What Are Reactive Oxygen Species, Free Radicals, and Oxidative Stress in Skin Diseases?
Abstract
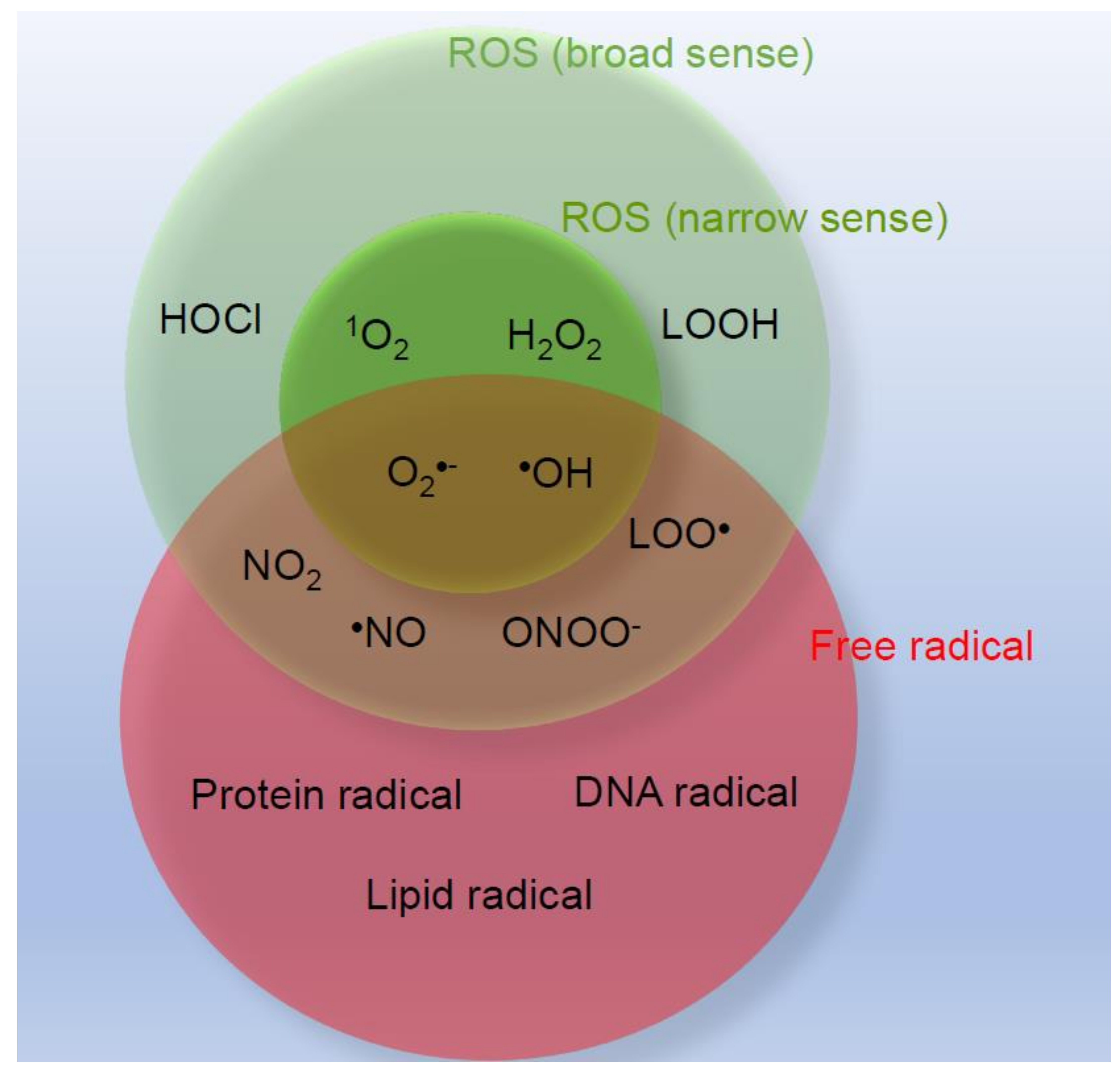
1. Introduction
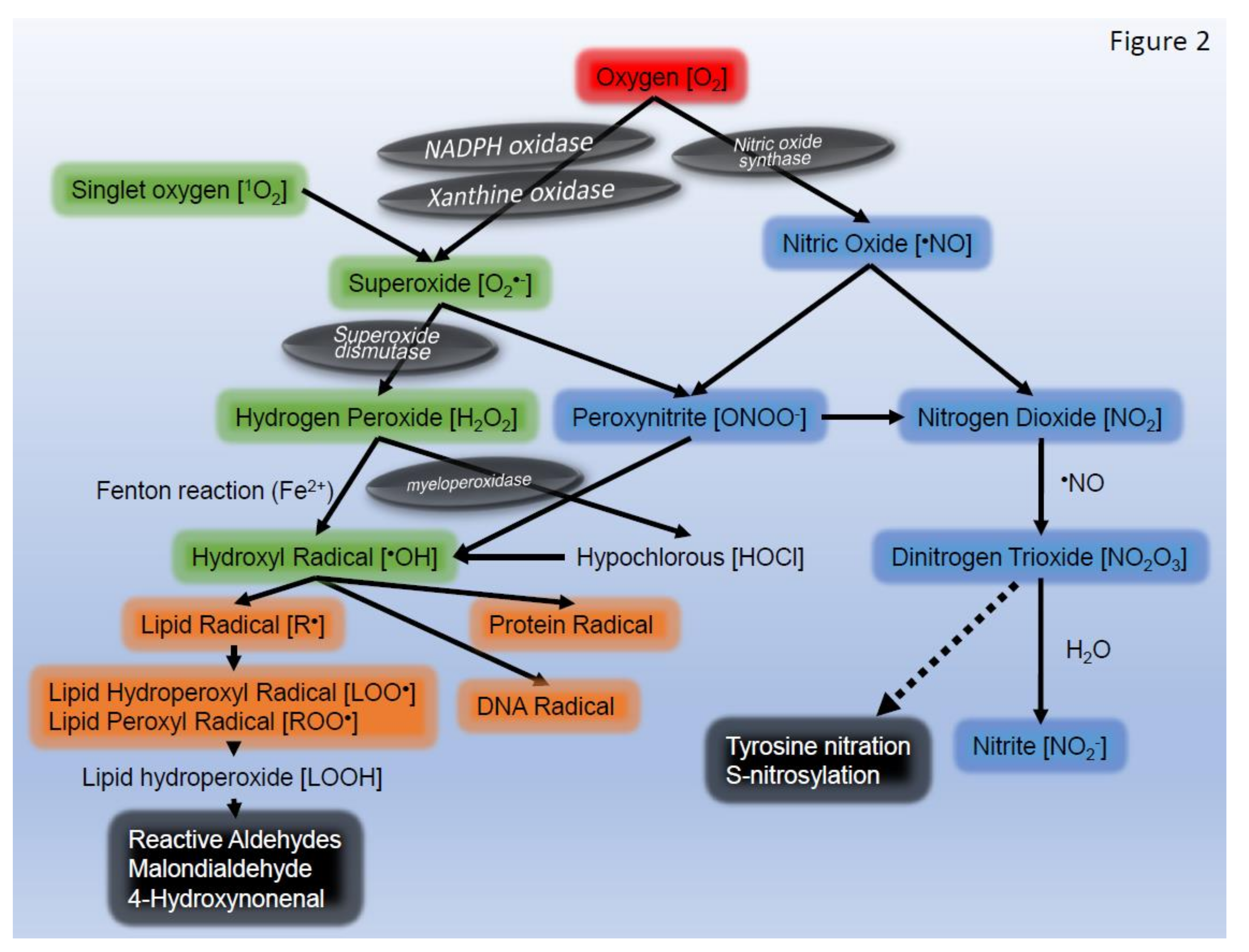
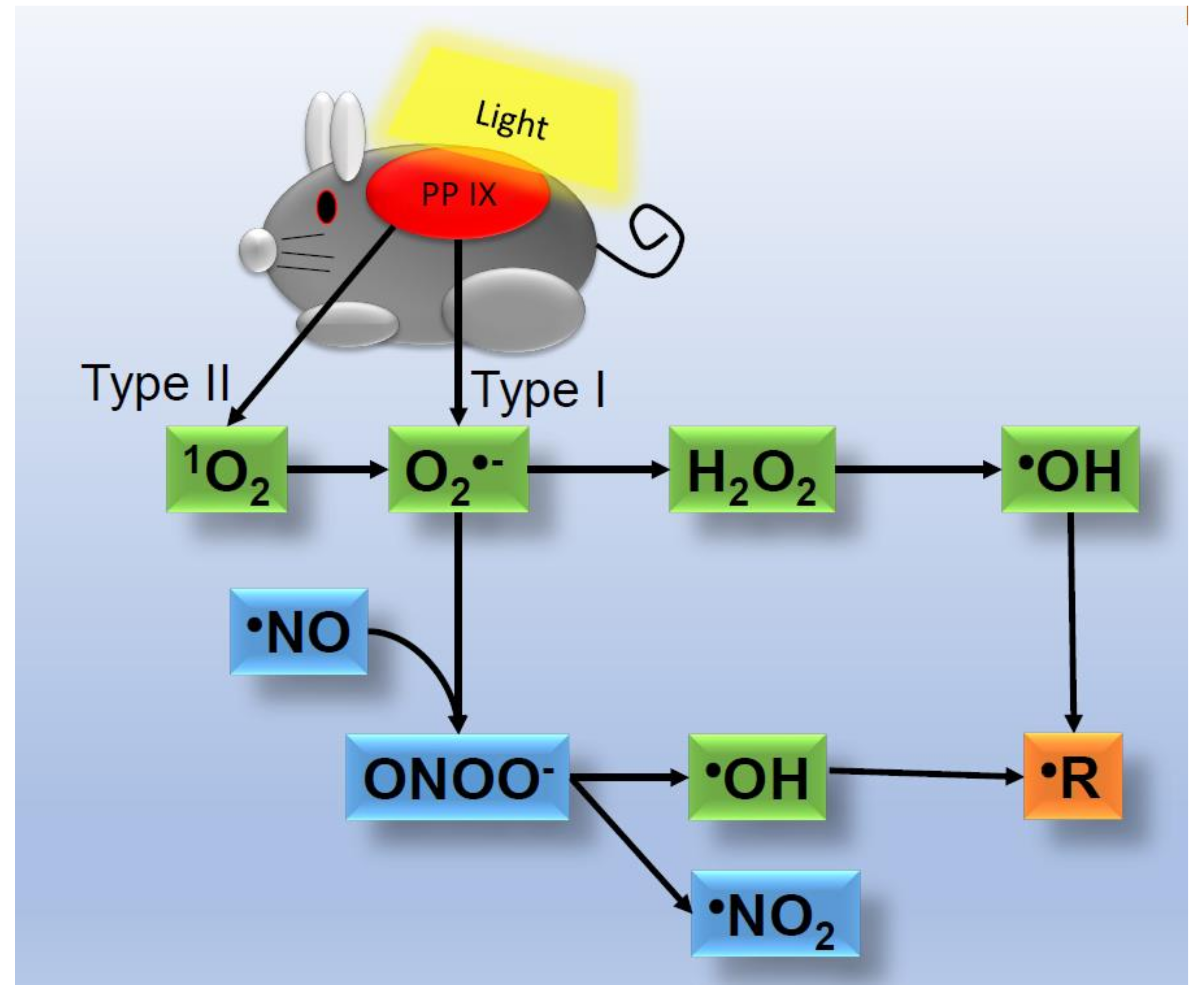
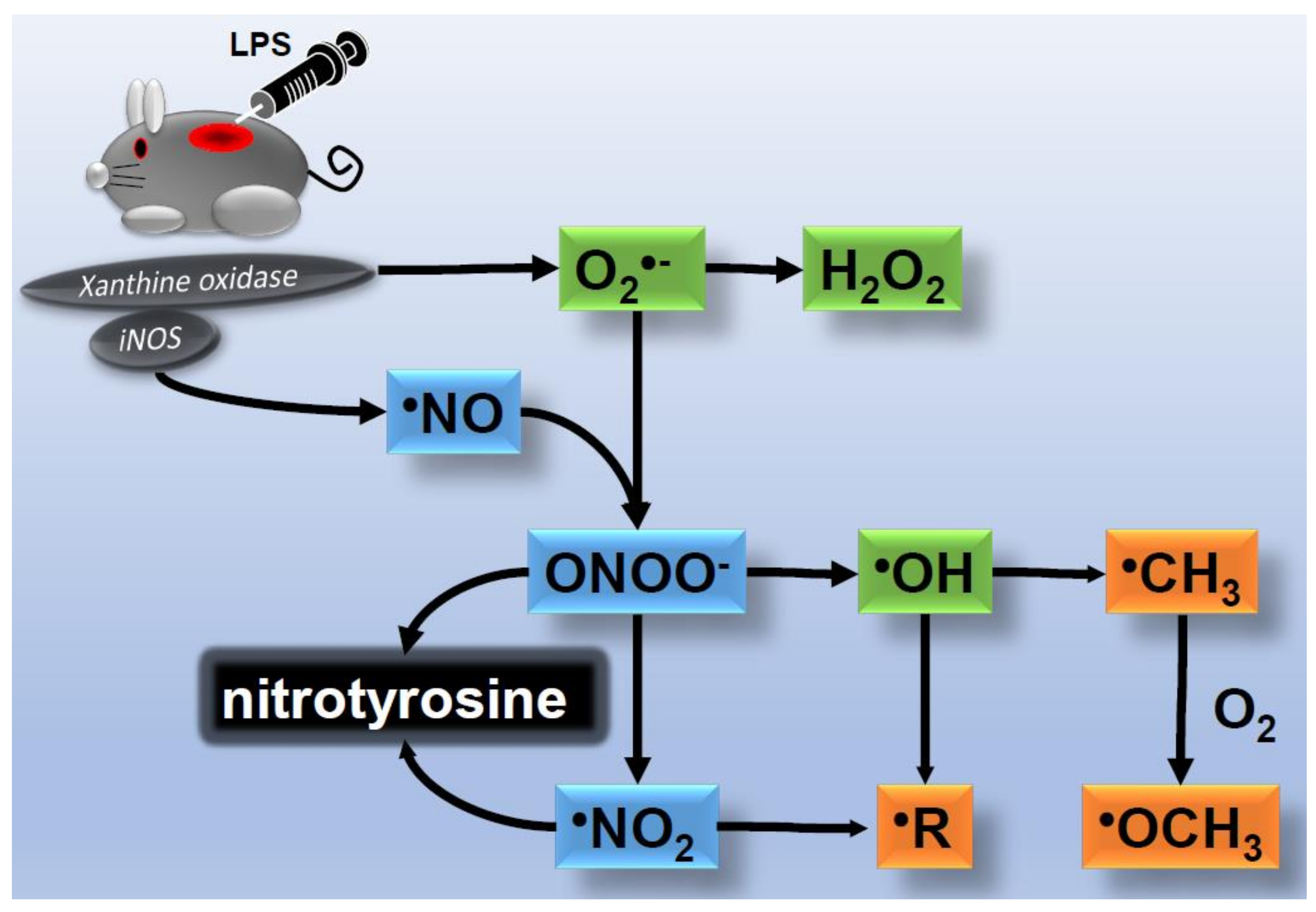
2. ROS Generation in Skin
3. Roles of ROS Generated in Skin
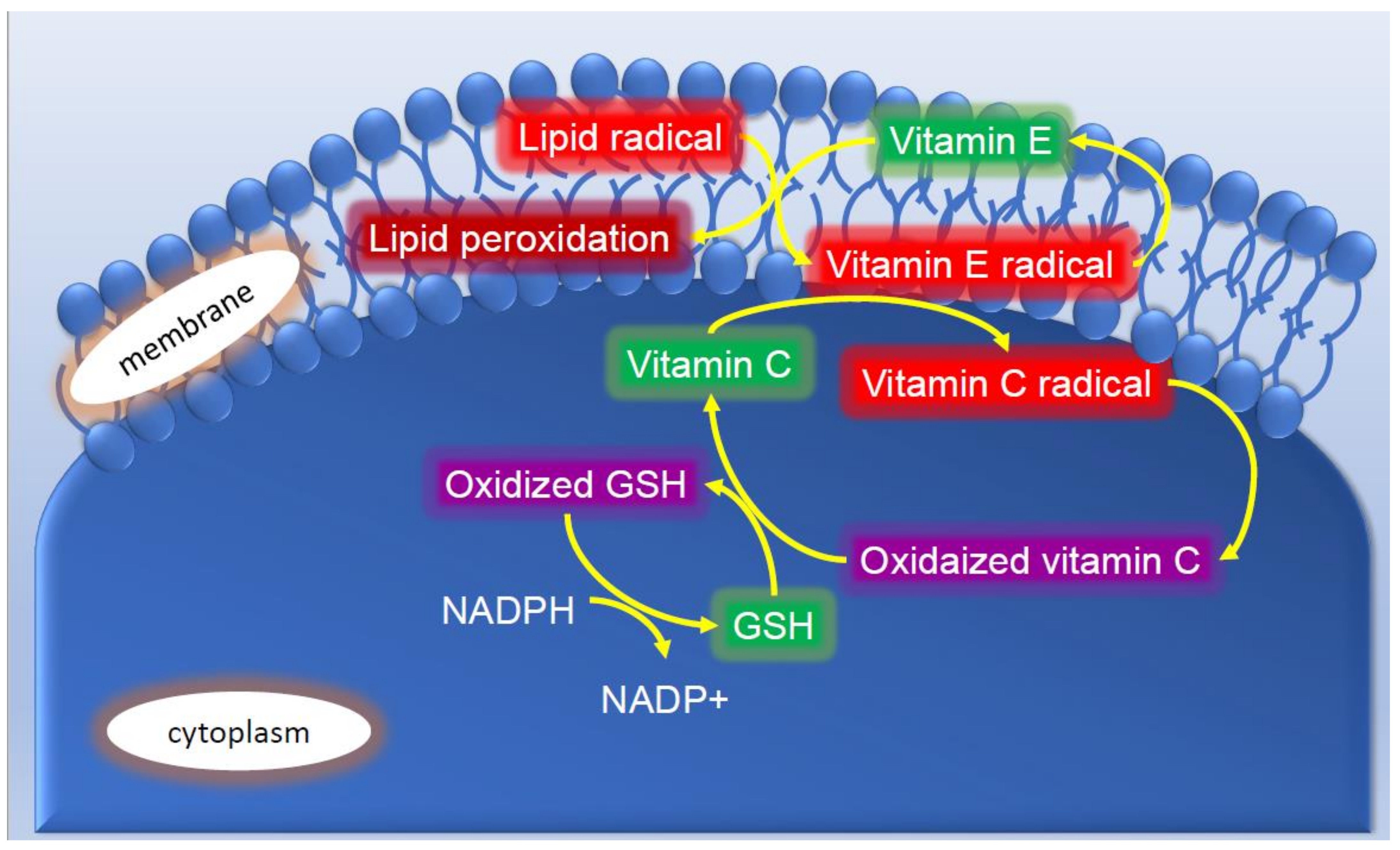
4. Antioxidants in Skin Tissue
5. ROS and Skin Diseases
5.1. Contact Dermatitis
5.2. Urticaria
5.3. Atopic Dermatitis
5.4. Psoriasis Vulgaris
5.5. Acne Vulgaris
5.6. Skin Cancer
5.7. Aging
5.8. Vitiligo Vulgaris
5.9. Alopecia Areata
5.10. Wound Healing
5.11. Granuloma
5.12. Other Skin Diseases
6. Concludings
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Carraro, C.; Pathak, M.A. Studies on the nature of in vitro and in vivo photosensitization reactions by psoralens and porphyrins. J. Investig. Dermatol. 1988, 90, 267–275. [Google Scholar] [CrossRef]
- Poppe, W.; Grossweiner, L.I. Photodynamic sensitization by 8-methoxypsoralen via the singlet oxygen mechanism. Photochem. Photobiol. 1975, 22, 217–219. [Google Scholar] [CrossRef]
- Nakai, K.; Motten, A.G.; Chignell, C.F. An in vivo study of free radicals generated in murine skin by protoporphyrin IX and visible light. Photochem. Photobiol. 2006, 82, 738–740. [Google Scholar] [CrossRef] [PubMed]
- Hasan, T.; Kochevar, I.E.; McAuliffe, D.J.; Cooperman, B.S.; Abdulah, D. Mechanism of tetracycline phototoxicity. J. Investig. Dermatol. 1984, 83, 179–183. [Google Scholar] [CrossRef]
- Al-Nu’airat, J.; Oluwoye, I.; Zeinali, N.; Altarawneh, M.; Dlugogorski, B.Z. Review of Chemical Reactivity of Singlet Oxygen with Organic Fuels and Contaminants. Chem. Rec. 2021, 21, 315–342. [Google Scholar] [CrossRef]
- Lui, H.; Hobbs, L.; Tope, W.D.; Lee, P.K.; Elmets, C.; Provost, N.; Chan, A.; Neyndorff, H.; Su, X.Y.; Jain, H.; et al. Photodynamic therapy of multiple nonmelanoma skin cancers with verteporfin and red light-emitting diodes: Two-year results evaluating tumor response and cosmetic outcomes. Arch. Dermatol. 2004, 140, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, F.H.; Lopes, J.D.; Anderson, R.R. Photodynamic therapy for acne vulgaris: A critical review from basics to clinical practice: Part I. Acne vulgaris: When and why consider photodynamic therapy? J. Am. Acad. Dermatol. 2010, 63, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, F.H.; Torezan, L.; Anderson, R.R. Photodynamic therapy for acne vulgaris: A critical review from basics to clinical practice: Part II. Understanding parameters for acne treatment with photodynamic therapy. J. Am. Acad. Dermatol. 2010, 63, 195–211. [Google Scholar] [CrossRef] [PubMed]
- Belambri, S.A.; Rolas, L.; Raad, H.; Hurtado-Nedelec, M.; Dang, P.M.; El-Benna, J. NADPH oxidase activation in neutrophils: Role of the phosphorylation of its subunits. Eur. J. Clin. Invest. 2018, 48 (Suppl. 2), e12951. [Google Scholar] [CrossRef] [PubMed]
- Moghadam, Z.M.; Henneke, P.; Kolter, J. From Flies to Men: ROS and the NADPH Oxidase in Phagocytes. Front. Cell Dev. Biol. 2021, 9, 628991. [Google Scholar] [CrossRef]
- Manea, A. NADPH oxidase-derived reactive oxygen species: Involvement in vascular physiology and pathology. Cell Tissue Res. 2010, 342, 325–339. [Google Scholar] [CrossRef]
- Steinbrenner, H.; Ramos, M.C.; Stuhlmann, D.; Mitic, D.; Sies, H.; Brenneisen, P. Tumor promoter TPA stimulates MMP-9 secretion from human keratinocytes by activation of superoxide-producing NADPH oxidase. Free Radic. Res. 2005, 39, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Papparella, I.; Ceolotto, G.; Lenzini, L.; Mazzoni, M.; Franco, L.; Sartori, M.; Ciccariello, L.; Semplicini, A. Angiotensin II-induced over-activation of p47phox in fibroblasts from hypertensives: Which role in the enhanced ERK1/2 responsiveness to angiotensin II? J. Hypertens. 2005, 23, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Rudolf, J.; Raad, H.; Taieb, A.; Rezvani, H.R. NADPH Oxidases and Their Roles in Skin Homeostasis and Carcinogenesis. Antioxid. Redox Signal. 2018, 28, 1238–1261. [Google Scholar] [CrossRef] [PubMed]
- Deliconstantinos, G.; Villiotou, V.; Stavrides, J.C. Alterations of nitric oxide synthase and xanthine oxidase activities of human keratinocytes by ultraviolet B radiation. Potential role for peroxynitrite in skin inflammation. Biochem. Pharmacol. 1996, 51, 1727–1738. [Google Scholar] [CrossRef]
- Meneshian, A.; Bulkley, G.B. The physiology of endothelial xanthine oxidase: From urate catabolism to reperfusion injury to inflammatory signal transduction. Microcirculation 2002, 9, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Rees, R.; Smith, D.; Li, T.D.; Cashmer, B.; Garner, W.; Punch, J.; Smith, D.J., Jr. The role of xanthine oxidase and xanthine dehydrogenase in skin ischemia. J. Surg. Res. 1994, 56, 162–167. [Google Scholar] [CrossRef][Green Version]
- Siegel, S.M.; Weintraub, R.L. The effect of light on xanthine oxidase activity. Biochem. J. 1952, 52, 324–326. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Kadiiska, M.B.; Jiang, J.J.; Stadler, K.; Mason, R.P. Free radical production requires both inducible nitric oxide synthase and xanthine oxidase in LPS-treated skin. Proc. Natl. Acad. Sci. USA 2006, 103, 4616–4621. [Google Scholar] [CrossRef]
- Abreu, I.A.; Cabelli, D.E. Superoxide dismutases-a review of the metal-associated mechanistic variations. Biochim. Biophys. Acta 2010, 1804, 263–274. [Google Scholar] [CrossRef]
- Kadiiska, M.B.; Burkitt, M.J.; Xiang, Q.H.; Mason, R.P. Iron supplementation generates hydroxyl radical in vivo. An ESR spin-trapping investigation. J. Clin. Invest. 1995, 96, 1653–1657. [Google Scholar] [CrossRef]
- Pelegrino, M.T.; Paganotti, A.; Seabra, A.B.; Weller, R.B. Photochemistry of nitric oxide and S-nitrosothiols in human skin. Histochem. Cell Biol. 2020, 153, 431–441. [Google Scholar] [CrossRef]
- Sirsjo, A.; Karlsson, M.; Gidlof, A.; Rollman, O.; Torma, H. Increased expression of inducible nitric oxide synthase in psoriatic skin and cytokine-stimulated cultured keratinocytes. Br. J. Dermatol. 1996, 134, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Belmont, H.M.; Levartovsky, D.; Goel, A.; Amin, A.; Giorno, R.; Rediske, J.; Skovron, M.L.; Abramson, S.B. Increased nitric oxide production accompanied by the up-regulation of inducible nitric oxide synthase in vascular endothelium from patients with systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1810–1816. [Google Scholar] [CrossRef]
- Stallmeyer, B.; Kampfer, H.; Kolb, N.; Pfeilschifter, J.; Frank, S. The function of nitric oxide in wound repair: Inhibition of inducible nitric oxide-synthase severely impairs wound reepithelialization. J. Investig. Dermatol. 1999, 113, 1090–1098. [Google Scholar] [CrossRef]
- Nakai, K.; Fujii, S.; Yamamoto, A.; Igarashi, J.; Kubota, Y.; Kosaka, H. Effects of high glucose on NO synthesis in human keratinocyte cell line (HaCaT). J. Dermatol. Sci. 2003, 31, 211–218. [Google Scholar] [CrossRef]
- Nakai, K.; Kubota, Y.; Kosaka, H. Inhibition of nuclear factor kappa B activation and inducible nitric oxide synthase transcription by prolonged exposure to high glucose in the human keratinocyte cell line HaCaT. Br. J. Dermatol. 2004, 150, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Mason, R.P. Immunochemical detection of nitric oxide and nitrogen dioxide trapping of the tyrosyl radical and the resulting nitrotyrosine in sperm whale myoglobin. Free Radic. Biol. Med. 2005, 39, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.K.; Yim, M.B.; Stadtman, E.R.; Chock, P.B. Peroxynitrite disables the tyrosine phosphorylation regulatory mechanism: Lymphocyte-specific tyrosine kinase fails to phosphorylate nitrated cdc2(6-20)NH2 peptide. Proc. Natl. Acad. Sci. USA 1996, 93, 3377–3382. [Google Scholar] [CrossRef] [PubMed]
- Gutteridge, J.M. Lipid peroxidation and antioxidants as biomarkers of tissue damage. Clin. Chem. 1995, 41 Pt 2, 1819–1828. [Google Scholar] [CrossRef]
- Shigenaga, M.K.; Gimeno, C.J.; Ames, B.N. Urinary 8-hydroxy-2′-deoxyguanosine as a biological marker of in vivo oxidative DNA damage. Proc. Natl. Acad. Sci. USA 1989, 86, 9697–9701. [Google Scholar] [CrossRef] [PubMed]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Nakai, K.; Yoneda, K.; Moriue, T.; Igarashi, J.; Kosaka, H.; Kubota, Y. HB-EGF-induced VEGF production and eNOS activation depend on both PI3 kinase and MAP kinase in HaCaT cells. J. Dermatol. Sci. 2009, 55, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Yoneda, K.; Ishihara, Y.; Ohmori, K.; Moriue, T.; Igarashi, J.; Kohno, M.; Kosaka, H.; Kubota, Y. Lipid peroxidation-induced VEGF expression in the skin of KKAy obese mice. Exp. Dermatol. 2011, 20, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Yoneda, K.; Igarashi, J.; Moriue, T.; Kosaka, H.; Kubota, Y. Angiotensin II enhances EGF receptor expression levels via ROS formation in HaCaT cells. J. Dermatol. Sci. 2008, 51, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Karita, S.; Igarashi, J.; Tsukamoto, I.; Hirano, K.; Kubota, Y. COA-Cl prevented TGF-β1-induced CTGF expression by Akt dephosphorylation in normal human dermal fibroblasts, and it attenuated skin fibrosis in mice models of systemic sclerosis. J. Dermatol. Sci. 2019, 94, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Arbiser, J.L.; Bonner, M.Y.; Ward, N.; Elsey, J.; Rao, S. Selenium unmasks protective iron armor: A possible defense against cutaneous inflammation and cancer. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 2518–2527. [Google Scholar] [CrossRef]
- Gagliardi, M.; Saverio, V.; Monzani, R.; Ferrari, E.; Piacentini, M.; Corazzari, M. Ferroptosis: A new unexpected chance to treat metastatic melanoma? Cell Cycle 2020, 19, 2411–2425. [Google Scholar] [CrossRef]
- Li, P.; Jiang, M.; Li, K.; Li, H.; Zhou, Y.; Xiao, X.; Xu, Y.; Krishfield, S.; Lipsky, P.E.; Tsokos, G.C.; et al. Glutathione peroxidase 4-regulated neutrophil ferroptosis induces systemic autoimmunity. Nat. Immunol. 2021, 22, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Nandi, A.; Yan, L.J.; Jana, C.K.; Das, N. Role of Catalase in Oxidative Stress- and Age-Associated Degenerative Diseases. Oxid. Med. Cell. Longev. 2019, 2019, 9613090. [Google Scholar] [CrossRef]
- Ursini, F.; Maiorino, M.; Brigelius-Flohe, R.; Aumann, K.D.; Roveri, A.; Schomburg, D.; Flohe, L. Diversity of glutathione peroxidases. Methods Enzymol. 1995, 252, 38–53. [Google Scholar]
- Brigelius-Flohe, R.; Maiorino, M. Glutathione peroxidases. Biochim. Biophys. Acta 2013, 1830, 3289–3303. [Google Scholar] [CrossRef]
- Mironczuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med. Sci. 2018, 63, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Urushihara, M.; Kubota, Y.; Kosaka, H. Ascorbate enhances iNOS activity by increasing tetrahydrobiopterin in RAW 264.7 cells. Free Radic. Biol. Med. 2003, 35, 929–937. [Google Scholar] [CrossRef]
- Hsu, S. Green tea and the skin. J. Am. Acad. Dermatol. 2005, 52, 1049–1059. [Google Scholar] [CrossRef] [PubMed]
- Mehling, R.; Schwenck, J.; Lemberg, C.; Trautwein, C.; Zizmare, L.; Kramer, D.; Muller, A.; Fehrenbacher, B.; Gonzalez-Menendez, I.; Quintanilla-Martinez, L.; et al. Immunomodulatory role of reactive oxygen species and nitrogen species during T cell-driven neutrophil-enriched acute and chronic cutaneous delayed-type hypersensitivity reactions. Theranostics 2021, 11, 470–490. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Zilmer, M.; Eisen, M.; Rehema, A.; Kullisaar, T.; Vihalemm, T.; Zilmer, K. Nickel sulphate and epoxy resin: Differences in iron status and glutathione redox ration at the time of patch testing. Arch. Dermatol. Res. 2004, 295, 517–520. [Google Scholar] [CrossRef]
- Brans, R.; Dickel, H.; Bruckner, T.; Coenraads, P.J.; Heesen, M.; Merk, H.F.; Blomeke, B. MnSOD polymorphisms in sensitized patients with delayed-type hypersensitivity reactions to the chemical allergen para-phenylene diamine: A case-control study. Toxicology 2005, 212, 148–154. [Google Scholar] [CrossRef]
- Bruchhausen, S.; Zahn, S.; Valk, E.; Knop, J.; Becker, D. Thiol antioxidants block the activation of antigen-presenting cells by contact sensitizers. J. Investig. Dermatol. 2003, 121, 1039–1044. [Google Scholar] [CrossRef]
- Ormerod, A.D.; Dwyer, C.M.; Reid, A.; Copeland, P.; Thompson, W.D. Inducible nitric oxide synthase demonstrated in allergic and irritant contact dermatitis. Acta Derm. Venereol. 1997, 77, 436–440. [Google Scholar]
- Ross, R.; Gillitzer, C.; Kleinz, R.; Schwing, J.; Kleinert, H.; Forstermann, U.; Reske-Kunz, A.B. Involvement of NO in contact hypersensitivity. Int. Immunol. 1998, 10, 61–69. [Google Scholar] [CrossRef]
- Nordberg, J.; Zhong, L.; Holmgren, A.; Arner, E.S. Mammalian thioredoxin reductase is irreversibly inhibited by dinitrohalobenzenes by alkylation of both the redox active selenocysteine and its neighboring cysteine residue. J. Biol. Chem. 1998, 273, 10835–10842. [Google Scholar] [CrossRef] [PubMed]
- Briganti, S.; Cristaudo, A.; D’Argento, V.; Cassano, N.; Turbino, L.; Guarrera, M.; Vena, G.; Picardo, M. Oxidative stress in physical urticarias. Clin. Exp. Dermatol. 2001, 26, 284–288. [Google Scholar] [CrossRef]
- Raho, G.; Cassano, N.; D’Argento, V.; Vena, G.A.; Zanotti, F. Over-expression of Mn-superoxide dismutase as a marker of oxidative stress in lesional skin of chronic idiopathic urticaria. Clin. Exp. Dermatol. 2003, 28, 318–320. [Google Scholar] [CrossRef] [PubMed]
- Yoshimaru, T.; Suzuki, Y.; Matsui, T.; Yamashita, K.; Ochiai, T.; Yamaki, M.; Shimizu, K. Blockade of superoxide generation prevents high-affinity immunoglobulin E receptor-mediated release of allergic mediators by rat mast cell line and human basophils. Clin. Exp. Allergy 2002, 32, 612–618. [Google Scholar] [CrossRef] [PubMed]
- Cassano, N.; Raho, G.; Filieri, M.; D’Argento, V.; Amoruso, A.; Filotico, R.; Vena, G.A. Influence of desloratadine on oxidative stress markers in patients with chronic idiopathic urticaria. Int. J. Dermatol. 2006, 45, 394–396. [Google Scholar] [CrossRef]
- Kasperska-Zajac, A.; Brzoza, Z.; Polaniak, R.; Rogala, B.; Birkner, E. Markers of antioxidant defence system and lipid peroxidation in peripheral blood of female patients with chronic idiopathic urticaria. Arch. Dermatol. Res. 2007, 298, 499–503. [Google Scholar] [CrossRef]
- Briganti, S.; Picardo, M. Antioxidant activity, lipid peroxidation and skin diseases. What’s new. J. Eur. Acad. Dermatol. Venereol. 2003, 17, 663–669. [Google Scholar] [CrossRef]
- Nakai, K.; Yoneda, K.; Maeda, R.; Munehiro, A.; Fujita, N.; Yokoi, I.; Moriue, J.; Moriue, T.; Kosaka, H.; Kubota, Y. Urinary biomarker of oxidative stress in patients with psoriasis vulgaris and atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 1405–1408. [Google Scholar] [CrossRef]
- Leveque, N.; Robin, S.; Muret, P.; Mac-Mary, S.; Makki, S.; Humbert, P. High iron and low ascorbic acid concentrations in the dermis of atopic dermatitis patients. Dermatology 2003, 207, 261–264. [Google Scholar] [CrossRef]
- Bengtsson, A.; Lundberg, M.; Avila-Carino, J.; Jacobsson, G.; Holmgren, A.; Scheynius, A. Thiols decrease cytokine levels and down-regulate the expression of CD30 on human allergen-specific T helper (Th) 0 and Th2 cells. Clin. Exp. Immunol. 2001, 123, 350–360. [Google Scholar] [CrossRef]
- Nakai, K.; Yoneda, K.; Murakami, Y.; Koura, A.; Maeda, R.; Tamai, A.; Ishikawa, E.; Yokoi, I.; Moriue, J.; Moriue, T.; et al. Effects of Topical N-Acetylcysteine on Skin Hydration/Transepidermal Water Loss in Healthy Volunteers and Atopic Dermatitis Patients. Ann. Dermatol. 2015, 27, 450–451. [Google Scholar] [CrossRef]
- Nakai, K.; Yoneda, K.; Hosokawa, Y.; Moriue, T.; Presland, R.B.; Fallon, P.G.; Kabashima, K.; Kosaka, H.; Kubota, Y. Reduced expression of epidermal growth factor receptor, E-cadherin, and occludin in the skin of flaky tail mice is due to filaggrin and loricrin deficiencies. Am. J. Pathol. 2012, 181, 969–977. [Google Scholar] [CrossRef]
- Drewa, G.; Krzyzynska-Malinowska, E.; Wozniak, A.; Protas-Drozd, F.; Mila-Kierzenkowska, C.; Rozwodowska, M.; Kowaliszyn, B.; Czajkowski, R. Activity of superoxide dismutase and catalase and the level of lipid peroxidation products reactive with TBA in patients with psoriasis. Med. Sci. Monit. 2002, 8, BR338–BR343. [Google Scholar] [PubMed]
- Ormerod, A.D.; Copeland, P.; Shah, S.A. Treatment of psoriasis with topical NG-monomethyl-L-arginine, an inhibitor of nitric oxide synthesis. Br. J. Dermatol. 2000, 142, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; He, Y.Y.; Nishiyama, F.; Naruse, F.; Haba, R.; Kushida, Y.; Katsuki, N.; Moriue, T.; Yoneda, K.; Kubota, Y. IL-17A induces heterogeneous macrophages, and it does not alter the effects of lipopolysaccharides on macrophage activation in the skin of mice. Sci. Rep. 2017, 7, 12473. [Google Scholar] [CrossRef]
- Nakai, K.; Haba, R.; Kushida, Y.; Kubota, Y. Macrophages express βKlotho in skin lesions of psoriatic patients and the skin of imiquimod-treated mice. J. Dermatol. 2018, 45, 1475–1477. [Google Scholar] [CrossRef] [PubMed]
- Akamatsu, H.; Horio, T.; Hattori, K. Increased hydrogen peroxide generation by neutrophils from patients with acne inflammation. Int. J. Dermatol. 2003, 42, 366–369. [Google Scholar] [CrossRef]
- Kurutas, E.B.; Arican, O.; Sasmaz, S. Superoxide dismutase and myeloperoxidase activities in polymorphonuclear leukocytes in acne vulgaris. Acta Dermatovenerol. Alp. Pannonica Adriat. 2005, 14, 39–42. [Google Scholar] [PubMed]
- Akamatsu, H.; Horio, T. The possible role of reactive oxygen species generated by neutrophils in mediating acne inflammation. Dermatology 1998, 196, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, M.; Alestas, T.; Flori, E.; Mastrofrancesco, A.; Zouboulis, C.C.; Picardo, M. Peroxidated squalene induces the production of inflammatory mediators in HaCaT keratinocytes: A possible role in acne vulgaris. J. Investig. Dermatol. 2006, 126, 2430–2437. [Google Scholar] [CrossRef]
- Miyachi, Y.; Yoshioka, A.; Imamura, S.; Niwa, Y. Effect of antibiotics on the generation of reactive oxygen species. J. Investig. Dermatol. 1986, 86, 449–453. [Google Scholar] [CrossRef]
- Borelli, C.; Merk, K.; Schaller, M.; Jacob, K.; Vogeser, M.; Weindl, G.; Berger, U.; Plewig, G. In vivo porphyrin production by P. acnes in untreated acne patients and its modulation by acne treatment. Acta Derm. Venereol. 2006, 86, 316–319. [Google Scholar] [CrossRef]
- Hongcharu, W.; Taylor, C.R.; Chang, Y.; Aghassi, D.; Suthamjariya, K.; Anderson, R.R. Topical ALA-photodynamic therapy for the treatment of acne vulgaris. J. Investig. Dermatol. 2000, 115, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Matsumoto, M.; Iizuka, H.; Suzuki, K.; Taniguchi, N. Superoxide dismutase in psoriasis, squamous cell carcinoma and basal cell epithelioma: An immunohistochemical study. Br. J. Dermatol. 1991, 124, 555–559. [Google Scholar] [CrossRef]
- Ekmekcioglu, S.; Ellerhorst, J.A.; Prieto, V.G.; Johnson, M.M.; Broemeling, L.D.; Grimm, E.A. Tumor iNOS predicts poor survival for stage III melanoma patients. Int. J. Cancer 2006, 119, 861–866. [Google Scholar] [CrossRef]
- Suk Kang, J.; Son, S.S.; Lee, J.H.; Lee, S.W.; Jeong, A.R.; Lee, E.S.; Cha, S.K.; Chung, C.H.; Lee, E.Y. Protective effects of klotho on palmitate-induced podocyte injury in diabetic nephropathy. PLoS ONE 2021, 16, e0250666. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Yoneda, K.; Haba, R.; Kushida, Y.; Katsuki, N.; Moriue, T.; Kosaka, H.; Kubota, Y.; Inoue, S. Deranged epidermal differentiation in kl/kl mouse and the effects of βKlotho siRNA on the differentiation of HaCaT cells. Exp. Dermatol. 2013, 22, 772–774. [Google Scholar] [CrossRef]
- Nakai, K.; Yoneda, K.; Haba, R.; Kushida, Y.; Katsuki, N.; Moriue, J.; Moriue, T.; Koura, A.; Yokoi, I.; Ishikawa, E.; et al. βKlotho expression is reduced in human non-melanoma skin cancer. Int. J. Dermatol. 2015, 54, e431–e433. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.H.; Rhie, G.E.; Kim, Y.K.; Park, C.H.; Cho, K.H.; Kim, K.H.; Eun, H.C.; Chung, J.H. H2O2 accumulation by catalase reduction changes MAP kinase signaling in aged human skin in vivo. J. Investig. Dermatol. 2005, 125, 221–229. [Google Scholar] [CrossRef]
- Bang, E.; Kim, D.H.; Chung, H.Y. Protease-activated receptor 2 induces ROS-mediated inflammation through Akt-mediated NF-kappaB and FoxO6 modulation during skin photoaging. Redox Biol. 2021, 44, 102022. [Google Scholar] [CrossRef]
- Mastore, M.; Kohler, L.; Nappi, A.J. Production and utilization of hydrogen peroxide associated with melanogenesis and tyrosinase-mediated oxidations of DOPA and dopamine. FEBS J. 2005, 272, 2407–2415. [Google Scholar] [CrossRef] [PubMed]
- Ines, D.; Sonia, B.; Riadh, B.M.; Amel el, G.; Slaheddine, M.; Hamida, T.; Hamadi, A.; Basma, H. A comparative study of oxidant-antioxidant status in stable and active vitiligo patients. Arch. Dermatol. Res. 2006, 298, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, M.; Baysal, V.; Inaloz, H.S.; Can, M. The role of oxidants and antioxidants in generalized vitiligo at tissue level. J. Eur. Acad. Dermatol. Venereol. 2004, 18, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Akar, A.; Arca, E.; Erbil, H.; Akay, C.; Sayal, A.; Gur, A.R. Antioxidant enzymes and lipid peroxidation in the scalp of patients with alopecia areata. J. Dermatol. Sci. 2002, 29, 85–90. [Google Scholar] [CrossRef]
- Acharya, P.; Mathur, M.C. Oxidative stress in alopecia areata: A systematic review and meta-analysis. Int. J. Dermatol. 2020, 59, 434–440. [Google Scholar] [CrossRef]
- Koca, R.; Armutcu, F.; Altinyazar, H.C.; Gurel, A. Evaluation of lipid peroxidation, oxidant/antioxidant status, and serum nitric oxide levels in alopecia areata. Med. Sci. Monit. 2005, 11, Cr296–Cr299. [Google Scholar]
- Alzolibani, A.A. Preferential recognition of hydroxyl radical-modified superoxide dismutase by circulating autoantibodies in patients with alopecia areata. Ann. Dermatol. 2014, 26, 576–583. [Google Scholar] [CrossRef]
- Kampfer, H.; Pfeilschifter, J.; Frank, S. Expression and activity of arginase isoenzymes during normal and diabetes-impaired skin repair. J. Investig. Dermatol. 2003, 121, 1544–1551. [Google Scholar] [CrossRef]
- Ackerman, Z.; Seidenbaum, M.; Loewenthal, E.; Rubinow, A. Overload of Iron in the Skin of Patients with Varicose Ulcers—Possible Contributing Role of Iron Accumulation in Progression of the Disease. Arch. Dermatol. 1988, 124, 1376–1378. [Google Scholar] [CrossRef]
- Ou, Q.; Zhang, S.; Fu, C.; Yu, L.; Xin, P.; Gu, Z.; Cao, Z.; Wu, J.; Wang, Y. More natural more better: Triple natural anti-oxidant puerarin/ferulic acid/polydopamine incorporated hydrogel for wound healing. J. Nanobiotechnol. 2021, 19, 237. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Chen, Y.; Huang, H.; Fan, S.; Yang, C.; Wang, L.; Li, W.; Niu, W.; Liao, J. ROS-Eliminating Carboxymethyl Chitosan Hydrogel to Enhance Burn Wound-Healing Efficacy. Front. Pharmacol. 2021, 12, 679580. [Google Scholar] [CrossRef] [PubMed]
- Brechtel, B.; Haas, N.; Henz, B.M.; Kolde, G. Allopurinol: A therapeutic alternative for disseminated cutaneous sarcoidosis. Br. J. Dermatol. 1996, 135, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Reisberger, E.M.; Landthaler, M.; Wiest, L.; Schroder, J.; Stolz, W. Foreign body granulomas caused by polymethylmethacrylate microspheres: Successful treatment with allopurinol. Arch. Dermatol. 2003, 139, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Morita, A.; Minami, H.; Sakakibara, N.; Sato, K.; Tsuji, T. Elevated plasma superoxide dismutase activity in patients with systemic sclerosis. J. Dermatol. Sci. 1996, 11, 196–201. [Google Scholar] [CrossRef]
- Georgescu, S.R.; Mitran, C.I.; Mitran, M.I.; Nicolae, I.; Matei, C.; Ene, C.D.; Popa, G.L.; Tampa, M. Oxidative Stress in Cutaneous Lichen Planus-A Narrative Review. J. Clin. Med. 2021, 10, 2692. [Google Scholar] [CrossRef]
- Sredoja Tisma, V.; Bulimbasic, S.; Galesic Ljubanovic, D.; Galesic, K.; Morovic-Vergles, J.; Mitrovic, J.; Uchida, K.; Tatzber, F.; Zarkovic, N.; Jaganjac, M. The Onset of Systemic Oxidative Stress Associated with the Accumulation of Lipid Peroxidation Product Acrolein in the Skin of Patients with Small-Vessel Vasculitis. Molecules 2021, 26, 2344. [Google Scholar] [CrossRef]
- Bergqvist, C.; Safi, R.; El Hasbani, G.; Abbas, O.; Kibbi, A.; Nassar, D. Neutrophil Extracellular Traps are Present in Immune-complex-mediated Cutaneous Small Vessel Vasculitis and Correlate with the Production of Reactive Oxygen Species and the Severity of Vessel Damage. Acta Derm. Venereol. 2020, 100, adv00281. [Google Scholar] [CrossRef]
- Emre, S.; Metin, A.; Demirseren, D.D.; Akoglu, G.; Oztekin, A.; Neselioglu, S.; Erel, O. The association of oxidative stress and disease activity in seborrheic dermatitis. Arch. Dermatol. Res. 2012, 304, 683–687. [Google Scholar] [CrossRef]
- Naziroglu, M.; Kokcam, I.; Simsek, H.; Karakilcik, A.Z. Lipid peroxidation and antioxidants in plasma and red blood cells from patients with pemphigus vulgaris. J. Basic Clin. Physiol. Pharmacol. 2003, 14, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Tisma, V.S.; Basta-Juzbasic, A.; Jaganjac, M.; Brcic, L.; Dobric, I.; Lipozencic, J.; Tatzber, F.; Zarkovic, N.; Poljak-Blazi, M. Oxidative stress and ferritin expression in the skin of patients with rosacea. J. Am. Acad. Dermatol. 2009, 60, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Hoque, S.R.; Ameen, M.; Holden, C.A. Acquired reactive perforating collagenosis: Four patients with a giant variant treated with allopurinol. Br. J. Dermatol. 2006, 154, 759–762. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakai, K.; Tsuruta, D. What Are Reactive Oxygen Species, Free Radicals, and Oxidative Stress in Skin Diseases? Int. J. Mol. Sci. 2021, 22, 10799. https://doi.org/10.3390/ijms221910799
Nakai K, Tsuruta D. What Are Reactive Oxygen Species, Free Radicals, and Oxidative Stress in Skin Diseases? International Journal of Molecular Sciences. 2021; 22(19):10799. https://doi.org/10.3390/ijms221910799
Chicago/Turabian StyleNakai, Kozo, and Daisuke Tsuruta. 2021. "What Are Reactive Oxygen Species, Free Radicals, and Oxidative Stress in Skin Diseases?" International Journal of Molecular Sciences 22, no. 19: 10799. https://doi.org/10.3390/ijms221910799
APA StyleNakai, K., & Tsuruta, D. (2021). What Are Reactive Oxygen Species, Free Radicals, and Oxidative Stress in Skin Diseases? International Journal of Molecular Sciences, 22(19), 10799. https://doi.org/10.3390/ijms221910799





