The Concentration of Organic Acids in Cranberry Juice Modulates the Gut Microbiota in Mice
Abstract
1. Introduction
2. Results
2.1. Cranberry Juices Composition
2.2. Animals
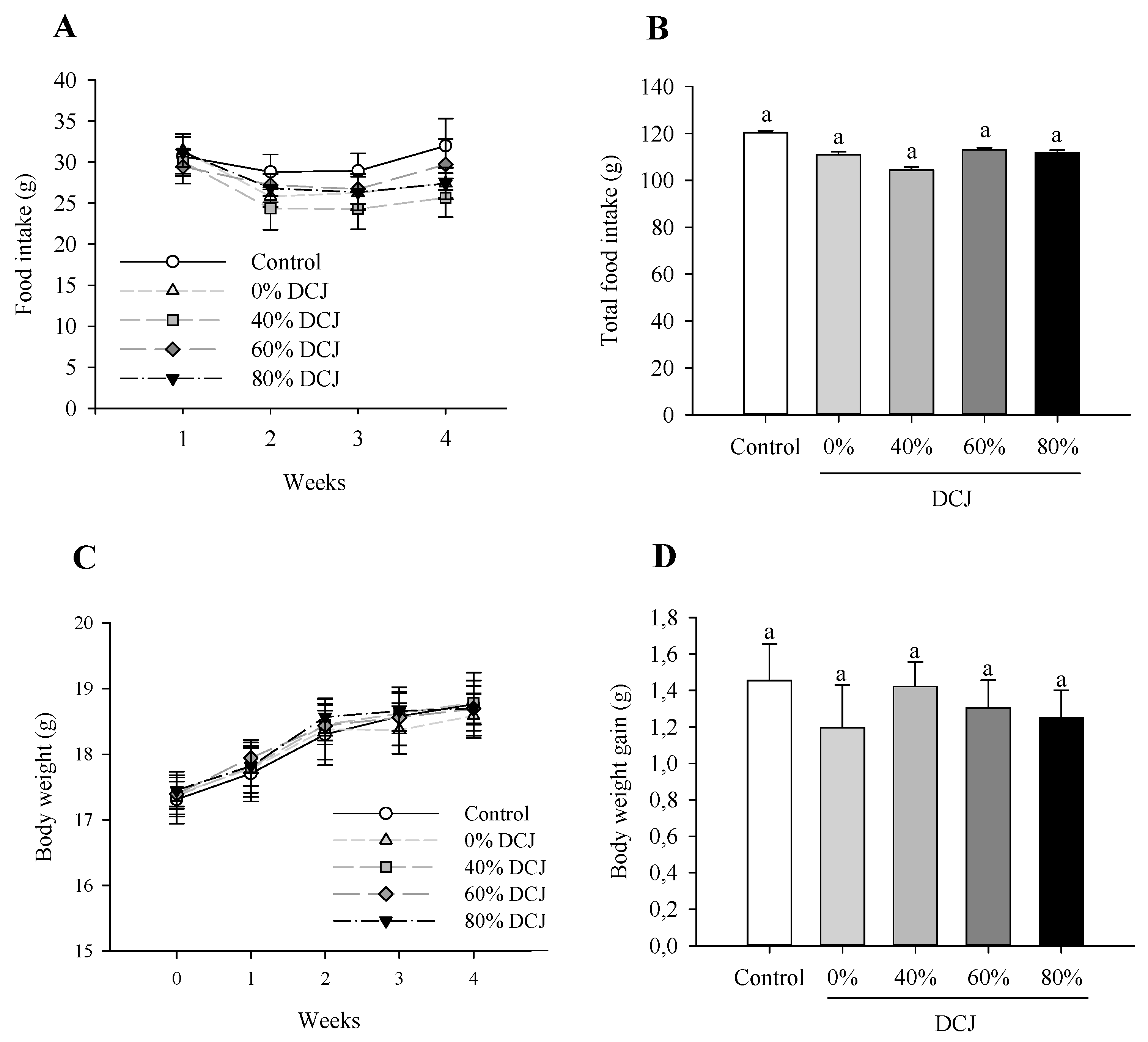
2.2.1. Effect of DCJ on the Food Intake and Body Weight
2.2.2. Feces Analyses
2.2.3. Post-Mortem Observations
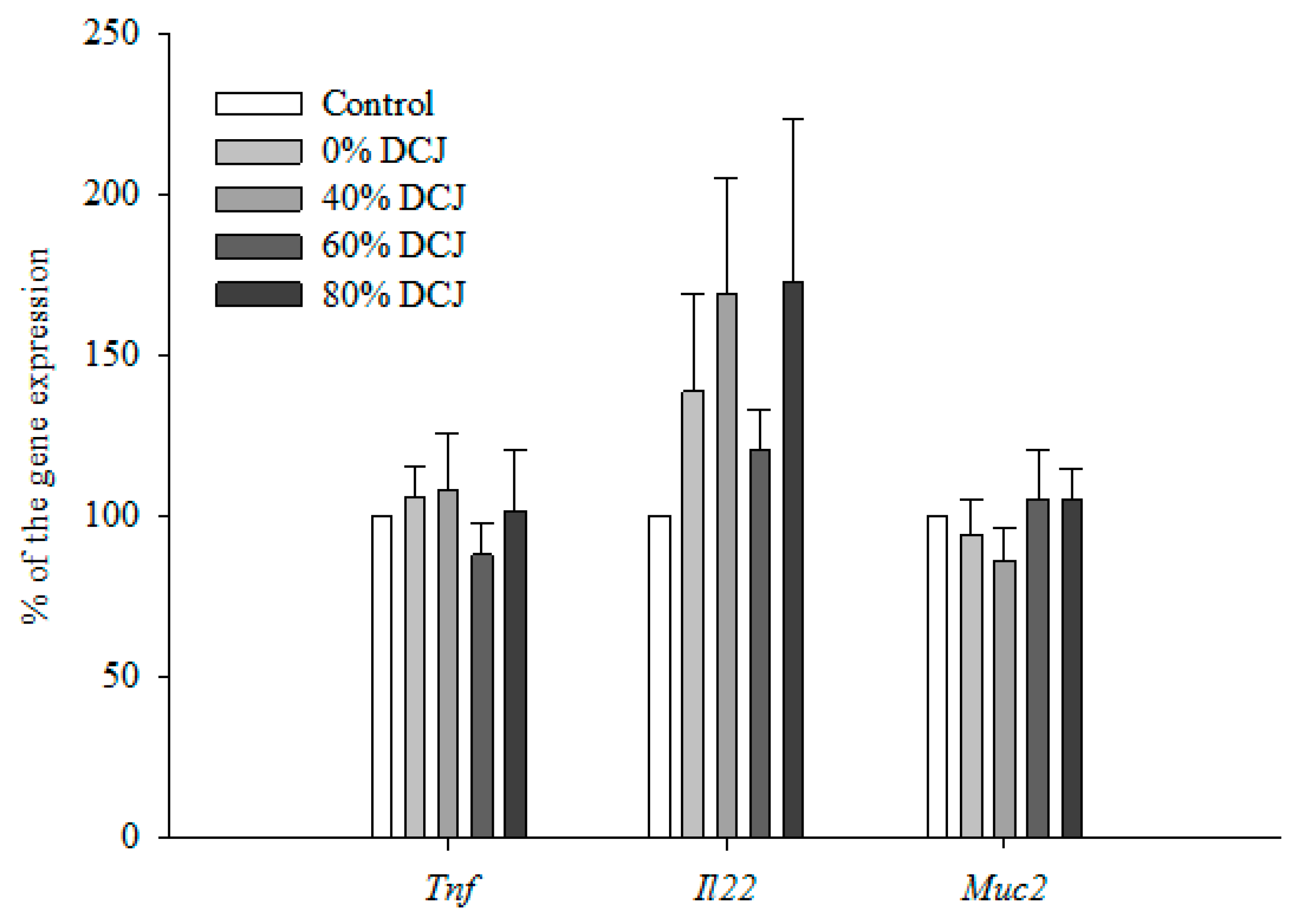
2.2.4. Gene Expression by qRT-PCR
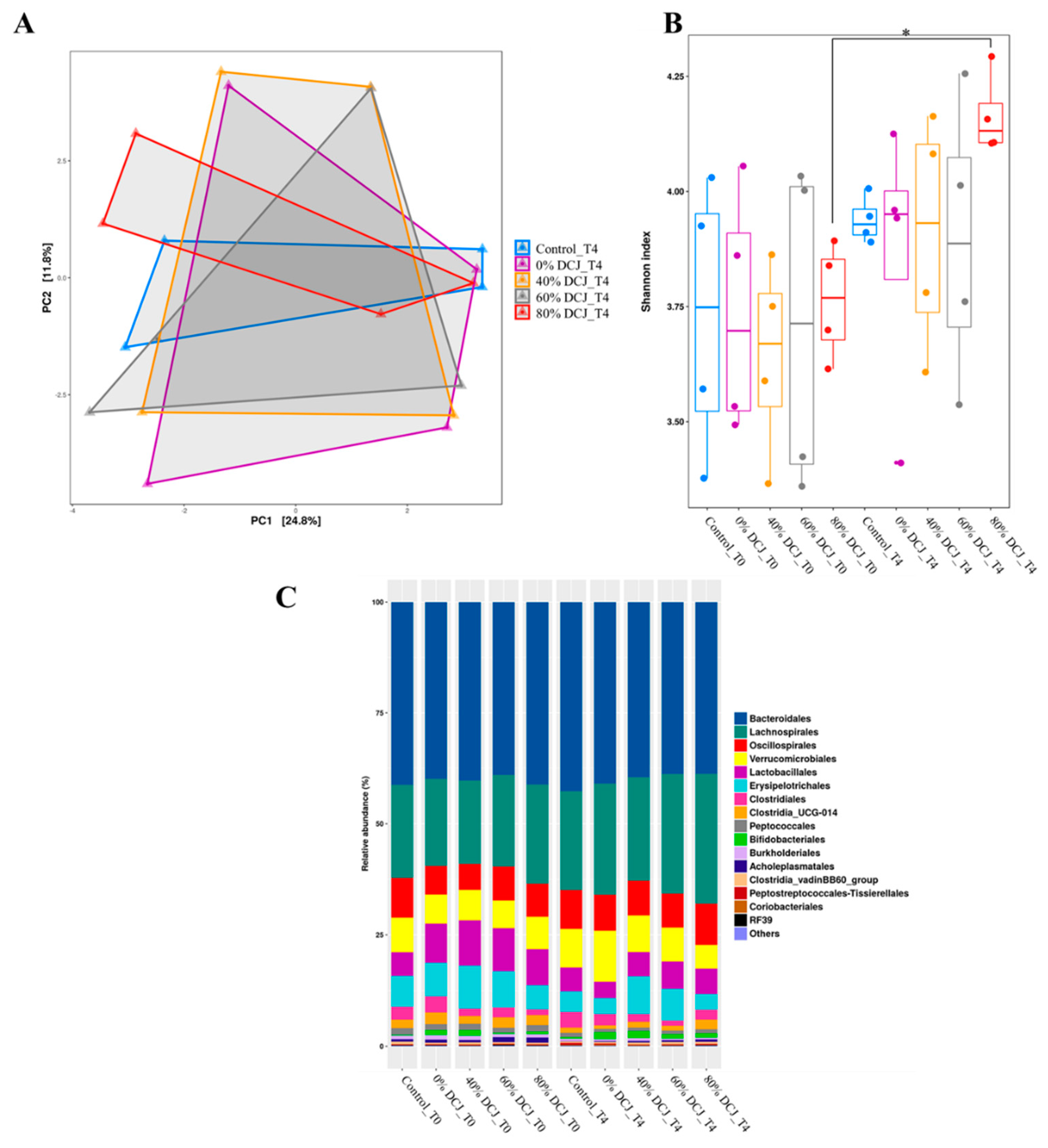
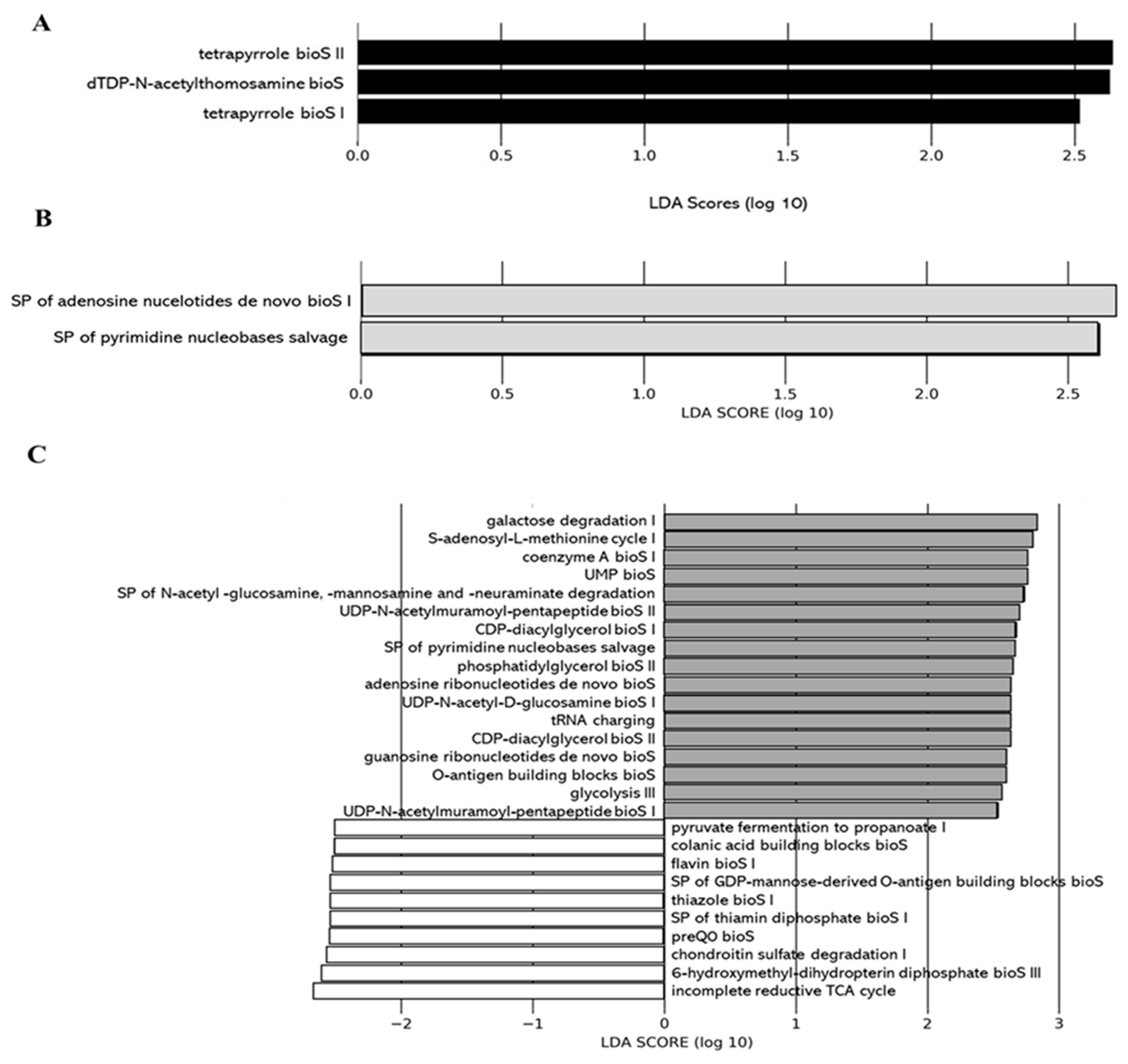
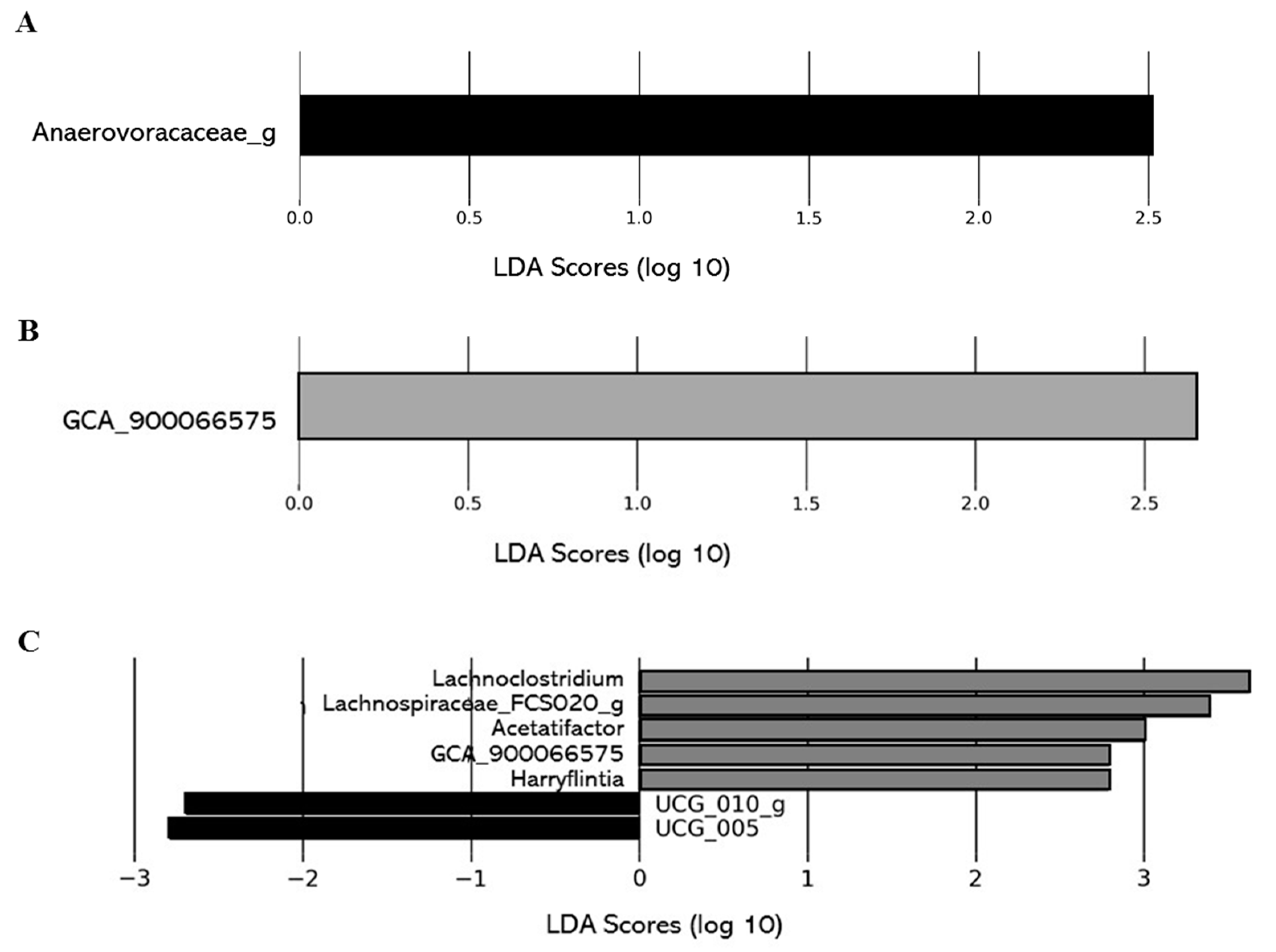
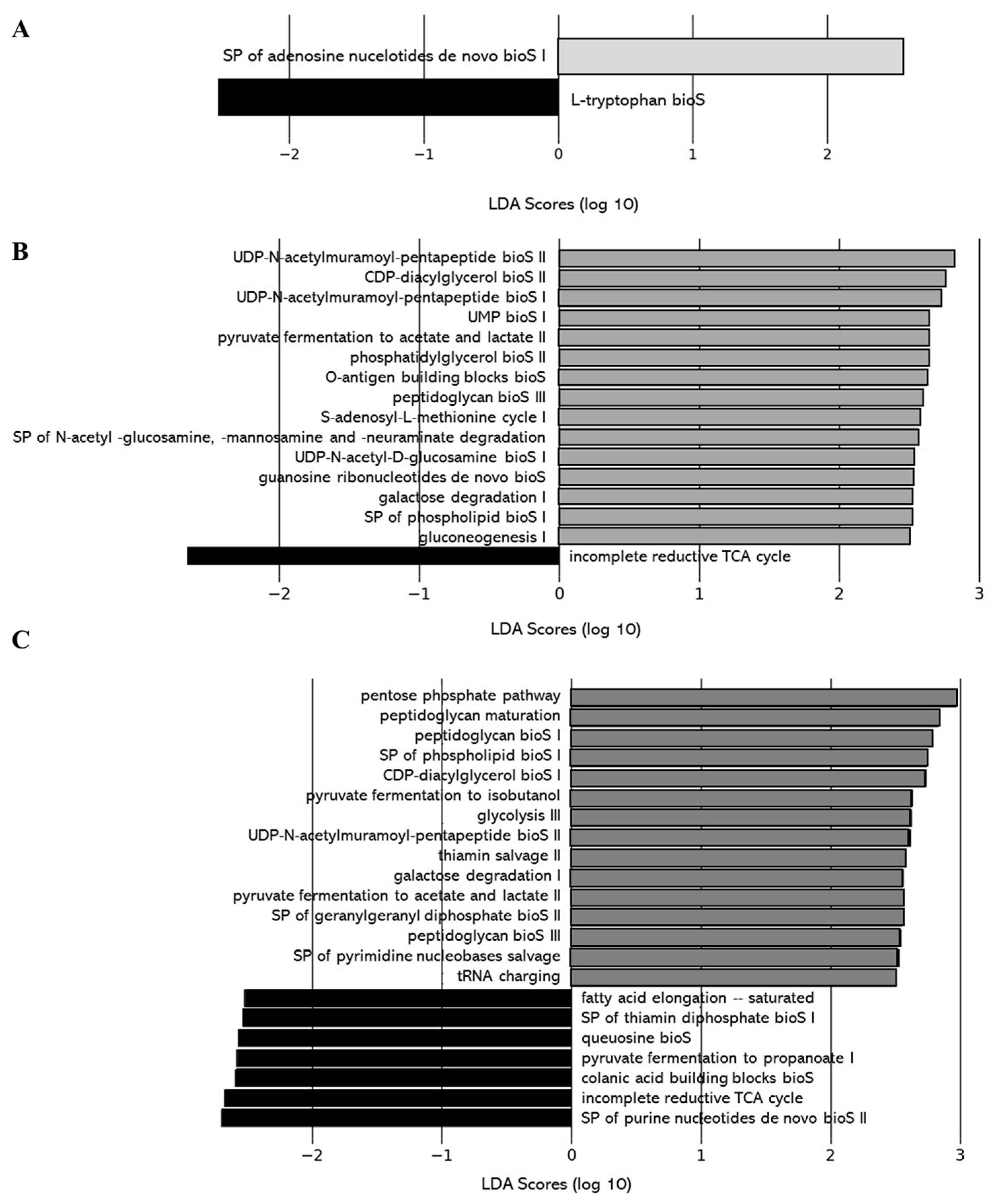
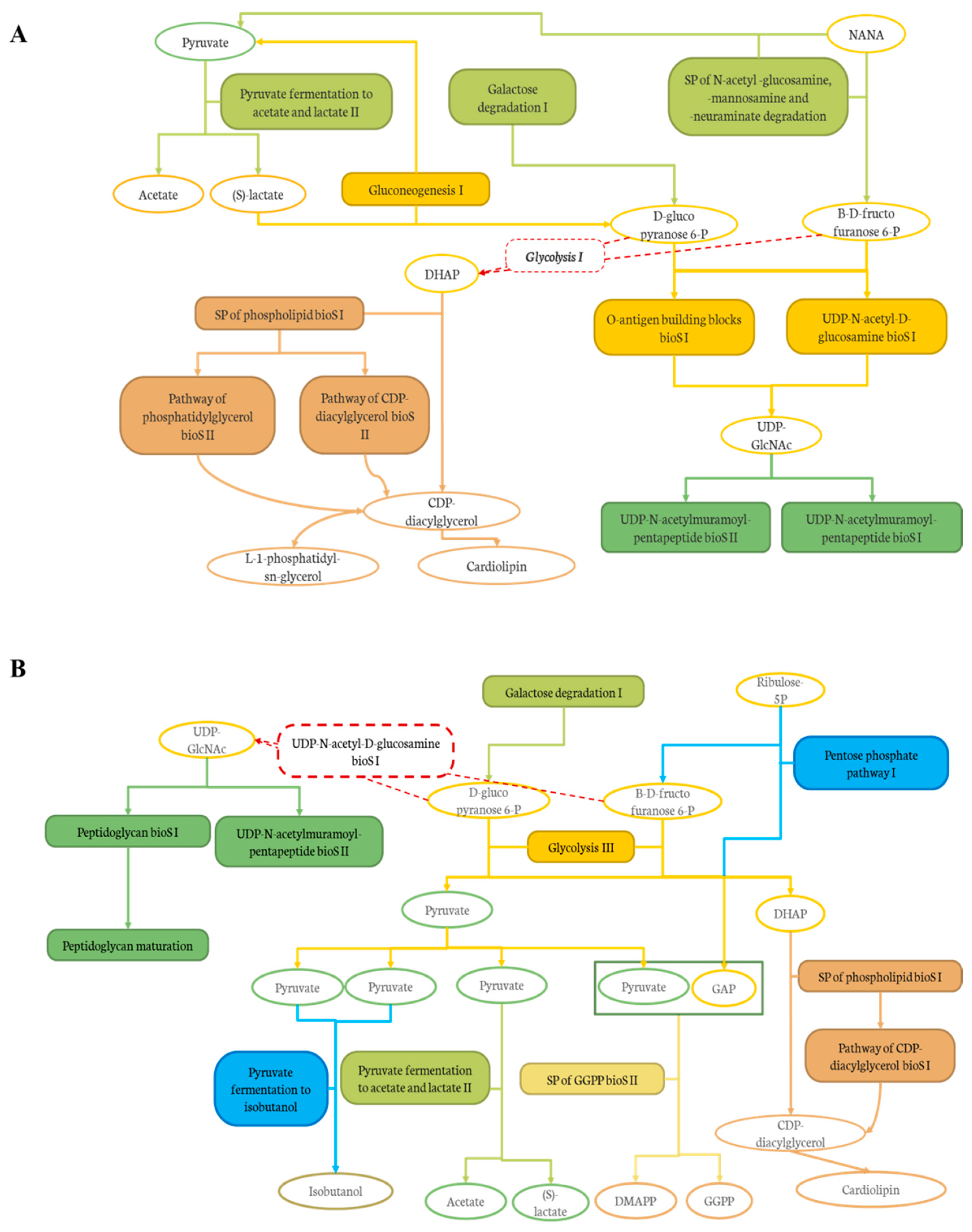
2.2.5. Impacts of DCJ on the Gut Microbiota
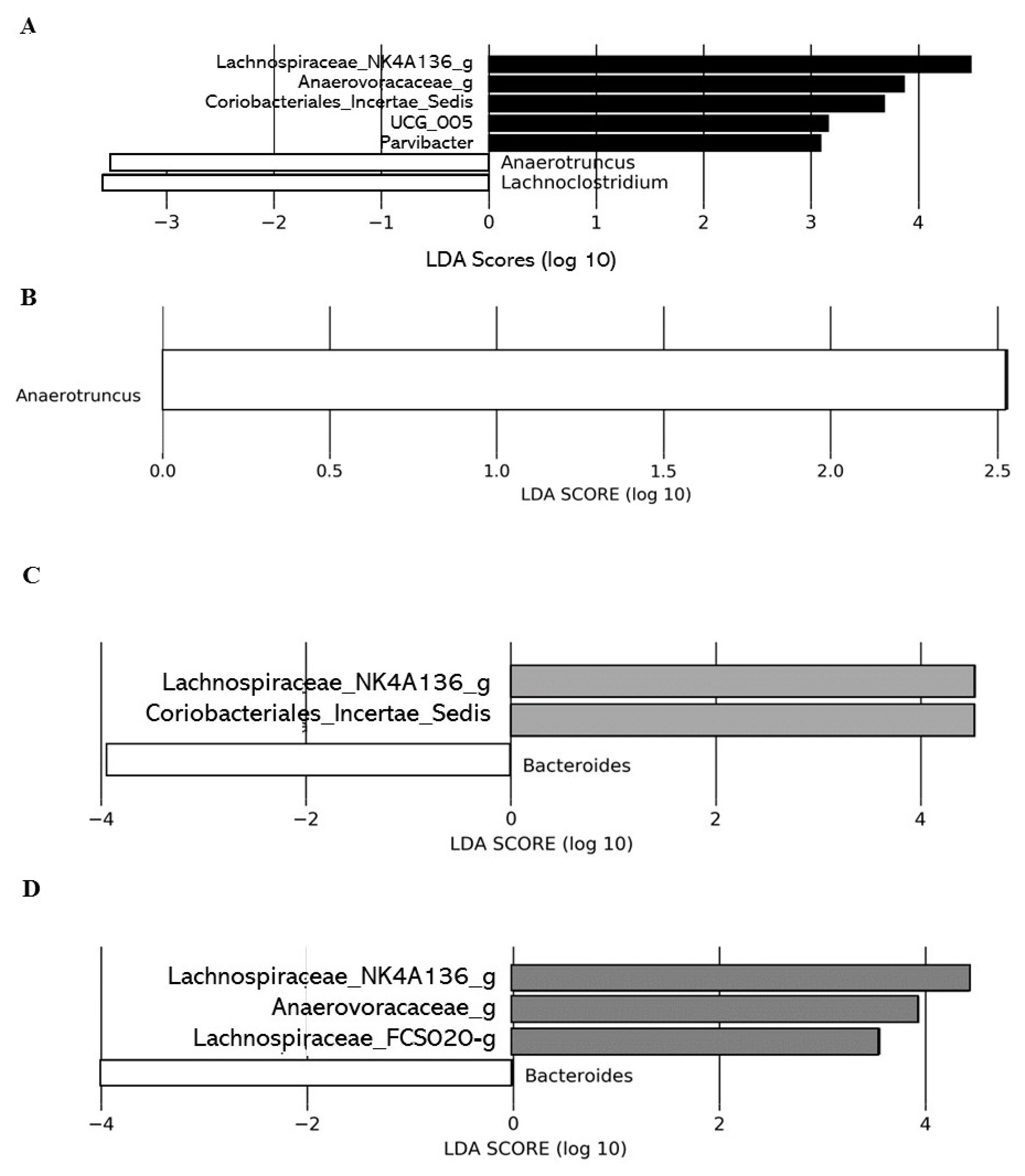
Effect of DCJ Administration on the Composition and the Functions of the Gut Microbiota
Effect of Organic Acid Removal on the Composition and the Functions of the Gut Microbiota
3. Discussion
4. Materials and Methods
4.1. Cranberry Juice
4.1.1. Cranberry Juice and Sample Preparation
4.1.2. Analyses of the Physicochemical Composition of DCJ
pH
Conductivity
Titratable Acidity
Organic Acid Contents
Polyphenol Contents
4.2. Animals
4.2.1. Animals and Dietary Treatments
4.2.2. Analyses Determining Intestinal Inflammation
Occult Blood Testing
Intestine Macroscopic Observations
Gene Expression Analyses by qRT-PCR
4.2.3. Bioinformatics Analyses Determining the Modulation of Gut Microbiota
Fecal Sample Processing and 16S rRNA Gene-Based Sequencing
Gut Microbiota Analyses
Functional Prediction of Gut Bacterial Communities
4.3. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cunningham, D.G.; Vannozzi, S.A.; Turk, R.; Roderick, R.; O’Shea, E.; Brilliant, K. Cranberry phytochemicals and their health benefits. In Nutraceutical Beverages; Shahidi, F., Weerasinghe, D.K., Eds.; American Chemical Society: Washington, DC, USA, 2003; Volume 871, pp. 35–51. [Google Scholar] [CrossRef]
- McKay, D.L.; Chen, C.-Y.O.; Zampariello, C.A.; Blumberg, J.B. Flavonoids and phenolic acids from cranberry juice are bioavailable and bioactive in healthy older adults. Food Chem. 2015, 168, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Mullen, W.; Marks, S.C.; Crozier, A. Evaluation of phenolic compounds in commercial fruit juices and fruit drinks. J. Agric. Food Chem. 2007, 55, 3148–3157. [Google Scholar] [CrossRef]
- Prior, R.L.; Lazarus, S.A.; Cao, G.; Muccitelli, H.; Hammerstone, J.F. Identification of procyanidins and anthocyanins in blueberries and cranberries (Vaccinium spp.) using high-performance liquid chromatography/mass spectrometry. J. Agric. Food Chem. 2001, 49, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Kelm, M.A.; Hammerstone, J.F.; Beecher, G.; Holden, J.; Haytowitz, D.; Prior, R.L. Screening of foods containing proanthocyanidins and their structural characterization using LC-MS/MS and thiolytic degradation. J. Agric. Food Chem. 2003, 51, 7513–7521. [Google Scholar] [CrossRef] [PubMed]
- Neto, C.C.; Krueger, C.G.; Lamoureaux, T.L.; Kondo, M.; Vaisberg, A.J.; Hurta, R.A.; Curtis, S.; Matchett, M.D.; Yeung, H.; Sweeney, M.I.; et al. MALDI-TOF MS characterization of proanthocyanidins from cranberry fruit (Vaccinium macrocarpon) that inhibit tumor cell growth and matrix metalloproteinase expression in vitro. J. Sci. Food Agric. 2006, 86, 18–25. [Google Scholar] [CrossRef]
- Serre, E.; Boutin, Y.; Langevin, M.-E.; Lutin, F.; Pedneault, K.; Lacour, S.; Bazinet, L. Deacidification of cranberry juice protects against disruption of in-vitro intestinal cell barrier integrity. J. Funct. Foods 2016, 26, 208–216. [Google Scholar] [CrossRef]
- Bazinet, L.; Brianceau, S.; Dubé, P.; Desjardins, Y. Evolution of cranberry juice physico-chemical parameters during phenolic antioxidant enrichment by electrodialysis with filtration membrane. Sep. Purif. Technol. 2012, 87, 31–39. [Google Scholar] [CrossRef]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.M.; Shenouda, S.M.; Chung, W.B.; Titas, M.; Kluge, M.A.; Wang, N.; Palmisano, J.; Milbury, P.E.; et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef] [PubMed]
- Duthie, S.J.; Jenkinson, A.M.E.; Crozier, A.; Mullen, W.; Pirie, L.; Kyle, J.; Yap, L.S.; Christen, P.; Duthie, G.G. The effects of cranberry juice consumption on antioxidant status and biomarkers relating to heart disease and cancer in healthy human volunteers. Eur. J. Nutr. 2006, 45, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Bansal, K.; Marwaha, M. Effect of high-molecular-weight component of cranberry on plaque and salivary streptococcus mutans counts in children: An in vivo study. J. Indian Soc. Pedod. Prev. Dent. 2015, 33, 128. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.L.; Blumberg, J.B. Cranberries (Vaccinium macrocarpon) and cardiovascular disease risk factors. Nutr. Rev. 2007, 65, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Neto, C.C.; Amoroso, J.W.; Liberty, A.M. Anticancer activities of cranberry phytochemicals: An update. Mol. Nutr. Food Res. 2008, 52, S18–S27. [Google Scholar] [CrossRef]
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive compounds and antioxidant activity in different types of berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef]
- Wada, L.; Ou, B. Antioxidant activity and phenolic content of oregon caneberries. J. Agric. Food Chem. 2002, 50, 3495–3500. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ma, J.; Pan, K.; Go, V.L.W.; Chen, J.; You, W. Efficacy of cranberry juice on helicobacter pylori infection: A double-blind, randomized placebo-controlled trial. Helicobacter 2005, 10, 139–145. [Google Scholar] [CrossRef]
- Sun, J.; Marais, J.P.J.; Khoo, C.; LaPlante, K.; Vejborg, R.M.; Givskov, M.; Tolker-Nielsen, T.; Seeram, N.P.; Rowley, D.C. Cranberry (Vaccinium macrocarpon) oligosaccharides decrease biofilm formation by uropathogenic escherichia coli. J. Funct. Foods 2015, 17, 235–242. [Google Scholar] [CrossRef]
- Kontiokari, T.; Salo, J.; Eerola, E.; Uhari, M. Cranberry juice and bacterial colonization in children—A placebo-controlled randomized trial. Clin. Nutr. 2005, 24, 1065–1072. [Google Scholar] [CrossRef]
- McMurdo, M.E.T.; Bissett, L.Y.; Price, R.J.G.; Phillips, G.; Crombie, I.K. Does ingestion of cranberry juice reduce symptomatic urinary tract infections in older people in hospital? A double-blind, placebo-controlled trial. Age Ageing 2005, 34, 256–261. [Google Scholar] [CrossRef] [PubMed]
- McMurdo, M.E.T.; Argo, I.; Phillips, G.; Daly, F.; Davey, P. Cranberry or trimethoprim for the prevention of recurrent urinary tract infections? A randomized controlled trial in older women. J. Antimicrob. Chemother. 2009, 63, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, A.E.; Dziura, J.; Hooton, T.M.; Cox, M.E.; Yarova-Yarovaya, Y.; Chen, S.; Gupta, K. Recurrent urinary tract infection and urinary escherichia coli in women ingesting cranberry juice daily: A randomized controlled trial. Mayo Clin. Proc. 2012, 87, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Stothers, L. A randomized trial to evaluate effectiveness and cost effectiveness of naturopathic cranberry products as prophylaxis against urinary tract infection in women. Can. J. Urol. 2002, 9, 1558–1562. [Google Scholar]
- Wing, D.A.; Rumney, P.J.; Preslicka, C.W.; Chung, J.H. Daily cranberry juice for the prevention of asymptomatic bacteriuria in pregnancy: A randomized, controlled pilot study. J. Urol. 2008, 180, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Renaud, V.; Faucher, M.; Perreault, V.; Serre, E.; Dubé, P.; Boutin, Y.; Bazinet, L. Evolution of cranberry juice compounds during in vitro digestion and identification of the organic acid responsible for the disruption of in vitro intestinal cell barrier integrity. J. Food Sci. Technol. 2020, 57, 2329–2342. [Google Scholar] [CrossRef] [PubMed]
- Renaud, V. Impacts du Retrait des Acides Organiques du jus de Canneberge, Par Électrodialyse Avec Membranes Bipolaires, sur sa Digestion, L’apparition de L’inflammation Intestinale et le Microbiote Intestinal. Ph.D. Thesis, University Laval, Québec, QC, Canada, 21 April 2021. [Google Scholar]
- Anhê, F.F.; Varin, T.V.; Le Barz, M.; Pilon, G.; Dudonné, S.; Trottier, J.; St-Pierre, P.; Harris, C.S.; Lucas, M.; Lemire, M.; et al. Arctic berry extracts target the gut-liver axis to alleviate metabolic endotoxaemia, insulin resistance and hepatic steatosis in diet-induced obese mice. Diabetologia 2018, 61, 919–931. [Google Scholar] [CrossRef] [PubMed]
- Anhê, F.F.; Nachbar, R.T.; Varin, T.V.; Vilela, V.; Dudonné, S.; Pilon, G.; Fournier, M.; Lecours, M.-A.; Desjardins, Y.; Roy, D.; et al. A polyphenol-rich cranberry extract reverses insulin resistance and hepatic steatosis independently of body weight loss. Mol. Metab. 2017, 6, 1563–1573. [Google Scholar] [CrossRef] [PubMed]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; et al. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. Population in the gut microbiota of mice. Gut 2015, 64, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Han, Y.; Gu, M.; Song, M.; Wu, X.; Li, Z.; Li, F.; Goulette, T.; Xiao, H. Dietary cranberry suppressed colonic inflammation and alleviated gut microbiota dysbiosis in dextran sodium sulfate-treated mice. Food Funct. 2019, 10, 6331–6341. [Google Scholar] [CrossRef]
- Heyman-Lindén, L.; Kotowska, D.; Sand, E.; Bjursell, M.; Plaza, M.; Turner, C.; Holm, C.; Fåk, F.; Berger, K. Lingonberries alter the gut microbiota and prevent low-grade inflammation in high-fat diet fed mice. Food Nutr. Res. 2016, 60, 29993. [Google Scholar] [CrossRef] [PubMed]
- Roopchand, D.E.; Carmody, R.N.; Kuhn, P.; Moskal, K.; Rojas-Silva, P.; Turnbaugh, P.J.; Raskin, I. Dietary polyphenols promote growth of the gut bacterium akkermansia muciniphila and attenuate high-fat diet-induced metabolic syndrome. Diabetes 2015, 64, 2847–2858. [Google Scholar] [CrossRef] [PubMed]
- Abdelrazek, H.; Abuzead, S.; Ali, A.; El-Genaidy, H.; Abdel-Hafez, S. Effect of Citric and Acetic Acid Water Acidification on Broiler’s Performance with Respect to Thyroid Hormones Levels. Adv. Anim. Vet. Sci. 2016, 4, 271–278. [Google Scholar] [CrossRef]
- Adil, S.; Banday, T.; Bhat, G.A.; Mir, M.S.; Rehman, M. Effect of Dietary Supplementation of Organic Acids on Performance, Intestinal Histomorphology, and Serum Biochemistry of Broiler Chicken. Vet. Med. Int. 2010, 2010. [Google Scholar] [CrossRef] [PubMed]
- Basmacioğlu-Malayoğlu, H.; Ozdemir, P.; Bağriyanik, H.A. Influence of an Organic Acid Blend and Essential Oil Blend, Individually or in Combination, on Growth Performance, Carcass Parameters, Apparent Digestibility, Intestinal Microflora and Intestinal Morphology of Broilers. Br. Poult. Sci. 2016, 57, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Orij, R.; Brul, S.; Smits, G.J. Quantitative Analysis of the Modes of Growth Inhibition by Weak Organic Acids in Saccharomyces Cerevisiae. Appl. Environ. Microbiol. 2012, 78, 8377–8387. [Google Scholar] [CrossRef] [PubMed]
- Rozoy, E.; Boudesocque, L.; Bazinet, L. Deacidification of Cranberry Juice by Electrodialysis with Bipolar Membranes. J. Agric. Food Chem. 2015, 63, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Serre, E.; Rozoy, E.; Pedneault, K.; Lacour, S.; Bazinet, L. Deacidification of Cranberry Juice by Electrodialysis: Impact of Membrane Types and Configurations on Acid Migration and Juice Physicochemical Characteristics. Sep. Purif. Technol. 2016, 163, 228–237. [Google Scholar] [CrossRef]
- Bazinet, L.; Ippersiel, D.; Mahdavi, B. Fractionation of Whey Proteins by Bipolar Membrane Electroacidification. Innov. Food Sci. Emerg. Technol. 2004, 5, 17–25. [Google Scholar] [CrossRef]
- Mani, K.N. Electrodialysis Water Splitting Technology. J. Membr. Sci. 1991, 58, 117–138. [Google Scholar] [CrossRef]
- Koelink, P.J.; Wildenberg, M.E.; Stitt, L.W.; Feagan, B.G.; Koldijk, M.; van ‘t Wout, A.B.; Atreya, R.; Vieth, M.; Brandse, J.F.; Duijst, S.; et al. Development of reliable, valid and responsive scoring systems for endoscopy and histology in animal models for inflammatory bowel disease. J. Crohns Colitis 2018, 12, 794–803. [Google Scholar] [CrossRef]
- Singh, H.; Kumar, H.; Handa, R.; Talapatra, P.; Ray, S.; Gupta, V. Use of clinical disease activity index score for assessment of disease activity in rheumatoid arthritis patients: An indian experience. Arthritis 2011, 2011, 146398. [Google Scholar] [CrossRef] [PubMed]
- Faucher, M.; Serre, É.; Langevin, M.-È.; Mikhaylin, S.; Lutin, F.; Bazinet, L. Drastic energy consumption reduction and ecoefficiency improvement of cranberry juice deacidification by electrodialysis with bipolar membranes at semi-industrial scale: Reuse of the recovery solution. J. Membr. Sci. 2018, 555, 105–114. [Google Scholar] [CrossRef]
- Pelletier, S.; Serre, É.; Mikhaylin, S.; Bazinet, L. Optimization of cranberry juice deacidification by electrodialysis with bipolar membrane: Impact of pulsed electric field conditions. Sep. Purif. Technol. 2017, 186, 106–116. [Google Scholar] [CrossRef]
- Cesquini, M.; Stoppa, G.R.; Prada, P.O.; Torsoni, A.S.; Romanatto, T.; Souza, A.; Saad, M.J.; Velloso, L.A.; Torsoni, M.A. Citrate diminishes hypothalamic acetyl-CoA carboxylase phosphorylation and modulates satiety signals and hepatic mechanisms involved in glucose homeostasis in rats. Life Sci. 2008, 82, 1262–1271. [Google Scholar] [CrossRef]
- Gatta, B.; Zuberbuehler, C.; Arnold, M.; Aubert, R.; Langhans, W.; Chapelot, D. Acute effects of pharmacological modifications of fatty acid metabolism on human satiety. Br. J. Nutr. 2008, 101, 1867–1877. [Google Scholar] [CrossRef] [PubMed]
- Josic, J.; Olsson, A.T.; Wickeberg, J.; Lindstedt, S.; Hlebowicz, J. Does green tea affect postprandial glucose, insulin and satiety in healthy subjects: A randomized controlled trial. Nutr. J. 2010, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Cremonini, E.; Daveri, E.; Mastaloudis, A.; Adamo, A.M.; Mills, D.; Kalanetra, K.; Hester, S.N.; Wood, S.M.; Fraga, C.G.; Oteiza, P.I. Anthocyanins protect the gastrointestinal tract from high fat diet-induced alterations in redox signaling, barrier integrity and dysbiosis. Redox Biol. 2019, 26, 101269. [Google Scholar] [CrossRef] [PubMed]
- Calcinotto, A.; Grioni, M.; Jachetti, E.; Curnis, F.; Mondino, A.; Parmiani, G.; Corti, A.; Bellone, M. Targeting TNF-α to neoangiogenic vessels enhances lymphocyte infiltration in tumors and increases the therapeutic potential of immunotherapy. J. Immunol. 2012, 188, 2687–2694. [Google Scholar] [CrossRef] [PubMed]
- Ruder, B.; Atreya, R.; Becker, C. Tumour necrosis factor alpha in intestinal homeostasis and gut related diseases. Int. J. Mol. Sci. 2019, 20, 1887. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Zhang, Z.; Inatomi, O.; Fujino, S.; Deguchi, Y.; Araki, Y.; Tsujikawa, T.; Kitoh, K.; Kim–Mitsuyama, S.; Takayanagi, A.; et al. Interleukin-22, a member of the IL-10 subfamily, induces inflammatory responses in colonic subepithelial myofibroblasts. Gastroenterology 2005, 129, 969–984. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, K.; Ogawa, A.; Mizoguchi, E.; Shimomura, Y.; Andoh, A.; Bhan, A.K.; Blumberg, R.S.; Xavier, R.J.; Mizoguchi, A. IL-22 ameliorates intestinal inflammation in a mouse model of ulcerative colitis. J. Clin. Investig. 2008, 118, 534–544. [Google Scholar] [CrossRef]
- Zenewicz, L.A.; Yancopoulos, G.D.; Valenzuela, D.M.; Murphy, A.J.; Stevens, S.; Flavell, R.A. Innate and adaptive interleukin-22 protects mice from inflammatory bowel disease. Immunity 2008, 29, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Pelaseyed, T.; Bergström, J.H.; Gustafsson, J.K.; Ermund, A.; Birchenough, G.M.H.; Schütte, A.; van der Post, S.; Svensson, F.; Rodríguez-Piñeiro, A.M.; Nyström, E.E.L.; et al. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol. Rev. 2014, 260, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Tolić, M.-T.; Krbavčić, I.P.; Vujević, P.; Milinović, B.; Jurčević, I.L.; Vahčić, N. Effects of weather conditions on phenolic content and antioxidant capacity in juice of chokeberries (Aronia melanocarpa L.). Pol. J. Food Nutr. Sci. 2017, 67, 67–74. [Google Scholar] [CrossRef]
- Chen, L.; Jiang, B.; Zhong, C.; Guo, J.; Zhang, L.; Mu, T.; Zhang, Q.; Bi, X. Chemoprevention of colorectal cancer by black raspberry anthocyanins involved the modulation of gut microbiota and SFRP2 demethylation. Carcinogenesis 2018, 39, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Flores, G.; Ruiz del Castillo, M.L.; Costabile, A.; Klee, A.; Bigetti Guergoletto, K.; Gibson, G.R. In vitro fermentation of anthocyanins encapsulated with cyclodextrins: Release, metabolism and influence on gut microbiota growth. J. Funct. Foods 2015, 16, 50–57. [Google Scholar] [CrossRef]
- Hidalgo, M.; Oruna-Concha, M.J.; Kolida, S.; Walton, G.E.; Kallithraka, S.; Spencer, J.P.E.; de Pascual-Teresa, S. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. J. Agric. Food Chem. 2012, 60, 3882–3890. [Google Scholar] [CrossRef] [PubMed]
- Hogan, S.; Canning, C.; Sun, S.; Sun, X.; Zhou, K. Effects of grape pomace antioxidant extract on oxidative stress and inflammation in diet induced obese mice. J. Agric. Food Chem. 2010, 58, 11250–11256. [Google Scholar] [CrossRef] [PubMed]
- Ky, C.; Se, B.; Tm, A.; Sm, A.; Ka, L.; Gs, H. Grape seed extract reduces the severity of selected disease markers in the proximal colon of dextran sulphate sodium-induced colitis in rats. Dig. Dis. Sci. 2013, 58, 970–977. [Google Scholar] [CrossRef]
- Shoji, T.; Mutsuga, M.; Nakamura, T.; Kanda, T.; Akiyama, H.; Goda, Y. Isolation and structural elucidation of some procyanidins from apple by low-temperature nuclear magnetic resonance. J. Agric. Food Chem. 2003, 51, 3806–3813. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, P.; Zhu, Y.; Lou, Q.; He, S. Antioxidant and prebiotic activity of five peonidin-based anthocyanins extracted from purple sweet potato (Ipomoea batatas (L.) Lam.). Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Vugic, L.; Colson, N.; Nikbakht, E.; Gaiz, A.; Holland, O.J.; Kundur, A.R.; Singh, I. Anthocyanin supplementation inhibits secretion of pro-inflammatory cytokines in overweight and obese individuals. J. Funct. Foods 2020, 64, 103596. [Google Scholar] [CrossRef]
- Yang, G.; Xue, Y.; Zhang, H.; Du, M.; Zhu, M.-J. Favourable effects of grape seed extract on intestinal epithelial differentiation and barrier function in IL10-deficient mice. Br. J. Nutr. 2015, 114, 15–23. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yoshioka, Y.; Akiyama, H.; Nakano, M.; Shoji, T.; Kanda, T.; Ohtake, Y.; Takita, T.; Matsuda, R.; Maitani, T. Orally administered apple procyanidins protect against experimental inflammatory bowel disease in mice. Int. Immunopharmacol. 2008, 8, 1802–1807. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Sun, H.; He, S.; Lou, Q.; Yu, M.; Tang, M.; Tu, L. Metabolism and prebiotics activity of anthocyanins from black rice (Oryza sativa L.) in vitro. PLoS ONE 2018, 13, e0195754. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wu, T.; Li, N.; Wang, X.; Chen, G.; Lyu, X. Bilberry anthocyanin extract promotes intestinal barrier function and inhibits digestive enzyme activity by regulating the gut microbiota in aging rats. Food Funct. 2019, 10, 333–343. [Google Scholar] [CrossRef]
- Yan, X.; Yang, C.; Lin, G.; Chen, Y.; Miao, S.; Liu, B.; Zhao, C. Antidiabetic potential of green seaweed enteromorpha prolifera flavonoids regulating insulin signaling pathway and gut microbiota in type 2 diabetic mice. J. Food Sci. 2019, 84, 165–173. [Google Scholar] [CrossRef] [PubMed]
- López, R.L.; Burgos, M.J.G.; Gálvez, A.; Pulido, R.P. The human gastrointestinal tract and oral microbiota in inflammatory bowel disease: A state of the science review. APMIS 2017, 125, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Daza, M.-C.; Daoust, L.; Boutkrabt, L.; Pilon, G.; Varin, T.; Dudonné, S.; Levy, É.; Marette, A.; Roy, D.; Desjardins, Y. Wild blueberry proanthocyanidins shape distinct gut microbiota profile and influence glucose homeostasis and intestinal phenotypes in high-fat high-sucrose fed mice. Sci. Rep. 2020, 10, 2217. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Grootaert, C.; Pitart, J.; Vidovic, N.K.; Kamiloglu, S.; Possemiers, S.; Glibetic, M.; Smagghe, G.; Raes, K.; de Wiele, T.V.; et al. Aronia (Aronia melanocarpa) polyphenols modulate the microbial community in a simulator of the human intestinal microbial ecosystem (SHIME) and decrease secretion of proinflammatory markers in a caco-2/endothelial cell coculture model. Mol. Nutr. Food Res. 2018, 62, 1800607. [Google Scholar] [CrossRef] [PubMed]
- den Besten, G.; Lange, K.; Havinga, R.; van Dijk, T.H.; Gerding, A.; van Eunen, K.; Müller, M.; Groen, A.K.; Hooiveld, G.J.; Bakker, B.M.; et al. Gut-derived short-chain fatty acids are vividly assimilated into host carbohydrates and lipids. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G900–G910. [Google Scholar] [CrossRef] [PubMed]
- Haines-Menges, B.L.; Whitaker, W.B.; Lubin, J.B.; Boyd, E.F. Host sialic acids: A delicacy for the pathogen with discerning taste. Microbiol. Spectr. 2015, 3, 321–342. [Google Scholar] [CrossRef] [PubMed]
- Sohlenkamp, C.; Geiger, O. Bacterial membrane lipids: Diversity in structures and pathways. FEMS Microbiol. Rev. 2016, 40, 133–159. [Google Scholar] [CrossRef]
- Pérez-Sánchez, A.; Borrás-Linares, I.; Barrajón-Catalán, E.; Arráez-Román, D.; González-Álvarez, I.; Ibáñez, E.; Segura-Carretero, A.; Bermejo, M.; Micol, V. Evaluation of the intestinal permeability of rosemary (Rosmarinus officinalis L.) extract polyphenols and terpenoids in caco-2 cell monolayers. PLoS ONE 2017, 12, e0172063. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Wächtler, B.; Temp, U.; Krekling, T.; Séguin, A.; Gershenzon, J. A bifunctional geranyl and geranylgeranyl diphosphate synthase is involved in terpene oleoresin formation in picea abies. Plant Physiol. 2010, 152, 639–655. [Google Scholar] [CrossRef] [PubMed]
- Anhê, F.F.; Nachbar, R.T.; Varin, T.V.; Trottier, J.; Dudonné, S.; Barz, M.L.; Feutry, P.; Pilon, G.; Barbier, O.; Desjardins, Y.; et al. Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut 2019, 68, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naïve bayesian classifier for rapid assignment of RRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Caspi, R.; Billington, R.; Ferrer, L.; Foerster, H.; Fulcher, C.A.; Keseler, I.M.; Kothari, A.; Krummenacker, M.; Latendresse, M.; Mueller, L.A.; et al. The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of pathway/genome databases. Nucleic Acids Res. 2016, 44, D471–D480. [Google Scholar] [CrossRef] [PubMed]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.; Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef] [PubMed]

| CJs | 0% DCJ | 40% DCJ | 60% DCJ | 80% DCJ |
|---|---|---|---|---|
| Deacidification (%) | 0 | 42 | 60 | 79 |
| pH | 2.59 ± 0.03 a | 2.71 ± 0.01 b | 2.87 ± 0.01 c | 3.24 ± 0.02 d |
| Conductivity (µS/cm) | 2 409.00 ± 5.00 a | 1 883.00 ± 15.72 b | 1 966.67 ± 15.89 d | 1 840.00 ± 4.36 c |
| Titratable acidity (g of citric acid eq/L) | 9.25 ± 0.05 a | 5.40 ± 0.05 b | 3.72 ± 0.02 c | 1.91 ± 0.05 d |
| °Brix | 6.90 ± 0.00 a | 6.10 ± 0.00 b | 5.80 ± 0.00 c | 5.60 ± 0.00 d |
| Organic acids (g/L) | ||||
| Quinic | 10.35 ± 0.31 a | 10.49 ± 0.17 a | 10.11 ± 0.14 a | 9.19 ± 0.11 b |
| Malic | 6.03 ± 0.10 a | 2.40 ± 0.11 b | 1.34 ± 0.06 c | 0.00 ± 0.00 d |
| Citric | 11.59 ± 0.20 a | 6.88 ± 0.15 b | 4.67 ± 0.09 c | 2.35 ± 0.06 d |
| Anthocyanins (mg of cyanidin eq/L) | ||||
| C-3-galactoside | 65.14 ± 0.51 a | 64.70 ± 0.32 a | 65.76 ± 1.22 a | 61.89 ± 0.61 b |
| C-3-glucoside | 2.15 ± 0.12 ab | 2.47 ± 0.10 b | 2.06 ± 0.06 a | 2.22 ± 0.19 ab |
| C-3-arabinoside | 51.12 ± 0.69 a | 50.99 ± 0.37 a | 50.57 ± 0.89 a | 48.24 ± 0.22 b |
| P-3-galactoside | 84.74 ± 0.54 a | 83.76 ± 0.51 a | 85.02 ± 1.21 a | 80.74 ± 0.71 b |
| P-3-glucoside | 8.50 ± 0.10 a | 9.01 ± 0.12 b | 8.71 ± 0.06 ab | 8.53 ± 0.10 a |
| P-3-arabinoside | 37.94 ± 0.62 a | 37.13 ± 0.36 a | 37.29 ± 0.34 a | 35.98 ± 0.30 b |
| Total | 249.58 ± 2.42 a | 248.05 ± 0.67 a | 249.40 ± 3.88 a | 237.60 ± 1.97 b |
| PACs (mg of epicatechin eq/L) | ||||
| Monomers | 39.35 ± 0.64 a | 36.05 ± 2.74 a | 36.67 ± 2.41 a | 37.43 ± 1.97 a |
| 2–3 mers | 148.36 ± 1.80 a | 142.12 ± 18.47 a | 157.39 ± 3.86 a | 159.30 ± 9.67 a |
| 4–6 mers | 59.92 ± 1.24 a | 57.64 ± 7.06 a | 62.55 ± 1.88 a | 62.29 ± 3.58 a |
| 7–10 mers | 4.28 ± 0.27 a | 4.06 ± 0.54 a | 4.41 ± 0.48 a | 4.53 ± 0.50 a |
| Polymers | 5.55 ± 0.52 a | 5.90 ± 0.38 a | 5.88 ± 0.05 a | 5.78 ± 0.05 a |
| Total | 257.46 ± 2.36 a | 245.78 ± 29.00 a | 266.91 ± 8.35 a | 269.33 ± 15.61 a |
| Total polyphenols (mg of gallic acid eq/L) | 1 074.79 ± 4.90 a | 978.42 ± 47.79 b | 1 075.52 ± 34.87 a | 984.21 ± 53.66 b |
| (A) | ||||
| Groups | Inflammation | Vascularization | Thickening | Total Score |
| Control | 0.00 ± 0.00 a | 0.08 ± 0.29 a | 0.00 ± 0.00 a | 0.03 ± 0.17 a |
| 0% DCJ | 0.25 ± 0.87 ab | 1.42 ± 1.5 b | 0.00 ± 0.00 a | 0.56 ± 1.16 ab |
| 40% DCJ | 1.27 ± 1.49 bc | 0.73 ± 0.90 a | 0.00 ± 0.00 a | 0.67 ± 1.11 b |
| 60% DCJ | 1.42 ± 1.51 c | 0.58 ± 1.16 a | 0.00 ± 0.00 a | 0.67 ± 1.22 b |
| 80% DCJ | 0.00 ± 0.00 a | 0.17 ± 0.58 a | 0.00 ± 0.00 a | 0.06 ± 0.33 a |
| (B) | ||||
| Groups | Inflammation | Vascularization | Thickening | Total Score |
| Control | 0.00 ± 0.00 a | 0.08 ± 0.29 a | 0.00 ± 0.00 a | 0.08 ± 0.29 a |
| 0% DCJ | 0.25 ± 0.45 a | 0.08 ± 0.29 a | 0.33 ± 0.65 a | 0.67 ± 0.89 a |
| 40% DCJ | 0.45 ± 0.93 a | 0.09 ± 0.30 a | 0.27 ± 0.65 a | 0.82 ± 1.40 a |
| 60% DCJ | 0.67 ± 0.89 a | 0.50 ± 0.67 b | 0.25 ± 0.45 a | 1.42 ± 1.38 a |
| 80% DCJ | 0.25 ± 0.45 a | 0.08 ± 0.29 a | 0.25 ± 0.45 a | 0.58 ± 0.90 a |
| Score | Inflammation | Vascularization | Thickening |
|---|---|---|---|
| 0 | None | None | None |
| 1 | 0.1 to 1 cm | 0.1 to 1 cm | 0.1 to 1 cm |
| 2 | 1.1 to 2 cm | 1.1 to 2 cm | 1.1 to 2 cm |
| 3 | 2.1 to 3 cm | 2.1 to 3 cm | 2.1 to 3 cm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renaud, V.; Houde, V.P.; Pilon, G.; Varin, T.V.; Roblet, C.; Marette, A.; Boutin, Y.; Bazinet, L. The Concentration of Organic Acids in Cranberry Juice Modulates the Gut Microbiota in Mice. Int. J. Mol. Sci. 2021, 22, 11537. https://doi.org/10.3390/ijms222111537
Renaud V, Houde VP, Pilon G, Varin TV, Roblet C, Marette A, Boutin Y, Bazinet L. The Concentration of Organic Acids in Cranberry Juice Modulates the Gut Microbiota in Mice. International Journal of Molecular Sciences. 2021; 22(21):11537. https://doi.org/10.3390/ijms222111537
Chicago/Turabian StyleRenaud, Valentine, Vanessa P. Houde, Geneviève Pilon, Thibault V. Varin, Cyril Roblet, André Marette, Yvan Boutin, and Laurent Bazinet. 2021. "The Concentration of Organic Acids in Cranberry Juice Modulates the Gut Microbiota in Mice" International Journal of Molecular Sciences 22, no. 21: 11537. https://doi.org/10.3390/ijms222111537
APA StyleRenaud, V., Houde, V. P., Pilon, G., Varin, T. V., Roblet, C., Marette, A., Boutin, Y., & Bazinet, L. (2021). The Concentration of Organic Acids in Cranberry Juice Modulates the Gut Microbiota in Mice. International Journal of Molecular Sciences, 22(21), 11537. https://doi.org/10.3390/ijms222111537







