Genus Parkia: Phytochemical, Medicinal Uses, and Pharmacological Properties
Abstract
1. Introduction
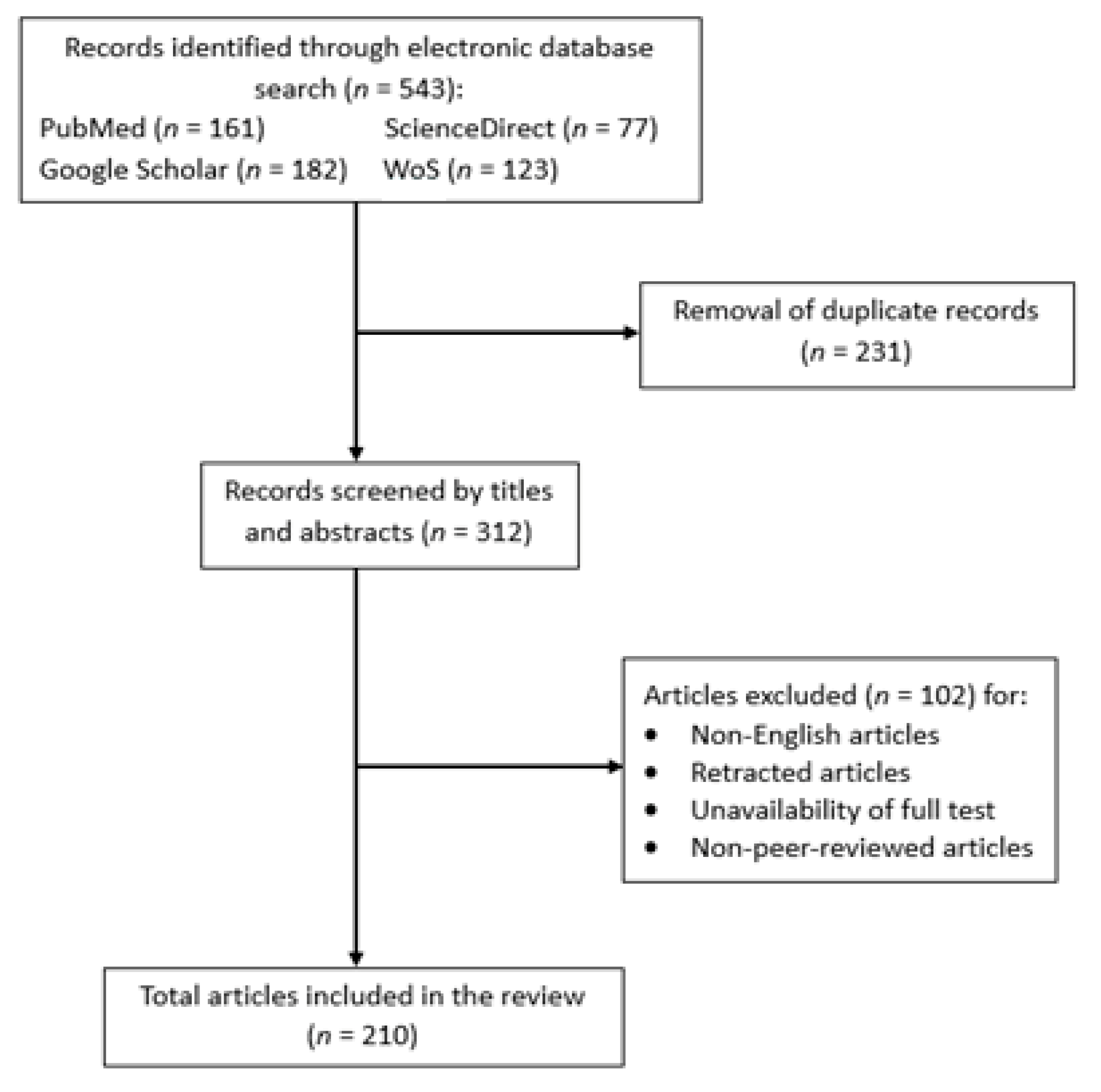
2. Methodology
3. Traditional Medicinal Uses
4. Phytochemistry of Genus Parkia
4.1. Polyphenolic Compounds
4.2. Terpenoid and Steroid
4.3. Miscellaneous Compounds
5. Pharmacological Activities of Parkia Species
5.1. Antimicrobial Activity
5.2. Antidiabetic Activity
5.3. Anticancer Activity
5.4. Antihypertensive Activity
5.5. Antidiarrheal Activity
5.6. Antiulcer Activity
5.7. Antianemic Activity
5.8. Anti-Inflammatory Activity
5.9. Antioxidant Activity
5.10. Other Pharmacological Activities
6. Toxicity
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Heymann, E.W.; Lüttmann, K.; Michalczyk, I.M.; Saboya, P.P.P.; Ziegenhagen, B.; Bialozyt, R. DNA fingerprinting validates seed dispersal curves from observational studies in the neotropical legume Parkia. PLoS ONE 2012, 7, e35480. [Google Scholar] [CrossRef] [PubMed]
- Orwa, C.; Mutua, A.; Kindt, R.; Jamnadass, R.; Simons, A. Agroforestree Database: A Tree Reference and Selection Guide, version 4; World Agroforestry Centre: Nairobi, Kenya, 2009. [Google Scholar]
- Luckow, M.; Hopkins, H.C.F. A cladistic analysis of Parkia (Leguminosae: Mimosoideae). Am. J. Bot. 1995, 82, 1300–1320. [Google Scholar] [CrossRef]
- Neill, D.A. Parkia nana (Leguminosae, Mimosoideae), a new species from the sub-Andean sandstone cordilleras of Peru. Novon A J. Bot. Nomencl. 2009, 19, 204–208. [Google Scholar] [CrossRef]
- Ching, L.S.; Mohamed, S. Alpha-tocopherol content in 62 edible tropical plants. J. Agric. Food Chem. 2001, 49, 3101–3105. [Google Scholar] [CrossRef] [PubMed]
- Suvachittanont, W.; Peutpaiboon, A. Lectin from Parkia speciosa seeds. Phytochemistry 1992, 31, 4065–4070. [Google Scholar] [CrossRef]
- Ogunyinka, B.I.; Oyinloye, B.E.; Osunsanmi, F.O.; Kappo, A.P.; Opoku, A.R. Comparative study on proximate, functional, mineral, and antinutrient composition of fermented, defatted, and protein isolate of Parkia biglobosa seed. Food Sci. Nutr. 2017, 5, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Alabi, D.A.; Akinsulire, O.R.; Sanyaolu, M.A. Qualitative determination of chemical and nutritional composition of Parkia biglobosa (Jacq.) Benth. Afr. J. Biotechnol. 2005, 4, 812–815. [Google Scholar] [CrossRef]
- Fetuga, B.L.; Babatunde, G.M.; Oyenuga, V.A. Protein quality of some unusual protein foodstuffs. Studies on the African locust-bean seed (Parkia filicoidea Welw.). Br. J. Nutr. 1974, 32, 27–36. [Google Scholar] [CrossRef]
- Hassan, L.G.; Umar, K.J. Protein and amino acids composition of African locust bean (Parkia biglobosa). Trop. Subtrop. Agroecosyst. 2005, 5, 45–50. [Google Scholar]
- Builders, M.; Alemika, T.; Aguiyi, J. Antimalarial Activity and isolation of phenolic compound from Parkia biglobosa. IOSR J. Pharm. Biol. Sci. 2014, 9, 78–85. [Google Scholar] [CrossRef]
- Ifesan, B.O.T.; Akintade, A.O.; Gabriel-Ajobiew, R.A.O. Physicochemical and nutritional properties of Mucuna pruriens and Parkia biglobosa subjected to controlled fermentation. Int. Food Res. J. 2017, 24, 2177–2184. [Google Scholar]
- Iheke, E.; Oshodi, A.; Omoboye, A.; Ogunlalu, O. Effect of fermentation on the physicochemical properties and nutritionally valuable minerals of locust bean (Parkia biglobosa). Am. J. Food Technol. 2017, 6, 379–384. [Google Scholar] [CrossRef]
- Abdullahi, I.N.; Chuwang, P.Z.; Anjorin, T.S.; Ikemefuna, H. Determination of Mineral Accumulation through Litter Fall of Parkia Biglobosa Jacq Benth and Vitellaria Paradoxa Lahm Trees in Abuja, Nigeria. Int. J. Sci. Res. Agric. Sci. 2015, 2, 0016–0021. [Google Scholar]
- Singh, N.P.; Gajurel, P.R.; Rethy, P. Ethnomedicinal value of traditional food plants used by the Zeliang tribe of Nagaland. Indian J. Tradit. Knowl. 2015, 14, 298–305. [Google Scholar]
- Mondal, P.; Bhuyan, N.; Das, S.; Kumar, M.; Borah, S.; Mahato, K. Herbal medicines useful for the treatment of diabetes in north-east India: A review. Int. J. Pharm. Biol. Sci. 2013, 3, 575–589. [Google Scholar]
- Boye, A.; Boampong, V.A.; Takyi, N.; Martey, O. Assessment of an aqueous seed extract of Parkia clappertoniana on reproductive performance and toxicity in rodents. J. Ethnopharmacol. 2016, 185, 155–161. [Google Scholar] [CrossRef]
- Sheikh, Y.; Maibam, B.C.; Talukdar, N.C.; Deka, D.C.; Borah, J.C. In vitro and in vivo anti-diabetic and hepatoprotective effects of edible pods of Parkia roxburghii and quantification of the active constituent by HPLC-PDA. J. Ethnopharmacol. 2016, 191, 21–28. [Google Scholar] [CrossRef]
- Singh, M.K. Potential of underutilized legume tree Parkia timoriana (DC.) Merr. In Eco-restoration of Jhum fallows of Manipur. J. Pharmacogn. Phytochem. 2019, 8, 1685–1687. [Google Scholar]
- Roosita, K.; Kusharto, C.M.; Sekiyama, M.; Fachrurozi, Y.; Ohtsuka, R. Medicinal plants used by the villagers of a Sundanese community in West Java, Indonesia. J. Ethnopharmacol. 2008, 115, 72–81. [Google Scholar] [CrossRef]
- Srisawat, T.; Suvarnasingh, A.; Maneenoon, K. Traditional medicinal plants notably used to treat skin disorders nearby Khao Luang mountain hills region, Nakhon si Thammarat, Southern Thailand. J. HerbsSpices Med. Plants 2016, 22, 35–56. [Google Scholar] [CrossRef]
- Aiyelaagbe, O.O.; Ajaiyeoba, E.O.; Ekundayo, O. Studies on the seed oils of Parkia biglobosa and Parkia bicolor. Plant Foods Hum. Nutr. 1996, 49, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Ajaiyeoba, E. 0 Phytochemical and antibacterial properties of Parkia biglobosa and Parkia bicolor leaf extracts. Afr. J. Biomed. Res. 2002, 5, 125–129. [Google Scholar] [CrossRef]
- Oladunmoye, M.K.; Kehinde, F.Y. Ethnobotanical survey of medicinal plants used in treating viral infections among Yoruba tribe of South Western Nigeria. Afr. J. Microbiol. Res. 2011, 5, 2991–3004. [Google Scholar] [CrossRef]
- Rathi, R.S.; Misra, A.K.; Somnath, R.; Verma, S.K.; Singh, S.K. Potential of a lesser known tree species Parkia roxburghii G. Don of North East India. Indian For. 2012, 138, 476–479. [Google Scholar]
- Ong, H.C.; Ahmad, N.; Milow, P. Traditional Medicinal Plants Used by the Temuan Villagers in Kampung Tering, Negeri Sembilan, Malaysia. Stud. Ethno-Med. 2011, 5, 169–173. [Google Scholar] [CrossRef]
- Ong, H.C.; Chua, S.; Milow, P. Ethno-medicinal plants used by the Temuan villagers in Kampung Jeram Kedah, Negeri Sembilan, Malaysia. Stud. Ethno-Med. 2011, 5, 95–100. [Google Scholar] [CrossRef]
- Fotie, J.; Nkengfack, A.E.; Peter, M.G.; Heydenreich, M.; Fomum, Z.T. Chemical constituents of the ethyl acetate extracts of the stem bark and fruits of Dichrostachys cinerea and the roots of Parkia bicolor. Bull. Chem. Soc. Ethiop. 2004, 18, 111–115. [Google Scholar] [CrossRef]
- Kouadio, F.; Kanko, C.; Juge, M.; Grimaud, N.; Jean, A.; N’Guessan, Y.T.; Petit, J.Y. Analgesic and antiinflammatory activities of an extract from Parkia biglobosa used in traditional medicine in the ivory coast. Phytother. Res. 2000, 14, 635–637. [Google Scholar] [CrossRef]
- Ong, H.C.; Zuki, R.M.; Milow, P. Traditional Knowledge of Medicinal Plants among the Malay Villagers in Kampung Mak Kemas, Terengganu, Malaysia. Stud. Ethno-Med. 2011, 2011, 175–185. [Google Scholar] [CrossRef]
- Millogo-Kone, H.; Guissou, I.P.; Nacoulma, O.; Traore, A.S. Antimicrobial effects of the stem bark extracts of Parika biglobosa (Jacq.)Benth. on Shigellae. Afr. J. Tradit. Complementary Altern. Med. 2007, 4, 392–396. [Google Scholar] [CrossRef]
- Enujiugha, V.N. The antioxidant and free radical-scavenging capacity of phenolics from African locust bean seeds (Parkia biglobosa). Adv. Food Sci. 2010, 32, 88–93. [Google Scholar]
- Gernah, D.I.; Inyang, C.U.; Ezeora, N.L. Incubation and fermentation of African locust beans (Parkia biglobosa) in production of ‘dawadawa’. J. Food Process. Preserv. 2007, 31, 227–239. [Google Scholar] [CrossRef]
- El-Mahmood, A.M.; Ameh, J.M. In vitro antibacterial activity of Parkia biglobosa (Jacq.) root bark extract against some microorganisms associated with urinary tract infections. Afr. J. Biotechnol. 2007, 6, 1272–1275. [Google Scholar] [CrossRef]
- Adaramola, T.F.; Ariwaodo, J.O.; Adeniji, K.A. Distribution, phytochemistry and antioxidant properties of the genus Parkia R.br. (mimosaceae) in Nigeria. Int. J. Pharmacogn. Phytochem. Res. 2012, 4, 172–178. [Google Scholar]
- Builders, M. Toxicity studies of the extracts of Parkia biglobosa Stem Bark in Rats. Br. J. Pharm. Res. 2012, 2, 1–16. [Google Scholar] [CrossRef]
- Chhikara, N.; Devi, H.R.; Jaglan, S.; Sharma, P.; Gupta, P.; Panghal, A. Bioactive compounds, food applications and health benefits of Parkia speciosa (stinky beans): A review. Agric. Food Secur. 2018, 7, 1–9. [Google Scholar] [CrossRef]
- Udobi, C.E.; Onaolapo, J.A. Phytochemical analysis and antibacterial evaluation of the leaf stem bark and root of the African locust bean (Parkia biglobosa). J. Med. Plants Res. 2009, 3, 338–344. [Google Scholar]
- Ezema, B.E.; Eze, F.U.; Ezeofor, C.C. Phytochemical and antibacterial studies of eastern nigerian mistletoe (Loranthus micranthus) parasitic on Pentacletra macrophylla and Parkia biglobosa. Int. J. Pharm. Technol. Res. 2016, 9, 360–365. [Google Scholar]
- Fred-Jaiyesimi, A.A.; Abo, K.A. Hypoglycaemic effects of Parkia biglobosa (Jacq) Benth seed extract in glucose-loaded and NIDDM rats. Int. J. Biol. Chem. Sci. 2009, 3, 545–550. [Google Scholar] [CrossRef]
- Mohan, V.R.; Janardhanan, K. Chemical and nutritional evaluation of raw seeds of the tribal pulses Parkia roxburghii G. Don. and Entada phaseoloides (L.) Merr. Int. J. Food Sci. Nutr. 1993, 44, 47–53. [Google Scholar] [CrossRef]
- Dinda, B.; Chandra Mohanta, B.; Debnath, S.; Ghosh, B.; Arima, S.; Sato, N.; Harigaya, Y. Iridoid glucosides from leaves and stem barks of Parkia javanica. J. Asian Nat. Prod. Res. 2009, 11, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Abioye, E.O.; Akinpelu, D.A.; Aiyegoro, O.A.; Adegboye, M.F.; Oni, M.O.; Okoh, A.I. Preliminary phytochemical screening and antibacterial properties of crude stem bark extracts and fractions of Parkia biglobosa (Jacq.). Molecules 2013, 18, 8485–8499. [Google Scholar] [CrossRef] [PubMed]
- Chanu, K.V.; Geeta Devi, L.; Kumar Srivastava, S.; Telang, A.; Khangembam Victoria Chanu, C.; Thakuria, D.; Kataria, M. Phytochemical analysis and evaluation of anticancer activity of Parkia javanica seeds. Pharma Innov. J. 2018, 7, 305–311. [Google Scholar]
- Tijani, A.Y.; Okhale, S.E.; Salawu, T.A.; Onigbanjo, H.O.; Obianodo, L.A.; Akingbasote, J.A.; Salawu, O.A.; Okogun, J.I.; Kunle, F.O.; Emeje, M. Antidiarrhoeal and antibacterial properties of crude aqueous stem bark extract and fractions of Parkia biglobosa (Jacq) R. Br. Ex G. Don. Afr. J. Pharm. Pharmacol. 2009, 3, 347–353. [Google Scholar]
- Awotedu, O.L.; Ogunbamowo, P.O.; Emmanuel, I.B.; Lawal, I.O. Phytominerals and Phytochemical Studies of Azadiracthta indica, Leea guineensis and Parkia biglobosa Leaves. Int. Ann. Sci. 2018, 6, 28–34. [Google Scholar] [CrossRef]
- Fayinminnu, O.O.; Adeniyi, O.O.; Alabi, O.Y.; Omobusuyi, D.O. Potentials of Aqueous Extract of Pod Husk Parkia biglobosa (Jacq.) Benth as a Biopesticide in Okra (Abelmoschus esculentus (L.) Moench) Production. J. Agric. Ecol. Res. Int. 2017, 1–12. [Google Scholar] [CrossRef]
- Sani, U.M. Phytochemical screening and antifeedant activity of the seed extracts of Parkia biglobosa against cowpea vean (Vigna unguiculata) storage pest (Callosobruchus maculatus). Int. J. Innov. Sci. Eng. Technol. 2014, 3, 15991–15995. [Google Scholar] [CrossRef]
- Soetan, K.O.; Lasisi, O.T.; Agboluaje, A.K. Comparative assessment of in-vitro anthelmintic effects of the aqueous extracts of the seeds and leaves of the African locust bean (Parkia biglobosa) on bovine nematode eggs. J. Cell Anim. Biol. 2011, 5, 109–112. [Google Scholar]
- Iyamu, M.I.; Ekozien, M.I.; Omoigberale, M.N.O. Phytochemical screening and antibacterial activity of the stem back of African Locust bean plant (Parkia Filicoidea Welw.). Glob. J. Biol. Agric. Health Sci. 2014, 3, 36–43. [Google Scholar]
- Salam, S.; Jamir, N.S.; Singh, P.K. Traditional uses of medicinal plants by the Tangkhul–Naga tribe in Manipur, India. Pleione 2009, 3, 157–162. [Google Scholar]
- Salam, J.S.; Salam, P.; Potshangbam, K.S.; Kumar, D.B. Effect of processing methods on secondary metabolites and enzyme inhibitors in different developmental stages of Parkia roxburghii G. Don pods. Am. J. Food Technol. 2014, 9, 89–96. [Google Scholar]
- Costa, B.A.; de Oliveira, J.M.; Sales, P.A.; Lira, S.R.D.S.; Silva, S.M.D.S.; Costa, L.M.; Muratori, M.; Costa, A.P. Systemic and reproductive toxicity induced by Parkia platycephala ethanolic extract in female Wistar rats. Braz. J. Pharmacogn. 2013, 23, 920–926. [Google Scholar] [CrossRef]
- SáSantos, M.M.; da Silva, F.M.P.; da Silva, J.F.M.; Pimenta, R.S. Phytochemistry and antibacterial activity of aqueous and hydroalcoholic extracts of three medicinal plants against food pathogens. Acta Sci. Biol. Sci. 2018, 40, 1–6. [Google Scholar] [CrossRef]
- Lawal, I.O.; Uzokwe, N.E.; Igboanugo, A.B.I.; Adio, A.F.; Awosan, E.A.; Nwogwugwu, J.O.; Faloye, B.; Olatunji, B.P.; Adesoga, A.A. Ethno medicinal information on collation and identification of some medicinal plants in Research Institutes of South-west Nigeria. Afr. J. Pharm. Pharmacol. 2010, 4, 1–7. [Google Scholar]
- Henry, S.G.; Francis, A.; Kofi, A. Ethnobotanical survey of medicinal plants used for the treatment of diarrhoea and skin ulcer in the Brong Ahafo region of Ghana. J. Med. Plants Res. 2013, 7, 3280–3285. [Google Scholar] [CrossRef]
- Campbell-Platt, G. African locust bean (Parkia species) and its west african fermented food product, dawadawa. Ecol. Food Nutr. 1980, 9, 123–132. [Google Scholar] [CrossRef]
- Igoli, J.O.; Ogaji, O.G.; Tor-Anyiin, T.A.; Igoli, N.P. Traditional medicine practice amongst the Igede people of Nigeria. Part II. Afr. J. Tradit. Complementary Altern. Med. 2005, 2, 134–152. [Google Scholar] [CrossRef]
- Agunu, A.; Yusuf, S.; Andrew, G.O.; Zezi, A.U.; Abdurahman, E.M. Evaluation of five medicinal plants used in diarrhoea treatment in Nigeria. J. Ethnopharmacol. 2005, 101, 27–30. [Google Scholar] [CrossRef]
- Asuzu, I.U.; Harvey, A.L. The antisnake venom activities of Parkia biglobosa (Mimosaceae) stem bark extract. Toxicon 2003, 42, 763–768. [Google Scholar] [CrossRef]
- Karou, S.D.; Tchacondo, T.; Djikpo Tchibozo, M.A.; Abdoul-Rahaman, S.; Anani, K.; Koudouvo, K.; Batawila, K.; Agbonon, A.; Simpore, J.; de Souza, C. Ethnobotanical study of medicinal plants used in the management of diabetes mellitus and hypertension in the Central Region of Togo. Pharm. Biol. 2011, 49, 1286–1297. [Google Scholar] [CrossRef]
- Grønhaug, T.E.; Glæserud, S.; Skogsrud, M.; Ballo, N.; Bah, S.; Diallo, D.; Paulsen, B.S. Ethnopharmacological survey of six medicinal plants from Mali, West-Africa. J. Ethnobiol. Ethnomed. 2008, 4, 26. [Google Scholar] [CrossRef] [PubMed]
- Abo, K.A.; Fred-Jaiyesimi, A.A.; Jaiyesimi, A.E.A. Ethnobotanical studies of medicinal plants used in the management of diabetes mellitus in South Western Nigeria. J. Ethnopharmacol. 2008, 115, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Pare, D.; Hilou, A.; Ouedraogo, N.; Guenne, S. Ethnobotanical study of medicinal plants used as anti-obesity remedies in the nomad and hunter communities of Burkina Faso. Medicines 2016, 3, 9. [Google Scholar] [CrossRef] [PubMed]
- Millogo-Kone, H.; Guissoe, P.I.; Nacoulma, O.; Traore, A.S. Study of the antibacterial activity of the stem bark and leaf extracts of Parkia biglobosa (Jacq.) Benth. on Satphylococcus aureus. Afr. J. Tradit. Complementary Altern. Med. 2006, 3, 74–78. [Google Scholar] [CrossRef]
- Quansah, L.; Mahunu, G.K.; Tahir, H.E.; Mariod, A.A. Parkia biglobosa: Phytochemical Constituents, Bioactive Compounds, Traditional and Medicinal Uses. In Wild Fruits: Composition, Nutritional Value and Products; Springer: Berlin/Heidelberg, Germany, 2019; pp. 271–284. [Google Scholar]
- Abreu, P.M.; Martins, E.S.; Kayser, O.; Bindseil, K.U.; Siems, K.; Seemann, A.; Frevert, J. Antimicrobial, antitumor and antileishmania screening of medicinal plants from Guinea-Bissau. Phytomedicine 1999, 6, 187–195. [Google Scholar] [CrossRef]
- Rupesh, P.; Pal, S.C.; Pavani, A.; Gadge, M.S. Quantitave estimation of the active constituents of Parkia biglandulosa by using HPTLC and FTIR. Int. J. Pharma Bio Sci. 2010, 1, 315–332. [Google Scholar]
- Khond, M.; Bhosale, J.D.; Arif, T.; Mandal, T.K.; Padhi, M.M.; Dabur, R. Screening of some selected medicinal plants extracts for in-vitro antimicrobial activity. Middle-East J. Sci. Res. 2009, 4, 271–278. [Google Scholar]
- Pingale, R.; Pokharkar, D.; Phadatare, S.P.; Gorle, A. Pharmacognostic Evaluation of Parkia biglandulosa bark. Int. J. Pharm. Phytochem. Res. 2016, 8, 1160–1163. [Google Scholar]
- Banwo, G.O.; Abdullahi, I.; Duguryil, M. The antimicrobial activity of the stem-bark and leaf of Parkia clappertoniana Keay family Leguminosae against selected microorganisms. Niger. J. Pharm. Res. 2004, 3, 16–22. [Google Scholar] [CrossRef]
- Nwodo, N.J.; Ibezim, A.; Ntie-Kang, F.; Adikwu, M.U.; Mbah, C.J. Anti-trypanosomal activity of Nigerian plants and their constituents. Molecules 2015, 20, 7750–7771. [Google Scholar] [CrossRef]
- Lawal, M.S.; Sani, A.M.; Dangmwan, D.S.; Yahaya, U. Antimicrobial potentials of Parkia clappertoniana Jacq, Boswellia dalzielli hutch and Carica papaya L. ethanolic extract on multi-drug resistant Diarrheal salmonallae and Shigellae Bacteria. Biochem. Mol. Biol. 2016, 1, 27. [Google Scholar]
- Muazu, J.; Kaita, M.H. A review of traditional plants used in the treatment of epilepsy amongst the Hausa/Fulani tribes of northern Nigeria. Afr. J. Tradit. Complementary Altern. Med. 2008, 5, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Boye, A. Nephroprotective and curative assessment of an aqueous seed extract of Parkia clappertoniana keay in gentamicin-induced renal damage in Sprague-dawley rats. Eur. J. Med. Plants 2014, 4, 234–248. [Google Scholar] [CrossRef]
- Patrick-Iwuanyanwu, K.C.; Wegwu, M.O.; Okiyi, J.K. Hepatoprotective effects of African locust bean (Parkia clappertoniana) and negro pepper (Xylopia aethiopica) in CCl4-induced liver damage in wistar albino rats. Int. J. Pharmacol. 2010, 6, 744–749. [Google Scholar] [CrossRef]
- Obata, O.O.; Aigbokhan, E.I. Ethnobotanical practices among the people of Okaakoko, Nigeria. Plant Arch. 2012, 12, 627–638. [Google Scholar]
- Van Andel, T.; Behari-Ramdas, J.; Havinga, R.; Groenendijk, S. The medicinal plant trade in Suriname. Ethnobot. Res. Appl. 2007, 5, 351–372. [Google Scholar] [CrossRef]
- Ferreira, A.B.; Ming, L.C.; Haverroth, M.; Daly, D.C.; Caballero, J.; Ballesté, A.M. Plants used to treat malaria in the regions of Rio Branco-Acre state and southern Amazonas state—Brazil. Int. J. Phytocosmetics Nat. Ingred. 2015, 2, 9. [Google Scholar] [CrossRef]
- Mitra, R.; Orbell, J.; Muralitharan, M. Medicinal plants of Malaysia. Asia Pac. Biotech News 2007, 11, 105–110. [Google Scholar] [CrossRef]
- Siew, Y.Y.; Zareisedehizadeh, S.; Seetoh, W.G.; Neo, S.Y.; Tan, C.H.; Koh, H.L. Ethnobotanical survey of usage of fresh medicinal plants in Singapore. J. Ethnopharmacol. 2014, 155, 1450–1466. [Google Scholar] [CrossRef]
- Ripen, J.E.; Noweg, G.T. Economic valuation of medicinal plants in Jagoi Area, Bau, Malaysia. Procedia Soc. Behav. Sci. 2016, 224, 124–131. [Google Scholar] [CrossRef]
- Eswani, N.; Kudus, K.A.; Nazre, M.; Noor, A.G.A.; Ali, M. Medicinal plant diversity and vegetation analysis of logged over hill forest of Tekai Tembeling Forest Reserve, Jerantut, Pahang. J. Agric. Sci. 2010, 2, 189. [Google Scholar] [CrossRef]
- Sonia, N.; Dsouza, M.R. Alisha Pharmacological evaluation of Parkia speciosa Hassk for antioxidant, anti-inflammatory, anti-diabetic and anti-microbial activities in vitro. Int. J. Life Sci. Spec. Issue 2018, 11, 49–59. [Google Scholar]
- Bahtiar, A.; Vichitphan, K.; Han, J. Leguminous plants in the Indonesian Archipelago: Traditional uses and secondary metabolites. Nat. Prod. Commun. 2017, 12, 461–472. [Google Scholar] [CrossRef]
- Batoro, J.; Siswanto, D. Ethnomedicinal survey of plants used by local society in Poncokusumo district, Malang, East Java Province, Indonesia. Asian J. Med Biol. Res. 2017, 3, 158–167. [Google Scholar] [CrossRef]
- Samuel, A.J.S.J.; Kalusalingam, A.; Chellappan, D.K.; Gopinath, R.; Radhamani, S.; Husain, H.A.; Muruganandham, V.; Promwichit, P. Ethnomedical survey of plants used by the Orang Asli in Kampung Bawong, Perak, West Malaysia. J. Ethnobiol. Ethnomed. 2010, 6, 5. [Google Scholar] [CrossRef]
- Rai, P.K.; Lalramnghinglova, H. Ethnomedicinal plant resources of Mizoram, India: Implication of traditional knowledge in health care system. Ethnobot. Leafl. 2010, 2010, 6. [Google Scholar]
- Irvine, F.R. Woody Plants of Ghana; Oxford University Press: England, UK, 1961. [Google Scholar]
- Phumthum, M.; Balslev, H. Thai ethnomedicinal plants used for diabetes treatment. OBM ICM 2018, 3, 1–25. [Google Scholar] [CrossRef]
- Khumbongmayum, A.; Khan, M.; Tripathi, R. Ethnomedicinal plants in the sacred groves of Manipur. Indian J. Tradit. Knowl. (IJTK) 2005, 4, 21–32. [Google Scholar]
- Bhardwaj, S.; Gakhar, S.K. Ethnomedicinal plants used by the tribals of Mizoram to cure cuts & wounds. Indian J. Tradit. Knowl. 2005, 4, 75–80. [Google Scholar]
- Jamal, J.A.; Ghafar, Z.A.; Husain, K. Medicinal plants used for postnatal care in Malay traditional medicine in the Peninsular Malaysia. Pharmacogn. J. 2011, 3, 15–24. [Google Scholar] [CrossRef]
- Nanda, Y.; Singson, N.; Rao, A.N. Ethnomedicinal plants of Thadou tribe of Manipur (India)-1. Pleione 2013, 7, 138–145. [Google Scholar]
- Lalmuanpuii, J.; Rosangkima, G.; Lamin, H. Ethno-medicinal practices among the Mizo ethnic group in Lunglei district, Ethno-medicinal practices among the Mizo ethnic group in Lunglei district, Mizoram. Sci. Vis. 2013, 12, 24–34. [Google Scholar]
- Khan, M.H.; Yadava, P.S. Antidiabetic plants used in Thoubal district of Manipur, Northeast India. Indian J. Tradit. Knowl. 2010, 9, 510–514. [Google Scholar]
- Mullick, J.B.; Majumdar, T.; Reddy, K.V.R.; Mukherjee, S.; Sil, S.K. Activity of the medicinal plant Parkia Javanica against multidrug-resistant Neisseria gonorrhoeae and other clinical isolates. Asian J. Pharm. Clin. Res. 2019, 12, 83–86. [Google Scholar] [CrossRef]
- Quattrocchi, U. CRC World Dictionary of Medicinal and Poisonous Plants: Common Names, Scientific Names, Eponyms, Synonyms, and Etymology (5 Volume Set); CRC Press: Boca Raton, FL, USA, 2012; ISBN 142008044X. [Google Scholar]
- Das, A.; Das, M.C.; Sandhu, P.; Das, N.; Tribedi, P.; De, U.C.; Akhter, Y.; Bhattacharjee, S. Antibiofilm activity of Parkia javanica against Pseudomonas aeruginosa: A study with fruit extract. Rsc Adv. 2017, 7, 5497–5513. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Ovidi, E.; Tiezzi, A.; Craker, L. Phytochemical and Pharmacological Properties of Medicinal Plants from Uzbekistan: A Review. J. Med. Act. Plants 2016, 5, 59–75. [Google Scholar] [CrossRef]
- Saxena, M.; Saxena, J.; Nema, R.; Singh, D.; Gupta, A. Phytochemistry of medicinal plants. J. Pharmacogn. Phytochem. Phytochem. 2013, 1, 168–182. [Google Scholar] [CrossRef]
- Tariq, A.L.; Reyaz, A.L. Significances and importance of phytochemical present in Terminalia chebula. Int. J. Drug Dev. Res. 2013, 5, 256–262. [Google Scholar]
- Wadood, A.; Ghufran, M.; Jamal, S.B.; Naeem, M.; Khan, A.; Ghaffar, R. Phytochemical analysis of medicinal plants occurring in local area of Mardan. Biochem. Anal. Biochem. 2013, 2. [Google Scholar] [CrossRef]
- Ahmad, N.I.; Rahman, S.A.; Leong, Y.H.; Azizul, N.H. A review on the phytochemicals of Parkia speciosa, stinky beans as potential phytomedicine. J. Food Sci. Nutr. Res. 2019, 2, 151–173. [Google Scholar] [CrossRef]
- Sikolia, S.F.; Omondi, S. Phytochemical Analysis of Some Selected Plants and Families in the University Botanic Garden of Maseno, Kenya. IOSR J. Pharm. Biol. Sci. 2017, 12, 31–38. [Google Scholar] [CrossRef]
- Tala, V.R.S.; Da Silva, V.C.; Rodrigues, C.M.; Nkengfack, A.E.; Dos Santos, L.C.; Vilegas, W. Characterization of proanthocyanidins from Parkia biglobosa (Jacq.) G. Don. (Fabaceae) by flow injection analysis—electrospray ionization ion trap tandem mass spectrometry and liquid chromatography/electrospray ionization mass spectrometry. Molecules 2013, 18, 2803–2820. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.J.; Ang, L.H.; Ng, L.T. Antioxidant activities and polyphenolic constituents of bitter bean Parkia speciosa. Int. J. Food Prop. 2014, 17, 1977–1986. [Google Scholar] [CrossRef]
- Loukrakpam, B.; Rajendran, A.; Chyne, D.A.L.; Longvah, T. 12th IFDC 2017 Special Issue—Nutrient and phytonutrient profiles of some indigenous vegetables of Manipur, Northeast India. J. Food Compos. Anal. 2019, 79, 12–22. [Google Scholar] [CrossRef]
- Mohammad, M.; Garba, M.A.; Haruna, A.; Jimoh, A.A. Characterization of naringenin from the fruit pulp extract of Parkia biglobosa (FABACEAE). Fuw Trends Sci. Technol. J. 2018, 4, 918–920. [Google Scholar]
- Dinda, B.; Mohanta, B.C.; Ghosh, P.; Sato, N.; Harigaya, Y. ChemInform Abstract: Chemical Constituents of Parkia javanica, Alocasia indica and Premna latifolia. ChemInform 2011, 42. [Google Scholar] [CrossRef]
- Tringali, C.; Spatafora, C.; Longo, O.D. Bioactive constituents of the bark of Parkia biglobosa. Fitoterapia 2000, 71, 118–125. [Google Scholar] [CrossRef]
- Kamisah, Y.; Zuhair, J.S.F.; Juliana, A.H.; Jaarin, K. Parkia speciosa empty pod prevents hypertension and cardiac damage in rats given N(G)-nitro-L-arginine methyl ester. Biomed. Pharmacother. 2017, 96, 291–298. [Google Scholar] [CrossRef]
- Adewoye, R.O.; Ajayi, O.O. Flavonols, flavones and tannins of Parkia clapperoniana. J. Am. Leather Chem. Assoc. (USA) 1988, 83, 153–156. [Google Scholar]
- Adewoye, R.O.; Ajayi, O.O. Anthocyanidins of Parkia clappertoniana. J. Soc. Leather Technol. Chem. 1989, 73, 120–121. [Google Scholar]
- Lemmich, E.; Adewunmi, C.O.; Furu, P.; Kristensen, A.; Larsen, L.; Olsen, C.E. 5-Deoxyflavones from Parkia clappertoniana. Phytochemistry 1996, 42, 1011–1013. [Google Scholar] [CrossRef]
- Ouoba, L.I.I.; Diawara, B.; Annan, N.T.; Poll, L.; Jakobsen, M. Volatile compounds of Soumbala, a fermented African locust bean (Parkia biglobosa) food condiment. J. Appl. Microbiol. 2005, 99, 1413–1421. [Google Scholar] [CrossRef] [PubMed]
- Mohd Azizi, C.Y.; Salman, Z.; Nik Norulain, N.; Mohd Omar, A. Extraction and identification of compounds from Parkia Speciosa seeds by supercritical carbon dioxide. J. Chem. Nat. Resour. Eng. 2008, 2, 153–163. [Google Scholar]
- Bitchi, M.B.; Magid, A.A.; Yao-Kouassi, P.A.; Kabran, F.A.; Harakat, D.; Martinez, A.; Morjani, H.; Tonzibo, F.Z.; Voutquenne-Nazabadioko, L. Triterpene saponins from the roots of Parkia bicolor A. Chev. Fitoterapia 2019, 137, 104264. [Google Scholar] [CrossRef] [PubMed]
- Rahman, N.N.N.A.; Zhari, S.; Sarker, M.Z.I.; Ferdosh, S.; Yunus, M.A.C.; Kadir, M.O.A. Profile of Parkia speciosa hassk metabolites extracted with SFE using FTIR-PCA method. J. Chin. Chem. Soc. 2012, 59, 507–514. [Google Scholar] [CrossRef]
- Jamaluddin, F.; Mohamed, S.; Lajis, M.N. Hypoglycaemic effect of Parkia speciosa seeds due to the synergistic action of β-sitosterol and stigmasterol. Food Chem. 1994, 49, 339–345. [Google Scholar] [CrossRef]
- Akintayo, E.T. Characteristics and composition of Parkia biglobbossa and Jatropha curcas oils and cakes. Bioresour. Technol. 2004, 92, 307–310. [Google Scholar] [CrossRef]
- Olatunya, A.M.; Omojola, A.; Akinpelu, K.; Akintayo, E.T. Vitamin E, Phospholipid, and Phytosterol Contents of Parkia biglobosa and Citrullus colocynthis Seeds and Their Potential Applications to Human Health. Prev. Nutr. Food Sci. 2019, 24, 338–343. [Google Scholar] [CrossRef]
- Jamaluddin, F.; Mohameda, S.; Lajis, M.N. Hypoglycaemic effect of Stigmast-4-en-3-one, from Parkia speciosa empty pods. Food Chem. 1995, 54, 9–13. [Google Scholar] [CrossRef]
- Salman, Z.; Mohd Azizi, C.; Nik Norulaini, N.; Mohd Omar, A. Gas chromatography/time-of-flight mass spectrometry for identification of compounds from Parkia speciosa seeds extracted by supercritical carbon dioxide. In Proceedings of the First International Conference on Natural Resources Engineering & Technology, Putrajaya, Malaysia, 24–25 July 2006; pp. 112–120. [Google Scholar]
- Frérot, E.; Velluz, A.; Bagnoud, A.; Delort, E. Analysis of the volatile constituents of cooked petai beans (Parkia speciosa) using high-resolution GC/ToF–MS. Flavour Fragr. J. 2008, 23, 434–440. [Google Scholar] [CrossRef]
- Gmelin, R.; Susilo, R.; Fenwick, G.R. Cyclic polysulphides from Parkia speciosa. Phytochemistry 1981, 20, 2521–2523. [Google Scholar] [CrossRef]
- Miyazawa, M.; Osman, F. Headspace constituents of Parkia speciosa seeds. Nat. Prod. Lett. 2001, 15, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Tocmo, R.; Liang, D.; Wang, C.; Poh, J.; Huang, D. Organosulfide profile and hydrogen sulfide-releasing capacity of stinky bean (Parkia speciosa) oil: Effects of pH and extraction methods. Food Chem. 2016, 190, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Adeyeye, E.I. The effect of fermentation on the dietary quality of lipids from African locust bean (Parkia biglobosa) seeds. Elixir Food Sci. 2013, 58, 14912–14922. [Google Scholar]
- Olatunya, A.M.; Akintayo, C.O.; Akintayo, E.T. Determination of qualitative and quantitative fatty acid composition of Parkia biglobbossa seed oil using two different analytical techniques. Int. J. Adv. Res. 2015, 3, 463–473. [Google Scholar]
- Millogo-Kone, H.; Guissou, I.; Nacoulma, O.; Traore, A. Comparative study of leaf and stem bark extracts of Parkia biglobosa against enterobacteria. Afr. J. Tradit. Complementary Altern. Med. 2008, 5, 238–243. [Google Scholar] [CrossRef][Green Version]
- Yahaya, U.; Abubakar, S.; Salisu, A. Antifungal activity of Parkia biglobosa extract on pathogenic strain of Candida albicans. J. Appl. Sci. 2019, 19, 235–240. [Google Scholar] [CrossRef]
- Bukar, A.; Uba, A.; Oyeyi, T.I. Phytochemical analysis and antimicrobial activity of Parkia biglobosa (Jacq.) Benth. extracts againt some food--borne microrganisms. Adv. Environ. Biol. 2010, 74–80. [Google Scholar]
- Joshua, E.; Joshua, E.; Ifeanyichukwu, I.; Chika, E.; Okoro, N.; Carissa, D.; Emmanuel, N.; Chukwuka, A. In vitro evaluation of antibacterial activity of Parkia biglobosa, Hymenocardia acida and Zanthoxylum zanthoxyloides extracts on pathogenic Staphylococcus aureus Isolates. Int. J. Life Sci. 2016, 5, 72–77. [Google Scholar]
- Nounagnon, M.; Dah-Nouvlessounon, D.; N’tcha, C.; Nanoukon, C.; Assogba, F.; Lalèyè, F.O.A.; Baba-Moussa, L. Phytochemical composition, antimicrobial and cytotoxicity activities of Parkia biglobosa (Jacq) benth extracts from Benin. J. Pharmacogn. Phytochem. 2017, 6, 35–42. [Google Scholar]
- Millogo-Kone, H.; Lompo, M.; Kini, F.; Asimi, S.; Guissou, I.P.; Nacoulma, O. Evaluation of flavonoids and total phenolic contents of stem bark and leaves of Parkia biglobosa (Jacq.) Benth.(Mimosaceae)-free radical scavenging and antimicrobial activities. Res. J. Med Sci. 2009, 3, 70–74. [Google Scholar]
- Obajuluwa, A.F.; Onaolapo, J.A.; Oyi, A.R.; Olayinka, B.O. Susceptibility profile of methicillin-resistant Staphylococcus aureus (MRSA) isolates to antibiotics and methanolic extracts of Parkia biglobosa (Jacq.) Benth. Br. J. Pharm. Res. 2013, 3, 587–596. [Google Scholar] [CrossRef]
- Dosumu, O.O.; Oluwaniyi, O.O.; Awolola, G.V.; Oyedeji, O.O. Nutritional composition and antimicrobial properties of three Nigerian condiments. Niger. Food J. 2012, 30, 43–52. [Google Scholar] [CrossRef]
- Osemwegie, O.O.; Dahunsi, S.O. In-vitro effects of aqueous and ethanolic extracts of Parkia biglobossa (Jacq.) Benth on selected microorganisms. Niger. J. Biotechnol. 2015, 11–20. [Google Scholar] [CrossRef]
- Igwo-Ezikpe, M.N.; Ogbunugafor, H.A.; Gureje, A.P.; Ezeonwumelu, I.J. Phytochemical, antioxidant and antimicrobial properties of Parkia biglobosa (African Locust Bean) pods. Bioscientist 2013, 1, 182–191. [Google Scholar]
- Farias, D.F.; Souza, T.M.; Viana, M.P.; Soares, B.M.; Cunha, A.P.; Vasconcelos, I.M.; Ricardo, N.M.P.S.; Ferreira, P.M.P.; Melo, V.M.M.; Carvalho, A.F.U. Antibacterial, antioxidant, and anticholinesterase activities of plant seed extracts from Brazilian semiarid region. Biomed Res. Int. 2013, 2013, 510736. [Google Scholar] [CrossRef]
- Silva, R.R.S.; Silva, C.R.; Santos, V.F.; Barbosa, C.R.S.; Muniz, D.F.; Santos, A.L.E.; Santos, M.H.C.; Rocha, B.A.M.; Batista, K.L.R.; Costa-Júnior, L.M.; et al. Parkia platycephala lectin enhances the antibiotic activity against multi-resistant bacterial strains and inhibits the development of Haemonchus contortus. Microb. Pathog. 2019, 135, 103629. [Google Scholar] [CrossRef]
- Musa, N.; Wei, L.S.; Seng, C.T.; Wee, W.; Leong, L.K. Potential of Edible Plants as Remedies of Systemic Bacterial Disease Infection in Cultured Fish. Glob. J. Pharmacol. 2008, 2, 31–36. [Google Scholar]
- Sakunpak, A.; Panichayupakaranant, P. Antibacterial activity of Thai edible plants against gastrointestinal pathogenic bacteria and isolation of a new broad spectrum antibacterial polyisoprenylated benzophenone, chamuangone. Food Chem. 2012, 130, 826–831. [Google Scholar] [CrossRef]
- Fatimah, I. Green synthesis of silver nanoparticles using extract of Parkia speciosa Hassk pods assisted by microwave irradiation. J. Adv. Res. 2016, 7, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, V.; Vasanthi, S.; Shalini, S.; Shah, S.A.A.; Tripathy, M.; Paliwal, N. Green synthesis, characterization, antibacterial, antioxidant and photocatalytic activity of Parkia speciosa leaves extract mediated silver nanoparticles. Results Phys. 2019, 15, 102565. [Google Scholar] [CrossRef]
- Kawamura, F.; Ramle, S.F.M.; Sulaiman, O.; Hashim, R.; Ohara, S. Antioxidant and antifungal activities of extracts from 15 selected hardwood species of Malaysian timber. Eur. J. Wood Wood Prod. 2011, 69, 207–212. [Google Scholar] [CrossRef]
- Hasim, H.; Faridah, D.N. Antibacterial activity of Parkia speciosa Hassk. peel to Escherichia coli and Staphylococcus aureus bacteria. J. Chem. Pharm. Res. 2015, 7, 239–243. [Google Scholar]
- Thongbam, P.D.; Shakuntala, I.; Fiyaz, A.R.; Moirangthem, S.S.; Pajat, J.J.; Ngachan, S.V. Tree bean (Parkia roxburghii G. Don): A complete food and ethno-medicine for North East India. Res. Bull. 2012, 12–14. [Google Scholar]
- Zuhud, E.A.M.; Rahayu, W.P.; Wijaya, C.H.; Sari, P.P. Antimicrobial activity of kedawung extract (Parkia roxburghii G. Don) on food borne pathogens. J. Teknol. Dan Ind. Pangan 2001, 12, 1–5. [Google Scholar]
- Paul, B.; Bhuyan, B.; Purkayastha, D.D.; Dhar, S.S. Photocatalytic and antibacterial activities of gold and silver nanoparticles synthesized using biomass of Parkia roxburghii leaf. J. Photochem. Photobiol. B Biol. 2016, 154, 1–7. [Google Scholar] [CrossRef]
- Devi, T.P.; Shakuntala, I.; Devi, G.; Nonglait, K.K.L.; Singha, L.B.; Pattanayak, A.; Rahman, H. Antibacterial, nematicidal and nutritional properties of different parts of tree bean, Parkia roxburghii G. Don. Asian J. Microbiol. Biotechnol. Environ. Sci 2007, 9, 621–626. [Google Scholar]
- Shrisha, D.L.; Raveesha, K.A. Nagabhushan Bioprospecting of selected medicinal plants for antibacterial activity against some pathogenic bacteria. J. Med. Plants Res. 2011, 5, 4087–4093. [Google Scholar]
- Patel, J.R.; Gohil, T.G. Antibacterial efficacy of methanolic leaf extracts of some trees against some common pathogenic bacteria. J. Appl. Sci. Comput. 2018, 5, 404–408. [Google Scholar]
- Behuria, H.G.; Sahu, S.K. An Anti-microbial terpenoid fraction from Gymnema sylvestre induces flip-flop of fluorescent-phospholipid analogs in model membrane. Appl. Biochem. Biotechnol. 2020, 192, 1331–1345. [Google Scholar] [CrossRef] [PubMed]
- Tunsaringkarn, T.; Rungsiyothin, A.; Ruangrungs, N. α-glucosidase inhibitory activity of Thai mimosaceous plant extracts. J. Health Res. 2008, 22, 29–33. [Google Scholar]
- Jamaluddin, F.; Mohameda, S. Hypoglycemic effect of extracts of petai papan (Parkia speciosa, Hassk). Agric. Sci. 1993, 16, 161. [Google Scholar]
- Fitria, F.; Annisa, A.; Nikita, S.; Ranna, C. Alpha glukosidase inhibitory test and total phenolic content of ethanol extract of Parkia speciosa plant. Sci. Technol. Indones. 2019, 4, 1. [Google Scholar] [CrossRef]
- Saleh, M.S.M.; Siddiqui, M.J.; Mat So’ad, S.Z.; Roheem, F.O.; Saidi-Besbes, S.; Khatib, A. Correlation of FT-IR fingerprint and α-glucosidase inhibitory activity of salak (Salacca zalacca) fruit extracts utilizing orthogonal partial least square. Molecules 2018, 23, 1434. [Google Scholar] [CrossRef] [PubMed]
- Ramu, R.; Shirahatti, P.S.; Nayakavadi, S.; Vadivelan, R.; Zameer, F.; Dhananjaya, B.L.; Nagendra Prasad, M.N. The effect of a plant extract enriched in stigmasterol and β-sitosterol on glycaemic status and glucose metabolism in alloxan-induced diabetic rats. Food Funct. 2016, 7, 3999–4011. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Huang, M.; Yang, J.; Ma, X.; Zheng, S.; Deng, S.; Huang, Y.; Yang, X.; Zhao, P. Anti-diabetic activity of stigmasterol from soybean oil by targeting the GLUT4 glucose transporter. Food Nutr. Res. 2017, 61. [Google Scholar] [CrossRef]
- Ward, M.G.; Li, G.; Barbosa-Lorenzi, V.C.; Hao, M. Stigmasterol prevents glucolipotoxicity induced defects in glucose-stimulated insulin secretion. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Aiyer, P.V. Amylases and their applications. Afr. J. Biotechnol. 2005, 4, 1525–1529. [Google Scholar]
- Odetola, A.A.; Akinloye, O.; Egunjobi, C.; Adekunle, W.A.; Ayoola, A.O. Possible antidiabetic and antihyperlipidaemic effect of fermented Parkia biglobosa (Jacq) ex- tract in alloxan induced diabetic rats. Clin. Exp. Pharmacol. Physiol. 2006, 33, 808–812. [Google Scholar] [CrossRef]
- Sule, O.; Godwin, J.; Abdu, A. Preliminary study of hypoglycemic effect of locust bean (Parkia biglobosa) on wistar albino rat. J. Sci. Res. Rep. 2015, 4, 467–472. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Habila, J.D.; Koorbanally, N.A.; Islam, M.S. Butanol fraction of Parkia biglobosa (Jacq.) G. Don leaves enhance pancreatic β-cell functions, stimulates insulin secretion and ameliorates other type 2 diabetes-associated complications in rats. J. Ethnopharmacol. 2016, 183, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Chanvitan, A.; Ubolcholket, S.; Chongsuvivatwong, V.; Geater, A. Risk factors for squamous cell carcinoma in southern Thailand. Esophageal Canver Stud. South. Thail. 1990, 81–100. [Google Scholar]
- Tangkanakul, P.; Trakoontivakorn, G.; Saengprakai, J.; Auttaviboonkul, P.; Niyomwit, B.; Lowvitoon, N.; Nakahara, K. Antioxidant capacity and antimutagenicity of thermal processed Thai foods. Jpn. Agric. Res. Q. JARQ 2011, 45, 211–218. [Google Scholar] [CrossRef][Green Version]
- Murakami, A.; Ohigashi, H.; Koshimizu, K. Possible anti-tumour promoting properties of traditional Thai food items and some of their active constituents. Asia Pac. J. Clin. Nutr. 1994, 3, 185–191. [Google Scholar]
- Aisha, A.F.A.; Abu-Salah, K.M.; Alrokayan, S.A.; Ismail, Z.; Abdul Majid, A.M.S. Evaluation of antiangiogenic and antoxidant properties of Parkia speciosa Hassk extracts. Pak. J. Pharm. Sci. 2012, 25, 7–14. [Google Scholar]
- Singh, R.S.; Bhari, R.; Kaur, H.P. Mushroom lectins: Current status and future perspectives. Crit. Rev. Biotechnol. 2010, 30, 99–126. [Google Scholar] [CrossRef]
- Suvachittanont, W.; Jaranchavanapet, P. Mitogenic effect of Parkia speciosa seed lectin on human lymphocytes. Planta Med. 2000, 66, 699–704. [Google Scholar] [CrossRef]
- Kaur, N.; Singh, J.; Kamboj, S.; Agrewala, J.; Kaur, M. Two Novel Lectins from Parkia biglandulosa and Parkia roxburghii: Isolation, Physicochemical Characterization, Mitogenicity and Anti- Proliferative Activity. Protein Pept. Lett. 2005, 12, 589–595. [Google Scholar] [CrossRef]
- Fadeyi, S.A.; Fadeyi, O.O.; Adejumo, A.A.; Okoro, C.; Myles, E.L. In vitro anticancer screening of 24 locally used Nigerian medicinal plants. BMC Complementary Altern. Med. 2013, 13, 79. [Google Scholar] [CrossRef]
- Shete, S.V.; Mundada, S.J.; Dhande, S. Comparative effect of crude extract of Parkia biglandulosa and Its isolate on regenerative angiogenesis In adult Zebrafish. Indian Drug 2017, 54, 51–57. [Google Scholar]
- Kamisah, Y.; Othman, F.; Qodriyah, H.M.S.; Jaarin, K. Parkia speciosa Hassk.: A potential phytomedicine. Evid. Based Complementary Altern. Med. 2013, 2013, 709028. [Google Scholar] [CrossRef] [PubMed]
- Auyeung, K.K.; Han, Q.-B.; Ko, J.K. Astragalus membranaceus: A review of its protection against inflammation and gastrointestinal cancers. Am. J. Chin. Med. 2016, 44, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Ognatan, K.; Adi, K.; Lamboni, C.; Damorou, J.M.; Aklikokou, K.A.; Gbeassor, M.; Guilland, J.C. Effect of dietary intake of fermented seeds of Parkia biglobosa (Jacq) Benth (African locust bean) on hypertension in bogou and goumou-kope areas of togo. Trop. J. Pharm. Res. 2011, 10, 603–609. [Google Scholar] [CrossRef][Green Version]
- Kodjo, K.M.; Contesse, V.; Do Rego, J.L.; Aklikokou, K.; Titrikou, S.; Gbeassor, M.; Vaudry, H. In vitro effects of crude extracts of Parkia biglobosa (Mimosaceae), Stereospermum kunthianum (Bignoniaceae) and Biophytum petersianum (Oxalidaceae) on corticosteroid secretion in rat. J. Steroid Biochem. Mol. Biol. 2006, 100, 202–208. [Google Scholar] [CrossRef]
- Ouédraogoa, S.; Somé, N.; Ouattara, S.; Kini, F.B.; Traore, A.; Bucher, B.; Guissou, I.P. Acute toxicity and vascular properties of seed of Parkia biglobosa (JACQ) R. Br Gift (Mimosaceae) on rat aorta. Afr. J. Tradit. Complementary Altern. Med. 2012, 9, 260–265. [Google Scholar]
- Kassi, Y.; Aka, K.J.; Abo, K.J.C.; Mea, A.; Bi, S.A.N.; Ehile, E.E. Effet antihypertensif d’un extrait aqueux d’écorce de tronc de Parkia biglobosa (mimosaceae) sur la pression artérielle de lapin. Sci. Nat. 2008, 5, 133–143. [Google Scholar]
- Yi, Q.Y.; Li, H.B.; Qi, J.; Yu, X.J.; Huo, C.J.; Li, X.; Bai, J.; Gao, H.L.; Kou, B.; Liu, K.L.; et al. Chronic infusion of epigallocatechin-3-O-gallate into the hypothalamic paraventricular nucleus attenuates hypertension and sympathoexcitation by restoring neurotransmitters and cytokines. Toxicol. Lett. 2016, 262, 105–113. [Google Scholar] [CrossRef]
- Galleano, M.; Pechanova, O.; G Fraga, C. Hypertension, nitric oxide, oxidants, and dietary plant polyphenols. Curr. Pharm. Biotechnol. 2010, 11, 837–848. [Google Scholar] [CrossRef]
- Takagaki, A.; Nanjo, F. Effects of Metabolites Produced from (-)-Epigallocatechin Gallate by Rat Intestinal Bacteria on Angiotensin I-Converting Enzyme Activity and Blood Pressure in Spontaneously Hypertensive Rats. J. Agric. Food Chem. 2015, 63, 8262–8266. [Google Scholar] [CrossRef]
- Luo, D.; Xu, J.; Chen, X.; Zhu, X.; Liu, S.; Li, J.; Xu, X.; Ma, X.; Zhao, J.; Ji, X. (−)-Epigallocatechin-3-gallate (EGCG) attenuates salt-induced hypertension and renal injury in Dahl salt-sensitive rats. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ayo-Lawal, R.A.; Osoniyi, O.; Famurewa, A.J.; Lawal, O.A. Evaluation of antioxidant and hypolipidaemic effects of fermented Parkia biglobosa (Jacq) seeds in tyloxapol-induced hyperlipidaemic rats. Afr. J. Food Sci. 2014, 8, 225–232. [Google Scholar] [CrossRef]
- Rendu, F.; Saleun, S.; Auger, J. Parkia biglobosa seeds possess anti platelet activity. Thromb. Res. 1993, 71, 505–508. [Google Scholar] [CrossRef]
- Appeldoorn, M.M.; Venema, D.P.; Peters, T.H.F.; Koenen, M.E.; Arts, I.C.W.; Vincken, J.P.; Gruppen, H.; Keuer, J.; Hollman, P.C.H. Some phenolic compounds increase the nitric oxide level in endothelial cells in vitro. J. Agric. Food Chem. 2009, 57, 7693–7699. [Google Scholar] [CrossRef]
- Siow, H.L.; Gan, C.Y. Extraction of antioxidative and antihypertensive bioactive peptides from Parkia speciosa seeds. Food Chem. 2013, 141, 3435–3442. [Google Scholar] [CrossRef]
- Zaini, N.; Mustaffa, F. Review: Parkia speciosa as Valuable, Miracle of Nature. Asian J. Med. Health 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Khalid, N.M.; Babji, A.S. Antioxidative and antihypertensive activities of selected Malaysian ulam (salad), vegetables and herbs. J. Food Res. 2018, 7, 27–37. [Google Scholar] [CrossRef]
- Owolabi, O.J.; Ukoima, G.S.; Inninh, S.O.; Otokiti, I.O. The anti-diarrhoeal activity of the aqueous stem bark extract of Parkia filicoidea (Fabaceae). J. Med. Biomed. Res. 2016, 15, 12–20. [Google Scholar]
- Adebayo, O.L.; Marzuk, S.; Mumuni, S.I. An in vivo assessment of Anti-diarrheal activity of solvent extracts of leaf and stem bark of Ghanian Parkia biglobosa against castor oil induced diarrhea in albino rats. Int. J. Bioassays 2014, 310, 3358–3362. [Google Scholar]
- Sahoo, H.B.; Sagar, R.; Kumar, A.; Bhaiji, A.; Bhattamishra, S.K. Antidiarrhoeal investigation of Apium leptophyllum (Pers.) by modulation of Na+K+ATPase, nitrous oxide and intestinal transit in rats. Biomed. J. 2016, 39, 376–381. [Google Scholar] [CrossRef]
- Khan, T.; Ali, S.; Qayyum, R.; Hussain, I.; Wahid, F.; Shah, A.J. Intestinal and vascular smooth muscle relaxant effect of Viscum album explains its medicinal use in hyperactive gut disorders and hypertension. BMC Complementary Altern. Med. 2016, 16, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Imtiaz, S.M.; Aleem, A.; Saqib, F.; Ormenisan, A.N.; Neculau, A.E.; Anastasiu, C.V. The potential involvement of an ATP-dependent potassium channel-opening mechanism in the smooth muscle relaxant properties of Tamarix dioica roxb. Biomolecules 2019, 9, 722. [Google Scholar] [CrossRef] [PubMed]
- Maria, M.S.; Devarakonda, S.; Kumar, A.T.V.; Balakrishnan, N. Anti-ulcer activity of ethanol extract of Parkia speciosa against indomethacin induced peptic ulcer in albino rats. Int. J. Pharm. Sci. Res. 2015, 6, 895–902. [Google Scholar]
- Al Batran, R.; Al-Bayaty, F.; Al-Obaidi, M.M.J.; Abdualkader, A.M.; Hadi, H.A.; Ali, H.M.; Abdulla, M. A In vivo antioxidant and antiulcer activity of Parkia speciosa ethanolic leaf extract against ethanol-induced gastric ulcer in rats. PLoS ONE 2013, 8, e64751. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, H.B.; Silva, F.V.; B Passos, F.F.; S Bezerra, R.D.; Chaves, M.H.; Oliveira, F.A.; Meneses Oliveira, R.C. Gastroprotective effect of the ethanolic extract of Parkia platycephala benth. Leaves against acute gastric lesion models in rodents. Biol. Res. 2010, 43, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Hamaishi, K.; Kojima, R.; Ito, M. Anti-ulcer effect of tea catechin in rats. Biol. Pharm. Bull. 2006, 29, 2206–2213. [Google Scholar] [CrossRef]
- Ito, Y.; Ichikawa, T.; Iwai, T.; Saegusa, Y.; Ikezawa, T.; Goso, Y.; Ishihara, K. Effects of tea catechins on the gastrointestinal mucosa in rats. J. Agric. Food Chem. 2008, 56, 12122–12126. [Google Scholar] [CrossRef]
- Suzuki, Y.; Ishihara, M.; Segami, T.; Ito, M. Anti-ulcer effects of antioxidants, quercetin, α-tocopherol, nifedipine and tetracycline in rats. Jpn. J. Pharmacol. 1998, 78, 435–441. [Google Scholar] [CrossRef]
- De Barros, M.P.; Lemos, M.; Maistro, E.L.; Leite, M.F.; Sousa, J.P.B.; Bastos, J.K.; de Andrade, S.F. Evaluation of antiulcer activity of the main phenolic acids found in Brazilian Green Propolis. J. Ethnopharmacol. 2008, 120, 372–377. [Google Scholar] [CrossRef]
- Ijarotimi, O.S.; Keshinro, O.O. Protein quality, hematological properties and nutritional status of albino rats fed complementary foods with fermented popcorn, African locust bean, and bambara groundnut flour blends. Nutr. Res. Pract. 2012, 6, 381–388. [Google Scholar] [CrossRef]
- Nursucihta, S.; Thai’in, H.A.; Putri, D.M.; Utami, D.N.; Ghani, A.P. Antianemia activity of parkia speciosa hassk seed ethanolic extract. Maj. Obat Tradis. 2014, 19, 49–54. [Google Scholar]
- Nwaehujor, C.O.; Ezeigbo, I.I.; Udeh, N.E.; Ezeja, M.I.; Asuzu, I.U. Anti-inflammatory anti-oxidant Activities of the methanolic extracts of the stalk of Parkia biglobosa. Hygein J. Med. 2010, 3, 34–40. [Google Scholar]
- Bari, A.U.; Santiago, M.Q.; Osterne, V.J.S.; Pinto-Junior, V.R.; Pereira, L.P.; Silva-Filho, J.C.; Debray, H.; Rocha, B.A.M.; Delatorre, P.; Teixeira, C.S.; et al. Lectins from Parkia biglobosa and Parkia platycephala: A comparative study of structure and biological effects. Int. J. Biol. Macromol. 2016, 92, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Silva, H.C.; Bari, A.U.; Rocha, B.A.M.; Nascimento, K.S.; Ponte, E.L.; Pires, A.F.; Delatorre, P.; Teixeira, E.H.; Debray, H.; Assreuy, A.M.S. Purification and primary structure of a mannose/glucose-binding lectin from Parkia biglobosa Jacq. seeds with antinociceptive and anti-inflammatory properties. J. Mol. Recognit. 2013, 26, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Ukwuani, A.; Ahmad, H. In vitro anti-inflammatory activity of Parkia biglobosa fruit bark extract. Int. J. Life Sci. Sci. Res. 2015, 1, 8–11. [Google Scholar] [CrossRef]
- Badu, M.; Mensah, J.K.; Boadi, N.O. Antioxidant activity of methanol and ethanol/water extracts of Tetrapleura tetraptera and Parkia biglobosa. Int. J. Pharma Bio Sci. 2012, 3, 312–321. [Google Scholar]
- Oboh, G.; Alabi, K.B.; Akindahunsi, A.A. Fermentation changes the nutritive value, polyphenol distribution, and antioxidant properties of Parkia biglobosa seeds (African locust beans). Food Biotechnol. 2008, 22, 363–376. [Google Scholar] [CrossRef]
- Seal, T. Antioxidant activity of some wild edible plants of Meghalaya state of India: A comparison using two solvent extraction systems. Int. J. Nutr. Metab. 2012, 4, 51–56. [Google Scholar]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Bukhori, M.F.M.; Rahmat, M.H.; Rahmat, A. Assessment and comparison of phytochemical constituents and biological activities of bitter bean (Parkia speciosa Hassk.) collected from different locations in Malaysia. Chem. Cent. J. 2018, 12, 1–9. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Alam, B.; Ju, M.; Kwon, K.; Suk, Y. Antioxidant mechanism of polyphenol-rich Nymphaea nouchali leaf extract protecting DNA damage and attenuating oxidative stress-induced cell death via Nrf2-mediated heme-oxygenase-1 induction coupled with ERK/p38 signaling pathway. Biomed. Pharmacother. 2018, 103, 1397–1407. [Google Scholar] [CrossRef]
- Tunsaringkarn, T.; Soogarun, S.; Rungsiyothin, A.; Palasuwan, A. Inhibitory activity of Heinz body induction in vitro antioxidant model and tannin concentration of Thai mimosaceous plant extracts. J. Med. Plants Res. 2012, 6, 4096–4101. [Google Scholar] [CrossRef]
- Ajibola, M.; Olugbemi, O.; Joseph, D.; Denen, A. Hepatoprotective effect of Parkia biglobosa stem bark methanolic extract on paracetamol induced liver damage in wistar rats. Am. J. Biomed. Life Sci 2013, 1, 75–78. [Google Scholar] [CrossRef][Green Version]
- Coriolano, M.C.; de Melo, C.M.L.; de Oliveira Silva, F.; Schirato, G.V.; Porto, C.S.; dos Santos, P.J.P.; dos Santos Correia, M.T.; Porto, A.L.F.; dos Anjos Carneiro-Leão, A.M.; Coelho, L.C.B.B. Parkia pendula seed lectin: Potential use to treat cutaneous wounds in healthy and immunocompromised mice. Appl. Biochem. Biotechnol. 2014, 172, 2682–2693. [Google Scholar] [CrossRef] [PubMed]
- Angami, T.; Bhagawati, R.; Touthang, L.; Makdoh, B.; Nirmal; Lungmuana; Bharati, K.A.; Silambarasan, R.; Ayyanar, M. Traditional uses, phytochemistry and biological activities of Parkia timoriana (DC.) Merr., an underutilized multipurpose tree bean: A review. Genet. Resour. Crop Evol. 2018, 65, 679–692. [Google Scholar] [CrossRef]
- Hopkins, H.C. Floral biology and pollination ecology of the neotropical species of Parkia. J. Ecol. 1984, 72, 1–23. [Google Scholar] [CrossRef]
- Abalaka, S.E.; Fatihu, M.Y.; Ibrahim, N.D.G.; Kazeem, H.M. Histopathologic changes in the gills and skin of adult Clarias gariepinus exposed to ethanolic extract of Parkia biglobosa pods. Basic Appl. Pathol. 2010, 3, 109–114. [Google Scholar] [CrossRef]
- Oshimagye, M.I.; Ayuba, V.O.; Annune, P.A. Toxicity of aqueous extracts of Parkia biglobosa pods on Clarias gariepinus (Burchell, 1822) Juveniles. Niger. J. Fish. Aquac. 2014, 2, 24–29. [Google Scholar]
- Uyub, A.M.; Nwachukwu, I.N.; Azlan, A.A.; Fariza, S.S. In-vitro antibacterial activity and cytotoxicity of selected medicinal plant extracts from Penang Island Malaysia on some pathogenic bacteria. Ethnobot. Res. Appl. 2010, 8, 95–106. [Google Scholar] [CrossRef]
- Sil, S.K.; Saha, S.; Karmakar, P. Reactive oxygen species as possible mediator of antibacterial activity of Parkia javanica, against bacterial species predominantly found in chronic wound. J. Drug Deliv. Ther. 2018, 8, 43–47. [Google Scholar] [CrossRef][Green Version]
- Rupanjali, S.; Basu, J.M.; Syamal, R.; Biswanath, D.; Sil, S.K. In vitro activity of Parkia javanica extract against Leishmania donovani parasite. J. Appl. Biosci. 2010, 36, 85–89. [Google Scholar]
- Favacho, A.R.M.; Cintra, E.A.; Coelho, L.C.B.B.; Linhares, M.I.S. In vitro activity evaluation of Parkia pendula seed lectin against human cytomegalovirus and herpes virus 6. Biologicals 2007, 35, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Ogunyinka, B.I.; Oyinloye, B.E.; Osunsanmi, F.O.; Opoku, A.R.; Kappo, A.P. Modulatory influence of Parkia biglobosa protein isolate on testosterone and biomarkers of oxidative stress in brain and testes of streptozotocin-induced diabetic male rats Bolajoko. Int. J. Physiol. Pathophysiol. Pharm. 2016, 8, 78–86. [Google Scholar] [CrossRef]
- Patra, K.; Jana, S.; Sarkar, A.; Karmakar, S.; Jana, J.; Gupta, M.; Mukherjee, G.; De, U.C.; Mandal, D.P.; Bhattacharjee, S. Parkia javanica extract induces apoptosis in S-180 cells via the intrinsic pathway of apoptosis. Nutr. Cancer 2016, 68, 689–707. [Google Scholar] [CrossRef] [PubMed]
- Aisha, A.F.A.; Abu-Salah, K.M.; Darwis, Y.; Majid, A.M.S.A. Screening of antiangiogenic activity of some tropical plants by rat aorta ring assay. Int. J. Pharmacol. 2009, 5, 370–376. [Google Scholar] [CrossRef]
- Gui, J.S.; Jalil, J.; Jubri, Z.; Kamisah, Y. Parkia speciosa empty pod extract exerts anti-inflammatory properties by modulating NFκB and MAPK pathways in cardiomyocytes exposed to tumor necrosis factor-α. Cytotechnology 2019, 71, 79–89. [Google Scholar] [CrossRef]
- Mustafa, N.H.; Ugusman, A.; Jalil, J.; Kamisah, Y. Anti-inflammatory property of Parkia speciosa empty pod extract in human umbilical vein endothelial cells. J. Appl. Pharm. Sci. 2018, 8, 152–158. [Google Scholar] [CrossRef]
- Ruthiran, P.; Selvaraj, C.I. Phytochemical screening and in vitro antioxidant activity of Parkia timoriana (DC.) Merr. Res. J. Biotechnol. 2017, 12, 12. [Google Scholar]
- Chanu, K.V.; Ali, M.A.; Kataria, M. Antioxidant activities of two medicinal vegetables: Parkia javanica and Phlogacanthus thyrsiflorus. Int. J. Pharm. Pharm. Sci. 2012, 4, 102–106. [Google Scholar]
- Balaji, K.; Nedumaran, S.A.; Devi, T.; Sikarwar, M.S.; Fuloria, S. Phytochemical analysis and in vitro antioxidant activity of Parkia speciosa. Int. J. Green Pharm. 2015, 9, S50–S54. [Google Scholar] [CrossRef]
- Ramli, S.; Bunrathep, S.; Tansaringkarn, T.; Ruangrungsi, N. Screening for free radical scavenging activity from ethanolic extract of Mimosaceous plants Endemic to Thailand. J. Health Res. 2008, 22, 55–59. [Google Scholar]
- Komolafe, K.; Olaleye, T.M.; Omotuyi, O.I.; Boligon, A.A.; Athayde, M.L.; Akindahunsi, A.A.; da Rocha, J.B.T. In vitro antioxidant activity and effect of Parkia biglobosa bark extract on mitochondrial redox status. Jams J. Acupunct. Meridian Stud. 2014, 7, 202–210. [Google Scholar] [CrossRef] [PubMed]

| Species | Part Used | Method of Preparation | Medicinal Uses | Region/Country | Reference |
|---|---|---|---|---|---|
| P. bicolor | Stem bark | Pulverized powder | Wound healing | West coast of Africa and Nigeria | [23] |
| Tree | Diarrhea, dysentery | Southwest Nigeria | [55] | ||
| Stem barks | Decoction | Bad cough, measles, and woman infertility | Cameroon | [28] | |
| Stem barks | Decoction | Diarrhea and skin ulcers | Ghana | [56] | |
| P. biglobosa | Roots & bark | Paste | Dental disorder | Ivory Coast | [29] |
| Seed and stem bark | Fresh seeds | Fish poison | West Africa | [57,58] | |
| Root | Decoction combined with other plants | Infertility | Nigeria | [58] | |
| Bark infusion with lemon | Diarrhea | Nigeria | [59] | ||
| Stem bark | Anti-snake venom | Nigeria | [60] | ||
| Bark | Paste, decoction | Wound healing leprosy, hypertension, mouth wash, toothpaste | Nigeria | [22,23] | |
| Leaves and roots | Eyesore | Lotion | Gambia | [23] | |
| Bark | Hot decoction | Fever | Gambia | [23] | |
| Bark | Decoction | Malaria, diabetes, amenorrhea, and hypertension | Senegal, Mali, Ghana Togo, and South Africa | [11,40,61,62,63] | |
| Roots and bark | Decoction of the roots with Ximenia americana | Weight loss | Burkina Faso | [64] | |
| Stem bark | Boiled bark | Diarrhea, conjunctivitis, severe cough, and leprosy | West Coast Africa | [23,65,66] | |
| Leaves | Decoction | Violent colic chest and muscular pain | Northern Nigeria | [38] | |
| bark | Infusion | Dental caries and astringent | Guinea Bissau | [67] | |
| P. biglandulosa | Seed bark | Saponins | Astringent | India | [68] |
| Stem bark | Hemagglutination, ulcer | India | [69] | ||
| Tree | Inflammation and ulcer | India | [70] | ||
| P. clappertoniana | Tree | Hypertension | Southwest Nigeria | [55] | |
| Root | Dental caries and conjunctivitis | African | [71,72] | ||
| Seed | Crudely pounded | Labor induction | Ghana | [17] | |
| Tree | Diarrhea | Kaduna and Nigeria | [73] | ||
| Leaves and bark | Maceration | Epilepsy | Northern Nigeria | [74] | |
| Stem bark | Chickenpox and measles | Southwest Nigeria | [24] | ||
| Tree | Diabetes, leprosy, and ulcers | Ghana | [75] | ||
| Tree | Mouthwash and toothache | Nigeria | [76] | ||
| Tree | Eczema and skin diseases | Nigeria | [77] | ||
| Bark | Infusion | Hernia | Ghana | [75] | |
| P. pendula | Leaves bark | Genital bath | Netherland | [78] | |
| Bark | Decoction | Malaria | Brazil | [79] | |
| P. speciosa | Seed | Eaten raw or cooked oral decoction | Diabetes | Malaysia | [80] |
| Leaves | Pounded with rice and applied on the neck | Cough | Malaysia | [30] | |
| Root | Decoction | Skin problems | Southern Thailand | [21] | |
| Root | Decoction taken orally | Hypertension and diabetes | Malaysia | [26] | |
| Fruit | Eaten raw | Diabetes | Malaysia | [30] | |
| Seed | Eaten raw | Detoxification and hypertension | Singapore | [81] | |
| Ringworm | Malaysia | [82] | |||
| Leaf | Decoction | Dermatitis | Indonesia | [20] | |
| Root | Oral decoction | Toothache | Malaysia | [27] | |
| Tree | Heart problem, constipation and edema | India | [83,84] | ||
| Leaves | Dermatitis | Indonesia | [85] | ||
| Seed | Loss of appetite | Indonesia | [86] | ||
| Seed | Cooked | Kidney disorder | West Malaysia | [87] | |
| P. timoriana | Bark and twig | Decoction of bark and twig paste | Diarrhea, dysentery, and wound | India | [88] |
| Bark | Decoction used to bath | Fever | Gambia | [89] | |
| Pulp bark | Mixed with lemon | Ulcer and wound | Gambia | [89] | |
| Fruit | Diabetes | Thailand | [90] | ||
| Pod | Pounded in water | Hair washing, skin diseases, and ulcers | India | [19] | |
| Bark and leaves | Head washing, skin diseases, and ulcers | India | [19] | ||
| Bark | Decoction with Centella. asiatica and Ficus glomerata | Diabetes | India | [16] | |
| P. roxburghii | Tree | Tender pod and bark taken orally | Diarrhea, dysentery, intestinal disorder, and bleeding piles | India | [91] |
| The fruit or young shoot | Green portion of the fruit mixed with water to be taken orally | Dysentery, diarrhea, food poisoning, wound, and scabies | India | [92] | |
| Seed | Grounded and mixed with hot water | Postnatal care, diarrhea, edema and tonsillitis | Malaysia | [93] | |
| Pod | Diabetes, hypertension, and urinary tract infections | India | [18] | ||
| Leaves, pod, peals, and bark | Diarrhea and dysentery | India | [94] | ||
| Stem bark | Hot water extraction | Diarrhea and dysentery | India | [95] | |
| Bark | Turn into paste | Used as plaster for eczema | India | [25] | |
| P. javanica | Bark, pod, and seed | Taking orally as vegetable | Dysentery and diarrhea | India | [15] |
| Tree | Inflammation | India | [96] | ||
| Bark fruit | Dysentery and piles | India | [51] | ||
| Stomachache and cholera | India | [97] | |||
| Bark and leaves | Lotion | Sores and skin diseases | [98] | ||
| Tree | Diarrhea, cholera dysentery, and food poisoning | India | [99] |
| Structure Number | Type | Compound | Species | Part | Reference |
|---|---|---|---|---|---|
| Polyphenolics | |||||
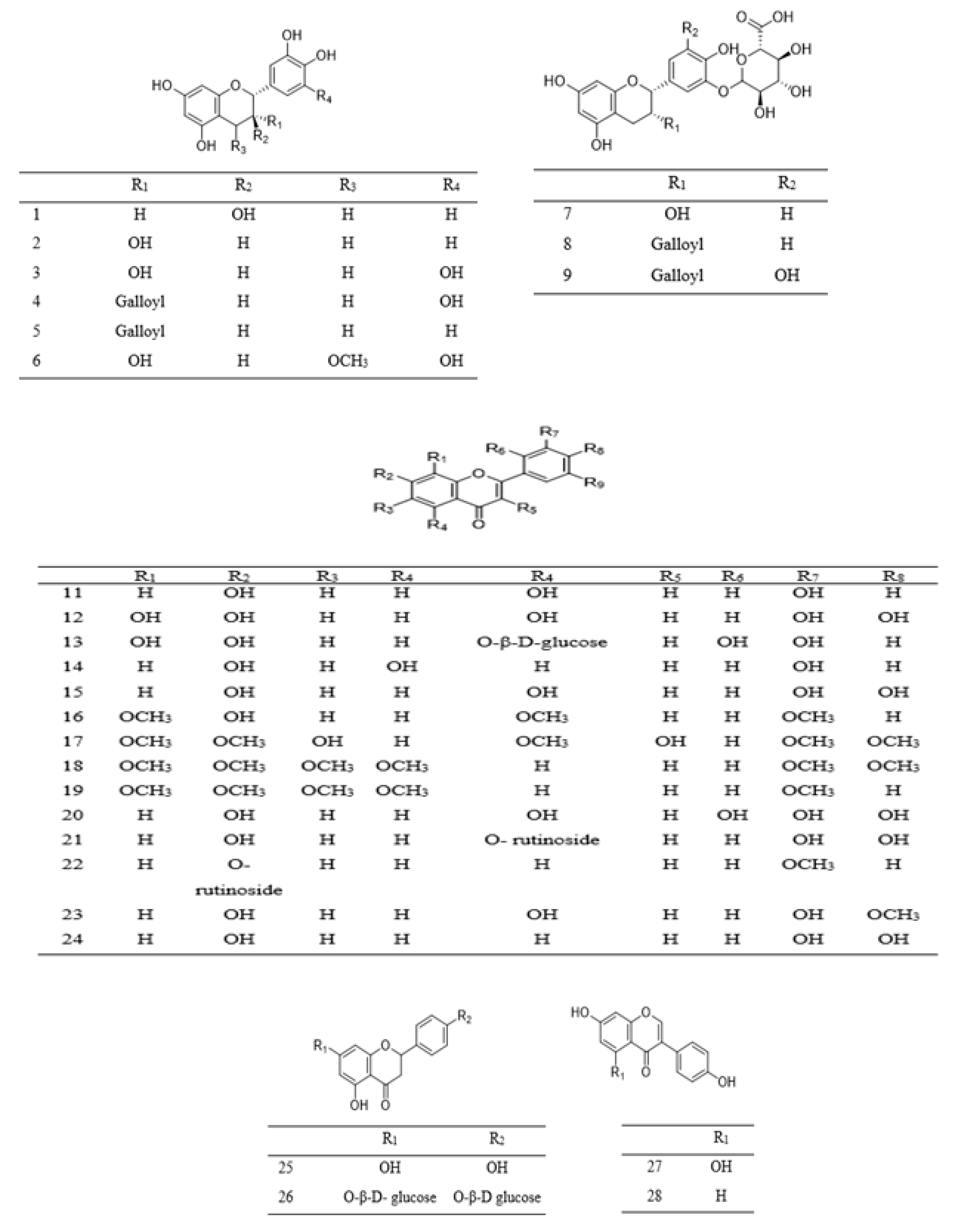
| 1 | Flavanol | Catechin | P. speciosa | Pod | [107] |
| P. biglobosa | Root/bark | [106] | |||
| P. javanica | Edible part | [108] | |||
| 2 | Flavanol | Epicatechin | P. speciosa | Pod | [107] |
| P. javanica | Edible part | [108] | |||
| 3 | Flavanol | Epigallocatechin | P. biglobosa | Root/bark | [111] |
| P. javanica | Edible part | [108] | |||
| 4 | Flavanol | Epigallocatechin gallate | P. roxburghii | Pod | [18] |
| P. biglobosa | Root/bark | [106,111] | |||
| 5 | Flavanol | Epicatechin-3-O-gallate | P. biglobosa | Bark | [111] |
| 6 | Flavanol | 4-O-methyl-epigallocate-chin | P. biglobosa | Bark | [111] |
| 7 | Flavanol | Epigallocatechin-O-glucuronide | P. biglobosa | Root/bark | [106] |
| 8 | Flavanol | Epicatechin-O-gallate-O-glucuronide | P. biglobosa | Root/bark | [106] |
| 9 | Flavanol | Epigallocatechin-O-gallate-O-glucuronide | P. biglobosa | Root/bark | [106] |
| 10 | Flavanol | Theaflavin gallate | P. speciosa | Pod | [112] |
| 11 | Flavonol | Kaempferol | P. speciosa | Pod | [107] |
| P. javanica | Edible part | [108] | |||
| 12 | Flavonol | Quercetin | P. speciosa | Pod | [107] |
| 13 | Flavonol | Hyperin | P. roxburghii | Pod | [18] |
| 14 | Flavonol | Apigenin | P. speciosa | Pod | [112] |
| 15 | Flavone | 3,7,3′,4′-Tetrahydroxyflavone | P. clappertoniana | Seeds | [113,114] |
| 16 | Flavone | 7-Hydroxy-3, 8, 4′-trimethoxyflavone | P. clappertoniana | Leaves | [115] |
| 17 | Flavone | 2′-Hydroxy-3,7,8,4′,5′′pentamethoxyflavone | P. clappertoniana | Leaves | [115] |
| 18 | Flavone | Nobiletin | P. speciosa | Pod | [112] |
| 19 | Flavone | Tangeritin | P. speciosa | Pod | [112] |
| 20 | Flavonol | Myricetin | P. javanica | Edible part | [108] |
| P. speciosa | Pod | [112] | |||
| 21 | Flavonol glycoside | Rutin | P. javanica | Edible part | [108] |
| P. speciosa | Pod | [112] | |||
| 22 | Flavonol glycoside | Didymin | P. speciosa | Pod | [112] |
| 23 | Methoxy flavonol | Isorhamnetin | P. javanica | Edible part | [108] |
| 24 | Flavone | Luteolin | P. javanica | Edible part | [108] |
| 25 | Flavanone | Naringenin | P. javanica | Edible part | [108] |
| 26 | Flavanone | Naringenin-1-4′-di-O-ß-d-glucopyranoside | P. biglobosa | Fruit pulp | [109] |
| 27 | Isoflavone | Genistein | P. javanica | Edible part | [108] |
| 28 | Isoflavone | Daidzein | P. javanica | Edible part | [108] |
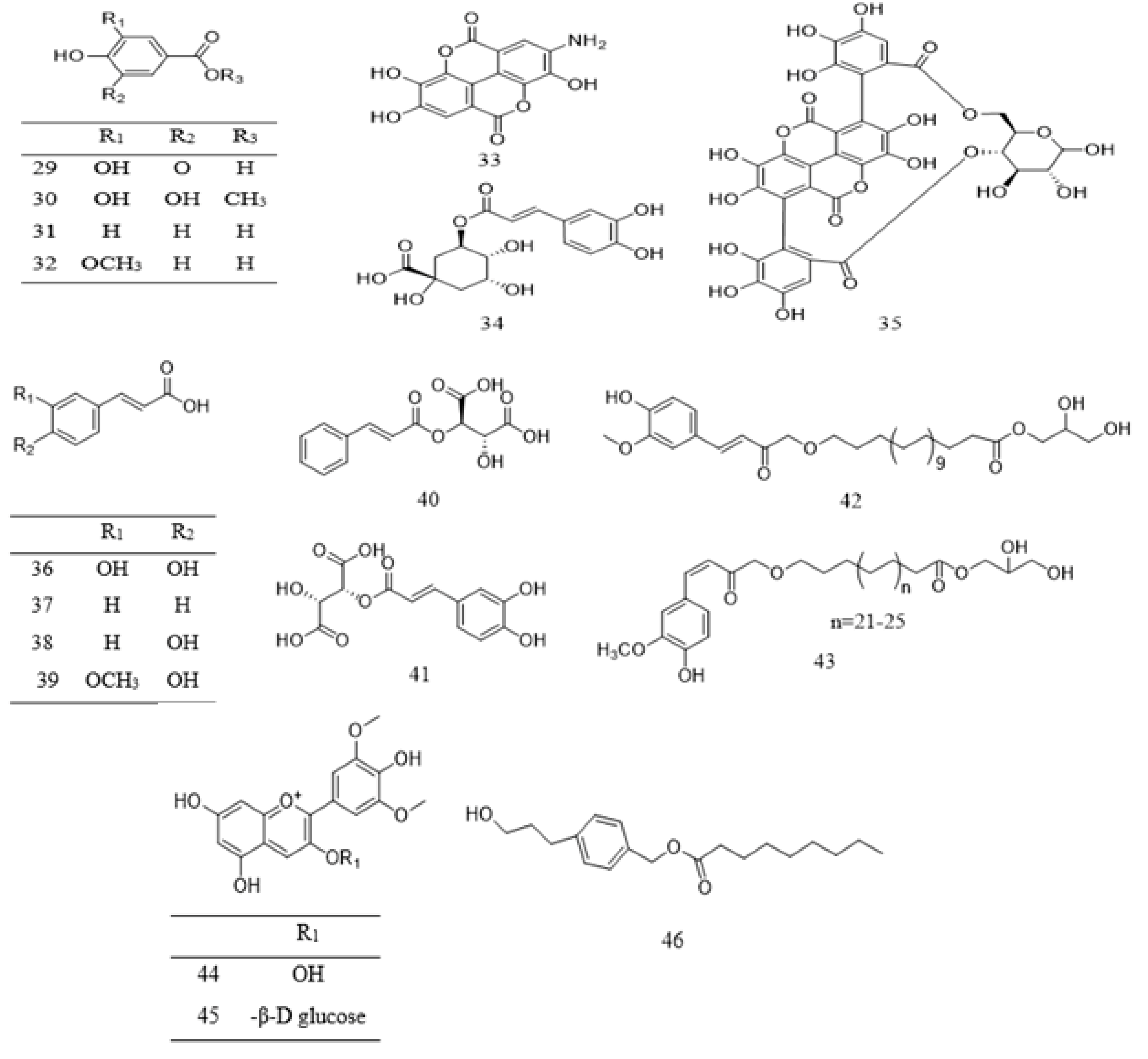
| 29 | Phenolic acid | Gallic acid | P. speciosa | Pod | [107] |
| P. bicolor | Root | [28] | |||
| 30 | Phenolic acid | Methyl gallate | P. bicolor | Root | [28] |
| 31 | Phenolic acid | Hydroxybenzoic acid | P. speciosa | Pod | [107] |
| 32 | Phenolic acid | Vanillic acid | P. speciosa | Pod | [107] |
| 33 | Phenolic acid | Chlorogenic acid | P. speciosa | Pod | [107] |
| P. javanica | Edible part | [108] | |||
| 34 | Phenolic acid | Ellagic acid | P. speciosa | Pod | [107] |
| 35 | Phenolic acid | Punicalin | P. speciosa | Pod | [112] |
| 36 | Phenolic acid | Caffeic acid | P. speciosa | Pod | [107] |
| P. javanica | Edible part | [108] | |||
| 37 | Phenolic acid | Cinnamic acid | P. speciosa | Pod | [107] |
| 38 | Phenolic acid | P-Coumaric acid | P. speciosa | Pod | [107] |
| P. javanica | Edible part | [108] | |||
| 39 | Phenolic acid | Ferulic acid | P. speciosa | Pod | [107] |
| P. javanica | Edible part | [108] | |||
| 40 | Phenolic acid | Coutaric acid | P. speciosa | Pod | [112] |
| 41 | Phenolic acid | Caftaric acid | P. speciosa | Pod | [112] |
| 42 | Phenolic | 1-(w-Feruloyllignoceryl) -glycerol | P. biglobosa | Bark | [111] |
| 43 | Phenolic | 1-(w-Isoferuloylalkanoyl) -glycerol | P. biglobosa | Bark | [111] |
| 44 | Phenolic | Malvidin | P. speciosa | Pod | [112] |
| 45 | Phenolic | Primulin | P. speciosa | Pod | [112] |
| 46 | Pheny propanoid | Parkinol | P. javanica | Leaves | [110] |
| 47 | Phenol | 2-Methoxy phenol | P. biglobosa | Seed | [116] |
| 48 | Phenol | 2,4-Disiopropyl-phenol | P. biglobosa | Seed | [116] |
| Terpenoid and steroid | |||||
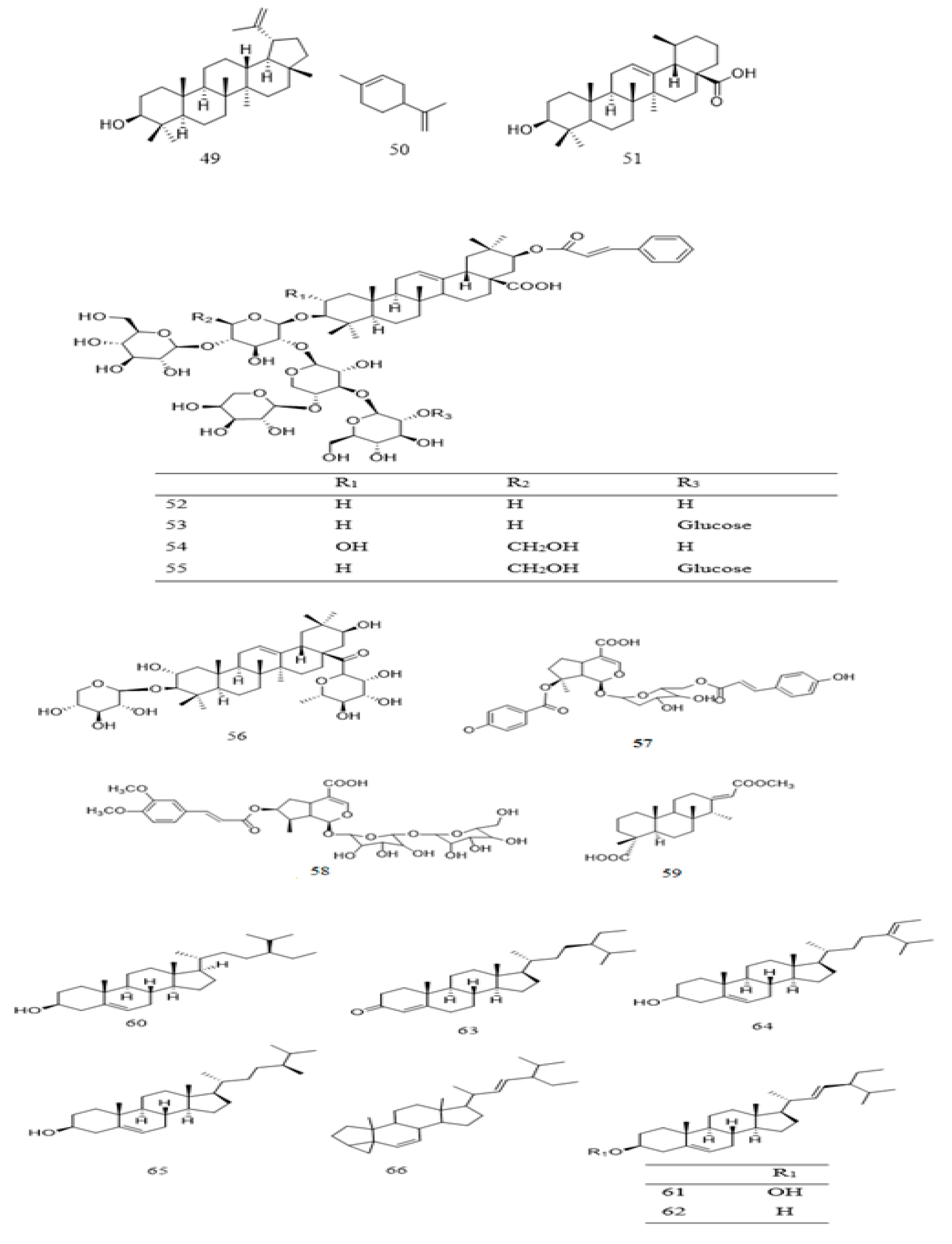
| 49 | Triterpenoid | Lupeol | P. biglobosa | Bark | [111] |
| P. bicolor | Root | [28] | |||
| P. speciosa | Seeds | [117] | |||
| 50 | Monoterpenoid | Limonene | P. biglobosa | Seed | [116] |
| 51 | Triterpenoid | Ursolic acid | P. javanica | Leaf/stem | [42] |
| 52 | Triterpenoid | Parkibicoloroside A | P. bicolor | Root | [118] |
| 53 | Triterpenoid | Parkibicoloroside B | P. bicolor | Root | [118] |
| 54 | Triterpenoid | Parkibicoloroside C | P. bicolor | Root | [118] |
| 55 | Triterpenoid | Parkibicoloroside D | P. bicolor | Root | [118] |
| 56 | Triterpenoid | Parkibicoloroside E | P. bicolor | Root | [118] |
| 57 | Monoterpenoidal glucoside | 8-O-p-Hydroxl-6′-O-p-coumaryl-missaeno-sidic acid | P. javanica | Leaf | [42] |
| 58 | Monoterpenoidal glucoside | 7-O-E-3,4-Dimethoxycinnamoyl-6′-O-ß-d-glucopyranosylloganic acid | P. javanica | Leaf | [42] |
| 59 | Diterpene | 16-O-Methyl-cass-13(15) ene-16,18-dionic acid | P. bicolor | Root | [118] |
| 60 | Steroid | β-Sitosterol | P. speciosa | Seed | [117,119,120] |
| P. javanica | Leaf/stem | [42] | |||
| P. biglobosa | Seed oil | [121,122] | |||
| 61 | Steroid | Stigmasterol | P. speciosa | Seed | [117,119,120] |
| P. biglobosa | Seed oil | [121,122] | |||
| 62 | Steroid | Stigmasterol methyl ester | P. speciosa | Seed | [117,119] |
| 63 | Steroid | Stigmast-4-en-3-one | P. speciosa | Seed | [123] |
| 64 | Steroid | Stigmasta-5,24(28)-diene-3-ol | P. speciosa | Seed | [117] |
| 65 | Steroid | Campesterol | P. speciosa | Seed | [117,119] |
| P. biglobosa | Seed oil | [121,122] | |||
| 66 | Steroid | Stigmastan-6,22-diien,3,6-dedihydo- | P. speciosa | Seed | [119] |
| Miscellaneous Compounds | |||||
| 67 | Fatty acid | Arachidonic acid | P. speciosa | Seed | [117,119] |
| P. bicolor | Seed | [22] | |||
| P. biglobosa | Seed | [22] | |||
| 68 | Fatty acid | Linoleic acid chloride | P. speciosa | Seed | [117,119] |
| 69 | Fatty acid | Linoleic acid | P. speciosa | Seed | [117,119] |
| P. biglobosa | Seed | [22] | |||
| P. bicolor | Seed | [22] | |||
| 70 | Fatty acid | Squalene | P. speciosa | Seed | [117,119] |
| 71 | Fatty acid | Lauric acid | P. speciosa | Seed | [117,124] |
| 72 | Fatty acid | Stearic acid | P. speciosa | Seed | [117,119,124] |
| P. biglobosa | Seed | [22] | |||
| P. bicolor | Seed | [22] | |||
| 73 | Fatty acid | Stearoic acid | P. speciosa | Seed | [124] |
| 74 | Fatty acid | Eicosanic acid | P. speciosa | Seed | [124] |
| 75 | Fatty acid | Oleic acid | P. speciosa | Seed | [117,119,124] |
| 76 | Fatty acid | Palmitic acid | P. speciosa | Seed | [117,119,124] |
| P. biglobosa | Seed | [22] | |||
| P. bicolor | Seed | [22] | |||
| 77 | Fatty acid | Myristic acid | P. speciosa | Seed | [117,119,124] |
| 78 | Fatty acid | Undecanoic acid | P. speciosa | Seed | [119,124] |
| 79 | Fatty acid | Stearolic acid | P. speciosa | Seed | [119] |
| 80 | Fatty acid | Hydnocarpic acid | P. speciosa | Seed | [124] |
| 81 | Cyclic polysulfide | 1,3-dithiabutane | P. speciosa | Seed | [125] |
| 82 | Cyclic polysulfide | 2,4- Dithiapentane | P. speciosa | Seed | [125] |
| 83 | Cyclic polysulfide | 2,3,5-Trithiahexane | P. speciosa | Seed | [125] |
| 84 | Cyclic polysulfide | 2,4,6-Trithiaheptane | P. speciosa | Seed | [125] |
| 85 | Cyclic polysulfide | 1,2,4-Trithiolane | P. biglobosa | Seed | [116,126] |
| P. speciosa | Seed | [126,127,128] | |||
| 86 | Cyclic polysulfide | 1,3,5-Trithiane | P. speciosa | Seed | [128] |
| 87 | Cyclic polysulfide | 3,5-Dimethyl-1,2,4-trithiolane | P. speciosa | Seed | [128] |
| 88 | Cyclic polysulfide | Dimethyl tetrasulfid | P. speciosa | Seed | [128] |
| 89 | Cyclic polysulfide | 1,2,5,6-Tetrathio-cane | P. speciosa | Seed | [128] |
| 90 | Cyclic polysulfide | 1,2,3,5-Tetrathiane | P. speciosa | Seed | [128] |
| 91 | Cyclic polysulfide | 1,2,4,5-Tetrathiane | P. speciosa | Seed | [128] |
| 92 | Cyclic polysulfide | 1,2,4,6-Tetrathie-pane | P. speciosa | Seed | [126,128] |
| 93 | Cyclic polysulfide | 1,2,4,5,7,8- Hexathiolnane | P. speciosa | Seed | [126] |
| 94 | Cyclic poly-sulfide | Lenthionine | P. speciosa | Seed | [117,124,126,128] |
| 95 | Esters | n-Tetradecyl acetate | P. speciosa | Seed | [124] |
| 96 | Esters | Methyl linoleate | P. speciosa | Seed | [124] |
| 97 | Esters | Ethyl linoleate | P. speciosa | Seed | [117,124] |
| P. biglobosa | Seed | [116] | |||
| 98 | Ester | Butyl palmitate | P. speciosa | Seed | [117] |
| 99 | Esters | Ethyl palmitate | P. speciosa | Seed | [124] |
| 100 | Esters | Methyl palmitate | P. speciosa | Seed | [124] |
| 101 | Esters | Methyl laurate | P. speciosa | Seed | [124] |
| 102 | Esters | Dodecyl acrylate | P. speciosa | Seed | [124] |
| 103 | Esters | Methyl hexadecanoate | P. biglobosa | Seed | [116] |
| 104 | Ester | Ethyl stearate | P. speciosa | Seed | [117,124] |
| 105 | Ester | Methyl octadecanoate | P. biglobosa | Seed | [116] |
| 106 | Ester | Butyl stearate | P. speciosa | Seed | [124] |
| 107 | Ester | Propanoic acid, 3,3′-thiobis-didodecyl ester | P. speciosa | Seed | [124] |
| 108 | Ester | Linoleaidic acid methyl ester | P. speciosa | Seed | [119] |
| 109 | Alcohol | 2,6,10,14-Hexadecatetraen-1-ol | P. speciosa | Seed | [117] |
| 110 | Alcohol | 1-Octen-3-ol | P. biglobosa | Seed | [116] |
| 111 | Alcohol | 3-Ethyl-4-nonanol | P. speciosa | Seed | [117] |
| 112 | Alcohol | 1-Tridecanol | P. speciosa | Seed | [117,124] |
| 113 | Acid | Eicosanoic acid | P. speciosa | Seed | [117] |
| 114 | Acid | 16-O-Methyl-cass-13(15)ene-16,18-dionic acid | P. bicolor | Root | [118] |
| 115 | Acid | Elaidic acid | P. speciosa | Seed | [117,124] |
| 116 | Pyrazine | 2,5-Dimethyl pyrazine | P. biglobosa | Seed | [116] |
| 117 | Pyrazine | Trimethyl pyrazine | P. biglobosa | Seed | [116] |
| 118 | Pyrazine | 2-Ethyl-3,5-dimethyl pyrazine | P. biglobosa | Seed | [116] |
| 119 | Ketone | 2-Nonade-canone | P. speciosa | Seed | [117,124] |
| 120 | Ketone | 2-Pyrrolidi-none | P. speciosa | Seed | [117] |
| 121 | Ketone | Cyclodecanone | P. speciosa | Seed | [124] |
| 122 | Alkane | Cyclododecane | P. biglobosa | Seed | [116] |
| 123 | Alkane | Tetradecane | P. speciosa | Seed | [119] |
| 124 | Benzene glucoside | 3,4,5-Trimethoxyphenyl-1-O-ß-d-glucopy-ranoside | P. bicolor | Root | [118] |
| 125 | Aldehyde | 2-Decenal | P. speciosa | Seed | [117] |
| 126 | Aldehyde | Cyclo-decanone-2,4-decadienal | P. speciosa | Seed | [117] |
| 127 | Aldehyde | Pentanal | P. biglobosa | Seed | [116] |
| P. speciosa | Seed | [125] | |||
| 128 | Aldehyde | 3-Methylthio-propanal | P. biglobosa | Seed | [116] |
| 129 | Aldehyde | Tetradecanal | P. speciosa | Seed | [119,124] |
| 130 | Aldehyde | Pentadecanal | P. speciosa | Seed | [117,124] |
| 131 | Aldehyde | Hexadecanal | P. speciosa | Seed | [117,124] |
| 132 | Amine | Hexanamide | P. speciosa | Seed | [117] |
| 133 | Oil | Vitamin E | P. speciosa | Seed | [117,124] |
| Activity | Species | Part | Type of Extract/Compound | Key Findings | References |
|---|---|---|---|---|---|
| Antimicrobial | P. biglobosa | Leaf, stem bark, and root | Methanolic and aqueous | Active against S. aureus, B. subtilis, E. coli, P. aeruginosa. | [38] |
| P. biglobosa | Root bark | Aqueous and methanol | Active against E. coli, S. aureus, K. pneumoniae, P. aeruginosa. Activity: Aqueous > methanol | [34] | |
| P. biglobosa | Leaves and pod | Aqueous and ethanol | Active against S. aureus, E. aerogenes, S. typi, S. typhimurium, Shigella spp., E. coli, and P. aeruginosa (bacteria), Mucor spp., and Rhizopus spp. (fungi) | [133] | |
| P. biglobosa | Bark and leaves | Hydro-alcohol and aqueous | Active against E. coli, S. enterica, and S. dysenteriae. Activity: hydroalcoholic > aqueous | [65] | |
| P. speciosa | Seeds | Water suspension | Active against S. aureus, A. hydrophila, S. agalactiae, S. anginosus, and V. parahaemolyticus isolated from moribund fishes and shrimps | [143] | |
| P. speciosa | Seed peel | Ethyl acetate (EA) Hexane Ethanol | EA: Four times higher than streptomycin against S. aureus and three times higher for E. coli. Hexane: 50% inhibitory ability of streptomycin for both bacteria. Ethanol: no inhibition | [148] | |
| P. speciosa | Pod extract and its silver | Aqueous | Pod: active against P. aeruginosa Silver particles: active against P. aeruginosa | [145] | |
| P. speciosa | Sapwood, heartwood, and bark | Methanol | Bark: Active against G. trabeum. Sapwood and heartwood: No effect | [147] | |
| P. speciosa | Seeds | Chloroform, petroleum ether, Aqueous and methanol | Active against H. pylori except aqueous extract. Activity: chloroform > methanol > petroleum ether | [222] | |
| P. speciosa | Seed | Methanol Ethyl acetate | Methanol: active against H. pylori. Ethyl acetate: active against E. coli Both: no effect on S. typhimurium, S. typhi, and S sonnei | [144] | |
| P. javanica | Stem bark | Methanol | Good inhibitory activity against E. coli, S. aureus S. pyogenes found in chronic wound | [223] | |
| P. javanica | Stem bark | Methanol | Active against four Vibrio cholerae strains | [224] | |
| P. javanica | Leaves | Gold and silver nanoparticles | Good inhibitory activity against S. aureus | [151] | |
| P. javanica | Bark | Methanol extract and semi-polar fractions (chloroform and ethyl acetate) | Active against Neisseria gonorrhoeae. Chloroform showed the best activity | [97] | |
| P. javanica | Seeds, leaves and skin pods | Aqueous | Active against S. aureus, A. hydrophila, and S. typhimurium Not active against E. coli | [152] | |
| P. clappertoniana | Leaves and barks | Ethanol | Active against Salmonellae and Shigella | [73] | |
| P. clappertoniana | Stem bark and leaves | Aqueous and methanol | Active against S. aureus and P. aeruginosa. Methanol extract was more potent | [71] | |
| P. biglandulosa | Leaf | Methanol | Active against E. coli, P. aeruginosa, and S. aureus | [154] | |
| P. filicoidea | Stem barks | Aqueous, acetone and ethanol | Active against S. aureus, K. pneumoniae, P. aeruginosa, S. viridans and B. subtilis. Not active against E. coli | [50] | |
| P. bicolor | Leaves | Ethyl acetate, ethanol and aqueous | Active against E. coli, S. aureus, P. aeruginosa, A. niger, B. cereus and a fungus, C. utilis | [23] | |
| P. bicolor | Roots | Methanol, ethyl acetate and Aqueous | Active against C. diphtheria, K. pneumoniae, P. mirabilis, S. typhi, and S. pyogenes | [28] | |
| P. pendula | Seeds | Lectin | Reduced cellular infectivity of human cytomegalovirus in human embryo lung (HEL) cells. | [225] | |
| Hypoglycemic | P. speciosa | Seeds and pods | Chloroform | Strong glucose-lowering activity in alloxan-induced diabetic rats Activity: seeds > pod | [157] |
| P. speciosa | Rind, leaves and seeds | Ethanol | Inhibited α-glucosidase activity in rat Activity: rind > leaf > seed | [158] | |
| P. speciosa | Seed | Chloroform | Reduced plasma glucose levels in alloxan-induced diabetic rats | [120] | |
| P. biglobosa | Fermented seeds | Methanol and aqueous | Reduced fasting plasma glucose in alloxan-induced diabetic rats | [160,161] | |
| P. biglobosa | Seeds | Protein | Significantly increased lipid peroxidation product levels in brain and testes of diabetic rats | [226] | |
| P. biglobosa | Seeds | Methanol and fractions (chloroform and n-hexane) | Showed glucose-lowering effect Activity: chloroform > methanol > n-hexane | [40] | |
| P. javanica | Fruits | Ethyl acetate fraction | Reduced blood glucose inhibited α-glucosidase and α-amylase in streptozotocin-induced diabetic rats | [18] | |
| Antitumor/ Anticancer | P javanica | Fruits | Aqueous methanol | Increased apoptosis in sarcoma-180 cancer cell lines | [227] |
| P javanica | Seeds | Methanol | Caused 50% death in HepG2 (liver cancer cell) but not cytotoxic to normal cells | [44] | |
| P javanica | Seeds | Lectin | Inhibited proliferation in cancerous cell lines; P388DI and J774, B-cell hybridoma and HB98 cell line | [173] | |
| P. speciosa | Seed coats | Methanol extract | Demonstrated selective cytotoxicity to MCG-7 and T47D (breast cancer), HCT-116 (colon cancer) | [228] | |
| P. speciosa | Pods | Methanolic ethyl acetate fraction | Showed selective cytotoxicity on breast cancer cells MCF-7 | [170] | |
| P. biglobosa | Leaves and stem | Methanol | Antiproliferative effect in human cancer cells T-549, BT-20, and PC-3 | [174] | |
| P. filicoidea | Leaves | Methanol | Antiproliferative effect in in human cancer cells T-549, BT-20, and PC-3 | [174] | |
| Antiproliferative and anti-mutagenic | P. biglandulosa | Seeds | Lectin | T cell mitogen and antiproliferative against P388DI and J774 cancer cell lines | [173] |
| Antihypertensive | P. speciosa | Seeds | Aqueous | Showed moderate ACE-inhibitory activity in in vitro | [191] |
| P. speciosa | Seeds | Peptide | Inhibited angiotensin-converting enzyme (ACE) in rats. No effect observed in non-hydrolyzed samples | [189,190] | |
| P. speciosa | Pods | Methanol | Prevented the increases in blood pressure and angiotensin-converting enzyme (ACE) and restored nitric oxide in hypertensive rat model | [112] | |
| P. biglobosa | Stem bark | Aqueous | Induced hypotension in adrenaline-induced hypertensive rabbits | [181] | |
| P. biglobosa | Roasted and fermented seeds | Aqueous | Induced relaxation in rat aorta precontracted with phenylephrine in the presence or absence of endothelium. | [180] | |
| P. biglobosa | fermented seeds | Aqueous | Lower blood pressure, blood glucose, and heart rate, high level of magnesium as well as improved lipid profile in patients with hypertension | [178] | |
| Antidiarrheal | P. biglobosa | Stem bark | Aqueous and fractions | The extract of stem bark exhibit dose-dependent antidiarrheal activity at different concentrations in albino rats with castor oil-induced diarrhea | [45] |
| P. biglobosa | Leaves and stem bark | Aqueous and ethanol | Reduced frequency of stooling in castor-oil induced diarrhea in rats | [193] | |
| P. biglobosa | Stem-bark | 70% Methanol | The extract exhibited 100% protections at 100 and 200 mg/kg bw in the diarrheal rats | [59] | |
| P. filicoidea | Stem bark | Aqueous | Reduced frequency of stooling and improved transit time at 100 and 200 mg/kg bw | [192] | |
| Antiulcer | P. speciosa | Leaves | Ethanol | Reduced mucosal injury and increased in periodic acid-Schiff (PAS) staining induced by ethanol | [198] |
| P. speciosa | Seed | Ethanol | Decreased gastric juice acidity, lesion length, collagen content and fibrosis in indomethacin-induced peptic ulcer in rats | [197] | |
| P. platycephala | Leaves | Ethanol | Reduced gastric mucosal lesion induced by ethanol, ischemia-reperfusion and ethanol-HCl | [199] | |
| Antianemic | P. biglobosa | Combination of fermented seed with other fermented products | Aqueous | Increased hemoglobin, red blood cell, white blood cell levels and packed cell volume in albino rats | [204] |
| P. biglobosa | Seeds | Ethanol | Increased hemoglobin levels in NaNO2-induced anemic mice | [205] | |
| P. speciosa | Seeds | Ethanol | Increased hemoglobin levels in NaNO2-induced anemic mice | [205] | |
| Antiangiogenic | P.biglandulosa | Fruit and β-sitosterol | Ethanol | The extract and the isolated compound showed antiangiogenic activity on the caudal fin of adult zebrafish | [175] |
| P. speciosa | Pods | Methanol and water sub-extract | Inhibited more than 50% micro vessel outgrowth in rat aortae and HUVECs | [170] | |
| Antimalarial | P. biglobosa | Stem bark | Methanol and fractions | Showed antiplasmodial activity caused by P. berghei and P. falciparum | [11] |
| Nephroprotective | P. clappertoniana | Seed | Aqueous | Reduced serum creatinine, Na, urine proteins and leukocytes and kidney weight in gentamicin-induced renal damage in rats | [75] |
| Hepatoprotective | P. biglobosa | Stem barks | Methanol | Reduced serum alanine and aspartate transaminases, and alkaline phosphatase in paracetamol-induced hepatotoxicity rat model | [216] |
| Wound healing | P. pendula | Seeds | Lectin | Increased skin wound repair in immunosuppressed mice | [217] |
| Anti-inflammatory | P. speciosa | Pods | Ethyl acetate fraction | Reduced iNOS activity, COX-2, VCAM-1 and NF-κB expressions in cardiomyocytes exposed to tumor necrosis factor-α | [229] |
| P. speciosa | Pods | Ethyl acetate fraction | Reduced iNOS activity, COX-2, VCAM-1 and NF-κB expressions in HUVECs exposed to tumor necrosis factor-α | [230] | |
| P. biglobosa | Stalk | Methanol | Inhibited croton pellet granuloma formation and carrageenin-induced rat paw edema | [206] | |
| P. biglobosa | Seeds | Lectin | Lectin showed anti-inflammatory effect by inhibition of pro-inflammatory cytokine release and stimulation of anti-inflammatory cytokine release on peritonitis induced model mice | [208] | |
| P. biglobosa | Stem bark | Hexane | Reduced carrageenan- and PMA-induced edema in mice | [29] | |
| P. biglobosa | Fruit | 70% Methanol | Increased percentage protection of the human red blood cell membrane | [209] | |
| P. platycephala | Seeds | Lectin | Lectin showed antinociceptive effect in the mouse model of acetic acid-induced | [207] | |
| Antioxidant | P. javanica | Leaves | Hexane, ethyl acetate, and methanol | Methanol extract showed the highest antioxidant potential activities (DPPH test) of about 85% and (FRAP test) of about 0.9 mM Fe (II)/g dry | [231] |
| P. javanica | Leaves | Aqueous, ethanol and methanol | All the extracts exhibited good antioxidant activity. The aqueous extract showed the highest values of 47.42 and 26.6 mg of ascorbic acid equivalent/g in DPPH and FRAP tests, respectively | [232] | |
| P. javanica | Pods | Methanol and acetone | High content of total phenolic and flavonoid. Showed high reducing power and strong radical scavenging activity. | [212] | |
| P. javanica | Fruit | Methanol | Showed increased DPPH and ferric-reducing power activities concentration-dependently | [210] | |
| P. speciosa | Pod | Methanol | Increased DPPH scavenging activity | [233] | |
| P. speciosa | Pod | Ethyl acetate fraction | Reduced NOX4, SOD1, p38 MAPK protein expressions and ROS level | [230] | |
| P. speciosa | Pod | Aqueous and ethanolic | Increased DPPH and ABTS scavenging activities, reduced lipid peroxidation Activity: ethanol > aqueous | [107] | |
| P. speciosa | Seeds | Ethanol | Extract exhibited significant activity (DPPH and FRAP tests) | [213] | |
| P. speciosa | Seed coats and pods | Ethanol | Reduced Heinz body formation in erythrocytes incubated with acetyl phenylhydrazine. Activity: seed coat > pods > | [215] | |
| P. speciosa | Pods | Ethanol | Increased DPPH scavenging activity | [234] | |
| P. biglobosa | Fermented and unfermented seed | Aqueous | Fermented seed increased reduction of Fe3+ to Fe2+. | [211] | |
| P. biglobosa | Stem bark | Aqueous-methanolic | Mitigated ferric-induced lipid peroxidation in rat tissues and increased scavenging activities against DPPH and ABTS, ferric-reducing ability | [235] | |
| P. biglobosa | Fruit | Methanol and hydro-ethanol | Increased DPPH scavenging activity and reducing power. | [210] | |
| P. biglobosa | Fruit | Hydroethanolic and methanol | Increased scavenging activity against DPPH free radical Activity: methanol > hydroethanolic | [210] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saleh, M.S.M.; Jalil, J.; Zainalabidin, S.; Asmadi, A.Y.; Mustafa, N.H.; Kamisah, Y. Genus Parkia: Phytochemical, Medicinal Uses, and Pharmacological Properties. Int. J. Mol. Sci. 2021, 22, 618. https://doi.org/10.3390/ijms22020618
Saleh MSM, Jalil J, Zainalabidin S, Asmadi AY, Mustafa NH, Kamisah Y. Genus Parkia: Phytochemical, Medicinal Uses, and Pharmacological Properties. International Journal of Molecular Sciences. 2021; 22(2):618. https://doi.org/10.3390/ijms22020618
Chicago/Turabian StyleSaleh, Mohammed S. M., Juriyati Jalil, Satirah Zainalabidin, Ahmad Yusof Asmadi, Nor Hidayah Mustafa, and Yusof Kamisah. 2021. "Genus Parkia: Phytochemical, Medicinal Uses, and Pharmacological Properties" International Journal of Molecular Sciences 22, no. 2: 618. https://doi.org/10.3390/ijms22020618
APA StyleSaleh, M. S. M., Jalil, J., Zainalabidin, S., Asmadi, A. Y., Mustafa, N. H., & Kamisah, Y. (2021). Genus Parkia: Phytochemical, Medicinal Uses, and Pharmacological Properties. International Journal of Molecular Sciences, 22(2), 618. https://doi.org/10.3390/ijms22020618






