The Protective Role of Sestrin2 in Atherosclerotic and Cardiac Diseases
Abstract
1. Introduction
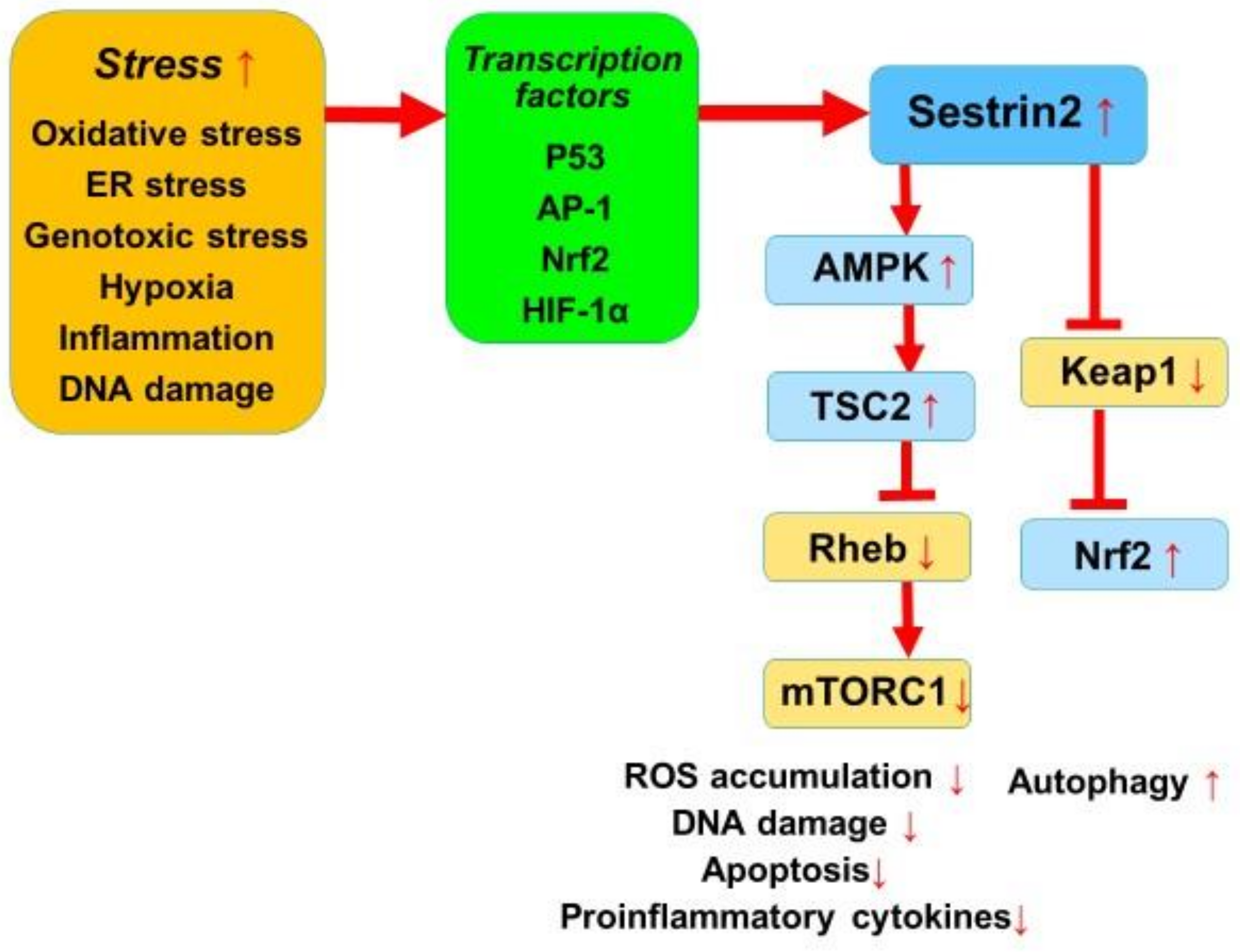
2. Physiopathological Mechanisms of Sestrin2
2.1. Sestrin2 Signaling Pathways
2.2. Sestrin2 and Oxidative Stress
2.3. Sestrin2 and Inflammation
3. Sestrin2 and Cardiac Diseases
3.1. Aging and Myocardial Infarction
3.2. Cardiomyopathy
4. Sestrin2 and Atherosclerotic Diseases
4.1. Coronary Artery Disease
4.2. Carotid Atherosclerosis
4.3. Miscellaneous
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative stress in atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef] [PubMed]
- Cervantes Gracia, K.; Llanas-Cornejo, D.; Husi, H. CVD and oxidative stress. J. Clin. Med. 2017, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Budanov, A.V.; Shoshani, T.; Faerman, A.; Zelin, E.; Kamer, I.; Kalinski, H.; Gorodin, S.; Fishman, A.; Chajut, A.; Einat, P.; et al. Identification of a novel stress-responsive gene Hi95 involved in regulation of cell viability. Oncogene 2002, 21, 6017–6031. [Google Scholar] [CrossRef] [PubMed]
- Budanov, A.V.; Sablina, A.A.; Feinstein, E.; Koonin, E.V.; Chumakov, P.M. Regeneration of peroxiredoxins by p53-regulated sestrins, homologs of bacterial AhpD. Science 2004, 304, 596–600. [Google Scholar] [CrossRef]
- Budanov, A.V.; Lee, J.H.; Karin, M. Stressin’ Sestrins take an aging fight. EMBO Mol. Med. 2010, 2, 388–400. [Google Scholar] [CrossRef]
- Wang, H.; Li, N.; Shao, X.; Li, J.; Guo, L.; Yu, X.; Sun, Y.; Hao, J.; Niu, H.; Xiang, J.; et al. Increased plasma sestrin2 concentrations in patients with chronic heart failure and predicted the occurrence of major adverse cardiac events: A 36-month follow-up cohort study. Clin. Chim. Acta 2019, 495, 338–344. [Google Scholar] [CrossRef]
- Wang, L.X.; Zhu, X.M.; Yao, Y.M. Sestrin2: Its potential role and regulatory mechanism in host immune response in diseases. Front. Immunol. 2019, 10, 2797. [Google Scholar] [CrossRef]
- Sun, W.; Wang, Y.; Zheng, Y.; Quan, N. The emerging role of sestrin2 in cell metabolism, and cardiovascular and age-related diseases. Aging Dis. 2020, 11, 154–163. [Google Scholar] [CrossRef]
- Hu, H.J.; Shi, Z.Y.; Lin, X.L.; Chen, S.M.; Wang, Q.Y.; Tang, S.Y. Upregulation of sestrin2 expression protects against macrophage apoptosis induced by oxidized low-density lipoprotein. DNA Cell Biol. 2015, 34, 296–302. [Google Scholar] [CrossRef]
- Kim, M.G.; Yang, J.H.; Kim, K.M.; Jang, C.H.; Jung, J.Y.; Cho, I.J.; Shin, S.M.; Ki, S.H. Regulation of toll-like receptor-mediated sestrin2 induction by AP-1, Nrf2, and the ubiquitin-proteasome system in macrophages. Toxicol. Sci. 2015, 144, 425–435. [Google Scholar] [CrossRef]
- Budanov, A.V.; Karin, M. p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell 2008, 134, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wu, Y.; Tang, S.; Liu, H.; Jiang, Y. Sestrin proteins in cardiovascular disease. Clin. Chim. Acta 2020, 508, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Pasha, M.; Eid, A.H.; Eid, A.A.; Gorin, Y.; Munusamy, S. Sestrin2 as a novel biomarker and therapeutic target for various diseases. Oxid. Med. Cell Longev. 2017, 2017, 3296294. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.H.; Sung, S.H.; Oh, S.Y.; Lim, J.M.; Lee, S.K.; Park, Y.N.; Lee, H.E.; Kang, D.; Rhee, S.G. Sestrins activate Nrf2 by promoting p62-dependent autophagic degradation of Keap1 and prevent oxidative liver damage. Cell Metab. 2013, 17, 73–84. [Google Scholar] [CrossRef]
- Alexander, A.; Cai, S.L.; Kim, J.; Nanez, A.; Sahin, M.; MacLean, K.H.; Inoki, K.; Guan, K.L.; Shen, J.; Person, M.D.; et al. ATM signals to TSC2 in the cytoplasm to regulate mTORC1 in response to ROS. Proc. Natl. Acad. Sci. USA 2010, 107, 4153–4158. [Google Scholar] [CrossRef]
- Wullschleger, S.; Loewith, R.; Hall, M.N. TOR signaling in growth and metabolism. Cell 2006, 124, 471–484. [Google Scholar] [CrossRef]
- Kishton, R.J.; Barnes, C.E.; Nichols, A.G.; Cohen, S.; Gerriets, V.A.; Siska, P.J.; Macintyre, A.N.; Goraksha-Hicks, P.; de Cubas, A.A.; Liu, T.; et al. AMPK is essential to balance glycolysis and mitochondrial metabolism to control T-ALL cell stress and survival. Cell Metab. 2016, 23, 649–662. [Google Scholar] [CrossRef]
- Hosokawa, N.; Hara, T.; Kaizuka, T.; Kishi, C.; Takamura, A.; Miura, Y.; Iemura, S.; Natsume, T.; Takehana, K.; Yamada, N.; et al. Nutrient-dependent mTORC1 association with the ULK1-Atg13-FIP200 complex required for autophagy. Mol. Biol. Cell 2009, 20, 1981–1991. [Google Scholar] [CrossRef]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef]
- Yang, Y.; Cuevas, S.; Yang, S.; Villar, V.A.; Escano, C.; Asico, L.; Yu, P.; Jiang, X.; Weinman, E.J.; Armando, I.; et al. Sestrin2 decreases renal oxidative stress, lowers blood pressure, and mediates dopamine D2 receptor-induced inhibition of reactive oxygen species production. Hypertension 2014, 64, 825–832. [Google Scholar] [CrossRef]
- Wang, L.X.; Zhu, X.M.; Luo, Y.N.; Wu, Y.; Dong, N.; Tong, Y.L.; Yao, Y.M. Sestrin2 protects dendritic cells against endoplasmic reticulum stress-related apoptosis induced by high mobility group box-1 protein. Cell Death Dis. 2020, 11, 125. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Frenis, K.; Oelze, M.; Kalinovic, S.; Kuntic, M.; Bayo Jimenez, M.T.; Vujacic-Mirski, K.; Helmstädter, J.; Kröller-Schön, S.; Münzel, T.; et al. Vascular inflammation and oxidative stress: Major triggers for cardiovascular disease. Oxid. Med. Cell Longev. 2019, 2019, 7092151. [Google Scholar] [CrossRef] [PubMed]
- Griendling, K.K.; FitzGerald, G.A. Oxidative stress and cardiovascular injury: Part I: Basic mechanisms and in vivo monitoring of ROS. Circulation 2003, 108, 1912–1916. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.H.; Kim, K.M.; Kim, M.G.; Seo, K.H.; Han, J.Y.; Ka, S.O.; Park, B.H.; Shin, S.M.; Ku, S.K.; Cho, I.J.; et al. Role of sestrin2 in the regulation of proinflammatory signaling in macrophages. Free Radic. Biol. Med. 2015, 78, 156–167. [Google Scholar] [CrossRef]
- Hwang, H.J.; Jung, T.W.; Choi, J.H.; Lee, H.J.; Chung, H.S.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Choi, K.M.; Choi, D.S.; et al. Knockdown of sestrin2 increases pro-inflammatory reactions and ER stress in the endothelium via an AMPK dependent mechanism. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1436–1444. [Google Scholar] [CrossRef]
- Li, D.; Wang, D.; Wang, Y.; Ling, W.; Feng, X.; Xia, M. Adenosine monophosphate-activated protein kinase induces cholesterol efflux from macrophage-derived foam cells and alleviates atherosclerosis in apolipoprotein E-deficient mice. J. Biol. Chem. 2010, 285, 33499–33509. [Google Scholar] [CrossRef]
- Brasier, A.R.; Recinos, A., 3rd; Eledrisi, M.S. Vascular inflammation and the renin-angiotensin system. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1257–1266. [Google Scholar] [CrossRef]
- Yi, L.; Li, F.; Yong, Y.; Jianting, D.; Liting, Z.; Xuansheng, H.; Fei, L.; Jiewen, L. Upregulation of sestrin-2 expression protects against endothelial toxicity of angiotensin II. Cell Biol. Toxicol. 2014, 30, 147–156. [Google Scholar] [CrossRef]
- Martinet, W.; De Loof, H.; De Meyer, G.R.Y. mTOR inhibition: A promising strategy for stabilization of atherosclerotic plaques. Atherosclerosis 2014, 233, 601–607. [Google Scholar] [CrossRef]
- Kurdi, A.; De Meyer, G.R.; Martinet, W. Potential therapeutic effects of mTOR inhibition in atherosclerosis. Br. J. Clin. Pharmacol. 2016, 82, 1267–1279. [Google Scholar] [CrossRef]
- Lee, J.H.; Budanov, A.V.; Talukdar, S.; Park, E.J.; Park, H.L.; Park, H.W.; Bandyopadhyay, G.; Li, N.; Aghajan, M.; Jang, I.; et al. Maintenance of metabolic homeostasis by Sestrin2 and Sestrin3. Cell Metab. 2012, 16, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Howell, J.J.; Ricoult, S.J.; Ben-Sahra, I.; Manning, B.D. A growing role for mTOR in promoting anabolic metabolism. Biochem. Soc. Trans. 2013, 41, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Sundararajan, S.; Jayachandran, I.; Balasubramanyam, M.; Mohan, V.; Venkatesan, B.; Manickam, N. Sestrin2 regulates monocyte activation through AMPK-mTOR nexus under high-glucose and dyslipidemic conditions. J. Cell Biochem. 2018, 10, 1002. [Google Scholar] [CrossRef]
- Gostner, J.M.; Fuchs, D. Biomarkers for the role of macrophages in the development and progression of atherosclerosis. Atherosclerosis 2016, 255, 117–118. [Google Scholar] [CrossRef] [PubMed]
- Mair, W.; Morantte, I.; Rodrigues, A.P.; Manning, G.; Montminy, M.; Shaw, R.J.; Dillin, A. Lifespan extension induced by AMPK and calcineurin is mediated by CRTC-1 and CREB. Nature 2011, 470, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Xue, R.; Sun, Y.; Dong, Y.; Liu, C. Sestrin 2 attenuates neonatal rat cardiomyocyte hypertrophy induced by phenylephrine via inhibiting ERK1/2. Mol. Cell Biochem. 2017, 433, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Wang, M.; Xu, Y.; Liu, J.; Jiang, H.; Wang, Z.; Lin, Y.; Wan, J. Sestrins increase in patients with coronary artery disease and associate with the severity of coronary stenosis. Clin. Chim. Acta 2017, 472, 51–57. [Google Scholar] [CrossRef]
- Chung, H.S.; Hwang, H.J.; Hwang, S.Y.; Kim, N.H.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; Choi, K.M.; Yoo, H.J. Association of serum Sestrin2 level with metabolic risk factors in newly diagnosed drug-naive type 2 diabetes. Diabetes Res. Clin. Pract. 2018, 144, 34–41. [Google Scholar] [CrossRef]
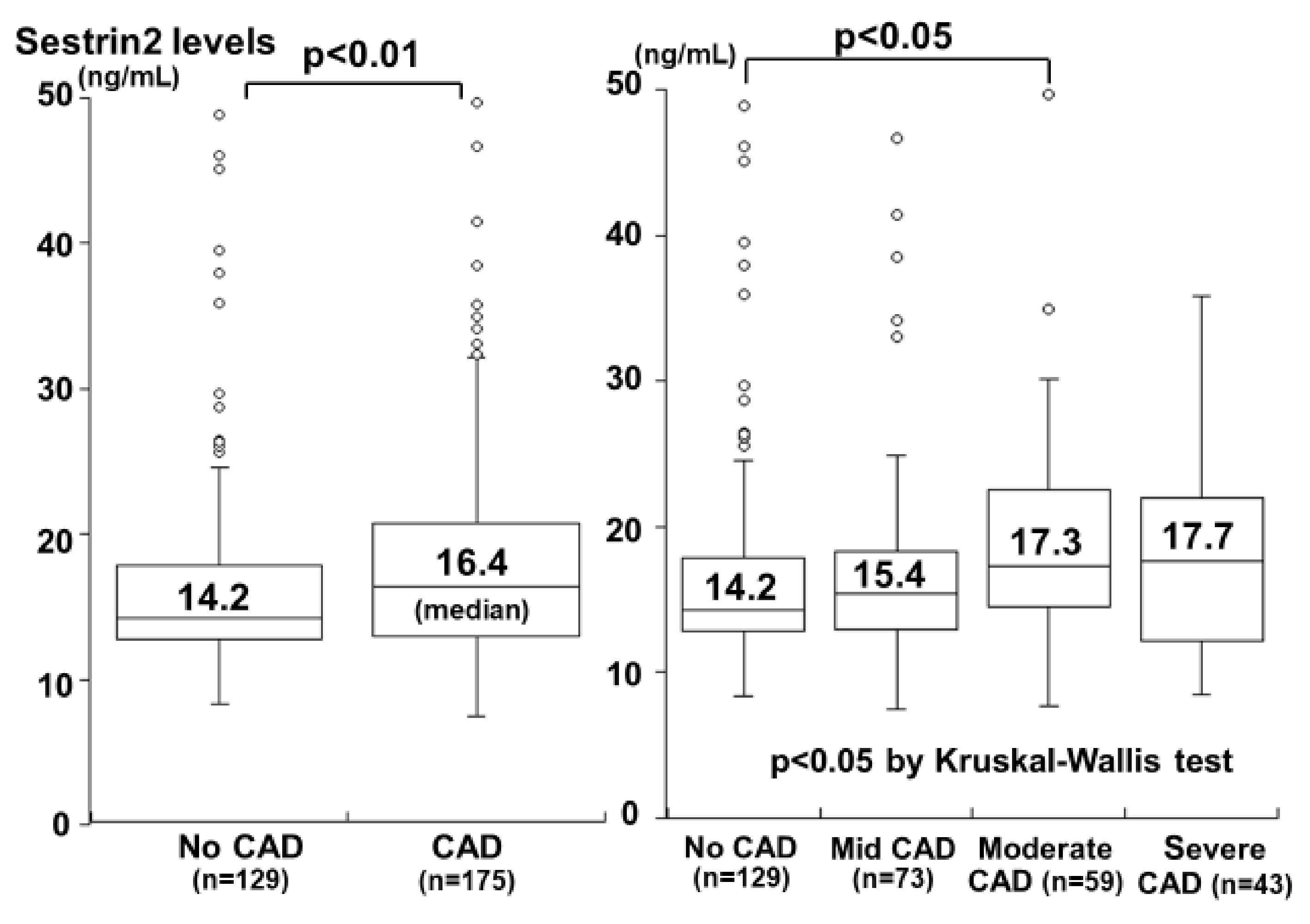
- Kishimoto, Y.; Aoyama, M.; Saita, E.; Ikegami, Y.; Ohmori, R.; Kondo, K.; Momiyama, Y. Association between plasma sestrin2 levels and the presence and severity of coronary artery disease. Dis. Markers 2020, 2020, 7439574. [Google Scholar] [CrossRef]
- Sanchis-Gomar, F. Sestrins: Novel antioxidant and AMPK-modulating functions regulated by exercise? J. Cell Physiol. 2013, 228, 1647–1650. [Google Scholar] [CrossRef]
- Rai, N.; Venugopalan, G.; Pradhan, R.; Ambastha, A.; Upadhyay, A.D.; Dwivedi, S.; Dey, A.B.; Dey, S. Exploration of novel anti-oxidant protein sestrin in frailty syndrome in elderly. Aging Dis. 2018, 9, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Buja, L.M. Myocardial ischemia and reperfusion injury. Cardiovasc. Pathol. 2005, 14, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Morrison, A.; Chen, L.; Wang, J.; Zhang, M.; Yang, H.; Ma, Y.; Budanov, A.; Lee, J.H.; Karin, M.; Li, J. Sestrin2 promotes LKB1-mediated AMPK activation in the ischemic heart. FASEB J. 2015, 29, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Quan, N.; Sun, W.; Wang, L.; Chen, X.; Bogan, J.S.; Zhou, X.; Cates, C.; Liu, Q.; Zheng, Y.; Li, J. Sestrin2 prevents age-related intolerance to ischemia and reperfusion injury by modulating substrate metabolism. FASEB J. 2017, 31, 4153–4167. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Quan, N.; Fedorova, J.; Zhang, J.; He, Z.; Li, J. Sestrin2 modulates cardiac inflammatory response through maintaining redox homeostasis during ischemia and reperfusion. Redox Biol. 2020, 34, 101556. [Google Scholar] [CrossRef]
- Frangogiannis, N.G.; Smith, C.W.; Entman, M.L. The inflammatory response in myocardial infarction. Cardiovasc. Res. 2002, 53, 31–47. [Google Scholar] [CrossRef]
- Yang, K.; Xu, C.; Zhang, Y.; He, S.; Li, D. Sestrin2 suppresses classically activated macrophages-mediated inflammatory response in myocardial infarction through inhibition of mTORC1 signaling. Front. Immunol. 2017, 8, 728. [Google Scholar] [CrossRef]
- Zeng, Y.C.; Chi, F.; Xing, R.; Zeng, J.; Gao, S.; Chen, J.J.; Wang, H.M.; Duan, Q.Y.; Sun, Y.N.; Niu, N.; et al. Sestrin2 protects the myocardium against radiation-induced damage. Radiat. Environ. Biophys. 2016, 55, 195–202. [Google Scholar] [CrossRef]
- Li, R.; Huang, Y.; Semple, I.; Kim, M.; Zhang, Z.; Lee, J.H. Cardioprotective roles of sestrin 1 and sestrin 2 against doxorubicin cardiotoxicity. Am. J. Physiol. Heart Circ. Physiol. 2019, 317, H39–H48. [Google Scholar] [CrossRef]
- Hwang, H.J.; Kim, J.W.; Chung, H.S.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Choi, K.M.; Baik, S.H.; Yoo, H.J. Knockdown of sestrin2 increases lipopolysaccharide-induced oxidative stress, apoptosis, and fibrotic reactions in H9c2 cells and heart tissues of mice via an AMPK-dependent mechanism. Mediat. Inflamm. 2018, 2018, 6209140. [Google Scholar] [CrossRef]
- Quan, N.; Li, X.; Zhang, J.; Han, Y.; Sun, W.; Ren, D.; Tong, Q.; Li, J. Substrate metabolism regulated by Sestrin2-mTORC1 alleviates pressure overload-induced cardiac hypertrophy in aged heart. Redox Biol. 2020, 36, 101637. [Google Scholar] [CrossRef] [PubMed]
- Zoncu, R.; Efeyan, A.; Sabatini, D.M. mTOR: From growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 2011, 12, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Sundararajan, S.; Jayachandran, I.; Subramanian, S.C.; Anjana, R.M.; Balasubramanyam, M.; Mohan, V.; Venkatesan, B.; Manickam, N. Decreased sestrin levels in patients with type 2 diabetes and dyslipidemia and their association with the severity of atherogenic index. J. Endocrinol. Investig. 2020. [Google Scholar] [CrossRef] [PubMed]
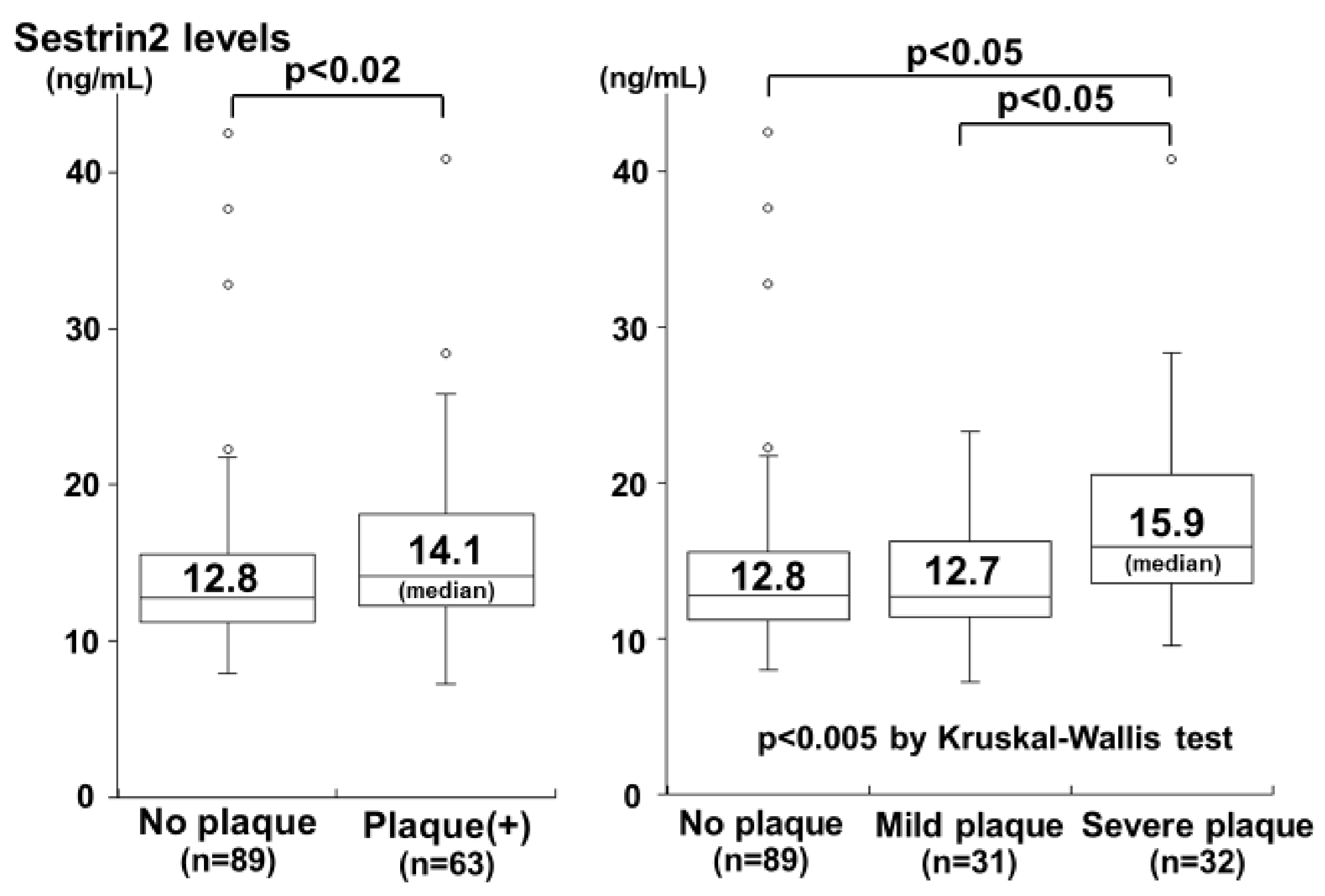
- Kishimoto, Y.; Saita, E.; Ohmori, R.; Kondo, K.; Momiyama, Y. Plasma sestrin2 concentrations and carotid atherosclerosis. Clin. Chim. Acta 2020, 504, 56–59. [Google Scholar] [CrossRef]
- Xiao, T.; Zhang, L.; Huang, Y.; Shi, Y.; Wang, J.; Ji, Q.; Ye, J.; Lin, Y.; Liu, H. Sestrin2 increases in aortas and plasma from aortic dissection patients and alleviates angiotensin II-induced smooth muscle cell apoptosis via the Nrf2 pathway. Life Sci. 2019, 218, 132–138. [Google Scholar] [CrossRef]
- Jiang, R.; Wang, Q.; Zhai, H.; Du, X.; Sun, S.; Wang, H. Explorating the involvement of plasma sestrin2 in obstructive sleep apnea. Can. Respir. J. 2019, 2019, 2047674. [Google Scholar] [CrossRef]
| Study | Serum/Plasma | Study Subjects | Results |
|---|---|---|---|
| Rai et al. [41] | Serum | 51 frail elderly vs. 41 non-frail elderly | Lower in frail elderly than in non-frail elderly. |
| Wang et al. [6] | Plasma | 220 patients with HF vs. 80 controls | Higher in patients with HF than in controls |
| Xiao et al. [55] | Plasma | 120 patients with aortic dissection vs. 40 without dissection | Higher in patients with aortic dissection than in those without dissection |
| Jiang et al. [56] | Plasma | 36 patients with OSA vs. 21 controls | Higher in patients with OSA than in controls |
| Ye et al. [37] | Plasma | 114 patients with CAD (44 SA, 41 UA, 29 AMI) vs. 35 without CAD | Higher in patients with CAD than in those without CAD Higher in patients with UA and AMI than in those with SA |
| Kishimoto et al. [39] | Plasma | 175 patients with CAD vs. 129 without CAD | Higher in patients with CAD than in those without CAD |
| Sundararajan et al. [53] | Serum | 81 patients with DM vs. 46 controls (NGT) | Lower in patients with DM than in controls |
| Chung et al. [38] | Serum | 194 patients with DM vs. 46 without DM | No difference between patients with and without DM |
| Chung et al. [38] | Serum | 80 DM patients with carotid atherosclerosis vs. 114 DM without carotid atherosclerosis | No difference between DM patients with and without carotid atherosclerosis |
| Kishimoto et al. [54] | Plasma | 63 subjects with carotid plaque vs. 89 without carotid plaque | Higher in subjects with carotid plaque than in those without carotid plaque |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kishimoto, Y.; Kondo, K.; Momiyama, Y. The Protective Role of Sestrin2 in Atherosclerotic and Cardiac Diseases. Int. J. Mol. Sci. 2021, 22, 1200. https://doi.org/10.3390/ijms22031200
Kishimoto Y, Kondo K, Momiyama Y. The Protective Role of Sestrin2 in Atherosclerotic and Cardiac Diseases. International Journal of Molecular Sciences. 2021; 22(3):1200. https://doi.org/10.3390/ijms22031200
Chicago/Turabian StyleKishimoto, Yoshimi, Kazuo Kondo, and Yukihiko Momiyama. 2021. "The Protective Role of Sestrin2 in Atherosclerotic and Cardiac Diseases" International Journal of Molecular Sciences 22, no. 3: 1200. https://doi.org/10.3390/ijms22031200
APA StyleKishimoto, Y., Kondo, K., & Momiyama, Y. (2021). The Protective Role of Sestrin2 in Atherosclerotic and Cardiac Diseases. International Journal of Molecular Sciences, 22(3), 1200. https://doi.org/10.3390/ijms22031200






