From Rheumatoid Factor to Anti-Citrullinated Protein Antibodies and Anti-Carbamylated Protein Antibodies for Diagnosis and Prognosis Prediction in Patients with Rheumatoid Arthritis
Abstract
1. Introduction: Overview of Autoantibodies in Rheumatoid Arthritis
2. Characteristics of RF, ACPA and anti-CarP Ab
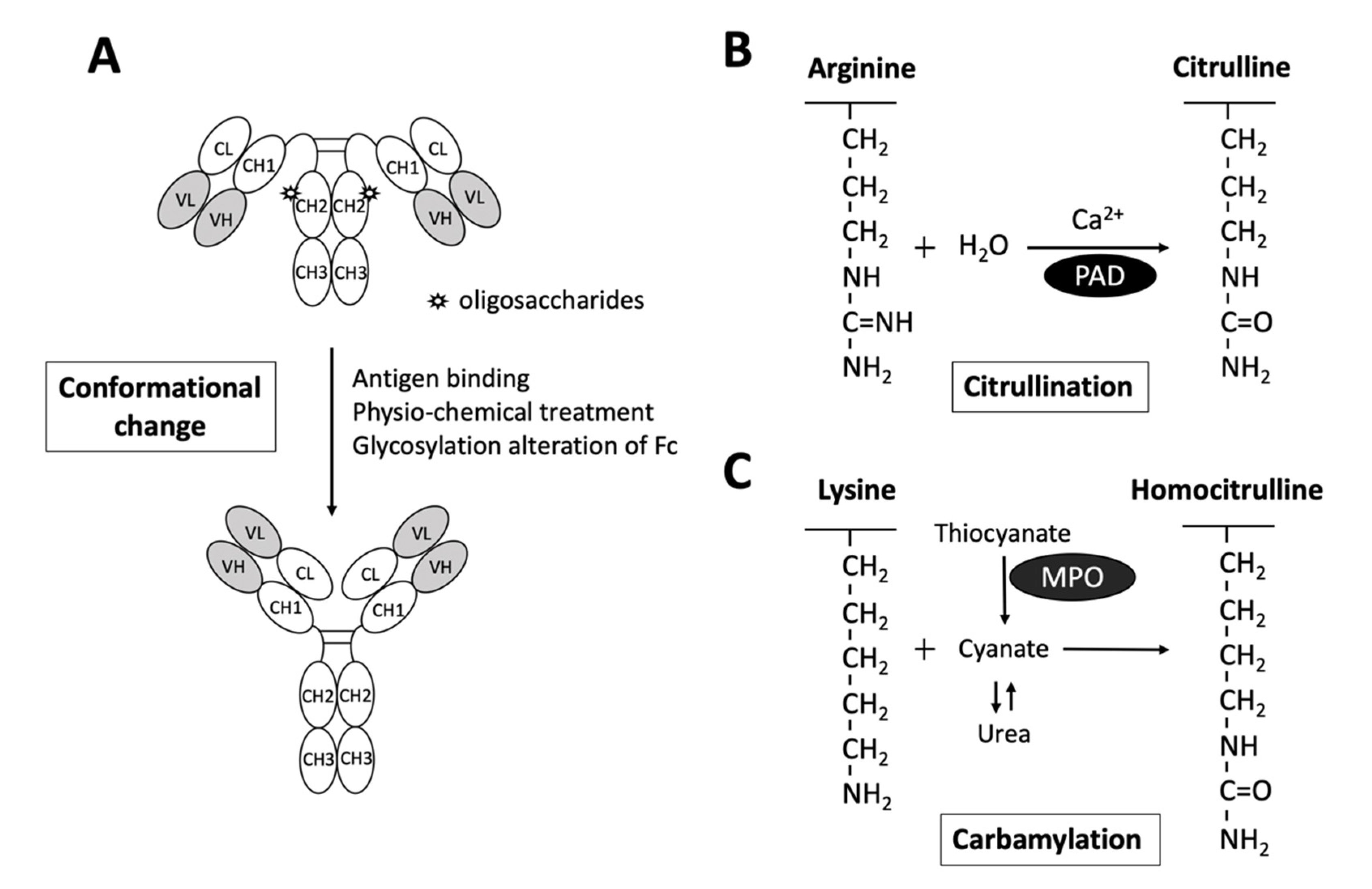
2.1. Rheumatoid Factors
2.2. Anti-Citrullinated Protein Antibodies
2.3. Anti-Carbamylated protein Antibodies
3. Risk Factors for the Production of RF, ACPA and Anti-CarP Ab
3.1. Genetic Risk Factors
3.2. Environmental Risk Factors
3.2.1. Tobacco Smoking
3.2.2. Microbial Triggers
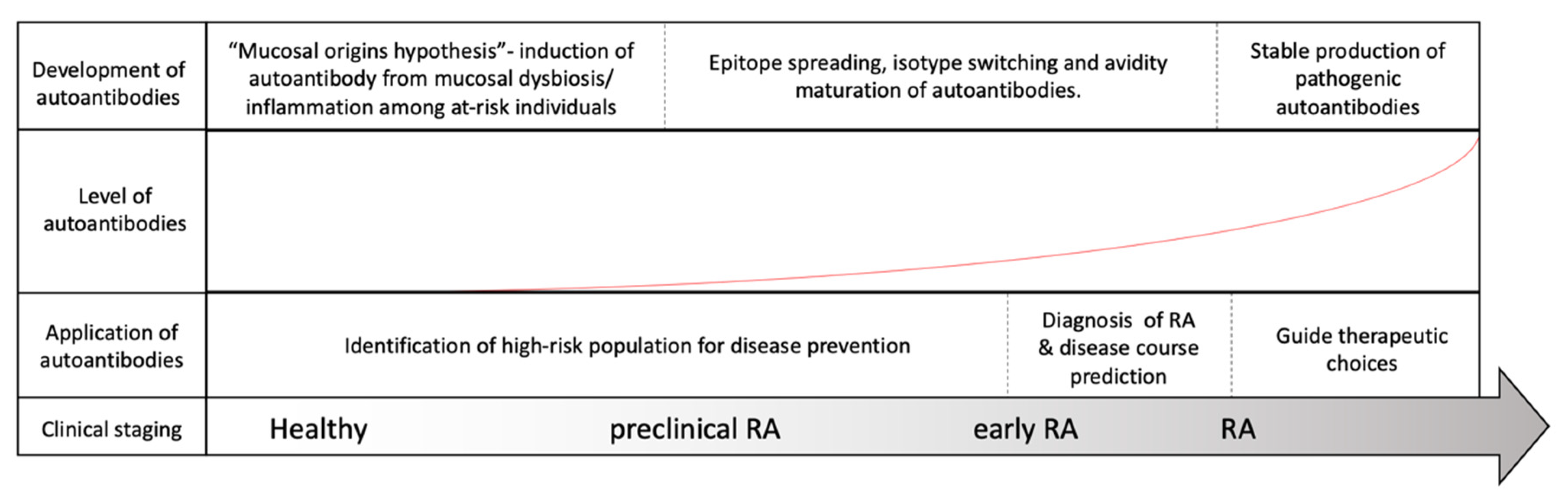
4. Predictive Values of RF, ACPA and Anti-CarP Ab in the Diagnosis of Presymptomatic RA Patients
4.1. Rheumatoid Factor
4.2. Anti-Citrullinated Protein Antibodies
4.3. Anti-Carbamylated Protein Antibodies
4.4. Additive Values of the RF, ACPA and Anti-CarP Ab
5. Monitoring RA Disease Activity and Progression with RF, ACPA and Anti-CarP Ab
5.1. Rheumatoid Factor
5.2. Anti-Citrullinated Protein Antibodies
5.3. Anti-Carbamylated Protein Antibodies
6. Summary and Future Prospects
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACPA | anti-citrullinated protein antibody |
| ACR | American College of Rheumatology |
| Anti-CarP Ab | Anti-carbamylated protein antibody |
| AutoAb | autoantibody |
| DMARDs | disease-modifying anti-rheumatic drugs |
| EULAR | European League Against Rheumatism |
| Fab | antigen-binding fragment |
| Fc | crystallizable fragment |
| GRP78 | 78-kDa glucose-regulated protein |
| HLA | human leukocyte antigen |
| IgG | immunoglobulins G |
| MPO | myeloperoxidase |
| MTX | methotrexate |
| NPV | negative predictive value |
| PAD | peptidyl arginine deiminase |
| PPV | positive predictive value |
| PTM | post-translational modified |
| RA | rheumatoid arthritis |
| RF | rheumatoid factor |
| SE | shared epitope |
| TNF | tumor necrosis factor |
References
- McInnes, I.B.; Schett, G. The pathogenesis of rheumatoid arthritis. N. Engl. J. Med. 2011, 365, 2205–2219. [Google Scholar] [CrossRef] [PubMed]
- Scott, I.C.; Machin, A.; Mallen, C.D.; Hider, S.L. The extra-articular impacts of rheumatoid arthritis: Moving towards holistic care. BMC Rheumatol. 2018, 2, 32. [Google Scholar] [CrossRef] [PubMed]
- Trouw, L.A.; Rispens, T.; Toes, R.E.M. Beyond citrullination: Other post-translational protein modifications in rheumatoid arthritis. Nat. Rev. Rheumatol. 2017, 13, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Aletaha, D.; Barton, A.; Burmester, G.R.; Emery, P.; Firestein, G.S.; Kavanaugh, A.; McInnes, I.B.; Solomon, D.H.; Strand, V.; et al. Rheumatoid arthritis. Nat. Rev. Dis. Primers 2018, 4, 18001. [Google Scholar] [CrossRef] [PubMed]
- Waaler, E.; Norberg, R. On the occurrence of a factor in human serum activating the specific agglutination of sheep blood corpuscles. Commentary. APMIS 2007, 115, 422–439. [Google Scholar]
- Pike, R.M.; Sulkin, S.E.; Coggeshall, H.C. Serological reactions in rheumatoid arthritis; factors affecting the agglutination of sensitized sheep erythrocytes in rheumatid-arthritis serum. J. Immunol. 1949, 63, 441–446. [Google Scholar]
- Franklin, E.C.; Holman, H.R.; Muller-Eberhard, H.J.; Kunkel, H.G. An unusual protein component of high molecular weight in the serum of certain patients with rheumatoid arthritis. J. Exp. Med. 1957, 105, 425–438. [Google Scholar] [CrossRef]
- Edelman, G.M.; Kunkel, H.G.; Franklin, E.C. Interaction of the rheumatoid factor with antigen-antibody complexes and aggregated gamma globulin. J. Exp. Med. 1958, 108, 105–120. [Google Scholar] [CrossRef]
- Carubbi, F.; Alunno, A.; Cipriani, P.; Bistoni, O.; Scipioni, R.; Liakouli, V.; Ruscitti, P.; Berardicurti, O.; Bartolomeo, S.D.; Gerli, R.; et al. Laboratory Assessment of Patients with Suspected Rheumatic Musculoskeletal Diseases: Challenges and Pitfalls. Curr. Rheumatol. Rev. 2019, 15, 27–43. [Google Scholar] [CrossRef]
- Nishimura, K.; Sugiyama, D.; Kogata, Y.; Tsuji, G.; Nakazawa, T.; Kawano, S.; Saigo, K.; Morinobu, A.; Koshiba, M.; Kuntz, K.M.; et al. Meta-analysis: Diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann. Intern. Med. 2007, 146, 797–808. [Google Scholar] [CrossRef]
- Regueiro, C.; Rodriguez-Martinez, L.; Nuno, L.; Ortiz, A.M.; Villalba, A.; Pascual-Salcedo, D.; Martinez-Feito, A.; Gonzalez-Alvaro, I.; Balsa, A.; Gonzalez, A. Improved RA classification among early arthritis patients with the concordant presence of three RA autoantibodies: Analysis in two early arthritis clinics. Arthritis Res. Ther. 2019, 21, 280. [Google Scholar] [CrossRef] [PubMed]
- Nienhuis, R.L.; Mandema, E. A New Serum Factor in Patients with Rheumatoid Arthritis; the Antiperinuclear Factor. Ann. Rheum. Dis. 1964, 23, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Valesini, G.; Gerardi, M.C.; Iannuccelli, C.; Pacucci, V.A.; Pendolino, M.; Shoenfeld, Y. Citrullination and autoimmunity. Autoimmun. Rev. 2015, 14, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Van Beers, J.J.; Schwarte, C.M.; Stammen-Vogelzangs, J.; Oosterink, E.; Bozic, B.; Pruijn, G.J. The rheumatoid arthritis synovial fluid citrullinome reveals novel citrullinated epitopes in apolipoprotein E, myeloid nuclear differentiation antigen, and beta-actin. Arthritis Rheum. 2013, 65, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.; Wang, D.; Wilhelm, M.; Zolg, D.P.; Schmidt, T.; Schnatbaum, K.; Reimer, U.; Ponten, F.; Uhlen, M.; Hahne, H.; et al. Mining the Human Tissue Proteome for Protein Citrullination. Mol. Cell Proteom. 2018, 17, 1378–1391. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Chen, F.F.; Gao, W.B.; Wang, H.Y.; Zhao, N.W.; Xu, M.; Gao, D.Y.; Yu, W.; Yan, X.L.; Zhao, J.N.; et al. Identification of citrullinated peptides in the synovial fluid of patients with rheumatoid arthritis using LC-MALDI-TOF/TOF. Clin. Rheumatol. 2016, 35, 2185–2194. [Google Scholar] [CrossRef]
- Tilvawala, R.; Nguyen, S.H.; Maurais, A.J.; Nemmara, V.V.; Nagar, M.; Salinger, A.J.; Nagpal, S.; Weerapana, E.; Thompson, P.R. The Rheumatoid Arthritis-Associated Citrullinome. Cell Chem. Biol. 2018, 25, 691–704 e696. [Google Scholar] [CrossRef]
- Romero, V.; Fert-Bober, J.; Nigrovic, P.A.; Darrah, E.; Haque, U.J.; Lee, D.M.; van Eyk, J.; Rosen, A.; Andrade, F. Immune-mediated pore-forming pathways induce cellular hypercitrullination and generate citrullinated autoantigens in rheumatoid arthritis. Sci. Transl. Med. 2013, 5, 209ra150. [Google Scholar] [CrossRef]
- Pinheiro, G.C.; Scheinberg, M.A.; Aparecida da Silva, M.; Maciel, S. Anti-cyclic citrullinated peptide antibodies in advanced rheumatoid arthritis. Ann. Intern. Med. 2003, 139, 234–235. [Google Scholar] [CrossRef]
- Lee, D.M.; Schur, P.H. Clinical utility of the anti-CCP assay in patients with rheumatic diseases. Ann. Rheum. Dis. 2003, 62, 870–874. [Google Scholar] [CrossRef]
- Pruijn, G.J.; Wiik, A.; van Venrooij, W.J. The use of citrullinated peptides and proteins for the diagnosis of rheumatoid arthritis. Arthritis Res. Ther. 2010, 12, 203. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Y.; Yang, H.Y.; Lai, J.H. Anti-Citrullinated Protein Antibodies in Patients with Rheumatoid Arthritis: Biological Effects and Mechanisms of Immunopathogenesis. Int. J. Mol. Sci. 2020, 21, 4015. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Rani, K.; Roychoudhury, A.; Chawla, A.; Nikolajeff, F.; Kumar, S. Novel Insights into Regulation of Human Teeth Biomineralization: Deciphering the Role of Post-Translational Modifications in a Tooth Protein Extract. Int. J. Mol. Sci. 2019, 20, 4035. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; van de Stadt, L.A.; Levarht, E.W.; Huizinga, T.W.; Hamann, D.; van Schaardenburg, D.; Toes, R.E.; Trouw, L.A. Anti-carbamylated protein (anti-CarP) antibodies precede the onset of rheumatoid arthritis. Ann. Rheum. Dis. 2014, 73, 780–783. [Google Scholar] [CrossRef] [PubMed]
- van Dijk, B.T.; Trouw, L.A.; van der Helm-van Mil, A.H.M.; Huizinga, T.W.J. Substitution of the quantitative serological component in the 2010 criteria for RA with qualitative presence of three autoantibodies yields similar performance: Response to the article by Regueiro et al. Arthritis Res. Ther. 2020, 22, 85. [Google Scholar] [CrossRef]
- Verheul, M.K.; Bohringer, S.; van Delft, M.A.M.; Jones, J.D.; Rigby, W.F.C.; Gan, R.W.; Holers, V.M.; Edison, J.D.; Deane, K.D.; Janssen, K.M.J.; et al. Triple Positivity for Anti-Citrullinated Protein Autoantibodies, Rheumatoid Factor, and Anti-Carbamylated Protein Antibodies Conferring High Specificity for Rheumatoid Arthritis: Implications for Very Early Identification of At-Risk Individuals. Arthritis Rheumatol. 2018, 70, 1721–1731. [Google Scholar] [CrossRef]
- Shi, J.; Knevel, R.; Suwannalai, P.; van der Linden, M.P.; Janssen, G.M.; van Veelen, P.A.; Levarht, N.E.; van der Helm-van Mil, A.H.; Cerami, A.; Huizinga, T.W.; et al. Autoantibodies recognizing carbamylated proteins are present in sera of patients with rheumatoid arthritis and predict joint damage. Proc. Natl. Acad. Sci. USA 2011, 108, 17372–17377. [Google Scholar] [CrossRef]
- Malmstrom, V.; Catrina, A.I.; Klareskog, L. The immunopathogenesis of seropositive rheumatoid arthritis: From triggering to targeting. Nat. Rev. Immunol. 2017, 17, 60–75. [Google Scholar] [CrossRef]
- Slaughter, L.; Carson, D.A.; Jensen, F.C.; Holbrook, T.L.; Vaughan, J.H. In vitro effects of Epstein-Barr virus on peripheral blood mononuclear cells from patients with rheumatoid arthritis and normal subjects. J. Exp. Med. 1978, 148, 1429–1434. [Google Scholar] [CrossRef]
- Welch, M.J.; Fong, S.; Vaughan, J.; Carson, D. Increased frequency of rheumatoid factor precursor B lymphocytes after immunization of normal adults with tetanus toxoid. Clin. Exp. Immunol. 1983, 51, 299–304. [Google Scholar] [PubMed]
- Van Snick, J.L.; Van Roost, E.; Markowetz, B.; Cambiaso, C.L.; Masson, P.L. Enhancement by IgM rheumatoid factor of in vitro ingestion by macrophages and in vivo clearance of aggregated IgG or antigen-antibody complexes. Eur. J. Immunol. 1978, 8, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Falkenburg, W.J.J.; von Richthofen, H.J.; Rispens, T. On the origin of rheumatoid factors: Insights from analyses of variable region sequences. Semin. Arthritis Rheum. 2019, 48, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Van Schaardenburg, D.; Lagaay, A.M.; Otten, H.G.; Breedveld, F.C. The relation between class-specific serum rheumatoid factors and age in the general population. Br. J. Rheumatol. 1993, 32, 546–549. [Google Scholar] [CrossRef] [PubMed]
- Sutton, B.; Corper, A.; Bonagura, V.; Taussig, M. The structure and origin of rheumatoid factors. Immunol. Today. 2000, 21, 177–183. [Google Scholar] [CrossRef]
- Nell, V.P.; Machold, K.P.; Stamm, T.A.; Eberl, G.; Heinzl, H.; Uffmann, M.; Smolen, J.S.; Steiner, G. Autoantibody profiling as early diagnostic and prognostic tool for rheumatoid arthritis. Ann. Rheum. Dis. 2005, 64, 1731–1736. [Google Scholar] [CrossRef]
- Malmstrom, V.; Gronwall, C. The parallel worlds of ACPA-positive and RF-positive B cells. Nat. Rev. Rheumatol. 2018, 14, 626–628. [Google Scholar] [CrossRef]
- Westwood, O.M.; Nelson, P.N.; Hay, F.C. Rheumatoid factors: What’s new? Rheumatology (Oxford) 2006, 45, 379–385. [Google Scholar] [CrossRef]
- Maibom-Thomsen, S.L.; Trier, N.H.; Holm, B.E.; Hansen, K.B.; Rasmussen, M.I.; Chailyan, A.; Marcatili, P.; Hojrup, P.; Houen, G. Immunoglobulin G structure and rheumatoid factor epitopes. PLoS ONE 2019, 14, e0217624. [Google Scholar] [CrossRef]
- Lu, M.C.; Lai, N.S.; Yu, H.C.; Huang, H.B.; Hsieh, S.C.; Yu, C.L. Anti-citrullinated protein antibodies bind surface-expressed citrullinated Grp78 on monocyte/macrophages and stimulate tumor necrosis factor alpha production. Arthritis Rheum. 2010, 62, 1213–1223. [Google Scholar] [CrossRef]
- Kampstra, A.S.B.; Dekkers, J.S.; Volkov, M.; Dorjee, A.L.; Hafkenscheid, L.; Kempers, A.C.; van Delft, M.; Kissel, T.; Reijm, S.; Janssen, G.M.C.; et al. Different classes of anti-modified protein antibodies are induced on exposure to antigens expressing only one type of modification. Ann. Rheum. Dis. 2019, 78, 908–916. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Willemze, A.; Janssen, G.M.; van Veelen, P.A.; Drijfhout, J.W.; Cerami, A.; Huizinga, T.W.; Trouw, L.A.; Toes, R.E. Recognition of citrullinated and carbamylated proteins by human antibodies: Specificity, cross-reactivity and the ‘AMC-Senshu’ method. Ann. Rheum. Dis. 2013, 72, 148–150. [Google Scholar] [CrossRef] [PubMed]
- Johansson, L.; Pratesi, F.; Brink, M.; Arlestig, L.; D’Amato, C.; Bartaloni, D.; Migliorini, P.; Rantapaa-Dahlqvist, S. Antibodies directed against endogenous and exogenous citrullinated antigens pre-date the onset of rheumatoid arthritis. Arthritis Res. Ther. 2016, 18, 127. [Google Scholar] [CrossRef]
- Ge, C.; Holmdahl, R. The structure, specificity and function of anti-citrullinated protein antibodies. Nat. Rev. Rheumatol. 2019, 15, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Kongpachith, S.; Lingampalli, N.; Ju, C.H.; Blum, L.K.; Lu, D.R.; Elliott, S.E.; Mao, R.; Robinson, W.H. Affinity Maturation of the Anti-Citrullinated Protein Antibody Paratope Drives Epitope Spreading and Polyreactivity in Rheumatoid Arthritis. Arthritis Rheumatol. 2019, 71, 507–517. [Google Scholar] [CrossRef]
- Rombouts, Y.; Ewing, E.; van de Stadt, L.A.; Selman, M.H.; Trouw, L.A.; Deelder, A.M.; Huizinga, T.W.; Wuhrer, M.; van Schaardenburg, D.; Toes, R.E.; et al. Anti-citrullinated protein antibodies acquire a pro-inflammatory Fc glycosylation phenotype prior to the onset of rheumatoid arthritis. Ann. Rheum. Dis. 2015, 74, 234–241. [Google Scholar] [CrossRef]
- Arlestig, L.; Mullazehi, M.; Kokkonen, H.; Rocklov, J.; Ronnelid, J.; Dahlqvist, S.R. Antibodies against cyclic citrullinated peptides of IgG, IgA and IgM isotype and rheumatoid factor of IgM and IgA isotype are increased in unaffected members of multicase rheumatoid arthritis families from northern Sweden. Ann. Rheum. Dis. 2012, 71, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, R.; Brandt, J.; Eggert, M.; Karberg, K.; Krause, A.; Neeck, G.; Mueller-Hilke, B. IgG1 and IgG4 are the predominant subclasses among auto-antibodies against two citrullinated antigens in RA. Rheumatology (Oxford) 2008, 47, 1489–1492. [Google Scholar] [CrossRef]
- Anumula, K.R. Quantitative glycan profiling of normal human plasma derived immunoglobulin and its fragments Fab and Fc. J. Immunol Methods. 2012, 382, 167–176. [Google Scholar] [CrossRef]
- Vergroesen, R.D.; Slot, L.M.; Hafkenscheid, L.; Koning, M.T.; van der Voort, E.I.H.; Grooff, C.A.; Zervakis, G.; Veelken, H.; Huizinga, T.W.J.; Rispens, T.; et al. B-cell receptor sequencing of anti-citrullinated protein antibody (ACPA) IgG-expressing B cells indicates a selective advantage for the introduction of N-glycosylation sites during somatic hypermutation. Ann. Rheum Dis. 2018, 77, 956–958. [Google Scholar]
- Hafkenscheid, L.; Bondt, A.; Scherer, H.U.; Huizinga, T.W.; Wuhrer, M.; Toes, R.E.; Rombouts, Y. Structural Analysis of Variable Domain Glycosylation of Anti-Citrullinated Protein Antibodies in Rheumatoid Arthritis Reveals the Presence of Highly Sialylated Glycans. Mol. Cell Proteomics 2017, 16, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Hafkenscheid, L.; de Moel, E.; Smolik, I.; Tanner, S.; Meng, X.; Jansen, B.C.; Bondt, A.; Wuhrer, M.; Huizinga, T.W.J.; Toes, R.E.M.; et al. N-Linked Glycans in the Variable Domain of IgG Anti-Citrullinated Protein Antibodies Predict the Development of Rheumatoid Arthritis. Arthritis Rheumatol. 2019, 71, 1626–1633. [Google Scholar] [CrossRef] [PubMed]
- Scherer, H.U.; van der Woude, D.; Ioan-Facsinay, A.; el Bannoudi, H.; Trouw, L.A.; Wang, J.; Haupl, T.; Burmester, G.R.; Deelder, A.M.; Huizinga, T.W.; et al. Glycan profiling of anti-citrullinated protein antibodies isolated from human serum and synovial fluid. Arthritis Rheum. 2010, 62, 1620–1629. [Google Scholar] [PubMed]
- Van Zanten, A.; Arends, S.; Roozendaal, C.; Limburg, P.C.; Maas, F.; Trouw, L.A.; Toes, R.E.M.; Huizinga, T.W.J.; Bootsma, H.; Brouwer, E. Presence of anticitrullinated protein antibodies in a large population-based cohort from the Netherlands. Ann. Rheum Dis. 2017, 76, 1184–1190. [Google Scholar] [CrossRef]
- van Beers, J.J.; Willemze, A.; Jansen, J.J.; Engbers, G.H.; Salden, M.; Raats, J.; Drijfhout, J.W.; van der Helm-van Mil, A.H.; Toes, R.E.; Pruijn, G.J. ACPA fine-specificity profiles in early rheumatoid arthritis patients do not correlate with clinical features at baseline or with disease progression. Arthritis Res. Ther. 2013, 15, R140. [Google Scholar] [CrossRef]
- Schwenzer, A.; Jiang, X.; Mikuls, T.R.; Payne, J.B.; Sayles, H.R.; Quirke, A.M.; Kessler, B.M.; Fischer, R.; Venables, P.J.; Lundberg, K.; et al. Identification of an immunodominant peptide from citrullinated tenascin-C as a major target for autoantibodies in rheumatoid arthritis. Ann. Rheum Dis. 2016, 75, 1876–1883. [Google Scholar]
- Shi, J.; van Veelen, P.A.; Mahler, M.; Janssen, G.M.; Drijfhout, J.W.; Huizinga, T.W.; Toes, R.E.; Trouw, L.A. Carbamylation and antibodies against carbamylated proteins in autoimmunity and other pathologies. Autoimmun. Rev. 2014, 13, 225–230. [Google Scholar] [CrossRef]
- Kraus, L.M.; Kraus Jr, A.P. Carbamoylation of amino acids and proteins in uremia. Kidney Int. Suppl. 2001, 78, S102–S107. [Google Scholar] [CrossRef]
- Ospelt, C.; Bang, H.; Feist, E.; Camici, G.; Keller, S.; Detert, J.; Kramer, A.; Gay, S.; Ghannam, K.; Burmester, G.R. Carbamylation of vimentin is inducible by smoking and represents an independent autoantigen in rheumatoid arthritis. Ann. Rheum Dis. 2017, 76, 1176–1183. [Google Scholar] [CrossRef]
- Nakabo, S. Clinical and etiological meaning of anti-carbamylated protein antibodies in rheumatoid arthritis. Immunol Med. 2018, 41, 147–153. [Google Scholar]
- Verheul, M.K.; Yee, A.; Seaman, A.; Janssen, G.M.; van Veelen, P.A.; Drijfhout, J.W.; Toes, R.E.M.; Mahler, M.; Trouw, L.A. Identification of carbamylated alpha 1 anti-trypsin (A1AT) as an antigenic target of anti-CarP antibodies in patients with rheumatoid arthritis. J. Autoimmun. 2017, 80, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.C.; Lai, P.H.; Lai, N.S.; Huang, H.B.; Koo, M.; Lu, M.C. Increased Serum Levels of Anti-Carbamylated 78-kDa Glucose-Regulated Protein Antibody in Patients with Rheumatoid Arthritis. Int. J. Mol. Sci. 2016, 17, 1510. [Google Scholar] [CrossRef] [PubMed]
- Van Delft, M.A.M.; van der Woude, D.; Toes, R.E.M.; Trouw, L.A. Secretory form of rheumatoid arthritis-associated autoantibodies in serum are mainly of the IgM isotype, suggesting a continuous reactivation of autoantibody responses at mucosal surfaces. Ann. Rheum. Dis. 2019, 78, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Van Delft, M.A.M.; Verheul, M.K.; Burgers, L.E.; Rantapaa-Dahlqvist, S.; van der Helm-van Mil, A.H.M.; Huizinga, T.W.J.; Toes, R.E.M.; Trouw, L.A. The anti-carbamylated protein antibody response is of overall low avidity despite extensive isotype switching. Rheumatology (Oxford) 2018, 57, 1583–1591. [Google Scholar]
- Jiang, X.; Trouw, L.A.; van Wesemael, T.J.; Shi, J.; Bengtsson, C.; Kallberg, H.; Malmstrom, V.; Israelsson, L.; Hreggvidsdottir, H.; Verduijn, W.; et al. Anti-CarP antibodies in two large cohorts of patients with rheumatoid arthritis and their relationship to genetic risk factors, cigarette smoking and other autoantibodies. Ann. Rheum. Dis. 2014, 73, 1761–1768. [Google Scholar] [CrossRef]
- Verheul, M.K.; Shiozawa, K.; Levarht, E.W.; Huizinga, T.W.; Toes, R.E.; Trouw, L.A.; Shiozawa, S. Anti-carbamylated protein antibodies in rheumatoid arthritis patients of Asian descent. Rheumatology (Oxford) 2015, 54, 1930–1932. [Google Scholar]
- Carubbi, F.; Alunno, A.; Gerli, R.; Giacomelli, R. Post-Translational Modifications of Proteins: Novel Insights in the Autoimmune Response in Rheumatoid Arthritis. Cells 2019, 8, 657. [Google Scholar]
- Verheul, M.K.; van Erp, S.J.; van der Woude, D.; Levarht, E.W.; Mallat, M.J.; Verspaget, H.W.; Stolk, J.; Toes, R.E.; van der Meulen-de Jong, A.E.; Hiemstra, P.S.; et al. Anti-carbamylated protein antibodies: A specific hallmark for rheumatoid arthritis. Comparison to conditions known for enhanced carbamylation; renal failure, smoking and chronic inflammation. Ann. Rheum. Dis. 2016, 75, 1575–1576. [Google Scholar] [CrossRef]
- Van Delft, M.A.M.; van Beest, S.; Kloppenburg, M.; Trouw, L.A.; Ioan-Facsinay, A. Presence of Autoantibodies in Erosive Hand Osteoarthritis and Association with Clinical Presentation. J. Rheumatol. 2019, 46, 101–105. [Google Scholar]
- Ziegelasch, M.; van Delft, M.A.; Wallin, P.; Skogh, T.; Magro-Checa, C.; Steup-Beekman, G.M.; Trouw, L.A.; Kastbom, A.; Sjowall, C. Antibodies against carbamylated proteins and cyclic citrullinated peptides in systemic lupus erythematosus: Results from two well-defined European cohorts. Arthritis Res. Ther. 2016, 18, 289. [Google Scholar] [CrossRef]
- Bell, D.A.; Elhayek, S.; Cairns, E.; Barra, L. Anti-homocitrullinated protein antibody isotype usage in rheumatoid arthritis and their unaffected first-degree relatives. Clin. Exp. Rheumatol. 2017, 35, 948–953. [Google Scholar] [PubMed]
- Muller, P.C.; Anink, J.; Shi, J.; Levarht, E.W.; Reinards, T.H.; Otten, M.H.; van Tol, M.J.; Jol-van der Zijde, C.M.; Brinkman, D.M.; Allaart, C.F.; et al. Anticarbamylated protein (anti-CarP) antibodies are present in sera of juvenile idiopathic arthritis (JIA) patients. Ann. Rheum. Dis. 2013, 72, 2053–2055. [Google Scholar] [CrossRef] [PubMed]
- Van Delft, M.A.M.; Verheul, M.K.; Burgers, L.E.; Derksen, V.; van der Helm-van Mil, A.H.M.; van der Woude, D.; Huizinga, T.W.J.; Toes, R.E.M.; Trouw, L.A. The isotype and IgG subclass distribution of anti-carbamylated protein antibodies in rheumatoid arthritis patients. Arthritis Res. Ther. 2017, 19, 190. [Google Scholar] [CrossRef]
- Challener, G.J.; Jones, J.D.; Pelzek, A.J.; Hamilton, B.J.; Boire, G.; de Brum-Fernandes, A.J.; Masetto, A.; Carrier, N.; Menard, H.A.; Silverman, G.J.; et al. Anti-carbamylated Protein Antibody Levels Correlate with Anti-Sa (Citrullinated Vimentin) Antibody Levels in Rheumatoid Arthritis. J. Rheumatol. 2016, 43, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Frisell, T.; Saevarsdottir, S.; Askling, J. Family history of rheumatoid arthritis: An old concept with new developments. Nat. Rev. Rheumatol. 2016, 12, 335–343. [Google Scholar] [CrossRef] [PubMed]
- MacGregor, A.J.; Bamber, S.; Carthy, D.; Vencovsky, J.; Mageed, R.A.; Ollier, W.E.; Silman, A.J. Heterogeneity of disease phenotype in monozygotic twins concordant for rheumatoid arthritis. Br. J. Rheumatol. 1995, 34, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Mackie, S.L.; Taylor, J.C.; Martin, S.G.; Consortium, Y.; Consortium, U.; Wordsworth, P.; Steer, S.; Wilson, A.G.; Worthington, J.; Emery, P.; et al. A spectrum of susceptibility to rheumatoid arthritis within HLA-DRB1: Stratification by autoantibody status in a large UK population. Genes Immun. 2012, 13, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Balsa, A.; Cabezon, A.; Orozco, G.; Cobo, T.; Miranda-Carus, E.; Lopez-Nevot, M.A.; Vicario, J.L.; Martin-Mola, E.; Martin, J.; Pascual-Salcedo, D. Influence of HLA DRB1 alleles in the susceptibility of rheumatoid arthritis and the regulation of antibodies against citrullinated proteins and rheumatoid factor. Arthritis Res. Ther. 2010, 12, R62. [Google Scholar] [CrossRef]
- Mattey, D.L.; Hassell, A.B.; Dawes, P.T.; Cheung, N.T.; Poulton, K.V.; Thomson, W.; Hajeer, A.H.; Ollier, W.E. Independent association of rheumatoid factor and the HLA-DRB1 shared epitope with radiographic outcome in rheumatoid arthritis. Arthritis Rheum. 2001, 44, 1529–1533. [Google Scholar] [CrossRef]
- Vehe, R.K.; Nepom, G.T.; Wilske, K.R.; Stage, D.; Begovich, A.B.; Nepom, B.S. Erosive rheumatoid factor negative and positive rheumatoid arthritis are immunogenetically similar. J. Rheumatol. 1994, 21, 194–196. [Google Scholar]
- Okada, Y.; Eyre, S.; Suzuki, A.; Kochi, Y.; Yamamoto, K. Genetics of rheumatoid arthritis: 2018 status. Ann. Rheum. Dis. 2019, 78, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Huizinga, T.W.; Amos, C.I.; van der Helm-van Mil, A.H.; Chen, W.; van Gaalen, F.A.; Jawaheer, D.; Schreuder, G.M.; Wener, M.; Breedveld, F.C.; Ahmad, N.; et al. Refining the complex rheumatoid arthritis phenotype based on specificity of the HLA-DRB1 shared epitope for antibodies to citrullinated proteins. Arthritis Rheum. 2005, 52, 3433–3438. [Google Scholar] [CrossRef] [PubMed]
- Kissel, T.; van Schie, K.A.; Hafkenscheid, L.; Lundquist, A.; Kokkonen, H.; Wuhrer, M.; Huizinga, T.W.; Scherer, H.U.; Toes, R.; Rantapaa-Dahlqvist, S. On the presence of HLA-SE alleles and ACPA-IgG variable domain glycosylation in the phase preceding the development of rheumatoid arthritis. Ann. Rheum. Dis. 2019, 78, 1616–1620. [Google Scholar] [PubMed]
- Plenge, R.M.; Padyukov, L.; Remmers, E.F.; Purcell, S.; Lee, A.T.; Karlson, E.W.; Wolfe, F.; Kastner, D.L.; Alfredsson, L.; Altshuler, D.; et al. Replication of putative candidate-gene associations with rheumatoid arthritis in >4000 samples from North America and Sweden: Association of susceptibility with PTPN22, CTLA4, and PADI4. Am. J. Hum. Genet. 2005, 77, 1044–1060. [Google Scholar] [CrossRef] [PubMed]
- Karami, J.; Aslani, S.; Jamshidi, A.; Garshasbi, M.; Mahmoudi, M. Genetic implications in the pathogenesis of rheumatoid arthritis; an updated review. Gene 2019, 702, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.P.; Zhu, B.Q.; Tao, S.S.; Fan, Y.G.; Li, X.M.; Pan, H.F.; Ye, D.Q. Long Non-coding RNAs Genes Polymorphisms and Their Expression Levels in Patients With Rheumatoid Arthritis. Front. Immunol. 2019, 10, 2529. [Google Scholar] [CrossRef] [PubMed]
- Verpoort, K.N.; van Gaalen, F.A.; van der Helm-van Mil, A.H.; Schreuder, G.M.; Breedveld, F.C.; Huizinga, T.W.; de Vries, R.R.; Toes, R.E. Association of HLA-DR3 with anti-cyclic citrullinated peptide antibody-negative rheumatoid arthritis. Arthritis Rheum. 2005, 52, 3058–3062. [Google Scholar] [CrossRef]
- Regueiro, C.; Rodriguez-Rodriguez, L.; Triguero-Martinez, A.; Nuno, L.; Castano-Nunez, A.L.; Villalva, A.; Perez-Pampin, E.; Lopez-Golan, Y.; Abasolo, L.; Ortiz, A.M.; et al. Specific Association of HLA-DRB1*03 With Anti-Carbamylated Protein Antibodies in Patients with Rheumatoid Arthritis. Arthritis Rheumatol. 2019, 71, 331–339. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Terao, C. The Impact of Cigarette Smoking on Risk of Rheumatoid Arthritis: A Narrative Review. Cells 2020, 9, 475. [Google Scholar] [CrossRef]
- Kallberg, H.; Ding, B.; Padyukov, L.; Bengtsson, C.; Ronnelid, J.; Klareskog, L.; Alfredsson, L.; Group, E.S. Smoking is a major preventable risk factor for rheumatoid arthritis: Estimations of risks after various exposures to cigarette smoke. Ann. Rheum. Dis. 2011, 70, 508–511. [Google Scholar] [CrossRef]
- Arnson, Y.; Shoenfeld, Y.; Amital, H. Effects of tobacco smoke on immunity, inflammation and autoimmunity. J. Autoimmun. 2010, 34, J258–J265. [Google Scholar] [CrossRef] [PubMed]
- Hensvold, A.H.; Magnusson, P.K.; Joshua, V.; Hansson, M.; Israelsson, L.; Ferreira, R.; Jakobsson, P.J.; Holmdahl, R.; Hammarstrom, L.; Malmstrom, V.; et al. Environmental and genetic factors in the development of anticitrullinated protein antibodies (ACPAs) and ACPA-positive rheumatoid arthritis: An epidemiological investigation in twins. Ann. Rheum. Dis. 2015, 74, 375–380. [Google Scholar] [CrossRef]
- Klareskog, L.; Malmstrom, V.; Lundberg, K.; Padyukov, L.; Alfredsson, L. Smoking, citrullination and genetic variability in the immunopathogenesis of rheumatoid arthritis. Semin. Immunol. 2011, 23, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Makrygiannakis, D.; Hermansson, M.; Ulfgren, A.K.; Nicholas, A.P.; Zendman, A.J.; Eklund, A.; Grunewald, J.; Skold, C.M.; Klareskog, L.; Catrina, A.I. Smoking increases peptidylarginine deiminase 2 enzyme expression in human lungs and increases citrullination in BAL cells. Ann. Rheum. Dis. 2008, 67, 1488–1492. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, Y.; Ikari, K.; Hashimoto, M.; Ohmura, K.; Tanaka, M.; Ito, H.; Taniguchi, A.; Yamanaka, H.; Mimori, T.; Terao, C. Shared epitope defines distinct associations of cigarette smoking with levels of anticitrullinated protein antibody and rheumatoid factor. Ann. Rheum. Dis. 2019, 78, 1480–1487. [Google Scholar] [CrossRef]
- Wang, Z.; Nicholls, S.J.; Rodriguez, E.R.; Kummu, O.; Horkko, S.; Barnard, J.; Reynolds, W.F.; Topol, E.J.; DiDonato, J.A.; Hazen, S.L. Protein carbamylation links inflammation, smoking, uremia and atherogenesis. Nat. Med. 2007, 13, 1176–1184. [Google Scholar] [CrossRef]
- Shmerling, R.H.; Delbanco, T.L. The rheumatoid factor: An analysis of clinical utility. Am. J. Med. 1991, 91, 528–534. [Google Scholar] [CrossRef]
- Ingegnoli, F.; Castelli, R.; Gualtierotti, R. Rheumatoid factors: Clinical applications. Dis. Markers 2013, 35, 727–734. [Google Scholar] [CrossRef]
- Konig, M.F.; Andrade, F. A Critical Reappraisal of Neutrophil Extracellular Traps and NETosis Mimics Based on Differential Requirements for Protein Citrullination. Front. Immunol. 2016, 7, 461. [Google Scholar] [CrossRef]
- Konig, M.F.; Abusleme, L.; Reinholdt, J.; Palmer, R.J.; Teles, R.P.; Sampson, K.; Rosen, A.; Nigrovic, P.A.; Sokolove, J.; Giles, J.T.; et al. Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci. Transl. Med. 2016, 8, 369ra176. [Google Scholar] [CrossRef]
- Mukherjee, A.; Jantsch, V.; Khan, R.; Hartung, W.; Fischer, R.; Jantsch, J.; Ehrenstein, B.; Konig, M.F.; Andrade, F. Rheumatoid Arthritis-Associated Autoimmunity Due to Aggregatibacter actinomycetemcomitans and Its Resolution with Antibiotic Therapy. Front. Immunol. 2018, 9, 2352. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Banuelos, E.; Mukherjee, A.; Darrah, E.; Andrade, F. Rheumatoid Arthritis-Associated Mechanisms of Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans. J. Clin. Med. 2019, 8, 1309. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, D.; Jia, H.; Feng, Q.; Wang, D.; Liang, D.; Wu, X.; Li, J.; Tang, L.; Li, Y.; et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015, 21, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wright, K.; Davis, J.M.; Jeraldo, P.; Marietta, E.V.; Murray, J.; Nelson, H.; Matteson, E.L.; Taneja, V. An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis. Genome Med. 2016, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Kurakawa, T.; Umemoto, E.; Motooka, D.; Ito, Y.; Gotoh, K.; Hirota, K.; Matsushita, M.; Furuta, Y.; Narazaki, M.; et al. Dysbiosis Contributes to Arthritis Development via Activation of Autoreactive T Cells in the Intestine. Arthritis Rheumatol. 2016, 68, 2646–2661. [Google Scholar] [CrossRef]
- Courbon, G.; Rinaudo-Gaujous, M.; Blasco-Baque, V.; Auger, I.; Caire, R.; Mijola, L.; Vico, L.; Paul, S.; Marotte, H. Porphyromonas gingivalis experimentally induces periodontis and an anti-CCP2-associated arthritis in the rat. Ann. Rheum. Dis. 2019, 78, 594–599. [Google Scholar] [CrossRef]
- Rantapaa Dahlqvist, S.; Andrade, F. Individuals at risk of seropositive rheumatoid arthritis: The evolving story. J. Intern. Med. 2019, 286, 627–643. [Google Scholar] [CrossRef]
- Turk, S.A.; van Beers-Tas, M.H.; van Schaardenburg, D. Prediction of future rheumatoid arthritis. Rheum. Dis. Clin. N. Am. 2014, 40, 753–770. [Google Scholar] [CrossRef]
- Deane, K.D.; Holers, V.M. Rheumatoid Arthritis: Pathogenesis, Prediction and Prevention–An Emerging Paradigm Shift. Arthritis Rheumatol. 2020. [Google Scholar] [CrossRef]
- Nielsen, S.F.; Bojesen, S.E.; Schnohr, P.; Nordestgaard, B.G. Elevated rheumatoid factor and long term risk of rheumatoid arthritis: A prospective cohort study. BMJ 2012, 345, e5244. [Google Scholar] [CrossRef]
- Rantapaa-Dahlqvist, S. What happens before the onset of rheumatoid arthritis? Curr. Opin. Rheumatol. 2009, 21, 272–278. [Google Scholar] [PubMed]
- Deane, K.D.; Norris, J.M.; Holers, V.M. Preclinical rheumatoid arthritis: Identification, evaluation, and future directions for investigation. Rheum. Dis. Clin. N. Am. 2010, 36, 213–241. [Google Scholar] [CrossRef] [PubMed]
- Deane, K.D.; O’Donnell, C.I.; Hueber, W.; Majka, D.S.; Lazar, A.A.; Derber, L.A.; Gilliland, W.R.; Edison, J.D.; Norris, J.M.; Robinson, W.H.; et al. The number of elevated cytokines and chemokines in preclinical seropositive rheumatoid arthritis predicts time to diagnosis in an age-dependent manner. Arthritis Rheum. 2010, 62, 3161–3172. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.; Liao, K.; Nair, R.; Ringold, S.; Costenbader, K.H. Anti-citrullinated peptide antibody assays and their role in the diagnosis of rheumatoid arthritis. Arthritis Rheum. 2009, 61, 1472–1483. [Google Scholar] [CrossRef] [PubMed]
- Nielen, M.M.; van Schaardenburg, D.; Reesink, H.W.; van de Stadt, R.J.; van der Horst-Bruinsma, I.E.; de Koning, M.H.; Habibuw, M.R.; Vandenbroucke, J.P.; Dijkmans, B.A. Specific autoantibodies precede the symptoms of rheumatoid arthritis: A study of serial measurements in blood donors. Arthritis Rheum. 2004, 50, 380–386. [Google Scholar] [CrossRef]
- Rantapaa-Dahlqvist, S.; de Jong, B.A.; Berglin, E.; Hallmans, G.; Wadell, G.; Stenlund, H.; Sundin, U.; van Venrooij, W.J. Antibodies against cyclic citrullinated peptide and IgA rheumatoid factor predict the development of rheumatoid arthritis. Arthritis Rheum. 2003, 48, 2741–2749. [Google Scholar]
- Bos, W.H.; Wolbink, G.J.; Boers, M.; Tijhuis, G.J.; de Vries, N.; van der Horst-Bruinsma, I.E.; Tak, P.P.; van de Stadt, R.J.; van der Laken, C.J.; Dijkmans, B.A.; et al. Arthritis development in patients with arthralgia is strongly associated with anti-citrullinated protein antibody status: A prospective cohort study. Ann. Rheum. Dis. 2010, 69, 490–494. [Google Scholar] [CrossRef]
- Van de Stadt, L.A.; Witte, B.I.; Bos, W.H.; van Schaardenburg, D. A prediction rule for the development of arthritis in seropositive arthralgia patients. Ann. Rheum. Dis. 2013, 72, 1920–1926. [Google Scholar] [CrossRef]
- Ten Brinck, R.M.; van Steenbergen, H.W.; van Delft, M.A.M.; Verheul, M.K.; Toes, R.E.M.; Trouw, L.A.; van der Helm-van Mil, A.H.M. The risk of individual autoantibodies, autoantibody combinations and levels for arthritis development in clinically suspect arthralgia. Rheumatology (Oxford) 2017, 56, 2145–2153. [Google Scholar]
- Rakieh, C.; Nam, J.L.; Hunt, L.; Hensor, E.M.; Das, S.; Bissell, L.A.; Villeneuve, E.; McGonagle, D.; Hodgson, R.; Grainger, A.; et al. Predicting the development of clinical arthritis in anti-CCP positive individuals with non-specific musculoskeletal symptoms: A prospective observational cohort study. Ann. Rheum. Dis. 2015, 74, 1659–1666. [Google Scholar]
- Ramos-Remus, C.; Castillo-Ortiz, J.D.; Aguilar-Lozano, L.; Padilla-Ibarra, J.; Sandoval-Castro, C.; Vargas-Serafin, C.O.; de la Mora-Molina, H.; Ramos-Gomez, A.; Sanchez-Ortiz, A.; Avila-Armengol, H.; et al. Autoantibodies in prediction of the development of rheumatoid arthritis among healthy relatives of patients with the disease. Arthritis Rheumatol. 2015, 67, 2837–2844. [Google Scholar] [CrossRef] [PubMed]
- Nielen, M.M.; van der Horst, A.R.; van Schaardenburg, D.; van der Horst-Bruinsma, I.E.; van de Stadt, R.J.; Aarden, L.; Dijkmans, B.A.; Hamann, D. Antibodies to citrullinated human fibrinogen (ACF) have diagnostic and prognostic value in early arthritis. Ann. Rheum. Dis. 2005, 64, 1199–1204. [Google Scholar] [CrossRef] [PubMed]
- van der Woude, D.; Rantapaa-Dahlqvist, S.; Ioan-Facsinay, A.; Onnekink, C.; Schwarte, C.M.; Verpoort, K.N.; Drijfhout, J.W.; Huizinga, T.W.; Toes, R.E.; Pruijn, G.J. Epitope spreading of the anti-citrullinated protein antibody response occurs before disease onset and is associated with the disease course of early arthritis. Ann. Rheum. Dis. 2010, 69, 1554–1561. [Google Scholar]
- Van de Stadt, L.A.; van der Horst, A.R.; de Koning, M.H.; Bos, W.H.; Wolbink, G.J.; van de Stadt, R.J.; Pruijn, G.J.; Dijkmans, B.A.; van Schaardenburg, D.; Hamann, D. The extent of the anti-citrullinated protein antibody repertoire is associated with arthritis development in patients with seropositive arthralgia. Ann. Rheum. Dis. 2011, 70, 128–133. [Google Scholar] [CrossRef]
- Janssen, K.M.; Westra, J.; Chalan, P.; Boots, A.M.; de Smit, M.J.; van Winkelhoff, A.J.; Vissink, A.; Brouwer, E. Regulatory CD4+ T-Cell Subsets and Anti-Citrullinated Protein Antibody Repertoire: Potential Biomarkers for Arthritis Development in Seropositive Arthralgia Patients? PLoS ONE 2016, 11, e0162101. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; van de Stadt, L.A.; Levarht, E.W.; Huizinga, T.W.; Toes, R.E.; Trouw, L.A.; van Schaardenburg, D. Anti-carbamylated protein antibodies are present in arthralgia patients and predict the development of rheumatoid arthritis. Arthritis Rheum. 2013, 65, 911–915. [Google Scholar] [CrossRef]
- Stoop, J.N.; Liu, B.S.; Shi, J.; Jansen, D.T.; Hegen, M.; Huizinga, T.W.; Trouw, L.A.; Toes, R.E. Antibodies specific for carbamylated proteins precede the onset of clinical symptoms in mice with collagen induced arthritis. PLoS ONE 2014, 9, e102163. [Google Scholar] [CrossRef]
- Brink, M.; Verheul, M.K.; Ronnelid, J.; Berglin, E.; Holmdahl, R.; Toes, R.E.; Klareskog, L.; Trouw, L.A.; Rantapaa-Dahlqvist, S. Anti-carbamylated protein antibodies in the pre-symptomatic phase of rheumatoid arthritis, their relationship with multiple anti-citrulline peptide antibodies and association with radiological damage. Arthritis Res. Ther. 2015, 17, 25. [Google Scholar] [CrossRef]
- Gan, R.W.; Trouw, L.A.; Shi, J.; Toes, R.E.; Huizinga, T.W.; Demoruelle, M.K.; Kolfenbach, J.R.; Zerbe, G.O.; Deane, K.D.; Edison, J.D.; et al. Anti-carbamylated protein antibodies are present prior to rheumatoid arthritis and are associated with its future diagnosis. J. Rheumatol. 2015, 42, 572–579. [Google Scholar] [CrossRef]
- Verheul, M.K.; Fearon, U.; Trouw, L.A.; Veale, D.J. Biomarkers for rheumatoid and psoriatic arthritis. Clin. Immunol. 2015, 161, 2–10. [Google Scholar] [CrossRef]
- Pecani, A.; Alessandri, C.; Spinelli, F.R.; Priori, R.; Riccieri, V.; Di Franco, M.; Ceccarelli, F.; Colasanti, T.; Pendolino, M.; Mancini, R.; et al. Prevalence, sensitivity and specificity of antibodies against carbamylated proteins in a monocentric cohort of patients with rheumatoid arthritis and other autoimmune rheumatic diseases. Arthritis Res. Ther. 2016, 18, 276. [Google Scholar] [CrossRef] [PubMed]
- Rantapaa-Dahlqvist, S. Diagnostic and prognostic significance of autoantibodies in early rheumatoid arthritis. Scand. J. Rheumatol. 2005, 34, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; van Steenbergen, H.W.; van Nies, J.A.; Levarht, E.W.; Huizinga, T.W.; van der Helm-van Mil, A.H.; Toes, R.E.; Trouw, L.A. The specificity of anti-carbamylated protein antibodies for rheumatoid arthritis in a setting of early arthritis. Arthritis Res. Ther. 2015, 17, 339. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, C.; Nuno, L.; Ortiz, A.M.; Peiteado, D.; Villalba, A.; Pascual-Salcedo, D.; Martinez-Feito, A.; Gonzalez-Alvaro, I.; Balsa, A.; Gonzalez, A. Value of Measuring Anti-Carbamylated Protein Antibodies for Classification on Early Arthritis Patients. Sci. Rep. 2017, 7, 12023. [Google Scholar] [CrossRef]
- Jonsson, T.; Steinsson, K.; Jonsson, H.; Geirsson, A.J.; Thorsteinsson, J.; Valdimarsson, H. Combined elevation of IgM and IgA rheumatoid factor has high diagnostic specificity for rheumatoid arthritis. Rheumatol. Int. 1998, 18, 119–122. [Google Scholar] [CrossRef]
- Bluett, J.; Sergeant, J.C.; MacGregor, A.J.; Chipping, J.R.; Marshall, T.; Symmons, D.P.M.; Verstappen, S.M.M. Risk factors for oral methotrexate failure in patients with inflammatory polyarthritis: Results from a UK prospective cohort study. Arthritis Res. Ther. 2018, 20, 50. [Google Scholar] [CrossRef]
- Tillmann, T.; Krishnadas, R.; Cavanagh, J.; Petrides, K.V. Possible rheumatoid arthritis subtypes in terms of rheumatoid factor, depression, diagnostic delay and emotional expression: An exploratory case-control study. Arthritis Res. Ther. 2013, 15, R45. [Google Scholar] [CrossRef]
- Barra, L.; Bykerk, V.; Pope, J.E.; Haraoui, B.P.; Hitchon, C.A.; Thorne, J.C.; Keystone, E.C.; Boire, G.; Catch, I. Anticitrullinated protein antibodies and rheumatoid factor fluctuate in early inflammatory arthritis and do not predict clinical outcomes. J. Rheumatol. 2013, 40, 1259–1267. [Google Scholar] [CrossRef]
- Isaacs, J.D.; Cohen, S.B.; Emery, P.; Tak, P.P.; Wang, J.; Lei, G.; Williams, S.; Lal, P.; Read, S.J. Effect of baseline rheumatoid factor and anticitrullinated peptide antibody serotype on rituximab clinical response: A meta-analysis. Ann. Rheum. Dis. 2013, 72, 329–336. [Google Scholar] [CrossRef]
- Maneiro, R.J.; Salgado, E.; Carmona, L.; Gomez-Reino, J.J. Rheumatoid factor as predictor of response to abatacept, rituximab and tocilizumab in rheumatoid arthritis: Systematic review and meta-analysis. Semin. Arthritis Rheum. 2013, 43, 9–17. [Google Scholar] [CrossRef]
- Smolen, J.S.; Landewe, R.B.M.; Bijlsma, J.W.J.; Burmester, G.R.; Dougados, M.; Kerschbaumer, A.; McInnes, I.B.; Sepriano, A.; van Vollenhoven, R.F.; de Wit, M.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann. Rheum. Dis. 2020, 79, 685–699. [Google Scholar] [CrossRef] [PubMed]
- Goekoop-Ruiterman, Y.P.; de Vries-Bouwstra, J.K.; Allaart, C.F.; van Zeben, D.; Kerstens, P.J.; Hazes, J.M.; Zwinderman, A.H.; Ronday, H.K.; Han, K.H.; Westedt, M.L.; et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): A randomized, controlled trial. Arthritis. Rheum. 2005, 52, 3381–3390. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, J.S.; Bergstra, S.A.; Chopra, A.; Tikly, M.; Fonseca, J.E.; Salomon-Escoto, K.; Huizinga, T.W.J.; van der Woude, D. Autoantibody status is not associated with early treatment response to first-line methotrexate in patients with early rheumatoid arthritis. Rheumatology (Oxford) 2019, 58, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, M.K.; Hensvold, A.H.; Hansson, M.; Aga, A.B.; Sexton, J.; Mathsson-Alm, L.; Cornillet, M.; Serre, G.; Lillegraven, S.; Fevang, B.S.; et al. The role of anti-citrullinated protein antibody reactivities in an inception cohort of patients with rheumatoid arthritis receiving treat-to-target therapy. Arthritis Res. Ther. 2018, 20, 146. [Google Scholar] [CrossRef]
- Wevers-de Boer, K.; Visser, K.; Heimans, L.; Ronday, H.K.; Molenaar, E.; Groenendael, J.H.; Peeters, A.J.; Westedt, M.L.; Collee, G.; de Sonnaville, P.B.; et al. Remission induction therapy with methotrexate and prednisone in patients with early rheumatoid and undifferentiated arthritis (the IMPROVED study). Ann. Rheum Dis. 2012, 71, 1472–1477. [Google Scholar] [CrossRef]
- Nordberg, L.B.; Lillegraven, S.; Aga, A.B.; Sexton, J.; Olsen, I.C.; Lie, E.; Berner Hammer, H.; Uhlig, T.; van der Heijde, D.; Kvien, T.K.; et al. Comparing the disease course of patients with seronegative and seropositive rheumatoid arthritis fulfilling the 2010 ACR/EULAR classification criteria in a treat-to-target setting: 2-year data from the ARCTIC trial. RMD Open 2018, 4, e000752. [Google Scholar] [CrossRef]
- Seegobin, S.D.; Ma, M.H.; Dahanayake, C.; Cope, A.P.; Scott, D.L.; Lewis, C.M.; Scott, I.C. ACPA-positive and ACPA-negative rheumatoid arthritis differ in their requirements for combination DMARDs and corticosteroids: Secondary analysis of a randomized controlled trial. Arthritis Res. Ther. 2014, 16, R13. [Google Scholar] [CrossRef]
- Courvoisier, D.S.; Chatzidionysiou, K.; Mongin, D.; Lauper, K.; Mariette, X.; Morel, J.; Gottenberg, J.E.; Bergstra, S.A.; Suarez, M.P.; Codreanu, C.; et al. The impact of seropositivity on the effectiveness of biologic anti-rheumatic agents: Results from a collaboration of 16 registries. Rheumatology (Oxford) 2020, keaa393. [Google Scholar] [CrossRef]
- Bugatti, S.; Manzo, A.; Montecucco, C.; Caporali, R. The Clinical Value of Autoantibodies in Rheumatoid Arthritis. Front. Med. (Lausanne) 2018, 5, 339. [Google Scholar] [CrossRef]
- Lorenzetti, R.; Janowska, I.; Smulski, C.R.; Frede, N.; Henneberger, N.; Walter, L.; Schleyer, M.T.; Huppe, J.M.; Staniek, J.; Salzer, U.; et al. Abatacept modulates CD80 and CD86 expression and memory formation in human B-cells. J. Autoimmun. 2019, 101, 145–152. [Google Scholar] [CrossRef]
- Bohler, C.; Radner, H.; Smolen, J.S.; Aletaha, D. Serological changes in the course of traditional and biological disease modifying therapy of rheumatoid arthritis. Ann. Rheum. Dis. 2013, 72, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Cambridge, G.; Perry, H.C.; Nogueira, L.; Serre, G.; Parsons, H.M.; De La Torre, I.; Dickson, M.C.; Leandro, M.J.; Edwards, J.C. The effect of B-cell depletion therapy on serological evidence of B-cell and plasmablast activation in patients with rheumatoid arthritis over multiple cycles of rituximab treatment. J. Autoimmun. 2014, 50, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Alivernini, S.; Galeazzi, M.; Peleg, H.; Tolusso, B.; Gremese, E.; Ferraccioli, G.; Naparstek, Y. Is ACPA positivity the main driver for rheumatoid arthritis treatment? Pros and cons. Autoimmun. Rev. 2017, 16, 1096–1102. [Google Scholar] [CrossRef] [PubMed]
- Bird, P.; Hall, S.; Nash, P.; Connell, C.A.; Kwok, K.; Witcombe, D.; Thirunavukkarasu, K. Treatment outcomes in patients with seropositive versus seronegative rheumatoid arthritis in Phase III randomised clinical trials of tofacitinib. RMD Open 2019, 5, e000742. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.H.; Verheul, M.K.; Barton, A.; MacGregor, A.J.; Lunt, M.; Toes, R.E.; Symmons, D.P.; Trouw, L.A.; Verstappen, S.M. Anticarbamylated protein antibodies are associated with long-term disability and increased disease activity in patients with early inflammatory arthritis: Results from the Norfolk Arthritis Register. Ann. Rheum. Dis. 2016, 75, 1139–1144. [Google Scholar] [CrossRef]
- Sidiras, P.; Spruyt, D.; Gangji, V.; Imbault, V.; Sokolova, T.; Durez, P.; Communi, D.; Rasschaert, J.; Badot, V. Antibodies against carbamylated proteins: Prevalence and associated disease characteristics in Belgian patients with rheumatoid arthritis or other rheumatic diseases. Scand. J. Rheumatol. 2020, 1–6. [Google Scholar] [CrossRef]
- Kumar, R.; Piantoni, S.; Boldini, M.; Garrafa, E.; Bazzani, C.; Fredi, M.; Ottaviani, R.; Cavazzana, I.; Tincani, A.; Franceschini, F. Anti-carbamylated protein antibodies as a clinical response predictor in rheumatoid arthritis patients treated with abatacept. Clin. Exp. Rheumatol. 2020. Online ahead of print. [Google Scholar]
- Greenblatt, H.K.; Kim, H.A.; Bettner, L.F.; Deane, K.D. Preclinical rheumatoid arthritis and rheumatoid arthritis prevention. Curr. Opin. Rheumatol. 2020, 32, 289–296. [Google Scholar] [CrossRef]
- Mahler, M.; Martinez-Prat, L.; Sparks, J.A.; Deane, K.D. Precision medicine in the care of rheumatoid arthritis: Focus on prediction and prevention of future clinically-apparent disease. Autoimmun. Rev. 2020, 19, 102506. [Google Scholar] [CrossRef]
- Hilliquin, S.; Hugues, B.; Mitrovic, S.; Gossec, L.; Fautrel, B. Ability of disease-modifying antirheumatic drugs to prevent or delay rheumatoid arthritis onset: A systematic literature review and meta-analysis. Ann. Rheum. Dis. 2018, 77, 1099–1106. [Google Scholar] [CrossRef]
- Finckh, A.; Escher, M.; Liang, M.H.; Bansback, N. Preventive Treatments for Rheumatoid Arthritis: Issues Regarding Patient Preferences. Curr. Rheumatol. Rep. 2016, 18, 51. [Google Scholar] [CrossRef] [PubMed]
- Emery, P.; Durez, P.; Dougados, M.; Legerton, C.W.; Becker, J.C.; Vratsanos, G.; Genant, H.K.; Peterfy, C.; Mitra, P.; Overfield, S.; et al. Impact of T-cell costimulation modulation in patients with undifferentiated inflammatory arthritis or very early rheumatoid arthritis: A clinical and imaging study of abatacept (the ADJUST trial). Ann. Rheum. Dis. 2010, 69, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Roelsgaard, I.K.; Thomsen, T.; Ostergaard, M.; Christensen, R.; Hetland, M.L.; Jacobsen, S.; Andersen, L.; Tonnesen, H.; Rollefstad, S.; Semb, A.G.; et al. The effect of an intensive smoking cessation intervention on disease activity in patients with rheumatoid arthritis: Study protocol for a randomised controlled trial. Trials 2017, 18, 570. [Google Scholar] [CrossRef]
- Roelsgaard, I.K.; Esbensen, B.A.; Ostergaard, M.; Rollefstad, S.; Semb, A.G.; Christensen, R.; Thomsen, T. Smoking cessation intervention for reducing disease activity in chronic autoimmune inflammatory joint diseases. Cochrane Database Syst. Rev. 2019, 9, CD012958. [Google Scholar] [CrossRef] [PubMed]
- Malm, K.; Bergman, S.; Bremander, A.; Larsson, I.; Andersson, M.L.E.; Group, B.S. Discussions of lifestyle habits as an integral part of care management: A cross-sectional cohort study in patients with established rheumatoid arthritis in Sweden. Rheumatol. Adv. Pract. 2019, 3, rkz039. [Google Scholar] [CrossRef]
| Rheumatoid Factor | Anti-Citrullinated Protein Antibodies | Anti-Carbamylated Protein Antibodies | |
|---|---|---|---|
| Year of Discovery | 1940s | 1964 | 2011 |
| Characteristics of the Autoantibody | |||
| Antigenic Targets |
|
|
|
| Isotypes |
|
|
|
| Characteristics |
|
|
|
| Clinical Significance | |||
| Role in RA Diagnosis |
|
|
|
| Sensitivity for RA |
|
|
|
| Specificity for RA |
|
|
|
| Other Conditions with Increased Antibody Titer |
|
|
|
| Clinical Presentations |
|
|
|
| Treatment Response |
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-Y.; Yang, H.-Y.; Luo, S.-F.; Lai, J.-H. From Rheumatoid Factor to Anti-Citrullinated Protein Antibodies and Anti-Carbamylated Protein Antibodies for Diagnosis and Prognosis Prediction in Patients with Rheumatoid Arthritis. Int. J. Mol. Sci. 2021, 22, 686. https://doi.org/10.3390/ijms22020686
Wu C-Y, Yang H-Y, Luo S-F, Lai J-H. From Rheumatoid Factor to Anti-Citrullinated Protein Antibodies and Anti-Carbamylated Protein Antibodies for Diagnosis and Prognosis Prediction in Patients with Rheumatoid Arthritis. International Journal of Molecular Sciences. 2021; 22(2):686. https://doi.org/10.3390/ijms22020686
Chicago/Turabian StyleWu, Chao-Yi, Huang-Yu Yang, Shue-Fen Luo, and Jenn-Haung Lai. 2021. "From Rheumatoid Factor to Anti-Citrullinated Protein Antibodies and Anti-Carbamylated Protein Antibodies for Diagnosis and Prognosis Prediction in Patients with Rheumatoid Arthritis" International Journal of Molecular Sciences 22, no. 2: 686. https://doi.org/10.3390/ijms22020686
APA StyleWu, C.-Y., Yang, H.-Y., Luo, S.-F., & Lai, J.-H. (2021). From Rheumatoid Factor to Anti-Citrullinated Protein Antibodies and Anti-Carbamylated Protein Antibodies for Diagnosis and Prognosis Prediction in Patients with Rheumatoid Arthritis. International Journal of Molecular Sciences, 22(2), 686. https://doi.org/10.3390/ijms22020686





