Scleroderma-like Impairment in the Network of Telocytes/CD34+ Stromal Cells in the Experimental Mouse Model of Bleomycin-Induced Dermal Fibrosis
Abstract
:1. Introduction
2. Results
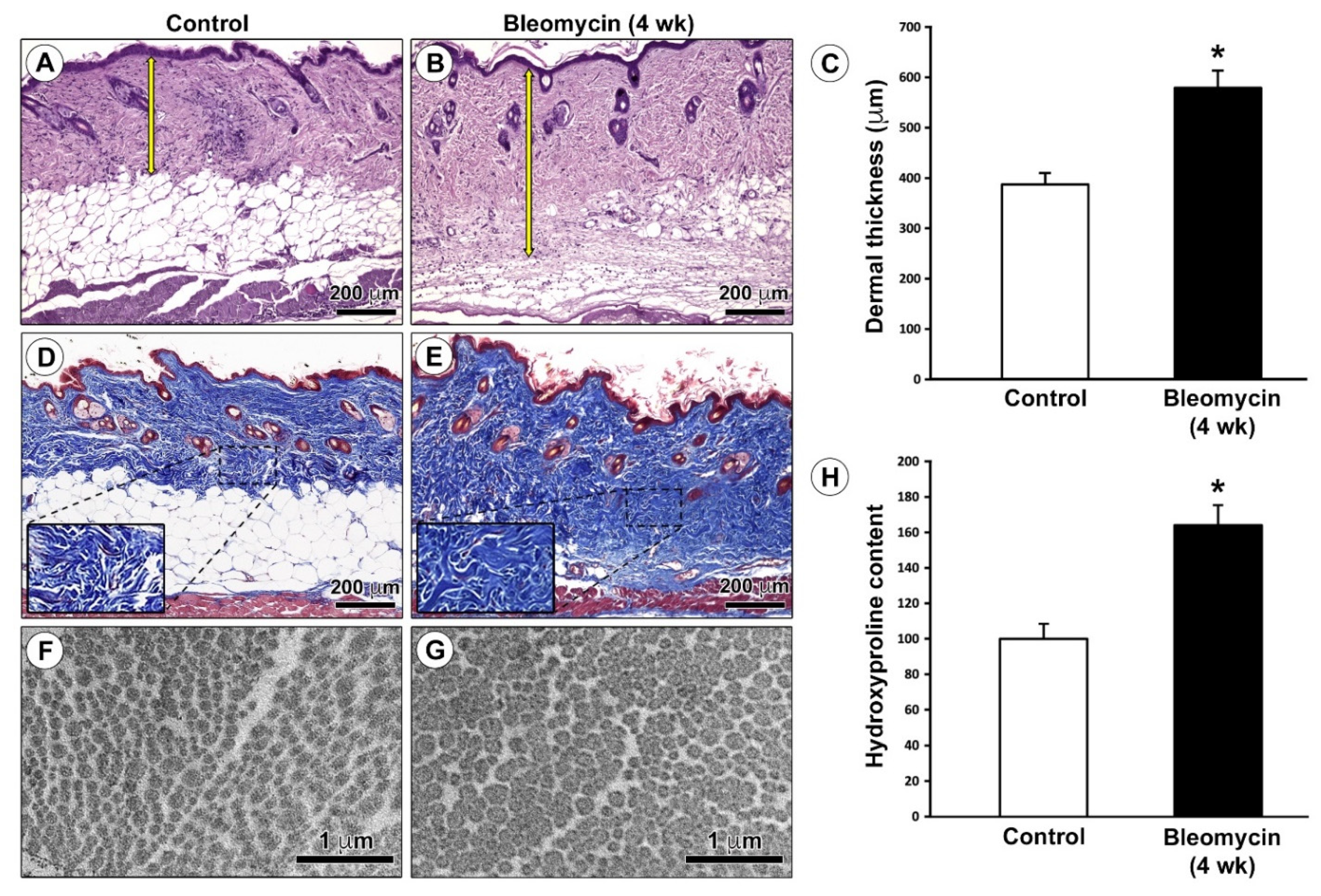
2.1. Verification of the Effective Development of the Bleomycin-Induced Dermal Fibrosis Mouse Model
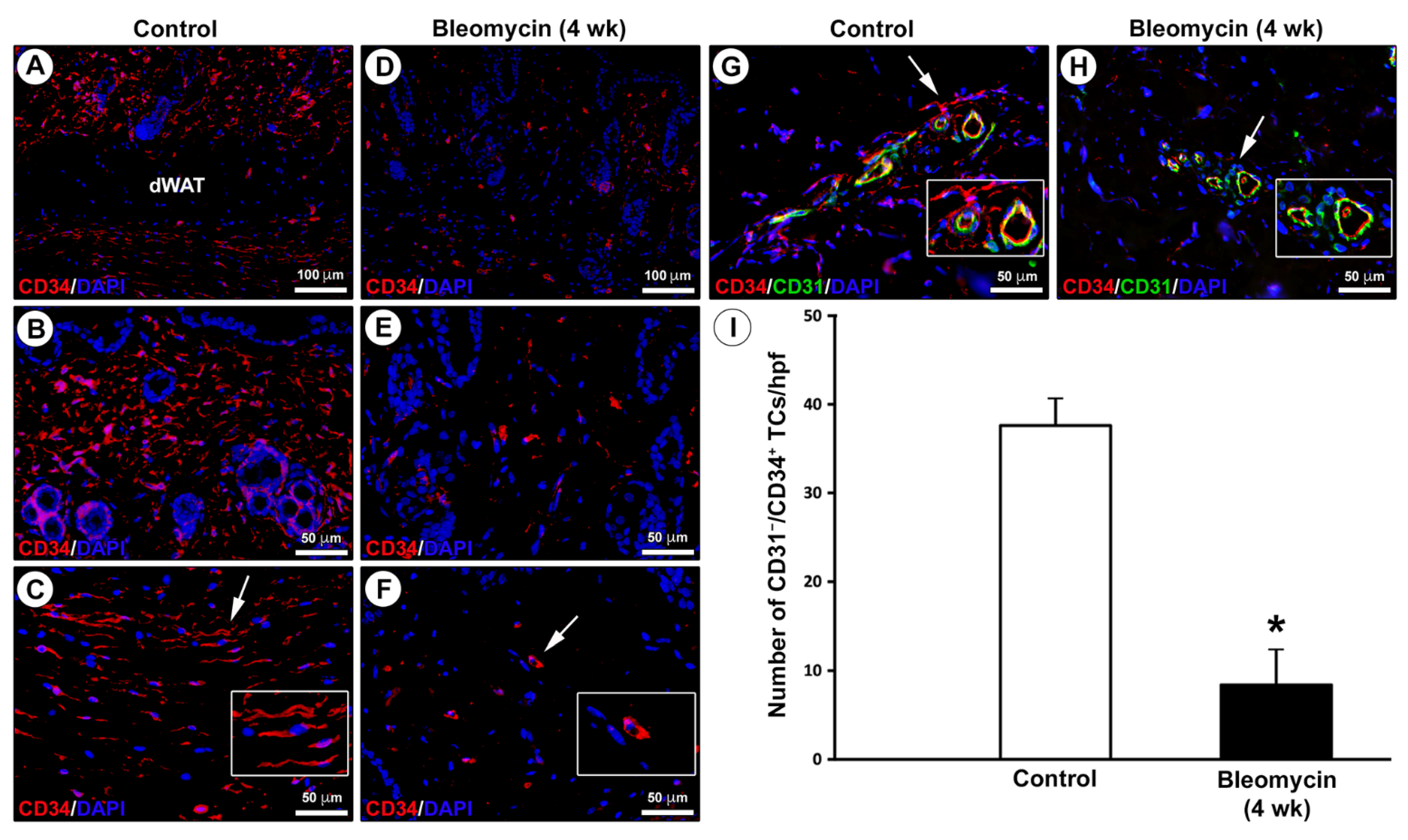
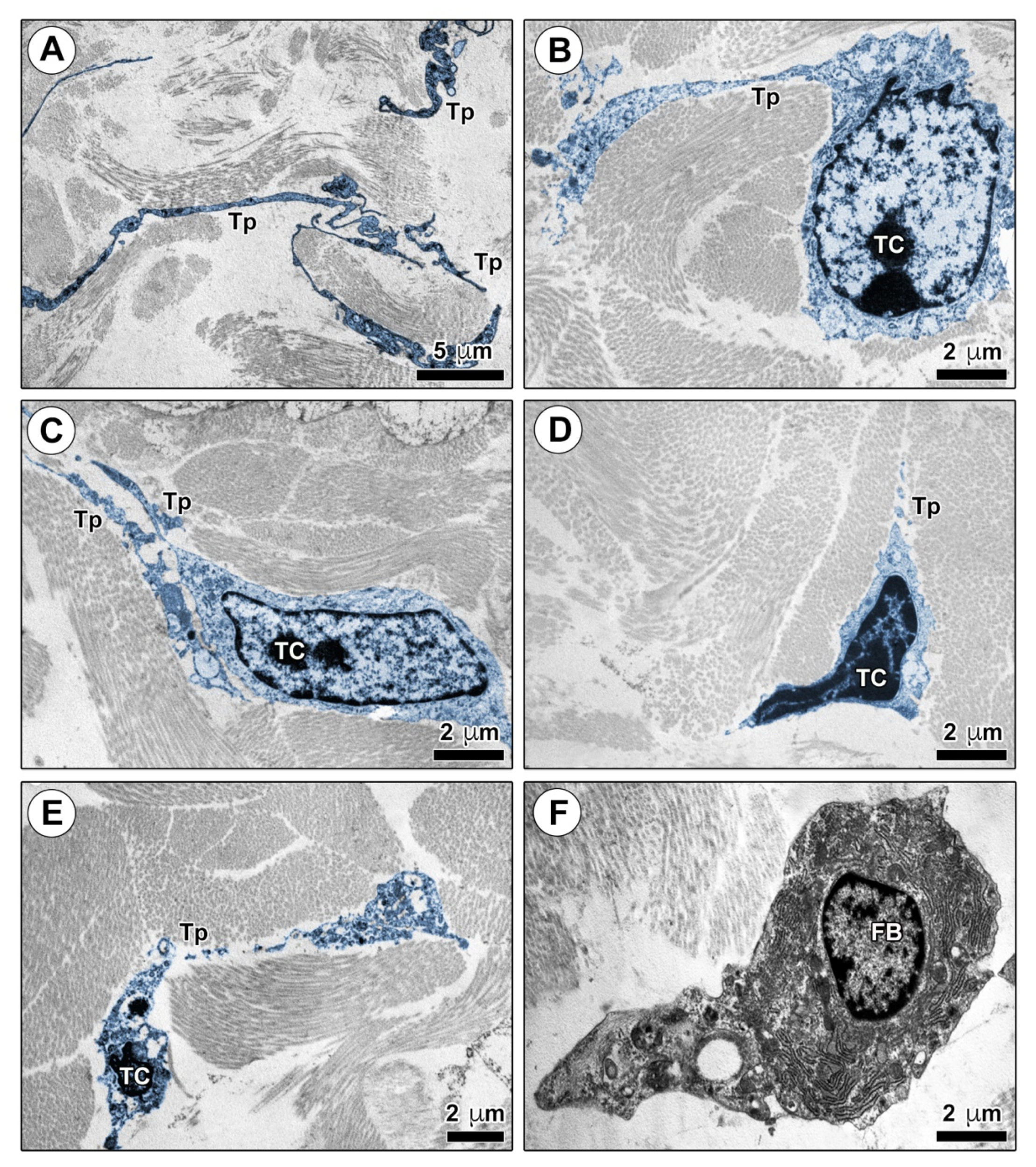
2.2. Impairment in the Network of Telocytes/CD34+ Stromal Cells in the Dermis of Bleomycin-Treated Mice
3. Discussion
4. Materials and Methods
4.1. Mouse Model of Bleomycin-Induced Dermal Fibrosis
4.2. Histochemical Analysis and Evaluation of Dermal Thickness
4.3. Hydroxyproline Assay
4.4. Transmission Electron Microscopy
4.5. Fluorescence Immunohistochemistry
4.6. Western Blotting
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Driskell, R.R.; Watt, F.M. Understanding fibroblast heterogeneity in the skin. Trends Cell. Biol. 2015, 25, 92–99. [Google Scholar] [CrossRef]
- Driskell, R.R.; Lichtenberger, B.M.; Hoste, E.; Kretzschmar, K.; Simons, B.D.; Charalambous, M.; Ferron, S.R.; Herault, Y.; Pavlovic, G.; Ferguson-Smith, A.C.; et al. Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 2013, 504, 277–281. [Google Scholar] [CrossRef] [Green Version]
- Cretoiu, S.M.; Popescu, L.M. Telocytes revisited. Biomol. Concepts 2014, 5, 353–369. [Google Scholar] [CrossRef]
- Kondo, A.; Kaestner, K.H. Emerging diverse roles of telocytes. Development 2019, 146, 175018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crețoiu, S.M. Telocytes and other interstitial cells: From structure to function. Int. J. Mol. Sci. 2021, 22, 5271. [Google Scholar] [CrossRef]
- Rosa, I.; Marini, M.; Manetti, M. Telocytes: An emerging component of stem cell niche microenvironment. J. Histochem. Cytochem. 2021, 221554211025489. [Google Scholar] [CrossRef] [PubMed]
- Cretoiu, D.; Radu, B.M.; Banciu, A.; Banciu, D.D.; Cretoiu, S.M. Telocytes heterogeneity: From cellular morphology to functional evidence. Semin. Cell Dev. Biol. 2017, 64, 26–39. [Google Scholar] [CrossRef]
- Díaz-Flores, L.; Gutiérrez, R.; García, M.P.; Sáez, F.J.; Díaz-Flores, L., Jr.; Valladares, F.; Madrid, J.F. CD34+ stromal cells/fibroblasts/fibrocytes/telocytes as a tissue reserve and a principal source of mesenchymal cells. Location, morphology, function and role in pathology. Histol. Histopathol. 2014, 29, 831–870. [Google Scholar] [CrossRef]
- Kang, Y.; Zhu, Z.; Zheng, Y.; Wan, W.; Manole, C.G.; Zhang, Q. Skin telocytes versus fibroblasts: Two distinct dermal cell populations. J. Cell. Mol. Med. 2015, 19, 2530–2539. [Google Scholar] [CrossRef] [PubMed]
- Romano, E.; Rosa, I.; Fioretto, B.S.; Lucattelli, E.; Innocenti, M.; Ibba-Manneschi, L.; Matucci-Cerinic, M.; Manetti, M. A two-step immunomagnetic microbead-based method for the isolation of human primary skin telocytes/CD34+ stromal cells. Int. J. Mol. Sci. 2020, 21, 5877. [Google Scholar] [CrossRef] [PubMed]
- Faussone Pellegrini, M.S.; Popescu, L.M. Telocytes. Biomol. Concepts 2011, 2, 481–489. [Google Scholar] [CrossRef]
- Vannucchi, M.G. The telocytes: Ten years after their introduction in the scientific literature. An update on their morphology, distribution, and potential roles in the gut. Int. J. Mol. Sci. 2020, 21, 4478. [Google Scholar] [CrossRef] [PubMed]
- Vannucchi, M.G.; Faussone-Pellegrini, M.S. The telocyte subtypes. Adv. Exp. Med. Biol. 2016, 913, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Flores, L.; Gutiérrez, R.; García, M.P.; González-Gómez, M.; Díaz-Flores, L., Jr.; Álvarez-Argüelles, H.; Luis Carrasco, J. Presence/absence and specific location of resident CD34+ stromal cells/telocytes condition stromal cell development in repair and tumors. Front. Cell Dev. Biol. 2020, 8, 544845. [Google Scholar] [CrossRef]
- Cretoiu, D.; Xu, J.; Xiao, J.; Cretoiu, S.M. Telocytes and their extracellular vesicles-Evidence and hypotheses. Int. J. Mol. Sci. 2016, 17, 1322. [Google Scholar] [CrossRef]
- Díaz-Flores, L.; Gutiérrez, R.; Díaz-Flores, L., Jr.; Goméz, M.G.; Sáez, F.J.; Madrid, J.F. Behaviour of telocytes during physiopathological activation. Semin. Cell Dev. Biol. 2016, 55, 50–61. [Google Scholar] [CrossRef]
- Bei, Y.; Wang, F.; Yang, C.; Xiao, J. Telocytes in regenerative medicine. J. Cell. Mol. Med. 2015, 19, 1441–1454. [Google Scholar] [CrossRef]
- Horch, R.E.; Weigand, A.; Beier, J.P.; Arkudas, A.; Boos, A.M. The potential role of telocytes for tissue engineering and regenerative medicine. Adv. Exp. Med. Biol. 2016, 913, 139–147. [Google Scholar] [CrossRef]
- Rusu, M.C.; Mirancea, N.; Mănoiu, V.S.; Vâlcu, M.; Nicolescu, M.I.; Păduraru, D. Skin telocytes. Ann. Anat. 2012, 194, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Cretoiu, D.; Gherghiceanu, M.; Hummel, E.; Zimmermann, H.; Simionescu, O.; Popescu, L.M. FIB-SEM tomography of human skin telocytes and their extracellular vesicles. J. Cell. Mol. Med. 2015, 19, 714–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ceafalan, L.; Gherghiceanu, M.; Popescu, L.M.; Simionescu, O. Telocytes in human skin--are they involved in skin regeneration? J. Cell. Mol. Med. 2012, 16, 1405–1420. [Google Scholar] [CrossRef]
- Arafat, E.A. Ultrastructural and immunohistochemical characteristics of telocytes in the skin and skeletal muscle of newborn rats. Acta Histochem. 2016, 118, 574–580. [Google Scholar] [CrossRef]
- Díaz-Flores, L.; Gutiérrez, R.; García, M.P.; González-Gómez, M.; Rodríguez-Rodriguez, R.; Hernández-León, N.; Díaz-Flores, L., Jr.; Carrasco, J.L. CD34+ stromal cells/telocytes in normal and pathological skin. Int. J. Mol. Sci. 2021, 22, 7342. [Google Scholar] [CrossRef]
- Yang, J.; Li, Y.; Xue, F.; Liu, W.; Zhang, S. Exosomes derived from cardiac telocytes exert positive effects on endothelial cells. Am. J. Transl. Res. 2017, 9, 5375–5387. [Google Scholar] [PubMed]
- Varga, I.; Polák, Š.; Kyselovič, J.; Kachlík, D.; Danišovič, Ľ.; Klein, M. Recently discovered interstitial cell population of telocytes: Distinguishing facts from fiction regarding their role in the pathogenesis of diverse diseases called “telocytopathies”. Medicina 2019, 55, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kostin, S. Cardiac telocytes in normal and diseased hearts. Semin. Cell Dev. Biol. 2016, 55, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Flores, L.; Gutiérrez, R.; García, M.P.; Gayoso, S.; Gutiérrez, E.; Díaz-Flores, L., Jr.; Carrasco, J.L. Telocytes in the normal and pathological peripheral nervous system. Int. J. Mol. Sci. 2020, 21, 4320. [Google Scholar] [CrossRef] [PubMed]
- Ibba-Manneschi, L.; Rosa, I.; Manetti, M. Telocytes in chronic inflammatory and fibrotic diseases. Adv. Exp. Med. Biol. 2016, 913, 51–76. [Google Scholar] [CrossRef]
- Manole, C.G.; Gherghiceanu, M.; Simionescu, O. Telocyte dynamics in psoriasis. J. Cell. Mol. Med. 2015, 19, 1504–1519. [Google Scholar] [CrossRef]
- Mirancea, N.; Moroşanu, A.M.; Mirancea, G.V.; Juravle, F.D.; Mănoiu, V.S. Infrastructure of the telocytes from tumor stroma in the skin basal and squamous cell carcinomas. Rom. J. Morphol. Embryol. 2013, 54, 1025–1037. [Google Scholar]
- Manetti, M.; Guiducci, S.; Ruffo, M.; Rosa, I.; Faussone-Pellegrini, M.S.; Matucci-Cerinic, M.; Ibba-Manneschi, L. Evidence for progressive reduction and loss of telocytes in the dermal cellular network of systemic sclerosis. J. Cell. Mol. Med. 2013, 17, 482–496. [Google Scholar] [CrossRef]
- Manetti, M.; Matucci-Cerinic, M. In search for the ideal anatomical composition of vascularized human skin equivalents for systemic sclerosis translational research: Should we recruit the telocytes? Ann. Rheum. Dis. 2021, 80, e149. [Google Scholar] [CrossRef] [Green Version]
- Allanore, Y.; Simms, R.; Distler, O.; Trojanowska, M.; Pope, J.; Denton, C.P.; Varga, J. Systemic sclerosis. Nat. Rev. Dis. Primers 2015, 1, 15002. [Google Scholar] [CrossRef]
- Varga, J.; Trojanowska, M.; Kuwana, M. Pathogenesis of systemic sclerosis: Recent insights of molecular and cellular mechanisms and therapeutic opportunities. J. Scleroderma Relat. Disord. 2017, 2, 137–152. [Google Scholar] [CrossRef]
- Asano, Y. Systemic sclerosis. J. Dermatol. 2018, 45, 128–138. [Google Scholar] [CrossRef]
- Korman, B. Evolving insights into the cellular and molecular pathogenesis of fibrosis in systemic sclerosis. Transl. Res. 2019, 209, 77–89. [Google Scholar] [CrossRef]
- Avouac, J. Mouse model of experimental dermal fibrosis: The bleomycin-induced dermal fibrosis. Methods Mol. Biol. 2014, 1142, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T. The bleomycin-induced scleroderma model: What have we learned for scleroderma pathogenesis? Arch. Dermatol. Res. 2006, 297, 333–344. [Google Scholar] [CrossRef]
- Rosa, I.; Taverna, C.; Novelli, L.; Marini, M.; Ibba-Manneschi, L.; Manetti, M. Telocytes constitute a widespread interstitial meshwork in the lamina propria and underlying striated muscle of human tongue. Sci. Rep. 2019, 9, 5858. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosa, I.; Faussone-Pellegrini, M.S.; Romano, E.; Ibba-Manneschi, L.; Matucci-Cerinic, M.; Manetti, M. Impairment in the telocyte/CD34+ stromal cell network in human rheumatoid arthritis synovium. J. Cell. Mol. Med. 2021, 25, 2274–2278. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Flores, L.; Gutiérrez, R.; García, M.P.; González, M.; Sáez, F.J.; Aparicio, F.; Díaz-Flores, L., Jr.; Madrid, J.F. Human resident CD34+ stromal cells/telocytes have progenitor capacity and are a source of alphaSMA+ cells during repair. Histol. Histopathol. 2015, 30, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Popescu, L.M.; Faussone-Pellegrini, M.S. TELOCYTES—A case of serendipity: The winding way from Interstitial Cells of Cajal (ICC), via Interstitial Cajal-Like Cells (ICLC) to TELOCYTES. J. Cell. Mol. Med. 2010, 14, 729–740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein, M.; Csöbönyeiová, M.; Žiaran, S.; Danišovič, Ľ.; Varga, I. Cardiac telocytes 16 years on—What have we learned so far, and how close are we to routine application of the knowledge in cardiovascular regenerative medicine? Int. J. Mol. Sci. 2021, 22, 10942. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Wang, F.; Cao, Y.; Huang, Q.; Xiao, J.; Yang, C.; Popescu, L.M. Telocytes in human liver fibrosis. J. Cell. Mol. Med. 2015, 19, 676–683. [Google Scholar] [CrossRef]
- Manetti, M.; Rosa, I.; Messerini, L.; Ibba-Manneschi, L. Telocytes are reduced during fibrotic remodelling of the colonic wall in ulcerative colitis. J. Cell. Mol. Med. 2015, 19, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Kostin, S. The failing human heart is characterized by decreased numbers of telocytes as result of apoptosis and altered extracellular matrix composition. J. Cell. Mol. Med. 2015, 19, 2597–2606. [Google Scholar] [CrossRef]
- Díaz-Flores, L.; Gutiérrez, R.; González-Gómez, M.; García, M.P.; Díaz-Flores, L., Jr.; Carrasco, J.L.; Martín-Vasallo, P. CD34+ stromal cells/telocytes as a source of cancer-associated fibroblasts (CAFs) in invasive lobular carcinoma of the breast. Int. J. Mol. Sci. 2021, 22, 3686. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Flores, L.; Gutiérrez, R.; García, M.P.; González-Gómez, M.; Carrasco, J.L.; Alvarez-Argüelles, H.; Díaz-Flores, L., Jr. Telocytes/CD34+ stromal cells in pathologically affected white adipose tissue. Int. J. Mol. Sci. 2020, 21, 9694. [Google Scholar] [CrossRef]
- Sanches, B.D.A.; Tamarindo, G.H.; Maldarine, J.D.S.; Da Silva, A.D.T.; Dos Santos, V.A.; Góes, R.M.; Taboga, S.R.; Carvalho, H.F. Telocytes of the male urogenital system: Interrelationships, possible functions, and pathological implications. Cell Biol. Int. 2021, 45, 1613–1623. [Google Scholar] [CrossRef]
- Vannucchi, M.G.; Traini, C. The telocytes/myofibroblasts 3-D network forms a stretch receptor in the human bladder mucosa. Is this structure involved in the detrusor overactive diseases? Ann. Anat. 2018, 218, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Aleksandrovych, V.; Pasternak, A.; Gil, K. Telocytes in the architecture of uterine fibroids. Folia Med. Cracov. 2019, 59, 33–44. [Google Scholar] [CrossRef]
- Marini, M.; Ibba-Manneschi, L.; Rosa, I.; Sgambati, E.; Manetti, M. Changes in the telocyte/CD34+ stromal cell and alpha-SMA+ myoid cell networks in human testicular seminoma. Acta Histochem. 2019, 121, 151442. [Google Scholar] [CrossRef]
- Manetti, M.; Rosa, I.; Messerini, L.; Guiducci, S.; Matucci-Cerinic, M.; Ibba-Manneschi, L. A loss of telocytes accompanies fibrosis of multiple organs in systemic sclerosis. J. Cell. Mol. Med. 2014, 18, 253–262. [Google Scholar] [CrossRef]
- Aleksandrovych, V.; Wrona, A.; Bereza, T.; Pityński, K.; Gil, K. Oviductal telocytes in patients with uterine myoma. Biomedicines 2021, 9, 1060. [Google Scholar] [CrossRef]
- Aleksandrovych, V.; Gil, K. Telocytes in the tumor microenvironment. Adv. Exp. Med. Biol. 2021, 1329, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Decato, B.E.; Ammar, R.; Reinke-Breen, L.; Thompson, J.R.; Azzara, A.V. Transcriptome analysis reveals key genes modulated by ALK5 inhibition in a bleomycin model of systemic sclerosis. Rheumatology 2021, 580. [Google Scholar] [CrossRef] [PubMed]
- Rius Rigau, A.; Luber, M.; Distler, J.H.W. Mouse models of skin fibrosis. Methods Mol. Biol. 2021, 2299, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Buechler, M.B.; Fu, W.; Turley, S.J. Fibroblast-macrophage reciprocal interactions in health, fibrosis, and cancer. Immunity 2021, 54, 903–915. [Google Scholar] [CrossRef]
- Manetti, M. Deciphering the alternatively activated (M2) phenotype of macrophages in scleroderma. Exp. Dermatol. 2015, 24, 576–578. [Google Scholar] [CrossRef] [Green Version]
- Jiang, X.J.; Cretoiu, D.; Shen, Z.J.; Yang, X.J. An in vitro investigation of telocytes-educated macrophages: Morphology, heterocellular junctions, apoptosis and invasion analysis. J. Transl. Med. 2018, 16, 85. [Google Scholar] [CrossRef]
- Huang, Y.L.; Zhang, F.L.; Tang, X.L.; Yang, X.J. Telocytes enhances M1 differentiation and phagocytosis while inhibits mitochondria-mediated apoptosis via activation of NF-κB in macrophages. Cell Transplant. 2021, 30, 9636897211002762. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, X.; Qian, M.; Zhang, M.; Zhang, D.; Bai, C.; Wang, Q.; Wang, X. Human lung telocytes could promote the proliferation and angiogenesis of human pulmonary microvascular endothelial cells in vitro. Mol. Cell. Ther. 2014, 2, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soliman, S.A. Telocytes are major constituents of the angiogenic apparatus. Sci. Rep. 2021, 11, 5775. [Google Scholar] [CrossRef] [PubMed]
- Asano, Y. The pathogenesis of systemic sclerosis: An understanding based on a common pathologic cascade across multiple organs and additional organ-specific pathologies. J. Clin. Med. 2020, 9, 2687. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Li, L.; Qi, G.; Hu, M.; Hu, C.; Wang, S.; Li, J.; Zhang, M.; Zhang, W.; Zeng, Y.; et al. Transplantation of telocytes attenuates unilateral ureter obstruction-induced renal fibrosis in rats. Cell. Physiol. Biochem. 2018, 46, 2056–2071. [Google Scholar] [CrossRef]
- Zhao, B.; Liao, Z.; Chen, S.; Yuan, Z.; Yilin, C.; Lee, K.K.; Qi, X.; Shen, X.; Zheng, X.; Quinn, T.; et al. Intramyocardial transplantation of cardiac telocytes decreases myocardial infarction and improves post-infarcted cardiac function in rats. J. Cell. Mol. Med. 2014, 18, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Takagawa, S.; Katayama, I.; Yamazaki, K.; Hamazaki, Y.; Shinkai, H.; Nishioka, K. Animal model of sclerotic skin. I: Local injections of bleomycin induce sclerotic skin mimicking scleroderma. J. Invest. Dermatol. 1999, 112, 456–462. [Google Scholar] [CrossRef] [Green Version]
- Woessner, J.F., Jr. The determination of hydroxyproline in tissue and protein samples containing small proportions of this imino acid. Arch. Biochem. Biophys. 1961, 93, 440–447. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosa, I.; Romano, E.; Fioretto, B.S.; Guasti, D.; Ibba-Manneschi, L.; Matucci-Cerinic, M.; Manetti, M. Scleroderma-like Impairment in the Network of Telocytes/CD34+ Stromal Cells in the Experimental Mouse Model of Bleomycin-Induced Dermal Fibrosis. Int. J. Mol. Sci. 2021, 22, 12407. https://doi.org/10.3390/ijms222212407
Rosa I, Romano E, Fioretto BS, Guasti D, Ibba-Manneschi L, Matucci-Cerinic M, Manetti M. Scleroderma-like Impairment in the Network of Telocytes/CD34+ Stromal Cells in the Experimental Mouse Model of Bleomycin-Induced Dermal Fibrosis. International Journal of Molecular Sciences. 2021; 22(22):12407. https://doi.org/10.3390/ijms222212407
Chicago/Turabian StyleRosa, Irene, Eloisa Romano, Bianca Saveria Fioretto, Daniele Guasti, Lidia Ibba-Manneschi, Marco Matucci-Cerinic, and Mirko Manetti. 2021. "Scleroderma-like Impairment in the Network of Telocytes/CD34+ Stromal Cells in the Experimental Mouse Model of Bleomycin-Induced Dermal Fibrosis" International Journal of Molecular Sciences 22, no. 22: 12407. https://doi.org/10.3390/ijms222212407









