzoug, S.A. Impact of ultrasound on solid–liquid extraction of phenolic compounds from maritime pine sawdust waste. Kinetics, optimization and large-scale experiments. Ultrason. Sonochem. 2016, 28, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Fierascu, R.C.; Sieniawska, E.; Ortan, A.; Fierascu, I.; Xiao, J. Fruits by-products—A source of valuable active principles. A short review. Front. Bioeng. Biotechnol. 2020, 8, 319. [Google Scholar] [CrossRef]
- Ferreira, L.F.; Minuzzi, N.M.; Rodrigues, R.F.; Pauletto, R.; Rodrigues, E.; Emanuelli, T.; Bochi, V.C. Citric acid water-based solution for blueberry bagasse anthocyanins recovery: Optimization and comparisons with microwave-assisted extraction (MAE). LWT 2020, 133, 110064. [Google Scholar] [CrossRef]
- Yang, Y.; Kayan, B.; Bozer, N.; Pate, B.; Baker, C.; Gizir, A.M. Terpene degradation and extraction from basil and oregano leaves using subcritical water. J. Chromatogr. A 2007, 1152, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.G.G.; Mazzutti, S.; Vitali, L.; Micke, G.A.; Ferreira, S.R.S. Recovery of bioactive phenolic compounds from papaya seeds agroindustrial residue using subcritical water extraction. Biocatal. Agric. Biotechnol. 2019, 22, 101367. [Google Scholar] [CrossRef]
- Majeed, M.; Hussain, A.I.; Chatha, S.A.; Khosa, M.K.; Kamal, G.M.; Kamal, M.A.; Zhang, X.; Liu, M. Optimization protocol for the extraction of antioxidant components from Origanum vulgare leaves using response surface methodology. Saudi J. Biol. Sci. 2016, 23, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Aziz, N.A.A.; Hasham, R.; Sarmidi, M.R.; Suhaimi, S.H.; Idris, M.K.H. A review on extraction techniques and therapeutic value of polar bioactives from Asian medicinal herbs: Case study on Orthosiphon aristatus, Eurycoma longifolia and Andrographis paniculate. Saudi Pharm. J. 2021. [Google Scholar] [CrossRef]
- Švarc-Gajić, J.; Stojanović, Z.; Carretero, A.S.; Román, D.A.; Borrás, I.; Vasiljević, I. Development of a microwave-assisted extraction for the analysis of phenolic compounds from Rosmarinus officinalis. J. Food Eng. 2013, 119, 525–532. [Google Scholar] [CrossRef]
- Thuy Pham, T.P.; Cho, C.W.; Yun, Y.S. Environmental fate and toxicity of ionic liquids: A review. Water Res. 2010, 44, 352–372. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Geng, Y.; Duan, W.; Wang, D.; Fu, M.; Wang, X. Ionic liquid-based ultrasoundassisted extraction of fangchinoline and tetrandrine from Stephaniae tetrandrae. J. Sep. Sci. 2009, 32, 3550–3554. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Ye, X.; Lu, Y.; Yu, Y.; Mo, W. Ionic liquid-based ultrasonic-assisted extraction of piperine from white pepper. Anal. Chim. Acta 2009, 640, 47–51. [Google Scholar] [CrossRef]
- Mbous, Y.P.; Hayyan, M.; Hayyan, A.; Wong, W.F.; Hashim, M.A.; Looi, C.Y. Applications of deep eutectic solvents in biotechnology and bioengineering—Promises and challenges. Biotechnol. Adv. 2017, 35, 105–134. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Ravi, H.K.; Khadhraoui, B.; Hilali, S.; Perino, S.; Tixier, A.S.F. Review of alternative solvents for green extraction of food and natural products: Panorama, principles, applications and prospects. Molecules 2019, 24, 3007. [Google Scholar] [CrossRef]
- Fierascu, R.C.; Fierascu, I.; Avramescu, S.M.; Sieniawska, E. Recovery of natural antioxidants from agro-industrial side streams through advanced extraction techniques. Molecules 2019, 24, 4212. [Google Scholar] [CrossRef] [PubMed]
- Skarpalezos, D.; Detsi, A. Deep eutectic solvents as extraction media for valuable flavonoids from natural sources. Appl. Sci. 2019, 9, 4169. [Google Scholar] [CrossRef]
- Pan, G.; Yu, G.; Zhu, C.; Qiao, J. Optimization of ultrasound-assisted extraction (UAE) of flavonoids compounds (FC) from hawthorn seed (HS). Ultrason. Sonochem. 2012, 19, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Sarfarazi, M.; Jafari, S.M.; Rajabzadeh, G.; Feizi, J. Development of an environmentally-friendly solvent-free extraction of saffron bioactives using subcritical water. LWT 2019, 114, 108428. [Google Scholar] [CrossRef]
- Fan, R.; Xiang, J.; Li, N.; Jiang, X.; Gao, Y. Impact of extraction parameters on chemical composition and antioxidant activity of bioactive compounds from Chinese licorice (Glycyrrhiza uralensis Fisch.) by subcritical water. Sep. Sci. Technol. 2016, 51, 609–621. [Google Scholar] [CrossRef]
- Munir, M.T.; Kheirkhah, H.; Baroutian, S.; Quek, S.Y.; Young, B.R. Subcritical water extraction of bioactive compounds from waste onion skin. J. Clean. Prod. 2018, 183, 487–494. [Google Scholar] [CrossRef]
- Amza, T.; Balla, A.; Tounkara, F.; Man, L.; Zhou, H.M. Effect of hydrolysis time on nutritional, functional and antioxidant properties of protein hydrolysates prepared from gingerbread plum (Neocarya macrophylla) seeds. Int. Food Res. J. 2013, 20, 2081–2090. [Google Scholar]
- Skoula, M.; Harborne, J.B. The taxonomy and chemistry of Origanum. In Oregano: The genera Origanum and Lippia; Kintzios, S.E., Ed.; Taylor & Francis: London, UK, 2002; pp. 67–108. [Google Scholar]
- Marrelli, M.; Statti, G.A.; Conforti, F. Origanum spp.: An update of their chemical and biological profiles. Phytochem. Rev. 2018, 17, 873–888. [Google Scholar] [CrossRef]
- Postu, P.A.; Gorgan, D.L.; Cioanca, O.; Russ, M.; Mikkat, S.; Glocker, M.O.; Hritcu, L. Memory-Enhancing Effects of Origanum majorana Essential Oil in an Alzheimer’s Amyloid beta1-42 Rat Model: A Molecular and Behavioral Study. Antioxidants 2020, 9, 919. [Google Scholar] [CrossRef]
- Alonazi, M.A.; Jemel, I.; Moubayed, N.; Alwhibi, M.; El-Sayed, N.N.E.; Bacha, A.B. Evaluation of the in vitro anti-inflammatory and cytotoxic potential of ethanolic and aqueous extracts of Origanum syriacum and Salvia lanigera leaves. Environ. Sci. Pollut. Res. 2021. [Google Scholar] [CrossRef]
- Han, F.; Ma, G.; Yang, M.; Yan, L.; Ziong, W.; Shu, J.; Zhao, Z.; Xu, H. Chemical composition and antioxidant activities of essential oils from different parts of the oregano. J. Zhejiang Univ. Sci. B 2017, 18, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.R.; Nadeem, M.; Bhutto, M.A.; Yu, F.; Xie, X.; El-Hamshary, H.; El-Faham, A.; Ibrahim, U.A.; Mo, X. Physico-chemical and biological evaluation of PLCL/SF nanofibers loaded with oregano essential oil. Pharmaceutics 2019, 11, 386. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Grijalva, E.P.; Antunes-Ricardo, M.; Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Heredia, J.B. Cellular antioxidant activity and in vitro inhibition of α-glucosidase, α-amylase and pancreatic lipase of oregano polyphenols under simulated gastrointestinal digestion. Food Res. Int. 2019, 116, 676–686. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Garcia, C.V.; Shin, G.H.; Kim, J.T. Antibacterial and antioxidant properties of hydroxypropyl methylcellulose-based active composite films incorporating oregano essential oil nanoemulsions. LWT 2019, 106, 164–171. [Google Scholar] [CrossRef]
- Dutra, T.V.; Castro, J.C.; Menezes, J.L.; Ramos, T.R.; Prado, I.N.; Machinski, M.; Mikcha, J.M.G.; de Abreu Filho, B.A. Bioactivity of oregano (Origanum vulgare) essential oil against Alicyclobacillus spp. Ind. Crops Prod. 2019, 129, 345–349. [Google Scholar] [CrossRef]
- Khan, A.R.; Huang, K.; Jinzhong, Z.; Zhu, T.; Morsi, Y.; Aldalbahi, A.; El-Newehy, M.; Yan, X.; Mo, X. PLCL/Silk fibroin based antibacterial nano wound dressing encapsulating oregano essential oil: Fabrication, characterization and biological evaluation. Colloid Surf. B Biointerfaces 2020, 196, 111352. [Google Scholar] [CrossRef]
- Avola, R.; Granata, G.; Geraci, C.; Napoli, E.; Graziano, A.C.E.; Cardile, V. Oregano (Origanum vulgare L.) essential oil provides anti-inflammatory activity and facilitates wound healing in a human keratinocytes cell model. Food Chem. Toxicol. 2020, 144, 111586. [Google Scholar] [CrossRef]
- Fernández-Marín, R.; Labidi, J.; Andrés, M.A.; Fernandes, S.C.M. Using α-chitin nanocrystals to improve the final properties of poly (vinyl alcohol) films with Origanum vulgare essential oil. Polym. Degrad. Stab. 2020, 179, 109227. [Google Scholar] [CrossRef]
- Salehi, B.; Ata, A.V.; Anil Kumar, N.; Sharopov, F.; Ramírez-Alarcón, K.; Ruiz-Ortega, A.; Abdulmajid Ayatollahi, S.; Valere Tsouh Fokou, P.; Kobarfard, F.; Amiruddin Zakaria, Z.; et al. Antidiabetic potential of medicinal plants and their active components. Biomolecules 2019, 9, 551. [Google Scholar] [CrossRef]
- Radünz, M.; Camargo, T.M.; dos Santos Hackbart, H.C.; Alves, P.I.C.; Radünz, A.L.; Gandra, E.A.; Zavareze, E.R. Chemical composition and in vitro antioxidant and antihyperglycemic activities of clove, thyme, oregano, and sweet orange essential oils. LWT 2021, 138, 110632. [Google Scholar] [CrossRef]
- Ayari, S.; Shankar, S.; Follett, P.; Hossain, F.; Lacroix, M. Potential synergistic antimicrobial efficiency of binary combinations of essential oils against Bacillus cereus and Paenibacillus amylolyticus-Part A. Microb. Pathog. 2020, 141, 104008. [Google Scholar] [CrossRef] [PubMed]
- Begum, T.; Follett, P.A.; Hossain, F.; Christopher, L.; Salmieri, S.; Lacroix, M. Microbicidal effectiveness of irradiation from Gamma and X-ray sources at different dose rates against the foodborne illness pathogens Escherichia coli, Salmonella Typhimurium and Listeria monocytogenes in rice. LWT 2020, 132, 109841. [Google Scholar] [CrossRef]
- Huang, W.; Tao, F.; Li, F.; Mortimer, M.; Guo, L.H. Antibacterial nanomaterials for environmental and consumer product applications. NanoImpact 2020, 20, 100268. [Google Scholar] [CrossRef]
- Augustine, R.; Hasan, A. Emerging applications of biocompatible phytosynthesized metal/metal oxide nanoparticles in healthcare. J. Drug Deliv. Sci. Technol. 2020, 56, 101516. [Google Scholar] [CrossRef]
- Beikzadeh, S.; Khezerlou, A.; Jafari, S.M.; Pilevar, Z.; Mortazavian, A.M. Seed mucilages as the functional ingredients for biodegradable films and edible coatings in the food industry. Adv. Colloid Interface Sci. 2020, 280, 102164. [Google Scholar] [CrossRef]
- Nabavi, S.M.; Marchese, A.; Izadi, M.; Curti, V.; Daglia, M.; Nabavi, S.F. Plants belonging to the genus Thymus as antibacterial agents: From farm to pharmacy. Food Chem. 2015, 173, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Abu-Darwish, M.S.; Tarawneh, A.H.; Cabral, C.; Gadetskaya, A.V.; Salgueiro, L.; Hosseinabadi, T.; Rajabi, S.; Chanda, W.; Sharifi-Rad, M.; et al. Thymus spp. plants-Food applications and phytopharmacy properties. Trends Food Sci. Technol. 2019, 85, 287–306. [Google Scholar] [CrossRef]
- Miguel, G.; Simoes, M.; Figueiredo, A.; Barroso, J.; Pedro, L.; Carvalho, L. Composition and antioxidant activities of the essential oils of Thymus caespititius, Hymus camphoratus and Thymus mastichina. Food Chem. 2004, 86, 183–188. [Google Scholar] [CrossRef]
- Giordani, R.; Hadef, Y.; Kaloustian, J. Compositions and antifungal activities of essential oils of some Algerian aromatic plants. Fitoterapia 2008, 79, 199–203. [Google Scholar] [CrossRef]
- Pavel, M.; Ristić, M.; Stević, T. Essential oils of Thymus pulegioides and Thymus glabrescens from Romania: Chemical composition and antimicrobial activity. J. Serb. Chem. Soc. 2010, 75, 27–34. [Google Scholar] [CrossRef]
- Muslim, S.N.; Hussin, Z.S. Chemical compounds and synergistic antifungal properties of Thymus kotschanus essential oil plus ketoconazole against Candida spp. Gene Rep. 2020, 21, 100916. [Google Scholar] [CrossRef]
- Najafloo, R.; Behyari, M.; Imani, R.; Nour, S. A mini-review of Thymol incorporated materials: Applications in antibacterial wound dressing. J. Drug Deliv. Sci. Technol. 2020, 60, 101904. [Google Scholar] [CrossRef]
- Guesmi, F.; Khantouche, L.; Mehrez, A.; Bellamine, H.; Landoulsi, A. Histopathological and biochemical effects of thyme essential oil on H2O2 stress in heart tissues. Heart Lung Circ. 2020, 29, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Abdelli, I.; Hassani, F.; Brikci, S.B.; Ghalem, S. In silico study the inhibition of angiotensin converting enzyme 2 receptor of COVID-19 by Ammoides verticillata components harvested from Western Algeria. J. Biomol. Struct. Dyn. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yepes-Pérez, A.F.; Herrera-Calderon, O.; Sánchez-Aparicio, J.E.; Tiessler-Sala, L.; Maréchal, J.D.; Cardona, G.W. Investigating potential inhibitory effect of Uncaria tomentosa (Cat’s Claw) against the main protease 3CLpro of SARS-CoV-2 by molecular modeling. Evid. Based Complement. Altern. Med. 2020, 2020, 4932572. [Google Scholar]
- Asgarpanah, J.; Oveyli, E.; Alidoust, S. Volatile components of the endemic species Salvia sharifii Rech f. & Esfand. J. Essent. Oil Bear. Plants 2017, 20, 578–582. [Google Scholar]
- Wei, T.; Gao, Y.; Deng, K.; Zhang, L.; Yang, M.; Liu, X.; Qi, C.; Wang, C.; Song, W.; Zhang, Y.; et al. Enhancement of tanshinone production in Salvia miltiorrhiza hairy root cultures by metabolic engineering. Plant Methods 2019, 15, 53. [Google Scholar] [CrossRef]
- Lin, Y.S.; Peng, W.H.; Shih, M.F.; Cherng, J.Y. Anxiolytic effect of an extract of Salvia miltiorrhiza Bunge (Danshen) in mice. J. Ethnopharmacol. 2021, 264, 113285. [Google Scholar] [CrossRef]
- Orgah, J.O.; Hea, S.; Wang, Y.; Jiang, M.; Wang, Y.; Orgah, E.A.; Duand, Y.; Zhao, B.; Zhang, B.; Hand, J.; et al. Pharmacological potential of the combination of Salvia miltiorrhiza (Danshen) and Carthamus tinctorius (Honghua) for diabetes mellitus and its cardiovascular complications. Pharmacol. Res. 2020, 153, 104654. [Google Scholar] [CrossRef]
- Arras, G.; Piga, A.; Agabbio, M. Effect of TBZ, acetaldehyde, citral and Thymus capitatus essential oil on “Minneola” tangelo fruit decay. Proc. Int. Soc. Citric. 1996, 1, 406–409. [Google Scholar]
- Martínez, K.; Ortiz, M.; Albis, A.; Castañeda, C.G.G.; Valencia, M.E.; Grande Tovar, C.D. The effect of edible chitosan coatings incorporated with Thymus capitatus essential oil on the shelf-life of strawberry (Fragaria x ananassa) during cold storage. Biomolecules 2018, 8, 155. [Google Scholar] [CrossRef] [PubMed]
- Jemaa, M.B.; Falleh, H.; Serairi, R.; Neves, M.A.; Snoussi, M.; Isoda, H.; Nakajima, M.; Ksouri, R. Nanoencapsulated Thymus capitatus essential oil as natural preservative. Innov. Food Sci. Emerg. Technol. 2018, 45, 92–97. [Google Scholar] [CrossRef]
- Arabpoor, B.; Yousefi, S.; Weisany, W.; Ghasemlou, M. Multifunctional coating composed of Eryngium campestre L. essential oil encapsulated in nano-chitosan to prolong the shelf-life of fresh cherry fruits. Food Hydrocoll. 2021, 111, 106394. [Google Scholar] [CrossRef]
- Mehdizadeh, T.; Tajik, H.; Langroodi, A.M.; Molaei, R.; Mahmoudian, A. Chitosan-starch film containing pomegranate peel extract and Thymus kotschyanus essential oil can prolong the shelf life of beef. Meat Sci. 2020, 163, 108073. [Google Scholar] [CrossRef] [PubMed]
- Javaherzadeh, R.; Tabatabaee Bafroee, A.S.; Kanjari, A. Preservation effect of Polylophium involucratum essential oil incorporated poly lactic acid/ nanochitosan composite film on shelf life and sensory properties of chicken fillets at refrigeration temperature. LWT 2020, 118, 108783. [Google Scholar] [CrossRef]
- Rehman, A.; Jafari, S.M.; Aadil, R.M.; Assadpour, E.; Randhawa, M.A.; Mahmood, S. Development of active food packaging via incorporation of biopolymeric nanocarriers containing essential oils. Trends Food Sci. Technol. 2020, 101, 106–121. [Google Scholar] [CrossRef]
- Hosseini, H.; Hamgini, E.Y.; Jafari, S.M.; Bolourian, S. Improving the oxidative stability of sunflower seed kernels by edible biopolymeric coatings loaded with rosemary extract. J. Stored Prod. Res. 2020, 89, 101729. [Google Scholar] [CrossRef]
- Choulitoudi, E.; Ganiari, S.; Tsironi, T.; Ntzimani, A.; Tsimogiannis, D.; Taoukis, P.; Oreopoulou, V. Edible coating enriched with rosemary extracts to enhance oxidative and microbial stability of smoked eel fillets. Food Packag. Shelf Life 2017, 12, 107–113. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G.; Canale, A.; Maggi, F.; Mártonfi, P. Exploring essential oils of Slovak medicinal plants for insecticidal activity: The case of Thymus alternans and Teucrium montanum subsp. Jailae. Food Chem. Toxicol. 2020, 138, 111203. [Google Scholar] [CrossRef]
- Bozkurt, M.; Tüzün, A.E. Application of aromatic plants and their extracts in diets of turkeys, In Feed Additives. Aromatic Plants and Herbs in Animal Nutrition and Health, 1st ed.; Florou-Paneri, P., Christaki, E., Giannenas, I., Eds.; Academic Press: London, UK, 2020; pp. 205–226. [Google Scholar]
- Saroj, A.; Oriyomi, O.V.; Nayak, A.K.; Haider, Z. Phytochemicals of plant-derived essential oils: A novel green approach against pests. In Natural Remedies for Pest, Disease and Weed Control; Egbuna, C., Sawicka, B., Eds.; Academic Press: London, UK, 2020; pp. 65–79. [Google Scholar]
- de Paris, M.; Stivanin, S.C.B.; Klein, C.P.; Vizzotto, E.F.; Passos, L.T.; Angelo, I.D.V.; Zanela, M.B.; Stone, V.; Matté, C.; Heisler, G.; et al. Calves fed with milk from cows receiving plant extracts improved redox status. Livest. Sci. 2020, 242, 104272. [Google Scholar] [CrossRef]
- Harhaun, R.; Kunik, O.; Saribekova, D.; Lazzara, G. Biologically active properties of plant extracts in cosmetic emulsions. Microchem. J. 2020, 154, 104543. [Google Scholar] [CrossRef]
- Bom, S.; Jorge, J.; Ribeiro, H.M.; Marto, J. A step forward on sustainability in the cosmetics industry: A review. J. Clean. Prod. 2019, 225, 270–290. [Google Scholar] [CrossRef]
- Chou, S.T.; Chang, W.L.; Chang, C.T.; Hsu, S.L.; Lin, Y.C.; Shih, Y. Cinnamomum cassia essential oil inhibits α-MSH-induced melanin production and oxidative stress in murine B16 melanoma cells. Int. J. Mol. Sci. 2013, 14, 19186–19201. [Google Scholar] [CrossRef] [PubMed]
- Aumeeruddy-Elalfi, Z.; Lall, N.; Fibrich, B.; Van Staden, A.B.; Hosenally, M.; Mahomoodally, M.F. Selected essential oils inhibit key physiological enzymes and possess intracellular and extracellular antimelanogenic properties in vitro. J. Food Drug Anal. 2018, 26, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Maria, M.F.F.; Ikhmal, W.M.K.W.M.; Amirah, M.N.N.S.; Manja, S.M.; Syaizwadi, S.M.; Chan, K.S.; Sabri, M.G.M.; Adnan, A. Green approach in anti-corrosion coating by using Andrographis paniculata leaves extract as additives of stainless steel 316L in seawater. Int. J. Corros. Scale Inhib. 2019, 8, 644–658. [Google Scholar]
- Ong, G.; Kasi, R.; Subramaniam, R. A review on plant extracts as natural additives in coating applications. Prog. Org. Coat. 2021, 151, 106091. [Google Scholar] [CrossRef]
- Brobbey, K.J.; Saarinen, J.; Alakomi, H.; Yang, B.; Toivakka, M. Efficacy of natural plant extracts in antimicrobial packaging systems. J. Appl. Packag. Res. 2017, 9, 60–71. [Google Scholar]
- Shlar, I.; Droby, S.; Choudhary, R.; Rodov, V. The mode of antimicrobial action of curcumin depends on the delivery system: Monolithic nanoparticles: Vs. Supramolecular inclusion complex. RSC Adv. 2017, 7, 42559–42569. [Google Scholar] [CrossRef]
- Agostini, V.O.; Macedo, A.J.; Muxagata, E.; da Silva, M.V.; Pinho, G.L.L. Non-toxic antifouling potential of Caatinga plant extracts: Effective inhibition of marine initial biofouling. Hydrobiologia 2020, 847, 45–60. [Google Scholar] [CrossRef]
- Tomak, E.D.; Yazici, O.A.; Parmak, E.D.S.; Gonultas, O. Influence of tannin containing coatings on weathering resistance of wood: Combination with zinc and cerium oxide nanoparticles. Polym. Degrad. Stab. 2018, 152, 289–296. [Google Scholar] [CrossRef]
- Peng, Y.; Wang, Y.; Chen, P.; Wang, W.; Cao, J. Enhancing weathering resistance of wood by using bark extractives as natural photostabilizers in polyurethane-acrylate coating. Prog. Org. Coat. 2020, 145, 105665. [Google Scholar] [CrossRef]
- Iamareerat, B.; Singh, M.; Sadiq, M.B.; Anal, A.K. Reinforced cassava starch based edible film incorporated with essential oil and sodium bentonite nanoclay as food packaging material. J. Food Sci. Technol. 2018, 55, 1953–1959. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Wang, X.; Zhang, D.; Wei, Y.; Cui, Y.; Shia, W.; Bao, Y. Synergistic use of florfenicol and Salvia miltiorrhiza polysaccharide can enhance immune responses in broilers. Ecotoxicol. Environ. Saf. 2021, 210, 111825. [Google Scholar] [CrossRef] [PubMed]
- Jokanović, M.; Snežan, M.; Vladimir Tomović, S.; Pavlić, B.; Šojić, B.; Zeković, Z.; Peulić, T.; Ikonić, P. Essential oil and supercritical extracts of winter savory (Satureja montana L.) as antioxidants in precooked pork chops during chilled storage. LWT 2020, 134, 110260. [Google Scholar] [CrossRef]
- Šojić, B.; Tomović, V.; Kocić-Tanackov, S.; Bursać Kovačević, D.; Putnik, P.; Mrkonjić, Z.; Đurović, S.; Jokanović, M.; Snežan, M.; Branimir Pavlić, S. Supercritical extracts of wild thyme (Thymus serpyllum L.) by-product as natural antioxidants in ground pork patties. LWT 2020, 130, 109661. [Google Scholar] [CrossRef]
- Badawy, M.E.I.; Lotfy, T.M.R.; Shawir, S.M.S. Facile synthesis and characterizations of antibacterial and antioxidant of chitosan monoterpene nanoparticles and their applications in preserving minced meat. Int. J. Biol. Macromol. 2020, 156, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Fierascu, I.; Fierascu, I.C.; Brazdis, R.I.; Baroi, A.M.; Fistos, T.; Fierascu, R.C. Phytosynthesized metallic nanoparticles—between nanomedicine and toxicology. A brief review of 2019′s findings. Materials 2020, 13, 574. [Google Scholar] [CrossRef]
- Sharma, V.; Kaushik, S.; Pandit, P.; Dhull, D.; Yadav, J.P.; Kaushik, S. Green synthesis of silver nanoparticles from medicinal plants and evaluation of their antiviral potential against chikungunya virus. Appl. Microbiol. Biotechnol. 2019, 103, 881–891. [Google Scholar] [CrossRef]
- Sundararajan, B.; Kumari, B.R. Novel synthesis of gold nanoparticles using Artemisia vulgaris L. leaf extract and their efficacy of larvicidal activity against dengue fever vector Aedes aegypti L. J. Trace Elem. Med. Biol. 2017, 43, 187–196. [Google Scholar] [CrossRef]
- Chandra, H.; Patel, D.; Kumari, P.; Jangwan, J.S.; Yadav, S. Phytomediated synthesis of zinc oxide nanoparticle of Berberis aristata: Characterisation, antioxidant activity and antibacterial activity with special reference to urinary tract infection. Mater. Sci. Eng. C 2019, 102, 212–220. [Google Scholar] [CrossRef]
- Fierascu, R.C.; Fierascu, I.; Lungulescu, E.M.; Nicula, N.; Somoghi, R.; Diţu, L.M.; Ungureanu, C.; Sutan, A.N.; Drăghiceanu, O.A.; Paunescu, A.; et al. Phytosynthesis and radiation-assisted methods for obtaining metal nanoparticles. J. Mater. Sci. 2020, 55, 1915–1932. [Google Scholar] [CrossRef]
- Wu, H.; Yin, J.J.; Wamer, W.G.; Zeng, M.; Lo, Y.M. Reactive oxygen species-related activities of nano-iron metal and nano-iron oxides. J. Food Drug Anal. 2014, 22, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Opris, R.; Tatomir, C.; Olteanu, D.; Moldovan, R.; Moldovan, B.; David, L.; Nagy, A.; Decea, N.; Kiss, M.L.; Filip, A.G. The effect of Sambucus nigra L. extract and phytosinthesized gold nanoparticles on diabetic rats. Colloids Surf. B Biointerfaces 2017, 150, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Bhardwaj, K.; Nepovimova, E.; Kuča, K.; Singh Dhanjal, D.; Bhardwaj, S.; Bhatia, S.K.; Verma, R.; Kumar, D. Antioxidant functionalized nanoparticles: A combat against oxidative stress. Nanomaterials 2020, 10, 1334. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, R.; Samadder, S.R. Aquatic and terrestrial weed mediated synthesis of iron nanoparticles for possible application in wastewater remediation. J. Clean. Prod. 2017, 168, 1201–1210. [Google Scholar] [CrossRef]
- Yuan, M.; Fu, X.; Yu, J.; Xu, Y.; Huang, J.; Lia, Q.; Sun, D. Green synthesized iron nanoparticles as highly efficient fenton-like catalyst for degradation of dyes. Chemosphere 2020, 261, 127618. [Google Scholar] [CrossRef] [PubMed]
- Alabi, M.A.; Muthusamy, A.; Kabekkodu, S.P.; Adebawo, O.O.; Satyamoorthy, K. Anticancer properties of recipes derived from nigeria and african medicinal plants on breast cancer cells in vitro. Sci. Afr. 2020, 8, e00446. [Google Scholar] [CrossRef]
- Dzah, C.S.; Duan, Y.; Zhang, H.; Adwo, N.; Boateng, S.; Ma, H. Latest developments in polyphenol recovery and purification from plant by-products: A review. Trends Food Sci. Technol. 2020, 99, 375–388. [Google Scholar] [CrossRef]
- Amin, F.U.; Hoshiar, A.K.; Do, T.D.; Noh, Y.; Shah, S.A.; Khan, M.S.; Yoon, J.; Kim, M.O. Osmotin-loaded magnetic nanoparticles with electromagnetic guidance for the treatment of Alzheimer’s disease. Nanoscale 2017, 9, 10619–10632. [Google Scholar] [CrossRef]
- Bilal, M.; Barani, M.; Sabir, F.; Rahdar, A.; Kyzas, G.Z. Nanomaterials for the treatment and diagnosis of Alzheimer’s disease: An overview. NanoImpact 2020, 20, 100251. [Google Scholar] [CrossRef]
| Plant | Extraction Method | Extraction Conditions | Obtained Compounds | Extraction Yield | Reference |
|---|---|---|---|---|---|
| Allium sativum Linn. | Microwave-assisted hydro-distillation | Solvent: deionized water/diethyl ether 2:1; 100 g vegetal material; MP = 700 W; t = 30 min; | Diallyl sulfides (mono-, di-, tri-, and tetra-); Methyl allyl sulfides (di- and tri-); Vinyl dithiins | 0.22% | [22] |
| Ultrasound-assisted extraction | Solvent: diethyl ether (50 mL); F = 35 kHz; T = 25 °C; t = 30 min. | 0.13% | |||
| Lickens–Nickerson apparatus | Solvent: water/diethyl ether = 1:10; 100 g. vegetal material; T = −10 °C; t = 2 h. | 0.23% | |||
| Hippophae rhamnoides L. | Solvent-free microwave-assisted extraction | 400 g vegetal material atmospheric pressure; P = 400 W; T = 20–100 °C; t = 15 min. | Polyphenols with an increased yield of recovery for microwave extraction method | 1147 mg GAE/g (d.w.) | [23] |
| Classical extraction | Solvent: methanol 80% (50 mL); 5 g vegetal material; 8000 rpm; t = 5 min. | 741.9 mg GAE/g (d.w.) | |||
| Matricaria chamomilla L. | Subcritical water extraction | Solvent: water (300 mL); 10 g vegetal material; P = 30, 45 and 60 bars; T = 100 °C; t = 30 min; | Polyphenols | 127–3226 mg/kg | [24] |
| Maceration | Solvent: water (100 mL); 2.5 g vegetal material—oven-dried chamomile at low temperatures (i.e., 40 °C); T = 100 °C; t = 120 min. | Polyphenols | 19.7 ± 0.5 mg/g (d.w.) | [25] | |
| Mentha spp. | Microwave hydro- diffusion | Solvent-free; 500 g vegetal material; MP 1 W/g; F 2.45 GHz t = 20 min. | Essential oil | 0.95% | [26] |
| Soxhlet extraction | Solvent: water: ethanol = 3:7 (250 mL); 1.5 g dry plant material; T = 95 °C | Polyphenols | 18,381–87,024 mg GAE/kg (d.w.) | [27] | |
| Origanum vulgare L., 1753 | Supercritical extraction | CO2 flow rate = 2.4 kg/h; 0.6 kg of vegetal material: CO2/plant ratio = 20 kg/kg; P = 30 MPa; T = 40 °C. | Carnosic acid | 3.18 ± 0.40% | [28] |
| Hydro-distillation | 300 g vegetal material; t = 45 min | Essential oil rich in terpenes | 0.75% (d.w.) | [29] | |
| Rosmarinus officinalis L. | Maceration | Solvent: dichloromethane/ethanol = 3/1 (15 mL); 1 g vegetal material; T = 35 °C; t 3 h. | Carnosic acid, rosmarinic acid, carnosol | 16.82; 0.12; 9.31 mg/g (f.w.) | [30] |
| Supercritical fluid extraction | Solvent-free CO2 extraction; flow rate: 5 g/min; 100 g vegetal material; P = 100–300 bar; T = 40 °C; t = 3 h. | Carnosic acid, rosmarinic acid, camphor, 1,8-cineole | 1.0730; 0.1242; 0.44; 0.029% (d.w.) | [31] | |
| Microwave-assisted extraction | Solvent: ethanol 96 %; 25 g milled leaves; Liquid/solid ratio = 6/1 (v/w); t = 7 min. | Carnosic acid, rosmarinic acid | 3.3 ± 0.2 % (w/v) 3.1 ± 1.2 % (w/v) | [32] | |
| Salvia officinalis L. | Supercritical CO2 extraction | Solvent-free CO2 extraction; flow rate: 1–3 kg/h; 50 g. of vegetal material; P = 15 or 20 MPa; T = 25 °C; t = 90 min. | Terpenes and phenolic compounds | 0.659–5.477 % (w/v) | [33] |
| Hydro distillation (Clevenger-type apparatus) | Solvent: water (1 L); 100 g vegetal material; t = 180 min. | Terpenes and phenolic compounds | 2.0–2.1% (v/w) | [34] | |
| Maceration | Solvent: ethanol (70%)—25 mL; 5 g of vegetal material; t = 2 days; | Rosmarinic acid, carnosic acid, carnosol and methyl carnosate | n.p. | [35] | |
| Satureja hortensis L. | Maceration | Solvent: ethanol 96% (300 mL); 10 g vegetal material; T = 22 °C; t = 7 days. | Phenolic compounds | 125.34 mg GAE/g | [36] |
| Soxhlet extraction | Solvent: ethanol 96% (600 mL); 75 g vegetal material; t = 8 h. | 119.28 mg GAE/g | |||
| Microwave extraction | Solvent: ethanol 96% (100 mL); 5 g vegetal material; t = 30 min | 147.21 mg GAE/g | |||
| Thymus daenensis Celak. and Thymus kotschyanus Boiss. and Hohen | Hydro-distillation (Clevenger-type apparatus) | 50 g vegetal material; t = 3 h. | Thymol, p-cymene, β-caryophyllene methyl carvacrol | 1.2–2.4% | [37] |
| Thymus munbyanus Boiss. & Reut., 1852 | Pressurized liquid extraction | Solvent: acetone; ethanol; water; 20 g. vegetal material; P = 45 MPa; T = 70 °C; t = 10 min | Oxygenated monoterpenoids; sesquiterpenoids and monoterpenoids | 21.2 ± 0.6% | [38] |
| Plant | Bioactive Compounds | Presentation Form | Activity | Industrial Application | Reference |
|---|---|---|---|---|---|
| Artemisia absinthium L., Calendula officinalis L., Lavandula vera DC, Syringa vulgaris L. | Water-soluble vitamins | Emulsion | Antioxidant activity | Cosmetic industry | [117] |
| Cinnamon | Essential oil | Starch based edible film | Antibacterial activity (Escherichia coli, Salmonella typhimurium and Staphylococcus aureus) | Natural packaging | [128] |
| Eryngium campestre L. | Essential oil | Chitosan nanoparticle | - | Prolonging shelf life of sweet cherries | [107] |
| Polylophium involucratum (Pall.) Boiss. | Essential oil | Poly lactic acid/ nanochitosan composite film | Antimicrobial activity (Pseudomonas spp.) | Prolonging shelf life of chicken fillet | [109] |
| Psiadia terebinthina A.J. Scott | Essential oil | - | Melanin inhibition | Cosmetic industry | [120] |
| Salvia miltiorrhiza Bunge | Polysaccharide | - | Increase the number of leukocytes in blood | Increase growth performance of broilers | [129] |
| Salvia rosmarinus Spenn. | Aqueous extract | Whey protein concentrate/carboxymethyl cellulose/glycerol coatings | - | Coatings for sun flower seeds | [111] |
| Satureja montana L. | Essential oil | - | Protein oxidative stability Lipid oxidative stability | Prolonging shelf life of pre-cooked pork chops | [130] |
| Thymus alternans Klokov and Teucrium montanum subsp. Jailae | Essential oil | - | Insecticide (Musca domestica L., Culex quinquefasciatus Say and Spodoptera littoralis (Boisd.)) | Natural insecticide | [113] |
| Thymus capitatus (L.) Hoffmanns et Link | Essential oil | Chitosan coatings | Antibacterial activity (Aerobic mesophylls, molds and yeasts) | Prolonged up to 1 day shelf life of strawberries stored under refrigeration conditions (5 ± 0.5 °C) | [105] |
| Thymus capitatus (L.) Hoffmanns et Link | Essential oil | Nano-emulsion | Antibacterial activity (Staphylococcus aureus) | Food preservative | [106] |
| Thymus kotschyanus Boiss. & Hohen. | Essential oil | Chitosan–starch composite film | Antibacterial activity (Pseudomonas spp. and Listeria monocytogenes) | Prolonging shelf life of beef during storage on a period of 21 days at 4 °C | [108] |
| Thymus serpyllum L. | Essential oil | - | Antimicrobial activity (Escherichia coli, Salmonella typhimurium, Staphylococcus aureus and Pseudomonas aeruginosa) | Ground pork patty | [131] |
| - | Limonene, linalool, menthol, and thymol | Chitosan nanoparticles | Antimicrobial activity (Escherichia coli and Salmonella typhimurium) | Preservation of minced meat | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fierascu, R.C.; Fierascu, I.; Baroi, A.M.; Ortan, A. Selected Aspects Related to Medicinal and Aromatic Plants as Alternative Sources of Bioactive Compounds. Int. J. Mol. Sci. 2021, 22, 1521. https://doi.org/10.3390/ijms22041521
Fierascu RC, Fierascu I, Baroi AM, Ortan A. Selected Aspects Related to Medicinal and Aromatic Plants as Alternative Sources of Bioactive Compounds. International Journal of Molecular Sciences. 2021; 22(4):1521. https://doi.org/10.3390/ijms22041521
Chicago/Turabian StyleFierascu, Radu Claudiu, Irina Fierascu, Anda Maria Baroi, and Alina Ortan. 2021. "Selected Aspects Related to Medicinal and Aromatic Plants as Alternative Sources of Bioactive Compounds" International Journal of Molecular Sciences 22, no. 4: 1521. https://doi.org/10.3390/ijms22041521
APA StyleFierascu, R. C., Fierascu, I., Baroi, A. M., & Ortan, A. (2021). Selected Aspects Related to Medicinal and Aromatic Plants as Alternative Sources of Bioactive Compounds. International Journal of Molecular Sciences, 22(4), 1521. https://doi.org/10.3390/ijms22041521








