Factors Affecting the Progression of Infection-Related Glomerulonephritis to Chronic Kidney Disease
Abstract
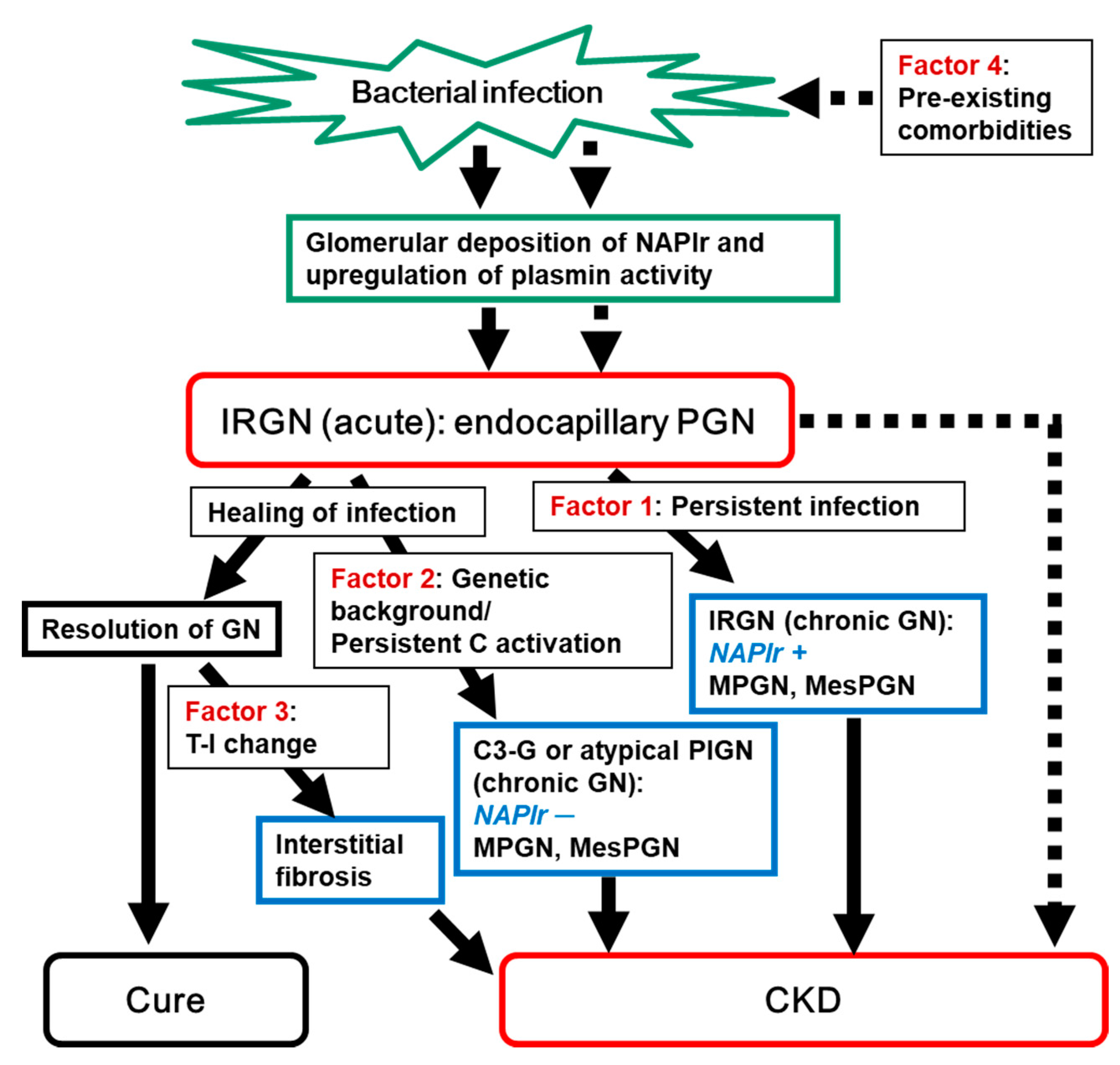
1. Introduction
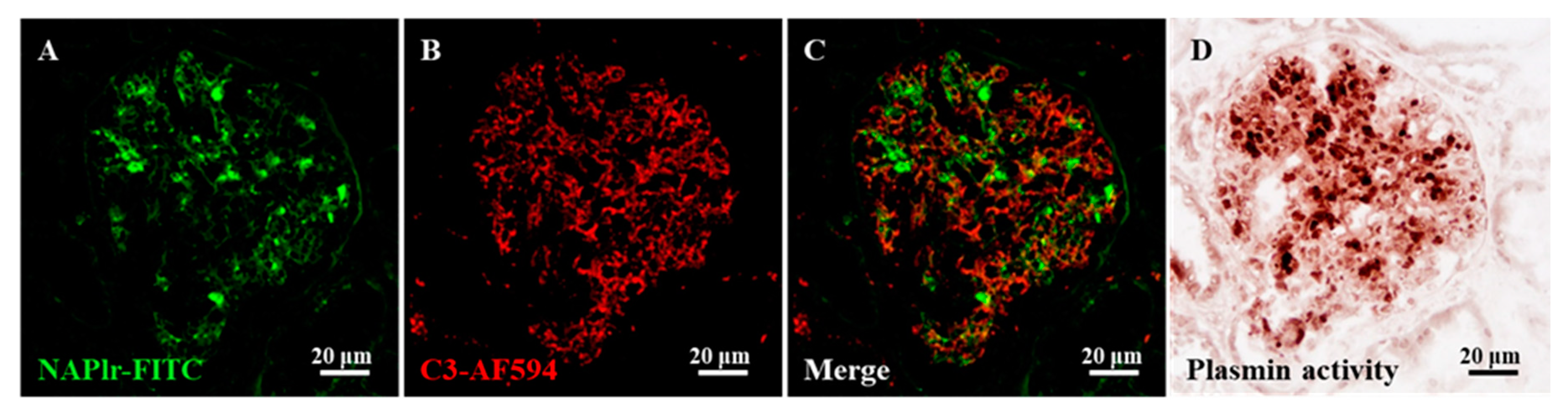
2. Persistent Infection as a Possible Cause of the Progression of IRGN into CKD
3. Genetic Background of the Host’s Complement Regulatory System as a Possible Cause of the Progression of IRGN into CKD
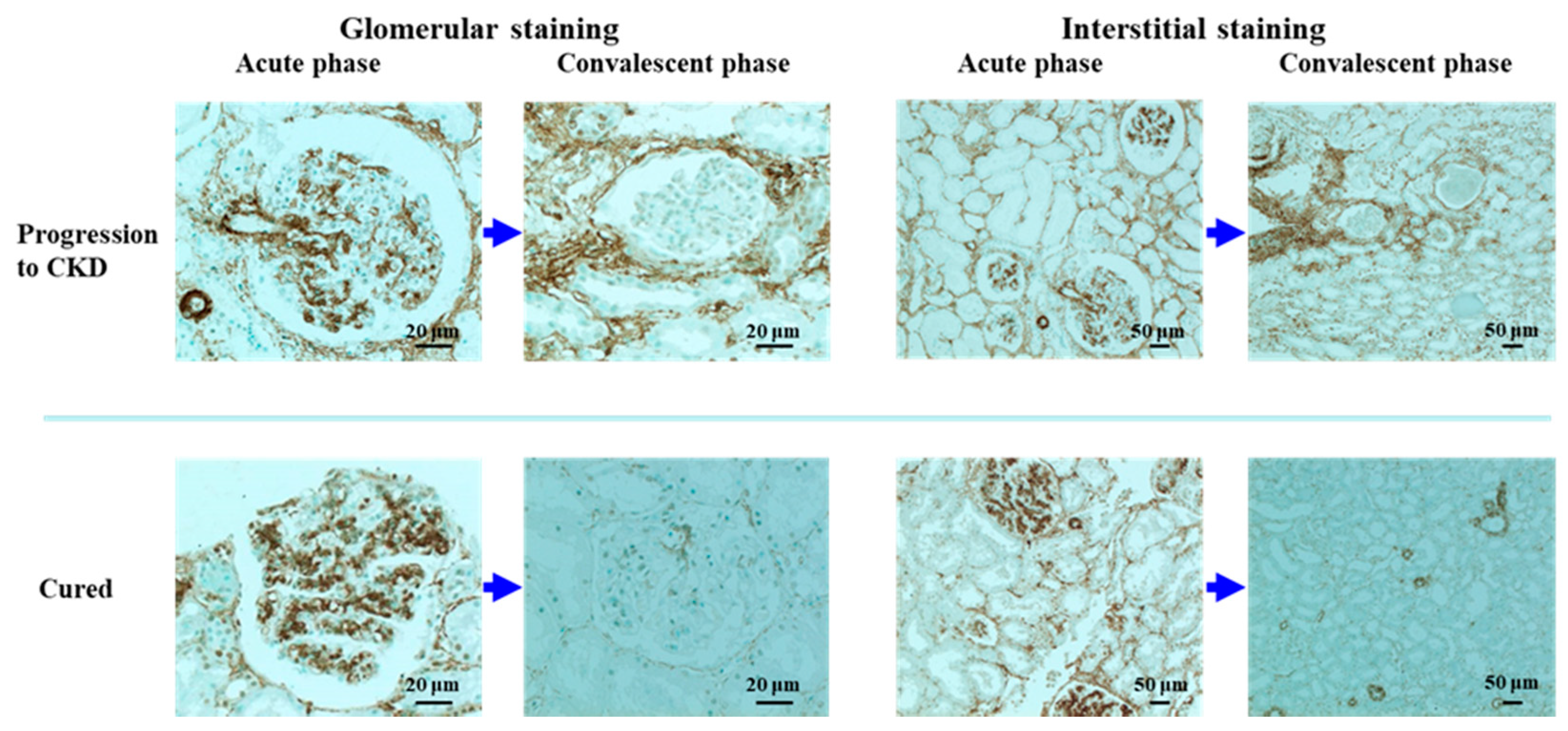
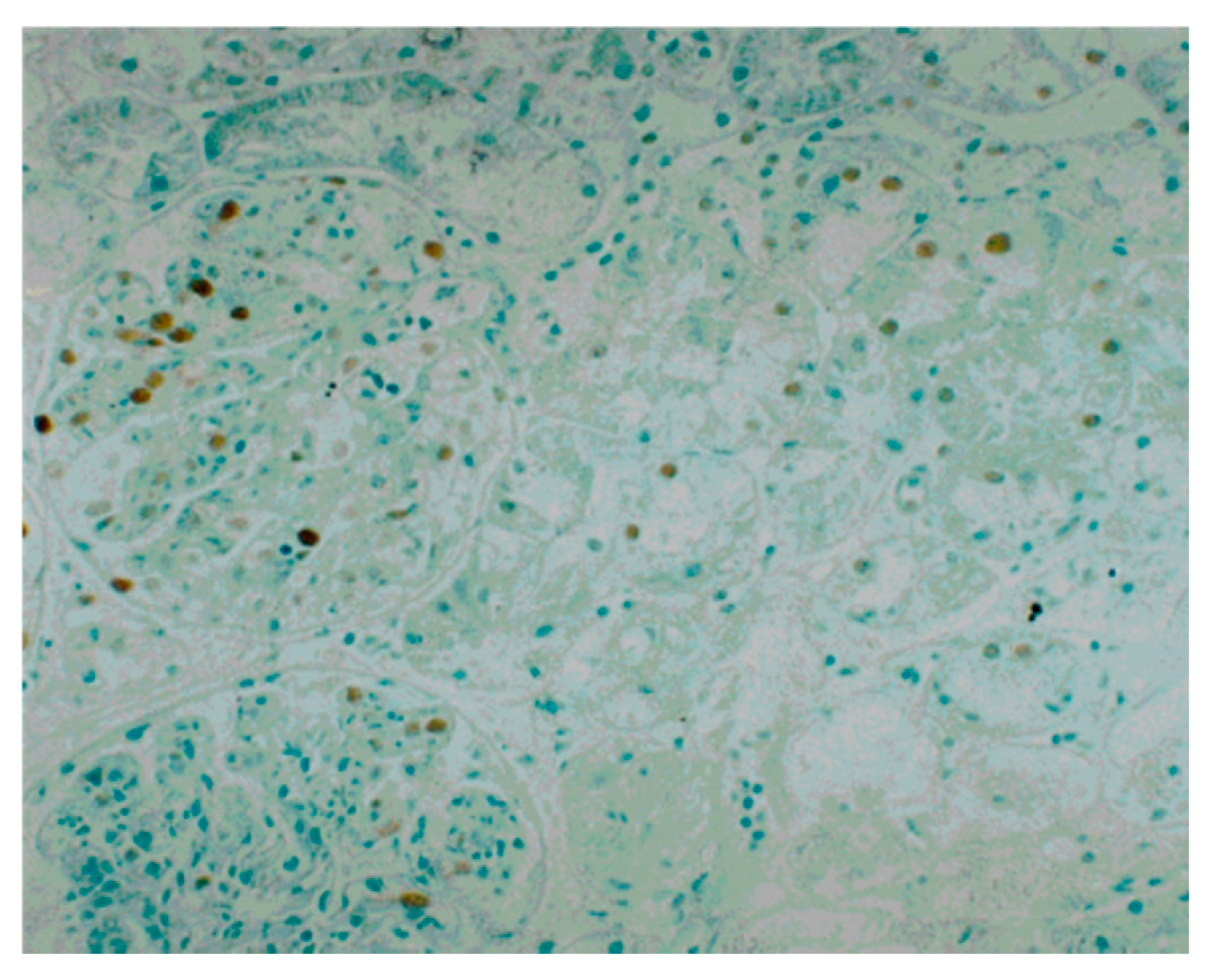
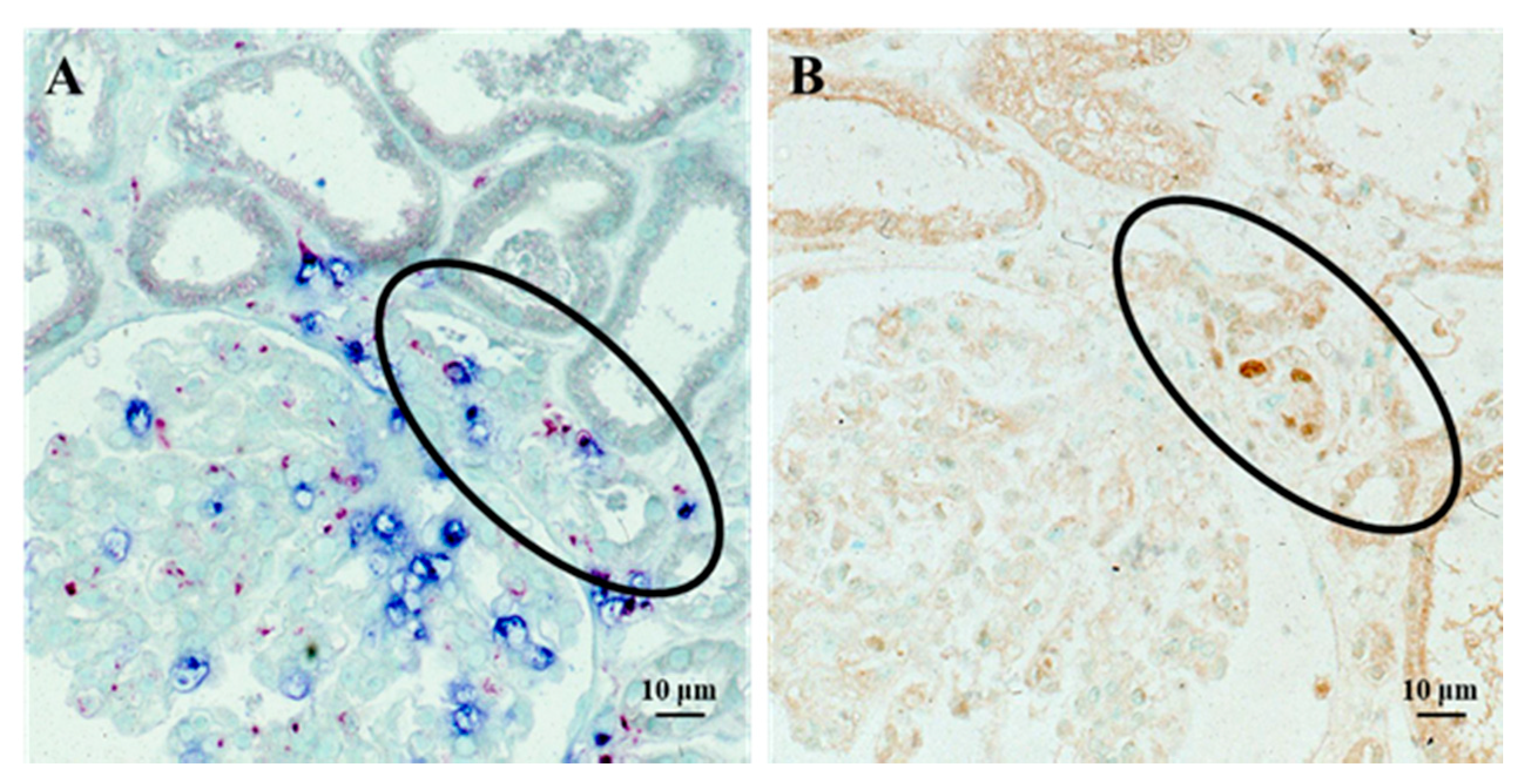
4. Continuous Tubulointerstitial Changes Independent of Glomerular Changes as a Possible Cause of the Progression of IRGN into CKD
5. Pre-Existing Kidney Injury as a Possible Cause of the Progression of IRGN into CKD
6. Detection of Autoantibodies in IRGN Patients
7. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coca, S.G.; Singanamala, S.; Parikh, C.R. Chronic kidney disease after acute kidney injury: A systematic review and meta-analysis. Kidney Int. 2012, 81, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Satoskar, A.A.; Parikh, S.V.; Nadasdy, T. Epidemiology, pathogenesis, treatment and outcomes of infection-associated glomerulonephritis. Nat. Rev. Nephrol. 2020, 16, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Radhakrishnan, J.; D’Agati, V.D. Bacterial infection-related glomerulonephritis in adults. Kidney Int. 2013, 83, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Fidler, M.E.; Valeri, A.M.; Cornell, L.D.; Sethi, S.; Zoller, A.; Stokes, M.B.; Markowitz, G.S.; D’Agati, V.D. Postinfectious glomerulonephritis in the elderly. J. Am. Soc. Nephrol. 2011, 22, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, D.S.; Gluck, M.C.; Schacht, R.G.; Gallo, G. The long-term course of poststreptococcal glomerulonephritis. Ann. Intern. Med. 1974, 80, 342–358. [Google Scholar] [CrossRef]
- Schacht, R.G.; Gluck, M.C.; Gallo, G.R.; Baldwin, D.S. Progression to uremia after remission of acute poststreptococcal glomerulonephritis. N. Engl. J. Med. 1976, 295, 977–981. [Google Scholar] [CrossRef]
- Gallo, G.R.; Feiner, H.D.; Steele, J.M., Jr.; Schacht, R.G.; Gluck, M.C.; Baldwin, D.S. Role of intrarenal vascular sclerosis in progression of poststreptococcal glomerulonephritis. Clin. Nephrol. 1980, 13, 49–57. [Google Scholar]
- Hoy, W.E.; White, A.V.; Dowling, A.; Sharma, S.K.; Bloomfield, H.; Tipiloura, B.T.; Swanson, C.E.; Mathews, J.D.; McCredie, D.A. Post-streptococcal glomerulonephritis is a strong risk factor for chronic kidney disease in later life. Kidney Int. 2012, 81, 1026–1032. [Google Scholar] [CrossRef]
- Hoy, W.E.; White, A.V.; Tipiloura, B.; Singh, G.; Sharma, S.K.; Bloomfield, H.; Swanson, C.E.; Dowling, A.; McCredie, D.A. The multideterminant model of renal disease in a remote Australian Aboriginal population in the context of early life risk factors: Lower birth weight, childhood post-streptococcal glomerulonephritis, and current body mass index influence levels of albuminuria in young Aboriginal adults. Clin. Nephrol. 2015, 83 (Suppl. 1), 75–81. [Google Scholar]
- Karmarkar, M.G.; Hule, G.P.; Hase, N.K.; Mehta, P.R.; Walter, S.R.; Sriprakash, K.S. Seroprevalence of Streptococcal Inhibitor of Complement (SIC) suggests association of streptococcal infection with chronic kidney disease. BMC Nephrol. 2013, 14, 101. [Google Scholar] [CrossRef]
- Oda, T.; Yamakami, K.; Omasu, F.; Suzuki, S.; Miura, S.; Sugisaki, T.; Yoshizawa, N. Glomerular plasmin-like activity in relation to nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. J. Am. Soc. Nephrol. 2005, 16, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Fervenza, F.C.; Zhang, Y.; Zand, L.; Meyer, N.C.; Borsa, N.; Nasr, S.H.; Smith, R.J. Atypical postinfectious glomerulonephritis is associated with abnormalities in the alternative pathway of complement. Kidney Int. 2012, 83, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, G.; Abeyaratne, A.; Sundaram, M.; Fernandes, D.K.; Pawar, B.; Perry, G.J.; Sajiv, C.; Majoni, S.W. Analysis of clinical presentation, pathological spectra, treatment and outcomes of biopsy-proven acute postinfectious glomerulonephritis in adult indigenous people of the Northern Territory of Australia. Nephrology 2017, 22, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Ying, C.M.; Yao, D.T.; Ding, H.H.; Yang, C.D. Infective endocarditis with antineutrophil cytoplasmic antibody: Report of 13 cases and literature review. PLoS ONE 2014, 9, e89777. [Google Scholar] [CrossRef] [PubMed]
- Cervi, A.; Kelly, D.; Alexopoulou, I.; Khalidi, N. ANCA-associated pauci-immune glomerulonephritis in a patient with bacterial endocarditis: A challenging clinical dilemma. Clin. Nephrol. Case Stud. 2017, 5, 32–37. [Google Scholar] [CrossRef]
- Bele, D.; Kojc, N.; Perše, M.; Černe Čerček, A.; Lindič, J.; Aleš Rigler, A.; Večerić-Haler, Ž. Diagnostic and treatment challenge of unrecognized subacute bacterial endocarditis associated with ANCA-PR3 positive immunocomplex glomerulonephritis: A case report and literature review. BMC Nephrol. 2020, 21, 40. [Google Scholar] [CrossRef]
- Satoskar, A.A.; Suleiman, S.; Ayoub, I.; Hemminger, J.; Parikh, S.; Brodsky, S.V.; Bott, C.; Calomeni, E.; Nadasdy, G.M.; Rovin, B.; et al. Staphylococcus infection-associated GN–Spectrum of IgA staining and prevalence of ANCA in a single-center cohort. Clin. J. Am. Soc. Nephrol. 2017, 12, 39–49. [Google Scholar] [CrossRef]
- Sève, P.; Ferry, T.; Koenig, M.; Cathebras, P.; Rousset, H.; Broussolle, C. Lupus-like presentation of parvovirus B19 infection. Semin. Arthritis Rheum. 2005, 34, 642–648. [Google Scholar] [CrossRef]
- Cugler, T.; Carvalho, L.M.; Facincani, I.; Yamamoto, A.Y.; Silva, G.E.; Costa, R.S.; Ferriani, V.P. Severe glomerulonephritis and encephalopathy associated with parvovirus B19 infection mimicking systemic lupus erythematosus. Scand. J. Rheumatol. 2012, 41, 79–81. [Google Scholar] [CrossRef]
- Amel, R.; Monia, K.; Anis, M.; Fatma, B.F.; Chadia, L. Systemic lupus erythematous revealed by cytomegalovirus infection. Pan Afr. Med. J. 2016, 24, 241. [Google Scholar] [CrossRef]
- Chauvet, S.; Berthaud, R.; Devriese, M.; Mignotet, M.; Vieira Martins, P.; Robe-Rybkine, T.; Miteva, M.A.; Gyulkhandanyan, A.; Ryckewaert, A.; Louillet, F.; et al. Anti-Factor B Antibodies and Acute Postinfectious GN in Children. Anti-Factor B Antibodies and Acute Postinfectious GN in Children. J. Am. Soc. Nephrol. 2020, 31, 829–840. [Google Scholar] [CrossRef]
- Yoshizawa, N.; Oda, T.; Oshikawa, Y.; Akashi, Y.; Suzuki, Y.; Shimizu, J.; Shimizu, E.; Niwa, H.; Treser, G. Cell-mediated immune response in acute poststreptococcal glomerulonephritis. Nihon Jinzo Gakkai Shi 1994, 36, 322–330. [Google Scholar] [PubMed]
- Oda, T.; Yoshizawa, N.; Takeuchi, A.; Nakabayashi, I.; Nishiyama, J.; Ishida, A.; Tazawa, K.; Murayama, M.; Hotta, O.; Taguma, Y. Glomerular proliferating cell kinetics in acute post-streptococcal glomerulonephritis (APSGN). J. Pathol. 1997, 183, 359–368. [Google Scholar] [CrossRef]
- Yoshizawa, N.; Yamakami, K.; Fujino, M.; Oda, T.; Tamura, K.; Matsumoto, K.; Sugisaki, T.; Boyle, M.D. Nephritis-associated plasmin receptor and acute poststreptococcal glomerulonephritis: Characterization of the antigen and associated immune response. J. Am. Soc. Nephrol. 2004, 15, 1785–1793. [Google Scholar] [CrossRef] [PubMed]
- Yamakami, K.; Yoshizawa, N.; Wakabayashi, K.; Takeuchi, A.; Tadakuma, T.; Boyle, M.D. The potential role for nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. Methods 2000, 21, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Sawanobori, E.; Umino, A.; Kanai, H.; Matsushita, K.; Iwasa, S.; Kitamura, H.; Oda, T.; Yoshizawa, N.; Sugita, K.; Higashida, K. A prolonged course of Group A streptococcus-associated nephritis: A mild case of dense deposit disease (DDD)? Clin. Nephrol. 2009, 71, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Suga, K.; Kondo, S.; Matsuura, S.; Kinoshita, Y.; Kitano, E.; Hatanaka, M.; Kitamura, H.; Hidaka, Y.; Oda, T.; Kagami, S. A case of dense deposit disease associated with a group A streptococcal infection without the involvement of C3NeF or complement factor H deficiency. Pediatr. Nephrol. 2010, 25, 1547–1550. [Google Scholar] [CrossRef] [PubMed]
- Okabe, M.; Tsuboi, N.; Yokoo, T.; Miyazaki, Y.; Utsunomiya, Y.; Hosoya, T. A case of idiopathic membranoproliferative glomerulonephritis with a transient glomerular deposition of nephritis-associated plasmin receptor antigen. Clin. Exp. Nephrol. 2012, 16, 337–341. [Google Scholar] [CrossRef]
- Iseri, K.; Iyoda, M.; Yamamoto, Y.; Kobayashi, N.; Oda, T.; Yamaguchi, Y.; Shibata, T. Streptococcal Infection-related Nephritis (SIRN) Manifesting Membranoproliferative Glomerulonephritis Type I. Intern. Med. 2016, 55, 647–650. [Google Scholar] [CrossRef][Green Version]
- Kikuchi, Y.; Yoshizawa, N.; Oda, T.; Imakiire, T.; Suzuki, S.; Miura, S. Streptococcal origin of a case of Henoch-Schoenlein purpura nephritis. Clin. Nephrol. 2006, 65, 124–128. [Google Scholar] [CrossRef]
- Yano, K.; Suzuki, H.; Oda, T.; Ueda, Y.; Tsukamoto, T.; Muso, E. Crescentic poststreptococcal acute glomerulonephritis accompanied by small vessel vasculitis: Case report of an elderly male. BMC Nephrol. 2019, 20, 471. [Google Scholar] [CrossRef] [PubMed]
- Odaka, J.; Kanai, T.; Ito, T.; Saito, T.; Aoyagi, J.; Betsui, H.; Oda, T.; Ueda, Y.; Yamagata, T. A case of post-pneumococcal acute glomerulonephritis with glomerular depositions of nephritis-associated plasmin receptor. CEN Case Rep. 2015, 4, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Komaru, Y.; Ishioka, K.; Oda, T.; Ohtake, T.; Kobayashi, S. Nephritis-associated plasmin receptor (NAPlr) positive glomerulonephritis caused by Aggregatibacter actinomycetemcomitans bacteremia: A case report. Clin. Nephrol. 2018, 90, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Hirano, D.; Oda, T.; Ito, A.; Yamada, A.; Kakegawa, D.; Miwa, S.; Umeda, C.; Takemasa, Y.; Tokunaga, A.; Wajima, T.; et al. Glyceraldehyde-3-phosphate dehydrogenase of Mycoplasma pneumoniae induces infection-related glomerulonephritis. Clin. Nephrol. 2019, 92, 263–272. [Google Scholar] [CrossRef]
- Uchida, T.; Oda, T. Glomerular deposition of nephritis-associated plasmin receptor (NAPlr) and related plasmin activity: Key diagnostic biomarkers of bacterial infection-related glomerulonephritis. Int. J. Mol. Sci. 2020, 21, 2595. [Google Scholar] [CrossRef] [PubMed]
- Noda, S.; Mandai, S.; Oda, T.; Shinoto, T.; Sato, H.; Sato, K.; Hirokawa, K.; Noda, Y.; Uchida, S. Asymptomatic sinusitis as an origin of infection-related glomerulonephritis manifesting steroid-resistant nephrotic syndrome: A case report. Medicine 2020, 99, e20572. [Google Scholar] [CrossRef]
- Uchida, T.; Oda, T.; Watanabe, A.; Yamamoto, K.; Katsurada, Y.; Shimazaki, H.; Tamai, S.; Kumagai, H. Transition from endocapillary proliferative glomerulonephritis to membranoproliferative glomerulonephritis in a patient with a prolonged human parvovirus B19 infection. Clin. Nephrol. 2014, 82, 62–67. [Google Scholar] [CrossRef]
- Vernon, K.A.; de Jorge, E.G.; Hall, A.E.; Fremeaux-Bacchi, V.; Aitman, T.J.; Cook, H.T.; Hangartner, R.; Koziell, A.; Pickering, M.C. Acute presentation and persistent glomerulonephritis following streptococcal infection in a patient with heterozygous complement factor H-related protein 5 deficiency. Am. J. Kidney Dis. 2012, 60, 121–125. [Google Scholar] [CrossRef]
- Prasto, J.; Kaplan, B.S.; Russo, P.; Chan, E.; Smith, R.J.; Meyers, K.E. Streptococcal infection as possible trigger for dense deposit disease (C3 glomerulopathy). Eur. J. Pediatr. 2014, 173, 767–772. [Google Scholar] [CrossRef]
- Jennings, R.B.; Earle, D.P. Post-streptococcal glomerulo-nephritis: Histopathologic and clinical studies of the acute, subsiding acute and early chronic latent phases. J. Clin. Investig. 1961, 40 Pt 1–2, 1525–1595. [Google Scholar] [CrossRef]
- Schmitt, H.; de Oliveira, V.C.; Bohle, A. Tubulo-interstitial alterations in type I membranoproliferative glomerulonephritis. An investigation of 259 cases. Pathol. Res. Pract. 1987, 182, 6–10. [Google Scholar] [CrossRef]
- Bogenschütz, O.; Bohle, A.; Batz, C.; Wehrmann, M.; Pressler, H.; Kendziorra, H.; Gärtner, H.V. IgA nephritis: On the importance of morphological and clinical parameters in the long-term prognosis of 239 patients. Am. J. Nephrol. 1990, 10, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Wehrmann, M.; Bohle, A.; Held, H.; Schumm, G.; Kendziorra, H.; Pressler, H. Long-term prognosis of focal sclerosing glomerulonephritis. An analysis of 250 cases with particular regard to tubulointerstitial changes. Clin. Nephrol. 1990, 33, 115–122. [Google Scholar] [PubMed]
- Bohle, A.; Wehrmann, M.; Bogenschütz, O.; Batz, C.; Müller, C.A.; Müller, G.A. The pathogenesis of chronic renal failure in diabetic nephropathy. Investigation of 488 cases of diabetic glomerulosclerosis. Pathol. Res. Pract. 1991, 187, 251–259. [Google Scholar] [CrossRef]
- Hewitson, T.D.; Becker, G.J. Interstitial myofibroblasts in IgA glomerulonephritis. Am. J. Nephrol. 1995, 15, 111–117. [Google Scholar] [CrossRef]
- Roberts, I.S.; Burrows, C.; Shanks, J.H.; Venning, M.; McWilliam, L.J. Interstitial myofibroblasts: Predictors of progression in membranous nephropathy. J. Clin. Pathol. 1997, 50, 123–127. [Google Scholar] [CrossRef]
- Essawy, M.; Soylemezoglu, O.; Muchaneta-Kubara, E.C.; Shortland, J.; Brown, C.B.; el Nahas, A.M. Myofibroblasts and the progression of diabetic nephropathy. Nephrol. Dial. Transpl. 1997, 12, 43–50. [Google Scholar] [CrossRef]
- Alpers, C.E.; Hudkins, K.L.; Gown, A.M.; Johnson, R.J. Enhanced expression of “muscle-specific” actin in glomerulonephritis. Kidney Int. 1992, 41, 1134–1142. [Google Scholar] [CrossRef][Green Version]
- Grgic, I.; Campanholle, G.; Bijol, V.; Wang, C.; Sabbisetti, V.S.; Ichimura, T.; Humphreys, B.D.; Bonventre, J.V. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney Int. 2012, 82, 172–183. [Google Scholar] [CrossRef]
- Endo, T.; Nakamura, J.; Sato, Y.; Asada, M.; Yamada, R.; Takase, M.; Takaori, K.; Oguchi, A.; Iguchi, T.; Higashi, A.Y.; et al. Exploring the origin and limitations of kidney regeneration. J. Pathol. 2015, 236, 251–263. [Google Scholar] [CrossRef]
- Takaori, K.; Nakamura, J.; Yamamoto, S.; Nakata, H.; Sato, Y.; Takase, M.; Nameta, M.; Yamamoto, T.; Economides, A.N.; Kohno, K.; et al. Severity and Frequency of Proximal Tubule Injury Determines Renal Prognosis. J. Am. Soc. Nephrol. 2016, 27, 2393–2406. [Google Scholar] [CrossRef] [PubMed]

| Factor | Evaluation of the Involvement of Each Factor (Biomarkers) | Potential Intervention |
|---|---|---|
| 1. Persistent infection | Histological staining for NAPlr and plasmin activity [11] | Use of antimicrobial agents Removal of indwelling device |
| 2. Genetic background of the host’s complement system [12] | Serum complement levels, histological deposition of complement components, genetic testing | Use of complement—regulating medications (in the future) |
| 3. Tubulointerstitial changes | Interstitial staining for α-SMA | Not yet determined |
| 4. Pre-existing renal histological damage due to comorbidities [4,13] | Histopathological evaluation | Adequate treatment for comorbidities, such as hypertension and DM |
| Patient Age (Years) | Acute Phase: Duration between Onset to First Biopsy (Days) | Convalescent Phase: Duration between Onset to Second Biopsy (Years) | Glomerular Staining Score | Interstitial Staining Score | |||
|---|---|---|---|---|---|---|---|
| First Biopsy | Second Biopsy | First Biopsy | Second Biopsy | ||||
| Progression into CKD (n = 3) | 39.3 ± 6.0 | 19.7 ± 9.9 | 3.0 ± 1.0 | 2.7 ± 1.2 | 0.7 ± 0.6 | 3.7 ± 0.6 | 3.0 ± 1.0 |
| Cured (n = 3) | 35.7 ± 7.8 | 23.3 ± 12.1 | 5.1 ± 4.5 | 3.0 ± 1.0 | 0.7 ± 0.6 | 2.7 ± 0.6 | 1.7 ± 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oda, T.; Yoshizawa, N. Factors Affecting the Progression of Infection-Related Glomerulonephritis to Chronic Kidney Disease. Int. J. Mol. Sci. 2021, 22, 905. https://doi.org/10.3390/ijms22020905
Oda T, Yoshizawa N. Factors Affecting the Progression of Infection-Related Glomerulonephritis to Chronic Kidney Disease. International Journal of Molecular Sciences. 2021; 22(2):905. https://doi.org/10.3390/ijms22020905
Chicago/Turabian StyleOda, Takashi, and Nobuyuki Yoshizawa. 2021. "Factors Affecting the Progression of Infection-Related Glomerulonephritis to Chronic Kidney Disease" International Journal of Molecular Sciences 22, no. 2: 905. https://doi.org/10.3390/ijms22020905
APA StyleOda, T., & Yoshizawa, N. (2021). Factors Affecting the Progression of Infection-Related Glomerulonephritis to Chronic Kidney Disease. International Journal of Molecular Sciences, 22(2), 905. https://doi.org/10.3390/ijms22020905




