Ferroptosis in Non-Small Cell Lung Cancer: Progression and Therapeutic Potential on It
Abstract
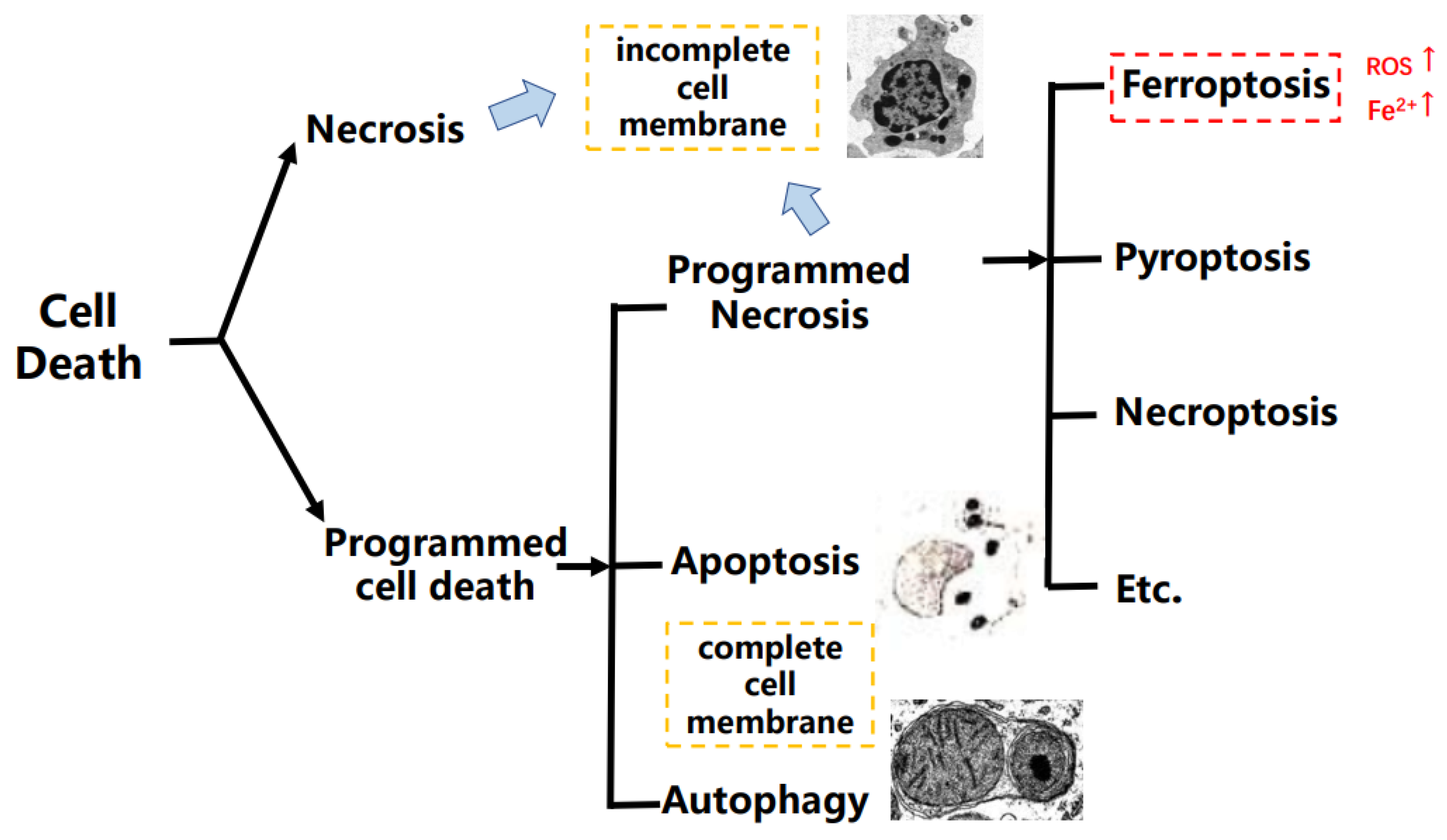
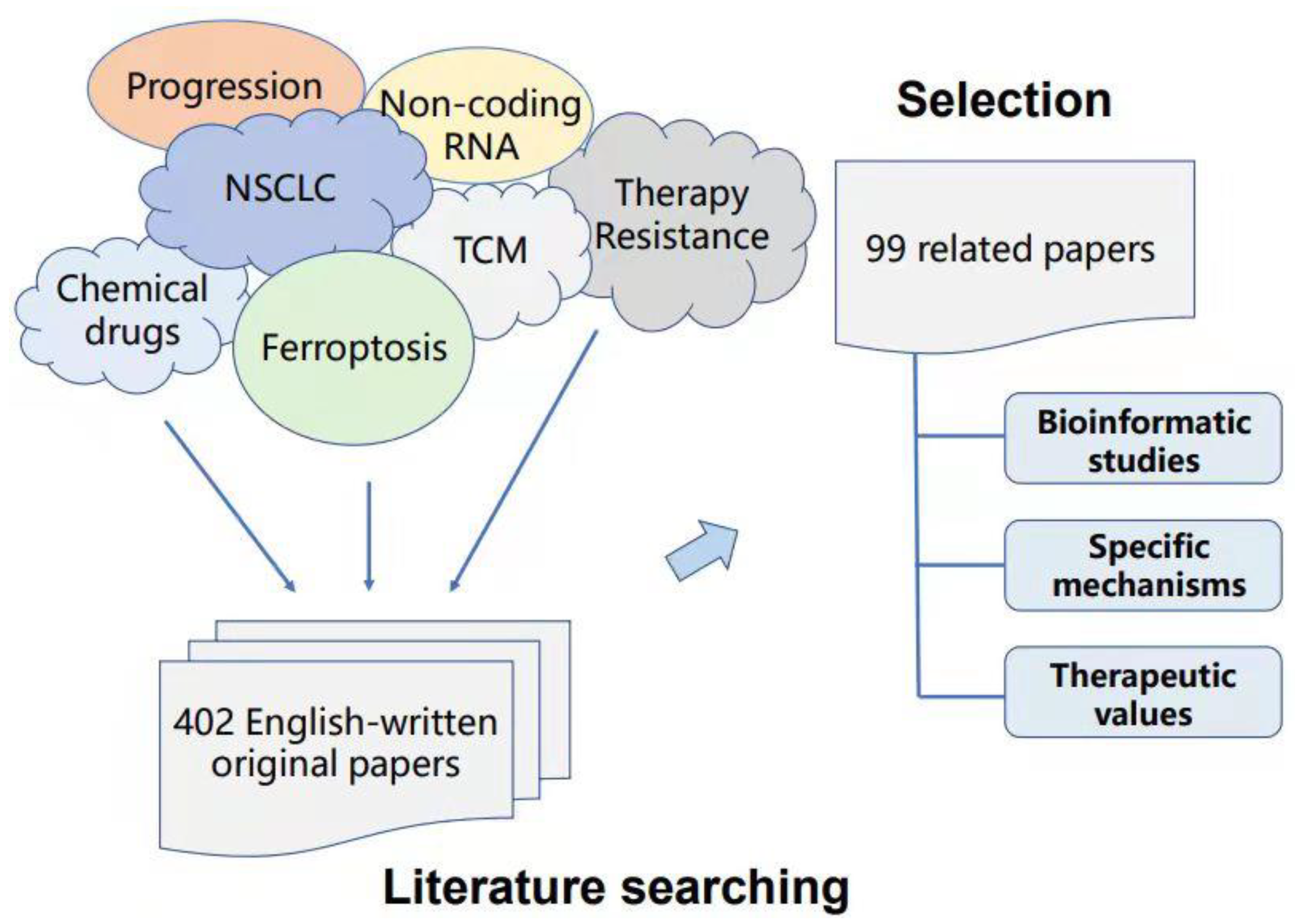
:1. Introduction
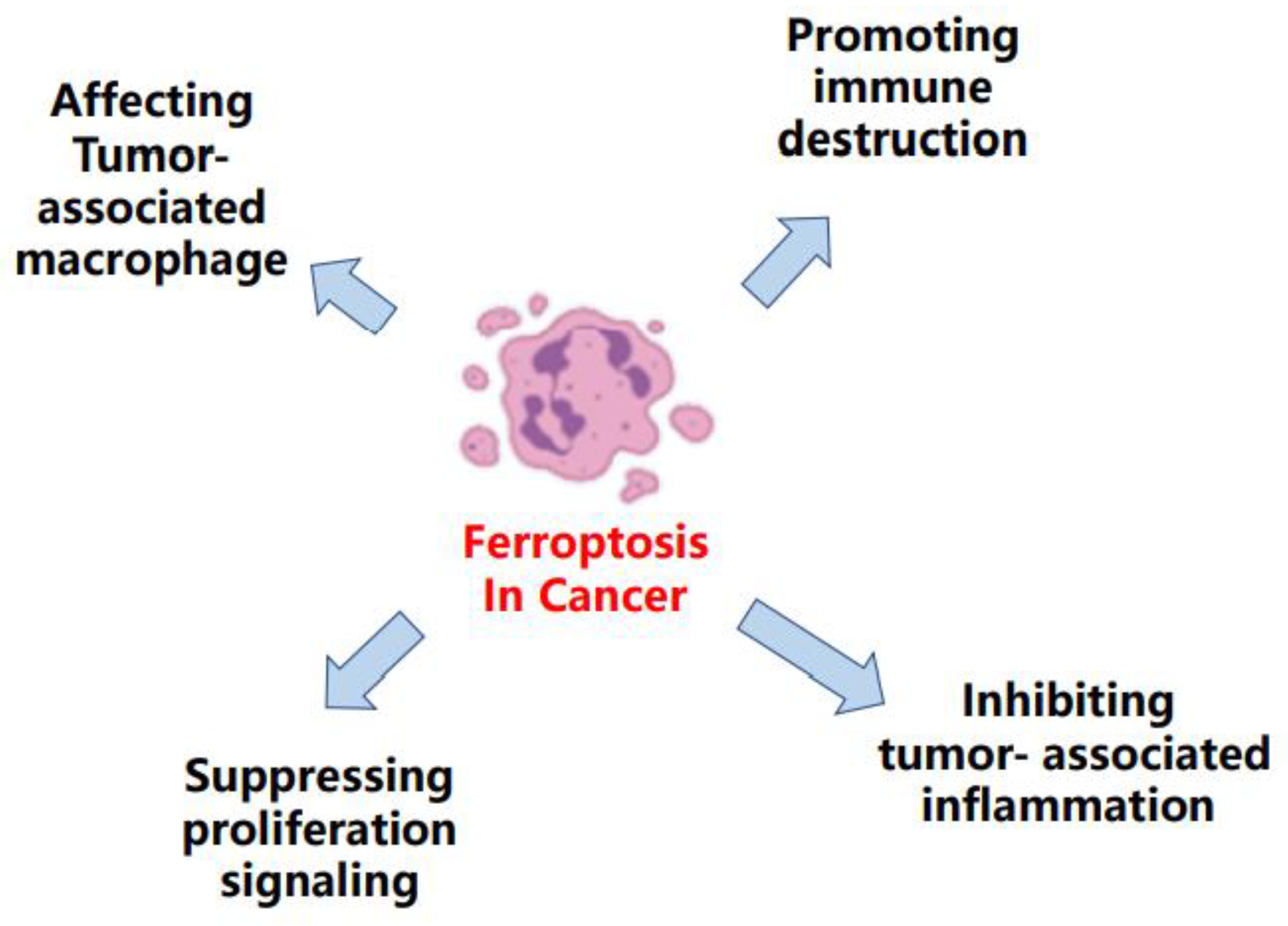
2. Ferroptosis in Cancer
2.1. Ferroptosis Was Observed in Diverse Cancers
2.2. Ferroptosis Could Be Triggered by Autophagy in Cancer
3. Mechanism of Ferroptosis in NSCLC
3.1. Brief Background of NSCLC
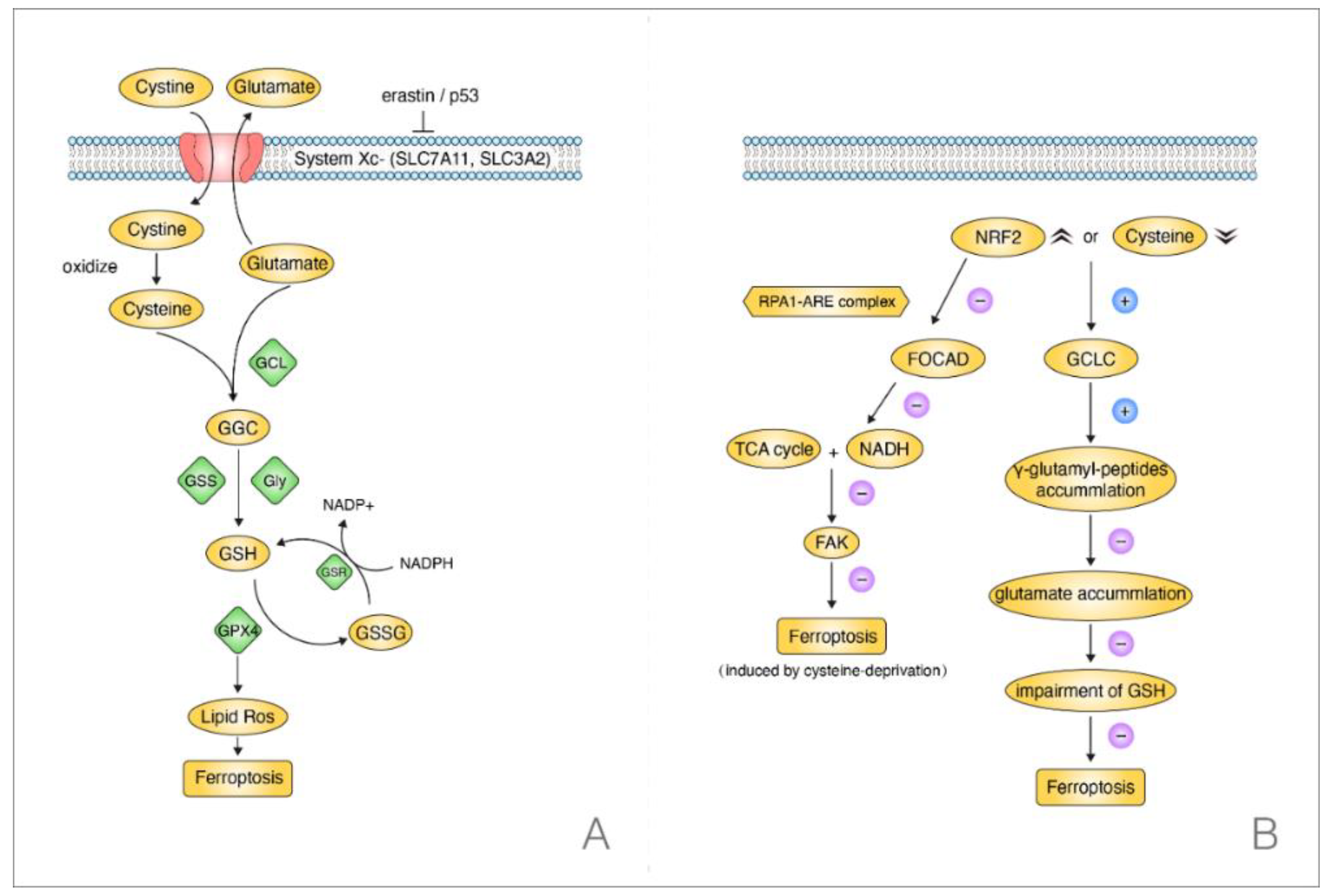
3.2. Current Discoveries on the Mechanisms of Ferroptosis in NSCLC
4. Specific Evidence Elucidating the Connection between Ferroptosis and NSCLC
4.1. Bioinformatics Implying the Relationship among Ferroptosis, NSCLC, and Clinical Immunotherapy
4.2. Non-Coding RNA Works as a Bridge Connecting Ferroptosis and NSCLC
4.2.1. Micro-RNA
4.2.2. LncRNA
4.3. Various Drugs Show A Regulatory Effect on Ferroptosis in NSCLC
4.3.1. Natural and Extracted Drugs
4.3.2. Chemical and Synthesized Drugs
5. Ferroptosis Works against Therapy Resistance of NSCLC
6. Conclusions and Discussions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AFC | Ammonium Ferric Citrate |
| ALK | Anaplastic lymphoma kinase |
| ARE | AU-rich element |
| ASCL4 | achaete-scute family bHLH transcription factor 4 |
| CBS | cellular cystathionine-beta-synthase |
| CCK-8 | cell counting kit-8 |
| circRNA | circular RNA |
| DEGs | differential expressed genes |
| EGFR | epithelial growth factor receptor |
| ETC | electron transport chain |
| FAK | focal adhesion kinase |
| FSP1 | ferroptosis suppressor protein 1 |
| GCL | glutamate-cysteine ligase |
| GCLC | γ-cysteine ligase catalytic subunit |
| GCO | Global Cancer Observatory |
| GEO | Gene Expression Omnibus |
| GGC | γ-Glutamylcysteine |
| GGT | γ-glutamyl transpeptadase |
| Gly | Glycine |
| GOT1 | cytosolic aspartate aminotransaminase |
| GPX4 | glutathione peroxidase 4 (phospholipid hydroperoxidase) |
| GSEA | gene set enrichment analysis |
| GSH | glutathione |
| GSS | glutamate synthetase |
| GSSG | L-Glutathione Oxidized |
| H&E staining | hematoxylin and eosin staining |
| HME cells | human mammary epithelial cells |
| HO-1 | Heme Oxygenase-1 |
| HP | haptoglobin |
| IHC | immunohistochemistry |
| IREB2 | iron-responsive element-binding protein 2 |
| Keap1 | kelch-like ECH-associated protein 1 |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| KRAS | kirsten rat sarcoma viral oncogene |
| LncRNA | long non-coding RNA |
| LUAD | Lung adenocarcinoma |
| LUSC | Lung squamous cell carcinoma |
| MDA | malondialdehyde |
| METTL3 | methyltransferase-like protein 3 |
| MREs | microRNA response elements |
| mTORC1 | mTOR complex 1 |
| NAC | N-Acetyl-L-cysteine |
| NFS1 | Cysteine desulfurase NFS1 |
| NRF2 | Subcellular localization of nuclear factor E2-related factor 2 |
| NSCLC | non-small cell lung cancer |
| PCD | programmed cell death |
| PD-1/PDL-1 | programmed cell death protein-1/programmed cell death protein-Ligand 1 |
| ROS | reactive oxygen species |
| SLC7A11 | solute carrier family 7, (cationic amino acid transporter, y+ system) member 11 |
| SLC3A2 | solute carrier family 3 (activators of dibasic and neutral amino acid transport), member 2 |
| SOD | superoxide dismutase |
| TCA | tricarboxylic acid cycle |
| TCGA | The Cancer Genome Atlas |
| TFRC | transferrin receptor |
| TKIs | tyrosine kinase inhibitors |
| TXN2 | thioredoxin 2 |
| VEGFA | vascular endothelial growth factor A |
| xCT | cystine/glutamate transporter |
| 3′-UTR′ | 3′-untranslational region |
References
- Bai, Y.; Lam, H.C.; Lei, X. Dissecting Programmed Cell Death with Small Molecules. Acc. Chem. Res. 2020, 53, 1034–1045. [Google Scholar] [CrossRef]
- Chen, X.; Zeh, H.J.; Kang, R.; Kroemer, G.; Tang, D. Cell death in pancreatic cancer: From pathogenesis to therapy. Nat. Rev. Gastroenterol. Hepatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhong, B.; Xiao, Q.; Du, L.; Hou, Y.; Sun, H.S.; Lu, J.J.; Chen, X. Induction of programmed necrosis: A novel anti-cancer strategy for natural compounds. Pharmacol. Ther. 2020, 214, 107593. [Google Scholar] [CrossRef]
- Man, S.M.; Karki, R.; Kanneganti, T.D. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol. Rev. 2017, 277, 61–75. [Google Scholar] [CrossRef] [Green Version]
- Khan, I.; Yousif, A.; Chesnokov, M.; Hong, L.; Chefetz, I. A decade of cell death studies: Breathing new life into necroptosis. Pharmacol. Ther. 2021, 220, 107717. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Minagawa, S.; Araya, J.; Sakamoto, T.; Hara, H.; Tsubouchi, K.; Hosaka, Y.; Ichikawa, A.; Saito, N.; Kadota, T.; et al. Involvement of cigarette smoke-induced epithelial cell ferroptosis in COPD pathogenesis. Nat. Commun. 2019, 10, 3145. [Google Scholar] [CrossRef] [PubMed]
- Kazan, K.; Kalaipandian, S. Ferroptosis: Yet Another Way to Die. Trends Plant Sci. 2019, 24, 479–481. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Kang, R.; Kroemer, G.; Tang, D. Broadening horizons: The role of ferroptosis in cancer. Nat. Rev. Clin. Oncol. 2021, 18, 280–296. [Google Scholar] [CrossRef]
- Schmitt, A.; Xu, W.; Bucher, P.; Grimm, M.; Konantz, M.; Horn, H.; Zapukhlyak, M.; Berning, P.; Brändle, M.; Jarboui, M.-A.; et al. Dimethyl fumarate induces ferroptosis and impairs NF-κB/STAT3 signaling in DLBCL. Blood 2021, 138, 871–884. [Google Scholar] [CrossRef]
- Zhang, H.; Deng, T.; Liu, R.; Ning, T.; Yang, H.; Liu, D.; Zhang, Q.; Lin, D.; Ge, S.; Bai, M.; et al. CAF secreted miR-522 suppresses ferroptosis and promotes acquired chemo-resistance in gastric cancer. Mol. Cancer 2020, 19, 43. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.F.; Niu, X.H.; Chen, R.C.; He, W.Y.; Chen, D.; Kang, R.; Tang, D.L. Metallothionein-1G Facilitates Sorafenib Resistance Through Inhibition of Ferroptosis. Hepatology 2016, 64, 488–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denton, D.; Chang, T.K.; Nicolson, S.; Shravage, B.; Simin, R.; Baehrecke, E.H.; Kumar, S. Relationship between growth arrest and autophagy in midgut programmed cell death in Drosophila. Cell Death Differ. 2012, 19, 1299–1307. [Google Scholar] [CrossRef] [Green Version]
- Emdad, L.; Qadeer, Z.A.; Bederson, L.B.; Kothari, H.P.; Uzzaman, M.; Germano, I.M. Is there a common upstream link for autophagic and apoptotic cell death in human high-grade gliomas? Neuro-Oncology 2011, 13, 725–735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kremer, D.M.; Nelson, B.S.; Lin, L.; Yarosz, E.L.; Halbrook, C.J.; Kerk, S.A.; Sajjakulnukit, P.; Myers, A.; Thurston, G.; Hou, S.W.; et al. GOT1 inhibition promotes pancreatic cancer cell death by ferroptosis. Nat. Commun. 2021, 12, 4860. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.H.; Monian, P.; Pan, Q.H.; Zhang, W.; Xiang, J.; Jiang, X.J. Ferroptosis is an autophagic cell death process. Cell Res. 2016, 26, 1021–1032. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Xu, S.; Zhang, L.; Cheng, X.; Yu, H.; Bao, J.; Lu, R. Vitamin C induces ferroptosis in anaplastic thyroid cancer cells by ferritinophagy activation. Biochem. Biophys. Res. Commun. 2021, 551, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Song, Z.; Liu, Y.J.; Ma, X.B.; Wang, W.; Ke, Y.; Xu, Y.C.; Yu, D.Q.; Liu, H.M. Identification of ferroptosis as a novel mechanism for antitumor activity of natural product derivative a2 in gastric cancer. Acta Pharm. Sin. B 2021, 11, 1513–1525. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, X.; Lu, S.; He, C.; Wang, C.; Wang, L.; Wang, X.; Ge, P.; Song, D. Erastin triggers autophagic death of breast cancer cells by increasing intracellular iron levels. Oncol. Lett. 2020, 20, 57. [Google Scholar] [CrossRef]
- Tang, X.; Ding, H.; Liang, M.; Chen, X.; Yan, Y.; Wan, N.; Chen, Q.; Zhang, J.; Cao, J. Curcumin induces ferroptosis in non-small-cell lung cancer via activating autophagy. Thorac. Cancer 2021, 12, 1219–1230. [Google Scholar] [CrossRef]
- Govindan, R.; Page, N.; Morgensztern, D.; Read, W.; Tierney, R.; Vlahiotis, A.; Spitznagel, E.L.; Piccirillo, J. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: Analysis of the surveillance, epidemiologic, and end results database. J. Clin. Oncol. 2006, 24, 4539–4544. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Abreu, D.; Powell, S.F.; Hochmair, M.J.; Gadgeel, S.; Esteban, E.; Felip, E.; Speranza, G.; De Angelis, F.; Domine, M.; Cheng, S.Y.; et al. Pemetrexed plus platinum with or without pembrolizumab in patients with previously untreated metastatic nonsquamous NSCLC: Protocol-specified final analysis from KEYNOTE-189. Ann. Oncol. 2021, 32, 881–895. [Google Scholar] [CrossRef]
- Gridelli, C.; Rossi, A.; Carbone, D.P.; Guarize, J.; Karachaliou, N.; Mok, T.; Petrella, F.; Spaggiari, L.; Rosell, R. Non-small-cell lung cancer. Nat. Rev. Dis. Primers 2015, 1, 15009. [Google Scholar] [CrossRef]
- Mayne, N.R.; Lin, B.K.; Darling, A.J.; Raman, V.; Patel, D.C.; Liou, D.Z.; D’Amico, T.A.; Yang, C.J. Stereotactic Body Radiotherapy Versus Delayed Surgery for Early-stage Non-small-cell Lung Cancer. Ann. Surg. 2020, 272, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Gridelli, C.; Morabito, A.; Cavanna, L.; Luciani, A.; Maione, P.; Bonanno, L.; Filipazzi, V.; Leo, S.; Cinieri, S.; Ciardiello, F.; et al. Cisplatin-Based First-Line Treatment of Elderly Patients With Advanced Non-Small-Cell Lung Cancer: Joint Analysis of MILES-3 and MILES-4 Phase III Trials. J. Clin. Oncol. 2018, 36, 2585–2592. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Garon, E.B.; Kim, D.W.; Cho, B.C.; Perez-Gracia, J.L.; Han, J.Y.; Arvis, C.D.; Majem, M.; Forster, M.D.; Monnet, I.; et al. Long-Term Outcomes and Retreatment Among Patients With Previously Treated, Programmed Death-Ligand 1Positive, Advanced NonSmall-Cell Lung Cancer in the KEYNOTE-010 Study. J. Clin. Oncol. 2020, 38, 1580–1590. [Google Scholar] [CrossRef]
- Mok, T.S.K.; Wu, Y.L.; Kudaba, I.; Kowalski, D.M.; Cho, B.C.; Turna, H.Z.; Castro, G., Jr.; Srimuninnimit, V.; Laktionov, K.K.; Bondarenko, I.; et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): A randomised, open-label, controlled, phase 3 trial. Lancet 2019, 393, 1819–1830. [Google Scholar] [CrossRef]
- Rittmeyer, A.; Barlesi, F.; Waterkamp, D.; Park, K.; Ciardiello, F.; von Pawel, J.; Gadgeel, S.M.; Hida, T.; Kowalski, D.M.; Dols, M.C.; et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): A phase 3, open-label, multicentre randomised controlled trial. Lancet 2017, 389, 255–265. [Google Scholar] [CrossRef]
- Wang, M.; Herbst, R.S.; Boshoff, C. Toward personalized treatment approaches for non-small-cell lung cancer. Nat. Med. 2021, 27, 1345–1356. [Google Scholar] [CrossRef]
- Patel, S.A.; Herynk, M.H.; Cascone, T.; Saigal, B.; Nilsson, M.B.; Tran, H.; Ramachandran, S.; Diao, L.; Wang, J.; Le, X.; et al. Estrogen Promotes Resistance to Bevacizumab in Murine Models of NSCLC. J. Thorac. Oncol. 2021. [Google Scholar] [CrossRef]
- Koga, T.; Suda, K.; Fujino, T.; Ohara, S.; Hamada, A.; Nishino, M.; Chiba, M.; Shimoji, M.; Takemoto, T.; Arita, T.; et al. KRAS Secondary Mutations That Confer Acquired Resistance to KRAS G12C Inhibitors, Sotorasib and Adagrasib, and Overcoming Strategies: Insights From In Vitro Experiments. J. Thorac. Oncol. 2021, 16, 1321–1332. [Google Scholar] [CrossRef]
- Sugawara, S.; Lee, J.S.; Kang, J.H.; Kim, H.R.; Inui, N.; Hida, T.; Lee, K.H.; Yoshida, T.; Tanaka, H.; Yang, C.T.; et al. Nivolumab with carboplatin, paclitaxel, and bevacizumab for first-line treatment of advanced nonsquamous non-small-cell lung cancer. Ann. Oncol. 2021, 32, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Peng, D.H.; Rodriguez, B.L.; Diao, L.; Gaudreau, P.O.; Padhye, A.; Konen, J.M.; Ochieng, J.K.; Class, C.A.; Fradette, J.J.; Gibson, L.; et al. Th17 cells contribute to combination MEK inhibitor and anti-PD-L1 therapy resistance in KRAS/p53 mutant lung cancers. Nat. Commun. 2021, 12, 2606. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-R.; Kim, T.M.; Lee, Y.; Kim, S.; Park, S.; Ju, Y.S.; Kim, M.; Keam, B.; Jeon, Y.K.; Kim, D.-W.; et al. Acquired Resistance to Third-Generation EGFR Tyrosine Kinase Inhibitors in Patients With De Novo EGFRT790M-Mutant NSCLC. J. Thorac. Oncol. 2021, 16, 1859–1871. [Google Scholar] [CrossRef] [PubMed]
- Felip, E.; Shaw, A.T.; Bearz, A.; Camidge, D.R.; Solomon, B.J.; Bauman, J.R.; Bauer, T.M.; Peters, S.; Toffalorio, F.; Abbattista, A.; et al. Intracranial and extracranial efficacy of lorlatinib in patients with ALK-positive non-small-cell lung cancer previously treated with second-generation ALK TKIs. Ann. Oncol. 2021, 32, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Tsakiridis, T.; Pond, G.R.; Wright, J.; Ellis, P.M.; Ahmed, N.; Abdulkarim, B.; Roa, W.; Robinson, A.; Swaminath, A.; Okawara, G.; et al. Metformin in Combination With Chemoradiotherapy in Locally Advanced Non-Small Cell Lung Cancer: The OCOG-ALMERA Randomized Clinical Trial. JAMA Oncol. 2021, 7, 1333. [Google Scholar] [CrossRef]
- Cao, L.; Hong, W.P.; Cai, P.H.; Xu, C.C.; Bai, X.P.; Zhao, Z.X.; Huang, M.; Jin, J. Cryptotanshinone strengthens the effect of gefitinib against non-small cell lung cancer through inhibiting transketolase. Eur. J. Pharmacol. 2021, 890, 173647. [Google Scholar] [CrossRef]
- Guohua, F.; Tieyuan, Z.; Xinping, M.; Juan, X. Melatonin protects against PM2.5-induced lung injury by inhibiting ferroptosis of lung epithelial cells in a Nrf2-dependent manner. Ecotoxicol. Environ. Saf. 2021, 223, 112588. [Google Scholar] [CrossRef]
- Tang, W.; Dong, M.; Teng, F.; Cui, J.; Zhu, X.; Wang, W.; Wuniqiemu, T.; Qin, J.; Yi, L.; Wang, S.; et al. Environmental allergens house dust mite-induced asthma is associated with ferroptosis in the lungs. Exp. Ther. Med. 2021, 22, 1483. [Google Scholar] [CrossRef]
- Yin, X.; Zhu, G.; Wang, Q.; Fu, Y.D.; Wang, J.; Xu, B. Ferroptosis, a New Insight Into Acute Lung Injury. Front. Pharmacol. 2021, 12, 709538. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Li, Z.; Yu, Z.; Li, J.; Zhang, J.; Wan, N.; Zhang, J.; Cao, J. Effect of curcumin on lung epithelial injury and ferroptosis induced by cigarette smoke. Hum. Exp. Toxicol. 2021, 9603271211059497. [Google Scholar] [CrossRef]
- Wu, S.; Zhu, C.; Tang, D.; Dou, Q.P.; Shen, J.; Chen, X. The role of ferroptosis in lung cancer. Biomark. Res. 2021, 9, 82. [Google Scholar] [CrossRef]
- Wei, D.; Duan, B.X.; Wang, C.Y.; Cao, P. MicroRNA-302a-3p induces ferroptosis of non-small cell lung cancer cells via targeting ferroportin. Free Radic. Res. 2021, 55, 821–830. [Google Scholar] [CrossRef]
- Song, Z.; Jia, G.; Ma, P.; Cang, S. Exosomal miR-4443 promotes cisplatin resistance in non-small cell lung carcinoma by regulating FSP1 m6A modification-mediated ferroptosis. Life Sci. 2021, 276, 119399. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.H.; Wu, D.M.; Li, L.; Liu, T.; Zhang, T.; Li, J.; Yu, Y.; He, M.; Zhao, Y.Y.; Han, R.; et al. miR-324-3p reverses cisplatin resistance by inducing GPX4-mediated ferroptosis in lung adenocarcinoma cell line A549. Biochem. Biophys. Res. Commun. 2021, 549, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Liu, A. Long non-coding RNA NEAT1 regulates ferroptosis sensitivity in non-small-cell lung cancer. J. Int. Med. Res. 2021, 49, 300060521996183. [Google Scholar] [CrossRef] [PubMed]
- Gai, C.; Liu, C.; Wu, X.; Yu, M.; Zheng, J.; Zhang, W.; Lv, S.; Li, W. MT1DP loaded by folate-modified liposomes sensitizes erastin-induced ferroptosis via regulating miR-365a-3p/NRF2 axis in non-small cell lung cancer cells. Cell Death Dis. 2020, 11, 751. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Mao, C.; Ouyang, L.; Liu, Y.; Lai, W.; Liu, N.; Shi, Y.; Chen, L.; Xiao, D.; Yu, F.; et al. Long noncoding RNA LINC00336 inhibits ferroptosis in lung cancer by functioning as a competing endogenous RNA. Cell Death Differ. 2019, 26, 2329–2343. [Google Scholar] [CrossRef]
- Zhang, Q.; Yi, H.; Yao, H.; Lu, L.; He, G.; Wu, M.; Zheng, C.; Li, Y.; Chen, S.; Li, L.; et al. Artemisinin Derivatives Inhibit Non-small Cell Lung Cancer Cells Through Induction of ROS-dependent Apoptosis/Ferroptosis. J. Cancer 2021, 12, 4075–4085. [Google Scholar] [CrossRef]
- Tian, Y.Y.; Yang, A.L.; Chen, X.N.; Ren, H.M.; Liu, Y.X.; Qiu, H.L.; Tang, L.M.; Huang, H.M.; Tu, P.F.; Hu, Z.D. Effect of Huaier aqueous extract on growth and metastasis of human non-small cell lung cancer NCI-H1299 cells and its underlying mechanisms. Zhongguo Zhong Yao Za Zhi 2020, 45, 3700–3706. [Google Scholar] [CrossRef] [PubMed]
- Palmer, L.D.; Jordan, A.T.; Maloney, K.N.; Farrow, M.A.; Gutierrez, D.B.; Gant-Branum, R.; Burns, W.J.; Romer, C.E.; Tsui, T.; Allen, J.L.; et al. Zinc intoxication induces ferroptosis in A549 human lung cells. Metallomics 2019, 11, 982–993. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.S.; Zhao, L.P.; Huang, Z.H.; Chen, X.Y.; Xu, J.T.; Tai, W.C.; Tsim, K.W.K.; Chen, Y.T.; Xie, T. Ginkgetin derived from Ginkgo biloba leaves enhances the therapeutic effect of cisplatin via ferroptosis-mediated disruption of the Nrf2/HO-1 axis in EGFR wild-type non-small-cell lung cancer. Phytomedicine 2021, 80, 153370. [Google Scholar] [CrossRef]
- Wu, W.; Geng, Z.; Bai, H.; Liu, T.; Zhang, B. Ammonium Ferric Citrate induced Ferroptosis in Non-Small-Cell Lung Carcinoma through the inhibition of GPX4-GSS/GSR-GGT axis activity. Int. J. Med. Sci. 2021, 18, 1899–1909. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Xu, B.; Han, Q.; Zhou, H.; Xia, Y.; Gong, C.; Dai, X.; Li, Z.; Wu, G. Ferroptosis: A Novel Anti-tumor Action for Cisplatin. Cancer Res. Treat. 2018, 50, 445–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Yan, H.; Xu, X.; Liu, H.; Wu, C.; Zhao, L. Erastin/sorafenib induces cisplatin-resistant non-small cell lung cancer cell ferroptosis through inhibition of the Nrf2/xCT pathway. Oncol. Lett. 2020, 19, 323–333. [Google Scholar] [CrossRef] [Green Version]
- Pan, X.; Lin, Z.; Jiang, D.; Yu, Y.; Yang, D.; Zhou, H.; Zhan, D.; Liu, S.; Peng, G.; Chen, Z.; et al. Erastin decreases radioresistance of NSCLC cells partially by inducing GPX4-mediated ferroptosis. Oncol. Lett. 2019, 17, 3001–3008. [Google Scholar] [CrossRef] [Green Version]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.; Yang, M.; Deng, J.; Li, P.; Su, W.; Jiang, R. Upregulation and activation of p53 by erastin induced reactive oxygen species contribute to cytotoxic and cytostatic effects in A549 lung cancer cells. Oncol. Rep. 2018, 40, 2363–2370. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Zhao, W.; Li, X.; Wang, L.; Meng, L.; Yu, R. Cisplatin synergizes with PRLX93936 to induce ferroptosis in non-small cell lung cancer cells. Biochem. Biophys. Res. Commun. 2021, 569, 79–85. [Google Scholar] [CrossRef]
- Gai, C.; Yu, M.; Li, Z.; Wang, Y.; Ding, D.; Zheng, J.; Lv, S.; Zhang, W.; Li, W. Acetaminophen sensitizing erastin-induced ferroptosis via modulation of Nrf2/heme oxygenase-1 signaling pathway in non-small-cell lung cancer. J. Cell Physiol. 2020, 235, 3329–3339. [Google Scholar] [CrossRef]
- Yu, H.; Han, Z.; Xu, Z.; An, C.; Xu, L.; Xin, H. RNA sequencing uncovers the key long non-coding RNAs and potential molecular mechanism contributing to XAV939-mediated inhibition of non-small cell lung cancer. Oncol. Lett. 2019, 17, 4994–5004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, M.; Huang, M.; Liu, C.; Wang, J.; Ren, W.; Cui, S.; Gu, J.; Xie, J.; Ma, B.; Yang, G.; et al. Local anesthetic levobupivacaine induces ferroptosis and inhibits progression by up-regulating p53 in non-small cell lung cancer. Aging 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Freire Boullosa, L.; Van Loenhout, J.; Flieswasser, T.; De Waele, J.; Hermans, C.; Lambrechts, H.; Cuypers, B.; Laukens, K.; Bartholomeus, E.; Siozopoulou, V.; et al. Auranofin reveals therapeutic anticancer potential by triggering distinct molecular cell death mechanisms and innate immunity in mutant p53 non-small cell lung cancer. Redox. Biol. 2021, 42, 101949. [Google Scholar] [CrossRef] [PubMed]
- Shui, S.; Zhao, Z.; Wang, H.; Conrad, M.; Liu, G. Non-enzymatic lipid peroxidation initiated by photodynamic therapy drives a distinct ferroptosis-like cell death pathway. Redox. Biol. 2021, 45, 102056. [Google Scholar] [CrossRef] [PubMed]
- Kain, H.S.; Glennon, E.K.K.; Vijayan, K.; Arang, N.; Douglass, A.N.; Fortin, C.L.; Zuck, M.; Lewis, A.J.; Whiteside, S.L.; Dudgeon, D.R.; et al. Liver stage malaria infection is controlled by host regulators of lipid peroxidation. Cell Death Differ. 2020, 27, 44–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, D.; Kroemer, G. Ferroptosis. Curr. Biol. 2020, 30, R1292–R1297. [Google Scholar] [CrossRef]
- Alvarez, S.W.; Sviderskiy, V.O.; Terzi, E.M.; Papagiannakopoulos, T.; Moreira, A.L.; Adams, S.; Sabatini, D.M.; Birsoy, K.; Possemato, R. NFS1 undergoes positive selection in lung tumours and protects cells from ferroptosis. Nature 2017, 551, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.P.; Mockabee-Macias, A.; Jiang, C.; Falzone, A.; Prieto-Farigua, N.; Stone, E.; Harris, I.S.; DeNicola, G.M. Non-canonical Glutamate-Cysteine Ligase Activity Protects against Ferroptosis. Cell Metab. 2021, 33, 174–189. [Google Scholar] [CrossRef]
- Liu, P.; Wu, D.; Duan, J.; Xiao, H.; Zhou, Y.; Zhao, L.; Feng, Y. NRF2 regulates the sensitivity of human NSCLC cells to cysteine deprivation-induced ferroptosis via FOCAD-FAK signaling pathway. Redox. Biol. 2020, 37, 101702. [Google Scholar] [CrossRef]
- Poursaitidis, I.; Wang, X.; Crighton, T.; Labuschagne, C.; Mason, D.; Cramer, S.L.; Triplett, K.; Roy, R.; Pardo, O.E.; Seckl, M.J.; et al. Oncogene-Selective Sensitivity to Synchronous Cell Death following Modulation of the Amino Acid Nutrient Cystine. Cell Rep. 2017, 18, 2547–2556. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Su, G.H.; Ma, D.; Xiao, Y.; Shao, Z.M.; Jiang, Y.Z. Technological advances in cancer immunity: From immunogenomics to single-cell analysis and artificial intelligence. Signal. Transduct. Target. Ther. 2021, 6, 312. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Yang, J.; Ma, C.; Li, F.; Luo, H. Development and Validation of a Robust Ferroptosis-Related Prognostic Signature in Lung Adenocarcinoma. Front. Cell Dev. Biol. 2021, 9, 616271. [Google Scholar] [CrossRef]
- Liu, T.; Luo, H.; Zhang, J.; Hu, X.; Zhang, J. Molecular identification of an immunity- and Ferroptosis-related gene signature in non-small cell lung Cancer. BMC Cancer 2021, 21, 783. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Wang, J.; Qian, K.; Zhao, T.; Liu, X.; Zhang, Y. Construction of a prognostic model for non-small-cell lung cancer based on ferroptosis-related genes. Biosci. Rep. 2021, 41, 41. [Google Scholar] [CrossRef]
- Mohapatra, S.; Pioppini, C.; Ozpolat, B.; Calin, G.A. Non-coding RNAs regulation of macrophage polarization in cancer. Mol. Cancer 2021, 20, 24. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Lim, J.; Dale, J.M.; Chen, H.; Shinn, P.; Palm, C.J.; Southwick, A.M.; Wu, H.C.; Kim, C.; Nguyen, M.; et al. Empirical analysis of transcriptional activity in the Arabidopsis genome. Science 2003, 302, 842–846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, Y.; Liao, H.; Pu, Q.; Ke, X.X.; Hu, X.T.; Ma, Y.F.; Luo, X.M.; Jiang, Q.Q.; Gong, Y.; Wu, M.; et al. miR-410 induces both epithelial-mesenchymal transition and radioresistance through activation of the PI3K/mTOR pathway in non-small cell lung cancer. Signal Transduct. Target. Ther. 2020, 5, 1–14. [Google Scholar] [CrossRef]
- Zhang, N.; Nan, A.R.; Chen, L.J.; Li, X.; Jia, Y.Y.; Qiu, M.Y.; Dai, X.; Zhou, H.Y.; Zhu, J.L.; Zhang, H.; et al. Circular RNA circSATB2 promotes progression of non-small cell lung cancer cells. Mol. Cancer 2020, 19, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, A.; Wang, Z.; Wang, B.; Chai, X.; Lu, W.; Cao, T.; Li, R.; Wu, M.; Lu, Z.; et al. LINC00173.v1 promotes angiogenesis and progression of lung squamous cell carcinoma by sponging miR-511-5p to regulate VEGFA expression. Mol. Cancer 2020, 19, 98. [Google Scholar] [CrossRef]
- Peng, F.; Xiong, L.; Peng, C. (-)-Sativan Inhibits Tumor Development and Regulates miR-200c/PD-L1 in Triple Negative Breast Cancer Cells. Front. Pharmacol. 2020, 11, 12. [Google Scholar] [CrossRef]
- Peng, F.; Tang, H.; Du, J.; Chen, J.; Peng, C. Isoliquiritigenin Suppresses EMT-Induced Metastasis in Triple-Negative Breast Cancer through miR-200c/C-JUN/ Formula: See text -Catenin. Am. J. Chin. Med. 2021, 49, 505–523. [Google Scholar] [CrossRef]
- Peng, F.; Xie, X.F.; Peng, C. Chinese Herbal Medicine-Based Cancer Therapy: Novel Anticancer Agents Targeting MicroRNAs to Regulate Tumor Growth and Metastasis. Am. J. Chin. Med. 2019, 47, 1711–1735. [Google Scholar] [CrossRef] [PubMed]
- Salmena, L.; Poliseno, L.; Tay, Y.; Kats, L.; Pandolfi, P.P. A ceRNA hypothesis: The Rosetta Stone of a hidden RNA language? Cell 2011, 146, 353–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Peng, F.; Peng, C.; Du, J.-R. Gut Microbiota in Tumor Microenvironment: A Critical Regulator in Cancer Initiation and Development as Potential Targets for Chinese Medicine. Am. J. Chin. Med. 2021, 49, 609–626. [Google Scholar] [CrossRef]
- Peng, F.; Xiong, L.; Xie, X.F.; Tang, H.L.; Huang, R.Z.; Peng, C. Isoliquiritigenin Derivative Regulates miR-374a/BAX Axis to Suppress Triple-Negative Breast Cancer Tumorigenesis and Development. Front. Pharmacol. 2020, 11, 11. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.; Li, J.L.; Wang, Q.W.; Zou, W.L.; Liu, Y.Q.; Jia, Z.Q.; Peng, F.; Han, B. Regiodivergent construction of medium-sized heterocycles from vinylethylene carbonates and allylidenemalononitriles. Chem. Sci. 2020, 11, 2888–2894. [Google Scholar] [CrossRef] [Green Version]
- Chen, G.Q.; Benthani, F.A.; Wu, J.; Liang, D.; Bian, Z.X.; Jiang, X. Artemisinin compounds sensitize cancer cells to ferroptosis by regulating iron homeostasis. Cell Death Differ. 2020, 27, 242–254. [Google Scholar] [CrossRef]
- Zhang, Q.; Qiao, H.; Wu, D.; Lu, H.; Liu, L.; Sang, X.; Li, D.; Zhou, Y. Curcumin potentiates the galbanic acid-induced anti-tumor effect in non-small cell lung cancer cells through inhibiting Akt/mTOR signaling pathway. Life Sci. 2019, 239, 117044. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Qiang, P.; Yu, J.; Miao, Y.; Chen, Z.; Qu, J.; Zhao, Q.; Chen, Z.; Liu, Y.; Yao, X.; et al. Identification of compound CA-5f as a novel late-stage autophagy inhibitor with potent anti-tumor effect against non-small cell lung cancer. Autophagy 2019, 15, 391–406. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Gao, S.; Yang, Y.; Zhao, X.; Fan, Y.; Ma, W.; Yang, D.; Yang, A.; Yu, Y. Antitumor activity of curcumin by modulation of apoptosis and autophagy in human lung cancer A549 cells through inhibiting PI3K/Akt/mTOR pathway. Oncol. Rep. 2018, 39, 1523–1531. [Google Scholar] [CrossRef]
- Liu, M.; Fan, Y.; Li, D.; Han, B.; Meng, Y.; Chen, F.; Liu, T.; Song, Z.; Han, Y.; Huang, L.; et al. Ferroptosis inducer erastin sensitizes NSCLC cells to celastrol through activation of the ROS-mitochondrial fission-mitophagy axis. Mol. Oncol. 2021, 15, 2084–2105. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Yang, J.; Zhao, G.; Shen, Z.; Yang, K.; Tian, L.; Zhou, Q.; Chen, Y.; Huang, Y. Dysregulation of ferroptosis may involve in the development of non-small-cell lung cancer in Xuanwei area. J. Cell Mol. Med. 2021, 25, 2872–2884. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Kon, N.; Li, T.; Wang, S.J.; Su, T.; Hibshoosh, H.; Baer, R.; Gu, W. Ferroptosis as a p53-mediated activity during tumour suppression. Nature 2015, 520, 57–62. [Google Scholar] [CrossRef] [Green Version]
- Zheng, D.W.; Lei, Q.; Zhu, J.Y.; Fan, J.X.; Li, C.X.; Li, C.; Xu, Z.; Cheng, S.X.; Zhang, X.Z. Switching Apoptosis to Ferroptosis: Metal-Organic Network for High-Efficiency Anticancer Therapy. Nano Lett. 2017, 17, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhao, M.; Cui, L.; Ren, Y.; Zhang, J.; Chen, J.; Jia, L.; Zhang, J.; Yang, J.; Chen, G.; et al. Characterization of a novel HDAC/RXR/HtrA1 signaling axis as a novel target to overcome cisplatin resistance in human non-small cell lung cancer. Mol. Cancer 2020, 19, 134. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Chen, K.; Zhang, J.; Zhang, X. Inhibition of GPX4 or mTOR overcomes resistance to Lapatinib via promoting ferroptosis in NSCLC cells. Biochem. Biophys. Res. Commun. 2021, 567, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Green, M.; Choi, J.E.; Gijon, M.; Kennedy, P.D.; Johnson, J.K.; Liao, P.; Lang, X.; Kryczek, I.; Sell, A.; et al. CD8(+) T cells regulate tumour ferroptosis during cancer immunotherapy. Nature 2019, 569, 270–274. [Google Scholar] [CrossRef]
- Kim, S.E.; Zhang, L.; Ma, K.; Riegman, M.; Chen, F.; Ingold, I.; Conrad, M.; Turker, M.Z.; Gao, M.; Jiang, X.; et al. Ultrasmall nanoparticles induce ferroptosis in nutrient-deprived cancer cells and suppress tumour growth. Nat. Nanotechnol. 2016, 11, 977–985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, J.; Luo, T.; Wang, J. Gene interfered-ferroptosis therapy for cancers. Nat. Commun. 2021, 12, 5311. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Wu, X.; Zhao, G.; Feng, K.; Ni, K.; Sun, X. Renal Clearable Ultrasmall Single-Crystal Fe Nanoparticles for Highly Selective and Effective Ferroptosis Therapy and Immunotherapy. J. Am. Chem. Soc. 2021, 143, 15812–15823. [Google Scholar] [CrossRef]
| Category | Molecular/Drug | Regulation Effect on Ferroptosis in NSCLC | Brief Mechanism | Techniques Used for Detection of Ferroptosis | Function Study | Reference |
|---|---|---|---|---|---|---|
| Micro-RNA | miR-302a-3p | ↑ | Directly bind ferroportin; Increase the lipid peroxidation and cellular iron level | _ | In vitro | [42] |
| miR-4443 | ↓ | Directly bind METTL3 + Regulate FSP1 in a m6A manner | RT-qPCR; Iron assay kit; Fluorescence staining; Xenograft model; H&E staining; Immunohistochemistry (IHC) | In vitro and In vivo | [43] | |
| miR-324-3p | ↑ | Directly bind GPX4, working as a GPX4 inhibitor | Annexin V/PI staining; Western blot; Luciferase report | In vitro | [44] | |
| LncRNA | NEAT1 | ↑ | Bind ACSL4 to Regulate SCL7A11 and GPX4 | RT-PCR; Western blot; Dual-luciferase reporter gene assay; Lipid Peroxidation MDA Assay Kit (Beyotime Biotechnology); Iron assay kit | In vitro | [45] |
| MT1DP | ↑ | Negatively regulate NFR2; Increase the Fe2+, ROS, and MDA level; Decrease the GSH level | Dichlorofluorescein diacetate fluorescent probe detection kit; Iron colorimetric assay kit; qRT-PCR; Western blot; xenografts; IHC | In vitro and In vivo | [46] | |
| LINC00336 | ↓ | Bind ELAVL1; Compete MIR6852 to regulate CBS level | Lipid ROS assays; Iron Assay Kit; RT-qPCR; Immunoblotting; | In vitro | [47] | |
| natural & extracted drugs | Artemisinin derivatives ART and DHA | ↑ | Downregulate xCT; Upregulate TFRC; | Western blot; RT-qPCR; Reactive oxygen species analysed by flow cytometry | In vitro | [48] |
| Extract from Huaier aqueous | ↑ | Upregulate cellular ROS level | Flow cytometry was used to examine the ROS level; Western blot | In vitro | [49] | |
| Zinc (high concentration) | ↑ | Decrease GSH; Increase GSSG | Fluorescence staining | In vitro | [50] | |
| Curcumin | ↑ | Ferroptosis is triggered by autophagy; Ferrostatin-1, chloroquine, and silencing IREB2 or Beclin1 could all reverse it. | Intracellular ROS, GSH, and iron contents were determined by corresponding assay kit | In vitro and In vivo | [19] | |
| Ginkgetin | ↑ | Ferroptosis is triggered by autophagy; Decrease SLC7A11 and GPX4 levels; Inactivate the NRF2/HO-1 axis + Upregulate the ROS formation. | lipid peroxidation assay; Labile iron pool assay; Western blot; qPCR | In vitro and In vivo | [51] | |
| chemical & synthesized drugs | Ammonium Ferric Citrate | ↑ | Decrease GPX4; Promote the oxidative stress injury; Ferroptosis is triggered by autophagy | qPCR; ROS detection kit | In vitro | [52] |
| Cisplatin | ↑ | Decrease the GSH level + Inactivate the glutathione peroxidase in A549 and H1299 cells | Intracellular ROS, GSH, and iron contents were determined by the related assay kit | In vitro | [53] | |
| Erastin | ↑ | Suppress the NRF2/xCT pathway in the NSCLC cells possessing cisplatin-resistance | RT-qPCR; Western blot; ROS detected by a FACSCalibur Flow Cytometer; Xenograft assay | In vitro and In vivo | [54] | |
| ↑ | Inhibit GPX4 in the NSCLC cells owning radio-resistance | Western blot | In vitro | [55] | ||
| ↑ | Affect the ROS generation and mitochondria when Cotreated with celastrol. | ROS, iron were detected by a FACSCalibur Flow Cytometer; RT-PCR; Western blot; Lipid peroxidation assay kit; Commercial GSH quantification kit | In vitro | [56] | ||
| ↑ | A feedback loop among the erastin-induced ROS, p53, and the erastin-induced ferroptosis in A549 cell; Increase ROS + Downregulate SLC7A11 | semiquantitative Western blot; ROS detection kit | In vitro | [57] | ||
| 3-(2-ethoxyphenyl)-2-(piperazin-1-ylmethyl)quinazolin-4(3H)-one | ↑ | Upregulate the ROS, lipid peroxidation, and Fe2+ levels when Cotreated with cisplatin; Downregulate the GPX4 and NRF2 expression; Regulate the NRF2/Keap1 pathway to avoid the cisplatin-resistance. | _ | In vitro | [58] | |
| Acetaminophen | ↑ | Decrease the NRF2 and heme oxygenase-1 expression | _ | In vitro and In vivo | [59] | |
| 2-(4-(trifluoromethyl)phenyl)-7,8-dihydro-5H-thiopyrano[4,3-d]pyrimidin-4-ol | ↑ | Decrease the SLC7A11 level in H1299 cell | RNA sequencing; Gene enrichment analysis | In vitro | [60] | |
| levobupivacaine | ↑ | Induce high levels of ROS, iron, and Fe2+; Upregulate p53 to trigger ferroptosis | The reactive oxygen species levels were detected using flow cytometry analysis; Iron Assay Kit; Xenograft model | In vitro and In vivo | [61] | |
| Auranofin | ↑ | Inhibit GPX4 in the mutant p53 R273H accumulating isogenic H1299 cell | GSH/GSSG-Glo™ Assay kit; Western blot; Fluorescence staining | In vitro | [62] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, J.; Wang, L.; Tang, H.; Liu, X.; Peng, F.; Peng, C. Ferroptosis in Non-Small Cell Lung Cancer: Progression and Therapeutic Potential on It. Int. J. Mol. Sci. 2021, 22, 13335. https://doi.org/10.3390/ijms222413335
Zou J, Wang L, Tang H, Liu X, Peng F, Peng C. Ferroptosis in Non-Small Cell Lung Cancer: Progression and Therapeutic Potential on It. International Journal of Molecular Sciences. 2021; 22(24):13335. https://doi.org/10.3390/ijms222413335
Chicago/Turabian StyleZou, Jiayu, Li Wang, Hailin Tang, Xiuxiu Liu, Fu Peng, and Cheng Peng. 2021. "Ferroptosis in Non-Small Cell Lung Cancer: Progression and Therapeutic Potential on It" International Journal of Molecular Sciences 22, no. 24: 13335. https://doi.org/10.3390/ijms222413335
APA StyleZou, J., Wang, L., Tang, H., Liu, X., Peng, F., & Peng, C. (2021). Ferroptosis in Non-Small Cell Lung Cancer: Progression and Therapeutic Potential on It. International Journal of Molecular Sciences, 22(24), 13335. https://doi.org/10.3390/ijms222413335





