The Role of Toll-like Receptors (TLRs) Mediated Inflammation in Pancreatic Cancer Pathophysiology
Abstract
1. Introduction
2. Toll-like Receptors
3. Toll-like Receptor 1
4. Toll-like Receptor 2
5. Toll-like Receptor 3
6. Toll-like Receptor 4
7. Toll-like Receptor 5
8. Toll-like Receptor 7
9. Toll-like Receptor 9
10. TLRs Agonists
11. TLRs Antagonists
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Baine, M.J.; Jain, M.; Sasson, A.R.; Batra, S.K. Early diagnosis of pancreatic cancer: Challenges and new developments. Biomarkers Med. 2012, 6, 597–612. [Google Scholar] [CrossRef]
- Lambert, A.; Schwarz, L.; Borbath, I.; Henry, A.; Van Laethem, J.-L.; Malka, D.; Ducreux, M.; Conroy, T. An update on treatment options for pancreatic adenocarcinoma. Ther. Adv. Med. Oncol. 2019, 11, 1758835919875568. [Google Scholar] [CrossRef]
- Teague, A.; Lim, K.-H.; Wang-Gillam, A. Advanced pancreatic adenocarcinoma: A review of current treatment strategies and developing therapies. Ther. Adv. Med. Oncol. 2015, 7, 68–84. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Hammel, P.; Hebbar, M.; Ben Abdelghani, M.; Wei, A.C.; Raoul, J.-L.; Choné, L.; Francois, E.; Artru, P.; Biagi, J.J.; et al. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N. Engl. J. Med. 2018, 379, 2395–2406. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-J.; Chang, J.S. Environmental Risk Factors of Pancreatic Cancer. J. Clin. Med. 2019, 8, 1427. [Google Scholar] [CrossRef]
- Goral, V. Pancreatic Cancer: Pathogenesis and Diagnosis. Asian Pac. J. Cancer Prev. 2015, 16, 5619–5624. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Fang, Y.; Yao, Q.; Chen, Z.; Xiang, J.; William, F.E.; Gibbs, R.A. Genetic and molecular alterations in pancreatic cancer: Implications for personalized medicine. Med. Sci. Monit. 2013, 19, 916–926. [Google Scholar] [CrossRef] [PubMed]
- Friess, H.; Guo, X.-Z.; Nan, B.-C.; Kleeff, J.; Buchler, M.W. Growth Factors and Cytokines in Pancreatic Carcinogenesis. Ann. N. Y. Acad. Sci. 1999, 880, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Van Duijneveldt, G.; Griffin, M.D.W.; Putoczki, T.L. Emerging roles for the IL-6 family of cytokines in pancreatic cancer. Clin. Sci. 2020, 134, 2091–2115. [Google Scholar] [CrossRef]
- Vaz, J.; Akbarshahi, H.; Andersson, R. Controversial role of toll-like receptors in acute pancreatitis. World J. Gastroenterol. 2013, 19, 616–630. [Google Scholar] [CrossRef]
- Shadhu, K.; Xi, C. Inflammation and pancreatic cancer: An updated review. Saudi J. Gastroenterol. 2019, 25, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Goto, Y.; Narita, N.; Hoon, D.S. Cancer Cells Expressing Toll-like Receptors and the Tumor Microenvironment. Cancer Microenviron. 2009, 2 (Suppl. S1), 205–214. [Google Scholar] [CrossRef] [PubMed]
- Lanki, M.A.; Seppänen, H.E.; Mustonen, H.K.; Böckelman, C.; Juuti, A.T.; Hagström, J.K.; Haglund, C.H. Toll-like receptor 2 and Toll-like receptor 4 predict favorable prognosis in local pancreatic cancer. Tumor Biol. 2018, 40, 1010428318801188. [Google Scholar] [CrossRef] [PubMed]
- Ochi, A.; Graffeo, C.S.; Zambirinis, C.P.; Rehman, A.; Hackman, M.; Fallon, N.; Barilla, R.; Henning, J.R.; Jamal, M.; Rao, R.; et al. Toll-like receptor 7 regulates pancreatic carcinogenesis in mice and humans. J. Clin. Investig. 2012, 122, 4118–4129. [Google Scholar] [CrossRef]
- Vaz, J.; Andersson, R. Intervention on toll-like receptors in pancreatic cancer. World J. Gastroenterol. 2014, 20, 5808–5817. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef]
- Mogensen, T.H. Pathogen Recognition and Inflammatory Signaling in Innate Immune Defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef]
- Anthoney, N.; Foldi, I.; Hidalgo, A. Toll and Toll-like receptor signalling in development. Development 2018, 145, dev156018. [Google Scholar] [CrossRef]
- Botos, I.; Segal, D.M.; Davies, D.R. The Structural Biology of Toll-like Receptors. Structure 2011, 19, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhu, C.; Wu, D.; Jiang, X. Possible Role of Toll-Like Receptor 4 in Acute Pancreatitis. Pancreas 2010, 39, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Muzio, M.; Polentarutti, N.; Bosisio, D.; Manoj Kumar, P.P.; Mantovani, A. Toll-like receptor family and signalling pathway. Biochem. Soc. Trans. 2000, 28, 563–566. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wang, L.; Chen, S. Endogenous toll-like receptor ligands and their biological significance. J. Cell. Mol. Med. 2010, 14, 2592–2603. [Google Scholar] [CrossRef]
- Deguine, J.; Barton, G.M. MyD88: A central player in innate immune signaling. F1000Prime Rep. 2014, 6, 97. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. NF-κB, inflammation, immunity and cancer: Coming of age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar] [CrossRef]
- Mlcochova, P.; Winstone, H.; Zuliani-Alvarez, L.; Gupta, R.K. TLR4-Mediated Pathway Triggers Interferon-Independent G0 Arrest and Antiviral SAMHD1 Activity in Macrophages. Cell Rep. 2020, 30, 3972–3980.e5. [Google Scholar] [CrossRef]
- Mazzone, P.; Congestri, M.; Scudiero, I.; Polvere, I.; Voccola, S.; Zerillo, L.; Telesio, G.; Vito, P.; Stilo, R.; Zotti, T. UBAC1/KPC2 Regulates TLR3 Signaling in Human Keratinocytes through Functional Interaction with the CARD14/CARMA2sh-TANK Complex. Int. J. Mol. Sci. 2020, 21, 9365. [Google Scholar] [CrossRef]
- Jialal, I.; Kaur, H.; Devaraj, S. Toll-like Receptor Status in Obesity and Metabolic Syndrome: A Translational Perspective. J. Clin. Endocrinol. Metab. 2014, 99, 39–48. [Google Scholar] [CrossRef]
- Subramanian, S.; Tus, K.; Li, Q.-Z.; Wang, A.; Tian, X.-H.; Zhou, J.; Liang, C.; Bartov, G.; McDaniel, L.D.; Zhou, X.J.; et al. A Tlr7 translocation accelerates systemic autoimmunity in murine lupus. Proc. Natl. Acad. Sci. USA 2006, 103, 9970–9975. [Google Scholar] [CrossRef]
- So, E.Y.; Ouchi, T. The application of Toll like receptors for cancer therapy. Int. J. Biol. Sci. 2010, 6, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Santoni, M.; Andrikou, K.; Sotte, V.; Bittoni, A.; Lanese, A.; Pellei, C.; Piva, F.; Conti, A.; Nabissi, M.; Santoni, G.; et al. Toll like receptors and pancreatic diseases: From a pathogenetic mechanism to a therapeutic target. Cancer Treat. Rev. 2015, 41, 569–576. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, L.; Jiang, Y.; Song, L.; Liu, Y.; Cheng, F.; Fan, X.; Cao, X.; Gong, A.; Wang, D.; et al. Radiotherapy-induced cell death activates paracrine HMGB1-TLR2 signaling and accelerates pancreatic carcinoma metastasis. J. Exp. Clin. Cancer Res. 2018, 37, 77. [Google Scholar] [CrossRef]
- Grimmig, T.; Moench, R.; Kreckel, J.; Haack, S.; Rueckert, F.; Rehder, R.; Tripathi, S.; Ribas, C.; Chandraker, A.; Germer, C.T.; et al. Toll Like Receptor 2, 4, and 9 Signaling Promotes Autoregulative Tumor Cell Growth and VEGF/PDGF Expression in Human Pancreatic Cancer. Int. J. Mol. Sci. 2016, 17, 2060. [Google Scholar] [CrossRef]
- Chen, X.; Cheng, F.; Liu, Y.; Zhang, L.; Song, L.; Cai, X.; You, T.; Fan, X.; Wang, D.; Gong, A.; et al. Toll-like receptor 2 and Toll-like receptor 4 exhibit distinct regulation of cancer cell stemness mediated by cell death-induced high-mobility group box 1. EBioMedicine 2019, 40, 135–150. [Google Scholar] [CrossRef]
- Schwartz, A.L.; Malgor, R.; Dickerson, E.; Weeraratna, A.T.; Slominski, A.; Wortsman, J.; Harii, N.; Kohn, A.D.; Moon, R.; Schwartz, F.L.; et al. Phenylmethimazole Decreases Toll-Like Receptor 3 and Noncanonical Wnt5a Expression in Pancreatic Cancer and Melanoma Together with Tumor Cell Growth and Migration. Clin. Cancer Res. 2009, 15, 4114–4122. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, M.; Kanda, T.; Shibata, T.; Sasaki, R.; Masuzaki, R.; Matsumoto, N.; Nirei, K.; Imazu, H.; Kuroda, K.; Sugitani, M.; et al. Involvement of the Interferon Signaling Pathways in Pancreatic Cancer Cells. Anticancer Res. 2020, 40, 4445–4455. [Google Scholar] [CrossRef]
- Shojaei, H.; Oberg, H.H.; Juricke, M.; Marischen, L.; Kunz, M.; Mundhenke, C.; Gieseler, F.; Kabelitz, D.; Wesch, D. Toll-like receptors 3 and 7 agonists enhance tumor cell lysis by human gammadelta T cells. Cancer Res. 2009, 69, 8710–8717. [Google Scholar] [CrossRef]
- Fan, P.; Zhang, J.-J.; Wang, B.; Wu, H.-Q.; Zhou, S.-X.; Wang, C.-Y.; Zhang, J.-H.; Tian, Y.; Wu, H.-S. Hypoxia-inducible factor-1 up-regulates the expression of Toll-like receptor 4 in pancreatic cancer cells under hypoxic conditions. Pancreatology 2012, 12, 170–178. [Google Scholar] [CrossRef]
- Zou, B.-B.; Wang, F.; Li, L.; Cheng, F.-W.; Jin, R.; Luo, X.; Zhu, L.-X.; Geng, X.; Zhang, S.-Q. Activation of Toll-like receptor 7 inhibits the proliferation and migration, and induces the apoptosis of pancreatic cancer cells. Mol. Med. Rep. 2015, 12, 6079–6085. [Google Scholar] [CrossRef]
- Grimmig, T.; Matthes, N.; Höland, K.; Tripathi, S.; Chandraker, A.; Grimm, M.; Moench, R.; Moll, E.-M.; Friess, H.; Tsaur, I.; et al. TLR7 and TLR8 expression increases tumor cell proliferation and promotes chemoresistance in human pancreatic cancer. Int. J. Oncol. 2015, 47, 857–866. [Google Scholar] [CrossRef]
- Wang, F.; Jin, R.; Zou, B.B.; Li, L.; Cheng, F.W.; Luo, X.; Geng, X.; Zhang, S.Q. Activation of Toll-like receptor 7 regulates the expression of IFN-lambda1, p53, PTEN, VEGF, TIMP-1 and MMP-9 in pancreatic cancer cells. Mol. Med. Rep. 2016, 13, 1807–1812. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-Q.; Wang, B.; Zhu, S.-K.; Tian, Y.; Zhang, J.-H.; Wu, H.-S. Effects of CPG ODN on biological behavior of PANC-1 and expression of TLR9 in pancreatic cancer. World J. Gastroenterol. 2011, 17, 996–1003. [Google Scholar] [CrossRef]
- Zambirinis, C.P.; Levie, E.; Nguy, S.; Avanzi, A.; Barilla, R.; Xu, Y.; Seifert, L.; Daley, D.; Greco, S.H.; Deutsch, M.; et al. TLR9 ligation in pancreatic stellate cells promotes tumorigenesis. J. Exp. Med. 2015, 212, 2077–2094. [Google Scholar] [CrossRef]
- Letiembre, M.; Liu, Y.; Walter, S.; Hao, W.; Pfander, T.; Wrede, A.; Schulz-Schaeffer, W.; Fassbender, K. Screening of innate immune receptors in neurodegenerative diseases: A similar pattern. Neurobiol. Aging 2009, 30, 759–768. [Google Scholar] [CrossRef]
- Miranda-Hernandez, S.; Baxter, A.G. Role of toll-like receptors in multiple sclerosis. Am. J. Clin. Exp. Immunol. 2013, 2, 75–93. [Google Scholar]
- Jin, M.S.; Kim, S.E.; Heo, J.Y.; Lee, M.E.; Kim, H.M.; Paik, S.-G.; Lee, H.; Lee, J.-O. Crystal Structure of the TLR1-TLR2 Heterodimer Induced by Binding of a Tri-Acylated Lipopeptide. Cell 2007, 130, 1071–1082. [Google Scholar] [CrossRef]
- Netea, M.G.; Van De Veerdonk, F.; Verschueren, I.; Van Der Meer, J.W.; Kullberg, B.J. Role of TLR1 and TLR6 in the host defense against disseminated candidiasis. FEMS Immunol. Med. Microbiol. 2008, 52, 118–123. [Google Scholar] [CrossRef]
- Lanki, M.; Seppänen, H.; Mustonen, H.; Hagström, J.; Haglund, C. Toll-like receptor 1 predicts favorable prognosis in pancreatic cancer. PLoS ONE 2019, 14, e0219245. [Google Scholar] [CrossRef]
- Huynh, A.S.; Chung, W.J.; Cho, H.-I.; Moberg, V.E.; Celis, E.; Morse, D.L.; Vagner, J. Novel Toll-like Receptor 2 Ligands for Targeted Pancreatic Cancer Imaging and Immunotherapy. J. Med. Chem. 2012, 55, 9751–9762. [Google Scholar] [CrossRef]
- Leppänen, J.; Helminen, O.; Huhta, H.; Kauppila, J.H.; Isohookana, J.; Haapasaari, K.-M.; Karihtala, P.; Parkkila, S.; Saarnio, J.; Lehenkari, P.P.; et al. Toll-like receptors 2, 4 and 9 and hypoxia markers HIF-1alpha and CAIX in pancreatic intraepithelial neoplasia. APMIS 2018, 126, 852–863. [Google Scholar] [CrossRef]
- Liu, C.Y.; Xu, J.Y.; Shi, X.Y.; Huang, W.; Ruan, T.Y.; Xie, P.; Ding, J.L. M2-polarized tumor-associated macrophages promoted epithelial-mesenchymal transition in pancreatic cancer cells, partially through TLR4/IL-10 signaling pathway. Lab. Investig. 2013, 93, 844–854. [Google Scholar] [CrossRef]
- Krock, B.L.; Skuli, N.; Simon, M.C. Hypoxia-Induced Angiogenesis: Good and Evil. Genes Cancer 2011, 2, 1117–1133. [Google Scholar] [CrossRef]
- Zhang, M.; Yan, L.; Wang, G.; Jin, R. Resistin effects on pancreatic cancer progression and chemoresistance are mediated through its receptors CAP1 and TLR4. J. Cell. Physiol. 2019, 234, 9457–9466. [Google Scholar] [CrossRef]
- Pushalkar, S.; Hundeyin, M.; Daley, D.; Zambirinis, C.P.; Kurz, E.; Mishra, A.; Mohan, N.; Aykut, B.; Usyk, M.; Torres, L.E.; et al. The Pancreatic Cancer Microbiome Promotes Oncogenesis by Induction of Innate and Adaptive Immune Suppression. Cancer Discov. 2018, 8, 403–416. [Google Scholar] [CrossRef]
- Sethi, V.; Vitiello, G.; Saxena, D.; Miller, G.; Dudeja, V. The Role of the Microbiome in Immunologic Development and its Implication For Pancreatic Cancer Immunotherapy. Gastroenterology 2019, 156, 2097–2115.e2. [Google Scholar] [CrossRef]
- Güngör, C.; Zander, H.; Effenberger, K.E.; Vashist, Y.K.; Kalinina, T.; Izbicki, J.R.; Yekebas, E.; Bockhorn, M. Notch Signaling Activated by Replication Stress–Induced Expression of Midkine Drives Epithelial–Mesenchymal Transition and Chemoresistance in Pancreatic Cancer. Cancer Res. 2011, 71, 5009–5019. [Google Scholar] [CrossRef]
- Leppänen, J.; Helminen, O.; Huhta, H.; Kauppila, J.H.; Isohookana, J.; Haapasaari, K.-M.; Lehenkari, P.; Saarnio, J.; Karttunen, T.J. High toll-like receptor (TLR) 9 expression is associated with better prognosis in surgically treated pancreatic cancer patients. Virchows Arch. 2017, 470, 401–410. [Google Scholar] [CrossRef]
- Jacobs, C.; Duewell, P.; Heckelsmiller, K.; Wei, J.; Bauernfeind, F.; Ellermeier, J.; Kisser, U.; Bauer, C.A.; Dauer, M.; Eigler, A.; et al. An ISCOM vaccine combined with a TLR9 agonist breaks immune evasion mediated by regulatory T cells in an orthotopic model of pancreatic carcinoma. Int. J. Cancer 2011, 128, 897–907. [Google Scholar] [CrossRef]
- Pratesi, G.; Petrangolini, G.; Tortoreto, M.; Addis, A.; Belluco, S.; Rossini, A.; Selleri, S.; Rumio, C.; Menard, S. Balsari, A. Therapeutic synergism of gemcitabine and CpG-oligodeoxynucleotides in an orthotopic human pancreatic carcinoma xenograft. Cancer Res. 2005, 65, 6388–6393. [Google Scholar] [CrossRef]
- Ciernikova, S.; Novisedlakova, M.; Cholujova, D.; Stevurkova, V.; Mego, M. The Emerging Role of Microbiota and Microbiome in Pancreatic Ductal Adenocarcinoma. Biomedicines 2020, 8, 565. [Google Scholar] [CrossRef]
- Lu, Y.-C.; Yeh, W.-C.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Gill, R.; Chen, M.L.; Zhang, F.; Linhardt, R.J.; Dudeja, P.K.; Tobacman, J.K. Toll-like receptor 4 mediates induction of the Bcl10-NFkappaB-interleukin-8 inflammatory pathway by carrageenan in human intestinal epithelial cells. J. Biol. Chem. 2008, 283, 10550–10558. [Google Scholar] [CrossRef]
- Mazzone, P.; Scudiero, I.; Ferravante, A.; Paolucci, M.; D’Andrea, L.E.; Varricchio, E.; Telesio, G.; De Maio, C.; Pizzulo, M.; Zotti, T.; et al. Functional Characterization of Zebrafish (Danio rerio) Bcl10. PLoS ONE 2015, 10, e0122365. [Google Scholar] [CrossRef]
- Calvo-Rodriguez, M.; García-Rodríguez, C.; Villalobos, C.; Núñez, L. Role of Toll Like Receptor 4 in Alzheimer’s Disease. Front. Immunol. 2020, 11, 1588. [Google Scholar] [CrossRef]
- Balducci, C.; Frasca, A.; Zotti, M.; La Vitola, P.; Mhillaj, E.; Grigoli, E.; Iacobellis, M.; Grandi, F.; Messa, M.; Colombo, L.; et al. Toll-like receptor 4-dependent glial cell activation mediates the impairment in memory establishment induced by beta-amyloid oligomers in an acute mouse model of Alzheimer’s disease. Brain Behav. Immun. 2017, 60, 188–197. [Google Scholar] [CrossRef]
- Smith, R.L.; Hebert, H.; Massey, J.; Bowes, J.; Marzo-Ortega, H.; Ho, P.; McHugh, N.J.; Worthington, J.; Barton, A.; Griffiths, C.; et al. Association of Toll-like receptor 4 (TLR4) with chronic plaque type psoriasis and psoriatic arthritis. Arch. Dermatol. Res. 2016, 308, 201–205. [Google Scholar] [CrossRef]
- Sun, L.; Liu, W.; Zhang, L.-J. The Role of Toll-Like Receptors in Skin Host Defense, Psoriasis, and Atopic Dermatitis. J. Immunol. Res. 2019, 2019, 1824624. [Google Scholar] [CrossRef]
- Scudiero, I.; Mazzone, P.; D’Andrea, L.E.; Ferravante, A.; Zotti, T.; Telesio, G.; De Rubis, G.; Reale, C.; Pizzulo, M.; Muralitharan, S.; et al. CARMA2sh and ULK2 control pathogen-associated molecular patterns recognition in human keratinocytes: Psoriasis-linked CARMA2sh mutants escape ULK2 censorship. Cell Death Dis. 2017, 8, e2627. [Google Scholar] [CrossRef]
- Telesio, G.; Scudiero, I.; Pizzulo, M.; Mazzone, P.; Zotti, T.; Voccola, S.; Polvere, I.; Vito, P.; Stilo, R. The E3 Ubiquitin Ligase RNF7 Negatively Regulates CARD14/CARMA2sh Signaling. Int. J. Mol. Sci. 2017, 18, 2581. [Google Scholar] [CrossRef]
- Jiang, N.; Xie, F.; Chen, L.; Chen, F.; Sui, L. The effect of TLR4 on the growth and local inflammatory microenvironment of HPV-related cervical cancer in vivo. Infect. Agents Cancer 2020, 15, 12. [Google Scholar] [CrossRef]
- Tang, X.; Zhu, Y. TLR4 Signaling Promotes Immune Escape of Human Colon Cancer Cells by Inducing Immunosuppressive Cytokines and Apoptosis Resistance. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2012, 20, 15–24. [Google Scholar] [CrossRef]
- Ou, T.; Lilly, M.; Jiang, W. The Pathologic Role of Toll-Like Receptor 4 in Prostate Cancer. Front. Immunol. 2018, 9, 1188. [Google Scholar] [CrossRef]
- Caballero, I.; Boyd, J.; Almiñana, C.; Sanchez-Lopez, J.A.; Basatvat, S.; Montazeri, M.; Lay, N.M.; Elliott, S.; Spiller, D.G.; White, M.R.H.; et al. Understanding the dynamics of Toll-like Receptor 5 response to flagellin and its regulation by estradiol. Sci. Rep. 2017, 7, 40981. [Google Scholar] [CrossRef]
- Rutkowski, M.R.; Stephen, T.L.; Svoronos, N.; Allegrezza, M.J.; Tesone, A.J.; Perales-Puchalt, A.; Brencicova, E.; Escovar-Fadul, X.; Nguyen, J.M.; Cadungog, M.G.; et al. Microbially Driven TLR5-Dependent Signaling Governs Distal Malignant Progression through Tumor-Promoting Inflammation. Cancer Cell 2015, 27, 27–40. [Google Scholar] [CrossRef]
- Petes, C.; Odoardi, N.; Gee, K. The Toll for Trafficking: Toll-Like Receptor 7 Delivery to the Endosome. Front. Immunol. 2017, 8, 1075. [Google Scholar] [CrossRef]
- Müller, T.; Hamm, S.; Bauer, S. TLR9-Mediated Recognition of DNA. Handb. Exp. Pharmacol. 2008, 183, 51–70. [Google Scholar] [CrossRef]
- Kumagai, Y.; Takeuchi, O.; Akira, S. TLR9 as a key receptor for the recognition of DNA. Adv. Drug Deliv. Rev. 2008, 60, 795–804. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Tseng, J.C.; Huang, L.R.; Huang, C.M.; Huang, C.F.; Chuang, T.H. Adjuvant Effect of Toll-Like Receptor 9 Activation on Cancer Immunotherapy Using Checkpoint Blockade. Front. Immunol. 2020, 11, 1075. [Google Scholar] [CrossRef]
- Patra, M.; Shah, M.; Choi, S. Toll-like receptor-induced cytokines as immunotherapeutic targets in cancers and autoimmune diseases. Semin. Cancer Biol. 2020, 64, 61–82. [Google Scholar] [CrossRef]
- Kauppila, J.H.; Korvala, J.; Siirilä, K.; Manni, M.; Mäkinen, L.K.; Hagström, J.; Atula, T.S.; Haglund, C.; Selander, K.S.; Saarnio, J.; et al. Toll-like receptor 9 mediates invasion and predicts prognosis in squamous cell carcinoma of the mobile tongue. J. Oral Pathol. Med. 2015, 44, 571–577. [Google Scholar] [CrossRef]
- Kauppila, J.H.; Takala, H.; Selander, K.S.; Lehenkari, P.P.; Saarnio, J.; Karttunen, T.J. Increased Toll-like receptor 9 expression indicates adverse prognosis in oesophageal adenocarcinoma. Histopathology 2011, 59, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Väisänen, M.-R.; Jukkola-Vuorinen, A.; Vuopala, K.S.; Selander, K.S.; Vaarala, M.H. Expression of Toll-like receptor-9 is associated with poor progression-free survival in prostate cancer. Oncol. Lett. 2013, 5, 1659–1663. [Google Scholar] [CrossRef]
- Ronkainen, H.; Hirvikoski, P.; Kauppila, S.; Vuopala, K.S.; Paavonen, T.K.; Selander, K.S.; Vaarala, M.H. Absent Toll-like receptor-9 expression predicts poor prognosis in renal cell carcinoma. J. Exp. Clin. Cancer Res. 2011, 30, 84. [Google Scholar] [CrossRef]
- Tuomela, J.; Sandholm, J.; Karihtala, P.; Ilvesaro, J.; Vuopala, K.S.; Kauppila, J.H.; Kauppila, S.; Chen, D.; Pressey, C.; Härkönen, P.; et al. Low TLR9 expression defines an aggressive subtype of triple-negative breast cancer. Breast Cancer Res. Treat. 2012, 135, 481–493. [Google Scholar] [CrossRef]
- Adams, S. Toll-like receptor agonists in cancer therapy. Immunotherapy 2009, 1, 949–964. [Google Scholar] [CrossRef]
- Iribarren, K.; Bloy, N.; Buqué, A.; Cremer, I.; Eggermont, A.; Fridman, W.H.; Fucikova, J.; Galon, J.; Špíšek, R.; Zitvogel, L.; et al. Trial Watch: Immunostimulation with Toll-like receptor agonists in cancer therapy. OncoImmunology 2016, 5, e1088631. [Google Scholar] [CrossRef]
- Hussein, W.M.; Liu, T.-Y.; Skwarczynski, M.; Toth, I. Toll-like receptor agonists: A patent review (2011–2013). Expert Opin. Ther. Pat. 2014, 24, 453–470. [Google Scholar] [CrossRef]
- Basith, S.; Manavalan, B.; Yoo, T.H.; Kim, S.G.; Choi, S. Roles of toll-like receptors in Cancer: A double-edged sword for defense and offense. Arch. Pharmacal Res. 2012, 35, 1297–1316. [Google Scholar] [CrossRef]
- Morr, M.; Takeuchi, O.; Akira, S.; Simon, M.M.; Muhlradt, P.F. Differential recognition of structural details of bacterial lipopeptides by toll-like receptors. Eur. J. Immunol. 2002, 32, 3337–3347. [Google Scholar] [CrossRef]
- Takeuchi, O.; Kaufmann, A.; Grote, K.; Kawai, T.; Hoshino, K.; Morr, M.; Mühlradt, P.F.; Akira, S. Cutting Edge: Preferentially theR-Stereoisomer of the Mycoplasmal Lipopeptide Macrophage-Activating Lipopeptide-2 Activates Immune Cells Through a Toll-Like Receptor 2- and MyD88-Dependent Signaling Pathway. J. Immunol. 2000, 164, 554–557. [Google Scholar] [CrossRef]
- Schneider, C.; Schmidt, T.; Ziske, C.; Tiemann, K.; Lee, K.-M.; Uhlinsky, V.; Behrens, P.; Sauerbruch, T.; Schmidt-Wolf, I.G.H.; Mühlradt, P.F.; et al. Tumour suppression induced by the macrophage activating lipopeptide MALP-2 in an ultrasound guided pancreatic carcinoma mouse model. Gut 2004, 53, 355–361. [Google Scholar] [CrossRef]
- Weigt, H.; Muhlradt, P.F.; Emmendorffer, A.; Krug, N.; Braun, A. Synthetic mycoplasma-derived lipopeptide MALP-2 induces maturation and function of dendritic cells. Immunobiology 2003, 207, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Sawahata, R.; Shime, H.; Yamazaki, S.; Inoue, N.; Akazawa, T.; Fujimoto, Y.; Fukase, K.; Matsumoto, M.; Seya, T. Failure of mycoplasma lipoprotein MALP-2 to induce NK cell activation through dendritic cell TLR2. Microbes Infect. 2011, 13, 350–358. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schmidt, J.; Welsch, T.; Jager, D.; Muhlradt, P.F.; Buchler, M.W.; Marten, A. Intratumoural injection of the toll-like receptor-2/6 agonist ‘macrophage-activating lipopeptide-2’ in patients with pancreatic carcinoma: A phase I/II trial. Br. J. Cancer 2007, 97, 598–604. [Google Scholar] [CrossRef]
- Rosendahl, A.H.; Sun, C.; Wu, D.; Andersson, R. Polysaccharide-K (PSK) increases p21WAF/Cip1 and promotes apoptosis in pancreatic cancer cells. Pancreatology 2012, 12, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, A.; Onishi, H.; Imaizumi, A.; Kawamoto, M.; Fujimura, A.; Oyama, Y.; Katano, M. Protein-bound Polysaccharide-K Inhibits Hedgehog Signaling Through Down-regulation of MAML3 and RBPJ Transcription Under Hypoxia, Suppressing the Malignant Phenotype in Pancreatic Cancer. Anticancer Res. 2016, 36, 3945–3952. [Google Scholar] [PubMed]
- Onishi, H.; Morisaki, T.; Nakao, F.; Odate, S.; Morisaki, T.; Katano, M. Protein-bound polysaccharide decreases invasiveness and proliferation in pancreatic cancer by inhibition of hedgehog signaling and HIF-1α pathways under hypoxia. Cancer Lett. 2013, 335, 289–298. [Google Scholar] [CrossRef]
- Kim, S.A.; Lee, Y.; Jung, D.E.; Park, K.H.; Park, J.Y.; Gang, J.; Jeon, S.B.; Park, E.C.; Kim, Y.-G.; Lee, B.; et al. Pancreatic adenocarcinoma up-regulated factor (PAUF), a novel up-regulated secretory protein in pancreatic ductal adenocarcinoma. Cancer Sci. 2009, 100, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.H.; Kang, T.H.; Song, J.S.; Kim, Y.S.; Chung, E.J.; Ylaya, K.; Kim, S.; Koh, S.S.; Chung, J.-Y.; Kim, J.-H.; et al. Elevated expression of pancreatic adenocarcinoma upregulated factor (PAUF) is associated with poor prognosis and chemoresistance in epithelial ovarian cancer. Sci. Rep. 2018, 8, 12161. [Google Scholar] [CrossRef]
- Kim, J.; Chung, J.-Y.; Kim, T.-J.; Lee, J.-W.; Kim, B.-G.; Bae, D.-S.; Choi, C.H.; Hewitt, S.M. Genomic Network-Based Analysis Reveals Pancreatic Adenocarcinoma Up-Regulating Factor-Related Prognostic Markers in Cervical Carcinoma. Front. Oncol. 2018, 8, 465. [Google Scholar] [CrossRef]
- Escudero-Paniagua, B.; Bartolome, R.A.; Rodriguez, S.; De Los Rios, V.; Pintado, L.; Jaen, M.; Lafarga, M.; Fernandez-Acenero, M.J.; Casal, J.I. PAUF/ZG16B promotes colorectal cancer progression through alterations of the mitotic functions and the Wnt/beta-catenin pathway. Carcinogenesis 2020, 41, 203–213. [Google Scholar]
- Park, H.D.; Lee, Y.; Oh, Y.K.; Jung, J.G.; Park, Y.W.; Myung, K.; Kim, K.-H.; Koh, S.S.; Lim, D.-S. Pancreatic adenocarcinoma upregulated factor promotes metastasis by regulating TLR/CXCR4 activation. Oncogene 2010, 30, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Kang, T.H.; Kim, Y.S.; Kim, S.; Yang, B.; Lee, J.-J.; Lee, H.-J.; Lee, J.; Jung, I.D.; Han, H.D.; Lee, S.-H.; et al. Pancreatic adenocarcinoma upregulated factor serves as adjuvant by activating dendritic cells through stimulation of TLR4. Oncotarget 2015, 6, 27751–27762. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Mount, A.; Krstevska, K.; Pejoski, D.; Hardy, M.P.; Owczarek, C.; Scotney, P.; Maraskovsky, E.; Morelli, A.B. The Combination of ISCOMATRIX Adjuvant and TLR Agonists Induces Regression of Established Solid Tumors In Vivo. J. Immunol. 2015, 194, 2199–2207. [Google Scholar] [CrossRef]
- Mehla, K.; Tremayne, J.; Grunkemeyer, J.A.; O’Connell, K.A.; Steele, M.M.; Caffrey, T.C.; Zhu, X.; Yu, F.; Singh, P.K.; Schultes, B.C.; et al. Combination of mAb-AR20.5, anti-PD-L1 and PolyICLC inhibits tumor progression and prolongs survival of MUC1.Tg mice challenged with pancreatic tumors. Cancer Immunol. Immunother. 2018, 67, 445–457. [Google Scholar] [CrossRef]
- Metzger, P.; Kirchleitner, S.V.; Kluge, M.; Koenig, L.M.; Horth, C.; Rambuscheck, C.A.; Bohmer, D.; Ahlfeld, J.; Kobold, S.; Friedel, C.C.; et al. Immunostimulatory RNA leads to functional reprogramming of myeloid-derived suppressor cells in pancreatic cancer. J Immunother. Cancer 2019, 7, 288. [Google Scholar] [CrossRef] [PubMed]
- Gonda, T.A.; Fang, J.; Salas, M.; Do, C.; Hsu, E.; Zhukovskaya, A.; Siegel, A.; Takahashi, R.; Lopez-Bujanda, Z.A.; Drake, C.G.; et al. A DNA Hypomethylating Drug Alters the Tumor Microenvironment and Improves the Effectiveness of Immune Checkpoint Inhibitors in a Mouse Model of Pancreatic Cancer. Cancer Res. 2020, 80, 4754–4767. [Google Scholar] [CrossRef]
- Bhoopathi, P.; Quinn, B.A.; Gui, Q.; Shen, X.-N.; Grossman, S.R.; Das, S.K.; Sarkar, D.; Fisher, P.B.; Emdad, L. Pancreatic Cancer–Specific Cell Death Induced In Vivo by Cytoplasmic-Delivered Polyinosine–Polycytidylic Acid. Cancer Res. 2014, 74, 6224–6235. [Google Scholar] [CrossRef]
- Michaelis, K.A.; Norgard, M.A.; Zhu, X.; Levasseur, P.R.; Sivagnanam, S.; Liudahl, S.M.; Burfeind, K.G.; Olson, B.; Pelz, K.R.; Angeles Ramos, D.M.; et al. The TLR7/8 agonist R848 remodels tumor and host responses to promote survival in pancreatic cancer. Nat. Commun. 2019, 10, 4682. [Google Scholar] [CrossRef]
- Xie, X.M.L.; Zhou, Y.; Shen, W.; Xu, D.; Dou, J.; Shen, B.; Zhou, C. Polysaccharide enhanced NK cell cytotoxicity against pancreatic cancer via TLR4/MAPKs/NF-κB pathway in vitro/vivo. Carbohydr. Polym. 2019, 225, 115223. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.X.; Sun, Y.L.; Yu, Y.; Zhang, J.; Wu, H.Y.; Yu, X.F. Triptolide enhances the sensitivity of pancreatic cancer PANC-1 cells to gemcitabine by inhibiting TLR4/NF-kappaB signaling. Am. J. Transl. Res. 2019, 11, 3750–3760. [Google Scholar]
- Hemmi, H.; Takeuchi, O.; Kawai, T.; Kaisho, T.; Sato, S.; Sanjo, H.; Matsumoto, M.; Hoshino, K.; Wagner, H.; Takeda, K.; et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000, 408, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Noack, J.; Jordi, M.; Zauner, L.; Alessi, D.; Burch, A.; Tinguely, M.; Hersberger, M.; Bernasconi, M.; Nadal, D. TLR9 agonists induced cell death in Burkitt’s lymphoma cells is variable and influenced by TLR9 polymorphism. Cell Death Dis. 2012, 3, e323. [Google Scholar] [CrossRef][Green Version]
- Krieg, A.M. Therapeutic potential of Toll-like receptor 9 activation. Nat. Rev. Drug Discov. 2006, 5, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Rosa, R.; Melisi, D.; Damiano, V.; Bianco, R.; Garofalo, S.; Gelardi, T.; Agrawal, S.; Di Nicolantonio, F.; Scarpa, A.; Bardelli, A.; et al. Toll-like Receptor 9 Agonist IMO Cooperates with Cetuximab in K-Ras Mutant Colorectal and Pancreatic Cancers. Clin. Cancer Res. 2011, 17, 6531–6541. [Google Scholar] [CrossRef]
- Damiano, V.; Caputo, R.; Bianco, R.; D’Armiento, F.P.; Leonardi, A.; De Placido, S.; Bianco, A.R.; Agrawal, S.; Ciardiello, F.; Tortora, G. Novel Toll-Like Receptor 9 Agonist Induces Epidermal Growth Factor Receptor (EGFR) Inhibition and Synergistic Antitumor Activity with EGFR Inhibitors. Clin. Cancer Res. 2006, 12, 577–583. [Google Scholar] [CrossRef]
- Carbone, C.; Piro, G.; Agostini, A.; Delfino, P.; De Sanctis, F.; Nasca, V.; Spallotta, F.; Sette, C.; Martini, M.; Ugel, S.; et al. Intratumoral injection of TLR9 agonist promotes an immunopermissive microenvironment transition and causes cooperative antitumor activity in combination with anti-PD1 in pancreatic cancer. J. Immunother. Cancer 2021, 9, e002876. [Google Scholar] [CrossRef]
- Dowling, J.K.; Dellacasagrande, J. Toll-Like Receptors: Ligands, Cell-Based Models, and Readouts for Receptor Action. Methods Mol. Biol. 2016, 1390, 3–27. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kim, S.J.; Park, H.D.; Park, E.H.; Huang, S.M.; Jeon, S.B.; Kim, J.-M.; Lim, D.-S.; Koh, S.S. PAUF functions in the metastasis of human pancreatic cancer cells and upregulates CXCR4 expression. Oncogene 2010, 29, 56–67. [Google Scholar] [CrossRef]
- Murata, M. Activation of Toll-like receptor 2 by a novel preparation of cell wall skeleton from Mycobacterium bovis BCG Tokyo (SMP-105) sufficiently enhances immune responses against tumors. Cancer Sci. 2008, 99, 1435–1440. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-K.; Balic, J.J.; Yu, L.; Jenkins, B. TLR Agonists as Adjuvants for Cancer Vaccines. Adv. Exp. Med. Biol. 2017, 1024, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Schulz, O.; Diebold, S.S.; Chen, M.; Näslund, T.I.; Nolte, M.A.; Alexopoulou, L.; Azuma, Y.-T.; Flavell, R.A.; Liljestrom, P.; Sousa, C.R.E. Toll-like receptor 3 promotes cross-priming to virus-infected cells. Nature 2005, 433, 887–892. [Google Scholar] [CrossRef]
- Kodama, S.; Nomi, N.; Suzuki, M.; Kodama, N. Toll-like receptor 3 signaling induces apoptosis in human head and neck cancer via survivin associated pathway. Oncol. Rep. 2010, 24, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Saxena, M.; Sabado, R.L.; La Mar, M.; Mohri, H.; Salazar, A.M.; Dong, H.; Da Rosa, J.C.; Markowitz, M.; Bhardwaj, N.; Miller, E. Poly-ICLC, a TLR3 Agonist, Induces Transient Innate Immune Responses in Patients With Treated HIV-Infection: A Randomized Double-Blinded Placebo Controlled Trial. Front. Immunol. 2019, 10, 725. [Google Scholar] [CrossRef]
- Martins, K.A.O.; Bavari, S.; Salazar, A.M. Vaccine adjuvant uses of poly-IC and derivatives. Expert Rev. Vaccines 2015, 14, 447–459. [Google Scholar] [CrossRef]
- Wang, X.; Smith, C.; Yin, H. Targeting Toll-like receptors with small molecule agents. Chem. Soc. Rev. 2013, 42, 4859–4866. [Google Scholar] [CrossRef]
- Seya, T.; Takeda, Y.; Matsumoto, M. A Toll-like receptor 3 (TLR3) agonist ARNAX for therapeutic immunotherapy. Adv. Drug Deliv. Rev. 2019, 147, 37–43. [Google Scholar] [CrossRef]
- Fensterheim, B.A.; Young, J.D.; Luan, L.; Kleinbard, R.R.; Stothers, C.L.; Patil, N.K.; McAtee-Pereira, A.G.; Guo, Y.; Trenary, I.; Hernandez, A.; et al. The TLR4 Agonist Monophosphoryl Lipid A Drives Broad Resistance to Infection via Dynamic Reprogramming of Macrophage Metabolism. J. Immunol. 2018, 200, 3777–3789. [Google Scholar] [CrossRef]
- Rebuffini, E.; Zuccarino, L.; Grecchi, E.; Carinci, F.; Merulla, V.E. Picibanil (OK-432) in the treatment of head and neck lymphangiomas in children. Dent. Res. J. 2012, 9 (Suppl. S2), S192–S196. [Google Scholar] [CrossRef]
- Didierlaurent, A.M.; Morel, S.; Lockman, L.; Giannini, S.L.; Bisteau, M.; Carlsen, H.; Kielland, A.; Vosters, O.; Vanderheyde, N.; Schiavetti, F.; et al. AS04, an Aluminum Salt- and TLR4 Agonist-Based Adjuvant System, Induces a Transient Localized Innate Immune Response Leading to Enhanced Adaptive Immunity. J. Immunol. 2009, 183, 6186–6197. [Google Scholar] [CrossRef]
- Lu, H.; Betancur, A.; Chen, M.; Ter Meulen, J.H. Toll-Like Receptor 4 Expression on Lymphoma Cells Is Critical for Therapeutic Activity of Intratumoral Therapy With Synthetic TLR4 Agonist Glucopyranosyl Lipid A. Front. Oncol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Sun, F.; Yang, G.; Wang, L.; Zhang, Q.; Zhang, Q.; Zhan, Y.; Chen, J.; Yu, M.; Li, C.; et al. CBLB502, a Toll-like receptor 5 agonist, offers protection against radiation-induced male reproductive system damage in mice †. Biol. Reprod. 2019, 100, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Eremina, N.V.; Kazey, V.I.; Mishugin, S.V.; Leonenkov, R.V.; Pushkar, D.Y.; Mett, V.L.; Gudkov, A.V. First-in-human study of anticancer immunotherapy drug candidate mobilan: Safety, pharmacokinetics and pharmacodynamics in prostate cancer patients. Oncotarget 2020, 11, 1273–1288. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Wang, M.; DeMaria, O.; Tang, J.; Rocchi, P.; Qu, F.; Iovanna, J.L.; Alexopoulou, L.; Peng, L. A Novel Bitriazolyl Acyclonucleoside Endowed with Dual Antiproliferative and Immunomodulatory Activity. J. Med. Chem. 2012, 55, 5642–5646. [Google Scholar] [CrossRef] [PubMed]
- Royer, C.M.; Rudolph, K.; Dietsch, G.N.; Hershberg, R.M.; Barrett, E.G. VTX-1463, a novel TLR-8 agonist, attenuates nasal congestion after ragweed challenge in sensitized beagle dogs. Immun. Inflamm. Dis. 2016, 4, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.L.; Davis, H.L.; Morris, M.L.; Efler, S.M.; Adhami, M.A.; Krieg, A.M.; Cameron, D.W.; Heathcote, J. CPG 7909, an immunostimulatory TLR9 agonist oligodeoxynucleotide, as adjuvant to Engerix-B HBV vaccine in healthy adults: A double-blind phase I/II study. J. Clin. Immunol. 2004, 24, 693–701. [Google Scholar] [CrossRef]
- Agrawal, S.; Kandimalla, E.R. Synthetic agonists of Toll-like receptors 7, 8 and 9. Biochem. Soc. Trans. 2007, 35, 1461–1467. [Google Scholar] [CrossRef]
- Wittig, B.; Schmidt, M.; Scheithauer, W.; Schmoll, H.-J. MGN1703, an immunomodulator and toll-like receptor 9 (TLR-9) agonist: From bench to bedside. Crit. Rev. Oncol. 2015, 94, 31–44. [Google Scholar] [CrossRef]
- Kapp, K.; Kleuss, C.; Schroff, M.; Wittig, B. Genuine Immunomodulation With dSLIM. Mol. Ther.—Nucleic Acids 2014, 3, e170. [Google Scholar] [CrossRef]
- Smith, M.; García-Martínez, E.; Pitter, M.R.; Fucikova, J.; Spisek, R.; Zitvogel, L.; Kroemer, G.; Galluzzi, L. Trial Watch: Toll-like receptor agonists in cancer immunotherapy. OncoImmunology 2018, 7, e1526250. [Google Scholar] [CrossRef]
- Qi, X.-F.; Zheng, L.; Kim, C.-S.; Lee, K.-J.; Kim, D.-H.; Cai, D.-Q.; Qin, J.-W.; Yu, Y.-H.; Wu, Z.; Kim, S.-K. CpG oligodeoxynucleotide induces apoptosis and cell cycle arrest in A20 lymphoma cells via TLR9-mediated pathways. Mol. Immunol. 2013, 54, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Lederman, M.M.; Salkowitz, J.R.; Rodriguez, B.; Harding, C.V.; Sieg, S.F. Impaired monocyte maturation in response to CpG oligodeoxynucleotide is related to viral RNA levels in human immunodeficiency virus disease and is at least partially mediated by deficiencies in alpha/beta interferon responsiveness and production. J. Virol. 2005, 79, 4109–4119. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.P.; O’Donnell, M.A.; Griffith, T.S. Role of neutrophils in BCG immunotherapy for bladder cancer. Urol. Oncol. Semin. Orig. Investig. 2008, 26, 341–345. [Google Scholar] [CrossRef]
- Kanzler, H.; Barrat, F.J.; Hessel, E.M.; Coffman, R.L. Therapeutic targeting of innate immunity with Toll-like receptor agonists and antagonists. Nat. Med. 2007, 13, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, N. Therapeutic targeting of pattern-recognition receptors. Int. Immunopharmacol. 2006, 6, 863–869. [Google Scholar] [CrossRef]
- Raja, S.G.; Dreyfus, G.D. Eritoran: The evidence of its therapeutic potential in sepsis. Core Évid. 2008, 2, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, A.; Tomita, T.; Ohto, U.; Takemura, K.; Kitao, A.; Akashi-Takamura, S.; Miyake, K.; Maru, Y. Eritoran inhibits S100A8-mediated TLR4/MD-2 activation and tumor growth by changing the immune microenvironment. Oncogene 2016, 35, 1445–1456. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.-T.; Lee, T.-C.; Yu, L.C.-H. Eritoran Suppresses Colon Cancer by Altering a Functional Balance in Toll-like Receptors That Bind Lipopolysaccharide. Cancer Res. 2016, 76, 4684–4695. [Google Scholar] [CrossRef]
- Ii, M.; Matsunaga, N.; Hazeki, K.; Nakamura, K.; Takashima, K.; Seya, T.; Hazeki, O.; Kitazaki, T.; Iizawa, Y. A Novel Cyclohexene Derivative, Ethyl (6R)-6-[N-(2-Chloro-4-fluorophenyl)sulfamoyl]cyclohex-1-ene-1-carboxylate (TAK-242), Selectively Inhibits Toll-Like Receptor 4-Mediated Cytokine Production through Suppression of Intracellular Signaling. Mol. Pharmacol. 2006, 69, 1288–1295. [Google Scholar] [CrossRef]
- Kashani, B.; Zandi, Z.; Bashash, D.; Zaghal, A.; Momeny, M.; Poursani, E.M.; Pourbagheri-Sigaroodi, A.; Mousavi, S.A.; Ghaffari, S.H. Small molecule inhibitor of TLR4 inhibits ovarian cancer cell proliferation: New insight into the anticancer effect of TAK-242 (Resatorvid). Cancer Chemother. Pharmacol. 2020, 85, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Zandi, Z.; Kashani, B.; Bashash, D.; Poursani, E.M.; Mousavi, S.A.; Chahardoli, B.; Ghaffari, S.H. The anticancer effect of the TLR4 inhibition using TAK-242 (resatorvid) either as a single agent or in combination with chemotherapy: A novel therapeutic potential for breast cancer. J. Cell. Biochem. 2019, 121, 1623–1634. [Google Scholar] [CrossRef] [PubMed]
- Zandi, Z.; Kashani, B.; Poursani, E.M.; Bashash, D.; Kabuli, M.; Momeny, M.; Mousavi-Pak, S.H.; Sheikhsaran, F.; Alimoghaddam, K.; Mousavi, S.A.; et al. TLR4 blockade using TAK-242 suppresses ovarian and breast cancer cells invasion through the inhibition of extracellular matrix degradation and epithelial-mesenchymal transition. Eur. J. Pharmacol. 2019, 853, 256–263. [Google Scholar] [CrossRef]
- Kashani, B.; Zandi, Z.; Karimzadeh, M.R.; Bashash, D.; Nasrollahzadeh, A.; Ghaffari, S.H. Blockade of TLR4 using TAK-242 (resatorvid) enhances anti-cancer effects of chemotherapeutic agents: A novel synergistic approach for breast and ovarian cancers. Immunol. Res. 2019, 67, 505–516. [Google Scholar] [CrossRef]
- Blohm-Mangone, K.; Burkett, N.B.; Tahsin, S.; Myrdal, P.B.; Aodah, A.; Ho, B.; Janda, J.; McComas, M.; Saboda, K.; Roe, D.J.; et al. Pharmacological TLR4 Antagonism Using Topical Resatorvid Blocks Solar UV-Induced Skin Tumorigenesis in SKH-1 Mice. Cancer Prev. Res. 2018, 11, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Brown, K.; King, C.; Awasthi, V.; Bondugula, R. A Toll-Like Receptor-4-Interacting Surfactant Protein-A-Derived Peptide Suppresses Tumor Necrosis Factor-α Release from Mouse JAWS II Dendritic Cells. J. Pharmacol. Exp. Ther. 2011, 336, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Rakhesh, M.; Cate, M.; Vijay, R.; Shrikant, A.; Shanjana, A. A TLR4-interacting peptide inhibits lipopolysaccharide-stimulated inflammatory responses, migration and invasion of colon cancer SW480 cells. OncoImmunology 2012, 1, 1495–1506. [Google Scholar] [CrossRef][Green Version]
- Awasthi, S.; Singh, B.; Ramani, V.; Xie, J.; Kosanke, S. TLR4-interacting SPA4 peptide improves host defense and alleviates tissue injury in a mouse model of Pseudomonas aeruginosa lung infection. PLoS ONE 2019, 14, e0210979. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Rahman, N.; Rui, B.; Kumar, G.; Awasthi, V.; Breshears, M.; Kosanke, S. Lung and general health effects of Toll-like receptor-4 (TLR4)-interacting SPA4 peptide. BMC Pulm. Med. 2020, 20, 179. [Google Scholar] [CrossRef]
- Baker, P.J.; Hraba, T.; Taylor, C.E.; Myers, K.R.; Takayama, K.; Qureshi, N.; Stuetz, P.; Kusumoto, S.; Hasegawa, A. Structural features that influence the ability of lipid A and its analogs to abolish expression of suppressor T cell activity. Infect. Immun. 1992, 60, 2694–2701. [Google Scholar] [CrossRef]
- Stöver, A.G.; Correia, J.D.S.; Evans, J.; Cluff, C.W.; Elliott, M.W.; Jeffery, E.W.; Johnson, D.A.; Lacy, M.J.; Baldridge, J.R.; Probst, P.; et al. Structure-Activity Relationship of Synthetic Toll-like Receptor 4 Agonists. J. Biol. Chem. 2004, 279, 4440–4449. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Zhang, Z.J.; Cao, C.B.; Wang, N.; Liu, F.F.; Peng, J.Q.; Ren, X.J.; Qian, J. The TLR4/NF-kappaB signaling pathway mediates the growth of colon cancer. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3834–3843. [Google Scholar] [PubMed]
- Huselton, E.C.A.; DiPersio, J.F.; Jacoby, M.; Pusic, I.; Romee, R.; Schroeder, M.; Uy, G.L.; Westervelt, P. Updated Study Results of CX-01, an Inhibitor of CXCL12/CXCR4, and Azacitidine for the Treatment of Hypomethylating Agent Refractory AML and MDS. Blood 2019, 134, 3915. [Google Scholar] [CrossRef]
- Kovacsovics, T.J.; Mims, A.; Salama, M.E.; Pantin, J.; Rao, N.; Kosak, K.M.; Ahorukomeye, P.; Glenn, M.J.; Deininger, M.W.N.; Boucher, K.M.; et al. Combination of the low anticoagulant heparin CX-01 with chemotherapy for the treatment of acute myeloid leukemia. Blood Adv. 2018, 2, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tu, Q.; Yan, W.; Xiao, D.; Zeng, Z.; Ouyang, Y.; Huang, L.; Cai, J.; Zeng, X.; Chen, Y.J.; et al. CXC195 suppresses proliferation and inflammatory response in LPS-induced human hepatocellular carcinoma cells via regulating TLR4-MyD88-TAK1-mediated NF-kappaB and MAPK pathway. Biochem. Biophys. Res. Commun. 2015, 456, 373–379. [Google Scholar] [CrossRef]
- Chen, X.-L.; Fu, J.-P.; Shi, J.; Wan, P.; Cao, H.; Tang, Z.-M. CXC195 induces apoptosis and endoplastic reticulum stress in human hepatocellular carcinoma cells by inhibiting the PI3K/Akt/mTOR signaling pathway. Mol. Med. Rep. 2015, 12, 8229–8236. [Google Scholar] [CrossRef]
- Zeng, T.; Peng, L.; Chao, C.; Fu, B.; Wang, G.; Wang, Y.; Zhu, X. WITHDRAWN: IRE1α-TRAF2-ASK1 complex-mediated endoplasmic reticulum stress and mitochondrial dysfunction contribute to CXC195-induced apoptosis in human bladder carcinoma T24 cells. Biochem. Biophys. Res. Commun. 2015, 460, 530–536. [Google Scholar] [CrossRef]
- Czeslick, E.; Struppert, A.; Simm, A.; Sablotzki, A. E5564 (Eritoran) inhibits lipopolysaccharide-induced cytokine production in human blood monocytes. Inflamm. Res. 2006, 55, 511–515. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, G.; Huang, J.; Ma, S.; Mi, K.; Cheng, J.; Zhu, Y.; Zha, X.; Huang, W. Atractylenolide I modulates ovarian cancer cell-mediated immunosuppression by blocking MD-2/TLR4 complex-mediated MyD88/NF-kappaB signaling in vitro. J. Transl. Med. 2016, 14, 104. [Google Scholar] [CrossRef]
- Premkumar, V.; Dey, M.; Dorn, R.; Raskin, I. MyD88-dependent and independent pathways of Toll-Like Receptors are engaged in biological activity of Triptolide in ligand-stimulated macrophages. BMC Chem. Biol. 2010, 10, 3. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, Q.; Qian, R.; Liu, S.; Hu, W.; Liu, Z. Paeonol antagonizes oncogenesis of osteosarcoma by inhibiting the function of TLR4/MAPK/NF-kappaB pathway. Acta Histochem. 2020, 122, 151455. [Google Scholar] [CrossRef] [PubMed]
- Monnet, E.; Lapeyre, G.; Van Poelgeest, E.; Jacqmin, P.; De Graaf, K.; Reijers, J.; Moerland, M.; Burggraaf, J.; De Min, C. Evidence of NI-0101 pharmacological activity, an anti-TLR4 antibody, in a randomized phase I dose escalation study in healthy volunteers receiving LPS. Clin. Pharmacol. Ther. 2017, 101, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.-L.; Wang, H.-Q.; Yang, J.; Yang, P.; Kang, P.; Zhao, T. Effects of nalmefene hydrochloride on TLR4 signaling pathway in rats with lung ischemia-reperfusion injury. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 461–468. [Google Scholar]
- Li, X.; Zou, Y.; Fu, Y.Y.; Xing, J.; Wang, K.Y.; Wan, P.Z.; Wang, M.; Zhai, X.Y. Ibudilast Attenuates Folic Acid-Induced Acute Kidney Injury by Blocking Pyroptosis Through TLR4-Mediated NF-kappaB and MAPK Signaling Pathways. Front. Pharmacol. 2021, 12, 650283. [Google Scholar] [CrossRef]
- Gowayed, M.A.; El Achy, S.; Kamel, M.A.; El-Tahan, R.A. Polymyxin B prevents the development of adjuvant arthritis via modulation of TLR/Cox-2 signaling pathway. Life Sci. 2020, 259, 118250. [Google Scholar] [CrossRef]
- Arslan, F.; Houtgraaf, J.H.; Keogh, B.; Kazemi, K.; de Jong, R.; McCormack, W.J.; O’Neill, L.A.; McGuirk, P.; Timmers, L.; Smeets, M.B.; et al. Treatment with OPN-305, a humanized anti-Toll-Like receptor-2 antibody, reduces myocardial ischemia/reperfusion injury in pigs. Circ. Cardiovasc. Interv. 2012, 5, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Lamphier, M.; Zheng, W.; Latz, E.; Spyvee, M.; Hansen, H.; Rose, J.; Genest, M.; Yang, H.; Shaffer, C.; Zhao, Y.; et al. Novel Small Molecule Inhibitors of TLR7 and TLR9: Mechanism of Action and Efficacy In Vivo. Mol. Pharmacol. 2014, 85, 429–440. [Google Scholar] [CrossRef] [PubMed]
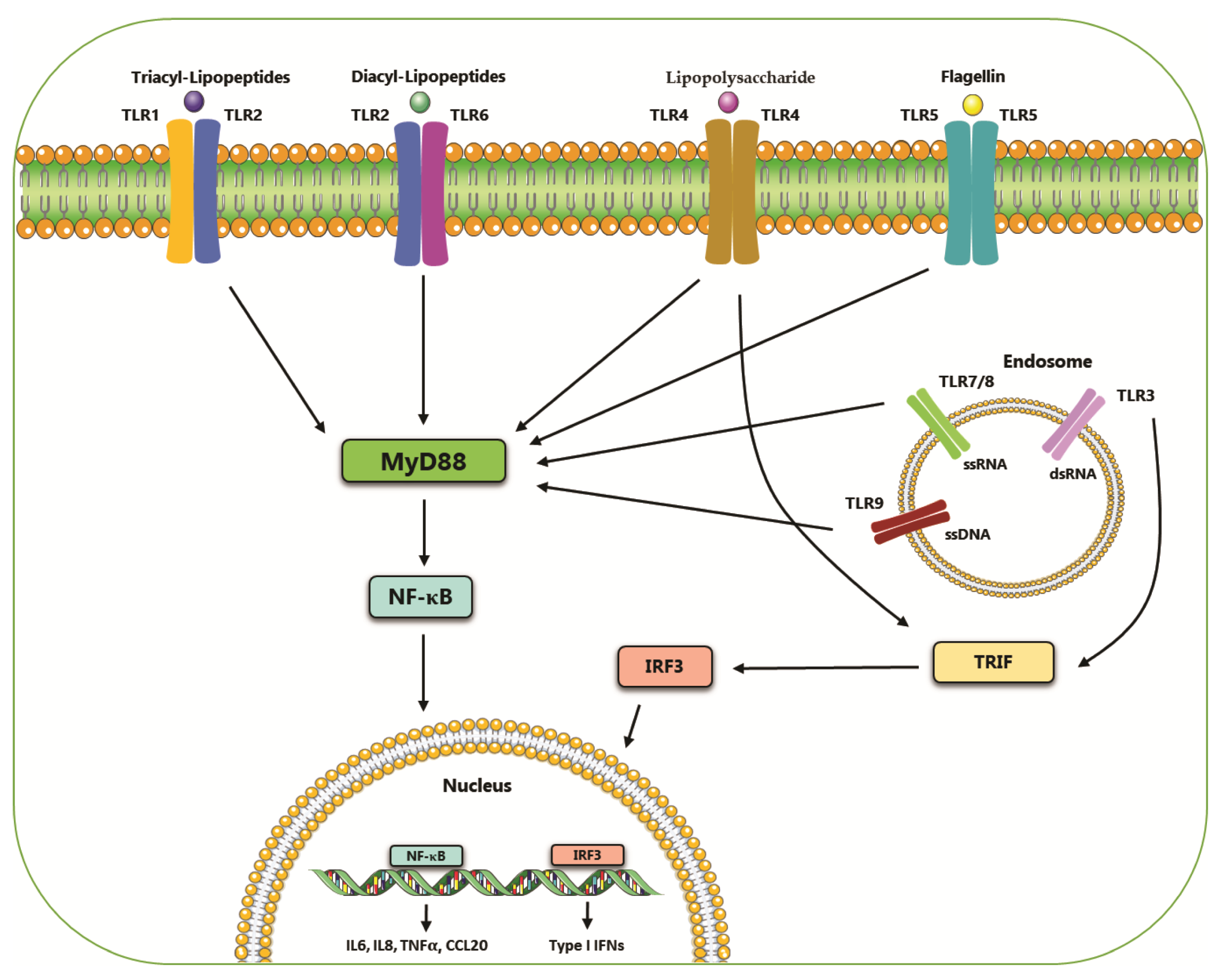
| TLRs | Localization | PAMPs | Adaptor | Pancreatic Cancer Cell Line | Refs. |
|---|---|---|---|---|---|
| TLR1 | Plasma membrane | Triacyl lipopeptides | Myd88/TIRAP | Not reported | [19,32] |
| TLR2 | Plasma membrane | Lycolipids, lipoprotein, lipoteichoic acid, peptidoglycan, zymosan | Myd88/TIRAP | BxPC-3; MIA PaCa-2, MDA Panc-28, SU 8686, SW-1990, AsPC-1, Panc-1 | [19,33,34,35] |
| TLR3 | Endosome | Double-stranded RNA | TRIF | AsPC-1, Colo357, Panc-89, PancTu-1, Pt45P1 | [36,37,38] |
| TLR4 | Plasma membrane | Lipopolysaccharide (LPS), heat shock proteins | Myd88/TIRAP | AsPC-1, BxPC-3, CFPAC, MIA PaCa-2, MDA Panc-28, Panc-1, Sw-1990 | [34,35,39] |
| TLR5 | Plasma membrane | Flagellin | Myd88 | Not reported | [32] |
| TLR6 | Plasma membrane | Diacyl lipopeptides, lipoteichoic acid | Myd88/TIRAP | Not reported | [32] |
| TLR7 | Endosome | Single-stranded RNA | Myd88 | Colo357, MIA PaCa-2, MDA Panc-28, Panc-1, Sw-1990, Panc-89, PancTu-1, BxPC-3 | [38,40,41,42] |
| TLR8 | Endosome | Single-stranded RNA | Myd88 | Panc-1 | [41] |
| TLR9 | Endosome | DNA (CpG) | Myd88 | GER, MIA PaCa-2, MDA Panc-28, Panc-1, Sw-1990, T3M4 | [34,43,44] |
| TLR10 | Endosome | Unknown | Unknown | Not reported | [32] |
| TLRs | Expression | Pancreatic Cancer Prognosis | Refs. |
|---|---|---|---|
| TLR1 | High | Favorable | [49] |
| TLR2 | High | Favorable | [15] |
| Unfavorable | [33,34,35,51] | ||
| TLR3 | High | Unfavorable | [36] |
| TLR4 | High | Favorable | [15] |
| Unfavorable | [39,52,53,54] | ||
| TLR5 | High | Unfavorable | [55,56] |
| TLR7 | High | Unfavorable | [16,41,57] |
| TLR8 | High | Unfavorable | [41,57] |
| TLR9 | High | Favorable | [43,58,59,60] |
| Unfavorable | [44,61] |
| Drug Name | Target | Drug Class | Refs. |
|---|---|---|---|
| Pam3CSK4 | TLR1/2 | Synthetic triacylated lipopeptide | [119] |
| MALP-2 | TLR2/6 | synthetic lipopeptide | [90,91] |
| PSK | TLR2 | Protein-bound polysaccharide | [96] |
| PAUF | TLR2/4 | Peptide | [103,120] |
| SMP-105 | TLR2 | Components of cell wall skeleton isolated from Mycobacterium Bovis | [121] |
| CBLB612 | TLR2/6 | Synthetic derivative of mycoplasma lipopeptide | [122] |
| Phenylmethimazole (C10) | TLR3 | Methimazole derivative | [36] |
| Poly I:C | TLR3 | Synthetic analog of viral dsRNA (polyinosinic-polycytidylic acid) | [123,124] |
| PolyICLC | TLR3 | Polyinosinic-polycytidylic acid mixed with the stabilizers carboxymethylcellulose and polylysine | [125] |
| Poly-IC12U | TLR3 | Poly I:C derivative with shorter half life and less toxicity | [126] |
| IPH 3102 | TLR3 | Synthetic dsRNA agent | [127] |
| ARNAX | TLR3 | Synthetic DNA/RNA hybrid molecule | [128] |
| MPLA | TLR4 | Lipid A derivative | [129] |
| OK-432 | TLR4 | Lyophilized mixture of group A Streptococcus pyogenes | [130] |
| AS04 | TLR4 | Combination of MPLA and aluminum salt | [131] |
| GLA-SE (G100) | TLR4 | Glucopyranosyl lipid-A oil-in-water emulsion | [132] |
| CBLB502 | TLR5 | derivative of Salmonella flagellin | [133] |
| M-VM3 (Mobilan) | TLR5 | Recombinant non-replicating adenovirus that directs expression of human Toll-like receptor 5 and of a flagellin derivative that acts as a selective agonist of TLR5 | [134] |
| ssRNA40 | TLR7 | 20-mer phosphorothioate protected single-stranded RNA oligonucleotide containing a GU-rich sequence | [16] |
| Gardiquimod | TLR7 | Imidazoquinoline compound | [42] |
| Resiquimod (R848) | TLR7 | Imidazoquinoline compound | [110] |
| Bistriazolyl | TLR7 | Small Molecule | [135] |
| VTX1463 | TLR8 | Small Molecule | [136] |
| CpG-1826 | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [122] |
| CpG-7909 | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [137] |
| IMO-2155 | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [138] |
| MGN1703 | TLR9 | Covalently closed natural DNA molecule | [139] |
| dSLIM | TLR9 | MGN1703 derivative | [140] |
| SD-101 | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [141] |
| KSK-CpG | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [142] |
| ODN2216 | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [143] |
| ODN M362 | TLR9 | Oligodeoxynucleotide containing immunostimulatory CpG motifs | [144] |
| Drug Name | Target | Drug Class | Refs. |
|---|---|---|---|
| TAK-242 (Resatorvid) | TLR4 | Small molecule inhibitor | [150] |
| CRX-526 | TLR4 | Synthetic lipopolysaccharide | [163] |
| CX-01 | TLR4 | heparin-derived olysaccharide | [164] |
| CXC195 | TLR4 | tetramethylpyrazine analogue | [165] |
| Eritoran (E5564) | TLR4 | Synthetic lipopolysaccharide | [168] |
| Atractylenolide-1 | TLR4 | sesquiterpene compound | [169] |
| Triptolide | TLR4 | diterpenoid epoxide | [170] |
| Paeonol | TLR4 | Small molecule inhibitor | [171] |
| NI-0101 | TLR4 | Monoclonal antibody | [172] |
| Nalmefene (JKB-121) | TLR4 | Small molecule inhibitor | [173] |
| Ibudilast (AV-411, N-166) | TLR4 | Small molecule inhibitor | [174] |
| Polymyxin B (PMB) | TLR4 | Cyclic polypeptide antibiotic | [175] |
| OPN305 | TLR2 | Monoclonal antibody | [176] |
| Hydroxychloroquine | TLR7,9 | Quinolone | [177] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orlacchio, A.; Mazzone, P. The Role of Toll-like Receptors (TLRs) Mediated Inflammation in Pancreatic Cancer Pathophysiology. Int. J. Mol. Sci. 2021, 22, 12743. https://doi.org/10.3390/ijms222312743
Orlacchio A, Mazzone P. The Role of Toll-like Receptors (TLRs) Mediated Inflammation in Pancreatic Cancer Pathophysiology. International Journal of Molecular Sciences. 2021; 22(23):12743. https://doi.org/10.3390/ijms222312743
Chicago/Turabian StyleOrlacchio, Arturo, and Pellegrino Mazzone. 2021. "The Role of Toll-like Receptors (TLRs) Mediated Inflammation in Pancreatic Cancer Pathophysiology" International Journal of Molecular Sciences 22, no. 23: 12743. https://doi.org/10.3390/ijms222312743
APA StyleOrlacchio, A., & Mazzone, P. (2021). The Role of Toll-like Receptors (TLRs) Mediated Inflammation in Pancreatic Cancer Pathophysiology. International Journal of Molecular Sciences, 22(23), 12743. https://doi.org/10.3390/ijms222312743







