Human Intravenous Immunoglobulin Alleviates Neuropathic Symptoms in a Rat Model of Paclitaxel-Induced Peripheral Neurotoxicity
Abstract
1. Introduction
2. Results
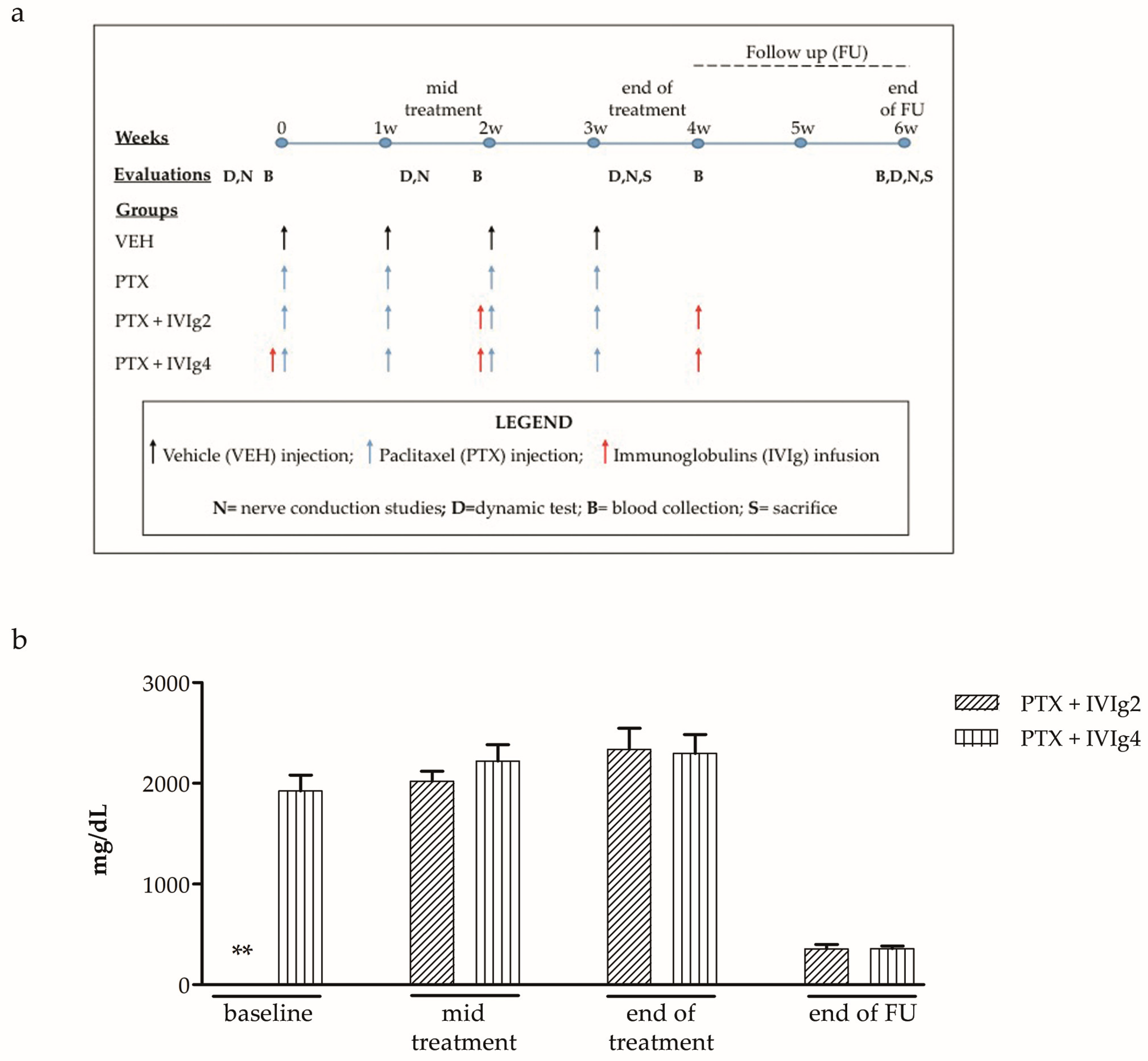
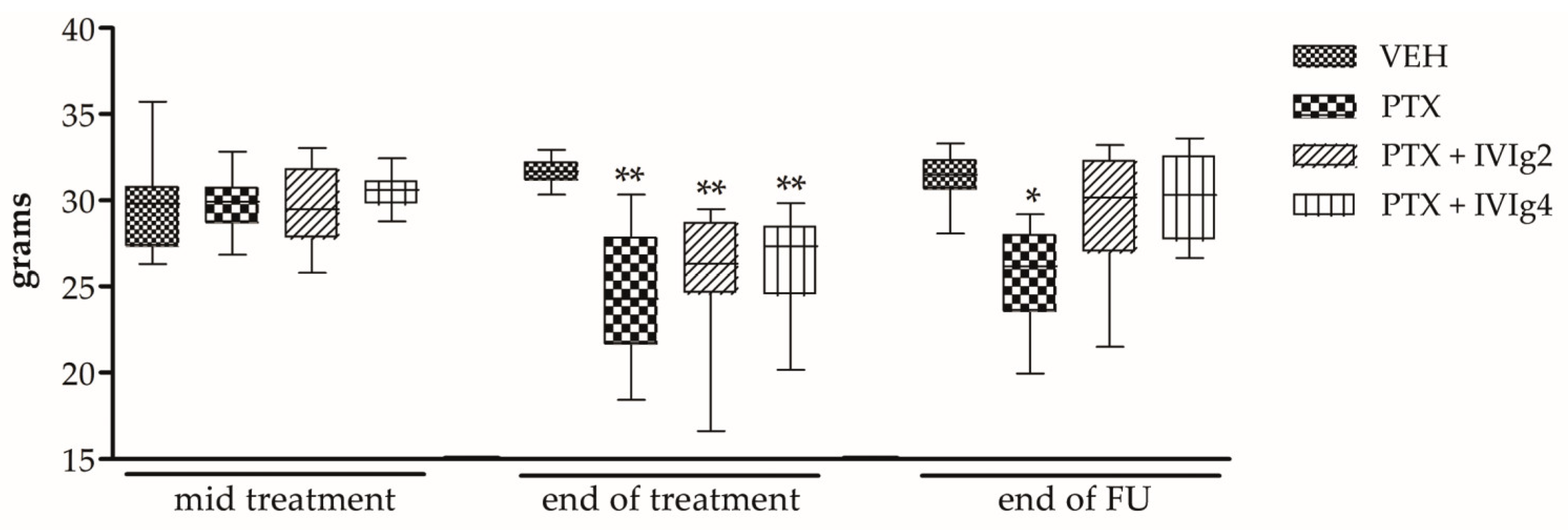
2.1. Safety and Tolerability of PTX and PTX + IVIg Co-Treatment
2.2. IVIg Significantly Attenuates Mechanical Allodynia
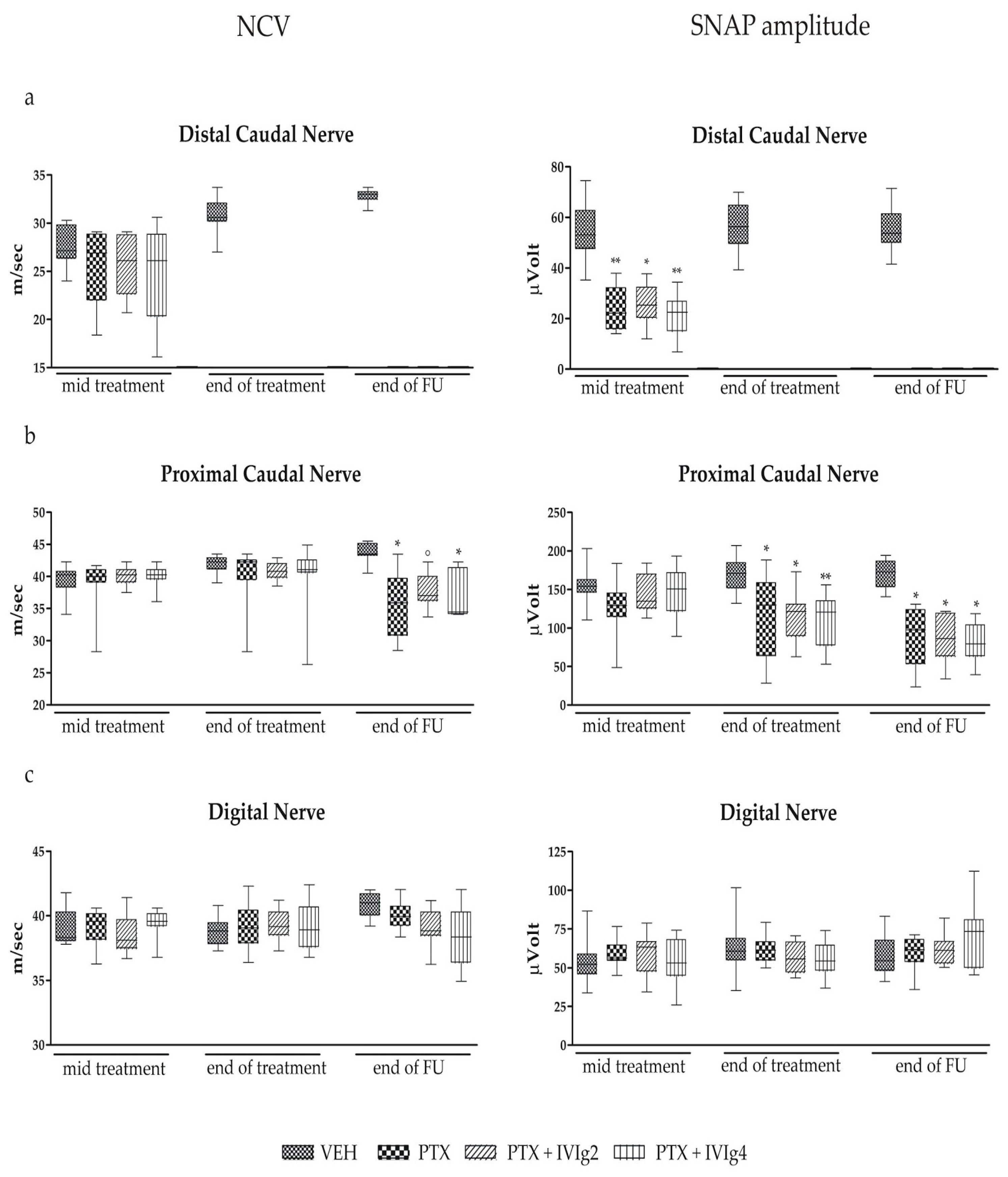
2.3. Nerve Conduction Studies
2.4. IVIg Reduced the Loss of IENF Induced by PTX
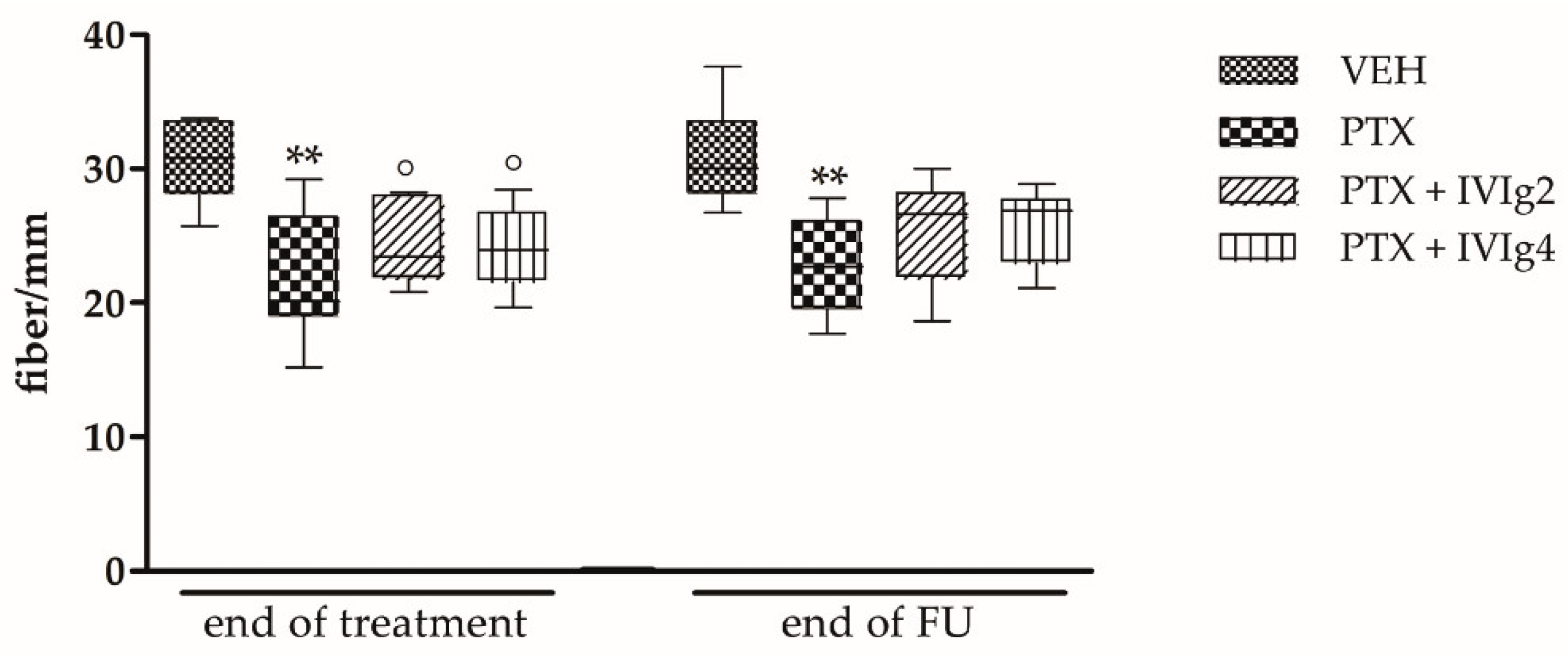
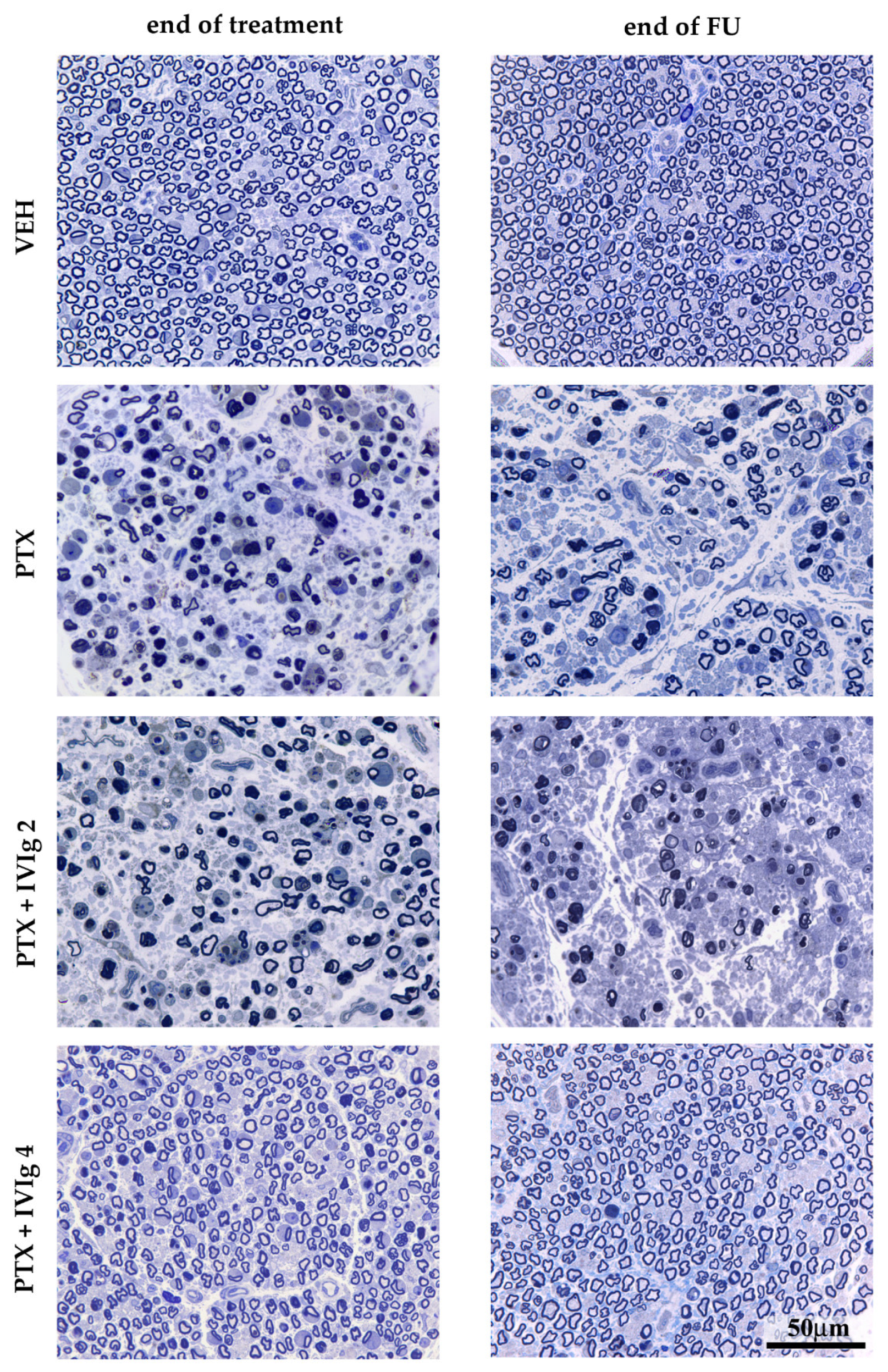
2.5. IVIg Reduced Axonal Degeneration Induced by PTX in Caudal Nerves
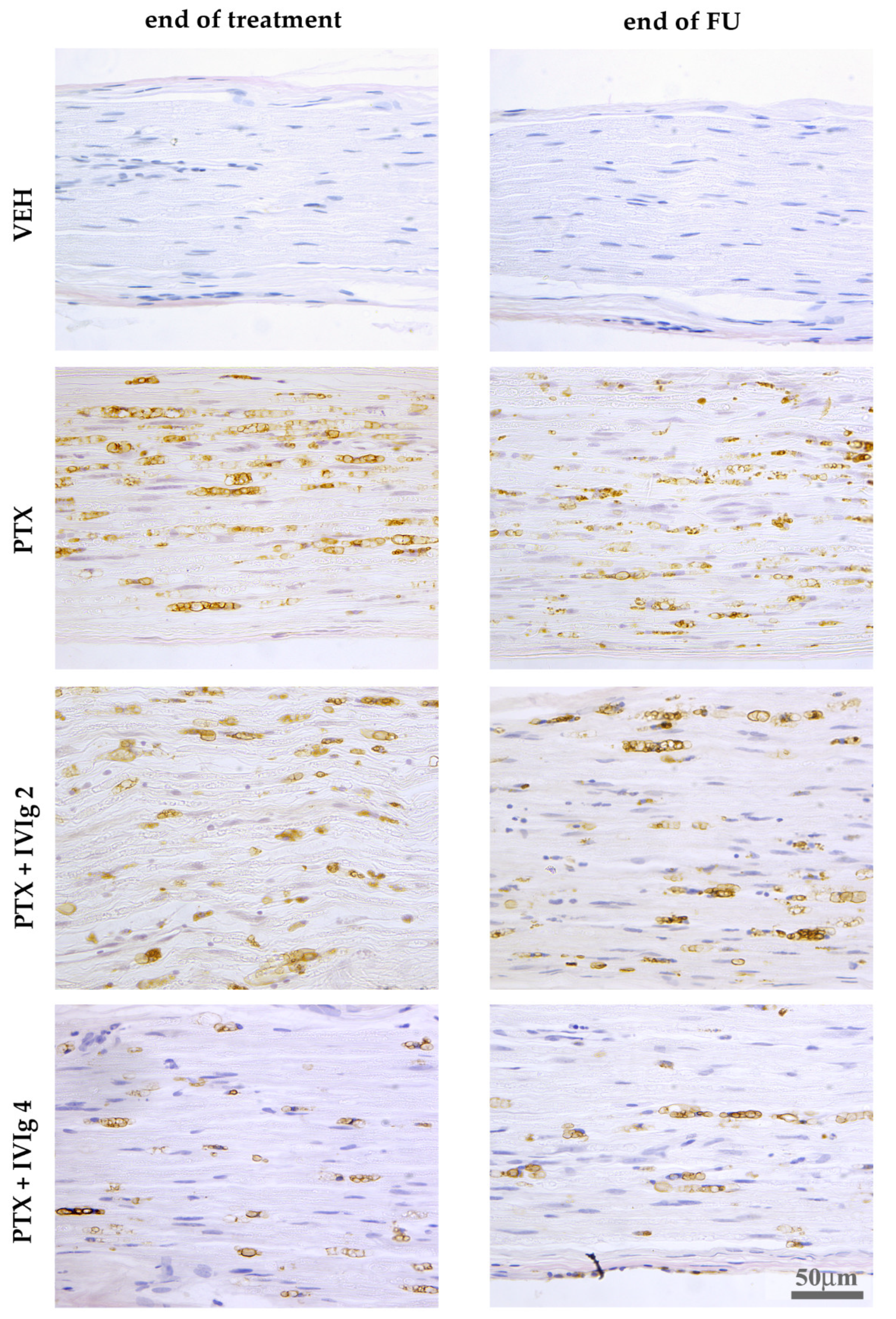
2.6. IVIg Induced a Reduction of PTX-Induced Macrophages Infiltration
3. Discussion
4. Materials and Methods
4.1. Animals and Drugs
4.2. IVIg Serum Levels Determination
4.3. Nerve Conduction Studies
4.4. Assessment of Mechanical Allodynia
4.5. Morphological Analysis
4.6. Immunohistochemical (IHC) Analysis of Infiltrating Cells
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CIPN | chemotherapy-induced peripheral neuropathy |
| DRG | dorsal root ganglia |
| FFPE | formalin-fixed paraffin-embedded |
| IENF | intra-epidermal nerve fibers |
| IHC | immunohistochemical |
| IVIg | human intravenous immunoglobulin |
| NCV | nerves conduction velocity |
| NLRP3 | nod-like receptor 3 inflammasome |
| PIPN | paclitaxel-induced peripheral neurotoxicity |
| PTX | paclitaxel |
| SNAP | sensory nerve action potential |
| TLR4 | toll-like receptor 4 |
| VEH | vehicle |
References
- Starobova, H.; Vetter, I. Pathophysiology of Chemotherapy-Induced Peripheral Neuropathy. Front. Mol. Neurosci. 2017, 10, 174. [Google Scholar] [CrossRef]
- Oneda, E.; Meriggi, F.; Zanotti, L.; Zaina, E.; Bighè, S.; Andreis, F.; Rueda, S.; Zaniboni, A. Innovative Approach for the Prevention of Chemotherapy-Induced Peripheral Neuropathy in Cancer Patients: A Pilot Study With the Hilotherm Device, the Poliambulanza Hospital Experience. Integr. Cancer Ther. 2020, 19, 1534735420943287. [Google Scholar] [CrossRef]
- Reeves, B.N.; Dakhil, S.R.; Sloan, J.A.; Wolf, S.L.; Burger, K.N.; Kamal, A.H.; Le-Lindqwister, N.A.; Soori, G.S.; Jaslowski, A.J.; Kelaghan, J.; et al. Further data supporting that paclitaxel-associated acute pain syndrome is associated with development of peripheral neuropathy. Cancer 2012, 118, 5171–5178. [Google Scholar] [CrossRef] [PubMed]
- Carozzi, V.A.; Canta, A.; Chiorazzi, A. Chemotherapy-induced peripheral neuropathy: What do we know about mechanisms? Neurosci. Lett. 2015, 596, 90–107. [Google Scholar] [CrossRef] [PubMed]
- Fehrenbacher, J.C. Chemotherapy-Induced Peripheral Neuropathy. Prog. Mol. Biol. Transl. Sci. 2015, 131, 471–508. [Google Scholar] [PubMed]
- Kim, J.H.; Dougherty, P.M.; Abdi, S. Basic science and clinical management of painful and non-painful chemotherapy-related neuropathy. Gynecol. Oncol. 2015, 136, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Marmiroli, P.; Nicolini, G.; Miloso, M.; Scuteri, A.; Cavaletti, G. The fundamental role of morphology in experimental neurotoxicology: The example of chemotherapy-induced peripheral neurotoxicity. Ital. J. Anat. Embryol. 2012, 117, 75–97. [Google Scholar] [PubMed]
- Velasco, R.; Bruna, J. Taxane-Induced Peripheral Neurotoxicity. Toxics 2015, 3, 152–169. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Adamek, P.; Zhang, H.; Tatsui, C.E.; Rhines, L.D.; Mrozkova, P.; Li, Q.; Kosturakis, A.K.; Cassidy, R.M.; Harrison, D.S.; et al. The Cancer Chemotherapeutic Paclitaxel Increases Human and Rodent Sensory Neuron Responses to TRPV1 by Activation of TLR4. J. Neurosci. 2015, 35, 13487–13500. [Google Scholar] [CrossRef]
- Yan, X.; Maixner, D.W.; Yadav, R.; Gao, M.; Li, P.; Bartlett, M.G.; Weng, H.-R. Paclitaxel induces acute pain via directly activating toll like receptor 4. Mol. Pain 2015, 11, 10–15. [Google Scholar] [CrossRef]
- Makker, P.G.S.; Duffy, S.S.; Lees, J.G.; Perera, C.J.; Tonkin, R.S.; Butovsky, O.; Park, S.B.; Goldstein, D.; Moalem-Taylor, G. Characterisation of Immune and Neuroinflammatory Changes Associated with Chemotherapy-Induced Peripheral Neuropathy. PLoS ONE 2017, 12, e0170814. [Google Scholar] [CrossRef] [PubMed]
- Lees, J.G.; Makker, P.G.; Tonkin, R.S.; Abdulla, M.; Park, S.B.; Goldstein, D.; Moalem-Taylor, G. Immune-mediated processes implicated in chemotherapy-induced peripheral neuropathy. Eur. J. Cancer 2017, 73, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.F.; Chen, L.H.; Yeh, Y.M.; Wu, P.Y.; Chen, Y.F.; Chang, L.Y.; Chang, J.Y.; Shen, M.R. Minoxidil is a potential neuroprotective drug for paclitaxel-induced peripheral neuropathy. Sci. Rep. 2017, 7, 45366. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.H.; Yeh, Y.M.; Chen, Y.F.; Hsu, Y.H.; Wang, H.H.; Lin, P.C.; Chang, L.Y.; Lin, C.C.K.; Chang, M.S.; Shen, M.R. Targeting interleukin-20 alleviates paclitaxel-induced peripheral neuropathy. Pain 2020, 161, 1237–1254. [Google Scholar] [CrossRef]
- Kalynovska, N.; Diallo, M.; Sotakova-Kasparova, D.; Palecek, J. Losartan attenuates neuroinflammation and neuropathic pain in paclitaxel-induced peripheral neuropathy. J. Cell. Mol. Med. 2020, 24, 7949–7958. [Google Scholar] [CrossRef] [PubMed]
- Peters, C.M.; Jimenez-Andrade, J.M.; Jonas, B.M.; Sevcik, M.A.; Koewler, N.J.; Ghilardi, J.R.; Wong, G.Y.; Mantyh, P.W. Intravenous paclitaxel administration in the rat induces a peripheral sensory neuropathy characterized by macrophage infiltration and injury to sensory neurons and their supporting cells. Exp. Neurol. 2007, 203, 42–54. [Google Scholar] [CrossRef]
- Sekiguchi, F.; Domoto, R.; Nakashima, K.; Yamasoba, D.; Yamanishi, H.; Tsubota, M.; Wake, H.; Nishibori, M.; Kawabata, A. Paclitaxel-induced HMGB1 release from macrophages and its implication for peripheral neuropathy in mice: Evidence for a neuroimmune crosstalk. Neuropharmacology 2018, 141, 201–213. [Google Scholar] [CrossRef]
- Gao, M.; Yan, X.; Weng, H.R. Inhibition of glycogen synthase kinase 3beta activity with lithium prevents and attenuates paclitaxel-induced neuropathic pain. Neuroscience 2013, 254, 301–311. [Google Scholar] [CrossRef][Green Version]
- Singhmar, P.; Huo, X.; Li, Y.; Dougherty, P.M.; Mei, F.; Cheng, X.; Heijnen, C.J.; Kavelaars, A. Orally active Epac inhibitor reverses mechanical allodynia and loss of intraepidermal nerve fibers in a mouse model of chemotherapy-induced peripheral neuropathy. Pain 2018, 159, 884–893. [Google Scholar] [CrossRef]
- Wu, J.; Hocevar, M.; Bie, B.; Foss, J.F.; Naguib, M. Cannabinoid Type 2 Receptor System Modulates Paclitaxel-Induced Microglial Dysregulation and Central Sensitization in Rats. J. Pain 2019, 20, 501–514. [Google Scholar] [CrossRef]
- Li, Y.; Yin, C.; Li, X.; Liu, B.; Wang, J.; Zheng, X.; Shao, X.; Liang, Y.; Du, J.; Fang, J.; et al. Electroacupuncture Alleviates Paclitaxel-Induced Peripheral Neuropathic Pain in Rats via Suppressing TLR4 Signaling and TRPV1 Upregulation in Sensory Neurons. Int. J. Mol. Sci. 2019, 20, 5917. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Y.; De Carvalho-Barbosa, M.; Kavelaars, A.; Heijnen, C.J.; Albrecht, P.J.; Dougherty, P.M. Dorsal Root Ganglion Infiltration by Macrophages Contributes to Paclitaxel Chemotherapy-Induced Peripheral Neuropathy. J. Pain 2016, 17, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Tasnim, A.; Rammelkamp, Z.; Slusher, A.B.; Wozniak, K.M.; Slusher, B.S.; Farah, M.H. Paclitaxel causes degeneration of both central and peripheral axon branches of dorsal root ganglia in mice. BMC Neurosci. 2016, 17, 47. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Wu, C.; Gao, F.; Xiang, H.; Sun, N.; Peng, P.; Li, J.; Yuan, X.; Li, H.; Meng, X.; et al. Activation of NLRP3 inflammasome in peripheral nerve contributes to paclitaxel-induced neuropathic pain. Mol. Pain 2017, 13, 1744806917719804. [Google Scholar] [CrossRef]
- Boyette-Davis, J.; Dougherty, P.M. Protection against oxaliplatin-induced mechanical hyperalgesia and intraepidermal nerve fiber loss by minocycline. Exp. Neurol. 2011, 229, 353–357. [Google Scholar] [CrossRef]
- Burgos, E.; Gómez-Nicola, D.; Pascual, D.; Martín, M.I.; Nieto-Sampedro, M.; Goicoechea, C. Cannabinoid agonist WIN 55,212-2 prevents the development of paclitaxel-induced peripheral neuropathy in rats. Possible involvement of spinal glial cells. Eur. J. Pharmacol. 2012, 682, 62–72. [Google Scholar] [CrossRef]
- Janes, K.; Esposito, E.; Doyle, T.; Cuzzocrea, S.; Tosh, D.K.; Jacobson, K.A.; Salvemini, D. A3 adenosine receptor agonist prevents the development of paclitaxel-induced neuropathic pain by modulating spinal glial-restricted redox-dependent signaling pathways. Pain 2014, 155, 2560–2567. [Google Scholar] [CrossRef]
- Deng, L.; Guindon, J.; Cornett, B.L.; Makriyannis, A.; Mackie, K.; Hohmann, A.G. Chronic Cannabinoid Receptor 2 Activation Reverses Paclitaxel Neuropathy Without Tolerance or Cannabinoid Receptor 1–Dependent Withdrawal. Biol. Psychiatry 2015, 77, 475–487. [Google Scholar] [CrossRef]
- Ochi-Ishi, R.; Nagata, K.; Inoue, T.; Tozaki-Saitoh, H.; Tsuda, M.; Inoue, K. Involvement of the Chemokine CCL3 and the Purinoceptor P2×7 in the Spinal Cord in Paclitaxel-Induced Mechanical Allodynia. Mol. Pain 2014, 10, 53. [Google Scholar] [CrossRef]
- Segat, G.C.; Manjavachi, M.N.; Matias, D.O.; Passos, G.F.; Freitas, C.S.; Costa, R.; Calixto, J.B. Antiallodynic effect of β-caryophyllene on paclitaxel-induced peripheral neuropathy in mice. Neuropharmacology 2017, 125, 207–219. [Google Scholar] [CrossRef]
- Xu, Y.; Cheng, G.; Zhu, Y.; Zhang, X.; Pu, S.; Wu, J.; Lv, Y.; Du, D. Anti-nociceptive roles of the glia-specific metabolic inhibitor fluorocitrate in paclitaxel-evoked neuropathic pain. Acta Biochim. Biophys. Sin. 2016, 48, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.C.; Park, D. Effectiveness of Intravenous Immunoglobulin for Management of Neuropathic Pain: A Narrative Review. J. Pain Res. 2020, 13, 2879–2884. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, S.H.; Sørensen, P.S. Intravenous immunoglobulin treatment of multiple sclerosis and its animal model, experimental autoimmune encephalomyelitis. J. Neurol. Sci. 2005, 233, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Niknami, M.; Wang, M.X.; Nguyen, T.; Pollard, J.D. Beneficial effect of a multimerized immunoglobulin Fc in an animal model of inflammatory neuropathy (experimental autoimmune neuritis). J. Peripher. Nerv. Syst. 2013, 18, 141–152. [Google Scholar] [CrossRef]
- Kajii, M.; Kobayashi, F.; Kashihara, J.; Yuuki, T.; Kubo, Y.; Nakae, T.; Kamizono, A.; Kuzumoto, Y.; Kusunoki, S. Intravenous immunoglobulin preparation attenuates neurological signs in rat experimental autoimmune neuritis with the suppression of macrophage inflammatory protein -1α expression. J. Neuroimmunol. 2014, 266, 43–48. [Google Scholar] [CrossRef]
- Meregalli, C.; Marjanovic, I.; Scali, C.; Monza, L.; Spinoni, N.; Galliani, C.; Brivio, R.; Chiorazzi, A.; Ballarini, E.; Rodriguez-Menendez, V.; et al. High-dose intravenous immunoglobulins reduce nerve macrophage infiltration and the severity of bortezomib-induced peripheral neurotoxicity in rats. J. Neuroinflamm. 2018, 15, 232. [Google Scholar] [CrossRef]
- Cavaletti, G.; Gilardini, A.; Canta, A.; Rigamonti, L.; Rodriguez-Menendez, V.; Ceresa, C.; Marmiroli, P.; Bossi, M.; Oggioni, N.; D’Incalci, M.; et al. Bortezomib-induced peripheral neurotoxicity: A neurophysiological and pathological study in the rat. Exp. Neurol. 2007, 204, 317–325. [Google Scholar] [CrossRef]
- Smith, E.L.; Pang, H.; Cirrincione, C.; Fleishman, S.; Paskett, E.D.; Ahles, T.; Bressler, L.R.; Fadul, C.E.; Knox, C.; Le-Lindqwister, N.; et al. Effect of Duloxetine on Pain, Function, and Quality of Life Among Patients With Chemotherapy-Induced Painful Peripheral Neuropathy. JAMA 2013, 309, 1359–1367. [Google Scholar] [CrossRef]
- Goebel, A. Immunoglobulin Responsive Chronic Pain. J. Clin. Immunol. 2010, 30, 103–108. [Google Scholar] [CrossRef]
- A Rajabally, Y. Overview of the pathogenesis and treatment of chronic inflammatory demyelinating polyneuropathy with intravenous immunoglobulins. Biol. Targets Ther. 2010, 4, 45–49. [Google Scholar] [CrossRef][Green Version]
- Meregalli, C.; Fumagalli, G.; Alberti, P.; Canta, A.; Chiorazzi, A.; Monza, L.; Pozzi, E.; Carozzi, V.A.; Blennow, K.; Zetterberg, H.; et al. Neurofilament light chain: A specific serum biomarker of axonal damage severity in rat models of Chemotherapy-Induced Peripheral Neurotoxicity. Arch. Toxicol. 2020, 94, 2517–2522. [Google Scholar] [CrossRef] [PubMed]
- Manjavachi, M.N.; Passos, G.F.; Trevisan, G.; Araújo, S.B.; Pontes, J.P.; Fernandes, E.S.; Da Costa, R.; Calixto, J.B. Spinal blockage of CXCL1 and its receptor CXCR2 inhibits paclitaxel-induced peripheral neuropathy in mice. Neuropharmacology 2019, 151, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Lv, N.; Cui, L.; Yang, T.; Zhao, Z.Q.; Xin, W.J.; Liu, X.G. Prevention of Paclitaxel-Induced Allodynia by Minocycline: Effect on Loss of Peripheral Nerve Fibers and Infiltration of Macrophages in Rats. Mol. Pain 2010, 6, 76. [Google Scholar] [CrossRef] [PubMed]
- Alberti, P.; Canta, A.; Chiorazzi, A.; Fumagalli, G.; Meregalli, C.; Monza, L.; Pozzi, E.; Ballarini, E.; Rodriguez-Menendez, V.; Oggioni, N.; et al. Topiramate prevents oxaliplatin-related axonal hyperexcitability and oxaliplatin induced peripheral neurotoxicity. Neuropharmacology 2020, 164, 107905. [Google Scholar] [CrossRef]
- Canta, A.; Chiorazzi, A.; Meregalli, C.; Carozzi, V.; Oggioni, N.; Lauria, G.; Lombardi, R.; Bianchi, R.; Porretta-Serapiglia, C.; Cavaletti, G. Continuous Buprenorphine Delivery Effect in Streptozotocine-Induced Painful Diabetic Neuropathy in Rats. J. Pain 2009, 10, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Siau, C.; Xiao, W.; Bennett, G.J. Paclitaxel- and vincristine-evoked painful peripheral neuropathies: Loss of epidermal innervation and activation of Langerhans cells. Exp. Neurol. 2006, 201, 507–514. [Google Scholar] [CrossRef]
- Lauria, G.; Lombardi, R. Skin biopsy: A new tool for diagnosing peripheral neuropathy. BMJ 2007, 334, 1159–1162. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meregalli, C.; Monza, L.; Chiorazzi, A.; Scali, C.; Guarnieri, C.; Fumagalli, G.; Alberti, P.; Pozzi, E.; Canta, A.; Ballarini, E.; et al. Human Intravenous Immunoglobulin Alleviates Neuropathic Symptoms in a Rat Model of Paclitaxel-Induced Peripheral Neurotoxicity. Int. J. Mol. Sci. 2021, 22, 1058. https://doi.org/10.3390/ijms22031058
Meregalli C, Monza L, Chiorazzi A, Scali C, Guarnieri C, Fumagalli G, Alberti P, Pozzi E, Canta A, Ballarini E, et al. Human Intravenous Immunoglobulin Alleviates Neuropathic Symptoms in a Rat Model of Paclitaxel-Induced Peripheral Neurotoxicity. International Journal of Molecular Sciences. 2021; 22(3):1058. https://doi.org/10.3390/ijms22031058
Chicago/Turabian StyleMeregalli, Cristina, Laura Monza, Alessia Chiorazzi, Carla Scali, Chiara Guarnieri, Giulia Fumagalli, Paola Alberti, Eleonora Pozzi, Annalisa Canta, Elisa Ballarini, and et al. 2021. "Human Intravenous Immunoglobulin Alleviates Neuropathic Symptoms in a Rat Model of Paclitaxel-Induced Peripheral Neurotoxicity" International Journal of Molecular Sciences 22, no. 3: 1058. https://doi.org/10.3390/ijms22031058
APA StyleMeregalli, C., Monza, L., Chiorazzi, A., Scali, C., Guarnieri, C., Fumagalli, G., Alberti, P., Pozzi, E., Canta, A., Ballarini, E., Rodriguez-Menendez, V., Oggioni, N., Cavaletti, G., & Marmiroli, P. (2021). Human Intravenous Immunoglobulin Alleviates Neuropathic Symptoms in a Rat Model of Paclitaxel-Induced Peripheral Neurotoxicity. International Journal of Molecular Sciences, 22(3), 1058. https://doi.org/10.3390/ijms22031058







