The Influence of Prenatal Fumonisin Exposure on Bone Properties, as well as OPG and RANKL Expression and Immunolocalization, in Newborn Offspring Is Sex and Dose Dependent
Abstract
1. Introduction
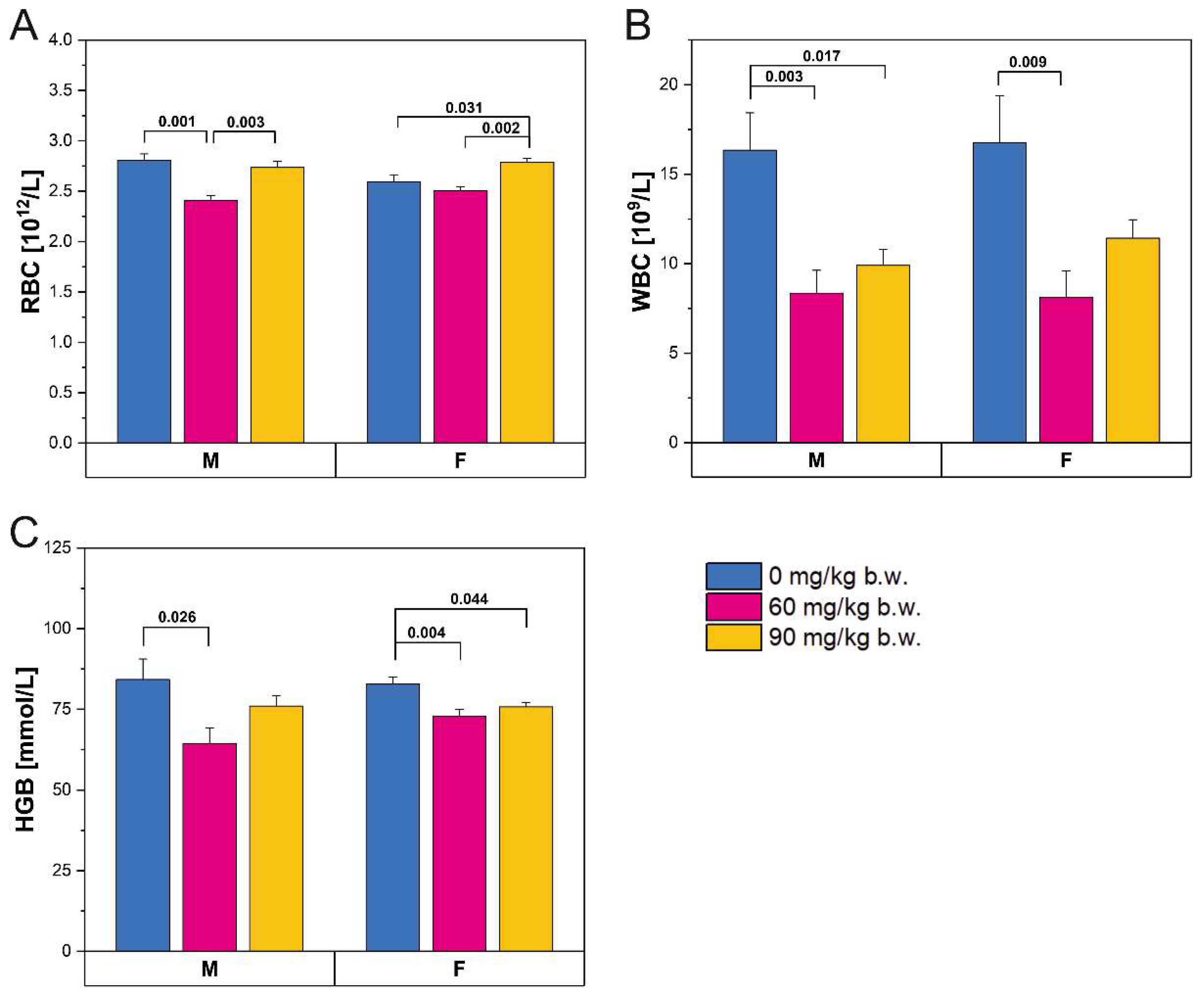
2. Results
2.1. Body Weight and Mechanical and Geometric Bone Properties
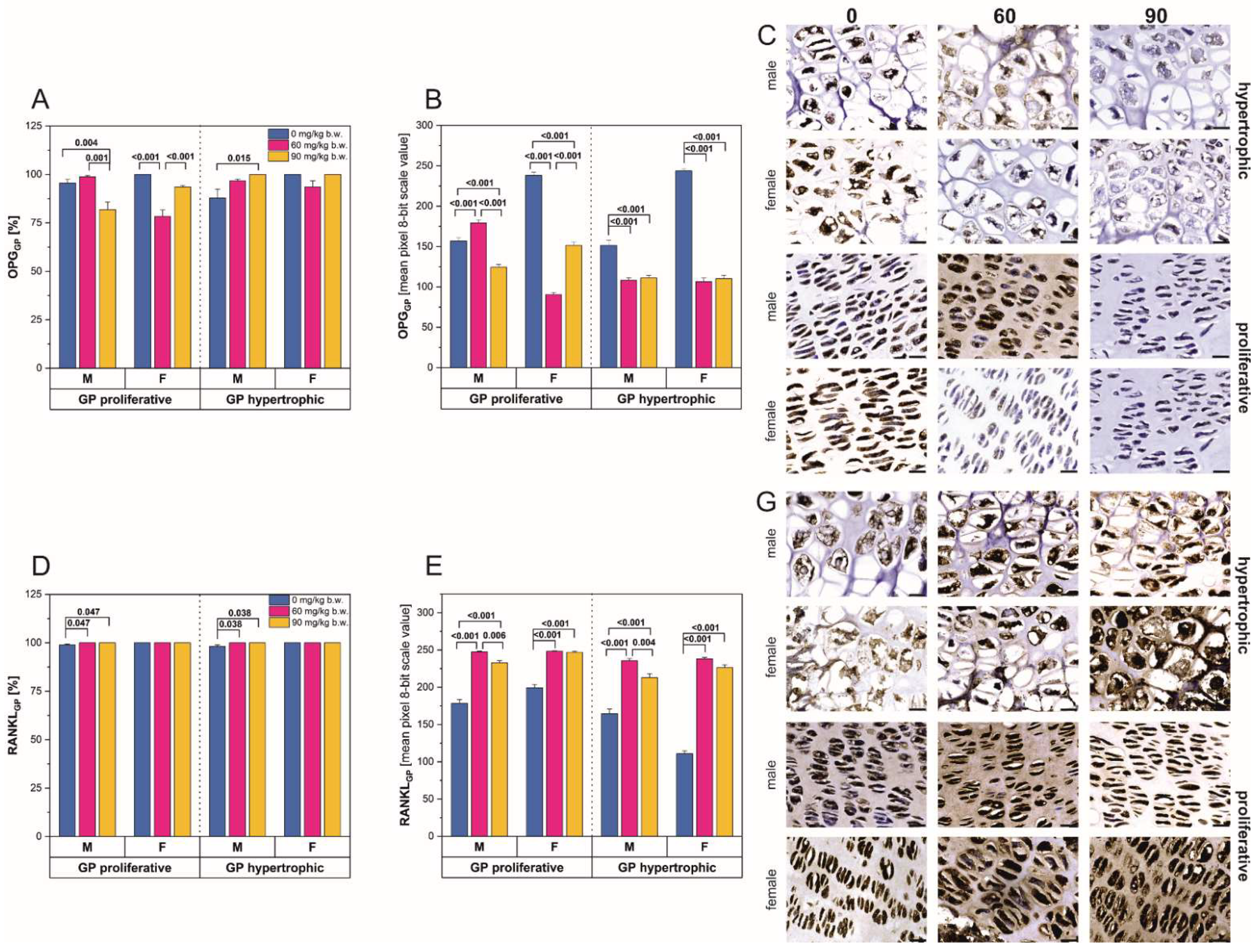
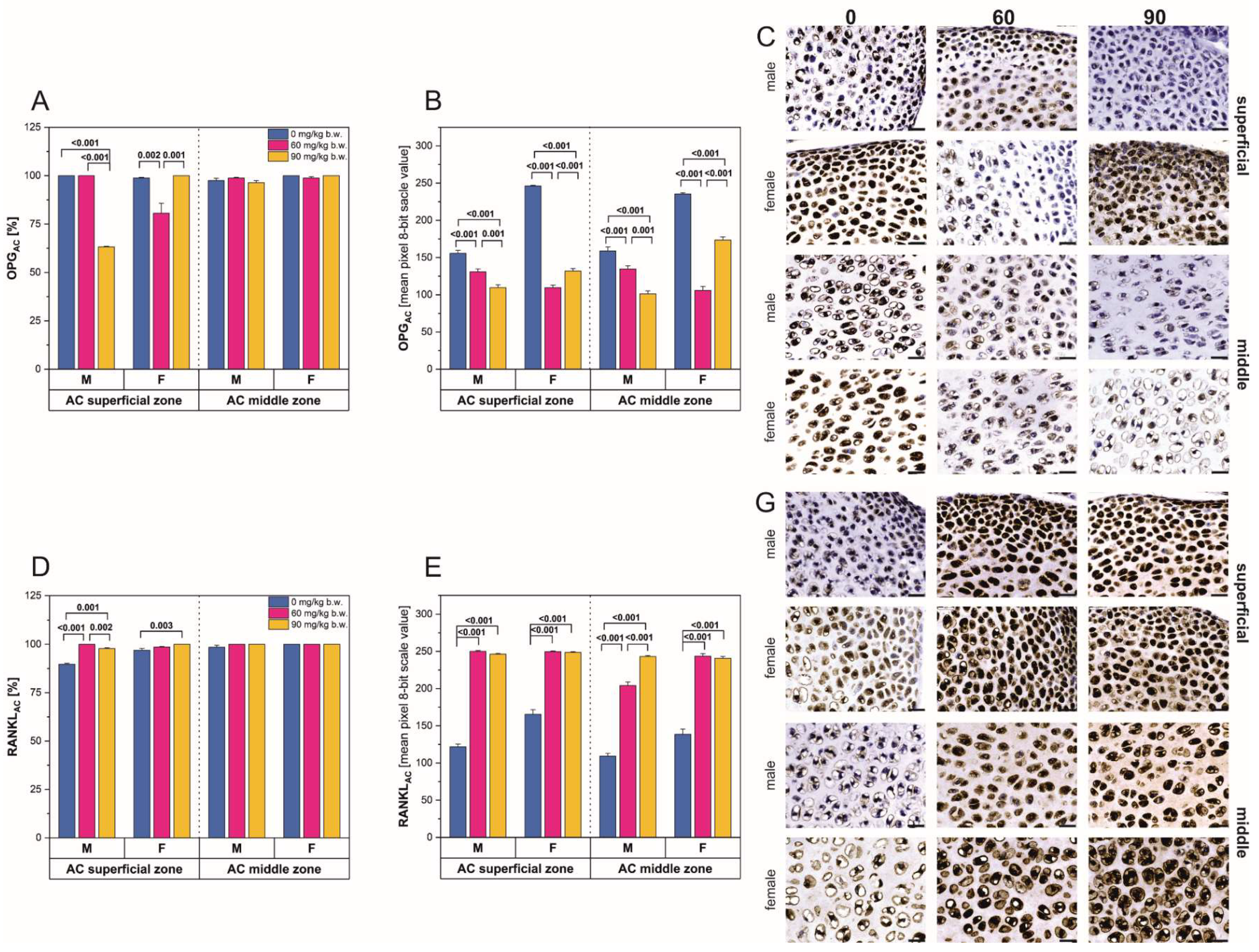
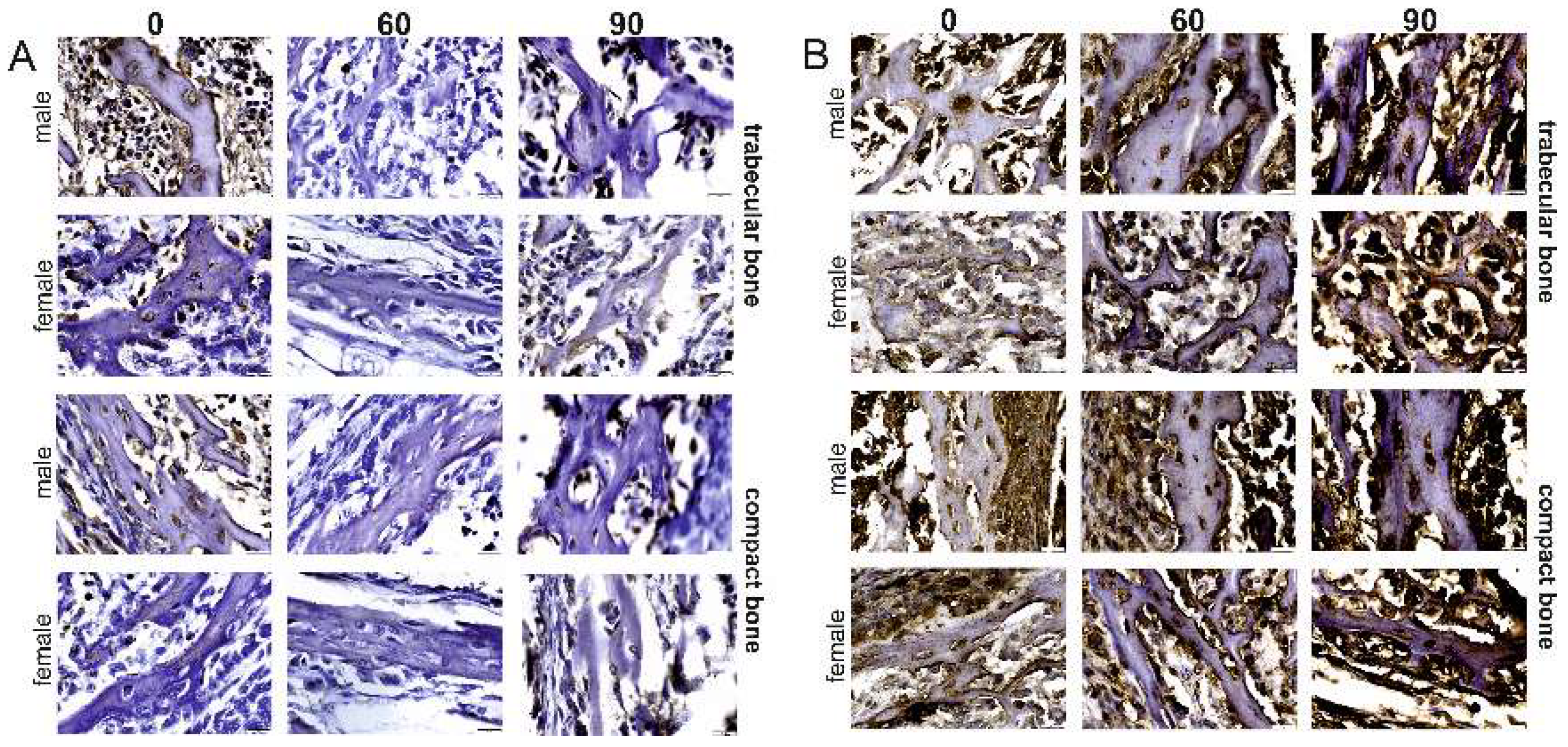
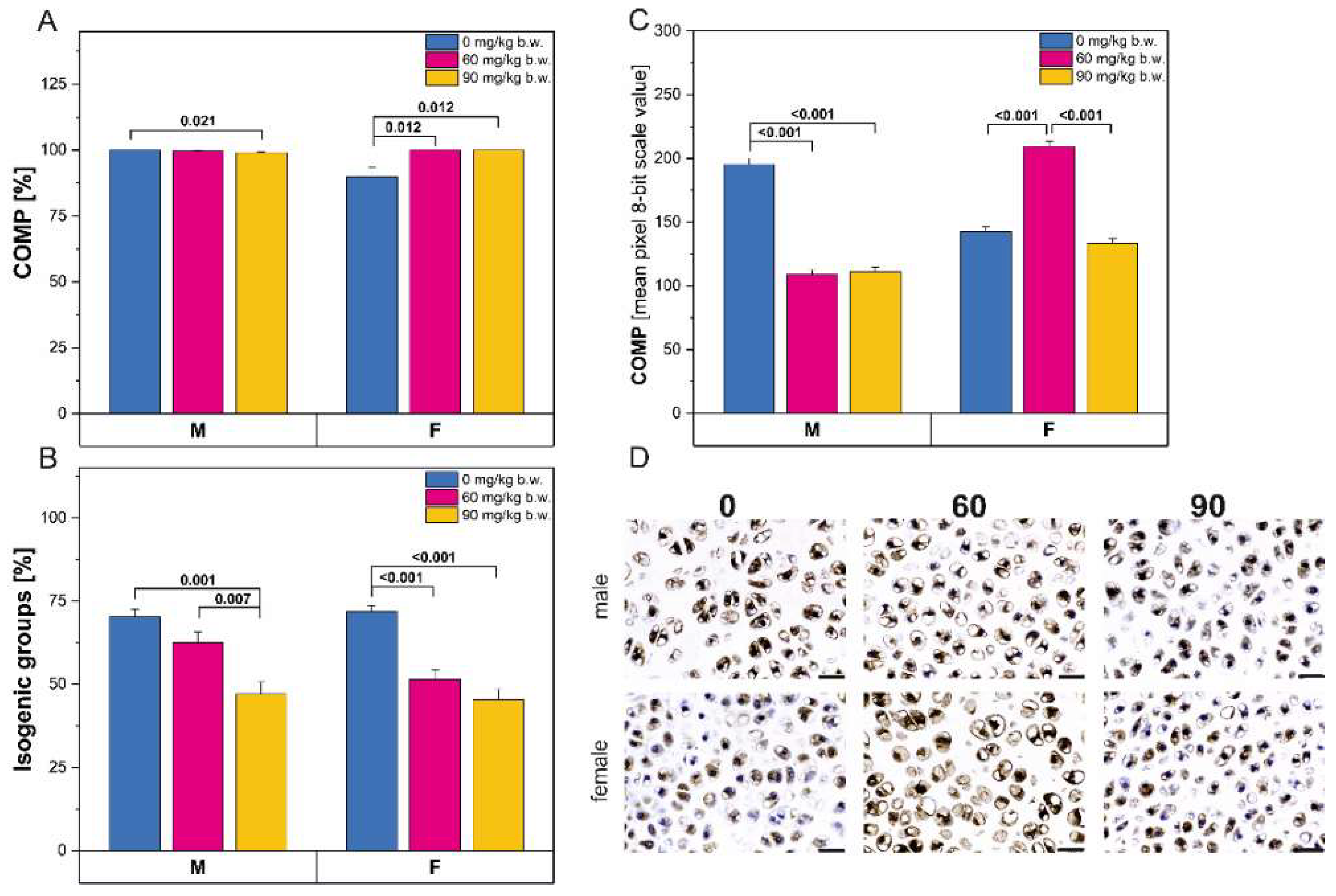
2.2. Immunostaining
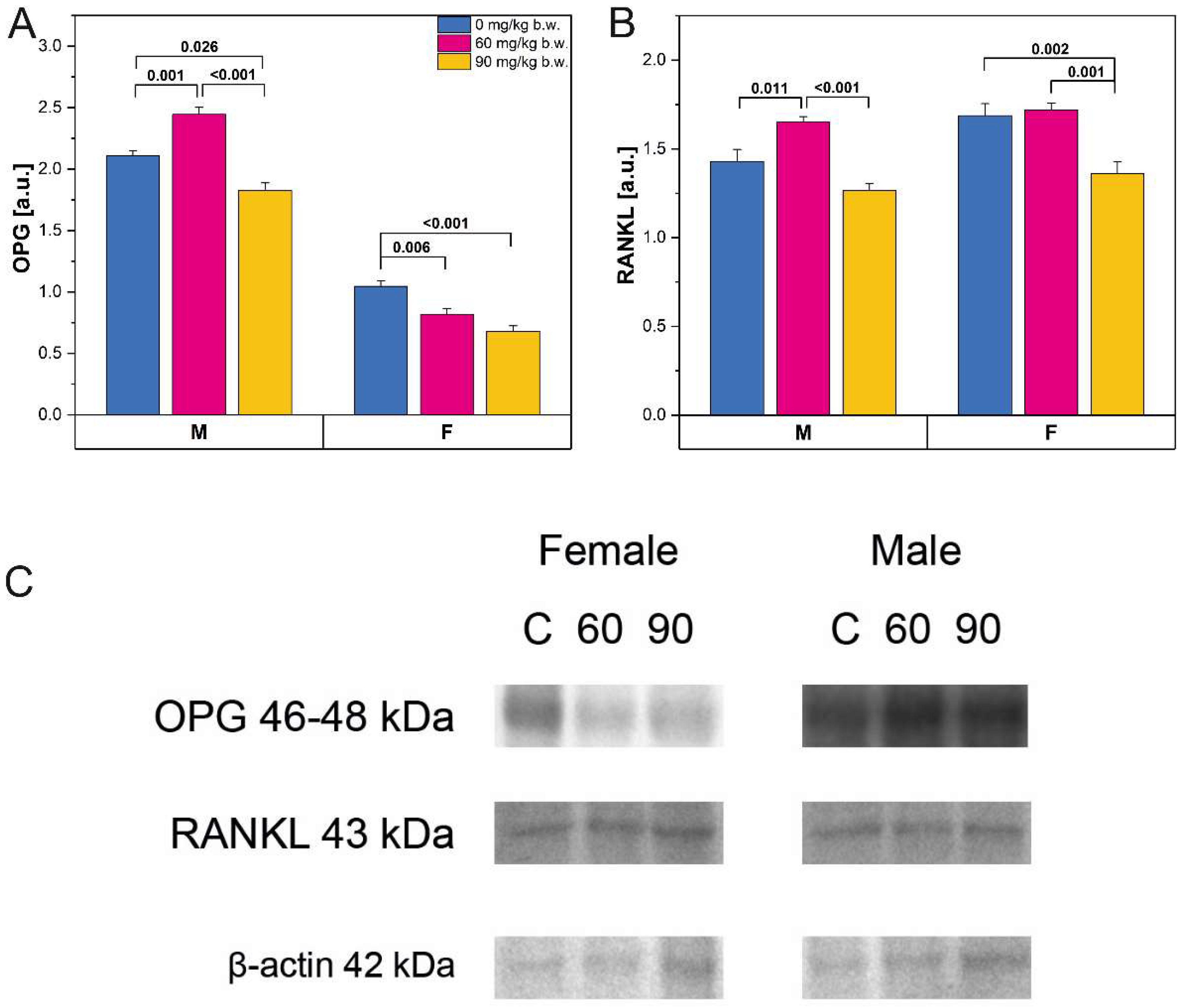
2.3. Western-Blot Protein Expression
3. Discussion
4. Materials and Methods
4.1. Animals and Experimental Design
4.2. Mechanical Testing
4.3. Immunohistochemistry
4.4. Western Blot
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wilson, D.A. Fumonisin toxicosis. In Clinical Veterinary Advisor: The Horse; Elsevier: Amsterdam, The Netherlands, 2012; pp. 214–215. [Google Scholar]
- Placinta, C.M.; D’Mello, J.P.F.; Macdonald, A.M.C. A review of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Anim. Feed Sci. Technol. 1999, 78, 21–37. [Google Scholar] [CrossRef]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide: Long-term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef]
- Wei, T.; Zhu, W.; Pang, M.; Liu, Y.; Dong, J. Natural occurrence of fumonisins B1 and B2 in corn in four provinces of China. Food Addit. Contam. B Surveill. 2013, 4, 270–274. [Google Scholar] [CrossRef] [PubMed]
- European Commision (EC). Commission Regulation (EU) 2005/856 amending Regulation 2001/466/EC as regards Fusarium toxins. Off. J. Eur. Union 2005, 143, 3–8. [Google Scholar]
- European Commission (EC). Commission Recommendation 2006/576/EC on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding. Off. J. Eur. Union 2006, 229, 7–9. [Google Scholar]
- Ledoux, D.R.; Brown, T.P.; Weibking, T.S.; Rottinghaus, G.E. Fumonisin toxicity in broiler chicks. J. Vet. Diagn. Investig. 1992, 4, 330–333. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA); Panel on contaminants in the food chain (CONTAM). Risks for animal health related to the presence of fumonisins, their modified forms and hidden forms in feed. EFSA J. 2018, 16, 5242. [Google Scholar]
- Riley, R.T.; Voss, K.A. Differential sensitivity of rat kidney and liver to fumonisin toxicity: Organ-specific differences in toxin accumulation and sphingoid base metabolism. Toxicol. Sci. 2006, 92, 335–345. [Google Scholar] [CrossRef]
- Rudyk, H.; Tomaszewska, E.; Kotsyumbas, I.; Muszyński, S.; Tomczyk-Warunek, A.; Szymańczyk, S.; Dobrowolski, P.; Wiącek, D.; Kamiński, D.; Brezvyn, O. Bone homeostasis in experimental fumonisins intoxication of rats. Ann. Anim. Sci. 2019, 19, 403–419. [Google Scholar] [CrossRef]
- Antonissen, G.; Devreese, M.; Van Immerseel, F.; De Baere, S.; Hessenberger, S.; Martel, A.; Croubels, S. Chronic exposure to deoxynivalenol has no influence on the oral bioavailability of fumonisin B1 in broiler chickens. Toxins 2015, 7, 560–571. [Google Scholar] [CrossRef]
- Devreese, M.; De Backer, P.; Croubels, S. Overview of the most important mycotoxins for the pig and poultry husbandry. Vlaam. Diergeneeskd. Tijdschr. 2013, 82, 171–180. [Google Scholar] [CrossRef]
- Tomaszewska, E.; Rudyk, H.; Dobrowolski, P.; Donaldson, J.; Świetlicka, I.; Puzio, I.; Kamiński, D.; Wiącek, D.; Kushnir, V.; Brezvyn, O. Changes in the intestinal histomorphometry, the expression of intestinal tight junction proteins, and the bone structure and liver of pre-laying hens following oral administration of fumonisins for 21 days. Toxins 2021, 13, 375. [Google Scholar] [CrossRef] [PubMed]
- Howard, P.C.; Warbritton, A.; Voss, K.A.; Lorentzen, R.J.; Thurman, J.D.; Kovach, R.M.; Bucci, T.J. Compensatory regeneration as a mechanism for renal tubule carcinogenesis of fumonisin B1 in the F344/N/Nctr BR rat. Environ. Health Perspect. 2001, 109 (Suppl. 2), 309–314. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Cawood, M.E.; Snyman, S.D.; Marasas, W.F.O. Fumonisin B1 dosimetry in relation to cancer initiation in rat liver. Carcinogenesis 1994, 15, 790. [Google Scholar] [CrossRef]
- Stockmann-Juvala, H.; Savolainen, K. A review of the toxic effects and mechanisms of action of fumonisin B1. Hum. Exp. Toxicol. 2008, 27, 799–809. [Google Scholar] [CrossRef]
- Coppock, R.W.; Jacobsen, B.J. Mycotoxins in animal and human patients. Toxicol. Ind. Health 2009, 25, 637–655. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, W.; Marasas, W. Controversies in fumonisin mycotoxicology and risk assessment. Hum. Exp. Toxicol. 2012, 31, 215–235. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.M. Recent research on fumonisins: A review. Food Addit. Contam. Chem. Anal. Control Expo. Risk Assess. 2012, 29, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). Guidance for industry: Fumonisin levels in human foods and animal feeds. Fed. Regist. 2001, 66, 56688–56689. [Google Scholar]
- Voss, K.A.; Smith, G.W.; Haschek, W.M. Fumonisins: Toxicokinetics, mechanism of action and toxicity. Anim. Feed Sci. Technol. 2007, 137, 299–325. [Google Scholar] [CrossRef]
- Didwania, N.; Joshi, M. Mycotoxins: A critical review on occurrence and significance. Int. J. Pharm. Sci. 2013, 5, 1014–1019. [Google Scholar]
- Muller, S.; Dekant, W.; Mally, A. Fumonisin B1 and the kidney: Modes of action for renal tumor formation by fumonisin B1 in rodents. Food Chem. Toxicol. 2012, 50, 3833–3846. [Google Scholar] [CrossRef] [PubMed]
- Dauncey, M.J.; Bicknell, R.J. Nutrition and neurodevelopment: Mechanisms of developmental dysfunction and disease in later life. Nutr. Res. Rev. 1999, 12, 231–253. [Google Scholar] [CrossRef]
- Tomaszewska, E.; Dobrowolski, P.; Puzio, I.; Donaldson, J.; Muszyński, S. Acrylamide-induced prenatal programming of bone structure in mammal model. Ann. Anim. Sci. 2020, 20, 1257–1287. [Google Scholar] [CrossRef]
- Chen, Q.; Bao, N.; Yao, Q.; Li, Z.Y. Fractal dimension: A complementary diagnostic indicator of osteoporosis to bone mineral density. Med. Hypotheses 2018, 116, 136–138. [Google Scholar] [CrossRef]
- Rudyk, H.; Tomaszewska, E.; Arciszewski, M.B.; Muszyński, S.; Tomczyk-Warunek, A.; Dobrowolski, P.; Donaldson, J.; Brezvyn, O.; Kotsyumbas, I. Histomorphometrical changes in intestine structure and innervation following experimental fumonisins intoxication in male Wistar rats. Pol. J. Vet. Sci. 2020, 23, 77–88. [Google Scholar] [PubMed]
- Voss, K.A.; Riley, R.T.; Gelineau-van Waes, J. Fumonisin B1 induced neural tube defects were not increased in LM/Bc mice fed folate-deficient diet. Mol. Nutr. Food Res. 2014, 58, 1190–1198. [Google Scholar] [CrossRef]
- Reddy, R.V.; Johnson, G.; Rottinghaus, G.E.; Casteel, S.W.; Reddy, C.S. Developmental effects of fumonisin B1 in mice. Mycopathologia 1996, 134, 161–166. [Google Scholar] [CrossRef]
- Voss, K.A.; Bacon, C.W.; Norred, W.P.; Chapin, R.E.; Chamberlain, W.J.; Plattner, R.D.; Meredith, F.I. Studies on the reproductive effects of Fusarium moniliforme culture material in rats and the biodistribution of [14C] fumonisin B1 in pregnant rats. Nat. Toxins 1996, 4, 24–33. [Google Scholar] [CrossRef]
- Abdel-Wahhab, M.A.; Hassan, A.M.; Amer, H.A.; Naguib, K.M. Prevention of fumonisin-induced maternal and developmental toxicity in rats by certain plant extracts. J. Appl. Toxicol. 2004, 24, 469–474. [Google Scholar] [CrossRef]
- Voss, K.A.; Gelineau-van Waes, J.B.; Riley, R.T. Fumonisins: Current research trends in developmental toxicology. Mycotoxin Res. 2006, 22, 61–69. [Google Scholar] [CrossRef]
- Tomaszewska, E.; Muszyński, S.; Dobrowolski, P.; Kostro, K.; Taszkun, I.; Żmuda, A.; Blicharski, T.; Kędzia, P. Bentonite diminishes DON-induced changes in bone development in mink dams. J. Vet. Res. 2016, 60, 349–355. [Google Scholar] [CrossRef][Green Version]
- Tomaszewska, E.; Dobrowolski, P.; Muszyński, S.; Kostro, K.; Taszkun, I.; Żmuda, A.; Blicharski, T.; Hułas-Stasiak, M. DON-induced changes in bone homeostasis in mink dams. J. Vet. Res. 2017, 61, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Shastak, Y.; Rodehuyscord, M. A review of the role of magnesium in poultry nutrition. Worlds Poult. Sci. J. 2015, 71, 125–138. [Google Scholar] [CrossRef]
- Torres, C.A.; Korver, D.R. Influences of trace mineral nutrition and maternal flock age on broiler embryo bone development. Poult. Sci. 2018, 97, 2996–3003. [Google Scholar] [CrossRef] [PubMed]
- Muszynski, S.; Tomaszewska, E.; Kwiecien, M.; Dobrowolski, P.; Tomczyk, A. Effect of dietary phytase supplementation on bone and hyaline cartilage development of broilers fed with organically complexed copper in a Cu-deficient diet. Biol. Trace Elem. Res. 2018, 182, 339–353. [Google Scholar] [CrossRef] [PubMed]
- Rath, N.C.; Huff, G.R.; Huff, W.E.; Balog, J.M. Factors regulating bone maturity and strength in poultry. Poult. Sci. 2000, 79, 1024–1032. [Google Scholar] [CrossRef]
- Parfitt, A.M. What is the normal rate of bone remodeling? Bone 2004, 35, 1–3. [Google Scholar] [CrossRef]
- Schoppet, M.; Preissner, K.T.; Hofbauer, L.C. RANK ligand and osteoprotegerin: Paracrine regulators of bone metabolism and vascular function. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 549–553. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Udagawa, N.; Takahashi, N. Action of RANKL and OPG for osteoclastogenesis. Crit. Rev. Eukaryot. Gene Expr. 2009, 19, 61–72. [Google Scholar] [CrossRef]
- Turner, C.H. Determinants of skeletal fragility and bone quality. J. Musculoskelet. Neuronal Interact. 2002, 2, 527–528. [Google Scholar]
- Paschalis, E.P.; Shane, E.; Lyritis, G.; Skarantavos, G.; Mendelsohn, R.; Boskey, A.L. Bone fragility and collagen cross-links. J. Bone Miner. Res. 2004, 19, 2000–2004. [Google Scholar] [CrossRef]
- Lebepe-Mazur, S.; Bal, H.; Hopmans, E.; Murphy, P.; Hendrich, S. Fumonisin B1 is fetotoxic in rats. Vet. Hum. Toxicol. 1995, 37, 126–130. [Google Scholar] [PubMed]
- Floss, J.L.; Casteel, S.W.; Johnson, G.C.; Rottinghaus, G.E.; Krause, G.F. Development toxicity of fumonisin in Syrian hamsters. Mycopathologia 1994, 128, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Huff, W.E. Discrepancies between bone ash and toe ash during aflatoxicosis. Poult. Sci. 1980, 59, 2213–2215. [Google Scholar] [CrossRef] [PubMed]
- Ruppel, M.E.; Miller, L.M.; Burr, D.B. The effect of the microscopic and nanoscale structure on bone fragility. Osteoporos. Int. 2008, 19, 1251–1265. [Google Scholar] [CrossRef] [PubMed]
- Shahnazari, M.; Lang, D.H.; Fosmire, G.J.; Sharkey, N.A.; Mitchell, A.D.; Leach, R.M. Strontium administration in young chickens improves bone volume and architecture but does not enhance bone structural and material strength. Calcif. Tissue Int. 2007, 80, 160–166. [Google Scholar] [CrossRef] [PubMed]
- O’Dell, B.L.; Hardwick, B.C.; Reynolds, G.; Savage, J.E. Connective tissue defect in the chick resulting from copper deficiency. Proc. Soc. Exp. Biol. Med. 1961, 108, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Leeson, S.; Summers, J.D. Some nutritional implications of leg problems with poultry. Brit. Vet. J. 1988, 144, 81–92. [Google Scholar] [CrossRef]
- Wu, W.; Li, G.; Liu, T.; Vesonder, R.R. The effect of fumonisin B1 on isolated chondrocytes and on bone formation. Poult. Sci. 1995, 74, 1431–1436. [Google Scholar] [CrossRef]
- Rogers, A.; Eastell, R. Circulating osteoprotegerin and receptor activator for nuclear factor kappaB ligand: Clinical utility in metabolic bone disease assessment. J. Clin. Endocrinol. Metab. 2005, 90, 6323–6331. [Google Scholar] [CrossRef] [PubMed]
- Hofbauer, L.C.; Schoppet, M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA 2004, 292, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Cawley, K.M.; Bustamante-Gomez, N.C.; Guha, A.G.; MacLeod, R.S.; Xiong, J.; Gubrij, I.; Liu, Y.; Mulkey, R.; Palmieri, M.; Thostenson, J.D. Local production of osteoprotegerin by osteoblasts suppresses bone resorption. Cell Rep. 2020, 32, 108052. [Google Scholar] [CrossRef]
- Mescher, A.L. Junqueira’s Basic Histology, Text and Atlas, 13th ed.; McGraw Hill Medical: New York, NY, USA, 2013; pp. 130–137. [Google Scholar]
- Zhang, L.; Hu, J.; Athanasiou, K.A. The role of tissue engineering in articular cartilage repair and regeneration. Crit. Rev. Biomed. Eng. 2009, 37, 1–57. [Google Scholar] [CrossRef] [PubMed]
- Francuski, J.V.; Radovanovic, A.; Andric, N.; Krstic, V.; Bogdanovic, D.; Hadzic, V.; Todorovic, V.; Lazarevic Macanovic, M.; Sourice Petit, S.; Beck-Cormier, S. Age-related changes in the articular cartilage of the stifle joint in non-working and working German Shepherd dogs. J. Comp. Pathol. 2014, 151, 363–374. [Google Scholar] [CrossRef]
- Posey, K.L.; Coustry, F.; Hecht, J.T. Cartilage oligomeric matrix protein: COMPopathies and beyond. Matrix Biol. 2018, 71–72s, 161–173. [Google Scholar] [CrossRef]
- Morozzi, G.; Fabbroni, M.; Bellisai, F.; Pucci, G.; Galeazzi, M. Cartilage oligomeric matrix protein level in rheumatic diseases: Potential use as a marker for measuring articular cartilage damage and/or the therapeutic efficacy of treatments. Ann. N. Y. Acad. Sci. 2007, 1108, 398–407. [Google Scholar] [CrossRef]
- Garnero, P. Use of biochemical markers to study and follow patients with osteoarthritis. Curr. Rheumatol. Rep. 2006, 8, 37–44. [Google Scholar] [CrossRef]
- Ferretti, J.L.; Capozza, R.F.; Mondelo, N.; Zanchetta, J.R. Interrelationships between densitometric, geometric, and mechanical properties of rat femora: Inferences concerning mechanical regulation of bone modeling. J. Bone Miner. Res. 1993, 8, 1389–1396. [Google Scholar] [CrossRef]
- Hułas-Stasiak, M.; Jakubowicz-Gil, J.; Dobrowolski, P.; Grzesiak, M.; Muszyński, S.; Świątkiewicz, M.; Tomaszewska, E. Regulation of folliculogenesis by growth factors in piglet ovary exposed prenatally to β-hydroxy-β-methylbutyrate (HMB). Ann. Anim. Sci. 2020, 20, 899–917. [Google Scholar] [CrossRef]
- Puzio, I.; Muszyński, S.; Dobrowolski, P.; Kapica, M.; Pawłowska-Olszewska, M.; Donaldson, J.; Tomaszewska, E. Alterations in small intestine and liver morphology, immunolocalization of leptin, ghrelin and nesfatin-1 as well as immunoexpression of tight junction proteins in intestinal mucosa after gastrectomy in rat model. J. Clin. Med. 2021, 10, 272. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
| Dependent Variable | Sex | FBs [mg/kg b.w.] | p-Value | ||
|---|---|---|---|---|---|
| 0 | 60 | 90 | |||
| Body weight [g] | M | 10.050 ± 0.438 a | 6.819 ± 0.105 b | 6.839 ± 0.203 b | <0.001 |
| Femur length [mm] | 11.546 ± 0.068 a | 10.794 ± 0.245 b | 10.200 ± 0.214 b | <0.001 | |
| Cross-sectional area [mm2] | 4.148 ± 0.194 a | 3.935 ± 0.288 a,b | 3.250 ± 0.212 b | 0.034 | |
| Ultimate force [N] | 1.693 ± 0.058 a | 1.190 ± 0.096 b | 1.170 ± 0.028 b | <0.001 | |
| Stiffness [N/mm] | 5.590 ± 0.409 a | 3.050 ± 0.163 b | 3.060 ± 0.127 b | <0.001 | |
| Collagentrabecular [%] | 13.188 ± 1.782 a,b | 15.948 ± 1.867 a | 8.003 ± 0.434 b | 0.004 | |
| Collagencompact [%] | 20.051 ± 2.034 a | 7.589 ± 0.978 b | 17.365 ± 1.528 a | <0.001 | |
| Body weight [g] | F | 7.766 ± 0.202 a | 6.747 ± 0.215 a,b | 5.903 ± 0.427 b | 0.001 |
| Femur length [mm] | 10.929 ± 0.075 a | 9.870 ± 0.174 b | 9.769 ± 0.173 b | <0.001 | |
| Cross-sectional area [mm2] | 3.945 ± 0.219 a | 3.726 ± 0.283 a,b | 3.115 ± 0.142 b | 0.040 | |
| Ultimate force [N] | 1.434 ± 0.099 a | 1.104 ± 0.101 b | 1.212 ± 0.082 b | 0.012 | |
| Stiffness [N/mm] | 4.508 ± 0.743 | 2.636 ± 0.324 | 3.770 ± 0.405 | 0.065 | |
| Collagentrabecular [%] | 14.231 ± 1.144 a,b | 13.634 ± 0.661 a | 18.699 ± 1.967 b | 0.032 | |
| Collagencompact [%] | 8.283 ± 0.764 a | 17.041 ± 3.056 b | 18.165 ± 1.782 b | 0.006 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomaszewska, E.; Rudyk, H.; Świetlicka, I.; Hułas-Stasiak, M.; Donaldson, J.; Arczewska, M.; Muszyński, S.; Dobrowolski, P.; Puzio, I.; Kushnir, V.; et al. The Influence of Prenatal Fumonisin Exposure on Bone Properties, as well as OPG and RANKL Expression and Immunolocalization, in Newborn Offspring Is Sex and Dose Dependent. Int. J. Mol. Sci. 2021, 22, 13234. https://doi.org/10.3390/ijms222413234
Tomaszewska E, Rudyk H, Świetlicka I, Hułas-Stasiak M, Donaldson J, Arczewska M, Muszyński S, Dobrowolski P, Puzio I, Kushnir V, et al. The Influence of Prenatal Fumonisin Exposure on Bone Properties, as well as OPG and RANKL Expression and Immunolocalization, in Newborn Offspring Is Sex and Dose Dependent. International Journal of Molecular Sciences. 2021; 22(24):13234. https://doi.org/10.3390/ijms222413234
Chicago/Turabian StyleTomaszewska, Ewa, Halyna Rudyk, Izabela Świetlicka, Monika Hułas-Stasiak, Janine Donaldson, Marta Arczewska, Siemowit Muszyński, Piotr Dobrowolski, Iwona Puzio, Volodymyr Kushnir, and et al. 2021. "The Influence of Prenatal Fumonisin Exposure on Bone Properties, as well as OPG and RANKL Expression and Immunolocalization, in Newborn Offspring Is Sex and Dose Dependent" International Journal of Molecular Sciences 22, no. 24: 13234. https://doi.org/10.3390/ijms222413234
APA StyleTomaszewska, E., Rudyk, H., Świetlicka, I., Hułas-Stasiak, M., Donaldson, J., Arczewska, M., Muszyński, S., Dobrowolski, P., Puzio, I., Kushnir, V., Brezvyn, O., Muzyka, V., & Kotsyumbas, I. (2021). The Influence of Prenatal Fumonisin Exposure on Bone Properties, as well as OPG and RANKL Expression and Immunolocalization, in Newborn Offspring Is Sex and Dose Dependent. International Journal of Molecular Sciences, 22(24), 13234. https://doi.org/10.3390/ijms222413234







