Rho/ROCK Pathway and Noncoding RNAs: Implications in Ischemic Stroke and Spinal Cord Injury
Abstract
:1. Introduction
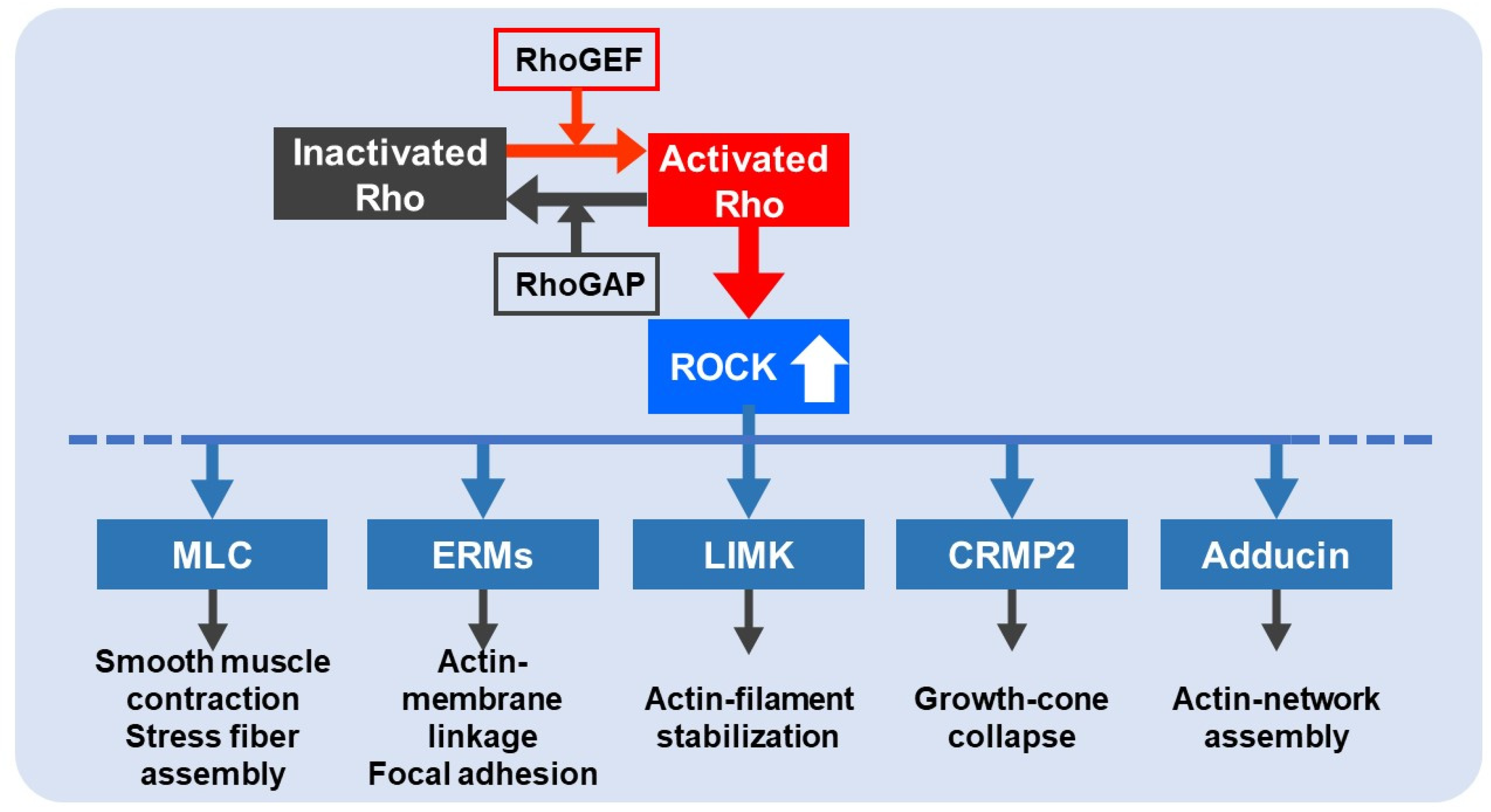
2. Rho/ROCK Pathway
3. Noncoding RNAs
4. Pathophysiology of IS
5. Rho/ROCK Pathway in IS
5.1. Rho/ROCK Pathway and Neuroprotection in IS
5.2. Rho/ROCK Pathway and Platelet Function in IS
5.3. Rho/ROCK Pathway and Neuroinflammation in IS
5.4. Rho/ROCK Pathway and BBB in IS
5.5. Rho/ROCK Pathway and Astrocytes in IS
5.6. Rho/ROCK Pathway and Axon Growth Inhibition in IS
5.7. Rho/ROCK Pathway and Neurogenesis/Angiogenesis in IS
6. Rho/ROCK Pathway in SCI
7. ncRNAs in IS
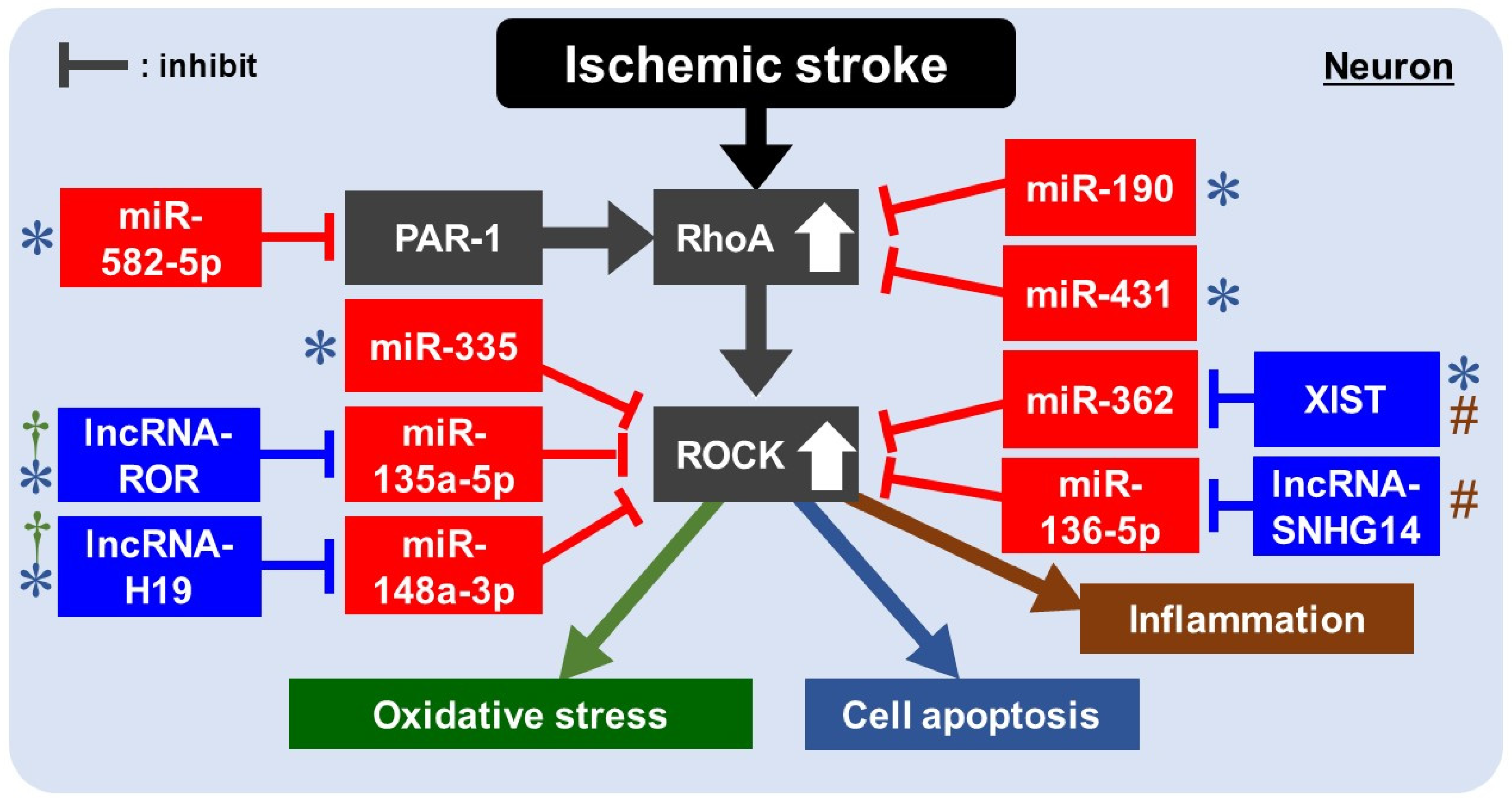
8. Rho/ROCK Pathway and ncRNAs in IS
8.1. Rho/ROCK Pathway and miRNAs for Apoptosis in IS
8.2. Rho/ROCK Pathway and lncRNAs/miRNAs for Apoptosis in IS
8.3. Rho/ROCK Pathway and lncRNAs/miRNAs for Oxidative Stress/Inflammation in IS
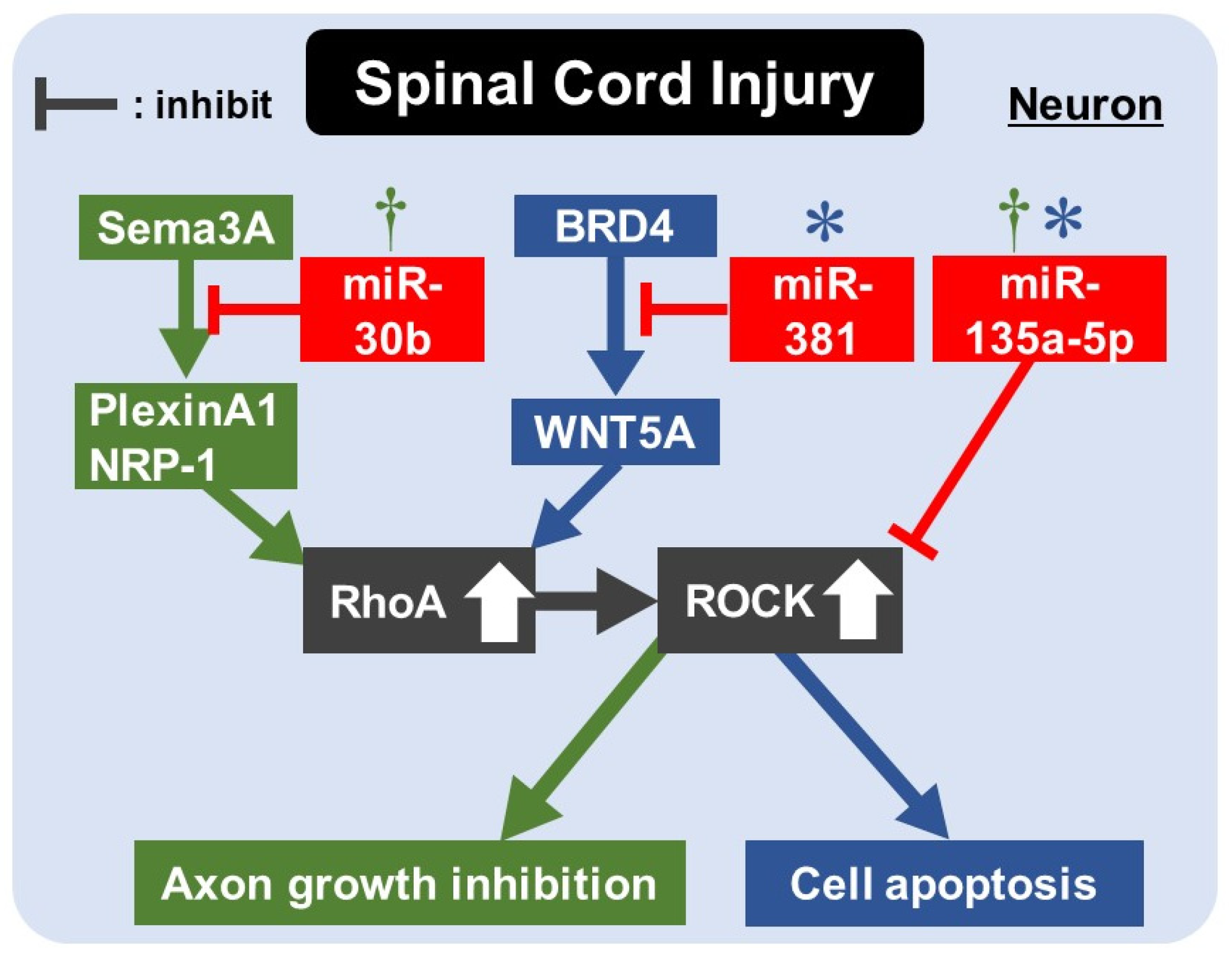
9. Rho/ROCK Pathway and miRNAs for Apoptosis/Axon Regeneration in SCI
| Model | miRNA | Expression after Insult | Target | Effects | Reference |
|---|---|---|---|---|---|
| SDCL in rat Primary sensory neuron of rat | miR-30b | Decreased | Sema3A | miR-30b agomir promoted neurite outgrowth, and antagomir inhibited it. miR-30b agomir regulates sema3A/PlexinA1-NRP-1/RhoA/ROCK pathway, promoting sensory conductive function recovery after SDCL. | [121] |
| SCI in rat DRG cells of rat | miR-381 | Decreased | BRD4 | BRD4 promoted WNT5A expression via binding to the promotor of WNT5A. WNT5A promoted apoptosis by activating the RhoA/ROCK pathway. miR-381 derived from EV in MSCs inhibited neuron apoptosis and promoted the recovery of SCI by inhibiting the BRD4/WNT5A axis. | [123] |
| SCI in rat PC12 cells under H2O2 stimulation | miR-135a-5p | Decreased | SP1 ROCK1/2 | miR-135a-5p-SP1-Bax/Bcl-2/caspase3 axis inhibited neuronal apoptosis. miR-135a-5p-ROCK-AKT/GSK3β pathway promoted axon regeneration during functional recovery after SCI. | [130] |
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sutherland, B.; Minnerup, J.; Balami, J.S.; Arba, F.; Buchan, A.M.; Kleinschnitz, C. Neuroprotection for Ischaemic Stroke: Translation from the Bench to the Bedside. Int. J. Stroke 2012, 7, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Feng, Z.; Du, L.; Huang, Y.; Ge, J.; Deng, Y.; Mei, Z. The Potential Role of MicroRNA-124 in Cerebral Ischemia Injury. Int. J. Mol. Sci. 2019, 21, 120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalkara, T.; Alarcon-Martinez, L. Cerebral microvascular pericytes and neurogliovascular signaling in health and disease. Brain Res. 2015, 1623, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Stankiewicz, T.R.; Linseman, D.A. Rho family GTPases: Key players in neuronal development, neuronal survival, and neurodegeneration. Front. Cell. Neurosci. 2014, 8, 314. [Google Scholar] [CrossRef] [Green Version]
- Loirand, G. Rho Kinases in Health and Disease: From Basic Science to Translational Research. Pharmacol. Rev. 2015, 67, 1074–1095. [Google Scholar] [CrossRef] [Green Version]
- Feng, Y.; Lograsso, P.V.; Defert, O.; Li, R. Rho Kinase (ROCK) Inhibitors and Their Therapeutic Potential. J. Med. Chem. 2015, 59, 2269–2300. [Google Scholar] [CrossRef] [PubMed]
- Koch, J.C.; Tatenhorst, L.; Roser, A.-E.; Saal, K.-A.; Tönges, L.; Lingor, P. ROCK inhibition in models of neurodegeneration and its potential for clinical translation. Pharmacol. Ther. 2018, 189, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.D.; Parsons, C.; Walker, L.; Zhang, W.C.; Slack, F.J. Targeting noncoding RNAs in disease. J. Clin. Investig. 2017, 127, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, O.; Fujisawa, K.; Ishizaki, T.; Saito, Y.; Nakao, K.; Narumiya, S. ROCK-I and ROCK-II, two isoforms of Rho-associated coiled-coil forming protein serine/threonine kinase in mice. FEBS Lett. 1996, 392, 189–193. [Google Scholar] [CrossRef] [Green Version]
- Riento, K.; Ridley, A.J. ROCKs: Multifunctional kinases in cell behaviour. Nat. Rev. Mol. Cell Biol. 2003, 4, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Amano, M.; Nakayama, M.; Kaibuchi, K. Rho-kinase/ROCK: A key regulator of the cytoskeleton and cell polarity. Cytoskeleton 2010, 67, 545–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, J.; Wei, L. Rho kinase in the regulation of cell death and survival. Arch. Immunol. Ther. Exp. 2007, 55, 61–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avalos, A.M.; Arthur, W.T.; Schneider, P.; Quest, A.F.G.; Burridge, K.; Leyton, L. Aggregation of Integrins and RhoA Activation Are Required for Thy-1-induced Morphological Changes in Astrocytes. J. Biol. Chem. 2004, 279, 39139–39145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loirand, G.; Guérin, P.; Pacaud, P. Rho Kinases in Cardiovascular Physiology and Pathophysiology. Circ. Res. 2006, 98, 322–334. [Google Scholar] [CrossRef]
- Sahai, E.; Marshall, C.J. RHO-GTPases and cancer. Nat. Rev. Cancer 2002, 2, 133–142. [Google Scholar] [CrossRef]
- Mueller, B.K.; Mack, H.; Teusch, N. Rho kinase, a promising drug target for neurological disorders. Nat. Rev. Drug Discov. 2005, 4, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.; Momeni, F.; Saadatpour, L.; Sahebkar, A.; Goodarzi, M.; Masoudifar, A.; Kouhpayeh, S.; Salehi, H.; Mirzaei, H.R.; Jaafari, M.R. MicroRNA: Relevance to stroke diagnosis, prognosis, and therapy. J. Cell. Physiol. 2018, 233, 856–865. [Google Scholar] [CrossRef]
- Lee, Y.; Jeon, K.; Lee, J.; Kim, S.; Kim, V.N. MicroRNA maturation: Stepwise processing and subcellular localization. EMBO J. 2002, 21, 4663–4670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basyuk, E.; Suavet, F.; Doglio, A.; Bordonné, R.; Bertrand, E. Human let-7 stem-loop precursors harbor features of RNase III cleavage products. Nucleic Acids Res. 2003, 31, 6593–6597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, J.; Lee, Y.; Yeom, K.-H.; Kim, Y.-K.; Jin, H.; Kim, V.N. The Drosha-DGCR8 complex in primary microRNA processing. Genes Dev. 2004, 18, 3016–3027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lau, N.C.; Lim, L.P.; Weinstein, E.G.; Bartel, D.P. An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science 2001, 294, 858–862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutvágner, G.; McLachlan, J.; Pasquinelli, A.E.; Bálint, E.; Tuschl, T.; Zamore, P.D. A cellular function for the RNA-interference en-zyme Dicer in the maturation of the let-7 small temporal RNA. Science 2001, 293, 834–838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chendrimada, T.P.; Gregory, R.I.; Kumaraswamy, E.; Norman, J.; Cooch, N.; Nishikura, K.; Shiekhattar, R. TRBP recruits the Dicer complex to Ago2 for microRNA processing and gene silencing. Nature 2005, 436, 740–744. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, Biogenesis, Mechanism, and Function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef] [Green Version]
- Khoshnam, S.E.; Winlow, W.; Farbood, Y.; Moghaddam, H.F.; Farzaneh, M. Emerging Roles of microRNAs in Ischemic Stroke: As Possible Therapeutic Agents. J. Stroke 2017, 19, 166–187. [Google Scholar] [CrossRef] [PubMed]
- Bam, M.; Yang, X.; Sen, S.; Zumbrun, E.E.; Dennis, L.; Zhang, J.; Nagarkatti, P.S.; Nagarkatti, M. Characterization of Dysregulated miRNA in Peripheral Blood Mononuclear Cells from Ischemic Stroke Patients. Mol. Neurobiol. 2018, 55, 1419–1429. [Google Scholar] [CrossRef]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long non-coding RNAs: Insights into functions. Nat. Rev. Genet. 2009, 10, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Mendis, S.; Davis, S.; Norrving, B. Organizational update: The world health organization global status report on noncom-municable diseases 2014; one more landmark step in the combat against stroke and vascular disease. Stroke 2015, 46, e121–e122. [Google Scholar] [CrossRef]
- Beal, C.C. Gender and stroke symptoms: A review of the current literature. J. Neurosci. Nurs. 2010, 42, 80–87. [Google Scholar] [CrossRef]
- Murphy, T.H.; Li, P.; Betts, K.; Liu, R. Two-Photon Imaging of Stroke Onset In Vivo Reveals That NMDA-Receptor Independent Ischemic Depolarization Is the Major Cause of Rapid Reversible Damage to Dendrites and Spines. J. Neurosci. 2008, 28, 1756–1772. [Google Scholar] [CrossRef] [Green Version]
- Hossmann, K.-A. Pathophysiology and Therapy of Experimental Stroke. Cell. Mol. Neurobiol. 2006, 26, 1055–1081. [Google Scholar] [CrossRef] [PubMed]
- Besancon, E.; Guo, S.; Lok, J.; Tymianski, M.; Lo, E.H. Beyond NMDA and AMPA glutamate receptors: Emerging mechanisms for ionic imbalance and cell death in stroke. Trends Pharmacol. Sci. 2008, 29, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Gilgen, M.; Slotboom, J.; El-Koussy, M.; Zubler, C.; Kiefer, C.; Luedi, R.; Mono, M.-L.; Heldner, M.R.; Weck, A.; et al. Factors that determine penumbral tissue loss in acute ischaemic stroke. Brain 2013, 136, 3554–3560. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Ning, N.; Zhou, Q.; Khoshnam, S.E.; Farzaneh, M. Mitochondria as a therapeutic target for ischemic stroke. Free. Radic. Biol. Med. 2020, 146, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.J.; Sharp, F.R. Implications of MMP9 for Blood Brain Barrier Disruption and Hemorrhagic Transformation Following Ischemic Stroke. Front. Cell. Neurosci. 2016, 10, 56. [Google Scholar] [CrossRef] [Green Version]
- Jin, R.; Liu, L.; Zhang, S.; Nanda, A.; Li, G. Role of Inflammation and Its Mediators in Acute Ischemic Stroke. J. Cardiovasc. Transl. Res. 2013, 6, 834–851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sladojevic, N.; Yu, B.; Liao, J.K. ROCK as a therapeutic target for ischemic stroke. Expert Rev. Neurother. 2017, 17, 1167–1177. [Google Scholar] [CrossRef] [PubMed]
- Yagita, Y.; Kitagawa, K.; Sasaki, T.; Terasaki, Y.; Todo, K.; Omura-Matsuoka, E.; Kaibuchi, K.; Hori, M. Rho-kinase activation in en-dothelial cells contributes to expansion of infarction after focal cerebral ischemia. J. Neurosci. Res. 2007, 85, 2460–2469. [Google Scholar] [CrossRef]
- Rikitake, Y.; Kim, H.-H.; Huang, Z.; Seto, M.; Yano, K.; Asano, T.; Moskowitz, M.A.; Liao, J.K. Inhibition of Rho Kinase (ROCK) Leads to Increased Cerebral Blood Flow and Stroke Protection. Stroke 2005, 36, 2251–2257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Satoh, S.; Toshima, Y.; Ikegaki, I.; Iwasaki, M.; Asano, T. Wide therapeutic time window for fasudil neuroprotection against is-chemia-induced delayed neuronal death in gerbils. Brain Res. 2007, 1128, 175–180. [Google Scholar] [CrossRef]
- Satoh, S.-I.; Toshima, Y.; Hitomi, A.; Ikegaki, I.; Seto, M.; Asano, T. Wide therapeutic time window for Rho-kinase inhibition therapy in ischemic brain damage in a rat cerebral thrombosis model. Brain Res. 2008, 1193, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, J.; Hu, H.; Liu, P.; Fang, Y.; Wu, D. Rho-Kinase Inhibitor, Fasudil, Prevents Neuronal Apoptosis via the Akt Activation and PTEN Inactivation in the Ischemic Penumbra of Rat Brain. Cell. Mol. Neurobiol. 2012, 32, 1187–1197. [Google Scholar] [CrossRef]
- Wang, G.Y.; Wang, T.Z.; Zhang, Y.Y.; Li, F.; Yu, B.Y.; Kou, J.P. NMMHC IIA Inhibition Ameliorates Cerebral Ischem-ic/Reperfusion-Induced Neuronal Apoptosis Through Caspase-3/ROCK1/MLC Pathway. Drug Des. Devel. Ther. 2020, 14, 13–25. [Google Scholar] [CrossRef] [Green Version]
- Jeon, B.T.; Jeong, E.A.; Park, S.-Y.; Son, H.; Shin, H.J.; Lee, N.H.; Kim, H.J.; Kang, S.S.; Cho, G.J.; Choi, W.S.; et al. The Rho-Kinase (ROCK) Inhibitor Y-27632 Protects Against Excitotoxicity-Induced Neuronal Death In Vivo and In Vitro. Neurotox. Res. 2012, 23, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Sladojevic, N.; Oh, G.T.; Kim, H.-H.; Beaulieu, L.M.; Falet, H.; Kamiński, K.; Freedman, J.E.; Liao, J.K. Decreased thromboembolic stroke but not atherosclerosis or vascular remodelling in mice with ROCK2-deficient platelets. Cardiovasc. Res. 2017, 113, 1307–1317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magnus, T.; Wiendl, H.; Kleinschnitz, C. Immune mechanisms of stroke. Curr. Opin. Neurol. 2012, 25, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Laufs, U.; Liao, J.K. Post-transcriptional Regulation of Endothelial Nitric Oxide Synthase mRNA Stability by Rho GTPase. J. Biol. Chem. 1998, 273, 24266–24271. [Google Scholar] [CrossRef] [Green Version]
- Satoh, S.; Kobayashi, T.; Hitomi, A.; Ikegaki, I.; Suzuki, Y.; Shibuya, M.; Yoshida, J.; Asano, T. Inhibition of neutrophil migration by a protein kinase inhibitor for the treatment of ischemic brain infarction. Jpn. J. Pharmacol. 1999, 80, 41–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Satoh, S.-I.; Utsunomiya, T.; Tsurui, K.; Kobayashi, T.; Ikegaki, I.; Sasaki, Y.; Asano, T. Pharmacological profile of hydroxy fasudil as a selective rho kinase inhibitor on ischemic brain damage. Life Sci. 2001, 69, 1441–1453. [Google Scholar] [CrossRef]
- Jin, R.; Yang, G.; Li, G. Inflammatory mechanisms in ischemic stroke: Role of inflammatory cells. J. Leukoc. Biol. 2010, 87, 779–789. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Li, Q.Y.; Wang, X.; Sun, C.H.; Lu, C.Z.; Xiao, B.G. Fasudil protects hippocampal neurons against hypoxia-reoxygenation in-jury by suppressing microglial inflammatory responses in mice. J. Neurochem. 2010, 114, 1619–1629. [Google Scholar] [CrossRef]
- Abeysinghe, H.C.; Phillips, E.L.; Chin-Cheng, H.; Beart, P.M.; Roulston, C.L. Modulating Astrocyte Transition after Stroke to Pro-mote Brain Rescue and Functional Recovery: Emerging Targets Include Rho Kinase. Int. J. Mol. Sci. 2016, 17, 288. [Google Scholar] [CrossRef] [Green Version]
- Moskowitz, M.A.; Grotta, J.C.; Koroshetz, W.J.; Stroke Progress Review Group; National Institute of Neurological Disorders and Stroke. The NINDS Stroke Progress Review Group final analysis and recommendations. Stroke 2013, 44, 2343–2350. [Google Scholar] [CrossRef] [Green Version]
- Liu, K.; Li, Z.; Wu, T.; Ding, S. Role of Rho Kinase in Microvascular Damage Following Cerebral Ischemia Reperfusion in Rats. Int. J. Mol. Sci. 2011, 12, 1222–1231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stamatovic, S.M.; Keep, R.; Kunkel, S.L.; Andjelkovic, A.V. Potential role of MCP-1 in endothelial cell tight junction ‘opening’: Signaling via Rho and Rho kinase. J. Cell Sci. 2003, 116, 4615–4628. [Google Scholar] [CrossRef] [Green Version]
- Seo, J.; Maki, T.; Miyamoto, N.; Choi, Y.; Chung, K.; Hamanaka, G.; Park, J.; Mandeville, E.; Takase, H.; Hayakawa, K.; et al. AKAP12 Supports Blood-Brain Barrier Integrity against Ischemic Stroke. Int. J. Mol. Sci. 2020, 21, 9078. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Engelhardt, B.; Sorokin, L. The blood–brain and the blood–cerebrospinal fluid barriers: Function and dysfunction. Semin. Immunopathol. 2009, 31, 497–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brabeck, C.; Mittelbronn, M.; Bekure, K.; Meyermann, R.; Schluesener, H.J.; Schwab, J.M. Effect of focal cerebral infarctions on le-sional RhoA and RhoB expression. Arch. Neurol. 2003, 60, 1245–1249. [Google Scholar] [CrossRef] [PubMed]
- Yano, K.; Kawasaki, K.; Hattori, T.; Tawara, S.; Toshima, Y.; Ikegaki, I.; Sasaki, Y.; Satoh, S.; Asano, T.; Seto, M. Demonstration of eleva-tion and localization of Rho-kinase activity in the brain of a rat model of cerebral infarction. Eur. J. Pharmacol. 2008, 594, 77–83. [Google Scholar] [CrossRef]
- LeComte, M.D.; Shimada, I.S.; Sherwin, C.; Spees, J.L. Notch1–STAT3–ETBR signaling axis controls reactive astrocyte proliferation after brain injury. Proc. Natl. Acad. Sci. USA 2015, 112, 8726–8731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujita, Y.; Yamashita, T. Axon growth inhibition by RhoA/ROCK in the central nervous system. Front. Neurosci. 2014, 8, 338. [Google Scholar] [CrossRef]
- Siebold, C.; Yamashita, T.; Monnier, P.; Mueller, B.K.; Pasterkamp, R.J. RGMs: Structural Insights, Molecular Regulation, and Downstream Signaling. Trends Cell Biol. 2017, 27, 365–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamashita, T.; Mueller, B.K.; Hata, K. Neogenin and repulsive guidance molecule signaling in the central nervous system. Curr. Opin. Neurobiol. 2007, 17, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Hata, K.; Fujitani, M.; Yasuda, Y.; Doya, H.; Saito, T.; Yamagishi, S.; Mueller, B.K.; Yamashita, T. RGMa inhibition promotes axonal growth and recovery after spinal cord injury. J. Cell Biol. 2006, 173, 47–58. [Google Scholar] [CrossRef] [Green Version]
- Monnier, P.P.; Sierra, A.; Schwab, J.; Henke-Fahle, S.; Mueller, B.K. The Rho/ROCK pathway mediates neurite growth-inhibitory activity associated with the chondroitin sulfate proteoglycans of the CNS glial scar. Mol. Cell. Neurosci. 2003, 22, 319–330. [Google Scholar] [CrossRef]
- Thored, P.; Arvidsson, A.; Cacci, E.; Ahlenius, H.; Kallur, T.; Darsalia, V.; Ekdahl, C.T.; Kokaia, Z.; Lindvall, O. Persistent Production of Neurons from Adult Brain Stem Cells During Recovery after Stroke. Stem Cells 2006, 24, 739–747. [Google Scholar] [CrossRef]
- Hayashi, T.; Noshita, N.; Sugawara, T.; Chan, P.H. Temporal profile of angiogenesis and expression of related genes in the brain after ischemia. J. Cereb. Blood Flow Metab. 2003, 23, 166–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruan, L.; Wang, B.; ZhuGe, Q.; Jin, K. Coupling of neurogenesis and angiogenesis after ischemic stroke. Brain Res. 2015, 1623, 166–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robin, A.M.; Zhang, Z.G.; Wang, L.; Zhang, R.L.; Katakowski, M.; Zhang, L.; Wang, Y.; Zhang, C.; Chopp, M. Stromal Cell-Derived Factor 1α Mediates Neural Progenitor Cell Motility after Focal Cerebral Ischemia. Br. J. Pharmacol. 2005, 26, 125–134. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.S.; Zhang, Z.G.; Zhang, R.L.; Gregg, S.; Morris, D.C.; Wang, Y.; Chopp, M. Stroke Induces Gene Profile Changes Associated with Neurogenesis and Angiogenesis in Adult Subventricular Zone Progenitor Cells. Br. J. Pharmacol. 2007, 27, 564–574. [Google Scholar] [CrossRef]
- Ding, J.; Yu, J.Z.; Li, Q.Y.; Wang, X.; Lu, C.Z.; Xiao, B.G. Rho kinase inhibitor Fasudil induces neuroprotection and neurogenesis partially through astrocyte-derived G-CSF. Brain Behav. Immun. 2009, 23, 1083–1088. [Google Scholar] [CrossRef]
- Dai, R.-L.; Zhu, S.-Y.; Xia, Y.-P.; Mao, L.; Mei, Y.-W.; Yao, Y.-F.; Xue, Y.-M.; Hu, B. Sonic Hedgehog Protects Cortical Neurons Against Oxidative Stress. Neurochem. Res. 2011, 36, 67–75. [Google Scholar] [CrossRef] [PubMed]
- He, Q.W.; Xia, Y.P.; Chen, S.C.; Wang, Y.; Huang, M.; Huang, Y.; Li, J.Y.; Li, Y.N.; Gao, Y.; Mao, L.; et al. Astrocyte-derived sonic hedgehog contributes to angiogenesis in brain microvascular endothelial cells via RhoA/ROCK pathway after oxy-gen-glucose deprivation. Mol. Neurobiol. 2013, 47, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Chen, Z.; Wen, J. RhoA/ROCK signaling pathway and astrocytes in ischemic stroke. Metab. Brain Dis. 2021, 36, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Z.Y.; Feng, J. Constraint-induced movement therapy enhances angiogenesis and neurogenesis after cerebral ischemia/reperfusion. Neural Regen. Res. 2019, 14, 1743–1754. [Google Scholar] [PubMed]
- Hara, M.; Takayasu, M.; Watanabe, K.; Noda, A.; Takagi, T.; Suzuki, Y.; Yoshida, J. Protein kinase inhibition by fasudil hydrochlo-ride promotes neurological recovery after spinal cord injury in rats. J. Neurosurg. 2000, 93, 94–101. [Google Scholar] [PubMed]
- Fournier, A.E.; Takizawa, B.T.; Strittmatter, S. Rho Kinase Inhibition Enhances Axonal Regeneration in the Injured CNS. J. Neurosci. 2003, 23, 1416–1423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boato, F.; Hendrix, S.; Huelsenbeck, S.C.; Hofmann, F.; Große, G.; Djalali, S.; Klimaschewski, L.; Auer, M.; Just, I.; Ahnert-Hilger, G.; et al. C3 peptide enhances recovery from spinal cord injury by improved regenerative growth of descending fiber tracts. J. Cell Sci. 2010, 123, 1652–1662. [Google Scholar] [CrossRef] [Green Version]
- Fehlings, M.G.; Kim, K.D.; Aarabi, B.; Rizzo, M.; Bond, L.M.; McKerracher, L.; Vaccaro, A.R.; Okonkwo, D.O. Rho Inhibitor VX-210 in Acute Traumatic Subaxial Cervical Spinal Cord Injury: Design of the SPinal Cord Injury Rho INhibition InvestiGation (SPRING) Clinical Trial. J. Neurotrauma 2018, 35, 1049–1056. [Google Scholar] [CrossRef]
- Sung, J.K.; Miao, L.; Calvert, J.W.; Huang, L.; Louis Harkey, H.; Zhang, J.H. A possible role of RhoA/Rho-kinase in experimental spinal cord injury in rat. Brain Res. 2003, 959, 29–38. [Google Scholar] [CrossRef]
- Fu, P.-C.; Tang, R.-H.; Wan, Y.; Xie, M.-J.; Wang, W.; Luo, X.; Yu, Z.-Y. ROCK inhibition with fasudil promotes early functional recovery of spinal cord injury in rats by enhancing microglia phagocytosis. Acta Acad. Med. Wuhan 2016, 36, 31–36. [Google Scholar] [CrossRef]
- Ohbuchi, M.; Kimura, T.; Nishikawa, T.; Horiguchi, T.; Fukuda, M.; Masaki, Y. Neuroprotective Effects of Fasudil, a Rho-Kinase Inhibitor, After Spinal Cord Ischemia and Reperfusion in Rats. Anesthesia Analg. 2018, 126, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.-G.; Hou, X.-L.; Chen, Y.; Yin, H. Combination of fasudil and celecoxib promotes the recovery of injured spinal cord in rats better than celecoxib or fasudil alone. Neural Regen. Res. 2015, 10, 1836–1840. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Joshi, H.P.; Kim, K.-T.; Kim, Y.Y.; Yeo, K.; Choi, H.; Kim, Y.W.; Choi, U.-Y.; Kumar, H.; Sohn, S.; et al. Combined Treatment with Fasudil and Menthol Improves Functional Recovery in Rat Spinal Cord Injury Model. Biomed 2020, 8, 258. [Google Scholar] [CrossRef]
- Streit, W.J.; Conde, J.R.; Fendrick, S.E.; Flanary, B.E.; Mariani, C.L. Role of microglia in the central nervous system’s immune re-sponse. Neurol Res. 2005, 27, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Gitik, M.; Reichert, F.; Rotshenker, S. Cytoskeleton plays a dual role of activation and inhibition in myelin and zymosan phagocytosis by microglia. FASEB J. 2010, 24, 2211–2221. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.-J.; Yu, Z.-Y.; Yang, H.-J.; Xie, M.-J.; Wang, W.; Luo, X. Cellular expression profile of RhoA in rats with spinal cord injury. Acta Acad. Med. Wuhan 2014, 34, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.C.; Tang, R.H.; Yu, Z.Y.; Xie, M.J.; Wang, W.; Luo, X. The Rho-associated kinase inhibitors Y27632 and fasudil promote micro-glial migration in the spinal cord via the ERK signaling pathway. Neural Regen Res. 2018, 13, 677–683. [Google Scholar] [PubMed]
- Serbanovic-Canic, J.; Cvejic, A.; Soranzo, N.; Stemple, D.L.; Ouwehand, W.H.; Freson, K. Silencing of RhoA nucleotide exchange factor, ARHGEF3, reveals its unexpected role in iron uptake. Blood 2011, 118, 4967–4976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, L.; Qian, Z.-Y.; Li, X.-Y.; Yang, D.-S.; Lei, B.-J.; Li, H.-J.; Hong, X. Disrupting RhoA activity by blocking Arhgef3 expression mitigates microglia-induced neuroinflammation post spinal cord contusion. J. Neuroimmunol. 2021, 359, 577688. [Google Scholar] [CrossRef]
- Luo, M.; Li, Y.Q.; Lu, Y.F.; Wu, Y.; Liu, R.; Zheng, Y.R.; Yin, M. Exploring the potential of RhoA inhibitors to improve exer-cise-recoverable spinal cord injury: A systematic review and meta-analysis. J. Chem. Neuroanat. 2021, 111, 101879. [Google Scholar] [CrossRef] [PubMed]
- Bulygin, K.V.; Beeraka, N.M.; Saitgareeva, A.R.; Nikolenko, V.N.; Gareev, I.; Beylerli, O.; Akhmadeeva, L.R.; Mikhaleva, L.M.; Solis, L.F.T.; Herrera, A.S.; et al. Can miRNAs Be Considered as Diagnostic and Therapeutic Molecules in Ischemic Stroke Pathogenesis?—Current Status. Int. J. Mol. Sci. 2020, 21, 6728. [Google Scholar] [CrossRef] [PubMed]
- Meissner, L.; Gallozzi, M.; Balbi, M.; Schwarzmaier, S.; Tiedt, S.; Terpolilli, N.A.; Plesnila, N. Temporal Profile of MicroRNA Ex-pression in Contused Cortex after Traumatic Brain Injury in Mice. J. Neurotrauma 2016, 33, 713–720. [Google Scholar] [CrossRef]
- Jiang, C.; Dong, N.; Feng, J.; Hao, M. MiRNA-190 exerts neuroprotective effects against ischemic stroke through Rho/Rho-kinase pathway. Pflügers Arch.-Eur. J. Physiol. 2021, 473, 121–130. [Google Scholar] [CrossRef]
- Han, X.R.; Wen, X.; Wang, Y.J.; Wang, S.; Shen, M.; Zhang, Z.F.; Fan, S.H.; Shan, Q.; Wang, L.; Li, M.Q.; et al. Protective effects of microRNA-431 against cerebral ischemia-reperfusion injury in rats by targeting the Rho/Rho-kinase signaling pathway. J. Cell. Physiol. 2018, 233, 5895–5907. [Google Scholar] [CrossRef]
- Si, W.; Ye, S.; Ren, Z.; Liu, X.; Wu, Z.; Li, Y.; Zhou, J.; Zhang, S.; Li, Y.; Deng, R.; et al. miR-335 promotes stress granule formation to inhibit apoptosis by targeting ROCK2 in acute ischemic stroke. Int. J. Mol. Med. 2019, 43, 1452–1466. [Google Scholar] [CrossRef]
- Beltran, E.G.; Moschou, P.N.; Smertenko, A.; Bozhkov, P. Tudor Staphylococcal Nuclease Links Formation of Stress Granules and Processing Bodies with mRNA Catabolism in Arabidopsis. Plant Cell 2015, 27, 926–943. [Google Scholar] [CrossRef] [Green Version]
- Sampuda, K.M.; Riley, M.; Boyd, L. Stress induced nuclear granules form in response to accumulation of misfolded proteins in Caenorhabditis elegans. BMC Cell Biol. 2017, 18, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, H.; Gao, S.; Wang, L.; Wei, Y.; Zhang, M. Overexpression of miR-582-5p Inhibits the Apoptosis of Neuronal Cells after Cerebral Ischemic Stroke Through Regulating PAR-1/Rho/Rho Axis. J. Stroke Cerebrovasc. Dis. 2019, 28, 149–155. [Google Scholar] [CrossRef]
- Stein, E.S.; Itsekson-Hayosh, Z.; Aronovich, A.; Reisner, Y.; Bushi, D.; Pick, C.G.; Tanne, D.; Chapman, J.; Vlachos, A.; Maggio, N. Thrombin induces ischemic LTP (iLTP): Implications for synaptic plasticity in the acute phase of ischemic stroke. Sci. Rep. 2015, 5, srep07912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olson, E.E.; Lyuboslavsky, P.; Traynelis, S.F.; McKeon, R.J. PAR-1 Deficiency Protects against Neuronal Damage and Neurologic Deficits after Unilateral Cerebral Hypoxia/Ischemia. Br. J. Pharmacol. 2004, 24, 964–971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Liu, W.; Zhang, Y.; Hu, Z.; Guo, H.; Lv, J.; Du, H. Dexmedetomidine had neuroprotective effects on hippocampal neu-ronal cells via targeting lncRNA SHNG16 mediated microRNA-10b-5p/BDNF axis. Mol. Cell. Biochem. 2020, 469, 41–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Zhang, Y. lncRNA ZFAS1 Improves Neuronal Injury and Inhibits Inflammation, Oxidative Stress, and Apoptosis by Sponging miR-582 and Upregulating NOS3 Expression in Cerebral Ischemia/Reperfusion Injury. Inflammation 2020, 43, 1337–1350. [Google Scholar] [CrossRef]
- Wang, H.; Zheng, X.; Jin, J.; Zheng, L.; Guan, T.; Huo, Y.; Xie, S.; Wu, Y.; Chen, W. LncRNA MALAT1 silencing protects against cerebral ischemia-reperfusion injury through miR-145 to regulate AQP4. J. Biomed. Sci. 2020, 27, 40. [Google Scholar] [CrossRef] [Green Version]
- Xiang, Y.; Zhang, Y.; Xia, Y.; Zhao, H.; Liu, A.; Chen, Y. LncRNA MEG3 targeting miR-424-5p via MAPK signaling pathway mediates neuronal apoptosis in ischemic stroke. Aging (Albany NY) 2020, 12, 3156–3174. [Google Scholar] [CrossRef] [PubMed]
- Bao, M.-H.; Szeto, V.; Yang, B.B.; Zhu, S.-Z.; Sun, H.-S.; Feng, Z.-P. Long non-coding RNAs in ischemic stroke. Cell Death Dis. 2018, 9, 281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, F.; Wei, W.-P.; Liu, Y.-B.; Wang, Y.; Zhang, H.-Y.; Liu, R. Long noncoding RNA XIST enhances cerebral ischemia-reperfusion injury by regulating miR-486-5p and GAB2. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 2013–2020. [Google Scholar]
- Wang, J.; Fu, Z.; Wang, M.; Lu, J.; Yang, H.; Lu, H. Knockdown of XIST Attenuates Cerebral Ischemia/Reperfusion Injury Through Regulation of miR-362/ROCK2 Axis. Neurochem. Res. 2021, 46, 2167–2180. [Google Scholar] [CrossRef]
- Zeng, J.; Zhu, L.; Liu, J.; Zhu, T.; Xie, Z.; Sun, X.; Zhang, H. Metformin Protects against Oxidative Stress Injury Induced by Ische-mia/Reperfusion via Regulation of the lncRNA-H19/miR-148a-3p/Rock2 Axis. Oxid. Med. Cell. Longev. 2019, 2019, 8768327. [Google Scholar] [CrossRef]
- Wang, J.; Cao, B.; Han, D.; Sun, M.; Feng, J. Long Non-coding RNA H19 Induces Cerebral Ischemia Reperfusion Injury via Activation of Autophagy. Aging Dis. 2017, 8, 71–84. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Y.; Yu, C.; Qin, W. LncRNA SNHG14 promotes inflammatory response induced by cerebral ischemia/reperfusion injury through regulating miR-136-5p/ROCK1. Cancer Gene Ther. 2019, 26, 234–247. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Li, X. LncRNA ROR is involved in cerebral hypoxia/reoxygenation-induced injury in PC12 cells via regulating miR-135a-5p/ROCK1/2. Am. J. Transl. Res. 2019, 11, 6145–6158. [Google Scholar]
- Wang, Y.; Xu, Z.; Jiang, J.; Xu, C.; Kang, J.; Xiao, L.; Wu, M.; Xiong, J.; Guo, X.; Liu, H. Endogenous miRNA sponge lincRNA-RoR regu-lates Oct4, Nanog, and Sox2 in human embryonic stem cell self-renewal. Dev. Cell. 2013, 25, 69–80. [Google Scholar] [CrossRef] [Green Version]
- Li, P.; Teng, Z.-Q.; Liu, C.-M. Extrinsic and Intrinsic Regulation of Axon Regeneration by MicroRNAs after Spinal Cord Injury. Neural Plast. 2016, 2016, 1279051. [Google Scholar] [CrossRef] [Green Version]
- Yan, H.; Hong, P.; Jiang, M.; Li, H. MicroRNAs as potential therapeutics for treating spinal cord injury. Neural Regen. Res. 2012, 7, 1352–1359. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Ding, F.; Gu, X. Non-coding RNAs as Emerging Regulators of Neural Injury Responses and Regeneration. Neurosci. Bull. 2016, 32, 253–264. [Google Scholar] [CrossRef] [Green Version]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef] [Green Version]
- Qi, L.; Tang, Y.-G.; Wang, L.; He, W.; Pan, H.-H.; Nie, R.-R.; Can, Y. Role of Rho-mediated ROCK-Semaphorin3A signaling pathway in the pathogenesis of Parkinson’s disease in a mouse model. J. Neurol. Sci. 2016, 370, 21–26. [Google Scholar] [CrossRef]
- Han, F.; Huo, Y.; Huang, C.-J.; Chen, C.-L.; Ye, J. MicroRNA-30b promotes axon outgrowth of retinal ganglion cells by inhibiting Semaphorin3A expression. Brain Res. 2015, 1611, 65–73. [Google Scholar] [CrossRef]
- Wang, X.; Li, B.; Wang, Z.; Wang, F.; Liang, J.; Chen, C.; Zhao, L.; Zhou, B.; Guo, X.; Ren, L.; et al. miR-30b Promotes spinal cord sensory function recovery via the Sema3A/NRP-1/PlexinA1/RhoA/ROCK Pathway. J. Cell. Mol. Med. 2020, 24, 12285–12297. [Google Scholar] [CrossRef] [PubMed]
- Sepramaniam, S.; Tan, J.R.; Tan, K.S.; DeSilva, D.A.; Tavintharan, S.; Woon, F.P.; Wang, C.W.; Yong, F.L.; Karolina, D.S.; Kaur, P.; et al. Circulating microRNAs as biomarkers of acute stroke. Int. J. Mol. Sci. 2014, 15, 1418–1432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, X.; Huang, G.; Wang, S.; Long, M.; Tang, X.; Feng, D.; Zhou, Q. Extracellular vesicles derived from mesenchymal stem cells con-taining microRNA-381 protect against spinal cord injury in a rat model via the BRD4/WNT5A axis. Bone Jt. Res. 2021, 10, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, T.; Koda, M.; Kitajo, K.; Yamazaki, M.; Takahashi, K.; Kikuchi, A.; Yamashita, T. Wnt-Ryk Signaling Mediates Axon Growth Inhibition and Limits Functional Recovery after Spinal Cord Injury. J. Neurotrauma 2009, 26, 955–964. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Chen, J.; Peng, S.; Zhang, J. Effects of Wnt5a protein on proliferation and apoptosis in JAR choriocarcinoma cells. Mol. Med. Rep. 2010, 4, 99–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Battum, E.Y.; Verhagen, M.G.; Vangoor, V.R.; Fujita, Y.; Derijck, A.A.H.A.; O’Duibhir, E.; Giuliani, G.; de Gunst, T.; Adolfs, Y.; Le-lieveld, D.; et al. An Image-Based miRNA Screen Identifies miRNA-135s As Regulators of CNS Axon Growth and Regeneration by Targeting Krüppel-like Factor 4. J. Neurosci. 2018, 38, 613–630. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liao, S.; Quan, H.; Lin, Y.; Li, J.; Yang, Q. Involvement of microRNA-135a-5p in the Protective Effects of Hydrogen Sulfide Against Parkinson’s Disease. Cell. Physiol. Biochem. 2016, 40, 18–26. [Google Scholar] [CrossRef] [PubMed]
- García-Morales, V.; Rodríguez-Bey, G.; Gómez-Pérez, L.; Domínguez-Vías, G.; González-Forero, D.; Portillo, F.; Campos-Caro, A.; Gento-Caro, Á.; Issaoui, N.; Soler, R.M.; et al. Sp1-regulated expression of p11 contributes to motor neuron degeneration by membrane insertion of TASK1. Nat. Commun. 2019, 10, 3784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, J.C.; Tönges, L.; Barski, E.; Michel, U.; Bahr, M.; Lingor, P. ROCK2 is a major regulator of axonal degeneration, neuronal death and axonal regeneration in the CNS. Cell Death Dis. 2014, 5, e1225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, N.; Yang, Y.; Pang, M.; Du, C.; Chen, Y.; Li, S.; Tian, Z.; Feng, F.; Wang, Y.; Chen, Z.; et al. MicroRNA-135a-5p Promotes the Functional Recovery of Spinal Cord Injury by Targeting SP1 and ROCK. Mol. Ther.-Nucleic Acids 2020, 22, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Deniaud, E.; Baguet, J.; Mathieu, A.L.; Pagès, G.; Marvel, J.; Leverrier, Y. Overexpression of Sp1 transcription factor induces apoptosis. Oncogene 2006, 25, 7096–7105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torabi, B.; Flashner, S.; Beishline, K.; Sowash, A.; Donovan, K.; Bassett, G.; Azizkhan-Clifford, J. Caspase cleavage of transcription factor Sp1 enhances apoptosis. Apoptosis 2017, 23, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Uchida, A.; Oh-hashi, K.; Kiuchi, K.; Hirata, Y. Manganese regulates caspase-3 gene promoter activity by inducing Sp1 phosphorylation in PC12 cells. Toxicology 2012, 302, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-Y.; Kim, Y.-W.; Cho, S.-J.; Lee, M.K.; Kook, M.-C.; Lee, J.H.; Lee, S.S.; Ashktorab, H.; Smoot, D.T.; Ryu, K.W.; et al. MicroRNA 135a Suppresses Lymph Node Metastasis through Down-Regulation of ROCK1 in Early Gastric Cancer. PLoS ONE 2014, 9, e85205. [Google Scholar] [CrossRef] [PubMed]
- Kroiss, A.; Vincent, S.; Decaussin-Petrucci, M.; Meugnier, E.; Viallet, J.; Ruffion, A.; Chalmel, F.; Samarut, J.; Allioli, N. Andro-gen-regulated microRNA-135a decreases prostate cancer cell migration and invasion through downregulating ROCK1 and ROCK2. Oncogene 2015, 34, 2846–2855. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Miao, L.; Yang, L.; Liang, F.; Wang, Q.; Zhuang, P.; Sun, Y.; Hu, Y. AKT-dependent and -independent pathways mediate PTEN deletion-induced CNS axon regeneration. Cell Death Dis. 2019, 10, 203. [Google Scholar] [CrossRef] [PubMed]
- Liz, M.A.; Mar, F.M.; Santos, T.E.; Pimentel, H.I.; Marques, A.M.; Morgado, M.M.; Vieira, S.; Sousa, V.F.; Pemble, H.; Wittmann, T.; et al. Neuronal deletion of GSK3β increases microtubule speed in the growth cone and enhances axon regeneration via CRMP-2 and independently of MAP1B and CLASP2. BMC Biol. 2014, 12, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kajikawa, M.; Noma, K.; Maruhashi, T.; Mikami, S.; Iwamoto, Y.; Iwamoto, A.; Matsumoto, T.; Hidaka, T.; Kihara, Y.; Chayama, K.; et al. Rho-Associated Kinase Activity Is a Predictor of Cardiovascular Outcomes. Hypertension 2014, 63, 856–864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Model | lncRNA/miRNA | Expression after Insult | Target | Effects | Reference |
|---|---|---|---|---|---|
| MCAO/R in rats Hippocampal neuron of rats under OGDR | miR-190 | Decreased | Rho | The overexpression of miR-190 decreased apoptosis. | [95] |
| MCAO/R in rats | miR-431 | Decresed | Rho | The overexpression of miR-431 decreased apoptosis and promoted proliferation. | [96] |
| MCAO in rats PC12 cells in serum-free medium | miR-335 | Decreased | ROCK2 | miR-335 treatment upregulated stress granule formation, alleviated infarction, decreased ROCK2 expression, and apoptosis. | [97] |
| MCAO in mice N2A cells under OGD/R | miR-582-5p | Decreased | PAR-1 | Overexpression of miR-582-5p inhibited the activation of the Rho/ROCK pathway by downregulating proteinase-activated receptors type-1 (PAR-1), reducing apoptosis. | [100] |
| MCAO/R in mice PC12 cells under OGD/R | XIST | Elevated | miR-362 | XIST negatively regulated miR-362. Depletion of XIST attenuated apoptosis and inflammation via miR-362/ROCK2 axis. | [109] |
| miR-362 | Decreased | ROCK2 | |||
| MCAO/R in mice N2a cells under OGB/R | lncRNA-H19 | Elevated | miR-148a-3p | lncRNA-H19 may act as a molecular sponge of miR-148a-3p. lncRNA-H19 altered OGD/R induced apoptosis and oxidative stress via the miR-148a-3p/ROCK2 axis. | [110] |
| miR-148a-3p | Decreased | ROCK2 | |||
| MCAO/R in rats PC12 cells under OGD/R | lncRNA-SNHG14 | Elevated | miR-136-5p | lncRNA-SNHG14 negatively regulated miR-136-5p as its ceRNA. lncRNA-SNHG14 promoted neurological impairment and inflammation via the miR-136-5p/ROCK1 axis. | [112] |
| miR-136-5p | Decreased | ROCK1 | |||
| PC12 cells under OGD/R | lncRNA-ROR | Elevated | miR-135a-5p | lncRNA-ROR promoted oxidative damage and apoptosis via the miR-135a-5p/ROCK1/2 axis. The overexpression of miR-135a-5p decreased cell damage by inhibiting ROCK1/2. | [113] |
| miR-135a-5p | Decreased | ROCK1/2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kimura, T.; Horikoshi, Y.; Kuriyagawa, C.; Niiyama, Y. Rho/ROCK Pathway and Noncoding RNAs: Implications in Ischemic Stroke and Spinal Cord Injury. Int. J. Mol. Sci. 2021, 22, 11573. https://doi.org/10.3390/ijms222111573
Kimura T, Horikoshi Y, Kuriyagawa C, Niiyama Y. Rho/ROCK Pathway and Noncoding RNAs: Implications in Ischemic Stroke and Spinal Cord Injury. International Journal of Molecular Sciences. 2021; 22(21):11573. https://doi.org/10.3390/ijms222111573
Chicago/Turabian StyleKimura, Tetsu, Yuta Horikoshi, Chika Kuriyagawa, and Yukitoshi Niiyama. 2021. "Rho/ROCK Pathway and Noncoding RNAs: Implications in Ischemic Stroke and Spinal Cord Injury" International Journal of Molecular Sciences 22, no. 21: 11573. https://doi.org/10.3390/ijms222111573
APA StyleKimura, T., Horikoshi, Y., Kuriyagawa, C., & Niiyama, Y. (2021). Rho/ROCK Pathway and Noncoding RNAs: Implications in Ischemic Stroke and Spinal Cord Injury. International Journal of Molecular Sciences, 22(21), 11573. https://doi.org/10.3390/ijms222111573






