Optical Properties of Composites Based on Poly(o-phenylenediamine), Poly(vinylenefluoride) and Double-Wall Carbon Nanotubes
Abstract
:1. Introduction
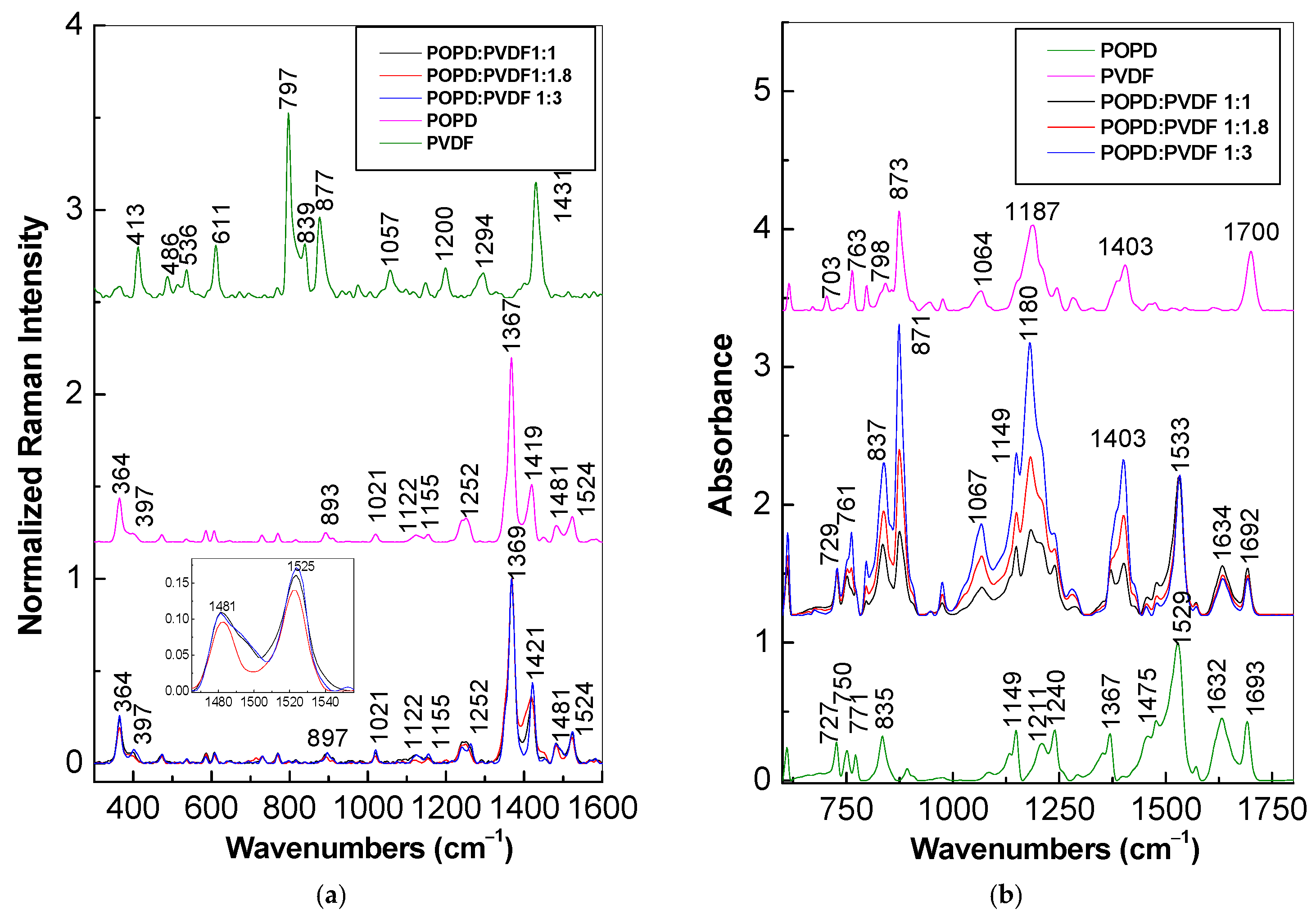
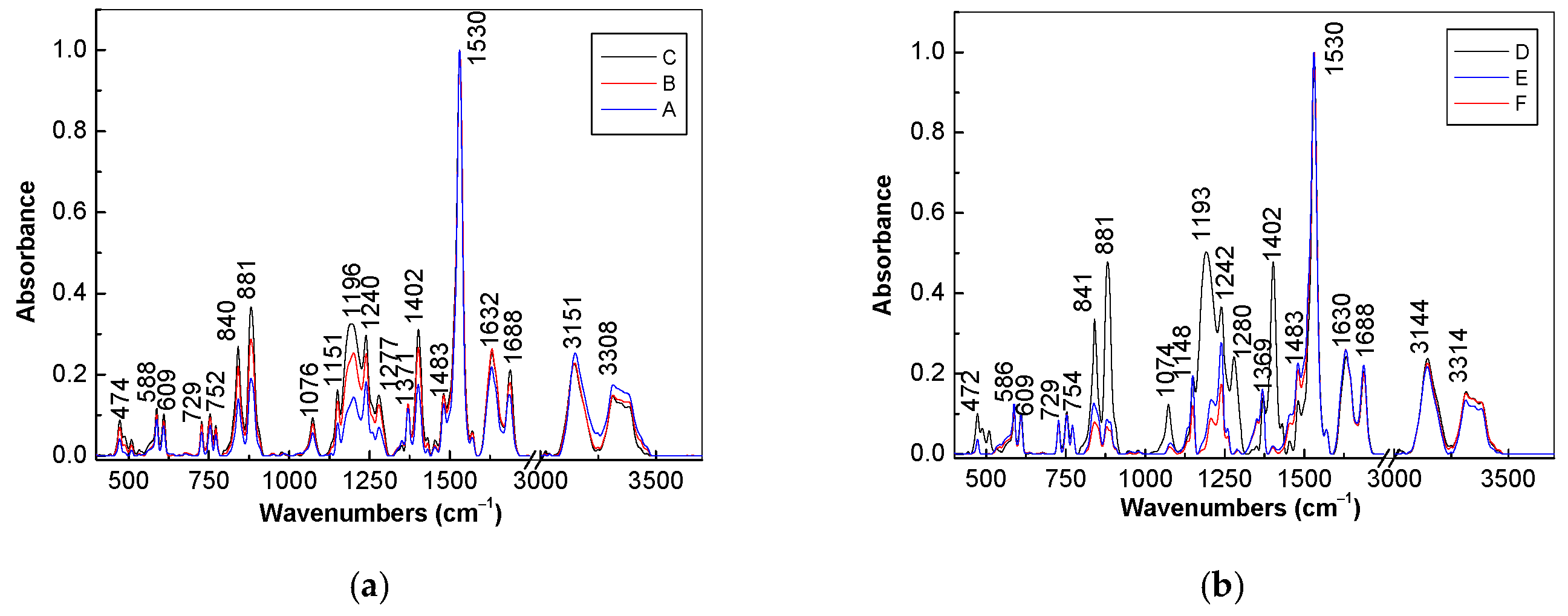
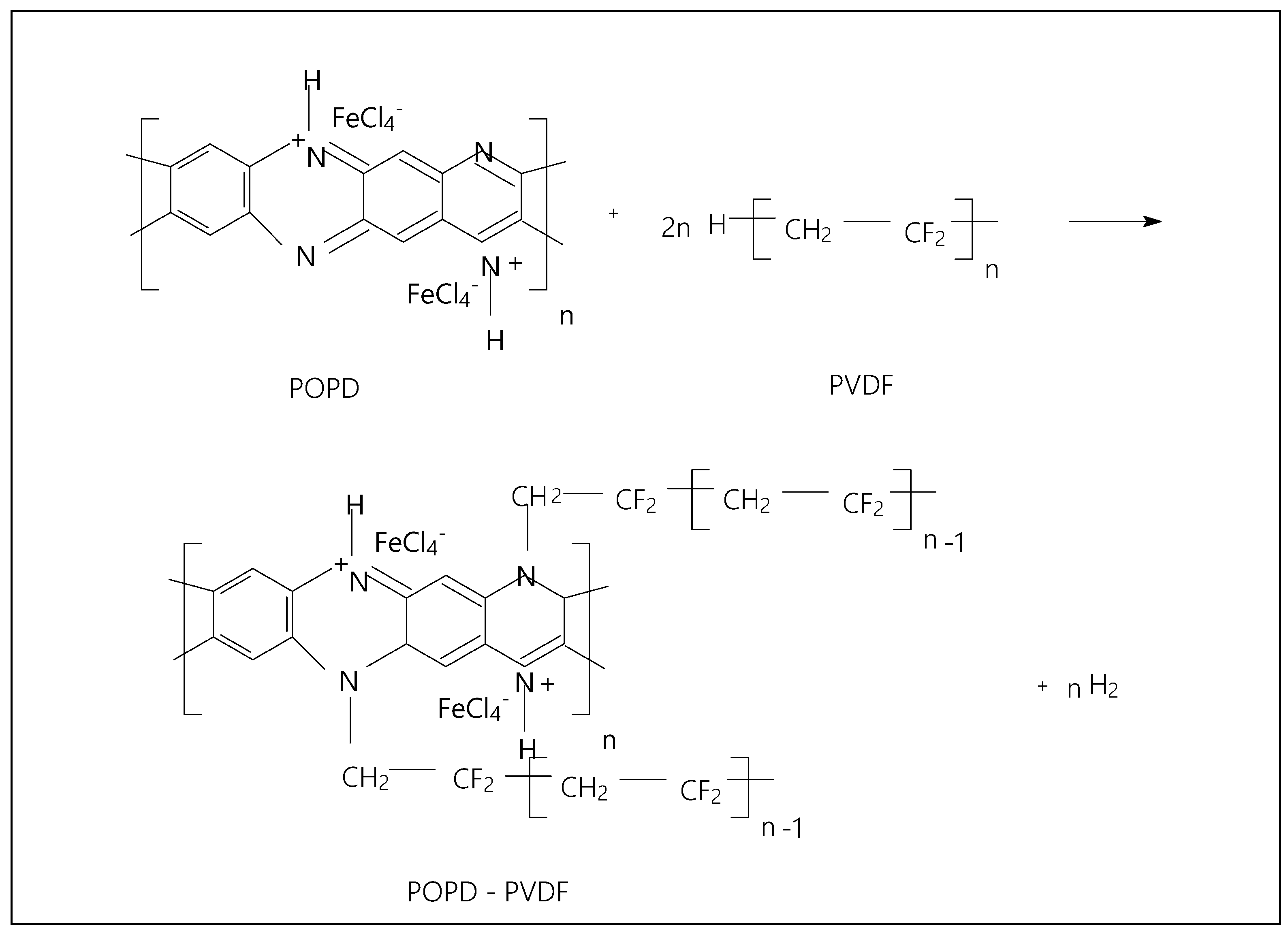
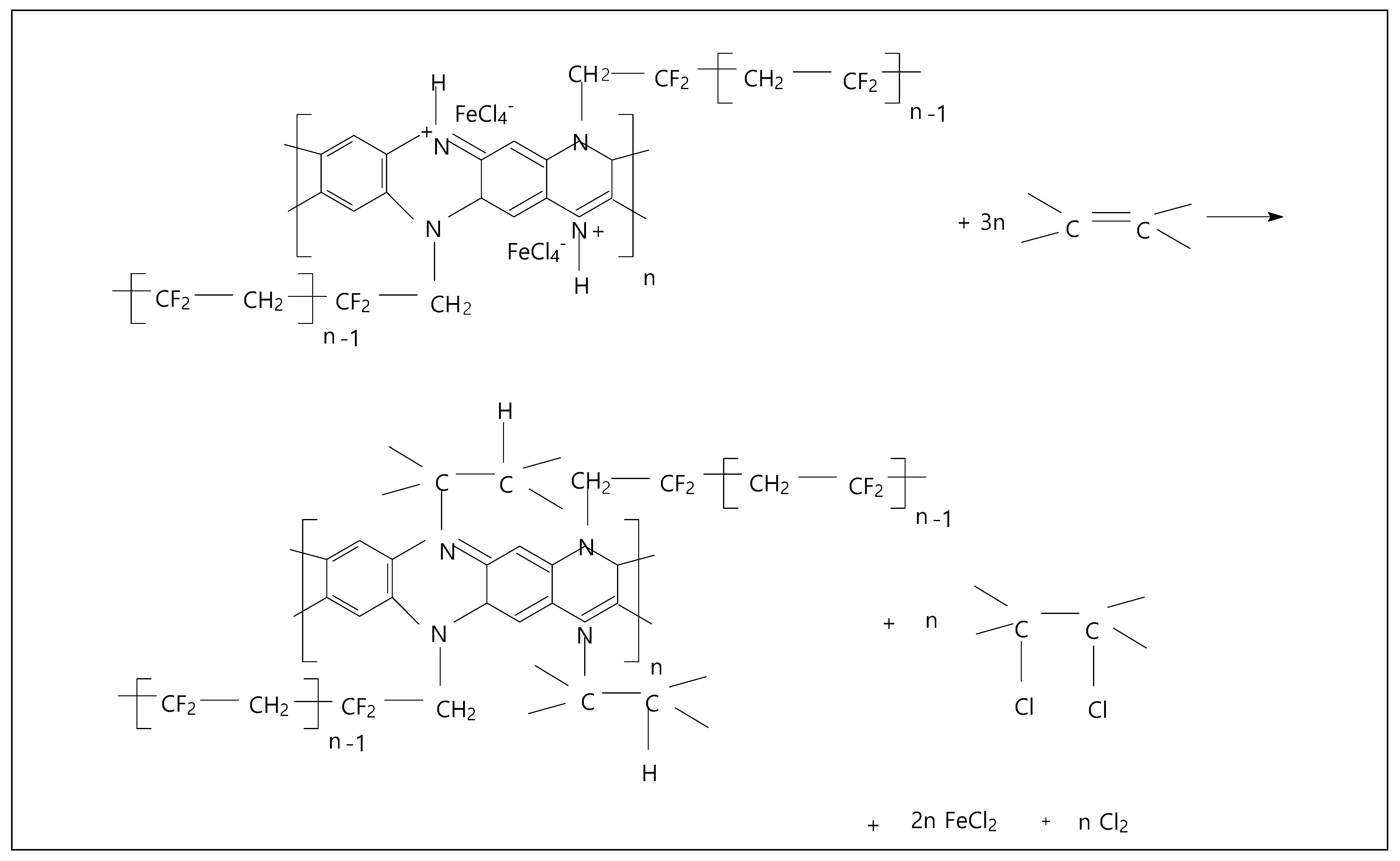
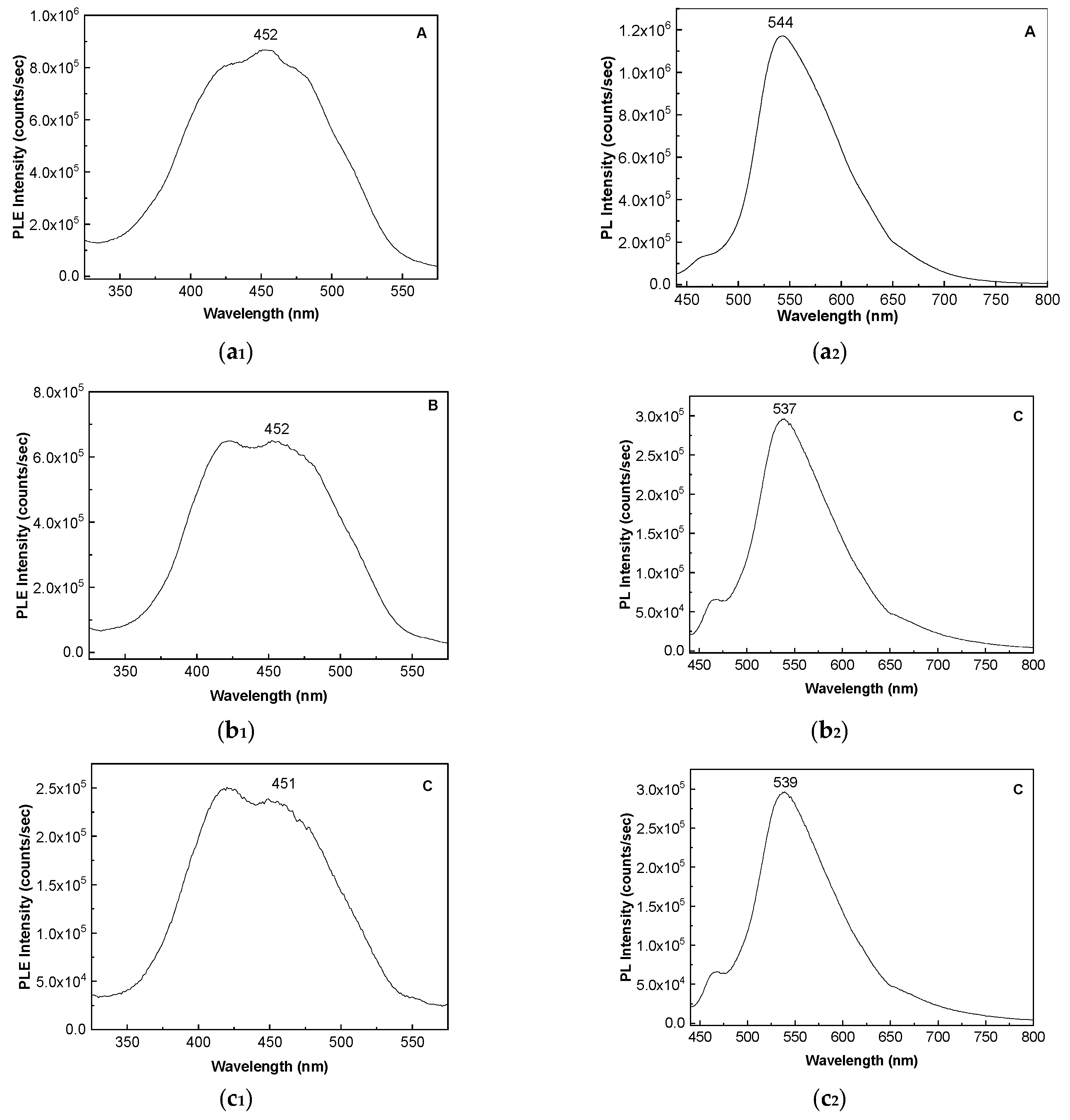
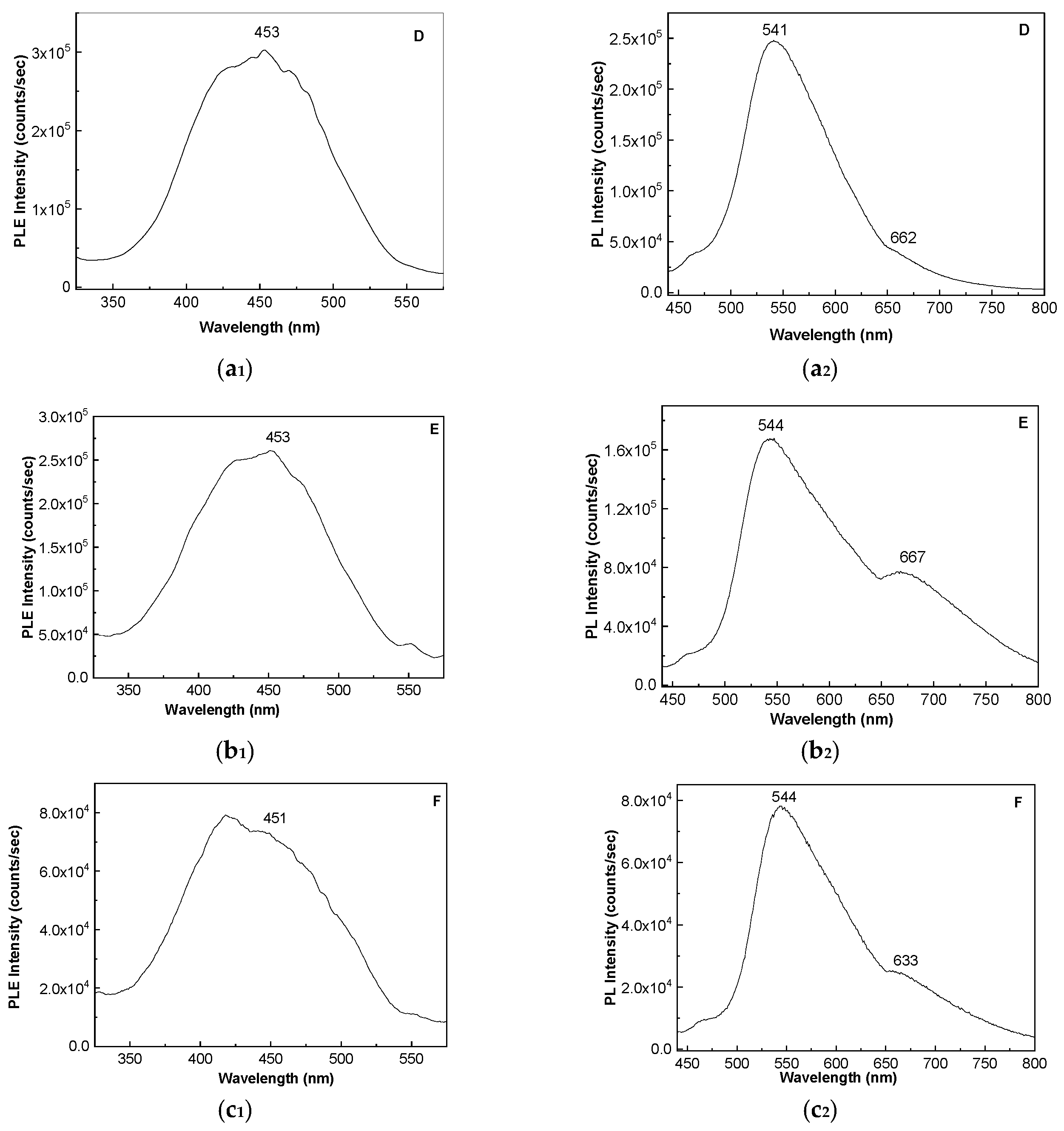
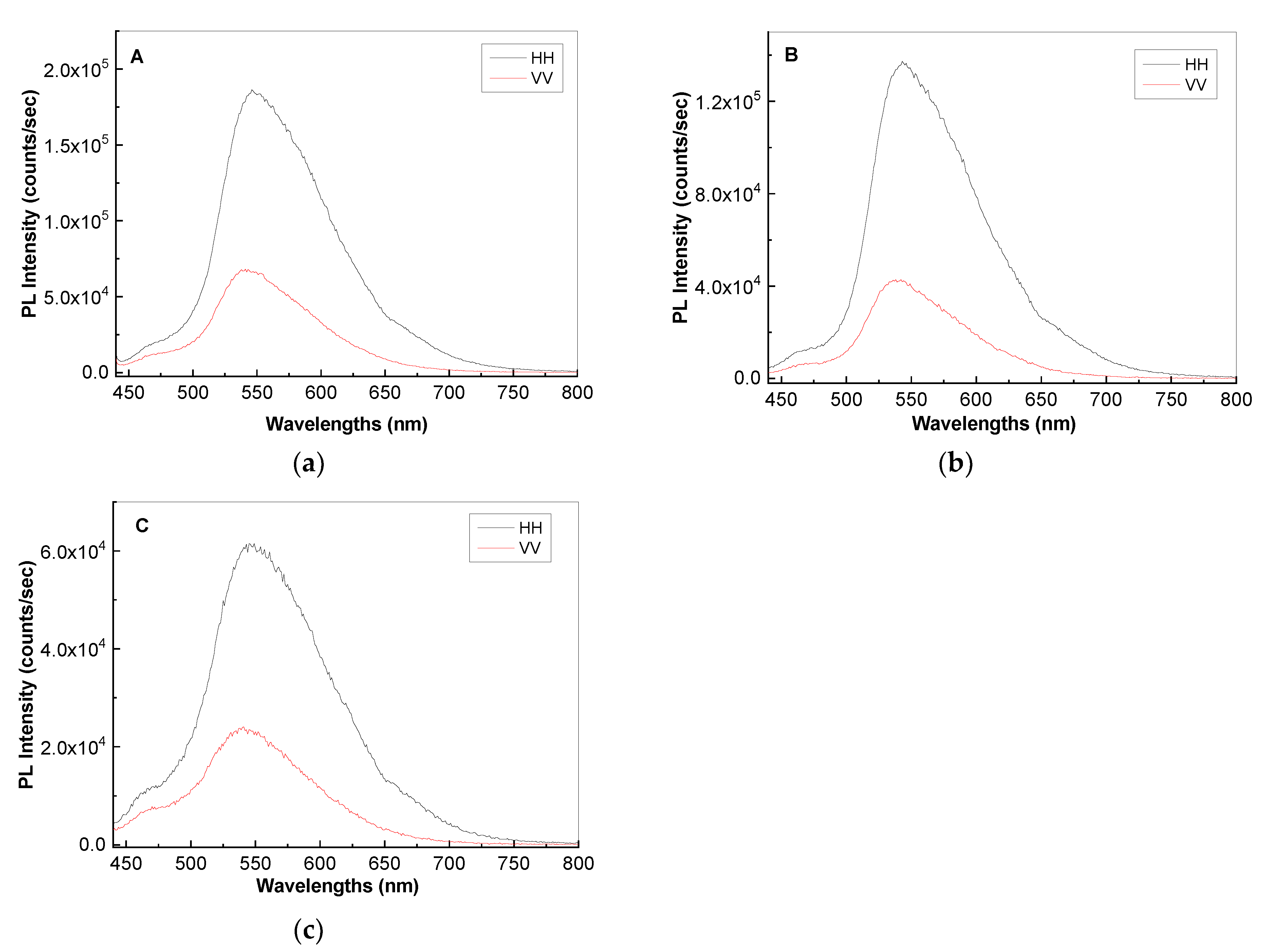
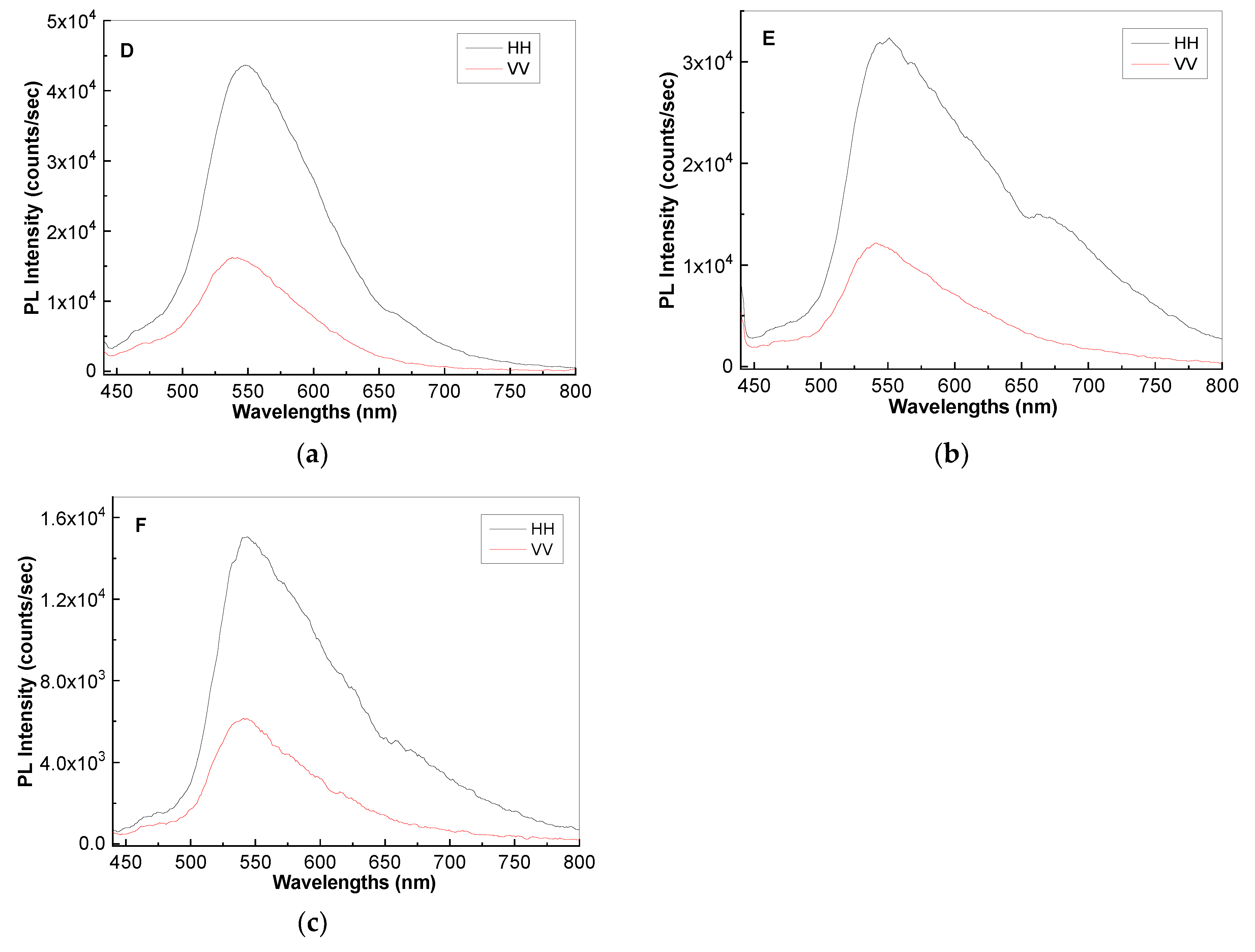
2. Results
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Esmaeil, H.; Habib, R. Kinetics of direct ethanol fuel cell based on Pt-PoPD nano particle anode catalyst. Int. J. Hydrogen Energy 2013, 38, 5442–5448. [Google Scholar]
- Kor, K.; Zarei, K. Development and characterization of an electrochemical sensor for furosemide detection based on electropolymerized molecularly imprinted polymer. Talanta 2016, 146, 181–187. [Google Scholar] [CrossRef]
- Couto, R.A.; Costa, S.S.; Mounosef, B.; Racheco, J.G.; Fernandes, E.; Carvalho, F.; Rodriques, C.M.P.; Selerue-Martos, C.; Braga, A.A.C.; Goncalves, L.M.; et al. Electrochemical sensing of ecstasy with electropolymerized molecularly imprinted polu(o-phenylenediamine) polymer on the surface of disposable screen-printed carbon electrodes. Sens. Actuators B 2019, 290, 378–386. [Google Scholar] [CrossRef]
- Zoromba, M.S.; Abdel-Aziz, M.H.; Bassyouni, M.; Bahaitham, H.; Al-Hossainy, A.F. Poly(o-phenylenediamine) thin film for organic solar cell applications. J. Solild State Electrochim. 2018, 22, 3673–3687. [Google Scholar] [CrossRef]
- Xu, L.; Sun, Y.; Han, B.; Su, C. Electrochemical performances on both poly(phenylenediamine) derivatives as anode of lithium ion batteries. J. Electrochem. Soc. 2019, 166, A1363–A1369. [Google Scholar] [CrossRef]
- Sayyach, S.M.; Khaliel, A.B.; Aboud, A.A.; Mohamed, S.M. Chemical polymerization kinetic of poly(o-phenylenediamine) and characterization of the obtained polymer in aqueous hydrochloric acid solution using K2Cr2O7 as oxidizing agent. Int. J. Polym. Sci. 2014, 2014, 520910. [Google Scholar]
- Jiang, K.; Ma, S.; Bi, H.; Chen, D.; Han, X. Morphology controllable fabrication of poly o-phenylenediamine microstructures tuned by the ionic strength and their applications in pH sensors. J. Mater. Chem. A 2014, 2, 19208. [Google Scholar] [CrossRef]
- Yan, S.; Yang, S.; He, L.; Ye, C.; Song, X.; Liao, F. Quantum size effect of poly(o-phenylenediamine) quantum dots: From controllable fabrication to tunable photoluminescence properties. Synth. Met. 2014, 198, 142–149. [Google Scholar] [CrossRef]
- Jiang, K.; Ma, S.; Zhang, Y.; Han, X. Formation of square prism-shaped poly(o-phenylenediamine) fibers triggered by high ionic strength. RSC Adv. 2016, 6, 21895. [Google Scholar] [CrossRef]
- Sun, X.; Hagner, M. Mixing aqueous ferric chloride and o-phenylenediamine solutions at room temperature Ka fast, economical route to ultra long microfibrils of assembling o-phenylenediamine dimers. Langmuir 2007, 23, 10441–10444. [Google Scholar] [CrossRef]
- Yang, C.; Zhang, M.; Dong, W.; Cui, G.; Ren, Z.; Wang, W. Highly efficient photocatalytic degradation of methylene blue by POPD/TiO2 nanocomposite. PLoS ONE 2017, 12, e0174104. [Google Scholar] [CrossRef] [PubMed]
- Ganash, A. Anticorrosive properties of poly(o-phenylenediamine)/ZnO nanocomposites coated stainless steel. J. Nanomater. 2014, 2014, 40. [Google Scholar] [CrossRef] [Green Version]
- Han, S.; Li, B.; Song, Z.; Pan, S.; Zhang, Z.; Yao, H.; Zhu, S.; Xu, G. Kanamycin sensor based on electrosynthesized molecularly imprinted poly o-phenylenediamine film on sinlge-walled carbon nanohorn modified glassy carbon electrode. Analyst 2017, 142, 218–223. [Google Scholar] [CrossRef]
- Pisarevskaya, E.Y.; Rychagov, A.Y.; Gorbunov, A.M.; Averin, A.A.; Rychagov, Y.B.; Gorbunov, A.M.; Averin, A.A.; Makarychev, Y.B.; Efirmov, O.N. Synthesis of nanostructured conducting composite films based on reduced graphene oxide and poly o-phenylenediamine. Synth. Met. 2018, 243, 1–7. [Google Scholar] [CrossRef]
- Jiang, J.; Nie, G.; Nie, P.; Li, Z.; Pan, Z.; Kou, Z.; Dou, H.; Zhang, X.; Wang, J. Nanohollow carbon for rechargeable batteries: Ongoing progresses and challenges. Nano Micro Lett. 2020, 12, 183. [Google Scholar] [CrossRef] [PubMed]
- Kuralay, F.; Bayramli, Y. Electrochemical determination of mitomycin C and its interaction with double-stranded DNA using a poly(ophenylenediamine)—Multi walled carbon nanotube modified pencil graphite electrode. Anal. Lett. 2020, 8, 1295–1308. [Google Scholar]
- Baibarac, M.; Baltog, I.; Scocioreanu, M.; Ballesteros, B.; Mevellec, J.Y.; Lefrant, S. One-dimensional composites based on single-walled carbon nanotubes and poly(o-phenylenediamine). Synth. Met. 2011, 16, 2344–2354. [Google Scholar] [CrossRef]
- Deng, W.; Zhang, Y.; Tan, Y.; Ma, M. Three-dimensional nitrogen-doped graphene derived from poly o-phenylenediamine for high-performance supercapacitors. J. Electroanal. Chem. 2017, 787, 103–109. [Google Scholar] [CrossRef]
- Yang, W.; Zhou, H.; Huang, Z.; Li, H.; Fu, C.; Chen, L.; Li, M.; Liu, S.; Kuang, Y. In situ growth of single-stranded like poly(o-phenylenediamine) onto graphene for high performance supercapacitors. Electrochim. Acta 2017, 245, 41–50. [Google Scholar] [CrossRef]
- Dukar, N.; Tunc, S.; Ozturk, K.; Demirci, S.; Dumangoz, M.; Celebi, M.S.; Kuralay, F. High sensitive dopamine sensing in biological fluids with one-pot prepared graphene/poly(o-phenylenediamine) modified electrodes. Mat. Chem. Phys. 2019, 228, 357–362. [Google Scholar] [CrossRef]
- Bas, S.Z. A gold nanoparticle functionalized multiwalled carbon nanotubes-poly(o-phenylenediamine) composite film for glucose biosensing applications. Anal. Methods 2014, 6, 752–7759. [Google Scholar] [CrossRef]
- Guny, S. Electrosynthesis of molecularly imprinted poly-o-phenylenediamine on MWCNT modified electrode for selective determination of meldonium. Electroanalysis 2019, 31, 661–670. [Google Scholar] [CrossRef]
- Jeyapragasam, T.; Raju, R.; Chen, S.M.; Saraswathi, R.; Hatamleh, A.A.; Chen, T.W.; Rwer, S.P. Poly(o-phenylenediamine)—Multiwalled carbon nanotube nanocomposite based electrochemical sensing platform for paraquat detection. Int. J. Electrochem. Sci. 2019, 14, 8326–8339. [Google Scholar] [CrossRef]
- Sousa, M.S.P.; de Sa Javo, A.C.; de Oliveira, P.J.; da Silva, M.J.; Santos, R.J.; Paim, L.L. Impedimetric sensor for pentoses based on electrodeposited carbon nanotubes and molecularly imprinted poly o-phenylenediamine. ECS J. Solid State Sci. Technol. 2020, 9, 041006. [Google Scholar] [CrossRef]
- Shi, X.; Ren, X.; Jing, N.; Zhang, J. Electrochemical determination of ampicillin based on an electropolymerization poly(o-phenylenediamine)/gold nanoparticle/single-walled carbon nanotube modified glassy carbon electrode. Anal. Lett. 2019, 53, 2854–2867. [Google Scholar] [CrossRef]
- Zare, E.N.; Lakouraj, M.M.; Ghasemi, S.; Moosavi, E. Emulsion polymerization for fabrication of poly(o-phenylenediamine)@multi-walled carbon nanotubes nanocomposite: Characterization and its application to the corrosion protection of 316LSS. RSC Adv. 2015, 84, 68788–68795. [Google Scholar] [CrossRef]
- Baibarac, M.; Baltog, I.; Lefrant, S.; Gomez-Romero, P. Polydiphenylamine/carbon nanotube composites for applications in rechargeable lithium batteries. Mat. Sci. Eng. B Adv. Funct. Solid State Mat. 2011, 176, 110–120. [Google Scholar] [CrossRef]
- Baibarac, M.; Baltig, I.; Frunza, S.; Magrez, A.; Schur, D.; Zaginaichenko, S.Y. Single-walled carbon nanotubes functionalized with polydiphenylamine as active materials for applications in the supercapacitors field. Diam. Relat. Mater. 2013, 32, 72–82. [Google Scholar] [CrossRef]
- Tonosaki, T.; Oho, T.; Isomura, K.; Ogura, K. Effect of the protonation level of poly(o-phenylenediamine) (POPD) on the ac impedance of humidity-sensitive POPD/poly(vinyl alcohol) composite film. J. Electroanal. Chem. 2002, 520, 89–93. [Google Scholar] [CrossRef]
- Iqbal, M.Z.; Khan, S. Progress in the performance of dye sensitized solar cells by incorporating cost effective counter electrodes. Sol. Energy 2018, 160, 130–152. [Google Scholar] [CrossRef]
- Jiang, H.; Sun, X.; Huang, M.; Wang, Y.; Li, D.; Dong, S. Rapid self-assembly of oligo(o-phenylenediamine) into one-dimensional structures through of facile reprecipitation route. Langmuir 2006, 22, 3358–3361. [Google Scholar] [CrossRef] [PubMed]
- Li, X.G.; Huang, M.R.; Duan, W. Novel multifunctional polymers from aromatic diamines by oxidative polymerizations. Chem. Rev. 2002, 102, 2925–3030. [Google Scholar] [CrossRef] [PubMed]
- Hill, P.S.; Schauble, E.A. Modeling the effects of bond environment on equilibrium iron isotope fractionation in ferric aquo-chloro complexes. Geochim. Cosmochim. Acta 2008, 72, 1939–1958. [Google Scholar] [CrossRef]
- Kobatasgum, M.; Tashiro, K.; Tadokoro, H. Molecular vibrations of three crystal forms of poly(vinylidene fluoride). Macromolecules 1975, 8, 158–171. [Google Scholar]
- Ramer, N.J.; Marrone, T.; Stiso, K.A. Structure and vibrational frequency determination for α-poly(vinylidene fluoride) using density-functional theory. Polymer 2006, 47, 7160–7165. [Google Scholar] [CrossRef]
- Matea, A.; Baibarac, M.; Baltog, I. Optical properties of single-walled carbon nanotubes highly separated in semiconducting and metallic tubes functionalized with poly(vinyl fluoride). J. Mol. Struct. 2017, 1130, 38–45. [Google Scholar] [CrossRef]
- Zhou, X.; Fogarasi, G.; Liu, R.; Pulay, P. Building a database of force constants based on scaled ab initio (SQM) results. I. Chlorobenzenes. Spectrochim. Acta 1993, 49, 1499–1514. [Google Scholar] [CrossRef]
- Bachmann, M.A.; Koenig, J.L. Vibrational analysis of phase II of poly(vinylidene fluoride). J. Chem. Phys. 1981, 74, 5896–5910. [Google Scholar] [CrossRef]
- Sestrem, R.H.; Ferreira, D.C.; Landers, R.; Temperini, M.L.A.; do Nascimento, G.M. Synthesis and spectroscopic characterization of polymer and oligomers of ortho-phenylenediamine. Eur. Polym. J. 2010, 46, 484–493. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, H. In situ external reflection FTIR spectroelectrochemical investigation of poly(o-phenylenediamine) film coated on a platinum electrode. Electrochim. Acta 1994, 41, 2019–2024. [Google Scholar] [CrossRef]
- Daescu, M.; Oprica, M.; Smaranda, I.; Matei, E.; Nastac, D.; Cramariuc, O.; Cramariuc, B.; Baibarac, M. Vibrational and photoluminescence properties of composites based on double-walled carbon nanotubes, poly(o-phenylenediamine) and poly(ethylene oxide). J. Nanosci. Nanotechnol. 2021, 21, 2334–2341. [Google Scholar] [CrossRef] [PubMed]
- Baibarac, M.; Ilie, M.; Baltog, I.; Lefrant, S.; Humbert, B. Infrared dichroism studies and anisotropic photoluminescence properties of poly(para-phenylene vinylene) functionalized reduced graphene oxide. RSC. Adv. 2017, 7, 6931–6942. [Google Scholar] [CrossRef] [Green Version]



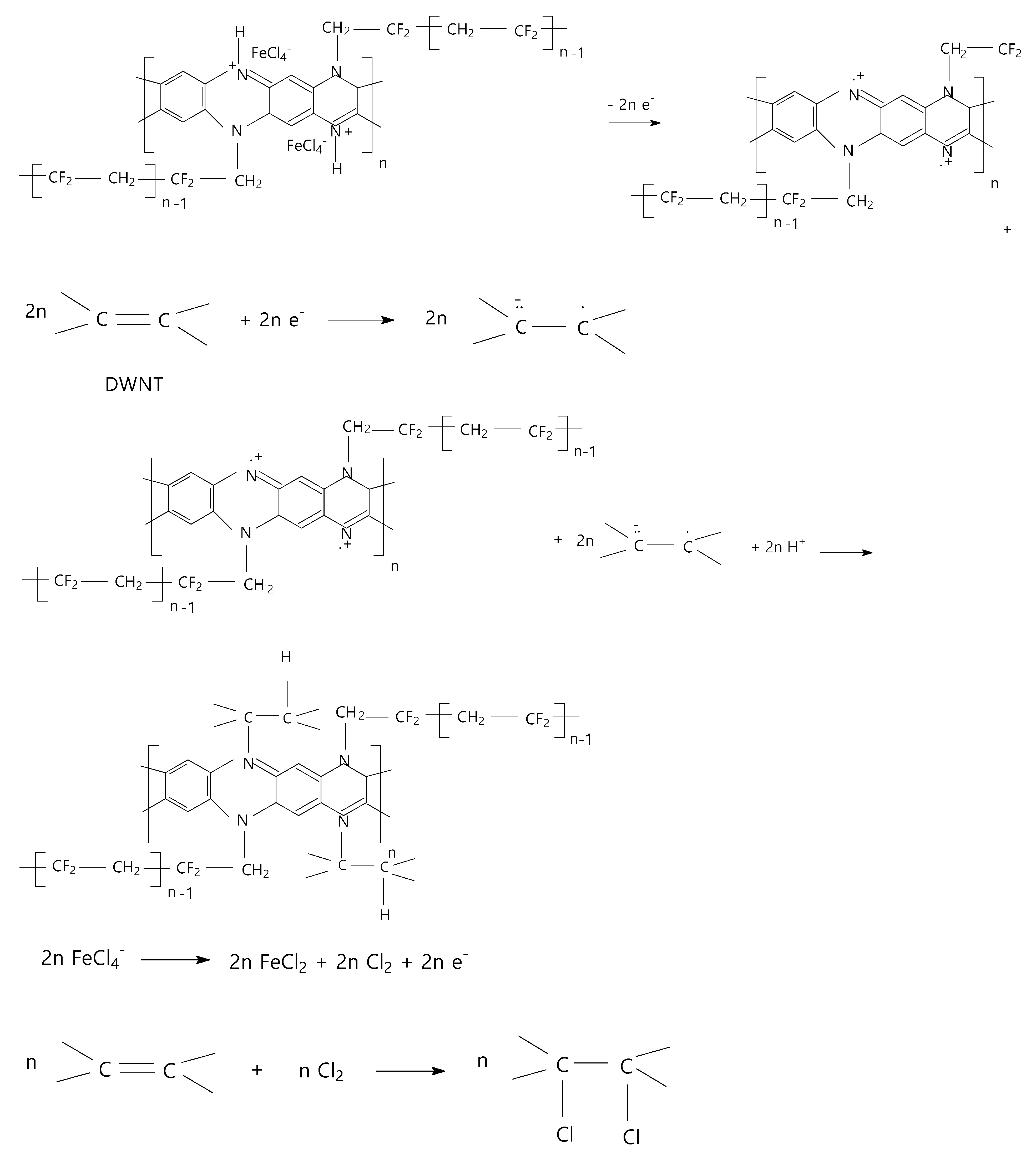
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baibarac, M.; Daescu, M.; Matei, E.; Nastac, D.; Cramariuc, O. Optical Properties of Composites Based on Poly(o-phenylenediamine), Poly(vinylenefluoride) and Double-Wall Carbon Nanotubes. Int. J. Mol. Sci. 2021, 22, 8260. https://doi.org/10.3390/ijms22158260
Baibarac M, Daescu M, Matei E, Nastac D, Cramariuc O. Optical Properties of Composites Based on Poly(o-phenylenediamine), Poly(vinylenefluoride) and Double-Wall Carbon Nanotubes. International Journal of Molecular Sciences. 2021; 22(15):8260. https://doi.org/10.3390/ijms22158260
Chicago/Turabian StyleBaibarac, Mihaela, Monica Daescu, Elena Matei, Daniela Nastac, and Oana Cramariuc. 2021. "Optical Properties of Composites Based on Poly(o-phenylenediamine), Poly(vinylenefluoride) and Double-Wall Carbon Nanotubes" International Journal of Molecular Sciences 22, no. 15: 8260. https://doi.org/10.3390/ijms22158260
APA StyleBaibarac, M., Daescu, M., Matei, E., Nastac, D., & Cramariuc, O. (2021). Optical Properties of Composites Based on Poly(o-phenylenediamine), Poly(vinylenefluoride) and Double-Wall Carbon Nanotubes. International Journal of Molecular Sciences, 22(15), 8260. https://doi.org/10.3390/ijms22158260






