Iron Complexes of Flavonoids-Antioxidant Capacity and Beyond
Abstract
:1. Introduction
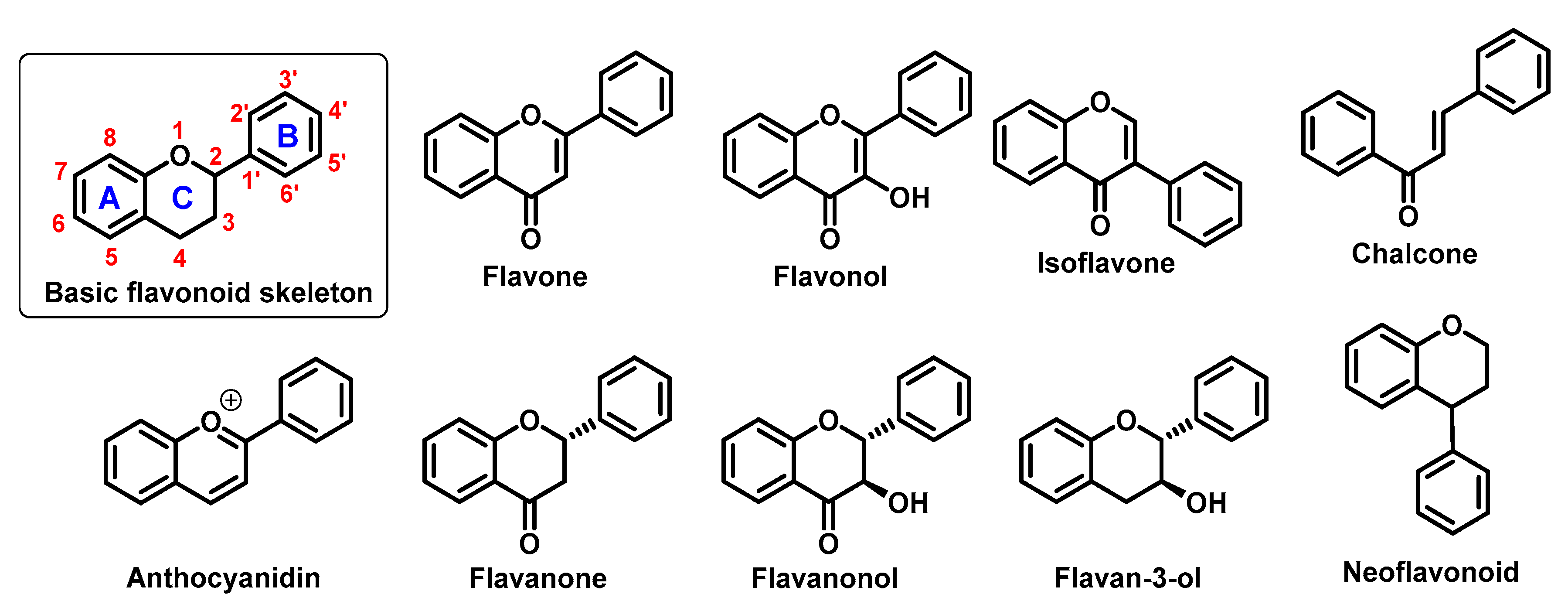
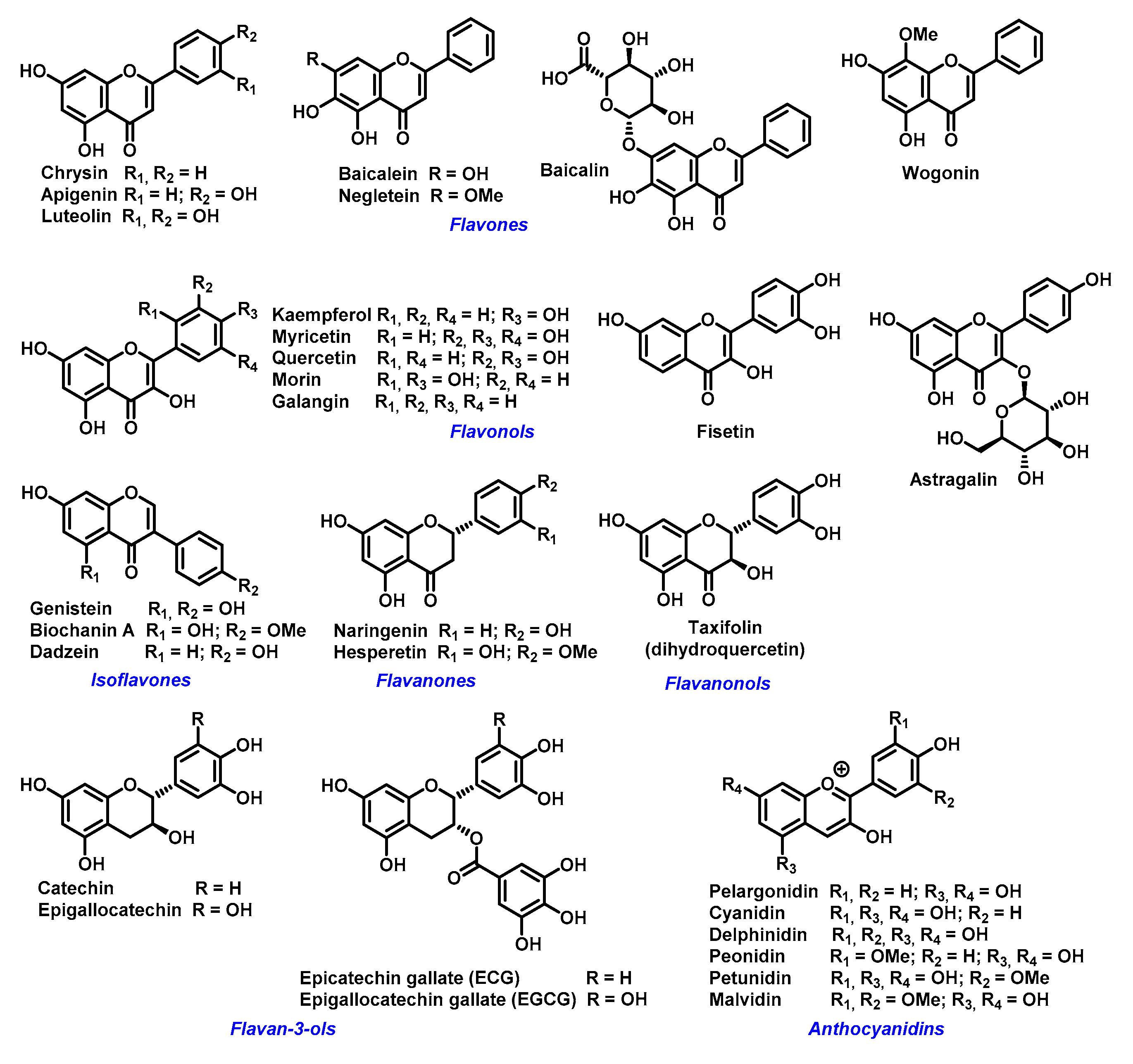
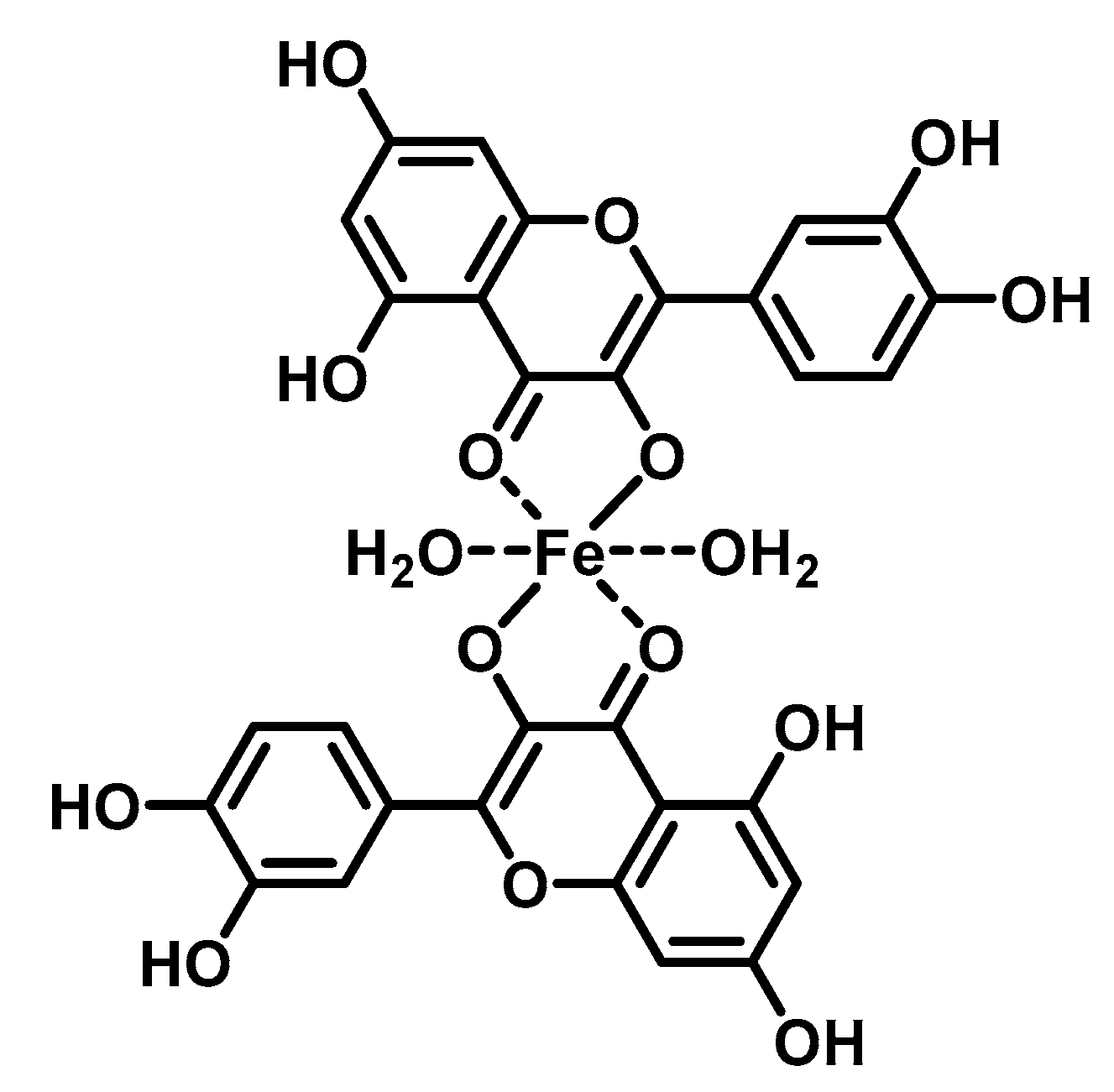
2. Chelating Properties of Flavonoids
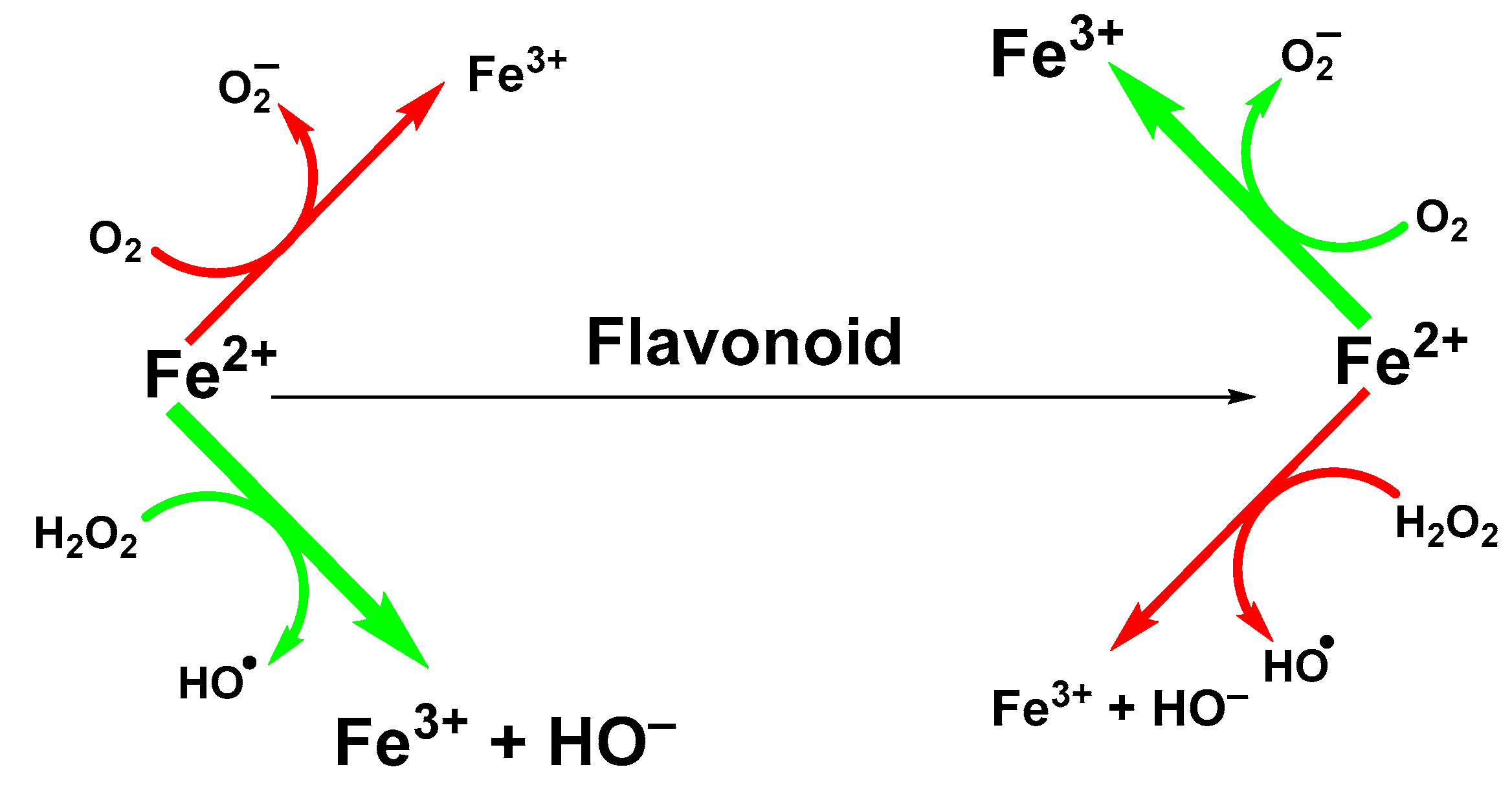
Influence of Chelated Iron Ions on the Antioxidative Effect of Flavonoids
3. Flavonoid Metallocomplexes in the Living Systems and Biological Effect of Their Interactions with Iron Ions
3.1. Influence of Flavonoids on Iron Dependent Enzymes
3.2. Influence of Flavonoids on the Heme Enzymes
4. Future Direction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 3-HF | 3-Hydroxyflavone |
| 4″-DHF | 3″,4″-Dihydroxyflavone |
| A549 | Human non-small cell lung carcinoma cell line |
| ATP | Adenosine tri-phosphate |
| cAMP | Cyclic adenosine monophosphate |
| DMSO | Dimethylsulfoxide |
| DMT1 | Divalent metal transporter 1 |
| EGCG | Epigallocatechin gallate |
| ECG | Epicatechin gallate |
| GLUT1 | Glucose transporter 1 |
| HeLa | Human cervical cancer cell line |
| H295R | Human adrenocortical cell line |
| HepG2 | Hepatoma G2 cell line |
| HIF-1α | Hypoxia-inducible factor 1-alpha |
| K | Binding constant |
| MDA-MB-231 | Human breast cancer cell line |
| MDA-MB-468 | Human breast cancer cell line |
| MAPK | Mitogen-activated protein kinase |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| PHD2 | HIF-prolyl hydroxylase |
| RKO | Human colon carcinoma cell line |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
| SMMC-7721 | Human hepatocellular cell line |
| St | Stoichiometry |
| TBARS | Thiobarbituric acid reactive substances |
| TfR1 | Transferrin receptor protein 1 |
| UV-Vis | UltraViolet-Visible Spectroscopy |
| y-CD | gamma-Cyclodextrin |
References
- Mathesius, U. Flavonoid Functions in Plants and Their Interactions with Other Organisms. Plants 2018, 7, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, K.; Luo, M.; Wei, S. The Bioprotective Effects of Polyphenols on Metabolic Syndrome against Oxidative Stress: Evidences and Perspectives. Oxid. Med. Cell. Longev. 2019, 2019, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belwal, T.; Singh, G.; Jeandet, P.; Pandey, A.; Giri, L.; Ramola, S.; Bhatt, I.D.; Venskutonis, P.R.; Georgiev, M.I.; Clément, C.; et al. Anthocyanins, multi-functional natural products of industrial relevance: Recent biotechnological advances. Biotechnol. Adv. 2020, 43, 107600. [Google Scholar] [CrossRef]
- Grosso, G.; Godos, J.; Lamuela-Raventos, R.; Ray, S.; Micek, A.; Pajak, A.; Sciacca, S.; D’Orazio, N.; Del Rio, D.; Galvano, F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food Res. 2016, 61. [Google Scholar] [CrossRef]
- Manish, P.; Wei Ling, L.; Seong Lin, T.; Mohamad Fairuz, Y. Flavonoids and its Neuroprotective Effects on Brain Ischemia and Neurodegenerative Diseases. Curr. Drug Targets 2018, 19, 1710–1720. [Google Scholar] [CrossRef]
- Kim, Y.; Je, Y. Flavonoid intake and mortality from cardiovascular disease and all causes: A meta-analysis of prospective cohort studies. Clin. Nutr. ESPEN 2017, 20, 68–77. [Google Scholar] [CrossRef]
- Mirossay, L.; Varinská, L.; Mojzis, J. Antiangiogenic Effect of Flavonoids and Chalcones: An Update. Int. J. Mol. Sci. 2017, 19, 27. [Google Scholar] [CrossRef] [Green Version]
- Luna-Vital, D.A.; De Mejia, E.G. Anthocyanins from purple corn activate free fatty acid-receptor 1 and glucokinase enhancing in vitro insulin secretion and hepatic glucose uptake. PLoS ONE 2018, 13, e0200449. [Google Scholar] [CrossRef] [Green Version]
- Sivamaruthi, B.S.; Kesika, P.; Chaiyasut, C. The Influence of Supplementation of Anthocyanins on Obesity-Associated Comorbidities: A Concise Review. Foods 2020, 9, 687. [Google Scholar] [CrossRef]
- Ciumărnean, L.; Milaciu, M.V.; Runcan, O.; Vesa, S.C.; Răchișan, A.L.; Negrean, V.; Perné, M.-G.; Donca, V.I.; Alexescu, T.-G.; Para, I.; et al. The Effects of Flavonoids in Cardiovascular Diseases. Molecules 2020, 25, 4320. [Google Scholar] [CrossRef]
- Singh, A.; Patel, S.K.; Kumar, P.; Das, K.C.; Verma, D.; Sharma, R.; Tripathi, T.; Giri, R.; Martins, N.; Garg, N. Quercetin acts as a P-gp modulator via impeding signal transduction from nucleotide-binding domain to transmembrane domain. J. Biomol. Struct. Dyn. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Shaji, S.K.; G, D.; Sunilkumar, D.; Pandurangan, N.; Kumar, G.B.; Nair, B.G. Nuclear factor-κB plays an important role in Tamarixetin-mediated inhibition of matrix metalloproteinase-9 expression. Eur. J. Pharmacol. 2020, 10. [Google Scholar] [CrossRef]
- Pilátová, M.; Stupáková, V.; Varinská, L.; Sarisský, M.; Mirossay, L.; Mirossay, A.; Gál, P.; Kraus, V.; Dianisková, K.; Mojzis, J. Effect of selected flavones on cancer and endothelial cells. Gen. Physiol. Biophys. 2010, 29, 134–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gates, M.A.; Tworoger, S.S.; Hecht, J.L.; De Vivo, I.; Rosner, B.; Hankinson, S.E. A prospective study of dietary flavonoid intake and incidence of epithelial ovarian cancer. Int. J. Cancer 2007, 121, 2225–2232. [Google Scholar] [CrossRef] [PubMed]
- Mojžišová, G.; Šarišský, M.; Mirossay, L.; Martinka, P.; Mojžiš, J. Effect of Flavonoids on Daunorubicin-induced Toxicity in H9c2 Cardiomyoblasts. Phytotherapy Res. 2008, 23, 136–139. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.J.; Symons, J.D.; Jalili, T. Therapeutic potential of quercetin to decrease blood pressure: Review of efficacy and mechanisms. Adv. Nutr. 2012, 3, 39–46. [Google Scholar] [CrossRef] [Green Version]
- Leeya, Y.; Mulvany, M.J.; Queiroz, E.F.; Marston, A.; Hostettmann, K.; Jansakul, C. Hypotensive activity of an n-butanol extract and their purified compounds from leaves of Phyllanthus acidus (L.) Skeels in rats. Eur. J. Pharmacol. 2010, 649, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Hang, Y.; Qin, X.; Ren, T.; Cao, J. Baicalin reduces blood lipids and inflammation in patients with coronary artery disease and rheumatoid arthritis: A randomized, double-blind, placebo-controlled trial. Lipids Heal. Dis. 2018, 17, 146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakkar, Z.A.; Fulford, J.; Gates, P.E.; Jackman, S.R.; Jones, A.M.; Bond, B.; Bowtell, J.L. Montmorency cherry supplementation attenuates vascular dysfunction induced by prolonged forearm occlusion in overweight, middle-aged men. J. Appl. Phsiol. 2019, 126, 246–254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dugo, P.; Mondello, L.; Morabito, D.; Dugo, G. Characterization of the anthocyanin fraction of sicilian blood orange juice by micro-HPLC-ESI/MS. J. Agric. Food Chem. 2003, 51, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Silveira, J.Q.; Dourado, G.K.Z.S.; Cesar, T.B. Red-fleshed sweet orange juice improves the risk factors for metabolic syndrome. Int. J. Food Sci. Nutr. 2015, 66, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Li, S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson’s disease. Geriatr. Gerontol. Int. 2013, 14, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Miyake, Y.; Fukushima, W.; Sasaki, S.; Kiyohara, C.; Tsuboi, Y.; Yamada, T.; Oeda, T.; Miki, T.; Kawamura, N.; et al. Intake of Japanese and Chinese teas reduces risk of Parkinson’s disease. Park. Relat. Disord. 2011, 17, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Unno, K.; Ohishi, T.; Tanabe, H.; Miyoshi, N.; Nakamura, Y. Beneficial Effects of Green Tea Catechins on Neurodegenerative Diseases. Molecules 2018, 23, 1297. [Google Scholar] [CrossRef] [Green Version]
- Fan, D.; Alamri, Y.; Liu, K.; MacAskill, M.; Harris, P.W.R.; Brimble, M.A.; Dalrymple-Alford, J.C.; Prickett, T.; Menzies, O.; Laurenson, A.; et al. Supplementation of Blackcurrant Anthocyanins Increased Cyclic Glycine-Proline in the Cerebrospinal Fluid of Parkinson Patients: Potential Treatment to Improve Insulin-Like Growth Factor-1 Function. Nutrients 2018, 10, 714. [Google Scholar] [CrossRef] [Green Version]
- Augustin, R.C.; Delgoffe, G.M.; Najjar, Y.G. Characteristics of the Tumor Microenvironment That Influence Immune Cell Functions: Hypoxia, Oxidative Stress, Metabolic Alterations. Cancers 2020, 12, 3802. [Google Scholar] [CrossRef]
- Wang, W.; Kang, P.M. Oxidative Stress and Antioxidant Treatments in Cardiovascular Diseases. Antioxidants 2020, 9, 1292. [Google Scholar] [CrossRef]
- Rehman, K.; Haider, K.; Jabeen, K.; Akash, M.S.H. Current perspectives of oleic acid: Regulation of molecular pathways in mitochondrial and endothelial functioning against insulin resistance and diabetes. Rev. Endocr. Metab. Disord. 2020, 21, 631–643. [Google Scholar] [CrossRef]
- Antonyová, V.; Kejík, Z.; Brogyányi, T.; Kaplánek, R.; Pajková, M.; Talianová, V.; Hromádka, R.; Masařík, M.; Sýkora, D.; Mikšátková, L.; et al. Role of mtDNA disturbances in the pathogenesis of Alzheimer’s and Parkinson’s disease. DNA Repair. 2020, 91–92, 102871. [Google Scholar] [CrossRef]
- Prochazkova, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Cherrak, S.A.; Mokhtari-Soulimane, N.; Berroukeche, F.; Bensenane, B.; Cherbonnel, A.; Merzouk, H.; Elhabiri, M. In Vitro Antioxidant versus Metal Ion Chelating Properties of Flavonoids: A Structure-Activity Investigation. PLoS ONE 2016, 11, e0165575. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, A.; Saidullah, B.; Sultana, S. Anti-carcinogenic effect of hesperidin against renal cell carcinoma by targeting COX-2/PGE2 pathway in Wistar rats. Environ. Toxicol. 2018, 33, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Haba, R.; Watanabe, S.; Arai, Y.; Chiba, H.; Miura, T. Suppression of Lipid-Hydroperoxide and DNA-Adduct Formation by Isoflavone-Containing Soy Hypocotyl Tea in Rats. Environ. Heal. Prev. Med. 2002, 7, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef] [Green Version]
- Koskenkorva-Frank, T.S.; Weiss, G.; Koppenol, W.H.; Burckhardt, S. The complex interplay of iron metabolism, reactive oxygen and reactive nitrogen species: Insights into the potential of different iron therapies to induce oxidative and nitrosative stress. Free Rad. Biol. Med. 2013, 65, 1174–1194. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.L.; Perez, C.; Wei, Y.B.; Rapoza, E.; Su, G.; Bou-Abdallah, F.; Chasteen, N.D. Iron-binding properties of plant phenolics and cranberry’s bio-effects. Dalton Trans. 2007, 10, 4951–4961. [Google Scholar] [CrossRef] [Green Version]
- Horniblow, R.D.; Henesy, D.; Iqbal, T.H.; Tselepis, C. Modulation of iron transport, metabolism and reactive oxygen status by quercetin-iron complexes in vitro. Mol. Nutr. Food Res. 2016, 61. [Google Scholar] [CrossRef]
- Miličević, A.; Raos, N. Modelling of Protective Mechanism of Iron(II)-polyphenol Binding with OH-related Molecular Descriptors. Croat. Chem. Acta 2016, 89, 89. [Google Scholar] [CrossRef]
- Perez, C.A.; Wei, Y.; Guo, M. Iron-binding and anti-Fenton properties of baicalein and baicalin. J. Inorg. Biochem. 2009, 103, 326–332. [Google Scholar] [CrossRef] [Green Version]
- Kostyuk, V.; Potapovich, A.; Strigunova, E.; Kostyuk, T.; Afanas’ev, I. Experimental evidence that flavonoid metal complexes may act as mimics of superoxide dismutase. Arch. Biochem. Biophys. 2004, 428, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Porfírio, D.A.; Ferreira, R.D.Q.; Malagutti, A.R.; Valle, E.M.A. Electrochemical study of the increased antioxidant capacity of flavonoids through complexation with iron(II) ions. Electrochim. Acta 2014, 141, 33–38. [Google Scholar] [CrossRef]
- Baccan, M.M.; Chiarelli-Neto, O.; Pereira, R.M.S.; Espósito, B.P. Quercetin as a shuttle for labile iron. J. Inorg. Biochem. 2012, 107, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Lee, H.-Y.; Ahn, D.-R.; Kim, S.-Y.; Kim, S.; Lee, K.B.; Lee, Y.M.; Park, H.; Yang, E.G. Baicalein induces functional hypoxia-inducible factor-1 alpha and angiogenesis. Mol. Pharmacol. 2008, 74, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.; Kim, H.; Choi, D.; Kim, D.; Park, S.-Y.; Kim, Y.-J.; Kim, Y.M.; Jung, Y. Quercetin activates an angiogenic pathway, hypoxia inducible factor (HIF)-1-vascular endothelial growth factor, by inhibiting HIF-prolyl hydroxylase: A structural analysis of quercetin for inhibiting HIF-prolyl hydroxylase. Mol. Pharmacol. 2007, 71, 1676–1684. [Google Scholar] [CrossRef] [PubMed]
- Mahesh, T.; Menon, V.P. Quercetin allievates oxidative stress in streptozotocin-induced diabetic rats. Phytother. Res. 2004, 18, 123–127. [Google Scholar] [CrossRef]
- Mascayano, C.; Núñez, G.; Acevedo, W.; Rezende, M.C. Binding of arachidonic acid and two flavonoid inhibitors to human 12-and 15-lipoxygenases: A steered molecular dynamics study. J. Mol. Model. 2010, 16, 1039–1045. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, H.; Iwanaga, S.; Kawazoe, K.; Ishizawa, K.; Orino, S.; Tomita, S.; Tsuchiya, K.; Kanematsu, Y.; Harada, N.; Mori, K.; et al. Effect of Iron-Quercetin Complex on Reduction of Nitrite in in Vitro and in Vivo Systems. J. Agric. Food Chem. 2008, 56, 10092–10098. [Google Scholar] [CrossRef]
- Raza, A.; Xu, X.; Xia, L.; Xia, C.; Tang, J.; Ouyang, Z. Quercetin-Iron Complex: Synthesis, Characterization, Antioxidant, DNA Binding, DNA Cleavage, and Antibacterial Activity Studies. J. Fluoresc. 2016, 26, 2023–2031. [Google Scholar] [CrossRef]
- Sakurai, M.; Rose, N.R.; Schultz, L.; Quinn, A.M.; Jadhav, A.; Ng, S.S.; Oppermann, U.; Schofield, C.J.; Simeonov, A. A miniaturized screen for inhibitors of Jumonji histone demethylases. Mol. Biosyst. 2010, 6, 357–364. [Google Scholar] [CrossRef] [Green Version]
- Sengupta, B. Biophysical Characterization of Genistein in Its Natural Carrier Human Hemoglobin Using Spectroscopic and Computational Approaches. Food Nutr. 2013, 4, 83–92. [Google Scholar]
- Yang, F.; Guo, Z.; Xiao, Y.; Zhu, J. Fe2+ and Co2+ Improved the Affinities of 7-Hydroxyflavone, Chrysin and Quercetin for Human Serum Albumin In Vitro. J. Solut. Chem. 2013, 42, 1717–1728. [Google Scholar] [CrossRef]
- Kim, Y.A.; Tarahovsky, Y.S.; Yagolnik, E.A.; Kuznetsova, S.M.; Muzafarov, E.N. Lipophilicity of flavonoid complexes with iron(II) and their interaction with liposomes. Biochem. Biophys. Res. Commun. 2013, 431, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.A.; Tarahovsky, Y.S.; Yagolnik, E.A.; Kuznetsova, S.M.; Muzafarov, E.N. Integration of Quercetin-Iron Complexes into Phosphatidylcholine or Phosphatidylethanolamine Liposomes. Appl. Biochem. Biotech. 2015, 176, 1904–1913. [Google Scholar] [CrossRef] [PubMed]
- Shatalin Iu, V.; Shubina, V.S. Partitioning of taxifolin-iron ions complexes in octanol-water system. Biofizika 2014, 59, 432–438. [Google Scholar] [CrossRef]
- Kostyuk, V.A.; Potapovich, A.; Vladykovskaya, E.; Korkina, L.; Afanas’ev, I. Influence of Metal Ions on Flavonoid Protection against Asbestos-Induced Cell Injury. Arch. Biochem. Biophys. 2001, 385, 129–137. [Google Scholar] [CrossRef]
- Leopoldini, M.; Russo, N.; Toscano, M. The molecular basis of working mechanism of natural polyphenolic antioxidants. Food Chem. 2011, 125, 288–306. [Google Scholar] [CrossRef]
- Xu, D.; Hu, M.-J.; Wang, Y.-Q.; Cui, Y.-L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules 2019, 24, 1123. [Google Scholar] [CrossRef] [Green Version]
- Bhuiyan, M.N.I.; Mitsuhashi, S.; Sigetomi, K.; Ubukata, M. Quercetin inhibits advanced glycation end product formation via chelating metal ions, trapping methylglyoxal, and trapping reactive oxygen species. Biosci. Biotechnol. Biochem. 2017, 81, 882–890. [Google Scholar] [CrossRef] [Green Version]
- Primikyri, A.; Mazzone, G.; Lekka, C.; Tzakos, A.G.; Russo, N.; Gerothanassis, I.P. Understanding Zinc(II) Chelation with Quercetin and Luteolin: A Combined NMR and Theoretical Study. J. Phys. Chem. B 2014, 119, 83–95. [Google Scholar] [CrossRef]
- Sun, S.; Chen, W.; Cao, W.; Zhang, F.; Song, J.; Tian, C. Research on the chelation between quercetin and Cr(III) ion by Density Functional Theory (DFT) method. J. Mol. Struct. Theochem. 2008, 860, 40–44. [Google Scholar] [CrossRef]
- Potapovich, A.I. Comparative study of antioxidant properties and cytoprotective activity of flavonoids. Biochemistry (Moscow) 2003, 68, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Mahesha, H.; Singh, S.A.; Rao, A.A. Inhibition of lipoxygenase by soy isoflavones: Evidence of isoflavones as redox inhibitors. Arch. Biochem. Biophys. 2007, 461, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.; Freitas, M.; Tomé, S.M.; Silva, A.M.S.; Laufer, S.; Lima, J.L.F.C.; Fernandes, E. Flavonoids Inhibit COX-1 and COX-2 Enzymes and Cytokine/Chemokine Production in Human Whole Blood. Inflammation 2014, 38, 858–870. [Google Scholar] [CrossRef]
- Nagao, A.; Seki, M.; Kobayashi, H. Inhibition of Xanthine Oxidase by Flavonoids. Biosci. Biotechnol. Biochem. 1999, 63, 1787–1790. [Google Scholar] [CrossRef]
- Bohmont, C.; Aaronson, L.M.; Mann, K.; Pardini, R.S. Inhibition of Mitochondrial NADH Oxidase, Succinoxidase, and ATPase by Naturally Occurring Flavonoids. J. Nat. Prod. 1987, 50, 427–433. [Google Scholar] [CrossRef]
- Krych, J.; Gebicka, L. Catalase is inhibited by flavonoids. Int. J. Biol. Macromol. 2013, 58, 148–153. [Google Scholar] [CrossRef]
- Loke, W.M.; Proudfoot, J.M.; McKinley, A.J.; Needs, P.W.; Kroon, P.A.; Hodgson, J.M.; Croft, K.D. Quercetin and its in vivo metabolites inhibit neutrophil-mediated low-density lipoprotein oxidation. J. Agric. Food Chem. 2008, 56, 3609–3615. [Google Scholar] [CrossRef]
- Yasuda, M.; Sonda, T.; Nagayama, K.; Tabata, M. Effects of pH and metal ions on antioxidative activities of catechins. Biosci. Biotechnol. Biochem. 2001, 65, 126–132. [Google Scholar] [CrossRef] [Green Version]
- Dowling, S.; Regan, F.; Hughes, H. The characterisation of structural and antioxidant properties of isoflavone metal chelates. J. Inorg. Biochem. 2010, 104, 1091–1098. [Google Scholar] [CrossRef]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florêncio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Loizzo, M.R.; Tundis, R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Avallone, L.; Menichini, F. Radical scavenging, antioxidant and metal chelating activities of Annona cherimola Mill. (cherimoya) peel and pulp in relation to their total phenolic and total flavonoid contents. J. Food Compos. Anal. 2012, 25, 179–184. [Google Scholar] [CrossRef]
- Macáková, K.; Mladěnka, P.; Filipský, T.; Říha, M.; Jahodář, L.; Trejtnar, F.; Bovicelli, P.; Silvestri, I.P.; Hrdina, R.; Saso, L. Iron reduction potentiates hydroxyl radical formation only in flavonols. Food Chem. 2012, 135, 2584–2592. [Google Scholar] [CrossRef] [PubMed]
- Laughton, M.J.; Halliwell, B.; Evans, P.J.; Robin, J.; Hoult, J.R. Antioxidant and Pro-Oxidant Actions of the Plant Phenolics Quercetin, Gossypol and Myricetin—Effects on Lipid-Peroxidation, Hydroxyl Radical Generation and Bleomycin-Dependent Damage to DNA. Biochem. Pharmacol. 1989, 38, 2859–2865. [Google Scholar] [CrossRef]
- Cheng, I.F.; Breen, K. On the ability of four flavonoids, baicilein, luteolin, naringenin, and quercetin, to suppress the fenton reaction of the iron-ATP complex. Biometals 2000, 13, 77–83. [Google Scholar] [CrossRef]
- Mladěnka, P.; Macáková, K.; Filipský, T.; Zatloukalová, L.; Jahodář, L.; Bovicelli, P.; Silvestri, I.P.; Hrdina, R.; Saso, L. In vitro analysis of iron chelating activity of flavonoids. J. Inorg. Biochem. 2011, 105, 693–701. [Google Scholar] [CrossRef]
- Fernandez, M.; Mira, M.; Florêncio, M.; Jennings, K.R. Iron and copper chelation by flavonoids: An electrospray mass spectrometry study. J. Inorg. Biochem. 2002, 92, 105–111. [Google Scholar] [CrossRef]
- Leopoldini, M.; Russo, N.; Chiodo, A.S.; Toscano, M. Iron chelation by the powerful antioxidant flavonoid quercetin. J. Agric. Food Chem. 2006, 54, 6343–6351. [Google Scholar] [CrossRef]
- Satterfield, M.; Brodbelt, J.S. Enhanced detection of flavonoids by metal complexation and electrospray ionization mass spectrometry. Anal. Chem. 2000, 72, 5898–5906. [Google Scholar] [CrossRef]
- Bodini, M.E.; Copia, G.; Tapia, R.; Leighton, F.; Herrera, L. Iron complexes of quercetin in aprotic medium. Redox chemistry and interaction with superoxide anion radical. Polyhedron 1999, 18, 2233–2239. [Google Scholar] [CrossRef]
- Shubina, V.; Shatalina, Y.V. Absorption Spectroscopy Study of Acid-Base and Metal-Binding Properties of Flavanones. J. Appl. Spectrosc. 2013, 80, 761–766. [Google Scholar] [CrossRef]
- Goyal, R.; Jialal, I. Hyperphosphatemia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Rizzo, S.C.; Eckel, R. Control of glycolysis in human erythrocytes by inorganic phosphate and sulfate. Am. J. Physiol. Content 1966, 211, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, C.R. Characterization and application of FerroZine iron reagent as a ferrous iron indicator. Anal. Chem. 1976, 48, 1197–1201. [Google Scholar] [CrossRef]
- Kumara, T.; Kaumal, M. The Photo-and Electro-Chemical Properties of Anthocyanin Extracts of Red Cabbage. Chem. Res. J. 2018, 3, 84–91. [Google Scholar]
- Sigurdson, G.T.; Robbins, R.; Collins, T.; Giusti, M. Spectral and colorimetric characteristics of metal chelates of acylated cyanidin derivatives. Food Chem. 2017, 221, 1088–1095. [Google Scholar] [CrossRef]
- Sigurdson, G.T.; Robbins, R.; Collins, T.; Giusti, M. Evaluating the role of metal ions in the bathochromic and hyperchromic responses of cyanidin derivatives in acidic and alkaline pH. Food Chem. 2016, 208, 26–34. [Google Scholar] [CrossRef]
- Kunsági-Máté, S.; Ortmann, E.; Kollár, L.; Szabó, K.; Pour Nikfardjam, M. Effect of ferrous and ferric ions on copigmentation in model solutions. J. Mol. Struct. 2008, 891, 471–474. [Google Scholar] [CrossRef]
- Yoshida, K.; Mori, M.; Kondo, T. Blue flower color development by anthocyanins: From chemical structure to cell physiology. Nat. Prod. Rep. 2009, 26, 884–915. [Google Scholar] [CrossRef]
- Areias, F.M.; Rego, A.; Oliveira, C.R.; Seabra, R.M. Antioxidant effect of flavonoids after ascorbate/Fe2+-induced oxidative stress in cultured retinal cells. Biochem. Pharmacol. 2001, 62, 111–118. [Google Scholar] [CrossRef]
- Vlachodimitropoulou, E.; Sharp, P.A.; Naftalin, R.J. Quercetin-iron chelates are transported via glucose transporters. Free Rad. Biol. Med. 2011, 50, 934–944. [Google Scholar] [CrossRef]
- Melidou, M.; Riganakos, K.; Galaris, D. Protection against nuclear DNA damage offered by flavonoids in cells exposed to hydrogen peroxide: The role of iron chelation. Free Rad. Biol. Med. 2005, 39, 1591–1600. [Google Scholar] [CrossRef]
- Tarahovsky, Y.S.; Yagolnik, E.A.; Muzafarov, E.N.; Abdrasilov, B.S.; Kim, Y.A. Calcium-dependent aggregation and fusion of phosphatidylcholine liposomes induced by complexes of flavonoids with divalent iron. Biochim. Biophys. Acta Biomembr. 2012, 1818, 695–702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, J.; Wang, B.; Zhu, L. DNA binding, cytotoxicity, apoptotic inducing activity, and molecular modeling study of quercetin zinc(II) complex. Bioorg. Med. Chem. 2009, 17, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.E.; Rekman, J.F.; Gunnink, L.K.; Busscher, B.M.; Scott, J.L.; Tidball, A.M.; Stehouwer, N.R.; Johnecheck, G.N.; Looyenga, B.D.; Louters, L.L. Quercetin inhibits glucose transport by binding to an exofacial site on GLUT1. Biochimie 2018, 151, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Takahama, U.; Oniki, T.; Hirota, S. Oxidation of quercetin by salivary components. Quercetin-dependent reduction of salivary nitrite under acidic conditions producing nitric oxide. J. Agric. Food Chem. 2002, 50, 4317–4322. [Google Scholar] [CrossRef]
- Takahama, U.; Yamamoto, A.; Hirota, S.; Oniki, T. Quercetin-dependent reduction of salivary nitrite to nitric oxide under acidic conditions and interaction between quercetin and ascorbic acid during the reduction. J. Agric. Food Chem. 2003, 51, 6014–6020. [Google Scholar] [CrossRef]
- Magar, R.T.; Sohng, J.K. A Review on Structure, Modifications and Structure-Activity Relation of Quercetin and Its Derivatives. J. Microbiol. Biotechnol. 2019, 30, 11–20. [Google Scholar] [CrossRef]
- Wen, L.; Jiang, Y.; Yang, J.; Zhao, Y.; Tian, M.; Yang, B. Structure, bioactivity, and synthesis of methylated flavonoids: Methylated flavonoids. Ann. N. Y. Acad. Sci. 2017, 1398, 120–129. [Google Scholar] [CrossRef]
- Ahmed, Q.U.; Sarian, M.N.; So’Ad, S.Z.M.; Latip, J.; Ichwan, S.J.A.; Hussein, N.N.; Taher, M.; Alhassan, A.M.; Hamidon, H.; Fakurazi, S. Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study. Biomolecules 2018, 8, 149. [Google Scholar] [CrossRef] [Green Version]
- Teles, Y.C.F.; Souza, M.D.F.V.D. Sulphated Flavonoids: Biosynthesis, Structures, and Biological Activities. Molecules 2018, 23, 480. [Google Scholar] [CrossRef] [Green Version]
- Huynh, N.T.; Van Camp, J.; Smagghe, G.; Raes, K. Improved Release and Metabolism of Flavonoids by Steered Fermentation Processes: A Review. Int. J. Mol. Sci. 2014, 15, 19369–19388. [Google Scholar] [CrossRef] [PubMed]
- Paik, W.K.; Kim, S. Enzymatic demethylation of calf thymus histones. Biochem. Biophys. Res. Commun. 1973, 51, 781–788. [Google Scholar] [CrossRef]
- Saraç, H.; Morova, T.; Pires, E.; McCullagh, J.; Kaplan, A.; Cingöz, A.; Bagci-Onder, T.; Önder, T.; Kawamura, A.; Lack, N.A. Systematic characterization of chromatin modifying enzymes identifies KDM3B as a critical regulator in castration resistant prostate cancer. Oncogene 2020, 39, 2187–2201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mashima, R.; Okuyama, T. The role of lipoxygenases in pathophysiology; new insights and future perspectives. Redox Biol. 2015, 6, 297–310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasquez-Martinez, Y.; Ohri, R.V.; Kenyon, V.; Holman, T.R.; Sepúlveda-Boza, S. Structure-activity relationship studies of flavonoids as potent inhibitors of human platelet 12-hLO, reticulocyte 15-hLO-1, and prostate epithelial 15-hLO-2. Bioorg. Med. Chem. 2007, 15, 7408–7425. [Google Scholar] [CrossRef] [Green Version]
- Triantafyllou, A.; Mylonis, I.; Simos, G.; Bonanou, S.; Tsakalof, A. Flavonoids induce HIF-1 alpha but impair its nuclear accumulation and activity. Free Radic. Biol. Med. 2008, 44, 657–670. [Google Scholar] [CrossRef]
- Triantafyllou, A.; Liakos, P.; Tsakalof, A.; Chachami, G.; Paraskeva, E.; Molyvdas, P.-A.; Georgatsou, E.; Simos, G.; Bonanou, S. The flavonoid quercetin induces hypoxia-inducible factor-1 alpha (HIF-1 alpha) and inhibits cell proliferation by depleting intracellular iron. Free Radic. Res. 2007, 41, 342–356. [Google Scholar] [CrossRef]
- Gebicka, L. Redox reactions of heme proteins with flavonoids. J. Inorg. Biochem. 2020, 208, 111095. [Google Scholar] [CrossRef]
- Poulos, T.L. Heme Enzyme Structure and Function. Chem. Rev. 2014, 114, 3919–3962. [Google Scholar] [CrossRef] [Green Version]
- Blakemore, J.; Naftolin, F. Aromatase: Contributions to Physiology and Disease in Women and Men. Physiology 2016, 31, 258–269. [Google Scholar] [CrossRef] [Green Version]
- Santen, R.J. Aromatase inhibitors for treatment of breast cancer: Current concepts and new perspectives. Breast Cancer Res. Treat. 1986, 7, S23–S35. [Google Scholar] [PubMed]
- Kellis, J.; Vickery, L. Inhibition of human estrogen synthetase (aromatase) by flavones. Science 1984, 225, 1032–1034. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.C.; Zhou, C.; Sherman, M.; Laughton, C.A.; Chen, S. Molecular basis of the inhibition of human aromatase (estrogen synthetase) by flavone and isoflavone phytoestrogens: A site-directed mutagenesis study. Environ. Heal. Perspect. 1998, 106, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Karkola, S.; Wähälä, K. The binding of lignans, flavonoids and coumestrol to CYP450 aromatase: A molecular modelling study. Mol. Cell. Endocrinol. 2009, 301, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Dawson, J.; Andersson, L.; Sono, M. Spectroscopic investigations of ferric cytochrome P-450-CAM ligand complexes. Identification of the ligand trans to cysteinate in the native enzyme. J. Biol. Chem. 1982, 257, 3606–3617. [Google Scholar] [CrossRef]
- Ibrahim, A.-R.S.; Abul-Hajj, Y.J. Aromatase inhibition by flavonoids. J. Steroid Biochem. Mol. Biol. 1990, 37, 257–260. [Google Scholar] [CrossRef]
- Sanderson, J.T.; Hordijk, J.; Denison, M.S.; Springsteel, M.F.; Nantz, M.H.; Berg, M.V.D. Induction and Inhibition of Aromatase (CYP19) Activity by Natural and Synthetic Flavonoid Compounds in H295R Human Adrenocortical Carcinoma Cells. Toxicol. Sci. 2004, 82, 70–79. [Google Scholar] [CrossRef] [Green Version]
- Irahara, N.; Miyoshi, Y.; Taguchi, T.; Tamaki, Y.; Noguchi, S. Quantitative analysis of aromatase mRNA expression derived from various promoters (I.4, I.3, PII and I.7) and its association with expression of TNF-alpha, IL-6 and COX-2 mRNAs in human breast cancer. Int. J. Cancer 2005, 118, 1915–1921. [Google Scholar] [CrossRef]
- Fita, I.; Rossmann, M.G. The active center of catalase. J. Mol. Biol. 1985, 185, 21–37. [Google Scholar] [CrossRef]
- Lin, Y.-L.; Cheng, C.-Y.; Lin, Y.-P.; Lau, Y.-W.; Juan, I.-M.; Lin, J.-K. Hypolipidemic Effect of Green Tea Leaves through Induction of Antioxidant and Phase II Enzymes Including Superoxide Dismutase, Catalase, and Glutathione S-Transferase in Rats. J. Agric. Food Chem. 1998, 46, 1893–1899. [Google Scholar] [CrossRef]
- Kampkötter, A.; Chovolou, Y.; Kulawik, A.; Röhrdanz, E.; Weber, N.; Proksch, P.; Wätjen, W. Isoflavone daidzein possesses no antioxidant activities in cell-free assays but induces the antioxidant enzyme catalase. Nutr. Res. 2008, 28, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Majumder, D.; Das, A.; Saha, C. Catalase inhibition an anti cancer property of flavonoids: A kinetic and structural evaluation. Int. J. Biol. Macromol. 2017, 104, 929–935. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Dey, S.K.; Saha, C. Inhibition of Catalase by Tea Catechins in Free and Cellular State: A Biophysical Approach. PLoS ONE 2014, 9, e102460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinecke, J.W.; Li, W.; Daehnke, H.L., 3rd; Goldstein, J.A. Dityrosine, a specific marker of oxidation, is synthesized by the myeloperoxidase-hydrogen peroxide system of human neutrophils and macrophages. J. Biol. Chem. 1993, 268, 4069–4077. [Google Scholar] [CrossRef]
- Nakagawara, A.; Nathan, C.F.; Cohn, Z.A. Hydrogen peroxide metabolism in human monocytes during differentiation in vitro. J. Clin. Investig. 1981, 68, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Shiba, Y.; Kinoshita, T.; Chuman, H.; Taketani, Y.; Takeda, E.; Kato, Y.; Naito, M.; Kawabata, K.; Ishisaka, A.; Terao, J.; et al. Flavonoids as substrates and inhibitors of myeloperoxidase: Molecular actions of aglycone and metabolites. Chem. Res. Toxicol. 2008, 21, 1600–1609. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, T.; Prasain, J.; Benton, M.R.; Botting, N.; Moore, R.; Darley-Usmar, V.; Patel, R.; Barnes, S. Polyphenols, Inflammatory Response, and Cancer Prevention: Chlorination of Isoflavones by Human Neutrophils. J. Nutr. 2003, 133, 3773S–3777S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Binsack, R.; Boersma, B.J.; Patel, R.P.; Kirk, M.; White, C.R.; Darley-Usmar, V.; Barnes, S.; Zhou, F.; Parks, D.A. Enhanced antioxidant activity after chlorination of quercetin by hypochlorous acid. Alcohol. Clinic. Experim. Res. 2001, 25, 434–443. [Google Scholar] [CrossRef]
- Kawai, Y.; Nishikawa, T.; Shiba, Y.; Saito, S.; Murota, K.; Shibata, N.; Kobayashi, M.; Kanayama, M.; Uchida, K.; Terao, J. Macrophage as a target of quercetin glucuronides in human atherosclerotic arteries: Implication in the anti-atherosclerotic mechanism of dietary flavonoids. J. Biol. Chem. 2008, 283, 9424–9434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibero-Baraibar, I.; Abete, I.; Navas-Carretero, S.; Massis-Zaid, A.; Martinez, J.A.; Zulet, M.A. Oxidised LDL levels decreases after the consumption of ready-to-eat meals supplemented with cocoa extract within a hypocaloric diet. Nutr. Metabol. Cardiovasc. Dis. 2014, 24, 416–422. [Google Scholar] [CrossRef] [Green Version]
- Basu, A.; Sanchez, K.; Leyva, M.J.; Wu, M.; Betts, N.M.; Aston, C.E.; Lyons, T.J. Green tea supplementation affects body weight, lipids, and lipid peroxidation in obese subjects with metabolic syndrome. J. Am. Coll. Nutr. 2010, 29, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Lowe, G.M.; Gana, K.; Rahman, K. Dietary supplementation with green tea extract promotes enhanced human leukocyte activity. J. Complement. Integr. Med. 2015, 12, 277–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yen, G.C.; Duh, P.D.; Tsai, H.L.; Huang, S.L. Pro-oxidative properties of flavonoids in human lymphocytes. Biosci. Biotechnol. Biochem. 2003, 67, 1215–1222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zubair, H.; Ullah, M.; Ahmad, A.; Khan, H.; Hadi, S.M. Soy isoflavone genistein in cancer chemoprevention: Role of endogenous copper and involvement of reactive oxygen species. In Soy: Nutrition, Consumption and Health; Nova Science Publisher: Hauppauge, NY, USA, 2012; pp. 333–344. [Google Scholar]
- Saul, D.; Gleitz, S.; Nguyen, H.H.; Kosinsky, R.L.; Sehmisch, S.; Hoffmann, D.B.; Wassmann, M.; Menger, B.; Komrakova, M. Effect of the lipoxygenase-inhibitors baicalein and zileuton on the vertebra in ovariectomized rats. Bone 2017, 101, 134–144. [Google Scholar] [CrossRef]
- Che, J.; Yang, J.; Zhao, B.; Zhang, G.; Wang, L.; Peng, S.; Shang, P. The Effect of Abnormal Iron Metabolism on Osteoporosis. Biol. Trace Elem. Res. 2019, 195, 353–365. [Google Scholar] [CrossRef]
- Toxqui, L.; Vaquero, M.P. Chronic iron deficiency as an emerging risk factor for osteoporosis: A hypothesis. Nutrients 2015, 7, 2324–2344. [Google Scholar] [CrossRef] [Green Version]
- Fung, E.B.; Harmatz, P.R.; Milet, M.; Coates, T.D.; Thompson, A.A.; Ranalli, M.; Mignaca, R.; Scher, C.; Giardina, P.; Robertson, S.; et al. Fracture prevalence and relationship to endocrinopathy in iron overloaded patients with sickle cell disease and thalassemia. Bone 2008, 43, 162–168. [Google Scholar] [CrossRef] [Green Version]
- Vasta, J.D.; Raines, R.T. Collagen Prolyl 4-Hydroxylase as a Therapeutic Target. J. Med. Chem. 2018, 61, 10403–10411. [Google Scholar] [CrossRef] [Green Version]
- Krol, B.J.; Murad, S.; Walker, L.C.; Marshall, M.K.; Clark, W.L.; Pinnell, S.R.; Yeowell, H.N. The expression of a functional, secreted human lysyl hydroxylase in a baculovirus system. J. Investig. Dermatol. 1996, 106, 11–16. [Google Scholar] [CrossRef] [Green Version]
- Bonifácio, B.; Silva, P.; Ramos, M.; Ramos, S.; Negri, K.; Bauab, T.; Chorilli, M. Nanotechnology-based drug delivery systems and herbal medicines: A review. Int. J. Nanomed. 2013, 9, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Suvarna, V.; Gujar, P.; Murahari, M. Complexation of phytochemicals with cyclodextrin derivatives – An insight. Biomed. Pharmacother. 2017, 88, 1122–1144. [Google Scholar] [CrossRef] [PubMed]
- Jakab, G.; Bogdán, D.; Mazák, K.; Deme, R.; Mucsi, Z.; Mándity, I.M.; Noszál, B.; Kállai-Szabó, N.; Antal, I. Physicochemical Profiling of Baicalin Along with the Development and Characterization of Cyclodextrin Inclusion Complexes. AAPS PharmSciTech 2019, 20, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahmanian, N.; Hamishehkar, H.; Dolatabadi, J.E.N.; Arsalani, N. Nano graphene oxide: A novel carrier for oral delivery of flavonoids. Coll. Surf. B Biointerfaces 2014, 123, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Maiti, K.; Murugan, V.; Saha, B.; Mukherjee, P. Exploring the Effect of Hesperetin–HSPC Complex—A Novel Drug Delivery System on the In Vitro Release, Therapeutic Efficacy and Pharmacokinetics. AAPS PharmSciTech 2009, 10, 943–950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
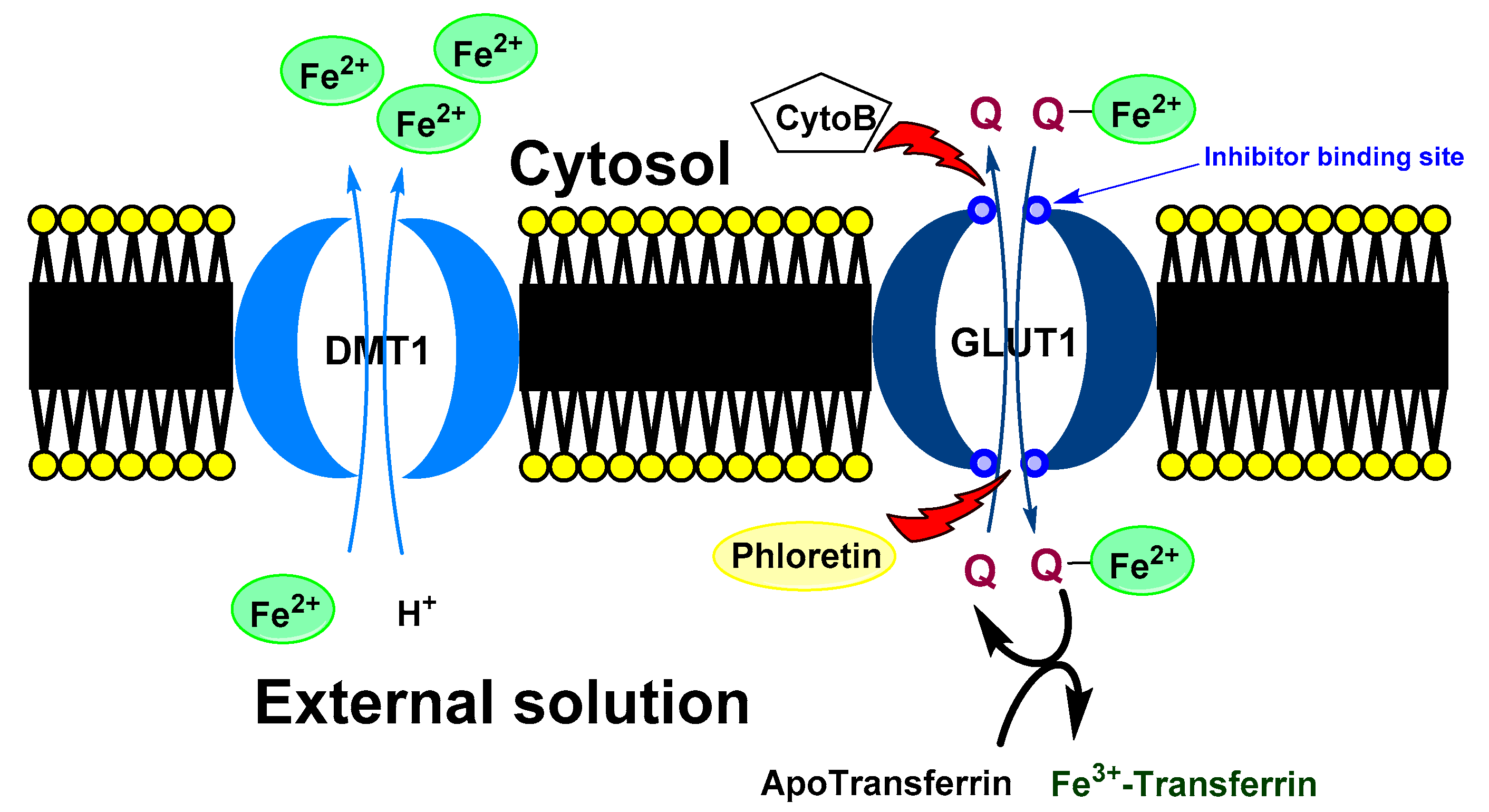
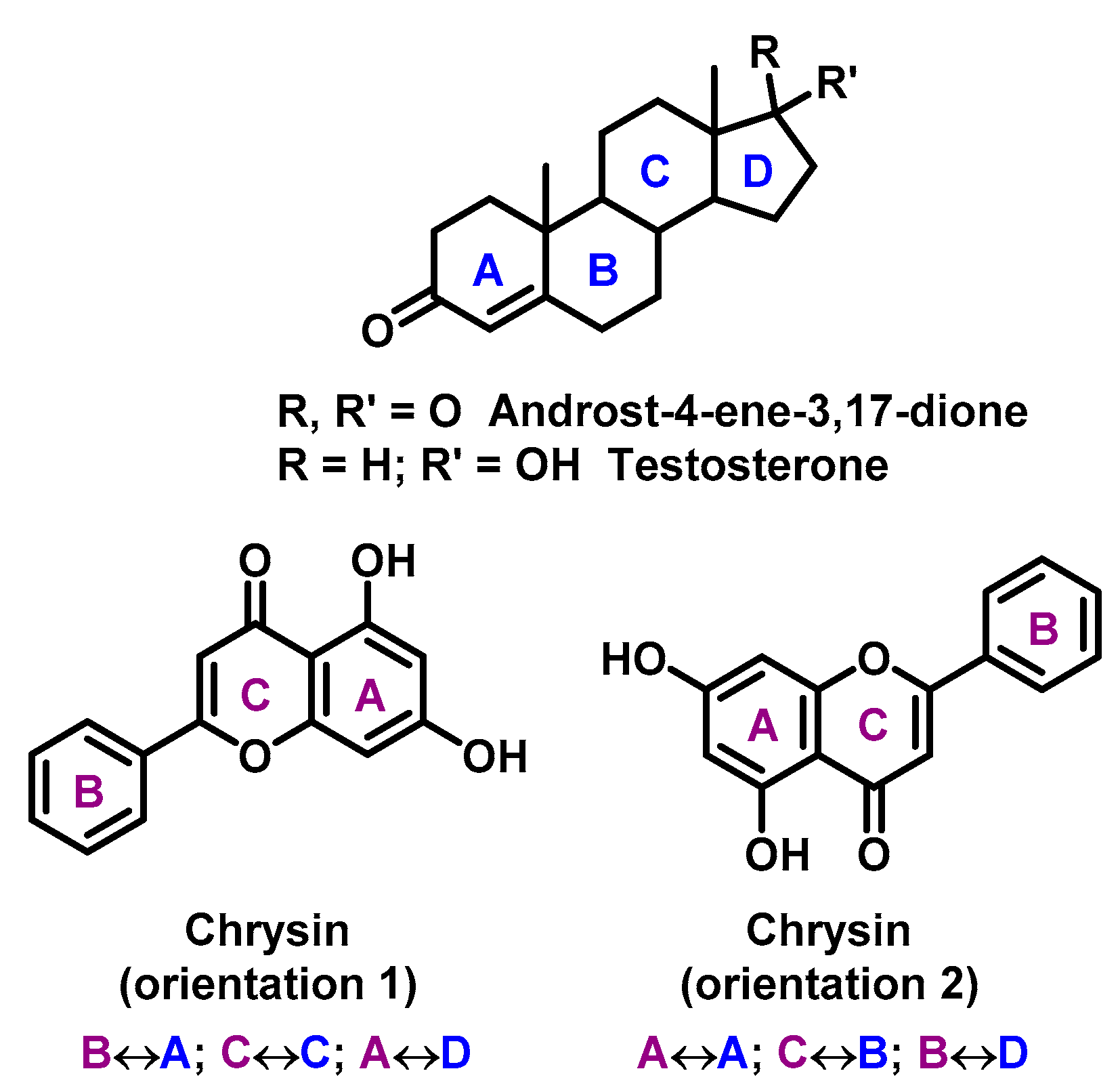
| Flavonoid | Method | System | K | St. | REF. |
|---|---|---|---|---|---|
| Fe(II) | |||||
| Quercetin | microcalorimetery | 0.1 M, pH = 7.2 | 3.6 × 106/M | 1:1 | [38] |
| UV-Vis | 0.02 M, pH = 7.2 | 2 × 106/M | 1:1 | [37] | |
| 5 × 1010/M2 | 1:2 | ||||
| Baicalein | UV-Vis | 0.02 M, pH = 7.2 | 9 × 1011/M2 | 1:2 | [40] |
| Rutin | microcalorimetery | 0.1 M, pH = 7 | 3.2 × 108/M | 1:1 | [38] |
| UV-Vis | 0.02 M, pH = 7.2 | 4 × 1011/M2 | 1:2 | [37] | |
| 3-HF | 2 × 1011/M2 | 1:2 | |||
| 3″,4″-DHF | 3 × 1010/M2 | 1:2 | |||
| Chrysin | 8 × 1010/M2 | 1:2 | |||
| Fe(III) | |||||
| Quercetin | microcalorimetery | 0.1 M, pH = 7 | 8.3 × 105/M2 | 1:2 | [38] |
| Rutin | 2.2 × 104/M2 | 1:1 | |||
| Enzymes | Flavonoids | IC50 (μmol/L) | Ref. |
|---|---|---|---|
| Iron Dependent Enzymes | |||
| Jumonji histone demethylase | baicalein | 4.0 | [50] |
| epigallocatechin | 5.0 | ||
| epigallocatechin gallate | 3.2 | ||
| myricetin | 3.6 | ||
| soy lypoxoygenase-1 | genistein | 0.136 | [63] |
| daidzein | 0.107 | ||
| human lypoxoygenase-5 | genistein | 0.157 | |
| daidzein | 0.125 | ||
| lipoxogenase-15 (human reticulocyte) | baicalin | 9.1 | [106] |
| quercetin | 0.25 | ||
| lipoxygenase-12 (human platelet) | baicalin | 0.86 μmol/L | |
| quercetin | 0.26 | ||
| Heme Enzymes | |||
| Aromatase (CYP450) | 7,8 benzoflavone | 0.07 1/0.06 2 | [113] |
| chrysin | 0.5/0.4 | ||
| apigenin | 1.2/1 | ||
| flavone | 8/5 | ||
| flavanone | 8/5 | ||
| quercetin | 12/10 | ||
| flavone | 10 1 | [117] | |
| 7-hydroxyflavone | 0.5 | ||
| 7,4′-dihydroxyflavone | 0.2 | ||
| flavanone | 8 | ||
| Catalase | Galangin | 46 | [67] |
| Kaempferol | 1.8 | ||
| Astragalin | 45 | ||
| Quercetin | 33 | ||
| Rutin | 36 | ||
| Myricetin | 0.014 | ||
| Catechin | 345 | ||
| Epigallocatechin | 16.6 | ||
| Epicatechin gallate | 0.029 | ||
| Epigallocatechin gallate | 0.33 | ||
| Apigenin | 49 | ||
| Luteolin | 56 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kejík, Z.; Kaplánek, R.; Masařík, M.; Babula, P.; Matkowski, A.; Filipenský, P.; Veselá, K.; Gburek, J.; Sýkora, D.; Martásek, P.; et al. Iron Complexes of Flavonoids-Antioxidant Capacity and Beyond. Int. J. Mol. Sci. 2021, 22, 646. https://doi.org/10.3390/ijms22020646
Kejík Z, Kaplánek R, Masařík M, Babula P, Matkowski A, Filipenský P, Veselá K, Gburek J, Sýkora D, Martásek P, et al. Iron Complexes of Flavonoids-Antioxidant Capacity and Beyond. International Journal of Molecular Sciences. 2021; 22(2):646. https://doi.org/10.3390/ijms22020646
Chicago/Turabian StyleKejík, Zdeněk, Robert Kaplánek, Michal Masařík, Petr Babula, Adam Matkowski, Petr Filipenský, Kateřina Veselá, Jakub Gburek, David Sýkora, Pavel Martásek, and et al. 2021. "Iron Complexes of Flavonoids-Antioxidant Capacity and Beyond" International Journal of Molecular Sciences 22, no. 2: 646. https://doi.org/10.3390/ijms22020646
APA StyleKejík, Z., Kaplánek, R., Masařík, M., Babula, P., Matkowski, A., Filipenský, P., Veselá, K., Gburek, J., Sýkora, D., Martásek, P., & Jakubek, M. (2021). Iron Complexes of Flavonoids-Antioxidant Capacity and Beyond. International Journal of Molecular Sciences, 22(2), 646. https://doi.org/10.3390/ijms22020646








