Design of High-Relaxivity Polyelectrolyte Nanocapsules Based on Citrate Complexes of Gadolinium(III) of Unusual Composition
Abstract
:1. Introduction
2. Results and Discussion
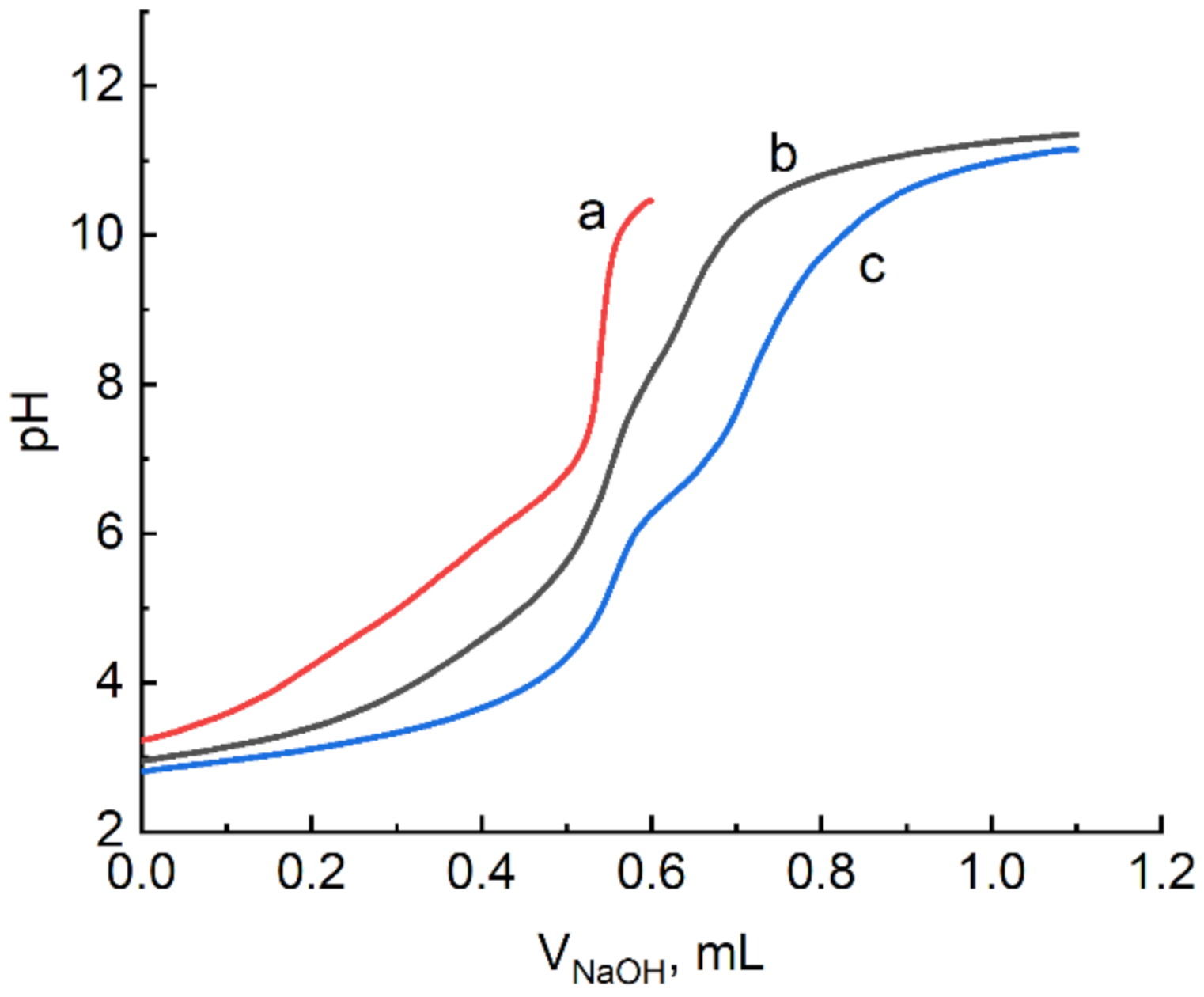
2.1. Formation of Gadolinium(III) Citrate Complexes in Water
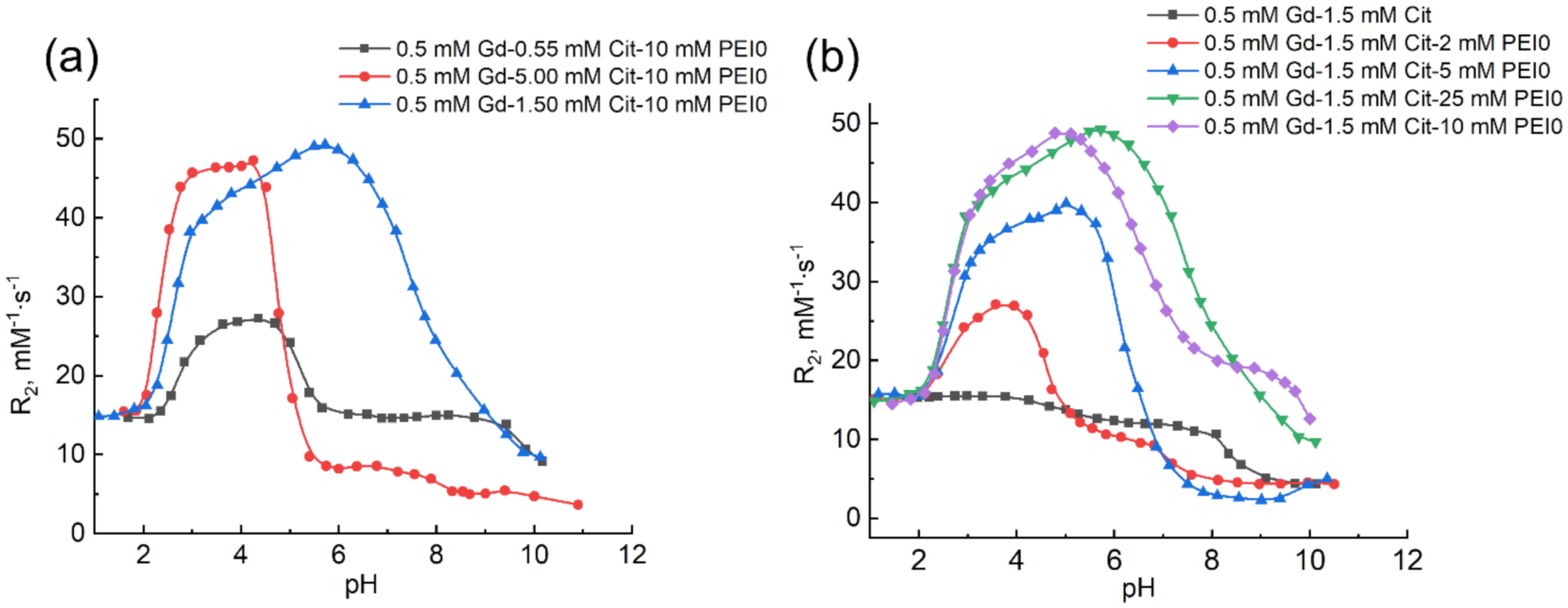
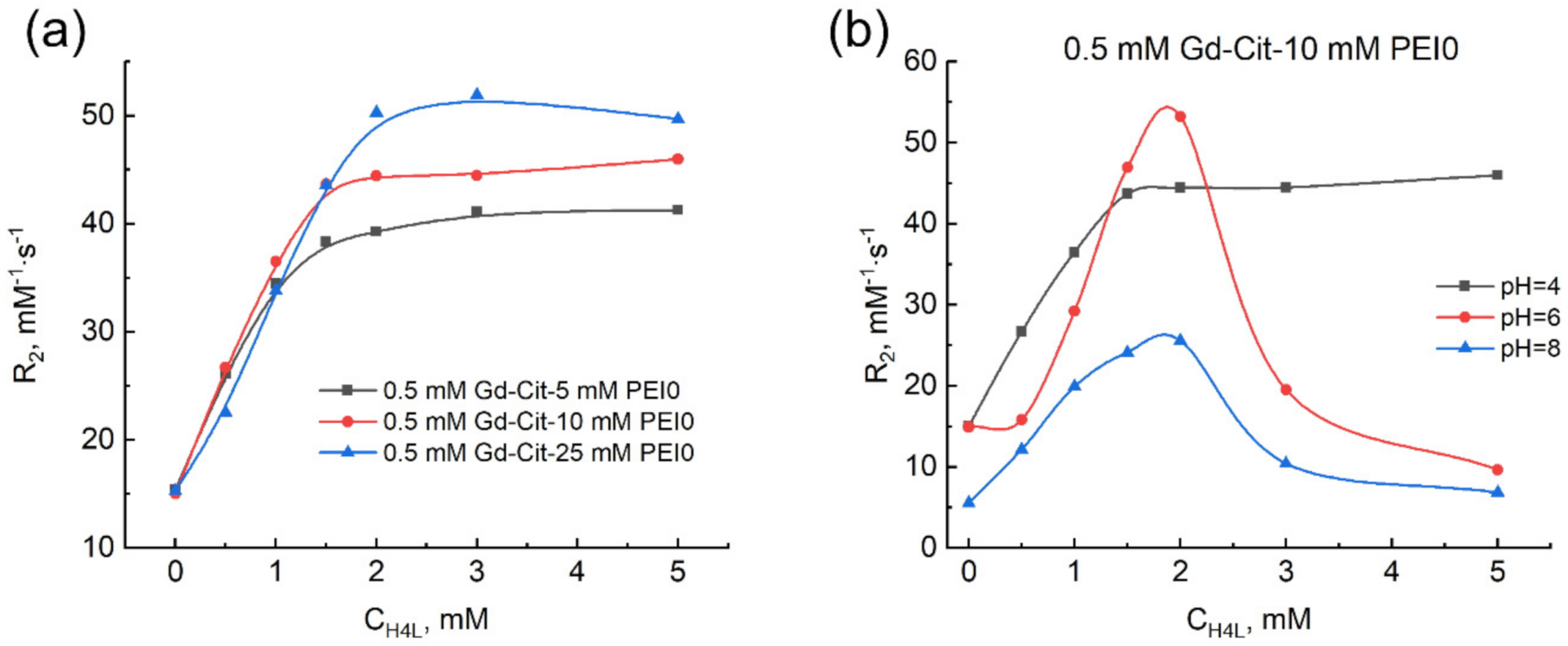
2.2. Effect of PEI on the Complexation of Gadolinium(III) Ions with Citric Acid
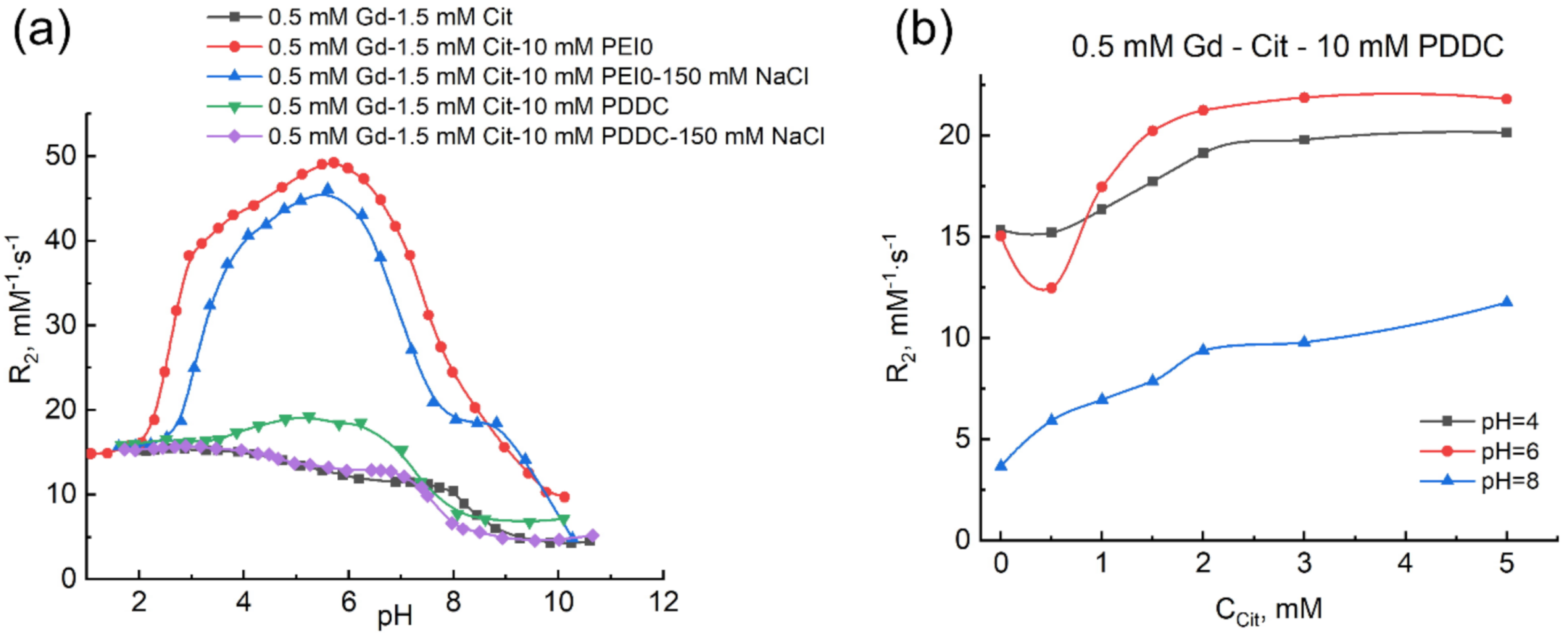
2.3. Effect of Molecular Mass and Nature of Cationic Polymer on the Complexation of Gadolinium(III) Ions with Citric Acid
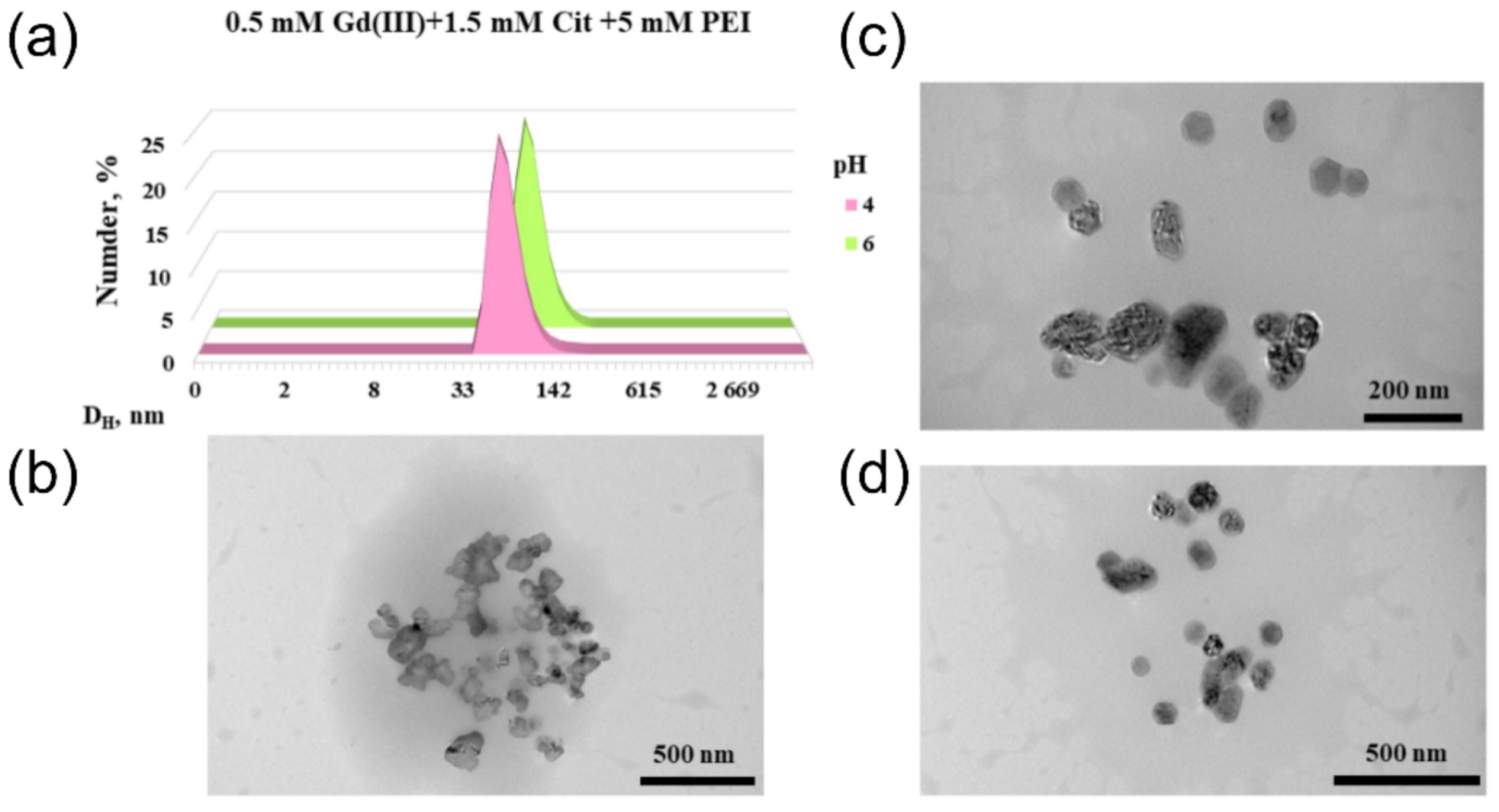
2.4. Morphology of Gadolinium(III) Complexes with Citric Acid in Polyethyleneimine Solution
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. NMR Relaxation
4.3. Modeling of Equilibria
4.4. Size and Morphology
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Geckeler, K.; Lange, G.; Eberhardt, H.; Bayer, E. Preparation and application of water soluble polymer-metal complexes. Pure Appl. Chem. 1980, 52, 1883–1905. [Google Scholar] [CrossRef]
- Rivas, B.L.; Maureira, A.E.; Mondaca, M.A. Aminodiacetic water-soluble polymer-metal ion interactions. Eur. Polym. J. 2008, 44, 2330–2338. [Google Scholar] [CrossRef]
- Zhu, Y.; Fan, W.; Zhou, T.; Li, X. Removal of chelated heavy metals from aqueous solution: A review of current methods and mechanisms. Sci. Total Environ. 2019, 678, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Feng, X. Polymer-enhanced ultrafiltration: Fundamentals, applications and recent developments. J. Memb. Sci. 2019, 586, 53–83. [Google Scholar] [CrossRef]
- Rivas, B.L.; Villoslada, I.M. Chelation properties of polymer complexes. Macromol. Chem. Phys. 1998, 1160, 1153–1160. [Google Scholar] [CrossRef]
- Rivas, B.L.; Moreno-Villoslada, I. Binding of Cd2+ and Na+ ions by poly(sodium 4-styrenesulfonate) analyzed by ultrafiltration and its relation with the counterion condensation theory. J. Phys. Chem. B 1998, 102, 6994–6999. [Google Scholar] [CrossRef]
- Juang, R.S.; Chen, M.N. Measurement of binding constants of poly(ethylenimine) with metal ions and metal chelates in aqueous media by ultrafiltration. Ind. Eng. Chem. Res. 1996, 35, 1935–1943. [Google Scholar] [CrossRef]
- Chandramouli, G.V.R.; Schläpfer, C.W.; von Zelewsky, A. EPR investigation of the second sphere coordination of [Cr(CN)5(NO)]3− with protonated polyethyleneimine. Magn. Reson. Chem. 1991, 29, S16–S20. [Google Scholar] [CrossRef]
- Moreno-Villoslada, I.; González, F.; Jofré, M.; Chandía, P.; Hess, S.; Rivas, B.L. Complexation behavior of Cu2+ in the presence of iminodiacetic acid and poly(ethyleneimine). Macromol. Chem. Phys. 2005, 206, 1541–1548. [Google Scholar] [CrossRef]
- Molinari, R.; Poerio, T.; Argurio, P. Polymer assisted ultrafiltration for copper-citric acid chelate removal from wash solutions of contaminated soil. J. Appl. Electrochem. 2005, 35, 375–380. [Google Scholar] [CrossRef]
- Molinari, R.; Poerio, T.; Argurio, P. Chemical and operational aspects in running the polymer assisted ultrafiltration for separation of copper(II)-citrate complexes from aqueous media. J. Memb. Sci. 2007, 295, 139–147. [Google Scholar] [CrossRef]
- Maketon, W.; Ogden, K.L. Synergistic effects of citric acid and polyethyleneimine to remove copper from aqueous solutions. Chemosphere 2009, 75, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.S.; Kwon, T.S.; Baek, K.; Yang, J.W. Centrifugal polyelectrolyte enhanced ultrafiltration for removal of copper-citrate complexes from aqueous solutions. Sep. Sci. Technol. 2006, 41, 1583–1592. [Google Scholar] [CrossRef]
- Lu, P.J.; Hu, W.W.; Chen, T.S.; Chern, J.M. Adsorption of copper-citrate complexes on chitosan: Equilibrium modeling. Bioresour. Technol. 2010, 101, 1127–1134. [Google Scholar] [CrossRef]
- De Robertis, A.; de Stefano, C.; Giuffré, O.; Sammartano, S. Binding of carboxylic ligands by protonated amines. J. Chem. Soc.—Faraday Trans. 1996, 92, 4219–4226. [Google Scholar] [CrossRef]
- De Robertis, A.; De Stefano, C.; Gianguzza, A.; Sammartano, S. Binding of polyanions by biogenic amines. III. Formation and stability of protonated spermidine and spermine complexes with carboxylic ligands. Talanta 1999, 48, 119–126. [Google Scholar] [CrossRef]
- Chen, G.E.; Sun, D.; Xu, Z.L. Rare earth ion from aqueous solution removed by polymer enhanced ultrafiltration process. Adv. Mater. Res. 2011, 233–235, 959–964. [Google Scholar] [CrossRef]
- Ramachandhran, V.; Samanta, S.K.; Misra, B.M. Radiocerium separation behavior of ultrafiltration membranes. J. Radioanal. Nucl. Chem. 1998, 237, 121–124. [Google Scholar] [CrossRef]
- Lauffer, R.B. Paramagnetic Metal Complexes as Water Proton Relaxation Agents for NMR Imaging: Theory and Design. Chem. Rev. 1987, 87, 901–927. [Google Scholar] [CrossRef]
- Caravan, P.; Ellison, J.J.; McMurry, T.J.; Lauffer, R.B. Gadolinium(III) chelates as MRI contrast agents: Structure, dynamics, and applications. Chem. Rev. 1999, 99, 2293–2352. [Google Scholar] [CrossRef] [PubMed]
- Sakol, N.; Egawa, A.; Fujiwara, T. Gadolinium complexes as contrast agent for cellular NMR spectroscopy. Int. J. Mol. Sci. 2020, 21, 4042. [Google Scholar] [CrossRef] [PubMed]
- Robb, I.D. The binding of counter ions to detergent micelles the nature of the stern layer. J. Colloid Interface Sci. 1971, 37, 521–527. [Google Scholar] [CrossRef]
- Amirov, R.R.; Saprykova, Z.A. Investigation of the sodium dodecylsulphate (SDS) micellization using paramagnetic relaxation probes. Russ. Colloid J. 1994, 56, 160–163. [Google Scholar]
- Amirov, R.R.; Nugaeva, Z.T.; Mustafina, A.R.; Fedorenko, S.V.; Morozov, V.I.; Kazakova, E.K.; Habicher, W.D.; Konovalov, A.I. Aggregation and counter ion binding ability of sulfonatomethylcalix[4] resorcinarenes in aqueous solutions. Colloids Surf. A Physicochem. Eng. Asp. 2004, 240, 35–43. [Google Scholar] [CrossRef]
- Amirov, R.R.; Ziyatdinova, A.B.; Burilova, E.A.; Zhukov, A.Y.; Antipin, I.S.; Stoikov, I.I. Complexation of GdIII with tetra-p-tert-butylthiacalix[4]arenoic acid in micellar media. Russ. Chem. Bull. 2009, 58, 1400–1407. [Google Scholar] [CrossRef]
- Amirov, R.R.; Burilova, E.A.; McMillan, Z.T.; Amirova, L.R.; Ziyatdinova, A.B.; Shayimova, J.R.; Bukharov, M.S.; Dimiev, A.M.; Zakharov, A.V. An NMR relaxivity and ESR study of the interaction of the paramagnetic manganese(II) and gadolinium(III) ions with anionic, cationic and neutral water-soluble polymers and their mixtures. J. Mol. Liq. 2017, 238, 184–192. [Google Scholar] [CrossRef]
- Amirov, R.R.; Shayimova, J.; Dimiev, A.M. Distribution of Gd(III) ions at the graphene oxide/water interface. J. Colloid Interface Sci. 2018, 527, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Solodov, A.N.; Shayimova, J.; Amirov, R.R.; Dimiev, A.M. Mimicking the graphene oxide structure in solutions by interaction of Fe(III) and Gd(III) with model small-size ligands. The NMR relaxation study. J. Mol. Liq. 2021, 321, 114344. [Google Scholar] [CrossRef]
- Amirov, R.R.; Litvinov, I.A.; Gubaidullin, A.T.; Matyugicheva, U.V. Complex of copper(II) with ethylenediamine and decyl sulfate ions. Formation in water and crystal structure. Russ. J. Gen. Chem. 2003, 73, 1860–1865. [Google Scholar] [CrossRef]
- Mirsaizyanova, S.A.; Ziyatdinova, A.B.; Amirov, R.R. Iron(III) Salicylate complexes in surfactant solutions. Colloid J. 2011, 73, 509–516. [Google Scholar] [CrossRef]
- Burilova, E.A.; Vagapova, L.I.; Nasirova, Z.A.; Nikitina, T.V.; Solodov, A.N.; Shaiymova, J.R.; Amirov, R.R. Peculiarities of manganese(II) interaction with hydroxyethylidene diphosphonic acid and aminomethylated calix[4]resorcinol in organized media. Uchenye Zap. Kazan. Univ. Seriya Estestv. Nauk. 2019, 161, 199–210. [Google Scholar] [CrossRef]
- Burilova, E.A.; Ziyatdinova, A.B.; Zyavkina, Y.I.; Amirov, R.R. Influence of Water-soluble Polymers on the Formation of Manganese(II) Complexonates in Solutions. 2. Complexes with DTPA. Proc. Kazan Univ. Nat. Sci. Ser. 2013, 155, 26–38. [Google Scholar]
- Pramauro, E.; Pelezetti, E. Chapter 3 Effect of surfactant aggregates on chemical kinetics and equilibria. Compr. Anal. Chem. 1996, 31, 131–202. [Google Scholar] [CrossRef]
- Hébrant, M. Metal ion extraction in microheterogeneous systems. Coord. Chem. Rev. 2009, 253, 2186–2192. [Google Scholar] [CrossRef]
- Shriadah, M.M.A.; Ohzeki, K. Effect of anion-exchange resin on the formation of iron(III)-Tiron complexes. Analyst 1986, 111, 197–200. [Google Scholar] [CrossRef]
- Amirov, R.R.; Saprykova, Z.A.; Ibragimova, Z.Z. Specific features of the complexation of copper(II), manganese(II), and gadolinium(III) with salicylic, benzoic, and sulfosalicylic acids in aqueous solutions of nonionic surfactant. Colloid J. Russ. Acad. Sci. 1998, 60, 263–269. [Google Scholar]
- Wahsner, J.; Gale, E.M.; Rodríguez-Rodríguez, A.; Caravan, P. Chemistry of MRI contrast agents: Current challenges and new frontiers. Chem. Rev. 2019, 119, 957–1057. [Google Scholar] [CrossRef] [PubMed]
- Lange, S.; Mędrzycka-Dąbrowska, W.; Zorena, K.; Dąbrowski, S.; Ślęzak, D.; Malecka-Dubiela, A.; Rutkowski, P. Nephrogenic systemic fibrosis as a complication after gadolinium-containing contrast agents: A rapid review. Int. J. Environ. Res. Public Health 2021, 18, 3000. [Google Scholar] [CrossRef]
- Aime, S.; Botta, M.; Fedeli, F.; Gianolio, E.; Terreno, E.; Anelli, P. High-relaxivity contrast agents for magnetic resonance imaging based on multisite interactions between a β-cyclodextrin oligomer and suitably functionalized GdIII chelates. Chem.—A Eur. J. 2001, 7, 5262–5269. [Google Scholar] [CrossRef]
- Biscotti, A.; Estour, F.; Sembo-Backonly, B.S.; Balieu, S.; Bosco, M.; Barbot, C.; Pallier, A.; Tóth, É.; Bonnet, C.S.; Gouhier, G. Gd3+ complexes conjugated to cyclodextrins: Hydroxyl functions influence the relaxation properties. Processes 2021, 9, 269. [Google Scholar] [CrossRef]
- Berki, T.R.; Martinelli, J.; Tei, L.; Willcock, H.; Butler, S.J. Polymerizable Gd(III) building blocks for the synthesis of high relaxivity macromolecular MRI contrast agents. Chem. Sci. 2021, 12, 3999–4013. [Google Scholar] [CrossRef]
- Gong, E.; Pauly, J.M.; Wintermark, M.; Zaharchuk, G. Deep learning enables reduced gadolinium dose for contrast-enhanced brain MRI. J. Magn. Reson. Imaging 2018, 48, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; He, Y.; Zhao, H.; Zhang, Y.; Yin, J.; Cao, Y.; Pei, R. A positively charged small-molecule T1 magnetic resonance imaging contrast agent for highly efficient labeling and tracking adipose tissue-derived stem cells. Mater. Today Commun. 2020, 25, 101627. [Google Scholar] [CrossRef]
- Yon, M.; Billotey, C.; Marty, J.D. Gadolinium-based contrast agents: From gadolinium complexes to colloidal systems. Int. J. Pharm. 2019, 569, 118577. [Google Scholar] [CrossRef] [PubMed]
- Xiong, R.; Cheng, L.; Tian, Y.; Tang, W.; Xu, K. Hyperbranched polyethylenimine based polyamine-N-oxide-carboxylate chelates of gadolinium for high relaxivity MRI contrast agents. RSC Adv. 2016, 6, 28063–28068. [Google Scholar] [CrossRef]
- Zhou, R.; Palmer, V.; Geckeler, K.E. Removal of inorganic ions by polymer-based colloid-enhanced membrane filtration in aqueous solution. Water Res. 1994, 28, 1257–1260. [Google Scholar] [CrossRef]
- Lide, D.R. Handbook of Chemistry and Physics, 84th ed.; CRC: Boca Raton, FL, USA, 2004. [Google Scholar]
- Nicolle, G.M.; Tóth, É.; Eisenwiener, K.P.; Mäcke, H.R.; Merbach, A.E. From monomers to micelles: Investigation of the parameters influencing proton relaxivity. J. Biol. Inorg. Chem. 2002, 7, 757–769. [Google Scholar] [CrossRef]
- Jackson, G.E.; Toit, J. Gadolinium(III) in vivo Speciation. Part I A. Potentiometric and Spectroscopic Study of Gadolinium(III) Citrate Complexes. J. Chem. Soc. Dalton Trans. 1991, 6, 1463–1466. [Google Scholar] [CrossRef]
- Zabiszak, M.; Nowak, M.; Taras-Goslinska, K.; Kaczmarek, M.T.; Hnatejko, Z.; Jastrzab, R. Carboxyl groups of citric acid in the process of complex formation with bivalent and trivalent metal ions in biological systems. J. Inorg. Biochem. 2018, 182, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Ohyoshi, A.; Ohyoshi, E.; Ono, H.; Yamakawa, S. A study of citrate complexes of several lanthanides. J. Inorg. Nucl. Chem. 1972, 34, 1955–1960. [Google Scholar] [CrossRef]
- Jain, A.; Singh, O.V.; Tandon, S.N. Extraction of Ce(III), Gd(III) And Yb(III) from Citrate Medium by High Molecular Weight Amines. Anal. Lett. 1988, 21, 1927–1934. [Google Scholar] [CrossRef]
- Tompkins, E.R.; Mayer, S.W. Ion Exchange as a Separations Method. III. Equilibrium Studies of the Reactions of Rare Earth Complexes with Synthetic Ion Exchange Resins. J. Am. Chem. Soc. 1947, 69, 2859–2865. [Google Scholar] [CrossRef] [PubMed]
- Baggio, R.; Calvo, R.; Garland, M.T.; Peña, O.; Perec, M.; Rizzi, A. Gadolinium and neodymium citrates: Evidence for weak ferromagnetic exchange between gadolinium(III) cations. Inorg. Chem. 2005, 44, 8979–8987. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, C.; Raptopoulou, C.P.; Drouza, C.; Lalioti, N.; Salifoglou, A. Synthesis, spectroscopic, structural and magnetic studies of new binary Cr(III)-citrate pH-specific structural variants from aqueous media. Polyhedron 2009, 28, 3209–3220. [Google Scholar] [CrossRef]
- Salnikov, Y.I.; Devyatov, F.V.; Amirov, R.R. Complexing of yttrium group REE ions with tricarballyl acid. Zhurnal Neorg. Khim. 1984, 29, 65–68. [Google Scholar]
- Zezin, A.A. Synthesis of hybrid materials in polyelectrolyte matrixes: Control over sizes and spatial organization of metallic nanostructures. Polym. Sci. Ser. C 2016, 58, 118–130. [Google Scholar] [CrossRef]
- Bronshtein, L.M.; Sidorov, S.N.; Valetskii, P.M. Nanostructured polymeric systems as nanoreactors for nanoparticle formation. Russ. Chem. Rev. 2004, 73, 501–515. [Google Scholar] [CrossRef]
- Khutoryanskiy, V.V.; Smyslov, R.Y.; Yakimansky, A.V. Modern Methods for Studying Polymer Complexes in Aqueous and Organic Solutions. Polym. Sci. Ser. A 2018, 60, 553–576. [Google Scholar] [CrossRef]
- von Harpe, A.; Petersen, H.; Li, Y.; Kissel, T. Characterization of commercially available and synthesized polyethylenimines for gene delivery. J. Control. Release 2000, 69, 309–322. [Google Scholar] [CrossRef]
- Li, Y.; Ghoreishi, S.M.; Warr, J.; Bloor, D.M.; Holzwarth, J.F.; Wyn-Jones, E. Binding of Sodium Dodecyl Sulfate to Some Polyethyleneimines and Their Ethoxylated Derivatives at Different pH Values. Electromotive Force and Microcalorimetry Studies. Langmuir 2000, 16, 3093–3100. [Google Scholar] [CrossRef]
- Zakharova, L.Y.; Valeeva, F.G.; Kudryavtsev, D.B.; Bilalov, A.V.; Tret´yakova, A.Y.; Kudryavtseva, L.A.; Konovalov, A.I.; Barabanov, V.P. Sodium dodecyl sulfate-polyethyleneimine-water system. Self-organization and catalytic activity. Russ. Chem. Bull. 2005, 3, 641–649. [Google Scholar] [CrossRef]
- Salnikov, Y.I.; Glebov, A.N.; Devyatov, F.V. Poliyadernye kompleksy v rastvorakh [Polynuclear Complexes in Solutions]; Izd-vo Kazanskogo Gos. Un-ta: Kazan, Russia, 1989; p. 287. Available online: https://www.webofscience.com/wos/woscc/full-record/WOS:A1984TK44700021 (accessed on 23 October 2021).


| No. | Equilibrium | lgK | pKa | lgβ | R2, mM−1s−1 |
|---|---|---|---|---|---|
| 1 | H4L ⇆ H3L− + H+ | −3.09 ± 0.01 | 3.09 | - | |
| 2 | H4L ⇆ H2L2− + H+ | −7.78 ± 0.01 | 4.69 | - | |
| 3 | H4L ⇆ HL3− + H+ | −13.95 ± 0.02 | 6.17 | - | |
| 4 | Gd3+ + H4L ⇆ GdH2L+ + 2H+ | −1.90 ± 0.03 | 5.88 | (15.5) | |
| 5 | Gd3+ + H4L ⇆ GdHL0 + 3H+ | −5.51 ± 0.02 | 8.44 | (15) | |
| 6 | Gd3+ + H4L ⇆ GdL− + 4H+ | −12.31 ± 0.04 | 15.64 | 7.2 ± 0.2 | |
| 7 | 3Gd3+ + 3H4L ⇆ Gd3L3H-25− + 14H+ | −50.14 ± 0.16 | 19.71 | 3.8 ± 0.4 | |
| 8 | Gd3+ + 2H4L ⇆ Gd(HL)23− + 6H+ | −15.15 ± 0.08 | 12.75 | 12.2 ± 0.2 | |
| 9 | Gd3+ + 2H4L ⇆ GdL25− + 8H+ | −32.25 ± 0.14 | 23.65 | 4 ± 0.1 |
| Reaction of Formation | lgKapp (R2, mM−1s−1) | ||
|---|---|---|---|
| 5 mM PEI | 10 mM PEI | 25 mM PEI | |
| Gd3+ + 3H4L ⇆ Gd(H2L)33− + 6H+ | −5.71 ± 0.07 (38 ± 0.2) | −5.48 ± 0.15 (44.6 ± 0.7) | −5.68 ± 0.14 (44.8 ± 0.9) |
| Gd3+ + 3H4L ⇆ Gd(H2L)2HL4− + 7H+ | −10.02 ± 0.22 (40.3 ± 0.3) | −10.01 ± 0.12 (48.5 ± 0.6) | −9.47 ± 0.10 (49.6 ± 0.6) |
| Reaction of Formation | lgKapp (R2, mM−1s−1) | ||
|---|---|---|---|
| PEI3 | PEI2 | PEI1 | |
| Gd3+ + 3H4L ⇆ Gd(H2L)33− + 6H+ | −5.94 ± 0.21 (22.5 ± 0.2) | −5.97 ± 0.26 (23.0 ± 0.3) | −5.71 ± 0.09 (44.8 ± 0.4) |
| Gd3+ + 3H4L ⇆ Gd(H2L)2HL4− + 7H+ | −10.98 ± 0.15 (22.6 ± 0.3) | −11.17 ± 0.12 (22.5 ± 0.3) | −11.01 ± 0.10 (49.8 ± 0.5) |
| Composition | ZP (mV) | |
|---|---|---|
| pH 4 | pH 6 | |
| 5 mM PEI0 | +17 ± 1 | +14 ± 1 |
| 1.5 mM Gd(III) + 5 mM PEI0 | +19 ± 1 | +13 ± 1 |
| 1.5 mM Cit + 5 mM PEI0 | +17 ± 1 | +4 ± 0.4 |
| 0.5 mM Gd(III) + 1.5 mM Cit + 5 mM PEI0 | +29 ± 0.3 | +18 ± 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burilova, E.; Solodov, A.; Shayimova, J.; Zhuravleva, J.; Shurtakova, D.; Evtjugin, V.; Zhiltsova, E.; Zakharova, L.; Kashapov, R.; Amirov, R. Design of High-Relaxivity Polyelectrolyte Nanocapsules Based on Citrate Complexes of Gadolinium(III) of Unusual Composition. Int. J. Mol. Sci. 2021, 22, 11590. https://doi.org/10.3390/ijms222111590
Burilova E, Solodov A, Shayimova J, Zhuravleva J, Shurtakova D, Evtjugin V, Zhiltsova E, Zakharova L, Kashapov R, Amirov R. Design of High-Relaxivity Polyelectrolyte Nanocapsules Based on Citrate Complexes of Gadolinium(III) of Unusual Composition. International Journal of Molecular Sciences. 2021; 22(21):11590. https://doi.org/10.3390/ijms222111590
Chicago/Turabian StyleBurilova, Evgenia, Alexander Solodov, Julia Shayimova, Julia Zhuravleva, Darya Shurtakova, Vladimir Evtjugin, Elena Zhiltsova, Lucia Zakharova, Ruslan Kashapov, and Rustem Amirov. 2021. "Design of High-Relaxivity Polyelectrolyte Nanocapsules Based on Citrate Complexes of Gadolinium(III) of Unusual Composition" International Journal of Molecular Sciences 22, no. 21: 11590. https://doi.org/10.3390/ijms222111590
APA StyleBurilova, E., Solodov, A., Shayimova, J., Zhuravleva, J., Shurtakova, D., Evtjugin, V., Zhiltsova, E., Zakharova, L., Kashapov, R., & Amirov, R. (2021). Design of High-Relaxivity Polyelectrolyte Nanocapsules Based on Citrate Complexes of Gadolinium(III) of Unusual Composition. International Journal of Molecular Sciences, 22(21), 11590. https://doi.org/10.3390/ijms222111590






