Investigating Pathogenicity and Virulence of Staphylococcus pettenkoferi: An Emerging Pathogen
Abstract
:1. Introduction
2. Results
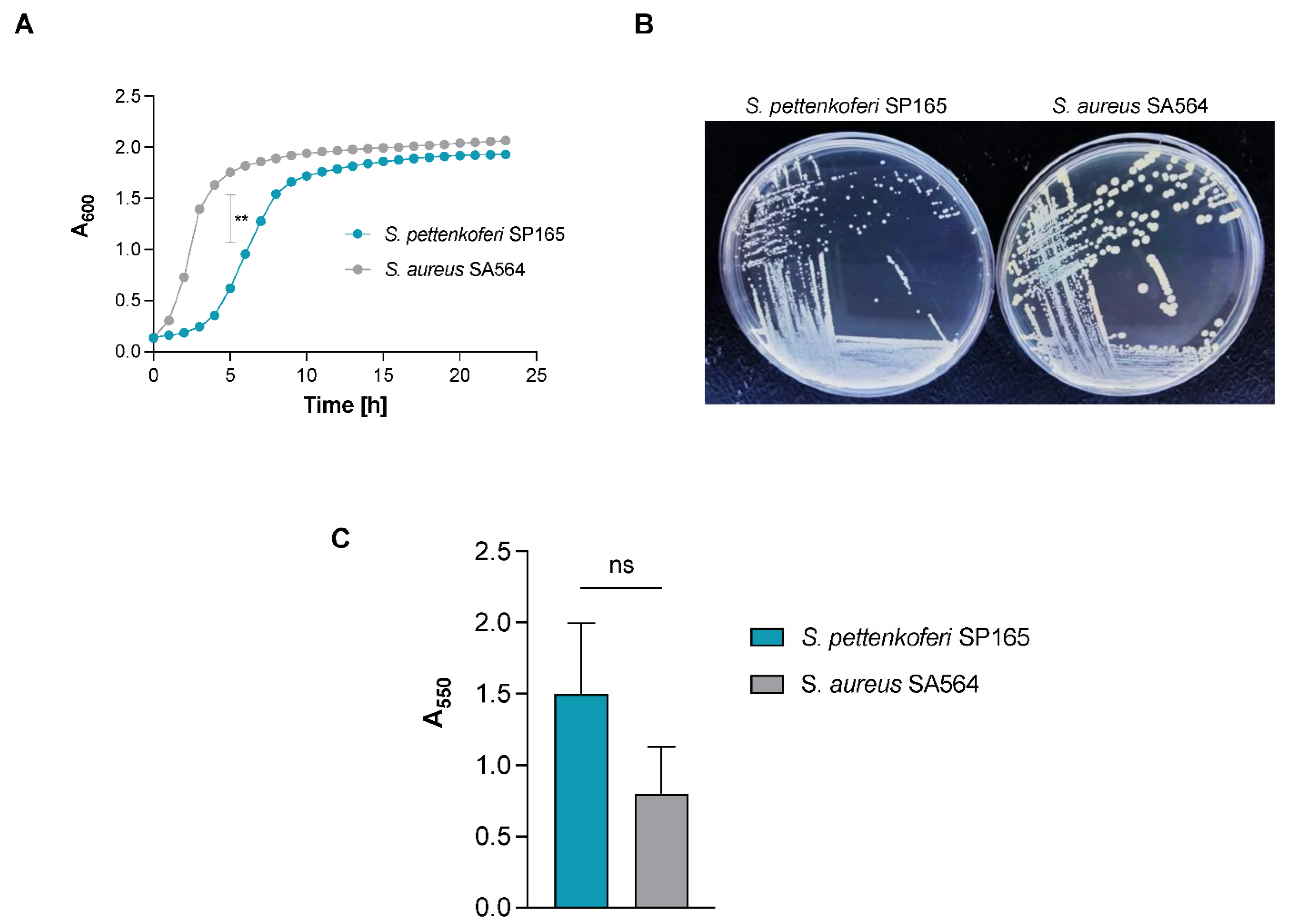
2.1. S. pettenkoferi Growth and Biofilm Formation
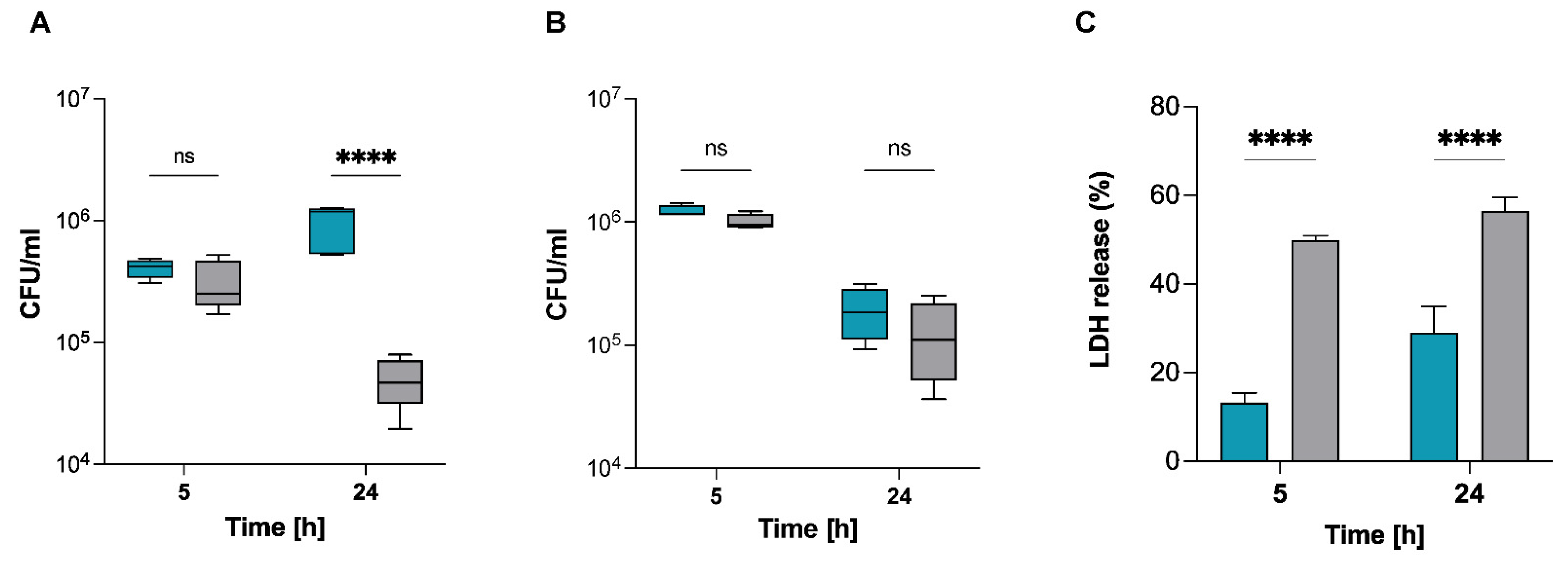
2.2. S. pettenkoferi Persists within Murine and Human Macrophages
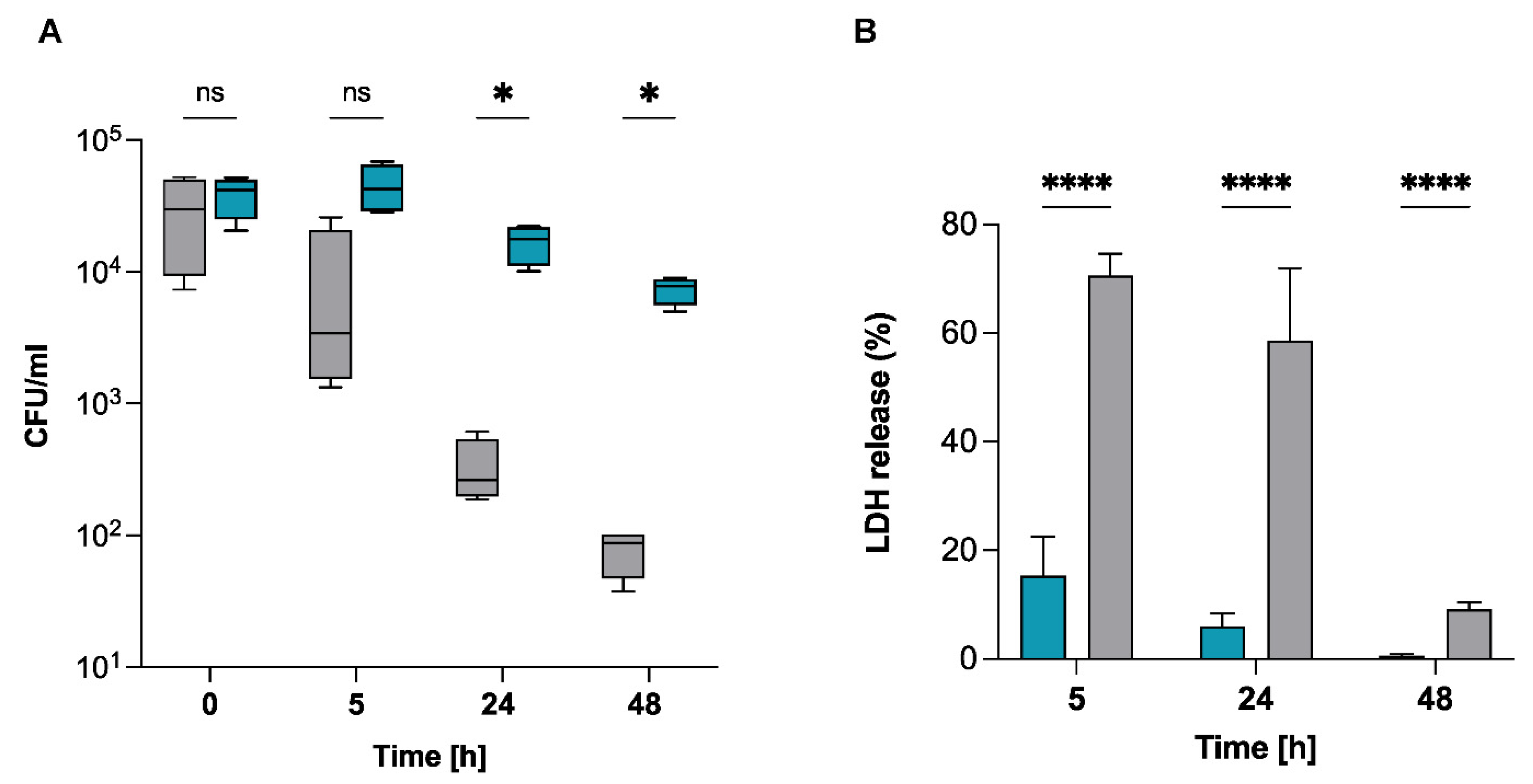
2.3. S. pettenkoferi Persists in Human Keratinocytes
2.4. S. pettenkoferi Avoids Whole Blood Killing but Is Sensitive to Plasma
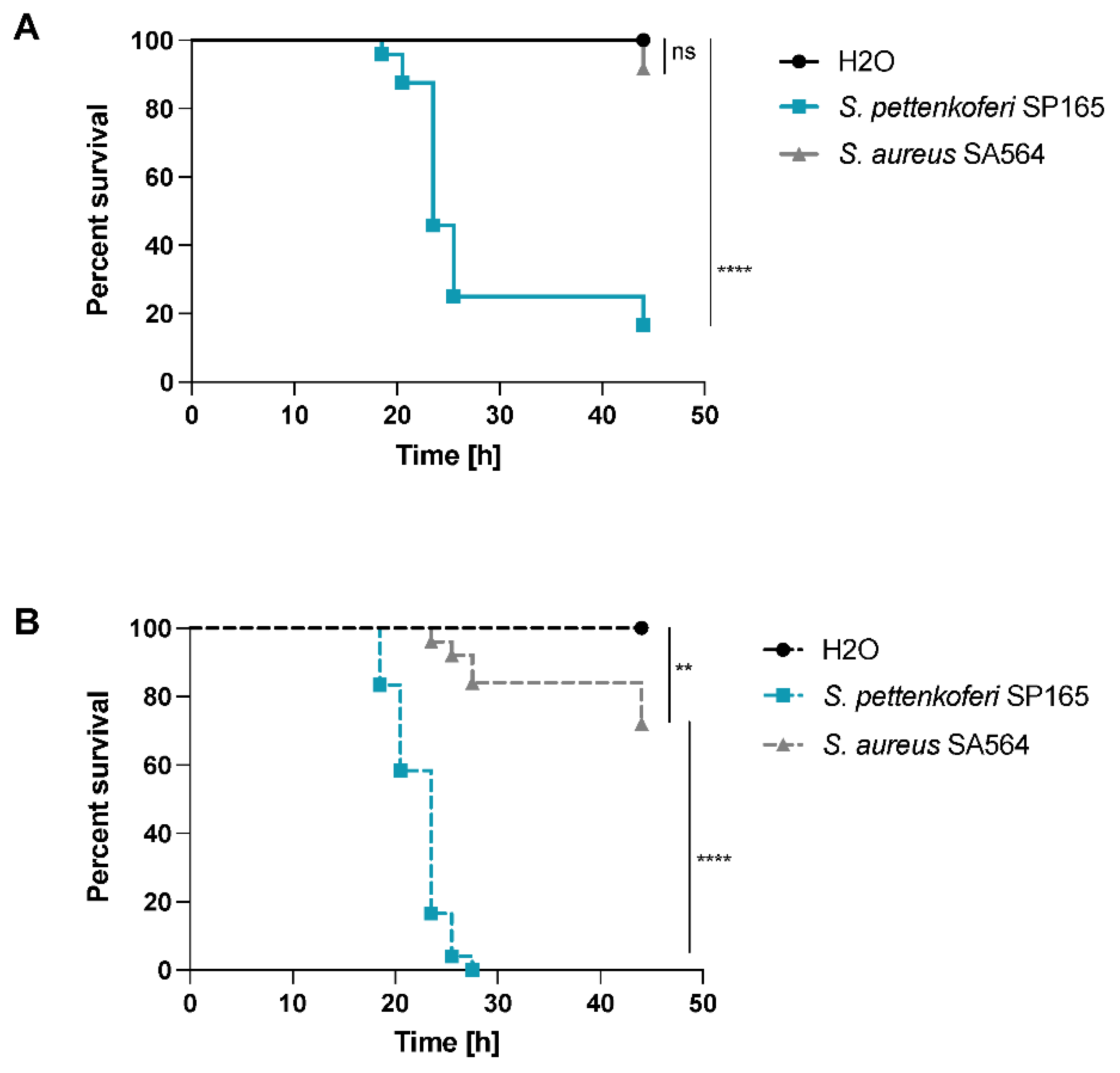
2.5. A Bath Infection Model Using Healthy and Wounded Zebrafish Embryos Was Used to Evaluate S. pettenkoferi Pathogenicity
2.6. Whole-Genome analysis of S. pettenkoferi SP165
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains, Media, and Growth Conditions
4.2. Macrophage Culture and Infection
4.3. Keratinocyte Culture and Infection
4.4. Cell Viability Assay
4.5. Whole Blood and Plasma Killing Assays
4.6. Infection of Danio Rerio Embryos
4.7. Crystal Violet Biofilm Assay
4.8. Ethical Statement
4.9. Whole-Genome Analysis
4.10. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bogut, A.; Magryś, A. The road to success of coagulase-negative staphylococci: Clinical significance of small colony variants and their pathogenic role in persistent infections. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 2249–2270. [Google Scholar] [CrossRef]
- Becker, K.; Heilmann, C.; Peters, G. Coagulase-Negative Staphylococci. Clin. Microbiol. Rev. 2014, 27, 870–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michalik, M.; Samet, A.; Podbielska-Kubera, A.; Savini, V.; Międzobrodzki, J.; Kosecka-Strojek, M. Coagulase-negative staphylococci (CoNS) as a significant etiological factor of laryngological infections: A review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 1–10. [Google Scholar] [CrossRef]
- Trülzsch, K.; Rinder, H.; Trček, J.; Bader, L.; Wilhelm, U.; Heesemann, J. “Staphylococcus pettenkoferi,” a novel staphylococcal species isolated from clinical specimens. Diagn. Microbiol. Infect. Dis. 2002, 43, 175–182. [Google Scholar] [CrossRef]
- Månsson, E.; Hellmark, B.; Stegger, M.; Andersen, P.S.; Sundqvist, M.; Söderquist, B. Genomic relatedness of Staphylococcus pettenkoferi isolates of different origins. J. Med Microbiol. 2017, 66, 601–608. [Google Scholar] [CrossRef] [Green Version]
- Hashi, A.A.; Delport, J.A.; Elsayed, S.; Silverman, M.S. Staphylococcus aureus Bacteremia: A Case Report and Review of the Literature. Can. J. Infect. Dis. Med. Microbiol. 2015, 26, 319–322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.; Chung, H.-S.; Lee, M. Clinical and Microbiological Characteristics of Six Staphylococcus pettenkoferi Isolates from Blood Samples. Ann. Lab. Med. 2015, 35, 250–253. [Google Scholar] [CrossRef] [Green Version]
- Vecchia, M.; Lombardi, A.; Zuccaro, V.; Mariani, B.; Morea, A.; Asperges, E.; Grecchi, C.; Sacchi, P.; Bruno, R. A Clinical Case of Sepsis Due to Staphylococcus pettenkoferi. Infez. Med. 2018, 26, 369–373. [Google Scholar] [PubMed]
- Eke, A.U.; Fairfax, M.R.; Mitchell, R.; Taylor, M.; Salimnia, H. Staphylococcus pettenkoferi-positive Blood cultures in Hospitalized Patients in a Multi-site Tertiary Center. Diagn. Microbiol. Infect. Dis. 2020, 99, 115284. [Google Scholar] [CrossRef]
- Dutta, T.K.; Chakraborty, S.; Das, M.; Mandakini, R.; Vanrahmlimphuii; Roychoudhury, P.; Ghorai, S.; Behera, S.K. Multidrug-resistant Staphylococcus pettenkoferi isolated from cat in India. Veter. World 2018, 11, 1380–1384. [Google Scholar] [CrossRef] [Green Version]
- Lipsky, B.A.; Senneville, É.; Abbas, Z.G.; Aragón-Sánchez, J.; Diggle, M.; Embil, J.M.; Kono, S.; Lavery, L.A.; Malone, M.; van Asten, S.A.; et al. Guidelines on the diagnosis and treatment of foot infection in persons with diabetes (IWGDF 2019 update). Diabetes/Metab. Res. Rev. 2020, 36 (Suppl. 1), e3280. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Ponsero, A.J.; Armstrong, D.G.; Lipsky, B.A.; Hurwitz, B.L. The dynamic wound microbiome. BMC Med. 2020, 18, 1–12. [Google Scholar] [CrossRef]
- Dunyach-Remy, C.; Essebe, C.N.; Sotto, A.; Lavigne, J.-P. Staphylococcus aureus Toxins and Diabetic Foot Ulcers: Role in Pathogenesis and Interest in Diagnosis. Toxins 2016, 8, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loïez, C.; Wallet, F.; Pischedda, P.; Renaux, E.; Senneville, E.; Mehdi, N.; Courcol, R.J. First Case of Osteomyelitis Caused by “Staphylococcus pettenkoferi”. J. Clin. Microbiol. 2007, 45, 1069–1071. [Google Scholar] [CrossRef] [Green Version]
- Flannagan, R.; Heit, B.; Heinrichs, D.E. Antimicrobial Mechanisms of Macrophages and the Immune Evasion Strategies of Staphylococcus aureus. Pathogens 2015, 4, 826–868. [Google Scholar] [CrossRef] [Green Version]
- Tribelli, P.M.; Luqman, A.; Nguyen, M.; Madlung, J.; Fan, S.; Macek, B.; Sass, P.; Bitschar, K.; Schittek, B.; Kretschmer, D.; et al. Staphylococcus aureusLpl protein triggers human host cell invasion via activation of Hsp90 receptor. Cell. Microbiol. 2019, 22, e13111. [Google Scholar] [CrossRef] [Green Version]
- Gupta, S.; Tang, C.; Tran, M.; Kadouri, D.E. Effect of Predatory Bacteria on Human Cell Lines. PLoS ONE 2016, 11, e0161242. [Google Scholar] [CrossRef] [PubMed]
- Jordana-Lluch, E.; Garcia, V.; Kingdon, A.D.H.; Singh, N.; Alexander, C.; Williams, P.; Hardie, K.R. A Simple Polymicrobial Biofilm Keratinocyte Colonization Model for Exploring Interactions between Commensals, Pathogens and Antimicrobials. Front. Microbiol. 2020, 11, 291. [Google Scholar] [CrossRef] [Green Version]
- Hoesl, C.; Zanuttigh, E.; Fröhlich, T.; Philippou-Massier, J.; Krebs, S.; Blum, H.; Dahlhoff, M. The secretome of skin cancer cells activates the mTOR/MYC pathway in healthy keratinocytes and induces tumorigenic properties. Biochim. Biophys. Acta (BBA)-Bioenerg. 2020, 1867, 118717. [Google Scholar] [CrossRef] [PubMed]
- Mempel, M.; Schnopp, C.; Hojka, M.; Fesq, H.; Weidinger, S.; Schaller, M.; Korting, H.; Ring, J.; Abeck, D. Invasion of human keratinocytes by Staphylococcus aureus and intracellular bacterial persistence represent haemolysin-independent virulence mechanisms that are followed by features of necrotic and apoptotic keratinocyte cell death. Br. J. Dermatol. 2002, 146, 943–951. [Google Scholar] [CrossRef]
- Soong, G.; Chun, J.; Parker, D.; Prince, A. Staphylococcus aureus Activation of Caspase 1/Calpain Signaling Mediates Invasion Through Human Keratinocytes. J. Infect. Dis. 2012, 205, 1571–1579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomes, M.C.; Mostowy, S. The Case for Modeling Human Infection in Zebrafish. Trends Microbiol. 2020, 28, 10–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torraca, V.; Mostowy, S. Zebrafish Infection: From Pathogenesis to Cell Biology. Trends Cell Biol. 2017, 28, 143–156. [Google Scholar] [CrossRef] [Green Version]
- Rasheed, S.; Fries, F.; Müller, R.; Herrmann, J. Zebrafish: An Attractive Model to Study Staphylococcus aureus Infection and Its Use as a Drug Discovery Tool. Pharmaceuticals 2021, 14, 594. [Google Scholar] [CrossRef] [PubMed]
- Nogaret, P.; El Garah, F.; Blanc-Potard, A.-B. A Novel Infection Protocol in Zebrafish Embryo to Assess Pseudomonas aeruginosa Virulence and Validate Efficacy of a Quorum Sensing Inhibitor In Vivo. Pathogens 2021, 10, 401. [Google Scholar] [CrossRef]
- Saraceni, P.R.; Romero, A.; Figueras, A.; Novoa, B. Establishment of Infection Models in Zebrafish Larvae (Danio rerio) to Study the Pathogenesis of Aeromonas hydrophila. Front. Microbiol. 2016, 7, 1219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef]
- Gutierrez, A.; Jain, S.; Bhargava, P.; Hamblin, M.; Lobritz, M.A.; Collins, J.J. Understanding and Sensitizing Density-Dependent Persistence to Quinolone Antibiotics. Mol. Cell 2017, 68, 1147–1154.e3. [Google Scholar] [CrossRef] [Green Version]
- Leggett, H.C.; Cornwallis, C.K.; Buckling, A.; West, S. Growth rate, transmission mode and virulence in human pathogens. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160094. [Google Scholar] [CrossRef] [Green Version]
- Tardy, L.; Giraudeau, M.; Hill, G.E.; McGraw, K.J.; Bonneaud, C. Contrasting evolution of virulence and replication rate in an emerging bacterial pathogen. Proc. Natl. Acad. Sci. USA 2019, 116, 16927–16932. [Google Scholar] [CrossRef] [Green Version]
- Heilmann, C.; Ziebuhr, W.; Becker, K. Are coagulase-negative staphylococci virulent? Clin. Microbiol. Infect. 2019, 25, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Laroux, F.S.; Romero, X.; Wetzler, L.; Engel, P.; Terhorst, C. Cutting Edge: MyD88 Controls Phagocyte NADPH Oxidase Function and Killing of Gram-Negative Bacteria. J. Immunol. 2005, 175, 5596–5600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canton, J.; Khezri, R.; Glogauer, M.; Grinstein, S. Contrasting phagosome pH regulation and maturation in human M1 and M2 macrophages. Mol. Biol. Cell 2014, 25, 3330–3341. [Google Scholar] [CrossRef] [PubMed]
- Huynh, K.K.; Eskelinen, E.-L.; Scott, C.; Malevanets, A.; Saftig, P.; Grinstein, S. LAMP proteins are required for fusion of lysosomes with phagosomes. EMBO J. 2007, 26, 313–324. [Google Scholar] [CrossRef]
- Flannagan, R.S.; Cosío, G.; Grinstein, S. Antimicrobial mechanisms of phagocytes and bacterial evasion strategies. Nat. Rev. Genet. 2009, 7, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Flannagan, R.; Watson, D.W.; Surewaard, B.G.J.; Kubes, P.; Heinrichs, D.E. The surreptitious survival of the emerging pathogenStaphylococcus lugdunensiswithin macrophages as an immune evasion strategy. Cell. Microbiol. 2018, 20, e12869. [Google Scholar] [CrossRef]
- Pelegrin, P.; Barroso-Gutierrez, C.; Surprenant, A. P2X7 Receptor Differentially Couples to Distinct Release Pathways for IL-1β in Mouse Macrophage. J. Immunol. 2008, 180, 7147–7157. [Google Scholar] [CrossRef] [Green Version]
- Bryan, N.B.; Dorfleutner, A.; Kramer, S.J.; Yun, C.; Rojanasakul, Y.; Stehlik, C. Differential splicing of the apoptosis-associated speck like protein containing a caspase recruitment domain (ASC) regulates inflammasomes. J. Inflamm. 2010, 7, 23. [Google Scholar] [CrossRef] [Green Version]
- Missiakas, D.; Winstel, V. Selective Host Cell Death by Staphylococcus aureus: A Strategy for Bacterial Persistence. Front. Immunol. 2021, 11. [Google Scholar] [CrossRef]
- Surewaard, B.G.; Deniset, J.F.; Zemp, F.J.; Amrein, M.; Otto, M.; Conly, J.; Omri, A.; Yates, R.M.; Kubes, P. Identification and treatment of the Staphylococcus aureus reservoir in vivo. J. Exp. Med. 2016, 213, 1141–1151. [Google Scholar] [CrossRef] [Green Version]
- Zeng, Z.; Surewaard, B.G.; Wong, C.; Geoghegan, J.A.; Jenne, C.N.; Kubes, P. CRIg Functions as a Macrophage Pattern Recognition Receptor to Directly Bind and Capture Blood-Borne Gram-Positive Bacteria. Cell Host Microbe 2016, 20, 99–106. [Google Scholar] [CrossRef] [Green Version]
- Lehar, S.M.; Pillow, T.; Xu, M.; Staben, L.; Kajihara, K.K.; Vandlen, R.; DePalatis, L.; Raab, H.; Hazenbos, W.L.; Morisaki, J.H.; et al. Novel antibody–antibiotic conjugate eliminates intracellular S. aureus. Nature 2015, 527, 323–328. [Google Scholar] [CrossRef]
- Mammina, C.; Bonura, C.; Verde, M.S.; Fasciana, T.; Palma, D.M. A Fatal Bloodstream Infection byStaphylococcus pettenkoferiin an Intensive Care Unit Patient. Case Rep. Crit. Care 2011, 2011, 1–3. [Google Scholar] [CrossRef] [Green Version]
- Malachowa, N.; DeLeo, F.R. Staphylococcus aureussurvival in human blood. Virulence 2011, 2, 567–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheung, G.Y.C.; Bae, J.S.; Otto, M. Pathogenicity and virulence of Staphylococcus aureus. Virulence 2021, 12, 547–569. [Google Scholar] [CrossRef]
- Guo, H.; Hall, J.; Yang, J.; Ji, Y. The SaeRS Two-Component System Controls Survival of Staphylococcus aureus in Human Blood through Regulation of Coagulase. Front. Cell. Infect. Microbiol. 2017, 7, 204. [Google Scholar] [CrossRef] [Green Version]
- Otto, M. Staphylococcus epidermidis—the ‘accidental’ pathogen. Nat. Rev. Genet. 2009, 7, 555–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taha, M.; Kyluik-Price, D.; Kumaran, D.; Scott, M.D.; Toyofuku, W.; Ramirez-Arcos, S. Bacterial survival in whole blood depends on plasma sensitivity and resistance to neutrophil killing. Transfusion 2019, 59, 3674–3682. [Google Scholar] [CrossRef]
- Masud, S.; Torraca, V.; Meijer, A.H. Modeling Infectious Diseases in the Context of a Developing Immune System. Curr. Top Dev. Biol. 2017, 124, 277–329. [Google Scholar] [CrossRef] [Green Version]
- Prajsnar, T.K.; Cunliffe, V.T.; Foster, S.J.; Renshaw, S.A. A novel vertebrate model ofStaphylococcus aureusinfection reveals phagocyte-dependent resistance of zebrafish to non-host specialized pathogens. Cell. Microbiol. 2008, 10, 2312–2325. [Google Scholar] [CrossRef] [PubMed]
- Prajsnar, T.K.; Hamilton, R.; Garcia-Lara, J.; McVicker, G.; Williams, A.; Boots, M.; Foster, S.J.; Renshaw, S.A. A privileged intraphagocyte niche is responsible for disseminated infection of S taphylococcus aureus in a zebrafish model. Cell. Microbiol. 2012, 14, 1600–1619. [Google Scholar] [CrossRef] [Green Version]
- McVicker, G.; Prajsnar, T.; Williams, A.; Wagner, N.L.; Boots, M.; Renshaw, S.; Foster, S.J. Clonal Expansion during Staphylococcus aureus Infection Dynamics Reveals the Effect of Antibiotic Intervention. PLoS Pathog. 2014, 10, e1003959. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boldock, E.; Surewaard, B.G.J.; Shamarina, D.; Na, M.; Fei, Y.; Ali, A.; Williams, A.; Pollitt, E.J.G.; Szkuta, P.; Morris, P.; et al. Human skin commensals augment Staphylococcus aureus pathogenesis. Nat. Microbiol. 2018, 3, 881–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keightley, M.-C.; Wang, C.-H.; Pazhakh, V.; Lieschke, G.J. Delineating the roles of neutrophils and macrophages in zebrafish regeneration models. Int. J. Biochem. Cell Biol. 2014, 56, 92–106. [Google Scholar] [CrossRef] [PubMed]
- Mateus, R.; Pereira, T.; Sousa, S.; de Lima, J.E.; Pascoal, S.; Saúde, L.; Jacinto, A. In Vivo Cell and Tissue Dynamics Underlying Zebrafish Fin Fold Regeneration. PLoS ONE 2012, 7, e51766. [Google Scholar] [CrossRef] [Green Version]
- Somerville, G.; Beres, S.B.; Fitzgerald, J.R.; DeLeo, F.; Cole, R.L.; Hoff, J.S.; Musser, J.M. In Vitro Serial Passage of Staphylococcus aureus: Changes in Physiology, Virulence Factor Production, and agr Nucleotide Sequence. J. Bacteriol. 2002, 184, 1430–1437. [Google Scholar] [CrossRef] [Green Version]
- Tsuchiya, S.; Yamabe, M.; Yamaguchi, Y.; Kobayashi, Y.; Konno, T.; Tada, K. Establishment and characterization of a human acute monocytic leukemia cell line (THP-1). Int. J. Cancer 1980, 26, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Stokes, R.W.; Doxsee, D. The Receptor-Mediated Uptake, Survival, Replication, and Drug Sensitivity of Mycobacterium tuberculosis within the Macrophage-like Cell Line THP-1: A Comparison with Human Monocyte-Derived Macrophages. Cell. Immunol. 1999, 197, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Boukamp, P.; Petrussevska, R.T.; Breitkreutz, D.; Hornung, J.; Markham, A.; Fusenig, N.E. Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J. Cell Biol. 1988, 106, 761–771. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Yang, J.; Yu, J.; Yao, Z.; Sun, L.; Shen, Y.; Jin, Q. VFDB: A reference database for bacterial virulence factors. Nucleic Acids Res. 2005, 33, D325–D328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zankari, E.; Hasman, H.; Cosentino, S.; Vestergaard, M.; Rasmussen, S.; Lund, O.; Aarestrup, F.M.; Larsen, M.V. Identification of acquired antimicrobial resistance genes. J. Antimicrob. Chemother. 2012, 67, 2640–2644. [Google Scholar] [CrossRef] [PubMed]
- Bortolaia, V.; Kaas, R.S.; Ruppe, E.; Roberts, M.C.; Schwarz, S.; Cattoir, V.; Philippon, A.; Allesoe, R.L.; Rebelo, A.R.; Florensa, A.F.; et al. ResFinder 4.0 for predictions of phenotypes from genotypes. J. Antimicrob. Chemother. 2020, 75, 3491–3500. [Google Scholar] [CrossRef] [PubMed]
- Alikhan, N.-F.; Petty, N.K.; Ben Zakour, N.L.; Beatson, S.A. BLAST Ring Image Generator (BRIG): Simple prokaryote genome comparisons. BMC Genom. 2011, 12, 402. [Google Scholar] [CrossRef] [PubMed] [Green Version]

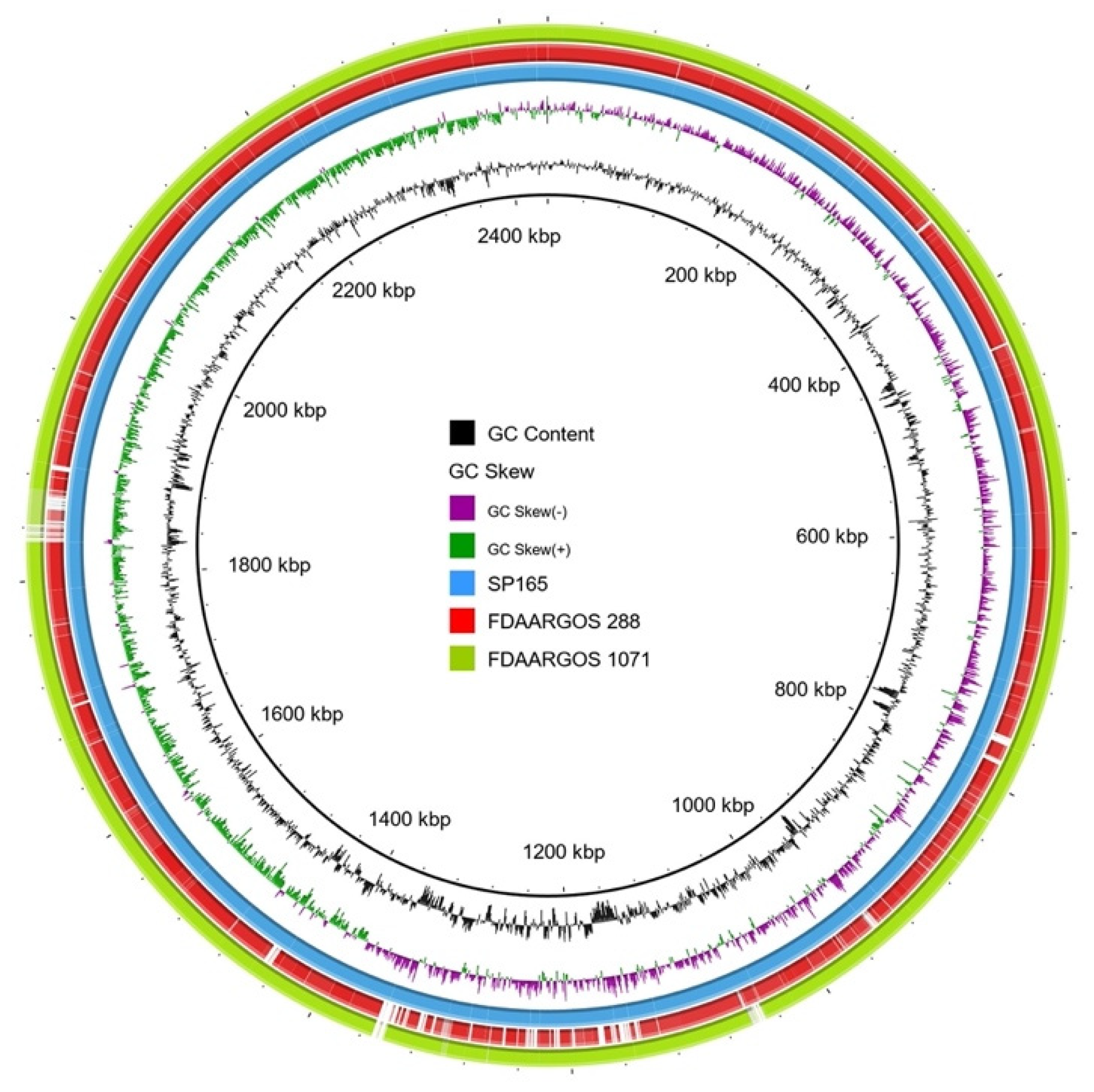
| Features | SP165 | FDAARGOS 288 | FDAARGOS 1071 |
|---|---|---|---|
| Biosample | SAMN22027592 | SAMN06173301 | SAMN16357240 |
| Assembly | SHOVILL 1.1.0 | CA v. 8.2 | SMRT v. 7.1.0, HGAP v. 4 |
| Genome size (bp) | 2,435,720 | 2,502,360 | 2,449,395 |
| Number contigs | 54 | ND | ND |
| GC Content (%) | 38.91 | 38.85 | 39.14 |
| Number CDS | 2298 | 2380 | 2318 |
| Year of collection | 2021 | 2014 | ND |
| Year of sequencing | 2021 | 2021 | 2020 |
| Locality | Nîmes (France) | Washington (USA) | Braunschweig (Germany) |
| Strain | Description | Resistance Profile | Reference |
|---|---|---|---|
| S. aureus SA564 | S. aureus clinical isolate, wild type | PEN | [56] |
| S. pettenkoferi SP165 | S. pettenkoferi clinical isolate from diabetic foot osteomyelitis, wild type | PEN, OXA, ERY, LIN, OFX, RIF, FOS | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad-Mansour, N.; Plumet, L.; Huc-Brandt, S.; Magnan, C.; Yahiaoui-Martinez, A.; Kissa, K.; Pantel, A.; Lavigne, J.-P.; Molle, V. Investigating Pathogenicity and Virulence of Staphylococcus pettenkoferi: An Emerging Pathogen. Int. J. Mol. Sci. 2021, 22, 13614. https://doi.org/10.3390/ijms222413614
Ahmad-Mansour N, Plumet L, Huc-Brandt S, Magnan C, Yahiaoui-Martinez A, Kissa K, Pantel A, Lavigne J-P, Molle V. Investigating Pathogenicity and Virulence of Staphylococcus pettenkoferi: An Emerging Pathogen. International Journal of Molecular Sciences. 2021; 22(24):13614. https://doi.org/10.3390/ijms222413614
Chicago/Turabian StyleAhmad-Mansour, Nour, Lucile Plumet, Sylvaine Huc-Brandt, Chloé Magnan, Alex Yahiaoui-Martinez, Karima Kissa, Alix Pantel, Jean-Philippe Lavigne, and Virginie Molle. 2021. "Investigating Pathogenicity and Virulence of Staphylococcus pettenkoferi: An Emerging Pathogen" International Journal of Molecular Sciences 22, no. 24: 13614. https://doi.org/10.3390/ijms222413614
APA StyleAhmad-Mansour, N., Plumet, L., Huc-Brandt, S., Magnan, C., Yahiaoui-Martinez, A., Kissa, K., Pantel, A., Lavigne, J.-P., & Molle, V. (2021). Investigating Pathogenicity and Virulence of Staphylococcus pettenkoferi: An Emerging Pathogen. International Journal of Molecular Sciences, 22(24), 13614. https://doi.org/10.3390/ijms222413614







