Kinase-Inhibitors in Iodine-Refractory Differentiated Thyroid Cancer—Focus on Occurrence, Mechanisms, and Management of Treatment-Related Hypertension
Abstract
1. Introduction
Multikinase Inhibitors
2. Methods
2.1. Eligibility Criteria
2.2. Search
2.3. Study Selection
3. Results
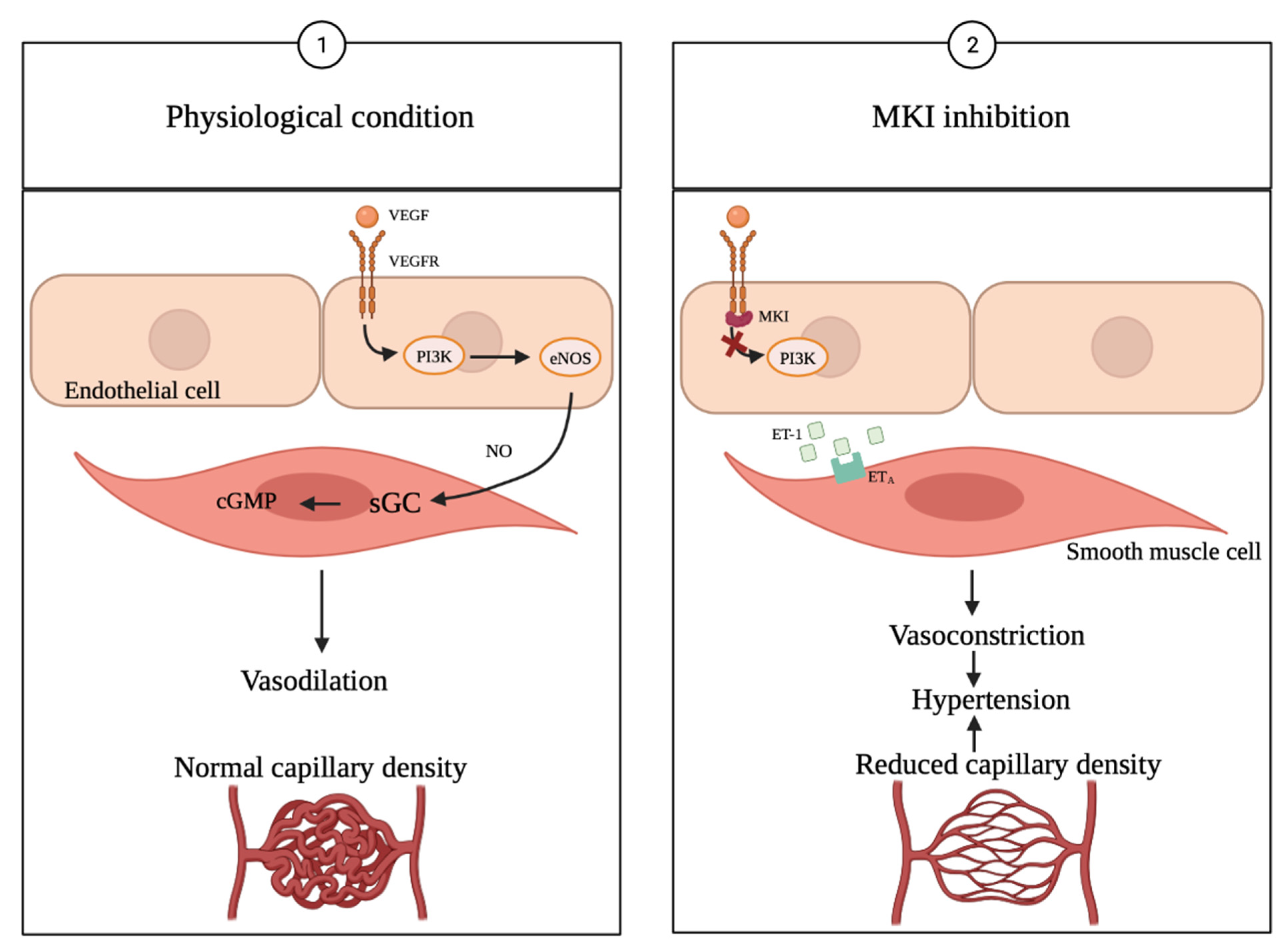
3.1. Multikinase Inhibitor Treatment Causes Hypertension
3.2. Clinical Study Findings
3.3. Management of MKI-Induced Hypertension
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AE(s) | adverse effect(s) |
| ATC | anaplastic thyroid cancer |
| BP | blood pressure |
| CV | cardiovascular |
| DBP | diastolic blood pressure |
| DTC | differentiated thyroid cancer |
| EGF | epidermal growth factor |
| EGFR | epidermal growth factor receptor |
| FGFR | fibroblast growth factor receptor |
| FTC | follicular thyroid cancer |
| HCC | hepatocellular carcinoma |
| HTC | Hürthle cell thyroid cancer |
| HGF | hepatocyte growth factor |
| HGFR | hepatocyte growth factor receptor |
| MTC | medullary thyroid cancer |
| MKI | multikinase inhibitor |
| ORR | overall response rate |
| OS | overall survival |
| PTC | papillary thyroid cancer |
| PDGF | platelet-derived growth factor |
| PDGFR | platelet-derived growth factor receptor |
| PDTC | poorly differentiated thyroid cancer |
| PFS | progression-free survival |
| RAI | radioactive iodine |
| RCC | Renal clear cell carcinoma |
| RR-DTC | RAI refractory DTC |
| RTK | receptor tyrosine kinases |
| SBP | systolic blood pressure |
| TEAE | treatment-emerged adverse effect |
| TE-HTN | treatment-emerged hypertension |
| TC | thyroid cancer |
| TK(s) | tyrosine kinase(s) |
| VEGFR | vascular endothelial growth factor receptor |
| VEGF | vascular endothelial growth factor |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Devesa, S.S.; Sosa, J.A.; Check, D.; Kitahara, C.M. Trends in Thyroid Cancer Incidence and Mortality in the United States, 1974–2013. JAMA 2017, 317, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Kiyota, N.; Yamazaki, T.; Chayahara, N.; Nakano, K.; Inagaki, L.; Toda, K.; Enokida, T.; Minami, H.; Imamura, Y.; et al. A Phase II study of the safety and efficacy of lenvatinib in patients with advanced thyroid cancer. Future Oncol. 2019, 15, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Carling, T.; Udelsman, R. Thyroid cancer. Annu. Rev. Med. 2014, 65, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, M.E.; McFadden, D.G.; Durante, C. Thyroid cancer. Lancet 2016, 388, 2783–2795. [Google Scholar] [CrossRef]
- Tirrò, E.; Martorana, F.; Romano, C.; Vitale, S.R.; Motta, G.; Di Gregorio, S.; Massimino, M.; Pennisi, M.S.; Stella, S.; Puma, A.; et al. Molecular Alterations in Thyroid Cancer: From Bench to Clinical Practice. Genes 2019, 10, 709. [Google Scholar] [CrossRef] [PubMed]
- John, W.; Baynes, M.H.D. Medical Biochemistry; Elsevier Health Sciences: Edinburgh, UK, 2019; Chapter 28. [Google Scholar]
- Zschäbitz, S.; Grüllich, C. Lenvantinib: A Tyrosine Kinase Inhibitor of VEGFR 1-3, FGFR 1-4, PDGFRα, KIT and RET. Recent Results Cancer Res. 2018, 211, 187–198. [Google Scholar] [CrossRef]
- Stjepanovic, N.; Capdevila, J. Multikinase inhibitors in the treatment of thyroid cancer: Specific role of lenvatinib. Biologics 2014, 8, 129–139. [Google Scholar] [CrossRef]
- Prete, A.; Borges de Souza, P.; Censi, S.; Muzza, M.; Nucci, N.; Sponziello, M. Update on Fundamental Mechanisms of Thyroid Cancer. Front. Endocrinol. 2020, 11, 102. [Google Scholar] [CrossRef]
- Marotta, V.; Sciammarella, C.; Colao, A.; Faggiano, A. Application of molecular biology of differentiated thyroid cancer for clinical prognostication. Endocr. Relat. Cancer 2016, 23, R499–R515. [Google Scholar] [CrossRef]
- Lorusso, L.; Cappagli, V.; Valerio, L.; Giani, C.; Viola, D.; Puleo, L.; Gambale, C.; Minaldi, E.; Campopiano, M.C.; Matrone, A.; et al. Thyroid Cancers: From Surgery to Current and Future Systemic Therapies through Their Molecular Identities. Int. J. Mol. Sci. 2021, 22, 3117. [Google Scholar] [CrossRef] [PubMed]
- Marotta, V.; Chiofalo, M.G.; Di Gennaro, F.; Daponte, A.; Sandomenico, F.; Vallone, P.; Costigliola, L.; Botti, G.; Ionna, F.; Pezzullo, L. Kinase-inhibitors for iodine-refractory differentiated thyroid cancer: Still far from a structured therapeutic algorithm. Crit. Rev. Oncol. Hematol. 2021, 162, 103353. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Wang, P. Lenvatinib in Management of Solid Tumors. Oncologist 2020, 25, e302–e310. [Google Scholar] [CrossRef]
- Resteghini, C.; Cavalieri, S.; Galbiati, D.; Granata, R.; Alfieri, S.; Bergamini, C.; Bossi, P.; Licitra, L.; Locati, L.D. Management of tyrosine kinase inhibitors (TKI) side effects in differentiated and medullary thyroid cancer patients. Best Pract. Res. Clin. Endocrinol. Metab. 2017, 31, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Marotta, V.; Sciammarella, C.; Vitale, M.; Colao, A.; Faggiano, A. The evolving field of kinase inhibitors in thyroid cancer. Crit. Rev. Oncol. Hematol. 2015, 93, 60–73. [Google Scholar] [CrossRef]
- Wirth, L.J.; Tahara, M.; Robinson, B.; Francis, S.; Brose, M.S.; Habra, M.A.; Newbold, K.; Kiyota, N.; Dutcus, C.E.; Mathias, E.; et al. Treatment-emergent hypertension and efficacy in the phase 3 Study of (E7080) lenvatinib in differentiated cancer of the thyroid (SELECT). Cancer 2018, 124, 2365–2372. [Google Scholar] [CrossRef]
- Ancker, O.V.; Wehland, M.; Bauer, J.; Infanger, M.; Grimm, D. The Adverse Effect of Hypertension in the Treatment of Thyroid Cancer with Multi-Kinase Inhibitors. Int. J. Mol. Sci. 2017, 18, 625. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Matsui, J.; Matsushima, T.; Obaishi, H.; Miyazaki, K.; Nakamura, K.; Tohyama, O.; Semba, T.; Yamaguchi, A.; Hoshi, S.S.; et al. Lenvatinib, an angiogenesis inhibitor targeting VEGFR/FGFR, shows broad antitumor activity in human tumor xenograft models associated with microvessel density and pericyte coverage. Vasc. Cell 2014, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Facemire, C.S.; Nixon, A.B.; Griffiths, R.; Hurwitz, H.; Coffman, T.M. Vascular endothelial growth factor receptor 2 controls blood pressure by regulating nitric oxide synthase expression. Hypertension 2009, 54, 652–658. [Google Scholar] [CrossRef]
- Costache, M.I.; Ioana, M.; Iordache, S.; Ene, D.; Costache, C.A.; Săftoiu, A. VEGF Expression in Pancreatic Cancer and Other Malignancies: A Review of the Literature. Rom. J. Intern. Med. 2015, 53, 199–208. [Google Scholar] [CrossRef]
- Shibuya, M.; Claesson-Welsh, L. Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp. Cell Res. 2006, 312, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Goel, H.L.; Mercurio, A.M. VEGF targets the tumour cell. Nat. Rev. Cancer 2013, 13, 871–882. [Google Scholar] [CrossRef]
- Mohamad Pakarul Razy, N.H.; Wan Abdul Rahman, W.F.; Win, T.T. Expression of Vascular Endothelial Growth Factor and Its Receptors in Thyroid Nodular Hyperplasia and Papillary Thyroid Carcinoma: A Tertiary Health Care Centre Based Study. Asian Pac. J. Cancer Prev. 2019, 20, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Laursen, R.; Wehland, M.; Kopp, S.; Pietsch, J.; Infanger, M.; Grosse, J.; Grimm, D. Effects and Role of Multikinase Inhibitors in Thyroid Cancer. Curr. Pharm. Des. 2016, 22, 5915–5926. [Google Scholar] [CrossRef]
- Marotta, V.; Sciammarella, C.; Capasso, M.; Testori, A.; Pivonello, C.; Chiofalo, M.G.; Gambardella, C.; Grasso, M.; Antonino, A.; Annunziata, A.; et al. Germline Polymorphisms of the VEGF Pathway Predict Recurrence in Nonadvanced Differentiated Thyroid Cancer. J. Clin. Endocrinol. Metab. 2017, 102, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Marotta, V.; Sciammarella, C.; Capasso, M.; Testori, A.; Pivonello, C.; Chiofalo, M.G.; Pivonello, R.; Pezzullo, L.; Botti, G.; Colao, A.; et al. Preliminary data of VEGF-A and VEGFR-2 polymorphisms as predictive factors of radiological response and clinical outcome in iodine-refractory differentiated thyroid cancer treated with sorafenib. Endocrine 2017, 57, 539–543. [Google Scholar] [CrossRef]
- Kish, J.K.; Chatterjee, D.; Wan, Y.; Yu, H.T.; Liassou, D.; Feinberg, B.A. Lenvatinib and Subsequent Therapy for Radioactive Iodine-Refractory Differentiated Thyroid Cancer: A Real-World Study of Clinical Effectiveness in the United States. Adv. Ther. 2020, 37, 2841–2852. [Google Scholar] [CrossRef] [PubMed]
- Eder, J.P.; Vande Woude, G.F.; Boerner, S.A.; LoRusso, P.M. Novel therapeutic inhibitors of the c-Met signaling pathway in cancer. Clin. Cancer Res. 2009, 15, 2207–2214. [Google Scholar] [CrossRef]
- Jayarangaiah, A.; Sidhu, G.; Brown, J.; Barrett-Campbell, O.; Bahtiyar, G.; Youssef, I.; Arora, S.; Skwiersky, S.; McFarlane, S.I. Therapeutic options for advanced thyroid cancer. Int. J. Clin. Endocrinol. Metab. 2019, 5, 26–34. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; de Souza, J.A.; Geyer, S.; Wirth, L.J.; Menefee, M.E.; Liu, S.V.; Shah, K.; Wright, J.; Shah, M.H. Cabozantinib as Salvage Therapy for Patients with Tyrosine Kinase Inhibitor-Refractory Differentiated Thyroid Cancer: Results of a Multicenter Phase II International Thyroid Oncology Group Trial. J. Clin. Oncol. 2017, 35, 3315–3321. [Google Scholar] [CrossRef]
- Takahashi, S.; Tahara, M.; Ito, K.; Tori, M.; Kiyota, N.; Yoshida, K.; Sakata, Y.; Yoshida, A. Safety and Effectiveness of Lenvatinib in 594 Patients with Unresectable Thyroid Cancer in an All-Case Post-Marketing Observational Study in Japan. Adv. Ther. 2020, 37, 3850–3862. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Sugino, K.; Matsuzu, K.; Masaki, C.; Akaishi, J.; Hames, K.; Tomoda, C.; Suzuki, A.; Uruno, T.; Ohkuwa, K.; et al. Rapid disease progression after discontinuation of lenvatinib in thyroid cancer. Medicine 2020, 99, e19408. [Google Scholar] [CrossRef] [PubMed]
- Aydemirli, M.D.; Kapiteijn, E.; Ferrier, K.R.M.; Ottevanger, P.B.; Links, T.P.; van der Horst-Schrivers, A.N.A.; Broekman, K.E.; Groenwold, R.H.H.; Zwaveling, J. Effectiveness and toxicity of lenvatinib in refractory thyroid cancer: Dutch real-life data. Eur. J. Endocrinol. 2020, 182, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Kiyota, N.; Robinson, B.; Shah, M.; Hoff, A.O.; Taylor, M.H.; Li, D.; Dutcus, C.E.; Lee, E.K.; Kim, S.B.; Tahara, M. Defining Radioiodine-Refractory Differentiated Thyroid Cancer: Efficacy and Safety of Lenvatinib by Radioiodine-Refractory Criteria in the SELECT Trial. Thyroid 2017, 27, 1135–1141. [Google Scholar] [CrossRef]
- Brose, M.S.; Smit, J.; Lin, C.C.; Pitoia, F.; Fellous, M.; DeSanctis, Y.; Schlumberger, M.; Tori, M.; Sugitani, I. Timing of multikinase inhibitor initiation in differentiated thyroid cancer. Endocr. Relat. Cancer 2017, 24, 237–242. [Google Scholar] [CrossRef]
- Tahara, M.; Schlumberger, M.; Elisei, R.; Habra, M.A.; Kiyota, N.; Paschke, R.; Dutcus, C.E.; Hihara, T.; McGrath, S.; Matijevic, M.; et al. Exploratory analysis of biomarkers associated with clinical outcomes from the study of lenvatinib in differentiated cancer of the thyroid. Eur. J. Cancer 2017, 75, 213–221. [Google Scholar] [CrossRef]
- Sugino, K.; Nagahama, M.; Kitagawa, W.; Ohkuwa, K.; Uruno, T.; Matsuzu, K.; Suzuki, A.; Masaki, C.; Akaishi, J.; Hames, K.Y.; et al. Clinical factors related to the efficacy of tyrosine kinase inhibitor therapy in radioactive iodine refractory recurrent differentiated thyroid cancer patients. Endocr. J. 2018, 65, 299–306. [Google Scholar] [CrossRef]
- Molina-Vega, M.; García-Alemán, J.; Sebastián-Ochoa, A.; Mancha-Doblas, I.; Trigo-Pérez, J.M.; Tinahones-Madueño, F. Tyrosine kinase inhibitors in iodine-refractory differentiated thyroid cancer: Experience in clinical practice. Endocrine 2018, 59, 395–401. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; Habra, M.A. Lenvatinib: Role in thyroid cancer and other solid tumors. Cancer Treat. Rev. 2016, 42, 47–55. [Google Scholar] [CrossRef]
- Hasskarl, J. Sorafenib. Recent Results Cancer Res. 2010, 184, 61–70. [Google Scholar] [CrossRef]
- Zimmerman, E.I.; Roberts, J.L.; Li, L.; Finkelstein, D.; Gibson, A.; Chaudhry, A.S.; Schuetz, E.G.; Rubnitz, J.E.; Inaba, H.; Baker, S.D. Ontogeny and sorafenib metabolism. Clin. Cancer Res. 2012, 18, 5788–5795. [Google Scholar] [CrossRef]
- White, P.T.; Cohen, M.S. The discovery and development of sorafenib for the treatment of thyroid cancer. Expert Opin. Drug Discov. 2015, 10, 427–439. [Google Scholar] [CrossRef]
- Lenvima, Lenvatinib. Available online: https://pro.medicin.dk/Medicin/Praeparater/7785 (accessed on 25 September 2021).
- Ancker, O.V.; Krüger, M.; Wehland, M.; Infanger, M.; Grimm, D. Multikinase Inhibitor Treatment in Thyroid Cancer. Int. J. Mol. Sci. 2019, 21, 10. [Google Scholar] [CrossRef] [PubMed]
- Cabometyx, Cabozantinib. Available online: https://pro.medicin.dk/Medicin/Praeparater/8195 (accessed on 25 September 2021).
- Tucker, N. Cabozantinib Lands Breakthrough Threapy Designation in RAI-Refractory Diffenrentiated Thyroid Cancer. Available online: https://www.targetedonc.com/view/cabozantinib-lands-breakthrough-therapy-designation-in-rai-refractory-differentiated-thyroid-cancer (accessed on 23 March 2021).
- Locati, L.D.; Piovesan, A.; Durante, C.; Bregni, M.; Castagna, M.G.; Zovato, S.; Giusti, M.; Ibrahim, T.; Puxeddu, E.; Fedele, G.; et al. Real-world efficacy and safety of lenvatinib: Data from a compassionate use in the treatment of radioactive iodine-refractory differentiated thyroid cancer patients in Italy. Eur. J. Cancer 2019, 118, 35–40. [Google Scholar] [CrossRef]
- Bair, S.M.; Choueiri, T.K.; Moslehi, J. Cardiovascular complications associated with novel angiogenesis inhibitors: Emerging evidence and evolving perspectives. Trends Cardiovasc. Med. 2013, 23, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Cicenas, E. Multi-kinase inhibitors, AURKs and cancer. Med. Oncol. 2016, 33, 43. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Yazici, Y.D.; Calzada, G.; Wang, Z.Y.; Younes, M.N.; Jasser, S.A.; El-Naggar, A.K.; Myers, J.N. Sorafenib inhibits the angiogenesis and growth of orthotopic anaplastic thyroid carcinoma xenografts in nude mice. Mol. Cancer Ther. 2007, 6, 1785–1792. [Google Scholar] [CrossRef] [PubMed]
- Gollob, J.A.; Wilhelm, S.; Carter, C.; Kelley, S.L. Role of Raf kinase in cancer: Therapeutic potential of targeting the Raf/MEK/ERK signal transduction pathway. Semin. Oncol. 2006, 33, 392–406. [Google Scholar] [CrossRef]
- Bentzien, F.; Zuzow, M.; Heald, N.; Gibson, A.; Shi, Y.; Goon, L.; Yu, P.; Engst, S.; Zhang, W.; Huang, D.; et al. In vitro and in vivo activity of cabozantinib (XL184), an inhibitor of RET, MET, and VEGFR2, in a model of medullary thyroid cancer. Thyroid 2013, 23, 1569–1577. [Google Scholar] [CrossRef]
- Brose, M.S.; Robinson, B.; Sherman, S.I.; Krajewska, J.; Lin, C.C.; Vaisman, F.; Hoff, A.O.; Hitre, E.; Bowles, D.W.; Hernando, J.; et al. Cabozantinib for radioiodine-refractory differentiated thyroid cancer (COSMIC-311): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021, 22, 1126–1138. [Google Scholar] [CrossRef]
- Worden, F.; Fassnacht, M.; Shi, Y.; Hadjieva, T.; Bonichon, F.; Gao, M.; Fugazzola, L.; Ando, Y.; Hasegawa, Y.; Park, D.J.; et al. Safety and tolerability of sorafenib in patients with radioiodine-refractory thyroid cancer. Endocr. Relat. Cancer 2015, 22, 877–887. [Google Scholar] [CrossRef]
- Brose, M.S.; Nutting, C.M.; Jarzab, B.; Elisei, R.; Siena, S.; Bastholt, L.; de la Fouchardiere, C.; Pacini, F.; Paschke, R.; Shong, Y.K.; et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: A randomised, double-blind, phase 3 trial. Lancet 2014, 384, 319–328. [Google Scholar] [CrossRef]
- Brose, M.S.; Nutting, C.M.; Sherman, S.I.; Shong, Y.K.; Smit, J.W.; Reike, G.; Chung, J.; Kalmus, J.; Kappeler, C.; Schlumberger, M. Rationale and design of decision: A double-blind, randomized, placebo-controlled phase III trial evaluating the efficacy and safety of sorafenib in patients with locally advanced or metastatic radioactive iodine (RAI)-refractory, differentiated thyroid cancer. BMC Cancer 2011, 11, 349. [Google Scholar] [CrossRef]
- Schlumberger, M.; Tahara, M.; Wirth, L.J.; Robinson, B.; Brose, M.S.; Elisei, R.; Habra, M.A.; Newbold, K.; Shah, M.H.; Hoff, A.O.; et al. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer. N. Engl. J. Med. 2015, 372, 621–630. [Google Scholar] [CrossRef]
- Taylor, M.H.; Takahashi, S.; Capdevila, J.; Tahara, M.; Leboulleux, S.; Kiyota, N.; Dutcus, C.E.; Xie, R.; Robinson, B.; Sherman, S.; et al. Correlation of Performance Status and Neutrophil-Lymphocyte Ratio with Efficacy in Radioiodine-Refractory Differentiated Thyroid Cancer Treated with Lenvatinib. Thyroid 2021, 31, 1226–1234. [Google Scholar] [CrossRef]
- Tahara, M.; Kiyota, N.; Hoff, A.O.; Badiu, C.; Owonikoko, T.K.; Dutcus, C.E.; Suzuki, T.; Ren, M.; Wirth, L.J. Impact of lung metastases on overall survival in the phase 3 SELECT study of lenvatinib in patients with radioiodine-refractory differentiated thyroid cancer. Eur. J. Cancer 2021, 147, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Tahara, M.; Brose, M.S.; Wirth, L.J.; Suzuki, T.; Miyagishi, H.; Fujino, K.; Dutcus, C.E.; Gianoukakis, A. Impact of dose interruption on the efficacy of lenvatinib in a phase 3 study in patients with radioiodine-refractory differentiated thyroid cancer. Eur. J. Cancer 2019, 106, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Robinson, B.; Schlumberger, M.; Wirth, L.J.; Dutcus, C.E.; Song, J.; Taylor, M.H.; Kim, S.B.; Krzyzanowska, M.K.; Capdevila, J.; Sherman, S.I.; et al. Characterization of Tumor Size Changes Over Time From the Phase 3 Study of Lenvatinib in Thyroid Cancer. J. Clin. Endocrinol. Metab. 2016, 101, 4103–4109. [Google Scholar] [CrossRef] [PubMed]
- Kiyota, N.; Schlumberger, M.; Muro, K.; Ando, Y.; Takahashi, S.; Kawai, Y.; Wirth, L.; Robinson, B.; Sherman, S.; Suzuki, T.; et al. Subgroup analysis of Japanese patients in a phase 3 study of lenvatinib in radioiodine-refractory differentiated thyroid cancer. Cancer Sci. 2015, 106, 1714–1721. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- PubMed.gov. Available online: https://pubmed.ncbi.nlm.nih.gov (accessed on 2 November 2021).
- Clinicaltrial.gov. Available online: https://clinicaltrials.gov (accessed on 2 November 2021).
- Pandey, A.K.; Singhi, E.K.; Arroyo, J.P.; Ikizler, T.A.; Gould, E.R.; Brown, J.; Beckman, J.A.; Harrison, D.G.; Moslehi, J. Mechanisms of VEGF (Vascular Endothelial Growth Factor) Inhibitor-Associated Hypertension and Vascular Disease. Hypertension 2018, 71, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Wasserstrum, Y.; Kornowski, R.; Raanani, P.; Leader, A.; Pasvolsky, O.; Iakobishvili, Z. Hypertension in cancer patients treated with anti-angiogenic based regimens. Cardiooncology 2015, 1, 6. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; Ryder, M.; Jimenez, C. Targeted Therapy for Advanced Thyroid Cancer: Kinase Inhibitors and Beyond. Endocr. Rev. 2019, 40, 1573–1604. [Google Scholar] [CrossRef] [PubMed]
- Yakes, F.M.; Chen, J.; Tan, J.; Yamaguchi, K.; Shi, Y.; Yu, P.; Qian, F.; Chu, F.; Bentzien, F.; Cancilla, B.; et al. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol. Cancer Ther. 2011, 10, 2298–2308. [Google Scholar] [CrossRef]
- Sueta, D.; Suyama, K.; Sueta, A.; Tabata, N.; Yamashita, T.; Tomiguchi, M.; Takeshita, T.; Yamamoto-Ibusuki, M.; Yamamoto, E.; Izumiya, Y.; et al. Lenvatinib, an oral multi-kinases inhibitor, -associated hypertension: Potential role of vascular endothelial dysfunction. Atherosclerosis 2017, 260, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Reed, N.; Glen, H.; Gerrard, G.; Good, J.; Lei, M.; Lyon, A.R.; Strachan, M.; Wadsley, J.; Newbold, K. Expert Consensus on the Management of Adverse Events During Treatment with Lenvatinib for Thyroid Cancer. Clin. Oncol. 2020, 32, e145–e153. [Google Scholar] [CrossRef] [PubMed]
- Dienstmann, R.; Braña, I.; Rodon, J.; Tabernero, J. Toxicity as a biomarker of efficacy of molecular targeted therapies: Focus on EGFR and VEGF inhibiting anticancer drugs. Oncologist 2011, 16, 1729–1740. [Google Scholar] [CrossRef]
- Small, H.Y.; Montezano, A.C.; Rios, F.J.; Savoia, C.; Touyz, R.M. Hypertension due to antiangiogenic cancer therapy with vascular endothelial growth factor inhibitors: Understanding and managing a new syndrome. Can. J. Cardiol. 2014, 30, 534–543. [Google Scholar] [CrossRef]
- de Jesus-Gonzalez, N.; Robinson, E.; Moslehi, J.; Humphreys, B.D. Management of antiangiogenic therapy-induced hypertension. Hypertension 2012, 60, 607–615. [Google Scholar] [CrossRef]
- Bæk Møller, N.; Budolfsen, C.; Grimm, D.; Krüger, M.; Infanger, M.; Wehland, M.; Magnusson, N.E. Drug-Induced Hypertension Caused by Multikinase Inhibitors (Sorafenib, Sunitinib, Lenvatinib and Axitinib) in Renal Cell Carcinoma Treatment. Int. J. Mol. Sci. 2019, 20, 4712. [Google Scholar] [CrossRef]
- Institute, N.c. Common Terminology Criteria for Adverse Events (CTCAE). Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5x11.pdf (accessed on 12 August 2021).
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 Practice Guidelines for the management of arterial hypertension of the European Society of Hypertension and the European Society of Cardiology: ESH/ESC Task Force for the Management of Arterial Hypertension. J. Hypertens. 2018, 36, 2284–2309. [Google Scholar] [CrossRef] [PubMed]
- Elliott, W.J. Systemic hypertension. Curr. Probl. Cardiol. 2007, 32, 201–259. [Google Scholar] [CrossRef] [PubMed]
- Capdevila, J.; Newbold, K.; Licitra, L.; Popovtzer, A.; Moreso, F.; Zamorano, J.; Kreissl, M.; Aller, J.; Grande, E. Optimisation of treatment with lenvatinib in radioactive iodine-refractory differentiated thyroid cancer. Cancer Treat. Rev. 2018, 69, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Nervo, A.; Gallo, M.; Samà, M.T.; Felicetti, F.; Alfano, M.; Migliore, E.; Marchisio, F.; Berardelli, R.; Arvat, E.; Piovesan, A. Lenvatinib in Advanced Radioiodine-refractory Thyroid Cancer: A Snapshot of Real-life Clinical Practice. Anticancer. Res. 2018, 38, 1643–1649. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Kiyota, N.; Tahara, M. Optimal use of lenvatinib in the treatment of advanced thyroid cancer. Cancers Head Neck 2017, 2, 7. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; Schlumberger, M.; Jarzab, B.; Martins, R.G.; Pacini, F.; Robinson, B.; McCaffrey, J.C.; Shah, M.H.; Bodenner, D.L.; Topliss, D.; et al. A phase 2 trial of lenvatinib (E7080) in advanced, progressive, radioiodine-refractory, differentiated thyroid cancer: A clinical outcomes and biomarker assessment. Cancer 2015, 121, 2749–2756. [Google Scholar] [CrossRef]
- Fierro-Maya, L.F.; González, G.G.; Melo, L.J.R.; Cuéllar, A.A.C.; Carreño, A.; Córdoba, C. Safety and efficacy of sorafenib in patients with advanced thyroid carcinoma: A phase II study (NCT02084732). Arch. Endocrinol. Metab. 2021, 65, 404–410. [Google Scholar] [CrossRef]
- Hoftijzer, H.; Heemstra, K.A.; Morreau, H.; Stokkel, M.P.; Corssmit, E.P.; Gelderblom, H.; Weijers, K.; Pereira, A.M.; Huijberts, M.; Kapiteijn, E.; et al. Beneficial effects of sorafenib on tumor progression, but not on radioiodine uptake, in patients with differentiated thyroid carcinoma. Eur. J. Endocrinol. 2009, 161, 923–931. [Google Scholar] [CrossRef]
- Brose, M.S.; Schlumberger, M.; Peña, C.; Kappeler, C. Sorafenib for patients with differentiated thyroid cancer—authors’ reply. Lancet 2015, 385, 228–229. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; Takahashi, S. Managing the adverse events associated with lenvatinib therapy in radioiodine-refractory differentiated thyroid cancer. Semin. Oncol. 2019, 46, 57–64. [Google Scholar] [CrossRef]
- Ptinopoulou, A.G.; Sprangers, B. Tyrosine kinase inhibitor-induced hypertension-marker of anti-tumour treatment efficacy or cardiovascular risk factor? Clin. Kidney J. 2020, 14, 14–17. [Google Scholar] [CrossRef] [PubMed]
- Budolfsen, C.; Faber, J.; Grimm, D.; Krüger, M.; Bauer, J.; Wehland, M.; Infanger, M.; Magnusson, N.E. Tyrosine Kinase Inhibitor-Induced Hypertension: Role of Hypertension as a Biomarker in Cancer Treatment. Curr. Vasc. Pharmacol. 2019, 17, 618–634. [Google Scholar] [CrossRef] [PubMed]
- Izzedine, H.; Ederhy, S.; Goldwasser, F.; Soria, J.C.; Milano, G.; Cohen, A.; Khayat, D.; Spano, J.P. Management of hypertension in angiogenesis inhibitor-treated patients. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2009, 20, 807–815. [Google Scholar] [CrossRef]
- Laffin, L.J.; Bakris, G.L. Endothelin antagonism and hypertension: An evolving target. Semin. Nephrol. 2015, 35, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M.; Lang, N.N.; Herrmann, J.; van den Meiracker, A.H.; Danser, A.H.J. Recent Advances in Hypertension and Cardiovascular Toxicities with Vascular Endothelial Growth Factor Inhibition. Hypertension 2017, 70, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Iglesias, L.; Klain, M.; Pitoia, F.; Schlumberger, M.J. Radioactive iodine-refractory differentiated thyroid cancer: An uncommon but challenging situation. Arch. Endocrinol. Metab. 2017, 61, 81–89. [Google Scholar] [CrossRef]
- Krajewska, J.; Kukulska, A.; Jarzab, B. Efficacy of lenvatinib in treating thyroid cancer. Expert Opin. Pharmacother. 2016, 17, 1683–1691. [Google Scholar] [CrossRef]
- Kreissl, M.C.; Janssen, M.J.R.; Nagarajah, J. Current Treatment Strategies in Metastasized Differentiated Thyroid Cancer. J. Nucl. Med. 2019, 60, 9–15. [Google Scholar] [CrossRef]
- De Leo, S.; Di Stefano, M.; Persani, L.; Fugazzola, L.; Colombo, C. Lenvatinib as first-line treatment for advanced thyroid cancer: Long progression-free survival. Endocrine 2021, 72, 462–469. [Google Scholar] [CrossRef]
- Berdelou, A.; Borget, I.; Godbert, Y.; Nguyen, T.; Garcia, M.E.; Chougnet, C.N.; Ferru, A.; Buffet, C.; Chabre, O.; Huillard, O.; et al. Lenvatinib for the Treatment of Radioiodine-Refractory Thyroid Cancer in Real-Life Practice. Thyroid 2018, 28, 72–78. [Google Scholar] [CrossRef]
- de la Fouchardiere, C.; Alghuzlan, A.; Bardet, S.; Borget, I.; Borson Chazot, F.; Do Cao, C.; Godbert, Y.; Leenhardt, L.; Zerdoud, S.; Leboulleux, S. The medical treatment of radioiodine-refractory differentiated thyroid cancers in 2019. A TUTHYREF(®) network review. Bull Cancer 2019, 106, 812–819. [Google Scholar] [CrossRef]
- Song, E.; Kim, M.; Kim, E.Y.; Kim, B.H.; Shin, D.Y.; Kang, H.C.; Ahn, B.C.; Kim, W.B.; Shong, Y.K.; Jeon, M.J.; et al. Lenvatinib for Radioactive Iodine-Refractory Differentiated Thyroid Carcinoma and Candidate Biomarkers Associated with Survival: A Multicenter Study in Korea. Thyroid 2020, 30, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.K.; Kim, S.M.; Kim, B.H.; Kim, M.J.; Lim, D.J.; Kim, M.H.; Shin, D.Y.; Kang, H.C.; Ahn, B.C.; Kim, S.W.; et al. Lesion-Based Evaluation Predicts Treatment Response to Lenvatinib for Radioactive Iodine-Refractory Differentiated Thyroid Cancer: A Korean Multicenter Retrospective Study. Thyroid 2019, 29, 1811–1819. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Wang, X.; Ohmoto, A.; Urasaki, T.; Sato, Y.; Nakano, K.; Nishizawa, M.; Yunokawa, M.; Ono, M.; Tomomatsu, J.; et al. Sequential Analysis of Neutrophil-to-lymphocyte Ratio for Differentiated Thyroid Cancer Patients Treated with Lenvatinib. In Vivo 2020, 34, 709–714. [Google Scholar] [CrossRef]
- Ito, Y.; Onoda, N.; Kudo, T.; Masuoka, H.; Higashiyama, T.; Kihara, M.; Miya, A.; Miyauchi, A. Sorafenib and Lenvatinib Treatment for Metastasis/Recurrence of Radioactive Iodine-refractory Differentiated Thyroid Carcinoma. In Vivo 2021, 35, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Werner, R.A.; Lückerath, K.; Schmid, J.S.; Higuchi, T.; Kreissl, M.C.; Grelle, I.; Reiners, C.; Buck, A.K.; Lapa, C. Thyroglobulin fluctuations in patients with iodine-refractory differentiated thyroid carcinoma on lenvatinib treatment—Initial experience. Sci. Rep. 2016, 6, 28081. [Google Scholar] [CrossRef] [PubMed]
- Brose, M.S.; Worden, F.P.; Newbold, K.L.; Guo, M.; Hurria, A. Effect of Age on the Efficacy and Safety of Lenvatinib in Radioiodine-Refractory Differentiated Thyroid Cancer in the Phase III SELECT Trial. J. Clin. Oncol. 2017, 35, 2692–2699. [Google Scholar] [CrossRef]
- Suzuki, C.; Kiyota, N.; Imamura, Y.; Goto, H.; Suto, H.; Chayahara, N.; Toyoda, M.; Ito, Y.; Miya, A.; Miyauchi, A.; et al. Exploratory analysis of prognostic factors for lenvatinib in radioiodine-refractory differentiated thyroid cancer. Head Neck 2019, 41, 3023–3032. [Google Scholar] [CrossRef]
- Jerkovich, F.; García Falcone, M.G.; Pitoia, F. The experience of an Endocrinology Division on the use of tyrosine multikinase inhibitor therapy in patients with radioiodine-resistant differentiated thyroid cancer. Endocrine 2019, 64, 632–638. [Google Scholar] [CrossRef]
- Balmelli, C.; Railic, N.; Siano, M.; Feuerlein, K.; Cathomas, R.; Cristina, V.; Güthner, C.; Zimmermann, S.; Weidner, S.; Pless, M.; et al. Lenvatinib in Advanced Radioiodine-Refractory Thyroid Cancer—A Retrospective Analysis of the Swiss Lenvatinib Named Patient Program. J. Cancer 2018, 9, 250–255. [Google Scholar] [CrossRef]
- Masaki, C.; Sugino, K.; Saito, N.; Akaishi, J.; Hames, K.Y.; Tomoda, C.; Suzuki, A.; Matsuzu, K.; Uruno, T.; Ohkuwa, K.; et al. Efficacy and Limitations of Lenvatinib Therapy for Radioiodine-Refractory Differentiated Thyroid Cancer: Real-World Experiences. Thyroid 2020, 30, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Jerkovich, F.; Califano, I.; Bueno, F.; Carrera, J.M.; Giglio, R.; Abelleira, E.; Pitoia, F. Real-life use of lenvatinib in patients with differentiated thyroid cancer: Experience from Argentina. Endocrine 2020, 69, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Gianoukakis, A.G.; Dutcus, C.E.; Batty, N.; Guo, M.; Baig, M. Prolonged duration of response in lenvatinib responders with thyroid cancer. Endocr. Relat. Cancer 2018, 25, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.S.; Khankin, E.V.; Karumanchi, S.A.; Humphreys, B.D. Hypertension induced by vascular endothelial growth factor signaling pathway inhibition: Mechanisms and potential use as a biomarker. Semin. Nephrol. 2010, 30, 591–601. [Google Scholar] [CrossRef]
- Tahara, M.; Takami, H.; Ito, Y.; Sugino, K.; Takahashi, S.; Takeyama, H.; Tsutsui, H.; Hara, H.; Mitsuma, A.; Yamashita, H.; et al. Cohort study exploring the effect of lenvatinib on differentiated thyroid cancer. Endocr. J. 2018, 65, 1071–1074. [Google Scholar] [CrossRef]
- Jasim, S.; Iniguez-Ariza, N.M.; Hilger, C.R.; Chintakuntlawar, A.V.; Ryder, M.M.; Morris, J.C., 3rd; Bible, K.C. Optimizing lenvatinib therapy in patients with metastatic radioactive iodine-resistant differentiated thyroid cancers. Endocr. Pract. 2017, 23, 1254–1261. [Google Scholar] [CrossRef]
- Suyama, K.; Tomiguchi, M.; Takeshita, T.; Sueta, A.; Yamamoto-Ibusuki, M.; Shimokawa, M.; Yamamoto, Y.; Iwase, H. Factors involved in early lenvatinib dose reduction: A retrospective analysis. Med. Oncol. 2018, 35, 19. [Google Scholar] [CrossRef]
- Haddad, R.I.; Schlumberger, M.; Wirth, L.J.; Sherman, E.J.; Shah, M.H.; Robinson, B.; Dutcus, C.E.; Teng, A.; Gianoukakis, A.G.; Sherman, S.I. Incidence and timing of common adverse events in Lenvatinib-treated patients from the SELECT trial and their association with survival outcomes. Endocrine 2017, 56, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Porcelli, T.; Luongo, C.; Sessa, F.; Klain, M.; Masone, S.; Troncone, G.; Bellevicine, C.; Schlumberger, M.; Salvatore, D. Long-term management of lenvatinib-treated thyroid cancer patients: A real-life experience at a single institution. Endocrine 2021, 73, 358–366. [Google Scholar] [CrossRef]
- Agarwal, M.; Thareja, N.; Benjamin, M.; Akhondi, A.; Mitchell, G.D. Tyrosine Kinase Inhibitor-Induced Hypertension. Curr. Oncol. Rep. 2018, 20, 65. [Google Scholar] [CrossRef]
- Oh, H.S.; Shin, D.Y.; Kim, M.; Park, S.Y.; Kim, T.H.; Kim, B.H.; Kim, E.Y.; Kim, W.B.; Chung, J.H.; Shong, Y.K.; et al. Extended Real-World Observation of Patients Treated with Sorafenib for Radioactive Iodine-Refractory Differentiated Thyroid Carcinoma and Impact of Lenvatinib Salvage Treatment: A Korean Multicenter Study. Thyroid 2019, 29, 1804–1810. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Iwasaki, H.; Takasaki, H.; Suganuma, N.; Sakai, R.; Masudo, K.; Nakayama, H.; Rino, Y.; Masuda, M. Efficacy and tolerability of initial low-dose lenvatinib to treat differentiated thyroid cancer. Medicine 2019, 98, e14774. [Google Scholar] [CrossRef] [PubMed]
- Keizer, R.J.; Gupta, A.; Mac Gillavry, M.R.; Jansen, M.; Wanders, J.; Beijnen, J.H.; Schellens, J.H.; Karlsson, M.O.; Huitema, A.D. A model of hypertension and proteinuria in cancer patients treated with the anti-angiogenic drug E7080. J. Pharmacokinet. Pharmacodyn. 2010, 37, 347–363. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Masaki, C.; Sugino, K.; Saito, N.; Saito, Y.; Tanaka, T.; Ogimi, Y.; Maeda, T.; Osaku, T.; Akaishi, J.; Hames, K.Y.; et al. Lenvatinib induces early tumor shrinkage in patients with advanced thyroid carcinoma. Endocr. J. 2017, 64, 819–826. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Markowitz, J.N.; Fancher, K.M. Cabozantinib: A Multitargeted Oral Tyrosine Kinase Inhibitor. Pharmacotherapy 2018, 38, 357–369. [Google Scholar] [CrossRef]
- Zheng, X.; Xu, Z.; Ji, Q.; Ge, M.; Shi, F.; Qin, J.; Wang, F.; Chen, G.; Zhang, Y.; Huang, R.; et al. A Randomized, Phase III Study of Lenvatinib in Chinese Patients with Radioiodine-Refractory Differentiated Thyroid Cancer. Clin. Cancer Res. 2021, 27, 5502. [Google Scholar] [CrossRef]
- Kim, S.Y.; Kim, S.M.; Chang, H.; Kim, B.W.; Lee, Y.S.; Chang, H.S.; Park, C.S. Safety of Tyrosine Kinase Inhibitors in Patients with Differentiated Thyroid Cancer: Real-World Use of Lenvatinib and Sorafenib in Korea. Front. Endocrinol. 2019, 10, 384. [Google Scholar] [CrossRef]
- Schlumberger, M.; Elisei, R.; Müller, S.; Schöffski, P.; Brose, M.; Shah, M.; Licitra, L.; Krajewska, J.; Kreissl, M.C.; Niederle, B.; et al. Overall survival analysis of EXAM, a phase III trial of cabozantinib in patients with radiographically progressive medullary thyroid carcinoma. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2017, 28, 2813–2819. [Google Scholar] [CrossRef]
- Elisei, R.; Schlumberger, M.J.; Müller, S.P.; Schöffski, P.; Brose, M.S.; Shah, M.H.; Licitra, L.; Jarzab, B.; Medvedev, V.; Kreissl, M.C.; et al. Cabozantinib in progressive medullary thyroid cancer. J. Clin. Oncol. 2013, 31, 3639–3646. [Google Scholar] [CrossRef]
- Verburg, F.A.; Amthauer, H.; Binse, I.; Brink, I.; Buck, A.; Darr, A.; Dierks, C.; Koch, C.; König, U.; Kreissl, M.C.; et al. Questions and Controversies in the Clinical Application of Tyrosine Kinase Inhibitors to Treat Patients with Radioiodine-Refractory Differentiated Thyroid Carcinoma: Expert Perspectives. Horm. Metab. Res. Horm. Stoffwechs. Horm. Metab. 2021, 53, 149–160. [Google Scholar] [CrossRef]

| Drug | Target | Half-Life in Plasma | Metabolism | Approval for DTC | Structure |
|---|---|---|---|---|---|
| Sorafenib | VEGFR-2 and -3 PDGFR, c-Kit, RET/PTC, RAF [41] | 36 h [41] | Hepatic CYP3A4 and UGT1A9 [42] | 2013 [43] | 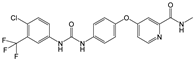 |
| Lenvatinib | , RET, c-KIT, FGFR 1-4 [3] | 28 h [44] | Hepatic CYP3A4 [45] | 2015 [28] | 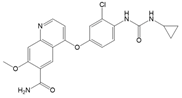 |
| Cabozantinib | VEGFR-2, c-MET, RET [31] | 100–120 h [46] | Hepatic CYP3A4 [46] | 2021 [47] | 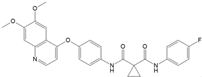 |
| Title | Design | Number of Participants | Doses and Cycles | Results | Status |
|---|---|---|---|---|---|
| A Study of Cabozantinib Compared with Placebo in Subjects with Radioiodine-refractory Differentiated Thyroid Cancer Who Have Progressed After Prior VEGFR-targeted Therapy (COSMIC-311 Trial) NCT03690388 [54] | Interventional, randomized, double blind | Planned 300 Enrolled187 | 60 mg or 20 mg cabozantinib or placebo equivalent once daily | PFS vs. placebo: median not reached (96% CI 5.7–not estimable) versus 1.9 months (1.8–3.6); hazard ratio 0.22 (96% CI 0.13–0.36; p < 0.0001) | Active, not recruiting |
| A Double-Blind Randomized Phase III Study Evaluating the Efficacy and Safety of Sorafenib Compared to Placebo in Locally Advanced/Metastatic RAI-Refractory Differentiated Thyroid Cancer (DECISISON) NCT0098428 [55,56,57] | Interventional, double blind, randomized | 417 | Sorafenib 800 mg/day (400 mg every 12 h) Placebo twice daily (approximately every 12 h). | PFS vs. placebo: hazard ratio, 0.59 (95% CI 0.45–0.76); p < 0.0001; median 10.8 vs. 5.8 months | Completed |
| A Multicenter, Randomized, Double-Blind, Placebo-Controlled Trial of Lenvatinib (E7080) in 131I-Refractory Differentiated Thyroid Cancer (SELECT) NCT01321554 [37,58,59,60,61,62,63] | Interventional, double blind, randomized | 392 | Randomization phase: Starting dose—24 mg Lenvatinib orally (two 10 mg tablets and one 4 mg tablet) once daily, continuously, or placebo matching the lenvatinib treatment. | PFS Treatment: 18.3 months Placebo: 3.6 months HR: 0.21 (99% CI: 0.14 to 0.31) ORR Treatment: 64.8% Placebo: 1.5%. | Completed |
| Condition | Systolic Blood Pressure (mmHg) | Diastolic Blood Pressure (mmHg) | |
|---|---|---|---|
| Optimal | <120 | and | <80 |
| Normal | 120–129 | and/or | 80–84 |
| High normal | 130–139 | and/or | 85–89 |
| Grade 1 hypertension | 140–159 | and/or | 90–99 |
| Grade 2 hypertension | 160–179 | and/or | 100–109 |
| Grade 3 hypertension | ≥180 | and/or | ≥110 |
| Isolated systolic hypertension | ≥140 | and | <90 |
| Title | Daily Dose | Most Frequent AEs (Not Including Serious) | Most Frequent Serious AEs |
|---|---|---|---|
| Evaluating the Safety and Efficacy of Oral Lenvatinib in Medullary and Iodine-131 Refractory, Unresectable Differentiated Thyroid Cancers, Stratified by Histology NCT00784303 [83] | 24 mg lenvatinib | Hypertension 77.59% Diarrhea 68.97% Fatigue 60.34% Decreased appetite 55.17% Nausea 51.72% | Hypotension 6.9% Dehydration 6.9% Hypertension 3.45% Renal failure 3.45 Pneumonia 3.45% |
| A Trial of Lenvatinib (E7080) in Subjects with Iodine-131 Refractory Differentiated Thyroid Cancer to Evaluate Whether an Oral Starting Dose of 18 Milligram (mg) Daily Will Provide Comparable Efficacy to a 24 mg Starting Dose, But Have a Better Safety Profile NCT02702388 | 24 mg or 18 mg lenvatinib (starting dose) | 24 mg: Hypertension 57.33% Diarrhea 56% Proteinuria 42.67% Nausea 40.00% Weight loss 36.00% 18 mg: Hypertension 51.95% Diarrhea 51.95% Weight loss 42.86% Stomatitis 38.57% Nausea 35.06% | 24 mg: Total 33.33% Malignant neoplasm progression 4% Pneumothorax 2.67% Hypertension 1.33% 18 mg: Total 40.26% Osteoarthritis 2.60% Pathological fracture 2.60% Malignant neoplasm progression 2.60% Malignant pleural effusion 2.60% |
| A Multicenter, Randomized, Double-Blind, Placebo-Controlled, Trial of Lenvatinib (E7080) in 131I-Refractory Differentiated Thyroid Cancer (DTC) (SELECT) NCT01321554 [58] | 24 mg lenvatinib Placebo matching the lenvatinib treatment. | Hypertension 69.35% Diarrhea 69.73% Loss of appetite 56.70% Weight loss 54.41% Nausea 48.66% | Pneumonia 4.6% Hypertension 3.83 Dehydration 3.45% General physical health deterioration 2.68% Pulmonary embolism 2.3% |
| A Study of E7080 in Subjects with Advanced Thyroid Cancer NCT01728623 | 24 mg lenvatinib | Hypertension 90.20% Palmar-plantar erythrodysaesthesia syndrome 76.47% Loss of appetite 76.47% Fatigue 72.55% Proteinuria 60.78% | Decreased appetite 13.73% Malignant neoplasm progression 7.84% Pneumonia 5.88% Nausea 3.92% Laryngeal stenosis 3.92% |
| Cabozantinib-S-Malate in Treating Patients with Refractory Thyroid Cancer NCT01811212 [31] | 60 mg cabozantinib S-malate | Liver transaminase elevation 80% Palmar-plantar erythrodysesthesia 76% Fatigue 76% Diarrhea 72% Nausea 64% … Hypertension 48% | Left ventricular systolic dysfunction 4% Osteonecrosis of the jaw 4% Asymptomatic increased lipase 4% Meningitis 4% Pneumonia 4% |
| A Study of Cabozantinib Compared with Placebo in Subjects with Radioiodine-refractory Differentiated Thyroid Cancer Who Have Progressed After Prior VEGFR-targeted Therapy (COSMIC-311 Trial) NCT03690388 [54] | 60 mg or 20 mg cabozantinib or placebo equivalent once daily | Diarrhea 44% Palmar-plantar erythrodysesthesia 35% Alanine aminotransferase increased 23% Aspartate aminotransferase increased 23% Nausea 21% … Hypertension 19% | Palmar-plantar erythrodysesthesia 10% Hypertension 9% Fatigue 8% Diarrhoea 7% Hypocalcaemia 7% |
| Safety and Efficacy of Sorafenib in Patients with Advanced Thyroid Cancer: a Phase II Clinical Study NCT02084732 [84] | 800 mg/day sorafenib | Hypertension 42.1% Hand/food Erythema 36.8% Diarrhea 31.5% Muscle pain 21% Rash 21% | Hypertension 56,8% Hand/food Erythema 31.5% Diarrhea 26.2% Rash 15.7% Acute myocardial infarction 5.2% |
| Sorafenib as Adjuvant to Radioiodine Therapy in Non-Medullary Thyroid Carcinoma NCT00887107 [85] | 800 mg/day sorafenib | Weight loss 47.6% Diarrhea 50% Alopecia 47% Rash 47% Hand foot syndrome 43.6% … Hypertension 25.4% | Hand foot syndrome 21.8% Hypertension 15% Weight loss 8.4% Myocardial infarction 3% Congestive heart disease 3% |
| A Double-Blind Randomized Phase III Study Evaluating the Efficacy and Safety of Sorafenib Compared to Placebo in Locally Advanced/Metastatic RAI-Refractory Differentiated Thyroid Cancer (DECISISON) NCT00984282 [56] | 800 mg/day sorafenib | Alopecia 67.1% Diarrhea 62.8% Hand–foot skin reaction 56% Rash 45.4% Fatigue 44% Hypertension 30.9% | Hand–foot skin reaction 20.3% Hypertension 9.7% Hypocalcemia 9.2 Diarrhea 5.8% Fatigue 5.8% |
| Drug | TE-HTN Grades 1–2 (%) | TE-HTN ≥ Grade 3 (%) | Dose Reductions (%) | Dose Interruptions (%) | Discontinuations (%) | TE-HTN-Related Deaths (%) |
|---|---|---|---|---|---|---|
| Lenvatinib [17,58] | 26 | 41.8 | 13 | 13 | 1 | 0 |
| Sorafenib [55,56] | 30.9 | 9.7 | 5.8 | 7.7 | 0.5 | 0 |
| Cabozantinib [54] | 19 | 9 | <7 | n/a | 0.8 | 0 |
| BP Level (mmHg) | Grade of AE | Half-life in plasma |
|---|---|---|
| SBP < 140, DBP < 90 | Grade 1 AE | No treatment needed |
| SBP 140–159, DBP 90–99 | Grade 2 AE | Need for antihypertensive medication |
| SBP ≥ 160, DBP ≥ 100 | Grade 3 AE | Need for antihypertensive medication If persistent for 3 consecutive months, dose reductions are required |
| Life-threatening | Grade 4 AE | Urgent intervention is indicated, possibly treatment interruption |
| Drug | Clinical Trial | Dose Level | Daily Dose | References |
|---|---|---|---|---|
| Lenvatinib (Lenvima®) | SELECT (NCT01321554) | Recommended daily dose | 24 mg p.o. once daily: 2 × 10 mg and 1 × 4 mg in capsules | [44,58,72] |
| Dose reduction No. 1 | 20 mg p.o. once daily: 2 × 10 mg in capsules | |||
| Dose reduction No. 2 | 14 mg orally once daily (1 × 10 mg capsule plus 1 × 4 mg capsule) | |||
| Dose reduction No. 3 | 10 mg p.o. once daily: 1 × 10 mg capsule | |||
| Lenvatinib (Lenvima®) | Lenvatinib in Chinese patients with RR-DTC | Starting dose | 24 mg/day p.o. | [123] |
| In case of intolerable grade 2 or grade 3 HTN; lenvatinib treatment interruption until the toxicity had resolved to grade ≤1 or baseline. | ||||
| Lenvatinib treatment was then resumed at a reduced dose: | ||||
| Dose reduction No. 1 | 20 mg/day | |||
| Dose reduction No. 2 | 14 mg/day | |||
| Dose reduction No. 3 | 10 mg/day | |||
| Lenvatinib (Lenvima®) Sorafenib (NEXAVAR®) | Lenvatinib and Sorafenib in South Korean patients with advanced and metastatic RR-DTC | Lenvatinib arm: starting dose | 20 mg/day capsules | [124] |
| Sorafenib arm: starting dose | 800 mg/day tablets | |||
| Final lenvatinib doses: | ||||
| N = 7 (30.4%) | 10 mg daily | |||
| N = 2 (8.7%) | 14 mg daily | |||
| N = 14 (60.9%) | 20 mg daily | |||
| Dose reduction for AEs: | ||||
| N = 8 (34.8%) | ||||
| Discontinuation of lenvatinib: | ||||
| AEs | ||||
| N = 1 (4.3%) | ||||
| Disease progression: | ||||
| N = 0 | ||||
| Death: | ||||
| N = 2 (8.7%) Financial issues: | ||||
| N = 1 (4.3%) | ||||
| Initial sorafenib dose: | ||||
| N = 12 (25.0) | ≤400 mg dail | |||
| N = 16 (33.3) | 600 mg daily | |||
| N = 20 (41.7) | 800 mg daily | |||
| Final sorafenib dose: | ||||
| N = 20 (41.7) | ≤400 mg daily | |||
| N = 21 (43.8) | 600 mg daily | |||
| N = 7 (14.6) | 800 mg daily | |||
| Dose reduction for AEs: | ||||
| N = 27 (56.3) | ||||
| Discontinuation of sorafenib: | ||||
| AE | ||||
| N = 4 (8.3) | ||||
| Disease progression: | ||||
| N = 17 (35.4) | ||||
| Death: | ||||
| N = 5 (10.4) | ||||
| Sorafenib (NEXAVAR®) | DECISION (NCT00984282) | Recommended daily dose | 2 × 400 mg p.o. | [56] |
| Study drug dose interruption or sequential reduction No. 1 | 600 mg (divided doses: 400 and 200) p.o. daily | |||
| Dose reduction No. 2 | 400 mg (divided 2 × 200) p.o. daily | |||
| Dose reduction No. 3 | 200 mg daily | |||
| Cabozantinib | COSMIC-311 (NCT03690388) | Cabozantinib | [54] | |
| AEs managed by dose modifications: | 60 mg/day tablets p.o. | |||
| The median daily dose was 42.0 mg (IQR 32.2–54.5) with cabozantinib and 60.0 mg (52.9–60.0) with placebo. | Reductions from 60 mg to 40 mg and then to 20 mg daily | |||
| Cabozantinib | EXAM (NCT00704730) MTC (medullary thyroid carcinoma) patients | Cabozantinib starting dose: | 140 mg/day | [125,126] |
| AEs managed with concomitant medications, dose interruptions, and dose reductions; | ||||
| SAEs were more frequent in cabozantinib- versus placebo-treated patients. For hypertension: 2.3% (5 of 214 cabozantinib) vs. 0% (0 of 109 placebo). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaae, A.C.; Kreissl, M.C.; Krüger, M.; Infanger, M.; Grimm, D.; Wehland, M. Kinase-Inhibitors in Iodine-Refractory Differentiated Thyroid Cancer—Focus on Occurrence, Mechanisms, and Management of Treatment-Related Hypertension. Int. J. Mol. Sci. 2021, 22, 12217. https://doi.org/10.3390/ijms222212217
Kaae AC, Kreissl MC, Krüger M, Infanger M, Grimm D, Wehland M. Kinase-Inhibitors in Iodine-Refractory Differentiated Thyroid Cancer—Focus on Occurrence, Mechanisms, and Management of Treatment-Related Hypertension. International Journal of Molecular Sciences. 2021; 22(22):12217. https://doi.org/10.3390/ijms222212217
Chicago/Turabian StyleKaae, Anne Christine, Michael C. Kreissl, Marcus Krüger, Manfred Infanger, Daniela Grimm, and Markus Wehland. 2021. "Kinase-Inhibitors in Iodine-Refractory Differentiated Thyroid Cancer—Focus on Occurrence, Mechanisms, and Management of Treatment-Related Hypertension" International Journal of Molecular Sciences 22, no. 22: 12217. https://doi.org/10.3390/ijms222212217
APA StyleKaae, A. C., Kreissl, M. C., Krüger, M., Infanger, M., Grimm, D., & Wehland, M. (2021). Kinase-Inhibitors in Iodine-Refractory Differentiated Thyroid Cancer—Focus on Occurrence, Mechanisms, and Management of Treatment-Related Hypertension. International Journal of Molecular Sciences, 22(22), 12217. https://doi.org/10.3390/ijms222212217









