Metal (Mo, W, Ti) Carbide Catalysts: Synthesis and Application as Alternative Catalysts for Dry Reforming of Hydrocarbons—A Review
Abstract
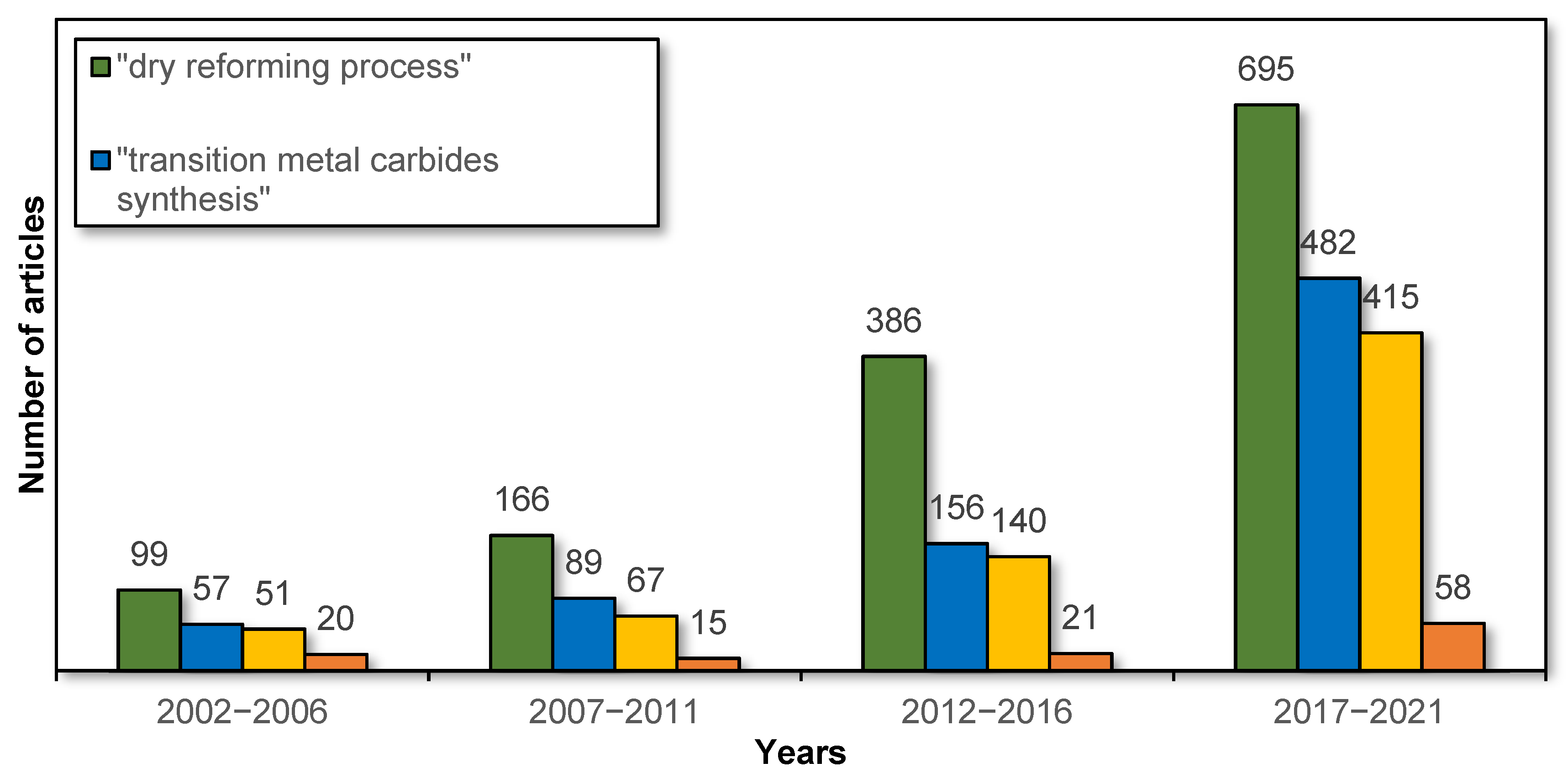
:1. Introduction
2. Metal Carbides
2.1. Tungsten Carbide
2.2. Molybdenum Carbide
2.3. Titanium Carbide
3. Synthesis of Metal Carbides
3.1. Reactive Sintering and Temperature-Programmed Reduction (TPR)
3.2. High-Energy Mechanical ball Milling Technique
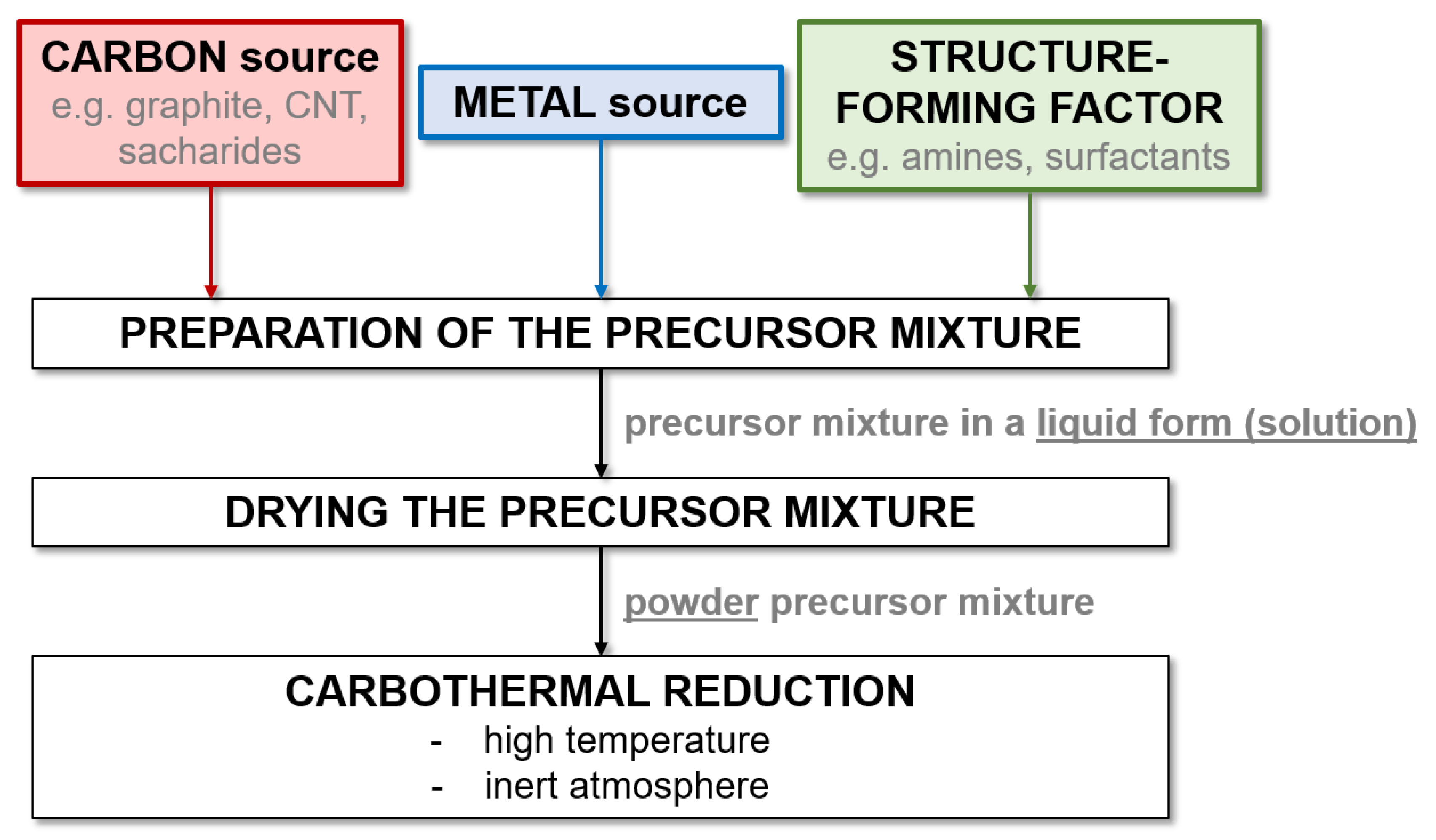
3.3. Structure-Directing Methods
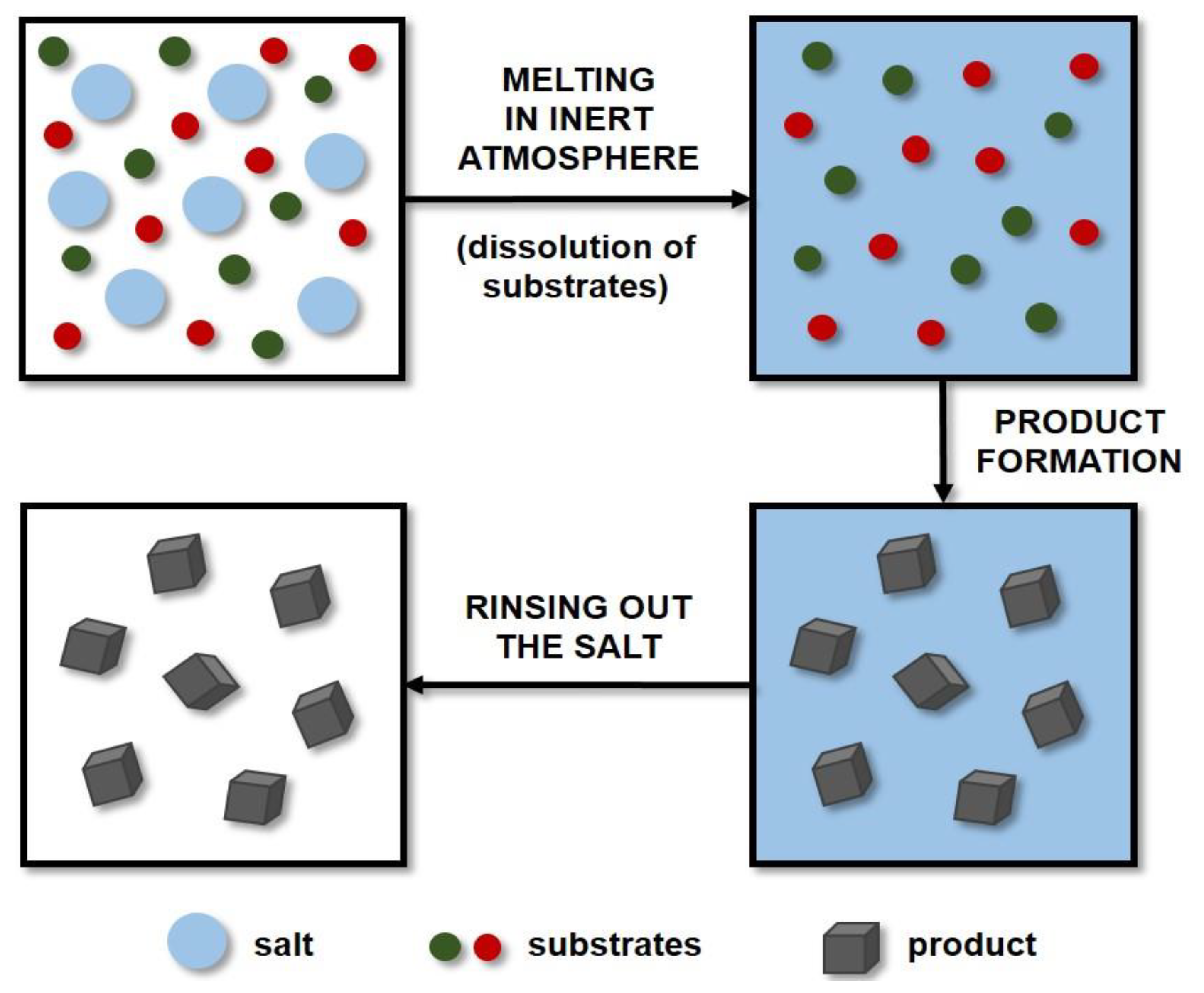
3.4. Molten Salt Synthesis
4. MAX Matrices and MXenes
4.1. Synthesis of MAX Matrices
4.2. Synthesis of MXenes
4.3. Modification of MXenes with Other Compounds
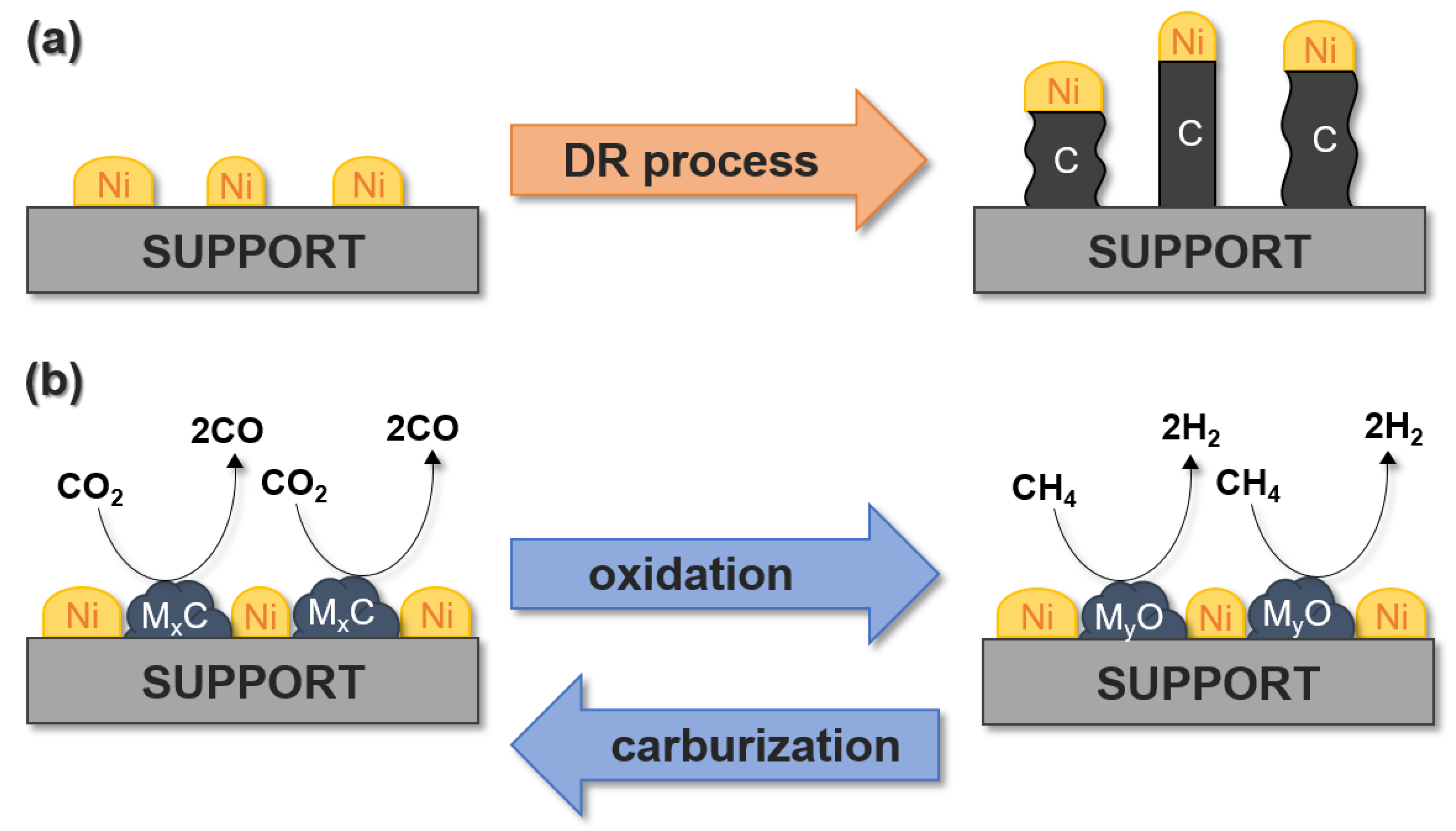
5. The Use of Metal Carbides for Dry Reforming
5.1. Tungsten Carbide
5.2. WC Combined with Nickel and Cobalt Particles
5.3. Molybdenum Carbide
5.4. Molybdenum Carbide Modified with Nickel Particles
5.5. MAX and MXenes for Dry Reforming of Hydrocarbons
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Pakhare, D.; Spivey, J. A Review of Dry (CO2) Reforming of Methane over Noble Metal Catalysts. Chem. Soc. Rev. 2014, 43, 7813–7837. [Google Scholar] [CrossRef]
- Yentekakis, I.V.; Goula, G. Biogas Management: Advanced Utilization for Production of Renewable Energy and Added-Value Chemicals. Front. Environ. Sci. 2017, 5, 7. [Google Scholar] [CrossRef] [Green Version]
- Usman, M.; Wan Daud, W.M.A.; Abbas, H.F. Dry Reforming of Methane: Influence of Process Parameters—A Review. Renew. Sustain. Energy Rev. 2015, 45, 710–744. [Google Scholar] [CrossRef] [Green Version]
- Guharoy, U.; Reina, T.R.; Liu, J.; Sun, Q.; Gu, S.; Cai, Q. A Theoretical Overview on the Prevention of Coking in Dry Reforming of Methane Using Non-Precious Transition Metal Catalysts. J. CO2 Util. 2021, 53, 101728. [Google Scholar] [CrossRef]
- Yentekakis, I.V.; Panagiotopoulou, P.; Artemakis, G. A Review of Recent Efforts to Promote Dry Reforming of Methane (DRM) to Syngas Production via Bimetallic Catalyst Formulations. Appl. Catal. B Environ. 2021, 296, 120210. [Google Scholar] [CrossRef]
- Mohamedali, M.; Henni, A.; Ibrahim, H. Recent Advances in Supported Metal Catalysts for Syngas Production from Methane. ChemEngineering 2018, 2, 9. [Google Scholar] [CrossRef] [Green Version]
- Arora, S.; Prasad, R. An Overview on Dry Reforming of Methane: Strategies to Reduce Carbonaceous Deactivation of Catalysts. RSC Adv. 2016, 6, 108668–108688. [Google Scholar] [CrossRef]
- Abdulrasheed, A.; Jalil, A.A.; Gambo, Y.; Ibrahim, M.; Hambali, H.U.; Shahul Hamid, M.Y. A Review on Catalyst Development for Dry Reforming of Methane to Syngas: Recent Advances. Renew. Sustain. Energy Rev. 2019, 108, 175–193. [Google Scholar] [CrossRef]
- Ma, Y.; Guan, G.; Hao, X.; Cao, J.; Abudula, A. Molybdenum Carbide as Alternative Catalyst for Hydrogen Production—A Review. Renew. Sustain. Energy Rev. 2017, 75, 1101–1129. [Google Scholar] [CrossRef]
- Silva, C.G.; Passos, F.B.; da Silva, V.T. Influence of the Support on the Activity of a Supported Nickel-Promoted Molybdenum Carbide Catalyst for Dry Reforming of Methane. J. Catal. 2019, 375, 507–518. [Google Scholar] [CrossRef]
- Wysocka, I.; Hupka, J.; Rogala, A. Catalytic Activity of Nickel and Ruthenium–Nickel Catalysts Supported on SiO2, ZrO2, Al2O3, and MgAl2O4 in a Dry Reforming Process. Catalysts 2019, 9, 540. [Google Scholar] [CrossRef] [Green Version]
- Wysocka, I.; Mielewczyk-Gryń, A.; Łapiński, M.; Cieślik, B.; Rogala, A. Effect of Small Quantities of Potassium Promoter and Steam on the Catalytic Properties of Nickel Catalysts in Dry/Combined Methane Reforming. Int. J. Hydrogen Energy 2021, 46, 3847–3864. [Google Scholar] [CrossRef]
- Shen, L.; Li, X.; Lindberg, D.; Taskinen, P. Tungsten Extractive Metallurgy: A Review of Processes and Their Challenges for Sustainability. Miner. Eng. 2019, 142, 105934. [Google Scholar] [CrossRef]
- Ghasali, E.; Ebadzadeh, T.; Alizadeh, M.; Razavi, M. Mechanical and Microstructural Properties of WC-Based Cermets: A Comparative Study on the Effect of Ni and Mo Binder Phases. Ceram. Int. 2018, 44, 2283–2291. [Google Scholar] [CrossRef]
- Gonzalez-Julian, J. Processing of MAX Phases: From Synthesis to Applications. J. Am. Ceram. Soc. 2021, 104, 659–690. [Google Scholar] [CrossRef]
- García, J.; Collado Ciprés, V.; Blomqvist, A.; Kaplan, B. Cemented Carbide Microstructures: A Review. Int. J. Refract. Met. Hard Mater. 2019, 80, 40–68. [Google Scholar] [CrossRef]
- Katiyar, P.K.; Singh, P.K.; Singh, R.; Kumar, A.L. Modes of Failure of Cemented Tungsten Carbide Tool Bits (WC/Co): A Study of Wear Parts. Int. J. Refract. Met. Hard Mater. 2016, 54, 27–38. [Google Scholar] [CrossRef]
- Jalaja, K.; Manwatkar, S.K.; Anand, P.; Rejith, R.; Narayana Murty, S.V.S. Metallurgical Analysis of Surface Distress on Balls during the Operation of AISI 440C Ball Bearings for Satellite Applications. Eng. Fail. Anal. 2021, 124, 105376. [Google Scholar] [CrossRef]
- Lee, J.S. Metal Carbides. Encycl. Catal. 2010, 4, 1–33. [Google Scholar] [CrossRef]
- De Paz Carmona, H.; Akhmetzyanova, U.; Tišler, Z.; Vondrová, P. Hydrotreating Atmospheric Gasoil and Co-Processing with Rapeseed Oil Using Supported Ni-Mo and Co-Mo Carbide Catalysts. Fuel 2020, 268, 117363. [Google Scholar] [CrossRef]
- Han, Y.; Gholizadeh, M.; Tran, C.C.; Kaliaguine, S.; Li, C.Z.; Olarte, M.; Garcia-Perez, M. Hydrotreatment of Pyrolysis Bio-Oil: A Review. Fuel Process. Technol. 2019, 195, 106140. [Google Scholar] [CrossRef]
- Lamic, A.F.; Pham, T.L.H.; Potvin, C.; Manoli, J.M.; Djéga-Mariadassou, G. Kinetics of Bifunctional Isomerization over Carbides (Mo, W). J. Mol. Catal. A Chem. 2005, 237, 109–114. [Google Scholar] [CrossRef]
- Morales-Garciá, Á.; Calle-Vallejo, F.; Illas, F. MXenes: New Horizons in Catalysis. ACS Catal. 2020, 10, 13487–13503. [Google Scholar] [CrossRef]
- Lalsare, A.D.; Leonard, B.; Robinson, B.; Sivri, A.C.; Vukmanovich, R.; Dumitrescu, C.; Rogers, W.; Hu, J. Self-Regenerable Carbon Nanofiber Supported Fe–Mo2C Catalyst for CH4-CO2 Assisted Reforming of Biomass to Hydrogen Rich Syngas. Appl. Catal. B Environ. 2021, 282, 119537. [Google Scholar] [CrossRef]
- Li, Z.; Wu, Y. 2D Early Transition Metal Carbides (MXenes) for Catalysis. Small 2019, 15, 1804736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hodala, J.L.; Kotni, S.; Ramachandrarao, B.; Chelliahn, B. Metal Carbide as a Potential Non Noble Metal Catalyst for Naphtha Reforming. Fuel 2021, 288, 119610. [Google Scholar] [CrossRef]
- Brungs, A.J.; York, A.P.E.; Green, M.L.H. Comparison of the Group V and VI Transition Metal Carbides for Methane Dry Reforming and Thermodynamic Prediction of Their Relative Stabilities. Catal. Lett. 1999, 57, 65–69. [Google Scholar] [CrossRef]
- Zhang, Q.; Pastor-Pérez, L.; Gu, S.; Reina, T.R. Transition Metal Carbides (TMCS) Catalysts for Gas Phase CO2 Upgrading Reactions: A Comprehensive Overview. Catalysts 2020, 10, 955. [Google Scholar] [CrossRef]
- Abou Hamdan, M.; Nassereddine, A.; Checa, R.; Jahjah, M.; Pinel, C.; Piccolo, L.; Perret, N. Supported Molybdenum Carbide and Nitride Catalysts for Carbon Dioxide Hydrogenation. Front. Chem. 2020, 8, 452. [Google Scholar] [CrossRef]
- Pasupulety, N.; Al-Zahrani, A.A.; Daous, M.A.; Driss, H.; Petrov, L.A. Methane Aromatization Study on M-Mo2C/HZSM-5 (M = Ce or Pd or Nb) Nano Materials. J. Mater. Res. Technol. 2021, 14, 363–373. [Google Scholar] [CrossRef]
- Khan, R.; Andreescu, S. Mxenes-Based Bioanalytical Sensors: Design, Characterization, and Applications. Sensors 2020, 20, 5434. [Google Scholar] [CrossRef]
- Wu, M.; An, Y.; Yang, R.; Tao, Z.; Xia, Q.; Hu, Q.; Li, M.; Chen, K.; Zhang, Z.; Huang, Q.; et al. V2CTx and Ti3C2Tx MXenes Nanosheets for Gas Sensing. ACS Appl. Nano Mater. 2021, 4, 6257–6268. [Google Scholar] [CrossRef]
- Yan, Z.; Cai, M.; Shen, P.K. Nanosized Tungsten Carbide Synthesized by a Novel Route at Low Temperature for High Performance Electrocatalysis. Sci. Rep. 2013, 3, 1646. [Google Scholar] [CrossRef] [Green Version]
- Levy, R.B.; Boudart, M. Platinum-like Behavior of Tungsten Carbide in Surface Catalysis. Science 1973, 181, 547–549. [Google Scholar] [CrossRef]
- Jewkes, J.; Sawers, D.; Stillerman, R. The Sources of Invention. In Tungsten Carbides; Palgrave Macmillan: London, UK, 1969; pp. 319–320. [Google Scholar] [CrossRef]
- Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Tungsten-carbide#section=Boiling-Point (accessed on 5 July 2021).
- Santos, J.B.O.; Valeņca, G.P.; Rodrigues, J.A.J. Catalytic Decomposition of Hydrazine on Tungsten Carbide: The Influence of Adsorbed Oxygen. J. Catal. 2002, 210, 1–6. [Google Scholar] [CrossRef]
- Moreno-Castilla, C.; Alvarez-Merino, M.A.; Carrasco-Marín, F.; Fierro, J.L.G. Tungsten and Tungsten Carbide Supported on Activated Carbon: Surface Structures and Performance for Ethylene Hydrogenation. Langmuir 2001, 17, 1752–1756. [Google Scholar] [CrossRef]
- Iyer, M.V.; Norcio, L.P.; Kugler, E.L.; Dadyburjor, D.B. Kinetic Modeling for Methane Reforming with Carbon Dioxide over a Mixed-Metal Carbide Catalyst. Ind. Eng. Chem. Res. 2003, 42, 2712–2721. [Google Scholar] [CrossRef]
- Ji, N.; Zhang, T.; Zheng, M.; Wang, A.; Wang, H.; Wang, X.; Chen, J.G. Direct Catalytic Conversion of Cellulose into Ethylene Glycol Using Nickel-Promoted Tungsten Carbide Catalysts. Angew. Chemie 2008, 120, 8638–8641. [Google Scholar] [CrossRef]
- Hwu, H.H.; Chen, J.G. Potential Application of Tungsten Carbides as Electrocatalysts: 4. Reactions of Methanol, Water, and Carbon Monoxide over Carbide-Modified W(110). J. Phys. Chem. B 2003, 107, 2029–2039. [Google Scholar] [CrossRef]
- Hsu, I.J.; Kimmel, Y.C.; Dai, Y.; Chen, S.; Chen, J.G. Rotating Disk Electrode Measurements of Activity and Stability of Monolayer Pt on Tungsten Carbide Disks for Oxygen Reduction Reaction. J. Power Sources 2012, 199, 46–52. [Google Scholar] [CrossRef]
- Yan, Z.; Meng, H.; Shen, P.K.; Wang, R.; Wang, L.; Shi, K.; Fu, H. A Facile Route to Carbide-Based Electrocatalytic Nanocomposites. J. Mater. Chem. 2012, 22, 5072–5079. [Google Scholar] [CrossRef]
- MacKenzie, K.J.D.; Meinhold, R.H.; McGavin, D.G.; Ripmeester, J.A.; Moudrakovski, I. Titanium Carbide, Nitride and Carbonitrides: A 13C, 14N, 15N and 47,49Ti Solid-State Nuclear Magnetic Resonance Study. Solid State Nucl. Magn. Reson. 1995, 4, 193–201. [Google Scholar] [CrossRef]
- Hart, G.L.W.; Klein, B.M.; Begay, S. Vacancy Ordering and Non-Stoichiometry in TiC1−x□x and TiN1−x□x. In Complex Inorganic Solids; Springer: Boston, MA, USA, 2005; pp. 99–100. [Google Scholar]
- Rasaki, S.A.; Zhang, B.; Anbalgam, K.; Thomas, T.; Yang, M. Synthesis and Application of Nano-Structured Metal Nitrides and Carbides: A Review. Prog. Solid State Chem. 2018, 50, 1–15. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, A.; Li, X.; Zhang, S.; Zhu, A.; Ma, Y.; Au, C. Ni-Modified Mo2C Catalysts for Methane Dry Reforming. Appl. Catal. A Gen. 2012, 431–432, 164–170. [Google Scholar] [CrossRef]
- Alaba, P.A.; Abbas, A.; Huang, J.; Daud, W.M.A.W. Molybdenum Carbide Nanoparticle: Understanding the Surface Properties and Reaction Mechanism for Energy Production towards a Sustainable Future. Renew. Sustain. Energy Rev. 2018, 91, 287–300. [Google Scholar] [CrossRef]
- Vasilevich, A.V.; Baklanova, O.N.; Lavrenov, A.V. Molybdenum Carbides: Synthesis and Application in Catalysis. Solid Fuel Chem. 2020, 54, 354–361. [Google Scholar] [CrossRef]
- Dridi, Z.; Bouhafs, B.; Ruterana, P.; Aourag, H. First-Principles Calculations of Vacancy Effects on Structural and Electronic Properties of TiCx and TiNx. J. Phys. Condens. Matter 2002, 14, 10237–10249. [Google Scholar] [CrossRef]
- Lohse, B.H.; Calka, A.; Wexler, D. Effect of Starting Composition on the Synthesis of Nanocrystalline TiC during Milling of Titanium and Carbon. J. Alloys Compd. 2005, 394, 148–151. [Google Scholar] [CrossRef]
- Prokudina, V.K. Titanium Carbide. In The Concise Encyclopedia of Self-Propagating High-Temperature Synthesis: History, Theory, Technology, and Products; Elsevier: Amsterdam, The Netherlands, 2017; pp. 394–397. [Google Scholar]
- Vidal, A.B.; Feria, L.; Evans, J.; Takahashi, Y.; Liu, P.; Nakamura, K.; Illas, F.; Rodriguez, J.A. CO2 Activation and Methanol Synthesis on Novel Au/TiC and Cu/TiC Catalysts. J. Phys. Chem. Lett. 2012, 3, 2275–2280. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Evans, J.; Feria, L.; Vidal, A.B.; Liu, P.; Nakamura, K.; Illas, F. CO2 Hydrogenation on Au/TiC, Cu/TiC, and Ni/TiC Catalysts: Production of CO, Methanol, and Methane. J. Catal. 2013, 307, 162–169. [Google Scholar] [CrossRef]
- Back, S.; Jung, Y. TiC- and TiN-Supported Single-Atom Catalysts for Dramatic Improvements in CO2 Electrochemical Reduction to CH4. ACS Energy Lett. 2017, 2, 969–975. [Google Scholar] [CrossRef]
- Huang, T.; Fang, H.; Mao, S.; Yu, J.; Qi, L. In-Situ Synthesized TiC@CNT as High-Performance Catalysts for Oxygen Reduction Reaction. Carbon 2018, 126, 566–573. [Google Scholar] [CrossRef]
- Yue, R.; Xia, M.; Wang, M.; Chen, P.; Gong, W.M.; Liao, S.; Li, Z.; Gao, F.; Zhang, L.; Wang, J. TiN and TiC as Stable and Promising Supports for Oxygen Reduction Reaction: Theoretical and Experimental Study. Appl. Surf. Sci. 2019, 495, 143620. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Ramírez, P.J.; Gutierrez, R.A. Highly Active Pt/MoC and Pt/TiC Catalysts for the Low-Temperature Water-Gas Shift Reaction: Effects of the Carbide Metal/Carbon Ratio on the Catalyst Performance. Catal. Today 2017, 289, 47–52. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, X.; Cheng, C.; Yang, Z. TiC Supported Single-Atom Platinum Catalyst for CO Oxidation: A Density Functional Theory Study. Appl. Surf. Sci. 2018, 453, 159–165. [Google Scholar] [CrossRef]
- Sahoo, S.K.; Ye, Y.; Lee, S.; Park, J.; Lee, H.; Lee, J.; Han, J.W. Rational Design of TiC-Supported Single-Atom Electrocatalysts for Hydrogen Evolution and Selective Oxygen Reduction Reactions. ACS Energy Lett. 2019, 4, 126–132. [Google Scholar] [CrossRef]
- Jin, Y.; Liu, D.; Li, X.; Yang, R. Synthesis of WC Nanopowders from Novel Precursors. Int. J. Refract. Met. Hard Mater. 2011, 29, 372–375. [Google Scholar] [CrossRef]
- Bolokang, S.; Banganayi, C.; Phasha, M. Effect of C and Milling Parameters on the Synthesis of WC Powders by Mechanical Alloying. Int. J. Refract. Met. Hard Mater. 2010, 28, 211–216. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, K.; Pandey, O.P. Reduction of WO3 to Nano-WC by Thermo-Chemical Reaction Route. Phys. E Low-Dimens. Syst. Nanostructures 2009, 41, 677–684. [Google Scholar] [CrossRef]
- Wanner, S.; Hilaire, L.; Wehrer, P.; Hindermann, J.P.; Maire, G. Obtaining Tungsten Carbides from Tungsten Bipyridine Complexes via Low Temperature Thermal Treatment. Appl. Catal. A Gen. 2000, 203, 55–70. [Google Scholar] [CrossRef]
- Zheng, H.; Huang, J.; Wang, W.; Ma, C. Preparation of Nano-Crystalline Tungsten Carbide Thin Film Electrode and Its Electrocatalytic Activity for Hydrogen Evolution. Electrochem. Commun. 2005, 7, 1045–1049. [Google Scholar] [CrossRef]
- Nersisyan, H.H.; Won, H.I.; Won, C.W. Combustion Synthesis of WC Powder in the Presence of Alkali Salts. Mater. Lett. 2005, 59, 3950–3954. [Google Scholar] [CrossRef]
- Nartowski, A.M.; Parkin, I.P.; MacKenzie, M.; Craven, A.J.; MacLeod, I. Solid State Metathesis Routes to Transition Metal Carbides. J. Mater. Chem. 1999, 9, 1275–1281. [Google Scholar] [CrossRef]
- Lin, H.; Tao, B.; Xiong, J.; Li, Q.; Li, Y. Tungsten Carbide (WC) Nanopowders Synthesized via Novel Core-Shell Structured Precursors. Ceram. Int. 2013, 39, 2877–2881. [Google Scholar] [CrossRef]
- Yao, Z.; Jiang, J.; Zhao, Y.; Luan, F.; Zhu, J.; Shi, Y.; Gao, H.; Wang, H. Insights into the Deactivation Mechanism of Metal Carbide Catalysts for Dry Reforming of Methane via Comparison of Nickel-Modified Molybdenum and Tungsten Carbides. RSC Adv. 2016, 6, 19944–19951. [Google Scholar] [CrossRef]
- Lee, J.S.; Oyama, S.T.; Boudart, M. Molybdenum Carbide Catalysts. I. Synthesis of Unsupported Powders. J. Catal. 1987, 106, 125–133. [Google Scholar] [CrossRef]
- Lee, J.S.; Yeom, M.H.; Lee, D.S. Catalysis by Molybdenum Carbide in Activation of C-C, C-O and C-H Bonds. J. Mol. Catal. 1990, 62, 45–51. [Google Scholar] [CrossRef]
- Bkour, Q.; Marin-Flores, O.G.; Norton, M.G.; Ha, S. A Highly Active and Stable Bimetallic Ni-Mo2C Catalyst for a Partial Oxidation of Jet Fuel. Appl. Catal. B Environ. 2019, 245, 613–622. [Google Scholar] [CrossRef]
- Zou, H.; Chen, S.; Huang, J.; Zhao, Z. Effect of Additives on the Properties of Nickel Molybdenum Carbides for the Tri-Reforming of Methane. Int. J. Hydrogen Energy 2016, 41, 16842–16850. [Google Scholar] [CrossRef]
- Mehdad, A.; Jentoft, R.E.; Jentoft, F.C. Passivation Agents and Conditions for Mo2C and W2C: Effect on Catalytic Activity for Toluene Hydrogenation. J. Catal. 2017, 347, 89–101. [Google Scholar] [CrossRef] [Green Version]
- Brungs, A.J.; York, A.P.E.; Claridge, J.B.; Márquez-Alvarez, C.; Green, M.L.H. Dry Reforming of Methane to Synthesis Gas over Supported Molybdenum Carbide Catalysts. Catal. Lett. 2000, 70, 117–122. [Google Scholar] [CrossRef]
- Darujati, A.R.S.; Thomson, W.J. Stability of Supported and Promoted-Molybdenum Carbide Catalysts in Dry-Methane Reforming. Appl. Catal. A Gen. 2005, 296, 139–147. [Google Scholar] [CrossRef]
- Li, R.; Shahbazi, A.; Wang, L.; Zhang, B.; Chung, C.C.; Dayton, D.; Yan, Q. Nanostructured Molybdenum Carbide on Biochar for CO2 Reforming of CH4. Fuel 2018, 225, 403–410. [Google Scholar] [CrossRef]
- Liang, P.; Gao, H.; Yao, Z.; Jia, R.; Shi, Y.; Sun, Y.; Fan, Q.; Wang, H. Simple Synthesis of Ultrasmall β-Mo2C and α-MoC1-x Nanoparticles and New Insights into Their Catalytic Mechanisms for Dry Reforming of Methane. Catal. Sci. Technol. 2017, 7, 3312–3324. [Google Scholar] [CrossRef]
- Gao, H.; Yao, Z.; Shi, Y.; Jia, R.; Liang, F.; Sun, Y.; Mao, W.; Wang, H. Simple and Large-Scale Synthesis of β-Phase Molybdenum Carbides as Highly Stable Catalysts for Dry Reforming of Methane. Inorg. Chem. Front. 2018, 5, 90–99. [Google Scholar] [CrossRef]
- Roca-Ayats, M.; Garcia, G.; Galante, J.L.; Pena, M.A.; Martinez-Huerta, M.V. TiC, TiCN, and TiN Supported Pt Electrocatalysts for CO and Methanol Oxidation in Acidic and Alkaline Media. J. Phys. Chem. C 2013, 117, 20769–20777. [Google Scholar] [CrossRef]
- Kirilin, A.V.; Hasse, B.; Tokarev, A.V.; Kustov, L.M.; Baeva, G.N.; Bragina, G.O.; Stakheev, A.Y.; Rautio, A.-R.; Salmi, T.; Etzold, B.J.M.; et al. Aqueous-Phase Reforming of Xylitol over Pt/C and Pt/TiC-CDC Catalytic Performance. Catal. Sci. Technol. 2014, 4, 387–401. [Google Scholar] [CrossRef] [Green Version]
- Rase, H.F.; Maddox, L.A. Titanium Carbide Catalysts, and the Catalyst Compositions. U.S. Patent 3,865,750, 11 February 1971. [Google Scholar]
- Xie, Z.; Deng, Y.; Yang, Y.; Su, H.; Zhou, D.; Liu, C.; Yang, W. Preparation of Nano-Sized Titanium Carbide Particles via a Vacuum Carbothermal Reduction Approach Coupled with Purification under Hydrogen/Argon Mixed Gas. RSC Adv. 2017, 7, 9037–9044. [Google Scholar] [CrossRef] [Green Version]
- Fu, Z.; Koc, R. Pressureless Sintering of Submicron Titanium Carbide Powders. Ceram. Int. 2017, 43, 17233–17237. [Google Scholar] [CrossRef]
- Cho, D.; Hoon Park, J.; Jeong, Y.; Lak Joo, Y. Synthesis of Titanium Carbide–Carbon Nanofibers via Carbothermal Reduction of Titania with Carbon. Ceram. Int. 2015, 41, 10974–10979. [Google Scholar] [CrossRef]
- Wu, K.H.; Jiang, Y.; Jiao, S.Q.; Chou, K.C.; Zhang, G.H. Preparations of Titanium Nitride, Titanium Carbonitride and Titanium Carbide via a Two-Step Carbothermic Reduction Method. J. Solid State Chem. 2019, 277, 793–803. [Google Scholar] [CrossRef]
- Sevastyanov, V.G.; Simonenko, E.P.; Ignatov, N.A.; Ezhov, Y.S.; Simonenko, N.P.; Kuznetsov, N.T. Low-Temperature Synthesis of Nanodispersed Titanium, Zirconium, and Hafnium Carbides. Russ. J. Inorg. Chem. 2011, 56, 661–672. [Google Scholar] [CrossRef]
- Kvashina, T.; Uvarov, N.; Ukhina, A. Synthesis of Titanium Carbide by Means of Pressureless Sintering. Ceramics 2020, 3, 306–311. [Google Scholar] [CrossRef]
- Niyomwas, S. Synthesis and Characterization of TiC and TiC-Al2O3 Composite from Wood Dust by Self-Propagating High Temperature Synthesis. Energy Procedia 2011, 9, 522–531. [Google Scholar] [CrossRef] [Green Version]
- Song, M.; Yang, Y.; Xiang, M.; Zhu, Q.; Zhao, H. Synthesis of Nano-Sized TiC Powders by Designing Chemical Vapor Deposition System in a Fluidized Bed Reactor. Powder Technol. 2021, 380, 256–264. [Google Scholar] [CrossRef]
- Ebrahimi-Kahrizsangi, R.; Alimardani, M.; Torabi, O. Investigation on Mechanochemical Behavior of the TiO2-Mg-C System Reactive Mixtures in the Synthesis of Titanium Carbide. Int. J. Refract. Met. Hard Mater. 2015, 52, 90–97. [Google Scholar] [CrossRef]
- Won, H.I.; Hayk, N.; Won, C.W.; Lee, H.H. Simple Synthesis of Nano-Sized Refractory Metal Carbides by Combustion Process. J. Mater. Sci. 2011, 46, 6000–6006. [Google Scholar] [CrossRef]
- Ghayour, H.; Abdellahi, M.; Bahmanpour, M. Optimization of the High Energy Ball-Milling: Modeling and Parametric Study. Powder Technol. 2016, 291, 7–13. [Google Scholar] [CrossRef]
- Abdellahi, M.; Bahmanpour, M. Rapid Synthesis of Nanopowders in High Energy Ball Milling; Optimization of Milling Parameters. Ceram. Int. 2015, 41, 1631–1639. [Google Scholar] [CrossRef]
- Kim, J.; Kang, S. WC Platelet Formation via High-Energy Ball Mill. Int. J. Refract. Met. Hard Mater. 2014, 47, 108–112. [Google Scholar] [CrossRef]
- Xia, Z.P.; Shen, Y.Q.; Shen, J.J.; Li, Z.Q. Mechanosynthesis of Molybdenum Carbides by Ball Milling at Room Temperature. J. Alloys Compd. 2008, 453, 185–190. [Google Scholar] [CrossRef]
- Ye, L.L.; Quan, M.X. Synthesis of Nanocrystalline TiC Powders by Mechanical Alloying. Nanostruct. Mater. 1995, 5, 25–31. [Google Scholar] [CrossRef]
- Razavi, M.; Rahimipour, M.R.; Rajabi-Zamani, A.H. Synthesis of Nanocrystalline TiC Powder from Impure Ti Chips via Mechanical Alloying. J. Alloys Compd. 2007, 436, 142–145. [Google Scholar] [CrossRef]
- Baklanova, O.N.; Vasilevich, A.V.; Lavrenov, A.V.; Drozdov, V.A.; Muromtsev, I.V.; Arbuzov, A.B.; Trenikhin, V.; Sigaeva, S.S.; Temerev, V.L.; Gorbunova, O.V.; et al. Molybdenum Carbide Synthesized by Mechanical Activation an Inert Medium. J. Alloys Compd. 2017, 698, 1018–1027. [Google Scholar] [CrossRef]
- Sheybani, K.; Javadpour, S. Mechano-Thermal Reduction of Molybdenite (MoS2) in the Presence of Sulfur Scavenger: New Method for Production of Molybdenum Carbide. Int. J. Refract. Met. Hard Mater. 2020, 92, 105277. [Google Scholar] [CrossRef]
- Vassilyeva, Y.Z.; Butenko, D.S.; Li, S.; Han, W.; Pak, A.Y. Synthesis of Molybdenum Carbide Catalyst by DC Arc Plasma in Ambient Air for Hydrogen Evolution. Mater. Chem. Phys. 2020, 254, 123509. [Google Scholar] [CrossRef]
- Tan, G.L.; Wu, X.J. Mechanochemical Synthesis of Nanocrystalline Tungsten Carbide Powders. Powder Metall. 1998, 41, 2–5. [Google Scholar] [CrossRef]
- El-Eskandarany, M.S.; Mahday, A.A.; Ahmed, H.A.; Amer, A.H. Synthesis and Characterizations of Ball-Milled Nanocrystalline WC and Nanocomposite WC-Co Powders and Subsequent Consolidations. J. Alloys Compd. 2000, 312, 315–325. [Google Scholar] [CrossRef]
- Wang, G.M.; Campbell, S.J.; Calka, A.; Kaczmarek, W.A. Synthesis and Structural Evolution of Tungsten Carbide Prepared by Ball Milling. J. Mater. Sci. 1997, 32, 1461–1467. [Google Scholar] [CrossRef]
- Rahaei, M.B.; Yazdani rad, R.; Kazemzadeh, A.; Ebadzadeh, T. Mechanochemical Synthesis of Nano TiC Powder by Mechanical Milling of Titanium and Graphite Powders. Powder Technol. 2012, 217, 369–376. [Google Scholar] [CrossRef]
- Sheybani, K.; Javadpour, S. Effect of Microwave Radiation and Mechanical Activation on the Carbothermic Reduction of Molybdenum Trioxide: Improving the Practical Efficiency. Int. J. Refract. Met. Hard Mater. 2020, 93, 105269. [Google Scholar] [CrossRef]
- Koc, R.; Kodambaka, S.K. ChemInform Abstract: Tungsten Carbide (WC) Synthesis from Novel Precursors. ChemInform 2010, 31. [Google Scholar] [CrossRef]
- Swift, G.A.; Koc, R. Formation of WC Powders Using Carbon. J. Mater. Sci. 2000, 35, 2109–2113. [Google Scholar] [CrossRef]
- Schärtl, W. Crosslinked Spherical Nanoparticles with Core-Shell Topology. Adv. Mater. 2000, 12, 1899–1908. [Google Scholar] [CrossRef]
- Wan, C.; Knight, N.A.; Leonard, B.M. Crystal Structure and Morphology Control of Molybdenum Carbide Nanomaterials Synthesized from an Amine-Metal Oxide Composite. Chem. Commun. 2013, 49, 10409–10411. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Zhang, C.; Xie, S.; Hua, W.; Zhang, Y.; Ren, N.; Xu, H.; Tang, Y. Synthesis of Nanoporous Molybdenum Carbide Nanowires Based on Organic - Inorganic Hybrid Nanocomposites with Sub-Nanometer Periodic Structures. Chem. Mater. 2009, 21, 5560–5562. [Google Scholar] [CrossRef]
- Jia, L.; Li, C.; Zhao, Y.; Liu, B.; Cao, S.; Mou, D.; Han, T.; Chen, G.; Lin, Y. Interfacial Engineering of Mo2C–Mo3C2 Heteronanowires for High Performance Hydrogen Evolution Reactions. Nanoscale 2019, 11, 23318–23329. [Google Scholar] [CrossRef]
- Wan, C.; Leonard, B.M. Iron-Doped Molybdenum Carbide Catalyst with High Activity and Stability for the Hydrogen Evolution Reaction. Chem. Mater. 2015, 27, 4281–4288. [Google Scholar] [CrossRef]
- Gavrilova, N.; Myachina, M.; Nazarov, V.; Skudin, V. Simple Synthesis of Molybdenum Carbides from Molybdenum Blue Nanoparticles. Nanomaterials 2021, 11, 873. [Google Scholar] [CrossRef]
- Mondal, A.; Sinha, K.; Paul, A.; Srivastava, D.N.; Panda, A.B. Large Scale Synthesis of Mo2C Nanoparticle Incorporated Carbon Nanosheet (Mo2C–C) for Enhanced Hydrogen Evolution Reaction. Int. J. Hydrogen Energy 2020, 45, 18623–18634. [Google Scholar] [CrossRef]
- Ren, P.; Zhao, Z. Unexpected Coke-Resistant Stability in Steam-CO2 Dual Reforming of Methane over the Robust Mo2C-Ni/ZrO2 Catalyst. Catal. Commun. 2019, 119, 71–75. [Google Scholar] [CrossRef]
- Guo, T.; Zhao, R.; Chen, X.; Du, Q.; Shuai, X.; Wang, Y.; Nie, X.; Du, J.; Li, J. Mo-Chelate Strategy for Synthesizing Ultrasmall Mo2C Nanoparticles Embedded in Carbon Nanosheets for Efficient Hydrogen Evolution. Int. J. Hydrogen Energy 2021, 46, 31598–31607. [Google Scholar] [CrossRef]
- Giordano, C.; Erpen, C.; Yao, W.; Antonietti, M. Synthesis of Mo and W Carbide and Nitride Nanoparticles via a Simple “Urea Glass” Route. Nano Lett. 2008, 8, 4659–4663. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Shang, R.; Zhong, X.; Xie, W.; Wang, X.; Huang, L. In-Situ Synthesis of Ni-Mo2C/Al2O3 Catalysts for Dry Reforming of Methane. Int. J. Hydrogen Energy 2016, 41, 21955–21964. [Google Scholar] [CrossRef]
- Wu, H.B.; Xia, B.Y.; Yu, L.; Yu, X.Y.; Lou, X.W. Porous Molybdenum Carbide Nano-Octahedrons Synthesized via Confined Carburization in Metal-Organic Frameworks for Efficient Hydrogen Production. Nat. Commun. 2015, 6, 6512. [Google Scholar] [CrossRef] [PubMed]
- Chi, J.Q.; Gao, W.K.; Lin, J.H.; Dong, B.; Qin, J.F.; Liu, Z.Z.; Liu, B.; Chai, Y.M.; Liu, C.G. Porous Core-Shell N-Doped Mo2C@C Nanospheres Derived from Inorganic-Organic Hybrid Precursors for Highly Efficient Hydrogen Evolution. J. Catal. 2018, 360, 9–19. [Google Scholar] [CrossRef]
- Koizumi, R.; Ozden, S.; Samanta, A.; Alves, A.P.P.; Mishra, A.; Ye, G.; Silva, G.G.; Vajtai, R.; Singh, A.K.; Tiwary, C.S.; et al. Origami-Inspired 3D Interconnected Molybdenum Carbide Nanoflakes. Adv. Mater. Interfaces 2018, 5, 3–9. [Google Scholar] [CrossRef]
- Gavrilova, N.; Dyakonov, V.; Myachina, M.; Nazarov, V.; Skudin, V. Synthesis of Mo2C by Thermal Decomposition of Molybdenum Blue Nanoparticles. Nanomaterials 2020, 10, 2053. [Google Scholar] [CrossRef]
- Gou, H.P.; Zhang, G.H.; Chou, K.C. Formation of Submicrometer Titanium Carbide from a Titanium Dioxide Encapsulated in Phenolic Resin. J. Mater. Sci. 2016, 51, 7008–7015. [Google Scholar] [CrossRef]
- Bae, S.T.; Shin, H.; Jung, H.S.; Hong, K.S. Synthesis of Titanium Carbide Nanoparticles with a High Specific Surface Area from a TiO2 Core-Sucrose Shell Precursor. J. Am. Ceram. Soc. 2009, 92, 2512–2516. [Google Scholar] [CrossRef]
- Shin, H.; Eun, J.H. Titanium Carbide Nanocrystals Synthesized from a Metatitanic Acid-Sucrose Precursor via a Carbothermal Reduction. J. Nanomater. 2015, 16, 217. [Google Scholar] [CrossRef] [Green Version]
- Park, S.K.; Shin, H. Microstructural Evolution of Metatitanic Acid with Temperature and Its Photosensitization Property. React. Kinet. Mech. Catal. 2013, 110, 237–249. [Google Scholar] [CrossRef]
- Yu, L.; Ji, W.; Zhang, S.; Song, Y.; Liu, H.; Wang, Z.; Liu, Q.; Wang, X. Design and Preparation of Continuous Titanium Carbide Fibers via Simple Precursor Route. Ceram. Int. 2020, 46, 25485–25492. [Google Scholar] [CrossRef]
- Wang, Y.; Song, S.; Shen, P.K.; Guo, C.; Li, C.M. Nanochain-Structured Mesoporous Tungsten Carbide and Its Superior Electrocatalysis. J. Mater. Chem. 2009, 19, 6149–6153. [Google Scholar] [CrossRef]
- Annalakshmi, M.; Balasubramanian, P.; Chen, S.M.; Chen, T.W.; Lin, P.H. Facile, Lowerature Synthesis of Tungsten Carbide (WC) Flakes for the Sensitive and Selective Electrocatalytic Detection of Dopamine in Biological Samples. Inorg. Chem. Front. 2019, 6, 2024–2034. [Google Scholar] [CrossRef]
- Kanerva, U.; Lagerbom, J.; Karhu, M.; Kronlöf, A.; Laitinen, T.; Turunen, E. Synthesis of Nano-WC from Water Soluble Raw Materials: Effects of Tungsten Source and Synthesis Atmosphere on Chemical and Phase Structure Evolution. Int. J. Refract. Met. Hard Mater. 2015, 50, 65–71. [Google Scholar] [CrossRef]
- Shanmugam, S.; Jacob, D.S.; Gedanken, A. Solid State Synthesis of Tungsten Carbide Nanorods and Nanoplatelets by a Single-Step Pyrolysis. J. Phys. Chem. B 2005, 109, 19056–19059. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, F.F.P.; De Oliveira, S.A.; De Souza, C.P.; Da Silva, A.G.P.; Gomes, U.U.; De Souza, J.F. Synthesis of Tungsten Carbide through Gas-Solid Reaction at Low Temperatures. Mater. Sci. Eng. A 2001, 315, 58–62. [Google Scholar] [CrossRef]
- Islam, M.; Martinez-Duarte, R. A Sustainable Approach for Tungsten Carbide Synthesis Using Renewable Biopolymers. Ceram. Int. 2017, 43, 10546–10553. [Google Scholar] [CrossRef] [Green Version]
- Keller, N.; Pietruszka, B.; Keller, V. A New One-Dimensional Tungsten Carbide Nanostructured Material. Mater. Lett. 2006, 60, 1774–1777. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, H.; Qin, M.; Li, Z.; Jia, B.; Chu, A.; Qu, X. Study on Influencing Factors and Mechanism of High-Quality Tungsten Carbide Nanopowders Synthesized via Carbothermal Reduction. J. Alloys Compd. 2021, 867, 158959. [Google Scholar] [CrossRef]
- Zhou, Y.; Niu, J.; Zhang, G.; Yu, M.; Yang, F. A Three-Dimensional Self-Standing Mo2C/Nitrogen-Doped Graphene Aerogel: Enhancement Hydrogen Production from Landfill Leachate Wastewater in MFCs-AEC Coupled System. Environ. Res. 2020, 184, 109283. [Google Scholar] [CrossRef] [PubMed]
- Tišler, Z.; Velvarská, R.; Skuhrovcová, L.; Pelíšková, L.; Akhmetzyanova, U. Key Role of Precursor Nature in Phase Composition of Supported Molybdenum Carbides and Nitrides. Materials 2019, 12, 415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Guan, G.; Phanthong, P.; Li, X.; Cao, J.; Hao, X.; Wang, Z.; Abudula, A. Steam Reforming of Methanol for Hydrogen Production over Nanostructured Wire-like Molybdenum Carbide Catalyst. Int. J. Hydrogen Energy 2014, 39, 18803–18811. [Google Scholar] [CrossRef] [Green Version]
- Kimura, T. Molten Salt Synthesis of Ceramic Powders. In Advances in Ceramics: Synthesis and Characterization, Processing and Specific Applications; Sikalidis, C., Ed.; InTech: Rijeka, Croatia, 2011; pp. 75–100. [Google Scholar]
- Li, X.; Dong, Z.; Westwood, A.; Brown, A.; Brydson, R.; Walton, A.; Yuan, G.; Cui, Z.; Cong, Y. Low-Temperature Preparation of Single Crystal Titanium Carbide Nanofibers in Molten Salts. Cryst. Growth Des. 2011, 11, 3122–3129. [Google Scholar] [CrossRef]
- Li, X.; Westwood, A.; Brown, A.; Brydson, R.; Rand, B. A Convenient, General Synthesis of Carbide Nanofibres via Templated Reactions on Carbon Nanotubes in Molten Salt Media. Carbon 2009, 47, 201–208. [Google Scholar] [CrossRef]
- Chen, X.; Li, Y.; Li, Y.; Zhu, J.; Jin, S.; Zhao, L.; Lei, Z.; Hong, X. Carbothermic Reduction Synthesis of Ti(C, N) Powder in the Presence of Molten Salt. Ceram. Int. 2008, 34, 1253–1259. [Google Scholar] [CrossRef]
- Dai, L.; Lu, Y.; Wang, X.; Zhu, J.; Li, Y.; Wang, L. Production of Nano-Sized Chromium Carbide Powders from Cr2O3/C Precursors by Direct Electrochemical Reduction in Molten Calcium Chloride. Int. J. Refract. Met. Hard Mater. 2015, 51, 153–159. [Google Scholar] [CrossRef]
- Yang, R.; Xing, T.; Xu, R.; Li, M. Molten Salt Synthesis of Tungsten Carbide Powder Using a Mechanically Activated Powder. Int. J. Refract. Met. Hard Mater. 2011, 29, 138–140. [Google Scholar] [CrossRef]
- Zhang, K.; Shi, Z.; Zhang, X.; Zhang, Z.; Ge, B.; Xia, H.; Guo, Y.; Qiao, G. Molten Salt Synthesis of Continuous Tungsten Carbide Coatings on Graphite Flakes. Ceram. Int. 2017, 43, 8089–8097. [Google Scholar] [CrossRef]
- Qiu, W.; Liu, Y.; Ye, J.; Fan, H.; Wang, G. Molten Salt Synthesis and Growth Mechanism of WC Platelet Powders. Powder Technol. 2017, 310, 228–233. [Google Scholar] [CrossRef]
- Yang, R.; Cui, L.; Zheng, Y.; Cai, X. Molten Salt Synthesis of Mo2C Powder Using a Mechanically Milled Powder. Mater. Lett. 2007, 61, 4815–4817. [Google Scholar] [CrossRef]
- Hu, Z.; Huang, J.; Luo, Y.; Liu, M.; Li, X.; Yan, M.; Ye, Z.; Chen, Z.; Feng, Z.; Huang, S. Wrinkled Ni-Doped Mo2C Coating on Carbon Fiber Paper: An Advanced Electrocatalyst Prepared by Molten-Salt Method for Hydrogen Evolution Reaction. Electrochim. Acta 2019, 319, 293–301. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, L.; Huang, J.; Feng, Z.; Xiong, Q.; Ye, Z.; Chen, Z.; Li, X.; Yu, Z. Self-Supported Nickel-Doped Molybdenum Carbide Nanoflower Clusters on Carbon Fiber Paper for an Efficient Hydrogen Evolution Reaction. Nanoscale 2021, 13, 8264–8274. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Zhu, H.; Weng, W.; Li, K.; Li, W.; Xiao, W. Electrochemical Fixation of CO2over a Mo Plate to Prepare a Mo2C Film for Electrocatalytic Hydrogen Evolution. Mater. Chem. Front. 2021, 5, 4963–4969. [Google Scholar] [CrossRef]
- Chen, Y.; Gao, B.; Wang, M.; Xiao, X.; Lv, A.; Jiao, S.; Chu, P.K. Dual-Phase MoC-Mo2C Nanosheets Prepared by Molten Salt Electrochemical Conversion of CO2 as Excellent Electrocatalysts for the Hydrogen Evolution Reaction. Nano Energy 2021, 90, 106533. [Google Scholar] [CrossRef]
- Ge, J.; Wang, S.; Zhang, F.; Zhang, L.; Jiao, H.; Zhu, H.; Jiao, S. Electrochemical Preparation of Carbon Films with a Mo2C Interlayer in LiCl-NaCl-Na2CO3 Melts. Appl. Surf. Sci. 2015, 347, 401–405. [Google Scholar] [CrossRef] [Green Version]
- Yan, M.; Xiong, Q.; Huang, J.; Hou, X.; Zhang, L.; Li, X.; Feng, Z. Molten Salt Synthesis of Titanium Carbide Using Different Carbon Sources as Templates. Ceram. Int. 2021, 47, 17589–17596. [Google Scholar] [CrossRef]
- Ding, J.; Deng, C.J.; Yuan, W.J.; Zhu, H.X.; Li, J. Preparation of Porous TiC/C Ceramics Using Wooden Template in Molten Salt Media. Adv. Appl. Ceram. 2013, 112, 131–135. [Google Scholar] [CrossRef]
- Yang, L.; Wang, Y.; Liu, R.; Liu, H.; Zhang, X.; Zeng, C.; Fu, C. In-Situ Synthesis of Nanocrystalline TiC Powders, Nanorods, and Nanosheets in Molten Salt by Disproportionation Reaction of Ti(II) Species. J. Mater. Sci. Technol. 2020, 37, 173–180. [Google Scholar] [CrossRef]
- Nadimi, H.; Soltanieh, M.; Sarpoolaky, H. The Formation Mechanism of Nanocrystalline TiC from KCl–LiCl Molten Salt Medium. Ceram. Int. 2020, 46, 18725–18733. [Google Scholar] [CrossRef]
- Li, X.; Dong, Z.; Westwood, A.; Brown, A.; Zhang, S.; Brydson, R.; Li, N.; Rand, B. Preparation of a Titanium Carbide Coating on Carbon Fibre Using a Molten Salt Method. Carbon 2008, 46, 305–309. [Google Scholar] [CrossRef]
- Jiang, R.; Pi, L.; Deng, B.; Hu, L.; Liu, X.; Cui, J.; Mao, X.; Wang, D. Electric Field-Driven Interfacial Alloying for in Situ Fabrication of Nano-Mo2C on Carbon Fabric as Cathode toward Efficient Hydrogen Generation. ACS Appl. Mater. Interfaces 2019, 11, 38606–38615. [Google Scholar] [CrossRef]
- Ni, J.; Ruan, Z.; Zhu, S.; Kan, X.; Lu, L.; Liu, Y. Sandwiched NiO/β-Mo2C/RGO as Improved Electrocatalyst for Hydrogen Evolution Reaction: Solvothermal-Assisted Self-Assembly and Catalytic Mechanism. ChemElectroChem 2019, 6, 5958–5966. [Google Scholar] [CrossRef]
- Fu, L.; Xia, W. MAX Phases as Nanolaminate Materials: Chemical Composition, Microstructure, Synthesis, Properties, and Applications. Adv. Eng. Mater. 2021, 23, 2001191. [Google Scholar] [CrossRef]
- Shekhirev, M.; Shuck, C.E.; Sarycheva, A.; Gogotsi, Y. Progress in Materials Science Characterization of MXenes at Every Step, from Their Precursors to Single Flakes and Assembled Films. Prog. Mater. Sci. 2021, 120, 100757. [Google Scholar] [CrossRef]
- Chaudhari, N.K.; Jin, H.; Kim, B.; San Baek, D.; Joo, S.H.; Lee, K. MXene: An Emerging Two-Dimensional Material for Future Energy Conversion and Storage Applications. J. Mater. Chem. A 2017, 5, 24564–24579. [Google Scholar] [CrossRef]
- Zhang, C.; Ma, Y.; Zhang, X.; Abdolhosseinzadeh, S.; Sheng, H.; Lan, W.; Pakdel, A.; Heier, J.; Nüesch, F. Two-Dimensional Transition Metal Carbides and Nitrides (MXenes): Synthesis, Properties, and Electrochemical Energy Storage Applications. Energy Environ. Mater. 2020, 3, 29–55. [Google Scholar] [CrossRef]
- Meshkian, R.; Dahlqvist, M.; Lu, J.; Wickman, B.; Halim, J.; Thörnberg, J.; Tao, Q.; Li, S.; Intikhab, S.; Snyder, J.; et al. W-Based Atomic Laminates and Their 2D Derivative W1.33C MXene with Vacancy Ordering. Adv. Mater. 2018, 30, 1706409. [Google Scholar] [CrossRef]
- Tao, Q.; Dahlqvist, M.; Lu, J.; Kota, S.; Meshkian, R.; Halim, J.; Palisaitis, J.; Hultman, L.; Barsoum, M.W.; Persson, P.O.Å.; et al. Two-Dimensional Mo1.33C MXene with Divacancy Ordering Prepared from Parent 3D Laminate with in-Plane Chemical Ordering. Nat. Commun. 2017, 8, 14949. [Google Scholar] [CrossRef]
- Grzegórska, A.; Głuchowski, P.; Karczewski, J.; Ryl, J.; Wysocka, I.; Siuzdak, K.; Trykowski, G.; Grochowska, K.; Zielińska-Jurek, A. Enhanced Photocatalytic Activity of Accordion-like Layered Ti3C2 (MXene) Coupled with Fe-Modified Decahedral Anatase Particles Exposing {1 0 1} and {0 0 1} Facets. Chem. Eng. J. 2021, 426, 130801. [Google Scholar] [CrossRef]
- Li, R.; Zhang, L.; Shi, L.; Wang, P. MXene Ti3C2: An Effective 2D Light-to-Heat Conversion Material. ACS Nano 2017, 11, 3752–3759. [Google Scholar] [CrossRef] [Green Version]
- Liang, J.; Chen, T.; Liu, J.; Zhang, Q.; Peng, W.C.; Li, Y.; Zhang, F.; Fan, X. Chemoselective Hydrodeoxygenation of Palmitic Acid to Diesel-like Hydrocarbons over Ni/MoO2@Mo2CTx Catalyst with Extraordinary Synergic Effect. Chem. Eng. J. 2020, 391, 123472. [Google Scholar] [CrossRef]
- Zhou, W.; Mei, B.; Zhu, J. Fabrication of High-Purity Ternary Carbide Ti3AlC2 by Spark Plasma Sintering (SPS) Technique. Ceram. Int. 2007, 33, 1399–1402. [Google Scholar] [CrossRef]
- Wang, P.; Mei, B.; Hong, X.; Zhou, W. Synthesis of Ti2AlC by Hot Pressing and Its Mechanical and Electrical Properties. Trans. Nonferrous Matals Soc. China 2007, 17, 1001–1004. [Google Scholar] [CrossRef]
- Anasori, B.; Dahlqvist, M.; Halim, J.; Moon, E.J.; Lu, J.; Hosler, B.C.; Caspi, E.N.; May, S.J.; Hultman, L.; Eklund, P.; et al. Experimental and Theoretical Characterization of Ordered MAX Phases Mo2TiAlC2 and Mo2Ti2AlC3. J. Appl. Phys. 2015, 118, 17431. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.; Lai, C.C.; Tao, Q.; Lu, J.; Halim, J.; Sun, L.; Zhang, J.; Yang, J.; Anasori, B.; Wang, J.; et al. Mo2Ga2C: A New Ternary Nanolaminated Carbide. Chem. Commun. 2015, 51, 6560–6563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shuck, C.E.; Ventura-Martinez, K.; Goad, A.; Uzun, S.; Shekhirev, M.; Gogotsi, Y. Safe Synthesis of MAX and MXene: Guidelines to Reduce Risk During Synthesis. ACS Chem. Health Saf. 2021, 28, 326–338. [Google Scholar] [CrossRef]
- Dash, A.; Vaßen, R.; Guillon, O.; Gonzalez-Julian, J. Molten Salt Shielded Synthesis of Oxidation Prone Materials in Air. Nat. Mater. 2019, 18, 465–470. [Google Scholar] [CrossRef]
- Deysher, G.; Shuck, C.E.; Hantanasirisakul, K.; Frey, N.C.; Foucher, A.C.; Maleski, K.; Sarycheva, A.; Shenoy, V.B.; Stach, E.A.; Anasori, B.; et al. Synthesis of Mo4VAlC4 MAX Phase and Two-Dimensional Mo4VC4 MXene with Five Atomic Layers of Transition Metals. ACS Nano 2020, 14, 204–217. [Google Scholar] [CrossRef]
- Roy, C.; Banerjee, P.; Bhattacharyya, S. Molten Salt Shielded Synthesis (MS3) of Ti2AlN and V2AlC MAX Phase Powders in Open Air. J. Eur. Ceram. Soc. 2020, 40, 923–929. [Google Scholar] [CrossRef]
- Wu, M.; Wang, B.; Hu, Q.; Wang, L.; Zhou, A. The Synthesis Process and Thermal Stability of V2C MXene. Materials 2018, 11, 2112. [Google Scholar] [CrossRef] [Green Version]
- Anasori, B.; Lukatskaya, M.R.; Gogotsi, Y. 2D Metal Carbides and Nitrides (MXenes) for Energy Storage. Nat. Rev. Mater. 2017, 2, 16098. [Google Scholar] [CrossRef]
- Zhu, W.; Panda, S.; Lu, C.; Ma, Z.; Khan, D.; Dong, J.; Sun, F.; Xu, H.; Zhang, Q.; Zou, J. Using a Self-Assembled Two-Dimensional MXene-Based Catalyst (2D-Ni@Ti3C2) to Enhance Hydrogen Storage Properties of MgH2. ACS Appl. Mater. Interfaces 2020, 12, 50333–50343. [Google Scholar] [CrossRef] [PubMed]
- Alhabeb, M.; Maleski, K.; Anasori, B.; Lelyukh, P.; Clark, L.; Sin, S.; Gogotsi, Y. Guidelines for Synthesis and Processing of Two-Dimensional Titanium Carbide (Ti3C2Tx MXene). Chem. Mater. 2017, 29, 7633–7644. [Google Scholar] [CrossRef]
- Jin, S.; Shi, Z.; Jing, H.; Wang, L.; Hu, Q.; Chen, D.; Li, N.; Zhou, A. Mo2C-MXene/CdS Heterostructures as Visible-Light Photocatalysts with an Ultrahigh Hydrogen Production Rate. ACS Appl. Energy Mater. 2021. [Google Scholar] [CrossRef]
- Bauer, E.; Rogl, G.; Chen, X.Q.; Khan, R.T.; Michor, H.; Hilscher, G.; Royanian, E.; Kumagai, K.; Li, D.Z.; Li, Y.Y.; et al. Unconventional Superconducting Phase in the Weakly Correlated Noncentrosymmetric Mo3Al2C Compound. Phys. Rev. B-Condens. Matter Mater. Phys. 2010, 82, 2–6. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Z.; Guo, C.; Wang, E.; He, Z.; Liang, T.; Yang, T.; Hou, X. The Oxidation and Thermal Stability of Two-Dimensional Transition Metal Carbides and/or Carbonitrides (MXenes) and the Improvement Based on Their Surface State. Inorg. Chem. Front. 2021, 8, 2164–2182. [Google Scholar] [CrossRef]
- Thakur, R.; Vahidmohammadi, A.; Moncada, J.; Adams, W.R.; Chi, M.; Tatarchuk, B.; Beidaghi, M.; Carrero, C.A. Insights into the Thermal and Chemical Stability of Multilayered V2CTX MXene. Nanoscale 2019, 11, 10716–10726. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.; Qiu, Z.; Wu, X.; Zhou, P.; Zhou, T.; Zhao, J.; Miao, Z.; Zhou, J.; Zhuo, S. Fe3O4@Ti3C2 MXene Hybrids with Ultrahigh Volumetric Capacity as an Anode Material for Lithium-Ion Batteries. J. Mater. Chem. A 2018, 6, 11189–11197. [Google Scholar] [CrossRef]
- He, X.; Bi, T.; Zheng, X.; Zhu, W.; Jiang, J. Nickel Cobalt Sulfide Nanoparticles Grown on Titanium Carbide MXenes for High-Performance Supercapacitor. Electrochim. Acta 2020, 332, 135514. [Google Scholar] [CrossRef]
- Anasori, B.; Lu, J.; Rivin, O.; Dahlqvist, M.; Halim, J.; Voigt, C.; Rosen, J.; Hultman, L.; Barsoum, M.W.; Caspi, E.N. A Tungsten-Based Nanolaminated Ternary Carbide: (W,Ti)4C4-X. Inorg. Chem. 2019, 58, 1100–1106. [Google Scholar] [CrossRef]
- Galvin, T.; Hyatt, N.C.; Rainforth, W.M.; Reaney, I.M.; Shepherd, D. Molten Salt Synthesis of MAX Phases in the Ti-Al-C System. J. Eur. Ceram. Soc. 2018, 38, 4585–4589. [Google Scholar] [CrossRef]
- Xu, L.; Miao, Z.; Song, H.; Chen, W.; Chou, L. Significant Roles of Mesostructure and Basic Modifier for Ordered Mesoporous Ni/CaO-Al2O3 Catalyst towards CO2 Reforming of CH4. Catal. Sci. Technol. 2014, 4, 1759–1770. [Google Scholar] [CrossRef]
- Wang, N.; Shen, K.; Yu, X.; Qian, W.; Chu, W. Preparation and Characterization of a Plasma Treated NiMgSBA-15 Catalyst for Methane Reforming with CO2 to Produce Syngas. Catal. Sci. Technol. 2013, 3, 2278–2287. [Google Scholar] [CrossRef]
- Mo, L.; Leong, K.K.M.; Kawi, S. A Highly Dispersed and Anti-Coking Ni-La2O3/SiO2 Catalyst for Syngas Production from Dry Carbon Dioxide Reforming of Methane. Catal. Sci. Technol. 2014, 4, 2107–2114. [Google Scholar] [CrossRef]
- Wang, X.H.; Zhang, M.H.; Li, W.; Tao, K.Y. Synthesis and Characterization of Cobalt-Molybdenum Bimetallic Carbides Catalysts. Catal. Today 2008, 131, 111–117. [Google Scholar] [CrossRef]
- Lamont, D.C.; Thomson, W.J. Dry Reforming Kinetics over a Bulk Molybdenum Carbide Catalyst. Chem. Eng. Sci. 2005, 60, 3553–3559. [Google Scholar] [CrossRef]
- Erdöhelyi, A.; Cserényi, J.; Papp, E.; Solymosi, F. Catalytic Reaction of Methane with Carbon Dioxide over Supported Palladium. Appl. Catal. A Gen. 1994, 108, 205–219. [Google Scholar] [CrossRef] [Green Version]
- Mounfield, W.P.; Harale, A.; Román-Leshkov, Y. Impact of Morphological Effects on the Activity and Stability of Tungsten Carbide Catalysts for Dry Methane Reforming. Energy Fuels 2019, 33, 5544–5550. [Google Scholar] [CrossRef]
- Shao, H.; Kugler, E.L.; Dadyburjor, D.B. Ni-W-C and Co-W-C as Alternative Catalysts for Dry Reforming and Steam Reforming of Methane. Carbon 2006, 2712, 26506. [Google Scholar]
- Zhang, Y.; Zhang, S.; Zhang, X.; Qiu, J.; Yu, L.; Shi, C. Ni Modified WC x Catalysts for Methane Dry Reforming. In Advances in CO2 Capture, Sequestration, and Conversion; American Chemical Society: Washington, DC, USA, 2015; pp. 171–189. [Google Scholar] [CrossRef]
- Yan, Q.; Lu, Y.; To, F.; Li, Y.; Yu, F. Synthesis of Tungsten Carbide Nanoparticles in Biochar Matrix as a Catalyst for Dry Reforming of Methane to Syngas. Catal. Sci. Technol. 2015, 5, 3270–3280. [Google Scholar] [CrossRef]
- Hunt, S.T.; Kokumai, T.M.; Zanchet, D.; Román-Leshkov, Y. Alloying Tungsten Carbide Nanoparticles with Tantalum: Impact on Electrochemical Oxidation Resistance and Hydrogen Evolution Activity. J. Phys. Chem. C 2015, 119, 13691–13699. [Google Scholar] [CrossRef]
- Vijayakumar, P.; Senthil Pandian, M.; Lim, S.P.; Pandikumar, A.; Huang, N.M.; Mukhopadhyay, S.; Ramasamy, P. Facile Synthesis of Tungsten Carbide Nanorods and Its Application as Counter Electrode in Dye Sensitized Solar Cells. Mater. Sci. Semicond. Process. 2015, 39, 292–299. [Google Scholar] [CrossRef]
- Michalsky, R.; Zhang, Y.J.; Medford, A.J.; Peterson, A.A. Departures from the Adsorption Energy Scaling Relations for Metal Carbide Catalysts. J. Phys. Chem. C 2014, 118, 13026–13034. [Google Scholar] [CrossRef]
- Michalsky, R.; Zhang, Y.J.; Peterson, A.A. Trends in the Hydrogen Evolution Activity of Metal Carbide Catalysts. ACS Catal. 2014, 4, 1274–1278. [Google Scholar] [CrossRef]
- Xiao, T.; Wang, H.; York, A.P.E.; Williams, V.C.; Green, M.L.H. Preparation of Nickel-Tungsten Bimetallic Carbide Catalysts. J. Catal. 2002, 209, 318–330. [Google Scholar] [CrossRef]
- Shao, H.; Kugler, E.L.; Ma, W.; Dadyburjor, D.B. Effect of Temperature on Structure and Performance of In-House Cobalt-Tungsten Carbide Catalyst for Dry Reforming of Methane. Ind. Eng. Chem. Res. 2005, 44, 4914–4921. [Google Scholar] [CrossRef]
- Hunt, S.T.; Milina, M.; Alba-Rubio, A.C.; Hendon, C.H.; Dumesic, J.A.; Roman-Leshkov, Y. Self-Assembly of Noble Metalmonolayers on Transition Metalcarbide Nanoparticle Catalysts. Science 2016, 352, 974–978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wannakao, S.; Artrith, N.; Limtrakul, J.; Kolpak, A.M. Engineering Transition-Metal-Coated Tungsten Carbides for Efficient and Selective Electrochemical Reduction of CO2 to Methane. ChemSusChem 2015, 8, 2745–2751. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.S.; Cai, N.S. Modeling of Multiple Cycles for Sorption-Enhanced Steam Methane Reforming and Sorbent Regeneration in Fixed Bed Reactor. Energy Fuels 2007, 21, 2909–2918. [Google Scholar] [CrossRef]
- Barbosa, R.D.; Baldanza, M.A.S.; de Resende, N.S.; Passos, F.B.; da Silva, V.L.d.S.T. Nickel–Promoted Molybdenum or Tungsten Carbides as Catalysts in Dry Reforming of Methane: Effects of Variation in CH4/CO2 Molar Ratio. Catal. Lett. 2021, 151, 1578–1591. [Google Scholar] [CrossRef]
- York, A.P.E.; Claridge, J.B.; Márquez-Alvarez, C.; Brungs, A.J.; Tsang, S.C.; Green, M.L.H. Synthesis of Early Transition Metal Carbides and Their Application for the Reforming of Methane to Synthesis Gas. Stud. Surf. Sci. Catal. 1997, 110, 711–720. [Google Scholar] [CrossRef]
- Gavrilova, N.N.; Sapunov, V.N.; Skudin, V.V. Intensification of Dry Reforming of Methane on Membrane Catalyst. Chem. Eng. J. 2019, 374, 983–991. [Google Scholar] [CrossRef]
- Grigoryan, R.R.; Aloyan, S.G.; Harutyunyan, V.R.; Arsentev, S.D.; Tavadyan, L.A. Dry Reforming of Methane over Nanosized Tungsten Carbide Powders Obtained by Mechanochemical and Plasma-Mechanochemical Methods. Pet. Chem. 2019, 59, 1256–1263. [Google Scholar] [CrossRef]
- Iyer, M.V.; Norcio, L.P.; Punnoose, A.; Kugler, E.L.; Seehra, M.S.; Dadyburjor, D.B. Catalysis for Synthesis Gas Formation from Reforming of Methane. Top. Catal. 2004, 29, 197–200. [Google Scholar] [CrossRef]
- Li, S.; Zhang, G.; Wang, J.; Liu, J. Enhanced Activity of Co Catalysts Supported on Tungsten Carbide-Activated Carbon for CO2 Reforming of CH4 to Produce Syngas. Int. J. Hydrogen Energy 2021, 46, 28613–28625. [Google Scholar] [CrossRef]
- Solymosi, F.; Németh, R.; Oszkó, A. The Oxidative Dehydrogenation of Propane with CO2 over Supported Mo2C Catalyst. Stud. Surf. Sci. Catal. 2001, 136, 339–344. [Google Scholar] [CrossRef]
- Roohi, P.; Alizadeh, R.; Fatehifar, E. Dry Reforming of Methane over Nano-Mo2C/Al2O3 Catalyst: Effect of Carburization Conditions on Excess Carbon Deposition. Energy Sources Part A Recover. Util. Environ. Eff. 2016, 38, 3565–3571. [Google Scholar] [CrossRef]
- Oshikawa, K.; Nagai, M.; Omi, S. Active Species of Molybdenum Carbide Catalysts in Methane Reforming: η-Mo3C2. Chem. Lett. 2000, 29, 1086–1087. [Google Scholar] [CrossRef]
- Kurlov, A.; Huang, X.; Deeva, E.B.; Abdala, P.M.; Fedorov, A.; Müller, C.R. Molybdenum Carbide and Oxycarbide from Carbon-Supported MoO3 Nanosheets: Phase Evolution and DRM Catalytic Activity Assessed by TEM and: In Situ XANES/XRD Methods. Nanoscale 2020, 12, 13086–13094. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhang, A.J.; Zhu, A.M.; Xu, Y.; Au, C.T.; Shi, C. A Carbide Catalyst Effective for the Dry Reforming of Methane at Atmospheric Pressure. ACS Symp. Ser. 2010, 1056, 181–196. [Google Scholar] [CrossRef]
- Porosoff, M.D.; Myint, M.N.Z.; Kattel, S.; Xie, Z.; Gomez, E.; Liu, P.; Chen, J.G. Identifying Different Types of Catalysts for CO2 Reduction by Ethane through Dry Reforming and Oxidative Dehydrogenation. Angew. Chemie Int. Ed. 2015, 54, 15501–15505. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.M.; Bhan, A. Effects of Oxygen Coverage on Rates and Selectivity of Propane-CO2 Reactions on Molybdenum Carbide. J. Catal. 2018, 357, 195–205. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Y.; Yao, Z.; Yan, S.; Kang, X. Finding of a New Cycle Route in Ni/Mo2C Catalyzed CH4-CO2 reforming. Catal. Sci. Technol. 2021, 11, 479–483. [Google Scholar] [CrossRef]
- Du, X.; France, L.J.; Kuznetsov, V.L.; Xiao, T.; Edwards, P.P.; AlMegren, H.; Bagabas, A. Dry Reforming of Methane over ZrO2-Supported Co–Mo Carbide Catalyst. Appl. Petrochem. Res. 2014, 4, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Shi, C.; Chen, B.; Zhang, Y.; Zhu, Y.; Qiu, J.; Au, C. Catalytic Role of β-Mo2C in DRM Catalysts That Contain Ni and Mo. Catal. Today 2015, 258, 676–683. [Google Scholar] [CrossRef]
- Sugiyama, S.; Oribe, K.; Endo, S.; Yoshida, T.; Shimoda, N.; Katoh, M.; Kato, Y.; Ninomiya, W. Enhancement of the Catalytic Activity Associated with Carbon Deposition Formed on NiO/γ-Al2O3 Catalysts during the Direct Dehydrogenation of Isobutane. J. Chem. Eng. Jpn. 2021, 54, 35–43. [Google Scholar] [CrossRef]
- Thakur, R.; Vahidmohammadi, A.; Smith, J.; Hoffman, M.; Moncada, J.; Beidaghi, M.; Carrero, C.A. Insights into the Genesis of a Selective and Coke-Resistant MXene-Based Catalyst for the Dry Reforming of Methane. ACS Catal. 2020, 10, 5124–5134. [Google Scholar] [CrossRef]
- Diao, Y.; Zhang, X.; Liu, Y.; Chen, B.; Wu, G.; Shi, C. Plasma-Assisted Dry Reforming of Methane over Mo2C-Ni/Al2O3 Catalysts: Effects of β-Mo2C Promoter. Appl. Catal. B Environ. 2022, 301, 120779. [Google Scholar] [CrossRef]
- Han, B.; Zhong, J.; Li, W.; Zhang, Z.; Bi, G.; Xie, J. The Promotional Role of β-Cyclodextrin on Ni-Mo2C/MgO Catalyst for Biogas Reforming. Mol. Catal. 2021, 515, 111897. [Google Scholar] [CrossRef]
- Shamsi, A. Methane Dry Reforming over Carbide, Nickel-Based, and Noble Metal Catalysts. ACS Symp. Ser. 2002, 809, 182–196. [Google Scholar] [CrossRef]
- Ronda-Lloret, M.; Marakatti, V.S.; Sloof, W.G.; Delgado, J.J.; Sepúlveda-Escribano, A.; Ramos-Fernandez, E.V.; Rothenberg, G.; Shiju, N.R. Butane Dry Reforming Catalyzed by Cobalt Oxide Supported on Ti2AlC MAX Phase. ChemSusChem 2020, 13, 6401–6408. [Google Scholar] [CrossRef] [PubMed]
- Kurlov, A.; Deeva, E.B.; Abdala, P.M.; Lebedev, D.; Tsoukalou, A.; Comas-Vives, A.; Fedorov, A.; Müller, C.R. Exploiting Two-Dimensional Morphology of Molybdenum Oxycarbide to Enable Efficient Catalytic Dry Reforming of Methane. Nat. Commun. 2020, 11, 4920. [Google Scholar] [CrossRef] [PubMed]
| Process | Main Reaction | Enthalpy ΔH0298 K [kJ/mol] | Pressure [bar] | H2/CO Ratio |
|---|---|---|---|---|
| Dry reforming of methane (DRM) | CH4 + CO2 = 2CO + 3 H2 | +247 | 1 | 1:1 |
| Steam reforming of methane (STM) | CH4 + H2O = CO + 3H2O | +206 | 3–25 | 3:1 |
| Partial oxidation of methane (POM) | CH4 + ½ O2 = CO + 2H2 | −35.2 | 100 | 2:1 |
| Autothermal reforming (ATR) | CH4 + H2O = CO + 3H2O CH4 + ½ O2 = CO + 2H2 | +206 −35.2 | 1–50 | 1:1 or 1:2 |
| Type of Mill (Balls and Vessel) | Substrates | Ball-to-Powder Weight Ratio | Rotation Speed | Inert Gas Atmosphere | Milling Time | Additional Process Stage | Characteristics of Obtained Particles | Ref. |
|---|---|---|---|---|---|---|---|---|
| powder mixture (99.9% purity) of W and amorphous C in a stoichiometric amount (6.12 wt.% C) | 10:1 | 300 rpm | Ar | 8 h 12 h | - |
| [62] |
| powder mixture of WO3 (99.9% purity, ~20 µm) and graphite (99.9% purity, ~1.7 µm) | 40:1 | 250 rpm | - | 20 h |
|
| [13] |
| powder mixture of WO3 (>99% purity), Mg (99.9% purity), and graphite (99.9% purity) at atomic ratio of 1:1:1 | 20:1 to 50:1 | 250 rpm | H2/Ar | 50 h | - |
| [102] |
| powder mixture (99.9% purity) of W (−75 mesh) and C (−350 mesh) | 10:1 | n.d. | Ar | 71 h 120 h | - |
| [103] |
| powder mixture (8 g) of W (99% purity, mean particle size ~5 µm) and granulated activated carbon (particle size ~0.9–1.7 mm) in the atomic ratio 50:50 | 30:1 | n.d. | - | 310 h |
|
| [104] |
| Type of Mill (Balls and Vessel) | Substrates | Ball-to-Powder Weight Ratio | Rotation Speed | Inert Gas Atmosphere | Milling Time | Additional Process Stage | Characteristics of Obtained Particles | Ref. |
|---|---|---|---|---|---|---|---|---|
|
| 20:1 | n.d. | Ar | 10 min | Pressureless sintering in a hot press: The mechanically activated mixture was loaded into a die. The die was set into a hot-pressing plant. P = 1 MPa, T = 900 and 1000°C, t = 30 min, argon atmosphere. |
| [88] |
|
| 40:1 | n.d. | Ar | 30 h | - |
| [97] |
|
| 10:1 | n.d. | Ar | 15, 20 h | - |
| [98] |
|
| 10:1 | 300 rpm | Ar | 8, 16 h | Using stearic acid as a PCA (process-control agent) during milling to prevent sticking of the powder to the balls and vial. |
| [105] |
|
| n.d. | n.d. | He | 60, 82, 96 h | - |
| [51] |
| Type of Mill (Balls and Vessel) | Substrates | Ball-to-Powder Weight Ratio | Rotation Speed | Inert Gas Atmosphere | Milling Time | Additional Process Stage | Characteristics of Obtained Particles | Ref. |
|---|---|---|---|---|---|---|---|---|
| Planetary mill with stainless-steel vials and balls | MoO3, Al, graphite at different molar ratios | 20:1 | 300 rpm | Ar | 2–150 h | Annealing after the milling under air or Ar atmosphere |
| [96] |
| Planetary mill with stainless-steel vials and balls | MoO3, graphite | 15:1 | 450 rpm | Ar | 5, 10, 15 and 24 h | Pressing at 4–0 MPa, heating, and microwave irradiation (800 W, 60 s). After microwave treatment, annealing at 700 °C and 1000 °C for 30 min |
| [106] |
| Planetary mill with stainless-steel vials and balls | MoS2, graphite, Na2CO3 | 15:1 | n.d. | Ar | 10, 20, 40, 50, and 70 h | - |
| [100] |
| Planetary ball mill | Graphite, Mo powder, melamine | n.d. | n.d. | n.d. | n.d. | DC arc discharge plasma sintering |
| [101] |
| High-energy planetary mill with stainless-steel balls (10 mm) | Carbon black P145 (18–25 nm), (NH4)6Mo7O24x 4H2O | 40:1 | 600–1000 m/s2 | yes | 30–60 min | Calcination at 760 °C and 800 °C in inert atmosphere |
| [99] |
| Precursors | Preparation of the Precursor Mixture | Carbothermal Reduction | Characteristics of Obtained Particles | Ref. | |||
|---|---|---|---|---|---|---|---|
| Type of Furnace | Flowing Gas | Temperature and Heating Rate | Time | ||||
|
| vacuum coal tube furnace (3.8 × 10−2 Pa) | - | 1000 °C 7–10 °C/min | 2 h |
| [61] |
|
| vacuum tube furnace | - | 980 °C 10 °C/min | 1 h |
| [68] |
|
| tube furnace | H2/N2 (VH2/VN2 1:3, 50 mL/min) | 900 °C | 3 h |
| [129] |
|
| tube furnace | N2 | 800 °C | 2 h |
| [130] |
|
| tube furnace | Ar H2/Ar (4% vol. H2, 96% vol. Ar) | 1300 °C 3.3 °C/min | 2 h (400 °C) 0.5 h (1300 °C) |
| [131] |
|
| tube furnace | - | 1000 °C 40 °C/min | 10 h |
| [132] |
|
| horizontal fixed bed alumina reactor | CH4/H2 (95% H2, 5% CH4, 20 l/h) | 850 °C (APT) 820 °C (TBO) 5 °C/min | 2 h |
| [133] |
|
| alumina tube furnace | N2 (283.17 l/h) | 1300 °C 5 °C/min (holding at 300 °C for 30 min to remove excess O2) | 3 h |
| [134] |
|
| vacuum furnace (shape memory synthesis) | - | 1300 °C 1200 °C | 7 h 30 h |
| [135] |
|
| vacuum furnace (4 Ñ 10−1 Pa) | - | 1100–1200 °C 10 °C/min | 3 h |
| [136] |
| Substrates for Organic–Inorganic Precursor Preparation | Preparation of the Precursor | Carbothermal Reduction | Characteristics of Obtained Particles | Ref. | ||
|---|---|---|---|---|---|---|
| Flowing Gas | Temperature | Time | ||||
|
| N2 | 700 °C, 800 °C, and 900 °C | - |
| [115] |
|
| N2 | 900 °C | - |
| [114] |
|
| N2 | 800 °C | 3 h |
| [118] |
|
| N2 | 900 °C | 2 h |
| [117] |
|
| Ar | 650, 750, 850, and 950 °C | 3 h |
| [121] |
|
| Inert | 600 °C, 700 °C, 800 °C, 900 °C | - |
| [112] |
|
| n.d. | 800°C | 2 h |
| [137] |
|
After carburization step, removal of copper particles by washing with iron chloride solution. | N2 | 800°C | 6 h |
| [120] |
| Method I:
| N2 | 1st step: 200 °C for 12 h, 2nd step: heating to carburization temperature under inert gas, 3rd step: heating under 20% CH4/H2 | 3 h | - | [138] |
|
| Ar | 625 °C, 725 °C, 750 °C | 5 h |
| [139] |
| Ti Source | C Source | Salts | Salt:Ti Molar Ratio | Temperature | Time | Characteristic of Obtained Product | Ref. |
|---|---|---|---|---|---|---|---|
| Ti powder: fine particles (1–3 μm) and large irregular particles (20–40 μm) |
| LiCl-KCl-KF | n.d. | 1100 °C (10 °C/min, 5 °C/min from 600 °C) | 6 h |
| [154] |
| Ti powder (99% purity) |
| KCl-LiCl | 9:1 (salt:reactants mass ratio) C:Ti molar ratio of 2:1 | 700, 815, 880, 950 °C | 2, 3, 4 h |
| [157] |
| Ti powder (99.9% purity) with the diameter of 100–150 μm |
| KCl-KF | 2:1, 3:1, 4:1 | 700–1000 °C | 3, 5 h |
| [155] |
| Ti powder (99.8% purity) with the size of 325 mesh |
| NaCl-KCl | 7:1 (salt:reactants mass ratio) 1.5 g of 1:1 Ti:C mixture | 750, 800, 850, 900 °C | 1, 2, 3, 4 h |
| [156] |
| Ti powder (99.5% purity) with the size of 200 mesh |
| LiCl-KCl-KF | 1.5:1, 2.5:1 | 900 and 950 °C | 1–5 h |
| [158] |
| Mo and C Source | Salts | Ratio of MS Components | Temperature | Time | Electrochemical Reaction Parameters | Characteristics of Obtained Product | Ref. |
|---|---|---|---|---|---|---|---|
| Mo and C powders (200 mesh) | NaCl and KCl | equimolar | 1000°C | 1 h | - |
| [148] |
| MoS2, graphite powder | NaCl and KCl | equimolar | 1st step: 800 °C, 2nd step: 700 °C | 2 h 10 min | Cell voltage 2.6 V |
| [159] |
| Mo foil, CO2 | CaCl2, CaO | CaO:CaCl2 = 1:10 | 850 °C | 5 h | Voltage 2.5 V |
| [152] |
| Mo plate CO2 | LiCO3 | - | 800 °C | 1 h 2 h | Cell voltage 3.1 V |
| [151] |
| MoO3 CFP-carbon fiber paper, carbon black | NaCl, KCl | equimolar | 1000 °C | 3 h 6 h 9 h | - |
| [150] |
| Mo powder, carbon nanotubes | LiCl, KCl, KF | mole ratio: 58/40/2 | 950 °C | 1 h |
| [160] |
| MAX Matrix, Synthesis Method | Substrates | Ratio of Substrates | Preliminary Preparation of Substrates for Synthesis | Temperature and Time of Solid-State Synthesis | Comments | Ref. |
|---|---|---|---|---|---|---|
| Mo2Ga2C Reactive sintering | Molybdenum and graphite powders, gallium shots | Mo:C molar ratio = 2: 1 Mo2C:Ga molar ratio = 1:8 | Ball milling of Mo and C powders for 24 h, grinding formed Mo2C with gallium | 850 °C for 48 h | 20% contamination of Mo2Ga2C phase with unreacted Mo2C, Ga, or Ga2O3 | [173] |
| Mo3Al2C Reactive sintering | Elemental powders of particular constituents | n.d. | n.d. | 24 h at 1500 °C with one intermediate grinding and compacting step, followed by ball milling and hot pressing at 1250 °C and at 56 MPa | Obtained MAX revealed unconventional superconductivity with possibly a nodal structure of the superconducting gap | [183] |
| Mo4ValC4 Reactive sintering | Molybdenum (250 mesh), vanadium (325 mesh), vanadium (III) oxide, aluminum (325 mesh), and graphite (325 mesh) powders | Mo:V:V2O3:Al:C = 4:0.9:0.05:1.2:3.5 | Grinding and pestling in agate mortar for 5 min | 1650 °C for 4 h under argon atmosphere | The synthesized MAX phase contained impurities of intermetallic and oxide compounds. They were removed by dissolution in 12 M HCl. | [176] |
| Mo2TiAlC2 Mo2Ti2AlC3 Reactive sintering | Elemental powders: Mo (325 mesh), Ti (325 mesh), and Al (300 mesh) | mMo:(3-m) Ti:1.1Al:2C, where m = 1.5, 1.8. 2, or 2.2. | Ball milling for 18 h | 1600 °C for 4 under Ar flow | The different ratios of starting materials led to formation of different major phases: m ≥ 2 led to (Mo2Ti)AlC2; for m < 2, (Mo2Ti2)AlC3 was the major product | [172] |
| (Mo2/3Sc1/3)2AlC Reactive sintering | Elemental powders: graphite, Mo, Al, and Sc | Stoichiometric ratio | Mixing in agate mortar | 1500 °C for 20 h under Ar flow | - | [166] |
| (W,Ti)4C4-x, x = 1.4 Reactive sintering | Powders of W, Ti, Al, and C | Molar ratio of W:Ti:Al:C = 2:1:1.1:2 | n.d. | 1600 °C for 4 h | Actual composition ≈ W2.1(1)Ti1.6(1)C2.6(1) | [188] |
| (W2/3Sc1/3)2AlC (W2/3Y1/3)2AlC Reactive sintering | Elemental powders of W (12 µm), Sc (−200 mesh), Y (40 mesh), Al, and C (−200 mesh) | Stoichiometric ratio | n.d. | 1450 °C for 2 h under Ar flow | (W2/3Sc1/3)2AlC sample contained 43 wt.% of (W2/3Sc1/3)2AlC and 31 wt.% of unreacted W | [165] |
| Ti2AlC Hot pressing | TiC (11.8 um), aluminum (11.8 um), active carbon (13.2 um), Ti (10.6 um) | TiC:Ti:Al:C: = 0.5:1.5:1.0:0.5 | Mixing in ethanol for 24 h, pressing at high temperatures (1300 °C, 1400 °C, 1450 °C, and 1500 °C) at 30 Mpa under Ar flow | Sintering at pressing temperature: 1300 °C, 1400 °C, 1450 °C, or 1500 °C, the soaking time: 60 min | The main identified phase was Ti2AlC; however, with temperature increase (more than 1450 °C), Ti3AlC2 phase became more significant. Intermetallic impurities of Ti-Al were also detected. | [171] |
| Ti3AlC2 Ti3SiC2 Spark plasma sintering (SPS) | Ti (10.6 um), Si (9.5 um), Al (12.8 um), and TiC (8.4 um) | n.d. | Mixing in ethanol for 24 h | Spark plasma sintering 1150–1300 °C, the soaking time 8 min | High-purity Ti3AlC2 can be obtained at temperatures 1200–1250 °C and molar ratio of TiC:Ti:Al:Si = 2:1:1:0.2 | [170] |
| Ti2AlC, Ti3AlC2 Molten salt synthesis method | Ti powder (74 um), aluminum (44–420 um), graphite (44 um), sodium chloride, potassium chloride (eutectic phase) | Molar ratio for Ti2AlC preparation: Ti:Al:C = 2:1.2:1, for Ti3AlC2: Ti:Al:C = 3:1.2:2. The salt-to-MAX constituents weight ratio: 1:1 | Ball milling (1800 rpm) in heptane to prevent dissolution of salts by adsorbed water fallowed by drying at 95 °C for 8 h and pressing at 140 MPa to form disks | For Ti2AlC from 900 to 1000 °C, the reaction time: 2 h; for Ti3AlC2 1300 °C, reaction time 2 h | The excess of Al element was required due to its volatility. Formation of MAX phase in shape of globular and long needles. | [189] |
| Type of Catalyst | Catalyst Properties | Catalyst Synthesis Method | Dry Methane Reforming | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catalyst Mass | Temperature | Pressure | Time | WHSV/ GHSV | CH4 /CO2 Ratio | CO2 Conversion | CH4 Conversion | CO Yield | H2/CO Ratio | ||||
| WC | n.d. |
| 3 g | 850 °C | 0.5 MPa | 500 h | 4000–12,000 h−1 | 0.7 0.9 1.0 1.1 1.4 | 85% 89% 93% 100% 100% | 92.5% 88% 82% 78% 76% | 83% 90% 92% 86% 79% | 0.61 0.69 0.79 0.85 1.03 | [199] |
| α-WC/γAl2O3 | nanorods |
| 0.2 g | 900 °C | 1 atm | 5000 TON1) | n.d. | 1 | 55% | 45% | 48% | n.d. | [196] |
| β-W2C/γAl2O3 | nanoparticles | 81% | 90% | 76% | n.d. | ||||||||
| α-WC/W2C | 39 m2/g (CH4), 71 m2/g (C2H6) |
| n.d. | 850 °C | 1 bar 8.3 bar | 8 h 140 h | 2870 h−1 | 1 | 93.1% 75.4% | 92% 62.7% | 92.6% 68.6% | 0.94 0.79 | [210] |
| WC | 20.6 m2/g d = 80 nm |
| 1.066 g | 900 °C 950 °C 970 °C | n.d. | 50 h | n.d. | 1 | 61.0% 79.6% 82.8% | 28.4% 57.6% 62.2% | n.d. | 0.43 0.69 0.71 | [211] |
| WC | d = 18 nm |
| 1.2 g | 843 °C 950 °C | 0.867 bar | 60 h | n.d. | 1 | n.d. | 34% 55% | n.d. | 1.22 1.48 | [212] |
| Co6W6C | 2–3 m2/g |
| 0.3 g | 850 °C | 3.4 atm | 100 h | n.d. | 1 | 70% | 75% | 61% | 0.91 | [205] |
| Co6W6C | 5 m2/g, particle size < 38 μm |
| 0.3 g | 850 °C | 5 atm | 20 h 90 h | 11,200 cm3/h/gcat | 1 | 78% 78% | 82% 81% | 76% | 1.01 0.99 | [213] |
| Co-βW2C/α-WC | 438.1 m2/g, 0.58 cm3/g |
| 2 g | 800 °C | 1 atm | 1 h 10 h | 36,000–72,000 cm3/h/gcat | 1 | 90% 78% | 82% 69% | n.d. | 0.86 0.69 | [214] |
| Ni-WC | 25 m2/g, 0.09 cm3/g |
| n.d. | 800 °C | 1 atm | 20 h | n.d. | 0.67 1.00 1.50 | 75% 85% 85% | 99% 75% 59% | 80%2) 83%2) 83%2) | 0.68 0.79 0.80 | [209] |
| Ni-WCx | Ni/W = 0.5 NiW = 4 |
| 0.2 g | 800 °C | 1 atm | 18,000 cm3/h/gcat | 1 | 71% 68% | 58% 55% | n.d. | 0.69 0.68 | [198] | |
| Type of Catalyst | Mo/C Ratio | Catalyst Synthesis Method | Dry Methane Reforming | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature | Pressure | WHSV/ GHSV [mlg−1h−1] | Sample | CO2 Conversion [%] | CH4 Conversion [%] | TOS | H2/CO Ratio | ||||||
| CO20 | CO2 | CH40 | CH4 | ||||||||||
| β-Mo2C | 0 5 10 15 30 60 100 wt.% |
| 850 °C | atm | 18,000 | 0 5 10 15 30 60 100 | 90 90 n.d. 90 90 90 5 | 25 15 n.d. 25 25 25 5 | 80 85 n.d. 85 85 85 5 | 15 10 n.d 15. 10 10 5 | 4 h 5 h n.d. 6 h 14 h 6h 6 h | n.d. | [79] |
| Mo2C | Ascorbic acid/Mo = 1.0 |
| 650 °C 725 °C 775 °C 825 °C 850 °C | n.d. | 30 | Mo2C | 20 30 70 70 75 | 10 15 35 85 90 | n.d. | 0.1 0.2 0.35 0.6 0.6 | [123] | ||
| β-Mo2C α-MoC1-x | n.d. |
| 850 °C | atm | 6000 | IE(Ar) IWI(Ar) MM(Ar) IE(H2) | 95 95 64 80 | 95 30 35 25 | 98 95 55 68 | 98 25 23 17 | 12 h 12 h 4 h 4 h | n.d n.d. n.d. n.d. | [78] |
| Mo2C/Al2O3 | 5 wt.% Mo, 12.5 wt.% Mo 20 wt.% |
| 650 °C 750 °C 800 °C | atm | 18,000 | 5Mo 12.5Mo 20Mo | 18 20 25 | 20 25 30 | 12 h 12 h 12 h | 0.6 0.65 0.6 | [216] | ||
| 5Mo 12.5Mo 20Mo | 55 65 60 | 50 60 60 | 12 h 12 h 12 h | 0.65 0.7 0.72 | |||||||||
| 5Mo 12.5Mo 20Mo | 85 95 93 | 85 90 90 | 12 h 12 h 12 h | 0.82 0.85 0.8 | |||||||||
| Mo2C (me) Mo2C (et) Mo2C/TiO2 |
| 850 °C | n.d. | 5040 | Mo2C (me) Mo2C (et) Mo2C/TiO2 | 99.8 99.9 88.7 | 92.1 89.5 70.1 | n.d. | 1.0 1.1 0.9 | [229] | |||
| Type of Catalyst | Mo, Ni, and C Contents/ Ratio | Catalyst Synthesis Method | Dry Methane Reforming | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature | Pressure | WHSV/ GHSV [mlg−1h−1] | Sample | CO2 Conversion [%] | CH4 Conversion [%] | TOS | H2/CO Ratio | ||||||
| CO20 | CO2 | CH40 | CH4 | ||||||||||
| Ni-Mo2C/MgO | Ni+Mo:β-cyclodextrin = 60:1, Ni: 7 wt.%, Mo: 15 wt.% |
| 850 °C | 1 atm | 30,000 | Ni-Mo2C/MgO | 95 | 95 | 95 | 90 | 200 h | n.d. | [228] |
| Ni/Mo2C/CNT | Ni:Mo = 0.5 (0.5Ni) NI:Mo = 1.0 (1Ni) Ni:Mo = 1.5 (1.5Ni) Ni:Mo = 2.0 (2Ni) |
| 850 °C | 1 atm | 60,000 | 0.5Ni 1Ni 1.5Ni 2Ni | 85 80 85 85 | 65 70 25 40 | 70 70 75 70 | 65 65 10 30 | 22 h 36 h 5 h 8 h | n.d. | [222] |
| Ni-Mo2C/La2O3 | Ni: 4.4 wt.% Mo 14.6 wt.%, Ni:Mo = 1:2 |
| 800 °C | 1 atm | 12,000 18,000 | Ni-Mo2C/La2O3 | 70 | 80 | 50 | 60 | 50 h | n.d. | [224] |
| Ni-Mo2C | Ni:Mo = 0 Ni:Mo = 1 Ni:Mo = 2 Ni:Mo = 3 |
| 800 °C | 1 atm | Ni:Mo = 0 Ni:Mo = 1 Ni:Mo = 2 Ni:Mo = 3 | 0 95 92 8 | 0 70 90 0 | 0 80 80 40 | 0 30 80 15 | 6 h 9 h 22 h | 0 0.5 0.52 0.15 | [47] | |
| Ni-Mo2C/Al2O3 | Al.:urea = 1:2.5 nStarch:nAl. Salt: 0.125 n(Ni+Mo) = 1.5 Ni content: 15 mol.% Mo content: 0. 3. 5 and 10 mol.% |
| 800°C | 1 at | 12,000 | 0Mo 3Mo 5Mo 10Mo | 89 92 92 94 | 89 93 94 90 | 84 87 90 85 | 84 87 88 84 | 15 h 15 h 15 h 15 | 0.99 0.97 0.99 0.99 | [116] |
| Mo2C-Ni/Al2O3 | Ni:Mo = 2:1 Ni:Mo = 1:2 Ni:Mo = 1:5 |
| 480 °C Plasma treatment | 1 atm | 50,000 | Ni:Mo = 2:1 Ni:Mo = 1:2 Ni:Mo = 1:5 | 85 7 605 | 80 75 60 | 80 75 61 | 80 75 61 | 11 h n.d. n.d. | 0.9 n.d n.d. | [227] |
| Ni/MoCx/SiO2Ni/MoCx/Al2O3Ni/MoCx/SiC | 20% mol. of Mo, Ni: Mo = 0.2 (0.2NiMo/support) 0.3 (0.3NiMo/support) and 0.4 (0.4NiMo/support) |
| 800 °C | 1 atm | 10,000 | 0.2NiMo/SiC 0.3NiMo/SiC 0.4NiMo/SiC | 90 90 90 | 85 85 85 | 85 85 85 | 75 75 75 | 0.8 0.8 0.8 | 20 h | [10] |
| 0.2NiMo/SiO2 0.3NiMo/SiO2 0.4NiMo/SiO2 | 25 25 75 | 25 25 90 | 13 13 65 | 13 13 75 | 0.25 0.25 0.75 | ||||||||
| 0.2NiMo/Al2O3 0.3NiMo/Al2O3 0.4NiMo/Al2O3 | 25 95 95 | 12 95 95 | 12 80 80 | 5 80 80 | 0.25 0.8 0.8 | ||||||||
| Method | Advantages | Disadvantages |
|---|---|---|
| Reactive sintering |
|
|
| TPC, TPR |
|
|
| High-energy milling |
|
|
| Structure-directing method |
|
|
| Molten salt synthesis |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czaplicka, N.; Rogala, A.; Wysocka, I. Metal (Mo, W, Ti) Carbide Catalysts: Synthesis and Application as Alternative Catalysts for Dry Reforming of Hydrocarbons—A Review. Int. J. Mol. Sci. 2021, 22, 12337. https://doi.org/10.3390/ijms222212337
Czaplicka N, Rogala A, Wysocka I. Metal (Mo, W, Ti) Carbide Catalysts: Synthesis and Application as Alternative Catalysts for Dry Reforming of Hydrocarbons—A Review. International Journal of Molecular Sciences. 2021; 22(22):12337. https://doi.org/10.3390/ijms222212337
Chicago/Turabian StyleCzaplicka, Natalia, Andrzej Rogala, and Izabela Wysocka. 2021. "Metal (Mo, W, Ti) Carbide Catalysts: Synthesis and Application as Alternative Catalysts for Dry Reforming of Hydrocarbons—A Review" International Journal of Molecular Sciences 22, no. 22: 12337. https://doi.org/10.3390/ijms222212337






