Highly Porous Fluorapatite/β-1,3-Glucan Composite for Bone Tissue Regeneration: Characterization and In-Vitro Assessment of Biomedical Potential
Abstract
1. Introduction
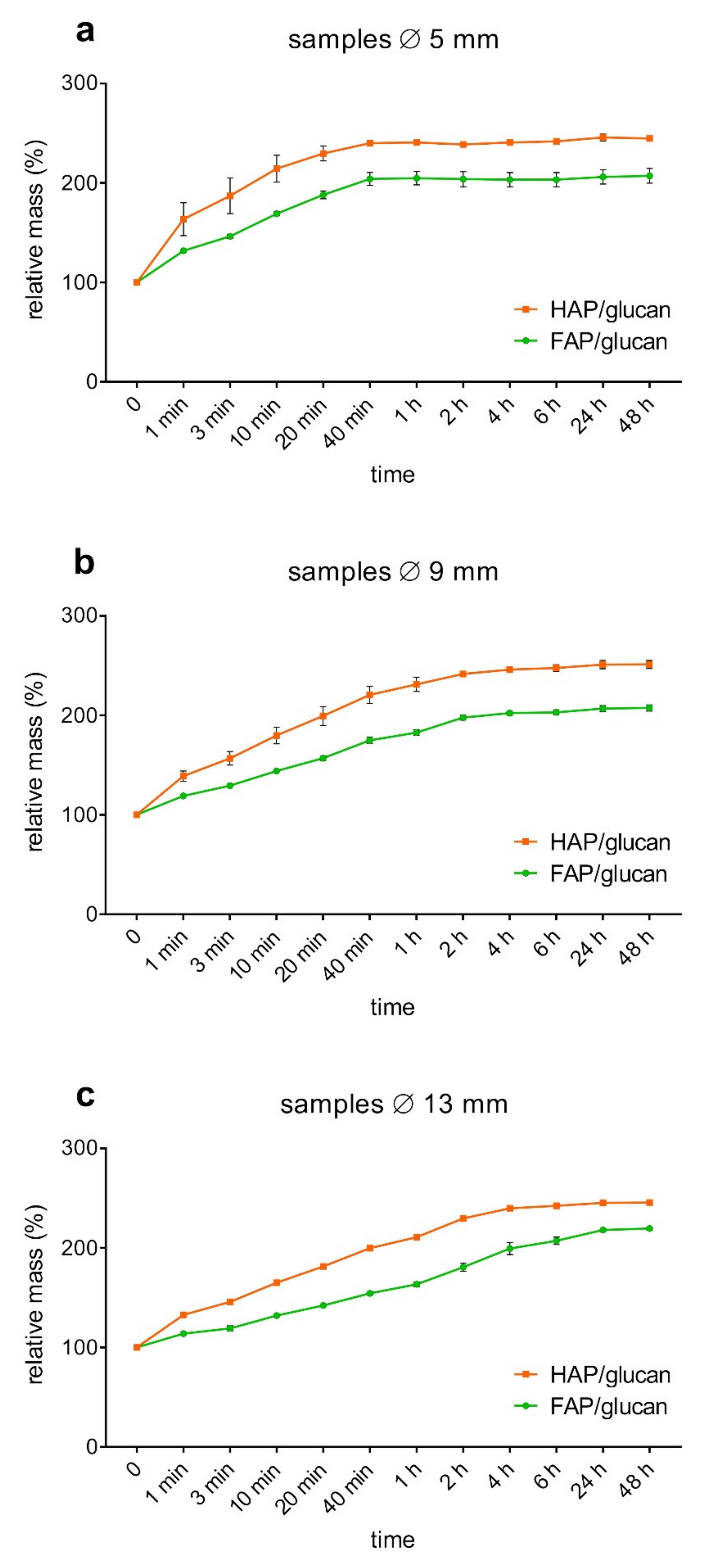
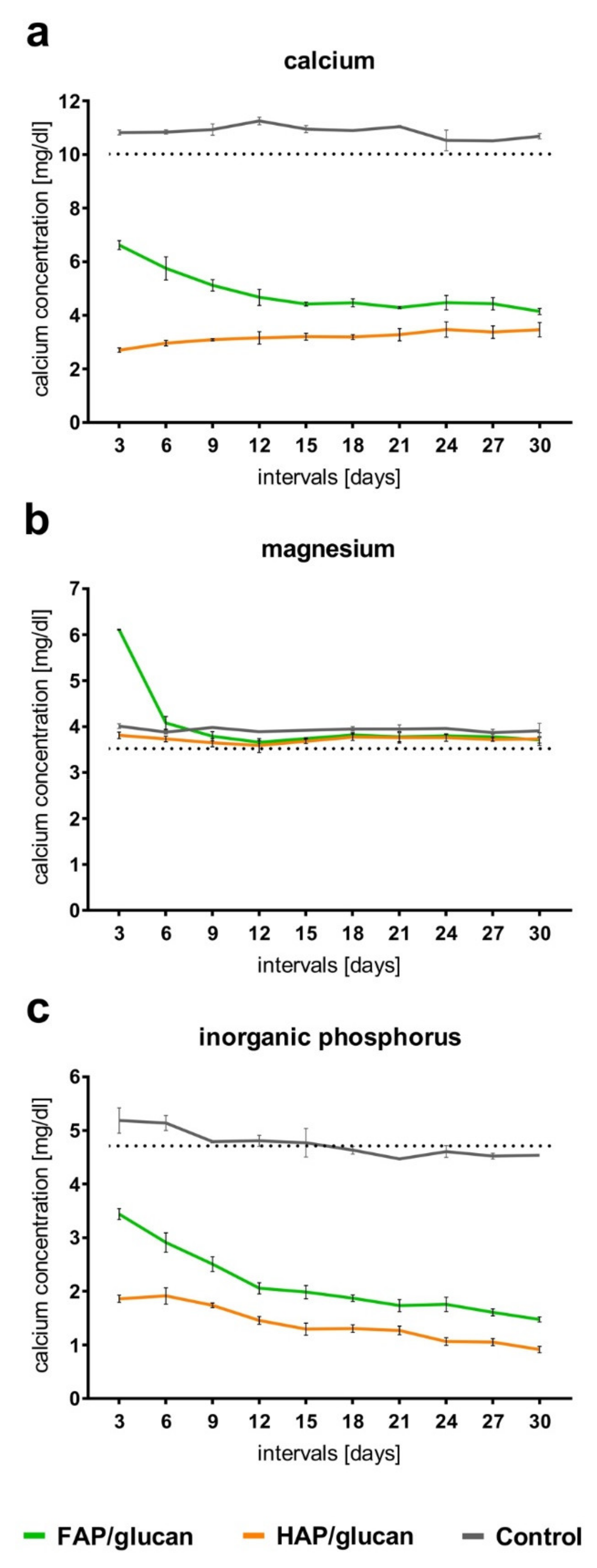
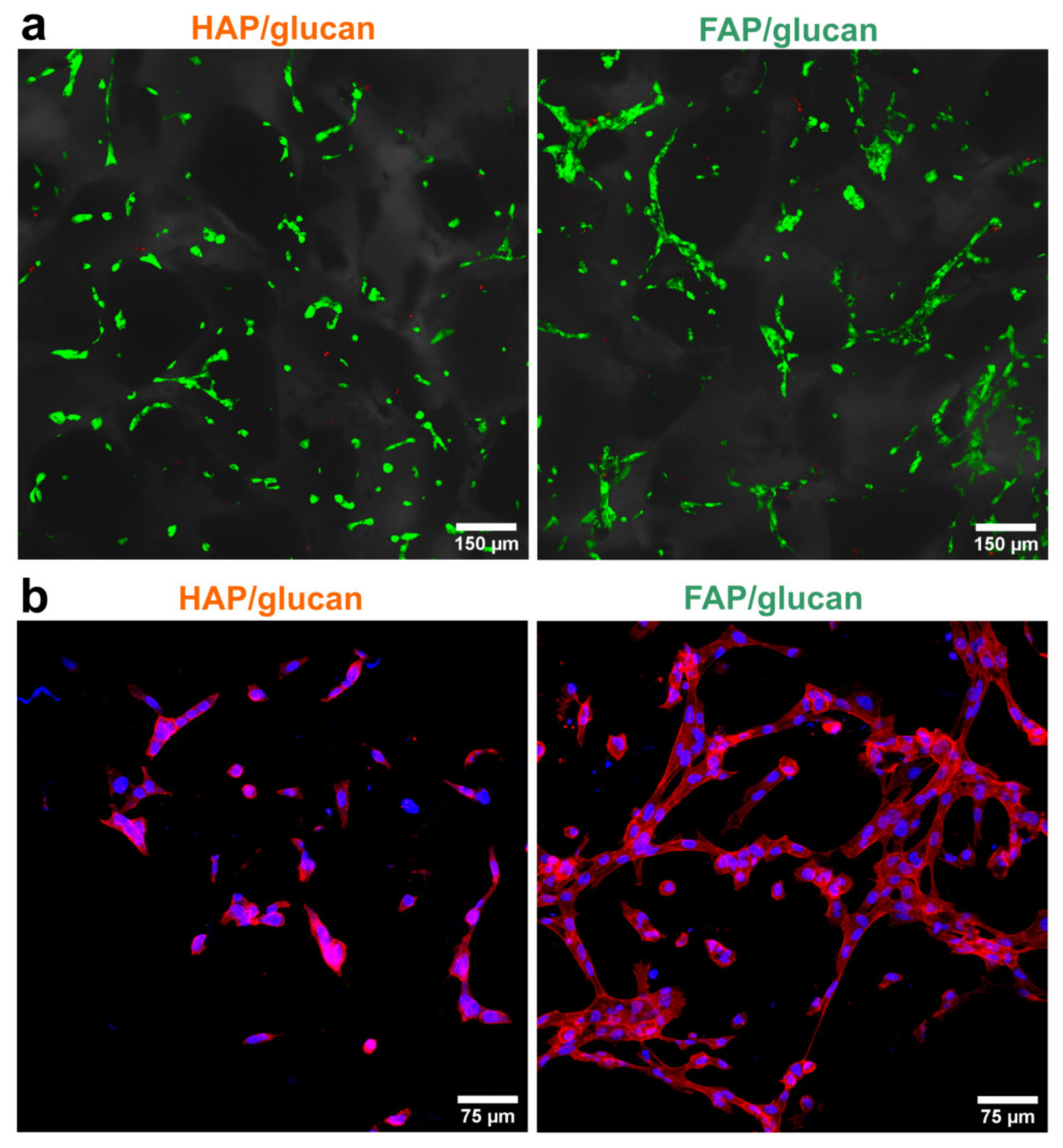
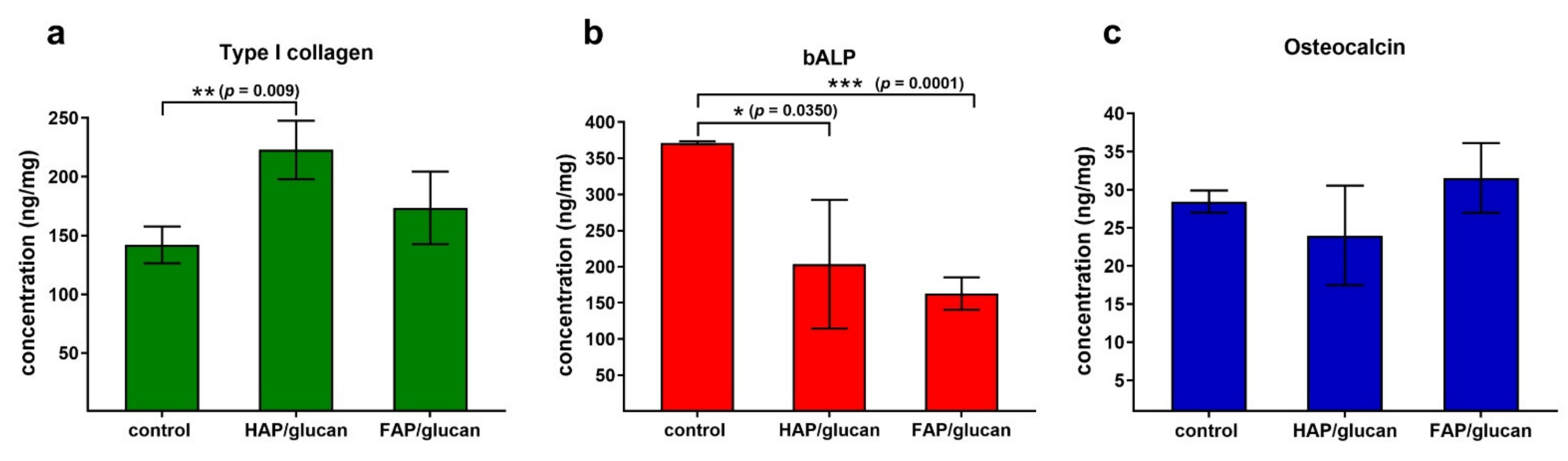
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Preparation of Samples (FAP/glucan and HAP/glucan Composites)
- FAP/glucan
- HAP/glucan
3.3. Characterization of Biomaterials
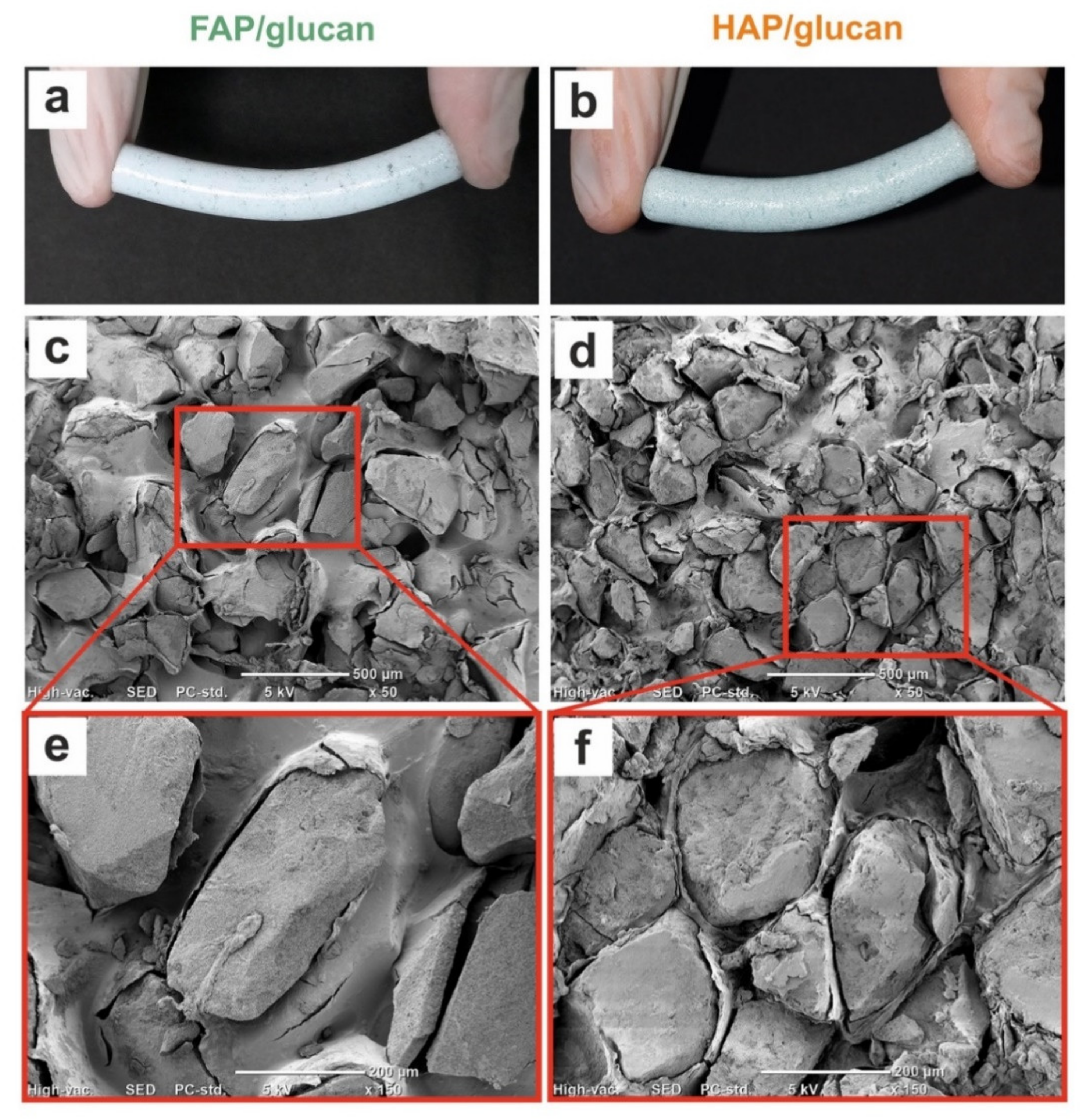
3.3.1. SEM Imaging
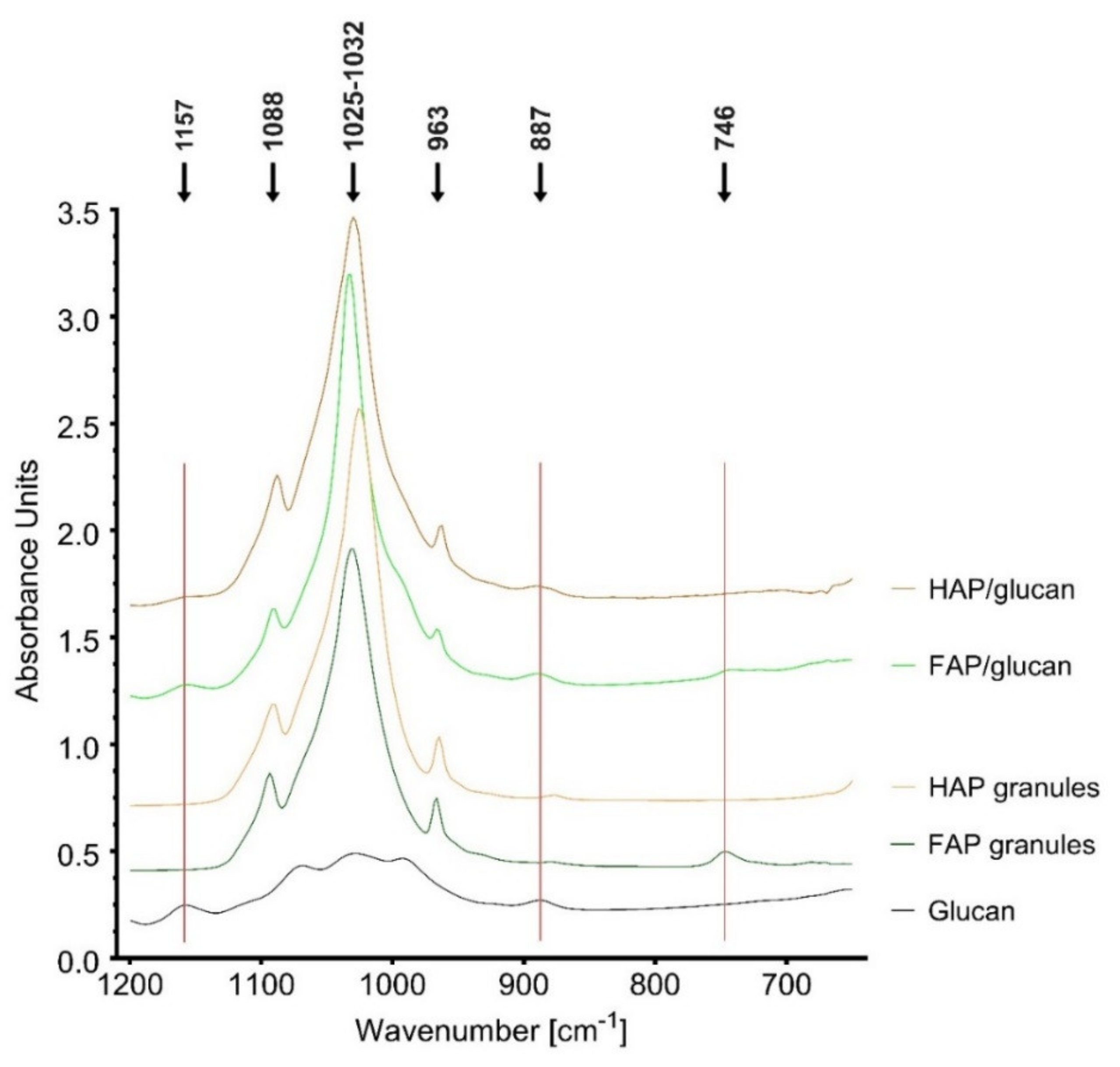
3.3.2. FTIR Analysis
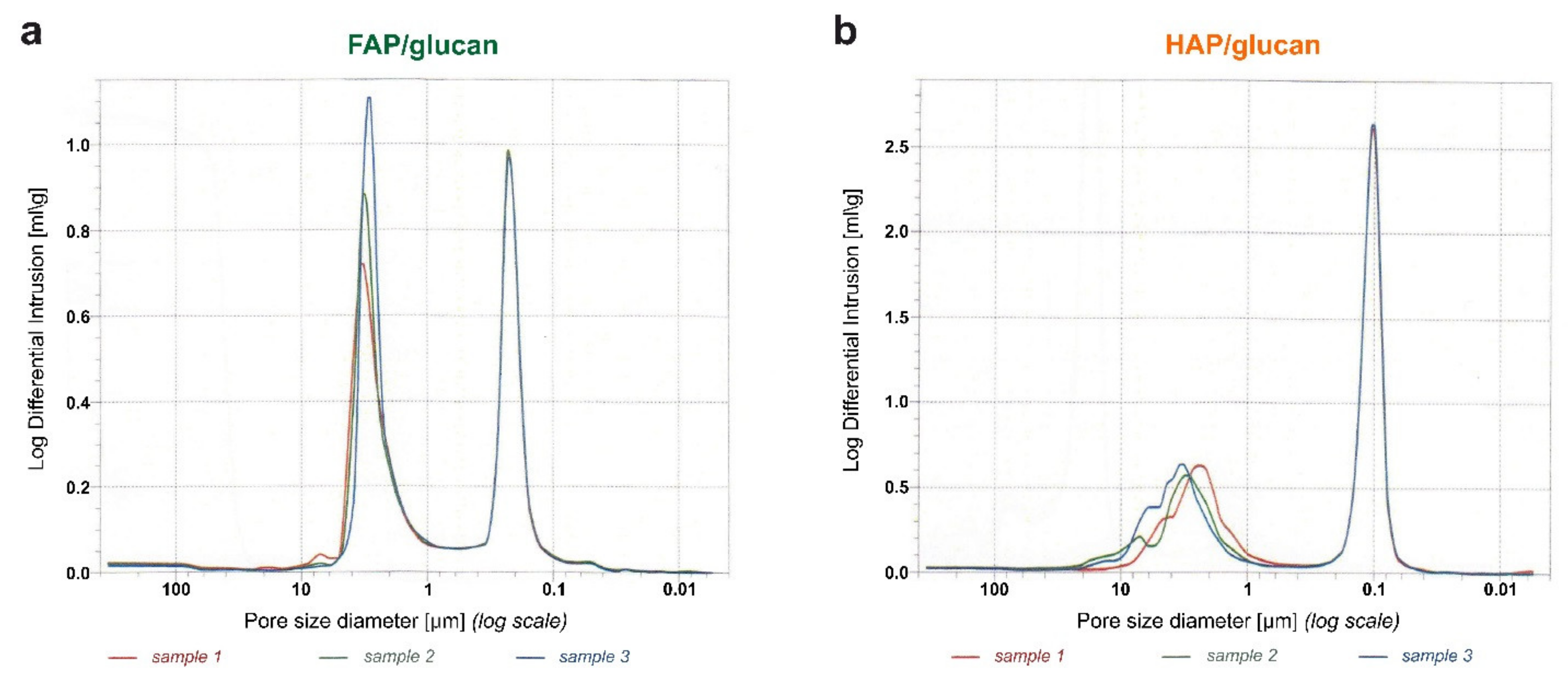
3.3.3. Porosity Determination
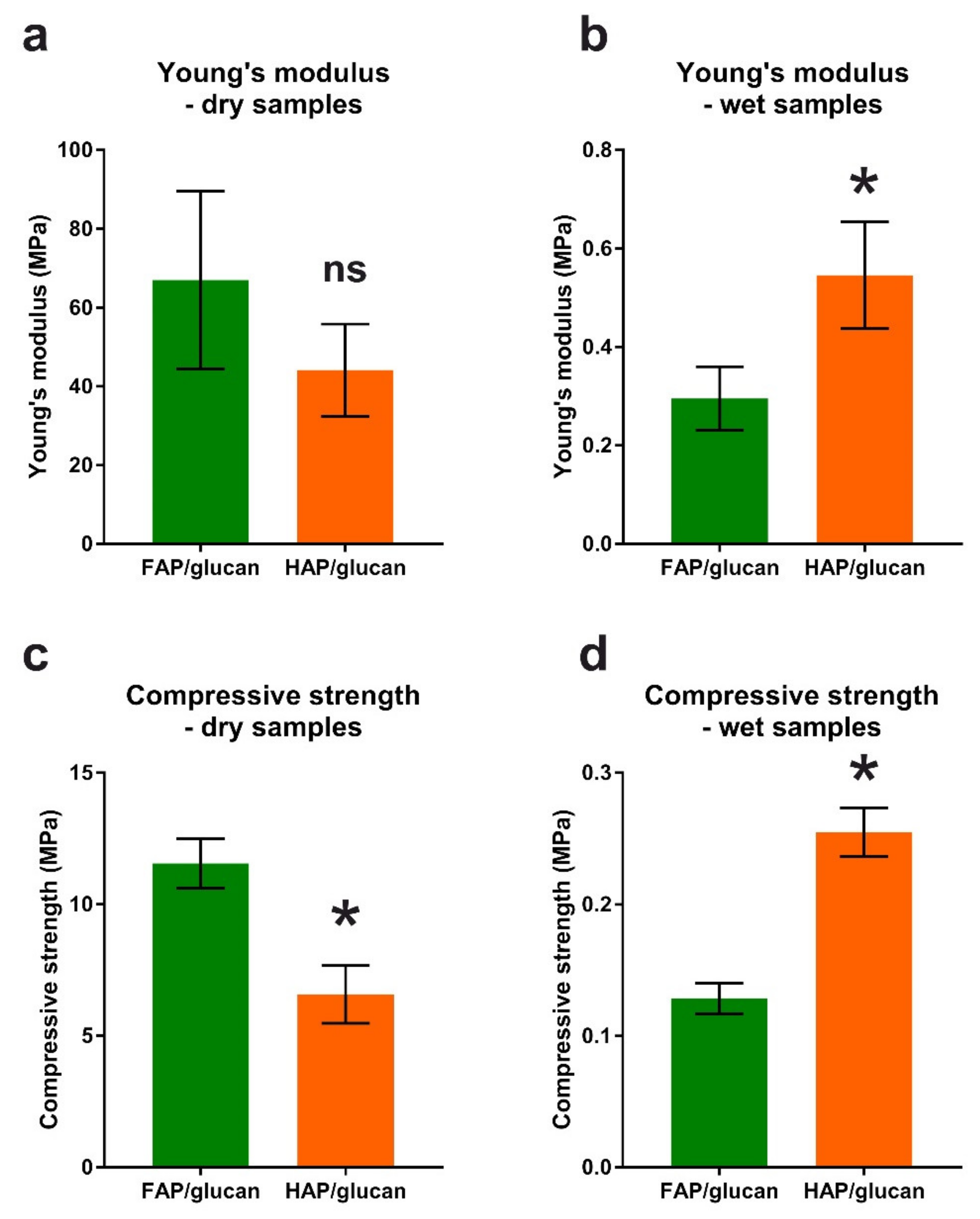
3.3.4. Mechanical Property Evaluation by Compression Test
3.3.5. Liquid Absorption Behavior of Composites
3.3.6. Bioactivity Assessment
3.3.7. Fluoride Ion Release Test
3.4. In-Vitro Cell Culture Experiments
3.4.1. Quantitative Cytotoxicity Assessment (MTT)
3.4.2. Direct-Contact Test for Qualitative Cytotoxicity Assessment (Live-Dead Staining)
3.4.3. Cell Growth Evaluation (Cytoskeleton Imaging—DAPI, Phalloidin)
3.4.4. Osteogenic Differentiation Evaluation (ELISAs)
4. Conclusions
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eliaz, N.; Metoki, N. Calcium Phosphate Bioceramics: A Review of Their History, Structure, Properties, Coating Technologies and Biomedical Applications. Materials 2017, 10, 334. [Google Scholar] [CrossRef] [PubMed]
- Van der Stok, J.; Van Lieshout, E.M.; El-Massoudi, Y.; Van Kralingen, G.H.; Patka, P. Bone substitutes in the Netherlands—A systematic literature review. Acta Biomater. 2010, 7, 739–750. [Google Scholar] [CrossRef] [PubMed]
- Papynov, E.; Shichalin, O.; Apanasevich, V.; Plekhova, N.; Buravlev, I.Y.; Zinoviev, S.; Mayorov, V.Y.; Fedorets, A.; Merkulov, E.; Shlyk, D.K.; et al. Synthetic nanostructured wollastonite: Composition, structure and “in vitro” biocompatibility investigation. Ceram. Int. 2021, 47, 22487–22496. [Google Scholar] [CrossRef]
- Parikh, S.N. Bone graft substitutes: Past, present, future. J. Postgrad. Med. 2002, 48, 142–148. [Google Scholar] [PubMed]
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36, S20–S27. [Google Scholar] [CrossRef]
- Przekora, A.; Ginalska, G. Chitosan/β-1,3-glucan/hydroxyapatite bone scaffold enhances osteogenic differentiation through TNF-α-mediated mechanism. Mater. Sci. Eng. C 2017, 73, 225–233. [Google Scholar] [CrossRef]
- Borkowski, L.; Pawłowska, M.; Radzki, R.; Bieńko, M.; Polkowska, I.; Belcarz, A.; Karpiński, M.; Słowik, T.; Matuszewski, L.; Ślósarczyk, A.; et al. Effect of a carbonated HAP/β-glucan composite bone substitute on healing of drilled bone voids in the proximal tibial metaphysis of rabbits. Mater. Sci. Eng. C 2015, 53, 60–67. [Google Scholar] [CrossRef]
- Klimek, K.; Ginalska, G. Proteins and Peptides as Important Modifiers of the Polymer Scaffolds for Tissue Engineering Applications—A Review. Polymers 2020, 12, 844. [Google Scholar] [CrossRef]
- Zhou, H.; Lawrence, J.G.; Bhaduri, S.B. Fabrication aspects of PLA-CaP/PLGA-CaP composites for orthopedic applications: A review. Acta Biomater. 2012, 8, 1999–2016. [Google Scholar] [CrossRef] [PubMed]
- Prabaharan, M.; Rodriguez-Perez, M.A.; de Saja, J.A.; Mano, J.F. Preparation and characterization of poly(L-lactic acid)-chitosan hybrid scaffolds with drug release capability. J. Biomed. Mater. Res. Part B Appl. Biomater. 2006, 81B, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Macedo, F.A.; Nunes, E.H.M.; Vasconcelos, W.L.; Santos, R.A.; Sinisterra, R.D.; Cortes, M.E. A biodegradable porous composite scaffold of PCL/BCP containing Ang-(1-7) for bone tissue engineering. Cerâmica 2012, 58, 481–488. [Google Scholar] [CrossRef][Green Version]
- Narbat, M.K.; Orang, F.; Hashtjin, M.S.; Goudarzi, A. Fabrication of porous hydroxyapatite-gelatin composite scaffolds for bone tissue engineering. Iran. Biomed. J. 2006, 10, 215–223. [Google Scholar]
- Zhang, Y.; Ni, M.; Zhang, M.; Ratner, B. Calcium Phosphate—Chitosan Composite Scaffolds for Bone Tissue Engineering. Tissue Eng. 2003, 9, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Jouyandeh, M.; Vahabi, H.; Rabiee, N.; Rabiee, M.; Bagherzadeh, M.; Saeb, M.R. Green composites in bone tissue engineering. Emergent Mater 2021, 9, 1–18. [Google Scholar]
- Kus-Liśkiewicz, M.; Rzeszutko, J.; (Bobitski), Y.B.; Barylyak, A.; Nechyporenko, G.; Zinchenko, V.; Zebrowski, J. Alternative Approach for Fighting Bacteria and Fungi: Use of Modified Fluorapatite. J. Biomed. Nanotechnol. 2019, 15, 848–855. [Google Scholar] [CrossRef]
- Tredwin, C.J.; Young, A.M.; Neel, E.A.A.; Georgiou, G.; Knowles, J.C. Hydroxyapatite, fluor-hydroxyapatite and fluorapatite produced via the sol–gel method: Dissolution behaviour and biological properties after crystallisation. J. Mater. Sci. Mater. Electron. 2013, 25, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-C.; Huang, S.-T.; Lin, H.-C.; Tseng, T.-W.; Rao, Q.-L.; Chen, M.-Y. Expression of Osteopontin and Type I Collagen of hFOB 1.19 Cells on Sintered Fluoridated Hydroxyapatite Composite Bone Graft Materials. Implant. Dent. 2010, 19, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Bhadang, K.A.; Gross, K.A. Influence of fluorapatite on the properties of thermally sprayed hydroxyapatite coatings. Biomaterials 2004, 25, 4935–4945. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.; Melville, A.; Forsythe, J.; Muddle, B.; Trounson, A.; Gross, K.; Mollard, R. Sintered hydroxyfluorapatites—IV: The effect of fluoride substitutions upon colonisation of hydroxyapatites by mouse embryonic stem cells. Biomaterials 2004, 25, 4977–4986. [Google Scholar] [CrossRef] [PubMed]
- Gentleman, E.; Stevens, M.; Hill, R.; Brauer, D. Surface properties and ion release from fluoride-containing bioactive glasses promote osteoblast differentiation and mineralization in vitro. Acta Biomater. 2012, 9, 5771–5779. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, B.; Mai, S.; Wu, X.; Zhang, H.; Qiao, W.; Luo, X.; Chen, Z. Effects of fluoridation of porcine hydroxyapatite on osteoblastic activity of human MG63 cells. Sci. Technol. Adv. Mater. 2015, 16, 035006. [Google Scholar] [CrossRef]
- Shah, F.A.; Brauer, D.S.; Wilson, R.M.; Hill, R.G.; Hing, K.A. Influence of cell culture medium composition onin vitrodissolution behavior of a fluoride-containing bioactive glass. J. Biomed. Mater. Res. Part A 2013, 102, 647–654. [Google Scholar] [CrossRef]
- Barandehfard, F.; Rad, M.K.; Hosseinnia, A.; Khoshroo, K.; Tahriri, M.; Jazayeri, H.; Moharamzadeh, K.; Tayebi, L. The addition of synthesized hydroxyapatite and fluorapatite nanoparticles to a glass-ionomer cement for dental restoration and its effects on mechanical properties. Ceram. Int. 2016, 42, 17866–17875. [Google Scholar] [CrossRef]
- Kim, H.-W.; Lee, S.Y.; Bae, C.-J.; Noh, Y.-J.; Kim, H.-E.; Kim, H.-M.; Ko, J.S. Porous ZrO2 bone scaffold coated with hydroxyapatite with fluorapatite intermediate layer. Biomaterials 2003, 24, 3277–3284. [Google Scholar] [CrossRef]
- Tredwin, C.J.; Georgiou, G.; Kim, H.-W.; Knowles, J.C. Hydroxyapatite, fluor-hydroxyapatite and fluorapatite produced via the sol–gel method: Bonding to titanium and scanning electron microscopy. Dent. Mater. 2013, 29, 521–529. [Google Scholar] [CrossRef]
- Wang, J.; Chao, Y.; Wan, Q.; Zhu, Z.; Yu, H. Fluoridated hydroxyapatite coatings on titanium obtained by electrochemical deposition. Acta Biomater. 2009, 5, 1798–1807. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-W.; Kim, H.-E.; Knowles, J.C. Fluor-hydroxyapatite sol–gel coating on titanium substrate for hard tissue implants. Biomaterials 2004, 25, 3351–3358. [Google Scholar] [CrossRef] [PubMed]
- Gineste, L.; Gineste, M.; Ranz, X.; Ellefterion, A.; Guilhem, A.; Rouquet, N.; Frayssinet, P. Degradation of hydroxylapatite, fluorapatite, and fluorhydroxyapatite coatings of dental implants in dogs. J. Biomed. Mater. Res. 1999, 48, 224–234. [Google Scholar] [CrossRef]
- Dhert, W.J.A.; Klein, C.P.A.T.; Jansen, J.A.; Van Der Velde, E.A.; Vriesde, R.C.; Rozing, P.M.; De Groot, K. A histological and histomorphometrical investigation of fluorapatite, magnesiumwhitlockite, and hydroxylapatite plasma-sprayed coatings in goats. J. Biomed. Mater. Res. 1993, 27, 127–138. [Google Scholar] [CrossRef]
- Borkowski, L.; Belcarz, A.; Przekora, A.; Ginalska, G. Production Method for Biocompatible Implant Material. Polish Patent no. 235803, 6 October 2020. [Google Scholar]
- Borkowski, L.; Przekora, A.; Belcarz, A.; Palka, K.; Jozefaciuk, G.; Lübek, T.; Jojczuk, M.; Nogalski, A.; Ginalska, G. Fluorapatite ceramics for bone tissue regeneration: Synthesis, characterization and assessment of biomedical potential. Mater. Sci. Eng. C 2020, 116, 111211. [Google Scholar] [CrossRef]
- Borkowski, L.; Belcarz, A.; Przekora, A.; Ginalska, G. Method of Obtaining Bone Scaffold based on Fluoroapatite Ceramics and Polymer and Bone Scaffold. Polish Patent no. 236369, 29 January 2021. [Google Scholar]
- Wahl, D.; Czernuszka, J. Collagen-Hydroxyapatite Composites for Hard Tissue Repair. Eur. Cells Mater. 2006, 11, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Antebi, B.; Cheng, X.; Harris, J.N.; Gower, L.B.; Chen, X.-D.; Ling, J. Biomimetic Collagen–Hydroxyapatite Composite Fabricated via a Novel Perfusion-Flow Mineralization Technique. Tissue Eng. Part C Methods 2013, 19, 487–496. [Google Scholar] [CrossRef]
- Pighinelli, L.; Kucharska, M. Chitosan–hydroxyapatite composites. Carbohydr. Polym. 2013, 93, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Sancilio, S.; Gallorini, M.; Di Nisio, C.; Marsich, E.; Di Pietro, R.; Schweikl, H.; Cataldi, A. Alginate/Hydroxyapatite-Based Nanocomposite Scaffolds for Bone Tissue Engineering Improve Dental Pulp Biomineralization and Differentiation. Stem Cells Int. 2018, 2018, 9643721. [Google Scholar] [CrossRef]
- Belcarz, A.; Ginalska, G.; Pycka, T.; Zima, A.; Ślósarczyk, A.; Polkowska, I.; Paszkiewicz, Z.; Piekarczyk, W. Application of β-1,3-glucan in production of ceramics-based elastic composite for bone repair. Open Life Sci. 2013, 8, 534–548. [Google Scholar] [CrossRef]
- Gross, K.A.; Bhadang, K.A. Sintered hydroxyfluorapatites. Part III: Sintering and resultant mechanical properties of sintered blends of hydroxyapatite and fluorapatite. Biomaterials 2003, 25, 1395–1405. [Google Scholar] [CrossRef] [PubMed]
- Gross, K.A.; Rodríguez-Lorenzo, L.M. Sintered hydroxyfluorapatites. Part I: Sintering ability of precipitated solid solution powders. Biomaterials 2004, 25, 1375–1384. [Google Scholar] [CrossRef]
- Gao, Y.; Karpukhina, N.; Law, R.V. Phase segregation in hydroxyfluorapatite solid solution at high temperatures studied by combined XRD/solid state NMR. RSC Adv. 2016, 6, 103782–103790. [Google Scholar] [CrossRef]
- Smyth, M.; M’Bengue, M.-S.; Terrien, M.; Picart, C.; Bras, J.; Foster, E.J. The effect of hydration on the material and mechanical properties of cellulose nanocrystal-alginate composites. Carbohydr. Polym. 2018, 179, 186–195. [Google Scholar] [CrossRef]
- Misch, C.E.; Qu, Z.; Bidez, M.W. Mechanical properties of trabecular bone in the human mandible: Implications for dental implant treatment planning and surgical placement. J. Oral Maxillofac. Surg. 1999, 57, 700–706. [Google Scholar] [CrossRef]
- Borkowski, L.; Lübek, T.; Jojczuk, M.; Nogalski, A.; Belcarz, A.; Palka, K.; Hajnos, M.; Ginalska, G. Behavior of new hydroxyapatite/glucan composite in human serum. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 2653–2664. [Google Scholar] [CrossRef] [PubMed]
- Przekora-Kuśmierz, A.; Ginalska, G. Enhanced differentiation of osteoblastic cells on novel chitosan/β-1,3-glucan/bioceramic scaffolds for bone tissue regeneration. Biomed. Mater. 2015, 10, 015009. [Google Scholar] [CrossRef] [PubMed]
- Slosarczyk, A.; Paszkiewicz, Z.; Zima, A. Method for Fabrication of Highly Porous, Calcium Phosphate Bioactive Implant Ma-terial. European Patent no. EP 2229961 A2, 12 March 2014. [Google Scholar]
- Belcarz, A.; Ginalska, G.; Slosarczyk, A.; Paszkiewicz, Z. Bioactive Composite and Process for the Production of the Bioactive Composite. European Patent no. EP 2421570 B1, 25 February 2015. [Google Scholar]
- Standard ISO 15901-1:2005. Pore Size Distribution and Porosity of Solid Materials by Mercury Porosimetry and Gas Adsorption; International Organization for Standardization: Geneva, Switzerland, 2005. [Google Scholar]
- Kokubo, T.; Kushitani, H.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Solutions able to reproducein vivo surface-structure changes in bioactive glass-ceramic A-W3. J. Biomed. Mater. Res. 1990, 24, 721–734. [Google Scholar] [CrossRef] [PubMed]
- Standard ISO 10993-5:2009. Biological Evaluation of Medical Devices—Part5: Tests for In Vitro Cytotoxicity; International Organization for Standardization: Geneva, Switzerland, 2009. [Google Scholar]
- Standard ISO 10993-12:2012. Biological Evaluation of Medical Devices—Part12: Sample Preparation and Reference Materials; International Organization for Standardization: Geneve, Switzerland, 2012. [Google Scholar]
- Przekora, A.; Czechowska, J.; Pijocha, D.; Ślósarczyk, A.; Ginalska, G. Do novel cement-type biomaterials reveal ion reactivity that affects cell viability in vitro? Open Life Sci. 2014, 9, 277–289. [Google Scholar] [CrossRef]
- Kazimierczak, P.; Palka, K.; Przekora, A. Development and Optimization of the Novel Fabrication Method of Highly Macroporous Chitosan/Agarose/Nanohydroxyapatite Bone Scaffold for Potential Regenerative Medicine Applications. Biomolecules 2019, 9, 434. [Google Scholar] [CrossRef]


| Parameter | Sample | Statistically Significant Difference * | |
|---|---|---|---|
| FAP/Glucan Mean (±SD) | HAP/Glucan Mean (±SD) | ||
| Total intrusion volume [cm3/g] | 0.47 (±0.004) | 0.88 (±0.014) | yes, p ≤ 0.0001 |
| Total pore area [m2/g] | 5.16 (±0.21) | 20.78 (±0.3) | yes, p ≤ 0.0001 |
| Bulk density [g/cm3] | 1.14 (±0.006) | 0.75 (±0.01) | yes, p ≤ 0.0001 |
| Apparent (skeletal) density [g/cm3] | 2.43 (±0.035) | 2.20 (±0.05) | yes, p ≤ 0.01 |
| Porosity [%] | 53.19 (±0.58) | 65.93 (±0.32) | yes, p ≤ 0.0001 |
| Median pore diameter (Volume based) [µm] | 1.41 (±0.11) | 0.13 (±0) | yes, p ≤ 0.0001 |
| Median pore diameter (Area based) [µm] | 0.21 (±0.006) | 0.1 (±0) | yes, p ≤ 0.0001 |
| Average pore diameter [µm] | 0.36 (±0.015) | 0.17 (±0.006) | yes, p ≤ 0.0001 |
| Calcium | Phosphorus | Carbon | Oxygen | Fluorine | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| HAP/glucan | 4.27 | 2.13 | 2.59 | 1.01 | 49.12 | 4.87 | 44.03 | 2.58 | 0 | 0 |
| FAP/glucan | 1.74 | 1.53 | 0.98 | 0.58 | 53.84 | 3.21 | 43.14 | 1.53 | 0.29 | 0.27 |
| statistically significant difference * | yes, p ≤ 0.0001 | yes, p ≤ 0.0001 | yes, p ≤ 0.01 | no, p > 0.05 | yes, p ≤ 0.0001 | |||||
| Experiment | Sample | ||
|---|---|---|---|
| Shape | Diameter (mm) (± 0.5 mm) | Height (mm) (± 0.5 mm) | |
| SEM imaging, Live-Dead staining, Cytoskeleton Imaging, ELISA tests | flat disc | 8 | 2 |
| Bioactivity assessment | flat disc | 10 | 2 |
| Absorbability testing | cylindrical | 5 | 9 |
| 9 | 12 | ||
| 13 | 15 | ||
| Mechanical testing | cylindrical | 8 | 8 |
| Porosimetry testing, Fluoride ion release test | cylindrical | 11 | 11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borkowski, L.; Przekora, A.; Belcarz, A.; Palka, K.; Jojczuk, M.; Lukasiewicz, P.; Nogalski, A.; Ginalska, G. Highly Porous Fluorapatite/β-1,3-Glucan Composite for Bone Tissue Regeneration: Characterization and In-Vitro Assessment of Biomedical Potential. Int. J. Mol. Sci. 2021, 22, 10414. https://doi.org/10.3390/ijms221910414
Borkowski L, Przekora A, Belcarz A, Palka K, Jojczuk M, Lukasiewicz P, Nogalski A, Ginalska G. Highly Porous Fluorapatite/β-1,3-Glucan Composite for Bone Tissue Regeneration: Characterization and In-Vitro Assessment of Biomedical Potential. International Journal of Molecular Sciences. 2021; 22(19):10414. https://doi.org/10.3390/ijms221910414
Chicago/Turabian StyleBorkowski, Leszek, Agata Przekora, Anna Belcarz, Krzysztof Palka, Mariusz Jojczuk, Piotr Lukasiewicz, Adam Nogalski, and Grazyna Ginalska. 2021. "Highly Porous Fluorapatite/β-1,3-Glucan Composite for Bone Tissue Regeneration: Characterization and In-Vitro Assessment of Biomedical Potential" International Journal of Molecular Sciences 22, no. 19: 10414. https://doi.org/10.3390/ijms221910414
APA StyleBorkowski, L., Przekora, A., Belcarz, A., Palka, K., Jojczuk, M., Lukasiewicz, P., Nogalski, A., & Ginalska, G. (2021). Highly Porous Fluorapatite/β-1,3-Glucan Composite for Bone Tissue Regeneration: Characterization and In-Vitro Assessment of Biomedical Potential. International Journal of Molecular Sciences, 22(19), 10414. https://doi.org/10.3390/ijms221910414








