A Novel In Vitro Assay Using Human iPSC-Derived Sensory Neurons to Evaluate the Effects of External Chemicals on Neuronal Morphology: Possible Implications in the Prediction of Abnormal Skin Sensation
Abstract
:1. Introduction
2. Results
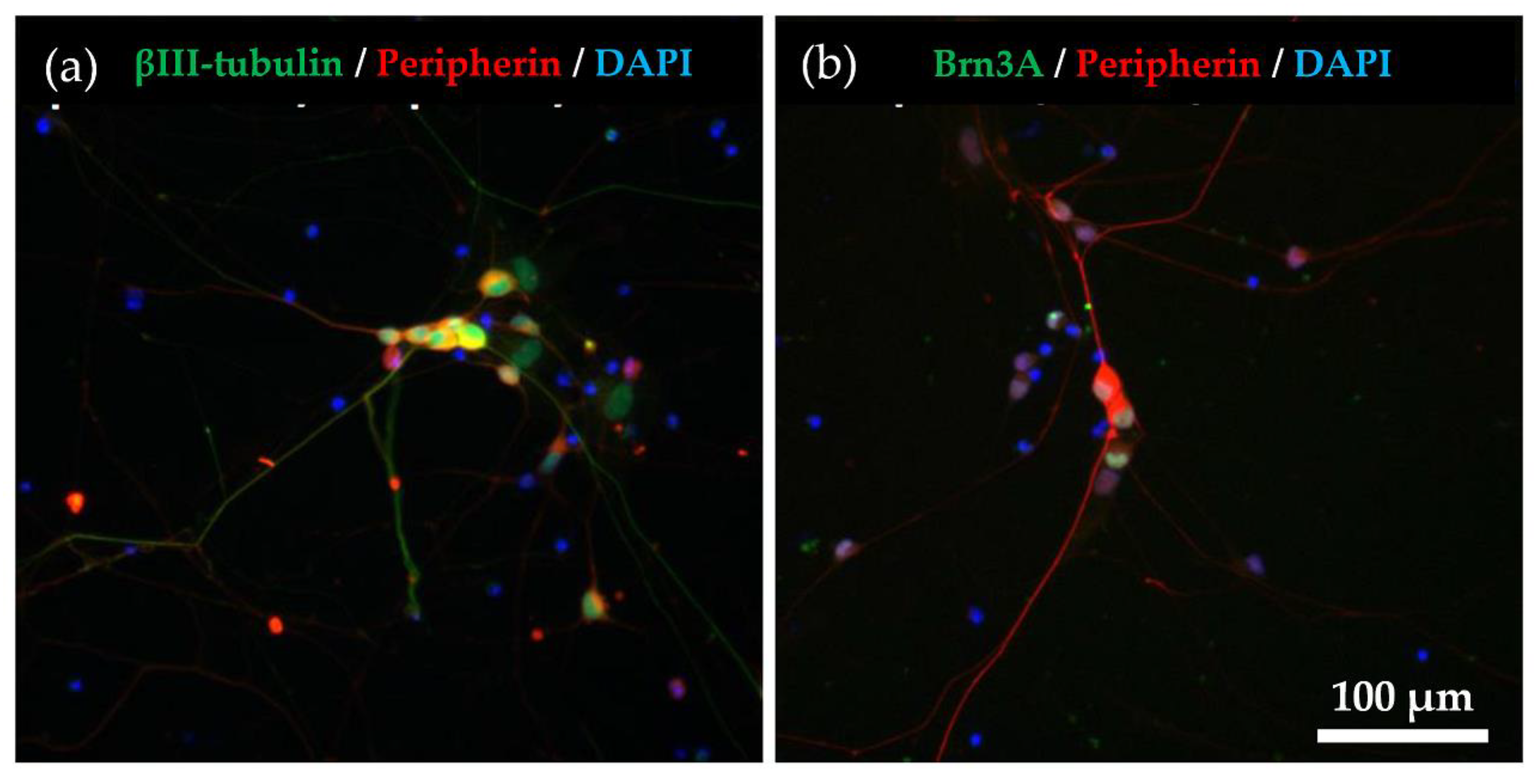
2.1. Immunocytochemical Characterisation of Differentiated hiPSC-SNs
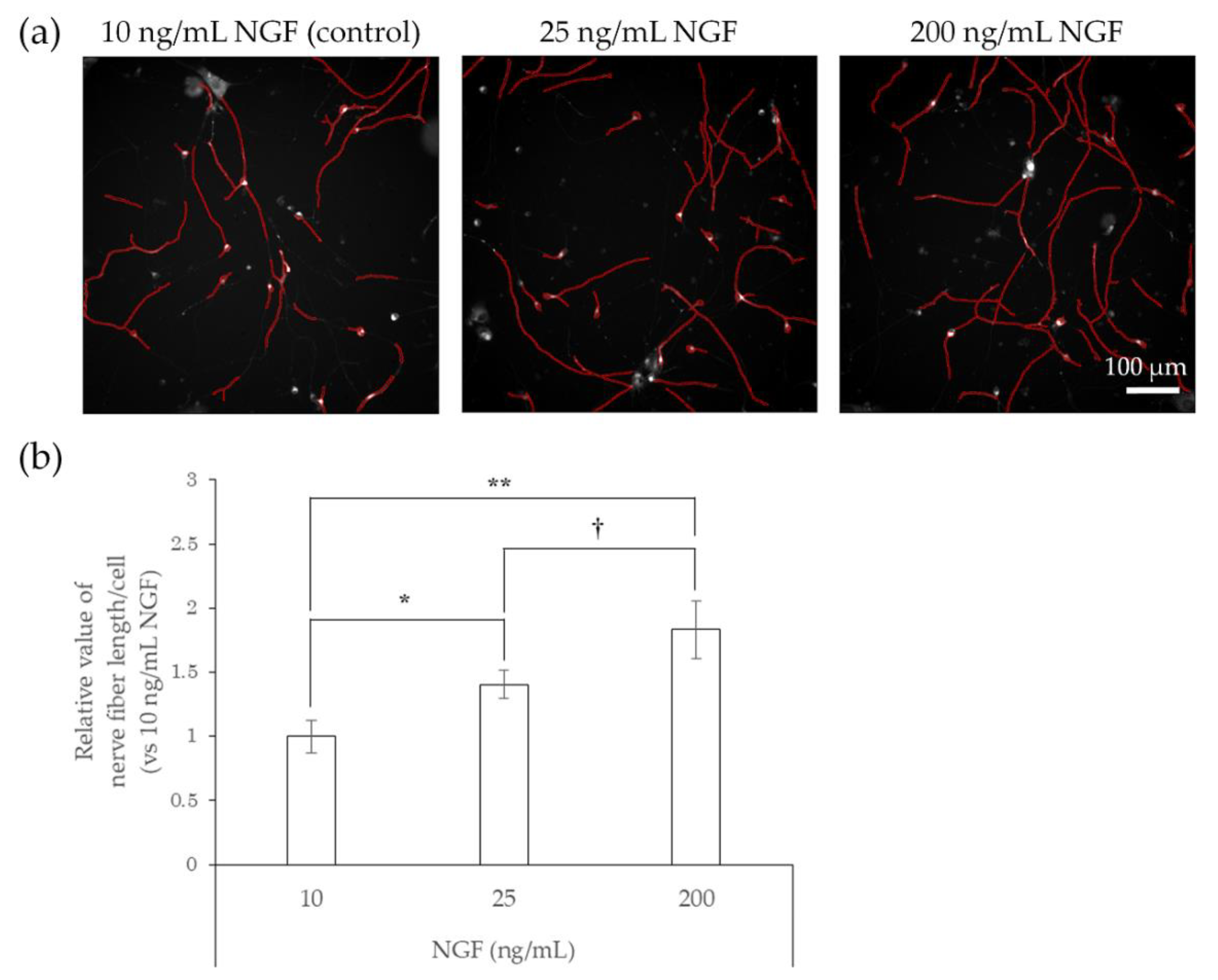
2.2. Responsiveness of hiPSC-SNs to NGF
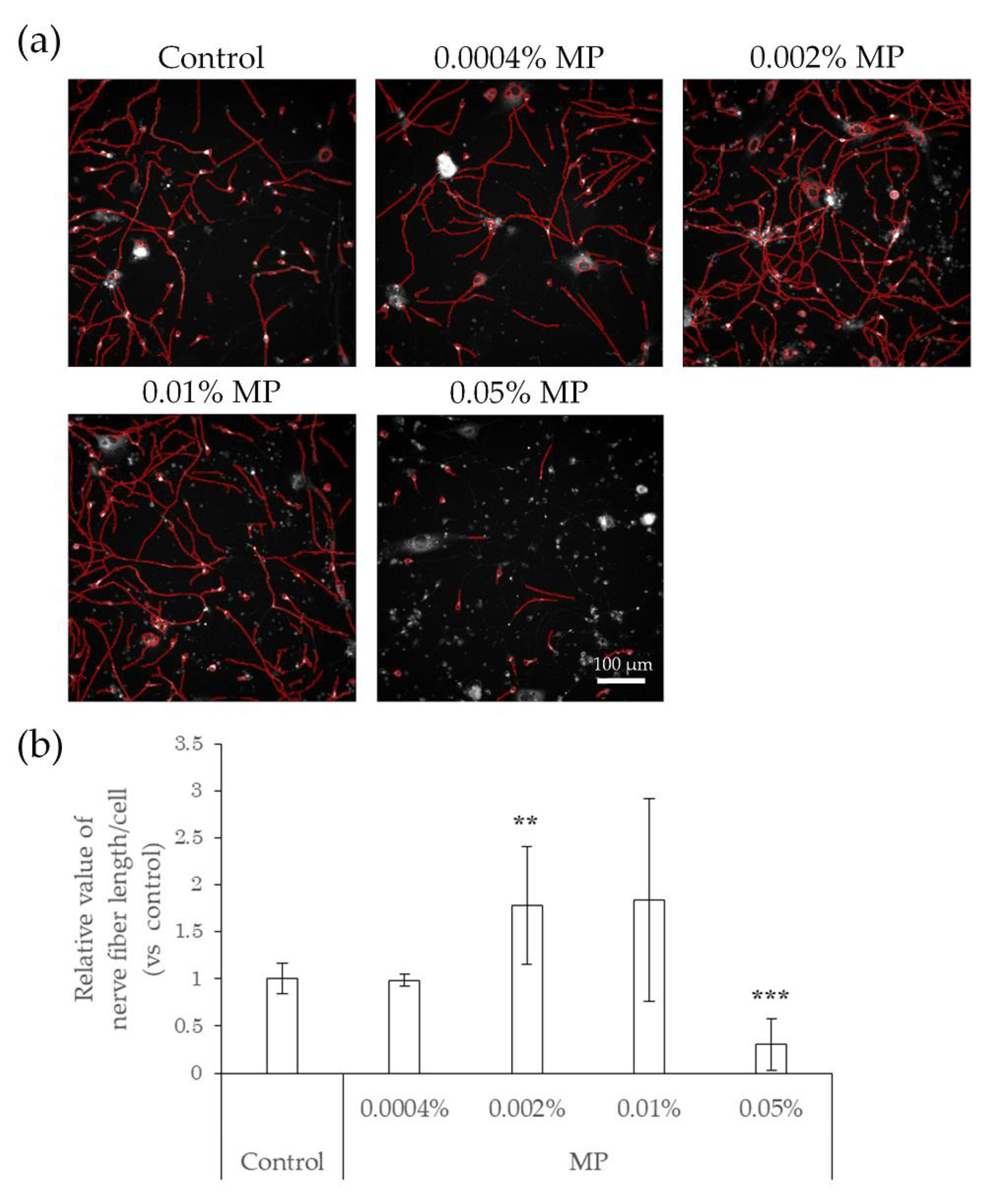
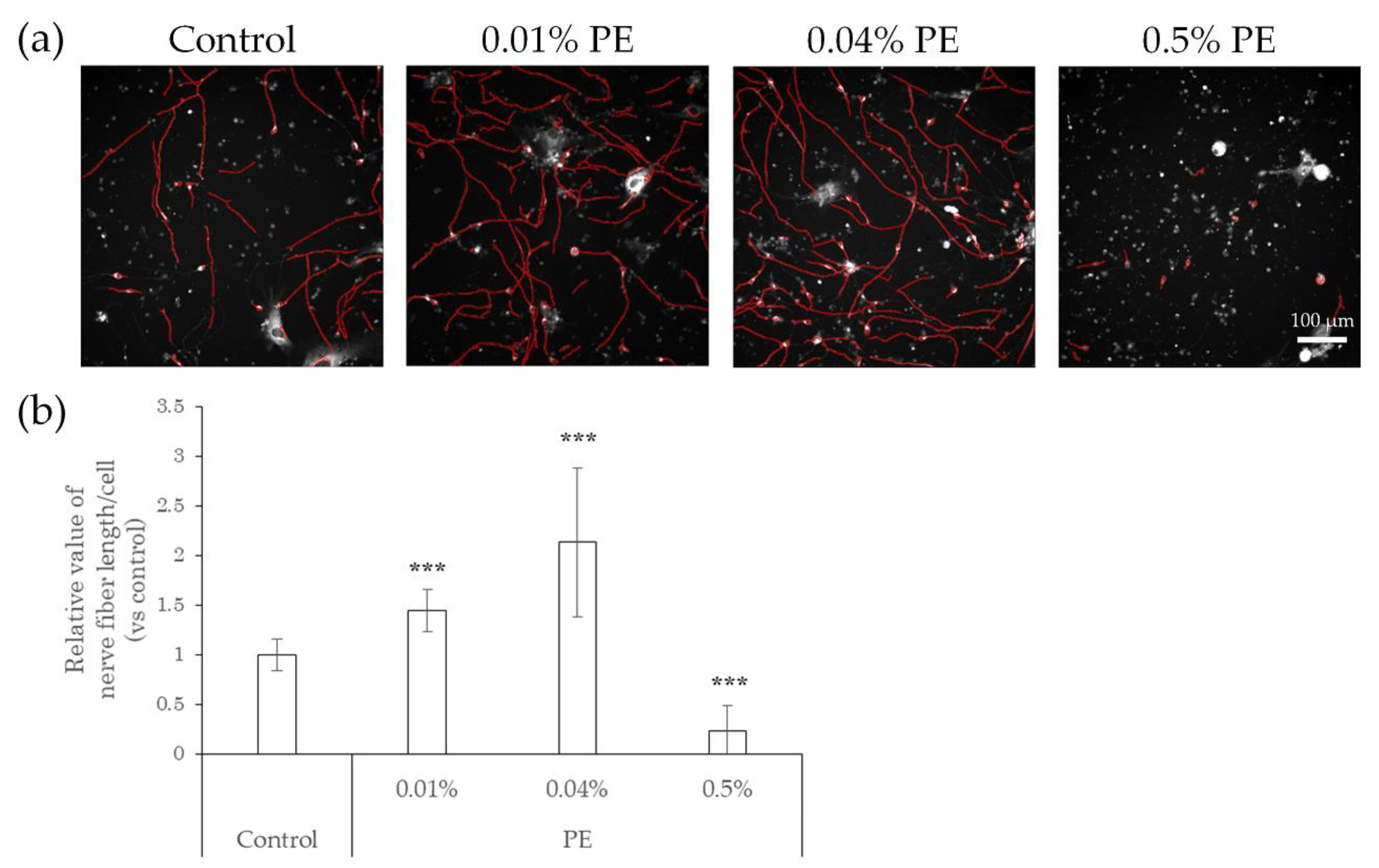
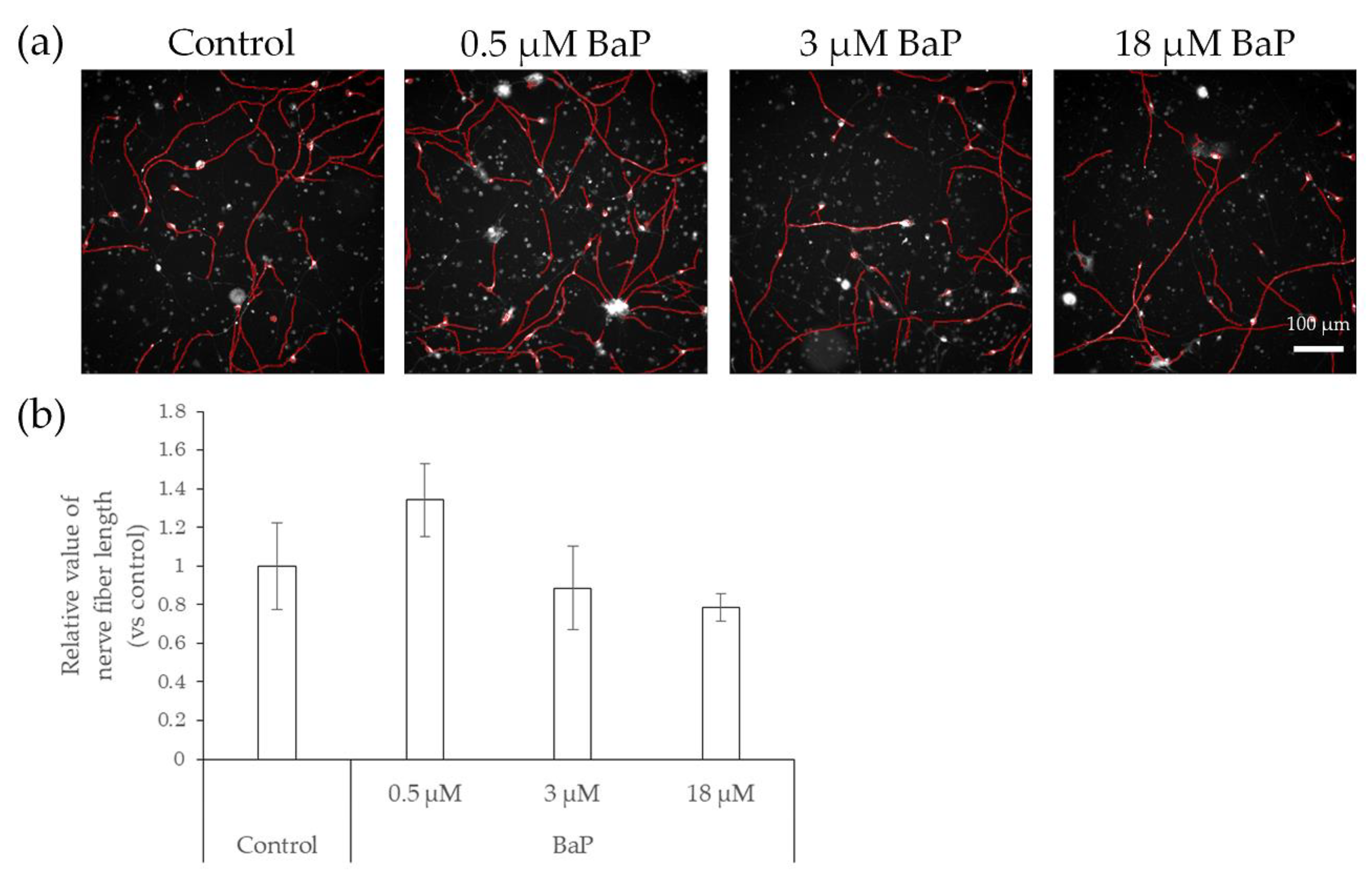
2.3. Elongation of Nerve Fibers in Response to Interaction with Preservatives and Air Pollutant
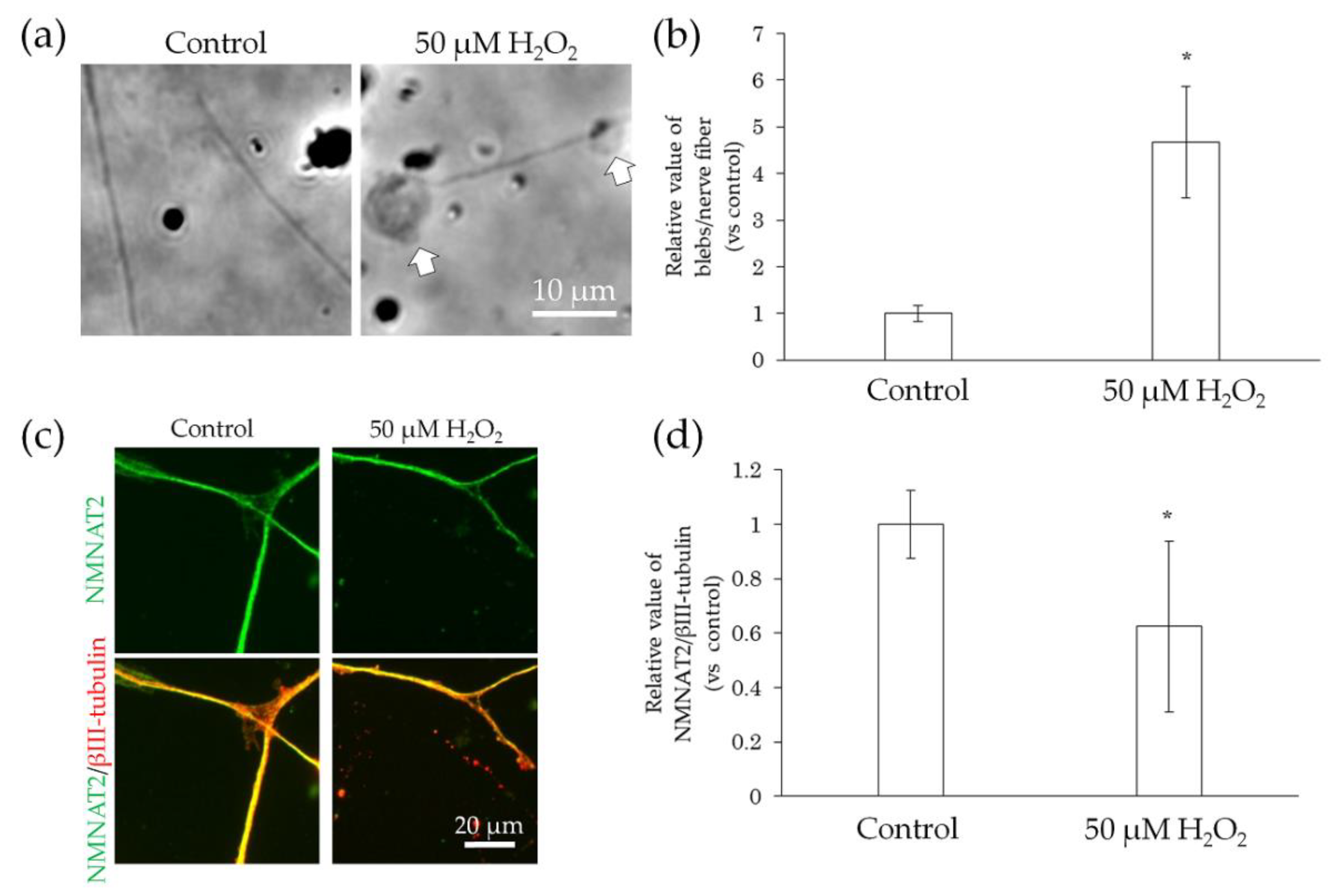
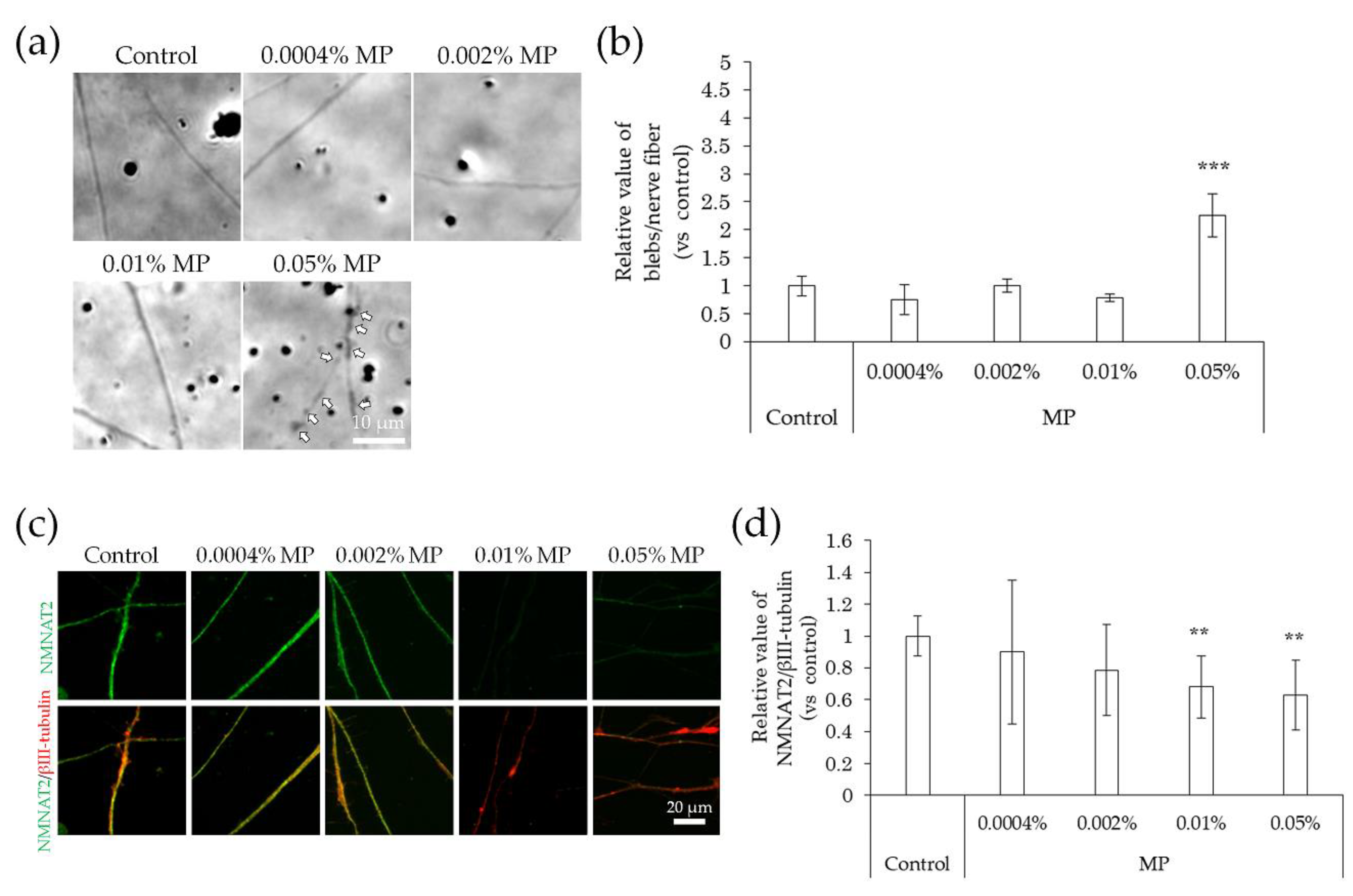
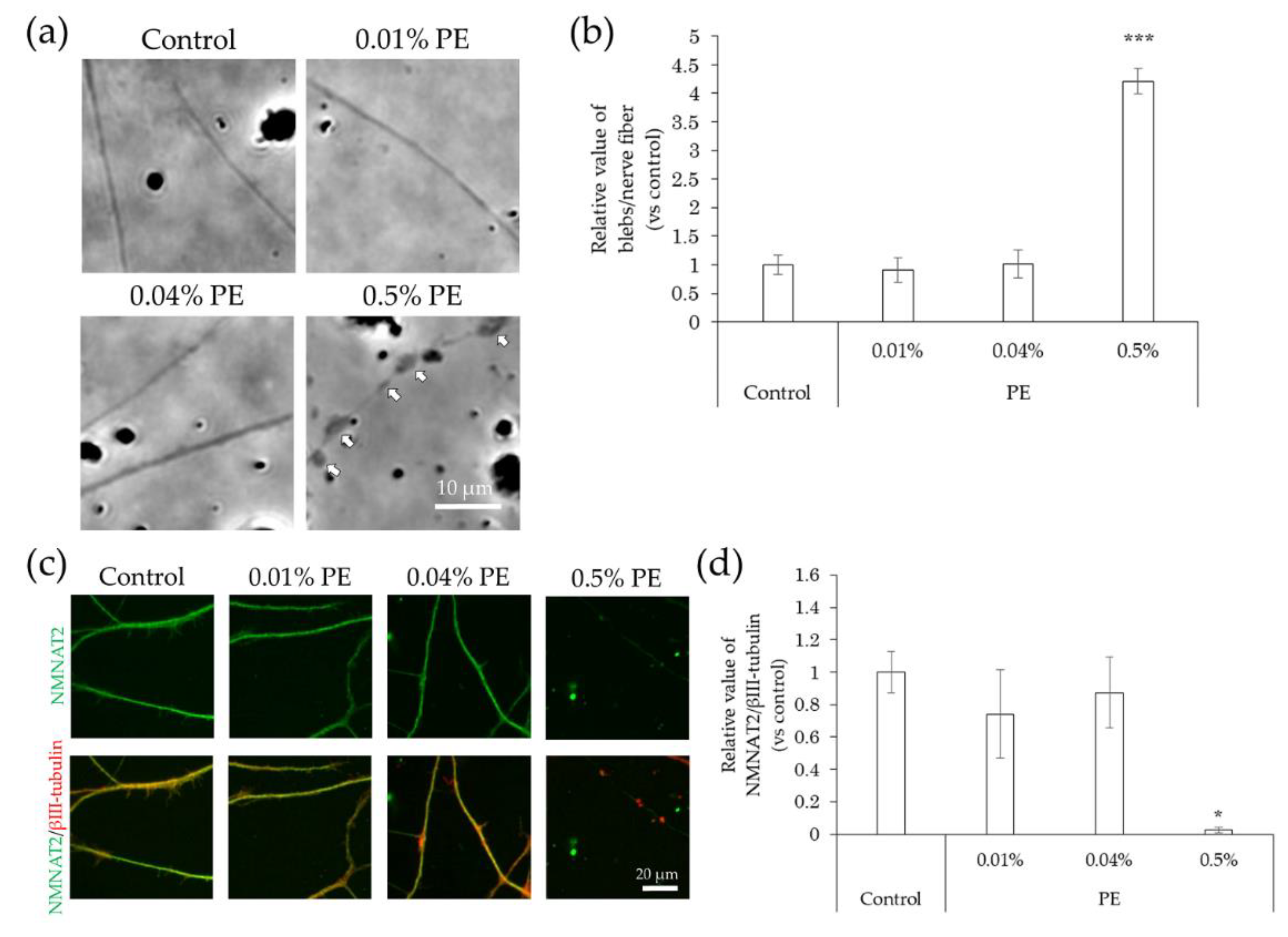
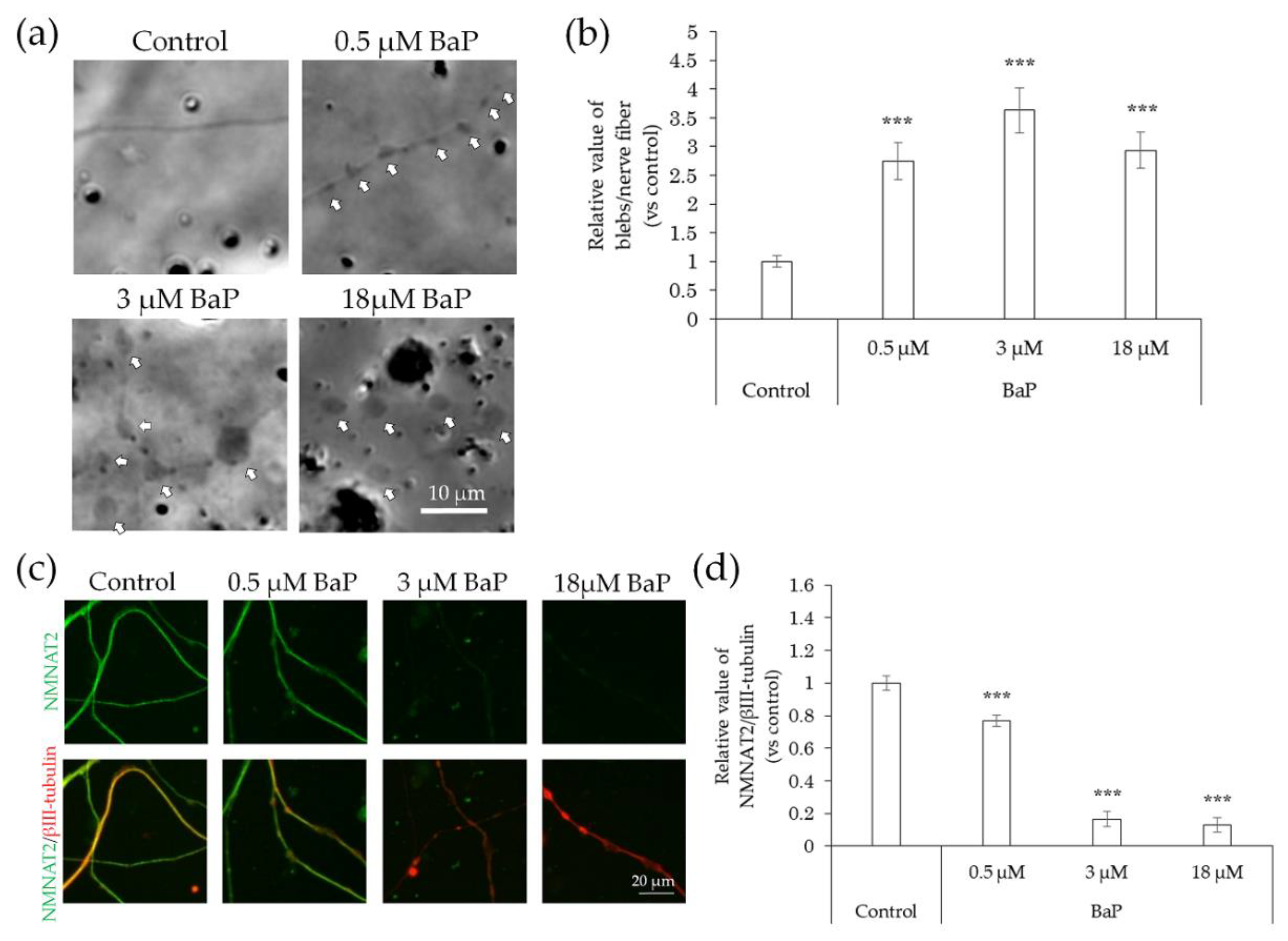
2.4. Neurodegeneration Due to Interaction with Substances
3. Discussion
4. Materials and Methods
4.1. Culture of hiPSC-SNs
4.2. Immunocy to Chemistry
4.3. Quantitative Evaluation of Neuronal Differentiation
4.4. Quantitative Evaluation of Nerve Degeneration
4.5. Quantitative Analyses of Nerve Fiber Length
4.6. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Talagas, M.; Misery, L. Role of keratinocytes in sensitive skin. Front. Med. 2019, 6, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Farage, M.A. Perceptions of sensitive skin: Changes in perceived severity and associations with environmental causes. Contact Dermat. 2008, 59, 226–232. [Google Scholar] [CrossRef]
- Farage, M.A. How do perceptions of sensitive skin differ at different anatomical sites? An epidemiological study. Clin. Exp. Dermatol. 2009, 34, e521–e530. [Google Scholar] [CrossRef]
- Yokota, Y. Classification of sensitive skin with analysis of skin physiological parameter. Nippon. Koshohin Kagakkaishi. 2005, 29, 44–49. [Google Scholar]
- Buhé, V.; Vié, K.; Guéré, C.; Natalizio, A.; Lhéritier, C.; Gall-Ianotto, C.L.; Huet, F.; Talagas, M.; Lebonvallet, N.; Marcorelles, P.; et al. Pathophysiological study of sensitive skin. Acta Derm.-Venereol. 2016, 96, 314–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saint-Martory, C.; Roguedas-Contios, A.M.; Sibaud, V.; Degouy, A.; Schmitt, A.M.; Misery, L. Sensitive skin is not limited to the face. Br. J. Dermatol. 2008, 158, 130–133. [Google Scholar] [CrossRef]
- Kabashima, K. New concept of the pathogenesis of atopic dermatitis: Interplay among the barrier, allergy, and pruritus as a trinity. J. Dermatol. Sci. 2013, 70, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Takamori, K. Itch and nerve fibers with special reference to atopic dermatitis: Therapeutic implications. J. Dermatol. 2014, 41, 205–212. [Google Scholar] [CrossRef]
- Bray, E.R.; Chéret, J.; Yosipovitch, G.; Paus, R. Schwann cells as underestimated, major players in human skin physiology and pathology. Exp. Dermatol. 2020, 29, 93–101. [Google Scholar] [CrossRef] [Green Version]
- Patil, M.J.; Salas, M.; Bialuhin, S.; Boyd, J.T.; Jeske, N.A.; Akopian, A.N. Sensitization of small-diameter sensory neurons is controlled by TRPV1 and TRPA1 association. FASEB J. 2020, 34, 287–302. [Google Scholar] [CrossRef] [Green Version]
- Bianchi, B.R.; Zhang, X.F.; Reilly, R.M.; Kym, P.R.; Yao, B.B.; Chen, J. Species comparison and pharmacological characterization of human, monkey, rat, and mouse TRPA1 channels. J. Pharmacol. Exp. Ther. 2012, 341, 360–368. [Google Scholar] [CrossRef] [Green Version]
- Markus, A.; Grigoryan, S.; Sloutskin, A.; Yee, M.B.; Zhu, H.; Yang, I.H.; Thakor, N.V.; Sarid, R.; Kinchington, P.R.; Goldstein, R.S. Varicella-zoster virus (VZV) infection of neurons derived from human embryonic stem cells: Direct demonstration of axonal infection, transport of VZV, and productive neuronal infection. J. Virol. 2011, 13, 6220–6233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pomp, O.; Brokhman, I.; Ziegler, L.; Almog, M.; Korngreen, A.; Tavian, M.; Goldstein, R.S. PA6-induced human embryonic stem cell-derived neurospheres: A new source of human peripheral sensory neurons and neural crest cells. Brain Res. 2008, 1230, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Valensi-Kurtz, M.; Lefler, S.; Cohen, M.A.; Aharonowiz, M.R.; Cohen-Kupiec, R.; Sheinin, A.; Ashery, U.; Reubinoff, B.; Weil, M. Enriched population of PNS neurons derived from human embryonic stem cells as a platform for studying peripheral neuropathies. PLoS ONE 2010, 5, e9290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brokhman, I.; Gamarnik-Ziegler, L.; Pomp, O.; Aharonowiz, M.; Reubinoff, B.E.; Goldstein, R.S. Peripheral sensory neurons differentiate from neural precursors derived from human embryonic stem cells. Differentiation 2008, 76, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Takamori, K. Recent advances in pathophysiological mechanisms of itch. Expert Rev. Dermatol. 2010, 5, 197–212. [Google Scholar] [CrossRef]
- Er, J.C.; Leong, C.; Teoh, C.L.; Yuan, Q.; Merchant, P.; Dunn, M.; Sulzer, D.; Sames, D.; Bhinge, A.; Kim, D.; et al. NeuO: A fluorescent chemical probe for live neuron labeling. Angew. Chem. Int. Ed. Engl. 2015, 54, 2442–2446. [Google Scholar] [CrossRef]
- Ishiwatari, S.; Suzuki, T.; Hitomi, T.; Yoshino, T.; Matsukuma, S.; Tsuji, T. Effects of methyl paraben on skin keratinocytes. J. Appl. Toxicol. 2007, 27, 1–9. [Google Scholar] [CrossRef]
- McClean, M.D.; Rinehart, R.D.; Sapkota, A.; Cavallari, J.M.; Herrick, R.F. Dermal exposure and urinary 1-hydroxypyrene among asphalt roofing workers. J. Occup. Environ. Hyg. 2007, 4, 118–126. [Google Scholar] [CrossRef]
- Bourgart, E.; Barbeau, D.; Marques, M.; Koschembahr, A.; Béal, D.; Persoons, R.; Leccia, M.; Douki, T.; Maitre, A. A realistic human skin model to study benzo[a]pyrene cutaneous absorption in order to determine the most relevant biomarker for carcinogenic exposure. Arch. Toxicol. 2019, 93, 81–93. [Google Scholar] [CrossRef]
- Sugimura, K. Acute intermittent porphyria. Nippon Rinsho 1995, 53, 1418–1421. [Google Scholar]
- Obrosova, I.G. Diabetic painful and insensate neuropathy: Pathogenesis and potential treatments. Neurotherapeutics 2009, 6, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Kamo, A.; Tominaga, M.; Kamata, Y.; Kaneda, K.; Ko, K.C.; Matsuda, H.; Kimura, U.; Ogawa, H.; Takamori, K. The excimer lamp induces cutaneous nerve degeneration and reduces scratching in a dry-skin mouse model. J. Investig. Dermatol. 2014, 134, 2977–2984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coleman, M. Axon degeneration mechanisms: Commonality amid diversity. Nat. Rev. Neurosci. 2005, 6, 889–898. [Google Scholar] [CrossRef]
- Wang, J.T.; Medress, Z.A.; Barres, B.A. Axon degeneration: Molecular mechanisms of a self-destruction pathway. J. Cell Biol. 2012, 196, 7–18. [Google Scholar] [CrossRef] [Green Version]
- Fukui, K.; Ushiki, K.; Takatsu, H.; Koike, T.; Urano, S. Tocotrienols prevent hydrogen peroxide-induced axon and dendrite degeneration in cerebellar granule cells. Free Radic. Res. 2012, 46, 184–193. [Google Scholar] [CrossRef]
- Sone, T.; Yamada, H.; Endo, H. Pharmacological studies of stinging caused by parabens. J. Jpn. Cosmet. Sci. Soc. 1990, 14, 8–16. [Google Scholar]
- Li, D.-G.; Du, H.-Y.; Gerhard, S.; Imke, M.; Liu, W. Inhibition of TRPV1 prevented skin irritancy induced by phenoxyethanol. A preliminary in vitro and in vivo study. Int. J. Cosmet. Sci. 2017, 39, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Nakazawa, K.; Inoue, K.; Fujimori, K.; Ohno, Y.; Takanaka, A.; Itagaki, H.; Kato, S.; Kobayashi, T.; Kuroiwa, Y. Evaluation of stinging-inducing chemicals using cultured neuronal cells: An electrophysiological approach. Toxicol. In Vitro 1996, 10, 455–462. [Google Scholar] [CrossRef]
- Wilkinson, J.D.; Shaw, S.; Andersen, K.E.; Brandao, F.M.; Bruynzeel, D.P.; Bruze, M.; Camarasa, J.M.G.; Diepgen, T.L.; Ducombs, G.; Frosch, P.J.; et al. Monitoring levels of preservative sensitivity in Europe. A 10-year overview (1991–2000). Contact Dermat. 2002, 46, 207–210. [Google Scholar] [CrossRef]
- Sasseville, D. Hypersensitivity to preservatives. Dermatol. Ther. 2004, 17, 251–263. [Google Scholar] [CrossRef]
- Fujita, F.; Moriyama, T.; Higashi, T.; Shima, A.; Tominaga, M. Methyl p-hydroxybenzoate causes pain sensation through activation of TRPA1 channels. Br. J. Pharmacol. 2007, 151, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.-I.; Lin, H.-C.; Lee, H.-T.; Tsai, F.-C.; Lee, T.-S. Loss of transient receptor potential ankyrin 1 channel deregulates emotion, learning and memory, cognition, and social behavior in mice. Mol. Neurobiol. 2017, 54, 3606–3617. [Google Scholar] [CrossRef]
- Caterina, M.J.; Pang, Z. TRP channels in skin biology and pathophysiology. Pharmaceuticals 2016, 9, 77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, C.; Catania, S.; Pasquale, R.D.; Stancanelli, R.; Scribano, G.M.; Melchini, A. Exposure of human skin to benzo[a]pyrene: Role of CYP1A1 and aryl hydrocarbon receptor in oxidative stress generation. Toxicology 2010, 271, 83–86. [Google Scholar] [CrossRef]
- Podechard, N.; Lecureur, V.; Ferrec, E.L.; Guenon, I.; Sparfel, L.; Gilot, D.; Gordon, J.R.; Lagente, V.; Fardel, O. Interleukin-8 induction by the environmental contaminant benzo(a)pyrene is aryl hydrocarbon receptor-dependent and leads to lung inflammation. Toxicol. Lett. 2008, 177, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Basbaum, A.I.; Bautista, D.M.; Scherrer, G.; Julius, D. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, M.-C.; Yoshimura, M. Effects of benzo[a]pyrene on conduction velocity of the rat dorsal root and synaptic transmission in substantia gelatinosa (SG) neurons of the rat spinal dorsal horn. Fukuoka Igaku Zasshi 2017, 108, 75–82. [Google Scholar]
- Akahane, M.; Matsumoto, S.; Kanagawa, Y.; Mitoma, C.; Uchi, H.; Yoshimura, T.; Furue, M.; Imamura, T. Long-term health effects of PCBs and related compounds: A comparative analysis of patients suffering from Yusho and the general population. Arch. Environ. Contam. Toxicol. 2018, 74, 203–217. [Google Scholar] [CrossRef]
- More, S.V.; Koppula, S.; Kim, I.-S.; Kumar, H.; Kim, B.-W.; Choi, D.-K. The role of bioactive compounds on the promotion of neurite outgrowth. Molecules 2012, 17, 6728–6753. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Satoh, M.; Suzuki, T.; Sakurai, T.; Toyama, S.; Kamata, Y.; Kondo, S.; Suga, Y.; Tominaga, M.; Takamori, K. A Novel In Vitro Assay Using Human iPSC-Derived Sensory Neurons to Evaluate the Effects of External Chemicals on Neuronal Morphology: Possible Implications in the Prediction of Abnormal Skin Sensation. Int. J. Mol. Sci. 2021, 22, 10525. https://doi.org/10.3390/ijms221910525
Satoh M, Suzuki T, Sakurai T, Toyama S, Kamata Y, Kondo S, Suga Y, Tominaga M, Takamori K. A Novel In Vitro Assay Using Human iPSC-Derived Sensory Neurons to Evaluate the Effects of External Chemicals on Neuronal Morphology: Possible Implications in the Prediction of Abnormal Skin Sensation. International Journal of Molecular Sciences. 2021; 22(19):10525. https://doi.org/10.3390/ijms221910525
Chicago/Turabian StyleSatoh, Masahiko, Tamie Suzuki, Tetsuhito Sakurai, Sumika Toyama, Yayoi Kamata, Shinya Kondo, Yasushi Suga, Mitsutoshi Tominaga, and Kenji Takamori. 2021. "A Novel In Vitro Assay Using Human iPSC-Derived Sensory Neurons to Evaluate the Effects of External Chemicals on Neuronal Morphology: Possible Implications in the Prediction of Abnormal Skin Sensation" International Journal of Molecular Sciences 22, no. 19: 10525. https://doi.org/10.3390/ijms221910525
APA StyleSatoh, M., Suzuki, T., Sakurai, T., Toyama, S., Kamata, Y., Kondo, S., Suga, Y., Tominaga, M., & Takamori, K. (2021). A Novel In Vitro Assay Using Human iPSC-Derived Sensory Neurons to Evaluate the Effects of External Chemicals on Neuronal Morphology: Possible Implications in the Prediction of Abnormal Skin Sensation. International Journal of Molecular Sciences, 22(19), 10525. https://doi.org/10.3390/ijms221910525





