New Insights into the Structure of Kappa/Beta-Carrageenan: A Novel Potential Inhibitor of HIV-1
Abstract
:1. Introduction
2. Results and Discussion
2.1. Carrageenan and Oligosaccharides
2.2. The Enzyme-Resistant Fraction of κ/β-Carrageenan RF/CRG
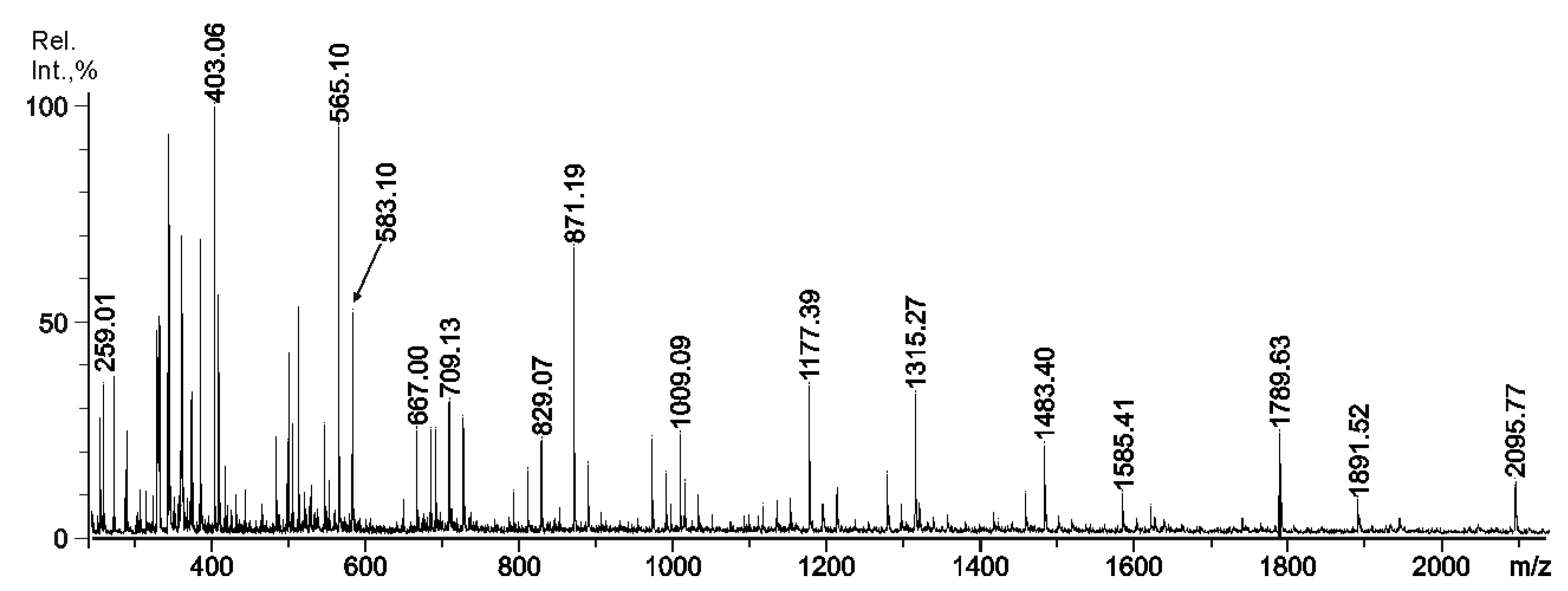
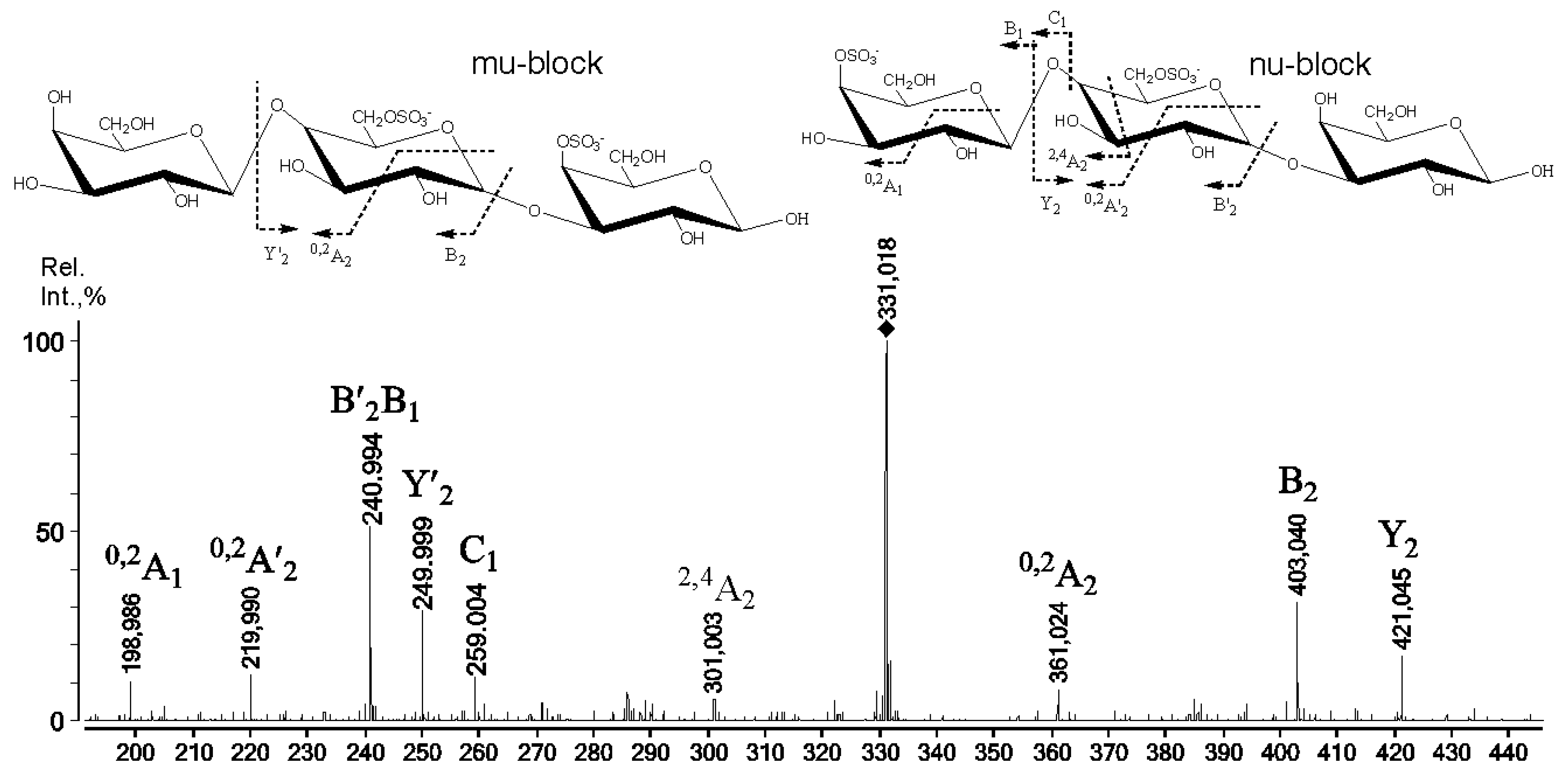
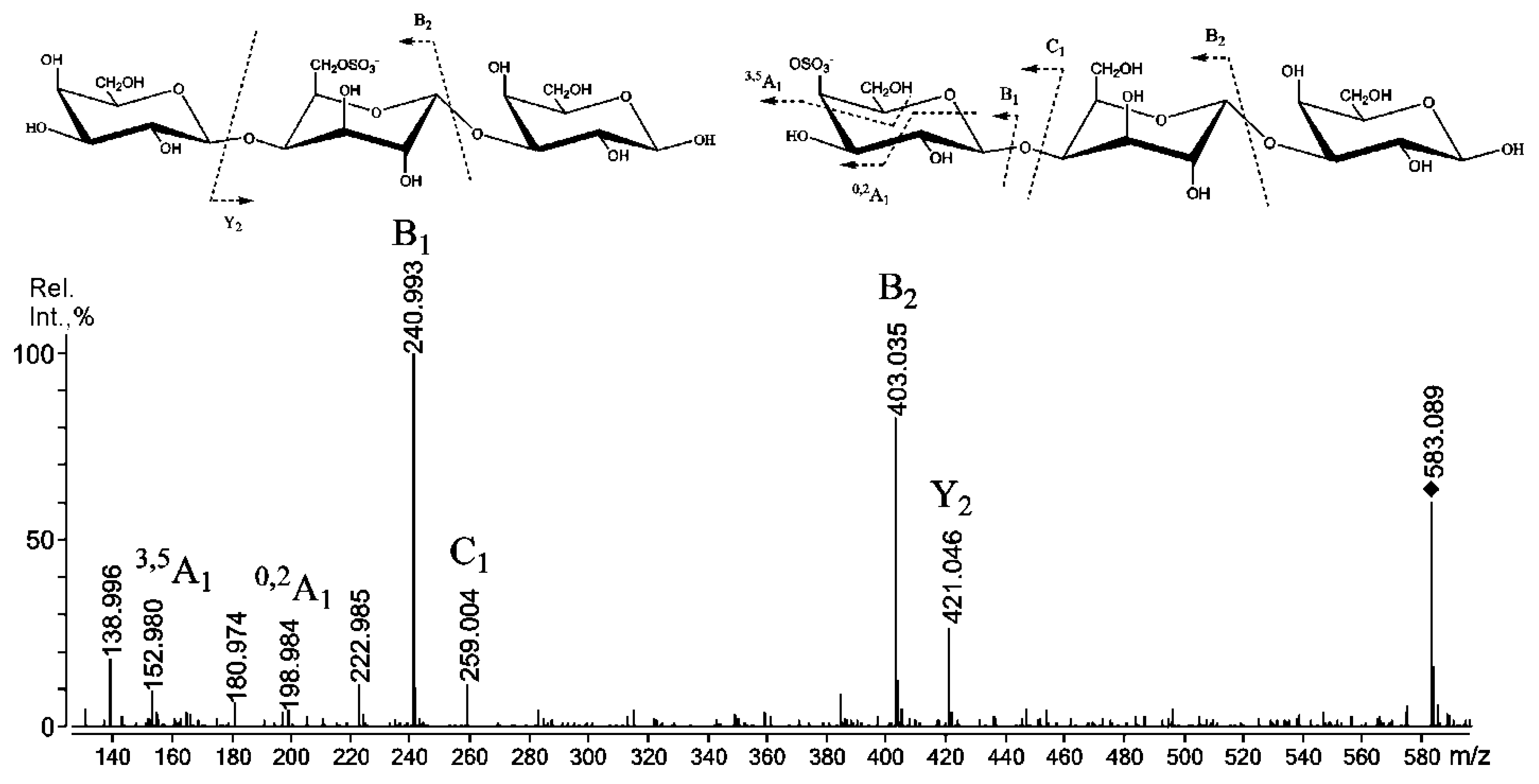
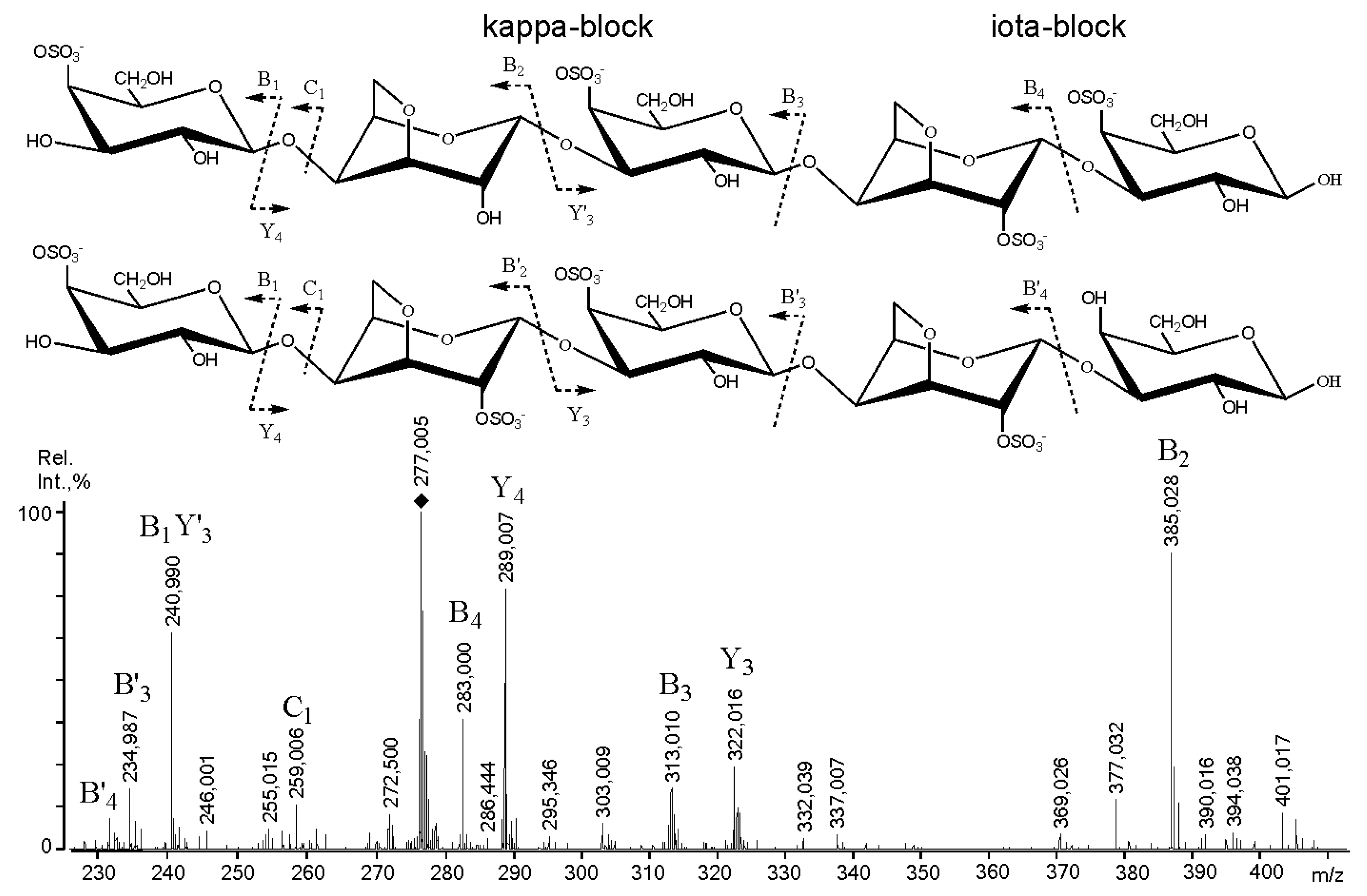
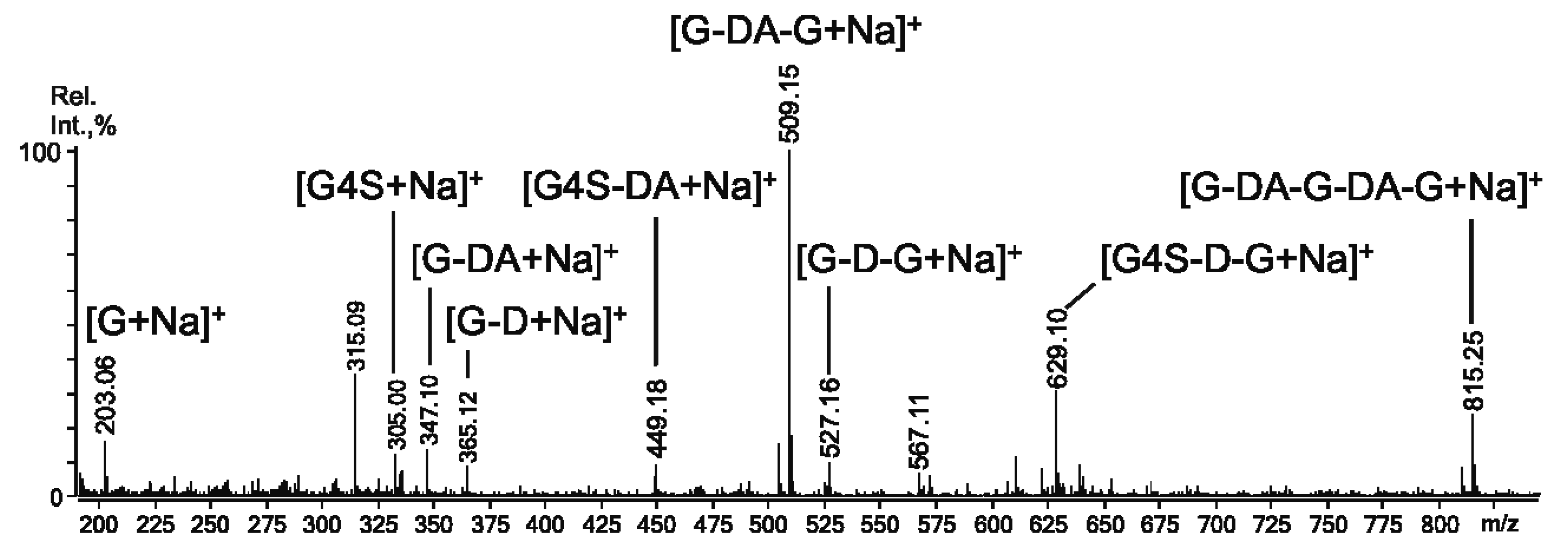
2.3. Mass Spectrometry of Oligosaccharides Obtained by Mild Acid Hydrolysis of RF/CRG
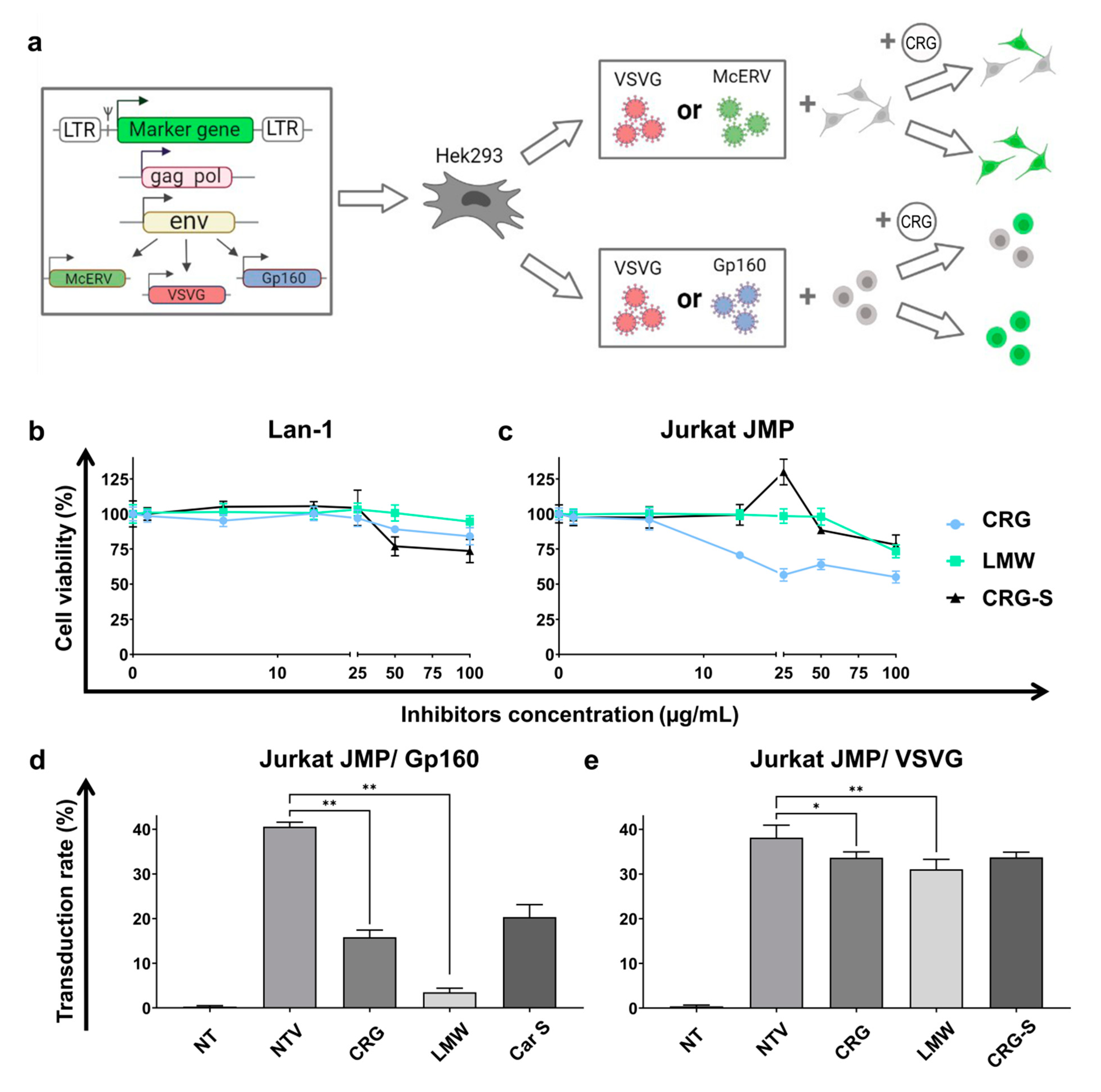
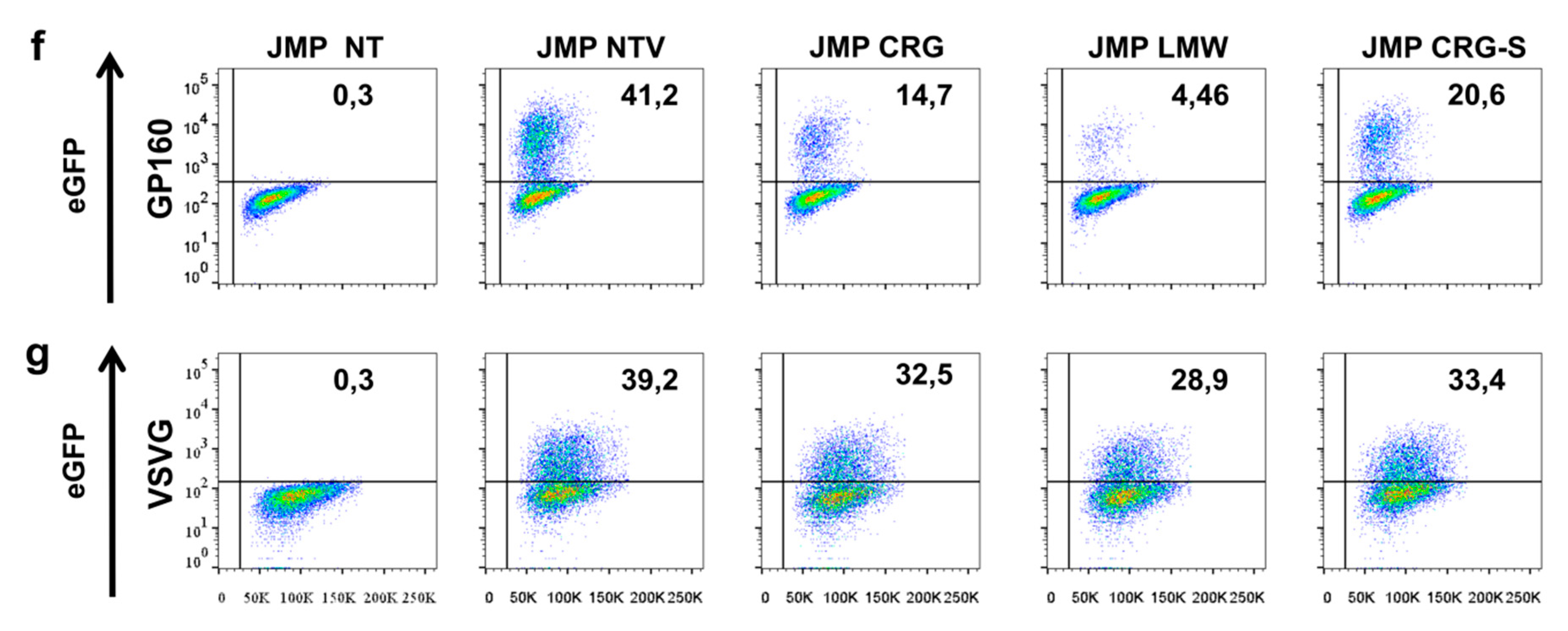
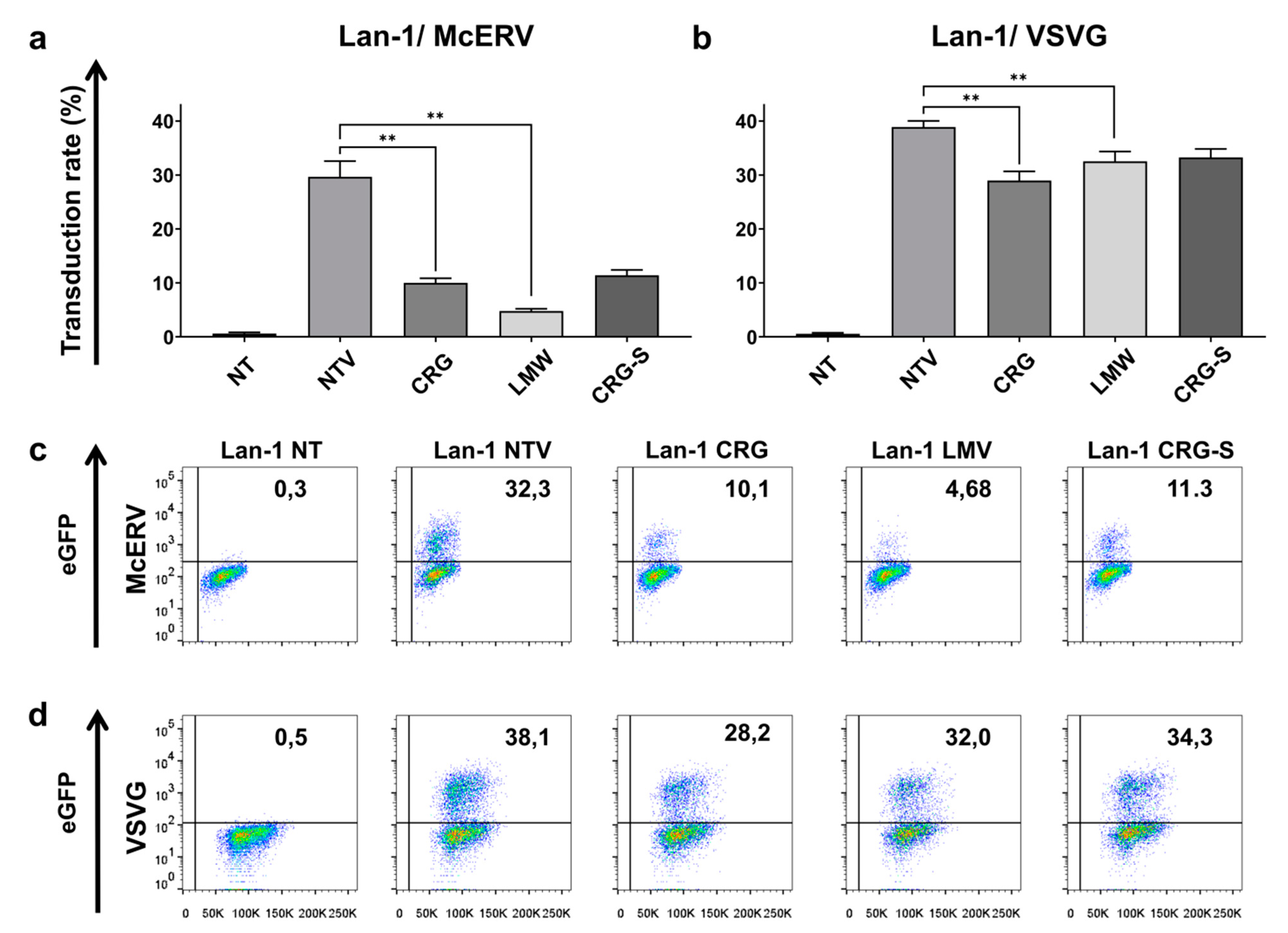
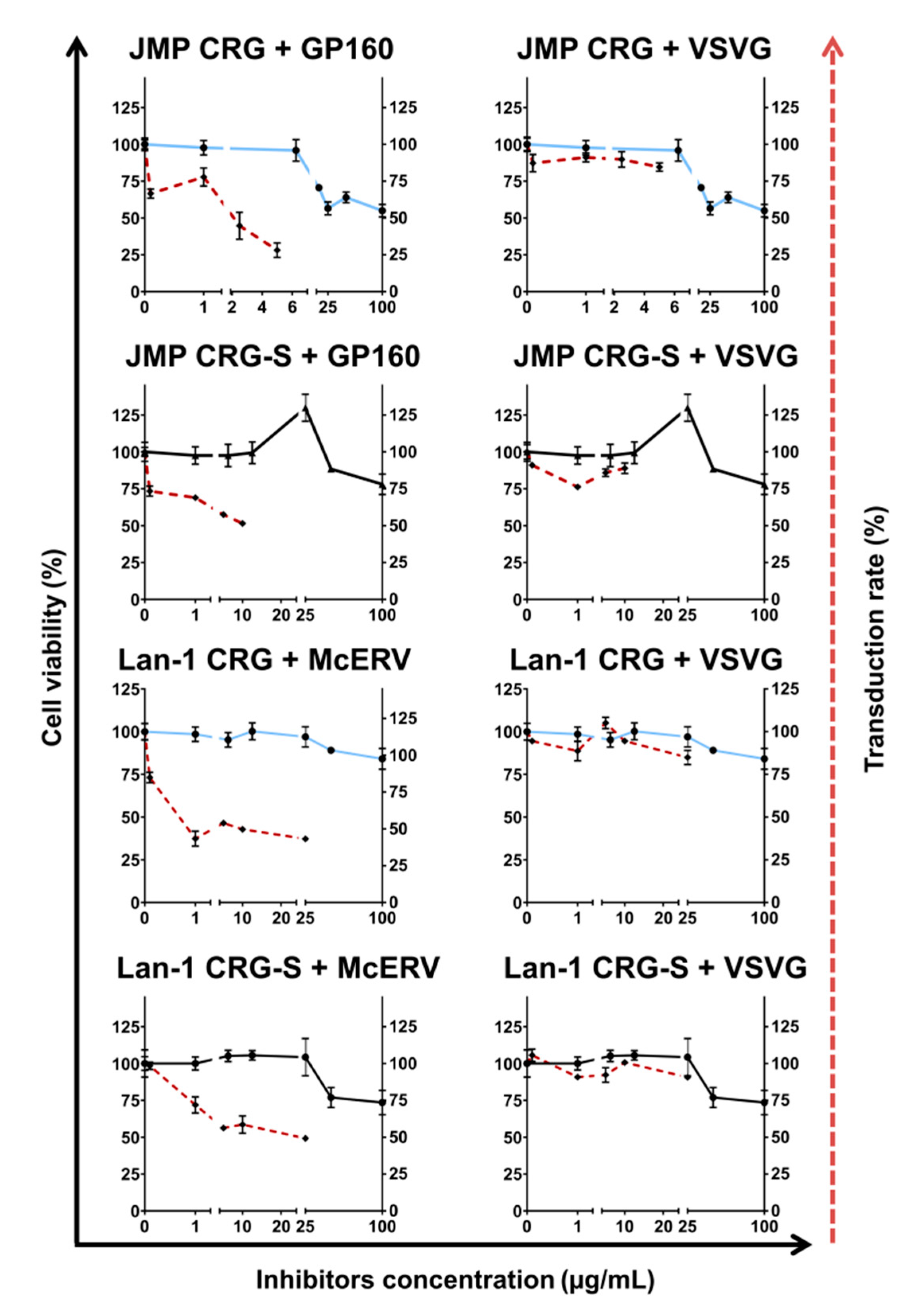
2.4. Antiviral Activity of κ/β-CRG and Its Oligosaccharides
3. Materials and Methods
3.1. Algae
3.2. Extraction of Carrageenan
3.3. Preparation of Oligosaccharides from the κ/β-CRG
3.4. Enzymatic Degradation of the κ/β-CRG
Degradation of RF/CRG (κ/β-CRG Resistant Fraction)
3.5. Instruments
3.6. General Methods
3.6.1. Analytical Procedures
3.6.2. Molecular Weight Estimation
3.7. Antiviral Activity
3.7.1. Cell Culture
3.7.2. Cytotoxicity Assay
3.7.3. Obtaining of Pseudotyped Lentiviral Vectors Particles
3.7.4. Antiviral Activity Assay
3.7.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knutsen, S.H.; Myslabodski, D.E.; Larsen, B.; Usov, A.I. A modified system of nomenclature for red algal galactans. Bot. Mar. 1994, 37, 163–169. [Google Scholar] [CrossRef]
- Rees, D.A. The carrageenans system of polysaccharides. 1. The relation between the κ- and λ-components. J. Chem. Soc. 1963, 1, 1821–1832. [Google Scholar] [CrossRef]
- Greer, C.W.; Yaphe, W. Purification and properties of iota-carrageenase from a marine bacterium. Can. J. Microbiol. 1984, 30, 1500–1506. [Google Scholar] [CrossRef]
- Renn, D.W.; Santos, G.A.; Dumont, L.E.; Parent, C.A.; Stanley, N.F.; Stancioff, D.J.; Guiseley, K.B. β-carrageenan: Isolation and characterization. Carbohydr. Polym. 1993, 22, 247–252. [Google Scholar] [CrossRef]
- Craigie, J.S. Cell Walls. In Biology of the Red Algae; Cole, K.M., Sheath, R.Y., Eds.; Cambridge University Press: Cambridge, UK, 1990; pp. 221–257. [Google Scholar]
- Guibet, M.; Colin, S.; Barbeyron, T.; Genicot, S.; Kloareg, B.; Michel, G.; Helbert, W. Degradation of lambda-carrageenan by Pseudoalteromonas carrageenovora lambda-carrageenase: A new family of glycoside hydrolases unrelated to kappa- and iota-carrageenases. Biochem. J. 2007, 404, 105–114. [Google Scholar] [CrossRef] [Green Version]
- Greer, C.W.; Yaphe, W. Characterization of hybrid (betha, kappa and gamma) carrageenan from Eucheuma gelatinae J. Agardh (Rhodophyta, Solieriaceae) using carragenases, infrared and C-NMR spectroscopy. Bot. Mar. 1984, 27, 473–478. [Google Scholar]
- Knutsen, S.H.; Grasdalen, H. Characterisation of water-extractable polysaccharides from Norwegian Furcellaria lumbricalis (Huds.) Lam. (Gigartinales, Rhodophyceae) by IR and NMR spectroscopy. Bot. Mar. 1987, 30, 497–505. [Google Scholar] [CrossRef]
- Barabanova, A.O.; Yermak, I.M.; Glazunov, V.P.; Isakov, V.V.; Titlyanov, E.A.; Solov’eva, T.F. Comparative study of carrageenans from reproductive and sterile forms of Tichocarpus crinitus (Gmel.) Rupr (Rhodophyta, Tichocarpaceae). Biochemistry (Moscow) 2005, 70, 350–356. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Barabanova, A.O.; Correc, G.; Nazarenko, E.L.; Davydova, V.N.; Helbert, W.; Dmitrenok, P.S.; Yermak, I.M. Analysis of structural heterogeneity of kappa/beta-carrageenan oligosaccharides from Tichocarpus crinitus by negative-ion ESI and tandem MALDI mass spectrometry. Carbohydr. Polym. 2011, 86, 546–554. [Google Scholar] [CrossRef]
- Correc, G.; Barabanova, A.; Tuvikene, R.; Truus, K.; Yermak, I.; Helbert, W. Comparison of the structures of hybrid κ/β-carrageenans extracted from Furcellaria lumbricalis and Tichocarpus crinitus. Carbohydr. Polym. 2012, 88, 31–36. [Google Scholar] [CrossRef]
- Reunov, A.; Nagorskaya, V.; Lapshina, L.; Yermak, I.; Barabanova, A. Effect of κ/β-carrageenan from red alga Tichocarpus crinitus (Tichocarpaceae) on infection of detached tobacco leaves with tobacco mosaic virus. J. Plant Dis. Prot. 2004, 111, 165–172. [Google Scholar] [CrossRef]
- Klimyte, E.M.; Smith, S.E.; Oreste, P.; Lembo, D.; Dutch, R.E. Inhibition of human metapneumovirus binding to heparan sulfate blocks infection in human lung cells and airway tissues. J. Virol. 2016, 90, 9237–9250. [Google Scholar] [CrossRef] [Green Version]
- Luo, Z.; Tian, D.; Zhou, M.; Xiao, W.; Zhang, Y.; Li, M.; Sui, B.; Wang, W.; Guan, H.; Chen, H.; et al. Lambda-carrageenan P32 is a potent inhibitor of rabies virus infection. PLoS ONE 2015, 10, e0140586. [Google Scholar] [CrossRef] [PubMed]
- Inic-Kanada, A.; Stein, E.; Stojanovic, M.; Schuerer, N.; Ghasemian, E.; Filipovic, A.; Marinkovic, E.; Kosanovic, D.; Barisani-Asenbauer, T. Effects of iota-carrageenan on ocular Chlamydia trachomatis infection in vitro and in vivo. J. Appl. Phycol. 2018, 30, 2601–2610. [Google Scholar] [CrossRef] [Green Version]
- Levendosky, K.; Mizenina, O.; Martinelli, E.; Jean-Pierre, N.; Kizima, L.; Rodriguez, A.; Kleinbeck, K.; Bonnaire, T.; Robbiani, M.; Zydowsky, T.M.; et al. Griffithsin and carrageenan combination to target herpes simplex virus 2 and human papillomavirus. Antimicrob. Agents Chemother. 2015, 59, 7290–7298. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez, A.; Kleinbeck, K.; Mizenina, O.; Kizima, L.; Levendosky, K.; Jean-Pierre, N.; Villegas, G.; Ford, B.; Cooney, M.L.; Teleshova, N.; et al. In vitro and in vivo evaluation of two carrageenan-based formulations to prevent HPV acquisition. Antivir. Res. 2014, 108, 88–93. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, T.; Chattopadhyay, K.; Marschall, M.; Karmakar, P.; Mandal, P.; Ray, B. Focus on antivirally active sulfated polysaccharides: From structure-activity analysis to clinical evaluation. Glycobiology 2009, 19, 2–15. [Google Scholar] [CrossRef]
- Tang, Z.-X.; Fang, X.-J.; Zhang, Z.-L.; Zhou, T.; Zhang, X.-Y.; Shi, L.-E. Nanosize MgO as antibacterial agent: Preparation and characteristics. Braz. J. Chem. Eng. 2012, 29, 775–781. [Google Scholar] [CrossRef]
- Kalitnik, A.A.; Byankina–Barabanova, A.O.; Nagorskaya, V.P.; Reunov, A.V.; Glazunov, V.P.; Solov’eva, T.F.; Yermak, I.M. Low molecular weight derivatives of different carrageenan types and their antiviral activity. J. Appl. Phycol. 2013, 25, 65–72. [Google Scholar] [CrossRef]
- Stancioff, D.J.; Stanley, N.F. Infrared and chemical studies on algal polysaccharides. Proc. Int. Seaweed Symp. 1969, 6, 595–609. [Google Scholar]
- Tissot, B.; Salpin, J.Y.; Martinez, M.; Gaigeot, M.P.; Daniel, R. Differentiation of the fucoidan sulfated L-fucose isomers constituents by CE-ESIMS and molecular modeling. Carbohydr. Res. 2006, 341, 598–609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, G.; Guan, H.; Ioanoviciu, A.S.; Sikkander, S.A.; Thanawiroon, C.; Tobacman, J.K.; Toida, T.; Linhardt, R.J. Structural studies on κ-carrageenan derived oligosaccharides. Carbohydr. Res. 2002, 337, 433–440. [Google Scholar] [CrossRef]
- Goncalves, A.G.; Ducatti, D.R.B.; Grindley, T.B.; Duarte, M.E.R.; Noseda, M.D. ESI-MS differential fragmentation of positional isomers of sulfated oligosaccharides derived from carrageenans and agarans. J. Am. Soc. Mass Spectrom. 2010, 21, 1404–1416. [Google Scholar] [CrossRef] [Green Version]
- Pomin, V.H.; Valente, A.P.; Pereira, M.S.; Mourão, P.A.S. Mild acid hydrolysis of sulfated fucans: A selective 2-desulfation reaction and an alternative approach for preparing tailored sulfated oligosaccharides. Glycobiology 2005, 15, 1376–1385. [Google Scholar] [CrossRef] [PubMed]
- Anastyuk, S.D.; Shevchenko, N.M.; Nazarenko, E.L.; Dmitrenok, P.S.; Zvyagintseva, T.N. Structural analysis of a fucoidan from the brown alga Fucus evanescens by MALDI-TOF and tandem ESI mass spectrometry. Carbohydr. Res. 2009, 344, 779–787. [Google Scholar] [CrossRef]
- Usov, A.I.; Elashvili, M.Y. Quantitative-determination of 3,6-anhydrogalactose derivatives and specific fragmentation of the red algal galactans under reductive hydrolysis conditions. Bioorg. Khim. 1991, 17, 839–848. [Google Scholar] [CrossRef]
- Kravchenko, A.O.; Anastyuk, S.D.; Glazunov, V.P.; Sokolova, E.V.; Isakov, V.V.; Yermak, I.M. Structural characteristics of carrageenans of red alga Mastocarpus pacificus from sea of Japan. Carbohydr. Polym. 2020, 229, 115518. [Google Scholar] [CrossRef]
- Dodgson, K.S.; Price, R.G. A note on the determination of the ester sulphate content of sulphated polysaccharides. J. Biochem. 1962, 84, 106–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rochas, C.; Rinaudo, M.; Landry, S. Role of the molecular weight on the mechanical properties of kappa-carrageenan gels. Carbohydr. Polym. 1990, 12, 255–266. [Google Scholar] [CrossRef]
- Park, J.T.; Johnson, M.J. A submicrodetermination of glucose. J. Biol. Chem. 1949, 181, 149–151. [Google Scholar] [CrossRef]
| Source | Sample of CRGs | Composition, Dry Weight % | MW, kDa | ||
|---|---|---|---|---|---|
| Gal | 3,6 AnGal | SO4 −2 | |||
| CRG-T. crinitus | κ/β- | 39.2 | 33.5 | 20.8 | 350.0 |
| Oligosaccharides (LMW) | κ/β- | 38.1 | 33.0 | 21.0 | 2.1 |
| m/z | Composition and Structural Features of Oligosaccharides | |
|---|---|---|
| MALDI | ESI | |
| 259.0 | 259.004 | G4S |
| 403.1 | 403.034 | G4S-DA |
| 565.1 | 565.079 | G4S-DA-G, G-DA-G4S |
| 583.1 | 583.089 | G4S-D-G, G-D6S-G |
| 667.0 | 322.015 | G4S-DA-G4S, G-DA2S-G4S |
| 685.1 | 331.018 | G4S-D6S-G,G-D6S-G4S |
| 709.1 | - | fragment |
| 727.0 | - | fragment |
| 811.0 | 394.041 | (G4S-DA)2 |
| 829.1 | 403 | G4S-DA-G4S-G |
| 871.2 | 871.188 | G4S-DA-G-DA-G, G-DA-G4S-DA-G, G-DA-G-DA-G4S |
| 889.3 | - | fragment |
| 1009.1 | - | fragment from 355.0233- |
| 1033.2 | - | fragment from 295.0103- |
| 1177.4 | 277.005 | G4S-DA2S-G4S-DA2S-G |
| 1315.3 | 646.106 | G4S-D-G4S-D-G-DA |
| 1483.4 | - | fragment |
| 1585.4 | - | fragment |
| 1789.6 | - | fragment |
| 1891.5 | - | fragment |
| 2095.7 | - | fragment |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yermak, I.; Anastyuk, S.; Kravchenko, A.; Helbert, W.; Glazunov, V.; Shulgin, A.; Spirin, P.; Prassolov, V. New Insights into the Structure of Kappa/Beta-Carrageenan: A Novel Potential Inhibitor of HIV-1. Int. J. Mol. Sci. 2021, 22, 12905. https://doi.org/10.3390/ijms222312905
Yermak I, Anastyuk S, Kravchenko A, Helbert W, Glazunov V, Shulgin A, Spirin P, Prassolov V. New Insights into the Structure of Kappa/Beta-Carrageenan: A Novel Potential Inhibitor of HIV-1. International Journal of Molecular Sciences. 2021; 22(23):12905. https://doi.org/10.3390/ijms222312905
Chicago/Turabian StyleYermak, Irina, Stanislav Anastyuk, Anna Kravchenko, William Helbert, Valery Glazunov, Andrey Shulgin, Pavel Spirin, and Vladimir Prassolov. 2021. "New Insights into the Structure of Kappa/Beta-Carrageenan: A Novel Potential Inhibitor of HIV-1" International Journal of Molecular Sciences 22, no. 23: 12905. https://doi.org/10.3390/ijms222312905
APA StyleYermak, I., Anastyuk, S., Kravchenko, A., Helbert, W., Glazunov, V., Shulgin, A., Spirin, P., & Prassolov, V. (2021). New Insights into the Structure of Kappa/Beta-Carrageenan: A Novel Potential Inhibitor of HIV-1. International Journal of Molecular Sciences, 22(23), 12905. https://doi.org/10.3390/ijms222312905






