Effects of Microenvironmental Changes on the Fluorescence Signal of Alternariol: Magnesium Induces Strong Enhancement in the Fluorescence of the Mycotoxin
Abstract
:1. Introduction
2. Results and Discussion
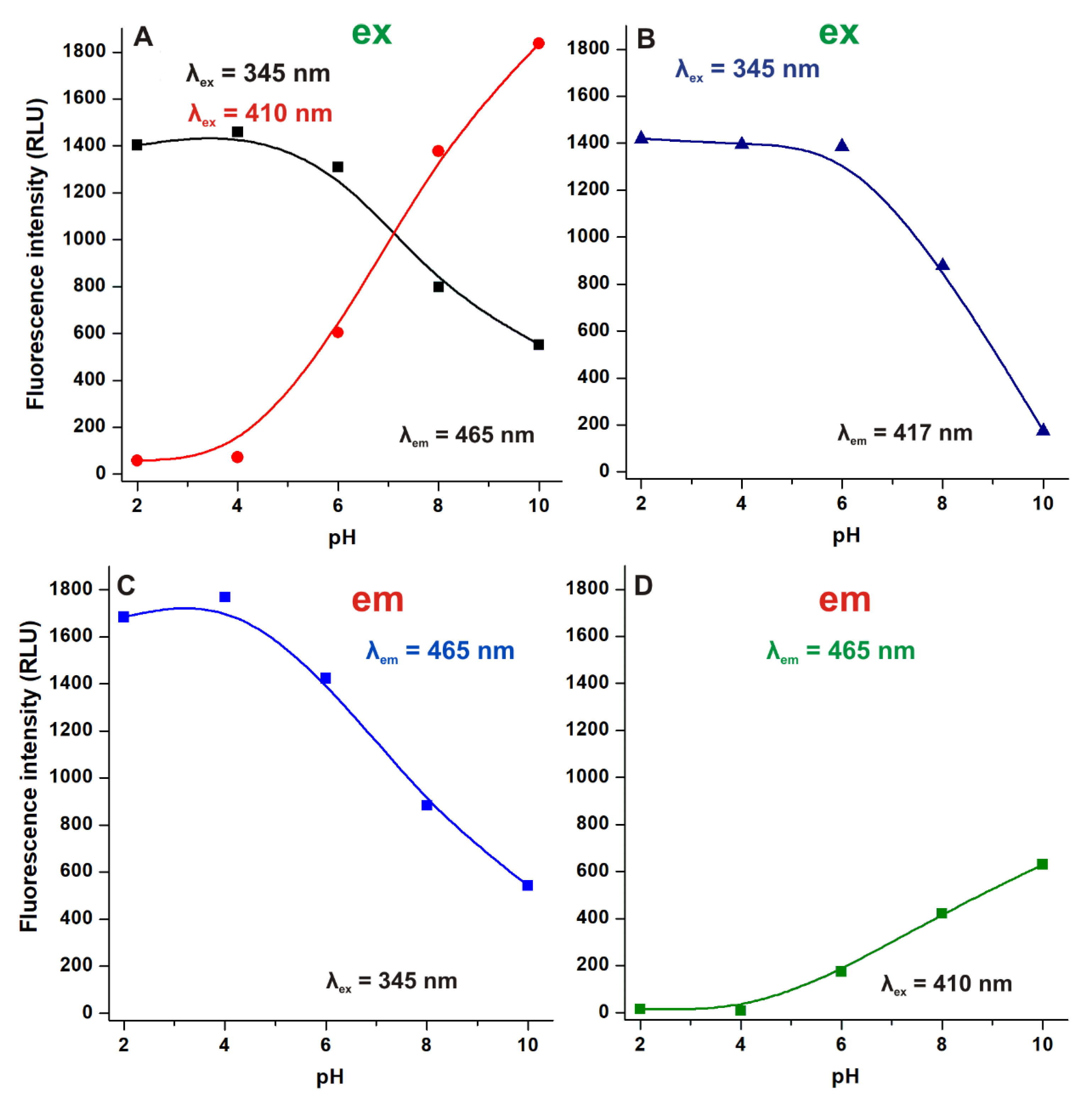
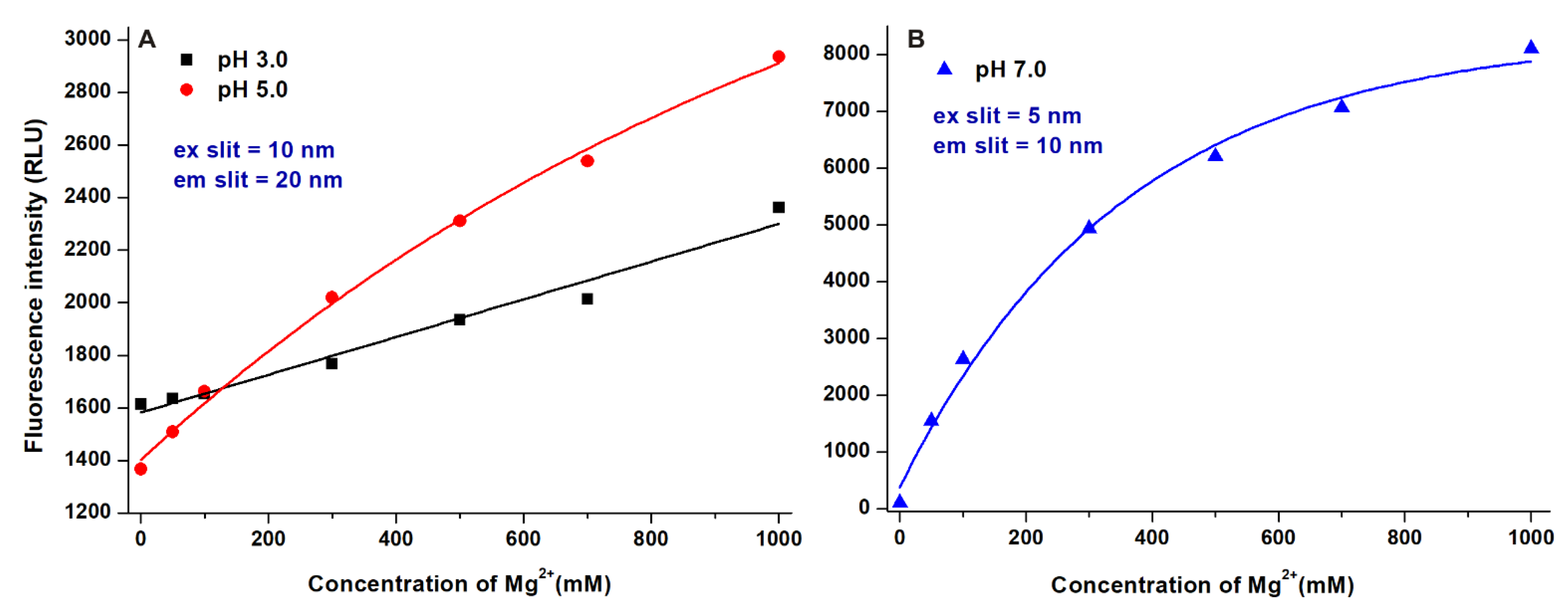
2.1. Effect of the Environmental pH on the Fluorescence Spectra of AOH
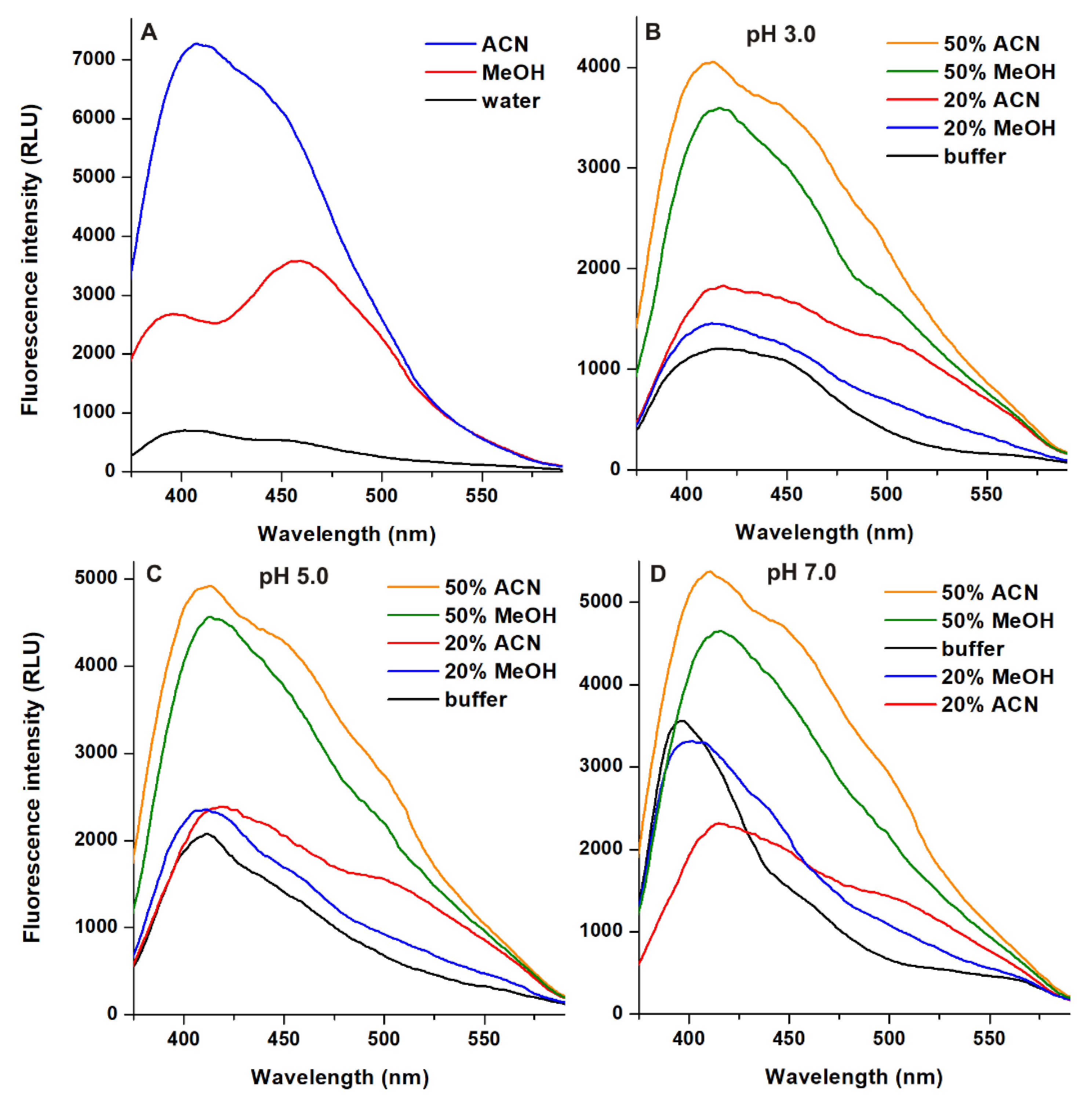
2.2. Effects of Acetonitrile and Methanol on the Emission Signal of AOH
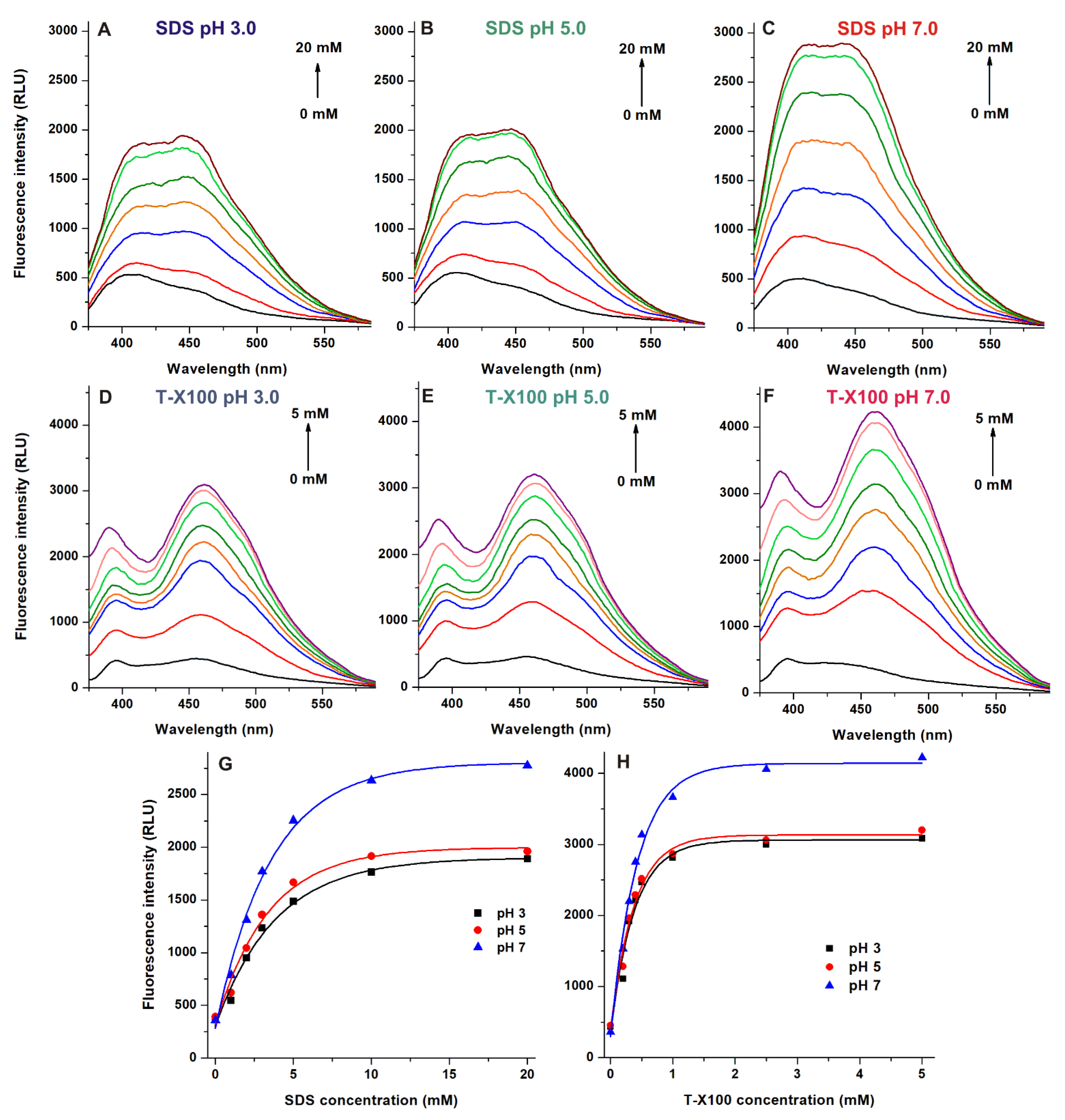
2.3. Effects of Detergents on the Emission Signal of AOH
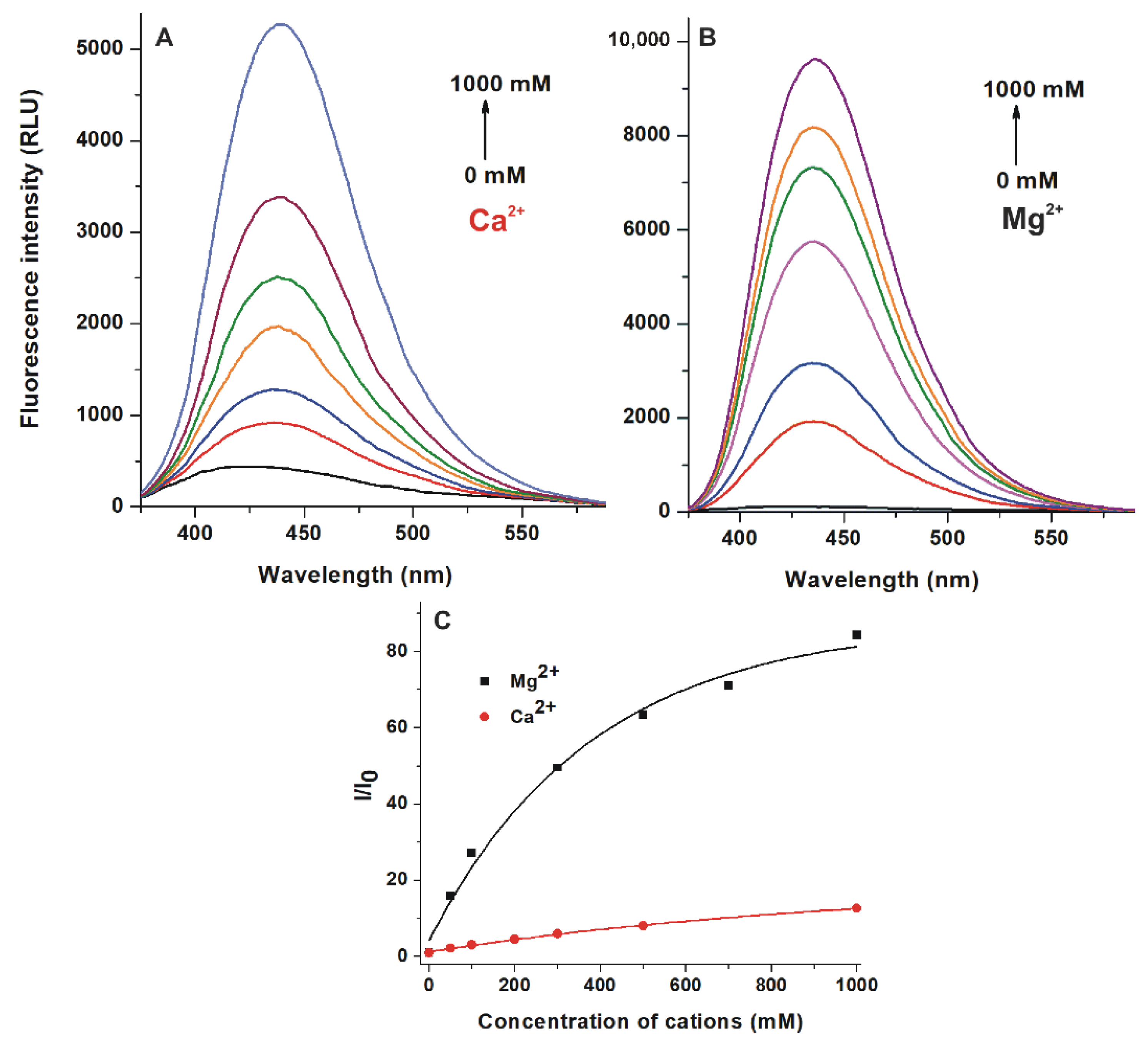
2.4. Effects of Cations on the Emission Signal of AOH
2.5. Investigation of AOH-Mg2+ and AOH-Ca2+ Interactions Employing Molecular Modeling Studies
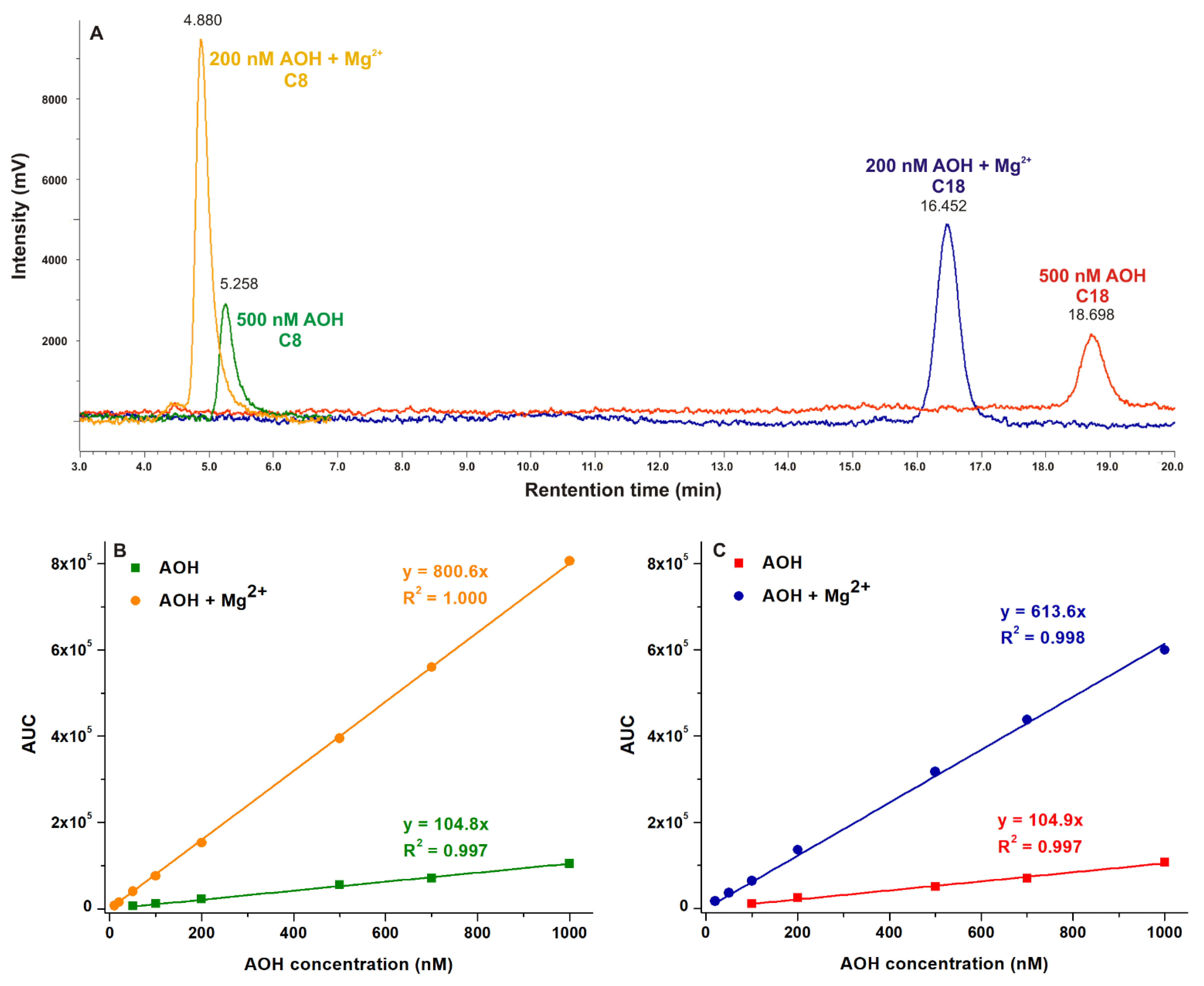
2.6. Probing the Analytical Application of AOH-Mg2+ Interaction to Increase the Sensitivity of HPLC-FLD Methods
3. Materials and Methods
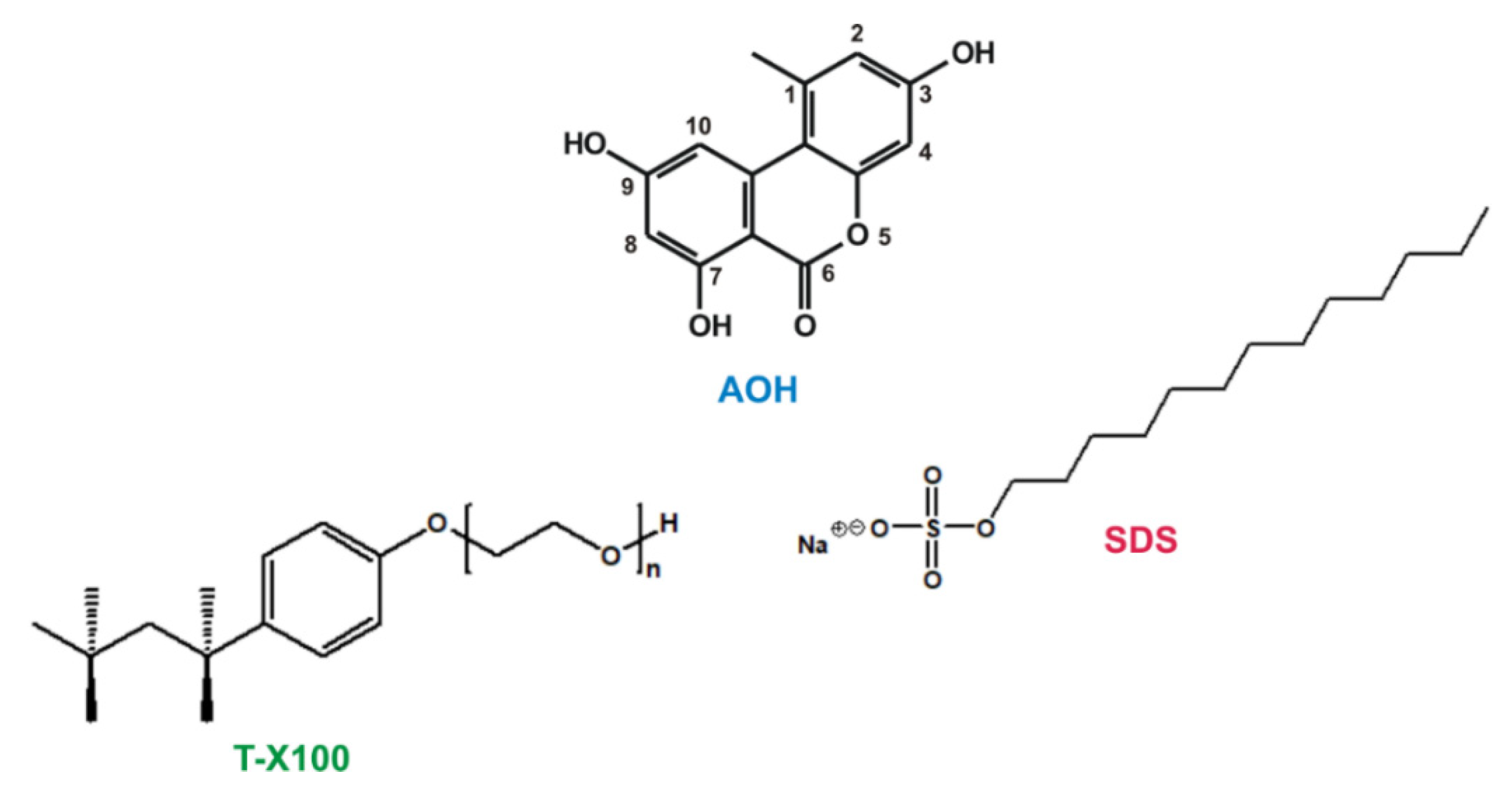
3.1. Reagents
3.2. Fluorescence Spectroscopic Measurements
3.3. Modeling Studies
3.4. HPLC Analyses
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dall’Asta, C.; Cirlini, M.; Falavigna, C. Mycotoxins from Alternaria: Toxicological implications. Adv. Mol. Toxicol. 2014, 8, 107–121. [Google Scholar] [CrossRef]
- Solhaug, A.; Eriksen, G.S.; Holme, J.A. Mechanisms of action and toxicity of the mycotoxin alternariol: A review. Basic Clin. Pharmacol. Toxicol. 2016, 119, 533–539. [Google Scholar] [CrossRef]
- Heussner, A.H.; Bingle, L.E.H. Comparative ochratoxin toxicity: A review of the available data. Toxins 2015, 7, 4253–4282. [Google Scholar] [CrossRef] [Green Version]
- McKean, C.; Tang, L.; Billam, M.; Tang, M.; Theodorakis, C.W.; Kendall, R.J.; Wang, J.-S. Comparative acute and combinative toxicity of aflatoxin B1 and T-2 toxin in animals and immortalized human cell lines. J. Appl. Toxicol. 2006, 26, 139–147. [Google Scholar] [CrossRef]
- Fraeyman, S.; Croubels, S.; Devreese, M.; Antonissen, G. Emerging Fusarium and Alternaria mycotoxins: Occurrence, toxicity and toxicokinetics. Toxins 2017, 9, 228. [Google Scholar] [CrossRef] [Green Version]
- ESFA. Scientific Opinion on the risks for animal and public health related to the presence of Alternaria toxins in feed and food. EFSA on Contaminants in the Food Chain (CONTAM). EFSA J. 2011, 9, 2407–2504. [Google Scholar] [CrossRef]
- Arcella, D.; Eskola, M.; Ruiz, J.A.G. Dietary exposure assessment to Alternaria toxins in the European population. EFSA J. 2016, 14, 4654. [Google Scholar] [CrossRef]
- Aichinger, G.; Del Favero, G.; Warth, B.; Marko, D. Alternaria toxins—Still emerging? Compr. Rev. Food Sci. Food Saf. 2021, 1–17. [Google Scholar] [CrossRef]
- Fliszár-Nyúl, E.; Lemli, B.; Kunsági-Máté, S.; Dellafiora, L.; Dall’Asta, C.; Cruciani, G.; Pethő, G.; Poór, M. Interaction of mycotoxin alternariol with serum albumin. Int. J. Mol. Sci. 2019, 20, 2352. [Google Scholar] [CrossRef] [Green Version]
- Fliszár-Nyúl, E.; Lemli, B.; Kunsági-Máté, S.; Szente, L.; Poór, M. Interactions of mycotoxin alternariol with cyclodextrins and its removal from aqueous solution by beta-cyclodextrin bead polymer. Biomolecules 2019, 9, 428. [Google Scholar] [CrossRef] [Green Version]
- Man, Y.; Liang, G.; Li, A.; Pan, L. Analytical methods for the determination of Alternaria mycotoxins. Chromatographia 2017, 80, 9–22. [Google Scholar] [CrossRef]
- Keller, J.; Moldenhauer, D.; Byrne, L.; Haase, H.; Resch-Genger, U.; Koch, M. Complexes of the mycotoxins citrinin and ochratoxin a with aluminum ions and their spectroscopic properties. Toxins 2018, 10, 538. [Google Scholar] [CrossRef] [Green Version]
- Hetmanski, M.T.; Scudamore, K.A. Detection of zearalenone in cereal extracts using high performance liquid chromatography with post-column derivatization. J. Chromatogr. 1991, 588, 41–52. [Google Scholar] [CrossRef]
- Appell, M.; Bosma, W.B. Effect of surfactants on the spectrofluorimetric properties of zearalenone. J. Lumin. 2011, 131, 2330–2334. [Google Scholar] [CrossRef]
- Manafi, M.H.; Allahyari, M.; Pourghazi, K.; Amoli-Diva, M.; Taherimaslak, Z. Surfactant-enhanced spectrofluorimetric determination of total aflatoxins from wheat samples after magnetic solid-phase extraction using modified Fe3O4 nanoparticles. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 146, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Poór, M.; Kunsági-Máté, S.; Matisz, G.; Li, Y.; Czibulya, Z.; Peles-Lemli, B.; Kőszegi, T. Interaction of alkali and alkaline earth ions with Ochratoxin, A. J. Lumin. 2013, 135, 276–280. [Google Scholar] [CrossRef]
- Poór, M.; Kuzma, M.; Matisz, G.; Li, Y.; Perjési, P.; Kunsági-Máté, S.; Kőszegi, T. Further Aspects of ochratoxin a-cation interactions: Complex formation with zinc ions and a novel analytical application of ochratoxin a-magnesium interaction in the HPLC-FLD system. Toxins 2014, 6, 1295–1307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez, E.M.R.; Alaejos, M.S.; Romero, C.D. Enhancement of the fluorescence intensity of Se-2,3-diaminonaphthalene complex in aqueous solution by adding organic solvents. Anal. Chim. Acta 1996, 334, 161–166. [Google Scholar] [CrossRef]
- Dobretsov, G.E.; Syrejschikova, T.I.; Smolina, N.V. On mechanisms of fluorescence quenching by water. Biophysics 2014, 59, 183–188. [Google Scholar] [CrossRef]
- Acharya, S.; Rebery, B. Fluorescence spectrometric study of eosin yellow dye-surfactant interactions. Arab. J. Chem. 2009, 2, 7–12. [Google Scholar] [CrossRef] [Green Version]
- Fery-Forgues, S.; Therese Le Bris, M.; Guette, J.P.; Valeur, B. Ion-responsive fluorescent compounds. 1. Effect of cation binding on photophysical properties of benzoxazinone derivative linked to monoaza-15-crown-5. J. Phys. Chem. 1988, 92, 6233–6237. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: Baltimore, MD, USA, 2006. [Google Scholar] [CrossRef]
- Rodriguez-Cruz, S.E.; Jockusch, R.A.; Williams, E.R. Hydration energies and structures of alkaline earth metal ions, M2+(H2O)n, n = 5–7, M = Mg, Ca, Sr, and Ba. J. Am. Chem. Soc. 1999, 121, 8898–8906. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rico-Yuste, A.; Gómez-Arribas, L.N.; Pérez-Conde, M.C.; Urraca, J.L.; Moreno-Bondi, M.C. Rapid determination of Alternaria mycotoxins in tomato samples by pressurised liquid extraction coupled to liquid chromatography with fluorescence detection. Food Addit. Contam. Part A 2018, 35, 2175–2182. [Google Scholar] [CrossRef] [PubMed]
- Janić Hajnal, E.; Orčić, D.; Torbica, A.; Kos, J.; Mastilović, J.; Škrinjar, M. Alternaria toxins in wheat from the autonomous Province of vojvodina, Serbia: A preliminary survey. Food Addit. Contam. Part A 2015, 32, 361–370. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Mañes, J.; Berrada, H.; Juan, C. Development and validation of a LC-ESI-MS/MS Method for the determination of Alternaria toxins alternariol, alternariol methyl-ether and tentoxin in tomato and tomato-based products. Toxins 2016, 8, 328. [Google Scholar] [CrossRef] [Green Version]
- Gans, P.; Sabatini, A.; Vacca, A. Investigation of equilibria in solution. Determination of equilibrium constants with the HYPERQUAD suite of programs. Talanta 1996, 43, 1739–1753. [Google Scholar] [CrossRef]
- Faisal, Z.; Lemli, B.; Szerencsés, D.; Kunsági-Máté, S.; Bálint, M.; Hetényi, C.; Kuzma, M.; Mayer, M.; Poór, M. Interactions of zearalenone and its reduced metabolites α-zearalenol and β-zearalenol with serum albumins: Species differences, binding sites, and thermodynamics. Mycotox. Res. 2018, 34, 269–278. [Google Scholar] [CrossRef]
- Poór, M.; Matisz, G.; Kunsági-Máté, S.; Derdák, D.; Szente, L.; Lemli, B. Fluorescence spectroscopic investigation of the interaction of citrinin with native and chemically modified cyclodextrins. J. Lumin. 2016, 172, 23–28. [Google Scholar] [CrossRef]
- Srinivasan, K.; Stalin, T.; Sivakumar, K. Spectral and electrochemical study of host–guest inclusion complex between 2,4-dinitrophenol and β-cyclodextrin. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2012, 94, 89–100. [Google Scholar] [CrossRef]

| Complex | K (L/mol) ± SEM Hyperquad | K (L/mol) ± SEM Benesi-Hildebrand |
|---|---|---|
| AOH-Mg2+ | 5.86 ± 0.62 | 4.11 ± 0.61 |
| AOH-Ca2+ | 1.49 ± 0.04 | 1.75 ± 0.03 |
| AOH | Interacting Ion | K (L/mol) |
|---|---|---|
| nonionic | Mg2+ | 5.54 |
| monoanionic | Mg2+ | 5.91 |
| nonionic | Ca2+ | 1.80 |
| monoanionic | Ca2+ | 4.80 |
| LOD | LOQ | |||
|---|---|---|---|---|
| Method | nM | μg/L | nM | μg/L |
| C8 without Mg2+ | 50 | 12.9 | 100 | 25.8 |
| C8 with Mg2+ | 10 | 2.6 | 20 | 5.2 |
| C18 without Mg2+ | 100 | 25.8 | 200 | 51.6 |
| C18 with Mg2+ | 20 | 5.2 | 50 | 12.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fliszár-Nyúl, E.; Lemli, B.; Kunsági-Máté, S.; Poór, M. Effects of Microenvironmental Changes on the Fluorescence Signal of Alternariol: Magnesium Induces Strong Enhancement in the Fluorescence of the Mycotoxin. Int. J. Mol. Sci. 2021, 22, 8692. https://doi.org/10.3390/ijms22168692
Fliszár-Nyúl E, Lemli B, Kunsági-Máté S, Poór M. Effects of Microenvironmental Changes on the Fluorescence Signal of Alternariol: Magnesium Induces Strong Enhancement in the Fluorescence of the Mycotoxin. International Journal of Molecular Sciences. 2021; 22(16):8692. https://doi.org/10.3390/ijms22168692
Chicago/Turabian StyleFliszár-Nyúl, Eszter, Beáta Lemli, Sándor Kunsági-Máté, and Miklós Poór. 2021. "Effects of Microenvironmental Changes on the Fluorescence Signal of Alternariol: Magnesium Induces Strong Enhancement in the Fluorescence of the Mycotoxin" International Journal of Molecular Sciences 22, no. 16: 8692. https://doi.org/10.3390/ijms22168692
APA StyleFliszár-Nyúl, E., Lemli, B., Kunsági-Máté, S., & Poór, M. (2021). Effects of Microenvironmental Changes on the Fluorescence Signal of Alternariol: Magnesium Induces Strong Enhancement in the Fluorescence of the Mycotoxin. International Journal of Molecular Sciences, 22(16), 8692. https://doi.org/10.3390/ijms22168692







