Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine—A Review
Abstract
1. Introduction
2. Medicinal Plants and Their Constituents with Antibacterial Activity Intended in the Treatment of Skin Disorders According to European Legislation
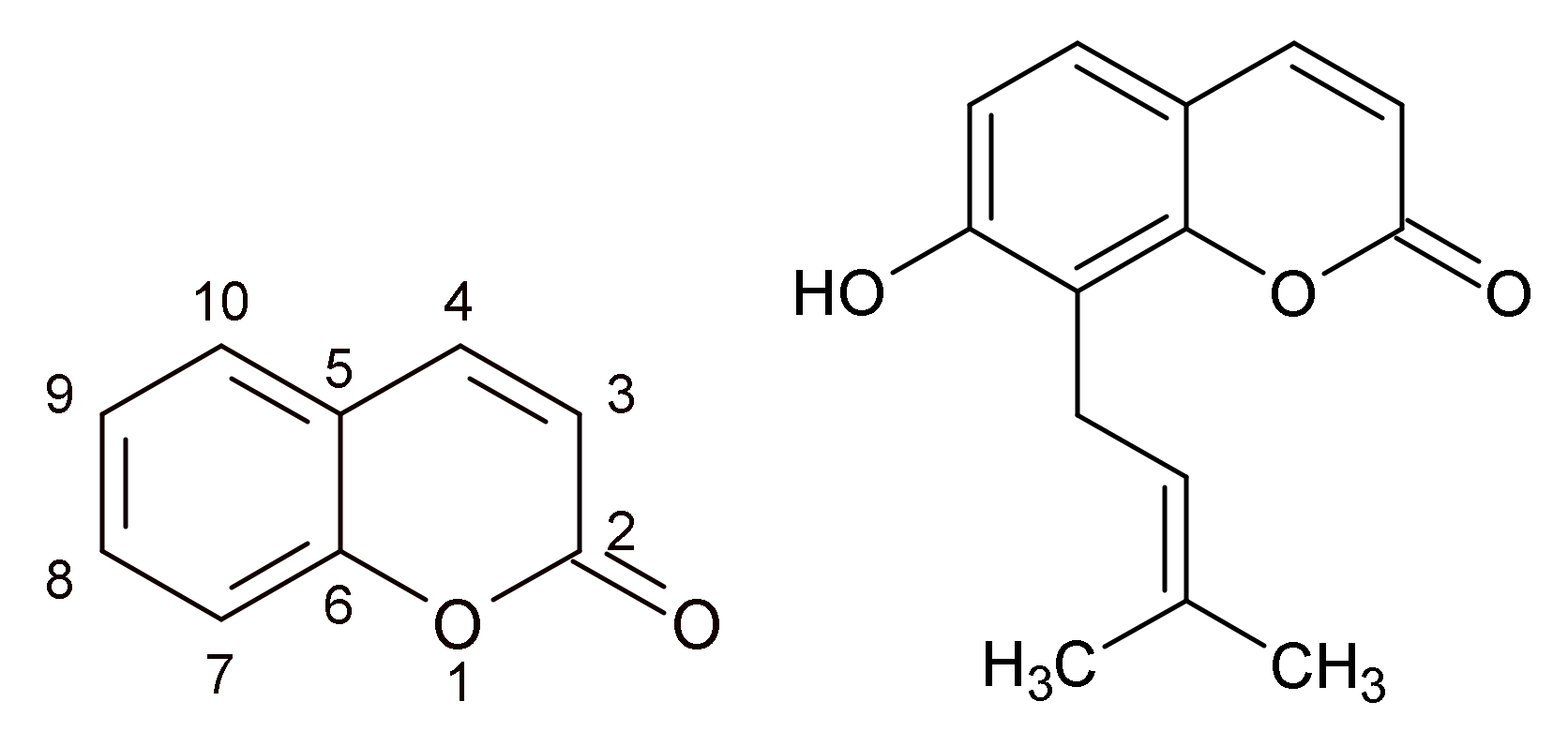
2.1. Coumarins
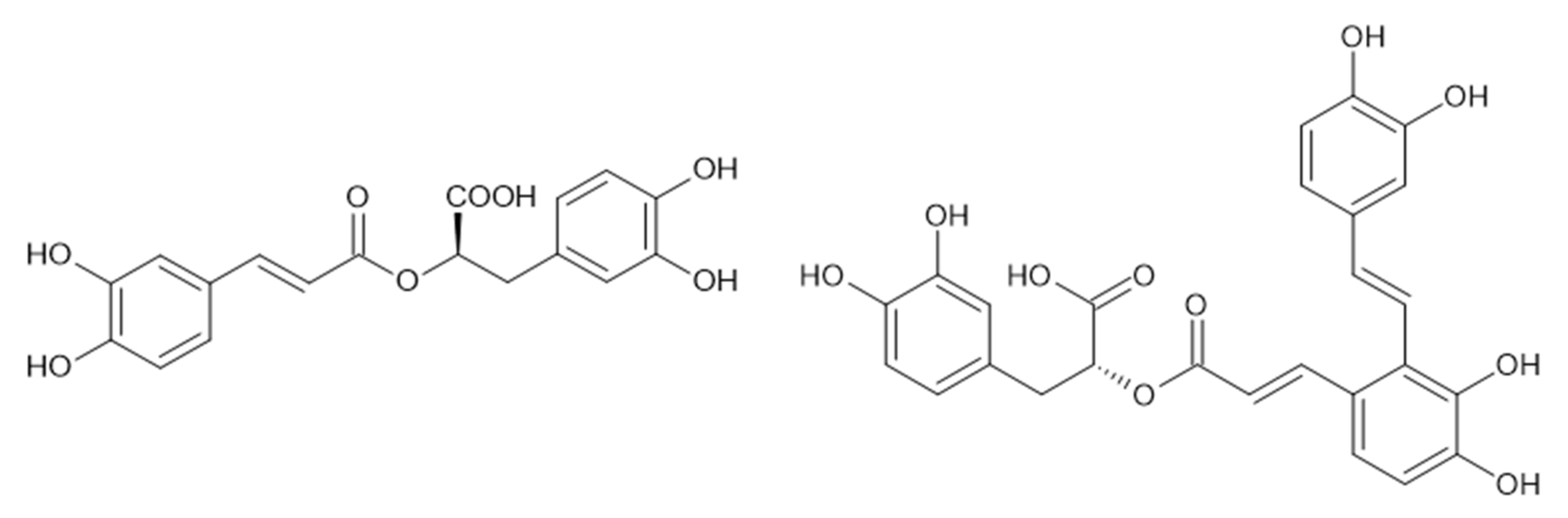
2.2. Cinnamic Acid Derivatives
2.3. Flavonoids
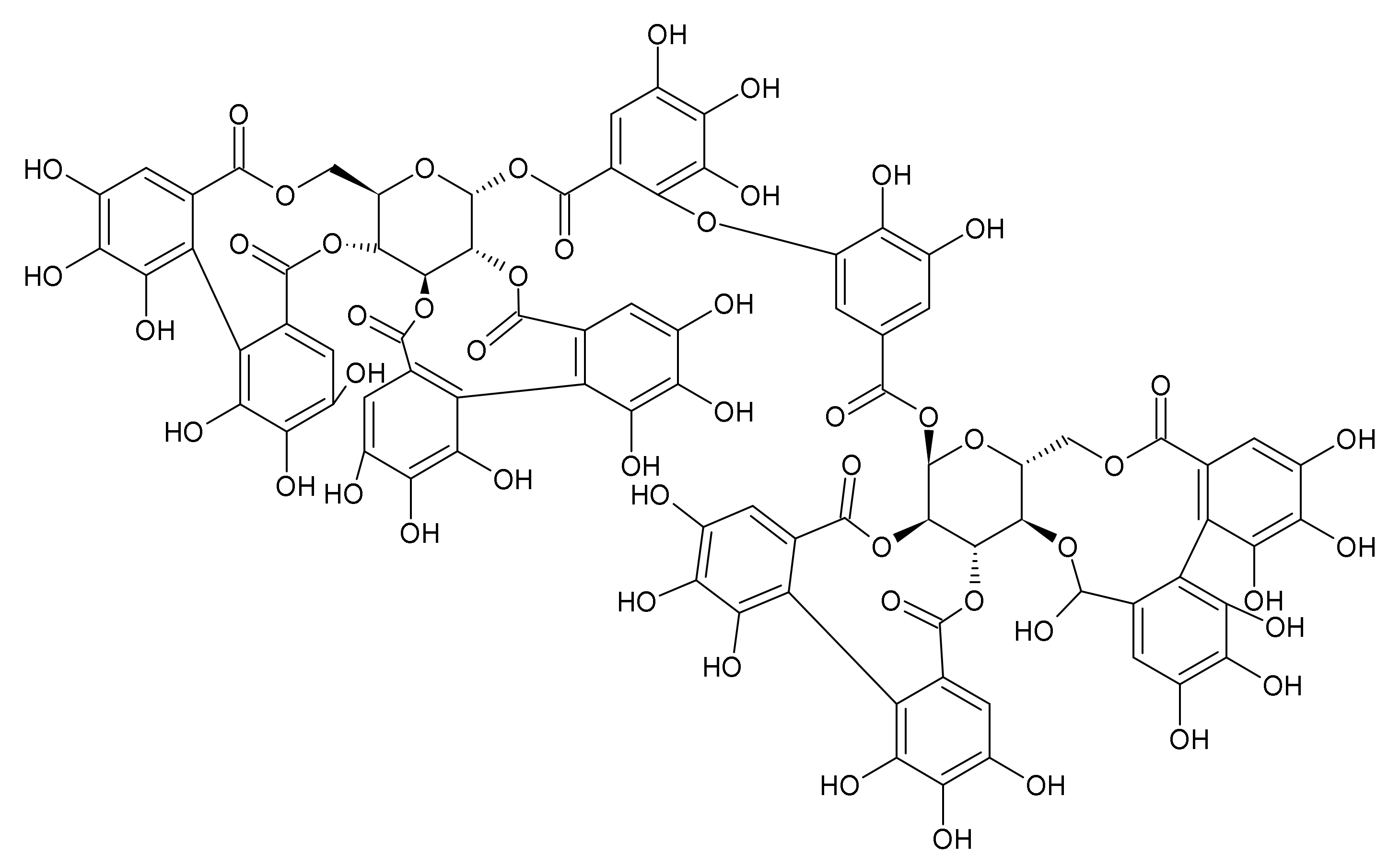
2.4. Tannins
2.5. Essential Oils
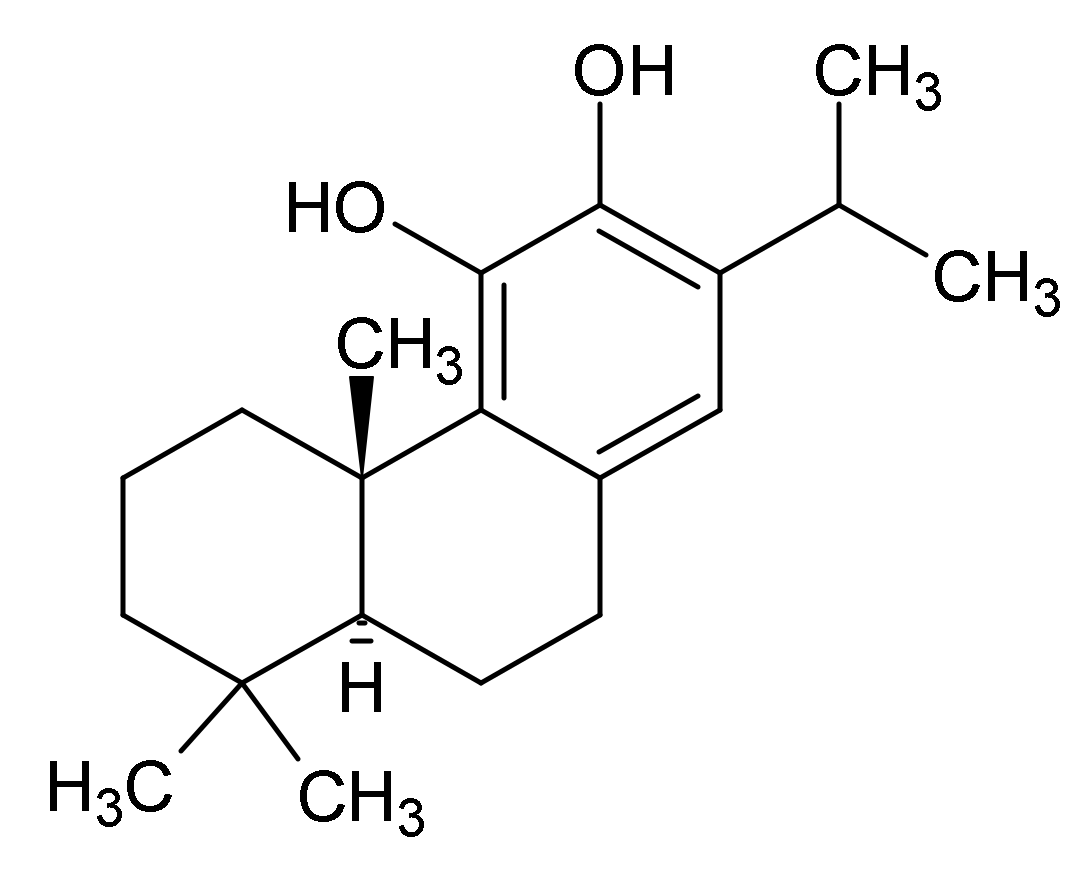
2.6. Diterpenoids
2.7. Saponins
2.8. Carotenoids
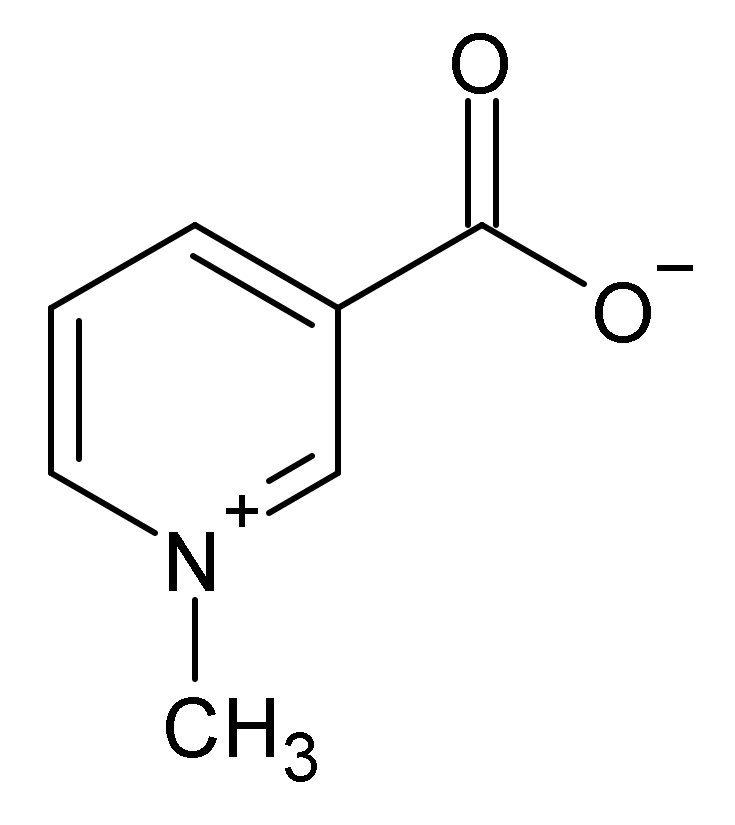
2.9. Alkaloids
2.10. Other Classes of Plant Constituents
3. Synergy of Plant Metabolites in Mixtures
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AIDS | acquired immunodeficiency syndrome |
| AITC | allylisothiocyanate |
| ATP | adenosine triphosphate |
| BITC | benzylisothiocyanate |
| EMA | European Medicines Agency |
| EO | essential oil |
| EOs | essential oils |
| EU | European Union |
| HMPC | Committee on Herbal Medicinal Products |
| IC50 | half maximal inhibitory concentration |
| IKKβ | inhibitor of nuclear factor kappa-B kinase subunit bet |
| IL-8 | interleukin 8 |
| ITCM | isothiocyanates mixture |
| ITCs | isothiocyanates |
| MBC | minimum bactericidal concentration |
| MIC | minimum inhibitory concentration |
| MRSA | methicillin-resistant Staphylococcus aureus |
| MSSA | methicillin-susceptible Staphylococcus aureus |
| NF-κB | the nuclear factor kappa-B |
| PEITC | phenylethyl isothiocyanate |
| TNF-α | tumor necrosis factor α |
| TRP | transient receptor potential |
| TRPA1 | transient receptor potential cation channel, subfamily A, member 1 |
| TRPM | transient receptor potential cation channel subfamily M, member 8 |
| TRPV3 | transient receptor potential cation channel, subfamily V, member 3 |
| TTO | tea tree oil |
| TU | traditional use |
| WHO | World Health Organization |
References
- Rahman, M.; Sarker, S.D. Antimicrobial natural products. In Annual Reports in Medicinal Chemistry: Medicinal Natural Products: A Disease-Focused Approach, 1st ed.; Sarker, S., Nahar, L., Eds.; Academic Press/Elsevier: Cambridge, MA, USA, 2020; Volume 55, pp. 77–113. [Google Scholar] [CrossRef]
- De Oliveira, D.M.P.; Forde, B.M.; Kidd, T.J.; Harris, P.N.A.; Schembri, M.A.; Beatson, S.A.; Paterson, D.L.; Walker, M.J. Antimicrobial Resistance in ESKAPE Pathogens. Clin. Microbiol. Rev. 2020, 33, e00181-19. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Buchvald, D. Anatomy and histology of the skin. In Dermatovenerology, 1st ed.; Šimaljaková, M., Buchvald, D., Eds.; Publishing House of Comenius University: Bratislava, Slovakia, 2019; pp. 21–24. [Google Scholar]
- Ki, V.; Rotstein, C. Bacterial skin and soft tissue infections in adults: A review of their epidemiology, pathogenesis, diagnosis, treatment and site of care. Can. J. Infect. Dis. Med. Microbiol. 2008, 19, 173–184. [Google Scholar] [CrossRef]
- Grice, E.A.; Segre, J.A. The skin microbiome. Nat. Rev. Microbiol. 2011, 9, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.C.; Chan, W.W.; Metelitsa, A.I.; Fiorillo, L.; Lin, A.N. Pseudomonas skin infection: Clinical features, epidemiology, and management. Am. J. Clin. Dermatol. 2011, 12, 157–169. [Google Scholar] [CrossRef]
- Zheng, Y.; Xu, N.; Pang, J.; Han, H.; Yang, H.; Qin, W.; Zhang, H.; Li, W.; Wang, H.; Chen, Y. Colonization with Extensively Drug-Resistant Acinetobacter baumannii and Prognosis in Critically Ill Patients: An Observational Cohort Study. Front. Med. 2021, 8, 667776. [Google Scholar] [CrossRef]
- Vila, T.; Sultan, A.S.; Montelongo-Jauregui, D.; Jabra-Rizk, M.A. Candida auris: A fungus with identity crisis. Pathog. Dis. 2020, 78, ftaa034. [Google Scholar] [CrossRef]
- Choi, J.H.; Seo, H.S.; Lim, S.Y.; Park, K. Cutaneous Immune Defenses Against Staphylococcus aureus Infections. J. Lifestyle Med. 2014, 4, 39–46. [Google Scholar] [CrossRef]
- Stevens, D.L.; Bisno, A.L.; Chambers, H.F.; Dellinger, E.P.; Goldstein, E.J.; Gorbach, S.L.; Hirschmann, J.V.; Kaplan, S.L.; Montoya, J.G.; Wade, J.C. Infectious Diseases Society of America. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2014, 59, 147–159. [Google Scholar] [CrossRef]
- Esposito, S.; Bassetti, M.; Concia, E.; De Simone, G.; De Rosa, F.G.; Grossi, P.; Novelli, A.; Menichetti, F.; Petrosillo, N.; Tinelli, M.; et al. Diagnosis and management of skin and soft-tissue infections (SSTI). A literature review and consensus statement: An update. J. Chemother. 2017, 29, 197–214. [Google Scholar] [CrossRef]
- Esposito, S.; Noviello, S.; Leone, S. Epidemiology and microbiology of skin and soft tissue infections. Curr. Opin. Infect. Dis. 2016, 29, 109–115. [Google Scholar] [CrossRef]
- Šimaljaková, M. Sebaceous glands diseases and related dermatoses. In Dermatovenerology, 1st ed.; Šimaljaková, M., Buchvald, D., Eds.; Publishing House of Comenius University: Bratislava, Slovakia, 2019; pp. 345–352. [Google Scholar]
- Khorvash, F.; Abdi, F.; Kashani, H.H.; Naeini, F.F.; Narimani, T. Staphylococcus aureus in Acne Pathogenesis: A Case-Control Study. N. Am. J. Med. Sci. 2012, 4, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Böni, R.; Nehrhoff, B. Treatment of gram-negative folliculitis in patients with acne. Am. J. Clin. Dermatol. 2003, 4, 273–276. [Google Scholar] [CrossRef]
- Vayalumkal, J.V.; Jadavji, T. Children hospitalized with skin and soft tissue infections: A guide to antibacterial selection and treatment. Paediatr. Drugs 2006, 8, 99–111. [Google Scholar] [CrossRef]
- O’Neill, A.M.; Gallo, R.L. Host-microbiome interactions and recent progress into understanding the biology of acne vulgaris. Microbiome 2018, 6, 177. [Google Scholar] [CrossRef]
- Karvonen, S.L.; Räsänen, L.; Cunliffe, W.J.; Holland, K.T.; Karvonen, J.; Reunala, T. Delayed hypersensitivity to Propionibacterium acnes in patients with severe nodular acne and acne fulminans. Dermatology 1994, 189, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Hassanzadeh, P.; Bahmani, M.; Mehrabani, D. Bacterial resistance to antibiotics in acne vulgaris: An in vitro study. Indian J. Dermatol. 2008, 53, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Buchvald, D. Viral infections of skin and mucosae. In Dermatovenerology, 1st ed.; Šimaljaková, M., Buchvald, D., Eds.; Publishing House of Comenius University: Bratislava, Slovakia, 2019; pp. 95–116. [Google Scholar]
- Šimaljaková, M. Mycotical infections of skin and mucosae. In Dermatovenerology, 1st ed.; Šimaljaková, M., Buchvald, D., Eds.; Publishing House of Comenius University: Bratislava, Slovakia, 2019; pp. 139–160. [Google Scholar]
- Buchvald, D. Dermatoses caused by protozoa and worms. In Dermatovenerology, 1st ed.; Šimaljaková, M., Buchvald, D., Eds.; Publishing House of Comenius University: Bratislava, Slovakia, 2019; pp. 161–167. [Google Scholar]
- Akers, K.S.; Mende, K.; Cheatle, K.A.; Zera, W.C.; Yu, X.; Beckius, M.L.; Aggarwal, D.; Li, P.; Sanchez, C.J.; Wenke, J.C.; et al. Biofilms and persistent wound infections in United States military trauma patients: A case-control analysis. BMC Infect. Dis. 2014, 14, 190. [Google Scholar] [CrossRef]
- Curcio, D. Resistant pathogen-associated skin and skin-structure infections: Antibiotic options. Expert Rev. Anti Infect. Ther. 2010, 8, 1019–1036. [Google Scholar] [CrossRef]
- Foster, T.J. Antibiotic resistance in Staphylococcus aureus. Current status and future prospects. FEMS Microbiol. Rev. 2017, 41, 430–449. [Google Scholar] [CrossRef]
- Kampf, G. Acquired resistance to chlorhexidine—Is it time to establish an ‘antiseptic stewardship’ initiative? J. Hosp. Infect. 2016, 94, 213–227. [Google Scholar] [CrossRef]
- Dadashi, M.; Hajikhani, B.; Darban-Sarokhalil, D.; van Belkum, A.; Goudarzi, M. Mupirocin resistance in Staphylococcus aureus: A systematic review and meta-analysis. J. Glob. Antimicrob. Resist. 2020, 20, 238–247. [Google Scholar] [CrossRef]
- Guan, H.; Dong, W.; Lu, Y.; Jiang, M.; Zhang, D.; Aobuliaximu, Y.; Dong, J.; Niu, Y.; Liu, Y.; Guan, B.; et al. Distribution and Antibiotic Resistance Patterns of Pathogenic Bacteria in Patients with Chronic Cutaneous Wounds in China. Front. Med. 2021, 8, 609584. [Google Scholar] [CrossRef]
- Bassetti, M.; Manno, G.; Collidà, A.; Ferrando, A.; Gatti, G.; Ugolotti, E.; Cruciani, M.; Bassetti, D. Erythromycin resistance in Streptococcus pyogenes in Italy. Emerg. Infect. Dis. 2000, 6, 180–183. [Google Scholar] [CrossRef]
- Lu, B.; Fang, Y.; Fan, Y.; Chen, X.; Wang, J.; Zeng, J.; Li, Y.; Zhang, Z.; Huang, L.; Li, H.; et al. High Prevalence of Macrolide-resistance and Molecular Characterization of Streptococcus pyogenes Isolates Circulating in China from 2009 to 2016. Front. Microbiol. 2017, 8, 1052. [Google Scholar] [CrossRef]
- Tsuzuki, S. Macrolide resistance in Streptococcus spp. Lancet Infect. Dis. 2019, 19, 243–244. [Google Scholar] [CrossRef]
- Amparo, T.R.; Seibert, J.B.; Vieira, P.M.A.; Teixeira, L.F.M.; Santos, O.D.H.D.; de Souza, G.H.B. Herbal medicines to the treatment of skin and soft tissue infections: Advantages of the multi-targets action. Phytother. Res. 2020, 34, 94–103. [Google Scholar] [CrossRef]
- Khameneh, B.; Iranshahy, M.; Soheili, V.; Fazly Bazzaz, B.S. Review on plant antimicrobials: A mechanistic viewpoint. Antimicrob. Resist. Infect. Control 2019, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Clinton, A.; Carter, T. Chronic Wound Biofilms: Pathogenesis and Potential Therapies. Lab. Med. 2015, 46, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Slobodníková, L.; Fialová, S.; Rendeková, K.; Kováč, J.; Mučaji, P. Antibiofilm Activity of Plant Polyphenols. Molecules 2016, 21, 1717. [Google Scholar] [CrossRef]
- Lewis, K. New approaches to antimicrobial discovery. Biochem. Pharmacol. 2017, 134, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Dubey, A.K. Diversity and Applications of Endophytic Actinobacteria of Plants in Special and Other Ecological Niches. Front. Microbiol. 2018, 9, 1767. [Google Scholar] [CrossRef]
- European Medicines Agency. Committee on Herbal Medicinal Products (HMPC). Available online: https://www.ema.europa.eu/en/committees/committee-herbal-medicinal-products-hmpc (accessed on 15 August 2021).
- European Medicines Agency. Herbal Medicinal Products. Available online: https://www.ema.europa.eu/en/medicines/field_ema_web_categories%253Aname_field/Herbal/search_api_aggregation_ema_therapeutic_area_name/Skin%20disorders%20and%20minor%20wounds (accessed on 26 June 2021).
- EUR-Lex. Access to European Union Law. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2004:136:0085:0090:en:PDF (accessed on 15 August 2021).
- European Medicines Agency. European Union monographs and list entries. Available online: https://www.ema.europa.eu/en/human-regulatory/herbal-products/european-union-monographs-list-entries (accessed on 15 August 2021).
- Nagy, M.; Mučaji, P.; Grančai, D. Pharmacognosy: Biologically Active Plant Metabolites and Their Sources, 2nd ed.; Osveta: Martin, Slovakia, 2017; pp. 69–178. [Google Scholar]
- Yuan, G.; Guan, Y.; Yi, H.; Lai, S.; Sun, Y.; Cao, S. Antibacterial activity and mechanism of plant flavonoids to gram-positive bacteria predicted from their lipophilicities. Sci. Rep. 2021, 11, 10471. [Google Scholar] [CrossRef] [PubMed]
- Sychrová, A.; Koláriková, I.; Žemlička, M.; Šmejkal, K. Natural compounds with dual antimicrobial and anti-inflammatory effects. Phytochem. Rev. 2020, 19, 1471–1502. [Google Scholar] [CrossRef]
- Slobodníková, L. Antibacterial therapeutics. In Medical Microbiology, 1st ed.; Liptáková, A., Predný, J., Buc, M., Slobodníková, L., Jalili, N., Krčméry, V., Koreň, J., Eds.; Herba: Bratislava, Slovakia, 2019; pp. 263–277. [Google Scholar]
- Slobodníková, L. Staphylococcus and the other aerobic and facultatively anaerobic Gram-positive cocci. In Medical Microbiology, 1st ed.; Liptáková, A., Predný, J., Buc, M., Slobodníková, L., Jalili, N., Krčméry, V., Koreň, J., Eds.; Herba: Bratislava, Slovakia, 2019; pp. 294–306. [Google Scholar]
- Stefanachi, A.; Leonetti, F.; Pisani, L.; Catto, M.; Carotti, A. Coumarin: A Natural, Privileged and Versatile Scaffold for Bioactive Compounds. Molecules 2018, 23, 250. [Google Scholar] [CrossRef]
- De Souza, S.M.; Delle Monache, F.; Smânia, A., Jr. Antibacterial activity of coumarins. Z. Naturforsch. C J. Biosci. 2005, 60, 693–700. [Google Scholar] [CrossRef]
- Feng, D.; Zhang, A.; Yang, Y.; Yang, P. Coumarin-containing hybrids and their antibacterial activities. Arch. Pharm. 2020, 353, e1900380. [Google Scholar] [CrossRef]
- Hu, C.F.; Zhang, P.L.; Sui, Y.F.; Lv, J.S.; Ansari, M.F.; Battini, N.; Li, S.; Zhou, C.H.; Geng, R.X. Ethylenic conjugated coumarin thiazolidinediones as new efficient antimicrobial modulators against clinical methicillin-resistant Staphylococcus aureus. Bioorg. Chem. 2020, 94, 103434. [Google Scholar] [CrossRef]
- Stefanović, O.D.; Tešić, J.D.; Čomić, R.L. Melilotus albus and Dorycnium herbaceum extracts as source of phenolic compounds and their antimicrobial, antibiofilm, and antioxidant potentials. J. Food Drug Anal. 2015, 23, 417–424. [Google Scholar] [CrossRef]
- Taofiq, O.; González-Paramás, A.M.; Barreiro, M.F.; Ferreira, I.C. Hydroxycinnamic Acids and Their Derivatives: Cosmeceutical Significance, Challenges and Future Perspectives, a Review. Molecules 2017, 22, 281. [Google Scholar] [CrossRef]
- Yingyongnarongkul, B.E.; Apiratikul, N.; Aroonrerk, N.; Suksamrarn, A. Synthesis of bis, tris and tetra(dihydrocaffeoyl)polyamine conjugates as antibacterial agents against VRSA. Arch. Pharm. Res. 2008, 31, 698–704. [Google Scholar] [CrossRef]
- El Hosary, R.; El-Mancy, M.S.S.; El Deeb, S.K.; Eid, H.H.; Tantawy, E.M.E.L.; Shams, M.M.; Samir, R.; Assar, H.N.; Sleem, A.A. Efficient wound healing composite hydrogel using Egyptian Avena sativa L. polysaccharide containing β-glucan. Int. J. Bio Macromol. 2020, 149, 1331–1338. [Google Scholar] [CrossRef]
- Veerasubramanian, P.K.; Thangavel, P.; Kannan, R.; Chakraborty, S.; Ramachandran, B.; Suguna, L.; Muthuvijayan, V. An investigation of konjac glucomannan-keratin hydrogel scaffold loaded with Avena sativa extracts for diabetic wound healing. Colloids Surf. B Biointerfaces 2018, 165, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Roumani, M.; Duval, R.E.; Ropars, A.; Risler, A.; Robin, C.; Larbat, R. Phenolamides: Plant specialized metabolites with a wide range of promising pharmacological and health-promoting interests. Biomed. Pharmacother. 2020, 131, 110762. [Google Scholar] [CrossRef]
- Yang, J.; Ou, B.; Wise, M.L.; Chu, Y. In vitro total antioxidant capacity and anti-inflammatory activity of three common oat-derived avenanthramides. Food Chem. 2014, 160, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Shin, W.S.; Yeo, D.; Lim, W.; Zhang, T.; Ji, L.L. Anti-inflammatory effect of avenanthramides via NF-κB pathways in C2C12 skeletal muscle cells. Free Radic. Biol. Med. 2018, 17, 30–36. [Google Scholar] [CrossRef]
- Gericke, S.; Lübken, T.; Wolf, D.; Kaiser, M.; Hannig, C.; Speer, K. Identification of New Compounds from Sage Flowers (Salvia officinalis L.) as Markers for Quality Control and the Influence of the Manufacturing Technology on the Chemical Composition and Antibacterial Activity of Sage Flower Extracts. J. Agric. Food Chem. 2018, 66, 1843–1853. [Google Scholar] [CrossRef] [PubMed]
- Moreno, S.; Scheyer, T.; Romano, C.S.; Vojnov, A.A. Antioxidant and antimicrobial activities of rosemary extracts linked to their polyphenol composition. Free Radic. Res. 2006, 40, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Slobodníková, L.; Fialová, S.; Hupková, H.; Grancai, D. Rosmarinic acid interaction with planktonic and biofilm Staphylococcus aureus. Nat. Prod. Commun. 2013, 8, 1747–1750. [Google Scholar] [CrossRef] [PubMed]
- Ekambaram, S.P.; Perumal, S.S.; Balakrishnan, A.; Marappan, N.; Gajendran, S.S.; Viswanathan, V. Antibacterial synergy between rosmarinic acid and antibiotics against methicillin-resistant Staphylococcus aureus. J. Intercult. Ethnopharmacol. 2016, 5, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Bamunuarachchi, N.I.; Tabassum, N.; Kim, Y.M. Caffeic Acid and Its Derivatives: Antimicrobial Drugs toward Microbial Pathogens. J. Agric. Food Chem. 2021, 69, 2979–3004. [Google Scholar] [CrossRef] [PubMed]
- Mu, D.; Luan, Y.; Wang, L.; Gao, Z.; Yang, P.; Jing, S.; Wang, Y.; Xiang, H.; Wang, T.; Wang, D. The combination of salvianolic acid A with latamoxef completely protects mice against lethal pneumonia caused by methicillin-resistant Staphylococcus aureus. Emerg. Microbes Infect. 2020, 9, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Nagy, M.; Grančai, D.; Mučaji, P. Pharmacognosy Biogenesis of Natural Substances, 1st ed.; Osveta: Martin, Slovakia, 2011; pp. 42–200. [Google Scholar]
- Bhattacharya, S. Are we in the polyphenols era? Pharmacogn. Res. 2011, 3, 147. [Google Scholar] [CrossRef]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Xie, Y.; Yang, W.; Tang, F.; Chen, X.; Ren, L. Antibacterial activities of flavonoids: Structure-activity relationship and mechanism. Curr. Med. Chem. 2015, 22, 132–149. [Google Scholar] [CrossRef]
- Farhadi, F.; Khameneh, B.; Iranshahi, M.; Iranshahy, M. Antibacterial activity of flavonoids and their structure–activity relationship: An update review. Phytother. Res. 2019, 33, 13–40. [Google Scholar] [CrossRef]
- Osonga, F.J.; Akgul, A.; Miller, R.M.; Eshun, G.B.; Yazgan, I.; Akgul, A.; Sadik, O.A. Antimicrobial Activity of a New Class of Phosphorylated and Modified Flavonoids. ACS Omega 2019, 4, 12865–12871. [Google Scholar] [CrossRef]
- Aslam, M.S.; Ahmad, M.S.; Riaz, H.; Raza, S.A.; Hussain, S.; Qureshi, O.S.; Maria, P.; Hamzah, Z.; Javed, O. Role of flavonoids as wound healing agent. In Phytochemicals—Source of Antioxidants and Role in Disease Prevention, 1st ed.; eBook; Asao, T., Asaduzzaman, M., Eds.; IntechOpen: London, UK, 2018; Available online: https://www.intechopen.com/chapters/62634 (accessed on 30 June 2021). [CrossRef]
- Wu, S.; Pang, Y.; He, Y.; Zhang, X.; Peng, L.; Guo, J.; Zeng, J. A comprehensive review of natural products against atopic dermatitis: Flavonoids, alkaloids, terpenes, glycosides and other compounds. Biomed. Pharmacother. 2021, 140, 111741. [Google Scholar] [CrossRef]
- Vieira, V.; Pereira, C.; Pires, T.C.S.P.; Calhelha, R.C.; Alves, M.J.; Ferreira, O.; Barros, L.; Ferreira, I.C.F.R. Phenolic profile, antioxidant and antibacterial properties of Juglans regia L. (walnut) leaves from the Northeast of Portugal. Ind. Crops Prod. 2019, 134, 347–355. [Google Scholar] [CrossRef]
- Baba, I.A.; Qureshi, A.B.N. Wound healing potential of methanolic extract of Juglans regia on albino rats. Eur. J. Mol. Clin. Med. 2021, 8, 997–1010. [Google Scholar]
- Pallag, A.; Filip, G.A.; Olteanu, D.; Clichici, S.; Baldea, I.; Jurca, T.; Micle, O.; Vicaş, L.; Marian, E.; Soriţău, O.; et al. Equisetum arvense L. Extract Induces Antibacterial Activity and Modulates Oxidative Stress, Inflammation, and Apoptosis in Endothelial Vascular Cells Exposed to Hyperosmotic Stress. Oxid. Med. Cell Longev. 2018, 3060525. [Google Scholar] [CrossRef]
- Kukric, Z.; Topalić-Trivunović, L.; Pavicic, S.; Zabic, M.; Matos, S.; Davidovic, A. Total phenolic content, antioxidant and antimicrobial activity of Equisetum arvense L. Chem. Ind. Chem. Eng. Q. 2013, 9, 37–43. [Google Scholar] [CrossRef]
- Gendron, F.; Nilson, S.; Ziffle, V.; Johnny, S.; Louie, D.; Diamente, P. Antimicrobial Effectiveness on Selected Bacterial Species and Alkaloid and Saponin Content of Rosa nutkana C. Presl (Nootka Rose) and Urtica dioica L. (Stinging Nettle) Extracts. Am. J. Plant Sci. 2021, 12, 720–733. [Google Scholar] [CrossRef]
- Zenão, S.; Aires, A.; Dias, C.; Saavedra, M.J.; Fernandes, C. Antibacterial potential of Urtica dioica and Lavandula angustifolia extracts against methicillin resistant Staphylococcus aureus isolated from diabetic foot ulcers. J. Herb. Med. 2017, 10, 53–58. [Google Scholar] [CrossRef]
- Pires, T.C.S.P.; Dias, M.I.; Barros, L.; Calhelha, R.C.; Alves, M.J.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Edible flowers as sources of phenolic compounds with bioactive potential. Food Res. Int. 2018, 105, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, N.; Tansho-Nagakawa, S.; Miyazaki, C.; Shimomura, K.; Ono, Y.; Abe, S. Inhibition of Neutrophil Adhesion and Antimicrobial Activity by Diluted Hydrosol Prepared from Rosa damascena. Biol. Pharm. Bull. 2017, 40, 161–168. [Google Scholar] [CrossRef]
- Hellinger, R.; Koehbach, J.; Fedchuk, H.; Sauer, B.; Huber, R.; Gruber, C.W.; Gründemann, C. Immunosuppressive activity of an aqueous Viola tricolor herbal extract. J. Ethnopharmacol. 2014, 151, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Witkowska-Banaszczak, E.; Bylka, W.; Matławska, I.; Goślińska, O.; Muszyński, Z. Antimicrobial activity of Viola tricolor herb. Fitoterapia 2005, 76, 458–461. [Google Scholar] [CrossRef]
- Das, A.K.; Islam, N.; Faruk, O.; Ashaduzzaman, M.; Dungani, R. Review on tannins: Extraction processes, applications and possibilities. S. Afr. J. Bot. 2020, 135, 58–70. [Google Scholar] [CrossRef]
- Farha, A.K.; Yang, Q.Q.; Kim, G.; Li, H.B.; Zhu, F.; Liu, H.Y.; Gan, R.Y.; Corke, H. Tannins as an alternative to antibiotics. Food Biosci. 2020, 38, 100751. [Google Scholar] [CrossRef]
- Engels, C.; Schieber, A.; Gänzle, M.G. Inhibitory spectra and modes of antimicrobial action of gallotannins from mango kernels (Mangifera indica L.). Appl. Environ. Microbiol. 2011, 77, 2215–2223. [Google Scholar] [CrossRef]
- Payne, D.E.; Martin, N.R.; Parzych, K.R.; Rickard, A.H.; Underwood, A.; Boles, B.R. Tannic acid inhibits Staphylococcus aureus surface colonization in an IsaA-dependent manner. Infect. Immun. 2013, 81, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Andrensek, S.; Simonovska, B.; Vovk, I.; Fyhrquist, P.; Vuorela, H.; Vuorela, P. Antimicrobial and antioxidative enrichment of oak (Quercus robur) bark by rotation planar extraction using Extra Chrom. Int. J. Food Microbiol. 2004, 92, 181–187. [Google Scholar] [CrossRef]
- Tolmacheva, A.A.; Rogozhin, E.A.; Deryabin, D.G. Antibacterial and quorum sensing regulatory activities of some traditional Eastern-European medicinal plants. Acta Pharm. 2014, 64, 173–186. [Google Scholar] [CrossRef]
- Nohynek, L.J.; Alakomi, H.L.; Kähkönen, M.P.; Heinonen, M.; Helander, I.M.; Oksman-Caldentey, K.M.; Puupponen-Pimiä, R.H. Berry phenolics: Antimicrobial properties and mechanisms of action against severe human pathogens. Nutr. Cancer 2006, 54, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Puljula, E.; Walton, G.; Woodward, M.J.; Karonen, M. Antimicrobial Activities of Ellagitannins against Clostridiales perfringens, Escherichia coli, Lactobacillus plantarum and Staphylococcus aureus. Molecules 2020, 25, 3714. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.L.; He, Y.B.; Liang, Y.Z.; Wang, Y.L.; Huang, L.F.; Xie, J.W. Comparative analysis of the volatile components of Agrimonia eupatoria from leaves and roots by gas chromatography-mass spectrometry and multivariate curve resolution. J. Anal. Methods Chem. 2013, 2013, 1–9. [Google Scholar] [CrossRef][Green Version]
- Granica, S.; Kluge, H.; Horn, G.; Matkowski, A.; Kiss, A.K. The phytochemical investigation of Agrimonia eupatoria L. and Agrimonia procera Wallr as valid sources of Agrimoniae herba—The pharmacopoeial plant material. J. Pharm. Biomed. Anal. 2015, 114, 272–279. [Google Scholar] [CrossRef]
- Al-Snafi, A.E. The pharmacological and therapeutic importance of Agrimonia eupatoria—A review. Asian J. Pharm. Sci. Technol. 2015, 5, 112–117. [Google Scholar]
- Muruzović, M.Ž.; Mladenović, K.G.; Stefanović, O.D.; Vasić, S.M.; Čomić, L.R. Extracts of Agrimonia eupatoria L. as sources of biologically active compounds and evaluation of their antioxidant, antimicrobial, and antibiofilm activities. J. Food Drug Anal. 2016, 24, 539–547. [Google Scholar] [CrossRef]
- Ghaima, K.K. Antibacterial and Wound Healing Activity of Some Agrimonia eupatoria Extracts. Baghdad Sci. J. 2013, 10, 152–160. [Google Scholar] [CrossRef][Green Version]
- Cheesman, M.J.; Alcorn, S.; Verma, V.; Cock, I.E. An assessment of the growth inhibition profiles of Hamamelis virginiana L. extracts against Streptococcus and Staphylococcus spp. J. Tradit. Complement. Med. 2021, 11, 457–465. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils-a review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Fialova, S.; Rendekova, K.; Mucaji, P.; Slobodnikova, L. Plant Natural Agents: Polyphenols, Alkaloids and Essential Oils as Perspective Solution of Microbial Resistance. Curr. Org. Chem. 2017, 21, 1875–1884. [Google Scholar] [CrossRef]
- Tariq, S.; Wani, S.; Rasool, W.; Shafi, K.; Bhat, M.A.; Prabhakar, A.; Shalla, A.H.; Rather, M.A. A comprehensive review of the antibacterial, antifungal and antiviral potential of essential oils and their chemical constituents against drug-resistant microbial pathogens. Microb. Pathog. 2019, 134, 103580. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils-Present Status and Future Perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef]
- Li, Z.H.; Cai, M.; Liu, Y.S.; Sun, P.L.; Luo, S.L. Antibacterial Activity and Mechanisms of Essential Oil from Citrus medica L. var. sarcodactylis. Molecules 2019, 24, 1577. [Google Scholar] [CrossRef]
- Wang, X.; Shen, Y.; Thakur, K.; Han, J.; Zhang, J.-G.; Hu, F.; Wei, Z.-J. Antibacterial Activity and Mechanism of Ginger Essential Oil against Escherichia coli and Staphylococcus aureus. Molecules 2020, 25, 3955. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, X.; Wang, Y.; Jiang, P.P.; Quek, S.Y. Antibacterial activity and mechanism of cinnamon essential oil against Escherichia coli and Staphylococcus aureus. Food Control 2016, 59, 282–289. [Google Scholar] [CrossRef]
- Szabó, M.Á.; Varga, G.Z.; Hohmann, J.; Schelz, Z.; Szegedi, E.; Amaral, L.; Molnár, J. Inhibition of quorum-sensing signals by essential oils. Phytother. Res. 2010, 24, 782–786. [Google Scholar] [CrossRef]
- Camele, I.; Elshafie, H.S.; Caputo, L.; De Feo, V. Anti-quorum Sensing and Antimicrobial Effect of Mediterranean Plant Essential Oils Against Phytopathogenic Bacteria. Front. Microbiol. 2019, 10, 2619. [Google Scholar] [CrossRef] [PubMed]
- Gadisa, E.; Weldearegay, G.; Desta, K.; Tsegaye, G.; Hailu, S.; Jote, K.; Takele, A. Combined antibacterial effect of essential oils from three most commonly used Ethiopian traditional medicinal plants on multidrug resistant bacteria. BMC Complement. Altern. Med. 2019, 19, 24. [Google Scholar] [CrossRef]
- Langeveld, W.T.; Veldhuizen, E.J.; Burt, S.A. Synergy between essential oil components and antibiotics: A review. Crit. Rev. Microbiol. 2014, 40, 76–94. [Google Scholar] [CrossRef]
- Kamatou, G.P.P.; Vermaak, I.; Viljoen, A.M.; Lawrence, B.M. Menthol: A simple monoterpene with remarkable biological properties. Phytochemistry 2013, 96, 15–25. [Google Scholar] [CrossRef]
- Paul, J. Wound pruritus: Pathophysiology and management. Chronic Wound Care Manag. Res. 2015, 2, 119–127. [Google Scholar] [CrossRef]
- Silva, J.M.; Pereira, C.V.; Mano, F.; Silva, E.; Castro, V.I.B.; Sá-Nogueira, I.; Reis, L.R.; Paiva, A.; Matias, A.A.; Duarte, A.R.C. Therapeutic Role of Deep Eutectic Solvents Based on Menthol and Saturated Fatty Acids on Wound Healing. ACS Appl. Bio Mater. 2019, 2, 4346–4355. [Google Scholar] [CrossRef]
- Karashima, Y.; Damann, N.; Prenen, J.; Talavera, K.; Segal, A.; Voets, T.; Nilius, B. Bimodal action of menthol on the transient receptor potential channel TRPA1. J. Neurosci. 2007, 27, 9874–9884. [Google Scholar] [CrossRef] [PubMed]
- Oz, M.; El Nebrisi, E.G.; Yang, K.S.; Howarth, F.C.; Al Kury, L.T. Cellular and Molecular Targets of Menthol Actions. Front. Pharmacol. 2017, 8, 472. [Google Scholar] [CrossRef] [PubMed]
- Tóth, B.I.; Oláh, A.; Szöllősi, A.G.; Bíró, T. TRP channels in the skin. Br. J. Pharmacol. 2014, 171, 2568–2581. [Google Scholar] [CrossRef]
- Kotan, R.; Kordali, S.; Cakir, A. Screening of antibacterial activities of twenty-one oxygenated monoterpenes. Z. Naturforsch. C J. Biosci. 2007, 62, 507–513. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Denkova, Z.; Stoyanova, A.; Murgov, I.; Schmidt, E.; Geissler, M. Antimicrobial testinas and gas chromatoaraphic analysis of pure oxvaenated monoterpenes 1.8-cineole, α-terpineol, terpinen-4-ol and camphor as well as target comoounds in essential oils of pine (Pinus pinaster), rosemary (Rosmarinus officinalis), tea tree (Melaleuca alternifolia). Sci. Pharm. 2005, 73, 27–39. [Google Scholar] [CrossRef]
- Sarkic, A.; Stappen, I. Essential Oils and Their Single Compounds in Cosmetics—A Critical Review. Cosmetics 2018, 5, 11. [Google Scholar] [CrossRef]
- Aumeeruddy-Elalfi, Z.; Gurib-Fakim, A.; Mahomoodally, F. Antimicrobial, antibiotic potentiating activity and phytochemical profile of essential oils from exotic and endemic medicinal plants of Mauritius. Ind. Crops Prod. 2015, 71, 197–204. [Google Scholar] [CrossRef]
- Casarin, M.; Pazinatto, J.; Santos, R.C.V.; Zanatta, F.B. Melaleuca alternifolia and its application against dental plaque and periodontal disease: A systematic review. Phytother. Res. 2017, 32, 230–242. [Google Scholar] [CrossRef]
- Labib, R.M.; Ayoub, I.M.; Michel, H.E.; Mehanny, M.; Kamil, V.; Hany, M.; Magdy, M.; Moataz, A.; Maged, B.; Mohamed, A. Appraisal on the wound healing potential of Melaleuca alternifolia and Rosmarinus officinalis L. essential oil-loaded chitosan topical preparations. PLoS ONE 2019, 14, e0219561. [Google Scholar] [CrossRef] [PubMed]
- Negut, I.; Grumezescu, V.; Grumezescu, A.M. Treatment Strategies for Infected Wounds. Molecules 2018, 23, 2392. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (Tea Tree) oil: A review of antimicrobial and other medicinal properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar] [CrossRef]
- Loughlin, R.; Gilmore, B.F.; McCarron, P.A.; Tunney, M.M. Comparison of the cidal activity of tea tree oil and terpinen-4-ol against clinical bacterial skin isolates and human fibroblast cells. Lett. Appl. Microbiol. 2008, 46, 428–433. [Google Scholar] [CrossRef]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial Activity of Terpenes and Terpenoids Present in Essential Oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Nazaruk, J.; Polito, L.; Morais-Braga, M.F.B.; Rocha, J.E.; Coutinho, H.D.M.; Salehi, B.; Tabanelli, G.; Montanari, C.; Contreras, M.M.; et al. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018, 215, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Horváth, B.; Šafranko, S.; Jokić, S.; Széchenyi, A.; Kőszegi, T. Antimicrobial Activity of Chamomile Essential Oil: Effect of Different Formulations. Molecules 2019, 24, 4321. [Google Scholar] [CrossRef]
- Khashan, A.A.; Hamad, M.A.; Jadaan. M.S. In vivo antimicrobial activity of Matricaria chamomilla extract against Pathogenic Bacteria induced skin infections in Mice. Sys. Rev. Pharm. 2020, 11, 672–676. [Google Scholar] [CrossRef]
- El-Kalamouni, C.; Venskutonis, P.R.; Zebib, B.; Merah, O.; Raynaud, C.; Talou, T. Antioxidant and Antimicrobial Activities of the Essential Oil of Achillea millefolium L. Grown in France. Medicines 2017, 4, 30. [Google Scholar] [CrossRef]
- Apel, L.; Lorenz, P.; Urban, S.; Sauer, S.; Spring, O.; Stintzing, F.C.; Kammerer, D.R. Phytochemical characterization of different yarrow species (Achillea sp.) and investigations into their antimicrobial activity. Z. Naturforsch. C 2021, 76, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Veneziani, R.C.S.; Ambrósio, S.R.; Martins, C.H.G.; Lemes, D.C.; Oliveira., L.C. Antibacterial Potential of Diterpenoids. In Studies in Natural Products Chemistry, 1st ed.; Atta-ur-Rahman, Ed.; Elsevier: Oxford, UK, 2017; Volume 54, pp. 109–139. [Google Scholar] [CrossRef]
- Pavić, V.; Jakovljević, M.; Molnar, M.; Jokić, S. Extraction of Carnosic Acid and Carnosol from Sage (Salvia officinalis L.) Leaves by Supercritical Fluid Extraction and Their Antioxidant and Antibacterial Activity. Plants 2019, 8, 16. [Google Scholar] [CrossRef]
- Ojeda-Sana, A.M.; Repetto, V.; Moreno, S. Carnosic acid is an efflux pumps modulator by dissipation of the membrane potential in Enterococcus faecalis and Staphylococcus aureus. World J. Microbiol. Biotechnol. 2013, 29, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, N.M.; Fiorilli, G.; Cáceres Guido, P.A.; Moreno, S. Carnosic acid acts synergistically with gentamicin in killing methicillin-resistant Staphylococcus aureus clinical isolates. Phytomedicine 2016, 23, 1337–1343. [Google Scholar] [CrossRef]
- Wijesundara, N.M.; Rupasinghe, H.P.V. Essential oils from Origanum vulgare and Salvia officinalis exhibit antibacterial and anti-biofilm activities against Streptococcus pyogenes. Microb. Pathog. 2018, 117, 118–128. [Google Scholar] [CrossRef]
- Khalil, N.; Fikry, S.; Salama, O. Bactericidal activity of Myrrh extracts and two dosage forms against standard bacterial strains and multidrug-resistant clinical isolates with GC/MS profiling. AMB Express 2020, 10, 21. [Google Scholar] [CrossRef]
- Papadopoulou, K.; Melton, R.E.; Leggett, M.; Daniels, M.J.; Osbourn, A.E. Compromised disease resistance in saponin-deficient plants. Proc. Natl. Acad. Sci. USA 1999, 96, 12923–12928. [Google Scholar] [CrossRef] [PubMed]
- Kavya, N.M.; Adil, L.; Senthilkumar, P.A. Review on Saponin Biosynthesis and its Transcriptomic Resources in Medicinal Plants. Plant Mol. Biol. Rep. 2021, 1–8. [Google Scholar] [CrossRef]
- Schmidt, S.; Heimesaat, M.M.; Fischer, A.; Bereswill, S.; Melzig, M.F. Saponins increase susceptibility of vancomycin-resistant enterococci to antibiotic compounds. Eur. J. Microbiol. Immunol. 2014, 4, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.T.S.; Berchová-Bímová, K.; Petráš, J.; Hassan, K.T.S. Cucurbitacin B interacts synergistically with antibiotics against Staphylococcus aureus clinical isolates and exhibits antiviral activity against HSV-1. S. Afr. J. Bot. 2017, 108, 90–94. [Google Scholar] [CrossRef]
- Nicolaus, C.; Junghanns, S.; Hartmann, A.; Murillo, R.; Ganzera, M.; Merfort, I. In vitro studies to evaluate the wound healing properties of Calendula officinalis extracts. J. Ethnopharmacol. 2017, 196, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.S.; Cheng, L.N.; Wu, J.H.; Chan, E.; Kwan, Y.W.; Lee, S.M.; Leung, G.P.; Yu, P.H.; Chan, S.W. A review of the pharmacological effects of Arctium lappa (burdock). Inflammopharmacology 2011, 19, 245–254. [Google Scholar] [CrossRef]
- Watkins, F.; Pendry, B.; Sanchez-Medina, A.; Corcoran, O. Antimicrobial assays of Tyree native British plants used In Anglo-Saxon medicine for wound healing formulations in 10th century England. J. Ethnopharmacol. 2012, 144, 408–415. [Google Scholar] [CrossRef]
- Lou, Z.; Li, C.; Kou, X.; Yu, F.; Wang, H.; Smith, G.M.; Zhu, S. Antibacterial, antibiofilm effect of Burdock (Arctium lappa L.) leaf fraction and its efficiency in meat preservation. J. Food Prof. 2016, 79, 1404–1409. [Google Scholar] [CrossRef]
- Miazga-Karska, M.; Michalak, K.; Ginalska, G. Anti-Acne Action of Peptides Isolated from Burdock Root-Preliminary Studies and Pilot Testing. Molecules 2020, 25, 2027. [Google Scholar] [CrossRef]
- Dabos, K.J.; Sfika, E.; Vlatta, L.J.; Giannikopoulos, G. The effect of mastic gum on Helicobacter pylori: A randomized pilot study. Phytomedicine 2010, 17, 296–299. [Google Scholar] [CrossRef]
- Mezni, F.; Aouadhi, C.; Khouja, M.L.; Khaldi, A.; Maaroufi, A. In vitro antimicrobial activity of Pistacia lentiscus L. edible oil and phenolic extract. Nat. Prod. Res. 2015, 29, 565–570. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.J.; Stinco, C.M.; Mapelli-Brahm, P. Skin Carotenoids in Public Health and Nutricosmetics: The Emerging Roles and Applications of the UV Radiation-Absorbing Colourless Carotenoids Phytoene and Phytofluene. Nutrients 2019, 11, 1093. [Google Scholar] [CrossRef] [PubMed]
- Zinder, R.; Cooley, R.; Vlad, L.G.; Molnar, J.A. Vitamin A and Wound Healing. Nutr. Clin. Pract. 2019, 34, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Toti, E.; Chen, C.O.; Palmery, M.; Villaño Valencia, D.; Peluso, I. Non-Provitamin A and Provitamin a Carotenoids as Immunomodulators: Recommended Dietary Allowance, Therapeutic Index, or Personalized Nutrition? Oxid. Med. Cell Longev. 2018, 2018, 4637861. [Google Scholar] [CrossRef] [PubMed]
- Balić, A.; Mokos, M. Do We Utilize Our Knowledge of the Skin Protective Effects of Carotenoids Enough? Antioxidants 2019, 8, 259. [Google Scholar] [CrossRef]
- Ibrahim, H.A.H. Antibacterial carotenoids of three Holothuria species in Hurghada, Egypt. Egypt. J. Aquat. Res. 2012, 38, 185–194. [Google Scholar] [CrossRef]
- Karpiński, T.M.; Adamczak, A. Fucoxanthin-An Antibacterial Carotenoid. Antioxidants 2019, 8, 239. [Google Scholar] [CrossRef]
- Xue, M.; Jackson, C. Extracellular matrix reorganization during wound healing and its impact on abnormal scarring. Adv. Wound Care 2015, 4, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Efstratiou, E.; Hussain, A.I.; Nigam, P.S.; Moore, J.E.; Ayub, M.A.; Rao, J.R. Antimicrobial activity of Calendula officinalis petal extracts against fungi, as well as Gram-negative and Gram-positive clinical pathogens. Complement. Ther. Clin. Pract. 2012, 18, 173–176. [Google Scholar] [CrossRef]
- Menda, J.; Reddy, T.; Deepika, R.; Pandima, D.; Sastry, T.P. Preparation and characterization of wound healing composites of chitosan, Aloe vera and Calendula officinalis—A comparative study. Am. J. Phytomed. Clin. Ther. 2014, 2, 61–76. [Google Scholar]
- Givol, O.; Kornhaber, R.; Visentin, D.; Cleary, M.; Haik, J.; Harats, M. A systematic review of Calendula officinalis extract for wound healing. Wound Repair Regen. 2019, 27, 548–561. [Google Scholar] [CrossRef]
- Moniruzzaman; Shahinuzzaman; Haque, A.; Khatun, R.; Yaakob, Z. Gas chromatography mass spectrometry analysis and in vitro antibacterial activity of essential oil from Trigonella foenum-graecum. Asian Pac. J. Trop. Biomed. 2015, 5, 1033–1036. [Google Scholar] [CrossRef]
- Kiyama, R. Estrogenic Activity of Coffee Constituents. Nutrients 2019, 11, 1401. [Google Scholar] [CrossRef]
- Horng, H.C.; Chang, W.H.; Yeh, C.C.; Huang, B.S.; Chang, C.P.; Chen, Y.J.; Tsui, K.H.; Wang, P.H. Estrogen Effects on Wound Healing. Int. J. Mol. Sci. 2017, 18, 2325. [Google Scholar] [CrossRef]
- Čoma, M.; Lachová, V.; Mitrengová, P.; Gál, P. Molecular Changes Underlying Genistein Treatment of Wound Healing: A Review. Curr. Issues Mol. Biol. 2021, 43, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Chan, L.; Zhou, S. Trigonelline: A plant alkaloid with therapeutic potential for diabetes and central nervous system disease. Curr. Med. Chem. 2012, 19, 3523–3531. [Google Scholar] [CrossRef] [PubMed]
- Benyagoub, E.; Nabbou, N.; Aguid, A.; Alkhudhairy, M.K.; Bendada, F. In vitro Antibacterial Activity of Fenugreek Seeds’ Phytoconstituents from Taghit Region (Southwest of Algeria) Against the Bacterial Strains Responsible for UTI. Curr. Bioact. Compd. 2021, 17, 339–355. [Google Scholar] [CrossRef]
- Turker, A.U.; Usta, C. Biological screening of some Turkish medicinal plant extracts for antimicrobial and toxicity activities. Nat. Prod. Res. 2008, 22, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Slobodníková, L.; Košálová, D.; Labudová, D.; Kotulová, D.; Kettmann, V. Antimicrobial activity of Mahonia aquifolium crude extract and its major isolated alkaloids. Phytother. Res. 2004, 18, 674–676. [Google Scholar] [CrossRef]
- Zuo, G.Y.; Meng, F.Y.; Hao, X.Y.; Zhang, Y.L.; Wang, G.C.; Xu, G.L. Antibacterial Alkaloids from Chelidonium Majus Linn (Papaveraceae) Against Clinical Isolates of Methicillin-Resistant Staphylococcus aureus. J. Pharm. Pharm. Sci. 2009, 11, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.; Cushnie, B.; Lamb, A.J. Alkaloids: An overview of their antibacterial, antibiotic-enhancing and antivirulence activities. Int. J. Antimicrob. Agents 2014, 44, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Grip, J.; Engstad, R.E.; Skjæveland, I.; Škalko-Basnet, N.; Holsæter, A.M. Sprayable Carbopol hydrogel with soluble beta-1,3/1,6-glucan as an active ingredient for wound healing—Development and in-vivo evaluation. Eur. J. Pharm. Sci. 2017, 107, 24–31. [Google Scholar] [CrossRef]
- Ciriminna, R.; Fidalgo, A.; Meneguzzo, F.; Presentato, A.; Scurria, A.; Nuzzo, D.; Alduina, R.; Ilharco, L.M.; Pagliaro, M. Pectin: A Long-Neglected Broad-Spectrum Antibacterial. Chem. Med. Chem. 2020, 15, 2228–2235. [Google Scholar] [CrossRef] [PubMed]
- Giusto, G.; Vercelli, C.; Comino, F.; Caramello, V.; Tursi, M.; Gandini, M. A new, easy-to-make pectin-honey hydrogel enhances wound healing in rats. BMC Complement. Altern. Med. 2017, 17, 266. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, S.J.; Mutters, N.T.; Blessing, B.; Günther, F. Natural isothiocyanates express antimicrobial activity against developing and mature biofilms of Pseudomonas aeruginosa. Fitoterapia 2017, 119, 57–63. [Google Scholar] [CrossRef]
- Dufour, V.; Stahl, M.; Baysse, C. The antibacterial properties of isothiocyanates. Microbiology 2015, 161, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Tajima, H.; Kimoto, H.; Taketo, A. Specific antimicrobial synergism of synthetic hydroxy isothiocyanates with aminoglycoside antibiotics. Biosci. Biotechnol. Biochem. 2001, 65, 1886–1888. [Google Scholar] [CrossRef]
- Saavedra, M.J.; Borges, A.; Dias, C.; Aires, A.; Bennett, R.N.; Rosa, E.S.; Simões, M. Antimicrobial activity of phenolics and glucosinolate hydrolysis products and their synergy with streptomycin against pathogenic bacteria. Med. Chem. 2010, 6, 174–183. [Google Scholar] [CrossRef]
- Avato, P.; Raffo, F.; Guglielmi, G.; Vitali, C.; Rosato, A. Extracts from St John’s wort and their antimicrobial activity. Phytother. Res. 2004, 18, 230–232. [Google Scholar] [CrossRef]
- Wölfle, U.; Seelinger, G.; Schempp, C.M. Topical application of St. John’s wort (Hypericum perforatum). Planta Med. 2014, 80, 109–120. [Google Scholar] [CrossRef]
- Lyles, J.T.; Kim, A.; Nelson, K.; Bullard-Roberts, A.L.; Hajdari, A.; Mustafa, B.; Quave, C.L. The Chemical and Antibacterial Evaluation of St. John’s Wort Oil Macerates Used in Kosovar Traditional Medicine. Front. Microbiol. 2017, 8, 1639. [Google Scholar] [CrossRef] [PubMed]
- Doğan, S.; Gökalsın, B.; Şenkardeş, I.; Doğan, A.; Cenk Sesal, N. Anti-quorum sensing and anti-biofilm activities of Hypericum perforatum extracts against Pseudomonas aeruginosa. J. Ethnopharmacol. 2019, 235, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Nazli, O.; Baygar, T.; Dönmez, C.E.D.; Dere, Ö.; Uysal, A.I.; Aksözek, A.; Işık, C.; Aktürk, S. Antimicrobial and anti-biofilm activity of polyurethane/Hypericum perforatum extract (PHPE) composite. Bioorgan. Chem. 2019, 82, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Saddiqe, Z.; Naeem, I.; Maimoona, A. A review of the antibacterial activity of Hypericum perforatum L. J. Ethnopharmacol. 2010, 131, 511–521. [Google Scholar] [CrossRef]
- Raclariu, A.C.; Paltinean, R.; Vlase, L.; Labarre, A.; Manzanilla, V.; Ichim, M.C.; Crisan, G.; Brysting, A.K.; De Boer, H. Comparative authentication of Hypericum perforatum herbal products using DNA metabarcoding, TLC and HPLC-MS. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.B.; Isik, K.; Marshall, J.R. Wound-Healing Potential of Oil of Hypercium perforatum in Excision Wounds of Male Sprague Dawley Rats. Adv. Wound Care 2017, 6, 401–406. [Google Scholar] [CrossRef]
- Schempp, C.M.; Hezel, S.; Simon, J.C. Topical treatment of atopic dermatitis with Hypericum cream. A randomised, placebo-controlled, double-blind half-side comparison study. Hautarzt 2003, 54, 248–253. [Google Scholar] [CrossRef]
- Silva, J.R.; Burger, B.; Kühl, C.; Candreva, T.; Dos Anjos, M.; Rodrigues, H.G. Wound Healing and Omega-6 Fatty Acids: From Inflammation to Repair. Mediat. Inflamm. 2018, 2018, 2503950. [Google Scholar] [CrossRef]
- Zheng, C.J.; Yoo, J.S.; Lee, T.G.; Cho, H.Y.; Kim, Y.H.; Kim, W.G. Fatty acid synthesis is a target for antibacterial activity of unsaturated fatty acids. FEBS Lett. 2005, 579, 5157–5162. [Google Scholar] [CrossRef]
- Desbois, A.P.; Lawlor, K.C. Antibacterial Activity of Long-Chain Polyunsaturated Fatty Acids against Propionibacterium acnes and Staphylococcus aureus. Mar. Drugs 2013, 11, 4544–4557. [Google Scholar] [CrossRef]
- Senapati, S.; Banerjee, S.; Gangopadhyay, D.N. Evening primrose oil is effective in atopic dermatitis: A randomized placebo-controlled trial. Indian J. Dermatol. Venereol. Leprol. 2008, 74, 447–452. [Google Scholar] [CrossRef]
- Williams, H.C. Evening primrose oil for atopic dermatitis. BMJ 2003, 327, 1358–1359. [Google Scholar] [CrossRef]
- Shawky, E.M.; Elgindi, M.R.; Ibrahim, H.A.; Baky, M.H. The potential and outgoing trends in traditional, phytochemical, economical, and ethnopharmacological importance of family Onagraceae: A comprehensive review. J. Ethnopharmacol. 2021, 281, 114450. [Google Scholar] [CrossRef] [PubMed]
- Kurin, E.; Mučaji, P.; Nagy, M. In Vitro Antioxidant Activities of Three Red Wine Polyphenols and Their Mixtures: An Interaction Study. Molecules 2012, 17, 14336–14348. [Google Scholar] [CrossRef] [PubMed]
- Imming, P.; Sinning, C.; Meyer, A. Drugs, their targets and the nature and number of drug targets. Nat. Rev. Drug Discov. 2006, 5, 821–834. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.; Ulrich-Merzenich, G. Synergy research: Approaching a new generation of phytopharmaceuticals. Phytomedicine 2009, 16, 97–110. [Google Scholar] [CrossRef]
- Caesar, L.K.; Cech, N.B. Synergy and antagonism in natural product extracts: When 1 + 1 does not equal 2. Nat. Prod. Rep. 2019, 36, 869–888. [Google Scholar] [CrossRef]
- Ettefagh, K.A.; Burns, J.T.; Junio, H.A.; Kaatz, G.W.; Cech, N.B. Goldenseal (Hydrastis canadensis L.) Extracts Synergistically Enhance the Antibacterial Activity of Berberine via Efflux Pump Inhibition. Planta Med. 2011, 77, 835–840. [Google Scholar] [CrossRef]
- Sanhueza, L.; Melo, R.; Montero, R.; Maisey, K.; Mendoza, L.; Wilkens, M. Synergistic interactions between phenolic compounds identified in grape pomace extract with antibiotics of different classes against Staphylococcus aureus and Escherichia coli. PLoS ONE 2017, 12, e0172273. [Google Scholar] [CrossRef]
- Rosato, A.; Carocci, A.; Catalano, A.; Clodoveo, M.L.; Franchini, C.; Corbo, F.; Carbonara, G.G.; Carrieri, A.; Fracchiolla, G. Elucidation of the synergistic action of Mentha piperita essential oil with common antimicrobials. PLoS ONE 2018, 13, e0200902. [Google Scholar] [CrossRef]
- Van Vuuren, S.; Orchard, A.; Viljoen, A. Essential oils: Fragrant pools of antimicrobial synergism explored. Synergy 2019, 9, 100051. [Google Scholar] [CrossRef]
- Bassolé, I.H.N.; Juliani, H.R. Essential Oils in Combination and Their Antimicrobial Properties. Molecules 2012, 17, 3989–4006. [Google Scholar] [CrossRef]
- Van Vuuren, S.; Viljoen, A. Plant-based antimicrobial studies—Methods and approaches to study the interaction between natural products. Planta Med. 2011, 77, 1168–1182. [Google Scholar] [CrossRef] [PubMed]
- Iten, F.; Saller, R.; Abel, G.; Reichling, J. Additive antimicrobial effects of the active components of the essential oil of Thymus vulgaris—Chemotype carvacrol. Planta Med. 2009, 75, 1231–1236. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.W.; Sharili, A.S.; Phee, L.M.; Wareham, D.W. In Vitro Activity of Epigallocatechin Gallate and Quercetin Alone and in Combination versus Clinical Isolates of Methicillin-Resistant Staphylococcus aureus. J. Nat. Prod. 2015, 78, 2145–2148. [Google Scholar] [CrossRef]
- Tomás-Menor, L.; Barrajón-Catalán, E.; Segura-Carretero, A.; Martí, N.; Saura, D.; Menéndez, J.A.; Joven, J.; Micol, V. The promiscuous and synergic molecular interaction of polyphenols in bactericidal activity: An opportunity to improve the performance of antibiotics? Phytother. Res. 2015, 29, 466–473. [Google Scholar] [CrossRef]
- Farooqui, A.; Khan, A.; Borghetto, I.; Kazmi, S.U.; Rubino, S.; Paglietti, B. Synergistic Antimicrobial Activity of Camellia sinensis and Juglans regia against Multidrug-Resistant Bacteria. PLoS ONE 2015, 10, e0118431. [Google Scholar] [CrossRef] [PubMed]
- Karioti, A.; Sokovic, M.; Ciric, A.; Koukoulitsa, C.; Bilia, A.R.; Skaltsa, H. Antimicrobial properties of Quercus ilex L. proanthocyanidin dimers and simple phenolics: Evaluation of their synergistic activity with conventional antimicrobials and prediction of their pharmacokinetic profile. J. Agric. Food Chem. 2011, 59, 6412–6422. [Google Scholar] [CrossRef] [PubMed]
- Cheesman, M.J.; Ilanko, A.; Blonk, B.; Cock, I.E. Developing New Antimicrobial Therapies: Are Synergistic Combinations of Plant Extracts/Compounds with Conventional Antibiotics the Solution? Pharmacogn. Rev. 2017, 11, 57–72. [Google Scholar] [CrossRef]

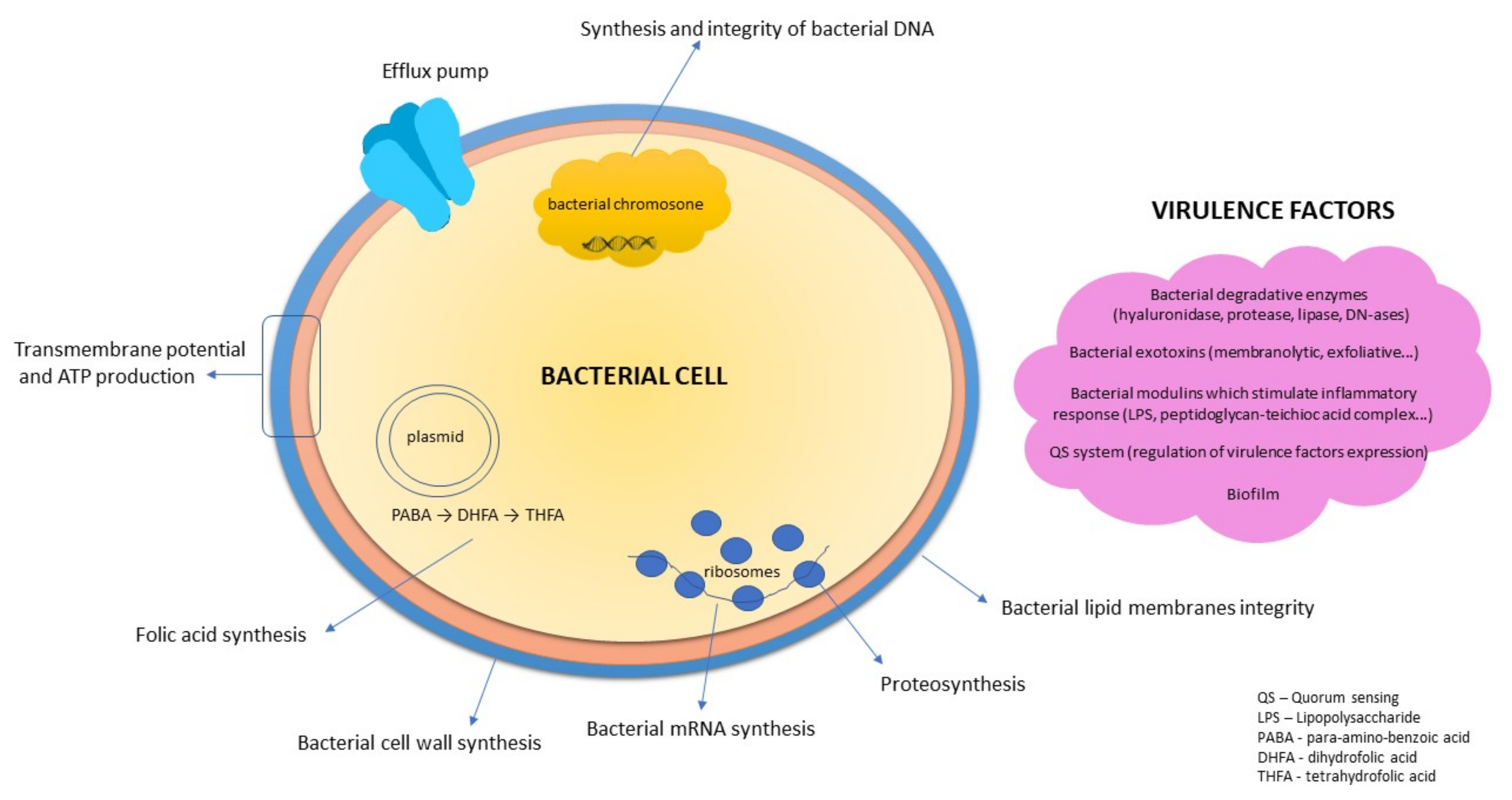
| Type of Infection | The Most Common Infectious Agents |
|---|---|
| Impetigo | Staphylococcus aureus, Streptococcus pyogenes |
| Abscess | S. aureus Polymicrobial |
| Furuncle, carbuncle | S. aureus |
| Erysipelas | S. pyogenes |
Cellulitis
| S. aureus, S. pyogenes S. aureus Aeromonas hydrophila Vibrio vulnificus Human oral microbiota Pasteurella multocida; animal oral microbiota |
| Ecthyma gangrenosum | Pseudomonas aeruginosa Gram-negative bacilli |
| Surgical site infection—clean surgery | Staphylococcus spp. |
| Surgical site infection—all types | Anaerobic bacteria Enterococcus spp. Gram-negative bacilli Staphylococcus pp. Streptococcus spp. |
| Infected pressure ulcers | Polymicrobial |
| Necrotising fasciitis | S. aureus, S. pyogenes Polymicrobial |
| Myonecrosis | Myonecrotic clostridia |
| Acne lesions | Cutibacterium acnes (Staphylococcus aureus, Gram-negative rods) |
| Medicinal Plant (Source) | Drug (Herbal Substance or Preparation) | EMA Recommended Therapeutic Indication for Skin (Traditional Use) [40] | Secondary Metabolites Responsible for Intended Indications [40,43] |
|---|---|---|---|
| Achillea millefolium L. | herb/flower (Millefolii herba/Millefolii flos) | Traditional herbal medicinal product for the treatment of small superficial wounds. | sequiterpenic lactones, flavonoids, cinnamic acid derivatives |
| Agrimonia eupatoria L. | herb (Agrimoniae herba) | Traditional herbal medicinal product for relief of minor skin inflammation and small, superficial wounds. | tannins (agrimoniin), flavonoids, phenolic acids, triterpenoids |
| Arctium lappa L. | root (Arctii radix) | Traditional herbal medicinal product used in treatment of seborrhoeic skin conditions. | triterpenes (α-amyrin and β-amyrin), lignans (arctiin), and hydroxycinnamic derivatives |
| Avena sativa L. | fruit (Avenae fructus) | Traditional herbal medicinal product for the symptomatic treatment of minor inflammations of the skin (such as sunburn) and as an aid in healing of minor wounds. | avenanthramides (A–C), steroidal saponins (avenacoside A and B), triterpene saponins (avenacines), flavonoids |
| Calendula officinalis L. | flower (Calendulae flos) | Traditional herbal medicinal product for the symptomatic treatment of minor inflammations of the skin (such as sunburn) and as an aid in healing of minor wounds. | triterpenic saponins (calendulosides), triterpenic alcohols (α-amyrin, β-amyrin, lupeol), carotenoids (flavoxanthin, zeaxanthine, lutein), flavonoids (quercetin, isorhamnetin), coumarins, polysaccharides, volatile compounds |
| Commiphora molmol Engler and/or other Commiphora species | rubber resin (Myrrha, gummi-resina) | Traditional herbal medicinal product for treatment of minor wounds and small boils (such as furuncles). | diterpenes (muculol), triterpenes (myrrhanol A, myrrhanone A), steroids, essential oil |
| Echinacea purpurea (L.) Moench | fresh herb (Echinaceae purpureae herba, recens) | Traditional herbal medicinal product for treatment of small superficial wounds. | cinnamic acid derivatives, alkamides, polysaccharides |
| Echinacea purpurea (L.) Moench | root (Echinaceae purpureae radix) | Traditional herbal medicinal product used for the relief of spots and pimples due to mild acne. | cinnamic acid derivatives, alkamides, polysaccharides, glycoproteins, essential oil |
| Equisetum arvense L. | herb (Equiseti herba) | Traditional herbal medicinal product used for supportive treatment of superficial wounds. | flavonoids (glycosides of quercetin and kaempferol), silicates |
| Glycine max (L.) Merr. | oil (Soiae oleum raffinatum) | Traditional herbal medicinal product used for the symptomatic relief of dry skin conditions associated with mild recurrent eczema. | fatty acids (linoleic acid, oleic acid, palmitic acid, linolenic acid, stearic acid) |
| Hamamelis virginiana L. | leaf/bark (Hamamelidis folium/Hamamelidis cortex) | Traditional herbal medicinal product for relief of minor skin inflammation and dryness of the skin. | tannins, flavonoids, phenolic acids |
| Hypericum perforatum L. | herb (Hyperici herba) | Traditional herbal medicinal product for the symptomatic treatment of minor inflammations of the skin (such as sunburn) and as an aid in healing of minor wounds. | phloroglucinols (hyperforin with its derivatives: adhyperforin, furohyperforin, fu-roadhyperforin), naftodianthrone (hypericin), flavonoids (derivatives of quercetin), biflavons, tannins, volatile compounds |
| Juglans regia L. | leaf (Juglandis folium) | Traditional herbal medicinal product for the relief of minor inflammatory conditions of the skin and in excessive perspiration of hands and feet. | flavonoids (quercetin derivatives), tannins, hydroxycinnamic derivatives |
| Matricaria recutita L. | flower (Matricariae flos) | Traditional herbal medicinal product for adjuvant therapy of irritations of skin and mucosae in the anal and genital region and for the treatment of minor inflammation of the skin (sunburn), superficial wounds and small boils (furuncles). | essential oil, flavonoids (apigenin, quercetin and luteolin derivatives), coumarins, N1,N5,N10,N14-tetra-p-coumaroylspermine, polysaccharides |
| Matricaria recutita L. | essential oil (Matricariae aetheroleum) | Traditional herbal medicinal product used for adjuvant therapy of irritations of skin and mucosae in the anal and genital region, after serious conditions have been excluded by a medical doctor. | sesquiterpenes ((-)-α-bisabolol, chamazulene, and bisabololoxides A, B, C) |
| Melaleuca alternifolia (Maiden and Betch) Cheel, M. linariifolia Smith, M. dissitiflora F. Mueller or other species of Melaleuca | essential oil (Melaleucae aetheroleum) | Traditional herbal medicinal product for treatment of small superficial wounds and insect bites, for treatment of small boils (furuncles and mild acne) and for the relief of itching and irritation in cases of mild athlete’s foot. | terpinen-4-ol, γ-terpinene, 1,8-cineole, α-terpinene, p-cymene, α-terpineole, aromadendrene, α-pinene, terpinolene, limonene, sabinene, α-phellandrene |
| Melilotus officinalis (L.) Lam. | herb (Meliloti herba) | Traditional herbal medicinal product used for the treatment of minor inflammations of the skin. | coumarins (coumarin, scopoletin, umbelliferone, melilotin), flavonoids (glycosides of quercetin and kaempferol), triterpene saponins (melilotoside A-C) |
| Mentha × piperita L. | essential oil (Menthae piperitae aetheroleum) | Traditional herbal medicinal product used for the symptomatic relief of localized pruritic conditions in intact skin. | menthol, menthone, 1,8-cineol |
| Oenothera biennis L., O. lamarckiana L. | oil (Oenotherae oleum) | Traditional herbal medicinal product for the symptomatic relief of itching in acute and chronic dry skin conditions exclusively based upon long-standing use. | fatty acids (cis-linoleic acid, γ-linolenic acid, oleic acid, palmitic acid) |
| Origanum dictamnus L. | herb (Origani dictamni herba) | Traditional herbal medicinal product used for the relief of minor skin inflammations and bruises. | essential oil (thymol, carvacrol), flavonoids, triterpenes, cinnamic acid derivatives |
| Origanum majorana L. | herb (Origani majoranae herba) | Traditional herbal medicinal product used for relief of irritated skin around the nostrils. | essential oil (terpinen-4-ol, (+)-cis-sabinene hydrat, α-terpinene, γ-terpinene, p-cymene), flavonoids, triterpenes, cinnamic acid derivatives |
| Pistacia lentiscus L. | resin (resinum/mastic) | Traditional herbal medicinal product used for the symptomatic treatment of minor inflammations of the skin and as an aid in healing of minor wounds | triterpenes (mastic acid, isomastic acid, oleanolic acid, tirucall), monoterpenes, sesquiterpenes, polyphenols |
| Quercus robur L., Q. petraea (Matt.) Liebl., Q. pubescens Willd. | bark (Quercus cortex) | Traditional herbal medicinal product for the symptomatic treatment of minor inflammation of the oral mucosa or skin | tannins (gallotannins, ellagitannins, flavano-ellagitannins, procyanidinoellagitannin) |
| Rosa gallica L., Rosa centifolia L., Rosa damascena Mill. | flower (Rosae flos) | Traditional herbal medicinal product used for relief of minor skin inflammation. | flavonoids (glycosides of quercetin and kaempferol), anthocyanins (cyanidin-3-O-β-glucoside), proanthocyanidins, carotenoids, essential oil |
| Salvia officinalis L. | leaf (Salviae officinalis folium) | Traditional herbal medicinal product for relief of minor skin inflammations. | cinnamic acid derivatives (rosmarinic acid, salvianolic acids, lithospermic acid), diterpenoids (carnosic acid, carnosol, rosmanol) triterpenes (oleanolic and ursolic acid), flavonoids (derivatives of apigenin and luteolin), essential oil |
| Solanum dulcamara L. | woody nightshade stem (Solani dulcamarae stipites) | Traditional herbal medicinal product for the symptomatic relief of mild recurrent eczema. | steroid alkaloids, steroid triterpenes, tropane alkaloids |
| Trigonella foenum-graecum L. | semen (Trigonellae foenugraeci semen) | Traditional herbal medicinal product for the symptomatic treatment of minor inflammations of the skin. | alkaloids (trigonelline), steroidal saponins, flavonoids, oil |
| Urtica dioica L., Urtica urens L. | herb (Urticae herba) | Traditional herbal medicinal product used in seborrhoeic skin conditions | flavonoids, cinnamic acid derivatives, triterpenes, coumarins |
| Viola tricolor L., and/or subsp. V. arvensis Murray (Gaud), V. vulgaris Koch (Oborny) | herb with flowers (Violae tricoloris herba cum flore) | Traditional herbal medicinal product for symptomatic treatment of mild seborrhoeic skin conditions. | flavonoids (rutin, C-glycosides of luteolin and apigenin), mucilage, caffeic acid, carotenoids (9-cis-violaxanthin), cyclopeptides |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bittner Fialová, S.; Rendeková, K.; Mučaji, P.; Nagy, M.; Slobodníková, L. Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine—A Review. Int. J. Mol. Sci. 2021, 22, 10746. https://doi.org/10.3390/ijms221910746
Bittner Fialová S, Rendeková K, Mučaji P, Nagy M, Slobodníková L. Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine—A Review. International Journal of Molecular Sciences. 2021; 22(19):10746. https://doi.org/10.3390/ijms221910746
Chicago/Turabian StyleBittner Fialová, Silvia, Katarína Rendeková, Pavel Mučaji, Milan Nagy, and Lívia Slobodníková. 2021. "Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine—A Review" International Journal of Molecular Sciences 22, no. 19: 10746. https://doi.org/10.3390/ijms221910746
APA StyleBittner Fialová, S., Rendeková, K., Mučaji, P., Nagy, M., & Slobodníková, L. (2021). Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine—A Review. International Journal of Molecular Sciences, 22(19), 10746. https://doi.org/10.3390/ijms221910746