RNA Binding Properties of the Ty1 LTR-Retrotransposon Gag Protein
Abstract
1. Introduction
2. Results
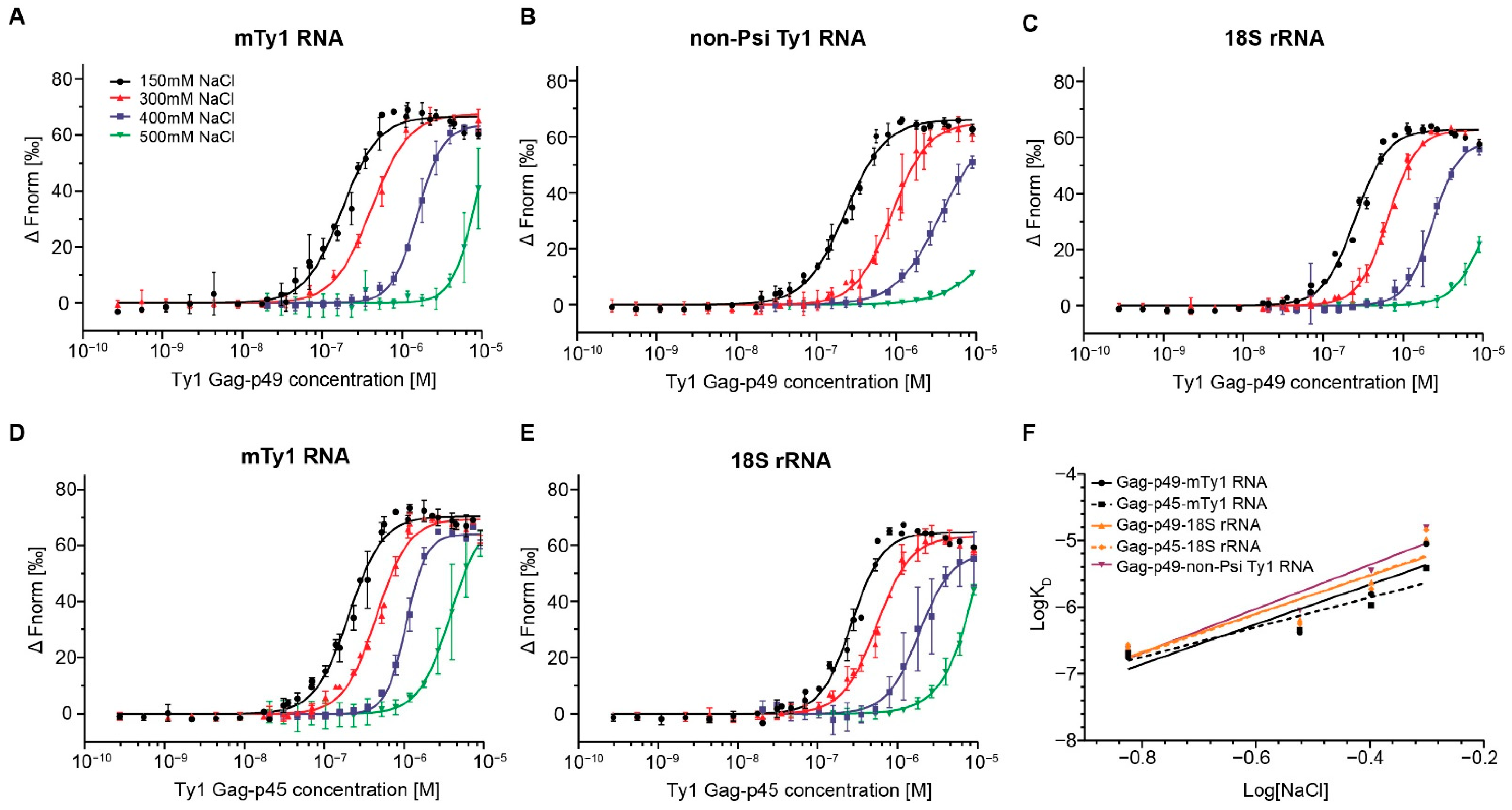
2.1. RNA Binding Affinity and Specificity of Immature and Mature Ty1 Gag
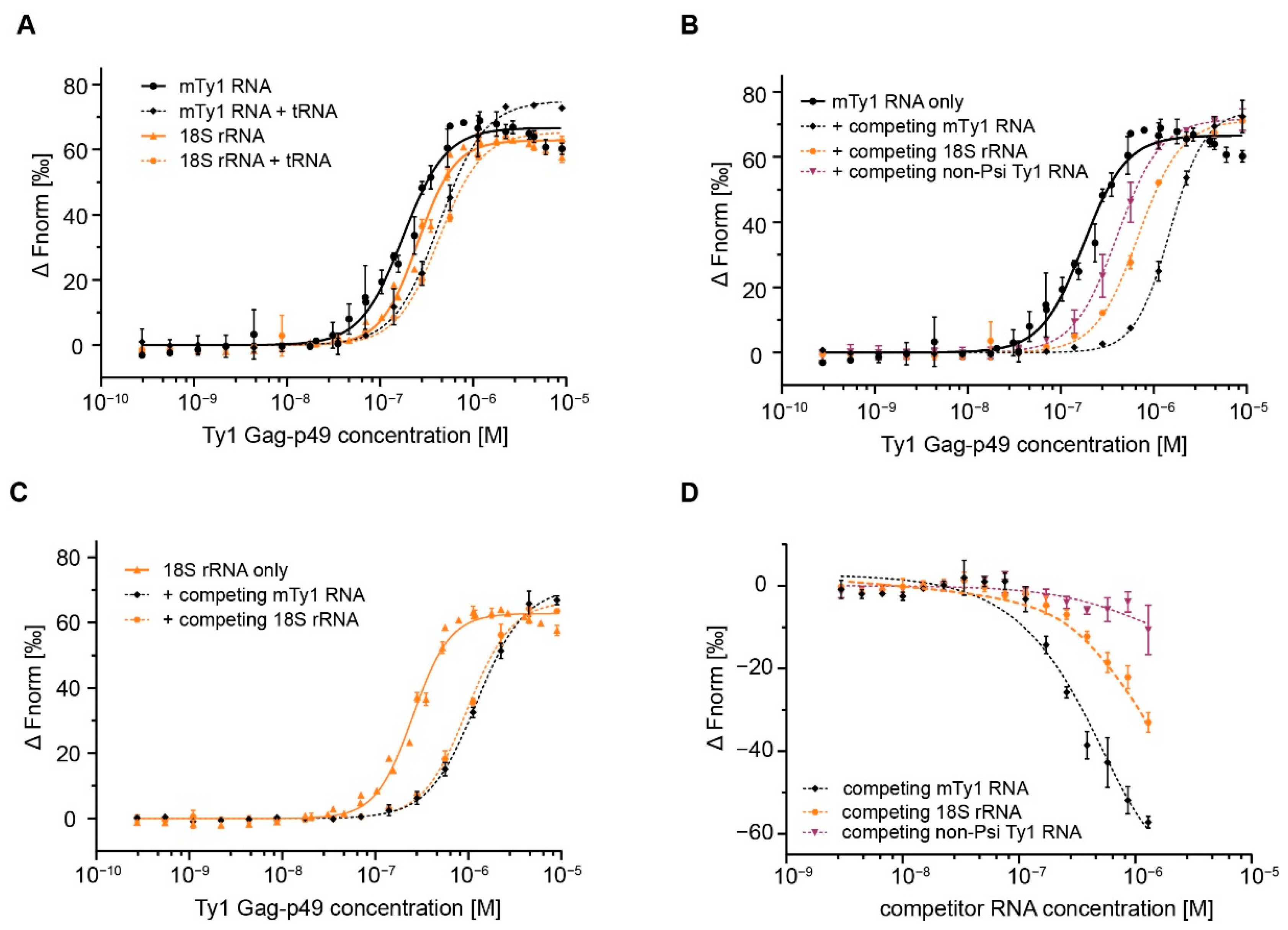
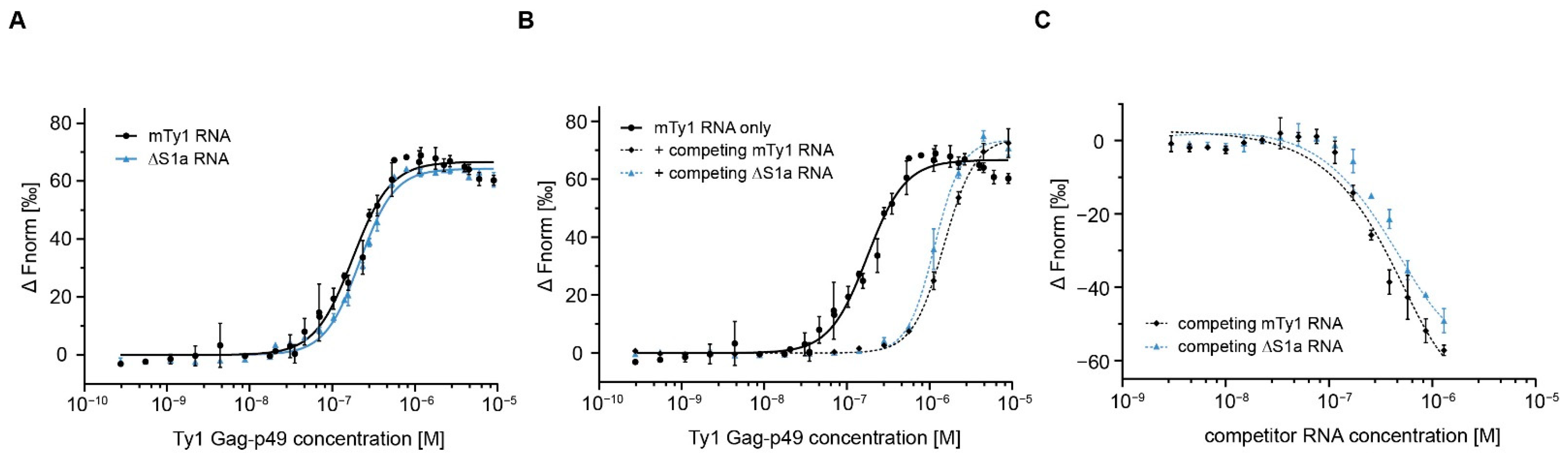
2.2. Gag Interactions in the Presence of Competitor RNA
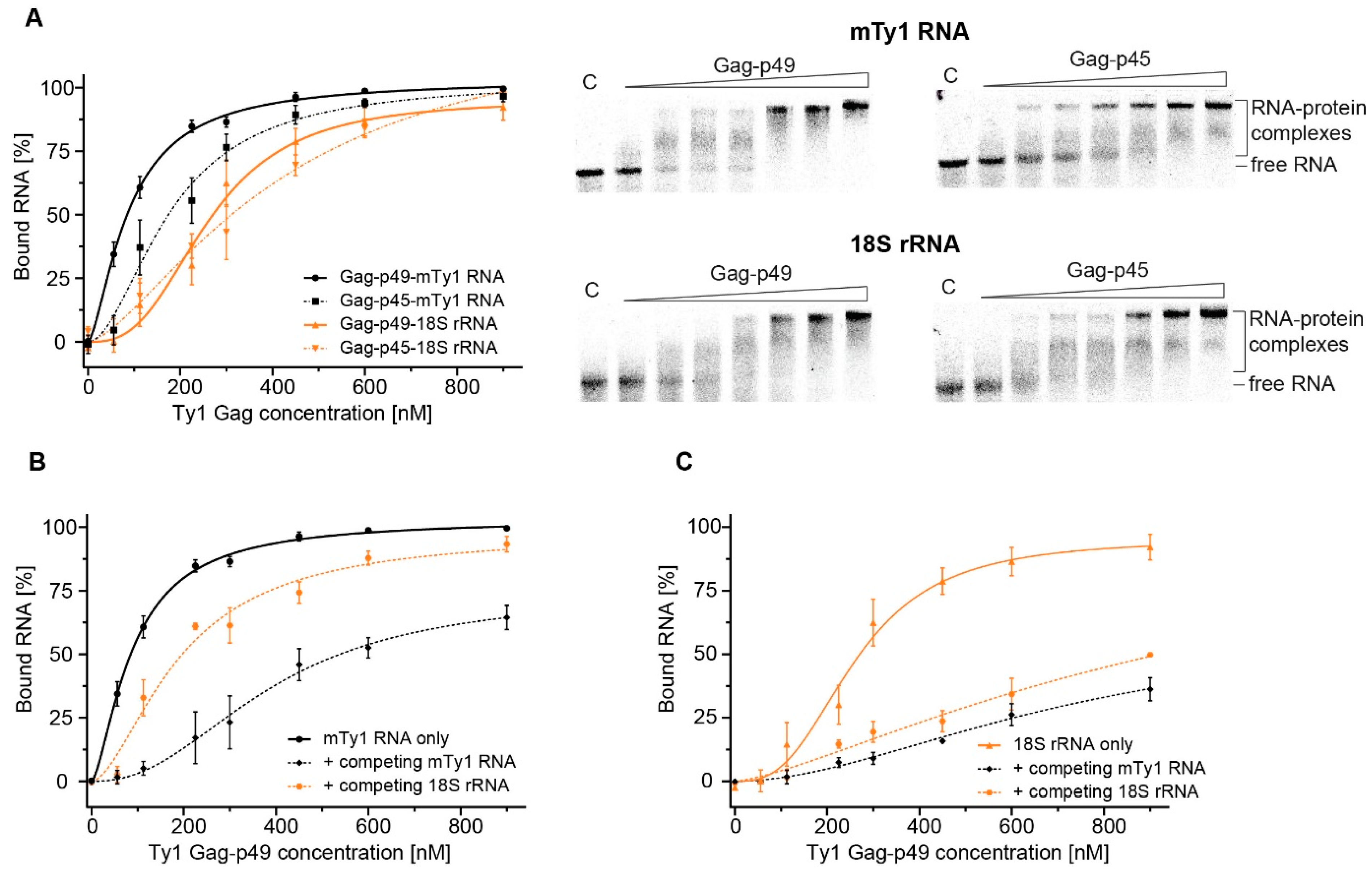
2.3. Comparing MST and EMSA to Detect Gag-RNA Complexes
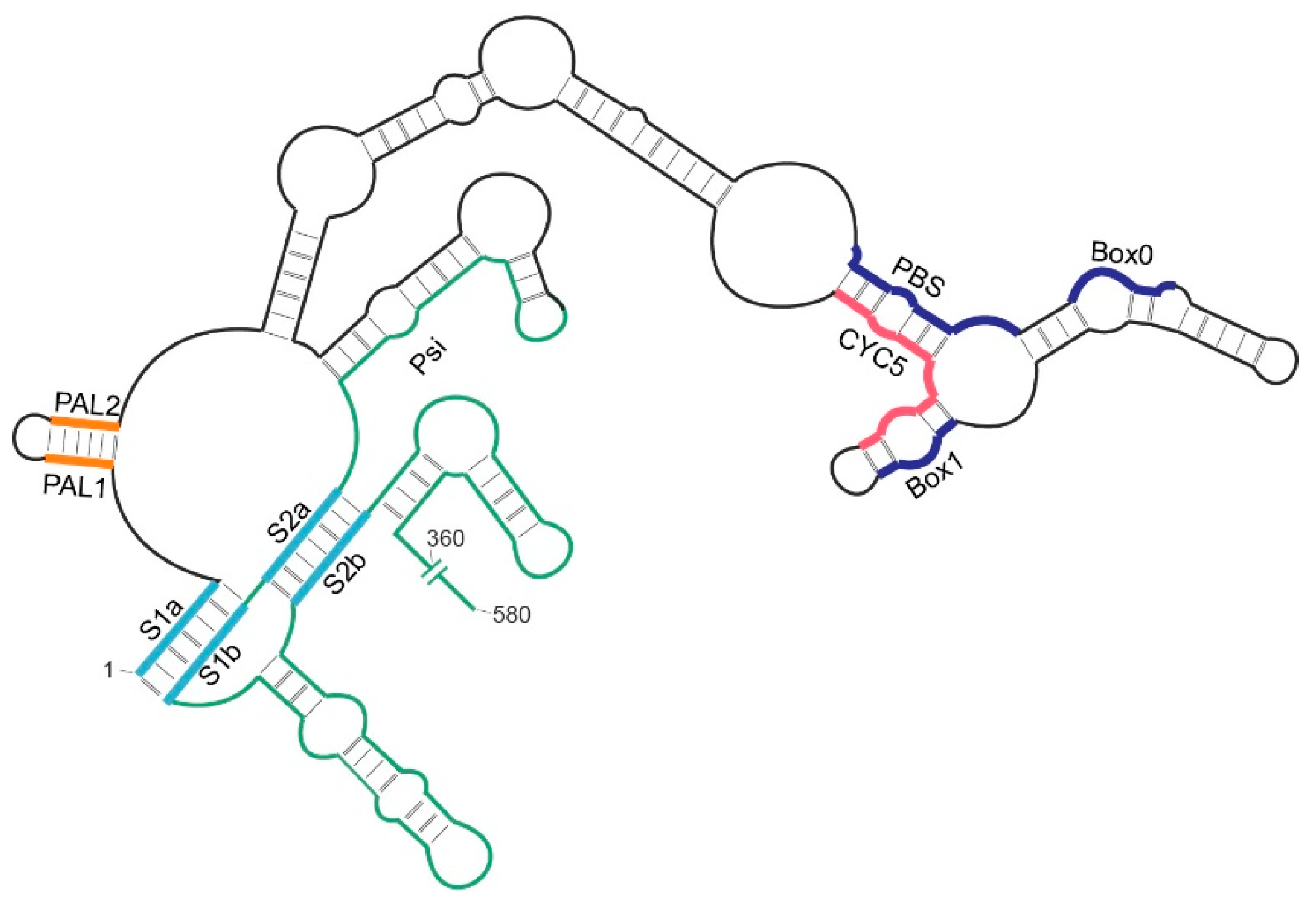
2.4. Role of the 5′ Pseudoknot in Ty1 RNA Binding
3. Discussion
4. Materials and Methods
4.1. Expression and Purification of Ty1 Gag Proteins
4.2. DNA and RNA Substrates
4.3. Microscale Thermophoresis
4.4. Electrophoretic Mobility Shift Assays
4.5. Hydroxyl Radical Footprinting, Primer Extension Reactions, and Data Processing
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eickbush, T.H.; Malik, H.S. Origin and Evolution of retrotransposons. In Mobile DNA II; Craig, N.L., Craigie, R., Gellert, M., Lambowitz, A.M., Eds.; ASM Press: Washington, DC, USA, 2002; pp. 1111–1144. [Google Scholar]
- Coffin, J.M.; Hughes, S.H.; Varmus, H.E. (Eds.) Retroviruses; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1997. [Google Scholar]
- Garfinkel, D.J.; Boeke, J.D.; Fink, G.R. Ty element transposition: Reverse transcriptase and virus-like particles. Cell 1985, 42, 507–517. [Google Scholar] [CrossRef]
- Shiba, T.; Saigo, K. Retrovirus-like particles containing RNA homologous to the transposable element copia in Drosophila melanogaster. Nature 1983, 302, 119–124. [Google Scholar] [CrossRef]
- Zhang, W.; Mendonça, L.M.; Mansky, L.M. The Retrovirus Capsid Core. In Virus Protein and Nucleoprotein Complexes; Harris, J.R., Bhella, D., Eds.; Springer Nature Singapore Pte Ltd.: Singapore, 2018; pp. 169–187. [Google Scholar]
- Olson, E.D.; Musier-Forsyth, K. Retroviral Gag protein–RNA interactions: Implications for specific genomic RNA packaging and virion assembly. Semin. Cell Dev. Biol. 2019, 86, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Rein, A.; Datta, S.; Jones, C.P.; Musier-Forsyth, K. Diverse interactions of retroviral Gag proteins with RNAs. Trends Biochem. Sci. 2011, 36, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Muriaux, D.; Darlix, J.-L. Properties and functions of the nucleocapsid protein in virus assembly. RNA Biol. 2010, 7, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Mailler, E.; Bernacchi, S.; Marquet, R.; Paillart, J.-C.; Vivet-Boudou, V.; Smyth, R.P. The Life-Cycle of the HIV-1 Gag–RNA Complex. Viruses 2016, 8, 248. [Google Scholar] [CrossRef] [PubMed]
- Pachulska-Wieczorek, K.; Le Grice, S.F.; Purzycka, K.J. Determinants of Genomic RNA Encapsidation in the Saccharomyces cerevisiae Long Terminal Repeat Retrotransposons Ty1 and Ty3. Viruses 2016, 8, 193. [Google Scholar] [CrossRef]
- Al-Khayat, A.H.; Bhella, D.; Kenney, J.M.; Roth, J.-F.; Kingsman, A.J.; Martin-Rendon, E.; Saibil, H.R. Yeast Ty retrotransposons assemble into virus-like particles whose T-numbers depend on the C-terminal length of the capsid protein. J. Mol. Biol. 1999, 292, 65–73. [Google Scholar] [CrossRef]
- Merkulov, G.V.; Swiderek, K.M.; Brachmann, C.B.; Boeke, J.D. A critical proteolytic cleavage site near the C terminus of the yeast retrotransposon Ty1 Gag protein. J. Virol. 1996, 70, 5548–5556. [Google Scholar] [CrossRef]
- Tucker, J.M.; Larango, M.E.; Wachsmuth, L.; Kannan, N.; Garfinkel, D.J. The Ty1 Retrotransposon Restriction Factor p22 Targets Gag. PLoS Genet. 2015, 11, e1005571. [Google Scholar] [CrossRef]
- Cristofari, G.; Ficheux, D.; Darlix, J.-L. The Gag-like Protein of the Yeast Ty1 Retrotransposon Contains a Nucleic Acid Chaperone Domain Analogous to Retroviral Nucleocapsid Proteins. J. Biol. Chem. 2000, 275, 19210–19217. [Google Scholar] [CrossRef]
- Checkley, M.A.; Mitchell, J.A.; Eizenstat, L.D.; Lockett, S.J.; Garfinkel, D.J. Ty1 Gag Enhances the Stability and Nuclear Export of Ty1 mRNA. Traffic 2012, 14, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Malagon, F.; Jensen, T.H. The T Body, a New Cytoplasmic RNA Granule in Saccharomyces cerevisiae. Mol. Cell. Biol. 2008, 28, 6022–6032. [Google Scholar] [CrossRef] [PubMed]
- Malagon, F.; Jensen, T.H. T-body formation precedes virus-like particle maturation in S. cerevisiae. RNA Biol. 2011, 8, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Checkley, M.A.; Nagashima, K.; Lockett, S.J.; Nyswaner, K.M.; Garfinkel, D.J. P-Body Components Are Required for Ty1 Retrotransposition during Assembly of Retrotransposition-Competent Virus-Like Particles. Mol. Cell. Biol. 2010, 30, 382–398. [Google Scholar] [CrossRef]
- Nishida, Y.; Pachulska-Wieczorek, K.; Blaszczyk, L.; Saha, A.; Gumna, J.; Garfinkel, D.J.; Purzycka, K.J. Ty1 retrovirus-like element Gag contains overlapping restriction factor and nucleic acid chaperone functions. Nucleic Acids Res. 2015, 43, 7414–7431. [Google Scholar] [CrossRef]
- Gumna, J.; Purzycka, K.J.; Ahn, H.W.; Garfinkel, D.J.; Pachulska-Wieczorek, K. Retroviral-like determinants and functions required for dimerization of Ty1 retrotransposon RNA. RNA Biol. 2019, 16, 1749–1763. [Google Scholar] [CrossRef]
- Cristofari, G.; Bampi, C.; Wilhelm, M.; Wilhelm, F.; Darlix, J. A 5’-3’ long-range interaction in Ty1 RNA controls its reverse transcription and retrotransposition. EMBO J. 2002, 21, 4368–4379. [Google Scholar] [CrossRef]
- Kuzembayeva, M.; Dilley, K.; Sardo, L.; Hu, W.-S. Life of psi: How full-length HIV-1 RNAs become packaged genomes in the viral particles. Virology 2014, 454-455, 362–370. [Google Scholar] [CrossRef]
- D’Souza, V.; Summers, M.F. How retroviruses select their genomes. Nat. Rev. Genet. 2005, 3, 643–655. [Google Scholar] [CrossRef]
- Webb, J.A.; Jones, C.P.; Parent, L.; Rouzina, I.; Musier-Forsyth, K. Distinct binding interactions of HIV-1 Gag to Psi and non-Psi RNAs: Implications for viral genomic RNA packaging. RNA 2013, 19, 1078–1088. [Google Scholar] [CrossRef]
- Comas-Garcia, M.; Datta, S.A.; Baker, L.; Varma, R.; Gudla, P.R.; Rein, A. Dissection of specific binding of HIV-1 Gag to the ’packaging signal’ in viral RNA. eLife 2017, 6, e27055. [Google Scholar] [CrossRef]
- Xu, H.; Boeke, J.D. Localization of sequences required in cis for yeast Ty1 element transposition near the long terminal repeats: Analysis of mini-Ty1 elements. Mol. Cell. Biol. 1990, 10, 2695–2702. [Google Scholar] [CrossRef]
- Bolton, E.C.; Coombes, C.; Eby, Y.; Cardell, M.; Boeke, J.D. Identification and characterization of critical cis-acting sequences within the yeast Ty1 retrotransposon. RNA 2005, 11, 308–322. [Google Scholar] [CrossRef] [PubMed]
- Luschnig, C.; Bachmair, A. RNA Packaging of Yeast Retrotransposon Ty1 in the Heterologous Host, Escherichia coli. Biol. Chem. 1997, 378, 39–46. [Google Scholar] [CrossRef]
- Friant, S.; Heyman, T.; Wilhelm, M.L.; Wilhelm, F.X. Extended Interactions Between the Primer tRNAiMet and Genomic RNA of the Yeast Ty1 Retrotransposon. Nucleic Acids Res. 1996, 24, 441–449. [Google Scholar] [CrossRef]
- Friant, S.; Heyman, T.; Byström, A.S.; Wilhelm, M.; Wilhelm, F.X. Interactions between Ty1 Retrotransposon RNA and the T and D Regions of the tRNAiMet Primer Are Required for Initiation of Reverse Transcription In Vivo. Mol. Cell. Biol. 1998, 18, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Purzycka, K.J.; Lusvarghi, S.; Li, D.; LeGrice, S.F.; Boeke, J.D. Retrotransposon Ty1 RNA contains a 5’-terminal long-range pseudoknot required for efficient reverse transcription. RNA 2013, 19, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Jerabek-Willemsen, M.; Wienken, C.J.; Braun, D.; Baaske, P.; Duhr, S. Molecular Interaction Studies Using Microscale Thermophoresis. ASSAY Drug Dev. Technol. 2011, 9, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Saha, A.; Mitchell, J.A.; Nishida, Y.; Hildreth, J.E.; Ariberre, J.A.; Gilbert, W.V.; Garfinkel, D.J. A trans -Dominant Form of Gag Restricts Ty1 Retrotransposition and Mediates Copy Number Control. J. Virol. 2015, 89, 3922–3938. [Google Scholar] [CrossRef] [PubMed]
- Błaszczyk, L.; Biesiada, M.; Saha, A.; Garfinkel, D.J.; Purzycka, K.J. Structure of Ty1 Internally Initiated RNA Influences Restriction Factor Expression. Viruses 2017, 9, 74. [Google Scholar] [CrossRef]
- Comas-Garcia, M.; Kroupa, T.; Datta, S.A.; Harvin, D.P.; Hu, W.-S.; Rein, A. Efficient support of virus-like particle assembly by the HIV-1 packaging signal. eLife 2018, 7. [Google Scholar] [CrossRef]
- El-Wahab, E.A.; Smyth, R.; Mailler, E.; Bernacchi, S.; Vivet-Boudou, V.; Hijnen, M.; Jossinet, F.; Mak, J.; Paillart, J.-C.; Marquet, R. Specific recognition of the HIV-1 genomic RNA by the Gag precursor. Nat. Commun. 2014, 5, 4304. [Google Scholar] [CrossRef]
- Record, M.T.; Lohman, T.M.; de Haseth, P. Ion effects on ligand-nucleic acid interactions. J. Mol. Biol. 1976, 107, 145–158. [Google Scholar] [CrossRef]
- Rouzina, I.; Bloomfield, V.A. Competitive electrostatic binding of charged ligands to polyelectrolytes: Practical approach using the non-linear Poisson-Boltzmann equation. Biophys. Chem. 1997, 64, 139–155. [Google Scholar] [CrossRef]
- Jones, C.P.; Datta, S.; Rein, A.; Rouzina, I.; Musier-Forsyth, K. Matrix Domain Modulates HIV-1 Gag’s Nucleic Acid Chaperone Activity via Inositol Phosphate Binding. J. Virol. 2010, 85, 1594–1603. [Google Scholar] [CrossRef] [PubMed]
- König, F.; Schubert, T.; Längst, G. The monoclonal S9.6 antibody exhibits highly variable binding affinities towards different R-loop sequences. PLoS ONE 2017, 12, e0178875. [Google Scholar] [CrossRef] [PubMed]
- Purzycka, K.J.; Legiewicz, M.; Matsuda, E.; Eizentstat, L.D.; Lusvarghi, S.; Saha, A.; Le Grice, S.F.J.; Garfinkel, D.J. Exploring Ty1 retrotransposon RNA structure within virus-like particles. Nucleic Acids Res. 2012, 41, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Nilsen, T.W. Mapping RNA–Protein Interactions Using Hydroxyl-Radical Footprinting. Cold Spring Harb. Protoc. 2014, 2014. [Google Scholar] [CrossRef]
- Kingsman, S.M.; Kingsman, A.J.; Martin-Rendon, E. Possible regulatory function of the Saccharomyces cerevisiae Ty1 retrotransposon core protein. Yeast 2000, 16, 921–932. [Google Scholar] [CrossRef]
- Muriaux, D.; Mirro, J.; Harvin, D.; Rein, A. RNA is a structural element in retrovirus particles. Proc. Natl. Acad. Sci. USA 2001, 98, 5246–5251. [Google Scholar] [CrossRef] [PubMed]
- Rye-McCurdy, T.; Olson, E.D.; Liu, S.; Binkley, C.; Reyes, J.-P.; Thompson, B.; Flanagan, J.M.; Parent, L.J.; Musier-Forsyth, K. Functional Equivalence of Retroviral MA Domains in Facilitating Psi RNA Binding Specificity by Gag. Viruses 2016, 8, 256. [Google Scholar] [CrossRef]
- Pachulska-Wieczorek, K.; Błaszczyk, L.; Biesiada, M.; Adamiak, R.W.; Purzycka, K.J. The matrix domain contributes to the nucleic acid chaperone activity of HIV-2 Gag. Retrovirology 2016, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kutluay, S.B.; Zang, T.; Blanco-Melo, D.; Powell, C.; Jannain, D.; Errando, M.; Bieniasz, P.D. Global Changes in the RNA Binding Specificity of HIV-1 Gag Regulate Virion Genesis. Cell 2014, 159, 1096–1109. [Google Scholar] [CrossRef] [PubMed]
- Berkowitz, R.D.; Luban, J.; Goff, S.P. Specific binding of human immunodeficiency virus type 1 gag polyprotein and nucleocapsid protein to viral RNAs detected by RNA mobility shift assays. J. Virol. 1993, 67, 7190–7200. [Google Scholar] [CrossRef] [PubMed]
- Comas-Garcia, M.; Davis, S.R.; Rein, A. On the Selective Packaging of Genomic RNA by HIV-1. Viruses 2016, 8, 246. [Google Scholar] [CrossRef]
- Corley, M.; Burns, M.C.; Yeo, G.W. How RNA-Binding Proteins Interact with RNA: Molecules and Mechanisms. Mol. Cell 2020, 78, 9–29. [Google Scholar] [CrossRef]
- Balcerak, A.; Trebinska-Stryjewska, A.; Konopiński, R.; Wakuła, M.; Grzybowska, E.A. RNA–protein interactions: Disorder, moonlighting and junk contribute to eukaryotic complexity. Open Biol. 2019, 9, 190096. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.S.; Hu, J.; Bériault, V.; Mouland, A.J.; Kleiman, L.; Wainberg, M.A.; Liang, C. Sequences Downstream of the 5′ Splice Donor Site Are Required for both Packaging and Dimerization of Human Immunodeficiency Virus Type 1 RNA. J. Virol. 2003, 77, 84–96. [Google Scholar] [CrossRef]
- Doria-Rose, N.A.; Vogt, V.M. In Vivo Selection of Rous Sarcoma Virus Mutants with Randomized Sequences in the Packaging Signal. J. Virol. 1998, 72, 8073–8082. [Google Scholar] [CrossRef] [PubMed]
- Gherghe, C.; Lombo, T.; Leonard, C.W.; Datta, S.; Bess, J.W.; Gorelick, R.J.; Rein, A.; Weeks, K.M. Definition of a high-affinity Gag recognition structure mediating packaging of a retroviral RNA genome. Proc. Natl. Acad. Sci. USA 2010, 107, 19248–19253. [Google Scholar] [CrossRef] [PubMed]
- Mougel, M.; Barklis, E. A role for two hairpin structures as a core RNA encapsidation signal in murine leukemia virus virions. J. Virol. 1997, 71, 8061–8065. [Google Scholar] [CrossRef] [PubMed]
- Andrzejewska, A.; Zawadzka, M.; Gumna, J.; Garfinkel, D.J.; Pachulska-Wieczorek, K. In vivo structure of the Ty1 retrotransposon RNA genome. Nucleic Acids Res. 2021, 49, 2878–2893. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, P.H.; Coombes, C.; Kenny, A.E.; Lawler, J.F.; Boeke, J.; Curcio, M.J. Ty1 Mobilizes Subtelomeric Y′ Elements in Telomerase-Negative Saccharomyces cerevisiae Survivors. Mol. Cell. Biol. 2004, 24, 9887–9898. [Google Scholar] [CrossRef]
- Maxwell, P.H.; Curcio, M.J. Retrosequence formation restructures the yeast genome. Genes Dev. 2007, 21, 3308–3318. [Google Scholar] [CrossRef]
- Dutko, J.A.; Kenny, A.E.; Gamache, E.R.; Curcio, M.J. 5′ to 3′ mRNA Decay Factors Colocalize with Ty1 Gag and Human APOBEC3G and Promote Ty1 Retrotransposition. J. Virol. 2010, 84, 5052–5066. [Google Scholar] [CrossRef]
- Vasa, S.M.; Guex, N.; Wilkinson, K.A.; Weeks, K.M.; Giddings, M.C. ShapeFinder: A software system for high-throughput quantitative analysis of nucleic acid reactivity information resolved by capillary electrophoresis. RNA 2008, 14, 1979–1990. [Google Scholar] [CrossRef]
- Gumna, J.; Zok, T.; Figurski, K.; Pachulska-Wieczorek, K.; Szachniuk, M. RNAthor—Fast, accurate normalization, visualization and statistical analysis of RNA probing data resolved by capillary electrophoresis. PLoS ONE 2020, 15, e0239287. [Google Scholar] [CrossRef]

| KD [nM] | nH | KD [nM] | nH | KD [nM] | nH | KD [nM] | nH | KD(1M) [M] | Zeff | |
|---|---|---|---|---|---|---|---|---|---|---|
| 150 mM NaCl | 300 mM NaCl | 400 mM NaCl | 500 mM NaCl | |||||||
| mTy1 RNA-Gag-p49 | 176.8 ± 11.4 | 1.9 | 414.7 ± 21.7 | 1.8 | 1566.2 ± 36 | 2.7 | 8935 ± 3544.5 | 2.8 | (3.4 ± 5.2) × 10−5 | 3.0 ± 0.9 |
| 18S rRNA-Gag-p49 | 253.9 ± 11.9 | 2.2 | 647.3 ± 17.2 | 2.4 | 2337.4 ± 127.7 | 2.6 | 10466 ± 4083.1 | 2.2 | (4.2 ± 4.4) × 10−5 | 2.9 ± 0.8 |
| non-Psi Ty1 RNA-Gag-p49 | 231.9 ± 10.5 | 1.7 | 886.4 ± 37.7 | 1.9 | 3520 ± 258.4 | 1.6 | 15635 ± 19989 | 1.4 | (9 ± 3.9) × 10−5 | 3.3 ± 0.7 |
| mTy1 RNA-Gag-p45 | 204.3 ± 10.8 | 2 | 451.1 ± 21.9 | 2.1 | 1064.7 ± 32.6 | 3.4 | 3823.2 ± 148.8 | 2.2 | (1.1 ± 3.4) × 10−5 | 2.2 ± 0.6 |
| 18S rRNA-Gag-p45 | 267.8 ± 15 | 2.2 | 552.4 ± 23.4 | 2 | 1871.3 ± 139.4 | 2.1 | 14525 ± 6330.2 | 1.7 | (4.6 ± 6.1) × 10−5 | 3.0 ± 1.1 |
| ΔS1a RNA-Gag-p49 | 214.7 ± 9.4 | 2.1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Labeled RNA | mTy1 RNA | 18S rRNA | |||
|---|---|---|---|---|---|
| Competitor | |||||
| KD [nM] | nH | KD [nM] | nH | ||
| competitor | 176.8 ± 11.4 | 1.9 | 253.9 ± 11.9 | 2.2 | |
| tRNA E.coli | 434.6 ± 15.8 | 1.8 | 430.2 ± 20.5 | 1.9 | |
| mTy1 RNA | 1491.6 ± 28.7 | 2.3 | 1224.9 ± 42.3 | 1.7 | |
| 18S rRNA | 679.2 ± 30.1 | 1.9 | 973.9 ± 37 | 1.8 | |
| non-Psi Ty1 RNA | 405.2 ± 7.5 | 1.8 | n.d. | n.d. | |
| ΔS1a RNA | 1150.2 ± 36.5 | 2.6 | n.d. | n.d. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gumna, J.; Andrzejewska-Romanowska, A.; Garfinkel, D.J.; Pachulska-Wieczorek, K. RNA Binding Properties of the Ty1 LTR-Retrotransposon Gag Protein. Int. J. Mol. Sci. 2021, 22, 9103. https://doi.org/10.3390/ijms22169103
Gumna J, Andrzejewska-Romanowska A, Garfinkel DJ, Pachulska-Wieczorek K. RNA Binding Properties of the Ty1 LTR-Retrotransposon Gag Protein. International Journal of Molecular Sciences. 2021; 22(16):9103. https://doi.org/10.3390/ijms22169103
Chicago/Turabian StyleGumna, Julita, Angelika Andrzejewska-Romanowska, David J. Garfinkel, and Katarzyna Pachulska-Wieczorek. 2021. "RNA Binding Properties of the Ty1 LTR-Retrotransposon Gag Protein" International Journal of Molecular Sciences 22, no. 16: 9103. https://doi.org/10.3390/ijms22169103
APA StyleGumna, J., Andrzejewska-Romanowska, A., Garfinkel, D. J., & Pachulska-Wieczorek, K. (2021). RNA Binding Properties of the Ty1 LTR-Retrotransposon Gag Protein. International Journal of Molecular Sciences, 22(16), 9103. https://doi.org/10.3390/ijms22169103






