PM2.5 Induces Early Epithelial Mesenchymal Transition in Human Proximal Tubular Epithelial Cells through Activation of IL-6/STAT3 Pathway
Abstract
:1. Introduction
2. Results
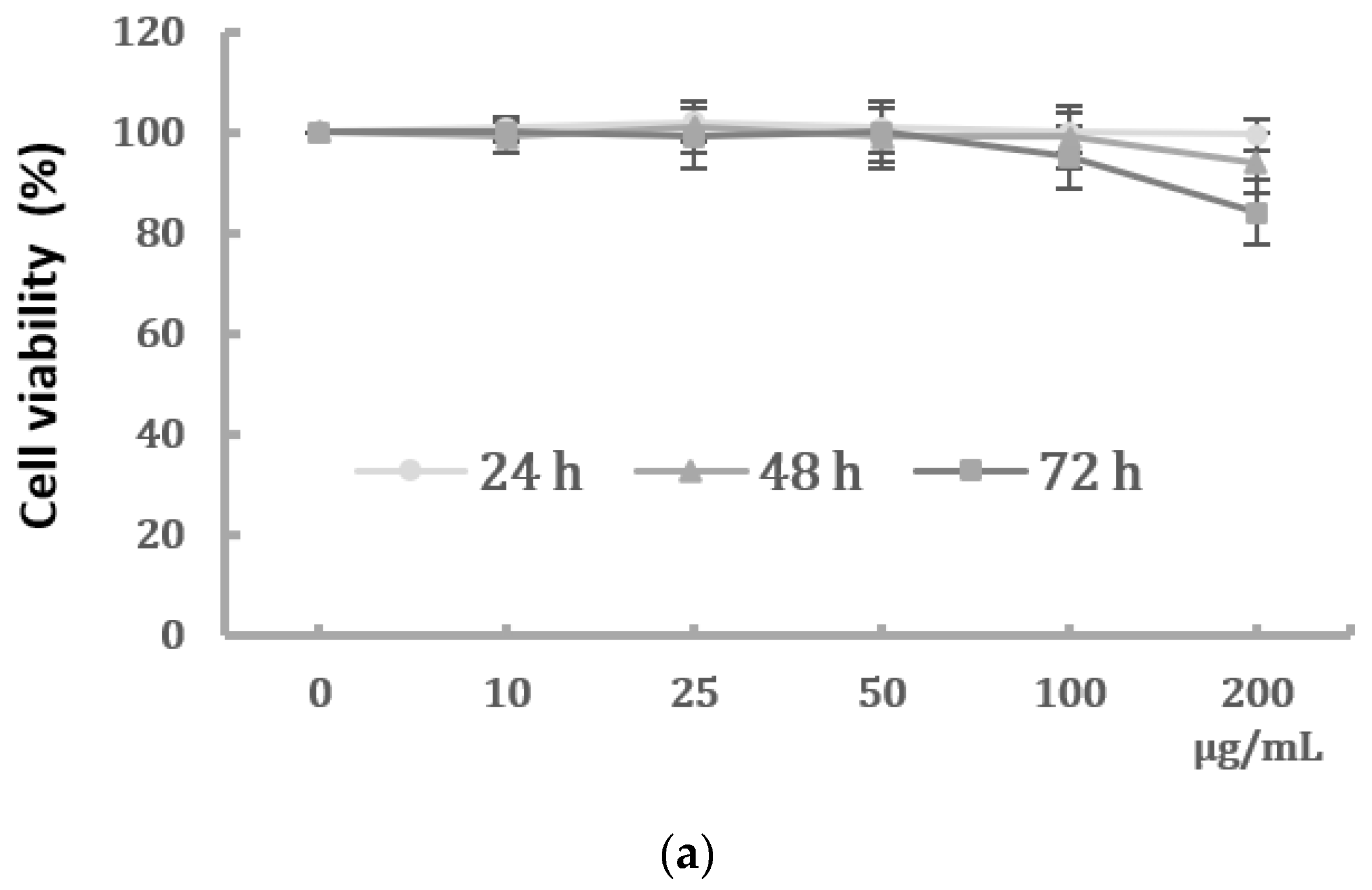
2.1. Effects of PM2.5 on Cell Viability and Morphological Changes of HK-2 Cells
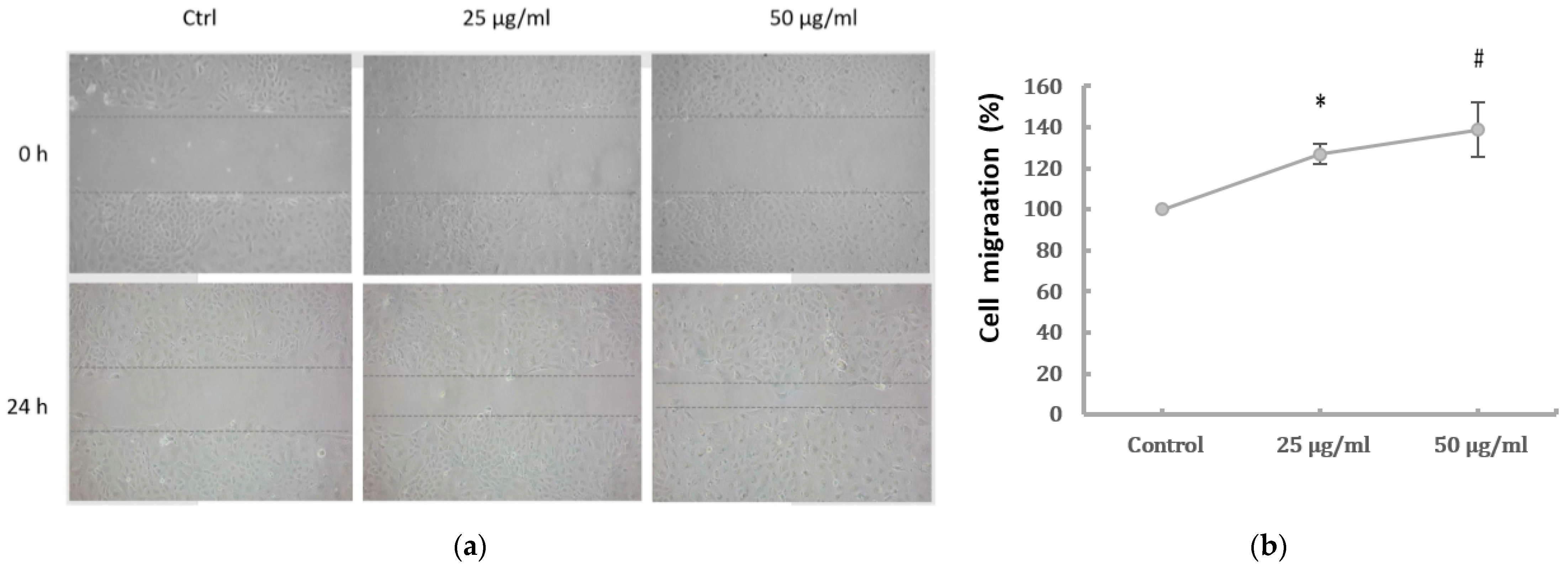
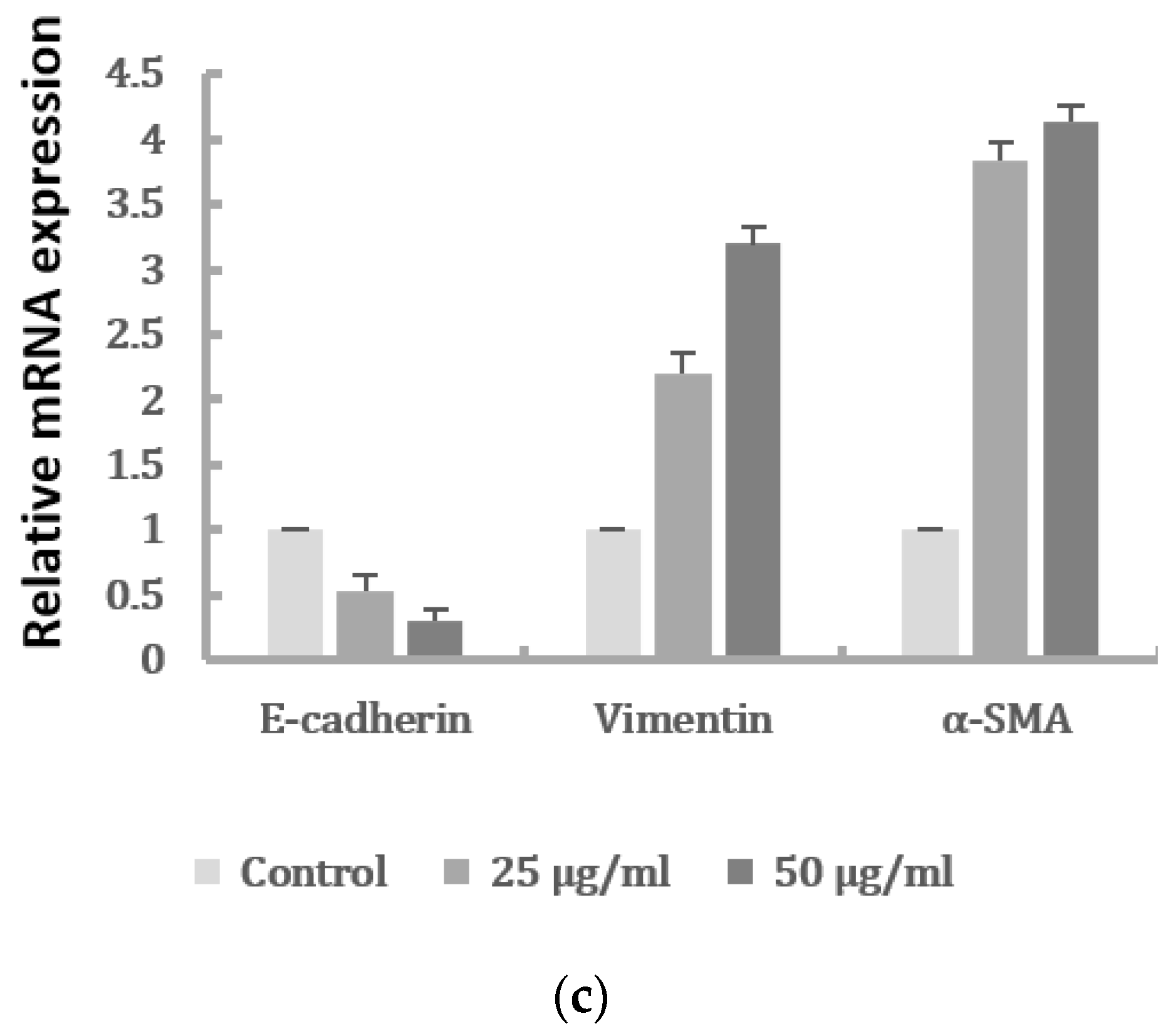
2.2. Cell Migration and Expression of EMT Markers of HK-2 Cells Induced by PM2.5
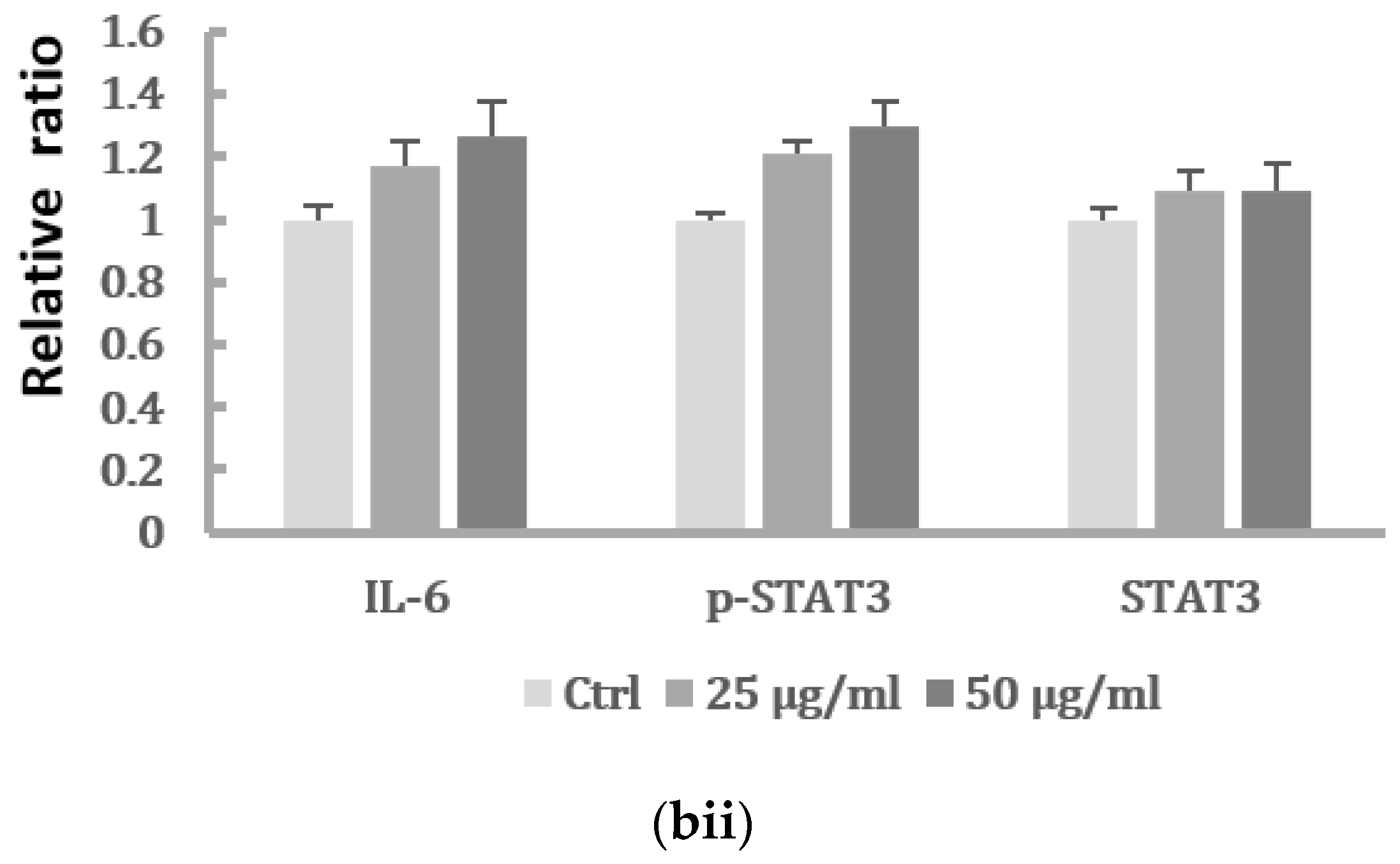
2.3. The Activation of IL-6/STAT3 Signaling Pathway in HK-2 Cells after PM2.5 Exposure
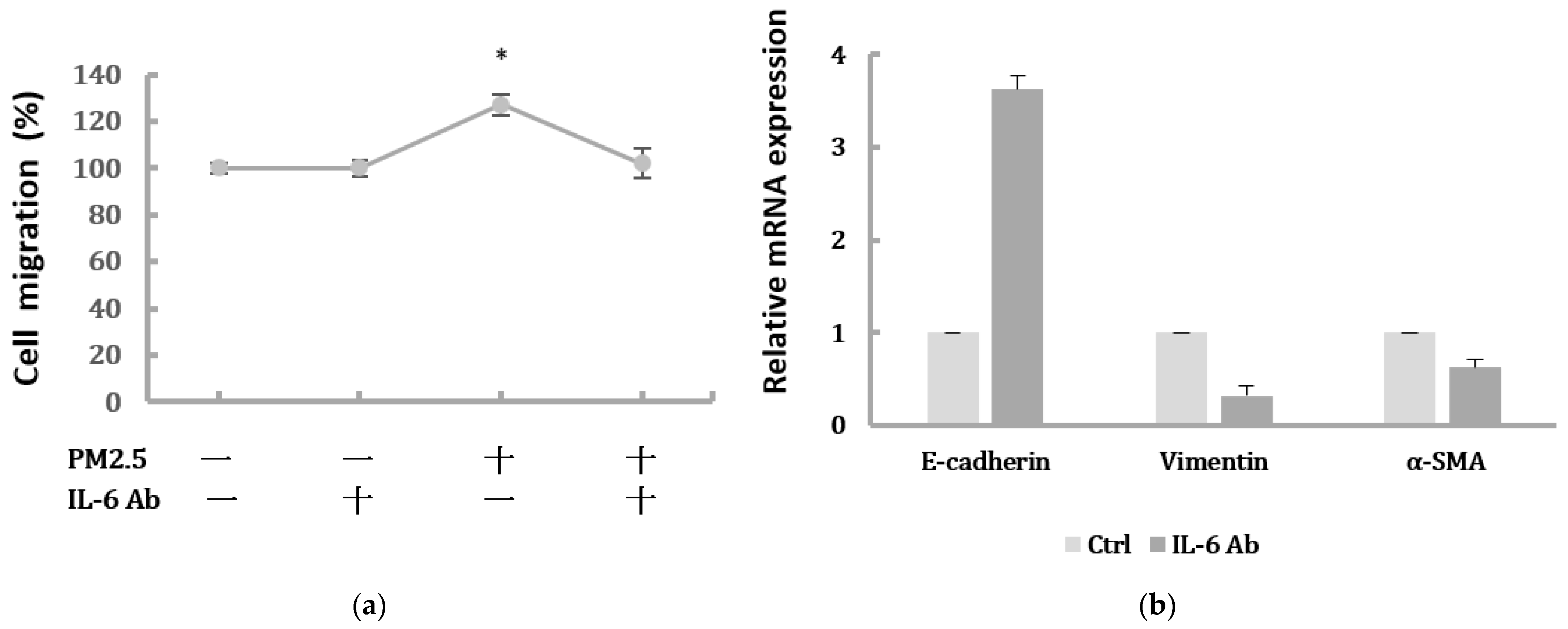
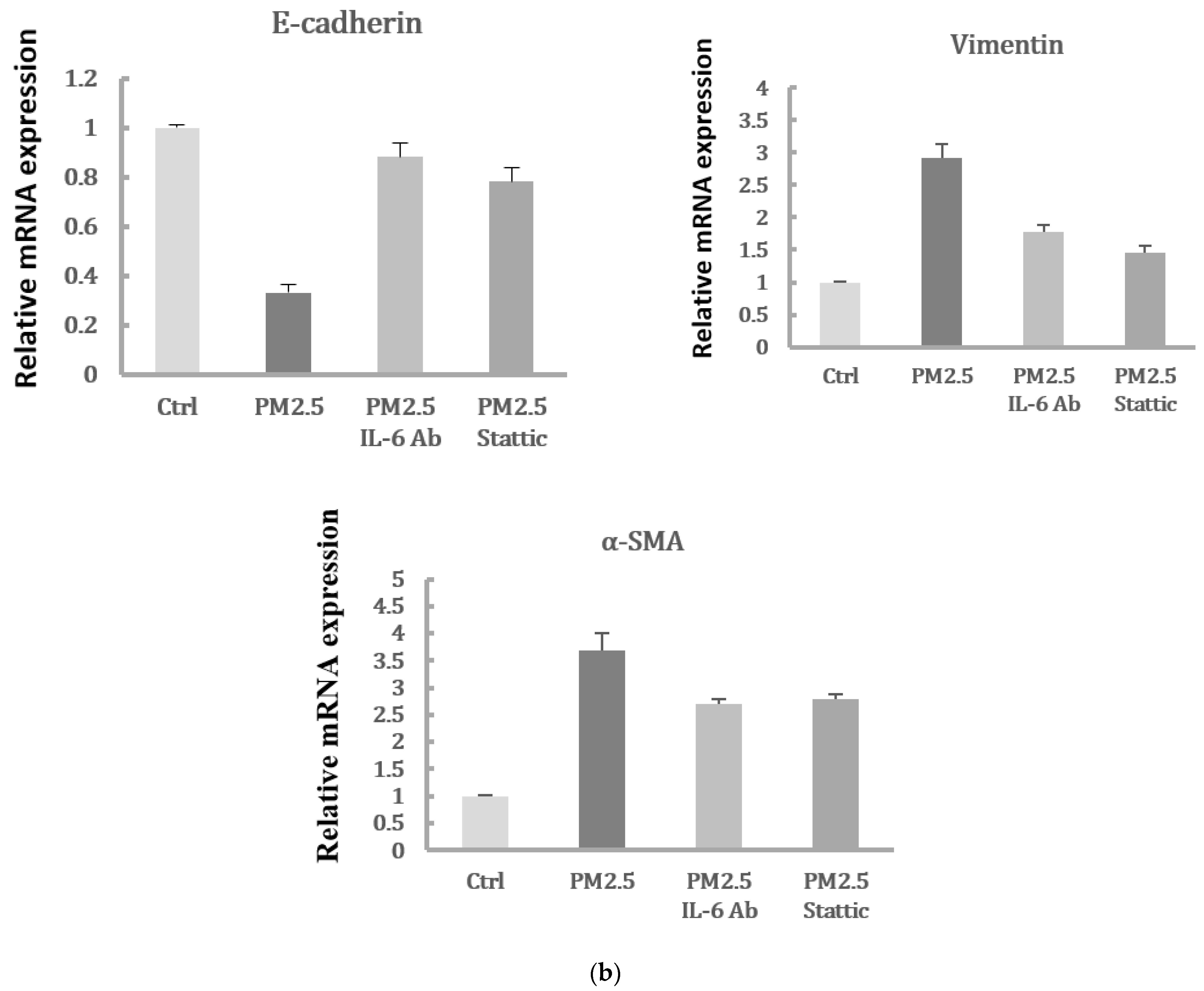
2.4. Suppression of IL-6 Inhibited EMT Features of HK-2 Cells Induced by PM2.5
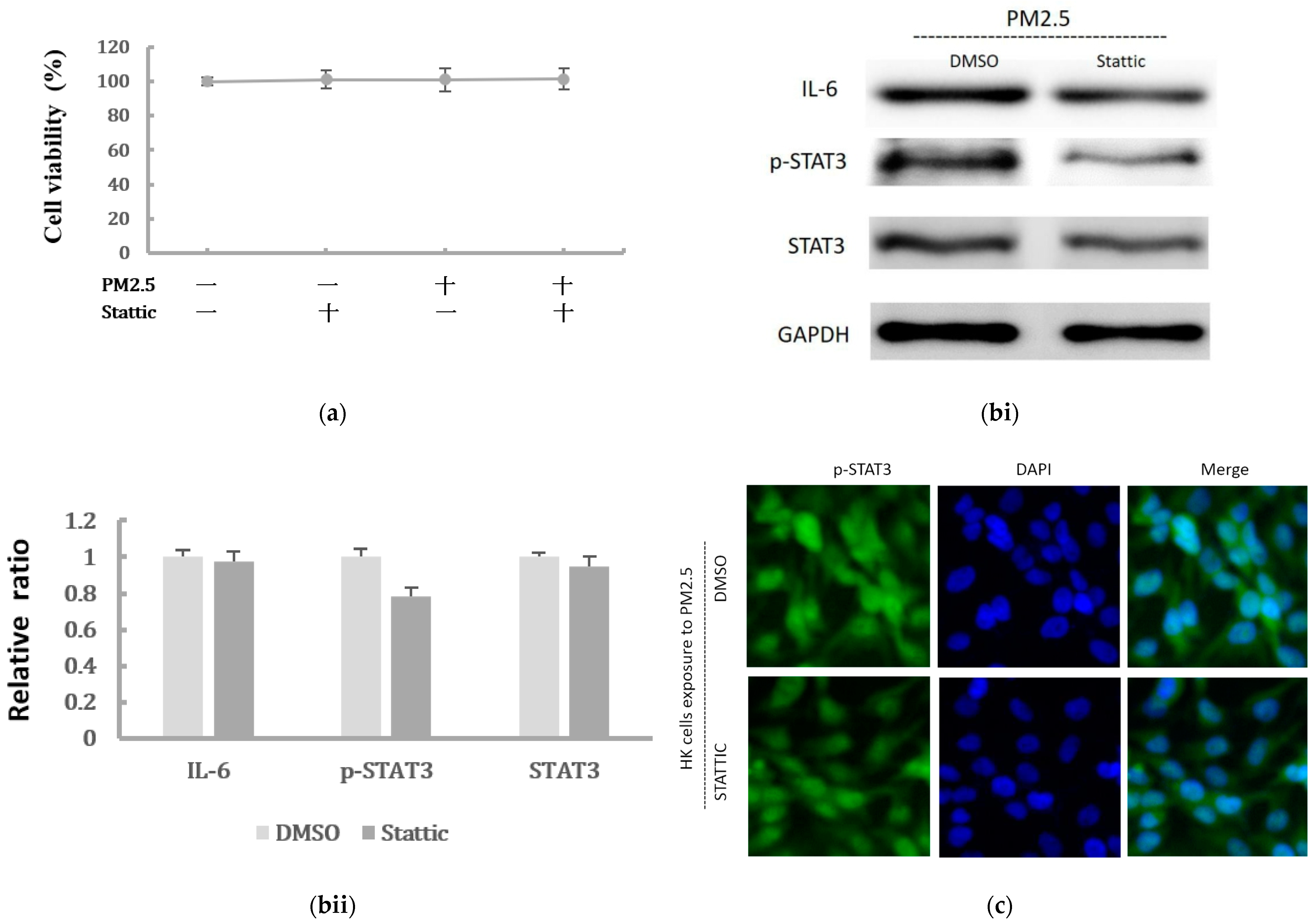
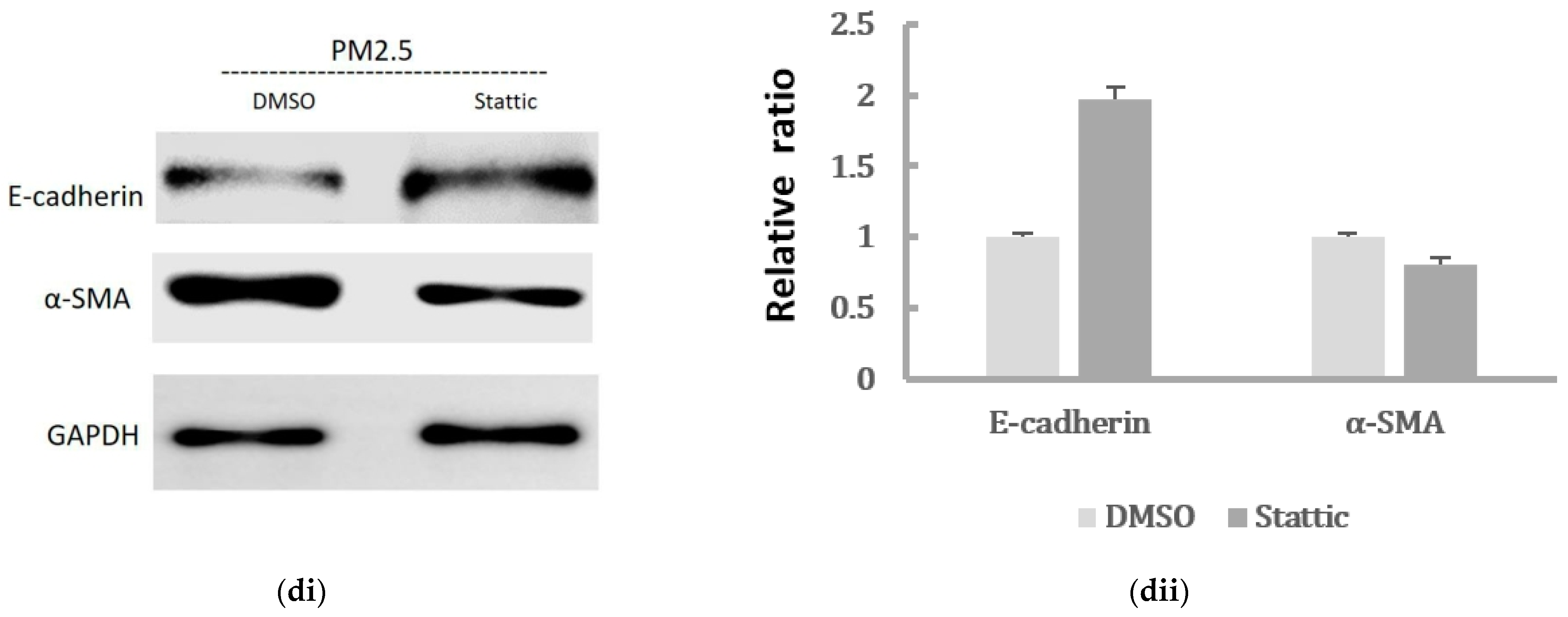
2.5. Inhibition of STAT3 Activation Decreased the EMT Expressions of HK-2 Cells Induced by PM2.5
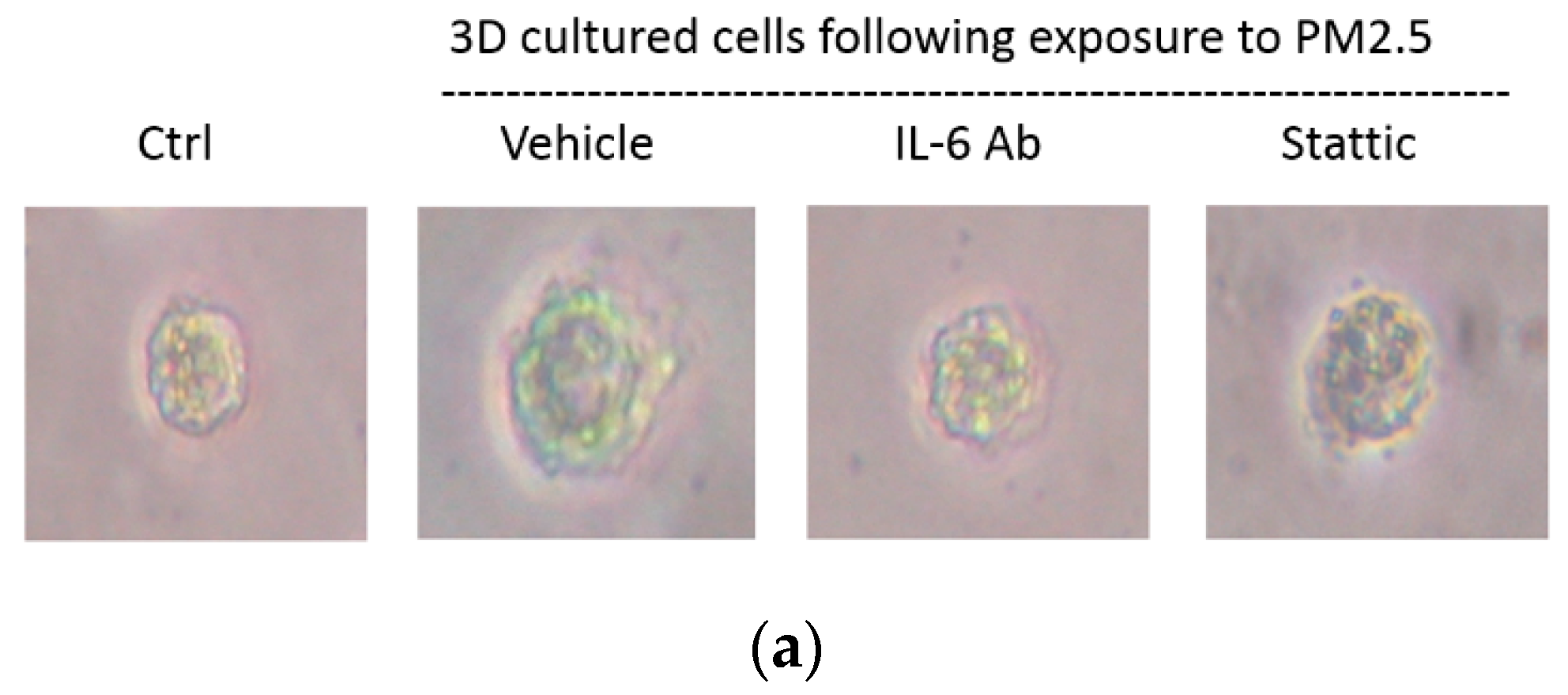
2.6. Migration of Spheroids and Expression of EMT Markers in HK-2 Cells Exposure to PM2.5 by 3D Culture Models
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Materials
4.2. Quantification of IL-6 Levels by Enzyme-Linked Immunosorbent Assay (ELISA)
4.3. Cell Viability Assay
4.4. Wound Healing and Transwell Migration Assays
4.5. RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
4.6. Western Blot Analysis
4.7. Immunofluorescence Staining
4.8. Cell Migration Assessed by 3D Cell Spheroids Culture
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Friedrich, M.J. Air Pollution Is Greatest Environmental Threat to Health. JAMA 2018, 319, 1085. [Google Scholar] [CrossRef]
- Boogaard, H.; Walker, K.; Cohen, A.J. Air pollution: The emergence of a major global health risk factor. Int. Health 2019, 11, 417–421. [Google Scholar] [CrossRef]
- Feng, S.; Gao, D.; Liao, F.; Zhou, F.; Wang, X. The health effects of ambient PM2.5 and potential mechanisms. Ecotoxicol. Environ. Saf. 2016, 128, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Guan, L.; Geng, X.; Stone, C.; Cosky, E.E.P.; Ji, Y.; Du, H.; Zhang, K.; Sun, Q.; Ding, Y. PM2.5 exposure induces systemic inflammation and oxidative stress in an intracranial atherosclerosis rat model. Environ. Toxicol. 2019, 34, 530–538. [Google Scholar] [CrossRef]
- Wyatt, L.H.; Xi, Y.; Kshirsagar, A.; Di, Q.; Ward-Caviness, C.; Wade, T.J.; Cascio, W.E.; Rappold, A.G. Association of short-term exposure to ambient PM2.5 with hospital admissions and 30-day readmissions in end-stage renal disease patients: Population-based retrospective cohort study. BMJ Open 2020, 10, e041177. [Google Scholar] [CrossRef] [PubMed]
- Lavigne, E.; Burnett, R.T.; Weichenthal, S. Association of short-term exposure to fine particulate air pollution and mortality: Effect modification by oxidant gases. Sci. Rep. 2018, 8, 16097. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, C.; Ji, G.; Liu, H.; Shao, W.; Zhang, C.; Gu, A.; Zhao, P. Effect of exposure to ambient PM2.5 pollution on the risk of respiratory tract diseases: A meta-analysis of cohort studies. J. Biomed. Res. 2017, 31, 130–142. [Google Scholar]
- Su, X.; Tian, J.; Li, B.; Zhou, L.; Kang, H.; Pei, Z.; Zhang, M.; Li, C.; Wu, M.; Wang, Q.; et al. Ambient PM2.5 caused cardiac dysfunction through FoxO1-targeted cardiac hypertrophy and macrophage-activated fibrosis in mice. Chemosphere 2020, 247, 125881. [Google Scholar] [CrossRef]
- Yitshak-Sade, M.; Nethery, R.; Schwartz, J.D.; Mealli, F.; Dominici, F.; Di, Q.; Abu Awad, Y.; Ifergane, G.; Zanobetti, A. PM2.5 and hospital admissions among Medicare enrollees with chronic debilitating brain disorders. Sci. Total Environ. 2021, 755, 142524. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Chen, G.; Huo, W.; Wang, C.; Liu, S.; Li, N.; Mao, S.; Hou, Y.; Lu, Y.; Xiang, H. Associations between long-term exposure to ambient air pollution and risk of type 2 diabetes mellitus: A systematic review and meta-analysis. Environ. Pollut. 2019, 252, 1235–1245. [Google Scholar] [CrossRef]
- Santibanez-Andrade, M.; Chirino, Y.I.; Gonzalez-Ramirez, I.; Sanchez-Perez, Y.; Garcia-Cuellar, C.M. Deciphering the Code between Air Pollution and Disease: The Effect of Particulate Matter on Cancer Hallmarks. Int. J. Mol. Sci. 2019, 21, 136. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Nie, S.; Ding, H.; Hou, F.F. Environmental pollution and kidney diseases. Nat. Rev. Nephrol. 2018, 14, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.Y.; Ju, S.W.; Lin, C.L.; Hsu, W.H.; Lin, C.C.; Ting, I.W.; Kao, C.H. Air pollutants and subsequent risk of chronic kidney disease and end-stage renal disease: A population-based cohort study. Environ. Pollut. 2020, 261, 114154. [Google Scholar] [CrossRef]
- Bowe, B.; Artimovich, E.; Xie, Y.; Yan, Y.; Cai, M.; Al-Aly, Z. The global and national burden of chronic kidney disease attributable to ambient fine particulate matter air pollution: A modelling study. BMJ Glob. Health 2020, 5, e002063. [Google Scholar] [CrossRef] [PubMed]
- Grgic, I.; Campanholle, G.; Bijol, V.; Wang, C.; Sabbisetti, V.S.; Ichimura, T.; Humphreys, B.D.; Bonventre, J.V. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney Int. 2012, 82, 172–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, B.C.; Tang, T.T.; Lv, L.L. How Tubular Epithelial Cell Injury Contributes to Renal Fibrosis. Adv. Exp. Med. Biol. 2019, 1165, 233–252. [Google Scholar] [PubMed]
- Sheng, L.; Zhuang, S. New Insights Into the Role and Mechanism of Partial Epithelial-Mesenchymal Transition in Kidney Fibrosis. Front. Physiol. 2020, 11, 569322. [Google Scholar] [CrossRef] [PubMed]
- Allison, S.J. Fibrosis: Targeting EMT to reverse renal fibrosis. Nat. Rev. Nephrol. 2015, 11, 565. [Google Scholar] [CrossRef]
- Rastaldi, M.P.; Ferrario, F.; Giardino, L.; Dell’Antonio, G.; Grillo, C.; Grillo, P.; Strutz, F.; Muller, G.A.; Colasanti, G.; D’Amico, G. Epithelial-mesenchymal transition of tubular epithelial cells in human renal biopsies. Kidney Int. 2002, 62, 137–146. [Google Scholar] [CrossRef] [Green Version]
- Dai, Y.; Zhang, W.; Wen, J.; Zhang, Y.; Kellems, R.E.; Xia, Y. A2B adenosine receptor-mediated induction of IL-6 promotes CKD. J. Am. Soc. Nephrol. 2011, 22, 890–901. [Google Scholar] [CrossRef] [Green Version]
- Amdur, R.L.; Feldman, H.I.; Gupta, J.; Yang, W.; Kanetsky, P.; Shlipak, M.; Rahman, M.; Lash, J.P.; Townsend, R.R.; Ojo, A.; et al. Inflammation and Progression of CKD: The CRIC Study. Clin. J. Am. Soc. Nephrol. 2016, 11, 1546–1556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Yan, H.; Jiang, N.; Yu, Z.; Yuan, J.; Ni, Z.; Fang, W. IL-6 trans-signaling drives a STAT3-dependent pathway that leads to structural alterations of the peritoneal membrane. Am. J. Physiol.-Renal Physiol. 2020, 318, F338–F353. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Gong, Y.; Chen, Y.; Yu, D.; Wang, X.; Zhang, X.; Dou, Y.; Liu, D.; Cheng, G.; Lu, S.; et al. IL-6 promotes epithelial-to-mesenchymal transition of human peritoneal mesothelial cells possibly through the JAK2/STAT3 signaling pathway. Am. J. Physiol.-Renal Physiol. 2017, 313, F310–F318. [Google Scholar] [CrossRef]
- Chan, T.C.; Zhang, Z.; Lin, B.C.; Lin, C.; Deng, H.B.; Chuang, Y.C.; Chan, J.W.M.; Jiang, W.K.; Tam, T.; Chang, L.Y.; et al. Long-Term Exposure to Ambient Fine Particulate Matter and Chronic Kidney Disease: A Cohort Study. Environ. Health Perspect. 2018, 126, 107002. [Google Scholar] [CrossRef] [PubMed]
- Paoin, K.; Ueda, K.; Vathesatogkit, P.; Ingviya, T.; Buya, S.; Dejchanchaiwong, R.; Phosri, A.; Seposo, X.T.; Kitiyakara, C.; Thongmung, N.; et al. Long-term air pollution exposure and decreased kidney function: A longitudinal cohort study in Bangkok Metropolitan Region, Thailand from 2002 to 2012. Chemosphere 2021, 287, 132117. [Google Scholar] [CrossRef]
- Li, G.; Huang, J.; Wang, J.; Zhao, M.; Liu, Y.; Guo, X.; Wu, S.; Zhang, L. Long-Term Exposure to Ambient PM2.5 and Increased Risk of CKD Prevalence in China. J. Am. Soc. Nephrol. 2021, 32, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Koutrakis, P.; Coull, B.; Lin, X.; Vokonas, P.; Schwartz, J.; Baccarelli, A.A. Short-term exposure to PM2.5 components and renal health: Findings from the Veterans Affairs Normative Aging Study. J. Hazard. Mater. 2021, 420, 126557. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.; Wei, H.; Guo, H.; Li, Y.; Feng, Y.; Bian, Q.; Wang, Y. LncRNA MALAT1, an lncRNA acting via the miR-204/ZEB1 pathway, mediates the EMT induced by organic extract of PM2.5 in lung bronchial epithelial cells. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2019, 317, L87–L98. [Google Scholar] [CrossRef]
- Xu, Z.; Li, Z.; Liao, Z.; Gao, S.; Hua, L.; Ye, X.; Wang, Y.; Jiang, S.; Wang, N.; Zhou, D.; et al. PM2.5 induced pulmonary fibrosis in vivo and in vitro. Ecotoxicol. Environ. Saf. 2019, 171, 112–121. [Google Scholar] [CrossRef]
- Wang, Z.; Divanyan, A.; Jourd’heuil, F.L.; Goldman, R.D.; Ridge, K.M.; Jourd’heuil, D.; Lopez-Soler, R.I. Vimentin expression is required for the development of EMT-related renal fibrosis following unilateral ureteral obstruction in mice. Am. J. Physiol.-Renal Physiol. 2018, 315, F769–F780. [Google Scholar] [CrossRef]
- Tennakoon, A.H.; Izawa, T.; Kuwamura, M.; Yamate, J. Pathogenesis of Type 2 Epithelial to Mesenchymal Transition (EMT) in Renal and Hepatic Fibrosis. J. Clin. Med. 2015, 5, 4. [Google Scholar] [CrossRef] [Green Version]
- Tan, Y.; Wang, Y.; Zou, Y.; Zhou, C.; Yi, Y.; Ling, Y.; Liao, F.; Jiang, Y.; Peng, X. LncRNA LOC101927514 regulates PM2.5-driven inflammation in human bronchial epithelial cells through binding p-STAT3 protein. Toxicol. Lett. 2020, 319, 119–128. [Google Scholar] [CrossRef]
- Jia, Y.; Li, X.; Nan, A.; Zhang, N.; Chen, L.; Zhou, H.; Zhang, H.; Qiu, M.; Zhu, J.; Ling, Y.; et al. Circular RNA 406961 interacts with ILF2 to regulate PM2.5-induced inflammatory responses in human bronchial epithelial cells via activation of STAT3/JNK pathways. Environ. Int. 2020, 141, 105755. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yuan, H.; Cao, W.; Wang, T.; Chen, W.; Yu, H.; Fu, Y.; Jiang, B.; Zhou, H.; Guo, H.; et al. Blocking interleukin-6 trans-signaling protects against renal fibrosis by suppressing STAT3 activation. Theranostics 2019, 9, 3980–3991. [Google Scholar] [CrossRef] [PubMed]
- Magno, A.L.; Herat, L.Y.; Carnagarin, R.; Schlaich, M.P.; Matthews, V.B. Current Knowledge of IL-6 Cytokine Family Members in Acute and Chronic Kidney Disease. Biomedicines 2019, 7, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Z.; Zou, C.; Yu, W.; Xu, S.; Huang, L.; Khan, Z.; Wang, J.; Liang, G.; Wang, Y. Inhibition of STAT3 activation mediated by toll-like receptor 4 attenuates angiotensin II-induced renal fibrosis and dysfunction. Br. J. Pharmacol. 2019, 176, 2627–2641. [Google Scholar] [CrossRef]
- Liu, J.; Zhong, Y.; Liu, G.; Zhang, X.; Xiao, B.; Huang, S.; Liu, H.; He, L. Role of Stat3 Signaling in Control of EMT of Tubular Epithelial Cells During Renal Fibrosis. Cell. Physiol. Biochem. 2017, 42, 2552–2558. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, C.-H.; Wan, C.; Liu, W.-S.; Wang, H.-H. PM2.5 Induces Early Epithelial Mesenchymal Transition in Human Proximal Tubular Epithelial Cells through Activation of IL-6/STAT3 Pathway. Int. J. Mol. Sci. 2021, 22, 12734. https://doi.org/10.3390/ijms222312734
Lin C-H, Wan C, Liu W-S, Wang H-H. PM2.5 Induces Early Epithelial Mesenchymal Transition in Human Proximal Tubular Epithelial Cells through Activation of IL-6/STAT3 Pathway. International Journal of Molecular Sciences. 2021; 22(23):12734. https://doi.org/10.3390/ijms222312734
Chicago/Turabian StyleLin, Chien-Hung, Chuan Wan, Wen-Sheng Liu, and Hsin-Hui Wang. 2021. "PM2.5 Induces Early Epithelial Mesenchymal Transition in Human Proximal Tubular Epithelial Cells through Activation of IL-6/STAT3 Pathway" International Journal of Molecular Sciences 22, no. 23: 12734. https://doi.org/10.3390/ijms222312734
APA StyleLin, C.-H., Wan, C., Liu, W.-S., & Wang, H.-H. (2021). PM2.5 Induces Early Epithelial Mesenchymal Transition in Human Proximal Tubular Epithelial Cells through Activation of IL-6/STAT3 Pathway. International Journal of Molecular Sciences, 22(23), 12734. https://doi.org/10.3390/ijms222312734





