Toll-Like Receptors in Acute Kidney Injury
Abstract
1. Introduction
2. Pathophysiology of AKI
3. Biomarkers in AKI
4. Toll-Like Receptors: Structure, Localization and Function
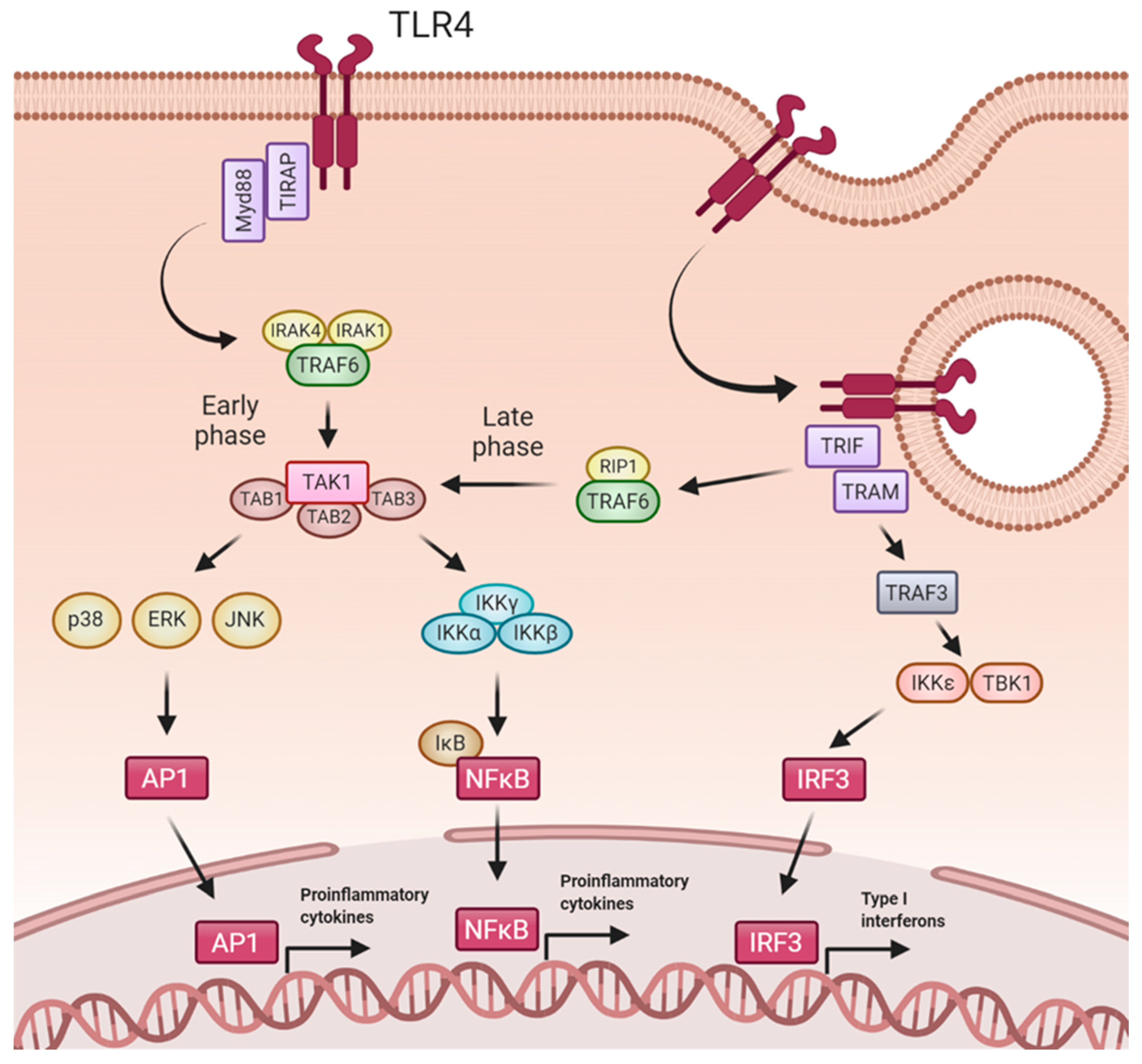
5. TLR4 Signaling Pathways
6. TLR4 Mediated Effects
7. Regulation of the TLR4 Pathway
8. TLRs and AKI
8.1. Ischemia-Reperfusion
8.2. Toxin Induced AKI
8.2.1. Endogenous Toxicity (Pigment Nephropathy)
8.2.2. Exogenous Toxicity
8.3. Sepsis-Induced AKI
8.4. Thrombotic Microangiopathy-Induced AKI
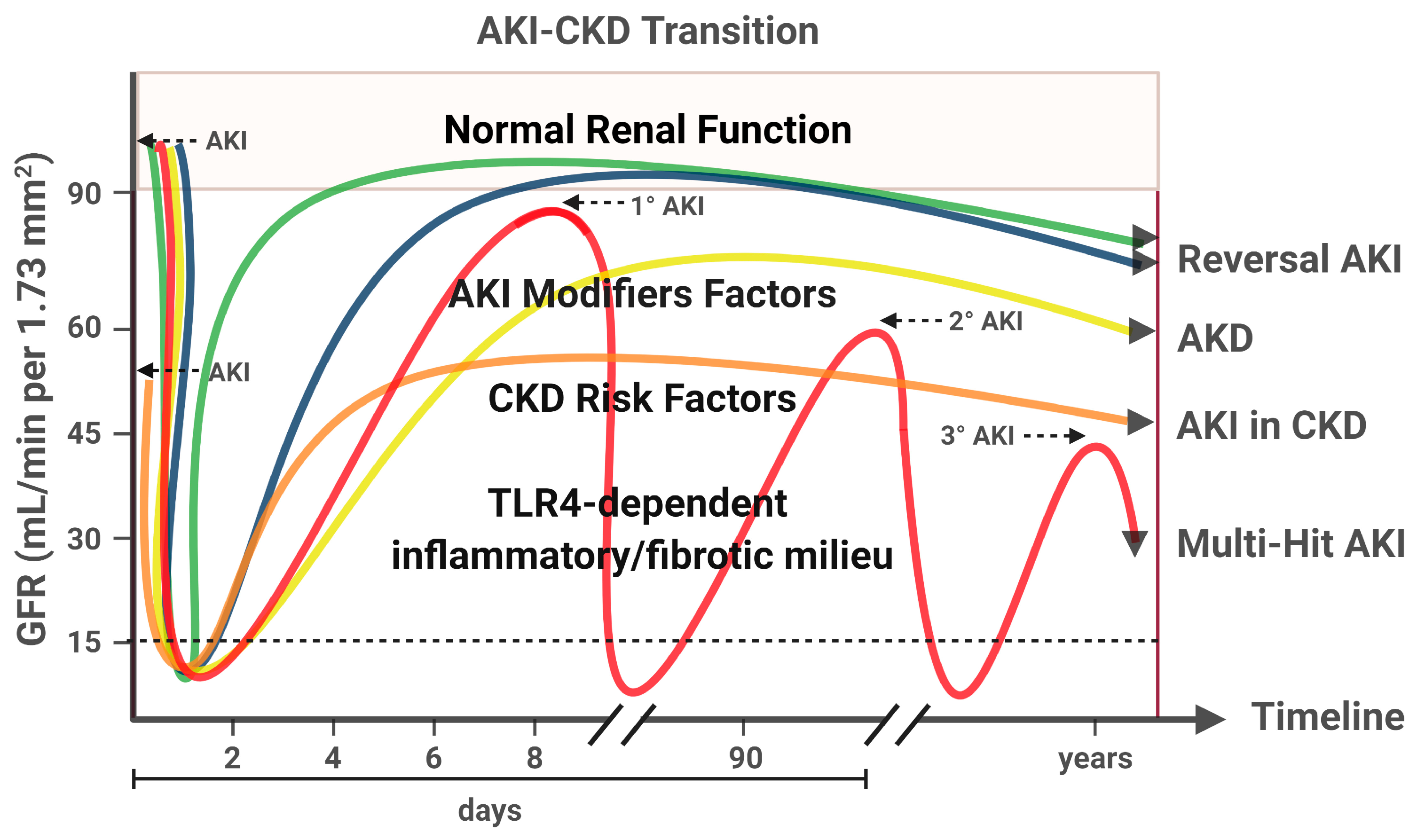
9. AKI to CKD Transition
10. Clinical Trials Targeting TLR4 in AKI
11. Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khwaja, A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef] [PubMed]
- Susantitaphong, P.; Cruz, D.N.; Cerda, J.; Abulfaraj, M.; Alqahtani, F.; Koulouridis, I.; Jaber, B.L. World incidence of AKI: A meta-analysis. Clin. J. Am. Soc. Nephrol. 2013, 8, 1482–1493. [Google Scholar] [CrossRef] [PubMed]
- Lewington, A.J.P.; Cerdá, J.; Mehta, R.L. Raising awareness of acute kidney injury: A global perspective of a silent killer. Kidney Int. 2013, 84, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Lameire, N.; Biesen, W.V.; Vanholder, R. Acute kidney injury. Lancet 2008, 372, 1863–1865. [Google Scholar] [CrossRef]
- Chawla, L.S.; Amdur, R.L.; Shaw, A.D.; Faselis, C.; Palant, C.E.; Kimmel, P.L. Association between AKI and long-term renal and cardiovascular outcomes in united states veterans. Clin. J. Am. Soc. Nephrol. 2014, 9, 448–456. [Google Scholar] [CrossRef]
- Eleftheriadis, T.; Pissas, G.; Liakopoulos, V.; Stefanidis, I.; Lawson, B.R. Toll-like receptors and their role in renal pathologies. Inflamm. Allergy Drug Targets 2012, 11, 464–477. [Google Scholar] [CrossRef]
- Bellomo, R.; Kellum, J.A.; Ronco, C. Acute kidney injury. Lancet 2012, 380, 756–766. [Google Scholar] [CrossRef]
- Ostermann, M.; Liu, K. Pathophysiology of AKI. Best Pract. Res. Clin. Anaesthesiol. 2017, 31, 305–314. [Google Scholar] [CrossRef]
- Basile, D.P.; Anderson, M.D.; Sutton, T.A. Pathophysiology of acute kidney injury. Compr. Physiol. 2012, 2, 1303–1353. [Google Scholar] [CrossRef]
- Hanif, M.O.; Ramphul, K. Renal Tubular Necrosis, Acute; StatPearls Publishing: Petersburg, FL, USA, 2018. [Google Scholar]
- Verma, S.K.; Molitoris, B.A. Renal Endothelial Injury and Microvascular Dysfunction in Acute Kidney Injury. Semin. Nephrol. 2015, 35, 96–107. [Google Scholar] [CrossRef]
- De Backer, D.; Creteur, J.; Preiser, J.C.; Dubois, M.J.; Vincent, J.L. Microvascular blood flow is altered in patients with sepsis. Am. J. Respir. Crit. Care Med. 2002, 166, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Andrade-Oliveira, V.; Foresto-Neto, O.; Watanabe, I.K.M.; Zatz, R.; Câmara, N.O.S. Inflammation in renal diseases: New and old players. Front. Pharmacol. 2019, 10, 1192. [Google Scholar] [CrossRef] [PubMed]
- Kundert, F.; Platen, L.; Iwakura, T.; Zhao, Z.; Marschner, J.A.; Anders, H.J. Immune mechanisms in the different phases of acute tubular necrosis. Kidney Res. Clin. Pract. 2018, 37, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Chawla, L.S.; Bellomo, R.; Bihorac, A.; Goldstein, S.L.; Siew, E.D.; Bagshaw, S.M.; Bittleman, D.; Cruz, D.; Endre, Z.; Fitzgerald, R.L.; et al. Acute kidney disease and renal recovery: Consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat. Rev. Nephrol. 2017, 13, 241–257. [Google Scholar] [CrossRef]
- Waikar, S.S.; Bonventre, J.V. Creatinine kinetics and the definition of acute kidney injury. J. Am. Soc. Nephrol. 2009, 20, 672–679. [Google Scholar] [CrossRef]
- Murray, P.T.; Mehta, R.L.; Shaw, A.; Ronco, C.; Endre, Z.; Kellum, J.A.; Chawla, L.S.; Cruz, D.; Ince, C.; Okusa, M.D. Potential use of biomarkers in acute kidney injury: Report and summary of recommendations from the 10th Acute Dialysis Quality Initiative consensus conference. Kidney Int. 2014, 85, 513–521. [Google Scholar] [CrossRef]
- Ostermann, M.; Zarbock, A.; Goldstein, S.; Kashani, K.; Macedo, E.; Murugan, R.; Bell, M.; Forni, L.; Guzzi, L.; Joannidis, M.; et al. Recommendations on Acute Kidney Injury Biomarkers From the Acute Disease Quality Initiative Consensus Conference: A Consensus Statement. JAMA Netw. Open 2020, 3, e2019209. [Google Scholar] [CrossRef]
- Vanmassenhove, J.; Vanholder, R.; Nagler, E.; Van Biesen, W. Urinary and serum biomarkers for the diagnosis of acute kidney injury: An in-depth review of the literature. Nephrol. Dial. Transpl. 2013, 28, 254–273. [Google Scholar] [CrossRef]
- Parikh, C.R.; Mansour, S.G. Perspective on clinical application of biomarkers in AKI. J. Am. Soc. Nephrol. 2017, 28, 1677–1685. [Google Scholar] [CrossRef]
- Yang, C.; Fan, P.C.; Chen, C.C.; Peng, C.C.; Chang, C.H.; Yang, C.H.; Chu, L.J.; Chen, Y.C.; Yang, C.W.; Chang, Y.S.; et al. A circulating miRNA signature for early diagnosis of acute kidney injury following acute myocardial infarction. J. Transl. Med. 2019, 17. [Google Scholar] [CrossRef]
- Wu, Y.L.; Li, H.F.; Chen, H.H.; Lin, H. MicroRNAs as biomarkers and therapeutic targets in inflammation-and ischemia-reperfusion-related acute renal injury. Int. J. Mol. Sci. 2020, 21, 1–16. [Google Scholar] [CrossRef]
- Vijay, K. Toll-like receptors in immunity and inflammatory diseases: Past, present, and future. Int. Immunopharmacol. 2018, 59, 391–412. [Google Scholar] [CrossRef]
- Akira, S.; Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 2004, 4, 499–511. [Google Scholar] [CrossRef]
- Jezierska, A.; Kolosova, I.A.; Verin, A.D. Toll Like Receptors Signaling Pathways as a Target for Therapeutic Interventions. Curr. Signal Transduct. Ther. 2011, 6, 428–440. [Google Scholar] [CrossRef]
- Gay, N.J.; Symmons, M.F.; Gangloff, M.; Bryant, C.E. Assembly and localization of Toll-like receptor signalling complexes. Nat. Rev. Immunol. 2014, 14, 546–558. [Google Scholar] [CrossRef]
- Sellge, G.; Kufer, T.A. PRR-signaling pathways: Learning from microbial tactics. Semin. Immunol. 2015, 27, 75–84. [Google Scholar] [CrossRef]
- Anders, H.J.; Banas, B.; Schlöndorff, D. Signaling Danger: Toll-Like Receptors and their Potential Roles in Kidney Disease. J. Am. Soc. Nephrol. 2004, 15, 854–867. [Google Scholar] [CrossRef]
- El-Achkar, T.M.; Dagher, P.C. Renal Toll-like receptors: Recent advances and implications for disease. Nat. Clin. Pract. Nephrol. 2006, 2, 568–581. [Google Scholar] [CrossRef]
- Doyle, S.L.; O’Neill, L.A.J. Toll-like receptors: From the discovery of NFκB to new insights into transcriptional regulations in innate immunity. Biochem. Pharmacol. 2006, 72, 1102–1113. [Google Scholar] [CrossRef]
- Pasare, C.; Medzhitov, R. Toll-like receptors: Linking innate and adaptive immunity. Microbes Infect. 2004, 6, 1382–1387. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, H.; Zhao, M.; Lu, Q. TLR2 and TLR4 in Autoimmune Diseases: A Comprehensive Review. Clin. Rev. Allergy Immunol. 2014, 47, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, R.T.; Fernandez, P.L.; Mourao-Sa, D.S.; Porto, B.N.; Dutra, F.F.; Alves, L.S.; Oliveira, M.F.; Oliveira, P.L.; Graça-Souza, A.V.; Bozza, M.T. Characterization of heme as activator of toll-like receptor 4. J. Biol. Chem. 2007, 282, 20221–20229. [Google Scholar] [CrossRef] [PubMed]
- Vaure, C.; Liu, Y. A comparative review of toll-like receptor 4 expression and functionality in different animal species. Front. Immunol. 2014, 5, 316. [Google Scholar] [PubMed]
- Leemans, J.C.; Kors, L.; Anders, H.J.; Florquin, S. Pattern recognition receptors and the inflammasome in kidney disease. Nat. Rev. Nephrol. 2014, 10, 398–414. [Google Scholar]
- Chen, J.; John, R.; Richardson, J.A.; Shelton, J.M.; Zhou, X.J.; Wang, Y.; Wu, Q.Q.; Hartono, J.R.; Winterberg, P.D.; Lu, C.Y. Toll-like receptor 4 regulates early endothelial activation during ischemic acute kidney injury. Kidney Int. 2011, 79, 288–299. [Google Scholar] [CrossRef]
- Wolfs, T.G.A.M.; Buurman, W.A.; van Schadewijk, A.; de Vries, B.; Daemen, M.A.R.C.; Hiemstra, P.S.; van’t Veer, C. In Vivo Expression of Toll-Like Receptor 2 and 4 by Renal Epithelial Cells: IFN-γ and TNF-α Mediated Up-Regulation during Inflammation. J. Immunol. 2002, 168, 1286–1293. [Google Scholar] [CrossRef]
- Wu, H.; Chen, G.; Wyburn, K.R.; Yin, J.; Bertolino, P.; Eris, J.M.; Alexander, S.I.; Sharland, A.F.; Chadban, S.J. TLR4 activation mediates kidney ischemia/reperfusion injury. J. Clin. Investig. 2007, 117, 2847–2859. [Google Scholar] [CrossRef]
- El-Achkar, T.M.; Huang, X.; Plotkin, Z.; Sandoval, R.M.; Rhodes, G.J.; Dagher, P.C. Sepsis induces changes in the expression and distribution of Toll-like receptor 4 in the rat kidney. Am. J. Physiol. Ren. Physiol. 2006, 290, F1034–F1043. [Google Scholar] [CrossRef]
- Zhang, B.; Ramesh, G.; Uematsu, S.; Akira, S.; Reeves, W.B. TLR4 signaling mediates inflammation and tissue injury in nephrotoxicity. J. Am. Soc. Nephrol. 2008, 19, 923–932. [Google Scholar] [CrossRef]
- Lim, S.W.; Li, C.; Ahn, K.O.; Kim, J.; Moon, I.S.; Ahn, C.; Lee, J.R.; Yang, C.W. Cyclosporine-induced renal injury induces toll-like receptor and maturation of dendritic cells. Transplantation 2005, 80, 691–699. [Google Scholar] [CrossRef]
- Pawar, R.D.; Castrezana-Lopez, L.; Allam, R.; Kulkarni, O.P.; Segerer, S.; Radomska, E.; Meyer, T.N.; Schwesinger, C.M.; Akis, N.; Gröne, H.J.; et al. Bacterial lipopeptide triggers massive albuminuria in murine lupus nephritis by activating Toll-like receptor 2 at the glomerular filtration barrier. Immunology 2009, 128, e206–e221. [Google Scholar] [CrossRef]
- Skuginna, V.; Lech, M.; Allam, R.; Ryu, M.; Clauss, S.; Susanti, H.E.; Römmele, C.; Garlanda, C.; Mantovani, A.; Anders, H.J. Toll-like receptor signaling and sigirr in renal fibrosis upon unilateral ureteral obstruction. PLoS ONE 2011, 6, e19204. [Google Scholar] [CrossRef]
- Lin, M.; Yiu, W.H.; Wu, H.J.; Chan, L.Y.Y.; Leung, J.C.K.; Au, W.S.; Chan, K.W.; Lai, K.N.; Tang, S.C.W. Toll-like receptor 4 promotes tubular inflammation in diabetic nephropathy. J. Am. Soc. Nephrol. 2012, 23, 86–102. [Google Scholar] [CrossRef]
- Guerrero-Hue, M.; García-Caballero, C.; Palomino-Antolín, A.; Rubio-Navarro, A.; Vázquez-Carballo, C.; Herencia, C.; Martín-Sanchez, D.; Farré-Alins, V.; Egea, J.; Cannata, P.; et al. Curcumin reduces renal damage associated with rhabdomyolysis by decreasing ferroptosis-mediated cell death. FASEB J. 2019, 33, 8961–8975. [Google Scholar] [CrossRef]
- Lu, Y.C.; Yeh, W.C.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef]
- Shimazu, R.; Akashi, S.; Ogata, H.; Nagai, Y.; Fukudome, K.; Miyake, K.; Kimoto, M. MD-2, a molecule that confers lipopolysaccharide responsiveness on toll- like receptor 4. J. Exp. Med. 1999, 189, 1777–1782. [Google Scholar] [CrossRef]
- Brubaker, S.W.; Bonham, K.S.; Zanoni, I.; Kagan, J.C. Innate immune pattern recognition: A cell biological perspective. Annu. Rev. Immunol. 2015, 33, 257–290. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef]
- Yamamoto, M.; Sato, S.; Hemmi, H.; Sanjo, H.; Uematsu, S.; Kaisho, T.; Hoshino, K.; Takeuchi, O.; Kobayashi, M.; Fujita, T.; et al. Essential role for TIRAP in activation of the signalling cascade shared by TLR2 and TLR4. Nature 2002, 420, 324–329. [Google Scholar] [CrossRef]
- Motshwene, P.G.; Moncrieffe, M.C.; Grossmann, J.G.; Kao, C.; Ayaluru, M.; Sandercock, A.M.; Robinson, C.V.; Latz, E.; Gay, N.J. An Oligomeric Signaling Platform formed by the toll-like receptor signal transducers MyD88 and IRAK-4. J. Biol. Chem. 2009, 284, 25404–25411. [Google Scholar] [CrossRef]
- Lin, S.C.; Lo, Y.C.; Wu, H. Helical assembly in the MyD88-IRAK4-IRAK2 complex in TLR/IL-1R signalling. Nature 2010, 465, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Suzuki, S.; Duncan, G.S.; Millar, D.G.; Wada, T.; Mirtsos, C.; Takada, H.; Wakeham, A.; Itie, A.; Li, S.; et al. Severe impairment of interleukin-1 and toll-like receptor signalling in mice lacking IRAK-4. Nature 2002, 416, 750–754. [Google Scholar] [CrossRef] [PubMed]
- Kawagoe, T.; Sato, S.; Matsushita, K.; Kato, H.; Matsui, K.; Kumagai, Y.; Saitoh, T.; Kawai, T.; Takeuchi, O.; Akira, S. Sequential control of Toll-like receptor-dependent responses by IRAK1 and IRAK2. Nat. Immunol. 2008, 9, 684–691. [Google Scholar] [CrossRef]
- Ajibade, A.A.; Wang, H.Y.; Wang, R.F. Cell type-specific function of TAK1 in innate immune signaling. Trends Immunol. 2013, 34, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Ben-Neriah, Y. Phosphorylation meets ubiquitination: The control of NF-κB activity. Annu. Rev. Immunol. 2000, 18, 621–663. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. The role of pattern-recognition receptors in innate immunity: Update on toll-like receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef]
- Marongiu, L.; Gornati, L.; Artuso, I.; Zanoni, I.; Granucci, F. Below the surface: The inner lives of TLR4 and TLR9. J. Leukoc. Biol. 2019, 106, 147–160. [Google Scholar] [CrossRef]
- Zanoni, I.; Ostuni, R.; Marek, L.R.; Barresi, S.; Barbalat, R.; Barton, G.M.; Granucci, F.; Kagan, J.C. CD14 controls the LPS-induced endocytosis of toll-like receptor 4. Cell 2011, 147, 868–880. [Google Scholar] [CrossRef]
- Fitzgerald, K.A.; McWhirter, S.M.; Faia, K.L.; Rowe, D.C.; Latz, E.; Golenbock, D.T.; Coyle, A.J.; Liao, S.M.; Maniatis, T. IKKE and TBKI are essential components of the IRF3 signalling pathway. Nat. Immunol. 2003, 4, 491–496. [Google Scholar] [CrossRef]
- Yoneyama, M.; Suhara, W.; Fukuhara, Y.; Fukuda, M.; Nishida, E.; Fujita, T. Direct triggering of the type I interferon system by virus infection: Activation of a transcription factor complex containing IRF-3 and CBP/p300. EMBO J. 1998, 17, 1087–1095. [Google Scholar] [CrossRef]
- Kawai, T.; Adachi, O.; Ogawa, T.; Takeda, K.; Akira, S. Unresponsiveness of MyD88-deficient mice to endotoxin. Immunity 1999, 11, 115–122. [Google Scholar] [CrossRef]
- Molteni, M.; Gemma, S.; Rossetti, C. The Role of Toll-Like Receptor 4 in Infectious and Noninfectious Inflammation. Mediat. Inflamm. 2016, 2016, 6978936. [Google Scholar] [CrossRef] [PubMed]
- Radi, Z.A. Immunopathogenesis of Acute Kidney Injury. Toxicol. Pathol. 2018, 46, 930–943. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Tarjuelo, R.; Cortegano, I.; Manosalva, J.; Rodríguez, M.; Ruíz, C.; Alía, M.; Prado, M.C.; Cano, E.M.; Ferrándiz, M.J.; de la Campa, A.G.; et al. The TLR4-MyD88 Signaling Axis Regulates Lung Monocyte Differentiation Pathways in Response to Streptococcus pneumoniae. Front. Immunol. 2020, 11, 2120. [Google Scholar] [CrossRef]
- Fan, J.; Malik, A.B. Toll-like receptor-4 (TLR4) signaling augments chemokine-induced neutrophil migration by modulating cell surface expression of chemokine receptors. Nat. Med. 2003, 9, 315–321. [Google Scholar] [CrossRef]
- Liu, Z.; Jiang, Y.; Li, Y.; Wang, J.; Fan, L.; Scott, M.J.; Xiao, G.; Li, S.; Billiar, T.R.; Wilson, M.A.; et al. TLR4 Signaling Augments Monocyte Chemotaxis by Regulating G Protein–Coupled Receptor Kinase 2 Translocation. J. Immunol. 2013, 191, 857–864. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Varga, J. Endogenous ligands of TLR4 promote unresolving tissue fibrosis: Implications for systemic sclerosis and its targeted therapy. Immunol. Lett. 2018, 195, 9–17. [Google Scholar] [CrossRef]
- Mantovani, A.; Biswas, S.K.; Galdiero, M.R.; Sica, A.; Locati, M. Macrophage plasticity and polarization in tissue repair and remodelling. J. Pathol. 2013, 229, 176–185. [Google Scholar]
- Geissmann, F.; Jung, S.; Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 2003, 19, 71–82. [Google Scholar] [CrossRef]
- Geissmann, F.; Manz, M.G.; Jung, S.; Sieweke, M.H.; Merad, M.; Ley, K. Development of monocytes, macrophages, and dendritic cells. Science 2010, 327, 656–661. [Google Scholar] [CrossRef]
- Zhou, H.; Andonegui, G.; Wong, C.H.Y.; Kubes, P. Role of Endothelial TLR4 for Neutrophil Recruitment into Central Nervous System Microvessels in Systemic Inflammation. J. Immunol. 2009, 183, 5244–5250. [Google Scholar] [CrossRef] [PubMed]
- Allam, R.; Scherbaum, C.R.; Darisipudi, M.N.; Mulay, S.R.; Hägele, H.; Lichtnekert, J.; Hagemann, J.H.; Rupanagudi, K.V.; Ryu, M.; Schwarzenberger, C.; et al. Histones from dying renal cells aggravate kidney injury via TLR2 and TLR4. J. Am. Soc. Nephrol. 2012, 23, 1375–1388. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Chadban, S.J.; Zhao, C.Y.; Chen, X.; Kwan, T.; Panchapakesan, U.; Pollock, C.A.; Wu, H. TLR4 activation promotes podocyte injury and interstitial fibrosis in diabetic nephropathy. PLoS ONE 2014, 9, e97985. [Google Scholar] [CrossRef] [PubMed]
- Vabulas, R.M.; Ahmad-Nejad, P.; Ghose, S.; Kirschning, C.J.; Issels, R.D.; Wagner, H. HSP70 as endogenous stimulus of the toll/interleukin-1 receptor signal pathway. J. Biol. Chem. 2002, 277, 15107–15112. [Google Scholar] [CrossRef]
- Lee, K.M.; Yeo, M.; Choue, J.S.; Jin, J.H.; Park, S.J.; Cheong, J.Y.; Lee, K.J.; Kim, J.H.; Hahm, K.B. Protective mechanism of epigallocatechin-3-gallate against Helicobocter pylori-induced gastric epithelial cytotoxicity via the blockage of TLR-4 signaling. Helicobacter 2004, 9, 632–642. [Google Scholar] [CrossRef]
- Deva, R.; Shankaranarayanan, P.; Ciccoli, R.; Nigam, S. Candida albicans Induces Selectively Transcriptional Activation of Cyclooxygenase-2 in HeLa Cells: Pivotal Roles of Toll-Like Receptors, p38 Mitogen-Activated Protein Kinase, and NF-κB. J. Immunol. 2003, 171, 3047–3055. [Google Scholar] [CrossRef]
- Chen, Y.; Jin, S.; Teng, X.; Hu, Z.; Zhang, Z.; Qiu, X.; Tian, D.; Wu, Y. Hydrogen sulfide attenuates LPS-induced acute kidney injury by inhibiting inflammation and oxidative stress. Oxid. Med. Cell. Longev. 2018, 2018, 6717212. [Google Scholar] [CrossRef]
- Kim, B.S.; Lim, S.W.; Li, C.; Kim, J.S.; Sun, B.K.; Ahn, K.O.; Han, S.W.; Kim, J.; Yang, C.W. Ischemia-reperfusion injury activates innate immunity in rat kidneys. Transplantation 2005, 79, 1370–1377. [Google Scholar] [CrossRef]
- Pulskens, W.P.; Teske, G.J.; Butter, L.M.; Roelofs, J.J.; van der Poll, T.; Florquin, S.; Leemans, J.C. Toll-like receptor-4 coordinates the innate immune response of the kidney to renal ischemia/reperfusion injury. PLoS ONE 2008, 3, e3596. [Google Scholar] [CrossRef]
- De Greef, K.E.; Ysebaert, D.K.; Persy, V.; Vercauteren, S.R.; De Broe, M.E. ICAM-1 expression and leukocyte accumulation in inner stripe of outer medulla in early phase of ischemic compared to HgCl2-induced ARF. Kidney Int. 2003, 63, 1697–1707. [Google Scholar] [CrossRef]
- Castoldi, A.; Braga, T.T.; Correa-Costa, M.; Aguiar, C.F.; Bassi, Ê.J.; Correa-Silva, R.; Elias, R.M.; Salvador, F.; Moraes-Vieira, P.M.; Cenedeze, M.A.; et al. TLR2, TLR4 and the Myd88 signaling pathway are crucial for neutrophil migration in acute kidney injury induced by sepsis. PLoS ONE 2012, 7, e37584. [Google Scholar] [CrossRef] [PubMed]
- Reiser, J.; Von Gersdorff, G.; Loos, M.; Oh, J.; Asanuma, K.; Giardino, L.; Rastaldi, M.P.; Calvaresi, N.; Watanabe, H.; Schwarz, K.; et al. Induction of B7-1 in podocytes is associated with nephrotic syndrome. J. Clin. Investig. 2004, 113, 1390–1397. [Google Scholar] [CrossRef] [PubMed]
- Gomez, H.; Ince, C.; De Backer, D.; Pickkers, P.; Payen, D.; Hotchkiss, J.; Kellum, J.A. A unified theory of sepsis-induced acute kidney injury: Inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to injury. Shock 2014, 41, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Fenhammar, J.; Rundgren, M.; Hultenby, K.; Forestier, J.; Taavo, M.; Kenne, E.; Weitzberg, E.; Eriksson, S.; Ozenci, V.; Wernerson, A.; et al. Renal effects of treatment with a TLR4 inhibitor in conscious septic sheep. Crit. Care 2014, 18. [Google Scholar] [CrossRef]
- Guo, R.; Wang, Y.; Minto, A.W.; Quigg, R.J.; Cunningham, P.N. Acute renal failure in endotoxemia is dependent on caspase activation. J. Am. Soc. Nephrol. 2004, 15, 3093–3102. [Google Scholar] [CrossRef]
- Cunningham, P.N.; Wang, Y.; Guo, R.; He, G.; Quigg, R.J. Role of Toll-Like Receptor 4 in Endotoxin-Induced Acute Renal Failure. J. Immunol. 2004, 172, 2629–2635. [Google Scholar] [CrossRef]
- Messaris, E.; Memos, N.; Chatzigianni, E.; Kataki, A.; Nikolopoulou, M.; Manouras, A.; Albanopoulos, K.; Konstadoulakis, M.M.; Bramis, J. Apoptotic death of renal tubular cells in experimental sepsis. Surg. Infect. (Larchmt.) 2008, 9, 377–388. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, J.; Yang, J.; Wang, S.; Yang, L. Role of TLR4/MyD88/NF-κB signaling in the contrast-induced injury of renal tubular epithelial cells. Exp. Ther. Med. 2020, 20, 115. [Google Scholar] [CrossRef]
- Tsan, M.-F.; Gao, B. Endogenous ligands of Toll-like receptors. J. Leukoc. Biol. 2004, 76, 514–519. [Google Scholar] [CrossRef]
- Krüger, B.; Krick, S.; Dhillon, N.; Lerner, S.M.; Ames, S.; Bromberg, J.S.; Lin, M.; Walsh, L.; Vella, J.; Fischereder, M.; et al. Donor toll-like receptor 4 contributes to ischemia and reperfusion injury following human kidney transplantation. Proc. Natl. Acad. Sci. USA 2009, 106, 3390–3395. [Google Scholar] [CrossRef]
- Watts, B.A.; George, T.; Good, D.W. Lumen LPS inhibits HCO3- absorption in the medullary thick ascending limb through TLR4-PI3K-Akt-mTOR-dependent inhibition of basolateral Na+/H+ exchange. Am. J. Physiol. Ren. Physiol. 2013, 305, F451–F462. [Google Scholar] [CrossRef] [PubMed]
- Iwami, K.; Matsuguchi, T.; Masuda, A.; Kikuchi, T.; Musikacharoen, T.; Yoshikai, Y. Cutting Edge: Naturally Occurring Soluble Form of Mouse Toll-Like Receptor 4 Inhibits Lipopolysaccharide Signaling. J. Immunol. 2000, 165, 6682–6686. [Google Scholar] [CrossRef] [PubMed]
- Hyakushima, N.; Mitsuzawa, H.; Nishitani, C.; Sano, H.; Kuronuma, K.; Konishi, M.; Himi, T.; Miyake, K.; Kuroki, Y. Interaction of Soluble Form of Recombinant Extracellular TLR4 Domain with MD-2 Enables Lipopolysaccharide Binding and Attenuates TLR4-Mediated Signaling. J. Immunol. 2004, 173, 6949–6954. [Google Scholar] [CrossRef] [PubMed]
- Basith, S.; Manavalan, B.; Govindaraj, R.G.; Choi, S. In Silico approach to inhibition of signaling pathways of toll-like receptors 2 and 4 by ST2L. PLoS ONE 2011, 6, e23989. [Google Scholar] [CrossRef] [PubMed]
- Wald, D.; Qin, J.; Zhao, Z.; Qian, Y.; Naramura, M.; Tian, L.; Towne, J.; Sims, J.E.; Stark, G.R.; Li, X. SIGIRR, a negative regulator of Toll-like receptor—Interleukin 1 receptor signaling. Nat. Immunol. 2003, 4, 920–927. [Google Scholar] [CrossRef]
- Divanovic, S.; Trompette, A.; Atabani, S.F.; Madan, R.; Golenbock, D.T.; Visintin, A.; Finberg, R.W.; Tarakhovsky, A.; Vogel, S.N.; Belkaid, Y.; et al. Negative regulation of Toll-like receptor 4 signaling by the Toll-like receptor homolog RP105. Nat. Immunol. 2005, 6, 571–578. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, T.; Han, C.; He, D.; Liu, H.; An, H.; Cai, Z.; Cao, X. Lysosome-associated small Rab GTPase Rab7b negatively regulates TLR4 signaling in macrophages by promoting lysosomal degradation of TLR4. Blood 2007, 110, 962–971. [Google Scholar] [CrossRef]
- Kobayashi, K.; Hernandez, L.D.; Galán, J.E.; Janeway, C.A.; Medzhitov, R.; Flavell, R.A. IRAK-M is a negative regulator of Toll-like receptor signaling. Cell 2002, 110, 191–202. [Google Scholar] [CrossRef]
- Zhang, G.; Ghosh, S. Negative regulation of toll-like receptor-mediated signaling by Tollip. J. Biol. Chem. 2002, 277, 7059–7065. [Google Scholar] [CrossRef]
- Mansell, A.; Smith, R.; Doyle, S.L.; Gray, P.; Fenner, J.E.; Crack, P.J.; Nicholson, S.E.; Hilton, D.J.; O’Neill, L.A.J.; Hertzog, P.J. Suppressor of cytokine signaling 1 negatively regulates Toll-like receptor signaling by mediating Mal degradation. Nat. Immunol. 2006, 7, 148–155. [Google Scholar] [CrossRef]
- Shembade, N.; Ma, A.; Harhaj, E.W. Inhibition of nf-kb signaling by a20 through disruption of ubiquitin enzyme complexes. Science 2010, 327, 1135–1139. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.H.; Ulevitch, R.J. Triad3A, an E3 ubiquitin-protein ligase regulating Toll-like receptors. Nat. Immunol. 2004, 5, 495–502. [Google Scholar] [CrossRef] [PubMed]
- An, H.; Hou, J.; Zhou, J.; Zhao, W.; Xu, H.; Zheng, Y.; Yu, Y.; Liu, S.; Cao, X. Phosphatase SHP-1 promotes TLR- and RIG-I-activated production of type I interferon by inhibiting the kinase IRAK1. Nat. Immunol. 2008, 9, 542–550. [Google Scholar] [CrossRef] [PubMed]
- An, H.; Zhao, W.; Hou, J.; Zhang, Y.; Xie, Y.; Zheng, Y.; Xu, H.; Qian, C.; Zhou, J.; Yu, Y.; et al. SHP-2 Phosphatase Negatively Regulates the TRIF Adaptor Protein-Dependent Type I Interferon and Proinflammatory Cytokine Production. Immunity 2006, 25, 919–928. [Google Scholar] [CrossRef] [PubMed]
- McCartney-Francis, N.; Jin, W.; Wahl, S.M. Aberrant Toll Receptor Expression and Endotoxin Hypersensitivity in Mice Lacking a Functional TGF-β1 Signaling Pathway. J. Immunol. 2004, 172, 3814–3821. [Google Scholar] [CrossRef]
- Naiki, Y.; Michelsen, K.S.; Zhang, W.; Chen, S.; Doherty, T.M.; Arditi, M. Transforming growth factor-β differentially inhibits MyD88-dependent, but not TRAM- and TRIF-dependent, lipopolysaccharide-induced TLR4 signaling. J. Biol. Chem. 2005, 280, 5491–5495. [Google Scholar] [CrossRef]
- Curtale, G.; Mirolo, M.; Renzi, T.A.; Rossato, M.; Bazzoni, F.; Locati, M. Negative regulation of Toll-like receptor 4 signaling by IL-10-dependent microRNA-146b. Proc. Natl. Acad. Sci. USA 2013, 110, 11499–11504. [Google Scholar] [CrossRef]
- Arenas-Padilla, M.; Mata-Haro, V. Regulation of TLR signaling pathways by microRNAs: Implications in inflammatory diseases. Cent. Eur. J. Immunol. 2018, 43, 482–489. [Google Scholar] [CrossRef]
- Shi, M.; Deng, W.; Bi, E.; Mao, K.; Ji, Y.; Lin, G.; Wu, X.; Tao, Z.; Li, Z.; Cai, X.; et al. TRIM30α negatively regulates TLR-mediated NF-κB activation by targeting TAB2 and TAB3 for degradation. Nat. Immunol. 2008, 9, 369–377. [Google Scholar] [CrossRef]
- Kuwata, H.; Matsumoto, M.; Atarashi, K.; Morishita, H.; Hirotani, T.; Koga, R.; Takeda, K. IκBNS inhibits induction of a subset of toll-like receptor-dependent genes and limits inflammation. Immunity 2006, 24, 41–51. [Google Scholar] [CrossRef]
- Carmody, R.J.; Ruan, Q.; Palmer, S.; Hilliard, B.; Chen, Y.H. Negative regulation of toll-like receptor signaling by NF-κB p50 ubiquitination blockade. Science 2007, 317, 675–678. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Y.; Li, L.C.; Yang, J.L. Emerging Roles of IL-33/ST2 axis in renal diseases. Int. J. Mol. Sci. 2017, 18, 783. [Google Scholar] [CrossRef] [PubMed]
- Lech, M.; Avila-Ferrufino, A.; Allam, R.; Segerer, S.; Khandoga, A.; Krombach, F.; Garlanda, C.; Mantovani, A.; Anders, H.-J. Resident Dendritic Cells Prevent Postischemic Acute Renal Failure by Help of Single Ig IL-1 Receptor-Related Protein. J. Immunol. 2009, 183, 4109–4118. [Google Scholar] [CrossRef] [PubMed]
- Leifer, C.A.; Medvedev, A.E. Molecular mechanisms of regulation of Toll-like receptor signaling. J. Leukoc. Biol. 2016, 100, 927–941. [Google Scholar] [CrossRef]
- Ling, H.; Chen, H.; Wei, M.; Meng, X.; Yu, Y.; Xie, K. The Effect of Autophagy on Inflammation Cytokines in Renal Ischemia/Reperfusion Injury. Inflammation 2016, 39, 347–356. [Google Scholar] [CrossRef]
- Watts, B.A.; Tamayo, E.; Sherwood, E.R.; Good, D.W. Monophosphoryl lipid A induces protection against LPS in medullary thick ascending limb through induction of Tollip and negative regulation of IRAK-1. Am. J. Physiol. Ren. Physiol. 2019, 317, F705–F719. [Google Scholar] [CrossRef]
- Wu, H.; Steenstra, R.; De Boer, E.C.S.; Zhao, C.Y.; Ma, J.; Van Der Stelt, J.M.; Chadban, S.J. Preconditioning with recombinant high-mobility group box 1 protein protects the kidney against ischemia-reperfusion injury in mice. Kidney Int. 2014, 85, 824–832. [Google Scholar] [CrossRef]
- Yang, C.; Zhang, Y.; Wang, J.; Li, L.; Wang, L.; Hu, M.; Xu, M.; Long, Y.; Rong, R.; Zhu, T. A novel cyclic helix B peptide inhibits dendritic cell maturation during amelioration of acute kidney graft rejection through Jak-2/STAT3/SOCS1. Cell Death Dis. 2015, 6, e1993. [Google Scholar] [CrossRef][Green Version]
- Tsogbadrakh, B.; Ryu, H.; Ju, K.D.; Lee, J.; Yun, S.; Yu, K.S.; Kim, H.J.; Ahn, C.; Oh, K.H. AICAR, an AMPK activator, protects against cisplatin-induced acute kidney injury through the JAK/STAT/SOCS pathway. Biochem. Biophys. Res. Commun. 2019, 509, 680–686. [Google Scholar] [CrossRef]
- Opazo-Ríos, L.; Sanchez Matus, Y.; Rodrigues-Díez, R.R.; Carpio, D.; Droguett, A.; Egido, J.; Gomez-Guerrero, C.; Mezzano, S. Anti-inflammatory, antioxidant and renoprotective effects of SOCS1 mimetic peptide in the BTBR ob/ob mouse model of type 2 diabetes. BMJ Open Diabetes Res. Care 2020, 8, e001242. [Google Scholar] [CrossRef]
- Recio, C.; Lazaro, I.; Oguiza, A.; Lopez-Sanz, L.; Bernal, S.; Blanco, J.; Egido, J.; Gomez-Guerrero, C. Suppressor of cytokine signaling-1 peptidomimetic limits progression of diabetic nephropathy. J. Am. Soc. Nephrol. 2017, 28, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Sanz, L.; Bernal, S.; Recio, C.; Lazaro, I.; Oguiza, A.; Melgar, A.; Jimenez-Castilla, L.; Egido, J.; Gomez-Guerrero, C. SOCS1-targeted therapy ameliorates renal and vascular oxidative stress in diabetes via STAT1 and PI3K inhibition. Lab. Investig. 2018, 98, 1276–1290. [Google Scholar] [CrossRef] [PubMed]
- Kunter, U.; Daniel, S.; Arvelo, M.B.; Choi, J.; Shukri, T.; Patel, V.I.; Longo, C.R.; Scali, S.T.; Shrikhande, G.; Rocha, E.; et al. Combined expression of A1 and A20 achieves optimal protection of renal proximal tubular epithelial cells. Kidney Int. 2005, 68, 1520–1532. [Google Scholar] [CrossRef] [PubMed]
- Lutz, J.; Luong, L.A.; Strobl, M.; Deng, M.; Huang, H.; Anton, M.; Zakkar, M.; Enesa, K.; Chaudhury, H.; Haskard, D.O.; et al. The A20 gene protects kidneys from ischaemia/reperfusion injury by suppressing pro-inflammatory activation. J. Mol. Med. 2008, 86, 1329–1339. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Zhang, X.; Ye, Q.; Yang, Y.; Chen, X. The transfection of A20 gene prevents kidney from ischemia reperfusion injury in rats. Mol. Med. Rep. 2017, 16, 1486–1492. [Google Scholar] [CrossRef]
- Chen, N.K.F.; Chong, T.W.; Loh, H.L.; Lim, K.H.; Gan, V.H.L.; Wang, M.; Kon, O.L. Negative regulatory responses to metabolically triggered inflammation impair renal epithelial immunity in diabetes mellitus. J. Mol. Med. 2013, 91, 587–598. [Google Scholar] [CrossRef][Green Version]
- Tian, H.; Tan, R.; Ye, B.; Yan, S.; Sui, M.; Zhao, W.; Zhang, L.; Zhu, Y.; Zeng, L. SHP-1 inhibits renal ischemia reperfusion injury via dephosphorylating ASK1 and suppressing apoptosis. Biochem. Biophys. Res. Commun. 2019, 513, 360–367. [Google Scholar] [CrossRef]
- Jiang, J.; Hu, B.; Chung, C.S.; Chen, Y.; Zhang, Y.; Tindal, E.W.; Li, J.; Ayala, A. SHP2 inhibitor PHPS1 ameliorates acute kidney injury by Erk1/2-STAT3 signaling in a combined murine hemorrhage followed by septic challenge model. Mol. Med. 2020, 26. [Google Scholar] [CrossRef]
- Teng, J.F.; Wang, K.; Li, Y.; Qu, F.J.; Yuan, Q.; Cui, X.G.; Wang, Q.X.; Xu, D.F. Conditional knockout of Src homology 2 domain-containing protein tyrosine phosphatase-2 in myeloid cells attenuates renal fibrosis after unilateral ureter obstruction. Chin. Med. J. 2015, 128, 1196–1201. [Google Scholar] [CrossRef]
- Teng, J.F.; Wang, K.; Jia, Z.M.; Guo, Y.J.; Guan, Y.W.; Li, Z.H.; Ai, X. Lentivirus-mediated silencing of src homology 2 domain-containing protein tyrosine phosphatase 2 inhibits release of inflammatory cytokines and apoptosis in renal tubular epithelial cells via inhibition of the TLR4/NF-KB pathway in renal ischemia-reperfusion injury. Kidney Blood Press. Res. 2018, 43, 1084–1103. [Google Scholar] [CrossRef]
- Zhang, W.; Li, X.; Tang, Y.; Chen, C.; Jing, R.; Liu, T.; Liu, T. MiR-155-5p Implicates in the Pathogenesis of Renal Fibrosis via Targeting SOCS1 and SOCS6. Oxid. Med. Cell. Longev. 2020, 2020, 6263921. [Google Scholar] [CrossRef] [PubMed]
- Poveda, J.; Sanz, A.B.; Carrasco, S.; Ruiz-Ortega, M.; Cannata-Ortiz, P.; Sanchez-Niño, M.D.; Ortiz, A. Bcl3: A regulator of NF-κB inducible by TWEAK in acute kidney injury with anti-inflammatory and antiapoptotic properties in tubular cells. Exp. Mol. Med. 2017, 49, e352. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.E.; Arslan, F.; Keogh, B.; McGuirk, P. TLR2 and TLR4 in ischemia reperfusion injury. Mediat. Inflamm. 2010, 2010, 704202. [Google Scholar] [CrossRef]
- Ikeda, T.; Yanaga, K.; Kishikawa, K.; Kakizoe, S.; Shimada, M.; Sugimachi, K. Ischemic injury in liver transplantation: Difference in injury sites between warm and cold ischemia in rats. Hepatology 1992, 16, 454–461. [Google Scholar] [CrossRef]
- Baldwin, W.M.; Larsen, C.P.; Fairchild, R.L. Innate immune responses to transplants: A significant variable with cadaver donors. Immunity 2001, 14, 369–376. [Google Scholar] [CrossRef]
- Weiser, M.R.; Williams, J.P.; Moore, F.D.; Kobzik, L.; Ma, M.; Hechtman, H.B.; Carroll, M.C. Reperfusion injury of ischemic skeletal muscle is mediated by natural antibody and complement. J. Exp. Med. 1996, 183, 2343–2348. [Google Scholar] [CrossRef]
- Weisman, H.F.; Bartow, T.; Leppo, M.K.; Marsh, H.C.; Carson, G.R.; Concino, M.F.; Boyle, M.P.; Roux, K.H.; Weisfeldt, M.L.; Fearon, D.T. Soluble human complement receptor type 1: In vivo inhibitor of complement suppressing post-ischemic myocardial inflammation and necrosis. Science 1990, 249, 146–151. [Google Scholar] [CrossRef]
- Paulus, P.; Rupprecht, K.; Baer, P.; Obermüller, N.; Penzkofer, D.; Reissig, C.; Scheller, B.; Holfeld, J.; Zacharowski, K.; Dimmeler, S.; et al. The early activation of toll-like receptor (TLR)-3 initiates kidney injury after ischemia and reperfusion. PLoS ONE 2014, 9, e94366. [Google Scholar] [CrossRef]
- Wu, H.; Ma, J.; Wang, P.; Corpuz, T.M.; Panchapakesan, U.; Wyburn, K.R.; Chadban, S.J. HMGB1 contributes to kidney ischemia reperfusion injury. J. Am. Soc. Nephrol. 2010, 21, 1878–1890. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, D.; Jin, Z. MiR-27a suppresses TLR4-induced renal ischemia-reperfusion injury. Mol. Med. Rep. 2019, 20, 967–976. [Google Scholar] [CrossRef]
- Leemans, J.C.; Stokman, G.; Claessen, N.; Rouschop, K.M.; Teske, G.J.D.; Kirschning, C.J.; Akira, S.; Van Der Poll, T.; Weening, J.J.; Florquin, S. Renal-associated TLR2 mediates ischemia/reperfusion injury in the kidney. J. Clin. Investig. 2005, 115, 2894–2903. [Google Scholar] [CrossRef] [PubMed]
- Shigeoka, A.A.; Holscher, T.D.; King, A.J.; Hall, F.W.; Kiosses, W.B.; Tobias, P.S.; Mackman, N.; McKay, D.B. TLR2 Is Constitutively Expressed within the Kidney and Participates in Ischemic Renal Injury through Both MyD88-Dependent and -Independent Pathways. J. Immunol. 2007, 178, 6252–6258. [Google Scholar] [CrossRef] [PubMed]
- Rusai, K.; Sollinger, D.; Baumann, M.; Wagner, B.; Strobl, M.; Schmaderer, C.; Roos, M.; Kirschning, C.; Heemann, U.; Lutz, J. Toll-like receptors 2 and 4 in renal ischemia/reperfusion injury. Pediatr. Nephrol. 2010, 25, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Dagher, P.C.; Hato, T.; Mang, H.E.; Plotkin, Z.; Richardson, Q.V.; Massad, M.; Mai, E.; Kuehl, S.E.; Graham, P.; Kumar, R.; et al. Inhibition of toll-like receptor 4 signaling mitigates microvascular loss but not fibrosis in a model of ischemic acute kidney injury. Int. J. Mol. Sci. 2016, 17, 647. [Google Scholar] [CrossRef]
- Amura, C.R.; Renner, B.; Lyubchenko, T.; Faubel, S.; Simonian, P.L.; Thurman, J.M. Complement activation and toll-like receptor-2 signaling contribute to cytokine production after renal ischemia/reperfusion. Mol. Immunol. 2012, 52, 249–257. [Google Scholar] [CrossRef][Green Version]
- Ben Mkaddem, S.; Pedruzzi, E.; Werts, C.; Coant, N.; Bens, M.; Cluzeaud, F.; Goujon, J.M.; Ogier-Denis, E.; Vandewalle, A. Heat shock protein gp96 and NAD(P)H oxidase 4 play key roles in Toll-like receptor 4-activated apoptosis during renal ischemia/reperfusion injury. Cell Death Differ. 2010, 17, 1474–1485. [Google Scholar] [CrossRef]
- Wang, S.; Schmaderer, C.; Kiss, E.; Schmidt, C.; Bonrouhi, M.; Porubsky, S.; Gretz, N.; Schaefer, L.; Kirschning, C.J.; Popovic, Z.V.; et al. Recipient toll-like receptors contribute to chronic graft dysfunction by both MyD88- and TRIF-dependent signaling. DMM Dis. Model. Mech. 2010, 3, 92–103. [Google Scholar] [CrossRef]
- Han, S.J.; Li, H.; Kim, M.; Shlomchik, M.J.; Lee, H.T. Kidney Proximal Tubular TLR9 Exacerbates Ischemic Acute Kidney Injury. J. Immunol. 2018, 201, 1073–1085. [Google Scholar] [CrossRef]
- Han, S.J.; Williams, R.M.; D’Agati, V.; Jaimes, E.A.; Heller, D.A.; Lee, H.T. Selective nanoparticle-mediated targeting of renal tubular Toll-like receptor 9 attenuates ischemic acute kidney injury. Kidney Int. 2020, 98, 76–87. [Google Scholar] [CrossRef]
- Fukuzawa, N.; Petro, M.; Baldwin, W.M.; Gudkov, A.V.; Fairchild, R.L. A TLR5 Agonist Inhibits Acute Renal Ischemic Failure. J. Immunol. 2011, 187, 3831–3839. [Google Scholar] [CrossRef]
- Qiu, Y.; Wu, Y.; Zhao, H.; Sun, H.; Gao, S. Maresin 1 mitigates renal ischemia/reperfusion injury in mice via inhibition of the TLR4/MAPK/ NF-κB pathways and activation of the Nrf2 pathway. Drug Des. Devel. Ther. 2019, 13, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Wei, M.; Wang, D.; Qu, X.; Zhang, K.; Zhang, N.; Li, X. The protective effect of hesperidin against renal ischemia-reperfusion injury involves the TLR-4/NF-kB/iNOS pathway in rats. Physiol. Int. 2020, 107, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.M.; Park, B.S.; Kim, J.I.; Kim, S.E.; Lee, J.; Oh, S.C.; Enkhbayar, P.; Matsushima, N.; Lee, H.; Yoo, O.J.; et al. Crystal Structure of the TLR4-MD-2 Complex with Bound Endotoxin Antagonist Eritoran. Cell 2007, 130, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Gu, M.; Xu, D.; Lv, Q.; Zhang, W.; Wu, Y. Protective Effects of Toll-like Receptor 4 Inhibitor Eritoran on Renal Ischemia-Reperfusion Injury. Transpl. Proc. 2010, 42, 1539–1544. [Google Scholar] [CrossRef]
- Youn, H.S.; Kim, Y.S.; Park, Z.Y.; Kim, S.Y.; Choi, N.Y.; Joung, S.M.; Seo, J.A.; Lim, K.-M.; Kwak, M.-K.; Hwang, D.H.; et al. Sulforaphane Suppresses Oligomerization of TLR4 in a Thiol-Dependent Manner. J. Immunol. 2010, 184, 411–419. [Google Scholar] [CrossRef]
- Yoon, H.Y.; Kang, N.I.; Lee, H.K.; Jang, K.Y.; Park, J.W.; Park, B.H. Sulforaphane protects kidneys against ischemia-reperfusion injury through induction of the Nrf2-dependent phase 2 enzyme. Biochem. Pharmacol. 2008, 75, 2214–2223. [Google Scholar] [CrossRef]
- Bosch, X.; Poch, E.; Grau, J.M. Rhabdomyolysis and Acute Kidney Injury. N. Engl. J. Med. 2009, 361, 62–72. [Google Scholar] [CrossRef]
- Janciauskiene, S.; Vijayan, V.; Immenschuh, S. TLR4 Signaling by Heme and the Role of Heme-Binding Blood Proteins. Front. Immunol. 2020, 11, 1964. [Google Scholar] [CrossRef]
- Zager, R.A.; Johnson, A.C.M.; Lund, S.; Randolph-Habecker, J. Toll-like receptor (TLR4) shedding and depletion: Acute proximal tubular cell responses to hypoxic and toxic injury. Am. J. Physiol. Ren. Physiol. 2007, 292, F304–F312. [Google Scholar] [CrossRef]
- Huang, R.S.; Zhou, J.J.; Feng, Y.Y.; Shi, M.; Guo, F.; Gou, S.J.; Salerno, S.; Ma, L.; Fu, P. Pharmacological inhibition of macrophage toll-like receptor 4/nuclear factor-kappa B alleviates rhabdomyolysis-induced acute kidney injury. Chin. Med. J. 2017, 130, 2163–2169. [Google Scholar] [CrossRef]
- Nath, K.A.; Belcher, J.D.; Nath, M.C.; Grande, J.P.; Croatt, A.J.; Ackerman, A.W.; Katusic, Z.S.; Vercellotti, G.M. Role of TLR4 signaling in the nephrotoxicity of Heme and heme proteins. Am. J. Physiol. Ren. Physiol. 2018, 314, F906–F914. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, N.; Tsuchimori, N.; Matsumoto, T.; Ii, M. TAK-242 (resatorvid), a small-molecule inhibitor of Toll-like receptor (TLR) 4 signaling, binds selectively to TLR4 and interferes with interactions between TLR4 and its adaptor molecules. Mol. Pharmacol. 2011, 79, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, Z.; Hou, S.; Liu, Z.; Lv, Q. TAK-242 Attenuates Crush Injury Induced Acute Kidney Injury through Inhibiting TLR4/NF-B Signaling Pathways in Rats. Prehosp. Disaster Med. 2020, 35, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Tan, Y.J.; Wang, M.Z.; Sun, Y.; Li, G.Y.; Wang, Q.L.; Yao, J.C.; Yue, J.; Liu, Z.; Zhang, G.M.; et al. Loganetin protects against rhabdomyolysis-induced acute kidney injury by modulating the toll-like receptor 4 signalling pathway. Br. J. Pharmacol. 2019, 176, 1106–1121. [Google Scholar] [CrossRef]
- Youn, H.S.; Saitoh, S.I.; Miyake, K.; Hwang, D.H. Inhibition of homodimerization of Toll-like receptor 4 by curcumin. Biochem. Pharmacol. 2006, 72, 62–69. [Google Scholar] [CrossRef]
- Arany, I.; Safirstein, R.L. Cisplatin nephrotoxicity. Semin. Nephrol. 2003, 23, 460–464. [Google Scholar] [CrossRef]
- Cenedeze, M.A.; Gonçalves, G.M.; Feitoza, C.Q.; Wang, P.M.H.; Damião, M.J.; Bertocchi, A.P.F.; Pacheco-Silva, A.; Câmara, N.O.S. The Role of Toll-Like Receptor 4 in Cisplatin-Induced Renal Injury. Transpl. Proc. 2007, 39, 409–411. [Google Scholar] [CrossRef]
- Andrade-Silva, M.; Cenedeze, M.A.; Perandini, L.A.; Felizardo, R.J.F.; Watanabe, I.K.M.; Agudelo, J.S.H.; Castoldi, A.; Gonçalves, G.M.; Origassa, C.S.T.; Semedo, P.; et al. TLR2 and TLR4 play opposite role in autophagy associated with cisplatin-induced acute kidney injury. Clin. Sci. 2018, 132, 1725–1739. [Google Scholar] [CrossRef]
- Guerrero-Beltrán, C.E.; Calderón-Oliver, M.; Tapia, E.; Medina-Campos, O.N.; Sánchez-González, D.J.; Martínez-Martínez, C.M.; Ortiz-Vega, K.M.; Franco, M.; Pedraza-Chaverri, J. Sulforaphane protects against cisplatin-induced nephrotoxicity. Toxicol. Lett. 2010, 192, 278–285. [Google Scholar] [CrossRef]
- Sharawy, M.H.; Serrya, M.S. Pirfenidone attenuates gentamicin-induced acute kidney injury by inhibiting inflammasome-dependent NLRP3 pathway in rats. Life Sci. 2020, 260, 118454. [Google Scholar] [CrossRef]
- Hassanein, E.H.M.; Ali, F.E.M.; Kozman, M.R.; Abd El-Ghafar, O.A.M. Umbelliferone attenuates gentamicin-induced renal toxicity by suppression of TLR-4/NF-κB-p65/NLRP-3 and JAK1/STAT-3 signaling pathways. Environ. Sci. Pollut. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- McCrae, J.C.; Morrison, E.E.; MacIntyre, I.M.; Dear, J.W.; Webb, D.J. Long-term adverse effects of paracetamol—a review. Br. J. Clin. Pharmacol. 2018, 84, 2218–2230. [Google Scholar] [PubMed]
- Salama, M.; Elgamal, M.; Abdelaziz, A.; Ellithy, M.; Magdy, D.; Ali, L.; Fekry, E.; Mohsen, Z.; Mostafa, M.; Elgamal, H.; et al. Toll-like receptor 4 blocker as potential therapy for acetaminophen-induced organ failure in mice. Exp. Ther. Med. 2015, 10, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.; Dhar, D.; El Zahraa Mohammed, F.; Habtesion, A.; Davies, N.A.; Jover-Cobos, M.; MacNaughtan, J.; Sharma, V.; Olde Damink, S.W.M.; Mookerjee, R.P.; et al. Prevention of acute kidney injury in a rodent model of cirrhosis following selective gut decontamination is associated with reduced renal TLR4 expression. J. Hepatol. 2012, 56, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.; Mohamed, F.E.; Jover-Cobos, M.; Macnaughtan, J.; Davies, N.; Moreau, R.; Paradis, V.; Moore, K.; Mookerjee, R.; Jalan, R. Increased renal expression and urinary excretion of TLR4 in acute kidney injury associated with cirrhosis. Liver Int. 2013, 33, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, I.; Matetzky, S. Nephropathy induced by contrast media: Pathogenesis, risk factors and preventive strategies. CMAJ 2005, 172, 1461–1471. [Google Scholar] [CrossRef]
- Tan, X.; Zheng, X.; Huang, Z.; Lin, J.; Xie, C.; Lin, Y. Involvement of S100A8/A9-TLR4-NLRP3 Inflammasome Pathway in Contrast-Induced Acute Kidney Injury. Cell. Physiol. Biochem. 2017, 43, 209–222. [Google Scholar] [CrossRef]
- Hoste, E.A.J.; Kellum, J.A.; Selby, N.M.; Zarbock, A.; Palevsky, P.M.; Bagshaw, S.M.; Goldstein, S.L.; Cerdá, J.; Chawla, L.S. Global epidemiology and outcomes of acute kidney injury. Nat. Rev. Nephrol. 2018, 14, 607–625. [Google Scholar]
- Qureshi, S.T.; Larivière, L.; Leveque, G.; Clermont, S.; Moore, K.J.; Gros, P.; Malo, D. Endotoxin-tolerant mice have mutations in toll-like receptor 4 (Tlr4). J. Exp. Med. 1999, 189, 615–625. [Google Scholar] [CrossRef]
- El-Achkar, T.M.; Hosein, M.; Dagher, P.C. Pathways of renal injury in systemic gram-negative sepsis. Eur. J. Clin. Investig. 2008, 38, 39–44. [Google Scholar] [CrossRef]
- Ilçe, F.; Gök, G.; Pandir, D. Acute effects of lipopolysaccharide (LPS) in kidney of rats and preventive role of vitamin E and sodium selenite. Hum. Exp. Toxicol. 2019, 38, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Roger, T.; Froidevaux, C.; Le Roy, D.; Reymond, M.K.; Chanson, A.L.; Mauri, D.; Burns, K.; Riederer, B.M.; Akira, S.; Calandra, T. Protection from lethal Gram-negative bacterial sepsis by targeting Toll-like receptor 4. Proc. Natl. Acad. Sci. USA 2009, 106, 2348–2352. [Google Scholar] [CrossRef] [PubMed]
- Daubeuf, B.; Mathison, J.; Spiller, S.; Hugues, S.; Herren, S.; Ferlin, W.; Kosco-Vilbois, M.; Wagner, H.; Kirschning, C.J.; Ulevitch, R.; et al. TLR4/MD-2 Monoclonal Antibody Therapy Affords Protection in Experimental Models of Septic Shock. J. Immunol. 2007, 179, 6107–6114. [Google Scholar] [CrossRef] [PubMed]
- Tulic, M.K.; Hurrelbrink, R.J.; Prêle, C.M.; Laing, I.A.; Upham, J.W.; Le Souef, P.; Sly, P.D.; Holt, P.G. TLR4 Polymorphisms Mediate Impaired Responses to Respiratory Syncytial Virus and Lipopolysaccharide. J. Immunol. 2007, 179, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Fenhammar, J.; Rundgren, M.; Forestier, J.; Kalman, S.; Eriksson, S.; Frithiof, R. Toll-like receptor 4 inhibitor TAK-242 attenuates acute kidney injury in endotoxemic sheep. Anesthesiology 2011, 114, 1130–1137. [Google Scholar] [CrossRef]
- Zhong, Y.; Wu, S.; Yang, Y.; Li, G.Q.; Meng, L.; Zheng, Q.Y.; Li, Y.; Xu, G.L.; Zhang, K.Q.; Peng, K.F. LIGHT aggravates sepsis-associated acute kidney injury via TLR4-MyD88-NF-κB pathway. J. Cell. Mol. Med. 2020, 24, 11936–11948. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, G.; Xie, W.J.; Wang, L.; Zhang, R.; Huang, K.S.; Zhou, Q.S.; Chen, D.C. Lipopolysaccharide stimulates surfactant protein-a in human renal epithelial hk-2 cells through upregulating toll-like receptor 4 dependent MEK1/2-ERK1/2-NF-κb pathway. Chin. Med. J. 2017, 130, 1236–1243. [Google Scholar] [CrossRef]
- Okechukwu, C.C.; Pirro, N.T.; Chappell, M.C. Evidence that angiotensin II does not directly stimulate the MD2-TLR4 innate inflammatory pathway. Peptides 2021, 136. [Google Scholar] [CrossRef]
- Mohammad, B.I.; Raheem, A.K.; Hadi, N.R.; Jamil, D.A.; Al-Aubaidy, H.A. Reno-protective effects of TAK-242 on acute kidney injury in a rat model. Biochem. Biophys. Res. Commun. 2018, 503, 304–308. [Google Scholar] [CrossRef]
- Chen, L.; Yang, S.; Zumbrun, E.E.; Guan, H.; Nagarkatti, P.S.; Nagarkatti, M. Resveratrol attenuates lipopolysaccharide-induced acute kidney injury by suppressing inflammation driven by macrophages. Mol. Nutr. Food Res. 2015, 59, 853–864. [Google Scholar] [CrossRef]
- Huang, Z.; Zhuang, X.; Xie, C.; Hu, X.; Dong, X.; Guo, Y.; Li, S.; Liao, X. Exogenous Hydrogen Sulfide Attenuates High Glucose-Induced Cardiotoxicity by Inhibiting NLRP3 Inflammasome Activation by Suppressing TLR4/NF-κB Pathway in H9c2 Cells. Cell. Physiol. Biochem. 2016, 40, 1578–1590. [Google Scholar] [CrossRef] [PubMed]
- Obi, A.T.; Andraska, E.; Kanthi, Y.; Kessinger, C.W.; Elfline, M.; Luke, C.; Siahaan, T.J.; Jaffer, F.A.; Wakefield, T.W.; Henke, P.K. Endotoxaemia-augmented murine venous thrombosis is dependent on TLR-4 and ICAM-1, and potentiated by neutropenia. Thromb. Haemost. 2017, 117, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Dayang, E.Z.; Plantinga, J.; Ter Ellen, B.; Van Meurs, M.; Molema, G.; Moser, J. Identification of LPS-activated endothelial subpopulations with distinct inflammatory phenotypes and regulatory signaling mechanisms. Front. Immunol. 2019, 10, 1169. [Google Scholar] [CrossRef] [PubMed]
- Hato, T.; Winfree, S.; Kalakeche, R.; Dube, S.; Kumar, R.; Yoshimoto, M.; Plotkin, Z.; Dagher, P.C. The macrophage mediates the renoprotective effects of endotoxin preconditioning. J. Am. Soc. Nephrol. 2015, 26, 1347–1362. [Google Scholar] [CrossRef] [PubMed]
- Patole, P.S.; Schubert, S.; Hildinger, K.; Khandoga, S.; Khandoga, A.; Segerer, S.; Henger, A.; Kretzler, M.; Werner, M.; Krombach, F.; et al. Toll-like receptor-4: Renal cells and bone marrow cells signal for neutrophil recruitment during pyelonephritis. Kidney Int. 2005, 68, 2582–2587. [Google Scholar] [CrossRef]
- Anderberg, S.B.; Luther, T.; Frithiof, R. Physiological aspects of Toll-like receptor 4 activation in sepsis-induced acute kidney injury. Acta Physiol. 2017, 219, 573–588. [Google Scholar] [CrossRef]
- Nakano, D.; Doi, K.; Kitamura, H.; Kuwabara, T.; Mori, K.; Mukoyama, M.; Nishiyama, A. Reduction of tubular flow rate as a mechanism of oliguria in the early phase of endotoxemia revealed by intravital imaging. J. Am. Soc. Nephrol. 2015, 26, 3035–3044. [Google Scholar] [CrossRef]
- Zarbock, A.; Gomez, H.; Kellum, J.A. Sepsis-induced acute kidney injury revisited: Pathophysiology, prevention and future therapies. Curr. Opin. Crit. Care 2014, 20, 588–595. [Google Scholar] [CrossRef]
- Peng, Y.; Zhang, X.; Wang, Y.; Li, S.; Wang, J.; Liu, L. Overexpression of toll-like receptor 2 in glomerular endothelial cells and podocytes in septic acute kidney injury mouse model. Ren. Fail. 2015, 37, 694–698. [Google Scholar] [CrossRef]
- Liaudet, L.; Murthy, K.G.K.; Mabley, J.G.; Pacher, P.; Soriano, F.G.; Salzman, A.L.; Szabó, C. Comparison of inflammation, organ damage, and oxidant stress induced by Salmonella enterica serovar muenchen flagellin and serovar enteritidis lipopolysaccharide. Infect. Immun. 2002, 70, 192–198. [Google Scholar] [CrossRef]
- Andersen-Nissen, E.; Hawn, T.R.; Smith, K.D.; Nachman, A.; Lampano, A.E.; Uematsu, S.; Akira, S.; Aderem, A. Cutting Edge: Tlr5 −/− Mice Are More Susceptible to Escherichia coli Urinary Tract Infection. J. Immunol. 2007, 178, 4717–4720. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, Y.; Hu, Z.; Su, J.; Huo, Y.; Tan, B.; Wang, X.; Liu, Y. Small interfering RNA targeting toll-like receptor 9 protects mice against polymicrobial septic acute kidney injury. Nephron Exp. Nephrol. 2013, 122, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, N.; Tsuji, T.; Ohashi, N.; Kato, A.; Fujigaki, Y.; Yasuda, H. Role of mitochondrial DNA in septic AKI via toll-like receptor 9. J. Am. Soc. Nephrol. 2016, 27, 2009–2020. [Google Scholar] [CrossRef] [PubMed]
- Naito, Y.; Tsuji, T.; Nagata, S.; Tsuji, N.; Fujikura, T.; Ohashi, N.; Kato, A.; Miyajima, H.; Yasuda, H. IL-17A activated by Toll-like receptor 9 contributes to the development of septic acute kidney injury. Am. J. Physiol. Ren. Physiol. 2020, 318, F238–F247. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, H.; Leelahavanichkul, A.; Tsunoda, S.; Dear, J.W.; Takahashi, Y.; Ito, S.; Hu, X.; Zhou, H.; Doi, K.; Childs, R.; et al. Chloroquine and inhibition of Toll-like receptor 9 protect from sepsis-induced acute kidney injury. Am. J. Physiol. Ren. Physiol. 2008, 294, F1050–F1058. [Google Scholar] [CrossRef] [PubMed]
- de Jesus Soares, T.; Volpini, R.A.; Francescato, H.D.C.; Costa, R.S.; da Silva, C.G.A.; Coimbra, T.M. Effects of resveratrol on glycerol-induced renal injury. Life Sci. 2007, 81, 647–656. [Google Scholar] [CrossRef]
- Wang, N.; Mao, L.; Yang, L.; Zou, J.; Liu, K.; Liu, M.; Zhang, H.; Xiao, X.; Wang, K. Resveratrol protects against early polymicrobial sepsis-induced acute kidney injury through inhibiting endoplasmic reticulum stress-activated NF-κB pathway. Oncotarget 2017, 8, 36449–36461. [Google Scholar] [CrossRef]
- Zhang, J.; Tang, L.; Li, G.S.; Wang, J. The anti-inflammatory effects of curcumin on renal ischemia-reperfusion injury in rats. Ren. Fail. 2018, 40, 680–686. [Google Scholar] [CrossRef]
- Awad, A.S.; El-Sharif, A.A. Curcumin immune-mediated and anti-apoptotic mechanisms protect against renal ischemia/reperfusion and distant organ induced injuries. Int. Immunopharmacol. 2011, 11, 992–996. [Google Scholar] [CrossRef]
- Rogers, N.M.; Stephenson, M.D.; Kitching, A.R.; Horowitz, J.D.; Coates, P.T.H. Amelioration of renal ischaemia-reperfusion injury by liposomal delivery of curcumin to renal tubular epithelial and antigen-presenting cells. Br. J. Pharmacol. 2012, 166, 194–209. [Google Scholar] [CrossRef]
- Wu, J.; Pan, X.; Fu, H.; Zheng, Y.; Dai, Y.; Yin, Y.; Chen, Q.; Hao, Q.; Bao, D.; Hou, D. Effect of curcumin on glycerol-induced acute kidney injury in rats. Sci. Rep. 2017, 7, 10114. [Google Scholar] [CrossRef] [PubMed]
- Mercantepe, F.; Mercantepe, T.; Topcu, A.; Yılmaz, A.; Tumkaya, L. Protective effects of amifostine, curcumin, and melatonin against cisplatin-induced acute kidney injury. Naunyn. Schmiedebergs. Arch. Pharmacol. 2018, 391, 915–931. [Google Scholar] [CrossRef] [PubMed]
- Soetikno, V.; Sari, S.D.P.; Ul Maknun, L.; Sumbung, N.K.; Rahmi, D.N.I.; Pandhita, B.A.W.; Louisa, M.; Estuningtyas, A. Pre-Treatment with Curcumin Ameliorates Cisplatin-Induced Kidney Damage by Suppressing Kidney Inflammation and Apoptosis in Rats. Drug Res. 2018, 69, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.Y.; Wang, X.K.; Li, X.; Ji, K.; Du, S.H.; Liu, Y.; Kong, L.L.; Xu, J.C.; Yang, G.Q.; Chen, D.Q.; et al. Curcumin, as a pleiotropic agent, improves doxorubicin-induced nephrotic syndrome in rats. J. Ethnopharmacol. 2020, 250, 112502. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Beltrán, C.E.; Mukhopadhyay, P.; Horváth, B.; Rajesh, M.; Tapia, E.; García-Torres, I.; Pedraza-Chaverri, J.; Pacher, P. Sulforaphane, a natural constituent of broccoli, prevents cell death and inflammation in nephropathy. J. Nutr. Biochem. 2012, 23, 494–500. [Google Scholar] [CrossRef]
- Zhang, D.; Li, Y.; Liu, Y.; Xiang, X.; Dong, Z. Paclitaxel ameliorates lipopolysaccharide-induced kidney injury by binding myeloid differentiation protein-2 to block toll-like receptor 4-mediated nuclear factor-κB activation and cytokine production. J. Pharmacol. Exp. Ther. 2013, 345, 69–75. [Google Scholar] [CrossRef]
- Karpman, D.; Sartz, L.; Johnson, S. Pathophysiology of typical hemolytic uremic syndrome. Semin. Thromb. Hemost. 2010, 36, 575–585. [Google Scholar] [CrossRef]
- Fakhouri, F.; Zuber, J.; Frémeaux-Bacchi, V.; Loirat, C. Haemolytic uraemic syndrome. Lancet 2017, 390, 681–696. [Google Scholar] [CrossRef]
- Brigotti, M.; Carnicelli, D.; Arfilli, V.; Tamassia, N.; Borsetti, F.; Fabbri, E.; Tazzari, P.L.; Ricci, F.; Pagliaro, P.; Spisni, E.; et al. Identification of TLR4 as the Receptor That Recognizes Shiga Toxins in Human Neutrophils. J. Immunol. 2013, 191, 4748–4758. [Google Scholar] [CrossRef]
- Fischer, H.; Ellström, P.; Ekström, K.; Gustafsson, L.; Gustafsson, M.; Svanborg, C. Ceramide as a TLR4 agonist; a putative signalling intermediate between sphingolipid receptors for microbial ligands and TLR4. Cell. Microbiol. 2007, 9, 1239–1251. [Google Scholar] [CrossRef]
- Lafalla Manzano, A.F.; Gil Lorenzo, A.F.; Bocanegra, V.; Costantino, V.V.; Cacciamani, V.; Benardon, M.E.; Vallés, P.G. Rab7b participation on the TLR4 (Toll-like receptor) endocytic pathway in Shiga toxin-associated Hemolytic Uremic Syndrome (HUS). Cytokine 2019, 121, 154732. [Google Scholar] [CrossRef] [PubMed]
- Brigotti, M.; Arfilli, V.; Carnicelli, D.; Ricci, F.; Tazzari, P.L.; Ardissino, G.; Scavia, G.; Morabito, S.; He, X. Soluble Toll-like receptor 4 impairs the interaction of Shiga toxin 2a with human serum amyloid P component. Toxins 2018, 10, 379. [Google Scholar] [CrossRef] [PubMed]
- Toledo, C.C.; Rogers, T.J.; Svensson, M.; Tati, R.; Fischer, H.; Svanborg, C.; Karpman, D. Shiga toxin-mediated disease in MyD88-deficient mice infected with Escherichia coli O157:H7. Am. J. Pathol. 2008, 173, 1428–1439. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, M.A.; Griffin, K.A.; Lan, R.; Geng, H.; Saikumar, P.; Bidani, A.K. Acute kidney injury: A springboard for progression in chronic kidney disease. Am. J. Physiol. Ren. Physiol. 2010, 298, F1078–F1094. [Google Scholar] [CrossRef]
- Venkatachalam, M.A.; Weinberg, J.M.; Kriz, W.; Bidani, A.K. Failed tubule recovery, AKI-CKD transition, and kidney disease progression. J. Am. Soc. Nephrol. 2015, 26, 1765–1776. [Google Scholar] [CrossRef]
- Basile, D.P.; Donohoe, D.; Roethe, K.; Osborn, J.L. Renal ischemic injury results in permanent damage to peritubular capillaries and influences long-term function. Am. J. Physiol. Physiol. 2001, 281, F887–F899. [Google Scholar] [CrossRef]
- Hainmhire, E.Ó.; Humphreys, B.D. Fibrotic Changes Mediating Acute Kidney Injury to Chronic Kidney Disease Transition. Nephron 2017, 137, 264–267. [Google Scholar] [CrossRef]
- Murashima, M.; Nishimoto, M.; Kokubu, M.; Hamano, T.; Matsui, M.; Eriguchi, M.; Samejima, K.I.; Akai, Y.; Tsuruya, K. Inflammation as a predictor of acute kidney injury and mediator of higher mortality after acute kidney injury in non-cardiac surgery. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Mulay, S.R.; Holderied, A.; Kumar, S.V.; Anders, H.J. Targeting Inflammation in So-Called Acute Kidney Injury. Semin. Nephrol. 2016, 36, 17–30. [Google Scholar] [CrossRef]
- Rabb, H.; Griffin, M.D.; McKay, D.B.; Swaminathan, S.; Pickkers, P.; Rosner, M.H.; Kellum, J.A.; Ronco, C. Inflammation in AKI: Current understanding, key questions, and knowledge gaps. J. Am. Soc. Nephrol. 2016, 27, 371–379. [Google Scholar] [CrossRef]
- Grgic, I.; Duffield, J.S.; Humphreys, B.D. The origin of interstitial myofibroblasts in chronic kidney disease. Pediatr. Nephrol. 2012, 27, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.L.; Kisseleva, T.; Brenner, D.A.; Duffield, J.S. Pericytes and perivascular fibroblasts are the primary source of collagen-producing cells in obstructive fibrosis of the kidney. Am. J. Pathol. 2008, 173, 1617–1627. [Google Scholar] [CrossRef] [PubMed]
- Curci, C.; Castellano, G.; Stasi, A.; Divella, C.; Loverre, A.; Gigante, M.; Simone, S.; Cariello, M.; Montinaro, V.; Lucarelli, G.; et al. Endothelial-to-mesenchymal transition and renal fibrosis in ischaemia/reperfusion injury are mediated by complement anaphylatoxins and Akt pathway. Nephrol. Dial. Transpl. 2014, 29, 799–808. [Google Scholar] [CrossRef]
- Chen, C.; Chou, K.; Fang, H.; Hsu, C.; Huang, W.; Huang, C.; Huang, C.; Chen, H.; Lee, P. Progenitor-like cells derived from mouse kidney protect against renal fibrosis in a remnant kidney model via decreased endothelial mesenchymal transition. Stem Cell Res. Ther. 2015, 6. [Google Scholar] [CrossRef]
- Castellano, G.; Stasi, A.; Franzin, R.; Sallustio, F.; Divella, C.; Spinelli, A.; Netti, G.S.; Fiaccadori, E.; Cantaluppi, V.; Crovace, A.; et al. LPS-binding protein modulates acute renal fibrosis by inducing pericyte-to-myofibroblast trans-differentiation through TLR-4 signaling. Int. J. Mol. Sci. 2019, 20, 3682. [Google Scholar] [CrossRef]
- Souza, A.C.P.; Tsuji, T.; Baranova, I.N.; Bocharov, A.V.; Wilkins, K.J.; Street, J.M.; Alvarez-Prats, A.; Hu, X.; Eggerman, T.; Yuen, P.S.T.; et al. TLR4 mutant mice are protected from renal fibrosis and chronic kidney disease progression. Physiol. Rep. 2015, 3, e12558. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, X.; Zou, F.; Xu, T.; Pan, P.; Hu, C. Toll-like receptor-4 deficiency alleviates chronic intermittent hypoxia-induced renal injury, inflammation, and fibrosis. Sleep Breath. 2019, 23, 503–513. [Google Scholar] [CrossRef]
- Fu, Y.; Tang, C.; Cai, J.; Chen, G.; Zhang, D.; Dong, Z. Rodent models of AKI-CKD transition. Am. J. Physiol. Ren. Physiol. 2018, 315, F1098–F1106. [Google Scholar] [CrossRef]
- Li, R.; Guo, Y.; Zhang, Y.; Zhang, X.; Zhu, L.; Yan, T. Salidroside ameliorates renal interstitial fibrosis by inhibiting the TLR4/NF-κB and MAPK signaling pathways. Int. J. Mol. Sci. 2019, 20, 1103. [Google Scholar] [CrossRef]
- Sun, Y.; Xun, L.; Jin, G.; Shi, L. Salidroside protects renal tubular epithelial cells from hypoxia/reoxygenation injury in vitro. J. Pharmacol. Sci. 2018, 137, 170–176. [Google Scholar] [CrossRef]
- ul Ain, Q.; Batool, M.; Choi, S. TLR4-targeting therapeutics: Structural basis and computer-aided drug discovery approaches. Molecules 2020, 25, 627. [Google Scholar] [CrossRef] [PubMed]
- Kuzmich, N.N.; Sivak, K.V.; Chubarev, V.N.; Porozov, Y.B.; Savateeva-Lyubimova, T.N.; Peri, F. TLR4 signaling pathway modulators as potential therapeutics in inflammation and sepsis. Vaccines 2017, 5, 34. [Google Scholar] [CrossRef] [PubMed]

| Biomarker | Function | Origin | Samples |
|---|---|---|---|
| Cystatin C | Extracellular cysteine protease inhibitor | All nuclear cells | Serum |
| NGAL | Limits bacterial growth by sequestering iron-containing siderophores | Neutrophils | Serum/Urine |
| IGFBP7 | Main regulator of IGFs availability in cells | Renal and inflammatory cells | Urine |
| TIMP2 | Peptidase involved in MMP inhibition and ECM degradation | ||
| CCL14 | Pro-inflammatory chemokine | ||
| IL-18 | Pro-inflammatory cytokine | ||
| KIM-1 | Phosphatidylserine receptor that recognizes apoptotic cells and oxidized lipoproteins | Tubular cells | |
| NAG | Hydrolytic lysosomal enzyme | ||
| NHE3 | Sodium–hydrogen exchanger in apical side of the epithelial cells | ||
| α/π-GST | Phase II enzyme involved in cellular detoxification | ||
| ALAP | Enzymes involved in tubular damage | ||
| AP | |||
| γGT | |||
| L-FABP | Cytoplasmic proteins involved in binding, transport and metabolism of LCFAs |
| Regulation | Regulator | Proposed Mechanism | References |
|---|---|---|---|
| Soluble receptor | sTLR4 | Decoy for TLR4 ligands and coreceptors, preventing their association with the functional transmembrane receptor | [93,94] |
| Transmembrane regulators | ST2L | Bind to Myd88 and IRAK to reduce their recruitment to TLR4 | [95,96] |
| SIGIRR | |||
| RP105 | Associates with TLR4 extracellular domain and prevents ligand binding | [97] | |
| Cellular trafficking | Rab7 | Promotes TLR4 endosomal internalization and degradation | [98] |
| Adaptor proteins | Myd88s | Heterodimerizes with Myd88 and inhibits its interaction with IRAK4 | [99] |
| TOLLIP | Reduces IRAK1 autophosphorylation and promotes its degradation | [100] | |
| SOCS1 | Induces TIRAP and p65 degradation | [101] | |
| Ubiquitination | A20 | Promotes deubiquitination of TRAF6 | [102] |
| TRIAD3A | Induces TLR4 ubiquitination and subsequent degradation | [103] | |
| Phosphorylation | SHP1 | Dephosphorylates IRAK1 | [104] |
| SHP2 | Dephosphorylates TBK1 | [105] | |
| Transcriptional regulation | TGFb | Inhibits TLR4 gene expression and promotes Myd88 degradation | [106,107] |
| IL10 | Reduces TLR4, Myd88, IRAK1 and TRAF6 expression | [108] | |
| miR-155-5p | Reduces Myd88 expression | [109] | |
| miR-210-5p | Inhibits NFkB1 mRNA subunit | [109] | |
| Feedback inhibition | TRIM30α | Promotes TAB2 and TAB3 degradation | [110] |
| IκBNS | Reduces IL6 gene expression | [111] | |
| Bcl3 | Blocks p50 ubiquitination and prevents DNA binding of NFkB | [112] |
| Compound | AKI Model | Beneficial Effects | Reference |
|---|---|---|---|
| TAK-242 | I/R | Reduces serum creatinine and urea concentration. Decreases IL-18 and malondialdehyde renal levels | [190] |
| Rhabdomyolysis | Decreases functional and histological renal damage. Reduces renal TNF-α, IL-6 and IL-1β levels and macrophage infiltration | [161] | |
| Decreases renal damage, serum creatinine and BUN levels. Reduces renal IL-6, TNF-α and TLR4 expression and NFκB signaling | [164] | ||
| Acetaminophen induced AKI | Decreases histological renal damage, serum creatinine concentration and partially restores glutathione levels | [174] | |
| Sepsis induced AKI | Improves creatinine clearance and urine output, reduces renal neutrophil accumulation and glomerular endothelial swelling | [85] | |
| Attenuates arterial pressure, plasma creatinine and BUN levels | [186] | ||
| Reduces TLR4-Myd88-NFκB signaling pathway, decreases renal levels of TNF-α, IL-1β and IL-6 and inflammatory cell infiltration | [187] | ||
| Eritoran | I/R | Decreases serum creatinine, renal histological damage, monocyte infiltration and inflammatory markers (TNF-α, IL-1β, CCL2) | [155] |
| Resveratrol | I/R | Ameliorates histological renal damage, serum creatinine and BUN, reduces renal levels of IL-6, TNF-α, IL-10, IFNγ, caspase-3 activity and improves redox balance (MDA, SOD) | [137,138] |
| Rhabdomyolysis | Attenuates creatinine levels, cortical macrophage infiltration and necrosis. Decreases renal NFκB activation as well as HO-1 and nitrotyrosine expression. | [207] | |
| Sepsis induced AKI | Improves renal function and tubular epithelial cell injury. Decreases serum content and renal mRNA expression of TNFα, IL-1β and IL-6. Inhibits IRE1 phosphorylation and NFκB activity in the kidney | [208] | |
| Improves renal function, reduces serum and renal inflammation, macrophage infiltration and TLR4 activation. Prevents endothelial cell permeability. Decreases expression of iNOS, Bcl2 and BclxL in macrophages | [191] | ||
| Curcumin | I/R | Decreases renal damage, serum creatinine and BUN. Reduces proinflammatory TNF-α and IL-6 levels. Attenuates NFκB signaling and increases p-JAK2 and p-STAT3 expression. | [209] |
| Reduces serum and renal level of TNF-α, IL-1β, IL-12, IL-18 and INF-γ | [210] | ||
| Decreases renal damage and serum creatinine. Decreases inflammatory chemokine expression, neutrophil infiltration, intracellular ROS production and cellular apoptosis. Reduces TLR4 and TNFα expression and inhibits NFκB and MAPK signaling pathways | [211] | ||
| Rhabdomyolysis | Decreases serum creatinine levels, endothelial damage, inflammatory markers, redox balance and tubular cell death. Decreases TLR4 and Myd88 gene expression and activation of NFκB and ERK1/2 pathways | [45] | |
| Ameliorates renal damage, inflammation and apoptosis. | [212] | ||
| Cisplatin induced AKI | Decreases serum urea and creatinine, reduces tubular necrosis, NFκB/p65 levels and caspase-3 expression | [213] | |
| Reduces renal histological injury, and plasma creatinine and BUN levels. Decreases renal MDA and restores renal GSH levels. Reduces ERK1/2 phosphorylation and NFκB expression. Decreases TNFα, IL-6, KIM-1 and NGAL mRNA expression. | [214] | ||
| Doxorubicin induced AKI | Reduces proteinuria and podocyte injury. Ameliorates renal function, decreases oxidative stress and inhibits NFκB activation. | [215] | |
| Sulforaphane | Cisplatin induced AKI | Reduces the activation of NFκB, p53, JNK and p38 pathways. Decreases TNF-α levels, expression of ICAM/VCAM and inflammatory infiltration | [216] |
| Paclitaxel | Sepsis induced AKI | Increases survival rate, downregulates TNF-α, IL-1β and IL-6 production, inhibits the expression and activation of NFκB. | [217] |
| NaHS (H2S donor) | Improves renal function and kidney histopathological changes, attenuates LPS-induced inflammation and oxidative stress, and reduces expression of TLR4, NLRP3, and caspase-1 | [78] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vázquez-Carballo, C.; Guerrero-Hue, M.; García-Caballero, C.; Rayego-Mateos, S.; Opazo-Ríos, L.; Morgado-Pascual, J.L.; Herencia-Bellido, C.; Vallejo-Mudarra, M.; Cortegano, I.; Gaspar, M.L.; et al. Toll-Like Receptors in Acute Kidney Injury. Int. J. Mol. Sci. 2021, 22, 816. https://doi.org/10.3390/ijms22020816
Vázquez-Carballo C, Guerrero-Hue M, García-Caballero C, Rayego-Mateos S, Opazo-Ríos L, Morgado-Pascual JL, Herencia-Bellido C, Vallejo-Mudarra M, Cortegano I, Gaspar ML, et al. Toll-Like Receptors in Acute Kidney Injury. International Journal of Molecular Sciences. 2021; 22(2):816. https://doi.org/10.3390/ijms22020816
Chicago/Turabian StyleVázquez-Carballo, Cristina, Melania Guerrero-Hue, Cristina García-Caballero, Sandra Rayego-Mateos, Lucas Opazo-Ríos, José Luis Morgado-Pascual, Carmen Herencia-Bellido, Mercedes Vallejo-Mudarra, Isabel Cortegano, María Luisa Gaspar, and et al. 2021. "Toll-Like Receptors in Acute Kidney Injury" International Journal of Molecular Sciences 22, no. 2: 816. https://doi.org/10.3390/ijms22020816
APA StyleVázquez-Carballo, C., Guerrero-Hue, M., García-Caballero, C., Rayego-Mateos, S., Opazo-Ríos, L., Morgado-Pascual, J. L., Herencia-Bellido, C., Vallejo-Mudarra, M., Cortegano, I., Gaspar, M. L., de Andrés, B., Egido, J., & Moreno, J. A. (2021). Toll-Like Receptors in Acute Kidney Injury. International Journal of Molecular Sciences, 22(2), 816. https://doi.org/10.3390/ijms22020816








