Selected Applications of Chitosan Composites
Abstract
1. Introduction
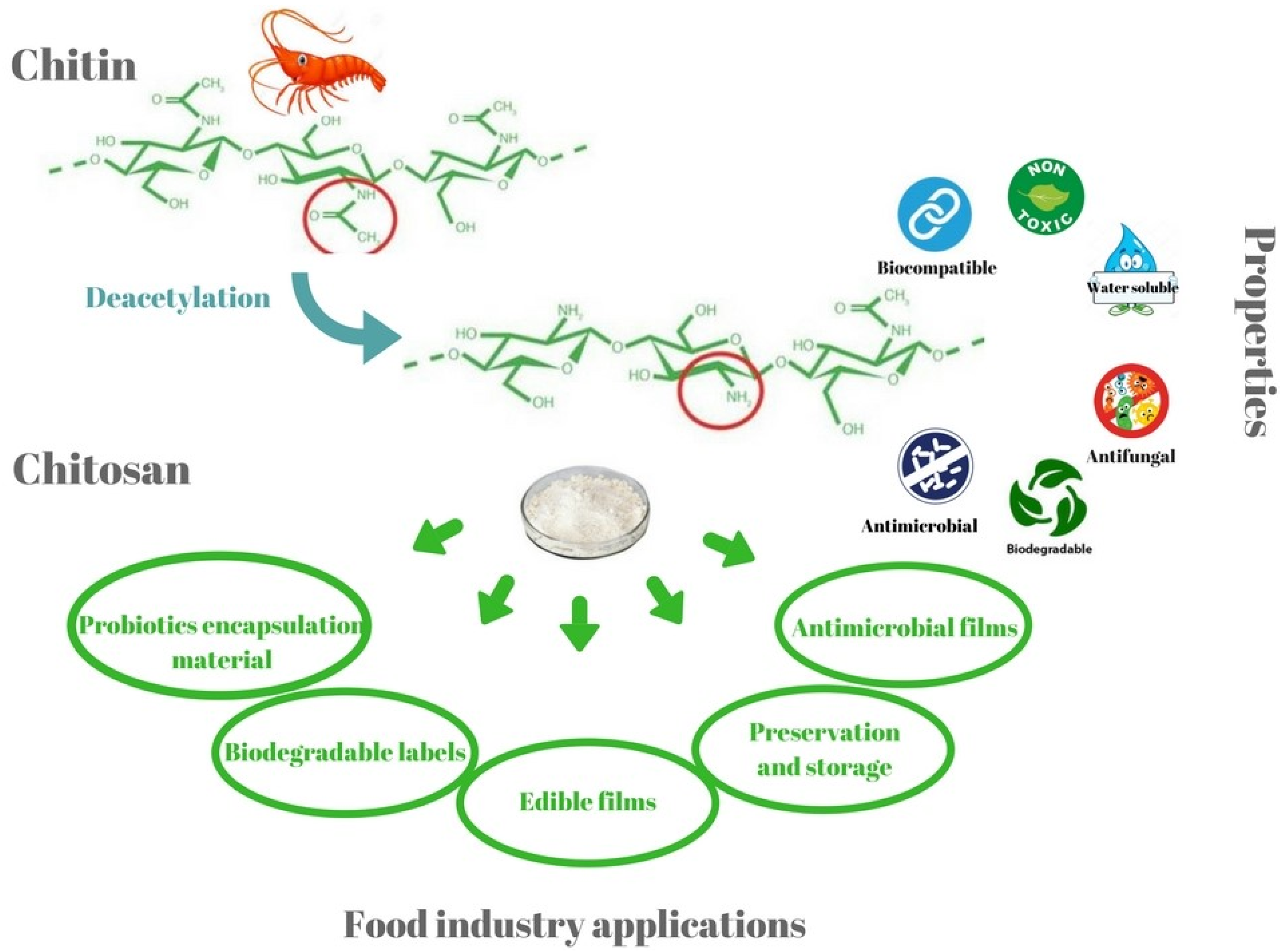
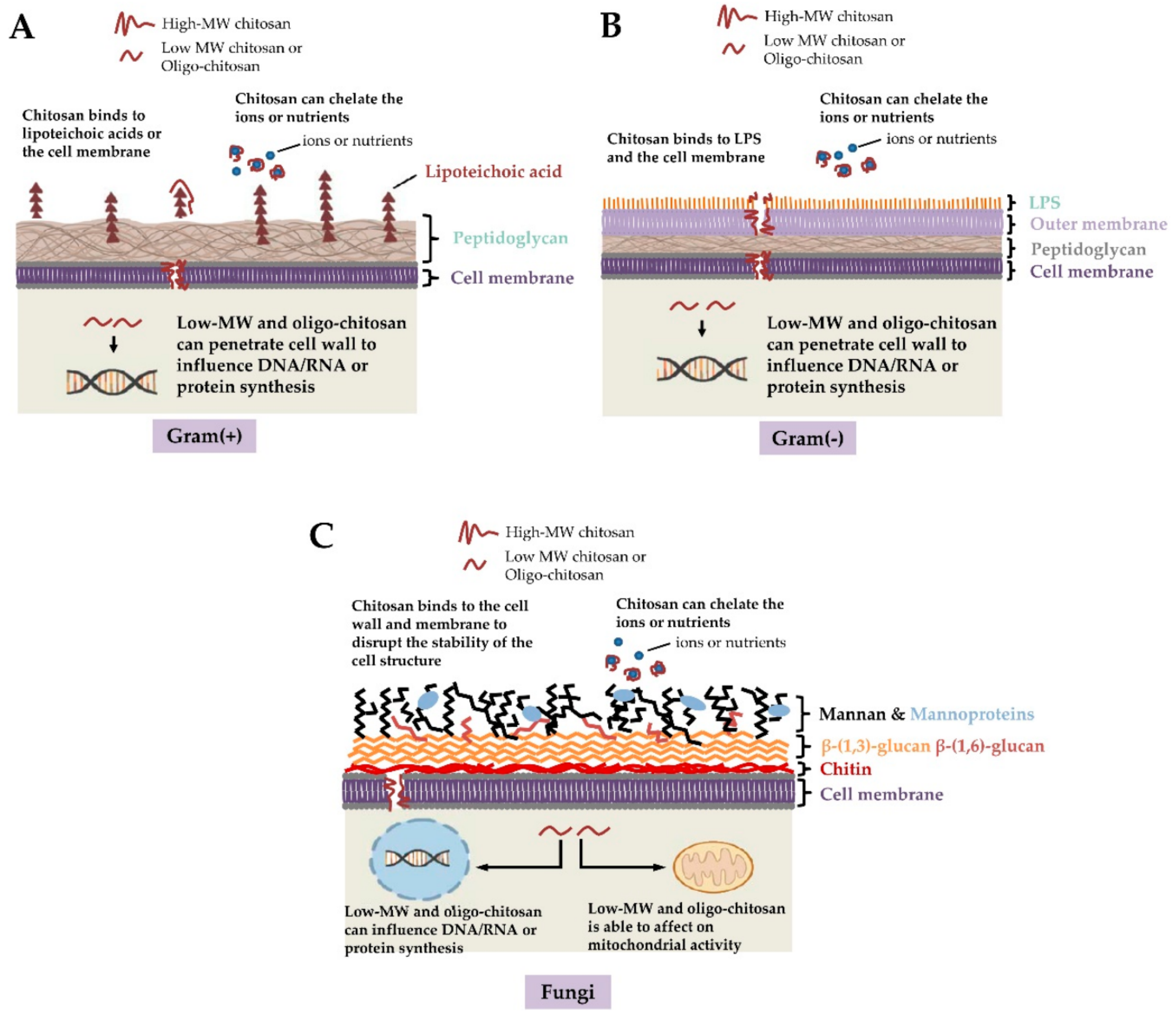
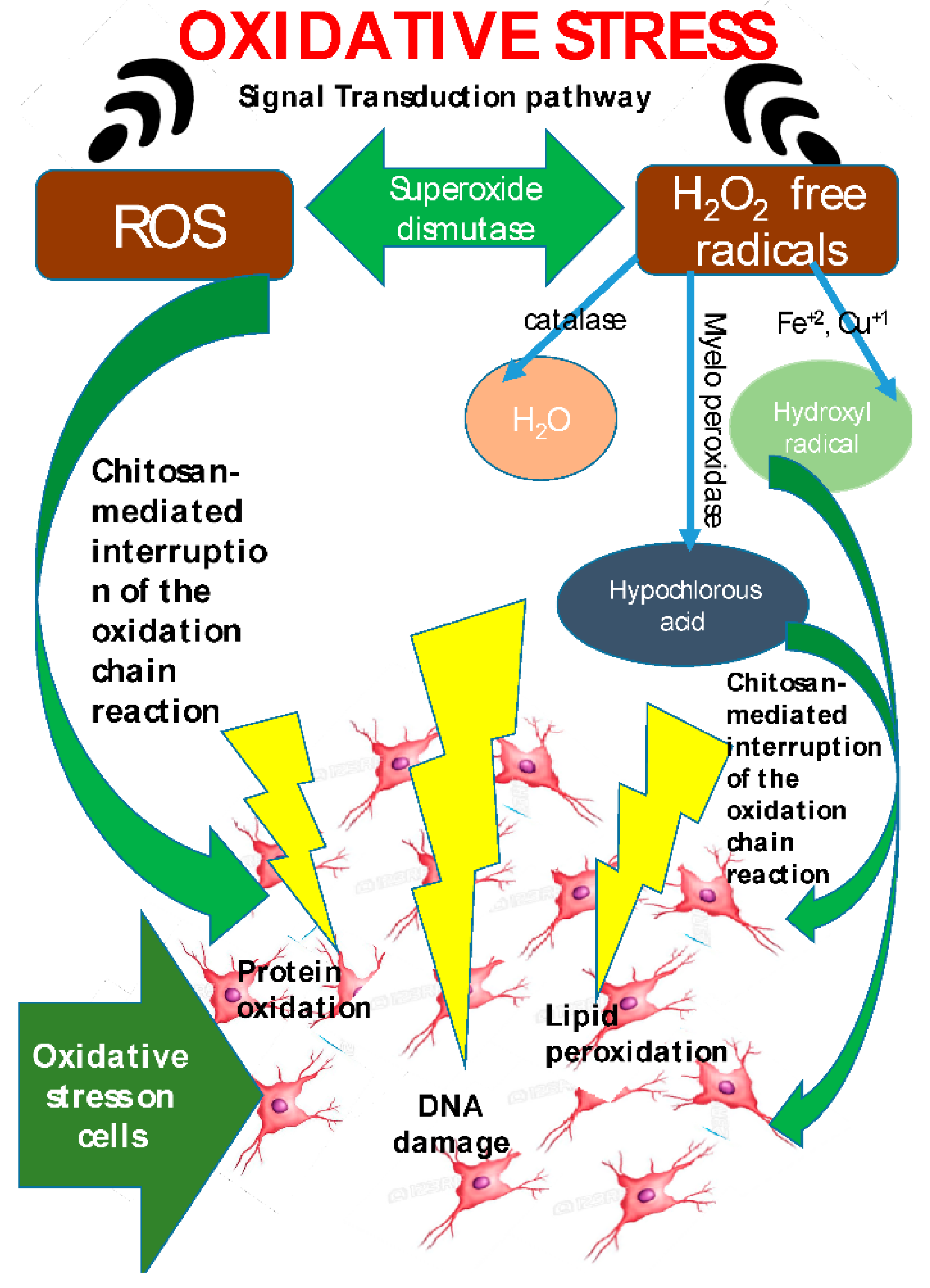
2. Properties of Chitosan
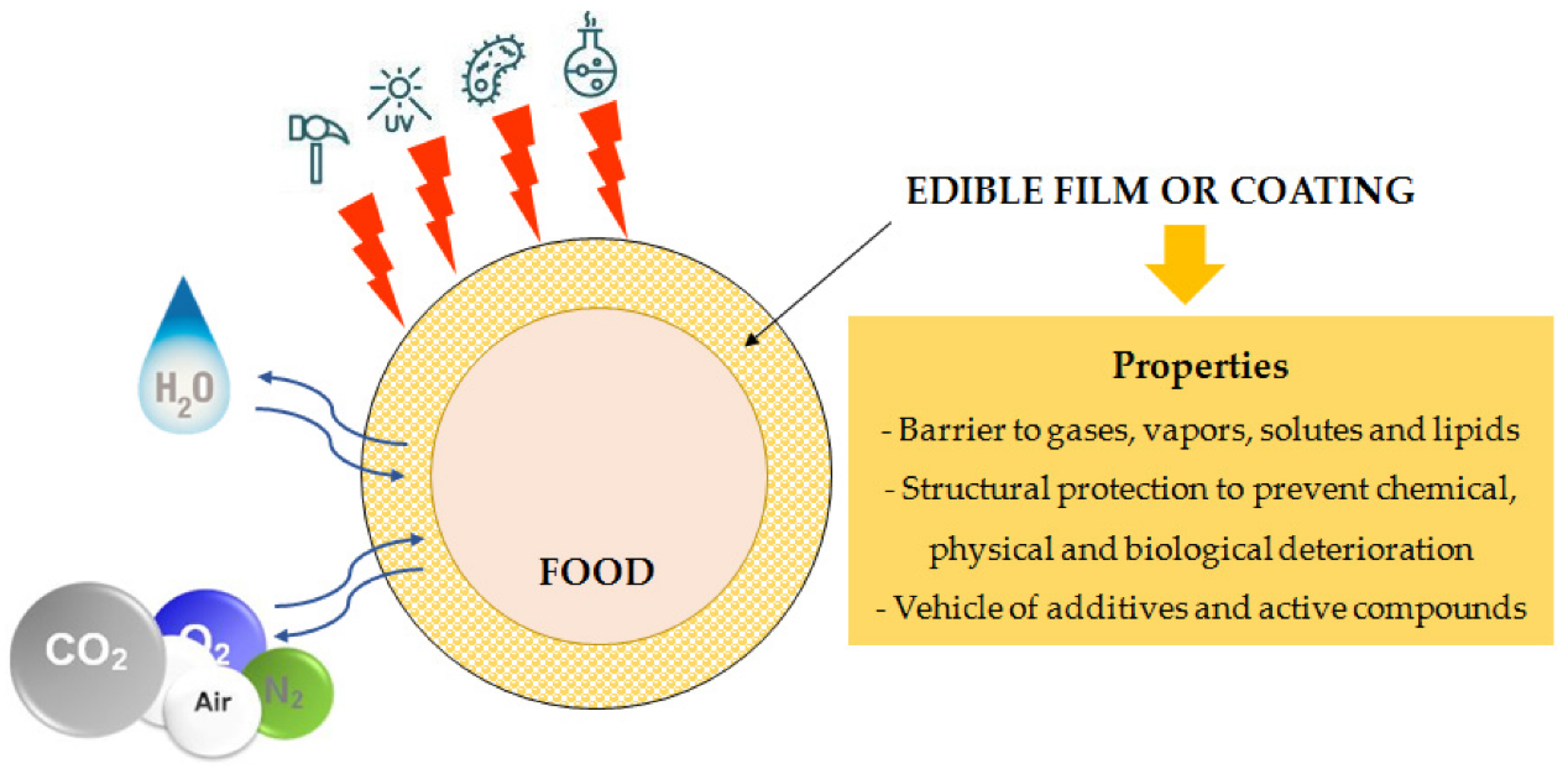
3. Methods for the Development of Food Packaging Systems
3.1. Solution Casting Method
3.2. Coating of Food Products
3.2.1. Spread Coating
3.2.2. Dip Coating
3.2.3. Spray Coating
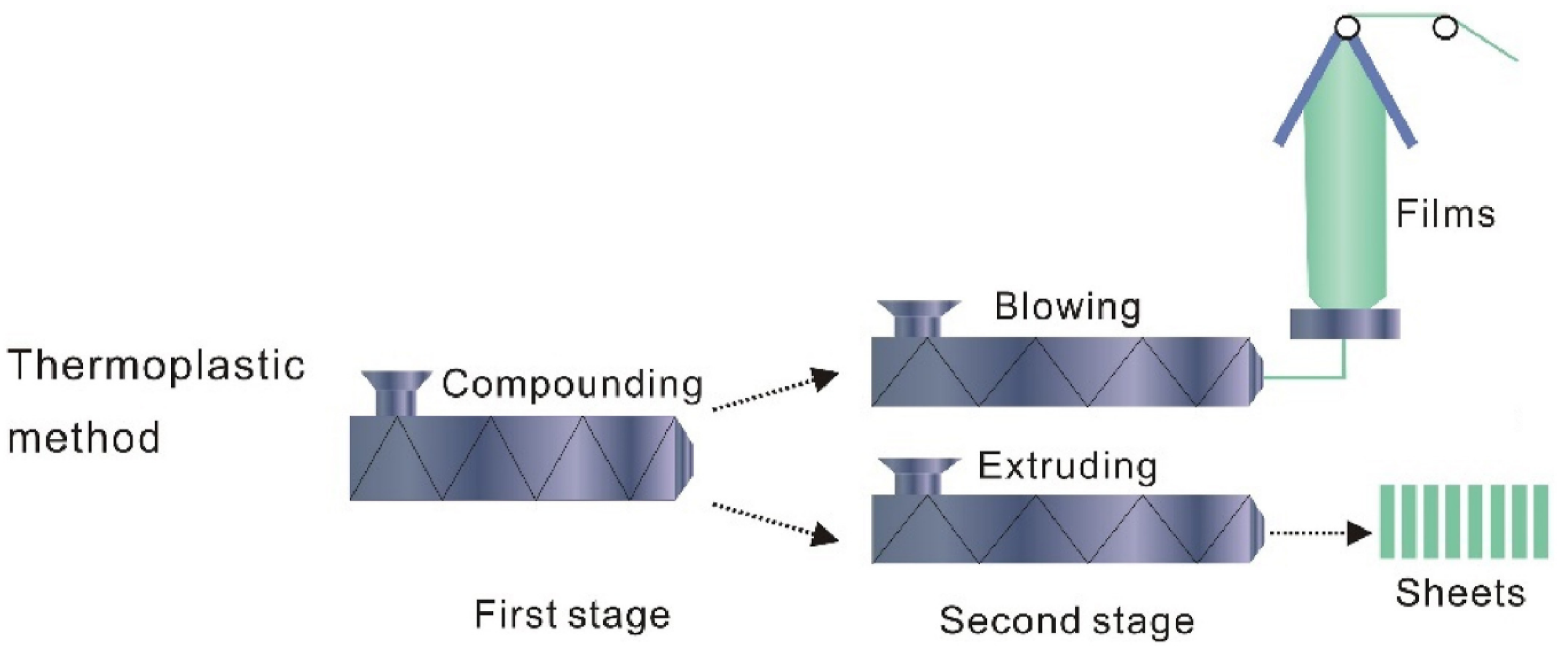
3.3. Extrusion Method
3.4. Multi-Layered Films
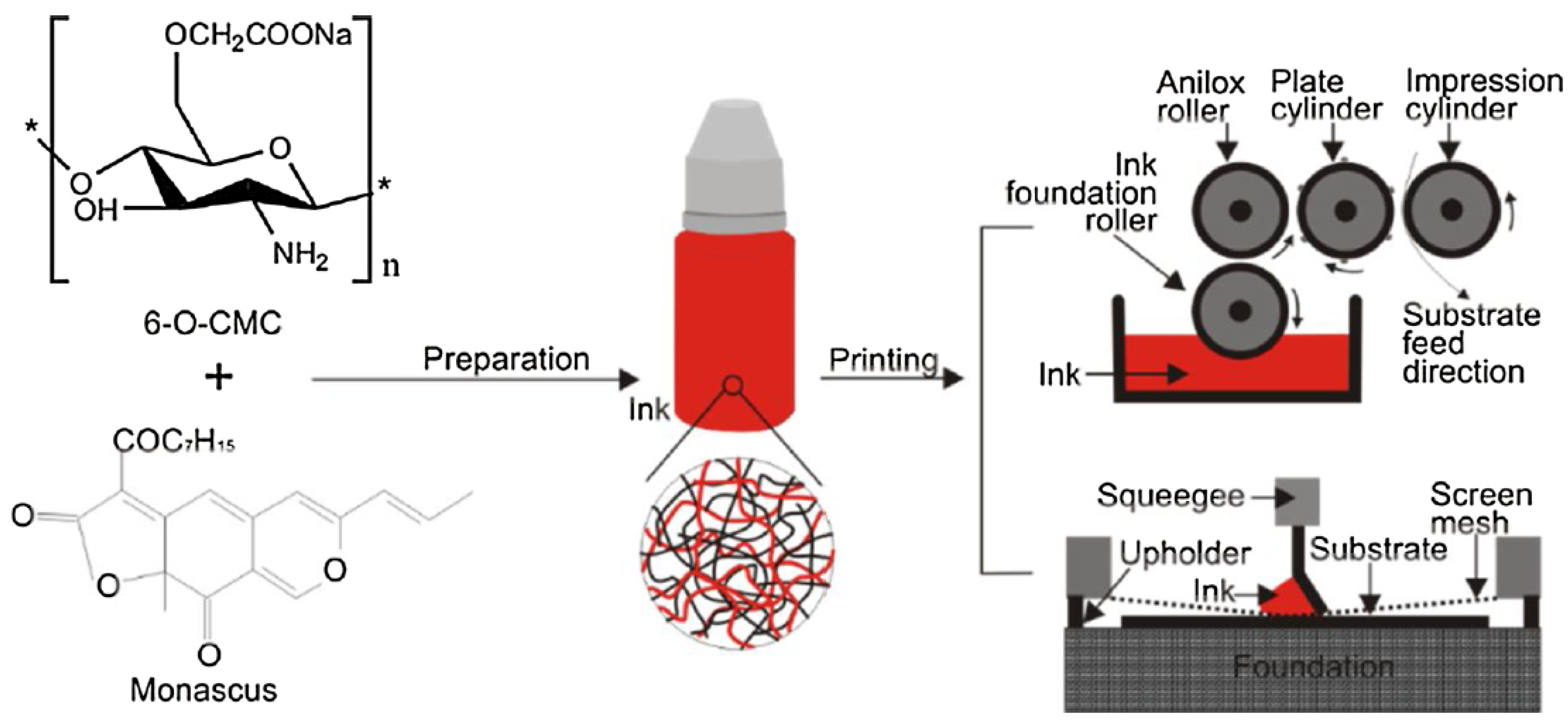
3.5. Flexographic and Screen Printing
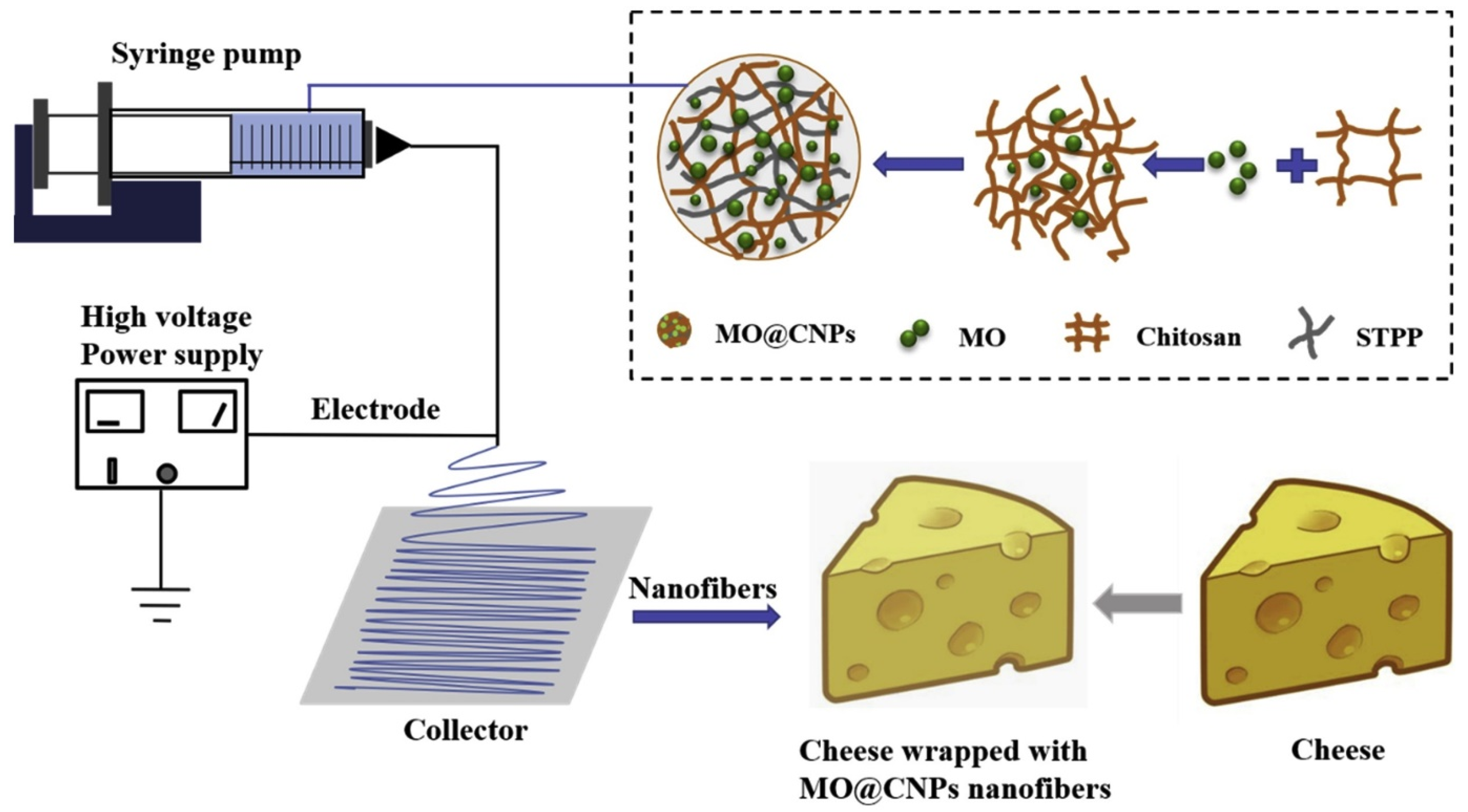
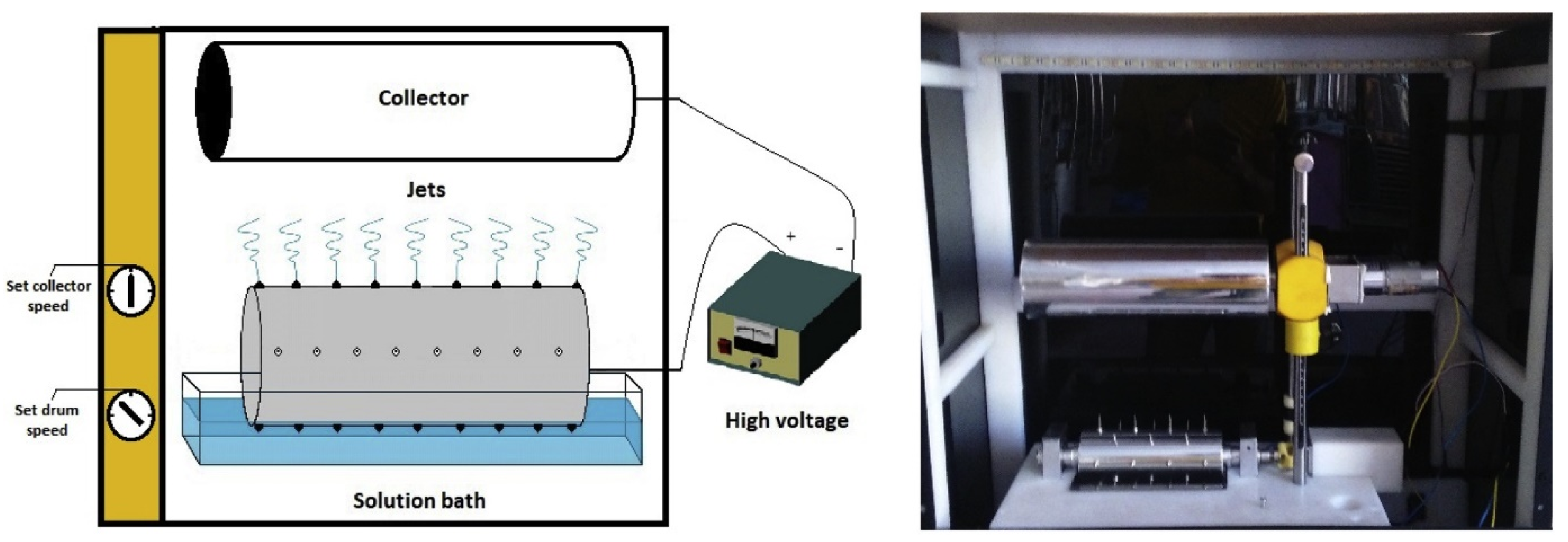
3.6. Electrospinning
4. Applications of Chitosan-Based Films
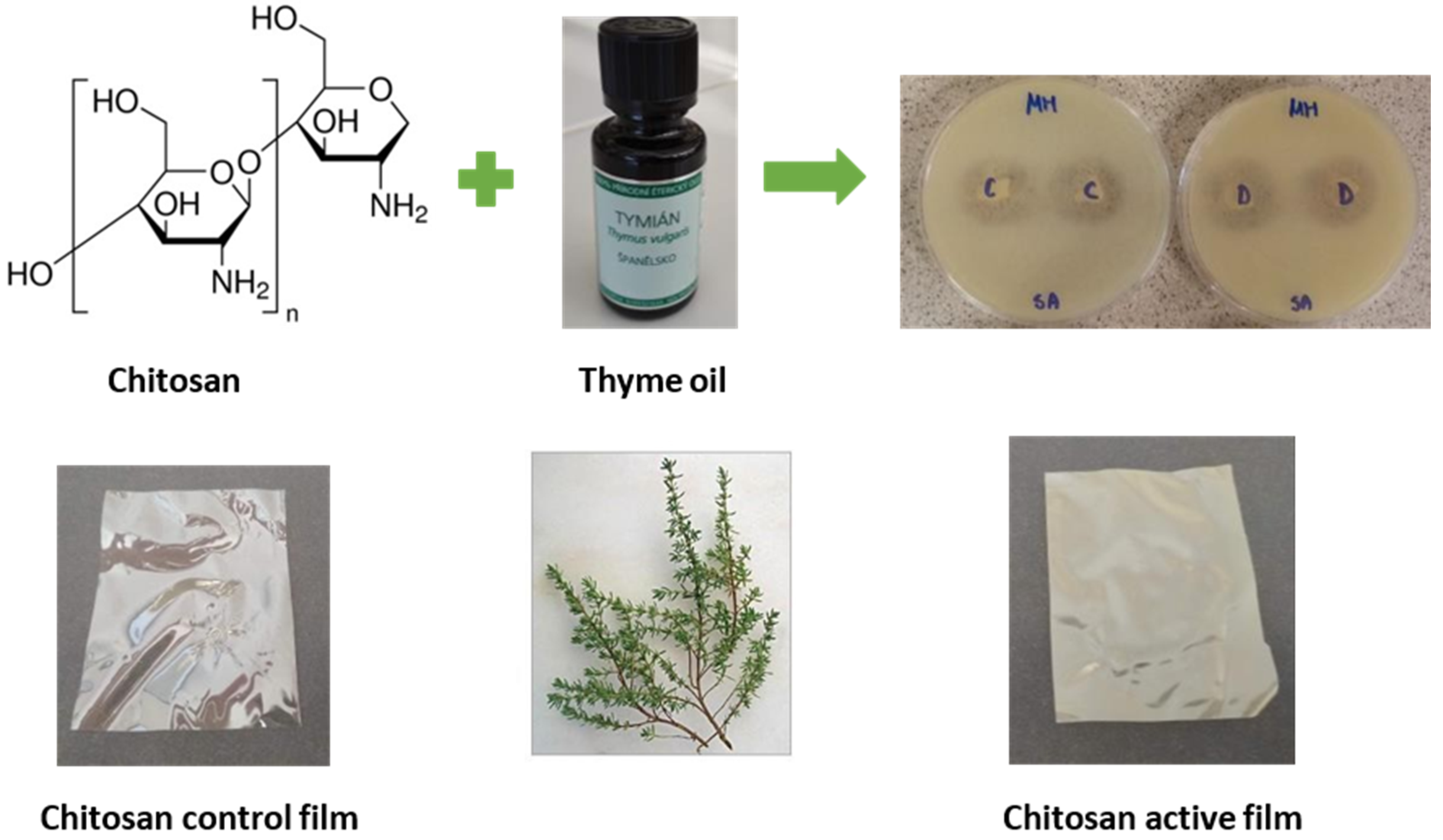
4.1. Chitosan Films Containing Natural Compounds and Essential Oil
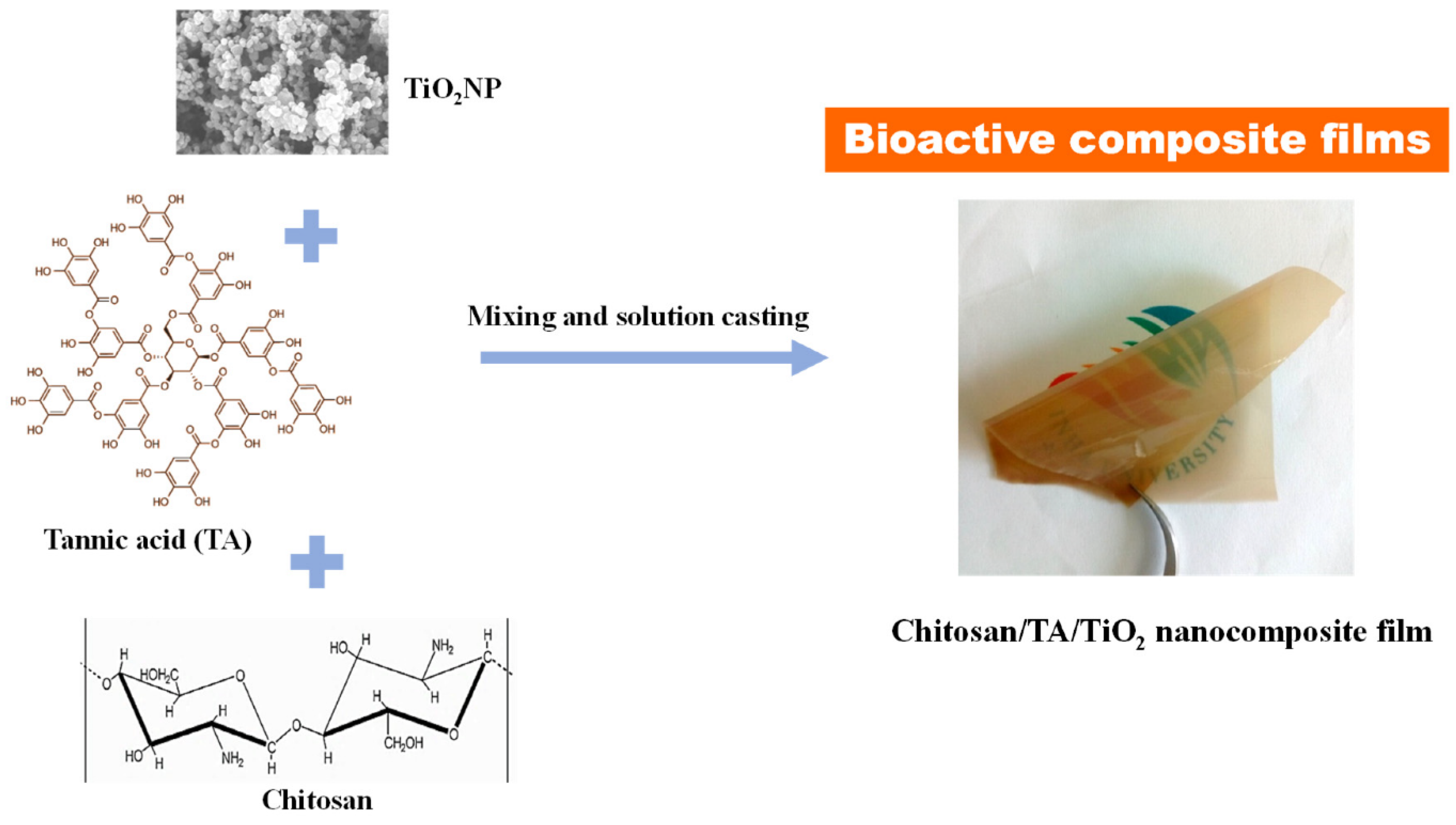
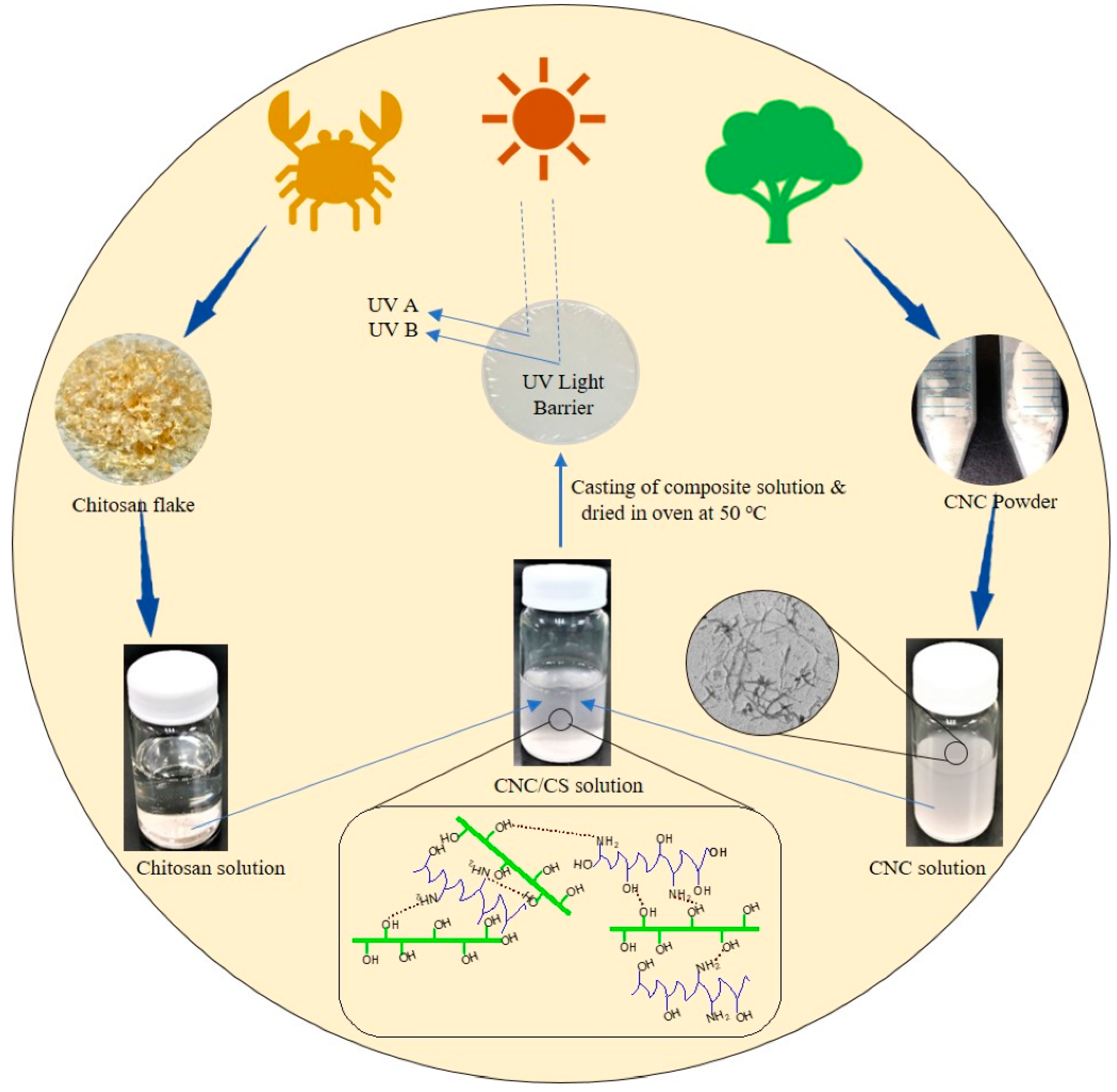
4.2. Chitosan-Based Nanocomposite Films
4.3. Chitosan as Nanofillers
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Haghighi, H.; Licciardello, F.; Fava, P.; Siesler, H.W.; Pulvirenti, A. Recent advances on chitosan-based films for sustainable food packaging applications. Food Packag. Shelf Life 2020, 26, 100551. [Google Scholar] [CrossRef]
- Han, J.H. A review of food packaging technologies and innovations. Innov. Food Packag. 2014, 1, 3–12. [Google Scholar] [CrossRef]
- Biji, K.B.; Ravishankar, C.N.; Mohan, C.O.; Gopal, T.K.S. Smart packaging systems for food applications: A review. J. Food Sci. Technol. 2015, 52, 6125–6135. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Rhim, J.-W. Chitosan-based biodegradable functional films for food packaging applications. Innov. Food Sci. Emerg. Technol. 2020, 62, 102346. [Google Scholar] [CrossRef]
- Akabueze, K.O.; Idu, M.; Erhabor, J.O.; Timothy, O. Antimicrobial and phytochemical attributes of Dennettia tripetala F. baker root and bark extracts. J. Microbiol. Biotechnol. Food Sci. 2016, 5, 297–300. [Google Scholar] [CrossRef][Green Version]
- Patil, A.S.; Waghmare, R.D.; Pawar, S.P.; Salunkhe, S.T.; Kolekar, G.B.; Sohn, D.; Gore, A.H. Photophysical insights of highly transparent, flexible and re-emissive PVA@ WTR-CDs composite thin films: A next generation food packaging material for UV blocking applications. J. Photochem. Photobiol. A Chem. 2020, 400, 112647. [Google Scholar] [CrossRef]
- Ezati, P.; Bang, Y.-J.; Rhim, J.-W. Preparation of a shikonin-based pH-sensitive color indicator for monitoring the freshness of fish and pork. Food Chem. 2021, 337, 127995. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C.; Patel, S.K.S.; Shanmugam, R.; Lee, J.-K. Polyhydroxyalkanoates: Trends and advances toward biotechnological applications. Bioresour. Technol. 2021, 326, 124737. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.S.; Kim, J.-H.; Kalia, V.C.; Lee, J.-K. Antimicrobial activity of amino-derivatized cationic polysaccharides. Indian J. Microbiol. 2019, 59, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhou, P.; Lv, X.; Liang, Y. Insight into the structure-function relationships of the solubility of chitin/chitosan in natural deep eutectic solvents. Mater. Today Commun. 2021, 27, 102374. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Muthu, M.; Gopal, J.; Chun, S.; Devadoss, A.; Hasan, N.; Sivanesan, I. Crustacean waste-derived chitosan: Antioxidant properties and future perspective. Antioxidants 2021, 10, 228. [Google Scholar] [CrossRef] [PubMed]
- da Silva Alves, D.C.; Healy, B.; Pinto, L.A.d.A.; Cadaval, T.R.S.; Breslin, C.B. Recent developments in chitosan-based adsorbents for the removal of pollutants from aqueous environments. Molecules 2021, 26, 594. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Gupta, R.K.; Kondaveeti, S.; Otari, S.V.; Kumar, A.; Kalia, V.C.; Lee, J.-K. Conversion of biogas to methanol by methanotrophs immobilized on chemically modified chitosan. Bioresour. Technol. 2020, 315, 123791. [Google Scholar] [CrossRef]
- Călinoiu, L.-F.; Ştefănescu, B.E.; Pop, I.D.; Muntean, L.; Vodnar, D.C. Chitosan coating applications in probiotic microencapsulation. Coatings 2019, 9, 194. [Google Scholar] [CrossRef]
- Ke, C.-L.; Deng, F.-S.; Chuang, C.-Y.; Lin, C.-H. Antimicrobial actions and applications of chitosan. Polymers 2021, 13, 904. [Google Scholar] [CrossRef] [PubMed]
- Trung, T.S.; Bao, H.N.D. Physicochemical properties and antioxidant activity of chitin and chitosan prepared from Pacific white shrimp waste. Int. J. Carbohydr. Chem. 2015, 2015, 706259. [Google Scholar] [CrossRef]
- Rajasree, S.R.; Gobalakrishnan, M.; Aranganathan, L.; Karthih, M. Fabrication and characterization of chitosan based collagen/ gelatin composite scaffolds from big eye snapper Priacanthus hamrur skin for antimicrobial and antioxidant applications. Mater. Sci. Eng. C 2020, 107, 110270. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, Q.; Wang, H. Synthesis and characterization of nanodiamond reinforced chitosan for bone tissue engineering. J. Funct. Biomater. 2016, 7, 27. [Google Scholar] [CrossRef]
- Roy, S.; Zhai, L.; Kim, H.C.; Pham, D.H.; Alrobei, H.; Kim, J. Tannic-acid-cross-linked and TiO2-nanoparticle-reinforced chitosan-based nanocomposite film. Polymers 2021, 13, 228. [Google Scholar] [CrossRef]
- Zambrano-Zaragoza, M.L.; Quintanar-Guerrero, D. Novel techniques for extrusion, agglomeration, encapsulation, gelation, and coating of foods. In Encyclopedia of Food Security and Sustainability; Ferranti, P., Berry, E.M., Anderson, J.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 379–392. [Google Scholar]
- Valdés, A.; Ramos, M.; Beltrán, A.; Jiménez, A.; Garrigós, M.D.C. State of the art of antimicrobial edible coatings for food packaging applications. Coatings 2017, 7, 56. [Google Scholar] [CrossRef]
- Suhag, R.; Kumar, N.; Petkoska, A.T.; Upadhyay, A. Film formation and deposition methods of edible coating on food products: A review. Food Res. Int. 2020, 136, 109582. [Google Scholar] [CrossRef]
- Wang, H.; Ding, F.; Ma, L.; Zhang, Y. Edible films from chitosan-gelatin: Physical properties and food packaging application. Food Biosci. 2021, 40, 100871. [Google Scholar] [CrossRef]
- Ding, F.; Hu, B.; Lan, S.; Wang, H. Flexographic and screen printing of carboxymethyl chitosan based edible inks for food packaging applications. Food Packag. Shelf Life 2020, 26, 100559. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, D.; Sun, Z.; Liu, F.; Du, L.; Wang, D. Preparation and characterization of gelatin/chitosan/3-phenylacetic acid food-packaging nanofiber antibacterial films by electrospinning. Int. J. Biol. Macromol. 2021, 169, 161–170. [Google Scholar] [CrossRef]
- Lin, L.; Gu, Y.; Cui, H. Moringa oil/chitosan nanoparticles embedded gelatin nanofibers for food packaging against Listeria monocytogenes and Staphylococcus aureus on cheese. Food Packag. Shelf Life 2019, 19, 86–93. [Google Scholar] [CrossRef]
- Ebrahimi, S.; Fathi, M.; Kadivar, M. Production and characterization of chitosan-gelatin nanofibers by nozzle-less electrospinning and their application to enhance edible film’s properties. Food Packag. Shelf Life 2019, 22, 100387. [Google Scholar] [CrossRef]
- Liu, Y.T.; Yuan, Y.; Duan, S.Q.; Li, C.; Hu, B.; Liu, A.P.; Wu, D.T.; Cui, H.Y.; Lin, L.; He, J.L.; et al. Preparation and characterization of chitosan films with three kinds of molecular weight for food packaging. Int. J. Biol. Macromol. 2020, 155, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Moalla, S.; Ammar, I.; Fauconnier, M.-L.; Danthine, S.; Blecker, C.; Besbes, S.; Attia, H. Development and characterization of chitosan films carrying Artemisia campestris antioxidants for potential use as active food packaging materials. Int. J. Biol. Macromol. 2021, 183, 254–266. [Google Scholar] [CrossRef]
- Min, T.; Zhu, Z.; Sun, X.; Yuan, Z.; Zha, J.; Wen, Y. Highly efficient antifogging and antibacterial food packaging film fabricated by novel quaternary ammonium chitosan composite. Food Chem. 2020, 308, 125682. [Google Scholar] [CrossRef]
- Gao, Q.; Lei, M.; Zhou, K.; Liu, X.; Wang, S.; Li, H. Preparation of a microfibrillated cellulose/chitosan/polypyrrole film for Active Food Packaging. Prog. Org. Coatings 2020, 149, 105907. [Google Scholar] [CrossRef]
- Chaparro-Hernández, S.; Ruiz-Cruz, S.; Márquez-Ríos, E.; Ornelas-Paz, J.D.J.; Del-Toro-Sánchez, C.L.; Gassos-Ortega, L.E.; Ocaño-Higuera, V.M.; López-Mata, M.A.; Devora-Isiordia, G.E. Effect of chitosan–tomato plant extract edible coating on the quality, shelf life, and antioxidant capacity of pork during refrigerated storage. Coatings 2019, 9, 827. [Google Scholar] [CrossRef]
- Sedlaříková, J.; Janalíková, M.; Rudolf, O.; Pavlačková, J.; Egner, P.; Peer, P.; Varaďová, V.; Krejčí, J. Chitosan/thyme oil systems as affected by stabilizing agent: Physical and antimicrobial properties. Coatings 2019, 9, 165. [Google Scholar] [CrossRef]
- Riaz, A.; Lagnika, C.; Luo, H.; Dai, Z.; Nie, M.; Hashim, M.M.; Liu, C.; Song, J.; Li, D. Chitosan-based biodegradable active food packaging film containing Chinese chive (Allium tuberosum) root extract for food application. Int. J. Biol. Macromol. 2020, 150, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Narasagoudr, S.; Hegde, V.G.; Chougale, R.B.; Masti, S.P.; Dixit, S. Influence of boswellic acid on multifunctional properties of chitosan/poly (vinyl alcohol) films for active food packaging. Int. J. Biol. Macromol. 2020, 154, 48–61. [Google Scholar] [CrossRef]
- Narasagoudr, S.S.; Hegde, V.G.; Chougale, R.B.; Masti, S.P.; Vootla, S.; Malabadi, R.B. Physico-chemical and functional properties of rutin induced chitosan/poly (vinyl alcohol) bioactive films for food packaging applications. Food Hydrocoll. 2020, 109, 106096. [Google Scholar] [CrossRef]
- Kadam, A.A.; Singh, S.; Gaikwad, K.K. Chitosan based antioxidant films incorporated with pine needles (Cedrus deodara) extract for active food packaging applications. Food Control 2021, 124, 107877. [Google Scholar] [CrossRef]
- Wang, K.; Lim, P.N.; Tong, S.Y.; Thian, E.S. Development of grapefruit seed extract-loaded poly(ε-caprolactone)/chitosan films for antimicrobial food packaging. Food Packag. Shelf Life 2019, 22, 100396. [Google Scholar] [CrossRef]
- Hu, H.; Yao, X.; Qin, Y.; Yong, H.; Liu, J. Development of multifunctional food packaging by incorporating betalains from vegetable amaranth (Amaranthus tricolor L.) into quaternary ammonium chitosan/fish gelatin blend films. Int. J. Biol. Macromol. 2020, 159, 675–684. [Google Scholar] [CrossRef]
- Annu, A.A.; Ahmed, S. Eco-friendly natural extract loaded antioxidative chitosan/polyvinyl alcohol based active films for food packaging. Heliyon 2021, 7, e06550. [Google Scholar] [CrossRef]
- Lei, Y.; Mao, L.; Yao, J.; Zhu, H. Improved mechanical, antibacterial and UV barrier properties of catechol-functionalized chitosan/polyvinyl alcohol biodegradable composites for active food packaging. Carbohydr. Polym. 2021, 264, 117997. [Google Scholar] [CrossRef]
- Hajji, S.; Kchaou, H.; Bkhairia, I.; Salem, R.B.S.B.; Boufi, S.; Debeaufort, F.; Nasri, M. Conception of active food packaging films based on crab chitosan and gelatin enriched with crustacean protein hydrolysates with improved functional and biological properties. Food Hydrocoll. 2021, 116, 106639. [Google Scholar] [CrossRef]
- Bhat, V.G.; Narasagoudr, S.S.; Masti, S.P.; Chougale, R.B.; Shanbhag, Y. Hydroxy citric acid cross-linked chitosan/guar gum/poly(vinyl alcohol) active films for food packaging applications. Int. J. Biol. Macromol. 2021, 177, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Huang, J.; Li, H.; Pan, Y.; Zhu, B.; Zhao, Y.; Liu, H. Chitosan-riboflavin composite film based on photodynamic inactivation technology for antibacterial food packaging. Int. J. Biol. Macromol. 2021, 172, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Guo, T.; Li, H. Evaluation of viscosity and printing quality of chitosan-based flexographic inks: The effect of chitosan molecular weight. J. Appl. Polym. Sci. 2016, 133, 43997. [Google Scholar] [CrossRef]
- Hidaka, M.; Kojima, M.; Nakahata, M.; Sakai, S. Visible light-curable chitosan ink for extrusion-based and vat polymerization-based 3D bioprintings. Polymers 2021, 13, 1382. [Google Scholar] [CrossRef]
- Zou, Y.; Zhang, C.; Wang, P.; Zhang, Y.; Zhang, H. Electrospun chitosan/polycaprolactone nanofibers containing chlorogenic acid-loaded halloysite nanotube for active food packaging. Carbohydr. Polym. 2020, 247, 116711. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Mehrotra, G.K.; Dutta, P.K. Chitosan based ZnO nanoparticles loaded gallic-acid films for active food packaging. Food Chem. 2021, 334, 127605. [Google Scholar] [CrossRef]
- Kaur, J.; Sood, K.; Bhardwaj, N.; Arya, S.K.; Khatri, M. Nanomaterial loaded chitosan nanocomposite films for antimicrobial food packaging. Mater. Today Proc. 2020, 28, 1904–1909. [Google Scholar] [CrossRef]
- Wang, Y.; Cen, C.; Chen, J.; Fu, L. MgO/carboxymethyl chitosan nanocomposite improves thermal stability, waterproof and antibacterial performance for food packaging. Carbohydr. Polym. 2020, 236, 116078. [Google Scholar] [CrossRef]
- Pandey, V.K.; Upadhyay, S.N.; Niranjan, K.; Mishra, P.K. Antimicrobial biodegradable chitosan-based composite nano-layers for food packaging. Int. J. Biol. Macromol. 2020, 157, 212–219. [Google Scholar] [CrossRef]
- Lin, D.; Yang, Y.; Wang, J.; Yan, W.; Wu, Z.; Chen, H.; Zhang, Q.; Wu, D.; Qin, W.; Tu, Z. Preparation and characterization of TiO2-Ag loaded fish gelatin-chitosan antibacterial composite film for food packaging. Int. J. Biol. Macromol. 2020, 154, 123–133. [Google Scholar] [CrossRef]
- Leceta, I.; Guerrero, P.; Ibarburu, I.; Dueñas, M.T.; de la Caba, K. Characterization and antimicrobial analysis of chitosan-based films. J. Food Eng. 2013, 116, 889–899. [Google Scholar] [CrossRef]
- Costa, S.M.; Ferreira, D.P.; Teixeira, P.; Ballesteros, L.F.; Teixeira, J.A.; Fangueiro, R. Active natural-based films for food packaging applications: The combined effect of chitosan and nanocellulose. Int. J. Biol. Macromol. 2021, 177, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.; Behera, K.; Chang, Y.-H.; Chiu, F.-C. Cellulose nanocrystal reinforced chitosan based UV barrier composite films for sustainable packaging. Polymers 2020, 12, 202. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, M.P.; Lopez, J.J.; Lopez, D.; Kenny, J.M.; Peponi, L. Effect of chitosan and catechin addition on the structural, thermal, mechanical and disintegration properties of plasticized electrospun PLA-PHB biocomposites. Polym. Degrad. Stab. 2016, 132, 145–156. [Google Scholar] [CrossRef]
- Toloue, E.B.; Karbasi, S.; Salehi, H.; Rafienia, M. Potential of an electrospun composite scaffold of poly (3-hydroxybutyrate)-chitosan/alumina nanowires in bone tissue engineering applications. Mater. Sci. Eng. C 2019, 99, 1075–1091. [Google Scholar] [CrossRef]
- Li, H.; Wang, Z.; Zhang, H.; Pan, Z. Nanoporous PLA/(chitosan nanoparticle) composite fibrous membranes with excellent air filtration and antibacterial performance. Polymers 2018, 10, 1085. [Google Scholar] [CrossRef]
- Garg, U.; Chauhan, S.; Nagaich, U.; Jain, N. Current advances in chitosan nanoparticles based drug delivery and targeting. Adv. Pharm. Bull. 2019, 9, 195–204. [Google Scholar] [CrossRef]
- Fauzi, B.; Nawawi, M.G.M.; Fauzi, R.; Mamauod, S.N.L. Physicochemical characteristics of sago starch-chitosan nanofillers film. BioResources 2019, 14, 8324–8330. [Google Scholar]
- Amjadi, S.; Emaminia, S.; Nazari, M.; Davudian, S.H.; Roufegarinejad, L.; Hamishehkar, H. Application of reinforced ZnO nanoparticle-incorporated gelatin bionanocomposite film with chitosan nanofiber for packaging of chicken fillet and cheese as food models. Food Bioprocess Technol. 2019, 12, 1205–1219. [Google Scholar] [CrossRef]
- Vahedikia, N.; Garavand, F.; Tajeddin, B.; Cacciotti, I.; Jafari, S.M.; Omidi, T.; Zahedi, Z. Biodegradable zein film composites reinforced with chitosan nanoparticles and cinnamon essential oil: Physical, mechanical, structural and antimicrobial attributes. Colloids Surf. B Biointerfaces 2019, 177, 25–32. [Google Scholar] [CrossRef]
- Pranoto, Y.; Rakshit, S.; Salokhe, V. Enhancing antimicrobial activity of chitosan films by incorporating garlic oil, potassium sorbate and nisin. LWT 2005, 38, 859–865. [Google Scholar] [CrossRef]
- Ai, H.; Wang, F.; Xia, Y.; Chen, X.; Lei, C. Antioxidant, antifungal and antiviral activities of chitosan from the larvae of housefly, Musca domestica L. Food Chem. 2012, 132, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xiao, G.; Wang, Y.; Zhao, Y.; Su, H.; Tan, T. Preparation of chitosan-TiO2 composite film with efficient antimicrobial activities under visible light for food packaging applications. Carbohydr. Polym. 2017, 169, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Riaz, A.; Lei, S.; Akhtar, H.M.S.; Wan, P.; Chen, D.; Jabbar, S.; Abid, M.; Hashim, M.M.; Zeng, X. Preparation and characterization of chitosan-based antimicrobial active food packaging film incorporated with apple peel polyphenols. Int. J. Biol. Macromol. 2018, 114, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Kumar, P.; Pandey, J. Binary grafted chitosan film: Synthesis, characterization, antibacterial activity and prospects for food packaging. Int. J. Biol. Macromol. 2018, 115, 341–348. [Google Scholar] [CrossRef]
- Xiao, W.; Xu, J.; Liu, X.; Hu, Q.; Huang, J. Antibacterial hybrid materials fabricated by nanocoating of microfibril bundles of cellulose substance with titania/chitosan/silver-nanoparticle composite films. J. Mater. Chem. B 2013, 1, 3477–3485. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, X.; Li, W.; Song, R.; Li, J.; Li, Y.; Li, B.; Liu, S. Green and biodegradable composite films with novel antimicrobial performance based on cellulose. Food Chem. 2016, 197, 250–256. [Google Scholar] [CrossRef]
- Hu, D.; Wang, H.; Wang, L. Physical properties and antibacterial activity of quaternized chitosan/carboxymethyl cellulose blend films. LWT 2016, 65, 398–405. [Google Scholar] [CrossRef]
- Davidovich-Pinhas, M.; Danin-Poleg, Y.; Kashi, Y.; Bianco-Peled, H. Modified chitosan: A step toward improving the properties of antibacterial food packages. Food Packag. Shelf Life 2014, 1, 160–169. [Google Scholar] [CrossRef]

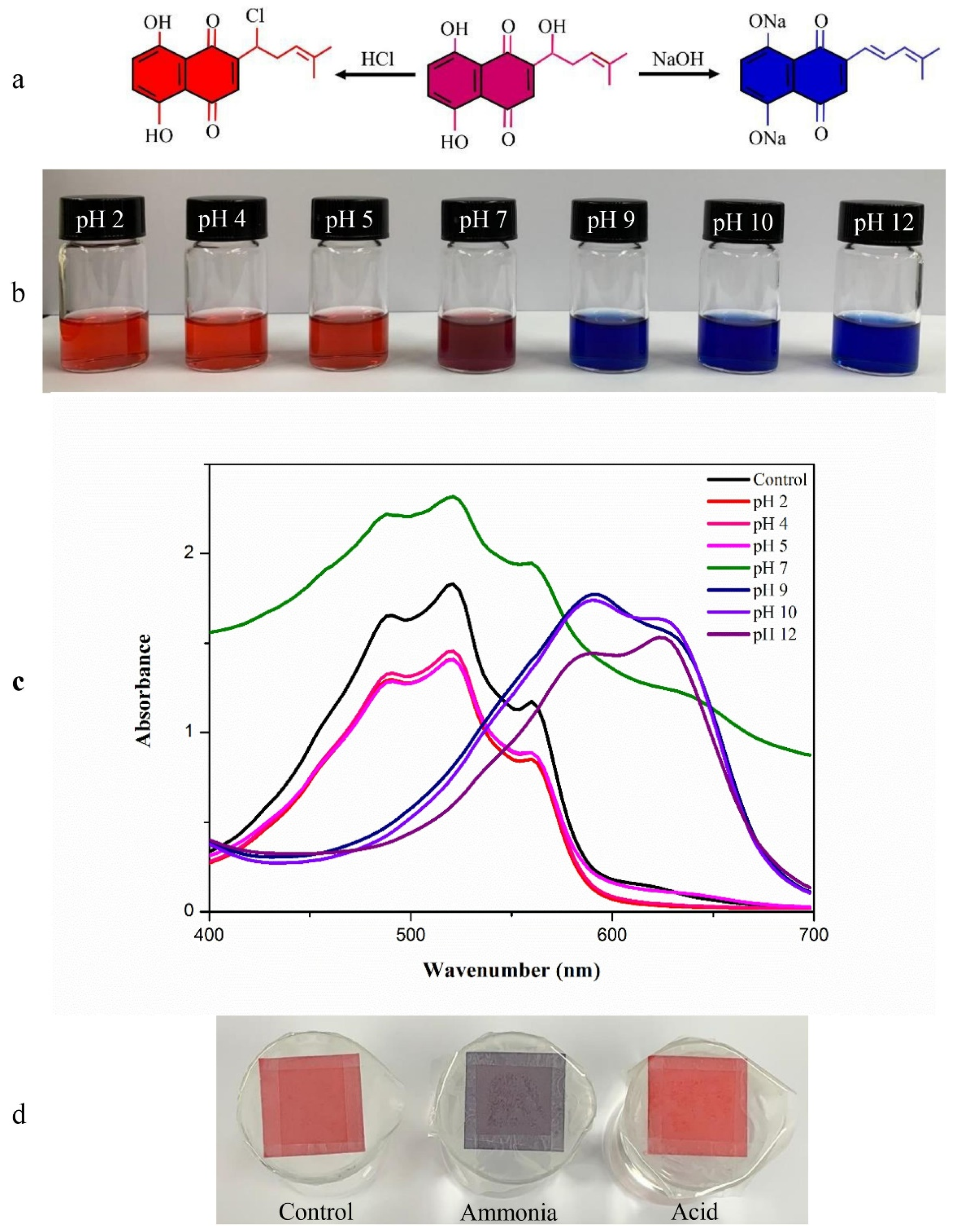
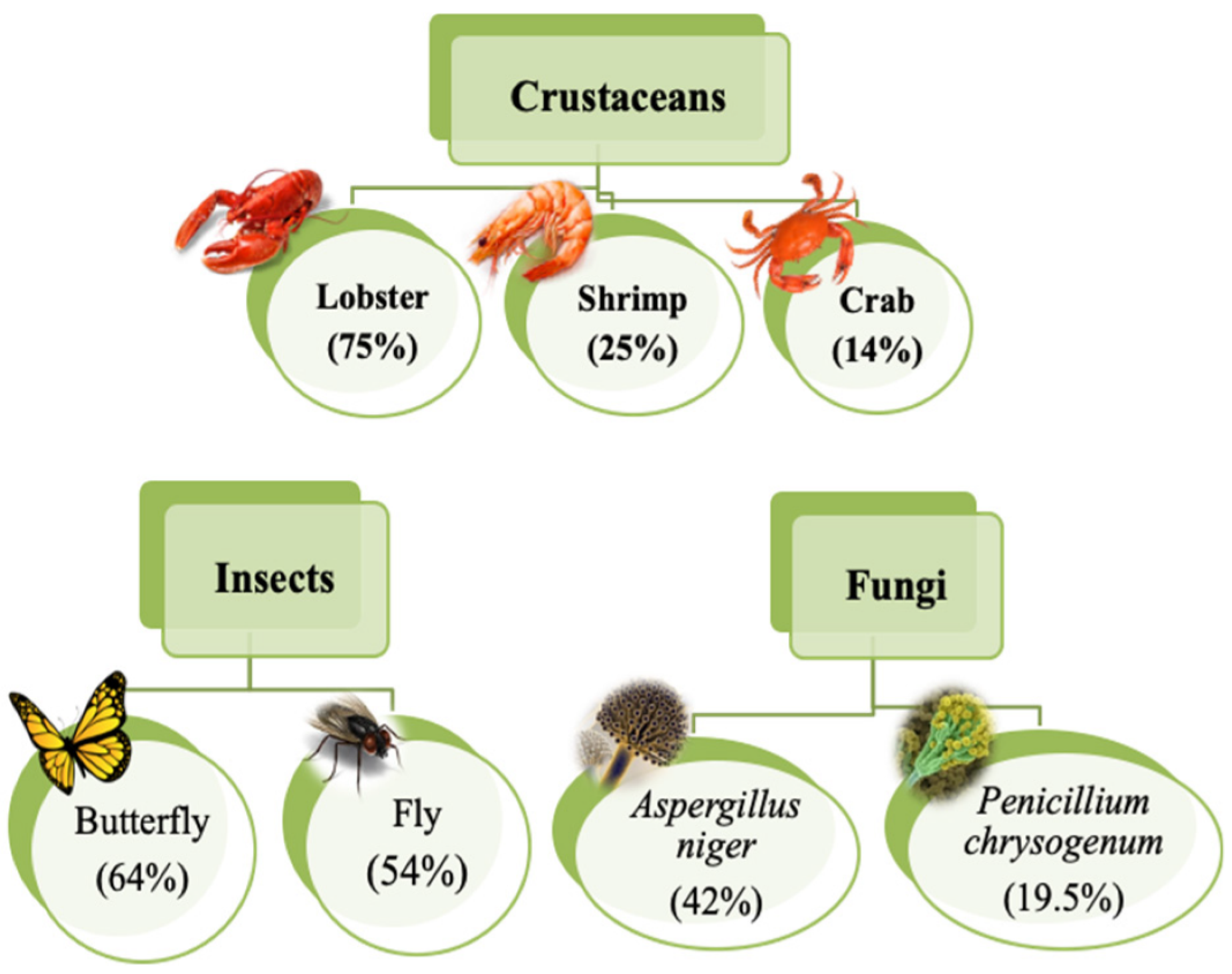
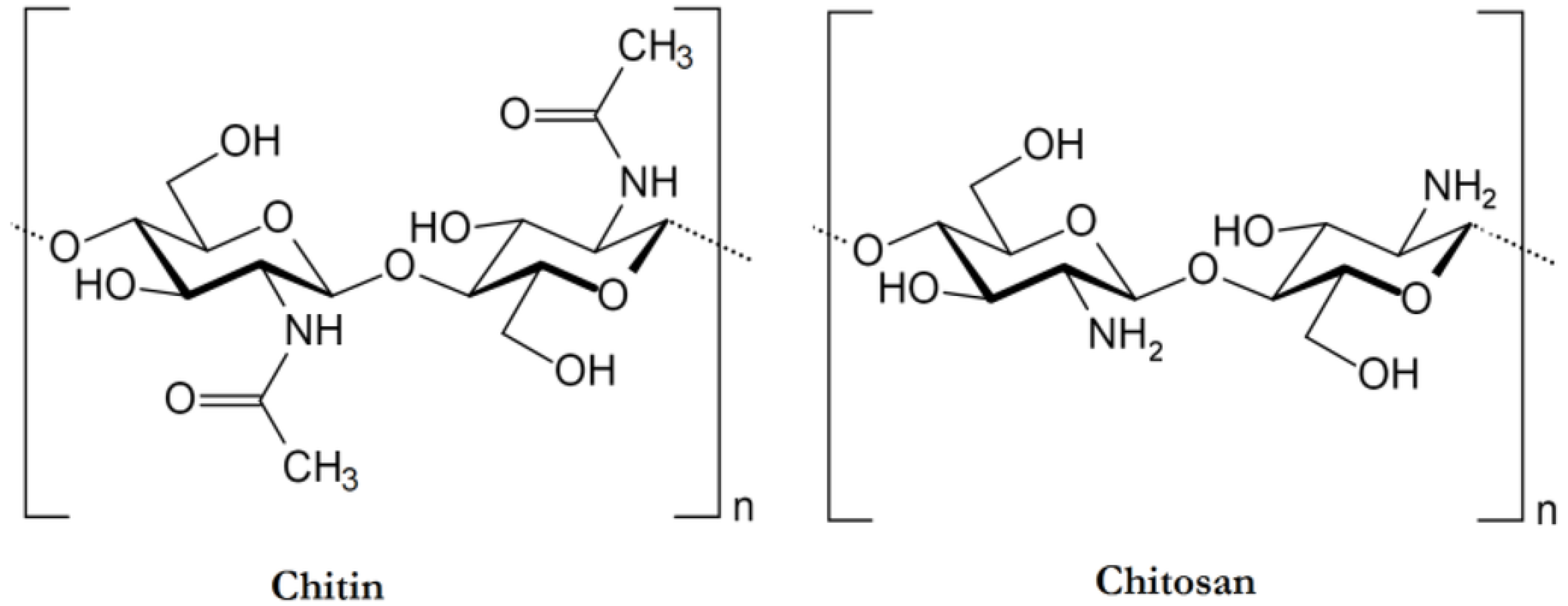
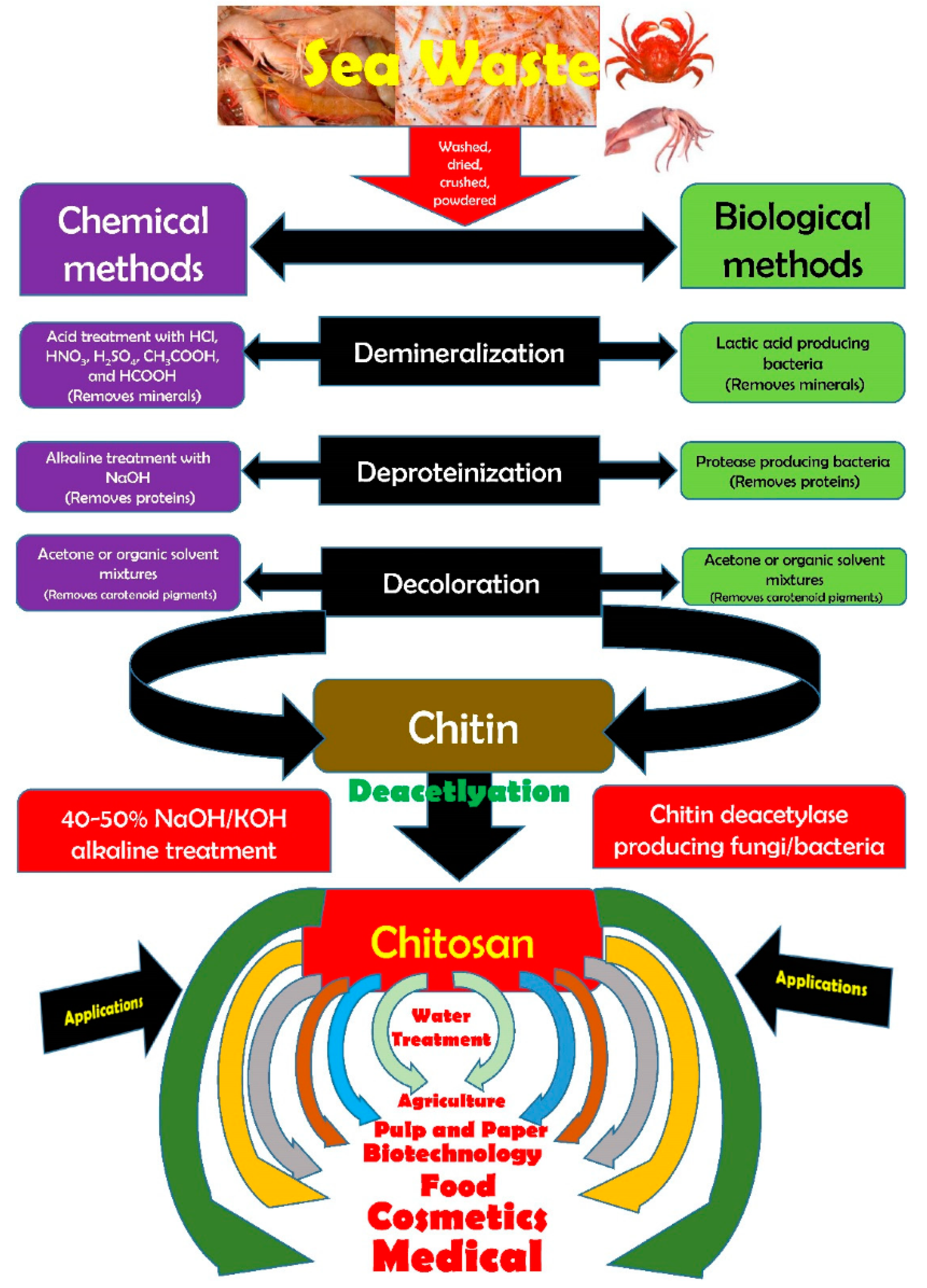




| Matrix Material(s) | Source of Chitosan | Antimicrobial Activity | Food Storage Effectiveness | References |
|---|---|---|---|---|
| Chitosan | Larvae of housefly | E. coli, Staphylococcus aureus, Salmonella typhimurium, Listeria monocytogenes, and Bacillus cereus. | Including garlic oil into chitosan film acts as a physical and antimicrobial barrier to food contamination. | [64] |
| Chitosan | Larvae of housefly | Rhizoctonia solani, Thanatephorus cucumeris, Sclerotina sclerotiorum, Curvularia lunata | Natural antioxidant for functional food products and holds the antifungal property. | [65] |
| PVA, Chitosan, and silver nanoparticle | - | E. coli and L. monocytogenes | PVA-CH-Ag composite nano-layer displayed better organoleptic qualities, such as visual appearance and smell of packaged meat. | [52] |
| Chitosan-TiO2 | Shrimp shells | E. coli, S. aureus, C. albicans, and A. niger | Chitosan-TiO2 film successfully shields red grapes from microbial spoilage and extends their shelf life. | [66] |
| Chitosan, apple peel polyphenols | - | E. coli, B. cereus, S. aureus and S. typhimurium | The incorporation of polyphenols in the chitosan film can show an enhanced shelf life of food products | [67] |
| Binary grafted chitosan film with acrylamide and acrylonitrile | - | Staphylococcus aureus, Escherichia coli, Pseudomonas aeroginosa | Protect apple and guava from microbial spoilage | [68] |
| Cellulose/titania/chitosan | Shrimp shells | E. coli and S. aureus | The formed hybrid material has enhanced antibacterial property essential for food packaging | [69] |
| Cellulose, chitosan | E. coli and Staphylococcus aureus. | Better performance than traditional polyethylene packaging of sausages | [70] | |
| Carboxymethyl cellulose, 2-N-Hydroxypropyl-3-trimethylammonium chloride chitosan | - | E. coli and Staphylococcus aureus. | The blended films have shown extended shelf life for packaging of banana | [71] |
| PEGylated chitosan | - | E.coli | Mechanical and thermally stable films for food packaging enhances anti-bacterial property | [72] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pal, K.; Bharti, D.; Sarkar, P.; Anis, A.; Kim, D.; Chałas, R.; Maksymiuk, P.; Stachurski, P.; Jarzębski, M. Selected Applications of Chitosan Composites. Int. J. Mol. Sci. 2021, 22, 10968. https://doi.org/10.3390/ijms222010968
Pal K, Bharti D, Sarkar P, Anis A, Kim D, Chałas R, Maksymiuk P, Stachurski P, Jarzębski M. Selected Applications of Chitosan Composites. International Journal of Molecular Sciences. 2021; 22(20):10968. https://doi.org/10.3390/ijms222010968
Chicago/Turabian StylePal, Kunal, Deepti Bharti, Preetam Sarkar, Arfat Anis, Doman Kim, Renata Chałas, Paweł Maksymiuk, Piotr Stachurski, and Maciej Jarzębski. 2021. "Selected Applications of Chitosan Composites" International Journal of Molecular Sciences 22, no. 20: 10968. https://doi.org/10.3390/ijms222010968
APA StylePal, K., Bharti, D., Sarkar, P., Anis, A., Kim, D., Chałas, R., Maksymiuk, P., Stachurski, P., & Jarzębski, M. (2021). Selected Applications of Chitosan Composites. International Journal of Molecular Sciences, 22(20), 10968. https://doi.org/10.3390/ijms222010968









