Transcriptome Sequencing in the Preoptic Region of Rat Dams Reveals a Role of Androgen Receptor in the Control of Maternal Behavior
Abstract
1. Introduction
2. Results
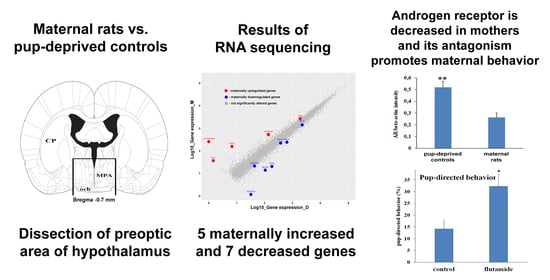
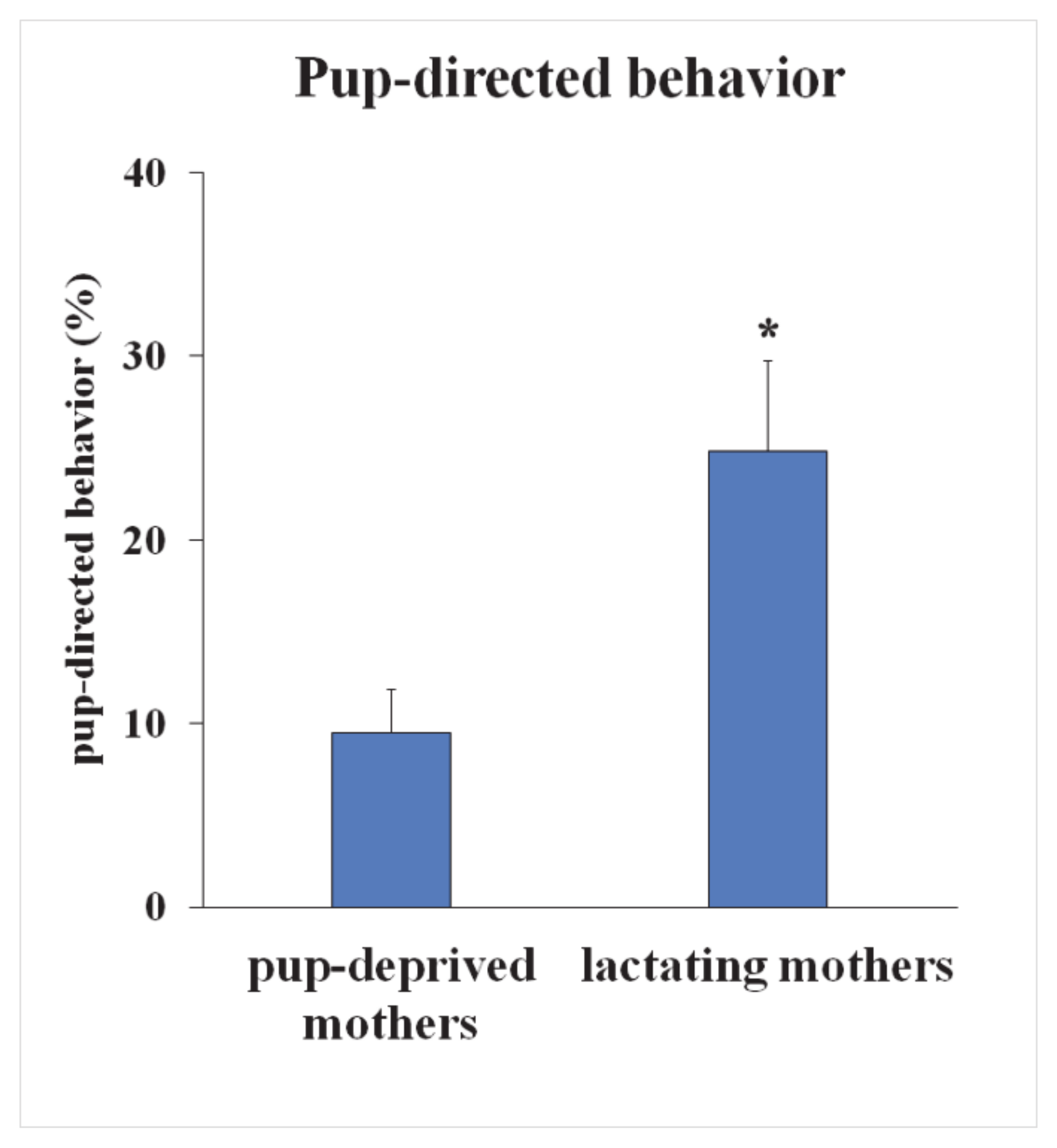
2.1. Pup-Deprived Mothers Showed Heavily Impaired Pup-Directed Behavior
2.2. Preoptic Transcriptome of Lactating and Pup-Deprived Mothers
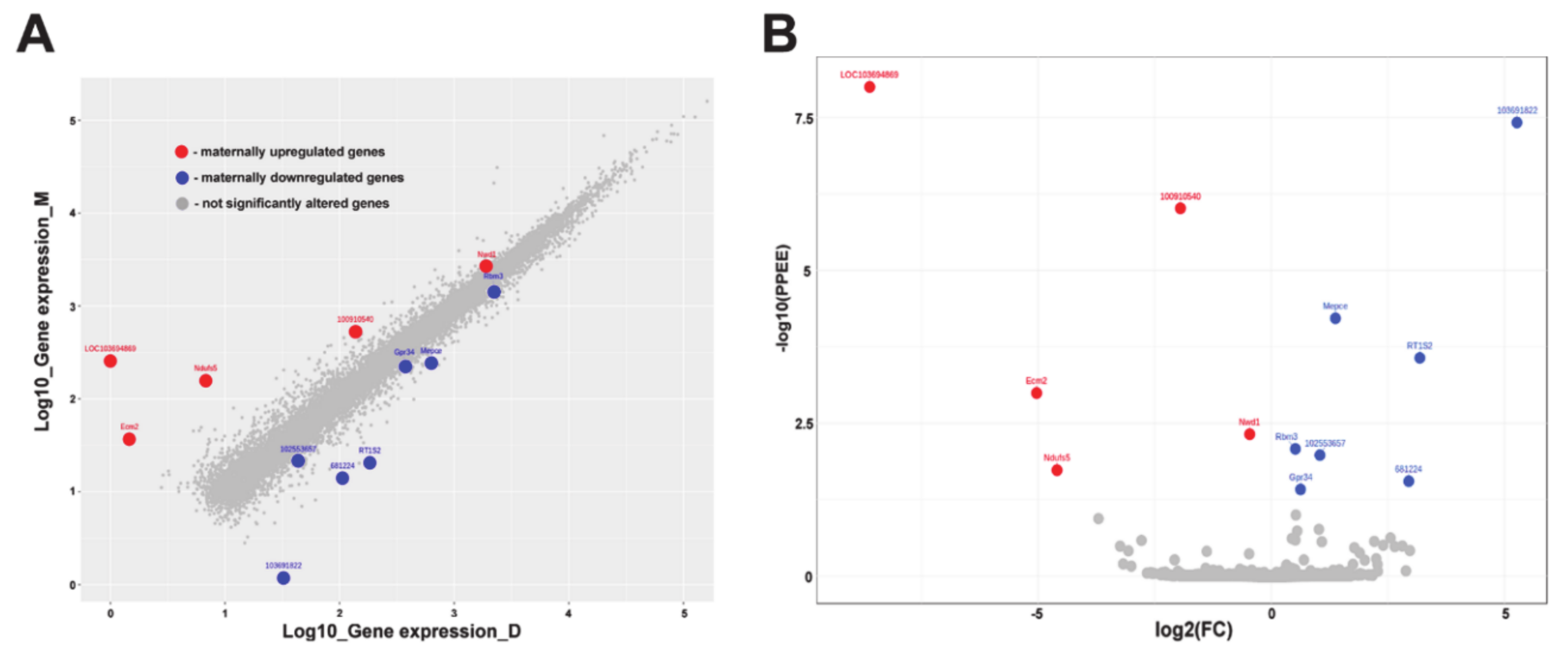
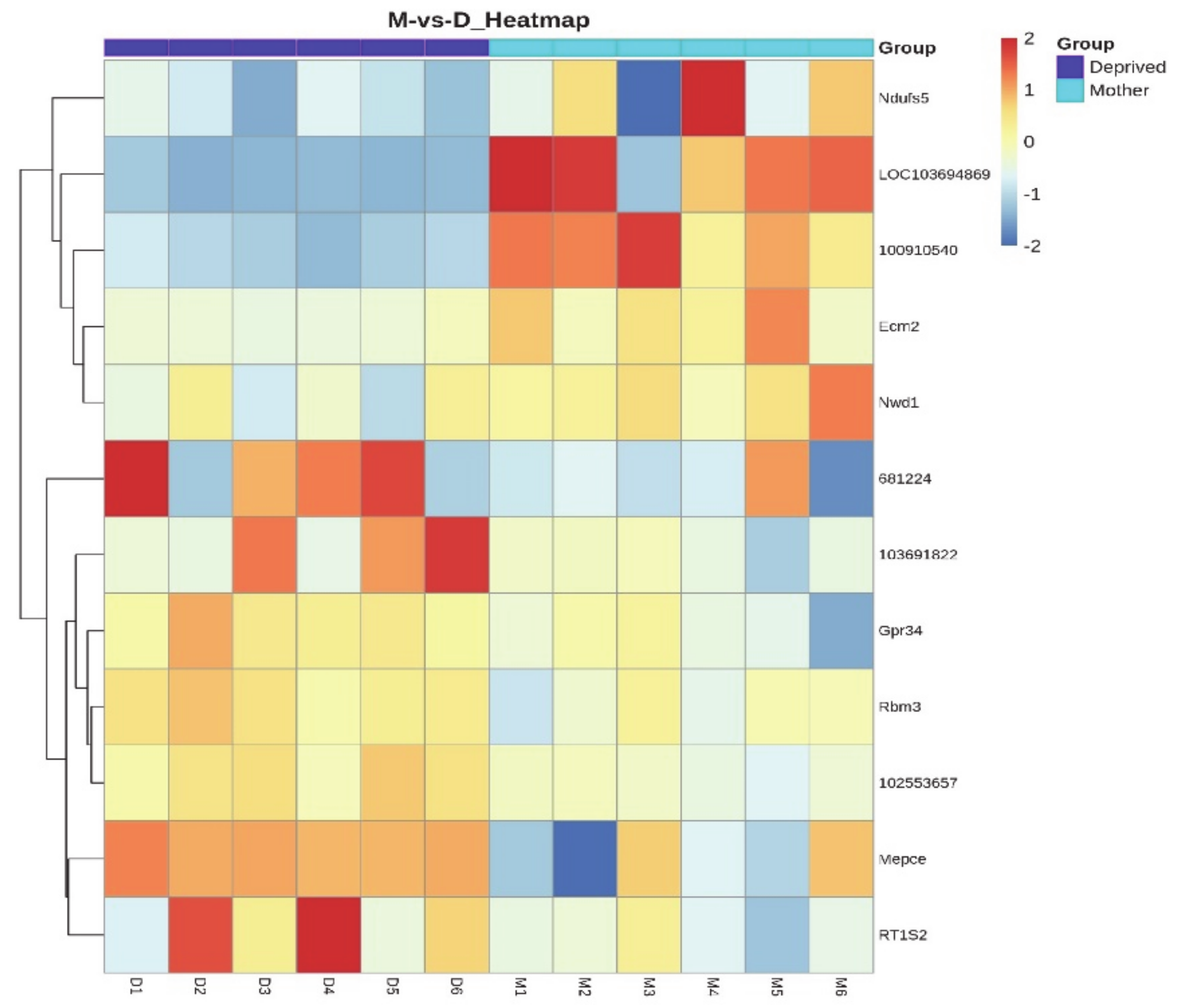
2.3. Differentially Expressed Genes in the MPOA of Lactating and Pup-Deprived Mothers
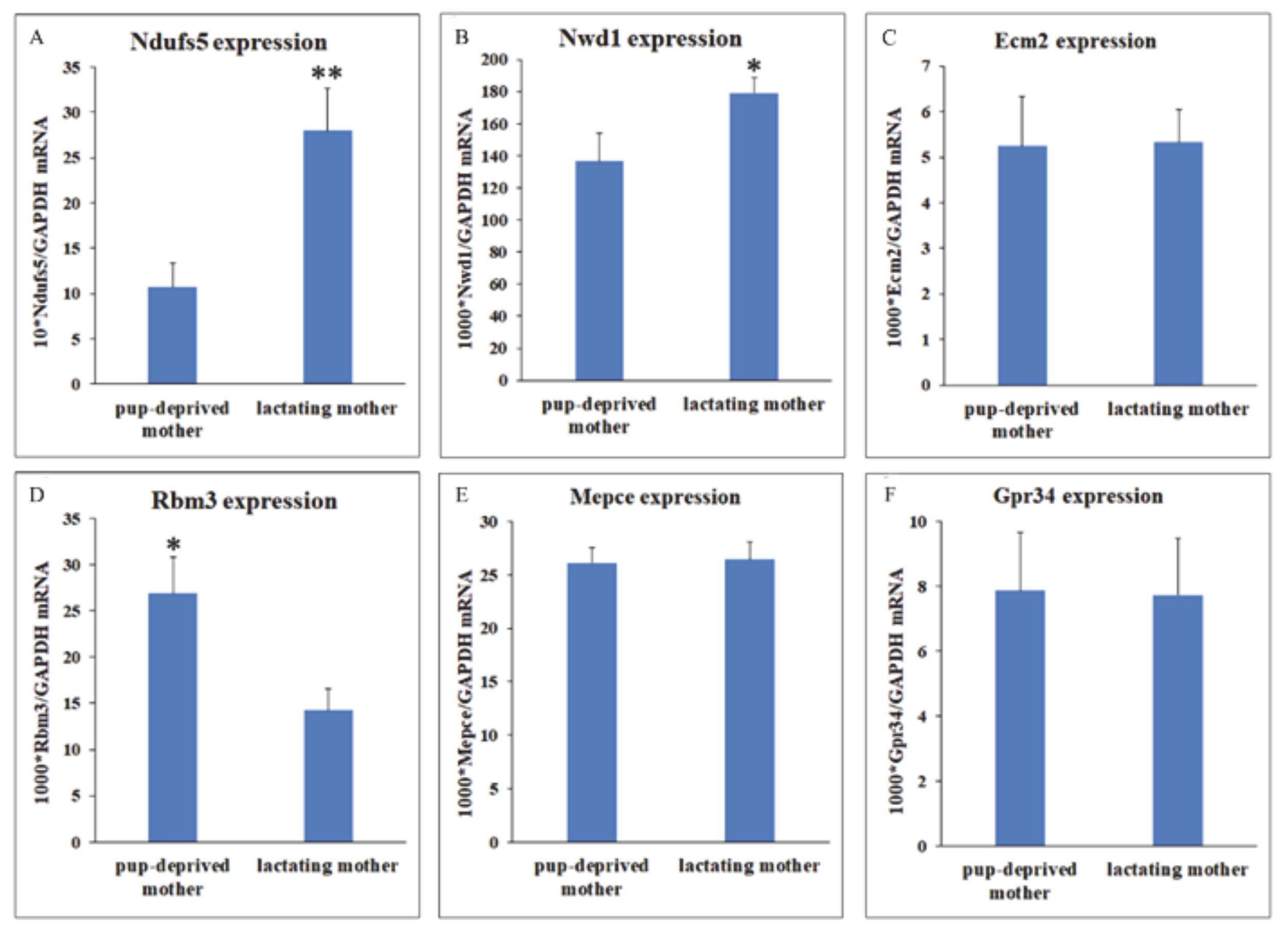
2.4. Validation of RNA-Seq
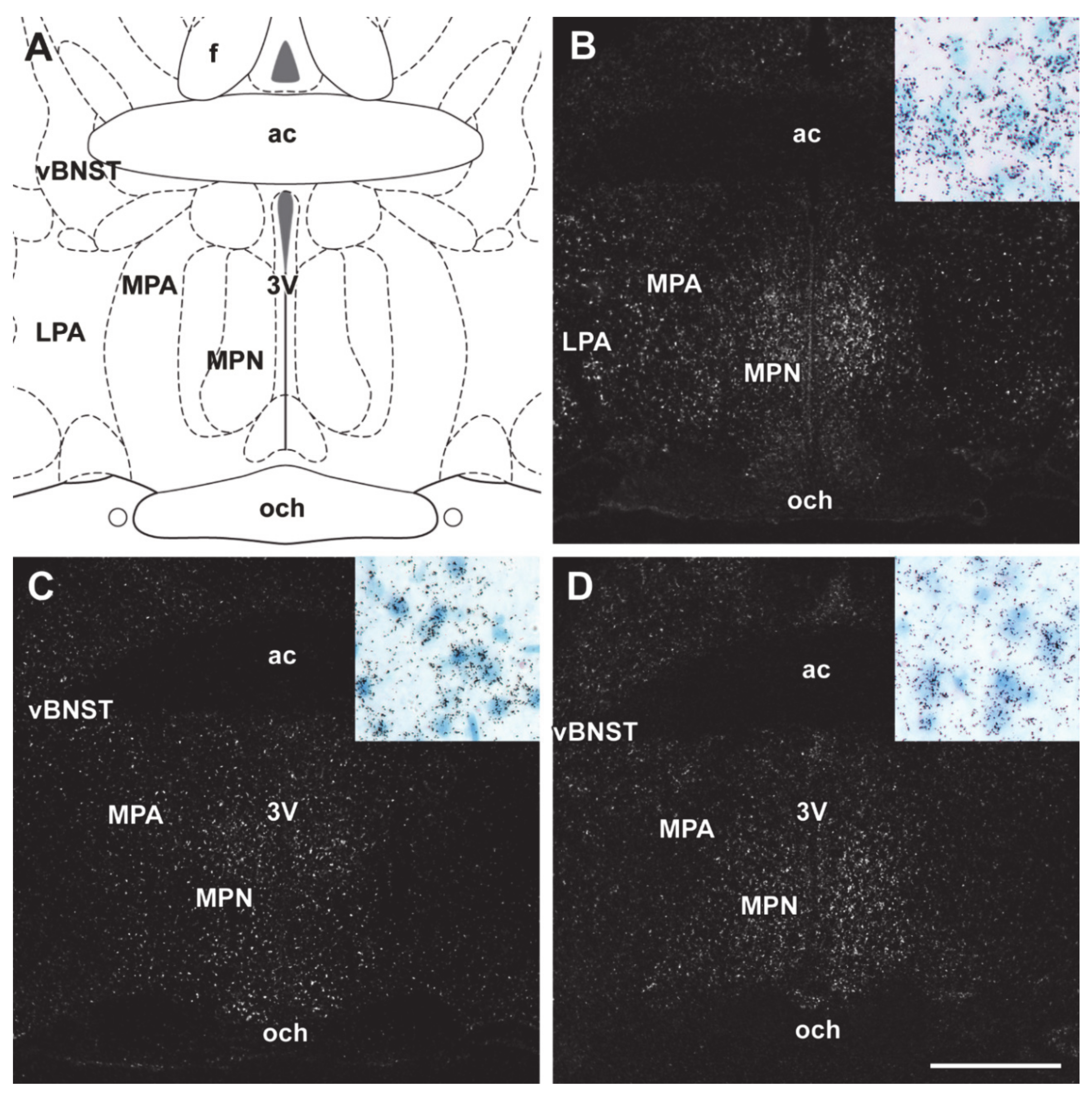
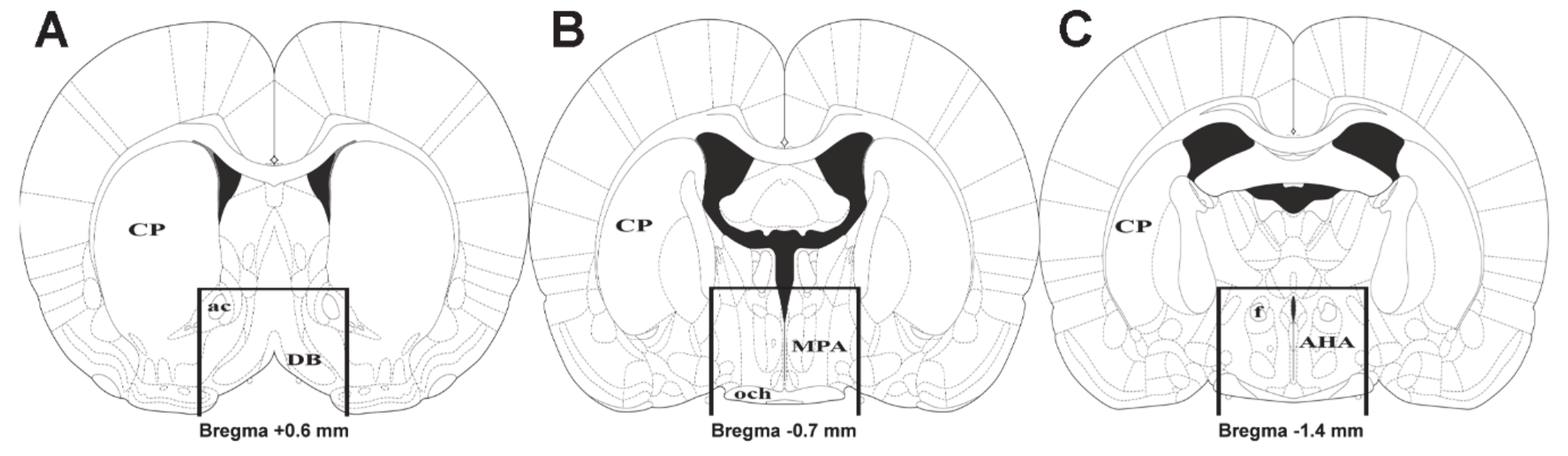
2.5. Distribution of Differentially Expressed Genes in the Preoptic Area
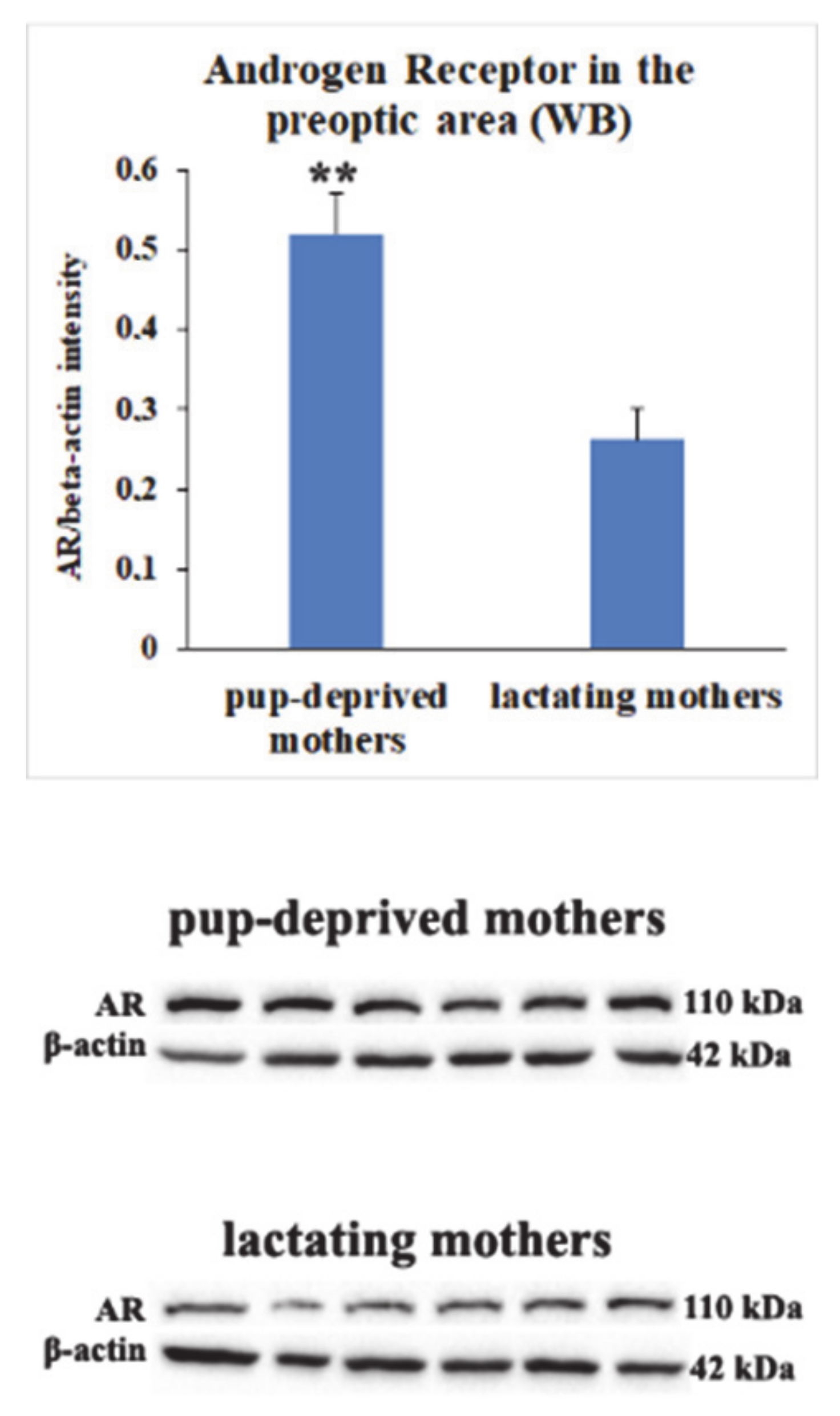
2.6. Decreased Androgen Receptor (AR) Protein Levels in MPOA of Lactating Mothers
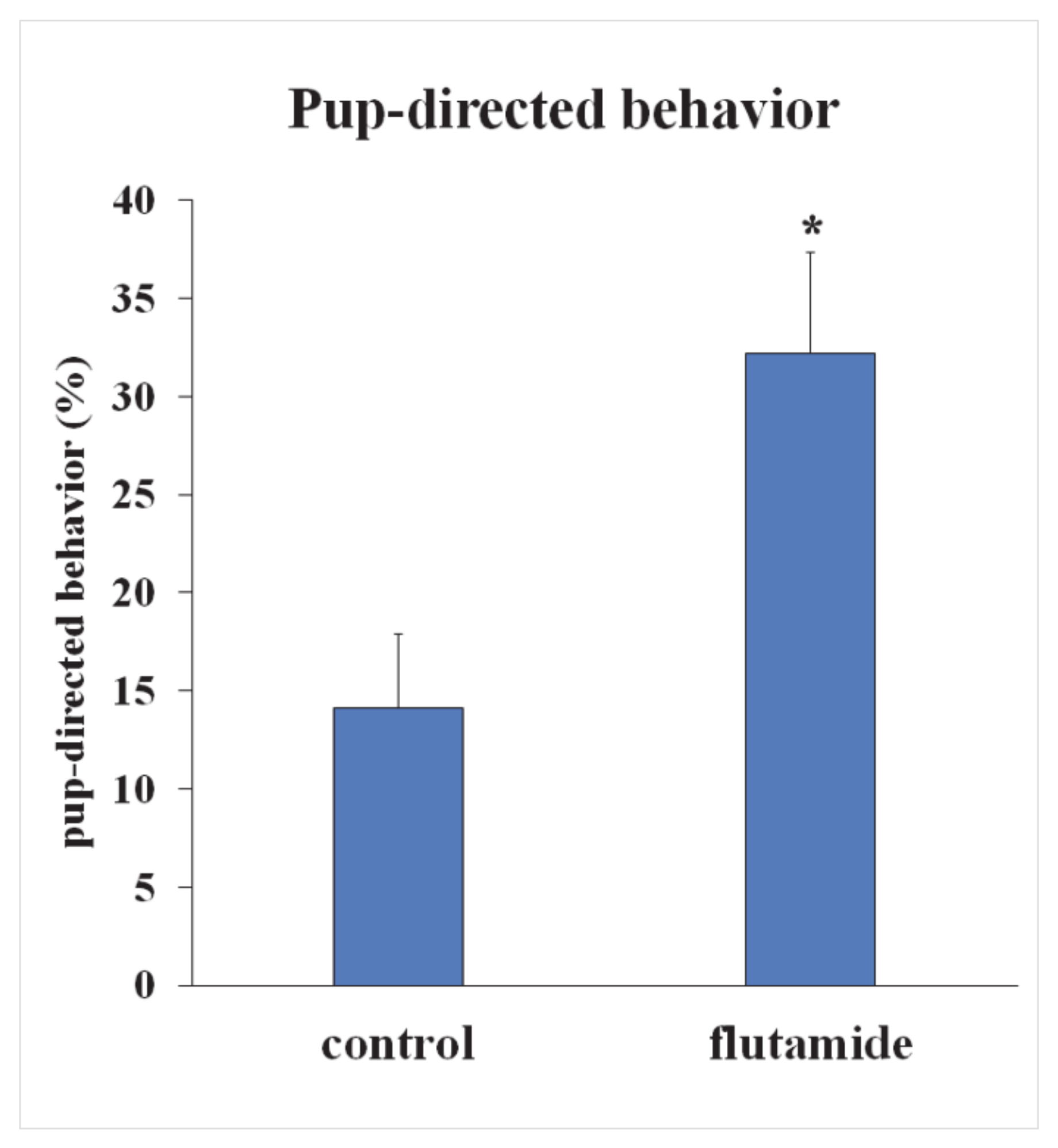
2.7. Intracerebroventricular (icv.) Administration of Androgen-Receptor Antagonist Flutamide Enhanced Pup-Directed Behavior
3. Discussion
3.1. Gene Expressional Changes in Mothers
3.2. DEGS in the MPOA of Lactating and Pup-Deprived Mothers
3.2.1. Ndufs5-NADH Dehydrogenase (Ubiquinone) Fe-S Protein 5
3.2.2. Nwd1-NACHT and WD Repeat Domain Containing Protein 1
3.2.3. Rbm3-RNA-Binding Motif Protein-3
3.3. Distribution of DEGS with the Preoptic Area
3.4. Androgen Receptor (AR) as a Possible Modulator of Central Maternal Adaptation
4. Materials and Methods
4.1. Animals
4.2. Analysis of Maternal Behavior
4.3. Microdissection of Brain Tissue Samples
4.4. RNA-Sequencing
4.4.1. Library Construction and Quality Check
4.4.2. Alignment and Quantification
4.5. Quantitiative RT-PCR
4.6. Production of Hybridization Probes for Ndufs5, Nwd1, and Rbm3
4.7. In Situ Hybridization Histochemistry
4.8. Androgen Receptor (AR) Immunohistochemistry
4.9. Western Blot Analysis
4.10. Implantation of Intracerebroventricular Cannulae
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ac | anterior commissure |
| ACTH | adrenocorticotropin |
| CP | caudatus putamen |
| Cx | cerebral cortex |
| DAB | 3,3-diaminobenzidine |
| f | fornix |
| FITC | fluorescein isothiocyanate |
| GAPDH | glyceraldehyde-3-phosphate-dehydrogenase |
| GnRH | gonadotropin releasing hormone |
| LH | gonadotropin releasing hormone |
| LS | lateral septal nucleus |
| LV | lateral ventricle |
| MPA | medial preoptic area |
| MPN | medial preoptic nucleus |
| och | optic chiasm |
| PB | phosphate buffer |
| PVN | paraventricular hypothalamic nucleus |
| vBNST | ventral subdivision of the bed nucleus of the stria terminalis |
| 3V | third ventricle |
References
- Dulac, C.; O’Connell, L.A.; Wu, Z. Neural control of maternal and paternal behaviors. Science 2014, 345, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Bridges, R.S. Neuroendocrine regulation of maternal behavior. Front. Neuroendocr. 2015, 36, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Ferreira, A. Neuroanatomical and neurochemical basis of parenting: Dynamic coordination of motivational, affective and cognitive processes. Horm. Behav. 2016, 77, 72–85. [Google Scholar] [CrossRef]
- Renier, N.; Adams, E.L.; Kirst, C.; Wu, Z.; Azevedo, R.; Kohl, J.; Autry, A.E.; Kadiri, L.; Umadevi Venkataraju, K.; Zhou, Y.; et al. Mapping of Brain Activity by Automated Volume Analysis of Immediate Early Genes. Cell 2016, 165, 1789–1802. [Google Scholar] [CrossRef] [PubMed]
- Numan, M.; Corodimas, K.P.; Numan, M.J.; Factor, E.M.; Piers, W.D. Axon-sparing lesions of the preoptic region and substantia innominata disrupt maternal behavior in rats. Behav. Neurosci. 1988, 102, 381–396. [Google Scholar] [CrossRef]
- Wu, Z.; Autry, A.E.; Bergan, J.F.; Watabe-Uchida, M.; Dulac, C.G. Galanin neurons in the medial preoptic area govern parental behaviour. Nature 2014, 509, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Numan, M.; Rosenblatt, J.S.; Komisaruk, B.R. Medial preoptic area and onset of maternal behavior in the rat. J. Comp. Physiol. Psychol. 1977, 91, 146–164. [Google Scholar] [CrossRef]
- Bridges, R.S.; Numan, M.; Ronsheim, P.M.; Mann, P.E.; Lupini, C.E. Central prolactin infusions stimulate maternal behavior in steroid-treated, nulliparous female rats. Proc. Natl. Acad. Sci. USA 1990, 87, 8003–8007. [Google Scholar] [CrossRef]
- Hashimoto, H.; Saito, T.R.; Furudate, S.; Takahashi, K.W. Prolactin levels and maternal behavior induced by ultrasonic vocalizations of the rat pup. Exp. Anim. 2001, 50, 307–312. [Google Scholar] [CrossRef]
- Okabe, S.; Nagasawa, M.; Kihara, T.; Kato, M.; Harada, T.; Koshida, N.; Mogi, K.; Kikusui, T. Pup odor and ultrasonic vocalizations synergistically stimulate maternal attention in mice. Behav. Neurosci. 2013, 127, 432–438. [Google Scholar] [CrossRef]
- Dobolyi, A. Central amylin expression and its induction in rat dams. J. Neurochem. 2009, 111, 1490–1500. [Google Scholar] [CrossRef] [PubMed]
- Driessen, T.M.; Eisinger, B.E.; Zhao, C.; Stevenson, S.A.; Saul, M.C.; Gammie, S.C. Genes showing altered expression in the medial preoptic area in the highly social maternal phenotype are related to autism and other disorders with social deficits. BMC Neurosci. 2014, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Akbari, E.M.; Shams, S.; Belay, H.T.; Kaiguo, M.; Razak, Z.; Kent, C.F.; Westwood, T.; Sokolowski, M.B.; Fleming, A.S. The effects of parity and maternal behavior on gene expression in the medial preoptic area and the medial amygdala in postpartum and virgin female rats: A microarray study. Behav. Neurosci. 2013, 127, 913–922. [Google Scholar] [CrossRef] [PubMed]
- Szabo, E.R.; Cservenak, M.; Dobolyi, A. Amylin is a novel neuropeptide with potential maternal functions in the rat. FASEB J. 2012, 26, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Orpen, B.G.; Fleming, A.S. Experience with pups sustains maternal responding in postpartum rats. Physiol. Behav. 1987, 40, 47–54. [Google Scholar] [CrossRef]
- Correa, R.G.; Krajewska, M.; Ware, C.F.; Gerlic, M.; Reed, J.C. The NLR-related protein NWD1 is associated with prostate cancer and modulates androgen receptor signaling. Oncotarget 2014, 5, 1666–1682. [Google Scholar] [CrossRef]
- Wang, T.A.; Teo, C.F.; Akerblom, M.; Chen, C.; Tynan-La Fontaine, M.; Greiner, V.J.; Diaz, A.; McManus, M.T.; Jan, Y.N.; Jan, L.Y. Thermoregulation via Temperature-Dependent PGD2 Production in Mouse Preoptic Area. Neuron 2019, 103, 349. [Google Scholar] [CrossRef]
- Kann, O.; Kovacs, R. Mitochondria and neuronal activity. Am. J. Physiol. Cell Physiol. 2007, 292, C641–C657. [Google Scholar] [CrossRef]
- Vyshkina, T.; Banisor, I.; Shugart, Y.Y.; Leist, T.P.; Kalman, B. Genetic variants of Complex I in multiple sclerosis. J. Neurol. Sci. 2005, 228, 55–64. [Google Scholar] [CrossRef]
- Seager, R.; Lee, L.; Henley, J.M.; Wilkinson, K.A. Mechanisms and roles of mitochondrial localisation and dynamics in neuronal function. Neuronal Signal. 2020, 4, NS20200008. [Google Scholar] [CrossRef]
- Volgyi, K.; Gulyassy, P.; Haden, K.; Kis, V.; Badics, K.; Kekesi, K.A.; Simor, A.; Gyorffy, B.; Toth, E.A.; Lubec, G.; et al. Synaptic mitochondria: A brain mitochondria cluster with a specific proteome. J. Proteom. 2015, 120, 142–157. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Ting, J.P. NLR, the nucleotide-binding domain leucine-rich repeat containing gene family. Curr. Opin. Immunol. 2008, 20, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Stein, C.; Caccamo, M.; Laird, G.; Leptin, M. Conservation and divergence of gene families encoding components of innate immune response systems in zebrafish. Genome Biol. 2007, 8, R251. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Sakakibara, S.I. Expression profile of the STAND protein Nwd1 in the developing and mature mouse central nervous system. J. Comp. Neurol. 2018, 526, 2099–2114. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Sato, A.; Sakakibara, S. Nwd1 Regulates Neuronal Differentiation and Migration through Purinosome Formation in the Developing Cerebral Cortex. Iscience 2020, 23, 45. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Huang, Z.F.; Luo, Y.F.; Zheng, F.S.; Hu, Y.D.; Liu, H.; Zhu, S.Z.; He, M.Q.; Xu, D.M.; Li, Y.; et al. Inhibition of Nwd1 activity attenuates neuronal hyperexcitability and GluN2B phosphorylation in the hippocampus. Ebiomedicine 2019, 47, 470–483. [Google Scholar] [CrossRef]
- Smart, F.; Aschrafi, A.; Atkins, A.; Owens, G.C.; Pilotte, J.; Cunningham, B.A.; Vanderklish, P.W. Two isoforms of the cold-inducible mRNA-binding protein RBM3 localize to dendrites and promote translation. J. Neurochem. 2007, 101, 1367–1379. [Google Scholar] [CrossRef]
- Peretti, D.; Bastide, A.; Radford, H.; Verity, N.; Molloy, C.; Martin, M.G.; Moreno, J.A.; Steinert, J.R.; Smith, T.; Dinsdale, D.; et al. RBM3 mediates structural plasticity and protective effects of cooling in neurodegeneration. Nature 2015, 518, 236–239. [Google Scholar] [CrossRef]
- Gellen, B.; Zelena, D.; Usdin, T.B.; Dobolyi, A. The parathyroid hormone 2 receptor participates in physiological and behavioral alterations of mother mice. Physiol. Behav. 2017, 181, 51–58. [Google Scholar] [CrossRef]
- Moffitt, J.R.; Bambah-Mukku, D.; Eichhorn, S.W.; Vaughn, E.; Shekhar, K.; Perez, J.D.; Rubinstein, N.D.; Hao, J.; Regev, A.; Dulac, C.; et al. Molecular, spatial, and functional single-cell profiling of the hypothalamic preoptic region. Science 2018, 362, 792. [Google Scholar] [CrossRef]
- Risold, P.Y.; Swanson, L.W. Chemoarchitecture of the rat lateral septal nucleus. Brain Res. Rev. 1997, 24, 91–113. [Google Scholar] [CrossRef]
- Gao, W.Q.; Bohl, C.E.; Dalton, J.T. Chemistry and structural biology of androgen receptor. Chem. Rev. 2005, 105, 3352–3370. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Weijdegard, B.; Wang, T.P.; Egecioglu, E.; Fernandez-Rodriguez, J.; Huhtaniemi, I.; Stener-Victorin, E.; Billig, H.; Shao, R.J. Spatiotemporal expression of androgen receptors in the female rat brain during the oestrous cycle and the impact of exogenous androgen administration: A comparison with gonadally intact males. Mol. Cell. Endocrinol. 2010, 321, 161–174. [Google Scholar] [CrossRef]
- Williamson, M.; Viau, V. Androgen receptor expressing neurons that project to the paraventricular nucleus of the hypothalamus in the male rat. J. Comp. Neurol. 2007, 503, 717–740. [Google Scholar] [CrossRef] [PubMed]
- Viau, V.; Meaney, M.J. The inhibitory effect of testosterone on hypothalamic-pituitary-adrenal responses to stress is mediated by the medial preoptic area. J. Neurosci. 1996, 16, 1866–1876. [Google Scholar] [CrossRef]
- Grattan, D.R.; Rocca, M.S.; Sagrillo, C.A.; McCarthy, M.M.; Selmanoff, M. Antiandrogen microimplants into the rostral medial preoptic area decrease gamma-aminobutyric acidergic neuronal activity and increase luteinizing hormone secretion in the intact male rat. Endocrinology 1996, 137, 4167–4173. [Google Scholar] [CrossRef]
- Foecking, E.M.; McDevitt, M.A.; Acosta-Martinez, M.; Horton, T.H.; Levine, J.E. Neuroendocrine consequences of androgen excess in female rodents. Horm. Behav. 2008, 53, 673–692. [Google Scholar] [CrossRef][Green Version]
- Diamantikandarakis, E.; Tolis, G.; Duleba, A.J. Androgens and therapeutic aspects of antiandrogens in women. J. Soc. Gynecol. Investig. 1995, 2, 577–592. [Google Scholar] [CrossRef]
- Mhaouty-Kodja, S. Role of the androgen receptor in the central nervous system. Mol. Cell. Endocrinol. 2018, 465, 103–112. [Google Scholar] [CrossRef]
- Dart, D.A.; Waxman, J.; Aboagye, E.O.; Bevan, C.L. Visualising androgen receptor activity in male and female mice. PLoS ONE 2013, 8, e71694. [Google Scholar] [CrossRef]
- Yasin, M.; Dalkin, A.C.; Haisenleder, D.J.; Marshall, J.C. Testosterone is required for gonadotropin-releasing hormone stimulation of luteinizing hormone-beta messenger ribonucleic acid expression in female rats. Endocrinology 1996, 137, 1265–1271. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dunlap, K.D.; Sridaran, R. Plasma-levels of dihydrotestosterone in the cycling rat—Implications for the regulation of lordosis behavior. Physiol. Behav. 1988, 42, 199–202. [Google Scholar] [CrossRef]
- Barkley, M.S.; Geschwind, I.I.; Bradford, G.E. The gestational pattern of estradiol, testosterone and progesterone secretion in selected strains of mice. Biol. Reprod. 1979, 20, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Kuzawa, C.W.; Gettler, L.T.; Huang, Y.Y.; McDade, T.W. Mothers have lower testosterone than non-mothers: Evidence from the Philippines. Horm. Behav. 2010, 57, 441–447. [Google Scholar] [CrossRef]
- Feng, Y.; Shao, R.J.; Weijdegard, B.; Wang, T.P.; Johansson, J.; Sun, S.; Wang, W.; Egecioglu, E.; Billig, H.; Stener-Victorin, E. Effects of androgen and leptin on behavioral and cellular responses in female rats. Horm. Behav. 2011, 60, 427–438. [Google Scholar] [CrossRef]
- Parizek, A.; Mikesova, M.; Jirak, R.; Hill, M.; Koucky, M.; Paskova, A.; Velikova, M.; Adamcova, K.; Sramkova, M.; Jandikova, H.; et al. Steroid Hormones in the Development of Postpartum Depression. Physiol. Res. 2014, 63, S277–S282. [Google Scholar] [CrossRef]
- Aswathi, A.; Rajendiren, S.; Nimesh, A.; Philip, R.R.; Kattimani, S.; Jayalakshmi, D.; Ananthanarayanan, P.H.; Dhiman, P. High serum testosterone levels during postpartum period are associated with postpartum depression. Asian J. Psychiatry 2015, 17, 85–88. [Google Scholar] [CrossRef]
- Marrs, C.R.; Ferraro, D.P.; Cross, C.L.; Rogers, S.L. A potential role for adrenal androgens in postpartum psychiatric distress. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 143, 127–128. [Google Scholar] [CrossRef]
- Olah, S.; Cservenak, M.; Keller, D.; Fazekas, E.A.; Renner, E.; Low, P.; Dobolyi, A. Prolactin-induced and neuronal activation in the brain of mother mice. Brain Struct. Funct. 2018, 223, 3229–3250. [Google Scholar] [CrossRef]
- Leko, A.H.; Cservenak, M.; Szabo, E.R.; Hanics, J.; Alpar, A.; Dobolyi, A. Insulin-like growth factor I and its binding protein-3 are regulators of lactation and maternal responsiveness. Sci. Rep. 2017, 7, 3396. [Google Scholar] [CrossRef]
- Leng, N.; Dawson, J.A.; Thomson, J.A.; Ruotti, V.; Rissman, A.I.; Smits, B.M.; Haag, J.D.; Gould, M.N.; Stewart, R.M.; Kendziorski, C. EBSeq: An empirical Bayes hierarchical model for inference in RNAseq experiments. Bioinformatics 2013, 29, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Vincze, C.; Pál, G.; Wappler, E.A.; Szabó, E.R.; Nagy, Z.G.; Lovas, G.; Dobolyi, A. Distribution of mRNAs encoding transforming growth factors-beta1, -2, and -3 in the intact rat brain and after experimentally induced focal ischemia. J. Comp. Neurol. 2010, 518, 3752–3770. [Google Scholar] [CrossRef] [PubMed]
- Udvari, E.B.; Völgyi, K.; Gulyássy, P.; Dimén, D.; Kis, V.; Barna, J.; Szabó, É.R.; Lubec, G.; Juhász, G.; Kékesi, K.A.; et al. Synaptic proteome changes in the hypothalamus of mother rats. J. Proteom. 2017, 159, 54–66. [Google Scholar] [CrossRef] [PubMed]

| Total Raw Reads (Mb) | Clean Reads (Mb) | Clean Reads Ratio (%) | Total Reads Mapping Ratio (%) | Uniquely Reads Mapping Ratio (%) | Total Gene Mapping Ratio (%) | |
|---|---|---|---|---|---|---|
| D1 | 57.27 | 52.8 | 92.19 | 75.12 | 61.22 | 57.25 |
| D2 | 58.08 | 54.54 | 93.9 | 74.14 | 60.16 | 58.19 |
| D3 | 51.25 | 48.8 | 95.23 | 94.28 | 76.39 | 70.65 |
| D4 | 60.63 | 57.1 | 94.17 | 93.51 | 76.47 | 72.67 |
| D5 | 62.55 | 59.42 | 95 | 74.3 | 60.26 | 58.78 |
| D6 | 61.46 | 58.47 | 95.14 | 74.43 | 60.17 | 57.58 |
| M1 | 102.26 | 96.28 | 94.15 | 94.69 | 77.19 | 75.08 |
| M2 | 90.68 | 87.18 | 96.14 | 94.52 | 76.76 | 74.15 |
| M3 | 92.07 | 87.73 | 95.28 | 93.71 | 76.05 | 72.68 |
| M4 | 120.24 | 114.73 | 95.42 | 94.58 | 76.41 | 75.51 |
| M5 | 100.04 | 96.01 | 95.97 | 94.58 | 76.04 | 75.19 |
| M6 | 60.36 | 57.6 | 95.42 | 73.27 | 59.06 | 56.96 |
| Gene ID | Unigen ID | Gene Symbol | Protein | Expression in Lactating Mothers | Expression in Pup-Deprived Mothers | Log2 (Lactating/Pup-Deprived) | PPEE | Function |
|---|---|---|---|---|---|---|---|---|
| 103694869 | Rn.1166. | LOC103694869 | Isochorismatase domain-containing protein 1 | 254.936 | 0,000 | 8.612 | 0 | Metabolic process |
| 100910978 | Rn.106785. | Ecm2 | Ecm2 (Extracellular matrix protein 2) | 35.792 | 0.46 | 6.281 | 0.00102 | Organization of extracellular matrix, collagen binding |
| 100363268 | Rn.156413. | Ndufs5 | NADH dehydrogenase (Ubiquinone) Fe-S protein 5 | 155.64 | 5.81 | 4.744 | 0.0186 | Accessory subunit of the mitochondrial membrane respiratory chain |
| 100910540 | - | LOC100910540 | - | 525.556 | 135.613 | 1.954 | 0.000000966 | - |
| 100364350 | - | Nwd1 | NACHT And WD Repeat Domain Containing 1 | 2644.637 | 1919.119 | 0.463 | 0.0048 | Control of androgen receptor (AR) protein steady-state levels. |
| 114488 | Rn.18057 | Rbm3 | RNA-binding protein 3 | 1530.588 | 2190.024 | -0.517 | 0.00834 | Regulation of translation, cold-inducible mRNA binding protein |
| 554353 | Rn.163444 | Gpr34 | G Protein-Coupled Receptor 34 | 235.54 | 364.333 | -0.629 | 0.0384 | G-protein coupled signal transduction |
| 102553657 | - | LOC102553657 | - | 20.299 | 42.477 | -1.065 | 0.0105 | - |
| 304361 | Rn.198943. | Mepce | Mepce protein (Methylphosphate Capping Enzyme) | 243.175 | 631.98 | -1.378 | 0.00006 | RNA-binding methyltransferase |
| 681224 | - | LOC681224 | - | 13.044 | 105.041 | -3.01 | 0.0281 | - |
| 24994 | Rn.222446 | RT1-S2 | RT1 Class 1 S2 | 19.498 | 182.707 | -3.228 | 0.0002 | Involved in the presentation of foreign antigens to the immune system. |
| 103691822 | - | LOC103691822 | - | 0.178 | 31.412 | -7.463 | 0.00000003 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lékó, A.H.; Kumari, R.; Dóra, F.; Keller, D.; Udvari, E.B.; Csikós, V.; Renner, É.; Dobolyi, A. Transcriptome Sequencing in the Preoptic Region of Rat Dams Reveals a Role of Androgen Receptor in the Control of Maternal Behavior. Int. J. Mol. Sci. 2021, 22, 1517. https://doi.org/10.3390/ijms22041517
Lékó AH, Kumari R, Dóra F, Keller D, Udvari EB, Csikós V, Renner É, Dobolyi A. Transcriptome Sequencing in the Preoptic Region of Rat Dams Reveals a Role of Androgen Receptor in the Control of Maternal Behavior. International Journal of Molecular Sciences. 2021; 22(4):1517. https://doi.org/10.3390/ijms22041517
Chicago/Turabian StyleLékó, András H., Rashmi Kumari, Fanni Dóra, Dávid Keller, Edina B. Udvari, Vivien Csikós, Éva Renner, and Arpád Dobolyi. 2021. "Transcriptome Sequencing in the Preoptic Region of Rat Dams Reveals a Role of Androgen Receptor in the Control of Maternal Behavior" International Journal of Molecular Sciences 22, no. 4: 1517. https://doi.org/10.3390/ijms22041517
APA StyleLékó, A. H., Kumari, R., Dóra, F., Keller, D., Udvari, E. B., Csikós, V., Renner, É., & Dobolyi, A. (2021). Transcriptome Sequencing in the Preoptic Region of Rat Dams Reveals a Role of Androgen Receptor in the Control of Maternal Behavior. International Journal of Molecular Sciences, 22(4), 1517. https://doi.org/10.3390/ijms22041517